The Two Level System: Resonance
|
|
|
- Duane Turner
- 5 years ago
- Views:
Transcription
1 Chapter 1 The Two Level System: Resonance 1.1 Introduction The cornerstone of contemporary Atomic, Molecular and Optical Physics (AMO Physics) is the study of atomic and molecular systems and their interactions through their resonant interaction with applied oscillating electromagnetic fields. The thrust of these studies has evolved continuously since Rabi performed the first resonance experiments in In the decade following World War II the edifice of quantum electrodynamics was constructed largely in response to resonance measurements of unprecedented accuracy on the properties of the electron and the fine and hyperfine structure of simple atoms. At the same time, nuclear magnetic resonance and electron paramagnetic resonance were developed and quickly became essential research tools for chemists and solid state physicists. Molecular beam magnetic and electric resonance studies yielded a wealth of information on the properties of nuclei and molecules, and provided invaluable data for the nuclear physicist and physical chemist. This work continues: the elucidation of basic theory such as quantum mechanics, tests of such quantum electrodynamics, the development of new techniques, the application of old techniques to more systems, and the universal move to even higher precision continues unabated. Molecular beams studies, periodically invigorated by new sources of higher intensity or new species (eg. clusters) are carried out in numerous laboratories - chemical as well as physical - and new methods for applying the techniques of nuclear magnetic resonance are still being developed. Properly practiced, resonance techniques controllably alter the quantum mechanical state of a system without adding any uncertainty. Thus resonance techniques may be used not only to learn about the structure of a system, but also to prepare it in a particular way for further use or study. Because of these two facets, resonance studies have lead physicists through a fundamental change in attitude - from the passive study of atoms to the active control of their internal quantum state and their interactions with the radiation field. This active approach is embodied in the existence of lasers and in the study and creation of coherence phenomena generally. One current frontier in AMO physics is the active control of the external (translational) degree of freedom. The chief technical legacy of the early work on resonance spectroscopy is the 1
2 2 family of lasers which have sprung up like the brooms of the sorcerer s apprentice. The scientific applications of these devices have been prodigious. They caused the resurrection of physical optics- now freshly christened quantum optics- and turned it into one of the liveliest fields in physics. They have had a similar impact on atomic and molecular spectroscopy. In addition they have led to new families of physical studies such as single particle spectroscopy, multiphoton excitation, cavity quantum electrodynamics, and laser cooling and trapping, to name but a few of the many developments. This chapter is about the interactions of a two state system with a sinusoidally oscillating field whose frequency is close to the natural resonance frequency of the system. The phrase two level (ie. two possibly degenerate levels with unspecified m quantum number) is less accurate than the phrase two state. However, its misusage is so widespread that we adopt it anyway - at least it correctly suggests that the two states have different energies. The oscillating field will be treated classically, and the linewidth of both states will be taken as zero until near the end of the chapter where relaxation will be treated phenomenologically Damped Resonance in a Classical System Because the terminology of classical resonance, as well as many of its features, are carried over into quantum mechanics, we start by reviewing an elementary resonant system. Consider a harmonic oscillator composed of a series RLC circuit. The charge obeys q + γ q + ω 2 0γ = 0 (1.1) where γ = R/L, ω0 2 = 1/LC. Assuming that the system is underdamped (i.e. γ 2 < 4ω0 2 ), the solution is a linear combination of ( exp γ ) exp ( ±iω t ) (1.2) 2 where ω = ω 0 1 γ 2 /4ω 2 0. If ω γ, which is often the case, we have ω ω 0. The energy in the circuit is W = 1 2C q L q2 = ω 0 e γt (1.3) where W 0 = W (t = 0). The lifetime of the stored energy if τ = 1 γ. If the circuit is driven by a voltage E 0 e iωt, the steady state solution is q o e iωt where q 0 = E 0 1 2ω 0 L (ω 0 ω + iγ/2. (1.4)
3 Adapted from: W.Ketterle MIT Department of Physics, Spring (We have made the usual resonance approximation: ω 2ω 0 (ω 0 ω).) average power delivered to the circuit is The P = 1 E0 2 2 R ( ω ω0 γ/2 ) 2 (1.5) The plot of P vs ω (Fig. 1) is a universal resonance curve often called a Lorentzian curve. The full width at half maximum ( FWHM ) is ω = γ. The quality factor of the oscillator is Q = ω 0 ω (1.6) Note that the decay time of the free oscillator and the linewidth of the driven oscillator obey τ ω = 1 (1.7) This can be regarded as an uncertainty relation. Assuming that energy and frequency are related by E = hω then the uncertainty in energy is E = h ω and τ E = h (1.8) It is important to realize that the Uncertainty Principle merely characterizes the spread of individual measurements. Ultimate precision depends on the experimenter s skill: the Uncertainty Principle essentially sets the scale of difficulty for his or her efforts. The precision of a resonance measurement is determined by how well one can split the resonance line. This depends on the signal to noise ratio (S/N). (see Fig. 2) As a rule of thumb, the uncertainty δω in the location of the center of the line is δω = ω S/N (1.9) In principle, one can make δω arbitrarily small by acquiring enough data to achieve the required statistical accuracy. In practice, systematic errors eventually limit the precision. Splitting a line by a factor of 10 4 is a formidable task which has only been achieved a few times, most notably in the measurement of the Lamb shift. A factor of 10 3, however, is not uncommon, and 10 2 is child s play.
4 4 1.2 Magnetic Resonance: Classical Spin in Time-varying B-Field Introduction: Electrons, Protons, and Nuclei The two-level system is basic to atomic physics because it approximates accurately many physical systems, particularly systems involving resonance phenomena. All two-level systems obey the same dynamical equations: thus to know one is to know all. The archetype two level system is a spin 1/2 particle such as an electron, proton or neutron. The spin motion of an electron or a proton in a magnetic field, for instance, displays the total range of phenomena in a two level system. To slightly generalize the subject, however, we shall also include the motion of atomic nuclei. Here is a summary of their properties. MASS electron m = g proton M p = g CHARGE ANGULAR MOMENTUM STATISTICS ELECTRON MAGNETIC MOMENT neutron nuclei M p M = AM p A = N + Z = mass number Z = atomic number N = neutron number electron -e e = esu proton +e neutron 0 nucleus Ze electron S = h/2 proton I = h/2 neutron I = h/2 nuclei even A:I/ h = 0, 1, 2,... odd A: I/ h = 1/2, 3/2,... electrons nucleons: Fermi-Dirac odd A, Fermi-Dirac even A, Bose-Einstein µ e = γ e S = g s µ 0 S/ h γ e = gyromagnetic ratio = e/mc = 2π 2.8MHz/gauss g s = free electron g-factor = 2 (Dirac Theory) µ 0 =Bohr magneton = e h/2mc = (erg/gauss) (Note that µ e is negative. We show this explicitly by taking g s to be positive, and writing µ e = g s µ 0 S)
5 Adapted from: W.Ketterle MIT Department of Physics, Spring NUCLEAR MAGNETIC MOMENTS µ N = γ I I = g I µ N I/ h γ I = gyromagnetic ratio of the nucleus µ N = nuclear magneton = e h/2mc = µ 0 (m/m p ) proton g p = 5.6, γ p = 2π 4.2 khz/gauss neutron g N = The Classical Motion of Spins in a Static Magnetic Field The interaction energy and equation of motion of a classical spin in a static magnetic field are given by W = µ B (1.10) F = W = ( µ B) (1.11) torque = µ B (1.12) In a uniform field, F = 0. The torque equation ( d L dt = torque) gives Since µ = γ j, we have h d J dt = µ B (1.13) d J dt = γ J B = γ B J (1.14) To see that the motion of J is pure precession about B, imagine that B is along ẑ and that the spin, J, is tipped at an angle θ from this axis, and then rotated at an angle φ(t) from the ˆx axis (ie., θ and φ are the conventionally chosen angles in spherical coordinates). The torque, γb J, has no component along J (that is, along ˆr), nor along ˆθ (because the J B plane contains ˆθ), hence γb J = δb J sin θ ˆφ. This implies that J maintains constant magnitude and constant tipping angle θ. Since the ˆφ - of component of J is dj/dt = J sin θ dφ dt it is clear that φ(t) = γbt. This solution shows that the moment precesses with angular velocity Ω L = γb (1.15) where Ω L is called the Larmor Frequency. For electrons, γ e /2π = 2.8 MHz/gauss, for protons γ p /2π = 4.2 khz/gauss. Note that Planck s constant does not appear in the equation of motion: the motion is classical.
6 Rotating Coordinate Transformation A second way to find the motion is to look at the problem in a rotating coordinate system. If some vector A rotates with angular velocity Ω, then d A dt = Ω A (1.16) If the rate of change of the vector in a system rotating at Ω is (d A/dt) rot, then the rate of change in an inertial system is the motion in plus the motion of the rotating coordinate system. ( ) da = dt inert ( ) da + Ω dt A (1.17) rot The operator prescription for transforming from an inertial to a rotating system is thus ( d dt) Applying this to Eq.1.14 gives If we let rot ( d = dt) inert Ω (1.18) ( ) dj = γj dt B Ω J = γj ( B + Ω/γ) (1.19) rot Eq becomes B eff = B + Ω/γ (1.20) ( ) dj = γj dt B eff (1.21) rot If B eff = 0, J is constant is the rotating system. The condition for this is as we have previously found in Eq Ω = γ B (1.22)
7 Adapted from: W.Ketterle MIT Department of Physics, Spring Larmor s Theorem Treating the effects of a magnetic field on a magnetic moment by transforming to a rotating co-ordinate system is closely related to Larmor s theorem, which asserts that the effect of a magnetic field on a free charge can be eliminated by a suitable rotating co-ordinate transformation. Consider the motion of a particle of mass m, charge q, under the influence of an applied force F 0 and the Lorentz force due to a static field B: F = F 0 + q c v B (1.23) Now consider the motion in a rotating coordinate system. By applying Eq twice to r, we have ( r) rot = ( r) inert 2 Ω v rot Ω ( Ω r) (1.24) F rot = F inert 2m( Ω v rot ) m Ω ( Ω r) (1.25) where F rot is the apparent force in the rotating system, and F inert is the true or inertial force. Substituting Eq gives F rot = F 0 q c v B + 2m v Ω m Ω ( Ω r) (1.26) If we choose Ω = (q/2mc)ẑ, we have F rot = F 0 mω 2 B 2 ẑ (ẑ r) (1.27) where B = ˆnB. The last term is usually small. If we drop it we have F rot = F 0 (1.28) The effect of the magnetic field is removed by going into a system rotating at the Larmor frequency qb/2mc. Although Larmor s theorem is suggestive of the rotating co-ordinate transformation, Eq. 1.19, it is important to realize that the two transformations, though identical in form, apply to fundamentally different systems. A magnetic moment is not necessarily charged- for example a neutral atom can have a net magnetic moment, and the neutron possesses a magnetic moment in spite of being neutral - and
8 8 it experiences no net force in a uniform magnetic field. Furthermore, the rotating co-ordinate transformation is exact for a magnetic moment, whereas Larmor s theorem for the motion of a charged particle is only valid when the Ω 2 is neglected. 1.3 Motion in a Rotating Magnetic Field Exact Resonance Consider a moment µ precessing about a static field B 0, which we take to lie along the z axis. Its motion might be described by µ x = µ sin θ cos ω 0 t (1.29) µ y = µ sin θ sin ω 0 t µ z = µ cos θ where ω 0 is the Larmor frequency, and θ is the angle the moment makes with B o. Now suppose we introduce a magnetic field B 1 which rotates in the x-y plane at the Larmor frequency ω 0 = γb 0. The magnetic field is B(t) = B 1 (ˆx cos ω 0 t ŷ sin ω 0 t) + B 0 ẑ. (1.30) The problem is to find the motion of µ. The solution is simple in a rotating coordinate system. Let system (ˆx, ŷ, ẑ = ẑ) precess around the z-axis at rate ω 0. In this system the field B 1 is stationary (and ˆx is chosen to lie along B 1 ), and we have B(t) eff = B(t) ω 0 /γẑ (1.31) = B 1ˆx + (B 0 ω 0 /γ)ẑ = B 1ˆx.
9 Adapted from: W.Ketterle MIT Department of Physics, Spring The effective field is static and has the value of B. The moment precesses about the field at rate ω R = γb 1, (1.32) often called the Rabi frequency This equation contains a lot of history: the RF magnetic resonance community conventionally calls this frequency ω 1, but the laser resonance community calls it the Rabi Frequency ω R in honor of Rabi s invention of the resonance technique. If the moment initially lies along the z axis, then its tip traces a circle in the ŷ ẑ plane. At time t it has precessed through an angle φ = ω R t. The moment s z-component is given by µ z (t) = µ cos ω R t (1.33) At time T = π/ω R, the moment points along the negative z-axis: it has turned over Off-Resonance Behavior Now suppose that the field B 1 rotates at frequency ω ω 0. In a coordinate frame rotating with B 1 the effective field is B eff = B 1ˆx + (B 0 ω/γ)ẑ. (1.34)
10 10 The effective field lies at angle θ with the z-axis, as shown. (Beware: there is a close correspondence between the resonance we are doing here and the dressed atom, but this θ, call it θ res = 2θ dressed.) The field is static, and the moment precesses about it at rate (called the effective Rabi frequency ) ω R = γb eff = γ (B 0 ω/γ) 2 + B1 2 (1.35) = (ω 0 ω) 2 + ωr 2 where ω 0 = γb 0, ω R = γb 1, as before. Assume that µ points initially along the +z-axis. Finding µ z (t) is a straightforward problem in geometry. The moment precesses about B eff at rate ω R, sweeping a circle as shown. The radius of the circle is µ sin θ, where sin θ = B 1 / (B 0 ω/γ) 2 + B1 2 (1.36) = ω R / (ω ω 0 ) 2 + ωr 2. (1.37) In time t the tip sweeps through angle The z-component of the moment is φ = ω R t. µ z (t) = µ cos α
11 Adapted from: W.Ketterle MIT Department of Physics, Spring where α is the angle between the moment and the z-axis after it has precessed through angle φ. As the drawing shows, cos α is found from A 2 = 2µ 2 (1 cos α). Since we have and A = 2µ sin θ sin(ω Rt/2) 4µ 2 sin 2 θ sin 2 (ω Rt/2) = 2µ 2 (1 cos α) µ z (t) = µ cos α = µ(1 2 sin 2 θ sin 2 1/2ω Rt) (1.38) [ ωr 2 = µ 1 2 (ω ω 0 ) 2 + ωr 2 sin 2 1 ] (ω ω 0 ) ω R t [ ] = µ 1 2(ω R /ω R) 2 sin 2 (ω Rt/2) The z-component of µ oscillates in time, but unless ω = ω 0, the moment never completely inverts. The rate of oscillation depends on the magnitude of the rotating field; the amplitude of oscillation depends on the frequency difference, ω ω 0, relative to ω R. The quantum mechanical result is identical.
Physics 221A Fall 1996 Notes 13 Spins in Magnetic Fields
 Physics 221A Fall 1996 Notes 13 Spins in Magnetic Fields A nice illustration of rotation operator methods which is also important physically is the problem of spins in magnetic fields. The earliest experiments
Physics 221A Fall 1996 Notes 13 Spins in Magnetic Fields A nice illustration of rotation operator methods which is also important physically is the problem of spins in magnetic fields. The earliest experiments
Spectral Broadening Mechanisms
 Spectral Broadening Mechanisms Lorentzian broadening (Homogeneous) Gaussian broadening (Inhomogeneous, Inertial) Doppler broadening (special case for gas phase) The Fourier Transform NC State University
Spectral Broadening Mechanisms Lorentzian broadening (Homogeneous) Gaussian broadening (Inhomogeneous, Inertial) Doppler broadening (special case for gas phase) The Fourier Transform NC State University
Spectral Broadening Mechanisms. Broadening mechanisms. Lineshape functions. Spectral lifetime broadening
 Spectral Broadening echanisms Lorentzian broadening (Homogeneous) Gaussian broadening (Inhomogeneous, Inertial) Doppler broadening (special case for gas phase) The Fourier Transform NC State University
Spectral Broadening echanisms Lorentzian broadening (Homogeneous) Gaussian broadening (Inhomogeneous, Inertial) Doppler broadening (special case for gas phase) The Fourier Transform NC State University
Chemistry 431. Lecture 23
 Chemistry 431 Lecture 23 Introduction The Larmor Frequency The Bloch Equations Measuring T 1 : Inversion Recovery Measuring T 2 : the Spin Echo NC State University NMR spectroscopy The Nuclear Magnetic
Chemistry 431 Lecture 23 Introduction The Larmor Frequency The Bloch Equations Measuring T 1 : Inversion Recovery Measuring T 2 : the Spin Echo NC State University NMR spectroscopy The Nuclear Magnetic
NMR Spectroscopy Laboratory Experiment Introduction. 2. Theory
 1. Introduction 64-311 Laboratory Experiment 11 NMR Spectroscopy Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful and theoretically complex analytical tool. This experiment will introduce to
1. Introduction 64-311 Laboratory Experiment 11 NMR Spectroscopy Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful and theoretically complex analytical tool. This experiment will introduce to
Part I. Principles and techniques
 Part I Principles and techniques 1 General principles and characteristics of optical magnetometers D. F. Jackson Kimball, E. B. Alexandrov, and D. Budker 1.1 Introduction Optical magnetometry encompasses
Part I Principles and techniques 1 General principles and characteristics of optical magnetometers D. F. Jackson Kimball, E. B. Alexandrov, and D. Budker 1.1 Introduction Optical magnetometry encompasses
The NMR Inverse Imaging Problem
 The NMR Inverse Imaging Problem Nuclear Magnetic Resonance Protons and Neutrons have intrinsic angular momentum Atoms with an odd number of proton and/or odd number of neutrons have a net magnetic moment=>
The NMR Inverse Imaging Problem Nuclear Magnetic Resonance Protons and Neutrons have intrinsic angular momentum Atoms with an odd number of proton and/or odd number of neutrons have a net magnetic moment=>
10.4 Continuous Wave NMR Instrumentation
 10.4 Continuous Wave NMR Instrumentation coherent detection bulk magnetization the rotating frame, and effective magnetic field generating a rotating frame, and precession in the laboratory frame spin-lattice
10.4 Continuous Wave NMR Instrumentation coherent detection bulk magnetization the rotating frame, and effective magnetic field generating a rotating frame, and precession in the laboratory frame spin-lattice
NMR, the vector model and the relaxation
 NMR, the vector model and the relaxation Reading/Books: One and two dimensional NMR spectroscopy, VCH, Friebolin Spin Dynamics, Basics of NMR, Wiley, Levitt Molecular Quantum Mechanics, Oxford Univ. Press,
NMR, the vector model and the relaxation Reading/Books: One and two dimensional NMR spectroscopy, VCH, Friebolin Spin Dynamics, Basics of NMR, Wiley, Levitt Molecular Quantum Mechanics, Oxford Univ. Press,
V27: RF Spectroscopy
 Martin-Luther-Universität Halle-Wittenberg FB Physik Advanced Lab Course V27: RF Spectroscopy ) Electron spin resonance (ESR) Investigate the resonance behaviour of two coupled LC circuits (an active rf
Martin-Luther-Universität Halle-Wittenberg FB Physik Advanced Lab Course V27: RF Spectroscopy ) Electron spin resonance (ESR) Investigate the resonance behaviour of two coupled LC circuits (an active rf
Spin resonance. Basic idea. PSC 3151, (301)
 Spin Resonance Phys623 Spring 2018 Prof. Ted Jacobson PSC 3151, (301)405-6020 jacobson@physics.umd.edu Spin resonance Spin resonance refers to the enhancement of a spin flipping probability in a magnetic
Spin Resonance Phys623 Spring 2018 Prof. Ted Jacobson PSC 3151, (301)405-6020 jacobson@physics.umd.edu Spin resonance Spin resonance refers to the enhancement of a spin flipping probability in a magnetic
Classical Description of NMR Parameters: The Bloch Equations
 Classical Description of NMR Parameters: The Bloch Equations Pascale Legault Département de Biochimie Université de Montréal 1 Outline 1) Classical Behavior of Magnetic Nuclei: The Bloch Equation 2) Precession
Classical Description of NMR Parameters: The Bloch Equations Pascale Legault Département de Biochimie Université de Montréal 1 Outline 1) Classical Behavior of Magnetic Nuclei: The Bloch Equation 2) Precession
Relativistic corrections of energy terms
 Lectures 2-3 Hydrogen atom. Relativistic corrections of energy terms: relativistic mass correction, Darwin term, and spin-orbit term. Fine structure. Lamb shift. Hyperfine structure. Energy levels of the
Lectures 2-3 Hydrogen atom. Relativistic corrections of energy terms: relativistic mass correction, Darwin term, and spin-orbit term. Fine structure. Lamb shift. Hyperfine structure. Energy levels of the
2m 2 Ze2. , where δ. ) 2 l,n is the quantum defect (of order one but larger
 PHYS 402, Atomic and Molecular Physics Spring 2017, final exam, solutions 1. Hydrogenic atom energies: Consider a hydrogenic atom or ion with nuclear charge Z and the usual quantum states φ nlm. (a) (2
PHYS 402, Atomic and Molecular Physics Spring 2017, final exam, solutions 1. Hydrogenic atom energies: Consider a hydrogenic atom or ion with nuclear charge Z and the usual quantum states φ nlm. (a) (2
Magnetic Resonance Spectroscopy EPR and NMR
 Magnetic Resonance Spectroscopy EPR and NMR A brief review of the relevant bits of quantum mechanics 1. Electrons have spin, - rotation of the charge about its axis generates a magnetic field at each electron.
Magnetic Resonance Spectroscopy EPR and NMR A brief review of the relevant bits of quantum mechanics 1. Electrons have spin, - rotation of the charge about its axis generates a magnetic field at each electron.
An Introduction to Hyperfine Structure and Its G-factor
 An Introduction to Hyperfine Structure and Its G-factor Xiqiao Wang East Tennessee State University April 25, 2012 1 1. Introduction In a book chapter entitled Model Calculations of Radiation Induced Damage
An Introduction to Hyperfine Structure and Its G-factor Xiqiao Wang East Tennessee State University April 25, 2012 1 1. Introduction In a book chapter entitled Model Calculations of Radiation Induced Damage
PHYSICAL SCIENCES PART A
 PHYSICAL SCIENCES PART A 1. The calculation of the probability of excitation of an atom originally in the ground state to an excited state, involves the contour integral iωt τ e dt ( t τ ) + Evaluate the
PHYSICAL SCIENCES PART A 1. The calculation of the probability of excitation of an atom originally in the ground state to an excited state, involves the contour integral iωt τ e dt ( t τ ) + Evaluate the
Magnetic Resonance Spectroscopy ( )
 Magnetic Resonance Spectroscopy In our discussion of spectroscopy, we have shown that absorption of E.M. radiation occurs on resonance: When the frequency of applied E.M. field matches the energy splitting
Magnetic Resonance Spectroscopy In our discussion of spectroscopy, we have shown that absorption of E.M. radiation occurs on resonance: When the frequency of applied E.M. field matches the energy splitting
Classical Description of NMR Parameters: The Bloch Equations
 Classical Description of NMR Parameters: The Bloch Equations Pascale Legault Département de Biochimie Université de Montréal 1 Outline 1) Classical Behavior of Magnetic Nuclei: The Bloch Equation 2) Precession
Classical Description of NMR Parameters: The Bloch Equations Pascale Legault Département de Biochimie Université de Montréal 1 Outline 1) Classical Behavior of Magnetic Nuclei: The Bloch Equation 2) Precession
Fundamental MRI Principles Module 2 N. Nuclear Magnetic Resonance. X-ray. MRI Hydrogen Protons. Page 1. Electrons
 Fundamental MRI Principles Module 2 N S 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons positively charged neutrons no significant charge electrons negatively charged Protons
Fundamental MRI Principles Module 2 N S 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons positively charged neutrons no significant charge electrons negatively charged Protons
Ion traps. Trapping of charged particles in electromagnetic. Laser cooling, sympathetic cooling, optical clocks
 Ion traps Trapping of charged particles in electromagnetic fields Dynamics of trapped ions Applications to nuclear physics and QED The Paul trap Laser cooling, sympathetic cooling, optical clocks Coulomb
Ion traps Trapping of charged particles in electromagnetic fields Dynamics of trapped ions Applications to nuclear physics and QED The Paul trap Laser cooling, sympathetic cooling, optical clocks Coulomb
Saturation Absorption Spectroscopy of Rubidium Atom
 Saturation Absorption Spectroscopy of Rubidium Atom Jayash Panigrahi August 17, 2013 Abstract Saturated absorption spectroscopy has various application in laser cooling which have many relevant uses in
Saturation Absorption Spectroscopy of Rubidium Atom Jayash Panigrahi August 17, 2013 Abstract Saturated absorption spectroscopy has various application in laser cooling which have many relevant uses in
Properties of Elementary Particles
 and of Elementary s 01/11/2018 My Office Hours: Thursday 1:00-3:00 PM 212 Keen Building Outline 1 2 3 Consider the world at different scales... Cosmology - only gravity matters XXXXX Input: Mass distributions
and of Elementary s 01/11/2018 My Office Hours: Thursday 1:00-3:00 PM 212 Keen Building Outline 1 2 3 Consider the world at different scales... Cosmology - only gravity matters XXXXX Input: Mass distributions
1 Magnetism, Curie s Law and the Bloch Equations
 1 Magnetism, Curie s Law and the Bloch Equations In NMR, the observable which is measured is magnetization and its evolution over time. In order to understand what this means, let us first begin with some
1 Magnetism, Curie s Law and the Bloch Equations In NMR, the observable which is measured is magnetization and its evolution over time. In order to understand what this means, let us first begin with some
B2.III Revision notes: quantum physics
 B.III Revision notes: quantum physics Dr D.M.Lucas, TT 0 These notes give a summary of most of the Quantum part of this course, to complement Prof. Ewart s notes on Atomic Structure, and Prof. Hooker s
B.III Revision notes: quantum physics Dr D.M.Lucas, TT 0 These notes give a summary of most of the Quantum part of this course, to complement Prof. Ewart s notes on Atomic Structure, and Prof. Hooker s
Atomic Structure. Chapter 8
 Atomic Structure Chapter 8 Overview To understand atomic structure requires understanding a special aspect of the electron - spin and its related magnetism - and properties of a collection of identical
Atomic Structure Chapter 8 Overview To understand atomic structure requires understanding a special aspect of the electron - spin and its related magnetism - and properties of a collection of identical
Atomic cross sections
 Chapter 12 Atomic cross sections The probability that an absorber (atom of a given species in a given excitation state and ionziation level) will interact with an incident photon of wavelength λ is quantified
Chapter 12 Atomic cross sections The probability that an absorber (atom of a given species in a given excitation state and ionziation level) will interact with an incident photon of wavelength λ is quantified
Uncertainty principle
 Chapter 3 Uncertainty principle Now it is Amperé s hypotheis that the source of all magnetic fields is the motion of charges. In particular, magnetic dipole moments arise from the circulation of charge.
Chapter 3 Uncertainty principle Now it is Amperé s hypotheis that the source of all magnetic fields is the motion of charges. In particular, magnetic dipole moments arise from the circulation of charge.
Nuclear Magnetic Resonance Imaging
 Nuclear Magnetic Resonance Imaging Simon Lacoste-Julien Electromagnetic Theory Project 198-562B Department of Physics McGill University April 21 2003 Abstract This paper gives an elementary introduction
Nuclear Magnetic Resonance Imaging Simon Lacoste-Julien Electromagnetic Theory Project 198-562B Department of Physics McGill University April 21 2003 Abstract This paper gives an elementary introduction
5.61 Physical Chemistry Lecture #36 Page
 5.61 Physical Chemistry Lecture #36 Page 1 NUCLEAR MAGNETIC RESONANCE Just as IR spectroscopy is the simplest example of transitions being induced by light s oscillating electric field, so NMR is the simplest
5.61 Physical Chemistry Lecture #36 Page 1 NUCLEAR MAGNETIC RESONANCE Just as IR spectroscopy is the simplest example of transitions being induced by light s oscillating electric field, so NMR is the simplest
Shimming of a Magnet for Calibration of NMR Probes UW PHYSICS REU 2013
 Shimming of a Magnet for Calibration of NMR Probes RACHEL BIELAJEW UW PHYSICS REU 2013 Outline Background The muon anomaly The g-2 Experiment NMR Design Helmholtz coils producing a gradient Results Future
Shimming of a Magnet for Calibration of NMR Probes RACHEL BIELAJEW UW PHYSICS REU 2013 Outline Background The muon anomaly The g-2 Experiment NMR Design Helmholtz coils producing a gradient Results Future
A Hands on Introduction to NMR Lecture #1 Nuclear Spin and Magnetic Resonance
 A Hands on Introduction to NMR 22.920 Lecture #1 Nuclear Spin and Magnetic Resonance Introduction - The aim of this short course is to present a physical picture of the basic principles of Nuclear Magnetic
A Hands on Introduction to NMR 22.920 Lecture #1 Nuclear Spin and Magnetic Resonance Introduction - The aim of this short course is to present a physical picture of the basic principles of Nuclear Magnetic
Optical Lattices. Chapter Polarization
 Chapter Optical Lattices Abstract In this chapter we give details of the atomic physics that underlies the Bose- Hubbard model used to describe ultracold atoms in optical lattices. We show how the AC-Stark
Chapter Optical Lattices Abstract In this chapter we give details of the atomic physics that underlies the Bose- Hubbard model used to describe ultracold atoms in optical lattices. We show how the AC-Stark
Fluorescence Spectroscopy
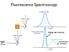 Fluorescence Spectroscopy Raleigh light scattering light all freqs Fluorescence emission Raleigh Scattering 10 nm Raleigh light scattering Fluorescence emission 400 nm Scattering - TWO particle 10 nm Particle
Fluorescence Spectroscopy Raleigh light scattering light all freqs Fluorescence emission Raleigh Scattering 10 nm Raleigh light scattering Fluorescence emission 400 nm Scattering - TWO particle 10 nm Particle
PRELIMINARY EXAMINATION Department of Physics University of Florida Part A, January, 2012, 09:00 12:00. Instructions
 Student ID Number: PRELIMINARY EXAMINATION Part A, January, 2012, 09:00 12:00 Instructions 1. You may use a calculator and CRC Math tables or equivalent. No other tables or aids are allowed or required.
Student ID Number: PRELIMINARY EXAMINATION Part A, January, 2012, 09:00 12:00 Instructions 1. You may use a calculator and CRC Math tables or equivalent. No other tables or aids are allowed or required.
10.3 NMR Fundamentals
 10.3 NMR Fundamentals nuclear spin calculations and examples NMR properties of selected nuclei the nuclear magnetic moment and precession around a magnetic field the spin quantum number and the NMR transition
10.3 NMR Fundamentals nuclear spin calculations and examples NMR properties of selected nuclei the nuclear magnetic moment and precession around a magnetic field the spin quantum number and the NMR transition
Biophysical Chemistry: NMR Spectroscopy
 Spin Dynamics & Vrije Universiteit Brussel 25th November 2011 Outline 1 Pulse/Fourier Transform NMR Thermal Equilibrium Effect of RF Pulses The Fourier Transform 2 Symmetric Exchange Between Two Sites
Spin Dynamics & Vrije Universiteit Brussel 25th November 2011 Outline 1 Pulse/Fourier Transform NMR Thermal Equilibrium Effect of RF Pulses The Fourier Transform 2 Symmetric Exchange Between Two Sites
Nuclear Magnetic Resonance Spectroscopy
 Nuclear Magnetic Resonance Spectroscopy Ecole Polytechnique Département de Chimie CHI 551 Dr. Grégory Nocton Bureau 01 30 11 A Tel: 44 02 Ecole polytechnique / CNRS Laboratoire de Chimie Moléculaire E-mail:
Nuclear Magnetic Resonance Spectroscopy Ecole Polytechnique Département de Chimie CHI 551 Dr. Grégory Nocton Bureau 01 30 11 A Tel: 44 02 Ecole polytechnique / CNRS Laboratoire de Chimie Moléculaire E-mail:
Table 1: Coefficients found for the damped sinusoids [asin(bx+c)+d]exp(-kx)
![Table 1: Coefficients found for the damped sinusoids [asin(bx+c)+d]exp(-kx) Table 1: Coefficients found for the damped sinusoids [asin(bx+c)+d]exp(-kx)](/thumbs/84/91193477.jpg) Searching harmonics in nuclei gyromagnetic ratios. Startling emergence of scaling, pseudo octaves, and the fine-structure constant from a seemingly random network. Bruno Galeffi, Chemist, PhD Québec, Canada
Searching harmonics in nuclei gyromagnetic ratios. Startling emergence of scaling, pseudo octaves, and the fine-structure constant from a seemingly random network. Bruno Galeffi, Chemist, PhD Québec, Canada
VIII. NUCLEAR MAGNETIC RESONANCE (NMR) SPECTROSCOPY
 1 VIII. NUCLEAR MAGNETIC RESONANCE (NMR) SPECTROSCOPY Molecules are extremely small entities; thus, their direct detection and direct investigation is still almost impossible. For the detection and detailed
1 VIII. NUCLEAR MAGNETIC RESONANCE (NMR) SPECTROSCOPY Molecules are extremely small entities; thus, their direct detection and direct investigation is still almost impossible. For the detection and detailed
Chem 325 NMR Intro. The Electromagnetic Spectrum. Physical properties, chemical properties, formulas Shedding real light on molecular structure:
 Physical properties, chemical properties, formulas Shedding real light on molecular structure: Wavelength Frequency ν Wavelength λ Frequency ν Velocity c = 2.998 10 8 m s -1 The Electromagnetic Spectrum
Physical properties, chemical properties, formulas Shedding real light on molecular structure: Wavelength Frequency ν Wavelength λ Frequency ν Velocity c = 2.998 10 8 m s -1 The Electromagnetic Spectrum
Physical Background Of Nuclear Magnetic Resonance Spectroscopy
 Physical Background Of Nuclear Magnetic Resonance Spectroscopy Michael McClellan Spring 2009 Department of Physics and Physical Oceanography University of North Carolina Wilmington What is Spectroscopy?
Physical Background Of Nuclear Magnetic Resonance Spectroscopy Michael McClellan Spring 2009 Department of Physics and Physical Oceanography University of North Carolina Wilmington What is Spectroscopy?
Fundamentals of Spectroscopy for Optical Remote Sensing. Course Outline 2009
 Fundamentals of Spectroscopy for Optical Remote Sensing Course Outline 2009 Part I. Fundamentals of Quantum Mechanics Chapter 1. Concepts of Quantum and Experimental Facts 1.1. Blackbody Radiation and
Fundamentals of Spectroscopy for Optical Remote Sensing Course Outline 2009 Part I. Fundamentals of Quantum Mechanics Chapter 1. Concepts of Quantum and Experimental Facts 1.1. Blackbody Radiation and
Biophysical Chemistry: NMR Spectroscopy
 Nuclear Magnetism Vrije Universiteit Brussel 21st October 2011 Outline 1 Overview and Context 2 3 Outline 1 Overview and Context 2 3 Context Proteins (and other biological macromolecules) Functional characterisation
Nuclear Magnetism Vrije Universiteit Brussel 21st October 2011 Outline 1 Overview and Context 2 3 Outline 1 Overview and Context 2 3 Context Proteins (and other biological macromolecules) Functional characterisation
(a) Write down the total Hamiltonian of this system, including the spin degree of freedom of the electron, but neglecting spin-orbit interactions.
 1. Quantum Mechanics (Spring 2007) Consider a hydrogen atom in a weak uniform magnetic field B = Bê z. (a) Write down the total Hamiltonian of this system, including the spin degree of freedom of the electron,
1. Quantum Mechanics (Spring 2007) Consider a hydrogen atom in a weak uniform magnetic field B = Bê z. (a) Write down the total Hamiltonian of this system, including the spin degree of freedom of the electron,
The Nuclear Emphasis
 The Nuclear Emphasis Atoms are composed of electrons and nuclei we ll focus almost exclusively on the physical properties of the nucleus and the chemicoelectronic attributes of its environment. The nucleus
The Nuclear Emphasis Atoms are composed of electrons and nuclei we ll focus almost exclusively on the physical properties of the nucleus and the chemicoelectronic attributes of its environment. The nucleus
Circular motion. Aug. 22, 2017
 Circular motion Aug. 22, 2017 Until now, we have been observers to Newtonian physics through inertial reference frames. From our discussion of Newton s laws, these are frames which obey Newton s first
Circular motion Aug. 22, 2017 Until now, we have been observers to Newtonian physics through inertial reference frames. From our discussion of Newton s laws, these are frames which obey Newton s first
Fundamental MRI Principles Module Two
 Fundamental MRI Principles Module Two 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons neutrons electrons positively charged no significant charge negatively charged Protons
Fundamental MRI Principles Module Two 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons neutrons electrons positively charged no significant charge negatively charged Protons
( ) /, so that we can ignore all
 Physics 531: Atomic Physics Problem Set #5 Due Wednesday, November 2, 2011 Problem 1: The ac-stark effect Suppose an atom is perturbed by a monochromatic electric field oscillating at frequency ω L E(t)
Physics 531: Atomic Physics Problem Set #5 Due Wednesday, November 2, 2011 Problem 1: The ac-stark effect Suppose an atom is perturbed by a monochromatic electric field oscillating at frequency ω L E(t)
OPTI 511R: OPTICAL PHYSICS & LASERS
 OPTI 511R: OPTICAL PHYSICS & LASERS Instructor: R. Jason Jones Office Hours: TBD Teaching Assistant: Robert Rockmore Office Hours: Wed. (TBD) h"p://wp.op)cs.arizona.edu/op)511r/ h"p://wp.op)cs.arizona.edu/op)511r/
OPTI 511R: OPTICAL PHYSICS & LASERS Instructor: R. Jason Jones Office Hours: TBD Teaching Assistant: Robert Rockmore Office Hours: Wed. (TBD) h"p://wp.op)cs.arizona.edu/op)511r/ h"p://wp.op)cs.arizona.edu/op)511r/
UNIVERSITY OF SOUTHAMPTON
 UNIVERSITY OF SOUTHAMPTON PHYS6012W1 SEMESTER 1 EXAMINATION 2012/13 Coherent Light, Coherent Matter Duration: 120 MINS Answer all questions in Section A and only two questions in Section B. Section A carries
UNIVERSITY OF SOUTHAMPTON PHYS6012W1 SEMESTER 1 EXAMINATION 2012/13 Coherent Light, Coherent Matter Duration: 120 MINS Answer all questions in Section A and only two questions in Section B. Section A carries
MR Fundamentals. 26 October Mitglied der Helmholtz-Gemeinschaft
 MR Fundamentals 26 October 2010 Mitglied der Helmholtz-Gemeinschaft Mitglied der Helmholtz-Gemeinschaft Nuclear Spin Nuclear Spin Nuclear magnetic resonance is observed in atoms with odd number of protons
MR Fundamentals 26 October 2010 Mitglied der Helmholtz-Gemeinschaft Mitglied der Helmholtz-Gemeinschaft Nuclear Spin Nuclear Spin Nuclear magnetic resonance is observed in atoms with odd number of protons
Electron spins in nonmagnetic semiconductors
 Electron spins in nonmagnetic semiconductors Yuichiro K. Kato Institute of Engineering Innovation, The University of Tokyo Physics of non-interacting spins Optical spin injection and detection Spin manipulation
Electron spins in nonmagnetic semiconductors Yuichiro K. Kato Institute of Engineering Innovation, The University of Tokyo Physics of non-interacting spins Optical spin injection and detection Spin manipulation
ELECTRON SPIN RESONANCE & MAGNETIC RESONANCE TOMOGRAPHY
 ELECTRON SPIN RESONANCE & MAGNETIC RESONANCE TOMOGRAPHY 1. AIM OF THE EXPERIMENT This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment
ELECTRON SPIN RESONANCE & MAGNETIC RESONANCE TOMOGRAPHY 1. AIM OF THE EXPERIMENT This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment
Electron spin resonance
 Quick reference guide Introduction This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment of the electron spin with a superimposed direct
Quick reference guide Introduction This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment of the electron spin with a superimposed direct
NANOSCALE SCIENCE & TECHNOLOGY
 . NANOSCALE SCIENCE & TECHNOLOGY V Two-Level Quantum Systems (Qubits) Lecture notes 5 5. Qubit description Quantum bit (qubit) is an elementary unit of a quantum computer. Similar to classical computers,
. NANOSCALE SCIENCE & TECHNOLOGY V Two-Level Quantum Systems (Qubits) Lecture notes 5 5. Qubit description Quantum bit (qubit) is an elementary unit of a quantum computer. Similar to classical computers,
Magnetic Resonance Imaging. Pål Erik Goa Associate Professor in Medical Imaging Dept. of Physics
 Magnetic Resonance Imaging Pål Erik Goa Associate Professor in Medical Imaging Dept. of Physics pal.e.goa@ntnu.no 1 Why MRI? X-ray/CT: Great for bone structures and high spatial resolution Not so great
Magnetic Resonance Imaging Pål Erik Goa Associate Professor in Medical Imaging Dept. of Physics pal.e.goa@ntnu.no 1 Why MRI? X-ray/CT: Great for bone structures and high spatial resolution Not so great
We have seen that the total magnetic moment or magnetization, M, of a sample of nuclear spins is the sum of the nuclear moments and is given by:
 Bloch Equations We have seen that the total magnetic moment or magnetization, M, of a sample of nuclear spins is the sum of the nuclear moments and is given by: M = [] µ i i In terms of the total spin
Bloch Equations We have seen that the total magnetic moment or magnetization, M, of a sample of nuclear spins is the sum of the nuclear moments and is given by: M = [] µ i i In terms of the total spin
Phys 622 Problems Chapter 5
 1 Phys 622 Problems Chapter 5 Problem 1 The correct basis set of perturbation theory Consider the relativistic correction to the electron-nucleus interaction H LS = α L S, also known as the spin-orbit
1 Phys 622 Problems Chapter 5 Problem 1 The correct basis set of perturbation theory Consider the relativistic correction to the electron-nucleus interaction H LS = α L S, also known as the spin-orbit
Measuring Spin-Lattice Relaxation Time
 WJP, PHY381 (2009) Wabash Journal of Physics v4.0, p.1 Measuring Spin-Lattice Relaxation Time L.W. Lupinski, R. Paudel, and M.J. Madsen Department of Physics, Wabash College, Crawfordsville, IN 47933 (Dated:
WJP, PHY381 (2009) Wabash Journal of Physics v4.0, p.1 Measuring Spin-Lattice Relaxation Time L.W. Lupinski, R. Paudel, and M.J. Madsen Department of Physics, Wabash College, Crawfordsville, IN 47933 (Dated:
Topics. The concept of spin Precession of magnetic spin Relaxation Bloch Equation. Bioengineering 280A Principles of Biomedical Imaging
 Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2006 MRI Lecture 1 Topics The concept of spin Precession of magnetic spin Relaxation Bloch Equation 1 Spin Intrinsic angular momentum of
Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2006 MRI Lecture 1 Topics The concept of spin Precession of magnetic spin Relaxation Bloch Equation 1 Spin Intrinsic angular momentum of
Physics 221 Lecture 31 Line Radiation from Atoms and Molecules March 31, 1999
 Physics 221 Lecture 31 Line Radiation from Atoms and Molecules March 31, 1999 Reading Meyer-Arendt, Ch. 20; Möller, Ch. 15; Yariv, Ch.. Demonstrations Analyzing lineshapes from emission and absorption
Physics 221 Lecture 31 Line Radiation from Atoms and Molecules March 31, 1999 Reading Meyer-Arendt, Ch. 20; Möller, Ch. 15; Yariv, Ch.. Demonstrations Analyzing lineshapes from emission and absorption
INTRODUCTION TO NMR and NMR QIP
 Books (NMR): Spin dynamics: basics of nuclear magnetic resonance, M. H. Levitt, Wiley, 2001. The principles of nuclear magnetism, A. Abragam, Oxford, 1961. Principles of magnetic resonance, C. P. Slichter,
Books (NMR): Spin dynamics: basics of nuclear magnetic resonance, M. H. Levitt, Wiley, 2001. The principles of nuclear magnetism, A. Abragam, Oxford, 1961. Principles of magnetic resonance, C. P. Slichter,
ATOMIC AND LASER SPECTROSCOPY
 ALAN CORNEY ATOMIC AND LASER SPECTROSCOPY CLARENDON PRESS OXFORD 1977 Contents 1. INTRODUCTION 1.1. Planck's radiation law. 1 1.2. The photoelectric effect 4 1.3. Early atomic spectroscopy 5 1.4. The postulates
ALAN CORNEY ATOMIC AND LASER SPECTROSCOPY CLARENDON PRESS OXFORD 1977 Contents 1. INTRODUCTION 1.1. Planck's radiation law. 1 1.2. The photoelectric effect 4 1.3. Early atomic spectroscopy 5 1.4. The postulates
9. Transitions between Magnetic Levels Spin Transitions Between Spin States. Conservation of Spin Angular Momentum
 9. Transitions between Magnetic Levels pin Transitions Between pin tates. Conservation of pin Angular Momentum From the magnetic energy diagram derived in the previous sections (Figures 14, 15 and 16),
9. Transitions between Magnetic Levels pin Transitions Between pin tates. Conservation of pin Angular Momentum From the magnetic energy diagram derived in the previous sections (Figures 14, 15 and 16),
Introduction to MRI. Spin & Magnetic Moments. Relaxation (T1, T2) Spin Echoes. 2DFT Imaging. K-space & Spatial Resolution.
 Introduction to MRI Spin & Magnetic Moments Relaxation (T1, T2) Spin Echoes 2DFT Imaging Selective excitation, phase & frequency encoding K-space & Spatial Resolution Contrast (T1, T2) Acknowledgement:
Introduction to MRI Spin & Magnetic Moments Relaxation (T1, T2) Spin Echoes 2DFT Imaging Selective excitation, phase & frequency encoding K-space & Spatial Resolution Contrast (T1, T2) Acknowledgement:
Intro to Nuclear and Particle Physics (5110)
 Intro to Nuclear and Particle Physics (5110) March 13, 009 Nuclear Shell Model continued 3/13/009 1 Atomic Physics Nuclear Physics V = V r f r L r S r Tot Spin-Orbit Interaction ( ) ( ) Spin of e magnetic
Intro to Nuclear and Particle Physics (5110) March 13, 009 Nuclear Shell Model continued 3/13/009 1 Atomic Physics Nuclear Physics V = V r f r L r S r Tot Spin-Orbit Interaction ( ) ( ) Spin of e magnetic
Time dependent perturbation theory 1 D. E. Soper 2 University of Oregon 11 May 2012
 Time dependent perturbation theory D. E. Soper University of Oregon May 0 offer here some background for Chapter 5 of J. J. Sakurai, Modern Quantum Mechanics. The problem Let the hamiltonian for a system
Time dependent perturbation theory D. E. Soper University of Oregon May 0 offer here some background for Chapter 5 of J. J. Sakurai, Modern Quantum Mechanics. The problem Let the hamiltonian for a system
The 3 dimensional Schrödinger Equation
 Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
The semi-empirical mass formula, based on the liquid drop model, compared to the data
 Nucleonic Shells The semi-empirical mass formula, based on the liquid drop model, compared to the data E shell = E total E LD (Z=82, N=126) (Z=28, N=50) Nature 449, 411 (2007) Magic numbers at Z or N=
Nucleonic Shells The semi-empirical mass formula, based on the liquid drop model, compared to the data E shell = E total E LD (Z=82, N=126) (Z=28, N=50) Nature 449, 411 (2007) Magic numbers at Z or N=
Quantum Physics II (8.05) Fall 2002 Outline
 Quantum Physics II (8.05) Fall 2002 Outline 1. General structure of quantum mechanics. 8.04 was based primarily on wave mechanics. We review that foundation with the intent to build a more formal basis
Quantum Physics II (8.05) Fall 2002 Outline 1. General structure of quantum mechanics. 8.04 was based primarily on wave mechanics. We review that foundation with the intent to build a more formal basis
Basic p rinciples COPYRIGHTED MATERIAL. Introduction. Atomic s tructure
 1 Basic p rinciples Introduction 1 Atomic structure 1 Motion in the atom 2 MR active nuclei 2 The hydrogen nucleus 4 Alignment 4 Precession 8 The Larmor equation 9 Introduction The basic principles of
1 Basic p rinciples Introduction 1 Atomic structure 1 Motion in the atom 2 MR active nuclei 2 The hydrogen nucleus 4 Alignment 4 Precession 8 The Larmor equation 9 Introduction The basic principles of
University of Illinois at Chicago Department of Physics
 University of Illinois at Chicago Department of Physics Electromagnetism Qualifying Examination January 4, 2017 9.00 am - 12.00 pm Full credit can be achieved from completely correct answers to 4 questions.
University of Illinois at Chicago Department of Physics Electromagnetism Qualifying Examination January 4, 2017 9.00 am - 12.00 pm Full credit can be achieved from completely correct answers to 4 questions.
Non-stationary States and Electric Dipole Transitions
 Pre-Lab Lecture II Non-stationary States and Electric Dipole Transitions You will recall that the wavefunction for any system is calculated in general from the time-dependent Schrödinger equation ĤΨ(x,t)=i
Pre-Lab Lecture II Non-stationary States and Electric Dipole Transitions You will recall that the wavefunction for any system is calculated in general from the time-dependent Schrödinger equation ĤΨ(x,t)=i
Physics 221A Fall 1996 Notes 21 Hyperfine Structure in Hydrogen and Alkali Atoms
 Physics 221A Fall 1996 Notes 21 Hyperfine Structure in Hydrogen and Alkali Atoms Hyperfine effects in atomic physics are due to the interaction of the atomic electrons with the electric and magnetic multipole
Physics 221A Fall 1996 Notes 21 Hyperfine Structure in Hydrogen and Alkali Atoms Hyperfine effects in atomic physics are due to the interaction of the atomic electrons with the electric and magnetic multipole
Lecture 02 Nuclear Magnetic Resonance Spectroscopy Principle and Application in Structure Elucidation
 Application of Spectroscopic Methods in Molecular Structure Determination Prof. S. Sankararaman Department of Chemistry Indian Institution of Technology Madras Lecture 02 Nuclear Magnetic Resonance Spectroscopy
Application of Spectroscopic Methods in Molecular Structure Determination Prof. S. Sankararaman Department of Chemistry Indian Institution of Technology Madras Lecture 02 Nuclear Magnetic Resonance Spectroscopy
5.61 Physical Chemistry Lecture #35+ Page 1
 5.6 Physical Chemistry Lecture #35+ Page NUCLEAR MAGNETIC RESONANCE ust as IR spectroscopy is the simplest example of transitions being induced by light s oscillating electric field, so NMR is the simplest
5.6 Physical Chemistry Lecture #35+ Page NUCLEAR MAGNETIC RESONANCE ust as IR spectroscopy is the simplest example of transitions being induced by light s oscillating electric field, so NMR is the simplest
ψ s a ˆn a s b ˆn b ψ Hint: Because the state is spherically symmetric the answer can depend only on the angle between the two directions.
 1. Quantum Mechanics (Fall 2004) Two spin-half particles are in a state with total spin zero. Let ˆn a and ˆn b be unit vectors in two arbitrary directions. Calculate the expectation value of the product
1. Quantum Mechanics (Fall 2004) Two spin-half particles are in a state with total spin zero. Let ˆn a and ˆn b be unit vectors in two arbitrary directions. Calculate the expectation value of the product
Let us go back to what you knew in high school, or even earlier...
 Lecture I Quantum-Mechanical Way of Thinking To cultivate QM way of thinking, we will not start with the fascinating historical approach, but instead begin with one of the most important expt, that sends
Lecture I Quantum-Mechanical Way of Thinking To cultivate QM way of thinking, we will not start with the fascinating historical approach, but instead begin with one of the most important expt, that sends
Spectroscopy in frequency and time domains
 5.35 Module 1 Lecture Summary Fall 1 Spectroscopy in frequency and time domains Last time we introduced spectroscopy and spectroscopic measurement. I. Emphasized that both quantum and classical views of
5.35 Module 1 Lecture Summary Fall 1 Spectroscopy in frequency and time domains Last time we introduced spectroscopy and spectroscopic measurement. I. Emphasized that both quantum and classical views of
Probing P & T-violation Beyond the Standard Model. Aaron E. Leanhardt
 An Electron EDM Search in HfF + : Probing P & T-violation Beyond the Standard Model Aaron E. Leanhardt Experiment: Laura Sinclair, Russell Stutz & Eric Cornell Theory: Ed Meyer & John Bohn JILA, NIST,
An Electron EDM Search in HfF + : Probing P & T-violation Beyond the Standard Model Aaron E. Leanhardt Experiment: Laura Sinclair, Russell Stutz & Eric Cornell Theory: Ed Meyer & John Bohn JILA, NIST,
Atomic Physics (Phys 551) Final Exam Solutions
 Atomic Physics (Phys 551) Final Exam Solutions Problem 1. For a Rydberg atom in n = 50, l = 49 state estimate within an order of magnitude the numerical value of a) Decay lifetime A = 1 τ = 4αω3 3c D (1)
Atomic Physics (Phys 551) Final Exam Solutions Problem 1. For a Rydberg atom in n = 50, l = 49 state estimate within an order of magnitude the numerical value of a) Decay lifetime A = 1 τ = 4αω3 3c D (1)
Square-wave External Force in a Linear System
 Square-wave External Force in a Linear System Manual Eugene Butikov Annotation. The manual includes a description of the simulated physical system and a summary of the relevant theoretical material for
Square-wave External Force in a Linear System Manual Eugene Butikov Annotation. The manual includes a description of the simulated physical system and a summary of the relevant theoretical material for
Nuclear spin maser with a novel masing mechanism and its application to the search for an atomic EDM in 129 Xe
 Nuclear spin maser with a novel masing mechanism and its application to the search for an atomic EDM in 129 Xe A. Yoshimi RIKEN K. Asahi, S. Emori, M. Tsukui, RIKEN, Tokyo Institute of Technology Nuclear
Nuclear spin maser with a novel masing mechanism and its application to the search for an atomic EDM in 129 Xe A. Yoshimi RIKEN K. Asahi, S. Emori, M. Tsukui, RIKEN, Tokyo Institute of Technology Nuclear
Modern Physics for Scientists and Engineers International Edition, 4th Edition
 Modern Physics for Scientists and Engineers International Edition, 4th Edition http://optics.hanyang.ac.kr/~shsong Review: 1. THE BIRTH OF MODERN PHYSICS 2. SPECIAL THEORY OF RELATIVITY 3. THE EXPERIMENTAL
Modern Physics for Scientists and Engineers International Edition, 4th Edition http://optics.hanyang.ac.kr/~shsong Review: 1. THE BIRTH OF MODERN PHYSICS 2. SPECIAL THEORY OF RELATIVITY 3. THE EXPERIMENTAL
Applied Nuclear Physics (Fall 2006) Lecture 2 (9/11/06) Schrödinger Wave Equation
 22.101 Applied Nuclear Physics (Fall 2006) Lecture 2 (9/11/06) Schrödinger Wave Equation References -- R. M. Eisberg, Fundamentals of Modern Physics (Wiley & Sons, New York, 1961). R. L. Liboff, Introductory
22.101 Applied Nuclear Physics (Fall 2006) Lecture 2 (9/11/06) Schrödinger Wave Equation References -- R. M. Eisberg, Fundamentals of Modern Physics (Wiley & Sons, New York, 1961). R. L. Liboff, Introductory
Biomedical Imaging Magnetic Resonance Imaging
 Biomedical Imaging Magnetic Resonance Imaging Charles A. DiMarzio & Eric Kercher EECE 4649 Northeastern University May 2018 Background and History Measurement of Nuclear Spins Widely used in physics/chemistry
Biomedical Imaging Magnetic Resonance Imaging Charles A. DiMarzio & Eric Kercher EECE 4649 Northeastern University May 2018 Background and History Measurement of Nuclear Spins Widely used in physics/chemistry
Nuclear Spin and Stability. PHY 3101 D. Acosta
 Nuclear Spin and Stability PHY 3101 D. Acosta Nuclear Spin neutrons and protons have s = ½ (m s = ± ½) so they are fermions and obey the Pauli- Exclusion Principle The nuclear magneton is eh m µ e eh 1
Nuclear Spin and Stability PHY 3101 D. Acosta Nuclear Spin neutrons and protons have s = ½ (m s = ± ½) so they are fermions and obey the Pauli- Exclusion Principle The nuclear magneton is eh m µ e eh 1
Physics Important Terms and their Definitions
 Physics Important Terms and their S.No Word Meaning 1 Acceleration The rate of change of velocity of an object with respect to time 2 Angular Momentum A measure of the momentum of a body in rotational
Physics Important Terms and their S.No Word Meaning 1 Acceleration The rate of change of velocity of an object with respect to time 2 Angular Momentum A measure of the momentum of a body in rotational
Supplementary Information to Non-invasive detection of animal nerve impulses with an atomic magnetometer operating near quantum limited sensitivity.
 Supplementary Information to Non-invasive detection of animal nerve impulses with an atomic magnetometer operating near quantum limited sensitivity. Kasper Jensen, 1 Rima Budvytyte, 1 Rodrigo A. Thomas,
Supplementary Information to Non-invasive detection of animal nerve impulses with an atomic magnetometer operating near quantum limited sensitivity. Kasper Jensen, 1 Rima Budvytyte, 1 Rodrigo A. Thomas,
arxiv: v1 [quant-ph] 11 Nov 2014
![arxiv: v1 [quant-ph] 11 Nov 2014 arxiv: v1 [quant-ph] 11 Nov 2014](/thumbs/80/80451705.jpg) Electric dipoles on the Bloch sphere arxiv:1411.5381v1 [quant-ph] 11 Nov 014 Amar C. Vutha Dept. of Physics & Astronomy, York Univerity, Toronto ON M3J 1P3, Canada email: avutha@yorku.ca Abstract The time
Electric dipoles on the Bloch sphere arxiv:1411.5381v1 [quant-ph] 11 Nov 014 Amar C. Vutha Dept. of Physics & Astronomy, York Univerity, Toronto ON M3J 1P3, Canada email: avutha@yorku.ca Abstract The time
Physics 351, Spring 2017, Homework #12. Due at start of class, Friday, April 14, 2017
 Physics 351, Spring 2017, Homework #12. Due at start of class, Friday, April 14, 2017 Course info is at positron.hep.upenn.edu/p351 When you finish this homework, remember to visit the feedback page at
Physics 351, Spring 2017, Homework #12. Due at start of class, Friday, April 14, 2017 Course info is at positron.hep.upenn.edu/p351 When you finish this homework, remember to visit the feedback page at
Coupled Electrical Oscillators Physics Advanced Physics Lab - Summer 2018 Don Heiman, Northeastern University, 5/24/2018
 Coupled Electrical Oscillators Physics 3600 - Advanced Physics Lab - Summer 08 Don Heiman, Northeastern University, 5/4/08 I. INTRODUCTION The objectives of this experiment are: () explore the properties
Coupled Electrical Oscillators Physics 3600 - Advanced Physics Lab - Summer 08 Don Heiman, Northeastern University, 5/4/08 I. INTRODUCTION The objectives of this experiment are: () explore the properties
PHYS3113, 3d year Statistical Mechanics Tutorial problems. Tutorial 1, Microcanonical, Canonical and Grand Canonical Distributions
 1 PHYS3113, 3d year Statistical Mechanics Tutorial problems Tutorial 1, Microcanonical, Canonical and Grand Canonical Distributions Problem 1 The macrostate probability in an ensemble of N spins 1/2 is
1 PHYS3113, 3d year Statistical Mechanics Tutorial problems Tutorial 1, Microcanonical, Canonical and Grand Canonical Distributions Problem 1 The macrostate probability in an ensemble of N spins 1/2 is
Physics 106a, Caltech 4 December, Lecture 18: Examples on Rigid Body Dynamics. Rotating rectangle. Heavy symmetric top
 Physics 106a, Caltech 4 December, 2018 Lecture 18: Examples on Rigid Body Dynamics I go through a number of examples illustrating the methods of solving rigid body dynamics. In most cases, the problem
Physics 106a, Caltech 4 December, 2018 Lecture 18: Examples on Rigid Body Dynamics I go through a number of examples illustrating the methods of solving rigid body dynamics. In most cases, the problem
Radiation Damping. 1 Introduction to the Abraham-Lorentz equation
 Radiation Damping Lecture 18 1 Introduction to the Abraham-Lorentz equation Classically, a charged particle radiates energy if it is accelerated. We have previously obtained the Larmor expression for the
Radiation Damping Lecture 18 1 Introduction to the Abraham-Lorentz equation Classically, a charged particle radiates energy if it is accelerated. We have previously obtained the Larmor expression for the
Lecture 2: Open quantum systems
 Phys 769 Selected Topics in Condensed Matter Physics Summer 21 Lecture 2: Open quantum systems Lecturer: Anthony J. Leggett TA: Bill Coish 1. No (micro- or macro-) system is ever truly isolated U = S +
Phys 769 Selected Topics in Condensed Matter Physics Summer 21 Lecture 2: Open quantum systems Lecturer: Anthony J. Leggett TA: Bill Coish 1. No (micro- or macro-) system is ever truly isolated U = S +
Overhauser Magnetometers For Measurement of the Earth s Magnetic Field
 Overhauser Magnetometers For Measurement of the Earth s Magnetic Field By: Dr. Ivan Hrvoic GEM Systems Inc. (Magnetic field Workshop on Magnetic Observatory Instrumentation Espoo, Finland. 1989) TABLE
Overhauser Magnetometers For Measurement of the Earth s Magnetic Field By: Dr. Ivan Hrvoic GEM Systems Inc. (Magnetic field Workshop on Magnetic Observatory Instrumentation Espoo, Finland. 1989) TABLE
QUALIFYING EXAMINATION, Part 1. 2:00 PM 5:00 PM, Thursday September 3, 2009
 QUALIFYING EXAMINATION, Part 1 2:00 PM 5:00 PM, Thursday September 3, 2009 Attempt all parts of all four problems. Please begin your answer to each problem on a separate sheet, write your 3 digit code
QUALIFYING EXAMINATION, Part 1 2:00 PM 5:00 PM, Thursday September 3, 2009 Attempt all parts of all four problems. Please begin your answer to each problem on a separate sheet, write your 3 digit code
The Wave Function. Chapter The Harmonic Wave Function
 Chapter 3 The Wave Function On the basis of the assumption that the de Broglie relations give the frequency and wavelength of some kind of wave to be associated with a particle, plus the assumption that
Chapter 3 The Wave Function On the basis of the assumption that the de Broglie relations give the frequency and wavelength of some kind of wave to be associated with a particle, plus the assumption that
