Lecture 9: Molecular Orbital theory for hydrogen molecule ion
|
|
|
- Claude Mills
- 6 years ago
- Views:
Transcription
1 Lecture 9: Molecular Orbital theory for hydrogen molecule ion Molecular Orbital Theory for Hydrogen Molecule Ion We have seen that the Schrödinger equation cannot be solved for many electron systems. The H + 2 molecule ion is a molecule that has only one electron. However, there are 2 nuclei so it becomes a 3-particle problem. To solve this problem, we invoke an approximation that is known as the Born- Oppenheimer approximation wherein we assume that the electronic and nuclear degrees of freedom can be solved independently. Further, assuming that the kinetic energy of the nuclei are very small, we can solve the electronic problem at fixed internuclear separation as illustrated below. ( Ĥ(R A, R B, r) = 2 ( 2 A + 2 2m B) 2 2 r e2 + ) p 2m e 4piɛ 0 r A r B R Under the BO approximation, we have Ĥ el (r; R) = 2 2m e 2 r e2 4piɛ 0 ( + ) r A r B R The electronic wavefunction is to be treated as a function of the electronic coordinate r for fixed internuclear separation R. Thus the Schrödinger equation is given by Ĥ el ψ(r; R) = E(R)ψ)(r; R) We can imagine solving this for different R. If there is a bond, then this energy should be minimum for some value of R as shown in the figure. This gives the equilibrium bond length R e and the dissociation energy D e of the
2 r A r B R + + A B E R D e R e 2
3 molecule. We will briefly describe this physical picture of the bond using Molecular Orbital theory. According to MO theory, the electron in a molecule is located in orbitals (single-electron functions) called molecular orbitals. The molecular orbitals are different from the atomic orbitals since they are not centered at atomic nuclei. One way to construct the functions corresponding to molecular orbitals is to use linear combination of atomic orbitals (LCA0). For example, we can consider the linear combination of the s wavefunctions centered on both atoms. ψ MO ( r) = N(ψ sa ( r) ± ψ sb ( r)) Note that ψ sa is a function centered at nucleus A. Thus we have ψ sa ( r) = N e r A/a 0 and similarly for ψ sb. Assuming that the wavefunctions for ψ sa and ψ sb are normalized, we can calculate the value of N as follows = ψmo( r)ψ MO ( r)d r = N 2 ψsa( r)ψ sa ( r)d r + 3moreterms We can easily show for real orbitals that the terms lead to = N 2 (2 ± 2 ψsa( r)ψ sb ( r)d r) The last term is denoted by S, so we can write N = 2( ± S) where S denotes the overlap between the orbitals. Of the two orbitals, the one with the positive sign turns out to have lower energy than the s orbital and is called the bonding MO and the one with negative sign has higher energy than s orbital and is called the antibonding MO. ψ MO Bonding = (ψ sa ( r) + ψ sb ( r)) 2( + S) Thus we can say that for a given R, the two s orbitals combine to give one bonding and one antibonding MO. The bonding MO turns out to have lower energy, but you could only know this if you could calculate the energies which 3
4 A B A B is difficult. The bonding MO has higher electron density in the internuclear axis, unlike in the case of the antibonding MO. The s bonding orbital is symmetric to inversion about the center and is referred to as gerade or simply g for short. In fact we will write the previous equation in short form as σ g (s A + s B ) The convention for naming this orbital is σ g. The antibonding orbital is labelled as σ u. We could also combine the two 2s orbitals to form a 2σ g and 2σ u MOs. With p orbitals things get a little more complicated because the phase of the orbitals matters. Consider 2p z orbital. This has opposite signs in the different halves. Now, when combining wavefunctions, we have to see the sign. For example, the bonding orbital is denoted as 3σ u is given by 3σ g 2pz A 2pz B where we assume that the Z-axis is along the internuclear axis. The antibonding MO is given by 3σ u 2pz A + 2pz B The sigma orbitals are spherically symmetric about the internuclear axis. We can also have orbitals which do not satisfy this condition. For example, if we take a linear combiation of 2px orbitals located on the two atoms, we will see that π u 2px A + 2px B and similarly for the 2py orbibtals. These two MOs are degenerate. The antibonding MO is denoted by π g 2px A 2px B 4
5 In the Hydrogen molecule ion, there is only one electron. The ground state corresponds to the electron in the σ g orbital so this orbital is referred to as the Highest Occupied Molecular Orbital(HOMO). The lowest unocupied MO(LUMO) is the σ u. These MOs were calculated at fixed internuclear separation. We can qualitatively see what happens when the internuclear separation is changed and define R e and D e as we had before for the σ g MO. The antibonding MO does not show a minimum energy at some separation. This qualitative picture is borne out by numerical calculations. 5
Chemistry 2000 Lecture 1: Introduction to the molecular orbital theory
 Chemistry 2000 Lecture 1: Introduction to the molecular orbital theory Marc R. Roussel January 5, 2018 Marc R. Roussel Introduction to molecular orbitals January 5, 2018 1 / 24 Review: quantum mechanics
Chemistry 2000 Lecture 1: Introduction to the molecular orbital theory Marc R. Roussel January 5, 2018 Marc R. Roussel Introduction to molecular orbitals January 5, 2018 1 / 24 Review: quantum mechanics
Chemistry 6 (9 am section) Spring Covalent Bonding
 Chemistry 6 (9 am section) Spring 000 Covalent Bonding The stability of the bond in molecules such as H, O, N and F is associated with a sharing (equal) of the VALENCE ELECTRONS between the BONDED ATOMS.
Chemistry 6 (9 am section) Spring 000 Covalent Bonding The stability of the bond in molecules such as H, O, N and F is associated with a sharing (equal) of the VALENCE ELECTRONS between the BONDED ATOMS.
7. Arrange the molecular orbitals in order of increasing energy and add the electrons.
 Molecular Orbital Theory I. Introduction. A. Ideas. 1. Start with nuclei at their equilibrium positions. 2. onstruct a set of orbitals that cover the complete nuclear framework, called molecular orbitals
Molecular Orbital Theory I. Introduction. A. Ideas. 1. Start with nuclei at their equilibrium positions. 2. onstruct a set of orbitals that cover the complete nuclear framework, called molecular orbitals
VALENCE Hilary Term 2018
 VALENCE Hilary Term 2018 8 Lectures Prof M. Brouard Valence is the theory of the chemical bond Outline plan 1. The Born-Oppenheimer approximation 2. Bonding in H + 2 the LCAO approximation 3. Many electron
VALENCE Hilary Term 2018 8 Lectures Prof M. Brouard Valence is the theory of the chemical bond Outline plan 1. The Born-Oppenheimer approximation 2. Bonding in H + 2 the LCAO approximation 3. Many electron
CHEMISTRY Topic #1: Bonding What Holds Atoms Together? Spring 2012 Dr. Susan Lait
 CHEMISTRY 2000 Topic #1: Bonding What Holds Atoms Together? Spring 2012 Dr. Susan Lait Why Do Bonds Form? An energy diagram shows that a bond forms between two atoms if the overall energy of the system
CHEMISTRY 2000 Topic #1: Bonding What Holds Atoms Together? Spring 2012 Dr. Susan Lait Why Do Bonds Form? An energy diagram shows that a bond forms between two atoms if the overall energy of the system
Chemistry 2. Lecture 1 Quantum Mechanics in Chemistry
 Chemistry 2 Lecture 1 Quantum Mechanics in Chemistry Your lecturers 8am Assoc. Prof Timothy Schmidt Room 315 timothy.schmidt@sydney.edu.au 93512781 12pm Assoc. Prof. Adam J Bridgeman Room 222 adam.bridgeman@sydney.edu.au
Chemistry 2 Lecture 1 Quantum Mechanics in Chemistry Your lecturers 8am Assoc. Prof Timothy Schmidt Room 315 timothy.schmidt@sydney.edu.au 93512781 12pm Assoc. Prof. Adam J Bridgeman Room 222 adam.bridgeman@sydney.edu.au
Lecture 10. Born-Oppenheimer approximation LCAO-MO application to H + The potential energy surface MOs for diatomic molecules. NC State University
 Chemistry 431 Lecture 10 Diatomic molecules Born-Oppenheimer approximation LCAO-MO application to H + 2 The potential energy surface MOs for diatomic molecules NC State University Born-Oppenheimer approximation
Chemistry 431 Lecture 10 Diatomic molecules Born-Oppenheimer approximation LCAO-MO application to H + 2 The potential energy surface MOs for diatomic molecules NC State University Born-Oppenheimer approximation
Using Symmetry to Generate Molecular Orbital Diagrams
 Using Symmetry to Generate Molecular Orbital Diagrams review a few MO concepts generate MO for XH 2, H 2 O, SF 6 Formation of a bond occurs when electron density collects between the two bonded nuclei
Using Symmetry to Generate Molecular Orbital Diagrams review a few MO concepts generate MO for XH 2, H 2 O, SF 6 Formation of a bond occurs when electron density collects between the two bonded nuclei
QUANTUM MECHANICS AND MOLECULAR STRUCTURE
 6 QUANTUM MECHANICS AND MOLECULAR STRUCTURE 6.1 Quantum Picture of the Chemical Bond 6.2 Exact Molecular Orbital for the Simplest Molecule: H + 2 6.3 Molecular Orbital Theory and the Linear Combination
6 QUANTUM MECHANICS AND MOLECULAR STRUCTURE 6.1 Quantum Picture of the Chemical Bond 6.2 Exact Molecular Orbital for the Simplest Molecule: H + 2 6.3 Molecular Orbital Theory and the Linear Combination
σ u * 1s g - gerade u - ungerade * - antibonding σ g 1s
 One of these two states is a repulsive (dissociative) state. Other excited states can be constructed using linear combinations of other orbitals. Some will be binding and others will be repulsive. Thus
One of these two states is a repulsive (dissociative) state. Other excited states can be constructed using linear combinations of other orbitals. Some will be binding and others will be repulsive. Thus
Molecular orbitals, potential energy surfaces and symmetry
 Molecular orbitals, potential energy surfaces and symmetry mathematical presentation of molecular symmetry group theory spectroscopy valence theory molecular orbitals Wave functions Hamiltonian: electronic,
Molecular orbitals, potential energy surfaces and symmetry mathematical presentation of molecular symmetry group theory spectroscopy valence theory molecular orbitals Wave functions Hamiltonian: electronic,
5.111 Lecture Summary #13 Monday, October 6, 2014
 5.111 Lecture Summary #13 Monday, October 6, 2014 Readings for today: Section 3.8 3.11 Molecular Orbital Theory (Same in 5 th and 4 th ed.) Read for Lecture #14: Sections 3.4, 3.5, 3.6 and 3.7 Valence
5.111 Lecture Summary #13 Monday, October 6, 2014 Readings for today: Section 3.8 3.11 Molecular Orbital Theory (Same in 5 th and 4 th ed.) Read for Lecture #14: Sections 3.4, 3.5, 3.6 and 3.7 Valence
In this lecture we will understand how the molecular orbitals are formed from the interaction of atomic orbitals.
 Lecture 7 Title: Understanding of Molecular Orbital Page-1 In this lecture we will understand how the molecular orbitals are formed from the interaction of atomic orbitals. We will see how the electrons
Lecture 7 Title: Understanding of Molecular Orbital Page-1 In this lecture we will understand how the molecular orbitals are formed from the interaction of atomic orbitals. We will see how the electrons
Symmetry and Molecular Orbitals (I)
 Symmetry and Molecular Orbitals (I) Simple Bonding Model http://chiuserv.ac.nctu.edu.tw/~htchiu/chemistry/fall-2005/chemical-bonds.htm Lewis Structures Octet Rule Resonance Formal Charge Oxidation Number
Symmetry and Molecular Orbitals (I) Simple Bonding Model http://chiuserv.ac.nctu.edu.tw/~htchiu/chemistry/fall-2005/chemical-bonds.htm Lewis Structures Octet Rule Resonance Formal Charge Oxidation Number
MO theory is better for spectroscopy (Exited State Properties; Ionization)
 CHEM 2060 Lecture 25: MO Theory L25-1 Molecular Orbital Theory (MO theory) VB theory treats bonds as electron pairs. o There is a real emphasis on this point (over-emphasis actually). VB theory is very
CHEM 2060 Lecture 25: MO Theory L25-1 Molecular Orbital Theory (MO theory) VB theory treats bonds as electron pairs. o There is a real emphasis on this point (over-emphasis actually). VB theory is very
would represent a 1s orbital centered on the H atom and φ 2px )+ 1 r 2 sinθ
 Physical Chemistry for Engineers CHEM 4521 Homework: Molecular Structure (1) Consider the cation, HeH +. (a) Write the Hamiltonian for this system (there should be 10 terms). Indicate the physical meaning
Physical Chemistry for Engineers CHEM 4521 Homework: Molecular Structure (1) Consider the cation, HeH +. (a) Write the Hamiltonian for this system (there should be 10 terms). Indicate the physical meaning
Activity Molecular Orbital Theory
 Activity 201 9 Molecular Orbital Theory Directions: This Guided Learning Activity (GLA) discusses the Molecular Orbital Theory and its application to homonuclear diatomic molecules. Part A describes the
Activity 201 9 Molecular Orbital Theory Directions: This Guided Learning Activity (GLA) discusses the Molecular Orbital Theory and its application to homonuclear diatomic molecules. Part A describes the
MOLECULAR STRUCTURE. The general molecular Schrödinger equation, apart from electron spin effects, is. nn ee en
 MOLECULAR STRUCTURE The Born-Oppenheimer Approximation The general molecular Schrödinger equation, apart from electron spin effects, is ( ) + + V + V + V =E nn ee en T T ψ ψ n e where the operators in
MOLECULAR STRUCTURE The Born-Oppenheimer Approximation The general molecular Schrödinger equation, apart from electron spin effects, is ( ) + + V + V + V =E nn ee en T T ψ ψ n e where the operators in
CHEM-UA 127: Advanced General Chemistry I
 CHEM-UA 7: Advanced General Chemistry I I. LINEAR COMBINATION OF ATOMIC ORBITALS Linear combination of atomic orbitals (LCAO) is a simple method of quantum chemistry that yields a qualitative picture of
CHEM-UA 7: Advanced General Chemistry I I. LINEAR COMBINATION OF ATOMIC ORBITALS Linear combination of atomic orbitals (LCAO) is a simple method of quantum chemistry that yields a qualitative picture of
Learning Objectives and Worksheet VIII. Chemistry 1B-AL Fall Lectures (13-14) Molecular Orbital Theory of Covalent Bonding
 Learning Objectives and Worksheet VIII Chemistry 1B-AL Fall 2016 Lectures (13-14) Molecular Orbital Theory of Covalent Bonding WE WILL BE COVERING CHAPTER 14 IN A DIFFERENT ORDER THAN THE TEXT: first we
Learning Objectives and Worksheet VIII Chemistry 1B-AL Fall 2016 Lectures (13-14) Molecular Orbital Theory of Covalent Bonding WE WILL BE COVERING CHAPTER 14 IN A DIFFERENT ORDER THAN THE TEXT: first we
5.111 Principles of Chemical Science
 MIT OpenCourseWare http://ocw.mit.edu 5.111 Principles of Chemical Science Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.111 Lecture Summary
MIT OpenCourseWare http://ocw.mit.edu 5.111 Principles of Chemical Science Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.111 Lecture Summary
we have to deal simultaneously with the motion of the two heavy particles, the nuclei
 157 Lecture 6 We now turn to the structure of molecules. Our first cases will be the e- quantum mechanics of the two simplest molecules, the hydrogen molecular ion, H +, a r A r B one electron molecule,
157 Lecture 6 We now turn to the structure of molecules. Our first cases will be the e- quantum mechanics of the two simplest molecules, the hydrogen molecular ion, H +, a r A r B one electron molecule,
CHEM-UA 127: Advanced General Chemistry I
 1 CHEM-UA 127: Advanced General Chemistry I I. OVERVIEW OF MOLECULAR QUANTUM MECHANICS Using quantum mechanics to predict the chemical bonding patterns, optimal geometries, and physical and chemical properties
1 CHEM-UA 127: Advanced General Chemistry I I. OVERVIEW OF MOLECULAR QUANTUM MECHANICS Using quantum mechanics to predict the chemical bonding patterns, optimal geometries, and physical and chemical properties
General Physical Chemistry II
 General Physical Chemistry II Lecture 10 Aleksey Kocherzhenko October 7, 2014" Last time " promotion" Promotion and hybridization" [He] 2s 2 2p x 1 2p y 1 2p z0 " 2 unpaired electrons" [He] 2s 1 2p x 1
General Physical Chemistry II Lecture 10 Aleksey Kocherzhenko October 7, 2014" Last time " promotion" Promotion and hybridization" [He] 2s 2 2p x 1 2p y 1 2p z0 " 2 unpaired electrons" [He] 2s 1 2p x 1
Symmetry III: Molecular Orbital Theory. Reading: Shriver and Atkins and , 6.10
 Lecture 9 Symmetry III: Molecular Orbital Theory Reading: Shriver and Atkins 2.7-2.9 and g 6.6-6.7, 6.10 The orbitals of molecules H H The electron energy in each H atom is -13.6 ev below vacuum. What
Lecture 9 Symmetry III: Molecular Orbital Theory Reading: Shriver and Atkins 2.7-2.9 and g 6.6-6.7, 6.10 The orbitals of molecules H H The electron energy in each H atom is -13.6 ev below vacuum. What
Molecular Orbitals. Based on Inorganic Chemistry, Miessler and Tarr, 4 th edition, 2011, Pearson Prentice Hall
 Molecular Orbitals Based on Inorganic Chemistry, Miessler and Tarr, 4 th edition, 2011, Pearson Prentice Hall Images from Miessler and Tarr Inorganic Chemistry 2011 obtained from Pearson Education, Inc.
Molecular Orbitals Based on Inorganic Chemistry, Miessler and Tarr, 4 th edition, 2011, Pearson Prentice Hall Images from Miessler and Tarr Inorganic Chemistry 2011 obtained from Pearson Education, Inc.
PHYSICAL CHEMISTRY I. Chemical Bonds
 PHYSICAL CHEMISTRY I Chemical Bonds Review The QM description of bonds is quite good Capable of correctly calculating bond energies and reaction enthalpies However it is quite complicated and sometime
PHYSICAL CHEMISTRY I Chemical Bonds Review The QM description of bonds is quite good Capable of correctly calculating bond energies and reaction enthalpies However it is quite complicated and sometime
Chapter 10 Chemical Bonding II
 Chapter 10 Chemical Bonding II Valence Bond Theory Valence Bond Theory: A quantum mechanical model which shows how electron pairs are shared in a covalent bond. Bond forms between two atoms when the following
Chapter 10 Chemical Bonding II Valence Bond Theory Valence Bond Theory: A quantum mechanical model which shows how electron pairs are shared in a covalent bond. Bond forms between two atoms when the following
CHAPTER 11 MOLECULAR ORBITAL THEORY
 CHAPTER 11 MOLECULAR ORBITAL THEORY Molecular orbital theory is a conceptual extension of the orbital model, which was so successfully applied to atomic structure. As was once playfuly remarked, a molecue
CHAPTER 11 MOLECULAR ORBITAL THEORY Molecular orbital theory is a conceptual extension of the orbital model, which was so successfully applied to atomic structure. As was once playfuly remarked, a molecue
CHEM3023: Spins, Atoms and Molecules
 CHEM3023: Spins, Atoms and Molecules Lecture 3 The Born-Oppenheimer approximation C.-K. Skylaris Learning outcomes Separate molecular Hamiltonians to electronic and nuclear parts according to the Born-Oppenheimer
CHEM3023: Spins, Atoms and Molecules Lecture 3 The Born-Oppenheimer approximation C.-K. Skylaris Learning outcomes Separate molecular Hamiltonians to electronic and nuclear parts according to the Born-Oppenheimer
Lecture 19: Building Atoms and Molecules
 Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r y even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r y even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
Molecular-Orbital Theory
 Prof. Dr. I. Nasser atomic and molecular physics -551 (T-11) April 18, 01 Molecular-Orbital Theory You have to explain the following statements: 1- Helium is monatomic gas. - Oxygen molecule has a permanent
Prof. Dr. I. Nasser atomic and molecular physics -551 (T-11) April 18, 01 Molecular-Orbital Theory You have to explain the following statements: 1- Helium is monatomic gas. - Oxygen molecule has a permanent
Molecular Bond Theory
 Molecular Bond Theory Short comings of the localized electron model: electrons are not really localized so the concept of resonance was added no direct information about bond energies Molecular Orbital
Molecular Bond Theory Short comings of the localized electron model: electrons are not really localized so the concept of resonance was added no direct information about bond energies Molecular Orbital
CB VII. Molecular Orbital (MO) Theory. General. Basic Principles. Basic Ideas. further improvement on Lewis, VSEPR & VB theory;
 chem101/3, D1 fa010 po 14 1 CB VII Molecular Orbital (MO) Theory chem101/3, D1 fa010 po 14 General further improvement on Lewis, VSEPR & VB theory; resulting in better info on: bond energy bond order magnetic
chem101/3, D1 fa010 po 14 1 CB VII Molecular Orbital (MO) Theory chem101/3, D1 fa010 po 14 General further improvement on Lewis, VSEPR & VB theory; resulting in better info on: bond energy bond order magnetic
Lecture B6 Molecular Orbital Theory. Sometimes it's good to be alone.
 Lecture B6 Molecular Orbital Theory Sometimes it's good to be alone. Covalent Bond Theories 1. VSEPR (valence shell electron pair repulsion model). A set of empirical rules for predicting a molecular geometry
Lecture B6 Molecular Orbital Theory Sometimes it's good to be alone. Covalent Bond Theories 1. VSEPR (valence shell electron pair repulsion model). A set of empirical rules for predicting a molecular geometry
Electron States of Diatomic Molecules
 IISER Pune March 2018 Hamiltonian for a Diatomic Molecule The hamiltonian for a diatomic molecule can be considered to be made up of three terms Ĥ = ˆT N + ˆT el + ˆV where ˆT N is the kinetic energy operator
IISER Pune March 2018 Hamiltonian for a Diatomic Molecule The hamiltonian for a diatomic molecule can be considered to be made up of three terms Ĥ = ˆT N + ˆT el + ˆV where ˆT N is the kinetic energy operator
wbt Λ = 0, 1, 2, 3, Eq. (7.63)
 7.2.2 Classification of Electronic States For all diatomic molecules the coupling approximation which best describes electronic states is analogous to the Russell- Saunders approximation in atoms The orbital
7.2.2 Classification of Electronic States For all diatomic molecules the coupling approximation which best describes electronic states is analogous to the Russell- Saunders approximation in atoms The orbital
Midterm Exam I. CHEM 181: Introduction to Chemical Principles September 24, 2015 Key
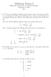 Midterm Exam I CHEM 8: Introduction to Chemical Principles September 24, 205 Key. A Li 2+ ion in an unknown, highly excited electronic state, first emits a photon at a wavelength of 4.36 µm, and following
Midterm Exam I CHEM 8: Introduction to Chemical Principles September 24, 205 Key. A Li 2+ ion in an unknown, highly excited electronic state, first emits a photon at a wavelength of 4.36 µm, and following
Lecture 19: Building Atoms and Molecules
 Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r ψ even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r ψ even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
We can model covalent bonding in molecules in essentially two ways:
 CHEM 2060 Lecture 22: VB Theory L22-1 PART FIVE: The Covalent Bond We can model covalent bonding in molecules in essentially two ways: 1. Localized Bonds (retains electron pair concept of Lewis Structures)
CHEM 2060 Lecture 22: VB Theory L22-1 PART FIVE: The Covalent Bond We can model covalent bonding in molecules in essentially two ways: 1. Localized Bonds (retains electron pair concept of Lewis Structures)
Molecular Bonding. Molecular Schrödinger equation. r - nuclei s - electrons. M j = mass of j th nucleus m 0 = mass of electron
 Molecular onding Molecular Schrödinger equation r - nuclei s - electrons 1 1 W V r s j i j1 M j m i1 M j = mass of j th nucleus m = mass of electron j i Laplace operator for nuclei Laplace operator for
Molecular onding Molecular Schrödinger equation r - nuclei s - electrons 1 1 W V r s j i j1 M j m i1 M j = mass of j th nucleus m = mass of electron j i Laplace operator for nuclei Laplace operator for
Molecular Structure Both atoms and molecules are quantum systems
 Molecular Structure Both atoms and molecules are quantum systems We need a method of describing molecules in a quantum mechanical way so that we can predict structure and properties The method we use is
Molecular Structure Both atoms and molecules are quantum systems We need a method of describing molecules in a quantum mechanical way so that we can predict structure and properties The method we use is
Rethinking Hybridization
 Rethinking Hybridization For more than 60 years, one of the most used concepts to come out of the valence bond model developed by Pauling was that of hybrid orbitals. The ideas of hybridization seemed
Rethinking Hybridization For more than 60 years, one of the most used concepts to come out of the valence bond model developed by Pauling was that of hybrid orbitals. The ideas of hybridization seemed
1s atomic orbital 2s atomic orbital 2s atomic orbital (with node) 2px orbital 2py orbital 2pz orbital
 Atomic Orbitals 1s atomic orbital 2s atomic orbital 2s atomic orbital (with node) 2px orbital 2py orbital 2pz orbital Valence Bond Theory and ybridized Atomic Orbitals Bonding in 2 1s 1s Atomic Orbital
Atomic Orbitals 1s atomic orbital 2s atomic orbital 2s atomic orbital (with node) 2px orbital 2py orbital 2pz orbital Valence Bond Theory and ybridized Atomic Orbitals Bonding in 2 1s 1s Atomic Orbital
Chapter 9 Molecular Geometry and Bonding Theories
 Chapter 9 Molecular Geometry and Bonding Theories 9.1 Molecular Shapes Lewis structures give atomic connectivity (which atoms are physically connected). By noting the number of bonding and nonbonding electron
Chapter 9 Molecular Geometry and Bonding Theories 9.1 Molecular Shapes Lewis structures give atomic connectivity (which atoms are physically connected). By noting the number of bonding and nonbonding electron
Wave Equations of Polyatomic Molecules
 Wave Equations of Polyatomic Molecules! Approximate wave functions are sought by combining atomic wave functions for the bonded atoms.! Several different approaches have been taken to constructing trial
Wave Equations of Polyatomic Molecules! Approximate wave functions are sought by combining atomic wave functions for the bonded atoms.! Several different approaches have been taken to constructing trial
Hybridization of Atomic Orbitals. (Chapter 1 in the Klein text)
 Hybridization of Atomic Orbitals (Chapter 1 in the Klein text) Basic Ideas The atomic structures, from the Periodic Table, of atoms such as C, N, and O do not adequately explain how these atoms use orbitals
Hybridization of Atomic Orbitals (Chapter 1 in the Klein text) Basic Ideas The atomic structures, from the Periodic Table, of atoms such as C, N, and O do not adequately explain how these atoms use orbitals
3: Many electrons. Orbital symmetries. l =2 1. m l
 3: Many electrons Orbital symmetries Atomic orbitals are labelled according to the principal quantum number, n, and the orbital angular momentum quantum number, l. Electrons in a diatomic molecule experience
3: Many electrons Orbital symmetries Atomic orbitals are labelled according to the principal quantum number, n, and the orbital angular momentum quantum number, l. Electrons in a diatomic molecule experience
Theoretical Chemistry - Level II - Practical Class Molecular Orbitals in Diatomics
 Theoretical Chemistry - Level II - Practical Class Molecular Orbitals in Diatomics Problem 1 Draw molecular orbital diagrams for O 2 and O 2 +. E / ev dioxygen molecule, O 2 dioxygenyl cation, O 2 + 25
Theoretical Chemistry - Level II - Practical Class Molecular Orbitals in Diatomics Problem 1 Draw molecular orbital diagrams for O 2 and O 2 +. E / ev dioxygen molecule, O 2 dioxygenyl cation, O 2 + 25
Be H. Delocalized Bonding. Localized Bonding. σ 2. σ 1. Two (sp-1s) Be-H σ bonds. The two σ bonding MO s in BeH 2. MO diagram for BeH 2
 The Delocalized Approach to Bonding: The localized models for bonding we have examined (Lewis and VBT) assume that all electrons are restricted to specific bonds between atoms or in lone pairs. In contrast,
The Delocalized Approach to Bonding: The localized models for bonding we have examined (Lewis and VBT) assume that all electrons are restricted to specific bonds between atoms or in lone pairs. In contrast,
ECE440 Nanoelectronics. Lecture 07 Atomic Orbitals
 ECE44 Nanoelectronics Lecture 7 Atomic Orbitals Atoms and atomic orbitals It is instructive to compare the simple model of a spherically symmetrical potential for r R V ( r) for r R and the simplest hydrogen
ECE44 Nanoelectronics Lecture 7 Atomic Orbitals Atoms and atomic orbitals It is instructive to compare the simple model of a spherically symmetrical potential for r R V ( r) for r R and the simplest hydrogen
Orbital approximation
 Orbital approximation Assign the electrons to an atomic orbital and a spin Construct an antisymmetrized wave function using a Slater determinant evaluate the energy with the Hamiltonian that includes the
Orbital approximation Assign the electrons to an atomic orbital and a spin Construct an antisymmetrized wave function using a Slater determinant evaluate the energy with the Hamiltonian that includes the
Activity Molecular Orbital Theory
 Activity 201 9 Molecular Orbital Theory Directions: This Guided Learning Activity (GLA) discusses the Molecular Orbital Theory and its application to homonuclear diatomic molecules. Part A describes the
Activity 201 9 Molecular Orbital Theory Directions: This Guided Learning Activity (GLA) discusses the Molecular Orbital Theory and its application to homonuclear diatomic molecules. Part A describes the
Chemistry 1B, Fall 2012 Lectures 15-16
 Chemistry 1B Fall 2012 Quantum Mechanics of the Covalent Bond for chapter 14 animations and links see: http://switkes.chemistry.ucsc.edu/teaching/chem1b/www_other_links/ch14_links.htm 1 LISTEN UP!!! WE
Chemistry 1B Fall 2012 Quantum Mechanics of the Covalent Bond for chapter 14 animations and links see: http://switkes.chemistry.ucsc.edu/teaching/chem1b/www_other_links/ch14_links.htm 1 LISTEN UP!!! WE
Chemistry 1B, Fall 2013 Lectures 15-16
 Chemistry 1, Fall 2013 Lectures 1516 Chemistry 1 Fall 2013 Lectures 1516 Quantum Mechanics of the Covalent ond LISTEN UP!!! WE WILL E COVERING SECOND PRT OF CHPTER 14 (pp 676688) FIRST You will go CRZY
Chemistry 1, Fall 2013 Lectures 1516 Chemistry 1 Fall 2013 Lectures 1516 Quantum Mechanics of the Covalent ond LISTEN UP!!! WE WILL E COVERING SECOND PRT OF CHPTER 14 (pp 676688) FIRST You will go CRZY
2 Electronic structure theory
 Electronic structure theory. Generalities.. Born-Oppenheimer approximation revisited In Sec..3 (lecture 3) the Born-Oppenheimer approximation was introduced (see also, for instance, [Tannor.]). We are
Electronic structure theory. Generalities.. Born-Oppenheimer approximation revisited In Sec..3 (lecture 3) the Born-Oppenheimer approximation was introduced (see also, for instance, [Tannor.]). We are
Molecular energy levels
 Molecular energy levels Hierarchy of motions and energies in molecules The different types of motion in a molecule (electronic, vibrational, rotational,: : :) take place on different time scales and are
Molecular energy levels Hierarchy of motions and energies in molecules The different types of motion in a molecule (electronic, vibrational, rotational,: : :) take place on different time scales and are
Diatomic Molecules. 14th May Chemical Bonds in Diatomic Molecules: Overlaps and Delocalization of Electrons
 Diatomic Molecules 14th May 2009 1 Chemical Bonds in Diatomic Molecules: Overlaps and Delocalization of Electrons 1.1 H + 2 Molecule Consider the process where 2 atomic nuclei and associated electron (1
Diatomic Molecules 14th May 2009 1 Chemical Bonds in Diatomic Molecules: Overlaps and Delocalization of Electrons 1.1 H + 2 Molecule Consider the process where 2 atomic nuclei and associated electron (1
Lecture 6. Tight-binding model
 Lecture 6 Tight-binding model In Lecture 3 we discussed the Krönig-Penny model and we have seen that, depending on the strength of the potential barrier inside the unit cell, the electrons can behave like
Lecture 6 Tight-binding model In Lecture 3 we discussed the Krönig-Penny model and we have seen that, depending on the strength of the potential barrier inside the unit cell, the electrons can behave like
Quantum mechanics can be used to calculate any property of a molecule. The energy E of a wavefunction Ψ evaluated for the Hamiltonian H is,
 Chapter : Molecules Quantum mechanics can be used to calculate any property of a molecule The energy E of a wavefunction Ψ evaluated for the Hamiltonian H is, E = Ψ H Ψ Ψ Ψ 1) At first this seems like
Chapter : Molecules Quantum mechanics can be used to calculate any property of a molecule The energy E of a wavefunction Ψ evaluated for the Hamiltonian H is, E = Ψ H Ψ Ψ Ψ 1) At first this seems like
(1/2) M α 2 α, ˆTe = i. 1 r i r j, ˆV NN = α>β
 Chemistry 26 Spectroscopy Week # The Born-Oppenheimer Approximation, H + 2. Born-Oppenheimer approximation As for atoms, all information about a molecule is contained in the wave function Ψ, which is the
Chemistry 26 Spectroscopy Week # The Born-Oppenheimer Approximation, H + 2. Born-Oppenheimer approximation As for atoms, all information about a molecule is contained in the wave function Ψ, which is the
The symmetry properties & relative energies of atomic orbitals determine how they react to form molecular orbitals. These molecular orbitals are then
 1 The symmetry properties & relative energies of atomic orbitals determine how they react to form molecular orbitals. These molecular orbitals are then filled with the available electrons according to
1 The symmetry properties & relative energies of atomic orbitals determine how they react to form molecular orbitals. These molecular orbitals are then filled with the available electrons according to
Born-Oppenheimer Approximation
 Born-Oppenheimer Approximation Adiabatic Assumption: Nuclei move so much more slowly than electron that the electrons that the electrons are assumed to be obtained if the nuclear kinetic energy is ignored,
Born-Oppenheimer Approximation Adiabatic Assumption: Nuclei move so much more slowly than electron that the electrons that the electrons are assumed to be obtained if the nuclear kinetic energy is ignored,
Lecture #30 page 1. Valence band approach: Molecular wavefunction described in terms of 1-electron atomic orbitals
 5.6 4 Lecture #3 page MOLCUL: MOLCULAR ORBITAL DCRIPTION Valence band approach: Molecular wavefunction described in terms of -ectron atomic orbitals Molecular orbital approach: Construct new set of -ectron
5.6 4 Lecture #3 page MOLCUL: MOLCULAR ORBITAL DCRIPTION Valence band approach: Molecular wavefunction described in terms of -ectron atomic orbitals Molecular orbital approach: Construct new set of -ectron
Molecular Orbital Theory
 Molecular Orbital Theory Paramagnetic properties of O 2 pranjoto utomo Covalent Bonding Theory Valence Bond Theory useful for deriving shapes/polarity simple but inaccurate/deficient Molecular Orbital
Molecular Orbital Theory Paramagnetic properties of O 2 pranjoto utomo Covalent Bonding Theory Valence Bond Theory useful for deriving shapes/polarity simple but inaccurate/deficient Molecular Orbital
From Atoms to Solids. Outline. - Atomic and Molecular Wavefunctions - Molecular Hydrogen - Benzene
 From Atoms to Solids Outline - Atomic and Molecular Wavefunctions - Molecular Hydrogen - Benzene 1 A Simple Approximation for an Atom Let s represent the atom in space by its Coulomb potential centered
From Atoms to Solids Outline - Atomic and Molecular Wavefunctions - Molecular Hydrogen - Benzene 1 A Simple Approximation for an Atom Let s represent the atom in space by its Coulomb potential centered
Chemical Bonding. Lewis Theory-VSEPR Valence Bond Theory Molecular Orbital Theory
 Chemical Bonding Lewis Theory-VSEPR Valence Bond Theory Molecular Orbital Theory Problems with Valence Bond Theory VB theory predicts properties better than Lewis theory bonding schemes, bond strengths,
Chemical Bonding Lewis Theory-VSEPR Valence Bond Theory Molecular Orbital Theory Problems with Valence Bond Theory VB theory predicts properties better than Lewis theory bonding schemes, bond strengths,
Molecular Orbital Approach to Bonding
 Molecular Orbital Approach to Bonding Chemistry 362; spring 2019 Marcetta Y. Darensbourg, Professor Xuemei Yang, Graduate Assistant Kyle Burns, Graduate Assistant The following slides were modified from
Molecular Orbital Approach to Bonding Chemistry 362; spring 2019 Marcetta Y. Darensbourg, Professor Xuemei Yang, Graduate Assistant Kyle Burns, Graduate Assistant The following slides were modified from
Physical Chemistry I Fall 2016 Second Hour Exam (100 points) Name:
 Physical Chemistry I Fall 2016 Second Hour Exam (100 points) Name: (20 points) 1. Quantum calculations suggest that the molecule U 2 H 2 is planar and has symmetry D 2h. D 2h E C 2 (z) C 2 (y) C 2 (x)
Physical Chemistry I Fall 2016 Second Hour Exam (100 points) Name: (20 points) 1. Quantum calculations suggest that the molecule U 2 H 2 is planar and has symmetry D 2h. D 2h E C 2 (z) C 2 (y) C 2 (x)
5.61 Physical Chemistry Exam III 11/29/12. MASSACHUSETTS INSTITUTE OF TECHNOLOGY Department of Chemistry Chemistry Physical Chemistry.
 MASSACHUSETTS INSTITUTE OF TECHNOLOGY Department of Chemistry Chemistry - 5.61 Physical Chemistry Exam III (1) PRINT your name on the cover page. (2) It is suggested that you READ THE ENTIRE EXAM before
MASSACHUSETTS INSTITUTE OF TECHNOLOGY Department of Chemistry Chemistry - 5.61 Physical Chemistry Exam III (1) PRINT your name on the cover page. (2) It is suggested that you READ THE ENTIRE EXAM before
Chemical Bonding & Structure
 Chemical Bonding & Structure Further aspects of covalent bonding and structure Hybridization Ms. Thompson - HL Chemistry Wooster High School Topic 14.2 Hybridization A hybrid orbital results from the mixing
Chemical Bonding & Structure Further aspects of covalent bonding and structure Hybridization Ms. Thompson - HL Chemistry Wooster High School Topic 14.2 Hybridization A hybrid orbital results from the mixing
Be H. Delocalized Bonding. Localized Bonding. σ 2. σ 1. Two (sp-1s) Be-H σ bonds. The two σ bonding MO s in BeH 2. MO diagram for BeH 2
 The Delocalized Approach to Bonding: The localized models for bonding we have examined (Lewis and VBT) assume that all electrons are restricted to specific bonds between atoms or in lone pairs. In contrast,
The Delocalized Approach to Bonding: The localized models for bonding we have examined (Lewis and VBT) assume that all electrons are restricted to specific bonds between atoms or in lone pairs. In contrast,
Structure of diatomic molecules
 Structure of diatomic molecules January 8, 00 1 Nature of molecules; energies of molecular motions Molecules are of course atoms that are held together by shared valence electrons. That is, most of each
Structure of diatomic molecules January 8, 00 1 Nature of molecules; energies of molecular motions Molecules are of course atoms that are held together by shared valence electrons. That is, most of each
Molecular Orbital Theory
 Molecular Orbital Theory While FMO theory allows prediction of reactions (by thermodynamics, regio or stereochemistry), all predictions seen so far have been qualitative We have predicted that HOMO or
Molecular Orbital Theory While FMO theory allows prediction of reactions (by thermodynamics, regio or stereochemistry), all predictions seen so far have been qualitative We have predicted that HOMO or
Chapter IV: Electronic Spectroscopy of diatomic molecules
 Chapter IV: Electronic Spectroscopy of diatomic molecules IV.2.1 Molecular orbitals IV.2.1.1. Homonuclear diatomic molecules The molecular orbital (MO) approach to the electronic structure of diatomic
Chapter IV: Electronic Spectroscopy of diatomic molecules IV.2.1 Molecular orbitals IV.2.1.1. Homonuclear diatomic molecules The molecular orbital (MO) approach to the electronic structure of diatomic
( R) MOLECULES: VALENCE BOND DESCRIPTION VALENCE BOND APPROXIMATION METHOD. Example: H 2. at some fixed R, repeat for many different values
 5.6 004 Lecture #9 page MOLECULES: VLENCE OND DESCRIPTION VLENCE OND PPROXIMTION METHOD Example: H H = + + r r r r r R Determine E ( R) at some fixed R, repeat for many different values Electronic Schrodinger
5.6 004 Lecture #9 page MOLECULES: VLENCE OND DESCRIPTION VLENCE OND PPROXIMTION METHOD Example: H H = + + r r r r r R Determine E ( R) at some fixed R, repeat for many different values Electronic Schrodinger
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer. Lecture 17, March 1, 2006
 Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer Lecture 17, March 1, 2006 (Some material in this lecture has been adapted from Cramer, C. J.
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer Lecture 17, March 1, 2006 (Some material in this lecture has been adapted from Cramer, C. J.
Exp. 4. Quantum Chemical calculation: The potential energy curves and the orbitals of H2 +
 Exp. 4. Quantum Chemical calculation: The potential energy curves and the orbitals of H2 + 1. Objectives Quantum chemical solvers are used to obtain the energy and the orbitals of the simplest molecules
Exp. 4. Quantum Chemical calculation: The potential energy curves and the orbitals of H2 + 1. Objectives Quantum chemical solvers are used to obtain the energy and the orbitals of the simplest molecules
Lecture 14 Chemistry 362 M. Darensbourg 2017 Spring term. Molecular orbitals for diatomics
 Lecture 14 Chemistry 362 M. Darensbourg 2017 Spring term Molecular orbitals for diatomics Molecular Orbital Theory of the Chemical Bond Simplest example - H 2 : two H atoms H A and H B Only two a.o.'s
Lecture 14 Chemistry 362 M. Darensbourg 2017 Spring term Molecular orbitals for diatomics Molecular Orbital Theory of the Chemical Bond Simplest example - H 2 : two H atoms H A and H B Only two a.o.'s
Page III-8-1 / Chapter Eight Lecture Notes MAR. Two s orbitals overlap. One s & one p. overlap. Two p orbitals. overlap MAR
 Bonding and Molecular Structure: Orbital ybridization and Molecular Orbitals Chapter 8 Page III-8-1 / Chapter Eight Lecture Notes Advanced Theories of Chemical Bonding Chemistry 222 Professor Michael Russell
Bonding and Molecular Structure: Orbital ybridization and Molecular Orbitals Chapter 8 Page III-8-1 / Chapter Eight Lecture Notes Advanced Theories of Chemical Bonding Chemistry 222 Professor Michael Russell
Wave Mechanical Model of Atom. Electron Configuration Pictures. The energy of the level increases as the value of n increases.
 Wave Mechanical Model of Atom Electron Configuration Pictures Mrs. Dormer Organic Chemistry Atoms have a series of energy levels called principal energy levels These are designated by whole numbers (symbolized
Wave Mechanical Model of Atom Electron Configuration Pictures Mrs. Dormer Organic Chemistry Atoms have a series of energy levels called principal energy levels These are designated by whole numbers (symbolized
Chapter 18 Molecular orbitals and spectroscopy Conjugation of bonds and resonance structures
 Chapter 18 Molecular orbitals and spectroscopy 18.1 Diatomic molecules 18.2 Polyatomic molecules 18.3 Conjugation of bonds and resonance structures 18.4 The interaction of light and matter (spectroscopy)
Chapter 18 Molecular orbitals and spectroscopy 18.1 Diatomic molecules 18.2 Polyatomic molecules 18.3 Conjugation of bonds and resonance structures 18.4 The interaction of light and matter (spectroscopy)
Chapter 9. Molecular Geometry and Bonding Theories
 Chapter 9. Molecular Geometry and Bonding Theories PART I Molecular Shapes Lewis structures give atomic connectivity: they tell us which atoms are physically connected to which atoms. The shape of a molecule
Chapter 9. Molecular Geometry and Bonding Theories PART I Molecular Shapes Lewis structures give atomic connectivity: they tell us which atoms are physically connected to which atoms. The shape of a molecule
23 The Born-Oppenheimer approximation, the Many Electron Hamiltonian and the molecular Schrödinger Equation M I
 23 The Born-Oppenheimer approximation, the Many Electron Hamiltonian and the molecular Schrödinger Equation 1. Now we will write down the Hamiltonian for a molecular system comprising N nuclei and n electrons.
23 The Born-Oppenheimer approximation, the Many Electron Hamiltonian and the molecular Schrödinger Equation 1. Now we will write down the Hamiltonian for a molecular system comprising N nuclei and n electrons.
Chemistry: The Central Science. Chapter 9: Molecular Geometry and Bonding Theory
 Chemistry: The Central Science Chapter 9: Molecular Geometry and Bonding Theory The shape and size of a molecule of a particular substance, together with the strength and polarity of its bonds, largely
Chemistry: The Central Science Chapter 9: Molecular Geometry and Bonding Theory The shape and size of a molecule of a particular substance, together with the strength and polarity of its bonds, largely
Bonding and Physical Properties The Molecular Orbital Theory
 Bonding and Physical Properties The Molecular Orbital Theory Ø Developed by F. Hund and R. S. Mulliken in 1932 Ø Diagram of molecular energy levels Ø Magnetic and spectral properties Paramagnetic vs. Diamagnetic
Bonding and Physical Properties The Molecular Orbital Theory Ø Developed by F. Hund and R. S. Mulliken in 1932 Ø Diagram of molecular energy levels Ø Magnetic and spectral properties Paramagnetic vs. Diamagnetic
NH 3 H 2 O N 2. Why do they make chemical bonds? Molecular Orbitals
 N 2 NH 3 H 2 O Why do they make chemical bonds? 5 Molecular Orbitals Why do they make chemical bonds? Stabilization Bond energy Types of Chemical Bonds Metallic Bond Ionic Bond Covalent Bond Covalent Bond
N 2 NH 3 H 2 O Why do they make chemical bonds? 5 Molecular Orbitals Why do they make chemical bonds? Stabilization Bond energy Types of Chemical Bonds Metallic Bond Ionic Bond Covalent Bond Covalent Bond
Valence bond theory accounts, at least qualitatively, for the stability of the covalent bond in terms of overlapping atomic orbitals.
 Molecular Orbital Theory Valence bond theory accounts, at least qualitatively, for the stability of the covalent bond in terms of overlapping atomic orbitals. Using the concept of hybridization, valence
Molecular Orbital Theory Valence bond theory accounts, at least qualitatively, for the stability of the covalent bond in terms of overlapping atomic orbitals. Using the concept of hybridization, valence
Shapes of Molecules. Lewis structures are useful but don t allow prediction of the shape of a molecule.
 Shapes of Molecules Lewis structures are useful but don t allow prediction of the shape of a molecule. H O H H O H Can use a simple theory based on electron repulsion to predict structure (for non-transition
Shapes of Molecules Lewis structures are useful but don t allow prediction of the shape of a molecule. H O H H O H Can use a simple theory based on electron repulsion to predict structure (for non-transition
Chancellor Phyllis Wise invites you to a birthday party!
 Chancellor Phyllis Wise invites you to a birthday party! 50 years ago, Illinois alumnus Nick Holonyak Jr. demonstrated the first visible light-emitting diode (LED) while working at GE. Holonyak returned
Chancellor Phyllis Wise invites you to a birthday party! 50 years ago, Illinois alumnus Nick Holonyak Jr. demonstrated the first visible light-emitting diode (LED) while working at GE. Holonyak returned
Chapter 10: Chemical Bonding II. Bonding Theories
 Chapter 10: Chemical Bonding II Dr. Chris Kozak Memorial University of Newfoundland, Canada Bonding Theories Previously, we saw how the shapes of molecules can be predicted from the orientation of electron
Chapter 10: Chemical Bonding II Dr. Chris Kozak Memorial University of Newfoundland, Canada Bonding Theories Previously, we saw how the shapes of molecules can be predicted from the orientation of electron
11/29/2014. Problems with Valence Bond Theory. VB theory predicts many properties better than Lewis Theory
 Problems with Valence Bond Theory VB theory predicts many properties better than Lewis Theory bonding schemes, bond strengths, bond lengths, bond rigidity however, there are still many properties of molecules
Problems with Valence Bond Theory VB theory predicts many properties better than Lewis Theory bonding schemes, bond strengths, bond lengths, bond rigidity however, there are still many properties of molecules
Chapter 9 Molecular Geometry and Bonding Theories
 Lecture Presentation Chapter 9 Geometry James F. Kirby Quinnipiac University Hamden, CT Shapes Lewis Structures show bonding and lone pairs, but do not denote shape. However, we use Lewis Structures to
Lecture Presentation Chapter 9 Geometry James F. Kirby Quinnipiac University Hamden, CT Shapes Lewis Structures show bonding and lone pairs, but do not denote shape. However, we use Lewis Structures to
Intro/Review of Quantum
 Intro/Review of Quantum QM-1 So you might be thinking I thought I could avoid Quantum Mechanics?!? Well we will focus on thermodynamics and kinetics, but we will consider this topic with reference to the
Intro/Review of Quantum QM-1 So you might be thinking I thought I could avoid Quantum Mechanics?!? Well we will focus on thermodynamics and kinetics, but we will consider this topic with reference to the
Intro/Review of Quantum
 Intro/Review of Quantum QM-1 So you might be thinking I thought I could avoid Quantum Mechanics?!? Well we will focus on thermodynamics and kinetics, but we will consider this topic with reference to the
Intro/Review of Quantum QM-1 So you might be thinking I thought I could avoid Quantum Mechanics?!? Well we will focus on thermodynamics and kinetics, but we will consider this topic with reference to the
ANNOUNCEMENTS. If you have questions about your exam 2 grade, write to me or Chapter 7 homework due Nov, 9 th.
 ANNOUNCEMENTS If you have questions about your exam 2 grade, write to me or Chem200@sdsu.edu. Chapter 7 homework due Nov, 9 th. Chapter 8 homework due Nov. 16 th. Exam 3 is 11/17 at 2 pm. LECTURE OBJECTIVES
ANNOUNCEMENTS If you have questions about your exam 2 grade, write to me or Chem200@sdsu.edu. Chapter 7 homework due Nov, 9 th. Chapter 8 homework due Nov. 16 th. Exam 3 is 11/17 at 2 pm. LECTURE OBJECTIVES
E nuc rep = Z AZ B r AB m =0.499 hartree =13.60 ev. E nuc rep (H 2 )= m/a m =0.714 hartree =19.43 ev.
 Chemistr 31 Phsical Chemistr Homework Assignment # 7 1. Sketch qualitative contour maps of the following molecular orbitals for a diatomic molecule AB. Identif each as a bonding or an anti-bonding molecular
Chemistr 31 Phsical Chemistr Homework Assignment # 7 1. Sketch qualitative contour maps of the following molecular orbitals for a diatomic molecule AB. Identif each as a bonding or an anti-bonding molecular
problem very complex is applied to bonding in a molecule as a whole i.e., includes interaction of all nuclei & e s
 CB VII Molecular Orbital (MO) Theory Ref 11: 5 14-1 General further improvement on Lewis, VSEPR & VB theory; resulting in better info on: bond energy bond order magnetic properties of molecules...... 14-2
CB VII Molecular Orbital (MO) Theory Ref 11: 5 14-1 General further improvement on Lewis, VSEPR & VB theory; resulting in better info on: bond energy bond order magnetic properties of molecules...... 14-2
Chapter 10. Structure Determines Properties! Molecular Geometry. Chemical Bonding II
 Chapter 10 Chemical Bonding II Structure Determines Properties! Properties of molecular substances depend on the structure of the molecule The structure includes many factors, including: the skeletal arrangement
Chapter 10 Chemical Bonding II Structure Determines Properties! Properties of molecular substances depend on the structure of the molecule The structure includes many factors, including: the skeletal arrangement
