Reprinted from the PKOCEEDrxas OF TUE NATIOXAL ACADEMY OF SCIENCES Vol. 54, No. 4, pp October, 1965.
|
|
|
- Cecil Lynch
- 5 years ago
- Views:
Transcription
1 Reprinted from the PKOCEEDrxas OF TUE NATIOXAL ACADEMY OF SCIENCES Vol. 54, No. 4, pp October, ISOPIESTIC TECHNIQUE FOR MEASURING LEAF WATER POTENTIALS WITH A THERMOCOUPLE PSYCHROMETER* BY JOHN S. BoYERf AND EDWARD B. KNIPLING DEPARTMENT OB' BOTANY, DUKE UNIVERSITY, DUHHAM, NORTH CAROLINA Communicated by Paul J. Kramer, August 18, 1965 Measurements of the water potential of plant tissue are made with thermocouple psychrometers by enclosing the tissue and thermocouple in a small container kept at a constant temperature and determining the degree of cooling of the thermocouple as water evaporates from it and is absorbed by the tissue. It is assumed that the rate of vapor transfer is proportional to the difference in potential between the thermocouple and plant material. Rawlins 1 suggested that leaf water potentials obtained with Richards and Ogata psychrometers 2 are too high, indicating too low a water stress, because of the leaf resistance to the diffusion of water vapor. In contrast, Barrs 3 concluded that Richards psychrometer values, when corrected for heat of respiration, 4 are the same as those obtained with Spanner 5 psychrometers and are therefore free of leaf resistance error. The lack of agreement in these studies indicates a need for further examination of the leaf resistance error in psychrometer measurements. The mathematics describing the diffusion process within a Richards psychrometer chamber (see below) indicates that there must be a leaf resistance error in the water potentials measured by this method. The close agreement in water potentials measured with the Richards and Spanner techniques suggests that the Spanner technique may also be in error. In this paper, we describe a new technique which is free of error attributable to leaf resistance, and this technique is used to evaluate the error in Richards and Spanner measurements. The new technique involves the use of a modified Richards thermocouple to determine the rate of vapor movement between the thermocouple and the leaf when solutions of various potentials are present on the thermocouple. No vapor movement occurs when the solution on the thermocouple has a potential equal to that
2 VOL. 54, 1965 BOTANY: BOYER AND KNIPLINO 1045 of the tissue, and therefore leaf resistance does not affect the measurement. The theoretical basis for this conclusion can be seen from the equations that follow. The pathway for vapor transfer between the thermocouple and the leaf can be divided into two segments. The first is the air between the thermocouple and the leaf surface. The second segment is the path between the leaf surface and the liquid phase inside the leaf. The rate of vapor transfer through the first segment may be represented by f dm\ a m,, -77 ) = (c,i - c tt ), (I) dt J d r a where (dm/dt) d is the rate of vapor transfer from the drop (gm sec" 1 ), Q is the concentration of water vapor in the air next to the drop (gin cm~ 3 ), c a is the concentration of vapor in the air next to the tissue, a,,, is the geometric mean area for flow (cm 2 ), arid? is the resistance of the air to vapor transfer (sec cm" 1 ) at a,,,. fi The calibration of Richards psychrometers shows that c d c a is proportional to the potential of the calibrating solution and therefore is proportional to the concentration difference, c 0 c a, where CD is the saturation vapor concentration of water at bath temperature. 7 Vapor transfer from the drop may then be expressed by substituting Co c a and the proportionality constant, L, in equation (1): dm\ -La m -7; ) = - (CD - c«), (2) dt Id r a where concentrations are expressed at bath temperature and the constant, L, may be considered to be a part of the coefficient, a m /r a, relating concentration differences to rate of vapor transfer. A similar equation describes the rate of water vapor movement through the second segment of the diffusion path: fdm\ -a, = (c a ~ ci), (3) dt, /a r, where c t is the concentration of water vapor inside the leaf, a, is the area of the leaf, and? ( is the resistance of the leaf to vapor diffusion. An equation describing steady-state vapor transfer for the entire diffusion path from the drop to the leaf can be determined by first dividing (2) and (3) by their respective coefficients relating vapor concentration differences to rate of vapor transfer. Adding the concentration differences in the two equations then gives an expression for the total concentration difference, CQ c (, at bath temperature in terms of flow in the two segments of the vapor pathway. Since the rate in each segment is the same at the steady state, vapor transfer in the total pathway may be described in terms of the rate of vapor transfer from the drop. Rearranging terms: (dm\ La m a t.. I =. (c 0 c,). (4) \ dt la LiOmTi + ttit a Equation (4) may be restated in terms of water potentials 8 f dm\ c 0 VLa m a t /T T N,. dt id RT(La m r t + a t r a )
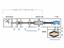
3 1046 BOTANY: BOYER AND KNIPLINO PROC. N. A. S. where V is the partial molal volume of water (liters mole"" 1 ), R is the ideal gas constant (liter bars mole" 1 cleg" 1 ), T is the Kelvin temperature, \?o is the water potential of the thermocouple drop at bath temperature, and ^f l is the water potential of the leaf. Equation (5) is the steady-state flow equation for Richards psychrometers when leaf tissue lines the walls in the chamber. It describes a straight line with a slope of (cvvla m ai)/[rt(la m ri + a ( r a )]. Since r t is included in the slope term, it will affect the rate of vapor transfer at all points except when ^o equals ^ and (dm/dt) a is zero. The point at which these two potentials are equal, the isopiestic point, TO GALVANOMETER, D FIG. 1. Thermocouple psychrometer for making isopiestic determinations. The psychrometer chamber and barrel are made of brass and are submerged in a constant-temperature water bath below the point A. Key to symbols: barrel, A; Plexiglas tube, B; plunger heat sink, P; diagrammatic representation of 0-ring seal for chamber and soft rubber seal for plunger, S; thermocouple with ring junction, T. may be determined by measuring thermocouple output when Sto is changed. A thermocouple output of zero indicates no transfer. An apparatus for measuring the isopiestic point is shown in Figure 1. The thermocouple is made of heavier wire (B and S gauge 38) than that of Richards and is mounted on a plunger which is sealed into the chamber by a soft rubber washer, permitting it to be removed and resealed rapidly. Leaf water potential is measured by first allowing the rate of vapor transfer between water or a solution on the thermocouple junction and the leaf tissue to reach the steady state. The plunger is then removed and rapidly replaced by a second plunger assembly with a solution of different potential on the thermocouple junction, and a second steady-state measurement is made.
4 VOL. 54, 1965 BOTANY: BOYER AND KNIPLING 1047 Figure 2 shows the graph obtained for a determination by the isopiestic method. To determine the line, four measurements of thermocouple output were made, one with water and three with solutions of different potential on the junction. The four values lie on a straight line, as predicted by equation (5), so that the line may be determined by a minimum of two measurements. The x intercept is represented by the determination at 3.9 bars and is the isopiestic point. This value, when corrected for respiration, is SP;, the true leaf water potential. The y intercept represents a typical Richards determination of 3.6 bars with water on the thermocouple junction. This value is essentially ^a, the water potential of the air at the leaf surface. -I POTENTIAL OF SOLUTION ON THERMOCOUPLE ( bar! ) FIG. 2. Isopiestic measurement of water potential of privet leaf tissue. The isopiestic value, 3.9 bars, is shown by a determination at the x intercept. The output at the y intercept corresponds to a Richards determination of 3.6 bars (galvanometer deflection = 1 cm bar" 1 ). The success of the isopiestic technique is strongly dependent on being able to change the solution on the ring without causing appreciable change in the water potential of the tissue or the air surrounding it. To determine whether the water potential changes when the apparatus in Figure 1 is opened, a steady-state readingwas obtained with water on the thermocouple junction and privet leaf tissue in the psychrometer chamber. The plunger-thermocouple assembly was then removed for 15 sec and replaced. Figure 3 indicates that the steady-state output remained the same even though the chamber had been opened three times. Similar results have been obtained with geranium and tomato. Since leaf resistance does not affect isopiestic values, it was possible to determine the influence of leaf resistance on Spanner and Richards measurements for leaf tissue from several plant species at various water potential levels. All three methods were used on the same sample of plant tissue by determining steady-state output with the apparatus shown in Figure 1, first with a Spanner thermocouple, then with
5 1048 BOTANY: BOYER AND KNIPLING PROC. N. A. S. PRIVET 3 O 4 TIME (hr) FIG. 3. The effect of removing the plunger on the steady-state output of the thermocouple shown in Fig. 1. The initial approach to the steady state is shown at A. The plunger was then removed for 15 sec and replaced at B, C, and D. a. Richards thermocouple, and finally by the isopiestic method. The Spanner thermocouple was then placed back in the chamber to check for drying of the tissue. No drying was detected. All measurements were corrected for the heat of respiration by inserting a dry Richards or Spanner thermocouple in the psychrometer chamber. 4 The results are shown in Table 1. Both the Spanner and the Richards techniques gave consistently more positive values of water potential than the isopiestic method, indicating that error due to leaf resistance is present in both techniques. Error was calculated as (ty t ^f^/^i for the Richards determinations and was pe.' - cent for all species 9 except geranium, which had smaller error (0-2.4%). A similar calculation was made for Spanner thermocouples, which had an error of per cent. At potentials of about 20 bars, Richards thermocouples differed by as much as 2.6 bars from isopiestic measurements. To check these results, diffusive resistances of leaf tissue were measured and theoretical errors based on leaf resistances were calculated for Richards psychrometer measurements. The theoretical error expression was derived from equations Plant Privet Geranium Tomato Magnolia Philodendron TABLE 1 WATER POTENTIAL OF LEAP TISSUE MEASURED BY ISOPIESTIC, SPANNER, AND RICHARDS TECHNIQIJES* potential. bars Potential, bars Error, % Potential, bars Error, % * Measurements by the three techniques were made on the same tissue sample at each water potential level. The calculation of error in Spanner and Richards techniques is described in the text.
6 VOL. 54, 1965 BOTANY: BOYER AND KNIPLING 1049 (2) and (3), which describe the vapor transfer occurring from the drop to the air and from the air to the leaf. Since the rate of vapor transfer through the two segments of the diffusion pathway is equal at steady state, La m,, Q>i,, /f,\ - (GO - c a ) = ~(c a - c ( ). («) ' The concentration differences of (6) may be replaced by water potential differences. 8 Noting that SPo is equal to zero and rearranging terms, the equation may be written The error expression can then be written e = ^J 3 = *' ~ * a = /X<W'- a ) ~ ~ *, ~ L(a m /r a ) + (ai/r,)' Leaf resistances were determined under psychrometer conditions of unstirred air and of a net transfer of water vapor to the leaf. Leaf disks of 2.4 cm diameter were placed across cylindrical chambers 1 cm away from water-saturated filter paper disks of the same diameter. The chambers containing the disks were maintained at 27.0 ± for hr prior to determining weight gains by the leaf disks. Concentrations c, were calculated from leaf water potentials measured isopiestically on parallel samples of tissue. Using the over-all concentration difference, CD Cf, and the cross-sectional area of the chamber, total diffusive resistances (i'a + i'i) were calculated from an equation similar to (1). Diffusive resistances of the lower leaf surfaces were then calculated from the differences between the total resistances and the air resistance in the chamber. Using the diffusivity of water in air (0.256 cm 2 sec~'), 10 the resistance of the air in the chambers was calculated to be 3.9 sec cm" 1. This was confirmed by two determinations with NaCl solutions on filter paper disks, which indicated air resistance values of 3.9 and 4.0 sec cm" 1. The data are shown in Table 2. Leaf resistances of privet and cotton 9 were about 14 sec cm" 1, which corresponded to theoretical errors in Richards determinations of about 7 per cent [calculated 11 from the right-hand side of equation (8)]. Tobacco, tomato, philodendron, and magnolia had leaf resistances ranging from about 19 to 23 sec cm~ l and corresponding errors in ^ of about per cent. TABLE 2 DIFFUSIVE RESISTANCES OF LEAVES AND THEORETICAL ERRORS IN DETERMINATIONS OF LEAF WATER POTENTIAL BY THE RICHARDS METHOD* Plant Potential, bars Resistance, sec cm" 1 Krror, % Privet Cotton Tobacco Philodendron Tomato Magnolia Geranium * Each resistance value is the mean of three observations. The errors tvere calculated from equation (8) (see text) for 20 cm 2 of leaf. (8)
7 1050 BOTANY: BOYER AND KNIPLING PHOC. N. A. S. Geranium had the lowest resistance, 6.2, which represented an error of approximately 3 per cent. Leaf resistance may also be determined for leaf tissue in the psychrometer chamber using equation (5) for comparison with the resistances quoted in Table 2. The slope of the line defined by the equation is determined by the resistance of the leaf, since the other terms are constant for a particular psychrometer. Calculations from (5) indicate that privet leaf tissue in the psychrometer chamber has a resistance of approximately 15 sec cm"" 1, which agrees closety with the values in Table 2. The theoretical errors calculated from leaf resistance are approximately the same as those determined directly for the Richards technique by the isopiestic method. This is particularly apparent with geranium which had a low resistance and a correspondingly low error when measured by the isopiestic method. The theoretical errors do not agree with those of Rawlins, 1 who calculated errors of up to 90 per cent for philodendron on the basis of leaf permeability measurements. Our calculations indicate that the leaf permeabilities which he measured correspond to leaf resistances as high as 1600 sec cm." 1 for philodendron. The data presented in this paper indicate a resistance for philodendron of 21.5, which is similar to that of the other species in Table 2 and also similar to that measured by other workers 12 for other species having closed stomata. The error found for the Richards and Spanner methods does not agree with the conclusion of Barrs 3 that there is no error in these techniques. Since his comparisons were made with paired samples of tissue, variations between samples may have obscured differences between methods. Although error is present in the Richards and Spanner techniques, it is small enough for tissues with higher potentials so that both methods remain useful for many purposes. However, it becomes quite large in the absolute sense when measurements are made on tissues with lower potentials. In these cases, isopiestic determinations should be used. There are several advantages to the use of the isopiestic technique in addition to its lack of resistance error. For instance, calibration is unnecessary because the thermocouple is used simply as a "null point" or zero transfer detector when solutions of known potential are placed on the junction. Since the rate of vapor transfer is a straight line function of the potential difference between the thermocouple and leaf, the line may be determined by a minimum of two points and extrapolated to the isopiestic value without actually placing an isopiestic solution on the thermocouple. However, highest precision is obtained when one of the solutions has a potential which is close to that of the leaf. The method can be used over a wide range of vapor pressures. Tissues drier than 30 to 50 bars have often been difficult to measure with Richards and Spanner techniques because the drop lasts only a short time and the steady state may not occur. This is not a problem with isopiestic measurements, since the potential difference between the solution and the plant tissue may be kept small. The theory of the isopiestic technique and the error in water potential determinations with ps3rchrometers may be summarized by the following statements: 1. The stead3 r -state vapor transfer equation for Richards and Ogata thermocouples is the equation of a line. Leaf resistance influences thermocouple output whenever vapor transfer is not zero. The isopiestic technique, based on this equa-
8 VOL. 54, 1965 BOTANY: BOYER AND KNIPLING 1051 tion, is free of leaf resistance error because it measures potential at zero vapor transfer. 2. The Richards and Ogata technique contains a resistance error of approximately per cent. The Spanner technique contains a resistance error of per cent. For many studies, this amount of error may be tolerated. 3. Error measured directly by the isopiestic technique is the same as the theoretical error calculated from leaf resistance measurements for Richards and Ogata thermocouples. 4. In addition to resistance error, the isopiestic, Richards, and Spanner techniques are affected by respiration error, but each type of measurement may be corrected by measuring chamber temperature with a dry junction, as Barrs 4 has suggested. We are indebted to Dr. K. 11. Knoerr for his helpful suggestions. * This work was partially supported by the U.S. Forest Service, Southeastern Forest Experiment Station (E. B. K.), and by National Science Foundation grant GB-82 (J. S. B. and E. B. K.). t Present address: Connecticut Agricultural Experiment Station, New Haven, Connecticut. 1 E.awlins, S. L., Science, 146, 644 (1964). 2 Richards, L. A., and G. Ogata, Science, 128, 1089 (1958). 3 B a rrs, H. D., Science, 149, 63 (1965). ' Barrs, H. D., Australian J. Biol. Sci., 18, 36 (1965). 6 Spanner, D. C., J. Exptl. Bot., 2, 145 (1951); Monteith, J. L., and P. C. Owen, /. Sci. Inst., 35, 443 (1958). 6 The geometric mean area, a,,,, is 4.irr,ir c, where rj and r c are the radii of the drop and chamber, which are considered to approximate spheres. The resistance of the air (diffusion path length divided by the diffusivity of water vapor in air) applies only at a,,,. See Crank, J., The Mathematics of Diffusion (Oxford: Clarendon Press, 1956), p. 84; McAdams, W. H., Heat Transmission (New York: McGraw-Hill, 1954), p The proportionality constant, L, is evaluated by determining Co, c,i, and c n in the psychrometer chamber for solutions of known potential. The concentration CD may be obtained from tables giving saturation vapor concentrations of water at various temperatures. The concentration Cd is determined similarly from saturation values at drop temperature (see ref. 1). The concentration c<, is calculated from the potential of calibrating solutions (see ref. 8). 8 The exponential relationship between water vapor concentration and water potentials higher than 200 bars may be estimated by the first two terms of the exponential series, exp x = 1 + x +..., so that Vf I F*\ ' = <*exp- = «(!+ ), (9) where Co is the saturation vapor concentration at the temperature T. The concentration diference to ci of equation (4) therefore may be expressed as: cof co - ci = (* - *,) (10) 9 Species used: privet, Liguslrum japonicum, Thunb.; cotton, Gossypium hirsutum, L.; tobacco, Nicoliana tabacum, L.; tomato, Lycopersicum esculentum, Mill.; philodendron, Philodendron hastatum, C. Koch & Sello; magnolia, Magnolia grandiflora, L.; geranium, Pelargonium zonale, L. 10 Perry, J. H., Chemical Engineers Handbook (New York: McGraw-Hill, 1950), p Characteristics of our psychrometers: chamber approximates a sphere with 1.2-cm radius; spherical water drop with 0.1-cm radius; 1.1-cm diffusion path; r a = 4.3 sec cm" 1 ; a m = 1.5 cm 2 ; L = Kuiper, P. J. C., Meded. Landbouwhogesch. Wageningen, 61, 1 (1961); Slatyer, R. O., and J. F. Bierhuizen, Australian J. Biol. Sci., 17, 115 (1964); Slatyer, R. O., and O. T. Denmead, National Symposium on Water Resources, Uses and Management, Aust. Acad. Sci., C2 (1963); Van Bavel, C. H. M., F. S. Nakayama, and W. L. Ehrler, Plant Physiol., 40, 535 (1965).
Reprinted from PLANT PHYSIOLOGY, VoL 42, No. 10, October, 1967, pages J (PRINTED IN U ; S.A.)
 Reprinted from PLANT PHYSIOLOGY, VoL 42, No. 10, October, 1967, pages 1315- J 1320. (PRINTED IN U ; S.A.) Comparison of the Dye Method with the Thermocouple Psychrometer for Measuring Leaf Water Potentials
Reprinted from PLANT PHYSIOLOGY, VoL 42, No. 10, October, 1967, pages 1315- J 1320. (PRINTED IN U ; S.A.) Comparison of the Dye Method with the Thermocouple Psychrometer for Measuring Leaf Water Potentials
Why Measure the Water Status of Plants and Soils?
 Chapter 1 Why Measure the Water Status of Plants and Soils? Plant tissues contain large amounts of water, and even larger amounts must be supplied to replace the water lost in transpiration. A maize plant
Chapter 1 Why Measure the Water Status of Plants and Soils? Plant tissues contain large amounts of water, and even larger amounts must be supplied to replace the water lost in transpiration. A maize plant
Relationship between Leaf Water Potential and Photosynthesis in Rice Plants
 Relationship between Leaf Water Potential and Photosynthesis in Rice Plants By KUNI ISHIHARA and HIDEO SAITO Faculty of Agriculture, Tokyo University of Agriculture and Technology (Saiwaicho,Fuchu, Tokyo,
Relationship between Leaf Water Potential and Photosynthesis in Rice Plants By KUNI ISHIHARA and HIDEO SAITO Faculty of Agriculture, Tokyo University of Agriculture and Technology (Saiwaicho,Fuchu, Tokyo,
Relationship between leaf and xylem water potentials in rice plants
 Plant & CcllPhysiol. 19(7): 1289-1294 (1978) Short communication Relationship between leaf and xylem water potentials in rice plants Kuni Ishihara and Tadashi Hirasawa Laboratory of Crop Science, Faculty
Plant & CcllPhysiol. 19(7): 1289-1294 (1978) Short communication Relationship between leaf and xylem water potentials in rice plants Kuni Ishihara and Tadashi Hirasawa Laboratory of Crop Science, Faculty
EFFECTS OF PHENYL-MERCURIC ACETATE ON, STOMATAL AND CUTICULAR RESISTANCE TO TRANSPIRATION
 New PhytoL (1975) 75,47-52.. :: i vi EFFECTS OF PHENYL-MERCURIC ACETATE ON, STOMATAL AND CUTICULAR RESISTANCE TO TRANSPIRATION BY S. MORESHET Agricultural Research Organization, The Volcani Centre, Bet
New PhytoL (1975) 75,47-52.. :: i vi EFFECTS OF PHENYL-MERCURIC ACETATE ON, STOMATAL AND CUTICULAR RESISTANCE TO TRANSPIRATION BY S. MORESHET Agricultural Research Organization, The Volcani Centre, Bet
Ideal Gas Law and Absolute Zero
 Experiment IX Ideal Gas Law and Absolute Zero I. Purpose The purpose of this lab is to examine the relationship between the pressure, volume and temperature of air in a closed chamber. To do this, you
Experiment IX Ideal Gas Law and Absolute Zero I. Purpose The purpose of this lab is to examine the relationship between the pressure, volume and temperature of air in a closed chamber. To do this, you
Physics 2135 Exam 3 April 18, 2017
 Physics 2135 Exam 3 April 18, 2017 Exam Total / 200 Printed Name: Rec. Sec. Letter: Solutions for problems 6 to 10 must start from official starting equations. Show your work to receive credit for your
Physics 2135 Exam 3 April 18, 2017 Exam Total / 200 Printed Name: Rec. Sec. Letter: Solutions for problems 6 to 10 must start from official starting equations. Show your work to receive credit for your
Chapter 17. Fundamentals of Atmospheric Modeling
 Overhead Slides for Chapter 17 of Fundamentals of Atmospheric Modeling by Mark Z. Jacobson Department of Civil & Environmental Engineering Stanford University Stanford, CA 94305-4020 August 21, 1998 Mass
Overhead Slides for Chapter 17 of Fundamentals of Atmospheric Modeling by Mark Z. Jacobson Department of Civil & Environmental Engineering Stanford University Stanford, CA 94305-4020 August 21, 1998 Mass
Cold and freeze protection of blueberries in Florida. Dr. Dorota Z. Haman Agricultural and Biological Dept. University of Florida
 Cold and freeze protection of blueberries in Florida Dr. Dorota Z. Haman Agricultural and Biological Dept. University of Florida Frost versus Freeze Frost is a local condition which occurs in your area
Cold and freeze protection of blueberries in Florida Dr. Dorota Z. Haman Agricultural and Biological Dept. University of Florida Frost versus Freeze Frost is a local condition which occurs in your area
 Unit II Thermal Physics Introduction- Modes of Heat Transfer Normally there are three modes of transfer of heat from one place to another viz., conduction, convection and radiation. Conduction : Conduction
Unit II Thermal Physics Introduction- Modes of Heat Transfer Normally there are three modes of transfer of heat from one place to another viz., conduction, convection and radiation. Conduction : Conduction
Gas Thermometer and Absolute Zero
 Chapter 1 Gas Thermometer and Absolute Zero Name: Lab Partner: Section: 1.1 Purpose Construct a temperature scale and determine absolute zero temperature (the temperature at which molecular motion ceases).
Chapter 1 Gas Thermometer and Absolute Zero Name: Lab Partner: Section: 1.1 Purpose Construct a temperature scale and determine absolute zero temperature (the temperature at which molecular motion ceases).
E6 PROPERTIES OF GASES Flow-times, density, phase changes, solubility
 E6 PROPERTIES OF GASES Flow-times, density, phase changes, solubility Introduction Kinetic-Molecular Theory The kinetic energy of an object is dependent on its mass and its speed. The relationship, given
E6 PROPERTIES OF GASES Flow-times, density, phase changes, solubility Introduction Kinetic-Molecular Theory The kinetic energy of an object is dependent on its mass and its speed. The relationship, given
4. Thermometry. Temperature and Heat Flow Temperature Scales Thermometers
 4. Thermometry Measuring temperature by sensation is very imprecise. That is why we need a temperature scale and a thermometer to measure temperature more accurately. Temperature and Heat Flow Temperature
4. Thermometry Measuring temperature by sensation is very imprecise. That is why we need a temperature scale and a thermometer to measure temperature more accurately. Temperature and Heat Flow Temperature
FACTORS AFFECTING THE RATE OF PHOTOSYNTHESIS
 AP BIOLOGY CELLULAR ENERGETICS ACTIVITY #5 NAME DATE HOUR FACTORS AFFECTING THE RATE OF PHOTOSYNTHESIS OBJECTIVES: The purpose of this activity is to: Demonstrate how photosynthetic rates in different
AP BIOLOGY CELLULAR ENERGETICS ACTIVITY #5 NAME DATE HOUR FACTORS AFFECTING THE RATE OF PHOTOSYNTHESIS OBJECTIVES: The purpose of this activity is to: Demonstrate how photosynthetic rates in different
TIME COURSE OF PHOTOSYNTHETIC RHYTHMS IN PHASEOLUS VULGARIS L. AS RELATED TO CHANGES IN DEGREE OF STOMATAL OPENING 12
 37S JOSEF N. KNULL Vol. 64 TIME COURSE OF PHOTOSYNTHETIC RHYTHMS IN PHASEOLUS VULGARIS L. AS RELATED TO CHANGES IN DEGREE OF STOMATAL OPENING 12 GEORGE F. HOWE 3 Department of Botany and Plant Pathology,
37S JOSEF N. KNULL Vol. 64 TIME COURSE OF PHOTOSYNTHETIC RHYTHMS IN PHASEOLUS VULGARIS L. AS RELATED TO CHANGES IN DEGREE OF STOMATAL OPENING 12 GEORGE F. HOWE 3 Department of Botany and Plant Pathology,
Name Partner. Thermal Physics. Part I: Heat of Vaporization of Nitrogen. Introduction:
 Name Partner Thermal Physics Part I: Heat of Vaporization of Nitrogen Introduction: The heat of vaporization of a liquid, L v, is the energy required to vaporize (boil) a unit mass of substance. Thus if
Name Partner Thermal Physics Part I: Heat of Vaporization of Nitrogen Introduction: The heat of vaporization of a liquid, L v, is the energy required to vaporize (boil) a unit mass of substance. Thus if
Transient Heat Transfer Experiment. ME 331 Introduction to Heat Transfer. June 1 st, 2017
 Transient Heat Transfer Experiment ME 331 Introduction to Heat Transfer June 1 st, 2017 Abstract The lumped capacitance assumption for transient conduction was tested for three heated spheres; a gold plated
Transient Heat Transfer Experiment ME 331 Introduction to Heat Transfer June 1 st, 2017 Abstract The lumped capacitance assumption for transient conduction was tested for three heated spheres; a gold plated
GENERAL PHYSICS (3) LABORATORY PHYS 203 LAB STUDENT MANUAL
 Haifaa altoumah& Rabab Alfaraj By Haifaa altoumah& Rabab Alfaraj GENERAL PHYSICS (3) LABORATORY PHYS 203 LAB STUDENT MANUAL Name:-. ID# KING ABDULAZIZ UNIVERSITY PHYSICS DEPARMENT 1st semester 1430H Contents
Haifaa altoumah& Rabab Alfaraj By Haifaa altoumah& Rabab Alfaraj GENERAL PHYSICS (3) LABORATORY PHYS 203 LAB STUDENT MANUAL Name:-. ID# KING ABDULAZIZ UNIVERSITY PHYSICS DEPARMENT 1st semester 1430H Contents
Chapter 10, Thermal Physics
 CHAPTER 10 1. If it is given that 546 K equals 273 C, then it follows that 400 K equals: a. 127 C b. 150 C c. 473 C d. 1 200 C 2. A steel wire, 150 m long at 10 C, has a coefficient of linear expansion
CHAPTER 10 1. If it is given that 546 K equals 273 C, then it follows that 400 K equals: a. 127 C b. 150 C c. 473 C d. 1 200 C 2. A steel wire, 150 m long at 10 C, has a coefficient of linear expansion
AE 3051, Lab #16. Investigation of the Ideal Gas State Equation. By: George P. Burdell. Group E3
 AE 3051, Lab #16 Investigation of the Ideal Gas State Equation By: George P. Burdell Group E3 Summer Semester 000 Abstract The validity of the ideal gas equation of state was experimentally tested for
AE 3051, Lab #16 Investigation of the Ideal Gas State Equation By: George P. Burdell Group E3 Summer Semester 000 Abstract The validity of the ideal gas equation of state was experimentally tested for
The calibration of Hygrometer (LectureandTraining )
 The calibration of Hygrometer (LectureandTraining ) HAGIYA Satoshi 8Nov.2016 Outline 1.Measu rementmethods of midity(theory) Hu 2.Traceab ili ty in JMA 3.Cali brationmethodsof Hyg rometer 4.Practice (Ca
The calibration of Hygrometer (LectureandTraining ) HAGIYA Satoshi 8Nov.2016 Outline 1.Measu rementmethods of midity(theory) Hu 2.Traceab ili ty in JMA 3.Cali brationmethodsof Hyg rometer 4.Practice (Ca
PHYS102 Previous Exam Problems. Temperature, Heat & The First Law of Thermodynamics
 PHYS102 Previous Exam Problems CHAPTER 18 Temperature, Heat & The First Law of Thermodynamics Equilibrium & temperature scales Thermal expansion Exchange of heat First law of thermodynamics Heat conduction
PHYS102 Previous Exam Problems CHAPTER 18 Temperature, Heat & The First Law of Thermodynamics Equilibrium & temperature scales Thermal expansion Exchange of heat First law of thermodynamics Heat conduction
ASSESSMENT OF WATER STATUS IN TREES FROM MEASUREMENTS OF STOMATAL CONDUCTANCE AND WATER POTENTIAL
 No. 1 159 ASSESSMENT OF WATER STATUS IN TREES FROM MEASUREMENTS OF STOMATAL CONDUCTANCE AND WATER POTENTIAL DAVID WHITEHEAD Forest Research Institute, Rotorua, New Zealand (Received for publication 24
No. 1 159 ASSESSMENT OF WATER STATUS IN TREES FROM MEASUREMENTS OF STOMATAL CONDUCTANCE AND WATER POTENTIAL DAVID WHITEHEAD Forest Research Institute, Rotorua, New Zealand (Received for publication 24
Mlchio KANECHI, Naotsugu UCHIDA, Takeshl YASUDA and Tadashi YAMAGUCHI Graduate School of Science and Technology, Kobe University, Rokko, Kobe 657
 Japan. J. Trop. Agr. 32 (1) : 16-21, 1988 Relationships between Leaf Water Potential and Photosynthesis of Coffea arabica L. Grown under Various Environmental Conditions as Affected by Withholding Irrigation
Japan. J. Trop. Agr. 32 (1) : 16-21, 1988 Relationships between Leaf Water Potential and Photosynthesis of Coffea arabica L. Grown under Various Environmental Conditions as Affected by Withholding Irrigation
Perfect Gas law. Interface. Heater stand Beaker water
 Perfect Gas law Objectives a. To find the value of absolute zero temperature (0K). b. To find the value of universal gas constant (R) Apparatus Required Gas bulb Computer Interface Pressure-Temp Sersors
Perfect Gas law Objectives a. To find the value of absolute zero temperature (0K). b. To find the value of universal gas constant (R) Apparatus Required Gas bulb Computer Interface Pressure-Temp Sersors
n rr 'ih ifetl! * fn>p' ' -.A SEP <[ NOV 2 0 bba LIBRARY J2 2_ TWO PHASE FLOW ABOUT A STAGNATION POINT IN FILM BOILING RESEARC NOTE 51
 CONVAIR S C I E N T I F I C R E S E A R C H L A B O R A T O R Y TWO PHASE FLOW ABOUT A STAGNATION POINT IN FILM BOILING RESEARC NOTE 51 ifetl! * W. H. Gallaher JULY 1961 Distribution of this document is
CONVAIR S C I E N T I F I C R E S E A R C H L A B O R A T O R Y TWO PHASE FLOW ABOUT A STAGNATION POINT IN FILM BOILING RESEARC NOTE 51 ifetl! * W. H. Gallaher JULY 1961 Distribution of this document is
Lab 12 Pressure-Temperature Relationship in Gases
 Lab 12 Pressure-Temperature Relationship in Gases INTRODUCTION /PURPOSE/PLE LAB QUESTION Gases are made up of molecules that are in constant motion and exert pressure when they collide with the walls of
Lab 12 Pressure-Temperature Relationship in Gases INTRODUCTION /PURPOSE/PLE LAB QUESTION Gases are made up of molecules that are in constant motion and exert pressure when they collide with the walls of
Chapter 17 Temperature and heat
 Chapter 17 Temperature and heat 1 Temperature and Thermal Equilibrium When we speak of objects being hot and cold, we need to quantify this by some scientific method that is quantifiable and reproducible.
Chapter 17 Temperature and heat 1 Temperature and Thermal Equilibrium When we speak of objects being hot and cold, we need to quantify this by some scientific method that is quantifiable and reproducible.
Remember that C is a constant and ë and n are variables. This equation now fits the template of a straight line:
 CONVERTING NON-LINEAR GRAPHS INTO LINEAR GRAPHS Linear graphs have several important attributes. First, it is easy to recognize a graph that is linear. It is much more difficult to identify if a curved
CONVERTING NON-LINEAR GRAPHS INTO LINEAR GRAPHS Linear graphs have several important attributes. First, it is easy to recognize a graph that is linear. It is much more difficult to identify if a curved
Chapter 26 & 27. Electric Current and Direct- Current Circuits
 Chapter 26 & 27 Electric Current and Direct- Current Circuits Electric Current and Direct- Current Circuits Current and Motion of Charges Resistance and Ohm s Law Energy in Electric Circuits Combination
Chapter 26 & 27 Electric Current and Direct- Current Circuits Electric Current and Direct- Current Circuits Current and Motion of Charges Resistance and Ohm s Law Energy in Electric Circuits Combination
A METHOD FOR DETERMINING TOTAL CARBON DIOXIDE IN SMALL VOLUMES OF LIQUID
 J. Exp. Biol. (1970). 5*> 39S-4OO 395 JVith 3 text-figures JPrinted in Great Britain A METHOD FOR DETERMINING TOTAL CARBON DIOXIDE IN SMALL VOLUMES OF LIQUID BY COLIN LITTLE AND GARY RUSTON Department
J. Exp. Biol. (1970). 5*> 39S-4OO 395 JVith 3 text-figures JPrinted in Great Britain A METHOD FOR DETERMINING TOTAL CARBON DIOXIDE IN SMALL VOLUMES OF LIQUID BY COLIN LITTLE AND GARY RUSTON Department
Part I.
 Part I bblee@unimp . Introduction to Mass Transfer and Diffusion 2. Molecular Diffusion in Gasses 3. Molecular Diffusion in Liquids Part I 4. Molecular Diffusion in Biological Solutions and Gels 5. Molecular
Part I bblee@unimp . Introduction to Mass Transfer and Diffusion 2. Molecular Diffusion in Gasses 3. Molecular Diffusion in Liquids Part I 4. Molecular Diffusion in Biological Solutions and Gels 5. Molecular
LEAF WATER POTENTIAL AND STOMATAL CONDUCTANCE OF RUBBER (Hevea brasiliensis) AS INFLUENCED BY SOIL MOISTURE AND LEAF AGE LALANI SAMARAPPULI ABSTRACT
 LEAF WATER POTENTIAL AND STOMATAL CONDUCTANCE OF RUBBER (Hevea brasiliensis) AS INFLUENCED BY SOIL MOISTURE AND LEAF AGE J BY LALANI SAMARAPPULI ABSTRACT Stomatal conductance and transpiration and leaf
LEAF WATER POTENTIAL AND STOMATAL CONDUCTANCE OF RUBBER (Hevea brasiliensis) AS INFLUENCED BY SOIL MOISTURE AND LEAF AGE J BY LALANI SAMARAPPULI ABSTRACT Stomatal conductance and transpiration and leaf
Soil Water Atmosphere Plant (SWAP) Model: I. INTRODUCTION AND THEORETICAL BACKGROUND
 Soil Water Atmosphere Plant (SWAP) Model: I. INTRODUCTION AND THEORETICAL BACKGROUND Reinder A.Feddes Jos van Dam Joop Kroes Angel Utset, Main processes Rain fall / irrigation Transpiration Soil evaporation
Soil Water Atmosphere Plant (SWAP) Model: I. INTRODUCTION AND THEORETICAL BACKGROUND Reinder A.Feddes Jos van Dam Joop Kroes Angel Utset, Main processes Rain fall / irrigation Transpiration Soil evaporation
CHAPTER 2. SOIL-WATER POTENTIAL: CONCEPTS AND MEASUREMENT
 SSC107 Fall 2000 Chapter 2, Page - 1 - CHAPTER 2. SOIL-WATER POTENTIAL: CONCEPTS AND MEASUREMENT Contents: Transport mechanisms Water properties Definition of soil-water potential Measurement of soil-water
SSC107 Fall 2000 Chapter 2, Page - 1 - CHAPTER 2. SOIL-WATER POTENTIAL: CONCEPTS AND MEASUREMENT Contents: Transport mechanisms Water properties Definition of soil-water potential Measurement of soil-water
A NEW HEAT PIPE COOLING DEVICE
 DAAAM INTERNATIONAL SCIENTIFIC BOOK 2010 pp. 593-606 CHAPTER 52 A NEW HEAT PIPE COOLING DEVICE COMANESCU, D.; COMANESCU, A.; FILIPOIU, I. D. & ALIONTE, C.G. Abstract: The paper presents a specific application
DAAAM INTERNATIONAL SCIENTIFIC BOOK 2010 pp. 593-606 CHAPTER 52 A NEW HEAT PIPE COOLING DEVICE COMANESCU, D.; COMANESCU, A.; FILIPOIU, I. D. & ALIONTE, C.G. Abstract: The paper presents a specific application
Temperature distribution and heat flow across the combustion chamber wall.
 ΜΕΤΑΔΟΣΗ ΘΕΡΜΟΤΗΤΑΣ ΣΤΟΝ ΚΥΛΙΝΔΡΟ (J.B. Heywood: Internal Combustion Engine Fundamentals McGraw Hill 1988) Temperature distribution and heat flow across the combustion chamber wall. Throughout each engine
ΜΕΤΑΔΟΣΗ ΘΕΡΜΟΤΗΤΑΣ ΣΤΟΝ ΚΥΛΙΝΔΡΟ (J.B. Heywood: Internal Combustion Engine Fundamentals McGraw Hill 1988) Temperature distribution and heat flow across the combustion chamber wall. Throughout each engine
BIOLOGY 1230: BIOLOGY I LABORATORY FALL SEMESTER 2000 OSMOSIS. September 25, 2000
 BIOLOGY 1230: BIOLOGY I LABORATORY FALL SEMESTER 2000 OSMOSIS September 25, 2000 Instructor Version Osmosis: The Biology of Water Movement Introduction The drawing below is a cartoon of the results of
BIOLOGY 1230: BIOLOGY I LABORATORY FALL SEMESTER 2000 OSMOSIS September 25, 2000 Instructor Version Osmosis: The Biology of Water Movement Introduction The drawing below is a cartoon of the results of
Physical Vapor Deposition
 Physical Vapor Deposition EVAPORATION SPUTTERING Typically used for metallization of semiconductors. Both Evaporation & Sputtering are done in vacuum environments. Typically: y Evaporation Pressures are
Physical Vapor Deposition EVAPORATION SPUTTERING Typically used for metallization of semiconductors. Both Evaporation & Sputtering are done in vacuum environments. Typically: y Evaporation Pressures are
Evapotranspiration. Rabi H. Mohtar ABE 325
 Evapotranspiration Rabi H. Mohtar ABE 325 Introduction What is it? Factors affecting it? Why we need to estimate it? Latent heat of vaporization: Liquid gas o Energy needed o Cooling process Saturation
Evapotranspiration Rabi H. Mohtar ABE 325 Introduction What is it? Factors affecting it? Why we need to estimate it? Latent heat of vaporization: Liquid gas o Energy needed o Cooling process Saturation
Introduction to Heat and Mass Transfer. Week 5
 Introduction to Heat and Mass Transfer Week 5 Critical Resistance Thermal resistances due to conduction and convection in radial systems behave differently Depending on application, we want to either maximize
Introduction to Heat and Mass Transfer Week 5 Critical Resistance Thermal resistances due to conduction and convection in radial systems behave differently Depending on application, we want to either maximize
Topic 10: Transpiration, transport and support in plants
 Topic 10: Transpiration, transport and support in plants 1. Transpiration is A. the loss of water vapour from the surfaces of plants due to evaporation B. the gain of water vapour from the surfaces of
Topic 10: Transpiration, transport and support in plants 1. Transpiration is A. the loss of water vapour from the surfaces of plants due to evaporation B. the gain of water vapour from the surfaces of
Teacher s notes 15 Temperature volume relationship in a gas (Charles law, Gay-Lussac s law)
 Sensors: Loggers: Temperature, Rotary Motion Any EASYSENSE Physics Logging time: EasyLog Teacher s notes 15 Temperature volume relationship in a gas (Charles law, Gay-Lussac s law) Read The relationship
Sensors: Loggers: Temperature, Rotary Motion Any EASYSENSE Physics Logging time: EasyLog Teacher s notes 15 Temperature volume relationship in a gas (Charles law, Gay-Lussac s law) Read The relationship
(D) Blv/R Counterclockwise
 1. There is a counterclockwise current I in a circular loop of wire situated in an external magnetic field directed out of the page as shown above. The effect of the forces that act on this current is
1. There is a counterclockwise current I in a circular loop of wire situated in an external magnetic field directed out of the page as shown above. The effect of the forces that act on this current is
A Method, tor Determining the Slope. or Neutron Moisture Meter Calibration Curves. James E. Douglass
 Station Paper No. 154 December 1962 A Method, tor Determining the Slope or Neutron Moisture Meter Calibration Curves James E. Douglass U.S. Department of Agriculture-Forest Service Southeastern Forest
Station Paper No. 154 December 1962 A Method, tor Determining the Slope or Neutron Moisture Meter Calibration Curves James E. Douglass U.S. Department of Agriculture-Forest Service Southeastern Forest
Temperature and light as ecological factors for plants
 PLB/EVE 117 Plant Ecology Fall 2005 1 Temperature and light as ecological factors for plants I. Temperature as an environmental factor A. The influence of temperature as an environmental factor is pervasive
PLB/EVE 117 Plant Ecology Fall 2005 1 Temperature and light as ecological factors for plants I. Temperature as an environmental factor A. The influence of temperature as an environmental factor is pervasive
CHAPTER 4 THERMAL CONDUCTIVITY AND VISCOSITY MEASUREMENTS
 50 CHAPTER 4 THERMAL CONDUCTIVITY AND VISCOSITY MEASUREMENTS 4.1 INTRODUCTION In the development of any energy-efficient heat transfer fluids for enhanced heat transfer performance, in practical applications,
50 CHAPTER 4 THERMAL CONDUCTIVITY AND VISCOSITY MEASUREMENTS 4.1 INTRODUCTION In the development of any energy-efficient heat transfer fluids for enhanced heat transfer performance, in practical applications,
Name : Applied Physics II Exam One Winter Multiple Choice ( 7 Points ):
 Name : e-mail: Applied Physics II Exam One Winter 2006-2007 Multiple Choice ( 7 Points ): 1. Pure nitrogen gas is contained in a sealed tank containing a movable piston. The initial volume, pressure and
Name : e-mail: Applied Physics II Exam One Winter 2006-2007 Multiple Choice ( 7 Points ): 1. Pure nitrogen gas is contained in a sealed tank containing a movable piston. The initial volume, pressure and
A).5 atm B) 1 atm C) 1.5 atm D) 2 atm E) it is impossible to tell
 1. ne atmosphere is equivalent to A) 1.00 g ml 1 B) 22,400 ml ) 273 K D) 760. mmhg E) 298 K 2. A cylinder contains 2.50 L of air at a pressure of 5.00 atmospheres. At what volume, will the air exert a
1. ne atmosphere is equivalent to A) 1.00 g ml 1 B) 22,400 ml ) 273 K D) 760. mmhg E) 298 K 2. A cylinder contains 2.50 L of air at a pressure of 5.00 atmospheres. At what volume, will the air exert a
Greenhouse Steady State Energy Balance Model
 Greenhouse Steady State Energy Balance Model The energy balance for the greenhouse was obtained by applying energy conservation to the greenhouse system as a control volume and identifying the energy terms.
Greenhouse Steady State Energy Balance Model The energy balance for the greenhouse was obtained by applying energy conservation to the greenhouse system as a control volume and identifying the energy terms.
7.6 Stress in symmetrical elastic beam transmitting both shear force and bending moment
 7.6 Stress in symmetrical elastic beam transmitting both shear force and bending moment à It is more difficult to obtain an exact solution to this problem since the presence of the shear force means that
7.6 Stress in symmetrical elastic beam transmitting both shear force and bending moment à It is more difficult to obtain an exact solution to this problem since the presence of the shear force means that
IGNITABILITY ANALYSIS USING THE CONE CALORIMETER AND LIFT APPARATUS
 189 IGNITABILITY ANALYSIS USING THE CONE CALORIMETER AND LIFT APPARATUS Mark A. Dietenberger USDA Forest Service Forest Products Laboratory* Madison, WI 53705-2398 ABSTRACT The irradiance plotted as function
189 IGNITABILITY ANALYSIS USING THE CONE CALORIMETER AND LIFT APPARATUS Mark A. Dietenberger USDA Forest Service Forest Products Laboratory* Madison, WI 53705-2398 ABSTRACT The irradiance plotted as function
fr>uafcji *> \E % jw r"'''f^,""'i;- ~^H^^
 NAME DATE Carolina Transpiration Kit for AP Biology Imagine that your family has received a bouquet of cut flowers as a gift. You place the flowers in a vase with a small volume of water, and return the
NAME DATE Carolina Transpiration Kit for AP Biology Imagine that your family has received a bouquet of cut flowers as a gift. You place the flowers in a vase with a small volume of water, and return the
If there is convective heat transfer from outer surface to fluid maintained at T W.
 Heat Transfer 1. What are the different modes of heat transfer? Explain with examples. 2. State Fourier s Law of heat conduction? Write some of their applications. 3. State the effect of variation of temperature
Heat Transfer 1. What are the different modes of heat transfer? Explain with examples. 2. State Fourier s Law of heat conduction? Write some of their applications. 3. State the effect of variation of temperature
EQUIPMENT Beta spectrometer, vacuum pump, Cs-137 source, Geiger-Muller (G-M) tube, scalar
 Modern Physics Laboratory Beta Spectroscopy Experiment In this experiment, electrons emitted as a result of the radioactive beta decay of Cs-137 are measured as a function of their momentum by deflecting
Modern Physics Laboratory Beta Spectroscopy Experiment In this experiment, electrons emitted as a result of the radioactive beta decay of Cs-137 are measured as a function of their momentum by deflecting
CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES
 Thermodynamics: An Engineering Approach 8th Edition in SI Units Yunus A. Çengel, Michael A. Boles McGraw-Hill, 2015 CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES Lecture slides by Dr. Fawzi Elfghi
Thermodynamics: An Engineering Approach 8th Edition in SI Units Yunus A. Çengel, Michael A. Boles McGraw-Hill, 2015 CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES Lecture slides by Dr. Fawzi Elfghi
Chapiter VII: Ionization chamber
 Chapiter VII: Ionization chamber 1 Types of ionization chambers Sensitive volume: gas (most often air direct measurement of exposure) ionization chamber Sensitive volume: semiconductor (silicon, germanium,
Chapiter VII: Ionization chamber 1 Types of ionization chambers Sensitive volume: gas (most often air direct measurement of exposure) ionization chamber Sensitive volume: semiconductor (silicon, germanium,
Cryogenic Instrumentation I Thermometry OUTLINE Thermometry Pt (pure metal) Temperature Ranges of Thermometer Application Typical Resistive Thermal
 Cryogenic Instrumentation I 1. Thermometry 2. anges of Application 3. Constant Volume 4. Thermocouples 5. Time esponse Data 6. 4 Terminal esistance Measurement OUTLINE 8. Pt (pure metal) 9. Typical esistive
Cryogenic Instrumentation I 1. Thermometry 2. anges of Application 3. Constant Volume 4. Thermocouples 5. Time esponse Data 6. 4 Terminal esistance Measurement OUTLINE 8. Pt (pure metal) 9. Typical esistive
Moment of inertia of different bodies
 Moment of inertia of different bodies Aim: 1) Study moment of inertia of different bodies Objectives of the experiment 1. Measuring the period of oscillation of a thin transverse rod with weights on a
Moment of inertia of different bodies Aim: 1) Study moment of inertia of different bodies Objectives of the experiment 1. Measuring the period of oscillation of a thin transverse rod with weights on a
Statistical Physics. Problem Set 4: Kinetic Theory
 Statistical Physics xford hysics Second year physics course Dr A. A. Schekochihin and Prof. A. Boothroyd (with thanks to Prof. S. J. Blundell) Problem Set 4: Kinetic Theory PROBLEM SET 4: Collisions and
Statistical Physics xford hysics Second year physics course Dr A. A. Schekochihin and Prof. A. Boothroyd (with thanks to Prof. S. J. Blundell) Problem Set 4: Kinetic Theory PROBLEM SET 4: Collisions and
TEMPERATURES OBTAINED IN TIMBERS WHEN THE SURFACE TEMPERATURE IS CHANGED AFTER VARIOUS PERIODS OF HEATING
 TEMPERATURES OBTAINED IN TIMBERS WHEN THE SURFACE TEMPERATURE IS CHANGED AFTER VARIOUS PERIODS OF HEATING April 1946 No. R1609 UNITED STATES DEPARTMENT OF AGRICULTURE FOREST SERVICE FOREST PRODUCTS LABORATORY
TEMPERATURES OBTAINED IN TIMBERS WHEN THE SURFACE TEMPERATURE IS CHANGED AFTER VARIOUS PERIODS OF HEATING April 1946 No. R1609 UNITED STATES DEPARTMENT OF AGRICULTURE FOREST SERVICE FOREST PRODUCTS LABORATORY
Thermodynamics. We can summarize the four laws of thermodynamics as follows:
 Thermodynamics Objective: To investigate the zeroth and first laws of thermodynamics. To calculate properties such as specific heat. To investigate the ideal gas law. To become familiar with basic P-V
Thermodynamics Objective: To investigate the zeroth and first laws of thermodynamics. To calculate properties such as specific heat. To investigate the ideal gas law. To become familiar with basic P-V
DEHRADUN PUBLIC SCHOOL I TERM ASSIGNMENT SUBJECT- PHYSICS (042) CLASS -XII
 Chapter 1(Electric charges & Fields) DEHRADUN PUBLIC SCHOOL I TERM ASSIGNMENT 2016-17 SUBJECT- PHYSICS (042) CLASS -XII 1. Why do the electric field lines never cross each other? [2014] 2. If the total
Chapter 1(Electric charges & Fields) DEHRADUN PUBLIC SCHOOL I TERM ASSIGNMENT 2016-17 SUBJECT- PHYSICS (042) CLASS -XII 1. Why do the electric field lines never cross each other? [2014] 2. If the total
HEAT- I Part - A C D A B. Te m p. Heat input
 e m p HE- I Part -. solid material is supplied with heat at a constant rate. he temperature of the material is changing with heat input as shown in the graph. Study the graph carefully and answer the following
e m p HE- I Part -. solid material is supplied with heat at a constant rate. he temperature of the material is changing with heat input as shown in the graph. Study the graph carefully and answer the following
Modern Physics Laboratory Beta Spectroscopy Experiment
 Modern Physics Laboratory Beta Spectroscopy Experiment Josh Diamond and John Cummings Fall 2009 Abstract In this experiment, electrons emitted as a result of the radioactive beta decay of 137 55 Cs are
Modern Physics Laboratory Beta Spectroscopy Experiment Josh Diamond and John Cummings Fall 2009 Abstract In this experiment, electrons emitted as a result of the radioactive beta decay of 137 55 Cs are
VACUUM PUMPING METHODS
 VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
The table lists some functions of parts of a plant. Match the part of the plant (A, B, C or D) to its function by writing the letters in the table.
 Low Demand Questions QUESTIONSHEET 1 The diagram shows a flowering plant. A Name the parts labelled A, B, C and D. (c) (d) B C D A... B C... D [4] The table lists some functions of parts of a plant. Match
Low Demand Questions QUESTIONSHEET 1 The diagram shows a flowering plant. A Name the parts labelled A, B, C and D. (c) (d) B C D A... B C... D [4] The table lists some functions of parts of a plant. Match
EXPERIMENT #4 Freezing Points, Cooling Curves, and Molar Mass Determination
 OBJECTIVES: EXPERIMENT #4 Freezing Points, Cooling Curves, and Molar Mass Determination Observe temperature versus time and record data for pure acetic acid cooled in an ice-water bath Plot temperature
OBJECTIVES: EXPERIMENT #4 Freezing Points, Cooling Curves, and Molar Mass Determination Observe temperature versus time and record data for pure acetic acid cooled in an ice-water bath Plot temperature
7. The weather instrument below can be used to determine relative humidity.
 1. What is the dewpoint temperature when the dry-bulb temperature is 12 C and the wet-bulb temperature is 7 C? A) 1 C B) -2 C C) -5 C D) 4 C 2. A parcel of air has a dry-bulb temperature reading of 16
1. What is the dewpoint temperature when the dry-bulb temperature is 12 C and the wet-bulb temperature is 7 C? A) 1 C B) -2 C C) -5 C D) 4 C 2. A parcel of air has a dry-bulb temperature reading of 16
Apparatus to measure high-temperature thermal conductivity and thermoelectric power of small specimens
 Apparatus to measure high-temperature thermal conductivity and thermoelectric power of small specimens T. Dasgupta and A. M. Umarji a Materials Research Centre, Indian Institute of Science, Bangalore-560012,
Apparatus to measure high-temperature thermal conductivity and thermoelectric power of small specimens T. Dasgupta and A. M. Umarji a Materials Research Centre, Indian Institute of Science, Bangalore-560012,
Calculus Relationships in AP Physics C: Electricity and Magnetism
 C: Electricity This chapter focuses on some of the quantitative skills that are important in your C: Mechanics course. These are not all of the skills that you will learn, practice, and apply during the
C: Electricity This chapter focuses on some of the quantitative skills that are important in your C: Mechanics course. These are not all of the skills that you will learn, practice, and apply during the
5. ELECTRIC CURRENTS
 5. ELECTRIC CURRENTS TOPIC OUTLINE Section Recommended Time Giancoli Section 5.1 Potential Difference, Current, Resistance 5.2 Electric Circuits 3h 19.1, 19.2 6.2 Electric Field and Force 6.3 Magnetic
5. ELECTRIC CURRENTS TOPIC OUTLINE Section Recommended Time Giancoli Section 5.1 Potential Difference, Current, Resistance 5.2 Electric Circuits 3h 19.1, 19.2 6.2 Electric Field and Force 6.3 Magnetic
On the Cooling and Evaporative Powers o f the, as Determined by the.
 438 On the Cooling and Evaporative Powers o f the, as Determined by the. B y L eonard H ill, F.R.S., and D. H argood-a sh. (Department of Applied Physiology, Medical Research Committee.) (Received December
438 On the Cooling and Evaporative Powers o f the, as Determined by the. B y L eonard H ill, F.R.S., and D. H argood-a sh. (Department of Applied Physiology, Medical Research Committee.) (Received December
8. ELECTROCHEMICAL CELLS. n Electrode Reactions and Electrode Potentials a. H 2 2H + + 2e. Cl 2 + 2e 2Cl. H 2 + Cl 2 2H + + 2Cl ; z = 2
 8. ELECTROCHEMICAL CELLS n Electrode Reactions and Electrode Potentials 8.1. a. H H + + e Cl + e Cl H + Cl H + + Cl ; z = E = E RT F ln ( a H +a Cl ) b. Hg(l)+ Cl Hg Cl + e H + + e H Hg + H + + Cl Hg Cl
8. ELECTROCHEMICAL CELLS n Electrode Reactions and Electrode Potentials 8.1. a. H H + + e Cl + e Cl H + Cl H + + Cl ; z = E = E RT F ln ( a H +a Cl ) b. Hg(l)+ Cl Hg Cl + e H + + e H Hg + H + + Cl Hg Cl
Chapter 11 Ideal gases
 OCR (A) specifications: 5.4.10c,d,e,i,j,k Chapter 11 Ideal gases Worksheet Worked examples Practical: Determining absolute zero of temperature from the pressure law End-of-chapter test Marking scheme:
OCR (A) specifications: 5.4.10c,d,e,i,j,k Chapter 11 Ideal gases Worksheet Worked examples Practical: Determining absolute zero of temperature from the pressure law End-of-chapter test Marking scheme:
Synoptic Biology: Water Potential
 Number 225 Synoptic Biology: Water Potential This Factsheet reviews the very wide range of exam questions that test your understanding of water potential. Osmosis: the diffusion of water through a partially
Number 225 Synoptic Biology: Water Potential This Factsheet reviews the very wide range of exam questions that test your understanding of water potential. Osmosis: the diffusion of water through a partially
Influence of Air Space on Multi-layered Material Water Vapor Permeability Measurement
 Influence of Air Space on Multi-layered Material Water Vapor Measurement Yang Wu 1, Mavinkal K. Kumaran 2, Paul Fazio 1 Abstract The effect of interface between material layers in building envelope is
Influence of Air Space on Multi-layered Material Water Vapor Measurement Yang Wu 1, Mavinkal K. Kumaran 2, Paul Fazio 1 Abstract The effect of interface between material layers in building envelope is
Botany Math Problems. What question was being investigated with this experiment? What is the possible hypothesis for this experiment?
 Botany Math Problems Problem set 1 A particular species of flower will only flower at particular times of the year. An experiment was done with 100 plants. 50 of the plants were exposed to 14 hours of
Botany Math Problems Problem set 1 A particular species of flower will only flower at particular times of the year. An experiment was done with 100 plants. 50 of the plants were exposed to 14 hours of
Mineral-insulated thermocouples to DIN and EN
 Data Sheet 90.1210 (90.1221) Page 1/7 s to DIN 43 710 and EN 60 584 for temperatures from -200 to +1200 C flexible sheath with shock-proof measuring insert protection tube diameter from 0.5mm fast response
Data Sheet 90.1210 (90.1221) Page 1/7 s to DIN 43 710 and EN 60 584 for temperatures from -200 to +1200 C flexible sheath with shock-proof measuring insert protection tube diameter from 0.5mm fast response
A cylinder in a magnetic field (Jackson)
 Problem 1. A cylinder in a magnetic field (Jackson) A very long hollow cylinder of inner radius a and outer radius b of permeability µ is placed in an initially uniform magnetic field B o at right angles
Problem 1. A cylinder in a magnetic field (Jackson) A very long hollow cylinder of inner radius a and outer radius b of permeability µ is placed in an initially uniform magnetic field B o at right angles
A half submerged metal sphere (UIC comprehensive
 Problem 1. exam) A half submerged metal sphere (UIC comprehensive A very light neutral hollow metal spherical shell of mass m and radius a is slightly submerged by a distance b a below the surface of a
Problem 1. exam) A half submerged metal sphere (UIC comprehensive A very light neutral hollow metal spherical shell of mass m and radius a is slightly submerged by a distance b a below the surface of a
- -2C. PH 102 Exam I F13 F23 +3C +2C +5C -2C -1C
 Name Date 10Feb07 LeClair PH 102 Exam I 1. A free electron and a free proton are placed in an identical electric field. Which of the following statements are true? Check all that apply. Each particle is
Name Date 10Feb07 LeClair PH 102 Exam I 1. A free electron and a free proton are placed in an identical electric field. Which of the following statements are true? Check all that apply. Each particle is
AN AUTOMATED ADSORPTION ISOTHERM DEVICE
 AN AUTOMATED ADSORPTION ISOTHERM DEVICE ALLAN WILKINSON, HAROLD J. MOROWITZ, and WALTER LUND From the Department of Molecular Biophysics and Biochemistry and the Department of Physics, Yale University,
AN AUTOMATED ADSORPTION ISOTHERM DEVICE ALLAN WILKINSON, HAROLD J. MOROWITZ, and WALTER LUND From the Department of Molecular Biophysics and Biochemistry and the Department of Physics, Yale University,
PROBLEM 14.6 ( )( ) (b) Applying a species balance to a control volume about the hydrogen, dt 6 dt 6RAT dt 6RT dt
 PROBLEM 14.6 KNOWN: Pressure and temperature of hydrogen stored in a spherical steel tank of prescribed diameter and thickness. FIND: (a) Initial rate of hydrogen mass loss from the tank, (b) Initial rate
PROBLEM 14.6 KNOWN: Pressure and temperature of hydrogen stored in a spherical steel tank of prescribed diameter and thickness. FIND: (a) Initial rate of hydrogen mass loss from the tank, (b) Initial rate
MEASUREMENT OF WBGT INDEX IN AXIAL FLOW AS THE MAIN THERMODYNAMIC PARAMETERS VARY G.
 MEASUREMENT OF WBGT INDEX IN AXIAL FLOW AS THE MAIN THERMODYNAMIC PARAMETERS VARY G. Buonanno, M. Dell'Isola, A. Frattolillo and L. Vanoli Di.M.S.A.T. - Università di Cassino via Di Biasio 43-03043 Cassino
MEASUREMENT OF WBGT INDEX IN AXIAL FLOW AS THE MAIN THERMODYNAMIC PARAMETERS VARY G. Buonanno, M. Dell'Isola, A. Frattolillo and L. Vanoli Di.M.S.A.T. - Università di Cassino via Di Biasio 43-03043 Cassino
Physical Pharmacy. Diffusion
 Physical Pharmacy Diffusion Diffusion Diffusion is defined as a process of mass transfer of individual molecules of a substance brought about by random molecular motion and associated with a driving force
Physical Pharmacy Diffusion Diffusion Diffusion is defined as a process of mass transfer of individual molecules of a substance brought about by random molecular motion and associated with a driving force
Effect of 1-MCP on Water Relations Parameters of Well-Watered and Water-Stressed Cotton Plants
 Effect of 1-MCP on Water Relations Parameters of Well-Watered and Water-Stressed Cotton Plants Eduardo M. Kawakami, Derrick M. Oosterhuis, and John L. Snider 1 RESEARCH PROBLEM The cotton crop in the U.S.
Effect of 1-MCP on Water Relations Parameters of Well-Watered and Water-Stressed Cotton Plants Eduardo M. Kawakami, Derrick M. Oosterhuis, and John L. Snider 1 RESEARCH PROBLEM The cotton crop in the U.S.
screw clip air bubble Transpiration itself is not measured directly by a potometer....
 1. Transpiration is the loss of water from plants by evaporation. The diagram below shows a potometer, an apparatus used to estimate transpiration rates. water reservoir leafy shoot screw clip air bubble
1. Transpiration is the loss of water from plants by evaporation. The diagram below shows a potometer, an apparatus used to estimate transpiration rates. water reservoir leafy shoot screw clip air bubble
1. Mark the correct statement(s)
 1. Mark the correct statement(s) Figure to the right shows a mass measurement scale using a spring. 1.1 The span of the scale is a) 16 kg b) 21 kg c) 11 kg d) 5-16 kg 1.2 The range of the scale is a) 16
1. Mark the correct statement(s) Figure to the right shows a mass measurement scale using a spring. 1.1 The span of the scale is a) 16 kg b) 21 kg c) 11 kg d) 5-16 kg 1.2 The range of the scale is a) 16
Theory and calibration of the Schi0tz tonometer
 Theory and calibration of the Schi0tz tonometer VI. Experimental results of tonometric measurements Scale reading versus tonometric pressure Robert A. Moses and Walter J. Grodzki Tonometer scale reading
Theory and calibration of the Schi0tz tonometer VI. Experimental results of tonometric measurements Scale reading versus tonometric pressure Robert A. Moses and Walter J. Grodzki Tonometer scale reading
Vacuum techniques (down to 1 K)
 Vacuum techniques (down to 1 K) For isolation (deep Knudsen regime) liquid helium dewar / inner vacuum jacket Leak testing at level 10-11 Pa m3/s (10-10 mbar l/s) liquid helium dewar & transfer syphon
Vacuum techniques (down to 1 K) For isolation (deep Knudsen regime) liquid helium dewar / inner vacuum jacket Leak testing at level 10-11 Pa m3/s (10-10 mbar l/s) liquid helium dewar & transfer syphon
The Molecular Weight of Carbon Dioxide
 The Molecular Weight of Carbon Dioxide Objectives The objectives of this laboratory are as follows: To generate and collect a sample of carbon dioxide gas, then measure its pressure, volume, temperature
The Molecular Weight of Carbon Dioxide Objectives The objectives of this laboratory are as follows: To generate and collect a sample of carbon dioxide gas, then measure its pressure, volume, temperature
Introduction to Mass Transfer
 Introduction to Mass Transfer Introduction Three fundamental transfer processes: i) Momentum transfer ii) iii) Heat transfer Mass transfer Mass transfer may occur in a gas mixture, a liquid solution or
Introduction to Mass Transfer Introduction Three fundamental transfer processes: i) Momentum transfer ii) iii) Heat transfer Mass transfer Mass transfer may occur in a gas mixture, a liquid solution or
Chapter 4: Transient Heat Conduction. Dr Ali Jawarneh Department of Mechanical Engineering Hashemite University
 Chapter 4: Transient Heat Conduction Dr Ali Jawarneh Department of Mechanical Engineering Hashemite University Objectives When you finish studying this chapter, you should be able to: Assess when the spatial
Chapter 4: Transient Heat Conduction Dr Ali Jawarneh Department of Mechanical Engineering Hashemite University Objectives When you finish studying this chapter, you should be able to: Assess when the spatial
Contents. 1 Introduction 4. 2 Methods Results and Discussion 15
 Contents 1 Introduction 4 2 Methods 11 3 Results and Discussion 15 4 Appendices 21 4.1 Variable Definitions................................ 21 4.2 Sample Calculations............................... 22
Contents 1 Introduction 4 2 Methods 11 3 Results and Discussion 15 4 Appendices 21 4.1 Variable Definitions................................ 21 4.2 Sample Calculations............................... 22
Diffusional Growth of Liquid Phase Hydrometeros.
 Diffusional Growth of Liquid Phase Hydrometeros. I. Diffusional Growth of Liquid Phase Hydrometeors A. Basic concepts of diffusional growth. 1. To understand the diffusional growth of a droplet, we must
Diffusional Growth of Liquid Phase Hydrometeros. I. Diffusional Growth of Liquid Phase Hydrometeors A. Basic concepts of diffusional growth. 1. To understand the diffusional growth of a droplet, we must
TOWARDS A BETTER UNDERSTANDING OF SIMULATING THE MULTICOMPONENT SOLUBILITY OF NATURAL EXTRACTS IN SUPERCRITICAL CARBON DIOXIDE
 TOWARDS A BETTER UNDERSTANDING OF SIMULATING THE MULTICOMPONENT SOLUBILITY OF NATURAL EXTRACTS IN SUPERCRITICAL CARBON DIOXIDE S. Maharaj *, D. R. McGaw, M.Watson, R.Skeene and B. Sellier * Sharad.Maharaj@sta.uwi.edu
TOWARDS A BETTER UNDERSTANDING OF SIMULATING THE MULTICOMPONENT SOLUBILITY OF NATURAL EXTRACTS IN SUPERCRITICAL CARBON DIOXIDE S. Maharaj *, D. R. McGaw, M.Watson, R.Skeene and B. Sellier * Sharad.Maharaj@sta.uwi.edu
1. Transpiration may be defined as the loss of water vapour by diffusion from a plant to its environment.
 1. Transpiration may be defined as the loss of water vapour by diffusion from a plant to its environment. The diagram below shows apparatus that can be used to estimate transpiration rates of a leafy shoot.
1. Transpiration may be defined as the loss of water vapour by diffusion from a plant to its environment. The diagram below shows apparatus that can be used to estimate transpiration rates of a leafy shoot.
Heat and Mass Transfer Prof. S.P. Sukhatme Department of Mechanical Engineering Indian Institute of Technology, Bombay
 Heat and Mass Transfer Prof. S.P. Sukhatme Department of Mechanical Engineering Indian Institute of Technology, Bombay Lecture No. 18 Forced Convection-1 Welcome. We now begin our study of forced convection
Heat and Mass Transfer Prof. S.P. Sukhatme Department of Mechanical Engineering Indian Institute of Technology, Bombay Lecture No. 18 Forced Convection-1 Welcome. We now begin our study of forced convection
WATER RELATIONS OF WHEAT ALTERNATED BETWEEN TWO ROOT TEMPERATURES
 Nezo Phytol. (1979) 82, 89-96 89 WATER RELATIONS OF WHEAT ALTERNATED BETWEEN TWO ROOT TEMPERATURES BY M. B. KIRKHAM Department of Agrofiomy, Oklahoma State University^ StiHwater, Oklahoma 74074, U,S.A.
Nezo Phytol. (1979) 82, 89-96 89 WATER RELATIONS OF WHEAT ALTERNATED BETWEEN TWO ROOT TEMPERATURES BY M. B. KIRKHAM Department of Agrofiomy, Oklahoma State University^ StiHwater, Oklahoma 74074, U,S.A.
