Chemical Engineering 412
|
|
|
- Arron Allen
- 5 years ago
- Views:
Transcription
1 Chemical Engineering 412 Introductory Nuclear Engineering Lecture 7 Nuclear Decay Behaviors
2 Spiritual Thought Sooner or later, I believe that all of us experience times when the very fabric of our world tears at the seams, leaving us feeling alone, frustrated, and adrift. It can happen to anyone. No one is immune. Everyone s situation is different, and the details of each life are unique. Nevertheless, I have learned that there is something that would take away the bitterness that may come into our lives. There is one thing we can do to make life sweeter, more joyful, even glorious. We can be grateful! Dieter F. Uchtdorf
3 Last Time: Decay Mechanisms Gamma (γγ) Alpha (αα) Beta (+/-) (ββ +, ββ ) Electron capture (EC) Proton (P) Neutron (N) Internal conversion (IC)
4 Decay Constant (λ) NN = number of atoms in the radionuclide sample NN = atoms that undergo decay during t λλ lim tt 0 NN NN tt Definition: the probability that a radionuclide will decay over an infinitesimally small unit of time λλ ff(tt, PP, NN ii, tt)
5 Exponential Decay Now we convert to continuous mathematics, and: NN(tt) = average or expected number of radionuclides at time tt dddd = λλnn tt dddd dddd(tt) dddd = λλnn tt N tt = NN oo ee λλλλ Radioactive Decay Law where NN oo = number of radionuclides in the sample at tt = 0
6 Half-Life (TT 1/2 ) Definition: constant representing the time it takes for a radionuclide to decay to ½ of its initial sample size NN TT 1 2 = NN oo 2 = NN ooee λλtt 1 2 TT 1/2 = lllll λλ λλ TT 1 2 ff(tt, PP, NN ii, tt) When solving for number of half-lives nn needed for a radioactive sample to decay to a fraction εε of its initial value: εε = NN nntt 1 2 NN oo = 1 2 nn ln εε nn = ln ln εε
7 Example A sample contains pure TTTT (Tellurium) which undergoes ββ decay and has a TT 1 2 of 9.35 h. (a) What is the decay constant (in h -1 ) for this radionuclide. λλ = ln 2 = ln 2 = h 1 TT 1 2 (9.35 h) (b) How long will it take for the sample to decay to 2 5 of the original number of atoms? ln εε nn = ln 2 = ln (2 5) = ln 2 tt = nn TT 1 2 = h = h
8 Half Life Histogram Age of Earth Age of Universe
9 Decay Probability & Avg. Lifetime Decay Probability for a Finite Time Interval: NN tt PP tt = = exp λλλλ NN 0 PP tt = 1 PP tt = 1 exp λλλλ pp tt = λλ exp λλλλ NN tt = number of atoms at time tt. PP tt = probability of existence during 0-tt. PP tt = decay probability in time 0-tt. pp tt = probability density Average Lifetime: TT aaaaaa = tt pp tt dddd = 1 0 λλ
10 Parallel Decay Routes 64 29CCCC ZZZZ ββ 39.0% = PPPPPPP NNNN ββ % = PPPPPPP 2 64 NNNN EEEE 43.6% = PPPPPPP 3 28 ddnn CCCC dddd 3 = 1 λλ ii NN CCCC (tt) = NN CCCC (tt) 3 1 λλ ii = λλ tttttt NN CCCC (tt)
11 ff ii = λλ ii λλ tttttttttt Parallel Decay Routes λλ tttttttttt λλ tttttttttt = ii λλ ii
12 Example 64 29CCCC ZZZZ ββ 39.0% = PPPPPPP NNNN ββ % = PPPPPPP 2 64 NNNN EEEE 43.6% = PPPPPPP 3 28 If CCCC has an overall TT 1 2 of 12.7 h, calculate the decay constants for the three decay modes in h -1. λλ tttttt = ln 2 = ln 2 = h 1 TT 1 2 (12.7 h) λλ ββ = ff ββ λλ tttttt = λλ tttttt = h 1 λλ ββ + = ff ββ +λλ tttttt = λλ tttttt = h 1 λλ EEEE = ff EEEE λλ tttttt = λλ tttttt = h 1
13 Activity Definition: number of decays or transmutations per unit of time that occur within the sample Units: Becquerel (Bq) = 1 transformation / sec Curie (Ci) = 3.7*10 10 Bq AA tt ddnn ii dddd = λλnn ii tt = AA 0 exp λλλλ Specific Activity = activity per unit mass AA tt = AA tt mm(tt) = λλnn aa MM Nuclides undergoing single decay mechanisms exhibit decreasing activity and constant specific activity with time.
14 Series Decay with Production When the decay of a radionuclide is accompanied by the creation of new atoms of the radionuclide: ddnn ii (tt) dddd = λλnn ii (tt) + QQ ii (tt) tt NN ii tt = NN ii,0 ee λλλλ + QQ ii ttt ee λλ tt ttt ddddd 0 For QQ = constant: NN ii tt = NN ii,0 exp λλλλ + QQ ii,0 λλ 1 exp λλλλ And as tt = : NN ii eeee = QQ ii,0 λλ
15 General Decay Chain Decay chain: when a parent radionuclide decays into a daughter, that then decays into another daughter, as so on, until a stable daughter is formed XX 1 λλ 1 XX 2 λλ 2 XX ii λλ ii λλ nn 1 XX nn (stable) ddnn 1 (tt) dddd = λλ 1 NN 1 (tt) ddnn 2 (tt) = λλ dddd 1 NN 1 (tt) λλ 2 NN 2 (tt) ddnn ii (tt) = λλ dddd ii 1 NN ii 1 (tt) λλ ii NN ii (tt) ddnn nn (tt) = λλ dddd nn 1 NN nn 1 (tt)
16 General Decay Chain For the case when only radionuclides of the parent XX 1 are initially present (NN 1 (0) 0 and NN ii 0 = 0, ii > 1): AA jj tt = λλ jj NN jj tt = NN 1 0 CC 1 ee λλ 1tt + CC 2 ee λλ 2tt + + CC jj ee λλ jjtt AA jj tt = NN 1 (0) CC mm ee λλ mmtt jj mm=1 CC mm = jj Π ii=1 ii mm jj Π ii=1 λλ ii λλ ii λλ mm = λλ 1 λλ 2 λλ 3 λλ jj λλ 1 λλ mm λλ 2 λλ mm λλ 3 λλ mm λλ jj λλ mm
17 Secular Equilibrium At long times compared to the half lives of the daughters (but short compared to the chain head), the activities of all species are the same. NN 1 λλ 1 = NN 2 λλ 2 = = NN jj λλ jj AA oo = AA 1 = AA 2 = = AA jj Species with short half lives (large λλ) have low concentrations, but concentrations can be estimated from species with longer half lives, in particular from the head of the chain. NN 1 λλ 1 = NN kk λλ kk NN 1 = NN kk (TT 1 2 ) 1 (TT 1 2 ) kk
18 Example Secular Equilibrium Example Problem
19 Natural Radionuclides 65 natural isotopes Cosmogenically produced 3 H, 7 Be, 14 C H and C used for dating. All three source of radioactivity in air samples. Primordial Isotopes Singly occurring (17) including 40 K and 87 Rb (parts of humans) Decay Products with Z > 83 form from 232 Th, 235 U, or 238 U via αα and ββ emission. Isolated natural radionuclides
20 Thorium Decay Chain (4n)
21 Uranium Decay Chain (4n+2)
22 Actinium Decay Chain (4n+3)
23 4 Possible decay chains Mass number given by 4n ( 232 Th), 4n+2 ( 238 U or 234 Np), and 4n+3 ( 235 U or 239 Pu) are near secular equilibrium. 4n+1 ( 241 Ac/Pu) has no step slow enough. Radioisotopes from it have long since decayed and are not found in nature.
Introduction to Nuclear Engineering. Ahmad Al Khatibeh
 Introduction to Nuclear Engineering Ahmad Al Khatibeh CONTENTS INTRODUCTION (Revision) RADIOACTIVITY Radioactive Decay Rates Units of Measurement for Radioactivity Variation of Radioactivity Over Time.
Introduction to Nuclear Engineering Ahmad Al Khatibeh CONTENTS INTRODUCTION (Revision) RADIOACTIVITY Radioactive Decay Rates Units of Measurement for Radioactivity Variation of Radioactivity Over Time.
Radiation Basics Cookie photo: Public domain.
 Radiation Basics Cookie photo: Public domain. 22. 02 DI eiger Counters Prof. Michael hort Old 22.01 Final Exam Question ou have four highl radioactive coo ies: α β γ n ou must: Put one in our poc et at
Radiation Basics Cookie photo: Public domain. 22. 02 DI eiger Counters Prof. Michael hort Old 22.01 Final Exam Question ou have four highl radioactive coo ies: α β γ n ou must: Put one in our poc et at
SOURCES of RADIOACTIVITY
 Section 9: SOURCES of RADIOACTIVITY This section briefly describes various sources of radioactive nuclei, both naturally occurring and those produced artificially (man-made) in, for example, reactors or
Section 9: SOURCES of RADIOACTIVITY This section briefly describes various sources of radioactive nuclei, both naturally occurring and those produced artificially (man-made) in, for example, reactors or
Answers to Practice Test Questions 2 Atoms, Isotopes and Nuclear Chemistry
 Answers to Practice Test Questions 2 Atoms, Isotopes and Nuclear Chemistry. Fluine has only one stable isotope. Its mass number is _9_. A neutral atom of fluine has 9 protons, 0 neutrons and 9 electrons.
Answers to Practice Test Questions 2 Atoms, Isotopes and Nuclear Chemistry. Fluine has only one stable isotope. Its mass number is _9_. A neutral atom of fluine has 9 protons, 0 neutrons and 9 electrons.
Nuclear Chemistry - HW
 Nuclear Chemistry - HW PSI AP Chemistry Name 1) In balancing the nuclear reaction 238 92U 234 90E + 4 2He, the identity of element E is. A) Pu B) Np C) U D) Pa E) Th 2) This reaction is an example of.
Nuclear Chemistry - HW PSI AP Chemistry Name 1) In balancing the nuclear reaction 238 92U 234 90E + 4 2He, the identity of element E is. A) Pu B) Np C) U D) Pa E) Th 2) This reaction is an example of.
1.1 ALPHA DECAY 1.2 BETA MINUS DECAY 1.3 GAMMA EMISSION 1.4 ELECTRON CAPTURE/BETA PLUS DECAY 1.5 NEUTRON EMISSION 1.6 SPONTANEOUS FISSION
 Chapter NP-3 Nuclear Physics Decay Modes and Decay Rates TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 RADIOACTIVE DECAY 1.1 ALPHA DECAY 1.2 BETA MINUS DECAY 1.3 GAMMA EMISSION 1.4 ELECTRON CAPTURE/BETA
Chapter NP-3 Nuclear Physics Decay Modes and Decay Rates TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 RADIOACTIVE DECAY 1.1 ALPHA DECAY 1.2 BETA MINUS DECAY 1.3 GAMMA EMISSION 1.4 ELECTRON CAPTURE/BETA
Recap from last time
 Recap from last time Nuclear Decay Occurs. when a nucleus is unstable (lower open energy levels) An unstable nucleus metamorphoses ( decays ) into a more stable (more tightly bound) nucleus Difference
Recap from last time Nuclear Decay Occurs. when a nucleus is unstable (lower open energy levels) An unstable nucleus metamorphoses ( decays ) into a more stable (more tightly bound) nucleus Difference
Strand J. Atomic Structure. Unit 2. Radioactivity. Text
 Strand J. Atomic Structure Unit 2. Radioactivity Contents Page Unstable Nuclei 2 Alpha, Beta and Gamma Radiation 5 Balancing Equations for Radioactive Decay 10 Half Life 12 J.2.1. Unstable Nuclei. The
Strand J. Atomic Structure Unit 2. Radioactivity Contents Page Unstable Nuclei 2 Alpha, Beta and Gamma Radiation 5 Balancing Equations for Radioactive Decay 10 Half Life 12 J.2.1. Unstable Nuclei. The
25.1. Nuclear Radiation
 Nuclear Radiation Marie Curie was a Polish scientist whose research led to many discoveries about radiation and radioactive elements. In 1934 she died from leukemia caused by her long-term exposure to
Nuclear Radiation Marie Curie was a Polish scientist whose research led to many discoveries about radiation and radioactive elements. In 1934 she died from leukemia caused by her long-term exposure to
Nuclear Fusion and Radiation
 Nuclear Fusion and Radiation Lecture 8 (Meetings 20, 21 & 22) Eugenio Schuster schuster@lehigh.edu Mechanical Engineering and Mechanics Lehigh University Nuclear Fusion and Radiation p. 1/66 The discovery
Nuclear Fusion and Radiation Lecture 8 (Meetings 20, 21 & 22) Eugenio Schuster schuster@lehigh.edu Mechanical Engineering and Mechanics Lehigh University Nuclear Fusion and Radiation p. 1/66 The discovery
General Physics (PHY 2140)
 General Physics (PHY 2140) Lecture 37 Modern Physics Nuclear Physics Radioactivity Nuclear reactions http://www.physics.wayne.edu/~apetrov/phy2140/ Chapter 29 1 Lightning Review Last lecture: 1. Nuclear
General Physics (PHY 2140) Lecture 37 Modern Physics Nuclear Physics Radioactivity Nuclear reactions http://www.physics.wayne.edu/~apetrov/phy2140/ Chapter 29 1 Lightning Review Last lecture: 1. Nuclear
Chapter 25. Nuclear Chemistry. Types of Radiation
 Chapter 25 Nuclear Chemistry Chemical Reactions 1. Bonds are broken and formed 2. Atoms may rearrange, but remain unchanged 3. Involve only valence electrons 4. Small energy changes 5. Reaction rate is
Chapter 25 Nuclear Chemistry Chemical Reactions 1. Bonds are broken and formed 2. Atoms may rearrange, but remain unchanged 3. Involve only valence electrons 4. Small energy changes 5. Reaction rate is
Radioactive Decay of 220 Rn and 232 Th Physics 2150 Experiment No. 10 University of Colorado
 Experiment 10 1 Introduction Radioactive Decay of 220 Rn and 232 Th Physics 2150 Experiment No. 10 University of Colorado Some radioactive isotopes formed billions of years ago have half-lives so long
Experiment 10 1 Introduction Radioactive Decay of 220 Rn and 232 Th Physics 2150 Experiment No. 10 University of Colorado Some radioactive isotopes formed billions of years ago have half-lives so long
Section 10: Natural Transmutation Writing Equations for Decay
 Section 10: Natural Transmutation Writing Equations for Decay Alpha Decay If a radioactive substance changes into another substance because particles are emitted from its nucleus, we say that the original
Section 10: Natural Transmutation Writing Equations for Decay Alpha Decay If a radioactive substance changes into another substance because particles are emitted from its nucleus, we say that the original
General, Organic, and Biological Chemistry, 3e (Frost) Chapter 2 Atoms and Radioactivity. 2.1 Multiple-Choice
 General, Organic, and Biological Chemistry, 3e (Frost) Chapter 2 Atoms and Radioactivity 2.1 Multiple-Choice 1) The smallest particle of an element that can be identified as that element is: A) a proton
General, Organic, and Biological Chemistry, 3e (Frost) Chapter 2 Atoms and Radioactivity 2.1 Multiple-Choice 1) The smallest particle of an element that can be identified as that element is: A) a proton
Chapter 3 Radioactivity
 Chapter 3 Radioactivity Marie Curie 1867 1934 Discovered new radioactive elements Shared Nobel Prize in physics in 1903 Nobel Prize in Chemistry in 1911 Radioactivity Radioactivity is the spontaneous emission
Chapter 3 Radioactivity Marie Curie 1867 1934 Discovered new radioactive elements Shared Nobel Prize in physics in 1903 Nobel Prize in Chemistry in 1911 Radioactivity Radioactivity is the spontaneous emission
Variations. ECE 6540, Lecture 02 Multivariate Random Variables & Linear Algebra
 Variations ECE 6540, Lecture 02 Multivariate Random Variables & Linear Algebra Last Time Probability Density Functions Normal Distribution Expectation / Expectation of a function Independence Uncorrelated
Variations ECE 6540, Lecture 02 Multivariate Random Variables & Linear Algebra Last Time Probability Density Functions Normal Distribution Expectation / Expectation of a function Independence Uncorrelated
Radioactive Decay. Becquerel. Atomic Physics. In 1896 Henri Becquerel. - uranium compounds would fog photographic plates as if exposed to light.
 Radioactive Decay Atomic Physics Becquerel In 1896 Henri Becquerel - uranium compounds would fog photographic plates as if exposed to light. - a magnetic field could deflect the radiation that caused the
Radioactive Decay Atomic Physics Becquerel In 1896 Henri Becquerel - uranium compounds would fog photographic plates as if exposed to light. - a magnetic field could deflect the radiation that caused the
LECTURE 26 RADIATION AND RADIOACTIVITY. Instructor: Kazumi Tolich
 LECTURE 26 RADIATION AND RADIOACTIVITY Instructor: Kazumi Tolich Lecture 26 2 30.4 Radiation and radioactivity Alpha decay Beta decay Gamma decay Decay series Nuclear radiation is a form of ionizing radiation
LECTURE 26 RADIATION AND RADIOACTIVITY Instructor: Kazumi Tolich Lecture 26 2 30.4 Radiation and radioactivity Alpha decay Beta decay Gamma decay Decay series Nuclear radiation is a form of ionizing radiation
Radioisotope Tracers
 Radioisotope Tracers OCN 623 Chemical Oceanography 23 March 2017 Reading: Emerson and Hedges, Chapter 5, p.153-169 2017 Frank Sansone Student Learning Outcomes At the completion of this class, students
Radioisotope Tracers OCN 623 Chemical Oceanography 23 March 2017 Reading: Emerson and Hedges, Chapter 5, p.153-169 2017 Frank Sansone Student Learning Outcomes At the completion of this class, students
General, Organic, and Biochemistry, 2e (Frost) Chapter 2 Atoms and Radioactivity. 2.1 Multiple-Choice
 General, Organic, and Biochemistry, 2e (Frost) Chapter 2 Atoms and Radioactivity 2.1 Multiple-Choice 1) Two atoms must represent the same element if they both have the same: A) number of electron shells
General, Organic, and Biochemistry, 2e (Frost) Chapter 2 Atoms and Radioactivity 2.1 Multiple-Choice 1) Two atoms must represent the same element if they both have the same: A) number of electron shells
Chapter IV: Radioactive decay
 Chapter IV: Radioactive decay 1 Summary 1. Law of radioactive decay 2. Decay chain/radioactive filiation 3. Quantum description 4. Types of radioactive decay 2 History Radioactivity was discover in 1896
Chapter IV: Radioactive decay 1 Summary 1. Law of radioactive decay 2. Decay chain/radioactive filiation 3. Quantum description 4. Types of radioactive decay 2 History Radioactivity was discover in 1896
Atomic Structure and Nuclear Chemistry Multiple Choice Questions PSI Chemistry
 Atomic Structure and Nuclear Chemistry Multiple Choice Questions PSI Chemistry Name: 1. What was the first particle discovered inside an atom? A. Proton C. Electron 2. What characteristic of cathode rays
Atomic Structure and Nuclear Chemistry Multiple Choice Questions PSI Chemistry Name: 1. What was the first particle discovered inside an atom? A. Proton C. Electron 2. What characteristic of cathode rays
Sources of Radiation
 Radioactivity Sources of Radiation Natural Sources Cosmic Radiation The Earth is constantly bombarded by radiation from outside our solar system. interacts in the atmosphere to create secondary radiation
Radioactivity Sources of Radiation Natural Sources Cosmic Radiation The Earth is constantly bombarded by radiation from outside our solar system. interacts in the atmosphere to create secondary radiation
Nuclear Study Packet. 1. What subatomic particles are involved in nuclear reactions? 2. What subatomic particles are involved in chemical reactions?
 Name Period Nuclear Study Packet Set 1 1. What subatomic particles are involved in nuclear reactions? 2. What subatomic particles are involved in chemical reactions? 3. If an atom undergoes a reaction
Name Period Nuclear Study Packet Set 1 1. What subatomic particles are involved in nuclear reactions? 2. What subatomic particles are involved in chemical reactions? 3. If an atom undergoes a reaction
Chemistry 201: General Chemistry II - Lecture
 Chemistry 201: General Chemistry II - Lecture Dr. Namphol Sinkaset Chapter 21 Study Guide Concepts 1. There are several modes of radioactive decay: (1) alpha (α) decay, (2) beta (β) decay, (3) gamma (γ)
Chemistry 201: General Chemistry II - Lecture Dr. Namphol Sinkaset Chapter 21 Study Guide Concepts 1. There are several modes of radioactive decay: (1) alpha (α) decay, (2) beta (β) decay, (3) gamma (γ)
Particle Physics. Question Paper 1. Save My Exams! The Home of Revision. International A Level. Exam Board Particle & Nuclear Physics
 For more awesome GSE and level resources, visit us at www.savemyexams.co.uk/ Particle Physics Question Paper 1 Level International Level Subject Physics Exam oard IE Topic Particle & Nuclear Physics Sub
For more awesome GSE and level resources, visit us at www.savemyexams.co.uk/ Particle Physics Question Paper 1 Level International Level Subject Physics Exam oard IE Topic Particle & Nuclear Physics Sub
Chapter 18: Radioactivity And Nuclear Transformation. Presented by Mingxiong Huang, Ph.D.,
 Chapter 18: Radioactivity And Nuclear Transformation Presented by Mingxiong Huang, Ph.D., mxhuang@ucsd.edu 18.1 Radionuclide Decay Terms and Relationships Activity Decay Constant Physical Half-Life Fundamental
Chapter 18: Radioactivity And Nuclear Transformation Presented by Mingxiong Huang, Ph.D., mxhuang@ucsd.edu 18.1 Radionuclide Decay Terms and Relationships Activity Decay Constant Physical Half-Life Fundamental
Lecture 3 Radioactivity
 Objectives In this lecture you will learn the following We shall begin with a general discussion on the nucleus. Learn about some characteristics of nucleons. Understand some concepts on stability of a
Objectives In this lecture you will learn the following We shall begin with a general discussion on the nucleus. Learn about some characteristics of nucleons. Understand some concepts on stability of a
Chapter 19 - Nuclear Chemistry Nuclear Stability and Modes of Decay
 Chapter 19 - Nuclear Chemistry Nuclear Stability and Modes of Decay History and Discovery of Radioactivity The Discovery of Radioactivity (1896) Antoine-Henri Bequerel designed experiment to determine
Chapter 19 - Nuclear Chemistry Nuclear Stability and Modes of Decay History and Discovery of Radioactivity The Discovery of Radioactivity (1896) Antoine-Henri Bequerel designed experiment to determine
Fiesta Ware. Nuclear Chemistry. 2009, Prentice-Hall, Inc.
 Fiesta Ware 2009, Prentice-Hall, Inc. Measuring Radioactivity One can use a device like this Geiger counter to measure the amount of activity present in a radioactive sample. The ionizing radiation creates
Fiesta Ware 2009, Prentice-Hall, Inc. Measuring Radioactivity One can use a device like this Geiger counter to measure the amount of activity present in a radioactive sample. The ionizing radiation creates
ECE 6540, Lecture 06 Sufficient Statistics & Complete Statistics Variations
 ECE 6540, Lecture 06 Sufficient Statistics & Complete Statistics Variations Last Time Minimum Variance Unbiased Estimators Sufficient Statistics Proving t = T(x) is sufficient Neyman-Fischer Factorization
ECE 6540, Lecture 06 Sufficient Statistics & Complete Statistics Variations Last Time Minimum Variance Unbiased Estimators Sufficient Statistics Proving t = T(x) is sufficient Neyman-Fischer Factorization
Nuclear Chemistry. Lecture 10
 Nuclear Chemistry Lecture 10 Atomic Nuclei The periodic table tells you about the average atom of an element. Atoms of an element can have different amounts of neutrons, this gives them different mass,
Nuclear Chemistry Lecture 10 Atomic Nuclei The periodic table tells you about the average atom of an element. Atoms of an element can have different amounts of neutrons, this gives them different mass,
Chapter 19 - Nuclear Chemistry Nuclear Stability and Modes of Decay
 Chapter 19 - Nuclear Chemistry Nuclear Stability and Modes of Decay History and Discovery of Radioactivity The Discovery of Radioactivity (1896) Antoine-Henri Bequerel designed experiment to determine
Chapter 19 - Nuclear Chemistry Nuclear Stability and Modes of Decay History and Discovery of Radioactivity The Discovery of Radioactivity (1896) Antoine-Henri Bequerel designed experiment to determine
Nuclear Physics Part 2: Radioactive Decay
 Nuclear Physics Part 2: Radioactive Decay Last modified: 17/10/2017 Part A: Decay Reactions What is a Decay? Alpha Decay Definition Q-value Example Not Every Alpha Decay is Possible Beta Decay β rays are
Nuclear Physics Part 2: Radioactive Decay Last modified: 17/10/2017 Part A: Decay Reactions What is a Decay? Alpha Decay Definition Q-value Example Not Every Alpha Decay is Possible Beta Decay β rays are
Some nuclei are unstable Become stable by ejecting excess energy and often a particle in the process Types of radiation particle - particle
 Radioactivity George Starkschall, Ph.D. Lecture Objectives Identify methods for making radioactive isotopes Recognize the various types of radioactive decay Interpret an energy level diagram for radioactive
Radioactivity George Starkschall, Ph.D. Lecture Objectives Identify methods for making radioactive isotopes Recognize the various types of radioactive decay Interpret an energy level diagram for radioactive
Radioactivity. General Physics II PHYS 111. King Saud University College of Applied Studies and Community Service Department of Natural Sciences
 King Saud University College of Applied Studies and Community Service Department of Natural Sciences Radioactivity General Physics II PHYS 111 Nouf Alkathran nalkathran@ksu.edu.sa Outline Radioactive Decay
King Saud University College of Applied Studies and Community Service Department of Natural Sciences Radioactivity General Physics II PHYS 111 Nouf Alkathran nalkathran@ksu.edu.sa Outline Radioactive Decay
H 1. Nuclear Physics. Nuclear Physics. 1. Parts of Atom. A. Nuclear Structure. 2b. Nomenclature. 2. Isotopes. AstroPhysics Notes
 AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia Nuclear Physics A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions Updated: 0Feb07 Rough draft A. Nuclear Structure. Parts of Atom. Parts of
AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia Nuclear Physics A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions Updated: 0Feb07 Rough draft A. Nuclear Structure. Parts of Atom. Parts of
CHAPTER 4 Structure of the Atom
 CHAPTER 4 Structure of the Atom Fall 2018 Prof. Sergio B. Mendes 1 Topics 4.1 The Atomic Models of Thomson and Rutherford 4.2 Rutherford Scattering 4.3 The Classic Atomic Model 4.4 The Bohr Model of the
CHAPTER 4 Structure of the Atom Fall 2018 Prof. Sergio B. Mendes 1 Topics 4.1 The Atomic Models of Thomson and Rutherford 4.2 Rutherford Scattering 4.3 The Classic Atomic Model 4.4 The Bohr Model of the
Chemical Engineering 412
 Chemical Engineering 412 Introductory Nuclear Engineering Lecture 5 Nuclear Energetics 1 Spiritual Thought 2 I add my voice to these wise and inspired brethren and say to you that one of the most important
Chemical Engineering 412 Introductory Nuclear Engineering Lecture 5 Nuclear Energetics 1 Spiritual Thought 2 I add my voice to these wise and inspired brethren and say to you that one of the most important
Types of radiation resulting from radioactive decay can be summarized in a simple chart. Only X-rays, Auger electrons and internal conversion
 General information Nuclei are composed of combinations of nucleons (protons and neutrons); certain combinations of these nucleons (i.e., certain nuclides) possess a high degree of stability while others
General information Nuclei are composed of combinations of nucleons (protons and neutrons); certain combinations of these nucleons (i.e., certain nuclides) possess a high degree of stability while others
Nuclear Chemistry. Decay Reactions The most common form of nuclear decay reactions are the following:
 Nuclear Chemistry Nuclear reactions are transmutation of the one element into another. We can describe nuclear reactions in a similar manner as regular chemical reactions using ideas of stoichiometry,
Nuclear Chemistry Nuclear reactions are transmutation of the one element into another. We can describe nuclear reactions in a similar manner as regular chemical reactions using ideas of stoichiometry,
H 1. Nuclear Physics. Nuclear Physics. 1. Parts of Atom. 2. Isotopes. AstroPhysics Notes. Dr. Bill Pezzaglia. Rough draft. A.
 AstroPhysics Notes Tom Lehrer: Elements Dr. Bill Pezzaglia Nuclear Physics Updated: 0Feb Rough draft Nuclear Physics A. Nuclear Structure A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions. Parts
AstroPhysics Notes Tom Lehrer: Elements Dr. Bill Pezzaglia Nuclear Physics Updated: 0Feb Rough draft Nuclear Physics A. Nuclear Structure A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions. Parts
Radioactivity. General Physics II PHYS 111. King Saud University College of Applied Studies and Community Service Department of Natural Sciences
 King Saud University College of Applied Studies and Community Service Department of Natural Sciences Radioactivity General Physics II PHYS 111 Nouf Alkathran nalkathran@ksu.edu.sa Outline Radioactive Decay
King Saud University College of Applied Studies and Community Service Department of Natural Sciences Radioactivity General Physics II PHYS 111 Nouf Alkathran nalkathran@ksu.edu.sa Outline Radioactive Decay
NUCLEAR PHYSICS AND RADIOACTIVITY
 CHAPTER 31 NUCLEAR PHYSICS AND RADIOACTIVITY CONCEPTUAL QUESTIONS 1. REASONING AND SOLUTION Isotopes are nuclei that contain the same number of protons, but a different number of neutrons. A material is
CHAPTER 31 NUCLEAR PHYSICS AND RADIOACTIVITY CONCEPTUAL QUESTIONS 1. REASONING AND SOLUTION Isotopes are nuclei that contain the same number of protons, but a different number of neutrons. A material is
Selected Topics in Physics a lecture course for 1st year students by W.B. von Schlippe Spring Semester 2007
 Selected Topics in Physics a lecture course for 1st year students by W.B. von Schlippe Spring Semester 2007 Lecture 10 Radioactive Decay of Nuclei 1 Some naturally occurring substances have the property
Selected Topics in Physics a lecture course for 1st year students by W.B. von Schlippe Spring Semester 2007 Lecture 10 Radioactive Decay of Nuclei 1 Some naturally occurring substances have the property
(1) Correspondence of the density matrix to traditional method
 (1) Correspondence of the density matrix to traditional method New method (with the density matrix) Traditional method (from thermal physics courses) ZZ = TTTT ρρ = EE ρρ EE = dddd xx ρρ xx ii FF = UU
(1) Correspondence of the density matrix to traditional method New method (with the density matrix) Traditional method (from thermal physics courses) ZZ = TTTT ρρ = EE ρρ EE = dddd xx ρρ xx ii FF = UU
Radioisotope Tracers
 Radioisotope Tracers OCN 623 Chemical Oceanography 31 March 2016 Reading: Emerson and Hedges, Chapter 5, p.153-169 2016 Frank Sansone and David Ho Student Learning Outcomes At the completion of this module,
Radioisotope Tracers OCN 623 Chemical Oceanography 31 March 2016 Reading: Emerson and Hedges, Chapter 5, p.153-169 2016 Frank Sansone and David Ho Student Learning Outcomes At the completion of this module,
Half Life Introduction
 Name: Date: Period: Half Life Introduction The half-life of an element is the time it will take half of the parent atoms to transmutate into different atoms (through alpha or beta decays, or another process).
Name: Date: Period: Half Life Introduction The half-life of an element is the time it will take half of the parent atoms to transmutate into different atoms (through alpha or beta decays, or another process).
Introduction to Nuclear Reactor Physics
 Introduction to Nuclear Reactor Physics J. Frýbort, L. Heraltová Department of Nuclear Reactors 19 th October 2017 J. Frýbort, L. Heraltová (CTU in Prague) Introduction to Nuclear Reactor Physics 19 th
Introduction to Nuclear Reactor Physics J. Frýbort, L. Heraltová Department of Nuclear Reactors 19 th October 2017 J. Frýbort, L. Heraltová (CTU in Prague) Introduction to Nuclear Reactor Physics 19 th
Interaction with matter
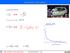 Interaction with matter accelerated motion: ss = bb 2 tt2 tt = 2 ss bb vv = vv 0 bb tt = vv 0 2 ss bb EE = 1 2 mmvv2 dddd dddd = mm vv 0 2 ss bb 1 bb eeeeeeeeeeee llllllll bbbbbbbbbbbbbb dddddddddddddddd
Interaction with matter accelerated motion: ss = bb 2 tt2 tt = 2 ss bb vv = vv 0 bb tt = vv 0 2 ss bb EE = 1 2 mmvv2 dddd dddd = mm vv 0 2 ss bb 1 bb eeeeeeeeeeee llllllll bbbbbbbbbbbbbb dddddddddddddddd
Chemistry: The Central Science. Chapter 21: Nuclear Chemistry
 Chemistry: The Central Science Chapter 21: Nuclear Chemistry A nuclear reaction involves changes in the nucleus of an atom Nuclear chemistry the study of nuclear reactions, with an emphasis in their uses
Chemistry: The Central Science Chapter 21: Nuclear Chemistry A nuclear reaction involves changes in the nucleus of an atom Nuclear chemistry the study of nuclear reactions, with an emphasis in their uses
Chapter Three (Nuclear Radiation)
 Al-Mustansiriyah University College of Science Physics Department Fourth Grade Nuclear Physics Dr. Ali A. Ridha Chapter Three (Nuclear Radiation) (3-1) Nuclear Radiation Whenever a nucleus can attain a
Al-Mustansiriyah University College of Science Physics Department Fourth Grade Nuclear Physics Dr. Ali A. Ridha Chapter Three (Nuclear Radiation) (3-1) Nuclear Radiation Whenever a nucleus can attain a
DEVIL PHYSICS THE BADDEST CLASS ON CAMPUS IB PHYSICS
 DEVIL PHYSICS THE BADDEST CLASS ON CAMPUS IB PHYSICS TSOKOS LESSON 6-6 NUCLEAR PHYSICS IB Assessment Statements Topic 13.2, Nuclear Physics 13.2.1. Explain how the radii of nuclei may be estimated from
DEVIL PHYSICS THE BADDEST CLASS ON CAMPUS IB PHYSICS TSOKOS LESSON 6-6 NUCLEAR PHYSICS IB Assessment Statements Topic 13.2, Nuclear Physics 13.2.1. Explain how the radii of nuclei may be estimated from
Physics 3204 UNIT 3 Test Matter Energy Interface
 Physics 3204 UNIT 3 Test Matter Energy Interface 2005 2006 Time: 60 minutes Total Value: 33 Marks Formulae and Constants v = f λ E = hf h f = E k + W 0 E = m c 2 p = h λ 1 A= A T 0 2 t 1 2 E k = ½ mv 2
Physics 3204 UNIT 3 Test Matter Energy Interface 2005 2006 Time: 60 minutes Total Value: 33 Marks Formulae and Constants v = f λ E = hf h f = E k + W 0 E = m c 2 p = h λ 1 A= A T 0 2 t 1 2 E k = ½ mv 2
Integrating Rational functions by the Method of Partial fraction Decomposition. Antony L. Foster
 Integrating Rational functions by the Method of Partial fraction Decomposition By Antony L. Foster At times, especially in calculus, it is necessary, it is necessary to express a fraction as the sum of
Integrating Rational functions by the Method of Partial fraction Decomposition By Antony L. Foster At times, especially in calculus, it is necessary, it is necessary to express a fraction as the sum of
Chemical Engineering 412
 Chemical Engineering 412 Introductory Nuclear Engineering Lecture 12 Radiation/Matter Interactions II 1 Neutron Flux The collisions of neutrons of all energies is given by FF = ΣΣ ii 0 EE φφ EE dddd All
Chemical Engineering 412 Introductory Nuclear Engineering Lecture 12 Radiation/Matter Interactions II 1 Neutron Flux The collisions of neutrons of all energies is given by FF = ΣΣ ii 0 EE φφ EE dddd All
Nuclear Physics. Radioactivity. # protons = # neutrons. Strong Nuclear Force. Checkpoint 4/17/2013. A Z Nucleus = Protons+ Neutrons
 Marie Curie 1867-1934 Radioactivity Spontaneous emission of radiation from the nucleus of an unstable isotope. Antoine Henri Becquerel 1852-1908 Wilhelm Roentgen 1845-1923 Nuclear Physics A Z Nucleus =
Marie Curie 1867-1934 Radioactivity Spontaneous emission of radiation from the nucleus of an unstable isotope. Antoine Henri Becquerel 1852-1908 Wilhelm Roentgen 1845-1923 Nuclear Physics A Z Nucleus =
Nuclear Science A Teacher s Guide to the Nuclear Science Wall Chart 1998 Contemporary Physics Education Project (CPEP)
 Nuclear Science A Teacher s Guide to the Nuclear Science Wall Chart 1998 Contemporary Physics Education Project (CPEP) Chapter 3 Radioactivity In radioactive processes, particles or electromagnetic radiation
Nuclear Science A Teacher s Guide to the Nuclear Science Wall Chart 1998 Contemporary Physics Education Project (CPEP) Chapter 3 Radioactivity In radioactive processes, particles or electromagnetic radiation
Lecture 10: Fission Conceptual process Fissionability Decay rate Decay branching Mass distribution Kinetic energy Neutrons
 Lecture 10: Fission Conceptual process Fissionability Decay rate Decay branching Mass distribution Kinetic energy Neutrons Lecture 10: Ohio University PHYS7501, Fall 2017, Z. Meisel (meisel@ohio.edu) Steps
Lecture 10: Fission Conceptual process Fissionability Decay rate Decay branching Mass distribution Kinetic energy Neutrons Lecture 10: Ohio University PHYS7501, Fall 2017, Z. Meisel (meisel@ohio.edu) Steps
PS-21 First Spring Institute say : Teaching Physical Science. Radioactivity
 PS-21 First Spring Institute say 2012-2013: Teaching Physical Science Radioactivity What Is Radioactivity? Radioactivity is the release of tiny, highenergy particles or gamma rays from the nucleus of an
PS-21 First Spring Institute say 2012-2013: Teaching Physical Science Radioactivity What Is Radioactivity? Radioactivity is the release of tiny, highenergy particles or gamma rays from the nucleus of an
da u g ht er + radiation
 RADIOACTIVITY The discovery of radioactivity can be attributed to several scientists. Wilhelm Roentgen discovered X-rays in 1895 and shortly after that Henri Becquerel observed radioactive behavior while
RADIOACTIVITY The discovery of radioactivity can be attributed to several scientists. Wilhelm Roentgen discovered X-rays in 1895 and shortly after that Henri Becquerel observed radioactive behavior while
NOTES: 25.2 Nuclear Stability and Radioactive Decay
 NOTES: 25.2 Nuclear Stability and Radioactive Decay Why does the nucleus stay together? STRONG NUCLEAR FORCE Short range, attractive force that acts among nuclear particles Nuclear particles attract one
NOTES: 25.2 Nuclear Stability and Radioactive Decay Why does the nucleus stay together? STRONG NUCLEAR FORCE Short range, attractive force that acts among nuclear particles Nuclear particles attract one
HOMEWORK 22-1 (pp )
 CHAPTER 22 HOMEWORK 22-1 (pp. 701 702) Define. 1. nucleons 2. nuclide 3. mass defect 4. nuclear binding energy Solve. Use masses of 1.0087 amu for the neutron, 1.00728 amu for the proton, and 5.486 x 10
CHAPTER 22 HOMEWORK 22-1 (pp. 701 702) Define. 1. nucleons 2. nuclide 3. mass defect 4. nuclear binding energy Solve. Use masses of 1.0087 amu for the neutron, 1.00728 amu for the proton, and 5.486 x 10
RADIOACTIVITY: spontaneous disintegration of the nucleus of certain atoms accompanied by the emission (release) of particles and/or energy
 RADIOACTIVITY: spontaneous disintegration of the nucleus of certain atoms accompanied by the emission (release) of particles and/or energy ~ TRANSMUTATION: the change of one element into another due to
RADIOACTIVITY: spontaneous disintegration of the nucleus of certain atoms accompanied by the emission (release) of particles and/or energy ~ TRANSMUTATION: the change of one element into another due to
Nuclear Chemistry. Nuclear Terminology
 Nuclear Chemistry Up to now, we have been concerned mainly with the electrons in the elements the nucleus has just been a positively charged things that attracts electrons The nucleus may also undergo
Nuclear Chemistry Up to now, we have been concerned mainly with the electrons in the elements the nucleus has just been a positively charged things that attracts electrons The nucleus may also undergo
Nuclear Chemistry. The Nucleus. Isotopes. Slide 1 / 43. Slide 2 / 43. Slide 3 / 43
 Slide 1 / 43 Nuclear Chemistry The Nucleus Slide 2 / 43 Remember that the nucleus is comprised of the two nucleons, protons and neutrons. The number of protons is the atomic number. The number of protons
Slide 1 / 43 Nuclear Chemistry The Nucleus Slide 2 / 43 Remember that the nucleus is comprised of the two nucleons, protons and neutrons. The number of protons is the atomic number. The number of protons
Natural Radiation K 40
 Natural Radiation There are a few radioisotopes that exist in our environment. Isotopes that were present when the earth was formed and isotopes that are continuously produced by cosmic rays can exist
Natural Radiation There are a few radioisotopes that exist in our environment. Isotopes that were present when the earth was formed and isotopes that are continuously produced by cosmic rays can exist
Lecture Presentation. Chapter 21. Nuclear Chemistry. James F. Kirby Quinnipiac University Hamden, CT Pearson Education, Inc.
 Lecture Presentation Chapter 21, Inc. James F. Kirby Quinnipiac University Hamden, CT Energy: Chemical vs. Chemical energy is associated with making and breaking chemical bonds. energy is enormous in comparison.
Lecture Presentation Chapter 21, Inc. James F. Kirby Quinnipiac University Hamden, CT Energy: Chemical vs. Chemical energy is associated with making and breaking chemical bonds. energy is enormous in comparison.
: When electrons bombarded surface of certain materials, invisible rays were emitted
 Nuclear Chemistry Nuclear Reactions 1. Occur when nuclei emit particles and/or rays. 2. Atoms are often converted into atoms of another element. 3. May involve protons, neutrons, and electrons 4. Associated
Nuclear Chemistry Nuclear Reactions 1. Occur when nuclei emit particles and/or rays. 2. Atoms are often converted into atoms of another element. 3. May involve protons, neutrons, and electrons 4. Associated
Chapter 13. I. Variables that Effect Reaction Rates. 1. Concentrations of reactants. 2. Concentration of a Catalyst. 3. Temperature.
 CHEMISTRY 112 LECTURE Chapter 13 Chemical Kinetics is the study of the rates of reactions Reaction Rate is the decrease of the concentration of reactants and the increase of the concentration of products
CHEMISTRY 112 LECTURE Chapter 13 Chemical Kinetics is the study of the rates of reactions Reaction Rate is the decrease of the concentration of reactants and the increase of the concentration of products
Time Domain Analysis of Linear Systems Ch2. University of Central Oklahoma Dr. Mohamed Bingabr
 Time Domain Analysis of Linear Systems Ch2 University of Central Oklahoma Dr. Mohamed Bingabr Outline Zero-input Response Impulse Response h(t) Convolution Zero-State Response System Stability System Response
Time Domain Analysis of Linear Systems Ch2 University of Central Oklahoma Dr. Mohamed Bingabr Outline Zero-input Response Impulse Response h(t) Convolution Zero-State Response System Stability System Response
Chapter 19 Radioactivity and Nuclear Chemistry
 Chapter 19 Radioactivity and Nuclear Chemistry The Discovery of Radioactivity! Antoine-Henri Becquerel designed an experiment to determine if phosphorescent minerals also gave off X-rays The Discovery
Chapter 19 Radioactivity and Nuclear Chemistry The Discovery of Radioactivity! Antoine-Henri Becquerel designed an experiment to determine if phosphorescent minerals also gave off X-rays The Discovery
Chapter 21 Nuclear Chemistry
 Chapter 21 Nuclear Chemistry The Nucleus Remember that the nucleus is comprised of the two nucleons, protons and neutrons. The number of protons is the atomic number. The number of protons and neutrons
Chapter 21 Nuclear Chemistry The Nucleus Remember that the nucleus is comprised of the two nucleons, protons and neutrons. The number of protons is the atomic number. The number of protons and neutrons
Nuclear Chemistry AP Chemistry Lecture Outline
 Nuclear Chemistry AP Chemistry Lecture Outline Name: involve changes with electrons. involve changes in atomic nuclei. Spontaneously-changing nuclei emit and are said to be. Radioactivity nucleons: mass
Nuclear Chemistry AP Chemistry Lecture Outline Name: involve changes with electrons. involve changes in atomic nuclei. Spontaneously-changing nuclei emit and are said to be. Radioactivity nucleons: mass
UNIT 13: NUCLEAR CHEMISTRY
 UNIT 13: NUCLEAR CHEMISTRY REVIEW: ISOTOPE NOTATION An isotope notation is written as Z A X, where X is the element, A is the mass number (sum of protons and neutrons), and Z is the atomic number. For
UNIT 13: NUCLEAR CHEMISTRY REVIEW: ISOTOPE NOTATION An isotope notation is written as Z A X, where X is the element, A is the mass number (sum of protons and neutrons), and Z is the atomic number. For
Nothing in life is to be feared. It is only to be understood. -Marie Curie. Segre Chart (Table of Nuclides)
 Nothing in life is to be feared. It is only to be understood. -Marie Curie Segre Chart (Table of Nuclides) Z N 1 Segre Chart (Table of Nuclides) Radioac8ve Decay Antoine Henri Becquerel Marie Curie, née
Nothing in life is to be feared. It is only to be understood. -Marie Curie Segre Chart (Table of Nuclides) Z N 1 Segre Chart (Table of Nuclides) Radioac8ve Decay Antoine Henri Becquerel Marie Curie, née
4 α or 4 2 He. Radioactivity. Exercise 9 Page 1. Illinois Central College CHEMISTRY 132 Laboratory Section:
 Exercise 9 Page 1 Illinois Central College CHEMISTRY 132 Laboratory Section: Radioactivity Name: Equipment Geiger Counter Alpha, Beta, and Gamma source Objectives The objectives of this experiment are
Exercise 9 Page 1 Illinois Central College CHEMISTRY 132 Laboratory Section: Radioactivity Name: Equipment Geiger Counter Alpha, Beta, and Gamma source Objectives The objectives of this experiment are
RADIOACTIVITY: spontaneous disintegration of the nucleus of certain atoms accompanied by the emission (release) of particles and/or energy
 RADIOACTIVITY: spontaneous disintegration of the nucleus of certain atoms accompanied by the emission (release) of particles and/or energy ~ TRANSMUTATION: the change of one element into another due to
RADIOACTIVITY: spontaneous disintegration of the nucleus of certain atoms accompanied by the emission (release) of particles and/or energy ~ TRANSMUTATION: the change of one element into another due to
Chemical Engineering 412
 Chemical Engineering 412 Introductory Nuclear Engineering Final Exam Review Spiritual Thought 2 Exam 3 Performance 3 Part a Part b Part c Part d Total Average 30.7 19.2 24.4 16.5 90.8 High 35.0 20.0 25.0
Chemical Engineering 412 Introductory Nuclear Engineering Final Exam Review Spiritual Thought 2 Exam 3 Performance 3 Part a Part b Part c Part d Total Average 30.7 19.2 24.4 16.5 90.8 High 35.0 20.0 25.0
Photons in the universe. Indian Institute of Technology Ropar
 Photons in the universe Photons in the universe Element production on the sun Spectral lines of hydrogen absorption spectrum absorption hydrogen gas Hydrogen emission spectrum Element production on the
Photons in the universe Photons in the universe Element production on the sun Spectral lines of hydrogen absorption spectrum absorption hydrogen gas Hydrogen emission spectrum Element production on the
Photon Interactions in Matter
 Radiation Dosimetry Attix 7 Photon Interactions in Matter Ho Kyung Kim hokyung@pusan.ac.kr Pusan National University References F. H. Attix, Introduction to Radiological Physics and Radiation Dosimetry,
Radiation Dosimetry Attix 7 Photon Interactions in Matter Ho Kyung Kim hokyung@pusan.ac.kr Pusan National University References F. H. Attix, Introduction to Radiological Physics and Radiation Dosimetry,
Atomic Notation (or Nuclear Symbol): Shorthand for keeping track of protons and neutrons in the nucleus
 Name Section CHM52LL: Nuclear Chemistry: Radioactivity, Decay, Dating, and Other Hazards There is no prelab assignment this week I. Radioactive Isotopes and Nuclear Equations Atoms are composed of three
Name Section CHM52LL: Nuclear Chemistry: Radioactivity, Decay, Dating, and Other Hazards There is no prelab assignment this week I. Radioactive Isotopes and Nuclear Equations Atoms are composed of three
Nuclear Physics Part 2A: Radioactive Decays
 Nuclear Physics Part 2A: Radioactive Decays Last modified: 23/10/2018 Links What is a Decay? Alpha Decay Definition Q-value Example Not Every Alpha Decay is Possible Beta Decay β rays are electrons Anti-particles
Nuclear Physics Part 2A: Radioactive Decays Last modified: 23/10/2018 Links What is a Decay? Alpha Decay Definition Q-value Example Not Every Alpha Decay is Possible Beta Decay β rays are electrons Anti-particles
Radioactivity. The Nobel Prize in Physics 1903 for their work on radioactivity. Henri Becquerel Pierre Curie Marie Curie
 Radioactivity Toward the end of the 19 th century, minerals were found that would darken a photographic plate even in the absence of light. This phenomenon is now called radioactivity. Marie and Pierre
Radioactivity Toward the end of the 19 th century, minerals were found that would darken a photographic plate even in the absence of light. This phenomenon is now called radioactivity. Marie and Pierre
Chemical Engineering 412
 Chemical Engineering 412 Introductory Nuclear Engineering Introduction 1 Spiritual Thought 2 The sweetest experience I know in life is to feel a prompting and act upon it and later find out that it was
Chemical Engineering 412 Introductory Nuclear Engineering Introduction 1 Spiritual Thought 2 The sweetest experience I know in life is to feel a prompting and act upon it and later find out that it was
Ch05. Radiation. Energy and matter that comes from the nucleus of an atom. version 1.6
 Ch05 Radiation Energy and matter that comes from the nucleus of an atom. version 1.6 Nick DeMello, PhD. 2007-2016 Ch05 Radiation The Discovery of Radioactivity Phosphorescence Radioactive history Antoine
Ch05 Radiation Energy and matter that comes from the nucleus of an atom. version 1.6 Nick DeMello, PhD. 2007-2016 Ch05 Radiation The Discovery of Radioactivity Phosphorescence Radioactive history Antoine
Worksheets for GCSE Mathematics. Algebraic Expressions. Mr Black 's Maths Resources for Teachers GCSE 1-9. Algebra
 Worksheets for GCSE Mathematics Algebraic Expressions Mr Black 's Maths Resources for Teachers GCSE 1-9 Algebra Algebraic Expressions Worksheets Contents Differentiated Independent Learning Worksheets
Worksheets for GCSE Mathematics Algebraic Expressions Mr Black 's Maths Resources for Teachers GCSE 1-9 Algebra Algebraic Expressions Worksheets Contents Differentiated Independent Learning Worksheets
WHAT IS IONIZING RADIATION
 WHAT IS IONIZING RADIATION Margarita Saraví National Atomic Energy Commission - Argentina Workshop on Ionizing Radiation SIM Buenos Aires 10 November 2011 What is ionizing radiation? What is ionizing radiation?
WHAT IS IONIZING RADIATION Margarita Saraví National Atomic Energy Commission - Argentina Workshop on Ionizing Radiation SIM Buenos Aires 10 November 2011 What is ionizing radiation? What is ionizing radiation?
 The previous images display some of our hopes and fears associated with nuclear radiation. We know the images, and some of the uses, but what is Nuclear Radiation and where does it come from? Nuclide In
The previous images display some of our hopes and fears associated with nuclear radiation. We know the images, and some of the uses, but what is Nuclear Radiation and where does it come from? Nuclide In
Units and Definition
 RADIATION SOURCES Units and Definition Activity (Radioactivity) Definition Activity: Rate of decay (transformation or disintegration) is described by its activity Activity = number of atoms that decay
RADIATION SOURCES Units and Definition Activity (Radioactivity) Definition Activity: Rate of decay (transformation or disintegration) is described by its activity Activity = number of atoms that decay
LECTURE 23 NUCLEI. Instructor: Kazumi Tolich
 LECTURE 23 NUCLEI Instructor: Kazumi Tolich Lecture 23 2 Reading chapter 32.1 to 32.2 Nucleus Radioactivity Mass and energy 3 The famous equation by Einstein tells us that mass is a form of energy. E =
LECTURE 23 NUCLEI Instructor: Kazumi Tolich Lecture 23 2 Reading chapter 32.1 to 32.2 Nucleus Radioactivity Mass and energy 3 The famous equation by Einstein tells us that mass is a form of energy. E =
Chemistry 52 Chapter 11 ATOMIC STRUCTURE. The general designation for an atom is shown below:
 ATOMIC STRUCTURE An atom is composed of a positive nucleus surrounded by negatively charged electrons. The nucleus is composed of protons and neutrons. The protons and neutrons in a nucleus are referred
ATOMIC STRUCTURE An atom is composed of a positive nucleus surrounded by negatively charged electrons. The nucleus is composed of protons and neutrons. The protons and neutrons in a nucleus are referred
PHYSICS OF NUCLEAR MEDICINE
 PHYSICS OF NUCLEAR MEDICINE Radioactivity A certain natural elements, heavy have unstable that disintegrate to emit various rays. Alpha(α ), Beta(β), and Gamma(γ )rays. Alpha(α ) Beta(β) Gamma(γ ) 1-Positive
PHYSICS OF NUCLEAR MEDICINE Radioactivity A certain natural elements, heavy have unstable that disintegrate to emit various rays. Alpha(α ), Beta(β), and Gamma(γ )rays. Alpha(α ) Beta(β) Gamma(γ ) 1-Positive
Quantum Mechanics. An essential theory to understand properties of matter and light. Chemical Electronic Magnetic Thermal Optical Etc.
 Quantum Mechanics An essential theory to understand properties of matter and light. Chemical Electronic Magnetic Thermal Optical Etc. Fall 2018 Prof. Sergio B. Mendes 1 CHAPTER 3 Experimental Basis of
Quantum Mechanics An essential theory to understand properties of matter and light. Chemical Electronic Magnetic Thermal Optical Etc. Fall 2018 Prof. Sergio B. Mendes 1 CHAPTER 3 Experimental Basis of
10.4 Half-Life. Investigation. 290 Unit C Radioactivity
 .4 Half-Life Figure Pitchblende, the major uranium ore, is a heavy mineral that contains uranium oxides, lead, and trace amounts of other radioactive elements. Pierre and Marie Curie found radium and polonium
.4 Half-Life Figure Pitchblende, the major uranium ore, is a heavy mineral that contains uranium oxides, lead, and trace amounts of other radioactive elements. Pierre and Marie Curie found radium and polonium
Radioactivity. Nuclear Physics. # neutrons vs# protons Where does the energy released in the nuclear 11/29/2010 A=N+Z. Nuclear Binding, Radioactivity
 Physics 1161: Lecture 25 Nuclear Binding, Radioactivity Sections 32-1 32-9 Marie Curie 1867-1934 Radioactivity Spontaneous emission of radiation from the nucleus of an unstable isotope. Antoine Henri Becquerel
Physics 1161: Lecture 25 Nuclear Binding, Radioactivity Sections 32-1 32-9 Marie Curie 1867-1934 Radioactivity Spontaneous emission of radiation from the nucleus of an unstable isotope. Antoine Henri Becquerel
The number of protons in the nucleus is known as the atomic number Z, and determines the chemical properties of the element.
 I. NUCLEAR PHYSICS I.1 Atomic Nucleus Very briefly, an atom is formed by a nucleus made up of nucleons (neutrons and protons) and electrons in external orbits. The number of electrons and protons is equal
I. NUCLEAR PHYSICS I.1 Atomic Nucleus Very briefly, an atom is formed by a nucleus made up of nucleons (neutrons and protons) and electrons in external orbits. The number of electrons and protons is equal
CHAPTER 25: NUCLEAR CHEMISTRY. Mrs. Brayfield
 CHAPTER 25: NUCLEAR CHEMISTRY Mrs. Brayfield CHEMICAL VS. NUCLEAR When you hear the word nuclear, what do you think of? What makes nuclear reactions different from chemical reactions? The speed of nuclear
CHAPTER 25: NUCLEAR CHEMISTRY Mrs. Brayfield CHEMICAL VS. NUCLEAR When you hear the word nuclear, what do you think of? What makes nuclear reactions different from chemical reactions? The speed of nuclear
ZX or X-A where X is chemical symbol of element. common unit: [unified mass unit = u] also known as [atomic mass unit = amu] or [Dalton = Da]
![ZX or X-A where X is chemical symbol of element. common unit: [unified mass unit = u] also known as [atomic mass unit = amu] or [Dalton = Da] ZX or X-A where X is chemical symbol of element. common unit: [unified mass unit = u] also known as [atomic mass unit = amu] or [Dalton = Da]](/thumbs/87/95084584.jpg) 1 Part 5: Nuclear Physics 5.1. The Nucleus = atomic number = number of protons N = neutron number = number of neutrons = mass number = + N Representations: X or X- where X is chemical symbol of element
1 Part 5: Nuclear Physics 5.1. The Nucleus = atomic number = number of protons N = neutron number = number of neutrons = mass number = + N Representations: X or X- where X is chemical symbol of element
