The partition function is so important because everything else one ever wants (and can) know about the macroscopic system can be calculated from it:
|
|
|
- Tabitha Quinn
- 6 years ago
- Views:
Transcription
1 5. Thermochemstry 5.. Molecular partton functon and thermodynamc quanttes One of the man goals of computatonal chemstry s to calculate macroscopc chemcal quanttes that we observe n the laboratory (e.g. equlbrum constants, rate constants etc.). ut what we actually do the calculatons on s a sngle molecule, whch s a mcroscopc system. The connecton between the mcro and macroscopc worlds s made through statstcal mechancs. The fundamental quantty of statstcal mechancs s the partton functon. For a canoncal ensemble, the partton functon Q s wrtten as: Q exp( E T) (5.) In ths equaton, ndex runs over all possble mcroscopc energy states of the system, s oltzmann constant (.3806 x 0-3 JK - ) and T s absolute temperature. The partton functon s so mportant because everythng else one ever wants (and can) now about the macroscopc system can be calculated from t: probablty that the system s found n the state wth energy E (at temperature T): exp( E T) exp( E T) P( E ) (5.) exp( E T) Q nternal energy (at temperature T), whch s the mean (average) value of the total energy (mean s denoted by ): enthalpy entropy U E P( E ) E E exp( E T) exp( E T) ln Q H U pv T pv T Gbbs free energy (from enthalpy and entropy): T ln Q T (5.3) (5.4) ln Q S T ln Q T (5.5) T G H TS (5.6) Partton functon Q establshes the connecton between macroscopc measurable quanttes (thermodynamcs) and mcroscopc energy states of the system. The ey to calculatng all macroscopc measurable ensemble propertes from mcroscopc quanttes s to get Q.
2 However, to calculate Q (and therefore everythng else), we need the energes E of all the possble states of the system. The number of possble states of a general system s enormous and the energy s an excrucatngly complcated functon. It s necessary to mae some smplfyng assumptons. Ideal gas assumpton Snce deal gas molecules do not nteract wth one another, s that we may rewrte the partton functon as: Q N! N! N! N q N! () exp (... ) energy levels exp( g () / exp( / T ) N T ) () N T exp( () / T )... ( N ) exp( ( N ) / T ) (5.7) where the factor of /N! derves from the quantum mechancal ndstngushablty of the partcles, s the total energy of an ndvdual molecule. On gong from the frst to the second lne the exponental of all possble sums of energes was expressed as a product of all possble sums of exponentals of ndvdual energes. That s just math. Gong from the second to the thrd lne, the sum has been changed so that t goes over dscrete energy levels, rather than ndvdual states, and g s the degeneracy of level. The quantty n bracets on the thrd lne defnes the molecular partton functon q. A second consequence of the deal gas assumpton s that pv N T nrt (5.8) where n s the number of moles and R s the unversal gas constant (8.345 J mol K ). Any pv n the above equatons can therefore be replace by NT or nrt. Separaton of energy components We have thus reduced the problem from fndng the ensemble partton functon Q to fndng the molecular partton functon q. In order to mae further progress, we assume that the molecular energy ε can be expressed as a separable sum of electronc, latonal, rotatonal, and vbratonal terms,.e.: (5.9) elect l rot vb
3 (we don t consder nuclear energy as t s generally not mportant n chemstry). Substtutng to (5.7) the molecular partton functon also separates: q q q q q (5.0) elect l rot vb where etc. eelctronc energy levels q elect g exp( elect, / T) (5.) Now we have to fnd expresson for the energes, and by (5.7) or (5.0) molecular partton functons for the ndvdual degrees of freedom. From those, usng (5.7) the canoncal partton functon Q can be obtaned and fnally from (5.) - (5.6) we can get all the thermodynamcs.. Molecular electronc partton functon: The electronc partton functon s usually the smplest to compute. For a typcal, closed shell snglet molecule, the degeneracy of the ground state s unty, and the varous excted states are so hgh n energy that, at least at temperatures below thousands of degrees, they mae no sgnfcant contrbuton to the partton functon. The only one that really matters s the ground electronc state energy: q elect exp( E, / T) (5.) elect ground The electronc component of U usng (5.3) s ndependent of temperature and equal to Eelec. However, f the ground state has multplcty other than one (total spn S greater than zero) a degeneracy factor S+ must be added.. Molecular latonal partton functon: To evaluate q, we assume that the molecule acts as a partcle n a three-dmensonal cubc box of dmenson a 3 where a s the length of one sde of the cube. In realty, the molecule s not confned to any such box, but to get around t, we wll assume that the box s very, very large (a s enormous compared to the sze of the molecule, n fact we ll call t macroscopc). The energy levels for ths elementary quantum mechancal system are gven by: h n x ny nz (5.3) 8Ma where M s the molecular mass, and each energy level has assocated wth t the three unque quantum numbers nx, ny, and nz. ecause the energy levels for the partcle n a box are very, very closely spaced (at least for a box of macroscopc dmensons), the partton functon sum may be replaced by an ndefnte ntegral, and ths ntegral can be evaluated analytcally as: 3
4 q M T h 3 / V (5.4) where volume V replaced a 3. Evaluatng equaton (5.3) for the nternal energy we get: 3 U nrt (5.5) The entropy (equaton 5.5) s slghtly more complcated, ncludng some addtonal terms from the expanson of the logarthm n Q (eqn. 5.7) gvng: S M nrln h T 3/ V N A 5 (5.6) Volume appears n the last expresson. Remember the deal gas assumpton (5.8). 3. Molecular rotatonal partton functon: The rotatonal partton functon can be evaluated from quantum mechancal rotatonal levels of the molecule. We wll not go nto detals here, just summarze the results. We have to dstngush between lnear and non-lnear molecules: lnear molecules the rotatonal nternal energy s: and the entropy U lnear rot nrt (5.7) S lnear rot 3/ I T nrln h (5.8) where I s the molecular moment of nerta and s a symmetry factor, = for asymmetrc molecules and = for symmetrc ones (belongng to Cv and Dh pont groups). non-lnear molecules For non-lnear molecules the rotatonal Schrodnger equaton cannot be n general solved exactly, but t can be suffcently well approxmated. The general formulas are: U non lnear rot n 3 RT (5.9) 4
5 S nonlnear rot nrln I A I I C 8 h T 3 / 3 (5.0) where IA, I, and IC are the prncpal moments of nerta, and σ s agan a symmetry number. 4. Molecular vbratonal partton functon: You mght have notced that for latonal and rotatonal motons (not electronc though), the nternal energy s derectly proportonal to temperature T. That means at zero absolute temperature (0K) the latonal and rotatonal energes are both zero. In other words, the laton and rotatonal motons are completely frozen at 0K. Vbratons are very dfferent. The quantum mechancal expresson for the energy of the harmonc oscllator (3.4) says that even n the ground state (n =0) there s stll a non-zero vbratonal energy: E0 h (5.) Ths s called a ZERO POINT ENERGY (ZPE) and s extremely mportant. A molecule wth N atoms wll have 3N - 6 vbratonal modes (3N -5 f t s lnear) and e ach of them wll contrbute to the ZPE. That means that at 0K the total energy of the molecule wll be ts electronc energy plus the sum of ZPE from all vbratonal modes: U 0 U( T 0K) E elect h (5.) 3N 6 ecause the zero pont energy always has to be counted, t s usually separated from the vbratonal partton functon, n only the hgher energy levels, whose populaton s dependent on temperature, are counted. The formulas for the vbratonal nternal energy and entropy are: U vb R h 3N 6 exp( h / T) (5.3) S vb R 3N 6 h exp( h / T) ln exp( h / T) (5.4) 5
6 5.. Thermochemstry n Gaussan All frequency calculatons nclude thermochemcal analyss of the system. y default, ths analyss s carred out at 98.5 K and atmosphere of pressure, usng the prncpal sotope for each element type. Here s the start of the thermochemstry output for formaldehyde: Thermochemstry Temperature Kelvn. Pressure Atm. Atom has atomc number 6 and mass Atom has atomc number 8 and mass Atom 3 has atomc number and mass Atom 4 has atomc number and mass Molecular mass: amu. Ths secton lsts the parameters used for the thermochemcal analyss: the temperature, pressure, and sotopes. Gaussan predcts varous mportant thermodynamc quanttes at the specfed temperature and pressure, ncludng the thermal energy correcton, heat capacty and entropy. It also gve the zero pont energy (ZPE). These tems are broen down nto ther source components n the output: Zero-pont correcton= (Hartree/Partcle) Thermal correcton to Energy= (E therm = ZPE+E +E rot+e vb) Thermal correcton to Enthalpy= (H therm= E therm+pv) Thermal correcton to Gbbs Free Energy= (G therm= H thermts) Sum of electronc and zero-pont Energes= (E 0=E elect+zpe) Sum of electronc and thermal Energes= (E= E 0+E +E rot+e vb= E elect +E therm) Sum of electronc and thermal Enthalpes= (H= E+pV= E elect+h therm) Sum of electronc and thermal Free Energes= (G= HTS= E elect+g therm) e careful here, because the descrpton s lttle confusng. The thermal correctons and the sums all contan the zero-pont energy, although t s not explctly stated! The Sum of electronc and thermal Energes s the same as the nternal energy we denoted U above. The expressons on the rght also gve you optons for calculatng the thermodynamc quanttes, U, H and G. The entropy s also gven n the output, along wth the heat capacty (Cv) and broen down to ndvdual contrbutons. The Entropy S can be used to calculate the Gbbs free energy from enthalpy. Note though that these are n dfferent unts than the quanttes above: E (Thermal) CV S KCal/Mol Cal/Mol-Kelvn Cal/Mol-Kelvn Total Electronc Translatonal Rotatonal Vbratonal
7 5.3. Scalng factors Raw frequency values computed at the Hartree-Foc level contan nown systematc errors due to the neglect of electron correlaton, resultng n overestmates of about 0%-%. Therefore, t s usual to scale frequences predcted at the Hartree-Foc level by an emprcal factor of Use of ths factor has been demonstrated to produce very good agreement wth experment for a wde range of systems. Our values must be expected to devate even a bt more from experment because of our choce of a medum-szed bass set (by around 5% n all). Frequences computed wth methods other than Hartree-Foc are also scaled to smlarly elmnate nown systematc errors n calculated frequences. The followng table lsts the recommended scale factors for frequences and for zero-pont energes and for use n computng thermal energy correctons (the latter two tems are dscussed later n ths chapter), for several mportant calculaton types (Table from Cramer: Essentals of Computatonal Chemstry: Theores and Models, nd Ed.) 7
8 Sometmes, dfferent scalng factors are used for ZPE and thermal energes than for frequences that are used for smulaton of spectra (Table from Foresman and Frsch: Explorng Chemstry wth Electronc Structure Methods, nd Ed.): Gaussan wll scale frequences for the thermochemstry calculatons, but the actual frequences wll always be unscaled (f you want to scale them you have to do t yourself) Changng thermochemstry parameters y default Gaussan wll carry out frequency and thermochemstry calculaton for the standard temperature and pressure (STP) and most abundant sotopes. y default the frequences are also unscaled. If dfferent condtons, sotopes and/or scalng factors are desred, there are several ways to change them:. An old way, whch does not seem to wor that well anymore s to use ReadIsotopes opton to the Freq eyword n the route secton. Values for all parameters must then be specfed n a separate nput secton followng the molecule specfcaton-and separated from t by a blan lne. #3LYP/6-3G(d) Opt Freq=ReadIsotopes Test Here s the general format for the ReadIsotopes nput secton: temp pressure [scale] sotope for atom sotope for atom. sotope for atom N The scale factor s optonal. Isotopes are specfed as ntegers although the program wll use the actual value. (e.g., 8 specfes 8 O, and Gaussan uses the value ). 8
9 . More drect way, whch now seems to be the only one that wors, s to set the temperature, pressure and scalng factors drectly n the route secton usng, respectvely, eywords Temperature, Pressure and Scale wth the desred values (for temperature n Kelvn, for pressure n atmospheres). For example: #3LYP/6-3G(d) Opt Freq Temperature=73.0 Pressure=0. Scale= Isotopes can be set n the molecule specfcaton secton usng (Iso= ) after the atom symbol. For example: 0 C(Iso=3) H(Iso=) wll mae the frst atom 3 C and the second atom Deuterum. 3. If you already calculated frequences and thermochemstry for one set of parameters and saved the checpont fle, a utlty program freqch can be used to just repeat the frequency calculaton (wthout dong any quantum mechancs) for dfferent condtons, sotopes or wth dfferent scalng factors. just type freqch at the prompt and enter the name of the checpont fle and desred parameters. For example, f I had done a calculaton for water, ths would do t for a heavy water (DO) at 300 K and.0 atmospheres: born 5% freqch Checpont fle? water.ch Wrte Hyperchem fles? n Temperature (K)? [0=>98.5] Pressure (Atm)? [0=> atm].0 Scale factor for frequences durng thermochemstry? [0=>/.].0 Do you want to use the prncpal sotope masses? [Y]: N For each atom, gve the nteger mass number. In each case, the default s the prncpal sotope. Atom number, atomc number 8: [6] 6 Atom number, atomc number : [] Atom number 3, atomc number : [] and the output would follow. You should always mae sure that your parameters are correctly set by checng the output. In ths case I have: Atom has atomc number 8 and mass Atom has atomc number and mass.040 Atom 3 has atomc number and mass.040 Temperature Kelvn. Pressure Atm. 9
10 If you don t use scalng (scale factor =) t wll not be reported. However, f you choose to scale the frequences, for example by , t wll appear n the thermochemstry output: Thermochemstry wll use frequences scaled by The only problem wth usng freqch s that only thermal correctons are reported, but not the total sums that also contan the electronc energy. Whle n the log fle you get: Zero-pont correcton= (Hartree/Partcle) Thermal correcton to Energy= Thermal correcton to Enthalpy= Thermal correcton to Gbbs Free Energy= Sum of electronc and zero-pont Energes= Sum of electronc and thermal Energes= Sum of electronc and thermal Enthalpes= Sum of electronc and thermal Free Energes= from freqch you get only part of t: Zero-pont correcton= (Hartree/Partcle) Thermal correcton to Energy= Thermal correcton to Enthalpy= Thermal correcton to Gbbs Free Energy= To calculate total energes, enthalpes and Gbbs free energes, you need to add the electronc energy to the thermal correctons. The electronc energy s n the log fle from the orgnal job: SCF Done: E(R3LYP) = A.U. after cycles Remember that the thermal correctons already contan the ZPE, so you don t add t agan (t would be there twce). To see that t wors, let s add the Thermal correctons from the freqch output to the electronc energy E(R3LYP) and compare to the summed energes: U = E(R3LYP) + Thermal correcton to Energy = A.U. = A.U. = Sum of electronc and thermal Energes H = E(R3LYP) + Thermal correcton to Enthalpy = A.U. = A.U. = Sum of electronc and thermal Enthalpes G = E(R3LYP) + Thermal correcton to Gbbs Free Energy = A.U. = A.U. = Sum of electronc and thermal Free Energes 0
11 5.5. Example: Enthalpy and Gbbs free energy of a reacton Let's consder the hydraton reacton: H + + HO H3O +. Our goal s to compute the enthalpy change H 98 and Gbbs free energy change G 98 at 98 K and atm pressure for the reacton, at 3LYP/6-3+G(df,p) level of theory. The enthalpy change can be calculated usng these expressons: H 98 = H 98 (products) H 98 (reactants) = H 98 (H3O + ) H 98 (HO) H 98 (H + ) Smlarly: G 98 = G 98 (products) G 98 (reactants) = G 98 (H3O + ) G 98 (HO) G 98 (H + ) We need to do calculatons frequency calculatons (whch also means optmzaton!) on H3O + and HO and get thermochemstry parameters under the specfed condtons (98K, atm happen to be the default). The proton H + not only cannot vbrate, but there s also no electron to even do any quantum calculaton. The only contrbuton to the enthalpy or free energy from the proton s latonal, gven by (for mol): U S H G 3 RT M T Rln h 3 5 RT pv RT H TS 3 / V N A 5 M T Rln h 3 / RT pn A 5 Substtutng the numbers for H + : U = J.mol - = cal.mol - H= J.mol - =.48 cal.mol - S = J.mol -.K - = 6.04 cal.mol -.K - G= 6.54 J.mol - = 6.75 cal.mol - You can chec these numbers because t turns out that Gaussan wll stll calculate thermochemstry, even for atoms and even for a naed proton! Just call Freq and remember that proton has a charge of +. The method does not matter, there are no electrons to calculate. Here s the nput:
12 # Freq Test h plus H and the output: Thermochemstry Temperature Kelvn. Pressure Atm. Atom has atomc number and mass Molecular mass: amu. Zero-pont vbratonal energy Vbratonal temperatures: (Kelvn) 0.0 (Joules/Mol) (Kcal/Mol) Zero-pont correcton= (Hartree/Partcle) Thermal correcton to Energy= Thermal correcton to Enthalpy= Thermal correcton to Gbbs Free Energy= Sum of electronc and zero-pont Energes= Sum of electronc and thermal Energes= Sum of electronc and thermal Enthalpes= Sum of electronc and thermal Free Energes= E (Thermal) CV S KCal/Mol Cal/Mol-Kelvn Cal/Mol-Kelvn Total Electronc Translatonal Rotatonal Vbratonal As you can see the enthalpy (under Sum of electronc and thermal Enthalpes) s A.U., whch s x 67.5 cal.mol - =.48 cal.mol -. The Gbbs free energy (under Sum of electronc and thermal Free Energes) s x 67.5 cal.mol - = 6.75 cal.mol -. Another way to get these numbers s from the E (Thermal), whch s the same as U: H = U + pv = E (Thermal) + RT = ( ) cal.mol - =.48 cal.mol - G = H TS =.48 cal.mol K x 6.04 cal.mol -.K - = 6.75 cal.mol - To get the thermochemstry for HO and H3O + we can buld them n Gabedt (agan remember the charge for the latter s plus one). Here s the nput fle for both jobs lned together (I used Opt Freq snce we are dong full optmzaton anyway):
13 # 3LYP/6-3+g(df,p) Opt Freq test water 0 O H H Ln-- # 3LYP/6-3+g(df,p) Opt Freq test H3O plus O H H H Ths s the useful output for water: Zero-pont correcton= (Hartree/Partcle). Sum of electronc and thermal Enthalpes= Sum of electronc and thermal Free Energes= And ths for the hydronum on: Sum of electronc and thermal Enthalpes= Sum of electronc and thermal Free Energes= Remember that these are the actual enthalpes and free energes, contanng all the contrbutons ncludng ZPE. All that needs to be done s smply convert these numbers to other unts. H 98 (HO) = Hartree = cal.mol - G 98 (HO) = Hartree = cal.mol - H 98 (H3O + ) = Hartree = 48.7 cal.mol - G 98 (H3O + ) = Hartree = 484. cal.mol - Substtutng nto products mnus reactants formulas above, we get the desred enthalpy and free energy changes for the reacton: H 98 = 48.7 (4796.4).5 cal.mol - = 78.0 cal.mol - G 98 = 484. (4797.8) ( 6.3) cal.mol - = 0.0 cal.mol - 3
1. Connection between QM and Thermodynamics
 Calculaton of hermodynamc and Knetc Propertes 1. Connecton between QM and hermodynamcs We have focused to ths pont on the many approaches and detals of calculatng the nternal tronc energy of a sngle molecule,
Calculaton of hermodynamc and Knetc Propertes 1. Connecton between QM and hermodynamcs We have focused to ths pont on the many approaches and detals of calculatng the nternal tronc energy of a sngle molecule,
Thermodynamics and statistical mechanics in materials modelling II
 Course MP3 Lecture 8/11/006 (JAE) Course MP3 Lecture 8/11/006 Thermodynamcs and statstcal mechancs n materals modellng II A bref résumé of the physcal concepts used n materals modellng Dr James Ellott.1
Course MP3 Lecture 8/11/006 (JAE) Course MP3 Lecture 8/11/006 Thermodynamcs and statstcal mechancs n materals modellng II A bref résumé of the physcal concepts used n materals modellng Dr James Ellott.1
Thermodynamics General
 Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Lecture 4. Macrostates and Microstates (Ch. 2 )
 Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
Molecular structure: Diatomic molecules in the rigid rotor and harmonic oscillator approximations Notes on Quantum Mechanics
 Molecular structure: Datomc molecules n the rgd rotor and harmonc oscllator approxmatons Notes on Quantum Mechancs http://quantum.bu.edu/notes/quantummechancs/molecularstructuredatomc.pdf Last updated
Molecular structure: Datomc molecules n the rgd rotor and harmonc oscllator approxmatons Notes on Quantum Mechancs http://quantum.bu.edu/notes/quantummechancs/molecularstructuredatomc.pdf Last updated
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM
 ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
THERMAL DISTRIBUTION IN THE HCL SPECTRUM OBJECTIVE
 ame: THERMAL DISTRIBUTIO I THE HCL SPECTRUM OBJECTIVE To nvestgate a system s thermal dstrbuton n dscrete states; specfcally, determne HCl gas temperature from the relatve occupatons of ts rotatonal states.
ame: THERMAL DISTRIBUTIO I THE HCL SPECTRUM OBJECTIVE To nvestgate a system s thermal dstrbuton n dscrete states; specfcally, determne HCl gas temperature from the relatve occupatons of ts rotatonal states.
Introduction to Vapor/Liquid Equilibrium, part 2. Raoult s Law:
 CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
STATISTICAL MECHANICS
 STATISTICAL MECHANICS Thermal Energy Recall that KE can always be separated nto 2 terms: KE system = 1 2 M 2 total v CM KE nternal Rgd-body rotaton and elastc / sound waves Use smplfyng assumptons KE of
STATISTICAL MECHANICS Thermal Energy Recall that KE can always be separated nto 2 terms: KE system = 1 2 M 2 total v CM KE nternal Rgd-body rotaton and elastc / sound waves Use smplfyng assumptons KE of
5.62 Physical Chemistry II Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Sprng 2008 Lecture 34 Page Transton
MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Sprng 2008 Lecture 34 Page Transton
Lecture 14: Forces and Stresses
 The Nuts and Bolts of Frst-Prncples Smulaton Lecture 14: Forces and Stresses Durham, 6th-13th December 2001 CASTEP Developers Group wth support from the ESF ψ k Network Overvew of Lecture Why bother? Theoretcal
The Nuts and Bolts of Frst-Prncples Smulaton Lecture 14: Forces and Stresses Durham, 6th-13th December 2001 CASTEP Developers Group wth support from the ESF ψ k Network Overvew of Lecture Why bother? Theoretcal
Econ107 Applied Econometrics Topic 3: Classical Model (Studenmund, Chapter 4)
 I. Classcal Assumptons Econ7 Appled Econometrcs Topc 3: Classcal Model (Studenmund, Chapter 4) We have defned OLS and studed some algebrac propertes of OLS. In ths topc we wll study statstcal propertes
I. Classcal Assumptons Econ7 Appled Econometrcs Topc 3: Classcal Model (Studenmund, Chapter 4) We have defned OLS and studed some algebrac propertes of OLS. In ths topc we wll study statstcal propertes
STATISTICAL MECHANICAL ENSEMBLES 1 MICROSCOPIC AND MACROSCOPIC VARIABLES PHASE SPACE ENSEMBLES. CHE 524 A. Panagiotopoulos 1
 CHE 54 A. Panagotopoulos STATSTCAL MECHACAL ESEMBLES MCROSCOPC AD MACROSCOPC ARABLES The central queston n Statstcal Mechancs can be phrased as follows: f partcles (atoms, molecules, electrons, nucle,
CHE 54 A. Panagotopoulos STATSTCAL MECHACAL ESEMBLES MCROSCOPC AD MACROSCOPC ARABLES The central queston n Statstcal Mechancs can be phrased as follows: f partcles (atoms, molecules, electrons, nucle,
University of Washington Department of Chemistry Chemistry 453 Winter Quarter 2015
 Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
( ) 1/ 2. ( P SO2 )( P O2 ) 1/ 2.
 Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
CHAPTER 5 NUMERICAL EVALUATION OF DYNAMIC RESPONSE
 CHAPTER 5 NUMERICAL EVALUATION OF DYNAMIC RESPONSE Analytcal soluton s usually not possble when exctaton vares arbtrarly wth tme or f the system s nonlnear. Such problems can be solved by numercal tmesteppng
CHAPTER 5 NUMERICAL EVALUATION OF DYNAMIC RESPONSE Analytcal soluton s usually not possble when exctaton vares arbtrarly wth tme or f the system s nonlnear. Such problems can be solved by numercal tmesteppng
5.60 Thermodynamics & Kinetics Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
5.04, Principles of Inorganic Chemistry II MIT Department of Chemistry Lecture 32: Vibrational Spectroscopy and the IR
 5.0, Prncples of Inorganc Chemstry II MIT Department of Chemstry Lecture 3: Vbratonal Spectroscopy and the IR Vbratonal spectroscopy s confned to the 00-5000 cm - spectral regon. The absorpton of a photon
5.0, Prncples of Inorganc Chemstry II MIT Department of Chemstry Lecture 3: Vbratonal Spectroscopy and the IR Vbratonal spectroscopy s confned to the 00-5000 cm - spectral regon. The absorpton of a photon
1 Matrix representations of canonical matrices
 1 Matrx representatons of canoncal matrces 2-d rotaton around the orgn: ( ) cos θ sn θ R 0 = sn θ cos θ 3-d rotaton around the x-axs: R x = 1 0 0 0 cos θ sn θ 0 sn θ cos θ 3-d rotaton around the y-axs:
1 Matrx representatons of canoncal matrces 2-d rotaton around the orgn: ( ) cos θ sn θ R 0 = sn θ cos θ 3-d rotaton around the x-axs: R x = 1 0 0 0 cos θ sn θ 0 sn θ cos θ 3-d rotaton around the y-axs:
Physics 240: Worksheet 30 Name:
 (1) One mole of an deal monatomc gas doubles ts temperature and doubles ts volume. What s the change n entropy of the gas? () 1 kg of ce at 0 0 C melts to become water at 0 0 C. What s the change n entropy
(1) One mole of an deal monatomc gas doubles ts temperature and doubles ts volume. What s the change n entropy of the gas? () 1 kg of ce at 0 0 C melts to become water at 0 0 C. What s the change n entropy
12. The Hamilton-Jacobi Equation Michael Fowler
 1. The Hamlton-Jacob Equaton Mchael Fowler Back to Confguraton Space We ve establshed that the acton, regarded as a functon of ts coordnate endponts and tme, satsfes ( ) ( ) S q, t / t+ H qpt,, = 0, and
1. The Hamlton-Jacob Equaton Mchael Fowler Back to Confguraton Space We ve establshed that the acton, regarded as a functon of ts coordnate endponts and tme, satsfes ( ) ( ) S q, t / t+ H qpt,, = 0, and
Physics 181. Particle Systems
 Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
Comparison of Regression Lines
 STATGRAPHICS Rev. 9/13/2013 Comparson of Regresson Lnes Summary... 1 Data Input... 3 Analyss Summary... 4 Plot of Ftted Model... 6 Condtonal Sums of Squares... 6 Analyss Optons... 7 Forecasts... 8 Confdence
STATGRAPHICS Rev. 9/13/2013 Comparson of Regresson Lnes Summary... 1 Data Input... 3 Analyss Summary... 4 Plot of Ftted Model... 6 Condtonal Sums of Squares... 6 Analyss Optons... 7 Forecasts... 8 Confdence
Linear Approximation with Regularization and Moving Least Squares
 Lnear Approxmaton wth Regularzaton and Movng Least Squares Igor Grešovn May 007 Revson 4.6 (Revson : March 004). 5 4 3 0.5 3 3.5 4 Contents: Lnear Fttng...4. Weghted Least Squares n Functon Approxmaton...
Lnear Approxmaton wth Regularzaton and Movng Least Squares Igor Grešovn May 007 Revson 4.6 (Revson : March 004). 5 4 3 0.5 3 3.5 4 Contents: Lnear Fttng...4. Weghted Least Squares n Functon Approxmaton...
Entropy generation in a chemical reaction
 Entropy generaton n a chemcal reacton E Mranda Área de Cencas Exactas COICET CCT Mendoza 5500 Mendoza, rgentna and Departamento de Físca Unversdad aconal de San Lus 5700 San Lus, rgentna bstract: Entropy
Entropy generaton n a chemcal reacton E Mranda Área de Cencas Exactas COICET CCT Mendoza 5500 Mendoza, rgentna and Departamento de Físca Unversdad aconal de San Lus 5700 San Lus, rgentna bstract: Entropy
Structure and Drive Paul A. Jensen Copyright July 20, 2003
 Structure and Drve Paul A. Jensen Copyrght July 20, 2003 A system s made up of several operatons wth flow passng between them. The structure of the system descrbes the flow paths from nputs to outputs.
Structure and Drve Paul A. Jensen Copyrght July 20, 2003 A system s made up of several operatons wth flow passng between them. The structure of the system descrbes the flow paths from nputs to outputs.
Physics 607 Exam 1. ( ) = 1, Γ( z +1) = zγ( z) x n e x2 dx = 1. e x2
 Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
C/CS/Phy191 Problem Set 3 Solutions Out: Oct 1, 2008., where ( 00. ), so the overall state of the system is ) ( ( ( ( 00 ± 11 ), Φ ± = 1
 C/CS/Phy9 Problem Set 3 Solutons Out: Oct, 8 Suppose you have two qubts n some arbtrary entangled state ψ You apply the teleportaton protocol to each of the qubts separately What s the resultng state obtaned
C/CS/Phy9 Problem Set 3 Solutons Out: Oct, 8 Suppose you have two qubts n some arbtrary entangled state ψ You apply the teleportaton protocol to each of the qubts separately What s the resultng state obtaned
A quote of the week (or camel of the week): There is no expedience to which a man will not go to avoid the labor of thinking. Thomas A.
 A quote of the week (or camel of the week): here s no expedence to whch a man wll not go to avod the labor of thnkng. homas A. Edson Hess law. Algorthm S Select a reacton, possbly contanng specfc compounds
A quote of the week (or camel of the week): here s no expedence to whch a man wll not go to avod the labor of thnkng. homas A. Edson Hess law. Algorthm S Select a reacton, possbly contanng specfc compounds
Open Systems: Chemical Potential and Partial Molar Quantities Chemical Potential
 Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Problem Points Score Total 100
 Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructions
 THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
CHAPTER 14 GENERAL PERTURBATION THEORY
 CHAPTER 4 GENERAL PERTURBATION THEORY 4 Introducton A partcle n orbt around a pont mass or a sphercally symmetrc mass dstrbuton s movng n a gravtatonal potental of the form GM / r In ths potental t moves
CHAPTER 4 GENERAL PERTURBATION THEORY 4 Introducton A partcle n orbt around a pont mass or a sphercally symmetrc mass dstrbuton s movng n a gravtatonal potental of the form GM / r In ths potental t moves
The ChemSep Book. Harry A. Kooijman Consultant. Ross Taylor Clarkson University, Potsdam, New York University of Twente, Enschede, The Netherlands
 The ChemSep Book Harry A. Koojman Consultant Ross Taylor Clarkson Unversty, Potsdam, New York Unversty of Twente, Enschede, The Netherlands Lbr Books on Demand www.bod.de Copyrght c 2000 by H.A. Koojman
The ChemSep Book Harry A. Koojman Consultant Ross Taylor Clarkson Unversty, Potsdam, New York Unversty of Twente, Enschede, The Netherlands Lbr Books on Demand www.bod.de Copyrght c 2000 by H.A. Koojman
...Thermodynamics. If Clausius Clapeyron fails. l T (v 2 v 1 ) = 0/0 Second order phase transition ( S, v = 0)
 If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
and Statistical Mechanics Material Properties
 Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Kernel Methods and SVMs Extension
 Kernel Methods and SVMs Extenson The purpose of ths document s to revew materal covered n Machne Learnng 1 Supervsed Learnng regardng support vector machnes (SVMs). Ths document also provdes a general
Kernel Methods and SVMs Extenson The purpose of ths document s to revew materal covered n Machne Learnng 1 Supervsed Learnng regardng support vector machnes (SVMs). Ths document also provdes a general
Lecture. Polymer Thermodynamics 0331 L Chemical Potential
 Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
Prof. Dr. I. Nasser Phys 630, T Aug-15 One_dimensional_Ising_Model
 EXACT OE-DIMESIOAL ISIG MODEL The one-dmensonal Isng model conssts of a chan of spns, each spn nteractng only wth ts two nearest neghbors. The smple Isng problem n one dmenson can be solved drectly n several
EXACT OE-DIMESIOAL ISIG MODEL The one-dmensonal Isng model conssts of a chan of spns, each spn nteractng only wth ts two nearest neghbors. The smple Isng problem n one dmenson can be solved drectly n several
Lecture 7: Boltzmann distribution & Thermodynamics of mixing
 Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
G4023 Mid-Term Exam #1 Solutions
 Exam1Solutons.nb 1 G03 Md-Term Exam #1 Solutons 1-Oct-0, 1:10 p.m to :5 p.m n 1 Pupn Ths exam s open-book, open-notes. You may also use prnt-outs of the homework solutons and a calculator. 1 (30 ponts,
Exam1Solutons.nb 1 G03 Md-Term Exam #1 Solutons 1-Oct-0, 1:10 p.m to :5 p.m n 1 Pupn Ths exam s open-book, open-notes. You may also use prnt-outs of the homework solutons and a calculator. 1 (30 ponts,
EPR Paradox and the Physical Meaning of an Experiment in Quantum Mechanics. Vesselin C. Noninski
 EPR Paradox and the Physcal Meanng of an Experment n Quantum Mechancs Vesseln C Nonnsk vesselnnonnsk@verzonnet Abstract It s shown that there s one purely determnstc outcome when measurement s made on
EPR Paradox and the Physcal Meanng of an Experment n Quantum Mechancs Vesseln C Nonnsk vesselnnonnsk@verzonnet Abstract It s shown that there s one purely determnstc outcome when measurement s made on
Robert Eisberg Second edition CH 09 Multielectron atoms ground states and x-ray excitations
 Quantum Physcs 量 理 Robert Esberg Second edton CH 09 Multelectron atoms ground states and x-ray exctatons 9-01 By gong through the procedure ndcated n the text, develop the tme-ndependent Schroednger equaton
Quantum Physcs 量 理 Robert Esberg Second edton CH 09 Multelectron atoms ground states and x-ray exctatons 9-01 By gong through the procedure ndcated n the text, develop the tme-ndependent Schroednger equaton
The Feynman path integral
 The Feynman path ntegral Aprl 3, 205 Hesenberg and Schrödnger pctures The Schrödnger wave functon places the tme dependence of a physcal system n the state, ψ, t, where the state s a vector n Hlbert space
The Feynman path ntegral Aprl 3, 205 Hesenberg and Schrödnger pctures The Schrödnger wave functon places the tme dependence of a physcal system n the state, ψ, t, where the state s a vector n Hlbert space
NUMERICAL DIFFERENTIATION
 NUMERICAL DIFFERENTIATION 1 Introducton Dfferentaton s a method to compute the rate at whch a dependent output y changes wth respect to the change n the ndependent nput x. Ths rate of change s called the
NUMERICAL DIFFERENTIATION 1 Introducton Dfferentaton s a method to compute the rate at whch a dependent output y changes wth respect to the change n the ndependent nput x. Ths rate of change s called the
Limited Dependent Variables
 Lmted Dependent Varables. What f the left-hand sde varable s not a contnuous thng spread from mnus nfnty to plus nfnty? That s, gven a model = f (, β, ε, where a. s bounded below at zero, such as wages
Lmted Dependent Varables. What f the left-hand sde varable s not a contnuous thng spread from mnus nfnty to plus nfnty? That s, gven a model = f (, β, ε, where a. s bounded below at zero, such as wages
Salmon: Lectures on partial differential equations. Consider the general linear, second-order PDE in the form. ,x 2
 Salmon: Lectures on partal dfferental equatons 5. Classfcaton of second-order equatons There are general methods for classfyng hgher-order partal dfferental equatons. One s very general (applyng even to
Salmon: Lectures on partal dfferental equatons 5. Classfcaton of second-order equatons There are general methods for classfyng hgher-order partal dfferental equatons. One s very general (applyng even to
Physics 5153 Classical Mechanics. D Alembert s Principle and The Lagrangian-1
 P. Guterrez Physcs 5153 Classcal Mechancs D Alembert s Prncple and The Lagrangan 1 Introducton The prncple of vrtual work provdes a method of solvng problems of statc equlbrum wthout havng to consder the
P. Guterrez Physcs 5153 Classcal Mechancs D Alembert s Prncple and The Lagrangan 1 Introducton The prncple of vrtual work provdes a method of solvng problems of statc equlbrum wthout havng to consder the
Economics 101. Lecture 4 - Equilibrium and Efficiency
 Economcs 0 Lecture 4 - Equlbrum and Effcency Intro As dscussed n the prevous lecture, we wll now move from an envronment where we looed at consumers mang decsons n solaton to analyzng economes full of
Economcs 0 Lecture 4 - Equlbrum and Effcency Intro As dscussed n the prevous lecture, we wll now move from an envronment where we looed at consumers mang decsons n solaton to analyzng economes full of
1 Derivation of Rate Equations from Single-Cell Conductance (Hodgkin-Huxley-like) Equations
 Physcs 171/271 -Davd Klenfeld - Fall 2005 (revsed Wnter 2011) 1 Dervaton of Rate Equatons from Sngle-Cell Conductance (Hodgkn-Huxley-lke) Equatons We consder a network of many neurons, each of whch obeys
Physcs 171/271 -Davd Klenfeld - Fall 2005 (revsed Wnter 2011) 1 Dervaton of Rate Equatons from Sngle-Cell Conductance (Hodgkn-Huxley-lke) Equatons We consder a network of many neurons, each of whch obeys
Session 7. Spectroscopy and Thermochemistry. Session 7 Overview: Part A I. Prediction of Vibrational Frequencies (IR)
 Sesson 7 Spectroscopy and Thermochemstry CCCE 2008 1 Sesson 7 Overvew: Part A I. Predcton of Vbratonal Frequences (IR) II. Thermochemstry Part B III. Predcton of Electronc Transtons (UV-Vs) IV. NMR Predctons
Sesson 7 Spectroscopy and Thermochemstry CCCE 2008 1 Sesson 7 Overvew: Part A I. Predcton of Vbratonal Frequences (IR) II. Thermochemstry Part B III. Predcton of Electronc Transtons (UV-Vs) IV. NMR Predctons
PHYS 705: Classical Mechanics. Canonical Transformation II
 1 PHYS 705: Classcal Mechancs Canoncal Transformaton II Example: Harmonc Oscllator f ( x) x m 0 x U( x) x mx x LT U m Defne or L p p mx x x m mx x H px L px p m p x m m H p 1 x m p m 1 m H x p m x m m
1 PHYS 705: Classcal Mechancs Canoncal Transformaton II Example: Harmonc Oscllator f ( x) x m 0 x U( x) x mx x LT U m Defne or L p p mx x x m mx x H px L px p m p x m m H p 1 x m p m 1 m H x p m x m m
PHYS 705: Classical Mechanics. Newtonian Mechanics
 1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
Grover s Algorithm + Quantum Zeno Effect + Vaidman
 Grover s Algorthm + Quantum Zeno Effect + Vadman CS 294-2 Bomb 10/12/04 Fall 2004 Lecture 11 Grover s algorthm Recall that Grover s algorthm for searchng over a space of sze wors as follows: consder the
Grover s Algorthm + Quantum Zeno Effect + Vadman CS 294-2 Bomb 10/12/04 Fall 2004 Lecture 11 Grover s algorthm Recall that Grover s algorthm for searchng over a space of sze wors as follows: consder the
3.1 Expectation of Functions of Several Random Variables. )' be a k-dimensional discrete or continuous random vector, with joint PMF p (, E X E X1 E X
 Statstcs 1: Probablty Theory II 37 3 EPECTATION OF SEVERAL RANDOM VARIABLES As n Probablty Theory I, the nterest n most stuatons les not on the actual dstrbuton of a random vector, but rather on a number
Statstcs 1: Probablty Theory II 37 3 EPECTATION OF SEVERAL RANDOM VARIABLES As n Probablty Theory I, the nterest n most stuatons les not on the actual dstrbuton of a random vector, but rather on a number
1 (1 + ( )) = 1 8 ( ) = (c) Carrying out the Taylor expansion, in this case, the series truncates at second order:
 68A Solutons to Exercses March 05 (a) Usng a Taylor expanson, and notng that n 0 for all n >, ( + ) ( + ( ) + ) We can t nvert / because there s no Taylor expanson around 0 Lets try to calculate the nverse
68A Solutons to Exercses March 05 (a) Usng a Taylor expanson, and notng that n 0 for all n >, ( + ) ( + ( ) + ) We can t nvert / because there s no Taylor expanson around 0 Lets try to calculate the nverse
9 Derivation of Rate Equations from Single-Cell Conductance (Hodgkin-Huxley-like) Equations
 Physcs 171/271 - Chapter 9R -Davd Klenfeld - Fall 2005 9 Dervaton of Rate Equatons from Sngle-Cell Conductance (Hodgkn-Huxley-lke) Equatons We consder a network of many neurons, each of whch obeys a set
Physcs 171/271 - Chapter 9R -Davd Klenfeld - Fall 2005 9 Dervaton of Rate Equatons from Sngle-Cell Conductance (Hodgkn-Huxley-lke) Equatons We consder a network of many neurons, each of whch obeys a set
Negative Binomial Regression
 STATGRAPHICS Rev. 9/16/2013 Negatve Bnomal Regresson Summary... 1 Data Input... 3 Statstcal Model... 3 Analyss Summary... 4 Analyss Optons... 7 Plot of Ftted Model... 8 Observed Versus Predcted... 10 Predctons...
STATGRAPHICS Rev. 9/16/2013 Negatve Bnomal Regresson Summary... 1 Data Input... 3 Statstcal Model... 3 Analyss Summary... 4 Analyss Optons... 7 Plot of Ftted Model... 8 Observed Versus Predcted... 10 Predctons...
Lecture 10 Support Vector Machines II
 Lecture 10 Support Vector Machnes II 22 February 2016 Taylor B. Arnold Yale Statstcs STAT 365/665 1/28 Notes: Problem 3 s posted and due ths upcomng Frday There was an early bug n the fake-test data; fxed
Lecture 10 Support Vector Machnes II 22 February 2016 Taylor B. Arnold Yale Statstcs STAT 365/665 1/28 Notes: Problem 3 s posted and due ths upcomng Frday There was an early bug n the fake-test data; fxed
CHAPTER 5: Lie Differentiation and Angular Momentum
 CHAPTER 5: Le Dfferentaton and Angular Momentum Jose G. Vargas 1 Le dfferentaton Kähler s theory of angular momentum s a specalzaton of hs approach to Le dfferentaton. We could deal wth the former drectly,
CHAPTER 5: Le Dfferentaton and Angular Momentum Jose G. Vargas 1 Le dfferentaton Kähler s theory of angular momentum s a specalzaton of hs approach to Le dfferentaton. We could deal wth the former drectly,
Transfer Functions. Convenient representation of a linear, dynamic model. A transfer function (TF) relates one input and one output: ( ) system
 Transfer Functons Convenent representaton of a lnear, dynamc model. A transfer functon (TF) relates one nput and one output: x t X s y t system Y s The followng termnology s used: x y nput output forcng
Transfer Functons Convenent representaton of a lnear, dynamc model. A transfer functon (TF) relates one nput and one output: x t X s y t system Y s The followng termnology s used: x y nput output forcng
763622S ADVANCED QUANTUM MECHANICS Solution Set 1 Spring c n a n. c n 2 = 1.
 7636S ADVANCED QUANTUM MECHANICS Soluton Set 1 Sprng 013 1 Warm-up Show that the egenvalues of a Hermtan operator  are real and that the egenkets correspondng to dfferent egenvalues are orthogonal (b)
7636S ADVANCED QUANTUM MECHANICS Soluton Set 1 Sprng 013 1 Warm-up Show that the egenvalues of a Hermtan operator  are real and that the egenkets correspondng to dfferent egenvalues are orthogonal (b)
CinChE Problem-Solving Strategy Chapter 4 Development of a Mathematical Model. formulation. procedure
 nhe roblem-solvng Strategy hapter 4 Transformaton rocess onceptual Model formulaton procedure Mathematcal Model The mathematcal model s an abstracton that represents the engneerng phenomena occurrng n
nhe roblem-solvng Strategy hapter 4 Transformaton rocess onceptual Model formulaton procedure Mathematcal Model The mathematcal model s an abstracton that represents the engneerng phenomena occurrng n
Frequency calculations can serve a number of different purposes:
 4. Frequency calculatons Frequency calculatons can serve a number of dfferent purposes: To predct the IR and Raman spectra of molecules (frequences and ntenstes. To compute force constants for a geometry
4. Frequency calculatons Frequency calculatons can serve a number of dfferent purposes: To predct the IR and Raman spectra of molecules (frequences and ntenstes. To compute force constants for a geometry
Physics 115. Molecular motion and temperature Phase equilibrium, evaporation
 Physcs 115 General Physcs II Sesson 9 Molecular moton and temperature Phase equlbrum, evaporaton R. J. Wlkes Emal: phy115a@u.washngton.edu Home page: http://courses.washngton.edu/phy115a/ 4/14/14 Physcs
Physcs 115 General Physcs II Sesson 9 Molecular moton and temperature Phase equlbrum, evaporaton R. J. Wlkes Emal: phy115a@u.washngton.edu Home page: http://courses.washngton.edu/phy115a/ 4/14/14 Physcs
Homework Chapter 21 Solutions!!
 Homework Chapter 1 Solutons 1.7 1.13 1.17 1.19 1.6 1.33 1.45 1.51 1.71 page 1 Problem 1.7 A mole sample of oxygen gas s confned to a 5 lter vessel at a pressure of 8 atm. Fnd the average translatonal knetc
Homework Chapter 1 Solutons 1.7 1.13 1.17 1.19 1.6 1.33 1.45 1.51 1.71 page 1 Problem 1.7 A mole sample of oxygen gas s confned to a 5 lter vessel at a pressure of 8 atm. Fnd the average translatonal knetc
Electron-Impact Double Ionization of the H 2
 I R A P 6(), Dec. 5, pp. 9- Electron-Impact Double Ionzaton of the H olecule Internatonal Scence Press ISSN: 9-59 Electron-Impact Double Ionzaton of the H olecule. S. PINDZOLA AND J. COLGAN Department
I R A P 6(), Dec. 5, pp. 9- Electron-Impact Double Ionzaton of the H olecule Internatonal Scence Press ISSN: 9-59 Electron-Impact Double Ionzaton of the H olecule. S. PINDZOLA AND J. COLGAN Department
Chapter Eight. Review and Summary. Two methods in solid mechanics ---- vectorial methods and energy methods or variational methods
 Chapter Eght Energy Method 8. Introducton 8. Stran energy expressons 8.3 Prncpal of statonary potental energy; several degrees of freedom ------ Castglano s frst theorem ---- Examples 8.4 Prncpal of statonary
Chapter Eght Energy Method 8. Introducton 8. Stran energy expressons 8.3 Prncpal of statonary potental energy; several degrees of freedom ------ Castglano s frst theorem ---- Examples 8.4 Prncpal of statonary
PHYS 705: Classical Mechanics. Hamilton-Jacobi Equation
 1 PHYS 705: Classcal Mechancs Hamlton-Jacob Equaton Hamlton-Jacob Equaton There s also a very elegant relaton between the Hamltonan Formulaton of Mechancs and Quantum Mechancs. To do that, we need to derve
1 PHYS 705: Classcal Mechancs Hamlton-Jacob Equaton Hamlton-Jacob Equaton There s also a very elegant relaton between the Hamltonan Formulaton of Mechancs and Quantum Mechancs. To do that, we need to derve
SPANC -- SPlitpole ANalysis Code User Manual
 Functonal Descrpton of Code SPANC -- SPltpole ANalyss Code User Manual Author: Dale Vsser Date: 14 January 00 Spanc s a code created by Dale Vsser for easer calbratons of poston spectra from magnetc spectrometer
Functonal Descrpton of Code SPANC -- SPltpole ANalyss Code User Manual Author: Dale Vsser Date: 14 January 00 Spanc s a code created by Dale Vsser for easer calbratons of poston spectra from magnetc spectrometer
Statistical mechanics handout 4
 Statstcal mechancs handout 4 Explan dfference between phase space and an. Ensembles As dscussed n handout three atoms n any physcal system can adopt any one of a large number of mcorstates. For a quantum
Statstcal mechancs handout 4 Explan dfference between phase space and an. Ensembles As dscussed n handout three atoms n any physcal system can adopt any one of a large number of mcorstates. For a quantum
Physics Nov The Direction of a Reaction A + B C,
 Physcs 301 12-Nov-2003 21-1 Suppose we have a reacton such as The Drecton of a Reacton A + B C whch has come to equlbrum at some temperature τ. Now we rase the temperature. Does the equlbrum shft to the
Physcs 301 12-Nov-2003 21-1 Suppose we have a reacton such as The Drecton of a Reacton A + B C whch has come to equlbrum at some temperature τ. Now we rase the temperature. Does the equlbrum shft to the
ESCI 341 Atmospheric Thermodynamics Lesson 10 The Physical Meaning of Entropy
 ESCI 341 Atmospherc Thermodynamcs Lesson 10 The Physcal Meanng of Entropy References: An Introducton to Statstcal Thermodynamcs, T.L. Hll An Introducton to Thermodynamcs and Thermostatstcs, H.B. Callen
ESCI 341 Atmospherc Thermodynamcs Lesson 10 The Physcal Meanng of Entropy References: An Introducton to Statstcal Thermodynamcs, T.L. Hll An Introducton to Thermodynamcs and Thermostatstcs, H.B. Callen
y i x P vap 10 A T SOLUTION TO HOMEWORK #7 #Problem
 SOLUTION TO HOMEWORK #7 #roblem 1 10.1-1 a. In order to solve ths problem, we need to know what happens at the bubble pont; at ths pont, the frst bubble s formed, so we can assume that all of the number
SOLUTION TO HOMEWORK #7 #roblem 1 10.1-1 a. In order to solve ths problem, we need to know what happens at the bubble pont; at ths pont, the frst bubble s formed, so we can assume that all of the number
THE CHINESE REMAINDER THEOREM. We should thank the Chinese for their wonderful remainder theorem. Glenn Stevens
 THE CHINESE REMAINDER THEOREM KEITH CONRAD We should thank the Chnese for ther wonderful remander theorem. Glenn Stevens 1. Introducton The Chnese remander theorem says we can unquely solve any par of
THE CHINESE REMAINDER THEOREM KEITH CONRAD We should thank the Chnese for ther wonderful remander theorem. Glenn Stevens 1. Introducton The Chnese remander theorem says we can unquely solve any par of
A how to guide to second quantization method.
 Phys. 67 (Graduate Quantum Mechancs Sprng 2009 Prof. Pu K. Lam. Verson 3 (4/3/2009 A how to gude to second quantzaton method. -> Second quantzaton s a mathematcal notaton desgned to handle dentcal partcle
Phys. 67 (Graduate Quantum Mechancs Sprng 2009 Prof. Pu K. Lam. Verson 3 (4/3/2009 A how to gude to second quantzaton method. -> Second quantzaton s a mathematcal notaton desgned to handle dentcal partcle
Inner Product. Euclidean Space. Orthonormal Basis. Orthogonal
 Inner Product Defnton 1 () A Eucldean space s a fnte-dmensonal vector space over the reals R, wth an nner product,. Defnton 2 (Inner Product) An nner product, on a real vector space X s a symmetrc, blnear,
Inner Product Defnton 1 () A Eucldean space s a fnte-dmensonal vector space over the reals R, wth an nner product,. Defnton 2 (Inner Product) An nner product, on a real vector space X s a symmetrc, blnear,
Section 8.3 Polar Form of Complex Numbers
 80 Chapter 8 Secton 8 Polar Form of Complex Numbers From prevous classes, you may have encountered magnary numbers the square roots of negatve numbers and, more generally, complex numbers whch are the
80 Chapter 8 Secton 8 Polar Form of Complex Numbers From prevous classes, you may have encountered magnary numbers the square roots of negatve numbers and, more generally, complex numbers whch are the
LINEAR REGRESSION ANALYSIS. MODULE IX Lecture Multicollinearity
 LINEAR REGRESSION ANALYSIS MODULE IX Lecture - 30 Multcollnearty Dr. Shalabh Department of Mathematcs and Statstcs Indan Insttute of Technology Kanpur 2 Remedes for multcollnearty Varous technques have
LINEAR REGRESSION ANALYSIS MODULE IX Lecture - 30 Multcollnearty Dr. Shalabh Department of Mathematcs and Statstcs Indan Insttute of Technology Kanpur 2 Remedes for multcollnearty Varous technques have
π e ax2 dx = x 2 e ax2 dx or x 3 e ax2 dx = 1 x 4 e ax2 dx = 3 π 8a 5/2 (a) We are considering the Maxwell velocity distribution function: 2πτ/m
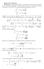 Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
Lecture 12: Discrete Laplacian
 Lecture 12: Dscrete Laplacan Scrbe: Tanye Lu Our goal s to come up wth a dscrete verson of Laplacan operator for trangulated surfaces, so that we can use t n practce to solve related problems We are mostly
Lecture 12: Dscrete Laplacan Scrbe: Tanye Lu Our goal s to come up wth a dscrete verson of Laplacan operator for trangulated surfaces, so that we can use t n practce to solve related problems We are mostly
Homework Assignment 3 Due in class, Thursday October 15
 Homework Assgnment 3 Due n class, Thursday October 15 SDS 383C Statstcal Modelng I 1 Rdge regresson and Lasso 1. Get the Prostrate cancer data from http://statweb.stanford.edu/~tbs/elemstatlearn/ datasets/prostate.data.
Homework Assgnment 3 Due n class, Thursday October 15 SDS 383C Statstcal Modelng I 1 Rdge regresson and Lasso 1. Get the Prostrate cancer data from http://statweb.stanford.edu/~tbs/elemstatlearn/ datasets/prostate.data.
NAME and Section No. it is found that 0.6 mol of O
 NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
Programming Project 1: Molecular Geometry and Rotational Constants
 Programmng Project 1: Molecular Geometry and Rotatonal Constants Center for Computatonal Chemstry Unversty of Georga Athens, Georga 30602 Summer 2012 1 Introducton Ths programmng project s desgned to provde
Programmng Project 1: Molecular Geometry and Rotatonal Constants Center for Computatonal Chemstry Unversty of Georga Athens, Georga 30602 Summer 2012 1 Introducton Ths programmng project s desgned to provde
Lecture 6: Diatomic gases (and others)
 Lecture 6: Datomc gases and others) General rule for calculatng n complex systems Ams: Deal wth a quantsed datomc molecule: Translatonal degrees of freedom last lecture); Rotaton and Vbraton. Partton functon
Lecture 6: Datomc gases and others) General rule for calculatng n complex systems Ams: Deal wth a quantsed datomc molecule: Translatonal degrees of freedom last lecture); Rotaton and Vbraton. Partton functon
Density matrix. c α (t)φ α (q)
 Densty matrx Note: ths s supplementary materal. I strongly recommend that you read t for your own nterest. I beleve t wll help wth understandng the quantum ensembles, but t s not necessary to know t n
Densty matrx Note: ths s supplementary materal. I strongly recommend that you read t for your own nterest. I beleve t wll help wth understandng the quantum ensembles, but t s not necessary to know t n
ECEN 5005 Crystals, Nanocrystals and Device Applications Class 19 Group Theory For Crystals
 ECEN 5005 Crystals, Nanocrystals and Devce Applcatons Class 9 Group Theory For Crystals Dee Dagram Radatve Transton Probablty Wgner-Ecart Theorem Selecton Rule Dee Dagram Expermentally determned energy
ECEN 5005 Crystals, Nanocrystals and Devce Applcatons Class 9 Group Theory For Crystals Dee Dagram Radatve Transton Probablty Wgner-Ecart Theorem Selecton Rule Dee Dagram Expermentally determned energy
Q e E i /k B. i i i i
 Water and Aqueous Solutons 3. Lattce Model of a Flud Lattce Models Lattce models provde a mnmalst, or coarse-graned, framework for descrbng the translatonal, rotatonal, and conformatonal degrees of freedom
Water and Aqueous Solutons 3. Lattce Model of a Flud Lattce Models Lattce models provde a mnmalst, or coarse-graned, framework for descrbng the translatonal, rotatonal, and conformatonal degrees of freedom
Numerical Heat and Mass Transfer
 Master degree n Mechancal Engneerng Numercal Heat and Mass Transfer 06-Fnte-Dfference Method (One-dmensonal, steady state heat conducton) Fausto Arpno f.arpno@uncas.t Introducton Why we use models and
Master degree n Mechancal Engneerng Numercal Heat and Mass Transfer 06-Fnte-Dfference Method (One-dmensonal, steady state heat conducton) Fausto Arpno f.arpno@uncas.t Introducton Why we use models and
(1) The saturation vapor pressure as a function of temperature, often given by the Antoine equation:
 CE304, Sprng 2004 Lecture 22 Lecture 22: Topcs n Phase Equlbra, part : For the remander of the course, we wll return to the subject of vapor/lqud equlbrum and ntroduce other phase equlbrum calculatons
CE304, Sprng 2004 Lecture 22 Lecture 22: Topcs n Phase Equlbra, part : For the remander of the course, we wll return to the subject of vapor/lqud equlbrum and ntroduce other phase equlbrum calculatons
Canonical transformations
 Canoncal transformatons November 23, 2014 Recall that we have defned a symplectc transformaton to be any lnear transformaton M A B leavng the symplectc form nvarant, Ω AB M A CM B DΩ CD Coordnate transformatons,
Canoncal transformatons November 23, 2014 Recall that we have defned a symplectc transformaton to be any lnear transformaton M A B leavng the symplectc form nvarant, Ω AB M A CM B DΩ CD Coordnate transformatons,
One-sided finite-difference approximations suitable for use with Richardson extrapolation
 Journal of Computatonal Physcs 219 (2006) 13 20 Short note One-sded fnte-dfference approxmatons sutable for use wth Rchardson extrapolaton Kumar Rahul, S.N. Bhattacharyya * Department of Mechancal Engneerng,
Journal of Computatonal Physcs 219 (2006) 13 20 Short note One-sded fnte-dfference approxmatons sutable for use wth Rchardson extrapolaton Kumar Rahul, S.N. Bhattacharyya * Department of Mechancal Engneerng,
Using T.O.M to Estimate Parameter of distributions that have not Single Exponential Family
 IOSR Journal of Mathematcs IOSR-JM) ISSN: 2278-5728. Volume 3, Issue 3 Sep-Oct. 202), PP 44-48 www.osrjournals.org Usng T.O.M to Estmate Parameter of dstrbutons that have not Sngle Exponental Famly Jubran
IOSR Journal of Mathematcs IOSR-JM) ISSN: 2278-5728. Volume 3, Issue 3 Sep-Oct. 202), PP 44-48 www.osrjournals.org Usng T.O.M to Estmate Parameter of dstrbutons that have not Sngle Exponental Famly Jubran
Moments of Inertia. and reminds us of the analogous equation for linear momentum p= mv, which is of the form. The kinetic energy of the body is.
 Moments of Inerta Suppose a body s movng on a crcular path wth constant speed Let s consder two quanttes: the body s angular momentum L about the center of the crcle, and ts knetc energy T How are these
Moments of Inerta Suppose a body s movng on a crcular path wth constant speed Let s consder two quanttes: the body s angular momentum L about the center of the crcle, and ts knetc energy T How are these
Note on EM-training of IBM-model 1
 Note on EM-tranng of IBM-model INF58 Language Technologcal Applcatons, Fall The sldes on ths subject (nf58 6.pdf) ncludng the example seem nsuffcent to gve a good grasp of what s gong on. Hence here are
Note on EM-tranng of IBM-model INF58 Language Technologcal Applcatons, Fall The sldes on ths subject (nf58 6.pdf) ncludng the example seem nsuffcent to gve a good grasp of what s gong on. Hence here are
Week 9 Chapter 10 Section 1-5
 Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Workshop: Approximating energies and wave functions Quantum aspects of physical chemistry
 Workshop: Approxmatng energes and wave functons Quantum aspects of physcal chemstry http://quantum.bu.edu/pltl/6/6.pdf Last updated Thursday, November 7, 25 7:9:5-5: Copyrght 25 Dan Dll (dan@bu.edu) Department
Workshop: Approxmatng energes and wave functons Quantum aspects of physcal chemstry http://quantum.bu.edu/pltl/6/6.pdf Last updated Thursday, November 7, 25 7:9:5-5: Copyrght 25 Dan Dll (dan@bu.edu) Department
2.3 Nilpotent endomorphisms
 s a block dagonal matrx, wth A Mat dm U (C) In fact, we can assume that B = B 1 B k, wth B an ordered bass of U, and that A = [f U ] B, where f U : U U s the restrcton of f to U 40 23 Nlpotent endomorphsms
s a block dagonal matrx, wth A Mat dm U (C) In fact, we can assume that B = B 1 B k, wth B an ordered bass of U, and that A = [f U ] B, where f U : U U s the restrcton of f to U 40 23 Nlpotent endomorphsms
3. Be able to derive the chemical equilibrium constants from statistical mechanics.
 Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
For now, let us focus on a specific model of neurons. These are simplified from reality but can achieve remarkable results.
 Neural Networks : Dervaton compled by Alvn Wan from Professor Jtendra Malk s lecture Ths type of computaton s called deep learnng and s the most popular method for many problems, such as computer vson
Neural Networks : Dervaton compled by Alvn Wan from Professor Jtendra Malk s lecture Ths type of computaton s called deep learnng and s the most popular method for many problems, such as computer vson
