CHM The Heat of Reaction (r14) Charles Taylor 1/6
|
|
|
- Diane McKinney
- 5 years ago
- Views:
Transcription
1 CHM The Heat of Reaction (r14) Charles Taylor 1/6 Introduction We've discussed heat briefly, and we've stated that the heat is an energy flow between systems that differ in temperature. Since we're chemists, though, we'd like to relate all this talk about heat flows, systems, and surroundings to what we're interested in - chemical reactions. This has important practical considerations. Heat is transferred in nearly all chemical reactions, and you have to take these into account when performing reactions. Sometimes this heat is desirable - you'd have a tough time cooking your food on a gas stove if the combustion process wasn't exothermic! In other cases, the heat is a serious safety concern. The reactions used to make plastics, for example, are often extremely exothermic. These reactions, if uncontrolled, can proceed very quickly and lead to explosions. (Consider what a large temperature increase can do to the pressure in a sealed vessel!) We will discuss what the heat of reaction is and how we can both measure (and predict) this quantity. We will then use it to calculate how much heat is transferred in a real chemical reaction where we have a given amount of reagent. Heat of reaction The heat of reaction is defined as the amount of energy required to bring a system back to its original temperature after a chemical reaction. Let's say we have a chemical reaction in a beaker. The reaction causes the temperature inside the beaker to drop. Illustration 1: Temperature change in a chemical reaction A, B, and C are all present in aqueous solution in this example. Why does the temperature in the beaker drop? What is removing energy from the contents of the beaker? Define the system as the substances involved in the chemical reaction, and the surroundings as the beaker of water. In the above illustration, the beaker's contents are cooled. But by what? The only apparent source of the cooling would be the reaction of A and B to form C. The formation of C is an endothermic process (and the reaction is said to be an endothermic reaction) - C has more internal energy than the starting materials. Energy flows from the water in the beaker to the reacting substances. This loss of energy from the water causes its temperature of the water to drop. The reacting substances accept this flow of energy and use it to complete the chemical reaction. Since energy flows into the system (the reaction), the measured Q (the heat of reaction) for this system it would be positive.
2 CHM The Heat of Reaction (r14) Charles Taylor 2/6 How you'd report a heat of reaction to others The energy absorbed or given off by a chemical reaction depends on the amount of reactants present. The more moles of chemicals react, the greater the heat transfer (in either direction). So it's not terribly useful to say something like "I did a reaction between A and B to get C and the heat of reaction (the Q) was kj." You've told your listener that the reaction was endothermic (because Q was positive), but you really haven't said much else. The number you've given is meaningless without some description of how much C you actually made, or how much A and B were reacted. Because of this, heat of reaction is typically expressed as tenergy units per mole of a substance involved in the reaction. We don't normally report values of Q for chemical reactions. Instead, we report what is called the enthalpy change of reaction (often just called enthalpy of reaction). Without going too deeply into the math and language of thermodynamics, we will say that there are several reasons that we express reaction heats in terms of this enthalpy. 1. Enthalpy is a mathematical construct, abbreviated as H. You can think of it as measuring the most efficient path from products to reactants. Consider the energy content of the products and the energy content of the reactants as two locations in South Carolina - say, Florence and Columbia. If you drive a Honda Civic to Columbia from here and someone else drive a Jeep Cherokee, you'll expend different amounts of energy, but you'll both arrive in the same place. You can think of the enthalpy as the idealized energy change between products and reactants. 2. Enthalpy is constructed so that the enthalpy change for a reaction it is equal to the heat change at constant pressure. Since we normally do chemical reactions in our lab at these conditions anyway (in a beaker on the desk, for example), enthalpy change is easy to measure. Someone who's taken a thermodynamics class or two can take that enthalpy change and use it to deal with other conditions. 3. Enthalpy is related to internal energy, which is the concept we discussed earlier. It is defined as H = U + PV, where P and V are pressure and volume. 4. Enthalpy is a state function. This means that the enthalpy content of a substance at a given set of conditions is the same no matter how the substance got to those conditions. The amount of heat you measure from a reaction may depend on how you performed the reaction - since energy takes forms other than heat, but the enthalpy change is always the same.
3 CHM The Heat of Reaction (r14) Charles Taylor 3/6 Enthalpy is a property of a substance. Heat, however, is an energy transfer. The heat of a reaction, then, is related to the enthalpy change between the products and reactants. You'd calculate this enthalpy change like this: Q reaction Δ H = r Number of moles where Δ H r is the enthalpy change of the reaction (abbreviated as the enthalpy of reaction, and often just called the heat of reaction) and Q reaction is the heat change of the reaction measured at constant pressure. We will measure the enthalpy change of some reactions in the lab. The thermochemical equation: Another way to express reaction heats Another useful (and unambiguous) way to express a heat of reaction is by the use of a thermochemical equation. A thermochemical equation is just a chemical equation with added heat data. For example, let's look at the chemical equation for the combustion of graphite (a common form of carbon): C(s) + O 2 (g) CO 2 (g) This is the chemical equation. A thermochemical equation contains the same information plus the heat of reaction as the reaction is written. For the combustion of graphite: C(s) + O 2 (g) CO 2 (g) ; Δ H= kj Some important things about the thermochemical equation are: The enthalpy change (or heat) given is for the equation as written. In the example, kj of heat are released if 1 mole C solid reacts with 1 mole O 2 gas to form 1 mole CO 2 gas. Phase labels are required, because phase changes involve energy. For example, production of liquid water involves a different amount of heat than the production of gaseous water or solid water. The same is true for any other substance. You can modify thermochemical equations much like you'd modify chemical equations. There are two important modifications you should be familiar with. 1. You can multiply a thermochemical equation by a factor. When you do so, you also multiply the enthalpy by the same factor. It's like the heat is simply another product of the reaction. For example, what if you were interested in the heat given off by the burning of two
4 CHM The Heat of Reaction (r14) Charles Taylor 4/6 moles of graphite? Well, you'd just multiply the reaction by two, just like we you scaled up chemical "recipes" (equations) earlier. becomes C(s) + O 2 (g) CO 2 (g) ; Δ H= kj 2 C(s) + 2 O 2 (g) 2 CO 2 (g) ; Δ H= kj So, two moles of graphite releases kj of heat when it is burned. 2. You can reverse a chemical reaction by switching products and reactants. If you do that, you must change the sign on the enthalpy. This should make logical sense. If a combustion, for example, releases energy, you would have to re-add that energy to get the original materials back. For example, let's see how much energy would be required to convert a mole of carbon dioxide back into carbon and oxygen.. We know: Reversing, we get C(s) + O 2 (g) CO 2 (g) ; Δ H= kj CO 2 (g) C(s) + O 2 (g) ; Δ H= kj So, it would require kj of energy to split carbon dioxide into graphite and molecular oxygen. (Though this reaction is theoretically possible with the right application of energy, this isn't a very practical way to get graphite.) Application: Heat production or requirement in real reactions As we said in the beginning, these heats of reaction (or enthalpies of reaction) are useful to us because they provide practical information like how much heat we need to supply to make a reaction go or how much heat we can expect a reaction to produce for us. It shouldn't be a surprise to you that we can use these heats to simply calculate how much heat is produced when a given amount of reagents reacts, or to calculate how much of a reagent is necessary to supply a required amount of heat. For example, let's go back to burning graphite. How much heat is produced when grams of carbon are burned in the presence of oxygen? The thermochemical equation is: C(s) + O 2 (g) CO 2 (g) ; Δ H= 393.5kJ This is a stoichiometry problem, You can treat the heat just like it is another product. To solve it, first we calculate the number of moles of carbon, then we relate the moles of carbon to kilojoules of heat using the thermochemical equation. This is similar to finding the moles of product produced in a simple stoichiometry problem.
5 CHM The Heat of Reaction (r14) Charles Taylor 5/6 Find the moles carbon: 2.524g C 1 mol C = mol C gc Now, find the amount of heat using the thermochemical equation. As long as the reaction takes place at constant pressure, the heat production will be the same as the enthalpy change: mol C kj = kj 1 mol C We've calculated the value of Q for this reaction. Q is negative, so the reaction is exothermic (it gives off heat, just as we'd expect from a combustion). To put it in plain English, kj of heat is released by the burning of g of graphite. Let's look at another problem, this time trying to figure out how much of a material it takes to generate a desired amount of heat. How many kilograms of benzene (C 6 H 6, FW = g/mol) must be burned to produce kj of heat? [Calculate your answer to four significant figures.] 2 C 6 H 6 (l) + 15 O 2 (g) --> 12 CO 2 (g) + 6 H 2 O(l) ; Δ H= kj We have an amount of heat we'd like to produce this time and are asked how much reactant we need to consume to produce it. This problem is worked just like you'd work a stoichiometry problem where you were asked how much of a reactant you'd need to produce an amount of a desired product. Again, the heat is just like another product! First, we calculate the number of moles benzene required to produce the given heat, assuming the reaction takes place at constant pressure. We use the thermochemical equation to get the conversion factor. Then we calculate the mass of benzene required from the formula weight. "Producing" heat means that the sign of Q is negative, so... 2 mol C H kj kj =45.91 mol C 6 H 6 required Now, we calculate the mass (remember, the problem specifies kilograms gC H mol C H kg 6 6 mol C H =3.586 kg C H 6 6 g
6 CHM The Heat of Reaction (r14) Charles Taylor 6/6 So, you must burn kg C 6 H 6 to produce kj of heat. Summary In this note pack, we've learned the definition of the heat of reaction, the notion of a thermochemical equation, and how to use the heat of reaction and thermochemical equations to find out information about heat transfer during real chemical reactions. In the next pack, we will talk about Hess's Law, a way to determine the heat of reaction without doing an experiment. We will also discuss how heat changes are measured.
Assuming that no heat is lost from the water to the surrounding air, Conservation of energy. The terms add to zero because they have opposite signs.
 CALORIMETRY - the measurement of heat. How do we measure heat flow? 0.20 mol A 100 g water A -> B + C When we add the reactant to water, it decomposes - heating the water. 100 g water A -> B + C... what
CALORIMETRY - the measurement of heat. How do we measure heat flow? 0.20 mol A 100 g water A -> B + C When we add the reactant to water, it decomposes - heating the water. 100 g water A -> B + C... what
How many ml of 0.250M potassium permangenate are needed to react with 3.36 g of iron(ii) sulfate?
 115 How many ml of 0.250M potassium permangenate are needed to react with 3.36 g of iron(ii) sulfate? 1) Convert 3.36 grams iron(ii) sulfate to moles. Use FORMULA WEIGHT. 2) Convert moles iron(ii) sulfate
115 How many ml of 0.250M potassium permangenate are needed to react with 3.36 g of iron(ii) sulfate? 1) Convert 3.36 grams iron(ii) sulfate to moles. Use FORMULA WEIGHT. 2) Convert moles iron(ii) sulfate
128 One problem... PATH. The amount of energy required for a process depends on how the process is carried out.
 128 One problem... PATH. The amount of energy required for a process depends on how the process is carried out. Example: Driving from Florence to Columbia. How much energy is required? (gas) Jeep Cherokee
128 One problem... PATH. The amount of energy required for a process depends on how the process is carried out. Example: Driving from Florence to Columbia. How much energy is required? (gas) Jeep Cherokee
- Joule (J): SI unit for energy. It's defined based on the equation for kinetic energy. from. mass. velocity
 153 ENERGY UNITS - calorie (cal): the amount of energy required to change the temperature of one gram of water by one degree Celsius (or Kelvin) 1g 1g add one calorie of energy - Calories in food? The
153 ENERGY UNITS - calorie (cal): the amount of energy required to change the temperature of one gram of water by one degree Celsius (or Kelvin) 1g 1g add one calorie of energy - Calories in food? The
This reaction is ENDOTHERMIC. Energy is being transferred from the room/flask/etc. (the SURROUNDINGS) to the reaction itself (the SYSTEM).
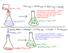 151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
- Kinetic energy: energy of matter in motion. gravity
 148 2500 L of chlorine gas at 25.0 C and 1.00 atm are used to make hydrochloric acid. How many grams of hydrochloric acid could be produced if all the chlorine reacts? 1 - Convert 2500 L chlorine gas to
148 2500 L of chlorine gas at 25.0 C and 1.00 atm are used to make hydrochloric acid. How many grams of hydrochloric acid could be produced if all the chlorine reacts? 1 - Convert 2500 L chlorine gas to
- The empirical gas laws (including the ideal gas equation) do not always apply.
 145 At 300 C, ammonium nitrate violently decomposes to produce nitrogen gas, oxygen gas, and water vapor. What is the total volume of gas that would be produced at 1.00 atm by the decomposition of 15.0
145 At 300 C, ammonium nitrate violently decomposes to produce nitrogen gas, oxygen gas, and water vapor. What is the total volume of gas that would be produced at 1.00 atm by the decomposition of 15.0
11B, 11E Temperature and heat are related but not identical.
 Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Measuring and Expressing Enthalpy Changes. Copyright Pearson Prentice Hall. Measuring and Expressing Enthalpy Changes. Calorimetry
 Measuring and Expressing Enthalpy Changes A burning match releases heat to its surroundings in all directions. How much heat does this exothermic reaction release? You will learn to measure heat flow in
Measuring and Expressing Enthalpy Changes A burning match releases heat to its surroundings in all directions. How much heat does this exothermic reaction release? You will learn to measure heat flow in
= (25.0 g)(0.137 J/g C)[61.2 C - (-31.4 C)] = 317 J (= kj)
![= (25.0 g)(0.137 J/g C)[61.2 C - (-31.4 C)] = 317 J (= kj) = (25.0 g)(0.137 J/g C)[61.2 C - (-31.4 C)] = 317 J (= kj)](/thumbs/89/101004910.jpg) CHEM 101A ARMSTRONG SOLUTIONS TO TOPIC D PROBLEMS 1) For all problems involving energy, you may give your answer in either joules or kilojoules, unless the problem specifies a unit. (In general, though,
CHEM 101A ARMSTRONG SOLUTIONS TO TOPIC D PROBLEMS 1) For all problems involving energy, you may give your answer in either joules or kilojoules, unless the problem specifies a unit. (In general, though,
ENTHALPY, INTERNAL ENERGY, AND CHEMICAL REACTIONS: AN OUTLINE FOR CHEM 101A
 ENTHALPY, INTERNAL ENERGY, AND CHEMICAL REACTIONS: AN OUTLINE FOR CHEM 101A PART 1: KEY TERMS AND SYMBOLS IN THERMOCHEMISTRY System and surroundings When we talk about any kind of change, such as a chemical
ENTHALPY, INTERNAL ENERGY, AND CHEMICAL REACTIONS: AN OUTLINE FOR CHEM 101A PART 1: KEY TERMS AND SYMBOLS IN THERMOCHEMISTRY System and surroundings When we talk about any kind of change, such as a chemical
Calculate the mass of L of oxygen gas at 25.0 C and 1.18 atm pressure.
 148 Calculate the mass of 22650 L of oxygen gas at 25.0 C and 1.18 atm pressure. 1 - Convert the volume of oxygen gas to moles using IDEAL GAS EQUATION 2 - Convert moles oxygen gas to mass using formula
148 Calculate the mass of 22650 L of oxygen gas at 25.0 C and 1.18 atm pressure. 1 - Convert the volume of oxygen gas to moles using IDEAL GAS EQUATION 2 - Convert moles oxygen gas to mass using formula
CHM Units and Dimensional Analysis (r14) Charles Taylor 1/6
 CHM 110 - Units and Dimensional Analysis (r14) - 2014 Charles Taylor 1/6 Introduction Units are critical in all measurements. If you don't tell someone a unit when you tell them a number, they're not likely
CHM 110 - Units and Dimensional Analysis (r14) - 2014 Charles Taylor 1/6 Introduction Units are critical in all measurements. If you don't tell someone a unit when you tell them a number, they're not likely
17.2 Thermochemical Equations
 17.2. Thermochemical Equations www.ck12.org 17.2 Thermochemical Equations Lesson Objectives Define enthalpy, and know the conditions under which the enthalpy change in a reaction is equal to the heat absorbed
17.2. Thermochemical Equations www.ck12.org 17.2 Thermochemical Equations Lesson Objectives Define enthalpy, and know the conditions under which the enthalpy change in a reaction is equal to the heat absorbed
- Why are phase labels required? Because phase changes either absorb or release energy. ... what does this mean?
 155 SINCE the enthalpy change does NOT depend on path, this means that we can use standard values for enthalpy to predict the heat change in reactions that we have not tested in a calorimeter. THERMOCHEMICAL
155 SINCE the enthalpy change does NOT depend on path, this means that we can use standard values for enthalpy to predict the heat change in reactions that we have not tested in a calorimeter. THERMOCHEMICAL
Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions
 Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions Terms, definitions, topics Joule, calorie (Re-read p 57-58) Thermochemistry Exothermic reaction Endothermic reaction
Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions Terms, definitions, topics Joule, calorie (Re-read p 57-58) Thermochemistry Exothermic reaction Endothermic reaction
Basic Concepts of Chemistry Notes for Students [Chapter 7, page 1] D J Weinkauff - Nerinx Hall High School
![Basic Concepts of Chemistry Notes for Students [Chapter 7, page 1] D J Weinkauff - Nerinx Hall High School Basic Concepts of Chemistry Notes for Students [Chapter 7, page 1] D J Weinkauff - Nerinx Hall High School](/thumbs/77/75754481.jpg) Basic Concepts of Chemistry Notes for Students [Chapter 7, page 1] Chapter 7 Quantitative Relationships in Chemical Reactions As you have begun to see, chemistry is a quantitative science involving a great
Basic Concepts of Chemistry Notes for Students [Chapter 7, page 1] Chapter 7 Quantitative Relationships in Chemical Reactions As you have begun to see, chemistry is a quantitative science involving a great
17.4 Calculating Heats Essential Understanding Heats of reaction can be calculated when it is difficult or
 17.4 Calculating Heats of Reaction Essential Understanding Heats of reaction can be calculated when it is difficult or impossible to measure them directly. Lesson Summary Hess s Law Hess s law provides
17.4 Calculating Heats of Reaction Essential Understanding Heats of reaction can be calculated when it is difficult or impossible to measure them directly. Lesson Summary Hess s Law Hess s law provides
Chemistry Slide 1 of 33
 Chemistry 17.2 1 of 33 17.2 Measuring and Expressing Enthalpy Changes A burning match releases heat to its surroundings in all directions. How much heat does this exothermic reaction release? You will
Chemistry 17.2 1 of 33 17.2 Measuring and Expressing Enthalpy Changes A burning match releases heat to its surroundings in all directions. How much heat does this exothermic reaction release? You will
The Nature of Energy. Chapter Six: Kinetic vs. Potential Energy. Energy and Work. Temperature vs. Heat
 The Nature of Energy Chapter Six: THERMOCHEMISTRY Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationship between chemical reactions and energy changes
The Nature of Energy Chapter Six: THERMOCHEMISTRY Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationship between chemical reactions and energy changes
CHM 111 An introduction to kinetics (r15) 2015 Charles Taylor 1/7
 CHM 111 An introduction to kinetics (r15) 2015 Charles Taylor 1/7 Introduction We've talked about chemical reactions many, many times in the past. Up to this point, we've focused on the ingredients you
CHM 111 An introduction to kinetics (r15) 2015 Charles Taylor 1/7 Introduction We've talked about chemical reactions many, many times in the past. Up to this point, we've focused on the ingredients you
Name Class Date. As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings.
 Name Class Date Thermochemistry 17.1 The Flow of Energy As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings. Process Cause Effect endothermic
Name Class Date Thermochemistry 17.1 The Flow of Energy As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings. Process Cause Effect endothermic
Energetics. Topic
 Energetics Topic 5.1 5.2 Topic 5.1 Exothermic and Endothermic Reactions?? total energy of the universe is a constant if a system loses energy, it must be gained by the surroundings, and vice versa Enthalpy
Energetics Topic 5.1 5.2 Topic 5.1 Exothermic and Endothermic Reactions?? total energy of the universe is a constant if a system loses energy, it must be gained by the surroundings, and vice versa Enthalpy
Chapter 5. Thermochemistry
 Chapter 5 Thermochemistry Energy Thermodynamics Study of the relationship between heat, work, and other forms of energy Thermochemistry A branch of thermodynamics Focuses on the study of heat given off
Chapter 5 Thermochemistry Energy Thermodynamics Study of the relationship between heat, work, and other forms of energy Thermochemistry A branch of thermodynamics Focuses on the study of heat given off
Energetics. These processes involve energy exchanges between the reacting system and its surroundings.
 Energetics Chemical reactions involve: the breaking of bonds between atoms the making of new bonds between atoms These processes involve energy exchanges between the reacting system and its surroundings.
Energetics Chemical reactions involve: the breaking of bonds between atoms the making of new bonds between atoms These processes involve energy exchanges between the reacting system and its surroundings.
Exothermic process is any process that gives off heat transfers thermal energy from the system to the surroundings. H 2 O (l) + energy
 Exothermic process is any process that gives off heat transfers thermal energy from the system to the surroundings. H 2 O (g) H 2 O (l) + energy Endothermic process is any process in which heat has to
Exothermic process is any process that gives off heat transfers thermal energy from the system to the surroundings. H 2 O (g) H 2 O (l) + energy Endothermic process is any process in which heat has to
Name Date Class THERMOCHEMISTRY
 Name Date Class 17 THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY HEAT AND WORK (pages 505 510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
Name Date Class 17 THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY HEAT AND WORK (pages 505 510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
UNIT ONE BOOKLET 6. Thermodynamic
 DUNCANRIG SECONDARY ADVANCED HIGHER CHEMISTRY UNIT ONE BOOKLET 6 Thermodynamic Can we predict if a reaction will occur? What determines whether a reaction will be feasible or not? This is a question that
DUNCANRIG SECONDARY ADVANCED HIGHER CHEMISTRY UNIT ONE BOOKLET 6 Thermodynamic Can we predict if a reaction will occur? What determines whether a reaction will be feasible or not? This is a question that
Gravity is a force which keeps us stuck to the earth. The Electrostatic force attracts electrons to protons in an atom.
 Energy Relations in Chemistry: Thermochemistry The Nature of Energy Sugar you eat is "combusted" by your body to produce CO 2 and H 2 O. During this process energy is also released. This energy is used
Energy Relations in Chemistry: Thermochemistry The Nature of Energy Sugar you eat is "combusted" by your body to produce CO 2 and H 2 O. During this process energy is also released. This energy is used
MITOCW ocw-5_60-s08-lec07_300k
 MITOCW ocw-5_60-s08-lec07_300k The following content is provided under a Creative Commons license. Your support will help MIT OpenCourseWare continue to offer high-quality educational resources for free.
MITOCW ocw-5_60-s08-lec07_300k The following content is provided under a Creative Commons license. Your support will help MIT OpenCourseWare continue to offer high-quality educational resources for free.
THERMOCHEMISTRY -1. Dr. Sapna Gupta
 THERMOCHEMISTRY -1 Dr. Sapna Gupta THERMODYNAMICS Thermodynamics: Relationship between heat and other forms of energy Thermochemistry: Study of heat absorbed or evolved by chemical reactions. Energy: Capacity
THERMOCHEMISTRY -1 Dr. Sapna Gupta THERMODYNAMICS Thermodynamics: Relationship between heat and other forms of energy Thermochemistry: Study of heat absorbed or evolved by chemical reactions. Energy: Capacity
Thermochemistry-Part 1
 Brad Collins Thermochemistry-Part 1 Chapter 7 Thermochemistry Thermodynamics: The study of energy Thermochemistry: The study of energy in chemical reactions Energy: The capacity to do work Work = force
Brad Collins Thermochemistry-Part 1 Chapter 7 Thermochemistry Thermodynamics: The study of energy Thermochemistry: The study of energy in chemical reactions Energy: The capacity to do work Work = force
Chemistry Chapter 16. Reaction Energy
 Chemistry Reaction Energy Section 16.1.I Thermochemistry Objectives Define temperature and state the units in which it is measured. Define heat and state its units. Perform specific-heat calculations.
Chemistry Reaction Energy Section 16.1.I Thermochemistry Objectives Define temperature and state the units in which it is measured. Define heat and state its units. Perform specific-heat calculations.
Chemical Reactions and Stoichiometry. Ms. Grobsky
 Chemical Reactions and Stoichiometry Ms. Grobsky Wrapping Up the Types of Chemical Reactions We just got done investigating the different types of chemical reactions We can now answer the two questions
Chemical Reactions and Stoichiometry Ms. Grobsky Wrapping Up the Types of Chemical Reactions We just got done investigating the different types of chemical reactions We can now answer the two questions
Lecture Presentation. Chapter 5. Thermochemistry. John D. Bookstaver St. Charles Community College Cottleville, MO
 Lecture Presentation Chapter 5 Thermochemistry John D. Bookstaver St. Charles Community College Cottleville, MO Thermochemistry Thermodynamics is the study of energy and its transformations. Thermochemistry
Lecture Presentation Chapter 5 Thermochemistry John D. Bookstaver St. Charles Community College Cottleville, MO Thermochemistry Thermodynamics is the study of energy and its transformations. Thermochemistry
Quantities in Chemical Reactions
 Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Unit 2: Chemical Kinetics Chemistry 30
 Practice Questions Section 3.2 Factors Influencing Reaction Rate - Activation Energy 1. Answer the following questions based on the potential energy diagram shown here: a. Does the graph represent an endothermic
Practice Questions Section 3.2 Factors Influencing Reaction Rate - Activation Energy 1. Answer the following questions based on the potential energy diagram shown here: a. Does the graph represent an endothermic
Ch. 6 Enthalpy Changes
 Ch. 6 Enthalpy Changes Energy: The capacity to do work. In Physics, there are 2 main types of energy Kinetic (energy of motion) = ½ mv 2 Potential (energy of position due to gravity)= mgh In Chemistry,
Ch. 6 Enthalpy Changes Energy: The capacity to do work. In Physics, there are 2 main types of energy Kinetic (energy of motion) = ½ mv 2 Potential (energy of position due to gravity)= mgh In Chemistry,
Enthalpy. Enthalpy. Enthalpy. Enthalpy. E = q + w. Internal Energy at Constant Volume SYSTEM. heat transfer in (endothermic), +q
 heat transfer in (endothermic), +q heat transfer out (exothermic), -q SYSTEM E = q + w w transfer in (+w) w transfer out (-w) Internal Energy at Constant Volume E = KE + PE ΔE = q + w Because most systems,
heat transfer in (endothermic), +q heat transfer out (exothermic), -q SYSTEM E = q + w w transfer in (+w) w transfer out (-w) Internal Energy at Constant Volume E = KE + PE ΔE = q + w Because most systems,
Name Chem 161, Section: Group Number: ALE 27. Hess s Law. (Reference: Chapter 6 - Silberberg 5 th edition)
 Name Chem 161, Section: Group Number: ALE 27. Hess s Law (Reference: Chapter 6 - Silberberg 5 th edition) Important!! For answers that involve a calculation you must show your work neatly using dimensional
Name Chem 161, Section: Group Number: ALE 27. Hess s Law (Reference: Chapter 6 - Silberberg 5 th edition) Important!! For answers that involve a calculation you must show your work neatly using dimensional
Section 9: Thermodynamics and Energy
 Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
Chapter 8. Thermochemistry 강의개요. 8.1 Principles of Heat Flow. 2) Magnitude of Heat Flow. 1) State Properties. Basic concepts : study of heat flow
 강의개요 Basic concepts : study of heat flow Chapter 8 Thermochemistry Calorimetry : experimental measurement of the magnitude and direction of heat flow Thermochemical Equations Copyright 2005 연세대학교이학계열일반화학및실험
강의개요 Basic concepts : study of heat flow Chapter 8 Thermochemistry Calorimetry : experimental measurement of the magnitude and direction of heat flow Thermochemical Equations Copyright 2005 연세대학교이학계열일반화학및실험
Quantitative Relationships in Chemical Reactions Chapter 7
 Quantitative Relationships in Chemical Reactions Chapter 7 The burning of charcoal releases heat (thermal energy) that grills our food. But the combustion of charcoal and fossil fuels also releases CO
Quantitative Relationships in Chemical Reactions Chapter 7 The burning of charcoal releases heat (thermal energy) that grills our food. But the combustion of charcoal and fossil fuels also releases CO
Lecture Outline. 5.1 The Nature of Energy. Kinetic Energy and Potential Energy. 1 mv
 Chapter 5. Thermochemistry Common Student Misconceptions Students confuse power and energy. Students confuse heat with temperature. Students fail to note that the first law of thermodynamics is the law
Chapter 5. Thermochemistry Common Student Misconceptions Students confuse power and energy. Students confuse heat with temperature. Students fail to note that the first law of thermodynamics is the law
Chapter 6: Thermochemistry
 Chapter 6: Thermochemistry Section 6.1: Introduction to Thermochemistry Thermochemistry refers to the study of heat flow or heat energy in a chemical reaction. In a study of Thermochemistry the chemical
Chapter 6: Thermochemistry Section 6.1: Introduction to Thermochemistry Thermochemistry refers to the study of heat flow or heat energy in a chemical reaction. In a study of Thermochemistry the chemical
Thermochemistry. Using Heats of Reaction - Hess s Law - Standard Enthalpies of Formation - Fuels Foods, Commercial Fuels, and Rocket Fuels
 Thermochemistry Understanding Heats of Reaction - Energy and Its Units - Heat of Reaction - Enthalpy and Enthalpy Change - Thermochemical Equations - Applying Stoichiometry to Heats of Reaction - Measuring
Thermochemistry Understanding Heats of Reaction - Energy and Its Units - Heat of Reaction - Enthalpy and Enthalpy Change - Thermochemical Equations - Applying Stoichiometry to Heats of Reaction - Measuring
Quantities in Chemical Reactions
 Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Enthalpy Chapter 5.3-4,7
 Enthalpy Chapter 5.3-4,7 heat transfer in (endothermic), +q heat transfer out (exothermic), -q SYSTEM E = q + w w transfer in (+w) w transfer out (-w) Internal Energy at Constant Volume E = E K + E P ΔE
Enthalpy Chapter 5.3-4,7 heat transfer in (endothermic), +q heat transfer out (exothermic), -q SYSTEM E = q + w w transfer in (+w) w transfer out (-w) Internal Energy at Constant Volume E = E K + E P ΔE
Chapter 16 Theories of Energy Changes
 {Read p. 624 and 626 to understand concepts} Class discussion for chapter 17.3 Chapter 16 Theories of Energy Changes Section 16.1A Temperature change and Heat THERMODYNAMICS - the study of energy and energy
{Read p. 624 and 626 to understand concepts} Class discussion for chapter 17.3 Chapter 16 Theories of Energy Changes Section 16.1A Temperature change and Heat THERMODYNAMICS - the study of energy and energy
Thermochemistry. Energy. 1st Law of Thermodynamics. Enthalpy / Calorimetry. Enthalpy of Formation
 THERMOCHEMISTRY Thermochemistry Energy 1st Law of Thermodynamics Enthalpy / Calorimetry Hess' Law Enthalpy of Formation The Nature of Energy Kinetic Energy and Potential Energy Kinetic energy is the energy
THERMOCHEMISTRY Thermochemistry Energy 1st Law of Thermodynamics Enthalpy / Calorimetry Hess' Law Enthalpy of Formation The Nature of Energy Kinetic Energy and Potential Energy Kinetic energy is the energy
Enthalpy and Internal Energy
 Enthalpy and Internal Energy H or ΔH is used to symbolize enthalpy. The mathematical expression of the First Law of Thermodynamics is: ΔE = q + w, where ΔE is the change in internal energy, q is heat and
Enthalpy and Internal Energy H or ΔH is used to symbolize enthalpy. The mathematical expression of the First Law of Thermodynamics is: ΔE = q + w, where ΔE is the change in internal energy, q is heat and
Thermochemistry Enthalpy & Hess Law. Packet #35
 Thermochemistry Enthalpy & Hess Law Packet #35 Introduction I Thermochemistry, is the branch of chemistry that, investigates the amount of energy that is gained or lost during a chemical reaction. Introduction
Thermochemistry Enthalpy & Hess Law Packet #35 Introduction I Thermochemistry, is the branch of chemistry that, investigates the amount of energy that is gained or lost during a chemical reaction. Introduction
CHEM 1105 S10 March 11 & 14, 2014
 CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
Limiting Reactants. In other words once the reactant that is present in the smallest amount is completely consumed the reaction will stop.
 In any type of chemical reaction, the amount of product that can be produced is determined by the reactant which is in the smallest amount. In any type of chemical reaction, the amount of product that
In any type of chemical reaction, the amount of product that can be produced is determined by the reactant which is in the smallest amount. In any type of chemical reaction, the amount of product that
Thermochemistry. Energy (and Thermochemistry) World of Chemistry Chapter 10. Energy. Energy
 Thermochemistry Thermodynamics is the science of the relationship between heat and other forms of energy. (and Thermochemistry) World of Chemistry Chapter 10 is defined as the ability to do work or produce
Thermochemistry Thermodynamics is the science of the relationship between heat and other forms of energy. (and Thermochemistry) World of Chemistry Chapter 10 is defined as the ability to do work or produce
ALE 27. Hess s Law. (Reference: Chapter 6 - Silberberg 5 th edition)
 Answer Key ALE 27. Hess s Law (Reference: Chapter 6 - Silberberg 5 th edition) Important!! For answers that involve a calculation you must show your work neatly using dimensional analysis with correct
Answer Key ALE 27. Hess s Law (Reference: Chapter 6 - Silberberg 5 th edition) Important!! For answers that involve a calculation you must show your work neatly using dimensional analysis with correct
Chapter 5. Thermochemistry
 Chapter 5 Thermochemistry Dr. A. Al-Saadi 1 Preview Introduction to thermochemistry: Potential energy and kinetic energy. Chemical energy. Internal energy, work and heat. Exothermic vs. endothermic reactions.
Chapter 5 Thermochemistry Dr. A. Al-Saadi 1 Preview Introduction to thermochemistry: Potential energy and kinetic energy. Chemical energy. Internal energy, work and heat. Exothermic vs. endothermic reactions.
I hope you aren't going to tear open the package and count the nails. We agree that. mass of nails = 1340 g g = g
 The Mole Concept Counting by weighing The size of molecule is so small that it is physically difficult if not impossible to directly count out molecules. this problem is solved using a common trick. Atoms
The Mole Concept Counting by weighing The size of molecule is so small that it is physically difficult if not impossible to directly count out molecules. this problem is solved using a common trick. Atoms
Since the rates of reactions in which acids and bases appear as catalysts depend strongly
 218 Chem 141 Lectures 29-32 Lecture 29 I stated at the beginning of our study of acids and bases that one reason that acids and bases are important is that they are the most common form of homogeneous
218 Chem 141 Lectures 29-32 Lecture 29 I stated at the beginning of our study of acids and bases that one reason that acids and bases are important is that they are the most common form of homogeneous
Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes
 Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes Students will be expected to: Compare the molar enthalpies of several combustion reactions involving organic compounds.
Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes Students will be expected to: Compare the molar enthalpies of several combustion reactions involving organic compounds.
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Learning Outcomes: Interconvert energy units Distinguish between the system and the surroundings in thermodynamics Calculate internal energy from heat and work and state sign
Chapter 5 Thermochemistry Learning Outcomes: Interconvert energy units Distinguish between the system and the surroundings in thermodynamics Calculate internal energy from heat and work and state sign
Chapter 6. Thermochemistry. Chapter 6. Chapter 6 Thermochemistry. Chapter 6 Thermochemistry Matter vs Energy 2/16/2016
 Chapter 6 Thermochemistry Chapter 6 Chapter 6 Thermochemistry 6.1 Chemical Hand Warmers 6.2 The Nature of Energy: Key Definitions 6.3 The First Law of Thermodynamics: There is no Free Lunch 6.4 6.5 Measuring
Chapter 6 Thermochemistry Chapter 6 Chapter 6 Thermochemistry 6.1 Chemical Hand Warmers 6.2 The Nature of Energy: Key Definitions 6.3 The First Law of Thermodynamics: There is no Free Lunch 6.4 6.5 Measuring
The reactions we have dealt with so far in chemistry are considered irreversible.
 1. Equilibrium Students: model static and dynamic equilibrium and analyse the differences between open and closed systems investigate the relationship between collision theory and reaction rate in order
1. Equilibrium Students: model static and dynamic equilibrium and analyse the differences between open and closed systems investigate the relationship between collision theory and reaction rate in order
Learning Check. How much heat, q, is required to raise the temperature of 1000 kg of iron and 1000 kg of water from 25 C to 75 C?
 Learning Check q = c * m * ΔT How much heat, q, is required to raise the temperature of 1000 kg of iron and 1000 kg of water from 25 C to 75 C? (c water =4.184 J/ C g, c iron =0.450 J/ C g) q Fe = 0.450
Learning Check q = c * m * ΔT How much heat, q, is required to raise the temperature of 1000 kg of iron and 1000 kg of water from 25 C to 75 C? (c water =4.184 J/ C g, c iron =0.450 J/ C g) q Fe = 0.450
5/14/14. How can you measure the amount of heat released when a match burns?
 CHEMISTRY & YOU Chapter 7 Thermochemistry How can you measure the amount of heat released when a match burns? 7. The Flow of Energy 7.3 Heat in Changes of State 7.4 Calculating Heats of Reaction Remember:
CHEMISTRY & YOU Chapter 7 Thermochemistry How can you measure the amount of heat released when a match burns? 7. The Flow of Energy 7.3 Heat in Changes of State 7.4 Calculating Heats of Reaction Remember:
10 Enthalpy changes Answers to Activity and Practice questions
 Page 150 151 Activity: Measuring the enthalpy change for the reaction of zinc with copper sulfate solution 1 The graph should have: axes with scales and labels points plotted accurately a clean, smooth
Page 150 151 Activity: Measuring the enthalpy change for the reaction of zinc with copper sulfate solution 1 The graph should have: axes with scales and labels points plotted accurately a clean, smooth
Section 1 - Thermochemistry
 Reaction Energy Section 1 - Thermochemistry Virtually every chemical reaction is accompanied by a change in energy. Chemical reactions usually absorb or release energy as heat. You learned in Chapter 12
Reaction Energy Section 1 - Thermochemistry Virtually every chemical reaction is accompanied by a change in energy. Chemical reactions usually absorb or release energy as heat. You learned in Chapter 12
Chapter 8. Thermochemistry
 Chapter 8 Thermochemistry Copyright 2001 by Harcourt, Inc. All rights reserved. Requests for permission to make copies of any part of the work should be mailed to the following address: Permissions Department,
Chapter 8 Thermochemistry Copyright 2001 by Harcourt, Inc. All rights reserved. Requests for permission to make copies of any part of the work should be mailed to the following address: Permissions Department,
Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy
 Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Name Date Class THE FLOW OF ENERGY HEAT AND WORK
 17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
CHM Accuracy, Precision, and Significant Figures (r14) C. Taylor 1/10
 CHM 110 - Accuracy, Precision, and Significant Figures (r14) - 2014 C. Taylor 1/10 Introduction Observations are vitally important to all of science. Some observations are qualitative in nature - such
CHM 110 - Accuracy, Precision, and Significant Figures (r14) - 2014 C. Taylor 1/10 Introduction Observations are vitally important to all of science. Some observations are qualitative in nature - such
Thermochemistry Chapter 4
 Thermochemistry Chapter 4 Thermochemistry is the study of energy changes that occur during chemical reactions Focus is on heat and matter transfer between the system and the surroundings Energy The ability
Thermochemistry Chapter 4 Thermochemistry is the study of energy changes that occur during chemical reactions Focus is on heat and matter transfer between the system and the surroundings Energy The ability
Stoichiometry. Please take out your notebooks
 Stoichiometry Please take out your notebooks Stoichiometry stochio = Greek for element metry = measurement Stoichiometry is about measuring the amounts of elements and compounds involved in a reaction.
Stoichiometry Please take out your notebooks Stoichiometry stochio = Greek for element metry = measurement Stoichiometry is about measuring the amounts of elements and compounds involved in a reaction.
Chemical Reactions. Chapter 17
 Chemical Reactions Chapter 17 Chemical Equations C+O 2 CO 2 C (s) +O 2 (g) CO 2 (g) Reactants on left, products on right Each are balanced because same number of atoms of reactants as products Some equations
Chemical Reactions Chapter 17 Chemical Equations C+O 2 CO 2 C (s) +O 2 (g) CO 2 (g) Reactants on left, products on right Each are balanced because same number of atoms of reactants as products Some equations
First Law of Thermodynamics: energy cannot be created or destroyed.
 1 CHEMICAL THERMODYNAMICS ANSWERS energy = anything that has the capacity to do work work = force acting over a distance Energy (E) = Work = Force x Distance First Law of Thermodynamics: energy cannot
1 CHEMICAL THERMODYNAMICS ANSWERS energy = anything that has the capacity to do work work = force acting over a distance Energy (E) = Work = Force x Distance First Law of Thermodynamics: energy cannot
Unit VI Stoichiometry. Applying Mole Town to Reactions
 Unit VI Stoichiometry Applying Mole Town to Reactions Learning Goals I can apply mole town to reactions to determine the amount of product based on the amount of a reactant. I can apply mole town to reaction
Unit VI Stoichiometry Applying Mole Town to Reactions Learning Goals I can apply mole town to reactions to determine the amount of product based on the amount of a reactant. I can apply mole town to reaction
Fundamentals Of Combustion (Part 1) Dr. D.P. Mishra Department of Aerospace Engineering Indian Institute of Technology, Kanpur
 Fundamentals Of Combustion (Part 1) Dr. D.P. Mishra Department of Aerospace Engineering Indian Institute of Technology, Kanpur Lecture 09 Stoichiometric calculations for air-gas mixture Let us start this
Fundamentals Of Combustion (Part 1) Dr. D.P. Mishra Department of Aerospace Engineering Indian Institute of Technology, Kanpur Lecture 09 Stoichiometric calculations for air-gas mixture Let us start this
Chapter 8 Thermochemistry: Chemical Energy
 Chapter 8 Thermochemistry: Chemical Energy 國防醫學院生化學科王明芳老師 2011-11-8 & 2011-11-15 Chapter 8/1 Energy and Its Conservation Conservation of Energy Law: Energy cannot be created or destroyed; it can only be
Chapter 8 Thermochemistry: Chemical Energy 國防醫學院生化學科王明芳老師 2011-11-8 & 2011-11-15 Chapter 8/1 Energy and Its Conservation Conservation of Energy Law: Energy cannot be created or destroyed; it can only be
Energy Relationships in Chemical Reactions
 Energy Relationships in Chemical Reactions What is heat? What is a state function? What is enthalpy? Is enthalpy a state function? What does this mean? How can we calculate this? How are the methods the
Energy Relationships in Chemical Reactions What is heat? What is a state function? What is enthalpy? Is enthalpy a state function? What does this mean? How can we calculate this? How are the methods the
Chemistry Crunch #3.2: Name: KEY What Is a Mole? Why? Learning Goals: Prerequisites:
 Chemistry Crunch #3.2: Name: KEY What Is a Mole? Why? 1) In the world around you, a variety of different measures are used to specify amounts. For example, at the store you might buy 10 apples or a 5 lb
Chemistry Crunch #3.2: Name: KEY What Is a Mole? Why? 1) In the world around you, a variety of different measures are used to specify amounts. For example, at the store you might buy 10 apples or a 5 lb
Today is Tuesday, May (!) 1 st, 2018
 In This Lesson: Stoichiometry (Lesson 4 of 4) Today is Tuesday, May (!) 1 st, 2018 Stuff You Need: Calculator Periodic Table Pre-Class: How does an airbag work? In solving a problem of this sort, the grand
In This Lesson: Stoichiometry (Lesson 4 of 4) Today is Tuesday, May (!) 1 st, 2018 Stuff You Need: Calculator Periodic Table Pre-Class: How does an airbag work? In solving a problem of this sort, the grand
MOLAR CONCENTRATION. - unit: MOLARITY (M): moles of dissolved substance per LITER of solution. molarity
 93 MOLAR CONCENTRATION - unit: MOLARITY (M): moles of dissolved substance per LITER of solution dissolved substance moles of SOLUTE molarity L SOLUTION If you have 0.250 L (250 ml) of 6.0 M HCl, how many
93 MOLAR CONCENTRATION - unit: MOLARITY (M): moles of dissolved substance per LITER of solution dissolved substance moles of SOLUTE molarity L SOLUTION If you have 0.250 L (250 ml) of 6.0 M HCl, how many
Energy and Chemical Change
 Energy and Chemical Change Section 15.1 Energy Section 15.2 Heat Section 15.3 Thermochemical Equations Section 15.4 Calculating Enthalpy Change Section 15.5 Reaction Spontaneity Click a hyperlink or folder
Energy and Chemical Change Section 15.1 Energy Section 15.2 Heat Section 15.3 Thermochemical Equations Section 15.4 Calculating Enthalpy Change Section 15.5 Reaction Spontaneity Click a hyperlink or folder
s Traditionally, we use the calorie as a unit of energy. The nutritional Calorie, Cal = 1000 cal. Kinetic Energy and Potential Energy
 AP Chemistry: Thermochemistry Lecture Outline 5.1 The Nature of Energy Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationships between chemical
AP Chemistry: Thermochemistry Lecture Outline 5.1 The Nature of Energy Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationships between chemical
General Chemistry 1 CHM201 Unit 3 Practice Test
 General Chemistry 1 CHM201 Unit 3 Practice Test 1. Heat is best defined as a. a substance that increases the temperature and causes water to boil. b. a form of potential energy. c. a form of work. d. the
General Chemistry 1 CHM201 Unit 3 Practice Test 1. Heat is best defined as a. a substance that increases the temperature and causes water to boil. b. a form of potential energy. c. a form of work. d. the
Atoms, Molecules, and the Mole
 The Mole Now that we know how to write and name chemical compounds, we need to understand how chemists use these formulas quantitatively. As chemists, we need to know how many atoms or molecules are reacting
The Mole Now that we know how to write and name chemical compounds, we need to understand how chemists use these formulas quantitatively. As chemists, we need to know how many atoms or molecules are reacting
DETERMINING AND USING H
 DETERMINING AND USING H INTRODUCTION CHANGES IN CHEMISTRY Chemistry is the science that studies matter and the changes it undergoes. Changes are divided into two categories: physical and chemical. During
DETERMINING AND USING H INTRODUCTION CHANGES IN CHEMISTRY Chemistry is the science that studies matter and the changes it undergoes. Changes are divided into two categories: physical and chemical. During
Chemistry I Chapter 9 Stoichiometry Objective Sheet. Equation 1. Objectives: 1. Define stoichiometry
 Chemistry I Chapter 9 Stoichiometry Objective Sheet Equation 1 2 C 2 H 2 (g) + 5 O 2 (g) 4 CO 2 (g) + 2 H 2 O (g), at STP C 2 H 2 (acetylene) 26 g/mol O 2 32 g/mol CO 2 44 g/mol H 2 O 18 g/mol Objectives:
Chemistry I Chapter 9 Stoichiometry Objective Sheet Equation 1 2 C 2 H 2 (g) + 5 O 2 (g) 4 CO 2 (g) + 2 H 2 O (g), at STP C 2 H 2 (acetylene) 26 g/mol O 2 32 g/mol CO 2 44 g/mol H 2 O 18 g/mol Objectives:
1.4 Enthalpy. What is chemical energy?
 1.4 Enthalpy What is chemical energy? Chemical energy is a form of potential energy which is stored in chemical bonds. Chemical bonds are the attractive forces that bind atoms together. As a reaction takes
1.4 Enthalpy What is chemical energy? Chemical energy is a form of potential energy which is stored in chemical bonds. Chemical bonds are the attractive forces that bind atoms together. As a reaction takes
AP Chemistry Chapter 16 Assignment. Part I Multiple Choice
 Page 1 of 7 AP Chemistry Chapter 16 Assignment Part I Multiple Choice 1984 47. CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O(l) H = 889.1 kj H f H 2 O(l) = 285.8 kj mol 1 H f CO 2 (g) = 393.3 kj mol 1 What is
Page 1 of 7 AP Chemistry Chapter 16 Assignment Part I Multiple Choice 1984 47. CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O(l) H = 889.1 kj H f H 2 O(l) = 285.8 kj mol 1 H f CO 2 (g) = 393.3 kj mol 1 What is
Bond Enthalpy and Activation Energy
 Bond Enthalpy and Activation Energy Energy of a Chemical Reaction ΔH = ΔH (bonds broken) - ΔH (bonds formed) Add up all the energies of the broken bonds Add up all the energies of the bonds that are reformed
Bond Enthalpy and Activation Energy Energy of a Chemical Reaction ΔH = ΔH (bonds broken) - ΔH (bonds formed) Add up all the energies of the broken bonds Add up all the energies of the bonds that are reformed
Chapter 5 - Thermochemistry
 Chapter 5 - Thermochemistry Study of energy changes that accompany chemical rx s. I) Nature of Energy Energy / Capacity to do work Mechanical Work w = F x d Heat energy - energy used to cause the temperature
Chapter 5 - Thermochemistry Study of energy changes that accompany chemical rx s. I) Nature of Energy Energy / Capacity to do work Mechanical Work w = F x d Heat energy - energy used to cause the temperature
Module 5: Combustion Technology. Lecture 32: Fundamentals of thermochemistry
 1 P age Module 5: Combustion Technology Lecture 32: Fundamentals of thermochemistry 2 P age Keywords : Heat of formation, enthalpy change, stoichiometric coefficients, exothermic reaction. Thermochemistry
1 P age Module 5: Combustion Technology Lecture 32: Fundamentals of thermochemistry 2 P age Keywords : Heat of formation, enthalpy change, stoichiometric coefficients, exothermic reaction. Thermochemistry
Chapter 6 Thermochemistry
 Chapter 6 Thermochemistry Contents and Concepts Understanding Heats of Reaction The first part of the chapter lays the groundwork for understanding what we mean by heats of reaction. 1. Energy and Its
Chapter 6 Thermochemistry Contents and Concepts Understanding Heats of Reaction The first part of the chapter lays the groundwork for understanding what we mean by heats of reaction. 1. Energy and Its
8.6 The Thermodynamic Standard State
 8.6 The Thermodynamic Standard State The value of H reported for a reaction depends on the number of moles of reactants...or how much matter is contained in the system C 3 H 8 (g) + 5O 2 (g) > 3CO 2 (g)
8.6 The Thermodynamic Standard State The value of H reported for a reaction depends on the number of moles of reactants...or how much matter is contained in the system C 3 H 8 (g) + 5O 2 (g) > 3CO 2 (g)
Energetics & Thermochemistry. Ms. Kiely IB Chemistry SL Coral Gables Senior High School
 Energetics & Thermochemistry Ms. Kiely IB Chemistry SL Coral Gables Senior High School Bell Ringer The following equation shows the formation of magnesium oxide from magnesium metal. 2Mg(s) + O2(g) 2MgO(s)
Energetics & Thermochemistry Ms. Kiely IB Chemistry SL Coral Gables Senior High School Bell Ringer The following equation shows the formation of magnesium oxide from magnesium metal. 2Mg(s) + O2(g) 2MgO(s)
Quiz I: Thermodynamics
 Quiz I: Thermodynamics SCH4U_2018-2019_V2 NAME: (Total Score: / 30) Multiple Choice (12) 1. What can be deduced from the following reaction profile? A. The reactants are less stable than the products and
Quiz I: Thermodynamics SCH4U_2018-2019_V2 NAME: (Total Score: / 30) Multiple Choice (12) 1. What can be deduced from the following reaction profile? A. The reactants are less stable than the products and
Quantity Relationships in Chemical Reactions
 Chapter 10 Relationships in Chemical Reactions Section 10.1 Conversion Factors from a Chemical Equation Goal 1 The coefficients in a chemical equation give us the conversion factors to get from the number
Chapter 10 Relationships in Chemical Reactions Section 10.1 Conversion Factors from a Chemical Equation Goal 1 The coefficients in a chemical equation give us the conversion factors to get from the number
SCIENCE 9 CONCEPT 4 CHEMICAL REACTIONS
 SCIENCE 9 CONCEPT 4 CHEMICAL REACTIONS VOCABULARY TERMS CONCEPT 4 Chemical reaction Reactants Products Exothermic Endothermic Combustion Corrosion Cellular respiration Conservation of mass Closed systems
SCIENCE 9 CONCEPT 4 CHEMICAL REACTIONS VOCABULARY TERMS CONCEPT 4 Chemical reaction Reactants Products Exothermic Endothermic Combustion Corrosion Cellular respiration Conservation of mass Closed systems
Lab #9- Calorimetry/Thermochemistry to the Rescue
 Chesapeake Campus Chemistry 111 Laboratory Lab #9- Calorimetry/Thermochemistry to the Rescue Objectives Determine whether a reaction is endothermic or exothermic. Determine the best ionic compound of to
Chesapeake Campus Chemistry 111 Laboratory Lab #9- Calorimetry/Thermochemistry to the Rescue Objectives Determine whether a reaction is endothermic or exothermic. Determine the best ionic compound of to
