Asymmetric oscillations during phase separation under continuous cooling - A simple model
|
|
|
- Elmer Watson
- 5 years ago
- Views:
Transcription
1 J. CHEM. PHYS. 29, 849 (28) symmetric oscillations during phase separation under continuous cooling - simple model Yumino Hayase a), Mika Kobayashi, Doris Vollmer, Harald Pleiner, and Günter K. uernhammer Max Planck Institute for Polymer Research, 5528 Mainz, Germany (Received 3 September 28; accepted 8 October 28; published online 28) We investigate the phase separation of binary mixtures under continuous cooling using the Cahn - Hilliard equation including the effect of gravity. In our simple model, sedimentation is accounted for by instantaneously removing droplets from the supersaturated mixture into the coexisting phase once the droplets have reached a defined maximum size. Our model predicts an oscillatory variation of turbidity. Depending on the composition either both phases oscillate (symmetric oscillations) or only one of the phases oscillates (asymmetric oscillations). In the asymmetric case, droplet sedimentation from the majority phase into the minority phase reduces supersaturation in the minority phase. This inhibits droplet formation in the minority phase. The cooling rate dependence of the period agrees with experimental results. c 28 merican Institute of Physics. [DOI:.63/.39867] I. INTRODUCTION The dynamics of phase separation strongly depends on the thermal protocol, for example whether phase separation is induced by a temperature quench from a one-phase to a twophase system,,2 by a double temperature quench, or by slow continuous ramping. In the latter case, qualitatively new features can arise, like oscillations of the turbidity and apparent heat capacity. 3 8 Oscillatory phase separation in volume-dominated systems has been observed in several systems as different as microemulsions, 3 polymer solutions 4 and even mixtures of olive oil and methanol. 5 It is a universal phenomenon that can be found, both in low molecular weight and polymer mixtures. Its qualitative behavior does not depend on whether the mixture phase separates under heating 3 or cooling. 4,5 Oscillatory phase separation proceeds as follows: 4,6 For simplicity, we adopt the notation of systems which are phase separating under cooling. binary mixture is prepared in the one-phase and cooled into the two-phase region (Fig.a). fter crossing the binodal curve (cloud point) supersaturation of the homogeneous mixture induces nucleation, which is followed by droplet growth and coarsening. When the droplets have reached a size of a few hundreds of nm, the mixture appears turbid due to the scattering of light in the generally not index matched mixtures. Upon further growth and coarsening of the droplets, gravity causes their sedimentation. fter sedimentation, the system is transparent again. However, the two coexisting phases are separated by an interface. Continuous cooling of the phase separated system, again leads to supersaturation in both coexisting phases. Depending on the supersaturation in each phase droplets nucleate in both phases or only in one. gain, the newly created droplets grow, coarsen and sediment. This cycle (Fig.b) may repeate itself several s (in some expea) Electronic mail: hayase@mpip-mainz.mpg.de riments up to 2 s) until the coexisting phases are sufficiently poor in their respective minority component such that the latter can be molecularly dissolved at all temperatures. Theoretical studies were started by J. Vollmer et al. 5,6,9. They pointed out that the oscillations arise due to an interplay of processes connected to well-separated scales, i.e. slow increase of supersaturation vs. fast relaxation (reduction of supersaturation). They predict that the oscillations arise from thermodynamics and are not of hydrodynamic origin. lthough sedimentation of droplets and surface tension cause convection in both phases, convection is assumed to homogenize the temperature, but does not determine the period of the oscillations. Vollmer et. al. observed an oscillatory phase separation and even a limit cycle for a well defined parameter range. 5,9 However, their model did not consider spatial degrees of freedom. Hydrodynamic effects are explicitly considered in a theoretical approach based on the Navier-Stokes equation, which is coupled to the nonlinear advection-diffusion equation., These studies were motivated by a weak container size dependence of the period of the oscillations in thin cells. 2 size dependence is characteristic for most hydrodynamic instabilities, for example for the Rayleigh-Bénard instability. In analogy to the latter instability, the oscillations were expected to be due to the density difference between the top and bottom of the container and the density close to the interface. Indeed, Lattice Boltzmann simulations showed an oscillatory instability. However, the numerical results differed by several orders of magnitude from the experimental parameter range., No previous theoretical studies discussed the difference of the dynamics between the upper and the lower phase, i.e. asymmetric oscillations. Experimentally, it was observed that often only the turbidity of the upper phase oscillates and the average turbidity of the lower phase remains almost constant (see in Fig.b,c). Our aim is to explain this asymmetry using a simple model. We use the Cahn - Hilliard (CH) equation 3,4 for phase sep-
2 Y. Hayase et al.: J. Chem. Phys. 29, 849 (28) symmetric oscillations in phase separation 2 4 one phase one phase.8 2 two phase.8 C meth gravity FIG. : Part of the phase diagram for mixtures of methanol - hexane. The points depict the experimentally determined phasetransition temperatures, as determined from turbidity measurements. 2 denotes the width of the biphasic region. Snapshots of the observed oscillatory phase separation under continuous cooling at a rate of K/h. Volume fraction of methanol C meth =.2. Images were taken after s (33.2 C), 85 s (33. C), 35 s (32.8 C), 5 s (32.8 C), 26 s (32.5 C), 385 s (32.2 C), from left to right, respectively. The white part indicates that droplets exist and dark part indicates that the system is transparent. The system suddenly becomes turbid, and after a while, it becomes transparent again due to droplet sedimentation. These processes are repeating. Space- plot of turbidity oscillations at K/h from 36 C to 26 C. It is obtained by assembling line data, which were integrated snapshots in horizontal direction. The total is 36 s. aration under continuous cooling, and include gravity-induced volume exchange as the coupling parameter between the upper and the lower phase. The CH equation is derived from a Ginzburg-Landau free energy functional. 3,4 It is a partial differential equation describing a system with a conserved order parameter. Phase-ordering dynamics after a temperature jump from the one-phase region to the two-phase region has been studied analytically and numerically using the CH equation. 5 7 In the case of continuous cooling, studies using the CH equation did not yield an oscillatory instability. 8 Various possibilities on how to include gravity in the CH equation are discussed in Refs Kitahara et al. 9 pointed out upper phase lower phase gravity upper phase lower phase Φ u Φ l "sedimentation" FIG. 2: The model system consists of two phases, in the upper and lower space, each of which is assumed to be homogeneous in the gravity direction. Each phase is described within a horizontal region (gray region). Scheme of the temperature - composition behavior. The thick lines indicate the composition of the upper/lower phase Φ u/l. t the black circle, droplets are created in the upper phase, which grow and sediment later by moving to the lower phase. The dashed arrow indicates the shift of composition due to droplets exchange. that the mobility should depend on the order parameter, when a gravity term is added to the free energy. More quantitative studies were able to predict droplet sedimentation during phase separation 22,23. ccording to our knowledge, the combination of gravity and continuous cooling has not yet been investigated using the CH model. In the sedimentation of droplets, of course, hydrodynamics affects coarsening. The phase separation dynamics including hydrodynamical effects has been studied by using model H in the Hohenberg-Halperin notation. 27 Since experimental findings point towards a thermodynamic origin of the oscillations, we only include strongly simplified hydrodynamic contributions. In this paper, we investigate the CH equation for continuous cooling including a model for gravity, focussing on the interplay between the up- Φ u
3 Y. Hayase et al.: J. Chem. Phys. 29, 849 (28) symmetric oscillations in phase separation 3 per and the lower phase. We find that the composition before the phase separation (total composition, Φ tot ) decides which phase oscillates. We also discuss the dependence of the oscillation frequency on the cooling rate. II. EXPERIMENTL. Sample Preparation Methanol and hexane (both analytic grade) were purchased from Sigma-ldrich and used as supplied. The samples were prepared by mixing the appropriate amounts of the respective components. The mixtures were loaded into teflon-sealed fluorescence cuvettes (width depth : 4cm 3 ). The mixture was continuously stirred and kept at a temperature well within the single-phase region before starting the experiments. B. Optical set-up The homogenized sample was placed in a transparent water bath (resembling a fish tank). The water temperature was controlled by a Haake thermostat to an accuracy of better than.k. Detailed temperature protocols can be followed automatically and the temperature setting is recorded together with the optical data. 4 The intensity of the scattered light was measured by video microscopy. The two-dimensional intensity distribution of individual snap shots (Fig.b) are averaged horizontally. The resulting vertical turbidity profiles of successive images are set together to the space- plots shown in Fig.c. Dark areas in the space- plot correspond to periods in which the sample was transparent and bright areas correspond to turbid periods (see Fig.c). III. MODEL. Phase separation The CH equation 3,4 describes the temporal evolution of the order parameter (r, t), which denotes the deviation of the composition from its average value, as a function of space, r and t. t = 2 ( ɛ [τ(t) 2 ]). () The total composition Φ tot is calculated from the order parameter (r, t) by Φ tot = V V d3 r(r, t). Here, V is the volume of the system. The CH equation is derived from the 4 free-energy functional. The parameter ɛ denotes the ratio between the interfacial width and the size of a nuclei of a mesoscopically phase separated domain. The phenomenological dimensionless parameter τ measures the temperature difference to the critical temperature T c. τ is negative if the mixture is one-phase, i.e. T > T c. For the CH equation one usually takes a positive value of τ to study phase separation processes after a temperature jump into the two phase region T < T c. For a continuously ramped temperature, τ is positive and an increasing function of. The average mass transfer between the coexisting phases depends on the width of the coexistence region 2 as shown in Fig.a. To keep the average mass transfer constant, we have adjusted the cooling rate to the slope of the phase diagram. This implies that the reduced cooling rate ξ has to be held constant: 4 ξ[τ(t)] = [τ(t)] [τ(t)]. (2) t Experimentally, it has been observed that the number of oscillations strongly increases, when keeping the mass transfer constant. 4 For the CH equation, the width of the coexistence region is equal to the square root of τ, = ± τ (3) Keeping ξ constant leads to an exponential increase of τ with. τ(t) = τ exp(2ξt). (4) τ(t) changes from τ = τ to τ = τ end. The experimental system is prepared in the one-phase region and rapidly cooled to a temperature just under the binodal line in oder to create an interface between the upper and lower phase. fter the system has equilibrated, it is slowly cooled into the two-phase region (Eq.(2)). In the simulation, the initial temperature τ is a value in the two-phase region close to the binodal line, Eq.(3). The initial temperature must be chosen below the binodal, i.e. τ < Φ 2 tot. For simplicity τ end = is used. The CH equation, modified by a term accounting for continuous cooling does not give rise to an oscillatory instability. 8 This can be understood from the following scaling argument: The scale of radius coarsening 28 is < R > t /3, while the mean square displacement of diffusion is proportional to t. Since the coarsening is slower than diffusion, the order parameter in the domains cannot reach supersaturation after the domains are formed. Gravity effectively increases the growth rate of interdroplet distances by sedimentation. B. Gravity The CH equation can be modified by adding a potential resembling the gravitational field 9 2. Using this modified CH equation, phase separation by a temperature jump has been investigated numerically. 22,23 Since these studies focused on sedimentation, they did not imply a zero flux boundary condition. The latter, however, is important in the present system, since is conserved and the system under investigation is finite and closed. Therefore one must enforce zero flux in the direction of gravity as a boundary condition. This considerably complicates the numerical solution of the CH equation, and therefore we do not take this approach. Instead we add a
4 Y. Hayase et al.: J. Chem. Phys. 29, 849 (28) symmetric oscillations in phase separation l V.3 V l.3 l V (d) [ ] (d) [ ].3.3 V l [ ] FIG. 4: symmetric oscillations: Evolution of the compositions Φ u/l. Time evolution of the total droplet area, which is a measure for the turbidity. Time evolution of the volume of the lower phase. The total composition Φ tot =.3, while the other parameters are identical to those in Fig FIG. 3: Symmetric oscillations: Evolution of the compositions Φ u/l. t the open circles, the upper/lower phase nucleates droplets. Time evolution of the total droplet area. The upper and lower parts of the figure correspond to the turbidity of the upper and lower phase, respectively. Time evolution of the volume of the lower phase. (d) Enlarged display of the first nucleation sequence shown in of the upper phase at t The total composition Φ tot =, the initial temperature τ =., the reduced cooling rate ξ = 2.3 5, the system size L = 32, the critical domain radius R c = 5/28.7, and the parameter in the CH equation ɛ =.. simple rule to the CH equation, Eq.(), mimicking sedimentation in a gravity field as explained now. We divide the sample into two systems representing the upper and lower phases. Both systems independently obey the CH equation modified to account for slow cooling. The coupling between the systems is accounted for by the following rule: when a droplet has reached its critical radius R c, it is taken out of one phase and added to the other. This resembles instantaneous sedimentation of droplets. This ansatz is motivated by the experimental fact that droplets are most efficient in collecting supersaturation when they are small and have a high number density and a large surface to volume ratio. s they grow, they merge, their number density strongly decreases, and droplets start to sediment. s a result of these processes, their efficiency to collect supersaturation strongly decreases. The critical droplet size can be estimated equating thermal and gravitational energy. The particles settle, if gravitational energy F G = 4/3πR 3 ρgh dominates over thermal energy, F B = k B T, where R denotes the radius of the droplet, g the earth s gravity constant, and ρ the density difference between the droplet and the surrounding matrix. To estimate this size, we take the difference h to be typically of the order of a particle diameter. Balancing both energies yields R c, the critical radius with Rc 4 = 3k B T/(4π ρg). For mixtures of methanol and hexane ( ρ.gcm 3 ) this gives R c = µm. For simplification, we assume homogeneity in the vertical direction, choose one horizontal region for each phase (Fig.2a) and solve the CH equation only on the two regions. To take into account the volume of both phases, we introduce the volume of the upper/lower phase, V u/l, and the composition of the upper/lower phase, Φ u/l. The value of Φ u/l is calculated from the order parameter u/l (r, t) in the upper/lower
5 Y. Hayase et al.: J. Chem. Phys. 29, 849 (28) symmetric oscillations in phase separation 5 phase by Φ u/l = V u/l V u/l d 3 r u/l (r, t). This setting avoids the difficulty of the zero flux boundary condition in the direction of gravity. When a droplet in the upper phase with volume V d and composition Φ d moves into the lower phase, the volume of the upper/lower phase and the composition change to the new values V u/v and Φ u/v. The system has to fulfill two conservation laws, i.e. volume conservation V l + V u = V l + V u = V ( ) and composition conservation Φ l V l + Φ u V u = Φ l V l + Φ uv u = Φ tot. Moving a droplet from the upper phase to the lower yields two additional equations V l = V l + V d (5) Φ l = Φ lv l + Φ d V d V l + V d. (6) When a droplet in the lower phase moves into the upper phase, the index l in the equations (5) and (6) are replaced by the index u. Fig.2b sketches a typical evolution of the compositions due to droplet exchange, i.e. due to instantaneous sedimentation. Under cooling, the compositions of the upper and the lower phases remain constant (the evolution is indicated by the vertical lines.). Supersaturation must be sufficiently high to make nucleation favorable. s we only allow for homogeneous nucleation, droplets nucleate only near the spinodal. They rapidly grow diffusively thereby reducing supersaturation. s soon as they have reached the critical size at the temperature marked by the black circle in Fig.2b, the composition of the upper phase moves toward the binodal line. Importantly, as the droplets are added to the lower phase also the composition of the lower phase moves towards the binodal line, i.e. supersaturation is reduced in both phases. Summing up, we have introduced gravity ignoring hydrodynamics, i.e. solvent flow. Our model should describe oscillatory phase separation well, if the built-up of supersaturation is slow compared to its relaxation. Our model fulfills all conservation laws and is very simple to implement numerically. The system has three length scales, the system size L, the critical domain radius R c and the domain width ɛ. In the numerical simulation, the critical radius R c is about s larger than ɛ, and the system size L is s larger than the critical radius R c, i.e. the three length scales are well separated. The oscillation properties described in the following are not sensitive to a change of the critical radius by at least a factor of 2. There are three relevant parameters, i.e. the total composition Φ tot, the initial temperature τ and the cooling rate ξ. Φ tot and τ define the initial condition. The simulations start after the formation of an interface between upper and lower phases. We assume that both phases have reached equilibrium at t =. This is equivalent to the experiments, in which the slow cooling starts after all processes due to the initial phase separation have relaxed, and an interface between the two phases has formed. The initial composition of the upper/lower phase, Φ u/l (t = ) is taken as its value on the binodal line at τ, and we take Φ l to be positive and Φ u negative. The volume of the upper/lower phase V u/l (t = ) is calculated considering volume and composition conservation as explained before. Each phase has an order parameter u/l (r, t), which describes the spatial variation of the composition in that phase. Initially, u/l (r, t = ) is uniformly distributed beside of random fluctuations around Φ u/l (t = ). We add conserved noise to the order parameter (r, t). The conserved noise 29 has the property < ζ(r, t) >= and ζ(r, t) = ζ (r, t).5[ζ (r dr, t) + ζ (r + dr, t)], with ζ (r, t)ζ (r + dr, t + dt) = ζ 2 δ(dr)δ(dt). The scenario described below is rather robust against changes in the noise strength. We also investigated the non-conserved noise, i.e. < ζ(r, t) >= and ζ(r, t)ζ(r + dr, t + dt) = ζ 2 δ(dr)δ(dt). However, the scenario described is robust against changes in the nature of the noise. When the noise strength increases, the period of the oscillation decreases. IV. RESULTS. Composition dependence C. Numerical conditions To test our ansatz we investigated the dependence of the resulting composition oscillations on the initial total composition and the cooling rate. The numerical simulations were carried out for ɛ =. and.2. We take the region as a -dimensional system. We chose three system sizes L = 6, L = 32, and L = 64, corresponding to 248, 496, and 892 points, respectively for a spatial mesh dr = / We chose a step dt =. and used periodic boundary conditions for each slice. We solved the CH equation using the Fast Fourier transformation method. 29 ll results presented have been checked to be qualitatively insensitive to changes in dr and dt. We took the critical domain radii R c = 5dr.7 and 25dr.95 corresponding to 5 and 25 points, respectively. Figure 3 shows the numerical results of our model at a total composition Φ tot =, an initial temperature τ =., and a reduced cooling rate ξ = t Φ tot =, the volume of the upper and the lower phases are equal, V l (t = ) = V u (t = ) =.5. ccording to Eq.(3), τ =. implies for the initial composition Φ u (t = ) = Φ l (t = ) = τ.32. Figure 3a depicts the evolution of Φ l/u. The right/left thick line indicates the temporal evolution of the compositions of the upper/lower phase. s Φ u (t = ) = Φ l (t = ), in the beginning the supersaturation in both phases increases identically. Nucleation of droplets is as probable in the upper as in the lower phase and therefore determined by noise. In this simulation, the upper phase happens to first nucleate droplets at about τ =.3. Figure 3b shows the evolution of the total area of the droplets, where N L i= r i, where N denotes the number of the droplets, and r i the radius of
6 Y. Hayase et al.: J. Chem. Phys. 29, 849 (28) symmetric oscillations in phase separation 6. τ.5 S S X S X S X X S X X S S X S X one phase region.2.25 Φ C asymmetric oscillation symmetric oscillation Φ C Φ tot Φ C asymmetric oscillation Φ tot FIG. 5: Phase-diagram in the Φ tot τ plane for systems for cooling rates ξ in the range < ξ < The area under the solid line corresponds to the one-phase region. indicates asymmetric oscillations for all ξ investigated. S indicates symmetric oscillations for all ξ. X indicates a region, where depending on ξ, either both phases or only the majority phase oscillates. Sketch of the phase separation mechanism as a function of the total composition Φ tot. For Φ c < Φ tot < Φ c, the turbidity of both phases oscillates. For Φ c < Φ tot, only the majority phase oscillates if the initial temperature τ is close to the binodal line. the i-th droplet. is a measure of the turbidity. round t = in Fig.3b, droplets are nucleated in the upper phase. When a droplet is taken from the upper phase and added to the lower phase, the volume of the lower phase V l increases (Fig.3c, around t = ). Thus the compositions of both phases move towards the binodal line (see the horizontal lines in Fig.3a). The nucleation looks like a delta function in Fig.3b, but when we zoom into the first sequence of nucleation events, it is rather a random sequence of events (Fig.3d). fter this nucleation wave, the upper phase still keeps significant supersaturation, i.e. does not reach the binodal. This is because the droplets have reached their critical size and are removed before they diminish supersaturation completely. fter the first nucleation wave, Φ u and Φ l are no longer equal. The lower phase, whose composition is now closer to the spinodal line, produces its next nucleation wave near t = (see Fig.3b). When droplets of the lower phase sediment into the upper phase, the volume of the lower phase V l decreases (Fig.3c close to t = ). t a cooling rate ξ = 2.3 5, three nucleation waves are observed for each phase, before the end temperature is reached. On these nucleation waves on average 8 droplets are exchanged for a FIG. 6: Space- plots for the composition dependence of turbidity oscillations of a binary mixture methanol/hexane. The volume fractions of methanol were C meth =.2, =.3, =.4. Before collecting data, the samples were prepared in the one phase region, cooled to the initial temperature (T c.5 K), and kept for one hour at this temperature. t the beginning, and are in the two-phase region, whereas still remains in the one-phase region. The samples were cooled at a rate of ξ exp =.75 h starting from (T c.5 K). Total is 4 s. system size of L = 32. In the symmetric case, Φ tot =., turbidity oscillations are observed in both phases. The volumes of the coexisting phases are changed during droplet exchange, but both volumes V l and V u always remain around.5 (see Fig.3c). In the experimentally investigated binary methanol-hexane mixture, Φ tot =. corresponds to an almost equal volume fraction of the lower and upper phase. This can be realized with a methanol concentration of C meth.3 (see Fig.a). The experimental results (Fig.6b) qualitatively match our numerical findings. Initially the volumes of both phases are similar, and the turbidity of both phases oscillates. For an asymmetric composition Φ tot =.3, the system exhibits a completely different scenario, Fig.4. s initial temperature we have chosen τ =. and for the cooling rate ξ = With these parameters, the values of Φ u (t = ) and Φ l (t = ) are the same as for the symmetric compo-
7 Y. Hayase et al.: J. Chem. Phys. 29, 849 (28) symmetric oscillations in phase separation 7 3 Δ t [ 4 ] 4.. ξ [ 5] 3 FIG. 7: The period t of turbidity oscillations in the upper phase is plotted as a function of the cooling rate ξ for a total composition Φ tot =.25 and an initial temperature τ =.7. The solid line corresponds to a system size L = 32, and the dashed line to L = 6. In the inset, the same data are shown in a log-log plot. Evolution of the composition in the upper phase u, for the same set of parameters as in, and L = 32. Solid line: ξ = 6.6 6, dashed line: ξ = sition, i.e. Φ u (t = ) = Φ l (t = ).32, but the initial volumes are different, V u (t = ) = V l (t = ).974 or V u /V l 4. The first droplets nucleate almost simultaneously in both phases (see Fig.4b near, t = 2.4). However, because of the volume difference, the number of droplets which move from the upper phase, the majority phase, into the lower phase, the minority phase, is significantly larger than those moving the other way round. The composition of the minority phase approaches the binodal after the first nucleation sequence as shown in Fig.4a. The composition of the upper phase always remains close to the spinodal. s a result, we observe turbidity oscillations only in the upper phase and not in the minority phase (see Fig.4b). The sedimentation of droplets increases the volume of the minority phase V l with each oscillation (Fig.4c). To understand the transitions from symmetric to asymmetric oscillations better, we have plotted a phase diagram in terms of the total composition Φ tot and the initial temperature τ, see Fig.5a. For each Φ tot and τ, we have investigated the cooling rates ξ between < ξ < In this range the turbidity of the system oscillates more than once and up to s after the initial nucleation sequence. The solid line in Fig.5a corresponds to the binodal line. Starting in the two-phase region τ > Φ 2 tot, we numerically determine the regions where asymmetric and symmetric oscillations show up. To the left of the dotted line, region S (symmetric oscillation) in Fig.5a, both phases nucleate droplets for all cooling rates ξ. To the right of the dotted line, the system shows asymmetric oscillations, i.e. the phase with the larger volume oscillates and the coexisting phase does not. In the region X, it depends on the actual value of ξ, whether the oscillations are symmetric or asymmetric. Below the critical composition, Φ tot < Φ c.2, both phases oscillate for all τ and ξ. symmetric oscillations are observed, when the volume difference between both phases is sufficiently large at the initial condition. In this case, the composition of the minority phase is changed by droplets expelled from the majority phase to such an extent that supersaturation remains below the threshold for nucleation. The volume ratio between the upper and the lower phase increases with increasing Φ tot. Furthermore, for an initial temperature τ close to the onephase region i.e, the binodal line in Fig.5, the volume ratio at the initial condition increases strongly due to volume and composition conservation laws. The large volume difference causes asymmetric oscillations. However, when we increase the initial temperature τ far into the two-phase region in Fig.5a, the volumes of both phases approach each other and finally, the minority phase has a chance to nucleate droplets, too. This holds even if Φ tot > Φ c. For large ξ, i.e. for very fast cooling, we observe only a small number of oscillations. The system resembles the asymmetric case, although both phases oscillate for small ξ (region indicated by X ). To compare our results with experimental data, the initial temperature τ should be assigned a value close to the one-phase region. With such an initial temperature, our model predicts that both phases oscillate in the region Φ tot < Φ c, while for Φ tot > Φ c, asymmetric oscillations are observed (see Fig.5b). To test our predictions experimentally, we have prepared different mixtures of hexane and methanol. For C meth =.2 (Fig.6a), the upper phase (hexane-rich phase) has initially the larger volume. In this case, the upper phase, i.e. the majority phase, shows turbidity oscillations and the lower does not. The volume of the lower phase increases due to droplet transfer from the upper phase. This scenario corresponds to the asymmetric oscillations in the model as shown in Fig. 4. s C meth increases, the initial volumes of the lower and the upper phase become equal. t C meth =.3, symmetric oscillations are observed, Fig.6b. Increasing C meth further the lower phase initially has the larger volume, shown in Fig.6c. In this case, the oscillations of the lower phase are more pronounced than those of the upper phase.
8 Y. Hayase et al.: J. Chem. Phys. 29, 849 (28) symmetric oscillations in phase separation 8 B. The period of the oscillation To check the cooling rate dependence, we used Φ tot =.25 and τ =.7, i.e. a composition where the upper phase always oscillates while the lower one does not (see Fig.5a). The cooling rate ξ was changed between < ξ < Even faster cooling rates do not cause any oscillation, because nucleation and coarsening are too slow compared to the the system needs to reach its end temperature τ end. The finite size of the system prevents to use of even lower cooling rates, since in the latter case, one can observe only one or two droplets at any oscillation. s the cooling rate was defined to keep the average mass transfer constant, the oscillation period of the upper phases t hardly varies with if ξ is constant. The average oscillation period t decreases for an increasing cooling rate, Fig.7a. log-log plot (inset of Fig.7a) indicates a power law relation between the cooling rate ξ and the oscillation period t, t ξ α, where α.6. The experimental results for the hexane-methanol binary mixture show a similar relation t ξ α with an exponent 4 α =.6 ±.5. V. DISCUSSION We investigated the CH equation with a special procedure to model gravity describing phase separation under continuous cooling. Other than gravity, further contributions might be added to the CH equation such as hydrodynamic flow and surface tension between both phases, etc. to achieve a realistic description of the experiments. The phase diagram used in our simulations is symmetric against, whereas in experimental systems, the phase diagram is slightly asymmetric. In the case of the methanol - hexane system, as shown in Fig.a, the left branch is steeper than the right one, and the position of the spinodal line is uncertain. nother important asymmetry between the upper and the lower phase is the airliquid interface at the top giving rise to Marangoni convection. Nevertheless, for the question, whether either asymmetric oscillation or symmetric oscillations occur, we consider the material exchange between the upper and the lower phase due to gravity to be the crucial effect. We note that, especially in the case of the hexane - methanol binary mixture, methanol wets the hexane-air interface bringing about a wetting layer of about 4nm thickness. 3 s the of the sample is a few cm, it exceeds the thickness of the wetting layer by more than 5 orders of magnitude. Therefore, we expect that the wetting layer does not influence the period of the oscillations. Indeed, we did not observe a change of the period when replacing the air by a solid interface. VI. CONCLUSION Motivated by experimental observations of turbidity and specific heat oscillations in binary mixtures under continuous cooling, we investigated the phase separation of binary mixtures using the CH equation with a simplified procedure to account for gravity. We numerically observed an oscillatory instability of droplet formation. Depending on the total composition, oscillations in either both or only one of the phases occur. In the latter case, the majority phase becomes the oscillating and the minority phase the silent phase, both, in experiment and in our simulations. In agreement with experimental findings and analytic studies, we found a power law relation between the oscillation period and the cooling rate. CKNOWLEDGEMENTS We are grateful to J. Vollmer, S. Puri, B. Dünweg, H. Tanaka, and. S. Mikhailov for useful discussions. This research was supported by the German Science Foundation, Vo639/-.. Onuki, Phase Transition Dynamics (Cambridge University Press, 22). 2 L.. Utracki, Polymer lloys and Blends. Thermodynamics and Rheology (Hanser, Munich, 99). 3 D. Vollmer, R. Strey, and J. Vollmer, J. Chem. Phys. 7, 369 (997). 4 G. K. uernhammer, D. Vollmer, and J. Vollmer, J. Chem. Phys. 23, 345 (25). 5 J. Vollmer, G. K. uernhammer, and D. Vollmer, Phys. Rev. Lett. 98, 3993 (27). 6 J. Vollmer and D. Vollmer, Faraday Discuss. 2, 5 (999). 7. Turchanin, R. Tsekov, and W. Freyland, J. Chem. Phys. 2, 7 (24). 8 R. S. Sparks, H. E. Huppert, T. Kozaguchi, and M.. Hallwood, Nature 36, 246 (993). 9 J. Vollmer, D. Vollmer, and R. Strey, J. Chem. Phys. 7, 3627 (997). M. E. Cates, J. Vollmer,. Wanger, and D. Vollmer, Phil. Trans. R. Soc. : Mathematical, Physical and Engineering Sciences, 36, 793 (23).. J. Wagner, M. E. Cates, and J. Vollmer, Proceedings of 3IC- CHMT, Paper Number Keynote 5. 2 D. Vollmer, J. Vollmer, and. Wagner, Phys. Chem. Chem. Phys (22). 3 J. W. Cahn and J. E. Hilliard, J. Chem. Phys. 28, 258 (958). 4 J. W. Cahn and J. E. Hilliard, J. Chem. Phys. 3, 688 (959). 5 Y. Oono and S. Puri, Phys. Rev. Lett. 58, 836 (987). 6 Y. Oono and S. Puri, Phys. Rev. 38, 434 (988). 7 S. Puri and Y. Oono, Phys. Rev. 38, 542 (988). 8 H.-O. Carmesin, D. W. Heermann, and K. Binder, Z. Phys. B 65, 89 (986). 9 K. Kitahara, Y. Oono, and D. Jasnow, Mod. Phys. Lett. B 2, 765 (988). 2 J. S. Langer, M. Baron, and H. D. Miller, Phys. Rev., 47 (975). 2 C. Yeung, T. Rogers,. Hernandes-Machado, and D. Jasnow, J.
9 Y. Hayase et al.: J. Chem. Phys. 29, 849 (28) symmetric oscillations in phase separation 9 Stat. Phys. 66, 7 (992). 22 S. Puri, N. Parekh, and S. Dattagupta, J. Stat. Phys. 75, 839 (994). 23 S. Puri, Physica 224, (996). 24 H. Tanaka, Phys. Rev. E 5, 33 (995). 25 H. Tanaka and T. raki, Phys. Rev. Lett. 8, 389 (998). 26 M. E. Cates, V. M. Kendon, P. Bladon, and J.-C. Desplat, Faraday Discuss., 2, (999). 27 P. C. Hohenberg and B. I. Halperin, Rev. Mod. Phys. 49, 435 (977). 28 I. M. Lifshitz and V. V. Slyozov, J. Phys. Chem. Solids 9, 35 (96). 29 W. H. Press, S.. Teukolsky, W. T. Vetterling, and B. P. Flannery, Numerical Recipes in Fortran (Cambridge University Press, London, 992). 3 D. Bonn and D. Ross, J. Rep. Prog. Phys. 64, 85 (2).
Long-time behavior and different shear regimes in quenched binary mixtures
 PHYSICAL REVIEW E 75, 011501 2007 Long-time behavior and different shear regimes in quenched binary mixtures G. Gonnella* Dipartimento di Fisica, Università di Bari, and INFN, Sezione di Bari, Via Amendola
PHYSICAL REVIEW E 75, 011501 2007 Long-time behavior and different shear regimes in quenched binary mixtures G. Gonnella* Dipartimento di Fisica, Università di Bari, and INFN, Sezione di Bari, Via Amendola
Complex Fluids. University of California - Santa Barbara, CA April 26, Abstract
 Gravitational Effects on Structure Development in Quenched Complex Fluids V. E. Badalassi, a H. D. Ceniceros b and S. Banerjee c a,c Department of Chemical Engineering, b Department of Mathematics University
Gravitational Effects on Structure Development in Quenched Complex Fluids V. E. Badalassi, a H. D. Ceniceros b and S. Banerjee c a,c Department of Chemical Engineering, b Department of Mathematics University
This is the accepted manuscript made available via CHORUS. The article has been published as:
 This is the accepted manuscript made available via CHORUS. The article has been published as: Similarity of the Signatures of the Initial Stages of Phase Separation in Metastable and Unstable Polymer Blends
This is the accepted manuscript made available via CHORUS. The article has been published as: Similarity of the Signatures of the Initial Stages of Phase Separation in Metastable and Unstable Polymer Blends
Hydrodynamic effects on phase separation of binary mixtures with reversible chemical reaction
 JOURNAL OF CHEMICAL PHYSICS VOLUME 118, NUMBER 21 1 JUNE 2003 Hydrodynamic effects on phase separation of binary mixtures with reversible chemical reaction Yanli Huo, Xiuli Jiang, Hongdong Zhang, and Yuliang
JOURNAL OF CHEMICAL PHYSICS VOLUME 118, NUMBER 21 1 JUNE 2003 Hydrodynamic effects on phase separation of binary mixtures with reversible chemical reaction Yanli Huo, Xiuli Jiang, Hongdong Zhang, and Yuliang
EFFECTS OF AN IMPOSED FLOW ON PHASE-SEPARATING BINARY MIXTURES
 Fractals, Vol. 11, Supplementary Issue (February 2003) 119 127 c World Scientific Publishing Company EFFECTS OF AN IMPOSED FLOW ON PHASE-SEPARATING BINARY MIXTURES F. CORBERI Istituto Nazionale per la
Fractals, Vol. 11, Supplementary Issue (February 2003) 119 127 c World Scientific Publishing Company EFFECTS OF AN IMPOSED FLOW ON PHASE-SEPARATING BINARY MIXTURES F. CORBERI Istituto Nazionale per la
Two-dimensional model of phase segregation in liquid binary mixtures
 PHYSICAL REVIEW E VOLUME 60, NUMBER 6 DECEMBER 1999 Two-dimensional model of phase segregation in liquid binary mixtures Natalia Vladimirova, 1 Andrea Malagoli, 2 and Roberto Mauri 1 1 Department of Chemical
PHYSICAL REVIEW E VOLUME 60, NUMBER 6 DECEMBER 1999 Two-dimensional model of phase segregation in liquid binary mixtures Natalia Vladimirova, 1 Andrea Malagoli, 2 and Roberto Mauri 1 1 Department of Chemical
Linear Theory of Evolution to an Unstable State
 Chapter 2 Linear Theory of Evolution to an Unstable State c 2012 by William Klein, Harvey Gould, and Jan Tobochnik 1 October 2012 2.1 Introduction The simple theory of nucleation that we introduced in
Chapter 2 Linear Theory of Evolution to an Unstable State c 2012 by William Klein, Harvey Gould, and Jan Tobochnik 1 October 2012 2.1 Introduction The simple theory of nucleation that we introduced in
A PRACTICALLY UNCONDITIONALLY GRADIENT STABLE SCHEME FOR THE N-COMPONENT CAHN HILLIARD SYSTEM
 A PRACTICALLY UNCONDITIONALLY GRADIENT STABLE SCHEME FOR THE N-COMPONENT CAHN HILLIARD SYSTEM Hyun Geun LEE 1, Jeong-Whan CHOI 1 and Junseok KIM 1 1) Department of Mathematics, Korea University, Seoul
A PRACTICALLY UNCONDITIONALLY GRADIENT STABLE SCHEME FOR THE N-COMPONENT CAHN HILLIARD SYSTEM Hyun Geun LEE 1, Jeong-Whan CHOI 1 and Junseok KIM 1 1) Department of Mathematics, Korea University, Seoul
Coarsening kinetics from a variable-mobility Cahn-Hilliard equation: Application of a semi-implicit Fourier spectral method
 PHYSICAL REVIEW E VOLUME 60, NUMBER 4 OCTOBER 1999 Coarsening kinetics from a variable-mobility Cahn-Hilliard equation: Application of a semi-implicit Fourier spectral method Jingzhi Zhu and Long-Qing
PHYSICAL REVIEW E VOLUME 60, NUMBER 4 OCTOBER 1999 Coarsening kinetics from a variable-mobility Cahn-Hilliard equation: Application of a semi-implicit Fourier spectral method Jingzhi Zhu and Long-Qing
Immigration, integration and ghetto formation
 Immigration, integration and ghetto formation arxiv:cond-mat/0209242v1 10 Sep 2002 Hildegard Meyer-Ortmanns School of Engineering and Science International University Bremen P.O.Box 750561 D-28725 Bremen,
Immigration, integration and ghetto formation arxiv:cond-mat/0209242v1 10 Sep 2002 Hildegard Meyer-Ortmanns School of Engineering and Science International University Bremen P.O.Box 750561 D-28725 Bremen,
Critical Phenomena under Shear Flow
 Critical Phenomena under Shear Flow Pavlik Lettinga, Hao Wang, Jan K.G. Dhont Close to a gas-liquid critical point, effective interactions between particles become very long ranged, and the dynamics of
Critical Phenomena under Shear Flow Pavlik Lettinga, Hao Wang, Jan K.G. Dhont Close to a gas-liquid critical point, effective interactions between particles become very long ranged, and the dynamics of
Title. Author(s)Nonomura, Makiko; Kobayashi, Ryo; Nishiura, Yasumasa. CitationJournal of the Physics Society Japan, 72(10): Issue Date
 Title Periodic Precipitation during Droplet Evaporation on AuthorsNonomura, Makiko; Kobayashi, Ryo; Nishiura, Yasumasa CitationJournal of the Physics Society Japan, 721: 268-2 Issue Date 23-1 Doc URL http://hdl.handle.net/2115/6
Title Periodic Precipitation during Droplet Evaporation on AuthorsNonomura, Makiko; Kobayashi, Ryo; Nishiura, Yasumasa CitationJournal of the Physics Society Japan, 721: 268-2 Issue Date 23-1 Doc URL http://hdl.handle.net/2115/6
Numerical Studies on Spinodal Decomposition of Liquid-Vapor Systems using Smoothed Particle Hydrodynamics. Martin Pütz
 Numerical Studies on Spinodal Decomposition of Liquid-Vapor Systems using Smoothed Particle Hydrodynamics Dissertation zur Erlangung des akademischen Grades eines Doktors der Naturwissenschaften vorgelegt
Numerical Studies on Spinodal Decomposition of Liquid-Vapor Systems using Smoothed Particle Hydrodynamics Dissertation zur Erlangung des akademischen Grades eines Doktors der Naturwissenschaften vorgelegt
Bubbles and Filaments: Stirring a Cahn Hilliard Fluid
 Bubbles and Filaments: Stirring a Cahn Hilliard Fluid Lennon Ó Náraigh and Jean-Luc Thiffeault Department of Mathematics Imperial College London APS-DFD Meeting, 21 November 2006 1 / 12 The classical Cahn
Bubbles and Filaments: Stirring a Cahn Hilliard Fluid Lennon Ó Náraigh and Jean-Luc Thiffeault Department of Mathematics Imperial College London APS-DFD Meeting, 21 November 2006 1 / 12 The classical Cahn
arxiv: v1 [cond-mat.stat-mech] 6 Mar 2008
![arxiv: v1 [cond-mat.stat-mech] 6 Mar 2008 arxiv: v1 [cond-mat.stat-mech] 6 Mar 2008](/thumbs/86/93817801.jpg) CD2dBS-v2 Convergence dynamics of 2-dimensional isotropic and anisotropic Bak-Sneppen models Burhan Bakar and Ugur Tirnakli Department of Physics, Faculty of Science, Ege University, 35100 Izmir, Turkey
CD2dBS-v2 Convergence dynamics of 2-dimensional isotropic and anisotropic Bak-Sneppen models Burhan Bakar and Ugur Tirnakli Department of Physics, Faculty of Science, Ege University, 35100 Izmir, Turkey
Breakdown of classical nucleation theory in nucleation kinetics
 Chapter 6 Breakdown of classical nucleation theory in nucleation kinetics In this chapter we present results of a study of nucleation of nematic droplets from the metastable isotropic phase. To the best
Chapter 6 Breakdown of classical nucleation theory in nucleation kinetics In this chapter we present results of a study of nucleation of nematic droplets from the metastable isotropic phase. To the best
A simple model for block-copolymer layer morphology formation
 A simple model for block-copolymer layer morphology formation A. J. Wagner and A. Croll, Department of Physics, North Dakota State University, Fargo, ND 58102 Prato Workshop, February 12, 2016 A big thanks
A simple model for block-copolymer layer morphology formation A. J. Wagner and A. Croll, Department of Physics, North Dakota State University, Fargo, ND 58102 Prato Workshop, February 12, 2016 A big thanks
Effects of Reversible Chemical Reaction on Morphology and Domain Growth of Phase Separating Binary Mixtures with Viscosity Difference
 280 Full Paper Summary: The effects of a reversible chemical reaction on morphology and dynamics of phase separating binary mixtures with viscosity difference are studied by numerically solving modified
280 Full Paper Summary: The effects of a reversible chemical reaction on morphology and dynamics of phase separating binary mixtures with viscosity difference are studied by numerically solving modified
Universality and scaling for the structure factor in dynamic order-disorder transitions
 PHYSICAL REVIEW E VOLUME 58, NUMBER 5 NOVEMBER 1998 Universality and scaling for the structure factor in dynamic order-disorder transitions Gregory Brown, 1,2 Per Arne Rikvold, 1,2,3 and Martin Grant 2
PHYSICAL REVIEW E VOLUME 58, NUMBER 5 NOVEMBER 1998 Universality and scaling for the structure factor in dynamic order-disorder transitions Gregory Brown, 1,2 Per Arne Rikvold, 1,2,3 and Martin Grant 2
Oscillantions in phase transitions
 Oscillantions in phase transitions Clémence Devailly, Caroline Crauste, Artyom Petrosyan, Sergio Ciliberto Laboratoire de Physique Ecole Normale Supérieure de Lyon and CNRS 46, Allée d'italie, 69364 Lyon
Oscillantions in phase transitions Clémence Devailly, Caroline Crauste, Artyom Petrosyan, Sergio Ciliberto Laboratoire de Physique Ecole Normale Supérieure de Lyon and CNRS 46, Allée d'italie, 69364 Lyon
Phenomenological Theories of Nucleation
 Chapter 1 Phenomenological Theories of Nucleation c 2012 by William Klein, Harvey Gould, and Jan Tobochnik 16 September 2012 1.1 Introduction These chapters discuss the problems of nucleation, spinodal
Chapter 1 Phenomenological Theories of Nucleation c 2012 by William Klein, Harvey Gould, and Jan Tobochnik 16 September 2012 1.1 Introduction These chapters discuss the problems of nucleation, spinodal
Finite Difference Solution of the Heat Equation
 Finite Difference Solution of the Heat Equation Adam Powell 22.091 March 13 15, 2002 In example 4.3 (p. 10) of his lecture notes for March 11, Rodolfo Rosales gives the constant-density heat equation as:
Finite Difference Solution of the Heat Equation Adam Powell 22.091 March 13 15, 2002 In example 4.3 (p. 10) of his lecture notes for March 11, Rodolfo Rosales gives the constant-density heat equation as:
Cellular Automata Model of Phase Transition in Binary Mixtures
 2892 Ind. Eng. Chem. Res. 2006, 45, 2892-2896 Cellular Automata Model of Phase Transition in Binary Mixtures Carolina Vannozzi, Diego Fiorentino, Matteo D Amore, David S. Rumshitzki, Andreas Dress, # and
2892 Ind. Eng. Chem. Res. 2006, 45, 2892-2896 Cellular Automata Model of Phase Transition in Binary Mixtures Carolina Vannozzi, Diego Fiorentino, Matteo D Amore, David S. Rumshitzki, Andreas Dress, # and
arxiv: v3 [cond-mat.stat-mech] 1 Dec 2016
![arxiv: v3 [cond-mat.stat-mech] 1 Dec 2016 arxiv: v3 [cond-mat.stat-mech] 1 Dec 2016](/thumbs/93/112509456.jpg) Effects of Landau-Lifshitz-Gilbert damping on domain growth Kazue Kudo Department of Computer Science, Ochanomizu University, 2-1-1 Ohtsuka, Bunkyo-ku, Tokyo 112-861, Japan (Dated: December 2, 216) arxiv:162.6673v3
Effects of Landau-Lifshitz-Gilbert damping on domain growth Kazue Kudo Department of Computer Science, Ochanomizu University, 2-1-1 Ohtsuka, Bunkyo-ku, Tokyo 112-861, Japan (Dated: December 2, 216) arxiv:162.6673v3
Spinodal decomposition
 Part V. Solid-solid transformations II Module 1 : Spinodal decomposition Part V. Solid-solid transformations II In this part, we discuss two transformations namely spinodal decomposition and ordering.
Part V. Solid-solid transformations II Module 1 : Spinodal decomposition Part V. Solid-solid transformations II In this part, we discuss two transformations namely spinodal decomposition and ordering.
Module 3: "Thin Film Hydrodynamics" Lecture 12: "" The Lecture Contains: Linear Stability Analysis. Some well known instabilities. Objectives_template
 The Lecture Contains: Linear Stability Analysis Some well known instabilities file:///e /courses/colloid_interface_science/lecture12/12_1.htm[6/16/2012 1:39:16 PM] Linear Stability Analysis This analysis
The Lecture Contains: Linear Stability Analysis Some well known instabilities file:///e /courses/colloid_interface_science/lecture12/12_1.htm[6/16/2012 1:39:16 PM] Linear Stability Analysis This analysis
Thermodynamic evolution of phase explosion during high-power nanosecond laser ablation
 Thermodynamic evolution of phase explosion during high-power nanosecond laser ablation Quanming Lu* School of Earth and Space Sciences, University of Science and Technology of China, Hefei, 230026, China
Thermodynamic evolution of phase explosion during high-power nanosecond laser ablation Quanming Lu* School of Earth and Space Sciences, University of Science and Technology of China, Hefei, 230026, China
Wetting Transitions at Fluid Interfaces and Related Topics
 Wetting Transitions at Fluid Interfaces and Related Topics Kenichiro Koga Department of Chemistry, Faculty of Science, Okayama University Tsushima-Naka 3-1-1, Okayama 7-853, Japan Received April 3, 21
Wetting Transitions at Fluid Interfaces and Related Topics Kenichiro Koga Department of Chemistry, Faculty of Science, Okayama University Tsushima-Naka 3-1-1, Okayama 7-853, Japan Received April 3, 21
Modeling, Simulation and Optimization of a Cross Flow Molten Carbonate Fuel Cell
 Modeling, Simulation and Optimization of a Cross Flow Molten Carbonate Fuel Cell P. Heidebrecht 1, M. Mangold 2, M. Gundermann 1, A. Kienle 2,3, K. Sundmacher 1,2 1 Otto-von-Guericke-University Magdeburg,
Modeling, Simulation and Optimization of a Cross Flow Molten Carbonate Fuel Cell P. Heidebrecht 1, M. Mangold 2, M. Gundermann 1, A. Kienle 2,3, K. Sundmacher 1,2 1 Otto-von-Guericke-University Magdeburg,
Double-diffusive lock-exchange gravity currents
 Abstract Double-diffusive lock-exchange gravity currents Nathan Konopliv, Presenting Author and Eckart Meiburg Department of Mechanical Engineering, University of California Santa Barbara meiburg@engineering.ucsb.edu
Abstract Double-diffusive lock-exchange gravity currents Nathan Konopliv, Presenting Author and Eckart Meiburg Department of Mechanical Engineering, University of California Santa Barbara meiburg@engineering.ucsb.edu
Network formation in viscoelastic phase separation
 INSTITUTE OF PHYSICSPUBLISHING JOURNAL OFPHYSICS: CONDENSED MATTER J. Phys.: Condens. Matter 15 (2003) S387 S393 PII: S0953-8984(03)54761-0 Network formation in viscoelastic phase separation Hajime Tanaka,
INSTITUTE OF PHYSICSPUBLISHING JOURNAL OFPHYSICS: CONDENSED MATTER J. Phys.: Condens. Matter 15 (2003) S387 S393 PII: S0953-8984(03)54761-0 Network formation in viscoelastic phase separation Hajime Tanaka,
with deterministic and noise terms for a general non-homogeneous Cahn-Hilliard equation Modeling and Asymptotics
 12-3-2009 Modeling and Asymptotics for a general non-homogeneous Cahn-Hilliard equation with deterministic and noise terms D.C. Antonopoulou (Joint with G. Karali and G. Kossioris) Department of Applied
12-3-2009 Modeling and Asymptotics for a general non-homogeneous Cahn-Hilliard equation with deterministic and noise terms D.C. Antonopoulou (Joint with G. Karali and G. Kossioris) Department of Applied
Modified Maxwell-Garnett equation for the effective transport coefficients in inhomogeneous media
 J. Phys. A: Math. Gen. 3 (998) 7227 7234. Printed in the UK PII: S0305-4470(98)93976-2 Modified Maxwell-Garnett equation for the effective transport coefficients in inhomogeneous media Juris Robert Kalnin
J. Phys. A: Math. Gen. 3 (998) 7227 7234. Printed in the UK PII: S0305-4470(98)93976-2 Modified Maxwell-Garnett equation for the effective transport coefficients in inhomogeneous media Juris Robert Kalnin
Thermodynamic expansion of nucleation free-energy barrier and size of critical nucleus near the vapor-liquid coexistence
 JOURNAL OF CHEMICAL PHYSICS VOLUME 110, NUMBER 7 15 FEBRUARY 1999 hermodynamic expansion of nucleation free-energy barrier and size of critical nucleus near the vapor-liquid coexistence Kenichiro Koga
JOURNAL OF CHEMICAL PHYSICS VOLUME 110, NUMBER 7 15 FEBRUARY 1999 hermodynamic expansion of nucleation free-energy barrier and size of critical nucleus near the vapor-liquid coexistence Kenichiro Koga
arxiv: v1 [cond-mat.stat-mech] 27 Apr 2017
![arxiv: v1 [cond-mat.stat-mech] 27 Apr 2017 arxiv: v1 [cond-mat.stat-mech] 27 Apr 2017](/thumbs/93/113959322.jpg) Effect of bond-disorder on the phase-separation kinetics of binary mixtures: a Monte Carlo simulation study arxiv:1704.08448v1 [cond-mat.stat-mech] 27 Apr 2017 Awaneesh Singh 1,2, Amrita Singh 2, and Anirban
Effect of bond-disorder on the phase-separation kinetics of binary mixtures: a Monte Carlo simulation study arxiv:1704.08448v1 [cond-mat.stat-mech] 27 Apr 2017 Awaneesh Singh 1,2, Amrita Singh 2, and Anirban
Chap. 2. Polymers Introduction. - Polymers: synthetic materials <--> natural materials
 Chap. 2. Polymers 2.1. Introduction - Polymers: synthetic materials natural materials no gas phase, not simple liquid (much more viscous), not perfectly crystalline, etc 2.3. Polymer Chain Conformation
Chap. 2. Polymers 2.1. Introduction - Polymers: synthetic materials natural materials no gas phase, not simple liquid (much more viscous), not perfectly crystalline, etc 2.3. Polymer Chain Conformation
Instantaneous gelation in Smoluchowski s coagulation equation revisited
 Instantaneous gelation in Smoluchowski s coagulation equation revisited Colm Connaughton Mathematics Institute and Centre for Complexity Science, University of Warwick, UK Collaborators: R. Ball (Warwick),
Instantaneous gelation in Smoluchowski s coagulation equation revisited Colm Connaughton Mathematics Institute and Centre for Complexity Science, University of Warwick, UK Collaborators: R. Ball (Warwick),
Polymer dynamics. Course M6 Lecture 5 26/1/2004 (JAE) 5.1 Introduction. Diffusion of polymers in melts and dilute solution.
 Course M6 Lecture 5 6//004 Polymer dynamics Diffusion of polymers in melts and dilute solution Dr James Elliott 5. Introduction So far, we have considered the static configurations and morphologies of
Course M6 Lecture 5 6//004 Polymer dynamics Diffusion of polymers in melts and dilute solution Dr James Elliott 5. Introduction So far, we have considered the static configurations and morphologies of
An electrokinetic LB based model for ion transport and macromolecular electrophoresis
 An electrokinetic LB based model for ion transport and macromolecular electrophoresis Raffael Pecoroni Supervisor: Michael Kuron July 8, 2016 1 Introduction & Motivation So far an mesoscopic coarse-grained
An electrokinetic LB based model for ion transport and macromolecular electrophoresis Raffael Pecoroni Supervisor: Michael Kuron July 8, 2016 1 Introduction & Motivation So far an mesoscopic coarse-grained
Phase-separation dynamics of a ternary mixture coupled with reversible chemical reaction
 JOURNAL OF CHEMICAL PHYSICS VOLUME 6, NUMBER 4 JANUARY 00 Phase-separation dynamics of a ternary mixture coupled with reversible chemical reaction Chaohui Tong and Yuliang Yang a) Department of Macromolecular
JOURNAL OF CHEMICAL PHYSICS VOLUME 6, NUMBER 4 JANUARY 00 Phase-separation dynamics of a ternary mixture coupled with reversible chemical reaction Chaohui Tong and Yuliang Yang a) Department of Macromolecular
2D Spinodal Decomposition in Forced Turbulence: Structure Formation in a Challenging Analogue of 2D MHD Turbulence
 2D Spinodal Decomposition in Forced Turbulence: Structure Formation in a Challenging Analogue of 2D MHD Turbulence 1 Xiang Fan 1, P H Diamond 1, Luis Chacon 2, Hui Li 2 1 University of California,San Diego
2D Spinodal Decomposition in Forced Turbulence: Structure Formation in a Challenging Analogue of 2D MHD Turbulence 1 Xiang Fan 1, P H Diamond 1, Luis Chacon 2, Hui Li 2 1 University of California,San Diego
APPLICATION OF KOHLER THEORY: MODELING CLOUD CONDENSATION NUCLEI ACTIVITY
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 APPLICATION OF KOHLER THEORY: MODELING CLOUD CONDENSATION NUCLEI ACTIVITY Gavin Cornwell, Katherine Nadler, Alex Nguyen, and Steven Schill Department of
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 APPLICATION OF KOHLER THEORY: MODELING CLOUD CONDENSATION NUCLEI ACTIVITY Gavin Cornwell, Katherine Nadler, Alex Nguyen, and Steven Schill Department of
Phase-Field Simulation of the Effect of Elastic Inhomogeneity on Microstructure Evolution in Ni-Based Superalloys
 Materials Transactions, Vol. 50, No. 4 (2009) pp. 744 to 748 #2009 The Japan Institute of Metals Phase-Field Simulation of the Effect of Elastic Inhomogeneity on Microstructure Evolution in Ni-Based Superalloys
Materials Transactions, Vol. 50, No. 4 (2009) pp. 744 to 748 #2009 The Japan Institute of Metals Phase-Field Simulation of the Effect of Elastic Inhomogeneity on Microstructure Evolution in Ni-Based Superalloys
CHAPTER 19. Fluid Instabilities. In this Chapter we discuss the following instabilities:
 CHAPTER 19 Fluid Instabilities In this Chapter we discuss the following instabilities: convective instability (Schwarzschild criterion) interface instabilities (Rayleight Taylor & Kelvin-Helmholtz) gravitational
CHAPTER 19 Fluid Instabilities In this Chapter we discuss the following instabilities: convective instability (Schwarzschild criterion) interface instabilities (Rayleight Taylor & Kelvin-Helmholtz) gravitational
Monte Carlo results for the Ising model with shear
 Monte Carlo results for the Ising model with shear E.N.M. Cirillo Dipartimento Me. Mo. Mat., via A. Scarpa 16-00161 Rome - Italy G. Gonnella and G. P. Saracco Dipartimento di Fisica, Università di Bari
Monte Carlo results for the Ising model with shear E.N.M. Cirillo Dipartimento Me. Mo. Mat., via A. Scarpa 16-00161 Rome - Italy G. Gonnella and G. P. Saracco Dipartimento di Fisica, Università di Bari
Simulation of mixing of heterogeneous HE components
 Chapter Simulation of mixing of heterogeneous HE components The majority on high explosives (HEs) used are blend ones. Properties of components differ that produces interaction on the grain scale (mesoprocesses).
Chapter Simulation of mixing of heterogeneous HE components The majority on high explosives (HEs) used are blend ones. Properties of components differ that produces interaction on the grain scale (mesoprocesses).
A Multi-Fluid Model of Membrane Formation by Phase-Inversion
 A Multi-Fluid Model of Membrane Formation by Phase-Inversion Douglas R. Tree 1 and Glenn Fredrickson 1,2 1 Materials Research Laboratory 2 Departments of Chemical Engineering and Materials University of
A Multi-Fluid Model of Membrane Formation by Phase-Inversion Douglas R. Tree 1 and Glenn Fredrickson 1,2 1 Materials Research Laboratory 2 Departments of Chemical Engineering and Materials University of
VIII. Phase Transformations. Lecture 38: Nucleation and Spinodal Decomposition
 VIII. Phase Transformations Lecture 38: Nucleation and Spinodal Decomposition MIT Student In this lecture we will study the onset of phase transformation for phases that differ only in their equilibrium
VIII. Phase Transformations Lecture 38: Nucleation and Spinodal Decomposition MIT Student In this lecture we will study the onset of phase transformation for phases that differ only in their equilibrium
Phase-field modeling of nanoscale island dynamics
 Title of Publication Edited by TMS (The Minerals, Metals & Materials Society), Year Phase-field modeling of nanoscale island dynamics Zhengzheng Hu, Shuwang Li, John Lowengrub, Steven Wise 2, Axel Voigt
Title of Publication Edited by TMS (The Minerals, Metals & Materials Society), Year Phase-field modeling of nanoscale island dynamics Zhengzheng Hu, Shuwang Li, John Lowengrub, Steven Wise 2, Axel Voigt
Coarsening: transient and self-similar dynamics in 1-D
 Coarsening: transient and self-similar dynamics in 1-D ½ 1 5 1 4 h ln(t) Ú ¼º N 1 3 N t 2/5 1 2 1 5 1 15 x ¼ 1 ¼ ¼º ½ 1 1 4 1 8 1 12 1 16 Ü t Michael Gratton (Northwestern) and Thomas Witelsi (Due) Cahn-Hilliard
Coarsening: transient and self-similar dynamics in 1-D ½ 1 5 1 4 h ln(t) Ú ¼º N 1 3 N t 2/5 1 2 1 5 1 15 x ¼ 1 ¼ ¼º ½ 1 1 4 1 8 1 12 1 16 Ü t Michael Gratton (Northwestern) and Thomas Witelsi (Due) Cahn-Hilliard
Dynamics of net-baryon density correlations near the QCD critical point
 Dynamics of net-baryon density correlations near the QCD critical point Marcus Bluhm and Marlene Nahrgang The work of M.B. is funded by the European Union s Horizon 22 research and innovation programme
Dynamics of net-baryon density correlations near the QCD critical point Marcus Bluhm and Marlene Nahrgang The work of M.B. is funded by the European Union s Horizon 22 research and innovation programme
Phase Transitions. µ a (P c (T ), T ) µ b (P c (T ), T ), (3) µ a (P, T c (P )) µ b (P, T c (P )). (4)
 Phase Transitions A homogeneous equilibrium state of matter is the most natural one, given the fact that the interparticle interactions are translationally invariant. Nevertheless there is no contradiction
Phase Transitions A homogeneous equilibrium state of matter is the most natural one, given the fact that the interparticle interactions are translationally invariant. Nevertheless there is no contradiction
Formation of valine microcrystals through rapid antisolvent precipitation
 Formation of valine microcrystals through rapid antisolvent precipitation Miroslav Variny a, Sandra Alvarez de Miguel b, Barry D. Moore c, Jan Sefcik b a Department of Chemical and Biochemical Engineering,
Formation of valine microcrystals through rapid antisolvent precipitation Miroslav Variny a, Sandra Alvarez de Miguel b, Barry D. Moore c, Jan Sefcik b a Department of Chemical and Biochemical Engineering,
Collective Effects. Equilibrium and Nonequilibrium Physics
 Collective Effects in Equilibrium and Nonequilibrium Physics: Lecture 3, 3 March 2006 Collective Effects in Equilibrium and Nonequilibrium Physics Website: http://cncs.bnu.edu.cn/mccross/course/ Caltech
Collective Effects in Equilibrium and Nonequilibrium Physics: Lecture 3, 3 March 2006 Collective Effects in Equilibrium and Nonequilibrium Physics Website: http://cncs.bnu.edu.cn/mccross/course/ Caltech
Stability Analysis of Stationary Solutions for the Cahn Hilliard Equation
 Stability Analysis of Stationary Solutions for the Cahn Hilliard Equation Peter Howard, Texas A&M University University of Louisville, Oct. 19, 2007 References d = 1: Commun. Math. Phys. 269 (2007) 765
Stability Analysis of Stationary Solutions for the Cahn Hilliard Equation Peter Howard, Texas A&M University University of Louisville, Oct. 19, 2007 References d = 1: Commun. Math. Phys. 269 (2007) 765
Supporting information for: A multi-fluid model for microstructure. formation in polymer membranes
 Electronic Supplementary Material ESI for Soft Matter. This journal is The Royal Society of Chemistry 2017 Supporting information for: A multi-fluid model for microstructure formation in polymer membranes
Electronic Supplementary Material ESI for Soft Matter. This journal is The Royal Society of Chemistry 2017 Supporting information for: A multi-fluid model for microstructure formation in polymer membranes
Thin Film Behavior after Ink Transfer in Printing Processes N. Bornemann, H. M. Sauer, E. Dörsam
 Thin Film Behavior after Ink Transfer in Printing Processes N. Bornemann, H. M. Sauer, E. Dörsam 15.04.2010 Institute of Printing Science and Technology Thin Film Behavior N. Bornemann Overview Thin Film
Thin Film Behavior after Ink Transfer in Printing Processes N. Bornemann, H. M. Sauer, E. Dörsam 15.04.2010 Institute of Printing Science and Technology Thin Film Behavior N. Bornemann Overview Thin Film
INFLUENCE OF THERMODIFFUSIVE PARTICLE TRANSPORT ON THERMOMAGNETIC CONVECTION IN MAGNETIC FLUIDS
 MAGNETOHYDRODYNAMICS Vol. 49 (2013), No. 3-4, pp. 473 478 INFLUENCE OF THERMODIFFUSIVE PARTICLE TRANSPORT ON THERMOMAGNETIC CONVECTION IN MAGNETIC FLUIDS TU Dresden, Chair of Magnetofluiddynamics, Measuring
MAGNETOHYDRODYNAMICS Vol. 49 (2013), No. 3-4, pp. 473 478 INFLUENCE OF THERMODIFFUSIVE PARTICLE TRANSPORT ON THERMOMAGNETIC CONVECTION IN MAGNETIC FLUIDS TU Dresden, Chair of Magnetofluiddynamics, Measuring
Gravitational Waves from Supernova Core Collapse: What could the Signal tell us?
 Outline Harald Dimmelmeier harrydee@mpa-garching.mpg.de Gravitational Waves from Supernova Core Collapse: What could the Signal tell us? Work done at the MPA in Garching Dimmelmeier, Font, Müller, Astron.
Outline Harald Dimmelmeier harrydee@mpa-garching.mpg.de Gravitational Waves from Supernova Core Collapse: What could the Signal tell us? Work done at the MPA in Garching Dimmelmeier, Font, Müller, Astron.
Miao Boya and An Yu Department of Physics, Tsinghua University, Beijing , People s Republic of China
 Localization in an acoustic cavitation cloud Miao Boya and An Yu Department of Physics, Tsinghua University, Beijing 100084, People s Republic of China Using a nonlinear sound wave equation for a bubbly
Localization in an acoustic cavitation cloud Miao Boya and An Yu Department of Physics, Tsinghua University, Beijing 100084, People s Republic of China Using a nonlinear sound wave equation for a bubbly
Entropic Crystal-Crystal Transitions of Brownian Squares K. Zhao, R. Bruinsma, and T.G. Mason
 Entropic Crystal-Crystal Transitions of Brownian Squares K. Zhao, R. Bruinsma, and T.G. Mason This supplementary material contains the following sections: image processing methods, measurements of Brownian
Entropic Crystal-Crystal Transitions of Brownian Squares K. Zhao, R. Bruinsma, and T.G. Mason This supplementary material contains the following sections: image processing methods, measurements of Brownian
Dynamics of transient metastable states in mixtures under coupled. phase ordering and chemical demixing
 Dynamics of transient metastable states in mixtures under coupled phase ordering and chemical demixing Ezequiel R. Soulé 1 and Alejandro D. Rey 2 1. Institute of Materials Science and Technology (INTEMA),
Dynamics of transient metastable states in mixtures under coupled phase ordering and chemical demixing Ezequiel R. Soulé 1 and Alejandro D. Rey 2 1. Institute of Materials Science and Technology (INTEMA),
Modified models of polymer phase separation
 Modified models of polymer phase separation Douglas Zhou, Pingwen Zhang,, * and Weinan E,2, LMAM, Peking University, Beijing 0087, People s Republic of China and School of Mathematical Science, Peking
Modified models of polymer phase separation Douglas Zhou, Pingwen Zhang,, * and Weinan E,2, LMAM, Peking University, Beijing 0087, People s Republic of China and School of Mathematical Science, Peking
Physics 598 ESM Term Paper Giant vortices in rapidly rotating Bose-Einstein condensates
 Physics 598 ESM Term Paper Giant vortices in rapidly rotating Bose-Einstein condensates Kuei Sun May 4, 2006 kueisun2@uiuc.edu Department of Physics, University of Illinois at Urbana- Champaign, 1110 W.
Physics 598 ESM Term Paper Giant vortices in rapidly rotating Bose-Einstein condensates Kuei Sun May 4, 2006 kueisun2@uiuc.edu Department of Physics, University of Illinois at Urbana- Champaign, 1110 W.
arxiv:cond-mat/ v2 [cond-mat.stat-mech] 8 Sep 1999
![arxiv:cond-mat/ v2 [cond-mat.stat-mech] 8 Sep 1999 arxiv:cond-mat/ v2 [cond-mat.stat-mech] 8 Sep 1999](/thumbs/95/123482736.jpg) BARI-TH 347/99 arxiv:cond-mat/9907149v2 [cond-mat.stat-mech] 8 Sep 1999 PHASE ORDERING IN CHAOTIC MAP LATTICES WITH CONSERVED DYNAMICS Leonardo Angelini, Mario Pellicoro, and Sebastiano Stramaglia Dipartimento
BARI-TH 347/99 arxiv:cond-mat/9907149v2 [cond-mat.stat-mech] 8 Sep 1999 PHASE ORDERING IN CHAOTIC MAP LATTICES WITH CONSERVED DYNAMICS Leonardo Angelini, Mario Pellicoro, and Sebastiano Stramaglia Dipartimento
ROBUST STABLE NONLINEAR CONTROL AND DESIGN OF A CSTR IN A LARGE OPERATING RANGE. Johannes Gerhard, Martin Mönnigmann, Wolfgang Marquardt
 ROBUST STABLE NONLINEAR CONTROL AND DESIGN OF A CSTR IN A LARGE OPERATING RANGE Johannes Gerhard, Martin Mönnigmann, Wolfgang Marquardt Lehrstuhl für Prozesstechnik, RWTH Aachen Turmstr. 46, D-5264 Aachen,
ROBUST STABLE NONLINEAR CONTROL AND DESIGN OF A CSTR IN A LARGE OPERATING RANGE Johannes Gerhard, Martin Mönnigmann, Wolfgang Marquardt Lehrstuhl für Prozesstechnik, RWTH Aachen Turmstr. 46, D-5264 Aachen,
Proposed approximation for contact angles in Shan-and-Chen-type multicomponent multiphase lattice Boltzmann models
 PHYSICAL REVIEW E 7, 0701 007 Proposed approximation for contact angles in Shan-and-Chen-type multicomponent multiphase lattice Boltzmann models Haibo Huang, 1 Daniel T. Thorne, Jr., Marcel G. Schaap,
PHYSICAL REVIEW E 7, 0701 007 Proposed approximation for contact angles in Shan-and-Chen-type multicomponent multiphase lattice Boltzmann models Haibo Huang, 1 Daniel T. Thorne, Jr., Marcel G. Schaap,
Supplemental Information - Glassy Dynamics in Composite Biopolymer Networks
 Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 2018 Supplemental Information - Glassy Dynamics in Composite Biopolymer Networks Tom Golde, 1 Constantin
Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 2018 Supplemental Information - Glassy Dynamics in Composite Biopolymer Networks Tom Golde, 1 Constantin
arxiv:cond-mat/ v1 [cond-mat.other] 4 Aug 2004
![arxiv:cond-mat/ v1 [cond-mat.other] 4 Aug 2004 arxiv:cond-mat/ v1 [cond-mat.other] 4 Aug 2004](/thumbs/74/69831888.jpg) Conservation laws for the voter model in complex networks arxiv:cond-mat/0408101v1 [cond-mat.other] 4 Aug 2004 Krzysztof Suchecki, 1,2 Víctor M. Eguíluz, 1 and Maxi San Miguel 1 1 Instituto Mediterráneo
Conservation laws for the voter model in complex networks arxiv:cond-mat/0408101v1 [cond-mat.other] 4 Aug 2004 Krzysztof Suchecki, 1,2 Víctor M. Eguíluz, 1 and Maxi San Miguel 1 1 Instituto Mediterráneo
arxiv:cond-mat/ v1 [cond-mat.stat-mech] 8 Oct 1996
![arxiv:cond-mat/ v1 [cond-mat.stat-mech] 8 Oct 1996 arxiv:cond-mat/ v1 [cond-mat.stat-mech] 8 Oct 1996](/thumbs/72/66812395.jpg) December 21, 2013 arxiv:cond-mat/9610066v1 [cond-mat.stat-mech] 8 Oct 1996 Some Finite Size Effects in Simulations of Glass Dynamics Jürgen Horbach, Walter Kob, Kurt Binder Institut für Physik, Johannes
December 21, 2013 arxiv:cond-mat/9610066v1 [cond-mat.stat-mech] 8 Oct 1996 Some Finite Size Effects in Simulations of Glass Dynamics Jürgen Horbach, Walter Kob, Kurt Binder Institut für Physik, Johannes
Interface Roughening in a Hydrodynamic Lattice- Gas Model with Surfactant
 Wesleyan University WesScholar Division III Faculty Publications Natural Sciences and Mathematics 1996 Interface Roughening in a Hydrodynamic Lattice- Gas Model with Surfactant Francis Starr Wesleyan University
Wesleyan University WesScholar Division III Faculty Publications Natural Sciences and Mathematics 1996 Interface Roughening in a Hydrodynamic Lattice- Gas Model with Surfactant Francis Starr Wesleyan University
Nuclear Science Research Group
 UR-NCUR 05-12 THE UNIVERSITY OF ROCHESTER Nuclear Science Research Group ROCHESTER, NEW YORK 14267-0216, USA Compound Nucleus with Surface Entropy; A Unified Phenomenology of Particle Evaporation, Fission,
UR-NCUR 05-12 THE UNIVERSITY OF ROCHESTER Nuclear Science Research Group ROCHESTER, NEW YORK 14267-0216, USA Compound Nucleus with Surface Entropy; A Unified Phenomenology of Particle Evaporation, Fission,
MOLECULAR DYNAMICS SIMULATION OF HETEROGENEOUS NUCLEATION OF LIQUID DROPLET ON SOLID SURFACE
 MOLECULAR DYNAMICS SIMULATION OF HETEROGENEOUS NUCLEATION OF LIQUID DROPLET ON SOLID SURFACE Tatsuto Kimura* and Shigeo Maruyama** *Department of Mechanical Engineering, The University of Tokyo 7-- Hongo,
MOLECULAR DYNAMICS SIMULATION OF HETEROGENEOUS NUCLEATION OF LIQUID DROPLET ON SOLID SURFACE Tatsuto Kimura* and Shigeo Maruyama** *Department of Mechanical Engineering, The University of Tokyo 7-- Hongo,
NUCLEATION, GROWTH AND COARSENING IN PHASE-SEPARATING SYSTEMS
 Scanning Microscopy Vol. 12, No. 1, 1998 (Pages 3-8) 0891-7035/98$5.00+.25 Scanning Microscopy International, Nucleation, Chicago growth (AMF O Hare), and coarsening IL 60666 in USA phase-separating systems
Scanning Microscopy Vol. 12, No. 1, 1998 (Pages 3-8) 0891-7035/98$5.00+.25 Scanning Microscopy International, Nucleation, Chicago growth (AMF O Hare), and coarsening IL 60666 in USA phase-separating systems
Macroscopic Simulation of Open Systems and Micro-Macro Link
 Macroscopic Simulation of Open Systems and Micro-Macro Link Ansgar Hennecke 1 and Martin Treiber 1 and Dirk Helbing 1 II Institute for Theoretical Physics, University Stuttgart, Pfaffenwaldring 57, D-7756
Macroscopic Simulation of Open Systems and Micro-Macro Link Ansgar Hennecke 1 and Martin Treiber 1 and Dirk Helbing 1 II Institute for Theoretical Physics, University Stuttgart, Pfaffenwaldring 57, D-7756
Hydrodynamics of Exploding Foil X-Ray Lasers with Time-Dependent Ionization Effect
 Hydrodynamics of Exploding Foil X-Ray Lasers with Time-Dependent Ionization Effect WANG Yu ( ), SU Dandan ( ), LI Yingjun ( ) State Key Laboratory for GeoMechanics and Deep Underground Engineering, China
Hydrodynamics of Exploding Foil X-Ray Lasers with Time-Dependent Ionization Effect WANG Yu ( ), SU Dandan ( ), LI Yingjun ( ) State Key Laboratory for GeoMechanics and Deep Underground Engineering, China
PORE-SCALE PHASE FIELD MODEL OF TWO-PHASE FLOW IN POROUS MEDIUM
 Excerpt from the Proceedings of the COMSOL Conference 2010 Paris PORE-SCALE PHASE FIELD MODEL OF TWO-PHASE FLOW IN POROUS MEDIUM Igor Bogdanov 1*, Sylvain Jardel 1, Anis Turki 1, Arjan Kamp 1 1 Open &
Excerpt from the Proceedings of the COMSOL Conference 2010 Paris PORE-SCALE PHASE FIELD MODEL OF TWO-PHASE FLOW IN POROUS MEDIUM Igor Bogdanov 1*, Sylvain Jardel 1, Anis Turki 1, Arjan Kamp 1 1 Open &
How Different is the Dynamics of a Lennard-Jones Binary Fluid from One-Component Lennard-Jones Fluid? 1
 How Different is the Dynamics of a Lennard-Jones Binary Fluid from One-Component Lennard-Jones Fluid? Takayuki NARUMI and Michio TOKUYAMA Summary We investigate the dynamics of liquids and supercooled
How Different is the Dynamics of a Lennard-Jones Binary Fluid from One-Component Lennard-Jones Fluid? Takayuki NARUMI and Michio TOKUYAMA Summary We investigate the dynamics of liquids and supercooled
Gravitational Waves from Supernova Core Collapse: Current state and future prospects
 Gravitational Waves from Core Collapse Harald Dimmelmeier harrydee@mpa-garching.mpg.de Gravitational Waves from Supernova Core Collapse: Current state and future prospects Work done with E. Müller (MPA)
Gravitational Waves from Core Collapse Harald Dimmelmeier harrydee@mpa-garching.mpg.de Gravitational Waves from Supernova Core Collapse: Current state and future prospects Work done with E. Müller (MPA)
v n,t n
 THE DYNAMICAL STRUCTURE FACTOR AND CRITICAL BEHAVIOR OF A TRAFFIC FLOW MODEL 61 L. ROTERS, S. L UBECK, and K. D. USADEL Theoretische Physik, Gerhard-Mercator-Universitat, 4748 Duisburg, Deutschland, E-mail:
THE DYNAMICAL STRUCTURE FACTOR AND CRITICAL BEHAVIOR OF A TRAFFIC FLOW MODEL 61 L. ROTERS, S. L UBECK, and K. D. USADEL Theoretische Physik, Gerhard-Mercator-Universitat, 4748 Duisburg, Deutschland, E-mail:
How DLS Works: Interference of Light
 Static light scattering vs. Dynamic light scattering Static light scattering measures time-average intensities (mean square fluctuations) molecular weight radius of gyration second virial coefficient Dynamic
Static light scattering vs. Dynamic light scattering Static light scattering measures time-average intensities (mean square fluctuations) molecular weight radius of gyration second virial coefficient Dynamic
Direct observation of aggregative nanoparticle growth: Kinetic modeling of the size distribution and growth rate
 Direct observation of aggregative nanoparticle growth: Kinetic modeling of the size distribution and growth rate Taylor J. Woehl, * Chiwoo Park, James E. Evans, Ilke Arslan, William D. Ristenpart, and
Direct observation of aggregative nanoparticle growth: Kinetic modeling of the size distribution and growth rate Taylor J. Woehl, * Chiwoo Park, James E. Evans, Ilke Arslan, William D. Ristenpart, and
Clouds associated with cold and warm fronts. Whiteman (2000)
 Clouds associated with cold and warm fronts Whiteman (2000) Dalton s law of partial pressures! The total pressure exerted by a mixture of gases equals the sum of the partial pressure of the gases! Partial
Clouds associated with cold and warm fronts Whiteman (2000) Dalton s law of partial pressures! The total pressure exerted by a mixture of gases equals the sum of the partial pressure of the gases! Partial
Chemical diffusion in a cuboidal cell
 Chemical diffusion in a cuboidal cell Chris Rycroft Department of Mathematics, MIT March 14, 28 1 Overview This article provides a short analysis of a chemical diffusion problem in a thin cuboidal cell
Chemical diffusion in a cuboidal cell Chris Rycroft Department of Mathematics, MIT March 14, 28 1 Overview This article provides a short analysis of a chemical diffusion problem in a thin cuboidal cell
Nucleation in a Fermi liquid at negative pressure
 Nucleation in a Fermi liquid at negative pressure Frédéric Caupin, Sébastien Balibar and Humphrey J. Maris Laboratoire de Physique Statistique de l Ecole Normale Supérieure associé aux Universités Paris
Nucleation in a Fermi liquid at negative pressure Frédéric Caupin, Sébastien Balibar and Humphrey J. Maris Laboratoire de Physique Statistique de l Ecole Normale Supérieure associé aux Universités Paris
Stabilization of Hyperbolic Chaos by the Pyragas Method
 Journal of Mathematics and System Science 4 (014) 755-76 D DAVID PUBLISHING Stabilization of Hyperbolic Chaos by the Pyragas Method Sergey Belyakin, Arsen Dzanoev, Sergey Kuznetsov Physics Faculty, Moscow
Journal of Mathematics and System Science 4 (014) 755-76 D DAVID PUBLISHING Stabilization of Hyperbolic Chaos by the Pyragas Method Sergey Belyakin, Arsen Dzanoev, Sergey Kuznetsov Physics Faculty, Moscow
Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers
 Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers Hong-ming Ding 1 & Yu-qiang Ma 1,2, 1 National Laboratory of Solid State Microstructures and Department
Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers Hong-ming Ding 1 & Yu-qiang Ma 1,2, 1 National Laboratory of Solid State Microstructures and Department
Self-organized Criticality and Synchronization in a Pulse-coupled Integrate-and-Fire Neuron Model Based on Small World Networks
 Commun. Theor. Phys. (Beijing, China) 43 (2005) pp. 466 470 c International Academic Publishers Vol. 43, No. 3, March 15, 2005 Self-organized Criticality and Synchronization in a Pulse-coupled Integrate-and-Fire
Commun. Theor. Phys. (Beijing, China) 43 (2005) pp. 466 470 c International Academic Publishers Vol. 43, No. 3, March 15, 2005 Self-organized Criticality and Synchronization in a Pulse-coupled Integrate-and-Fire
Supplementary information
 1 2 Supplementary information 3 4 5 6 Supplementary Figure 1 7 8 Supplementary Figure 1 ǀ Characterization of the lysozyme fibrils by atomic force microscopy 9 (AFM) and scanning electron microscopy (SEM).
1 2 Supplementary information 3 4 5 6 Supplementary Figure 1 7 8 Supplementary Figure 1 ǀ Characterization of the lysozyme fibrils by atomic force microscopy 9 (AFM) and scanning electron microscopy (SEM).
Synchronization of Limit Cycle Oscillators by Telegraph Noise. arxiv: v1 [cond-mat.stat-mech] 5 Aug 2014
![Synchronization of Limit Cycle Oscillators by Telegraph Noise. arxiv: v1 [cond-mat.stat-mech] 5 Aug 2014 Synchronization of Limit Cycle Oscillators by Telegraph Noise. arxiv: v1 [cond-mat.stat-mech] 5 Aug 2014](/thumbs/73/69324282.jpg) Synchronization of Limit Cycle Oscillators by Telegraph Noise Denis S. Goldobin arxiv:148.135v1 [cond-mat.stat-mech] 5 Aug 214 Department of Physics, University of Potsdam, Postfach 61553, D-14415 Potsdam,
Synchronization of Limit Cycle Oscillators by Telegraph Noise Denis S. Goldobin arxiv:148.135v1 [cond-mat.stat-mech] 5 Aug 214 Department of Physics, University of Potsdam, Postfach 61553, D-14415 Potsdam,
Abstract In mean-field theory, i.e. infinite-range interactions, the transition between metastable and unstable states of a thermodynamic system is sh
 Evidence for sharper than expected transition between metastable and unstable states Dieter W. Heermann and Claudette E. Cordeiro Λ Institut für Theoretische Physik Universität Heidelberg Philosophenweg
Evidence for sharper than expected transition between metastable and unstable states Dieter W. Heermann and Claudette E. Cordeiro Λ Institut für Theoretische Physik Universität Heidelberg Philosophenweg
SIMULATION OF DENDRITIC CRYSTAL GROWTH OF PURE Ni USING THE PHASE-FIELD MODEL
 46 Rev. Adv. Mater. Sci. 33 (13) 46-5 Yu. Zhao and H. Hou SIMULATION OF DENDRITIC CRYSTAL GROWTH OF PURE Ni USING THE PHASE-FIELD MODEL Yuhong Zhao and Hua Hou College of Materials Science & Engineering,
46 Rev. Adv. Mater. Sci. 33 (13) 46-5 Yu. Zhao and H. Hou SIMULATION OF DENDRITIC CRYSTAL GROWTH OF PURE Ni USING THE PHASE-FIELD MODEL Yuhong Zhao and Hua Hou College of Materials Science & Engineering,
Module 16. Diffusion in solids II. Lecture 16. Diffusion in solids II
 Module 16 Diffusion in solids II Lecture 16 Diffusion in solids II 1 NPTEL Phase II : IIT Kharagpur : Prof. R. N. Ghosh, Dept of Metallurgical and Materials Engineering Keywords: Micro mechanisms of diffusion,
Module 16 Diffusion in solids II Lecture 16 Diffusion in solids II 1 NPTEL Phase II : IIT Kharagpur : Prof. R. N. Ghosh, Dept of Metallurgical and Materials Engineering Keywords: Micro mechanisms of diffusion,
Growth oscillations. LElTER TO THE EDITOR. Zheming Cheng and Robert Savit
 J. Phys. A: Math. Gen. 19 (1986) L973-L978. Printed in Great Britain LElTER TO THE EDITOR Growth oscillations Zheming Cheng and Robert Savit Department of Physics, The University of Michigan, Ann Arbor,
J. Phys. A: Math. Gen. 19 (1986) L973-L978. Printed in Great Britain LElTER TO THE EDITOR Growth oscillations Zheming Cheng and Robert Savit Department of Physics, The University of Michigan, Ann Arbor,
Comparison of Heat and Mass Transport at the Micro-Scale
 Comparison of Heat and Mass Transport at the Micro-Scale E. Holzbecher, S. Oehlmann Georg-August Univ. Göttingen *Goldschmidtstr. 3, 37077 Göttingen, GERMANY, eholzbe@gwdg.de Abstract: Phenomena of heat
Comparison of Heat and Mass Transport at the Micro-Scale E. Holzbecher, S. Oehlmann Georg-August Univ. Göttingen *Goldschmidtstr. 3, 37077 Göttingen, GERMANY, eholzbe@gwdg.de Abstract: Phenomena of heat
2Heat transfer in critical
 2Heat transfer in critical fluids Chapter 2 This chapter starts with a brief overview of the properties, dynamic and static, of a fluid near its liquid-vapour critical point that are encountered in this
2Heat transfer in critical fluids Chapter 2 This chapter starts with a brief overview of the properties, dynamic and static, of a fluid near its liquid-vapour critical point that are encountered in this
Monte-Carlo simulations of spinodal ordering and decomposition in compositionally modulated alloys
 Monte-Carlo simulations of spinodal ordering and decomposition in compositionally modulated alloys Michael Atzmon Department of Nuclear Engineering, The University of Michigan, Ann Arbor, Michigan 48109
Monte-Carlo simulations of spinodal ordering and decomposition in compositionally modulated alloys Michael Atzmon Department of Nuclear Engineering, The University of Michigan, Ann Arbor, Michigan 48109
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa.
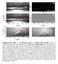 Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
MACROSCOPIC VARIABLES, THERMAL EQUILIBRIUM. Contents AND BOLTZMANN ENTROPY. 1 Macroscopic Variables 3. 2 Local quantities and Hydrodynamics fields 4
 MACROSCOPIC VARIABLES, THERMAL EQUILIBRIUM AND BOLTZMANN ENTROPY Contents 1 Macroscopic Variables 3 2 Local quantities and Hydrodynamics fields 4 3 Coarse-graining 6 4 Thermal equilibrium 9 5 Two systems
MACROSCOPIC VARIABLES, THERMAL EQUILIBRIUM AND BOLTZMANN ENTROPY Contents 1 Macroscopic Variables 3 2 Local quantities and Hydrodynamics fields 4 3 Coarse-graining 6 4 Thermal equilibrium 9 5 Two systems
PHASE BEHAVIOR OF WATER/FORMIC ACID/POLYAMIDE 6 IN PRESENCE OF TWO KINDS OF NANOCLAYS
 FPCM-9 (2008) The 9 th International Conference on Flow Processes in Composite Materials Montréal (Québec), Canada 8 ~ 10 July 2008 PHASE BEHAVIOR OF WATER/FORMIC ACID/POLYAMIDE 6 IN PRESENCE OF TWO KINDS
FPCM-9 (2008) The 9 th International Conference on Flow Processes in Composite Materials Montréal (Québec), Canada 8 ~ 10 July 2008 PHASE BEHAVIOR OF WATER/FORMIC ACID/POLYAMIDE 6 IN PRESENCE OF TWO KINDS
