6. X-ray Crystallography and Fourier Series
|
|
|
- Vanessa Wilkerson
- 5 years ago
- Views:
Transcription
1 6. X-ray Crystallography and Fourier Series Most of the information that we have on protein structure comes from x-ray crystallography. The basic steps in finding a protein structure using this method are: a high quality crystal is formed from a sample of protein the crystal is placed in an x-ray beam and the intensities of the diffraction spots are measured after finding the phases, an electron density map is computed from the diffraction intensities and phases using Fourier analysis. The coordinates of the atoms are found from the electron density. The structure is refined by checking that, for example, the atoms do not get too close to each other. We will briefly discuss some of the mathematics involved in finding the electron density from the diffraction intensities and phases. This requires studying Fourier series for functions periodic on lattices Lattices. The basic structure of a crystal is that of a lattice. A crystal is formed by many copies of the same protein in a pattern is formed which fills a unit cell. The unit cell is a parallelepiped which is used as a tile whose translations fill up space. For mathematical simplicity, we can suppose that there are an infinite number of copies of the same protein. A lattice is easy to describe mathematically. A lattice L in three dimensions is generated by three linearly independent vectors a, b, c; it is the set of all vectors h a+k b+lc where h, k and l are integers. A unit cell is the set of points xa+yb+zc for 0 x 1, 0 y 1, 0 z 1. Similarly we can define a lattice in two dimensions as the set of integer combinations of two linearly independent vectors a and b and the unit cell is the parallelogram formed by the points xa + yb for 0 x 1, 0 y 1. In the pdb file for a crystal structure, you can find the lengths and angles between the vectors a, b and c which generate the lattice for the crystal. The origin of the coordinate system can be put at any point crystallized protein. If the origin is placed at an atom in the protein, then every lattice point will be on exactly the same atom in a translation of the protein Examples of lattices. First consider two dimensions. The vectors a = (1, 0) and b = (0, 1) generate the square lattice. The vectors a = (1, 0) and b = (1/2, 3/2) generate the hexagonal lattice. 1
2 2 In three dimension the vectors a = (1, 0, 0), b = (0, 1, 0), and c = (0, 0, 1) generate the cubic lattice. The vectors a = (1, 1, 0), b = (1, 0, 1), c = (0, 1, 1) generate the face centered cubic lattice. The face centered cubic lattice can also be described as the set of points (x, y, z) with integer coordinates such that x + y + z is even. For an illustration of lattices see the Maple demo. The lattices described above are examples of lattices with symmetries. A symmetry of a lattice is a rotation such that the rotation and its inverse maps every point in the lattice onto another point of the lattice. We say the rotation leave the lattice unchanged. The square lattice, for example, is left unchanged by 90 degree rotation, an order 4 symmetry. The hexagonal lattice is left unchanged by 60 degree rotation, an order 6 symmetry. The cubic lattice is left fixed by 90 degree rotation about any axis. These are order 4 symmetries. The face centered cubic lattice is left fixed by 120 degree rotation about the axis in the direction (1, 1, 1). This is an order 3 symmetry The crystallographic restriction. A symmetry of the lattice for a crystal shows up in a symmetry of the diffraction pattern. It can be proved mathematically that the symmetry of a lattice has the crystallographic restriction: it has no rotational symmetries of order 5 or greater than 6. The crystallographic restriction can be stated as follows. A lattice in the plane or in three-dimensional space cannot have rotational symmetries of order 5 or of order greater than 6. Proof Here is a proof in dimension 2. The proof in dimension 3 is similar. Let L be a two dimensional lattice generated by vectors a and b, L = {h a + k b h and k are integers}. Suppose that the rotation ( ) cos θ sin θ (1) R = sin θ cos θ is a symmetry of L, that is, R moves L onto itself. Since R a and R b are in L, R a = h 1 a + k 1 b (2) R b = h 2 a + k 2 b for integers h 1, h 2, k 1, k 2. Using the frame (a, b) write (2) as R (a, b) = (a, b) M
3 3 or (3) (a, b) 1 R (a, b) = M where M = ( ) h1 h 2. k 1 k 2 The trace of a matrix A (tr A) is the sum of its diagonal entries. A basic fact in linear algebra is that tra = tr(b 1 AB) for all invertible matrices B. Applying this to (3), (4) tr R = 2 cos θ = tr M = h 1 + k 2. Now from (4) it follows that 2 cos θ is an integer, and so must be equal to 2, 1, 0, 1, or 2. Thus the only possibilities for θ are 0, ±60, ±90, ±120, and 180 and this proves the crystallographic restriction in dimension Diffraction. Molecules arranged in a lattice are arranged in many sets of parallel planes. You can see this by plotting a lattice, for example the face centered cubic lattice, in Maple and rotating it (Maple demo). An X-ray beam focused on a crystal creates a pattern of diffraction spots. The position of an X-ray diffraction spot depends on the distance d between the parallel planes creating it, the normal vector for the planes, and the wave length of the x-rays. The simplest and oldest rule of diffraction is Bragg s law derived by the English physicists Sir W.H. Bragg and his son Sir W.L. Bragg in 1913, (5) 2 d sin θ = n λ where θ is the angle of incidence of the x-ray, λ is the wave length of the x-ray, and n is some integer. The formula holds when refraction yields a bright spot at angle θ. The formula is explained by looking at figure 1. If d is the distance between the planes, the ray R 2 travels a distance of 2 d sin θ farther than R 1. If this is an integer multiple of the wavelength, the two rays are in phase after reflection and create a bright spot. Otherwise there is cancellation and no spot is seen. Here is a link to a Java applet illustrating Bragg s Law.
4 4 Figure 1. Illustration of Braggs Law. R 1 and R 2 are diffracted X-rays. The angle θ is the angle of incidence with planes containing the indicated atoms. Bragg s law can be derived by looking at the triangle ABC. The extra distance travelled by R 2 is 2BC Mathematical statement of Bragg s law. Bragg s law is an equation for the phase difference between two sine waves. We say the functions sin( 2π λ x) and sin( 2π λ x + p) have phase difference p/2π cycles. The wave length is λ. The amplitude at a point x in space of parallel sine waves with wavelength λ moving in the direction of the unit vector u 1 can be written as [ ] 2π (6) sin λ x u 1. This represents an x-ray beam moving in the direction u 1. The reflection off a point x 0 along a line through x 0 in the direction u 2, the wave has the equation [ ] 2π (7) sin λ (x u 2 + x 0 (u 1 u 2 )). This can be seen by checking that (6) and (7) agree at x = x 0. Similarly the reflection off the point x 1 along a line through x 1 in the same direction u 2, the wave has the equation [ ] 2π (8) sin λ (x u 2 + x 1 (u 1 u 2 )).
5 5 See figure 2. The difference in phase between (7) and (8) is Figure 2. x-rays of wavelength λ reflecting off two atoms at points x 0 and x 1. Incoming rays in direction u 1 are parallel. Outgoing rays in direction u 2 are possibly out of phase. 1 (9) λ (x 1 x 0 ) (u 1 u 2 ) and when the difference is a integer n, (10) (x 1 x 0 ) (u 1 u 2 ) = nλ, the outgoing waves are in phase at the point at infinity in the direction u 2. Suppose x 0 and x 1 are points in a lattice, so they differ by a lattice vector x 1 x 0 = v. Equation (10) shows that all such reflections are in phase if (11) v (u 1 u 2 ) = nλ for all lattice vectors v. For fixed n, (11) says that x 0 and x 1 lie in parallel planes perpendicular to u 1 u 2. Equation (11) is the mathematical expression of Bragg s law (5). It says, for example, that on the integer lattice we get reflections in the direction u 2 from the planes normal to u 1 u 2 only if (u 1 u 2 )/λ = (h, k, l) for integers h, k, and l. The reflection spot in the direction u 2 is labelled (h, k, l) Electron density. In crystallography, it is not the atoms of a molecule that are seen, but the electrons surrounding the atoms. To describe an electron cloud, a non-negative number giving the density of electrons is assigned to each point in the crystal. This number can be thought of as the amount of charge per unit volume. Although the information about protein structure is given in the form of a file containing the coordinates of each atom, in reality what the crystallographer sees is the electron density. The points representing the centers of the atoms are extrapolated from the electron density function using a computer.
6 6 Suppose that a structure is crystallized into a lattice consisting of all integer combinations of vectors a, b and c. Let r(x, y, z) be the electron density at the point x a + y b + zc, where x, y and z are real numbers, not necessarily integers. The function r is called the electron density function. If a level surface of the electron density function is plotted in one unit cell, it will look as in figure 3 for 6 Angstrom resolution and figure 4 for for 1A resolution. Figure 3. An electron density map of a molecule at 6 Angstrom resolution. The atoms are not clearly seen. The level surface shows more structural detail at higher (1 Angstrom) resolution. The electron density map is the level surface of a function computed from a Fourier series. Here is how Maple deals with level surfaces of functions. Since the crystal is unchanged if it translated by a, b or c, the electron density is the same at (x + 1)a + yb + zc as at Thus r has period 1 in the variable x, xa + yb + zc. r(x, y, z) = r(x + 1, y, z). A similar equation holds for the variables y and z. The electron density function is a periodic function, of period 1 in each of the three variables. Periodic functions are studied using Fourier series. We begin by looking at the theory for one variable. This will generalize to the three variable case which we are interested in.
7 7 Figure 4. Electron density function of a molecule at 1 Angstrom. The position of each atom is clearly seen Fourier series. The electron density function is constructed using Fourier Analysis. Fourier analysis is the approximation of periodic functions by sines and cosines. The basic idea of Fourier analysis is that any real valued function f(x) of period 1 can be approximated by sums of the type n (12) a 0 + [a j cos(2πjx) + b j sin(2πjx)] j=1 for suitable choice of the coefficients a j and b j and for n large enough. A sum of type (12) is called a Fourier series. The surprising part about Fourier s discovery is that even a function such as a square wave can be approximated by Fourier series. For example consider the period 1 square wave defined on the interval [0, 1] by the function { 0, if 1 (13) f(x) = 4 < x < 3 4 2, otherwise. This is a function of period 1, and 1 f(x) dx = 1. The Fourier cosine series 0 n (14) 1 + a j cos 2π(2j + 1)x with coefficients j=0 (15) a j = 4( 1)j π(2j + 1) approximates f closely for large n. Here is a Maple demo
8 Complex form of Fourier series. We will discuss how the coefficients of a Fourier series are computed. The simplest formula uses the complex form of the Fourier series. Complex numbers give a convenient way of writing Fourier series, even for real functions. The complex form of Fourier series is based on Euler s formula from which it follows that and e ix = cos x + i sin x. cos 2πjx = e2πijx + e 2πijx 2 sin 2πjx = e2πijx e 2πijx. 2i Substituting these expressions in the general form for the Fourier sum (12) get n (16) c j e 2πijx where j= n c j = 1 2 (a j ib j ), j = 1,..., n and c 0 = a 0 c j = c j, j = 1,..., n. So now we have a sum (16) of complex functions equal to the real sum (12). The sum (16) adds up to a real function since every term c j e 2πijx is added to its conjugate c j e 2πijx. If n (17) f(x) = c j e 2πijx j= n then the Fourier coefficients c j can be recovered from the function f by the formula (18) c j = 1 0 f(x)e 2πijx dx. Here is a demo in Maple of complex Fourier series. It is left as an exercise to show that formula (18) gives coefficients (15) for the square wave. The Fourier coefficients, like all complex numbers, have an absolute value and an argument, that is, we can write them in polar form as (19) c j = c j e iδj. The number δ j is called the phase and c j the norm.
9 Delta function. The function (13) is an example of an approximation to a periodic 1 Dirac delta function. The periodic delta function δ Z, or the delta function of the one dimensional integer lattice Z, is thought of as a function of period 1 such that (20) 1/2 1/2 δ Z (x)f(x) dx = f(0) for any smooth periodic function f(x). In particular, 1/2 1/2 δ Z (x) dx = 1. There is no function that has these properties, but the delta function can be thought of as the limit as M of functions such as { M (21) f(x) = 2, if n 1 M < x < n + 1 M, n an integer; 0, otherwise, of which (13) is the special case M = 4. Similarly by replacing 0 by a in (20), get the definition of the periodic delta function δ Z+a. A way to think about electron density function of a crystal and the phase problem is to use a delta function. Think of the periodic delta function δ Z+a in one dimension as describing the electron density of a crystal formed by a single atom of zero radius at each point a + n where n is an integer. By (18), the Fourier coefficients of r Z+a are given by (22) c j = e 2πija. So δ Z+a can be approximated by (23) δ Z+a (x) N k= N e 2πik(x a) for large N. Look at the Maple worksheet to see what this function looks like. In fact, simplifying (23) gives ) ] (x a) (24) δ Z+a (x) sin [ 2π ( N sin[π(x a)]. The phase of c j is 2πja and the norm is 1. If we did not know the phase but put 0 for the phase instead, then the coefficients c j would all be 1 and we would get an atom at x = 0 instead of x = a. This illustrates the importance of phase in computing the electron density function Three variable Fourier series. Fourier series can be used to analyze periodic functions in any number of variables, for example the electron density function ρ for a 3D crystal.
10 10 Given a real function ρ(x, y, z) of period 1 in each variable, it can be approximated by a triple sum of the form n n n c hkl e 2πi(hx+ky+lz) h= n written more compactly as k= n l= n (25) f(x, y, z) = hkl where c hkl e 2πi(hx+ky+lz) c h, k, l = c hkl since the function is real, and where h, j, and k are n where n is a large enough integer. Write the complex Fourier coefficient as The number α hkl is called the phase. c hkl = c hkl e iα hkl Similar to (18) the Fourier coefficients of (25) are (26) c hkl = f(x, y, z)e 2πi(hx+ky+lz) dx dy dz. In x-ray crystallography, the values c hkl are measured from the intensities of the spots in the diffraction pattern. The phases α hkl must be supplied by other means to find the electron density function. This is called the phase problem. There are methods for finding the phases but also some guess work is involved Delta function. Similar to the delta function for the one dimensional integer lattice, the delta function δ Z 3 for the three dimensional integer lattice, or cubic lattice, can be approximated by a sum of the form (25) with c hkl = 1 for all integer triples hkl. Also δ Z 3 +a is approximated by setting the Fourier coefficients c l = e 2πia l where l = (h, k, l) Diffraction pattern and Fourier series. Here we show how the diffraction intensities give the absolute values of the Fourier coefficients of the electron density function. Consider the electron density of a single atom of mass m modeled approximately as m times a delta function. For simplicity, assume the lattice of the crystal is the integer lattice, Z 3. In order to get a finite Fourier sum consider just a part of the lattice, L = {(x, y, z) x, y, z integers, N x N, N y N, N z N}
11 11 for N large. Now write the electron density function for atoms at points x 0 + l, l L as (27) ρ(x) = m l L e 2πix0 l e 2πix l. The Fourier coefficients of the electron density function are c l = me 2πix0 l for l L. Note that ρ(x) is an approximation of the delta function δ x0+z 3. Now look at the diffraction pattern. As before, consider the reflection of an x- ray beam in the direction u 1, reflected in the direction u 2 from all of these atoms. The equation for the beam reflected from the atom at x 0 is given as (7). The computation simplifies if we replace the sine function by the complex exponential. Also assume that the intensity of the reflected beam is proportional to the electron density m at the point. Then the outgoing beam has equation [ ] 2πi (28) m exp λ (x u 2 + x 0 (u 1 u 2 )) for the amplitude of the beam at a point x in space. The beam from the entire lattice of points is the sum of the intensities of each point in the lattice, (29) w(x) = [ ] 2πi m exp λ (x u 2 + (x 0 + l) (u 1 u 2 )) l L [ ] [ ] 2πi 2πi [ ] 2πi = m exp λ x u 2 exp λ x 0 (u 1 u 2 ) exp λ l (u 1 u 2 ). Writing l = (u 1 u 2 )/λ equation (29) becomes [ ] 2πi (30) w(x) m exp λ x u 2 exp [2πix 0 l ] δ Z 3(l ) since δ Z 3(l ) l L exp [2πil l ]. Integrating it with respect to l near a lattice point gives the value of 1. It follows that the total reflected wave for (u 1 u 2 )/λ near l is [ ] 2πi (31) m exp λ x u 2 exp [2πix 0 l ] where x is a point along the reflected beam. (Since all the beams are parallel in the direction u 2, we merge them all into one beam at infinity where they are collected.) The expression (31) is (c l ) exp [ 2πi λ x u 2] where cl is the l Fourier coefficient of the electron density function (27). Since only the amplitude of the beam can be measured from the intensity of the diffraction spot, the data gives only c l = m, the absolute value of the Fourier coefficient, and not the phase. l L The same result holds when we add electron densities of atoms to get the electron density of a molecule. Only the absolute value of the Fourier coefficient can be determined from the diffraction pattern.
Resolution: maximum limit of diffraction (asymmetric)
 Resolution: maximum limit of diffraction (asymmetric) crystal Y X-ray source 2θ X direct beam tan 2θ = Y X d = resolution 2d sinθ = λ detector 1 Unit Cell: two vectors in plane of image c* Observe: b*
Resolution: maximum limit of diffraction (asymmetric) crystal Y X-ray source 2θ X direct beam tan 2θ = Y X d = resolution 2d sinθ = λ detector 1 Unit Cell: two vectors in plane of image c* Observe: b*
GBS765 Electron microscopy
 GBS765 Electron microscopy Lecture 1 Waves and Fourier transforms 10/14/14 9:05 AM Some fundamental concepts: Periodicity! If there is some a, for a function f(x), such that f(x) = f(x + na) then function
GBS765 Electron microscopy Lecture 1 Waves and Fourier transforms 10/14/14 9:05 AM Some fundamental concepts: Periodicity! If there is some a, for a function f(x), such that f(x) = f(x + na) then function
3.012 Structure An Introduction to X-ray Diffraction
 3.012 Structure An Introduction to X-ray Diffraction This handout summarizes some topics that are important for understanding x-ray diffraction. The following references provide a thorough explanation
3.012 Structure An Introduction to X-ray Diffraction This handout summarizes some topics that are important for understanding x-ray diffraction. The following references provide a thorough explanation
CHEM-E5225 :Electron Microscopy. Diffraction 1
 CHEM-E5225 :Electron Microscopy Diffraction 1 2018-10-15 Yanling Ge Text book: Transmission electron microscopy by David B Williams & C. Barry Carter. 2009, Springer Outline Diffraction in TEM Thinking
CHEM-E5225 :Electron Microscopy Diffraction 1 2018-10-15 Yanling Ge Text book: Transmission electron microscopy by David B Williams & C. Barry Carter. 2009, Springer Outline Diffraction in TEM Thinking
Physical Chemistry I. Crystal Structure
 Physical Chemistry I Crystal Structure Crystal Structure Introduction Crystal Lattice Bravis Lattices Crytal Planes, Miller indices Distances between planes Diffraction patters Bragg s law X-ray radiation
Physical Chemistry I Crystal Structure Crystal Structure Introduction Crystal Lattice Bravis Lattices Crytal Planes, Miller indices Distances between planes Diffraction patters Bragg s law X-ray radiation
Fourier Syntheses, Analyses, and Transforms
 Fourier Syntheses, Analyses, and Transforms http://homepages.utoledo.edu/clind/ The electron density The electron density in a crystal can be described as a periodic function - same contents in each unit
Fourier Syntheses, Analyses, and Transforms http://homepages.utoledo.edu/clind/ The electron density The electron density in a crystal can be described as a periodic function - same contents in each unit
Crystals, X-rays and Proteins
 Crystals, X-rays and Proteins Comprehensive Protein Crystallography Dennis Sherwood MA (Hons), MPhil, PhD Jon Cooper BA (Hons), PhD OXFORD UNIVERSITY PRESS Contents List of symbols xiv PART I FUNDAMENTALS
Crystals, X-rays and Proteins Comprehensive Protein Crystallography Dennis Sherwood MA (Hons), MPhil, PhD Jon Cooper BA (Hons), PhD OXFORD UNIVERSITY PRESS Contents List of symbols xiv PART I FUNDAMENTALS
What is a Quasicrystal?
 July 23, 2013 Rotational symmetry An object with rotational symmetry is an object that looks the same after a certain amount of rotation. Rotational symmetry An object with rotational symmetry is an object
July 23, 2013 Rotational symmetry An object with rotational symmetry is an object that looks the same after a certain amount of rotation. Rotational symmetry An object with rotational symmetry is an object
Solid State Physics 460- Lecture 5 Diffraction and the Reciprocal Lattice Continued (Kittel Ch. 2)
 Solid State Physics 460- Lecture 5 Diffraction and the Reciprocal Lattice Continued (Kittel Ch. 2) Ewald Construction 2θ k out k in G Physics 460 F 2006 Lect 5 1 Recall from previous lectures Definition
Solid State Physics 460- Lecture 5 Diffraction and the Reciprocal Lattice Continued (Kittel Ch. 2) Ewald Construction 2θ k out k in G Physics 460 F 2006 Lect 5 1 Recall from previous lectures Definition
Protein Structure Determination. Part 1 -- X-ray Crystallography
 Protein Structure Determination Part 1 -- X-ray Crystallography Topics covering in this 1/2 course Crystal growth Diffraction theory Symmetry Solving phases using heavy atoms Solving phases using a model
Protein Structure Determination Part 1 -- X-ray Crystallography Topics covering in this 1/2 course Crystal growth Diffraction theory Symmetry Solving phases using heavy atoms Solving phases using a model
CS273: Algorithms for Structure Handout # 13 and Motion in Biology Stanford University Tuesday, 11 May 2003
 CS273: Algorithms for Structure Handout # 13 and Motion in Biology Stanford University Tuesday, 11 May 2003 Lecture #13: 11 May 2004 Topics: Protein Structure Determination Scribe: Minli Zhu We acknowledge
CS273: Algorithms for Structure Handout # 13 and Motion in Biology Stanford University Tuesday, 11 May 2003 Lecture #13: 11 May 2004 Topics: Protein Structure Determination Scribe: Minli Zhu We acknowledge
Handout 7 Reciprocal Space
 Handout 7 Reciprocal Space Useful concepts for the analysis of diffraction data http://homepages.utoledo.edu/clind/ Concepts versus reality Reflection from lattice planes is just a concept that helps us
Handout 7 Reciprocal Space Useful concepts for the analysis of diffraction data http://homepages.utoledo.edu/clind/ Concepts versus reality Reflection from lattice planes is just a concept that helps us
PSD '18 -- Xray lecture 4. Laue conditions Fourier Transform The reciprocal lattice data collection
 PSD '18 -- Xray lecture 4 Laue conditions Fourier Transform The reciprocal lattice data collection 1 Fourier Transform The Fourier Transform is a conversion of one space into another space with reciprocal
PSD '18 -- Xray lecture 4 Laue conditions Fourier Transform The reciprocal lattice data collection 1 Fourier Transform The Fourier Transform is a conversion of one space into another space with reciprocal
Basic Crystallography Part 1. Theory and Practice of X-ray Crystal Structure Determination
 Basic Crystallography Part 1 Theory and Practice of X-ray Crystal Structure Determination We have a crystal How do we get there? we want a structure! The Unit Cell Concept Ralph Krätzner Unit Cell Description
Basic Crystallography Part 1 Theory and Practice of X-ray Crystal Structure Determination We have a crystal How do we get there? we want a structure! The Unit Cell Concept Ralph Krätzner Unit Cell Description
Geometry of Crystal Lattice
 0 Geometry of Crystal Lattice 0.1 Translational Symmetry The crystalline state of substances is different from other states (gaseous, liquid, amorphous) in that the atoms are in an ordered and symmetrical
0 Geometry of Crystal Lattice 0.1 Translational Symmetry The crystalline state of substances is different from other states (gaseous, liquid, amorphous) in that the atoms are in an ordered and symmetrical
9. Nuclear Magnetic Resonance
 9. Nuclear Magnetic Resonance Nuclear Magnetic Resonance (NMR) is a method that can be used to find structures of proteins. NMR spectroscopy is the observation of spins of atoms and electrons in a molecule
9. Nuclear Magnetic Resonance Nuclear Magnetic Resonance (NMR) is a method that can be used to find structures of proteins. NMR spectroscopy is the observation of spins of atoms and electrons in a molecule
Roger Johnson Structure and Dynamics: X-ray Diffraction Lecture 6
 6.1. Summary In this Lecture we cover the theory of x-ray diffraction, which gives direct information about the atomic structure of crystals. In these experiments, the wavelength of the incident beam must
6.1. Summary In this Lecture we cover the theory of x-ray diffraction, which gives direct information about the atomic structure of crystals. In these experiments, the wavelength of the incident beam must
Understanding Single-Crystal X-Ray Crystallography Exercises and Solutions
 Understanding Single-Crystal X-Ray Crystallography Exercises and Solutions Dennis W. Bennett Department of Chemistry and Biochemistry University of Wisconsin-Milwaukee Chapter Crystal Lattices. The copper
Understanding Single-Crystal X-Ray Crystallography Exercises and Solutions Dennis W. Bennett Department of Chemistry and Biochemistry University of Wisconsin-Milwaukee Chapter Crystal Lattices. The copper
Protein Crystallography
 Protein Crystallography Part II Tim Grüne Dept. of Structural Chemistry Prof. G. Sheldrick University of Göttingen http://shelx.uni-ac.gwdg.de tg@shelx.uni-ac.gwdg.de Overview The Reciprocal Lattice The
Protein Crystallography Part II Tim Grüne Dept. of Structural Chemistry Prof. G. Sheldrick University of Göttingen http://shelx.uni-ac.gwdg.de tg@shelx.uni-ac.gwdg.de Overview The Reciprocal Lattice The
Crystal Structure SOLID STATE PHYSICS. Lecture 5. A.H. Harker. thelecture thenextlecture. Physics and Astronomy UCL
 Crystal Structure thelecture thenextlecture SOLID STATE PHYSICS Lecture 5 A.H. Harker Physics and Astronomy UCL Structure & Diffraction Crystal Diffraction (continued) 2.4 Experimental Methods Notes: examples
Crystal Structure thelecture thenextlecture SOLID STATE PHYSICS Lecture 5 A.H. Harker Physics and Astronomy UCL Structure & Diffraction Crystal Diffraction (continued) 2.4 Experimental Methods Notes: examples
Scattering by two Electrons
 Scattering by two Electrons p = -r k in k in p r e 2 q k in /λ θ θ k out /λ S q = r k out p + q = r (k out - k in ) e 1 Phase difference of wave 2 with respect to wave 1: 2π λ (k out - k in ) r= 2π S r
Scattering by two Electrons p = -r k in k in p r e 2 q k in /λ θ θ k out /λ S q = r k out p + q = r (k out - k in ) e 1 Phase difference of wave 2 with respect to wave 1: 2π λ (k out - k in ) r= 2π S r
General theory of diffraction
 General theory of diffraction X-rays scatter off the charge density (r), neutrons scatter off the spin density. Coherent scattering (diffraction) creates the Fourier transform of (r) from real to reciprocal
General theory of diffraction X-rays scatter off the charge density (r), neutrons scatter off the spin density. Coherent scattering (diffraction) creates the Fourier transform of (r) from real to reciprocal
Introduction to crystallography The unitcell The resiprocal space and unitcell Braggs law Structure factor F hkl and atomic scattering factor f zθ
 Introduction to crystallography The unitcell The resiprocal space and unitcell Braggs law Structure factor F hkl and atomic scattering factor f zθ Introduction to crystallography We divide materials into
Introduction to crystallography The unitcell The resiprocal space and unitcell Braggs law Structure factor F hkl and atomic scattering factor f zθ Introduction to crystallography We divide materials into
Keble College - Hilary 2012 Section VI: Condensed matter physics Tutorial 2 - Lattices and scattering
 Tomi Johnson Keble College - Hilary 2012 Section VI: Condensed matter physics Tutorial 2 - Lattices and scattering Please leave your work in the Clarendon laboratory s J pigeon hole by 5pm on Monday of
Tomi Johnson Keble College - Hilary 2012 Section VI: Condensed matter physics Tutorial 2 - Lattices and scattering Please leave your work in the Clarendon laboratory s J pigeon hole by 5pm on Monday of
Solid State Physics Lecture 3 Diffraction and the Reciprocal Lattice (Kittel Ch. 2)
 Solid State Physics 460 - Lecture 3 Diffraction and the Reciprocal Lattice (Kittel Ch. 2) Diffraction (Bragg Scattering) from a powder of crystallites - real example of image at right from http://www.uni-wuerzburg.de/mineralogie/crystal/teaching/pow.html
Solid State Physics 460 - Lecture 3 Diffraction and the Reciprocal Lattice (Kittel Ch. 2) Diffraction (Bragg Scattering) from a powder of crystallites - real example of image at right from http://www.uni-wuerzburg.de/mineralogie/crystal/teaching/pow.html
X-ray Crystallography
 2009/11/25 [ 1 ] X-ray Crystallography Andrew Torda, wintersemester 2009 / 2010 X-ray numerically most important more than 4/5 structures Goal a set of x, y, z coordinates different properties to NMR History
2009/11/25 [ 1 ] X-ray Crystallography Andrew Torda, wintersemester 2009 / 2010 X-ray numerically most important more than 4/5 structures Goal a set of x, y, z coordinates different properties to NMR History
7. Nuclear Magnetic Resonance
 7. Nuclear Magnetic Resonance Nuclear Magnetic Resonance (NMR) is another method besides crystallography that can be used to find structures of proteins. NMR spectroscopy is the observation of spins of
7. Nuclear Magnetic Resonance Nuclear Magnetic Resonance (NMR) is another method besides crystallography that can be used to find structures of proteins. NMR spectroscopy is the observation of spins of
Rigid Geometric Transformations
 Rigid Geometric Transformations Carlo Tomasi This note is a quick refresher of the geometry of rigid transformations in three-dimensional space, expressed in Cartesian coordinates. 1 Cartesian Coordinates
Rigid Geometric Transformations Carlo Tomasi This note is a quick refresher of the geometry of rigid transformations in three-dimensional space, expressed in Cartesian coordinates. 1 Cartesian Coordinates
Structure Factors. How to get more than unit cell sizes from your diffraction data.
 Structure Factors How to get more than unit cell sizes from your diffraction data http://homepages.utoledo.edu/clind/ Yet again expanding convenient concepts First concept introduced: Reflection from lattice
Structure Factors How to get more than unit cell sizes from your diffraction data http://homepages.utoledo.edu/clind/ Yet again expanding convenient concepts First concept introduced: Reflection from lattice
SOLID STATE 18. Reciprocal Space
 SOLID STATE 8 Reciprocal Space Wave vectors and the concept of K-space can simplify the explanation of several properties of the solid state. They will be introduced to provide more information on diffraction
SOLID STATE 8 Reciprocal Space Wave vectors and the concept of K-space can simplify the explanation of several properties of the solid state. They will be introduced to provide more information on diffraction
The following definition is fundamental.
 1. Some Basics from Linear Algebra With these notes, I will try and clarify certain topics that I only quickly mention in class. First and foremost, I will assume that you are familiar with many basic
1. Some Basics from Linear Algebra With these notes, I will try and clarify certain topics that I only quickly mention in class. First and foremost, I will assume that you are familiar with many basic
MS 3011 Exercises. December 11, 2013
 MS 3011 Exercises December 11, 2013 The exercises are divided into (A) easy (B) medium and (C) hard. If you are particularly interested I also have some projects at the end which will deepen your understanding
MS 3011 Exercises December 11, 2013 The exercises are divided into (A) easy (B) medium and (C) hard. If you are particularly interested I also have some projects at the end which will deepen your understanding
NATIONAL BOARD FOR HIGHER MATHEMATICS. M. A. and M.Sc. Scholarship Test. September 24, Time Allowed: 150 Minutes Maximum Marks: 30
 NATIONAL BOARD FOR HIGHER MATHEMATICS M. A. and M.Sc. Scholarship Test September 24, 2011 Time Allowed: 150 Minutes Maximum Marks: 30 Please read, carefully, the instructions on the following page 1 INSTRUCTIONS
NATIONAL BOARD FOR HIGHER MATHEMATICS M. A. and M.Sc. Scholarship Test September 24, 2011 Time Allowed: 150 Minutes Maximum Marks: 30 Please read, carefully, the instructions on the following page 1 INSTRUCTIONS
Quantum Condensed Matter Physics Lecture 5
 Quantum Condensed Matter Physics Lecture 5 detector sample X-ray source monochromator David Ritchie http://www.sp.phy.cam.ac.uk/drp2/home QCMP Lent/Easter 2019 5.1 Quantum Condensed Matter Physics 1. Classical
Quantum Condensed Matter Physics Lecture 5 detector sample X-ray source monochromator David Ritchie http://www.sp.phy.cam.ac.uk/drp2/home QCMP Lent/Easter 2019 5.1 Quantum Condensed Matter Physics 1. Classical
Crystal planes. Neutrons: magnetic moment - interacts with magnetic materials or nuclei of non-magnetic materials. (in Å)
 Crystallography: neutron, electron, and X-ray scattering from periodic lattice, scattering of waves by periodic structures, Miller indices, reciprocal space, Ewald construction. Diffraction: Specular,
Crystallography: neutron, electron, and X-ray scattering from periodic lattice, scattering of waves by periodic structures, Miller indices, reciprocal space, Ewald construction. Diffraction: Specular,
X-ray Data Collection. Bio5325 Spring 2006
 X-ray Data Collection Bio535 Spring 006 Obtaining I hkl and α (Ihkl) from Frame Images Braggs Law -predicts conditions for in-phase scattering by equivalent atoms lying in planes that transect a crystal.
X-ray Data Collection Bio535 Spring 006 Obtaining I hkl and α (Ihkl) from Frame Images Braggs Law -predicts conditions for in-phase scattering by equivalent atoms lying in planes that transect a crystal.
Quaternion Dynamics, Part 1 Functions, Derivatives, and Integrals. Gary D. Simpson. rev 00 Dec 27, 2014.
 Quaternion Dynamics, Part 1 Functions, Derivatives, and Integrals Gary D. Simpson gsim100887@aol.com rev 00 Dec 27, 2014 Summary Definitions are presented for "quaternion functions" of a quaternion. Polynomial
Quaternion Dynamics, Part 1 Functions, Derivatives, and Integrals Gary D. Simpson gsim100887@aol.com rev 00 Dec 27, 2014 Summary Definitions are presented for "quaternion functions" of a quaternion. Polynomial
SOLID STATE 9. Determination of Crystal Structures
 SOLID STATE 9 Determination of Crystal Structures In the diffraction experiment, we measure intensities as a function of d hkl. Intensities are the sum of the x-rays scattered by all the atoms in a crystal.
SOLID STATE 9 Determination of Crystal Structures In the diffraction experiment, we measure intensities as a function of d hkl. Intensities are the sum of the x-rays scattered by all the atoms in a crystal.
PX-CBMSO Course (2) of Symmetry
 PX-CBMSO Course (2) The mathematical description of Symmetry y PX-CBMSO-June 2011 Cele Abad-Zapatero University of Illinois at Chicago Center for Pharmaceutical Biotechnology. Lecture no. 2 This material
PX-CBMSO Course (2) The mathematical description of Symmetry y PX-CBMSO-June 2011 Cele Abad-Zapatero University of Illinois at Chicago Center for Pharmaceutical Biotechnology. Lecture no. 2 This material
Introduction to Group Theory
 Chapter 10 Introduction to Group Theory Since symmetries described by groups play such an important role in modern physics, we will take a little time to introduce the basic structure (as seen by a physicist)
Chapter 10 Introduction to Group Theory Since symmetries described by groups play such an important role in modern physics, we will take a little time to introduce the basic structure (as seen by a physicist)
Summary Chapter 2: Wave diffraction and the reciprocal lattice.
 Summary Chapter : Wave diffraction and the reciprocal lattice. In chapter we discussed crystal diffraction and introduced the reciprocal lattice. Since crystal have a translation symmetry as discussed
Summary Chapter : Wave diffraction and the reciprocal lattice. In chapter we discussed crystal diffraction and introduced the reciprocal lattice. Since crystal have a translation symmetry as discussed
Protein crystallography. Garry Taylor
 Protein crystallography Garry Taylor X-ray Crystallography - the Basics Grow crystals Collect X-ray data Determine phases Calculate ρ-map Interpret map Refine coordinates Do the biology. Nitrogen at -180
Protein crystallography Garry Taylor X-ray Crystallography - the Basics Grow crystals Collect X-ray data Determine phases Calculate ρ-map Interpret map Refine coordinates Do the biology. Nitrogen at -180
Chapter 2. X-ray X. Diffraction and Reciprocal Lattice. Scattering from Lattices
 Chapter. X-ray X Diffraction and Reciprocal Lattice Diffraction of waves by crystals Reciprocal Lattice Diffraction of X-rays Powder diffraction Single crystal X-ray diffraction Scattering from Lattices
Chapter. X-ray X Diffraction and Reciprocal Lattice Diffraction of waves by crystals Reciprocal Lattice Diffraction of X-rays Powder diffraction Single crystal X-ray diffraction Scattering from Lattices
Protein Structure Determination 9/25/2007
 One-dimensional NMR spectra Ethanol Cellulase (36 a.a.) Branden & Tooze, Fig. 18.16 1D and 2D NMR spectra of inhibitor K (57 a.a.) K. Wuthrich, NMR of Proteins and Nucleic Acids. (Wiley, 1986.) p. 54-55.
One-dimensional NMR spectra Ethanol Cellulase (36 a.a.) Branden & Tooze, Fig. 18.16 1D and 2D NMR spectra of inhibitor K (57 a.a.) K. Wuthrich, NMR of Proteins and Nucleic Acids. (Wiley, 1986.) p. 54-55.
Part IA. Vectors and Matrices. Year
 Part IA Vectors and Matrices Year 2018 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2018 Paper 1, Section I 1C Vectors and Matrices For z, w C define the principal value of z w. State de Moivre s
Part IA Vectors and Matrices Year 2018 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 2018 Paper 1, Section I 1C Vectors and Matrices For z, w C define the principal value of z w. State de Moivre s
X-ray analysis. 1. Basic crystallography 2. Basic diffraction physics 3. Experimental methods
 X-ray analysis 1. Basic crystallography 2. Basic diffraction physics 3. Experimental methods Introduction Noble prizes associated with X-ray diffraction 1901 W. C. Roentgen (Physics) for the discovery
X-ray analysis 1. Basic crystallography 2. Basic diffraction physics 3. Experimental methods Introduction Noble prizes associated with X-ray diffraction 1901 W. C. Roentgen (Physics) for the discovery
Rigid Geometric Transformations
 Rigid Geometric Transformations Carlo Tomasi This note is a quick refresher of the geometry of rigid transformations in three-dimensional space, expressed in Cartesian coordinates. 1 Cartesian Coordinates
Rigid Geometric Transformations Carlo Tomasi This note is a quick refresher of the geometry of rigid transformations in three-dimensional space, expressed in Cartesian coordinates. 1 Cartesian Coordinates
Candidates are expected to have available a calculator. Only division by (x + a) or (x a) will be required.
 Revision Checklist Unit C2: Core Mathematics 2 Unit description Algebra and functions; coordinate geometry in the (x, y) plane; sequences and series; trigonometry; exponentials and logarithms; differentiation;
Revision Checklist Unit C2: Core Mathematics 2 Unit description Algebra and functions; coordinate geometry in the (x, y) plane; sequences and series; trigonometry; exponentials and logarithms; differentiation;
Lattices Part II Dual Lattices, Fourier Transform, Smoothing Parameter, Public Key Encryption
 Lattices Part II Dual Lattices, Fourier Transform, Smoothing Parameter, Public Key Encryption Boaz Barak May 12, 2008 The first two sections are based on Oded Regev s lecture notes, and the third one on
Lattices Part II Dual Lattices, Fourier Transform, Smoothing Parameter, Public Key Encryption Boaz Barak May 12, 2008 The first two sections are based on Oded Regev s lecture notes, and the third one on
Analytical Methods for Materials
 Analytical Methods for Materials Lesson 15 Reciprocal Lattices and Their Roles in Diffraction Studies Suggested Reading Chs. 2 and 6 in Tilley, Crystals and Crystal Structures, Wiley (2006) Ch. 6 M. DeGraef
Analytical Methods for Materials Lesson 15 Reciprocal Lattices and Their Roles in Diffraction Studies Suggested Reading Chs. 2 and 6 in Tilley, Crystals and Crystal Structures, Wiley (2006) Ch. 6 M. DeGraef
Analysis II: Fourier Series
 .... Analysis II: Fourier Series Kenichi Maruno Department of Mathematics, The University of Texas - Pan American May 3, 011 K.Maruno (UT-Pan American) Analysis II May 3, 011 1 / 16 Fourier series were
.... Analysis II: Fourier Series Kenichi Maruno Department of Mathematics, The University of Texas - Pan American May 3, 011 K.Maruno (UT-Pan American) Analysis II May 3, 011 1 / 16 Fourier series were
High-Resolution. Transmission. Electron Microscopy
 Part 4 High-Resolution Transmission Electron Microscopy 186 Significance high-resolution transmission electron microscopy (HRTEM): resolve object details smaller than 1nm (10 9 m) image the interior of
Part 4 High-Resolution Transmission Electron Microscopy 186 Significance high-resolution transmission electron microscopy (HRTEM): resolve object details smaller than 1nm (10 9 m) image the interior of
The Reciprocal Lattice
 59-553 The Reciprocal Lattice 61 Because of the reciprocal nature of d spacings and θ from Bragg s Law, the pattern of the diffraction we observe can be related to the crystal lattice by a mathematical
59-553 The Reciprocal Lattice 61 Because of the reciprocal nature of d spacings and θ from Bragg s Law, the pattern of the diffraction we observe can be related to the crystal lattice by a mathematical
2. Diffraction as a means to determine crystal structure
 Page 1 of 22 2. Diffraction as a means to determine crystal structure Recall de Broglie matter waves: 2 p h E = where p = 2m λ h 1 E = ( ) 2m λ hc E = hυ = ( photons) λ ( matter wave) He atoms: [E (ev)]
Page 1 of 22 2. Diffraction as a means to determine crystal structure Recall de Broglie matter waves: 2 p h E = where p = 2m λ h 1 E = ( ) 2m λ hc E = hυ = ( photons) λ ( matter wave) He atoms: [E (ev)]
CSL361 Problem set 4: Basic linear algebra
 CSL361 Problem set 4: Basic linear algebra February 21, 2017 [Note:] If the numerical matrix computations turn out to be tedious, you may use the function rref in Matlab. 1 Row-reduced echelon matrices
CSL361 Problem set 4: Basic linear algebra February 21, 2017 [Note:] If the numerical matrix computations turn out to be tedious, you may use the function rref in Matlab. 1 Row-reduced echelon matrices
Molecular Biology Course 2006 Protein Crystallography Part I
 Molecular Biology Course 2006 Protein Crystallography Part I Tim Grüne University of Göttingen Dept. of Structural Chemistry November 2006 http://shelx.uni-ac.gwdg.de tg@shelx.uni-ac.gwdg.de Overview Overview
Molecular Biology Course 2006 Protein Crystallography Part I Tim Grüne University of Göttingen Dept. of Structural Chemistry November 2006 http://shelx.uni-ac.gwdg.de tg@shelx.uni-ac.gwdg.de Overview Overview
11.1 Three-Dimensional Coordinate System
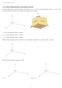 11.1 Three-Dimensional Coordinate System In three dimensions, a point has three coordinates: (x,y,z). The normal orientation of the x, y, and z-axes is shown below. The three axes divide the region into
11.1 Three-Dimensional Coordinate System In three dimensions, a point has three coordinates: (x,y,z). The normal orientation of the x, y, and z-axes is shown below. The three axes divide the region into
Chapter 1. Crystal structure. 1.1 Crystal lattices
 Chapter 1 Crystal structure 1.1 Crystal lattices We will concentrate as stated in the introduction, on perfect crystals, i.e. on arrays of atoms, where a given arrangement is repeated forming a periodic
Chapter 1 Crystal structure 1.1 Crystal lattices We will concentrate as stated in the introduction, on perfect crystals, i.e. on arrays of atoms, where a given arrangement is repeated forming a periodic
3.012 Fund of Mat Sci: Structure Lecture 18
 3.012 Fund of Mat Sci: Structure Lecture 18 X-RAYS AT WORK An X-ray diffraction image for the protein myoglobin. Source: Wikipedia. Model of helical domains in myoglobin. Image courtesy of Magnus Manske
3.012 Fund of Mat Sci: Structure Lecture 18 X-RAYS AT WORK An X-ray diffraction image for the protein myoglobin. Source: Wikipedia. Model of helical domains in myoglobin. Image courtesy of Magnus Manske
31. Diffraction: a few important illustrations
 31. Diffraction: a few important illustrations Babinet s Principle Diffraction gratings X-ray diffraction: Bragg scattering and crystal structures A lens transforms a Fresnel diffraction problem into a
31. Diffraction: a few important illustrations Babinet s Principle Diffraction gratings X-ray diffraction: Bragg scattering and crystal structures A lens transforms a Fresnel diffraction problem into a
Mathematics for Chemists 2 Lecture 14: Fourier analysis. Fourier series, Fourier transform, DFT/FFT
 Mathematics for Chemists 2 Lecture 14: Fourier analysis Fourier series, Fourier transform, DFT/FFT Johannes Kepler University Summer semester 2012 Lecturer: David Sevilla Fourier analysis 1/25 Remembering
Mathematics for Chemists 2 Lecture 14: Fourier analysis Fourier series, Fourier transform, DFT/FFT Johannes Kepler University Summer semester 2012 Lecturer: David Sevilla Fourier analysis 1/25 Remembering
X- ray crystallography. CS/CME/Biophys/BMI 279 Nov. 12, 2015 Ron Dror
 X- ray crystallography CS/CME/Biophys/BMI 279 Nov. 12, 2015 Ron Dror 1 Outline Overview of x-ray crystallography Crystals Electron density Diffraction patterns The computational problem: determining structure
X- ray crystallography CS/CME/Biophys/BMI 279 Nov. 12, 2015 Ron Dror 1 Outline Overview of x-ray crystallography Crystals Electron density Diffraction patterns The computational problem: determining structure
The Solid State. Phase diagrams Crystals and symmetry Unit cells and packing Types of solid
 The Solid State Phase diagrams Crystals and symmetry Unit cells and packing Types of solid Learning objectives Apply phase diagrams to prediction of phase behaviour Describe distinguishing features of
The Solid State Phase diagrams Crystals and symmetry Unit cells and packing Types of solid Learning objectives Apply phase diagrams to prediction of phase behaviour Describe distinguishing features of
Vector Geometry. Chapter 5
 Chapter 5 Vector Geometry In this chapter we will look more closely at certain geometric aspects of vectors in R n. We will first develop an intuitive understanding of some basic concepts by looking at
Chapter 5 Vector Geometry In this chapter we will look more closely at certain geometric aspects of vectors in R n. We will first develop an intuitive understanding of some basic concepts by looking at
Experimental Determination of Crystal Structure
 Experimental Determination of Crystal Structure Branislav K. Nikolić Department of Physics and Astronomy, University of Delaware, U.S.A. PHYS 624: Introduction to Solid State Physics http://www.physics.udel.edu/~bnikolic/teaching/phys624/phys624.html
Experimental Determination of Crystal Structure Branislav K. Nikolić Department of Physics and Astronomy, University of Delaware, U.S.A. PHYS 624: Introduction to Solid State Physics http://www.physics.udel.edu/~bnikolic/teaching/phys624/phys624.html
PURE MATHEMATICS AM 27
 AM SYLLABUS (2020) PURE MATHEMATICS AM 27 SYLLABUS 1 Pure Mathematics AM 27 (Available in September ) Syllabus Paper I(3hrs)+Paper II(3hrs) 1. AIMS To prepare students for further studies in Mathematics
AM SYLLABUS (2020) PURE MATHEMATICS AM 27 SYLLABUS 1 Pure Mathematics AM 27 (Available in September ) Syllabus Paper I(3hrs)+Paper II(3hrs) 1. AIMS To prepare students for further studies in Mathematics
1 Complex numbers and the complex plane
 L1: Complex numbers and complex-valued functions. Contents: The field of complex numbers. Real and imaginary part. Conjugation and modulus or absolute valued. Inequalities: The triangular and the Cauchy.
L1: Complex numbers and complex-valued functions. Contents: The field of complex numbers. Real and imaginary part. Conjugation and modulus or absolute valued. Inequalities: The triangular and the Cauchy.
Scattering and Diffraction
 Scattering and Diffraction Andreas Kreyssig, Alan Goldman, Rob McQueeney Ames Laboratory Iowa State University All rights reserved, 2018. Atomic scale structure - crystals Crystalline materials... atoms
Scattering and Diffraction Andreas Kreyssig, Alan Goldman, Rob McQueeney Ames Laboratory Iowa State University All rights reserved, 2018. Atomic scale structure - crystals Crystalline materials... atoms
4.1 Distance and Length
 Chapter Vector Geometry In this chapter we will look more closely at certain geometric aspects of vectors in R n. We will first develop an intuitive understanding of some basic concepts by looking at vectors
Chapter Vector Geometry In this chapter we will look more closely at certain geometric aspects of vectors in R n. We will first develop an intuitive understanding of some basic concepts by looking at vectors
Chapter 2 Kinematical theory of diffraction
 Graduate School of Engineering, Nagoya Institute of Technology Crystal Structure Analysis Taashi Ida (Advanced Ceramics Research Center) Updated Oct. 29, 2013 Chapter 2 Kinematical theory of diffraction
Graduate School of Engineering, Nagoya Institute of Technology Crystal Structure Analysis Taashi Ida (Advanced Ceramics Research Center) Updated Oct. 29, 2013 Chapter 2 Kinematical theory of diffraction
Complex numbers, the exponential function, and factorization over C
 Complex numbers, the exponential function, and factorization over C 1 Complex Numbers Recall that for every non-zero real number x, its square x 2 = x x is always positive. Consequently, R does not contain
Complex numbers, the exponential function, and factorization over C 1 Complex Numbers Recall that for every non-zero real number x, its square x 2 = x x is always positive. Consequently, R does not contain
Probing Atomic Crystals: Bragg Diffraction
 1 Probing Atomic Crystals: Bragg Diffraction OBJECTIVE: To learn how scientists probe the structure of solids, using a scaled-up version of X-ray Diffraction. APPARATUS: Steel ball "crystal", microwave
1 Probing Atomic Crystals: Bragg Diffraction OBJECTIVE: To learn how scientists probe the structure of solids, using a scaled-up version of X-ray Diffraction. APPARATUS: Steel ball "crystal", microwave
Crystallographic Symmetry. Jeremy Karl Cockcroft
 Crystallographic Symmetry Jeremy Karl Cockcroft Why bother? To describe crystal structures Simplifies the description, e.g. NaCl structure Requires coordinates for just 2 atoms + space group symmetry!
Crystallographic Symmetry Jeremy Karl Cockcroft Why bother? To describe crystal structures Simplifies the description, e.g. NaCl structure Requires coordinates for just 2 atoms + space group symmetry!
There and back again A short trip to Fourier Space. Janet Vonck 23 April 2014
 There and back again A short trip to Fourier Space Janet Vonck 23 April 2014 Where can I find a Fourier Transform? Fourier Transforms are ubiquitous in structural biology: X-ray diffraction Spectroscopy
There and back again A short trip to Fourier Space Janet Vonck 23 April 2014 Where can I find a Fourier Transform? Fourier Transforms are ubiquitous in structural biology: X-ray diffraction Spectroscopy
Donoghue, Golowich, Holstein Chapter 4, 6
 1 Week 7: Non linear sigma models and pion lagrangians Reading material from the books Burgess-Moore, Chapter 9.3 Donoghue, Golowich, Holstein Chapter 4, 6 Weinberg, Chap. 19 1 Goldstone boson lagrangians
1 Week 7: Non linear sigma models and pion lagrangians Reading material from the books Burgess-Moore, Chapter 9.3 Donoghue, Golowich, Holstein Chapter 4, 6 Weinberg, Chap. 19 1 Goldstone boson lagrangians
2. Diffraction as a means to determine crystal structure
 2. Diffraction as a means to determine crystal structure Recall de Broglie matter waves: He atoms: [E (ev)] 1/2 = 0.14 / (Å) E 1Å = 0.0196 ev Neutrons: [E (ev)] 1/2 = 0.28 / (Å) E 1Å = 0.0784 ev Electrons:
2. Diffraction as a means to determine crystal structure Recall de Broglie matter waves: He atoms: [E (ev)] 1/2 = 0.14 / (Å) E 1Å = 0.0196 ev Neutrons: [E (ev)] 1/2 = 0.28 / (Å) E 1Å = 0.0784 ev Electrons:
October 25, 2013 INNER PRODUCT SPACES
 October 25, 2013 INNER PRODUCT SPACES RODICA D. COSTIN Contents 1. Inner product 2 1.1. Inner product 2 1.2. Inner product spaces 4 2. Orthogonal bases 5 2.1. Existence of an orthogonal basis 7 2.2. Orthogonal
October 25, 2013 INNER PRODUCT SPACES RODICA D. COSTIN Contents 1. Inner product 2 1.1. Inner product 2 1.2. Inner product spaces 4 2. Orthogonal bases 5 2.1. Existence of an orthogonal basis 7 2.2. Orthogonal
Chapter 2: Vector Geometry
 Chapter 2: Vector Geometry Daniel Chan UNSW Semester 1 2018 Daniel Chan (UNSW) Chapter 2: Vector Geometry Semester 1 2018 1 / 32 Goals of this chapter In this chapter, we will answer the following geometric
Chapter 2: Vector Geometry Daniel Chan UNSW Semester 1 2018 Daniel Chan (UNSW) Chapter 2: Vector Geometry Semester 1 2018 1 / 32 Goals of this chapter In this chapter, we will answer the following geometric
13 Spherical geometry
 13 Spherical geometry Let ABC be a triangle in the Euclidean plane. From now on, we indicate the interior angles A = CAB, B = ABC, C = BCA at the vertices merely by A, B, C. The sides of length a = BC
13 Spherical geometry Let ABC be a triangle in the Euclidean plane. From now on, we indicate the interior angles A = CAB, B = ABC, C = BCA at the vertices merely by A, B, C. The sides of length a = BC
Solutions to Homework 11
 Solutions to Homework 11 Read the statement of Proposition 5.4 of Chapter 3, Section 5. Write a summary of the proof. Comment on the following details: Does the proof work if g is piecewise C 1? Or did
Solutions to Homework 11 Read the statement of Proposition 5.4 of Chapter 3, Section 5. Write a summary of the proof. Comment on the following details: Does the proof work if g is piecewise C 1? Or did
The ideal fiber pattern exhibits 4-quadrant symmetry. In the ideal pattern the fiber axis is called the meridian, the perpendicular direction is
 Fiber diffraction is a method used to determine the structural information of a molecule by using scattering data from X-rays. Rosalind Franklin used this technique in discovering structural information
Fiber diffraction is a method used to determine the structural information of a molecule by using scattering data from X-rays. Rosalind Franklin used this technique in discovering structural information
Texture, Microstructure & Anisotropy A.D. (Tony) Rollett
 1 Carnegie Mellon MRSEC 27-750 Texture, Microstructure & Anisotropy A.D. (Tony) Rollett Last revised: 5 th Sep. 2011 2 Show how to convert from a description of a crystal orientation based on Miller indices
1 Carnegie Mellon MRSEC 27-750 Texture, Microstructure & Anisotropy A.D. (Tony) Rollett Last revised: 5 th Sep. 2011 2 Show how to convert from a description of a crystal orientation based on Miller indices
Phys 460 Describing and Classifying Crystal Lattices
 Phys 460 Describing and Classifying Crystal Lattices What is a material? ^ crystalline Regular lattice of atoms Each atom has a positively charged nucleus surrounded by negative electrons Electrons are
Phys 460 Describing and Classifying Crystal Lattices What is a material? ^ crystalline Regular lattice of atoms Each atom has a positively charged nucleus surrounded by negative electrons Electrons are
Vectors in Function Spaces
 Jim Lambers MAT 66 Spring Semester 15-16 Lecture 18 Notes These notes correspond to Section 6.3 in the text. Vectors in Function Spaces We begin with some necessary terminology. A vector space V, also
Jim Lambers MAT 66 Spring Semester 15-16 Lecture 18 Notes These notes correspond to Section 6.3 in the text. Vectors in Function Spaces We begin with some necessary terminology. A vector space V, also
1 Matrices and vector spaces
 Matrices and vector spaces. Which of the following statements about linear vector spaces are true? Where a statement is false, give a counter-example to demonstrate this. (a) Non-singular N N matrices
Matrices and vector spaces. Which of the following statements about linear vector spaces are true? Where a statement is false, give a counter-example to demonstrate this. (a) Non-singular N N matrices
Qualification Exam: Mathematical Methods
 Qualification Exam: Mathematical Methods Name:, QEID#41534189: August, 218 Qualification Exam QEID#41534189 2 1 Mathematical Methods I Problem 1. ID:MM-1-2 Solve the differential equation dy + y = sin
Qualification Exam: Mathematical Methods Name:, QEID#41534189: August, 218 Qualification Exam QEID#41534189 2 1 Mathematical Methods I Problem 1. ID:MM-1-2 Solve the differential equation dy + y = sin
Electron Diffraction
 Electron iffraction o moving electrons display wave nature? To answer this question you will direct a beam of electrons through a thin layer of carbon and analyze the resulting pattern. Theory Louis de
Electron iffraction o moving electrons display wave nature? To answer this question you will direct a beam of electrons through a thin layer of carbon and analyze the resulting pattern. Theory Louis de
Materials 286C/UCSB: Class VI Structure factors (continued), the phase problem, Patterson techniques and direct methods
 Materials 286C/UCSB: Class VI Structure factors (continued), the phase problem, Patterson techniques and direct methods Ram Seshadri (seshadri@mrl.ucsb.edu) Structure factors The structure factor for a
Materials 286C/UCSB: Class VI Structure factors (continued), the phase problem, Patterson techniques and direct methods Ram Seshadri (seshadri@mrl.ucsb.edu) Structure factors The structure factor for a
Introduction to Materials Science Graduate students (Applied Physics)
 Introduction to Materials Science Graduate students (Applied Physics) Prof. Michael Roth Chapter Reciprocal Lattice and X-ray Diffraction Reciprocal Lattice - 1 The crystal can be viewed as made up of
Introduction to Materials Science Graduate students (Applied Physics) Prof. Michael Roth Chapter Reciprocal Lattice and X-ray Diffraction Reciprocal Lattice - 1 The crystal can be viewed as made up of
Bricklaying and the Hermite Normal Form
 Bricklaying and the Hermite Normal Form William J. Gilbert Pure Mathematics Department University of Waterloo Waterloo, Ontario Canada N2L 3G1 AMS Classifications: Primary: 15A36 Appeared in the American
Bricklaying and the Hermite Normal Form William J. Gilbert Pure Mathematics Department University of Waterloo Waterloo, Ontario Canada N2L 3G1 AMS Classifications: Primary: 15A36 Appeared in the American
Here are brief notes about topics covered in class on complex numbers, focusing on what is not covered in the textbook.
 Phys374, Spring 2008, Prof. Ted Jacobson Department of Physics, University of Maryland Complex numbers version 5/21/08 Here are brief notes about topics covered in class on complex numbers, focusing on
Phys374, Spring 2008, Prof. Ted Jacobson Department of Physics, University of Maryland Complex numbers version 5/21/08 Here are brief notes about topics covered in class on complex numbers, focusing on
PART 1 Introduction to Theory of Solids
 Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:1 Trim:165 240MM TS: Integra, India PART 1 Introduction to Theory of Solids Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:2
Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:1 Trim:165 240MM TS: Integra, India PART 1 Introduction to Theory of Solids Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:2
The Cross Product. Philippe B. Laval. Spring 2012 KSU. Philippe B. Laval (KSU) The Cross Product Spring /
 The Cross Product Philippe B Laval KSU Spring 2012 Philippe B Laval (KSU) The Cross Product Spring 2012 1 / 15 Introduction The cross product is the second multiplication operation between vectors we will
The Cross Product Philippe B Laval KSU Spring 2012 Philippe B Laval (KSU) The Cross Product Spring 2012 1 / 15 Introduction The cross product is the second multiplication operation between vectors we will
Wave diffraction and the reciprocal lattice
 Wave diffraction and the reciprocal lattice Dept of Phys M.C. Chang Braggs theory of diffraction Reciprocal lattice von Laue s theory of diffraction Braggs view of the diffraction (1912, father and son)
Wave diffraction and the reciprocal lattice Dept of Phys M.C. Chang Braggs theory of diffraction Reciprocal lattice von Laue s theory of diffraction Braggs view of the diffraction (1912, father and son)
de Broglie Waves h p de Broglie argued Light exhibits both wave and particle properties
 de Broglie argued de Broglie Waves Light exhibits both wave and particle properties Wave interference, diffraction Particle photoelectric effect, Compton effect Then matter (particles) should exhibit both
de Broglie argued de Broglie Waves Light exhibits both wave and particle properties Wave interference, diffraction Particle photoelectric effect, Compton effect Then matter (particles) should exhibit both
X-ray Crystallography. Kalyan Das
 X-ray Crystallography Kalyan Das Electromagnetic Spectrum NMR 10 um - 10 mm 700 to 10 4 nm 400 to 700 nm 10 to 400 nm 10-1 to 10 nm 10-4 to 10-1 nm X-ray radiation was discovered by Roentgen in 1895. X-rays
X-ray Crystallography Kalyan Das Electromagnetic Spectrum NMR 10 um - 10 mm 700 to 10 4 nm 400 to 700 nm 10 to 400 nm 10-1 to 10 nm 10-4 to 10-1 nm X-ray radiation was discovered by Roentgen in 1895. X-rays
Structure factors again
 Structure factors again Remember 1D, structure factor for order h F h = F h exp[iα h ] = I 01 ρ(x)exp[2πihx]dx Where x is fractional position along unit cell distance (repeating distance, origin arbitrary)
Structure factors again Remember 1D, structure factor for order h F h = F h exp[iα h ] = I 01 ρ(x)exp[2πihx]dx Where x is fractional position along unit cell distance (repeating distance, origin arbitrary)
NATIONAL BOARD FOR HIGHER MATHEMATICS. M. A. and M.Sc. Scholarship Test. September 25, Time Allowed: 150 Minutes Maximum Marks: 30
 NATIONAL BOARD FOR HIGHER MATHEMATICS M. A. and M.Sc. Scholarship Test September 25, 2010 Time Allowed: 150 Minutes Maximum Marks: 30 Please read, carefully, the instructions on the following page 1 INSTRUCTIONS
NATIONAL BOARD FOR HIGHER MATHEMATICS M. A. and M.Sc. Scholarship Test September 25, 2010 Time Allowed: 150 Minutes Maximum Marks: 30 Please read, carefully, the instructions on the following page 1 INSTRUCTIONS
6. Vectors. Given two points, P 0 = (x 0, y 0 ) and P 1 = (x 1, y 1 ), a vector can be drawn with its foot at P 0 and
 6. Vectors For purposes of applications in calculus and physics, a vector has both a direction and a magnitude (length), and is usually represented as an arrow. The start of the arrow is the vector s foot,
6. Vectors For purposes of applications in calculus and physics, a vector has both a direction and a magnitude (length), and is usually represented as an arrow. The start of the arrow is the vector s foot,
Basic Principles of Quasicrystallography
 Chapter 2 Basic Principles of Quasicrystallography Conventional crystals are constructed by a periodic repetition of a single unit-cell. This periodic long-range order only agrees with two-, three-, four-
Chapter 2 Basic Principles of Quasicrystallography Conventional crystals are constructed by a periodic repetition of a single unit-cell. This periodic long-range order only agrees with two-, three-, four-
