CHAPTER 3 LEWIS ACID MOLECULAR SIEVES AS DIELS-ALDER CYCLOADDITION CATALYSTS: REACTION OF FURAN WITH ACRYLIC ACID
|
|
|
- Derick Lester
- 5 years ago
- Views:
Transcription
1 40 CHAPTER 3 LEWIS ACID MOLECULAR SIEVES AS DIELS-ALDER CYCLOADDITION CATALYSTS: REACTION OF FURAN WITH ACRYLIC ACID 3.1 Diels-Alder cycloaddition between furan and acrylic acid The Diels-Alder reaction between furan and acrylic acid was studied using the series of Lewis acid molecular sieves as catalysts. These molecular sieve materials have not been previously reported as Diels-Alder catalysts, and so the aim of this work was to investigate the usefulness of these materials for improving the rates of the cycloaddition reaction. The target reaction is shown in Fig. 3.1 and did not involve the dehydration of the Diels-Alder adduct to the aromatic product, benzoic acid. Fig. 3.1 Lewis acid-catalyzed Diels-Alder cycloaddition between furan and acrylic acid. Lewis acid-containing solids are known to catalyze Diels-Alder reactions. 1 For example, Corma et al. have studied the Diels-Alder cycloaddition between cyclopentadiene and p-benzoquinone catalyzed by a series of silica-based Lewis acid-containing solids similar to the of Lewis acid molecular sieves that are
2 41 proposed here, that is Sn- and Ti-containing MCM-41 and ITQ-2. 2 MCM-41 is a mesoporous silica and ITQ-2 is a delaminated zeolitic material. Molecular sieves can have both acid catalysis 2 and molecular confinement 3 effects on improving rates and selectivities. Since these cycloaddition reactions are bi-molecular and have a negative change in entropy, the spatial confinement within the micropores of zeolites alone is expected to lead to improved rates. 3,4 The uncatalyzed cycloaddition reaction between furan and acrylic acid is very slow at ambient temperature, taking weeks to reach equilibrium conversions. 5 It can be catalyzed by various homogenous Lewis acids, such as ZnCl 2, FeCl 3, AlCl 3, and borane complexes, 6 decreasing the time to reach equilibrium at ambient temperature to within 24 hr. When the methyl ester of acrylic acid was used as the dienophile, ZnCl 2 and ZnI 2 supported on silica gel were shown to catalyze the reaction heterogeneously. 7 Here, Lewis acid Beta molecular sieves and scandium(iii) triflate were tested as catalysts in the Diels-Alder reaction between furan and acrylic acid. Scandium(III) triflate was chosen as a reference homogeneous Lewis acid catalyst since Toste and Shiramizu successfully used scandium(iii) triflate to catalyze a very similar and related Diels-Alder reaction between 2,5- dimethylfuran and acrolein. 8 An example of the concentration profiles for furan, acrylic acid, and the Diels-Alder adduct using scandium(iii) triflate as catalyst at 50 o C is shown in Fig.3.2. High (>95%) selectivity to the Diels-Alder adduct was achieved and
3 acrylic acid conversions reached equilibrium conversions of ~40% at reaction times of 4 hours. 42 Fig. 3.2 Concentration profiles [mmol/(g solution)] of the furan and acrylic acid Diels-Alder reaction using Sc(OTf) 3 as catalyst. Reaction conditions: T=50 o C, 3.0 g furan, 0.3 g acrylic acid, 6 mg catalyst. Fig. 3.3 compares the Diels-Alder product concentration profiles over time for the Sc(OTf) 3 and Lewis acid Beta molecular sieve (Sn-Beta, Zr-Beta, Ti-Beta) catalysts. A pure silica Beta molecular sieve (Si-Beta) was included as a control. The results clearly indicate that the solid Lewis acids, Sn-Beta and Zr-Beta, are catalysts for producing the Diels-Alder product and are comparable to the homogeneous Lewis acid. Ti-Beta is only marginally active when compared to
4 43 the Si-Beta control. At this reaction temperature (50 o C), the dehydration of the Diels-Alder product to form benzoic acid was not observed. This is the first time this series of Lewis acid Beta molecular sieve materials have been shown to be catalysts for any Diels-Alder reaction. In addition, this finding supports further investigation of using these materials as catalysts for the Diels-Alder reactions involving ethylene as the dienophile. Fig. 3.3 Diels-Alder product concentration profiles using Sn-Beta, Zr-Beta, Ti- Beta, and Sc(OTf) 3 catalysts. Reaction conditions: T=50 o C, 3.0 g furan, 0.3 g acrylic acid, 360 acrylic acid:metal molar ratio. Sn-BEA-2 profile used 784 acrylic acid:metal ratio. The conversion of the Diels-Alder adduct to benzoic acid (Fig. 3.4) was not accomplished using the Sn-Beta or Zr-Beta catalysts under the reaction
5 44 conditions used for the cycloaddition, but here it is shown that the conversion can be performed using a strong Bronsted acid. Toste and Shiramizu 8 reported methods for the conversion of a similar 7-oxabicyclo[2.2.1]hept-2-ene compound using concentrated sulfuric acid at 0 o C (Fig. 3.5). Fig. 3.4 Bronsted acid-catalyzed conversion of the Diels-Alder adduct, 7-oxabicyclo[2.2.1]hept-5-ene-2-carboxylic acid, to benzoic acid. Fig. 3.5 Dehydrative aromatization example from Toste and Shiramizu. 8 When these conditions were applied to the current Diels-Alder adduct, the formation of benzoic acid was confirmed by 1 H NMR. The product NMR spectrum obtained is given in the appendix (Fig. 3.1-A). This shows that the Diels-Alder adduct, 7-oxa-bicyclo[2.2.1]hept-5-ene-2-carboxylic acid, is capable of undergoing dehydrative aromatization to the substituted aromatic product under strong Bronsted acid conditions at low temperatures, but the Lewis acid
6 Beta molecular sieves were unable to perform the dehydration under the low temperature conditions. 45
7 References [1] Cativiela, C.; Fraile, J.M.; Garcia, J.I.; Mayoral, J.A.; Pires, E.; Royo, A.J.; Tetrahedron 1993, 49(19), [2] Gomez, M.V.; Cantin, A.; Corma, A.; de la Hoz, A.; J. Mol. Catal. A 2005, 240, [3] Dessau, R.M.; J. Chem. Soc., Chem. Commun. 1986, [4] Fringuelli, F. and Taticchi, A. (Eds.). (2002). The Diels-Alder Reaction: Selected Practical Methods. West Sussex, England: John Wiley & Sons, Ltd. [5] Moore, J.A.; Partain, E.M.; J. Org. Chem. 1983, 48, [6] Kuwayama, T.; Ujita, K.; Shimizu, K.; Terashima, S.; Process for preparation of 7-oxabicyclo[2.2.1]hept-5-ene-2-carboxylic acid derivatives. US Patent 6,677,464, [7] Fraile, J.M.; Garcia, J.I.; Massam, J.; Mayoral, J.A.; Pires,E.; J. Mol. Catal. 1997, 123, [8] Shiramizu, M.; Toste, F.D.; Chem. Eur. J. 2011, 17,
CHAPTER Identification of side products in the synthesis of MMBC. As shown in the previous chapter, MMBC can be produced with high
 113 CHAPTER 6 IDENTIFICATION OF SIDE PRODUCTS IN THE PRODUCTION OF METHYL 4- (METHOXYMETHYL) BENZENE CARBOXYLATE (MMBC) FROM METHYL 5- (METHOXYMETHYL)-FURAN-2-CARBOXYLATE (MMFC) AND ETHYLENE 6.1 Identification
113 CHAPTER 6 IDENTIFICATION OF SIDE PRODUCTS IN THE PRODUCTION OF METHYL 4- (METHOXYMETHYL) BENZENE CARBOXYLATE (MMBC) FROM METHYL 5- (METHOXYMETHYL)-FURAN-2-CARBOXYLATE (MMFC) AND ETHYLENE 6.1 Identification
CHAPTER 4 USING ETHYLENE AS A DIENOPHILE IN DIELS-ALDER-DEHYDRATION REACTIONS WITH VARIOUS FURANS: SUMMARY OF EXPLORATORY WORK
 47 CHAPTER 4 USING ETHYLENE AS A DIENOPHILE IN DIELS-ALDER-DEHYDRATION REACTIONS WITH VARIOUS FURANS: SUMMARY OF EXPLORATORY WORK Results from reaction screening studies involving ethylene and various
47 CHAPTER 4 USING ETHYLENE AS A DIENOPHILE IN DIELS-ALDER-DEHYDRATION REACTIONS WITH VARIOUS FURANS: SUMMARY OF EXPLORATORY WORK Results from reaction screening studies involving ethylene and various
CHAPTER 7. for two different Diels-Alder-dehydration reactions catalyzed by the Lewis acid
 131 CHAPTER 7 MEASUREMENT OF ACTIVATION ENERGIES FOR ETHYLENE DIELS-ALDER-DEHYDRATION REACTIONS This chapter provides a summary and discussion of kinetic data obtained for two different Diels-Alder-dehydration
131 CHAPTER 7 MEASUREMENT OF ACTIVATION ENERGIES FOR ETHYLENE DIELS-ALDER-DEHYDRATION REACTIONS This chapter provides a summary and discussion of kinetic data obtained for two different Diels-Alder-dehydration
New Catalysts for the Renewable Production of. Monomers for Bioplastics. Thesis by. Joshua J. Pacheco. In Partial Fulfillment of the Requirements for
 New Catalysts for the Renewable Production of Monomers for Bioplastics Thesis by Joshua J. Pacheco In Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy California Institute
New Catalysts for the Renewable Production of Monomers for Bioplastics Thesis by Joshua J. Pacheco In Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy California Institute
Diels-Alder Reactions
 WARNING NOTICE: The experiments described in these materials are potentially hazardous and require a high level of safety training, special facilities and equipment, and supervision by appropriate individuals.
WARNING NOTICE: The experiments described in these materials are potentially hazardous and require a high level of safety training, special facilities and equipment, and supervision by appropriate individuals.
Solvent-free Diels-Alder Reactions of in situ Generated Cyclopentadiene
 S1 Supplemental Materials Solvent-free Diels-Alder Reactions of in situ Generated Cyclopentadiene David Huertas, Melinda Florscher and Veljko Dragojlovic* Wilkes Honors College of Florida Atlantic University,
S1 Supplemental Materials Solvent-free Diels-Alder Reactions of in situ Generated Cyclopentadiene David Huertas, Melinda Florscher and Veljko Dragojlovic* Wilkes Honors College of Florida Atlantic University,
Lewis Acid Zeolites for Tandem Diels-Alder Cycloaddition and Dehydration of Biomass-Derived Dimethylfuran and Ethylene to Renewable p-xylene
 Green Chemistry Lewis Acid Zeolites for Tandem Diels-Alder Cycloaddition and Dehydration of Biomass-Derived Dimethylfuran and Ethylene to Renewable p-xylene Journal: Green Chemistry Manuscript ID GC-ART-09-2015-002164.R1
Green Chemistry Lewis Acid Zeolites for Tandem Diels-Alder Cycloaddition and Dehydration of Biomass-Derived Dimethylfuran and Ethylene to Renewable p-xylene Journal: Green Chemistry Manuscript ID GC-ART-09-2015-002164.R1
Asymmetric Catalysis by Lewis Acids and Amines
 Asymmetric Catalysis by Lewis Acids and Amines Asymmetric Lewis acid catalysis - Chiral (bisooxazoline) copper (II) complexes - Monodentate Lewis acids: the formyl -bond Amine catalysed reactions Asymmetric
Asymmetric Catalysis by Lewis Acids and Amines Asymmetric Lewis acid catalysis - Chiral (bisooxazoline) copper (II) complexes - Monodentate Lewis acids: the formyl -bond Amine catalysed reactions Asymmetric
Diels-Alder Cycloaddition of Cyclopentadiene with Ethylacrylate Catalyzed by Mesoporous Al-MCM-48 and Al-MCM-41 Catalysts
 Diels-Alder Reaction over Al-MCM-48 Catalyst Bull. Korean Chem. Soc. 2008, Vol. 29, No. 10 1993 Diels-Alder Cycloaddition of Cyclopentadiene with Ethylacrylate Catalyzed by Mesoporous Al-MCM-48 and Al-MCM-41
Diels-Alder Reaction over Al-MCM-48 Catalyst Bull. Korean Chem. Soc. 2008, Vol. 29, No. 10 1993 Diels-Alder Cycloaddition of Cyclopentadiene with Ethylacrylate Catalyzed by Mesoporous Al-MCM-48 and Al-MCM-41
SUPPORTING INFORMATION. and Mark E. Davis*
 SUPPORTING INFORMATION Active Sites in Sn-Beta for Glucose Isomerization to Fructose and Epimerization to Mannose Ricardo Bermejo-Deval, Marat Orazov, Rajamani Gounder** Son-Jong Hwang and Mark E. Davis*
SUPPORTING INFORMATION Active Sites in Sn-Beta for Glucose Isomerization to Fructose and Epimerization to Mannose Ricardo Bermejo-Deval, Marat Orazov, Rajamani Gounder** Son-Jong Hwang and Mark E. Davis*
Friedel-Crafts Acylation of Anisole with Phthalic Anhydride Catalyzed by Solid Superacid of Sulfated Zirconia
 276 Journal of the Japan Petroleum Institute, 53, (5), 276-282 (2010) [Regular Paper] Friedel-Crafts Acylation of Anisole with Phthalic Anhydride Catalyzed by Solid Superacid of Sulfated Zirconia Hideo
276 Journal of the Japan Petroleum Institute, 53, (5), 276-282 (2010) [Regular Paper] Friedel-Crafts Acylation of Anisole with Phthalic Anhydride Catalyzed by Solid Superacid of Sulfated Zirconia Hideo
Dual enantioselective control by heterocycles of (S)-indoline derivatives*
 Pure Appl. Chem., Vol. 77, No. 12, pp. 2053 2059, 2005. DOI: 10.1351/pac200577122053 2005 IUPAC Dual enantioselective control by heterocycles of (S)-indoline derivatives* Yong Hae Kim, Doo Young Jung,
Pure Appl. Chem., Vol. 77, No. 12, pp. 2053 2059, 2005. DOI: 10.1351/pac200577122053 2005 IUPAC Dual enantioselective control by heterocycles of (S)-indoline derivatives* Yong Hae Kim, Doo Young Jung,
Morita Baylis Hillman Reaction. Aaron C. Smith 11/10/04
 Morita Baylis Hillman Reaction Aaron C. Smith 11/10/04 Outline 1. Background 2. Development of Asymmetric Variants 3. Aza-Baylis Hillman Reaction 4. Applications of Baylis Hillman Adducts Outline 1. Background
Morita Baylis Hillman Reaction Aaron C. Smith 11/10/04 Outline 1. Background 2. Development of Asymmetric Variants 3. Aza-Baylis Hillman Reaction 4. Applications of Baylis Hillman Adducts Outline 1. Background
CHAPTER 7 FRIEDEL-CRAFTS ACYLATION OF TOLUENE WITH ACETIC ACID
 113 CHAPTER 7 FRIEDEL-CRAFTS ACYLATION OF TOLUENE WITH ACETIC ACID 7.1 INTRODUCTION Acylation of aromatic compounds are industrially prominent reaction as its products are intermediates in many organic
113 CHAPTER 7 FRIEDEL-CRAFTS ACYLATION OF TOLUENE WITH ACETIC ACID 7.1 INTRODUCTION Acylation of aromatic compounds are industrially prominent reaction as its products are intermediates in many organic
SUPPORTING INFORMATION. Framework and Extraframework Tin Sites in Zeolite Beta React Glucose Differently
 SUPPORTING INFORMATION Framework and Extraframework Tin Sites in Zeolite Beta React Glucose Differently Ricardo Bermejo-Deval, Rajamani Gounder and Mark E. Davis* Chemical Engineering, California Institute
SUPPORTING INFORMATION Framework and Extraframework Tin Sites in Zeolite Beta React Glucose Differently Ricardo Bermejo-Deval, Rajamani Gounder and Mark E. Davis* Chemical Engineering, California Institute
Synthesis and Crystal Structure of Zinc Iodide in the Sodalite Cavities of Zeolite A (LTA)
 Synthesis and Crystal Structure of Zinc Iodide in the Zeolite A Bull. Korean Chem. Soc. 2007, Vol. 28, No. 4 567 Synthesis and Crystal Structure of Zinc Iodide in the Sodalite Cavities of Zeolite A (LTA)
Synthesis and Crystal Structure of Zinc Iodide in the Zeolite A Bull. Korean Chem. Soc. 2007, Vol. 28, No. 4 567 Synthesis and Crystal Structure of Zinc Iodide in the Sodalite Cavities of Zeolite A (LTA)
The aza-baylis-hillman Reaction: Mechanism, Asymmetric Catalysis, & Abnormal Adducts. Larry Wolf SED Group Meeting
 The aza-baylis-hillman Reaction: Mechanism, Asymmetric Catalysis, & Abnormal Adducts Larry Wolf SED Group Meeting 04-10-07 Outline Brief historical account and Utility Mechanism Different methods for asymmetric
The aza-baylis-hillman Reaction: Mechanism, Asymmetric Catalysis, & Abnormal Adducts Larry Wolf SED Group Meeting 04-10-07 Outline Brief historical account and Utility Mechanism Different methods for asymmetric
Joshua J. Pacheco,, Jay A. Labinger, Alex L. Sessions, and Mark E. Davis*, 1. INTRODUCTION
 This is an open access article published under an ACS AuthorChoice License, which permits copying and redistribution of the article or any adaptations for non-commercial purposes. pubs.acs.org/acscatalysis
This is an open access article published under an ACS AuthorChoice License, which permits copying and redistribution of the article or any adaptations for non-commercial purposes. pubs.acs.org/acscatalysis
N-Heterocyclic Carbene Catalysis via Azolium Dienolates: An Efficient Strategy for Enantioselective Remote Functionalizations
 Angew. Chem. Int. Ed. 2017, 10.1002. 1 N-Heterocyclic Carbene Catalysis via Azolium Dienolates: An Efficient Strategy for Enantioselective Remote Functionalizations Reporter: En Li Supervisor: Prof. Yong
Angew. Chem. Int. Ed. 2017, 10.1002. 1 N-Heterocyclic Carbene Catalysis via Azolium Dienolates: An Efficient Strategy for Enantioselective Remote Functionalizations Reporter: En Li Supervisor: Prof. Yong
molecules ISSN by MDPI
 Molecules 2007, 12, 1674-1678 Communication molecules ISSN 1420-3049 2007 by MDPI www.mdpi.org/molecules Aqueous (IV)-Catalyzed Diels-Alder Reaction Kyle E. itz Applied Nanoworks, Inc. 9 University Place,
Molecules 2007, 12, 1674-1678 Communication molecules ISSN 1420-3049 2007 by MDPI www.mdpi.org/molecules Aqueous (IV)-Catalyzed Diels-Alder Reaction Kyle E. itz Applied Nanoworks, Inc. 9 University Place,
Catalytic Enantioselective Diels-Alder Reactions Chwee T.S 1 and Wong M.W 2
 Catalytic Enantioselective Diels-Alder Reactions Chwee T.S 1 and Wong M.W 2 Department of Chemistry,Faculty of Science, National University of Singapore 10 Kent Ridge Road, Singapore 117546 Abstract Theoretical
Catalytic Enantioselective Diels-Alder Reactions Chwee T.S 1 and Wong M.W 2 Department of Chemistry,Faculty of Science, National University of Singapore 10 Kent Ridge Road, Singapore 117546 Abstract Theoretical
CHAPTER 7. ACYLATION OF ANISOLE WITH ACETIC ANHYDRIDE OVER MnAPO-5 AND LEWIS ACID METAL ION-EXCHANGED MnAPO-5
 103 CHAPTER 7 ACYLATIN F ANISLE WITH ACETIC ANHYDRIDE VER MnAP-5 AND LEWIS ACID METAL IN-EXCHANGED MnAP-5 7.1 INTRDUCTIN Friedel-Crafts acylation is one of the most important methods for the synthesis
103 CHAPTER 7 ACYLATIN F ANISLE WITH ACETIC ANHYDRIDE VER MnAP-5 AND LEWIS ACID METAL IN-EXCHANGED MnAP-5 7.1 INTRDUCTIN Friedel-Crafts acylation is one of the most important methods for the synthesis
Borenium ionic liquids as catalysts for Diels Alder reaction: tuneable Lewis superacids for catalytic applications
 Borenium ionic liquids as catalysts for Diels Alder reaction: tuneable Lewis superacids for catalytic applications Matuszek, K., Coffie, S., Chrobok, A., & Swadzba-Kwasny, M. (2017). Borenium ionic liquids
Borenium ionic liquids as catalysts for Diels Alder reaction: tuneable Lewis superacids for catalytic applications Matuszek, K., Coffie, S., Chrobok, A., & Swadzba-Kwasny, M. (2017). Borenium ionic liquids
Diels-Alder Reaction
 Diels-Alder Reaction Method for synthesis of 6-membered ring ne-step, concerted reaction Termed [4+2] cycloaddition reaction where 4 and 2 electrons react. + Diels-Alder Reaction Discovered by. Diels and
Diels-Alder Reaction Method for synthesis of 6-membered ring ne-step, concerted reaction Termed [4+2] cycloaddition reaction where 4 and 2 electrons react. + Diels-Alder Reaction Discovered by. Diels and
Catalytic activity of the beta zeolite with enhanced textural properties in the Friedel-Crafts acylation of aromatic compounds
 Zeolites and Related Materials: Trends, Targets and Challenges Proceedings of 4 th International FEZA Conference A. Gedeon, P. Massiani and F. Babboneau (Editors) 28 Elsevier B.V. All rights reserved.
Zeolites and Related Materials: Trends, Targets and Challenges Proceedings of 4 th International FEZA Conference A. Gedeon, P. Massiani and F. Babboneau (Editors) 28 Elsevier B.V. All rights reserved.
Mechanistic and Theoretical Studies on Stereoselectivity in Diels-Alder reactions
 chanistic and Theoretical Studies on Stereoselectivity in Diels-Alder reactions (Formerly: Theoretical Studies on the Diels-Alder Reaction) Leading References: Evans Group Seminar Jeff Johnson 6/98 "chanistic
chanistic and Theoretical Studies on Stereoselectivity in Diels-Alder reactions (Formerly: Theoretical Studies on the Diels-Alder Reaction) Leading References: Evans Group Seminar Jeff Johnson 6/98 "chanistic
Supporting Information
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2014 Supporting Information Acceptorless dehydrogenative coupling of primary alcohols
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2014 Supporting Information Acceptorless dehydrogenative coupling of primary alcohols
Kinetic Characterisation of Zeolite Catalysts Using Cracking, Alkylation and Other Chemical Reactions
 Haldor Topsøe Catalysis Forum Munkerupgaard, 27-28 August 2015 Kinetic Characterisation of Zeolite Catalysts Using Cracking, Alkylation and Other Chemical Reactions Dmitry B. Lukyanov Catalysis & Reaction
Haldor Topsøe Catalysis Forum Munkerupgaard, 27-28 August 2015 Kinetic Characterisation of Zeolite Catalysts Using Cracking, Alkylation and Other Chemical Reactions Dmitry B. Lukyanov Catalysis & Reaction
Silesian University of Technology, Poland Recoverable and recyclable catalysts for sustainable chemical processes
 Silesian University of Technology, Poland Recoverable and recyclable catalysts for sustainable chemical processes Anna Chrobok What are the challenges for the sustainable chemical industry today? reduce
Silesian University of Technology, Poland Recoverable and recyclable catalysts for sustainable chemical processes Anna Chrobok What are the challenges for the sustainable chemical industry today? reduce
Exam I 19 April 2004 Name:
 Chem 317 S04 Page 1 of 7 Kantorowski Exam I 19 April 2004 Name: This exam contains 6 pages of questions confirm this once you begin The last page is a spectral data table that can be removed You will have
Chem 317 S04 Page 1 of 7 Kantorowski Exam I 19 April 2004 Name: This exam contains 6 pages of questions confirm this once you begin The last page is a spectral data table that can be removed You will have
Page 1 of 9. Sessional Examination (November 2017) Max Marks: 20 Date: Time: One Hour. Model Answers
 Page 1 of 9 Sessional Examination (November 2017) Class: B. Pharm-II yr (III sem) Subject: Pharma Org. Chem-II Max Marks: 20 Date: 14.11.2017 Time: One Hour Model Answers Q. 1. Solve the following (ANY
Page 1 of 9 Sessional Examination (November 2017) Class: B. Pharm-II yr (III sem) Subject: Pharma Org. Chem-II Max Marks: 20 Date: 14.11.2017 Time: One Hour Model Answers Q. 1. Solve the following (ANY
CHAPTER 5 SYNTHESIS OF 7-HYDROXY-4-METHYLCOUMARIN OVER ZAPO-5 AND LEWIS ACID METAL ION-EXCHANGED ZAPO-5 MOLECULAR SIEVES
 79 CHAPTER 5 SYNTHESIS F 7-HYDRXY-4-METHYLCUMARIN VER ZAP-5 AND LEWIS ACID METAL IN-EXCHANGED ZAP-5 MLECULAR SIEVES 5.1 INTRDUCTIN Coumarins are an important group of naturally occurring compounds widely
79 CHAPTER 5 SYNTHESIS F 7-HYDRXY-4-METHYLCUMARIN VER ZAP-5 AND LEWIS ACID METAL IN-EXCHANGED ZAP-5 MLECULAR SIEVES 5.1 INTRDUCTIN Coumarins are an important group of naturally occurring compounds widely
(12) Patent Application Publication (10) Pub. No.: US 2014/ A1
 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2014/0364631 A1 DAVIS et al. US 20140364631A1 (43) Pub. Date: (54) (71) (72) (21) (22) (60) DELS-ALDER REACTINS CATALYZED BY LEWIS
(19) United States (12) Patent Application Publication (10) Pub. No.: US 2014/0364631 A1 DAVIS et al. US 20140364631A1 (43) Pub. Date: (54) (71) (72) (21) (22) (60) DELS-ALDER REACTINS CATALYZED BY LEWIS
Niobium Pentachloride Activation of Enone Derivatives: Diels- Alder and Conjugate Addition Products
 Molecules 2002, 7, 456 465 molecules ISSN 1420-3049 http://www.mdpi.org Niobium Pentachloride Activation of Enone Derivatives: Diels- Alder and Conjugate Addition Products Mauricio Gomes Constantino*,
Molecules 2002, 7, 456 465 molecules ISSN 1420-3049 http://www.mdpi.org Niobium Pentachloride Activation of Enone Derivatives: Diels- Alder and Conjugate Addition Products Mauricio Gomes Constantino*,
Chemistry 263 Quiz 1/Exam 1 Practice Spring 2018
 Chemistry 263 Quiz 1/Exam 1 Practice Spring 2018 The following practice contains 36 questions covering chapters 14, 15, and 16 in Loudon and Parise s rganic Chemistry, 6 th edition. Quiz 1 will contain
Chemistry 263 Quiz 1/Exam 1 Practice Spring 2018 The following practice contains 36 questions covering chapters 14, 15, and 16 in Loudon and Parise s rganic Chemistry, 6 th edition. Quiz 1 will contain
Conjugated Systems & Pericyclic Reactions
 onjugated Systems & Pericyclic Reactions 1 onjugated Dienes from heats of hydrogenation-relative stabilities of conjugated vs unconjugated dienes can be studied: Name 1-Butene 1-Pentene Structural Formula
onjugated Systems & Pericyclic Reactions 1 onjugated Dienes from heats of hydrogenation-relative stabilities of conjugated vs unconjugated dienes can be studied: Name 1-Butene 1-Pentene Structural Formula
X UNIVERSITY COLLEGE OF THE FRASER VALLEY COURSE INFORMATION. DISCIPLINE/DEPARTMENT: Chemistry IMPLEMENTATION DATE: Fall 1993
 X UNIVERSITY COLLEGE OF THE FRASER VALLEY COURSE INFORMATION DISCIPLINE/DEPARTMENT: Chemistry IMPLEMENTATION DATE: Fall 1993 Course Revised Implementation Date: May 2006 Course to be Reviewed: December
X UNIVERSITY COLLEGE OF THE FRASER VALLEY COURSE INFORMATION DISCIPLINE/DEPARTMENT: Chemistry IMPLEMENTATION DATE: Fall 1993 Course Revised Implementation Date: May 2006 Course to be Reviewed: December
Solvent-free and aqueous Knoevenagel condensation of aromatic ketones with malononitrile
 Solvent-free and aqueous Knoevenagel condensation of aromatic ketones with malononitrile Guan-Wu Wang* and Bo Cheng Department of Chemistry, University of Science and Technology of China, Hefei, Anhui
Solvent-free and aqueous Knoevenagel condensation of aromatic ketones with malononitrile Guan-Wu Wang* and Bo Cheng Department of Chemistry, University of Science and Technology of China, Hefei, Anhui
Mesoporous molecular sieve Sn-MCM-41 as Baeyer-Villiger oxidation catalyst for sterically demanding aromatic and α,β-unsaturated aldehydes
 Issue in onor of Prof. J. Elguero and P. Molina ARKIVC 2005 (ix) 124-132 Mesoporous molecular sieve Sn-MCM-41 as Baeyer-Villiger oxidation catalyst for sterically demanding aromatic and α,β-unsaturated
Issue in onor of Prof. J. Elguero and P. Molina ARKIVC 2005 (ix) 124-132 Mesoporous molecular sieve Sn-MCM-41 as Baeyer-Villiger oxidation catalyst for sterically demanding aromatic and α,β-unsaturated
Photodecomposition of Water Catalyzed by Zr- and Ti-MCM-41
 ESO 25(6) #7759 Energy Sources, 25:591 596, 2003 Copyright 2003 Taylor & Francis 0090-8312/03 $12.00 +.00 DOI: 10.1080/00908310390195651 Photodecomposition of Water Catalyzed by Zr- and Ti-MCM-41 S.-H.
ESO 25(6) #7759 Energy Sources, 25:591 596, 2003 Copyright 2003 Taylor & Francis 0090-8312/03 $12.00 +.00 DOI: 10.1080/00908310390195651 Photodecomposition of Water Catalyzed by Zr- and Ti-MCM-41 S.-H.
Synthetic Efforts Towards Anthracyclines Using the Asymmetric Diels-Alder Reaction
 1 Synthetic Efforts Towards Anthracyclines Using the Asymmetric Diels-Alder Reaction D C B A Me Me N 2 Figure 1: Doxorubicin (DX) 1. Introduction Doxorubicin (1) is an effective drug used in chemotherapy,
1 Synthetic Efforts Towards Anthracyclines Using the Asymmetric Diels-Alder Reaction D C B A Me Me N 2 Figure 1: Doxorubicin (DX) 1. Introduction Doxorubicin (1) is an effective drug used in chemotherapy,
Institute of Paper Science and Technology Atlanta, Georgia
 ti Institute of Paper Science and Technology Atlanta, Georgia IPST Technical Paper Series Number 913 Pulping Catalysts from Lignin: The Diels-Alder Step D.R. Dimmel, J.J. Bozell, D.G. von Oepen, and M.G.
ti Institute of Paper Science and Technology Atlanta, Georgia IPST Technical Paper Series Number 913 Pulping Catalysts from Lignin: The Diels-Alder Step D.R. Dimmel, J.J. Bozell, D.G. von Oepen, and M.G.
Supporting Information
 Supporting Information Efficient Short Step Synthesis of Corey s Tamiflu Intermediate Nsiama Tienabe Kipassa, Hiroaki kamura, * Kengo Kina, Tetsuo Iwagawa, and Toshiyuki Hamada Department of Chemistry
Supporting Information Efficient Short Step Synthesis of Corey s Tamiflu Intermediate Nsiama Tienabe Kipassa, Hiroaki kamura, * Kengo Kina, Tetsuo Iwagawa, and Toshiyuki Hamada Department of Chemistry
Pericyclic Reactions and Organic Photochemistry S. Sankararaman Department of Chemistry Indian Institute of Technology, Madras
 Pericyclic Reactions and Organic Photochemistry S. Sankararaman Department of Chemistry Indian Institute of Technology, Madras Module No. #02 Lecture No. #09 Pericyclic Reactions Cycloaddition Reactions
Pericyclic Reactions and Organic Photochemistry S. Sankararaman Department of Chemistry Indian Institute of Technology, Madras Module No. #02 Lecture No. #09 Pericyclic Reactions Cycloaddition Reactions
10.5 Catalytic reactions Catalyzed reactions. Out-class extensive reading: Levine, p Catalysis Enzyme catalysis
 10.5 Catalytic reactions Catalyzed reactions Out-class extensive reading: Levine, p.577 17.16 Catalysis 17.17 Enzyme catalysis 5.1 Catalysts and catalysis Catalyst A substance of small amount that can
10.5 Catalytic reactions Catalyzed reactions Out-class extensive reading: Levine, p.577 17.16 Catalysis 17.17 Enzyme catalysis 5.1 Catalysts and catalysis Catalyst A substance of small amount that can
Ruthenium-catalyzed cross aldol reaction with aldehydes and ketones
 Ruthenium-catalyzed cross aldol reaction with aldehydes and ketones Khalil Tabatabaeian,* Manouchehr Mamaghani, Nosrat O. Mahmoodi, and Elahe Keshavarz Department of Chemistry, Faculty of Science, University
Ruthenium-catalyzed cross aldol reaction with aldehydes and ketones Khalil Tabatabaeian,* Manouchehr Mamaghani, Nosrat O. Mahmoodi, and Elahe Keshavarz Department of Chemistry, Faculty of Science, University
Supporting Information
 Supporting Information Tetraethylammonium hydroxide (35 wt.%) (TEAOH), aluminum powder, fumed silica, zinc acetate dehydrate, Ludox HS-40, pure silica MCM-41, and tetraethylammonium fluoride were purchased
Supporting Information Tetraethylammonium hydroxide (35 wt.%) (TEAOH), aluminum powder, fumed silica, zinc acetate dehydrate, Ludox HS-40, pure silica MCM-41, and tetraethylammonium fluoride were purchased
Diels-Alder Cycloaddition
 Diels-Alder ycloaddition A lab practice for the reaction between cyclopentadiene and maleic anhydride The Diels-Alder ycloaddition + onjugated diene Dienophile Diels-Alder reaction: * Stereospecific *
Diels-Alder ycloaddition A lab practice for the reaction between cyclopentadiene and maleic anhydride The Diels-Alder ycloaddition + onjugated diene Dienophile Diels-Alder reaction: * Stereospecific *
Cyclooctatetraene (2R)-2-phenylbutane benzyl chloride cycloocta-1,3,5,7-tetraene
 CM238-02 S05 Practice Material for xam 1 This is a compilation from relevant problems from last spring s exam 1&2. This is too long! 1. (4 pts) Draw 2 of the following 3: Cross one out or graded in order.
CM238-02 S05 Practice Material for xam 1 This is a compilation from relevant problems from last spring s exam 1&2. This is too long! 1. (4 pts) Draw 2 of the following 3: Cross one out or graded in order.
Conjugated Dienes and Ultraviolet Spectroscopy
 Conjugated Dienes and Ultraviolet Spectroscopy Key Words Conjugated Diene Resonance Structures Dienophiles Concerted Reaction Pericyclic Reaction Cycloaddition Reaction Bridged Bicyclic Compound Cyclic
Conjugated Dienes and Ultraviolet Spectroscopy Key Words Conjugated Diene Resonance Structures Dienophiles Concerted Reaction Pericyclic Reaction Cycloaddition Reaction Bridged Bicyclic Compound Cyclic
Chapter 7: Alcohols, Phenols and Thiols
 Chapter 7: Alcohols, Phenols and Thiols Nomenclature of Alcohols In the IUPAC system, the hydroxyl group in alcohols is indicated by the ending ol. In common names, the separate word alcohol is placed
Chapter 7: Alcohols, Phenols and Thiols Nomenclature of Alcohols In the IUPAC system, the hydroxyl group in alcohols is indicated by the ending ol. In common names, the separate word alcohol is placed
METAL-ORGANIC FRAMEWORKS
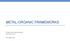 METAL-ORGANIC FRAMEWORKS Engle Lab Group Meeting Aug 10 th, 2017 Rei Matsuura A rapidly growing field Articles on "Metal-Organic Frameworks" 160000 140000 120000 100000 80000 60000 40000 20000 0 What are
METAL-ORGANIC FRAMEWORKS Engle Lab Group Meeting Aug 10 th, 2017 Rei Matsuura A rapidly growing field Articles on "Metal-Organic Frameworks" 160000 140000 120000 100000 80000 60000 40000 20000 0 What are
Reaction Mechanism of Polar Diels-Alder Reactions between 3-Nitrofuran and different Dienes. A Theoretical Study.
 Reaction Mechanism of Polar Diels-Alder Reactions between 3-Nitrofuran and different Dienes. A Theoretical Study. MAUR CAINELLI, CARLA RMACHEA, PEDR MANCINI, MARÍA KNEETEMAN* Área Química rgánica Departamento
Reaction Mechanism of Polar Diels-Alder Reactions between 3-Nitrofuran and different Dienes. A Theoretical Study. MAUR CAINELLI, CARLA RMACHEA, PEDR MANCINI, MARÍA KNEETEMAN* Área Química rgánica Departamento
Supporting Information
 1 Supporting Information Gold-catalyzed Synthesis of 3-arylindoles via Annulation of Nitrosoarenes and Alkynes Siva Murru, August A. Gallo and Radhey S. Srivastava* Department of Chemistry, University
1 Supporting Information Gold-catalyzed Synthesis of 3-arylindoles via Annulation of Nitrosoarenes and Alkynes Siva Murru, August A. Gallo and Radhey S. Srivastava* Department of Chemistry, University
{ReBr(CO) 3 (THF)} 2 (2.5 mol%) 4-Å molecular sieves toluene, 115 o C, 24 h
 VII Abstracts 2018 p1 10.2 Product Class 2: Benzo[c]furan and Its Derivatives. Kwiecień This chapter is a revision of the earlier cience of ynthesis contribution describing methods for the synthesis of
VII Abstracts 2018 p1 10.2 Product Class 2: Benzo[c]furan and Its Derivatives. Kwiecień This chapter is a revision of the earlier cience of ynthesis contribution describing methods for the synthesis of
Supporting Information For
 Supporting Information For Heteroatom-Substituted Delaminated Zeolites as Solid Lewis-Acid Catalysts Xiaoying Ouyang 1, Son-Jong Hwang 2, Dan Xie 3, Thomas Rea 3, Stacey I. Zones*,1,3, Alexander Katz*,1
Supporting Information For Heteroatom-Substituted Delaminated Zeolites as Solid Lewis-Acid Catalysts Xiaoying Ouyang 1, Son-Jong Hwang 2, Dan Xie 3, Thomas Rea 3, Stacey I. Zones*,1,3, Alexander Katz*,1
CHEM 203. Final Exam December 15, 2010 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
ROLE OF HETEROGENEOUS CATALYSTS IN CHEMICAL INDUSTRIES- A REVIEW
 ROLE OF HETEROGENEOUS CATALYSTS IN CHEMICAL INDUSTRIES- A REVIEW Priya Das 1, Yagnesh Joshi 2, Darshan Sarang 3, Dr. Yogesh Rotliwala 4 Student, Chemical Engg. Dept., SNPITRC, Bardoli, Gujarat, India 1
ROLE OF HETEROGENEOUS CATALYSTS IN CHEMICAL INDUSTRIES- A REVIEW Priya Das 1, Yagnesh Joshi 2, Darshan Sarang 3, Dr. Yogesh Rotliwala 4 Student, Chemical Engg. Dept., SNPITRC, Bardoli, Gujarat, India 1
Advanced Organic Chemistry
 D. A. Evans, G. Lalic Question of the day: Chemistry 530A TBS Me 2 C Me toluene, 130 C 70% TBS C 2 Me H H Advanced rganic Chemistry Me Lecture 16 Cycloaddition Reactions Diels _ Alder Reaction Photochemical
D. A. Evans, G. Lalic Question of the day: Chemistry 530A TBS Me 2 C Me toluene, 130 C 70% TBS C 2 Me H H Advanced rganic Chemistry Me Lecture 16 Cycloaddition Reactions Diels _ Alder Reaction Photochemical
Chiral Supramolecular Catalyst for Asymmetric Reaction
 Chiral Supramolecular Catalyst for Asymmetric Reaction 2017/1/21 (Sat.) Literature Seminar Taiki Fujita (B4) 1 Introduction Rational design of chiral ligands remains very difficult. Conventional chiral
Chiral Supramolecular Catalyst for Asymmetric Reaction 2017/1/21 (Sat.) Literature Seminar Taiki Fujita (B4) 1 Introduction Rational design of chiral ligands remains very difficult. Conventional chiral
An Eco-friendly Route to Synthesis of Quinolines
 An Eco-friendly Route to Synthesis of Quinolines A.D. Mishra Department of Chemistry, Tribhuvan University, P.N. Campus, Pokhara, Nepal E-mail: mishraad05@hotmail.com Abstract Some 2-hydroxy-4-methyl-6-
An Eco-friendly Route to Synthesis of Quinolines A.D. Mishra Department of Chemistry, Tribhuvan University, P.N. Campus, Pokhara, Nepal E-mail: mishraad05@hotmail.com Abstract Some 2-hydroxy-4-methyl-6-
Midterm #2 PRINTED LAST NAME 2 /20 3 /16 4 /9 5 /8 7 /24
 EM 234, Spring 2016 PRINTED FIRST complete this section ANSWER KEY Midterm #2 PRINTED LAST Ian R. Gould Person on your LEFT (or Empty or Aisle) Person on your RIGT (or Empty or Aisle) ASU or Posting ID
EM 234, Spring 2016 PRINTED FIRST complete this section ANSWER KEY Midterm #2 PRINTED LAST Ian R. Gould Person on your LEFT (or Empty or Aisle) Person on your RIGT (or Empty or Aisle) ASU or Posting ID
Organoselenium-Catalyzed Mild Dehydration of Aldoximes: An Unexpected Practical Method for Organonitrile Synthesis
 Supporting Information for Organoselenium-Catalyzed Mild Dehydration of Aldoximes: An Unexpected Practical Method for Organonitrile Synthesis Lei Yu,*,,, Hongyan Li, Xu Zhang,, Jianqing Ye, Jianping Liu,
Supporting Information for Organoselenium-Catalyzed Mild Dehydration of Aldoximes: An Unexpected Practical Method for Organonitrile Synthesis Lei Yu,*,,, Hongyan Li, Xu Zhang,, Jianqing Ye, Jianping Liu,
Reactivity and selectivity under microwaves in organic chemistry. Relation with medium effects and reaction mechanisms*
 Pure Appl. Chem., Vol. 73, No. 1, pp. 161 166, 2001. 2001 IUPAC Reactivity and selectivity under microwaves in organic chemistry. Relation with medium effects and reaction mechanisms* André Loupy 1,, Laurence
Pure Appl. Chem., Vol. 73, No. 1, pp. 161 166, 2001. 2001 IUPAC Reactivity and selectivity under microwaves in organic chemistry. Relation with medium effects and reaction mechanisms* André Loupy 1,, Laurence
Supplementary Figure S1: Solution NMR spectra for D-glucose with a 4:1 sugar:sb molar ratio in H 2 O. a) 13 C NMR of D-(1-13 C)glucose, b) 13 C NMR
 a c b d Supplementary Figure S1: Solution NMR spectra for D-glucose with a 4:1 sugar:sb molar ratio in H 2 O. a) 13 C NMR of D-(1-13 C)glucose, b) 13 C NMR of D-glucose, c) 11 B NMR, d) 1 H NMR a c b d
a c b d Supplementary Figure S1: Solution NMR spectra for D-glucose with a 4:1 sugar:sb molar ratio in H 2 O. a) 13 C NMR of D-(1-13 C)glucose, b) 13 C NMR of D-glucose, c) 11 B NMR, d) 1 H NMR a c b d
Two dimensional zeolites in catalysis
 Topsoe Catalysis Forum, August 27 28, 2015 Two dimensional zeolites in catalysis Jiří Čejka OUTLINE Two dimensional zeolites Synthesis routes Properties Catalytic applications Advantages Disadvantages
Topsoe Catalysis Forum, August 27 28, 2015 Two dimensional zeolites in catalysis Jiří Čejka OUTLINE Two dimensional zeolites Synthesis routes Properties Catalytic applications Advantages Disadvantages
CHEMISTRY MIDTERM # 2 ANSWER KEY July 10, 2002
 EMISTRY 314-81 MIDTERM # 2 ANSWER KEY July 10, 2002 Statistics: Average: 47 pts (67%); ighest: 69 pts (99%); Lowest: 26 pts (37%) Number of students performing at or above average: 12 (46%) 1. (7 pts)
EMISTRY 314-81 MIDTERM # 2 ANSWER KEY July 10, 2002 Statistics: Average: 47 pts (67%); ighest: 69 pts (99%); Lowest: 26 pts (37%) Number of students performing at or above average: 12 (46%) 1. (7 pts)
The following method was used to determine the percentage by mass of vanadium in a sample of ammonium vanadate(v).
 Q1. (a) Vanadium(V) oxide is used as a heterogeneous catalyst in the Contact Process. Explain what is meant by the terms heterogeneous and catalyst and state, in general terms, how a catalyst works. State
Q1. (a) Vanadium(V) oxide is used as a heterogeneous catalyst in the Contact Process. Explain what is meant by the terms heterogeneous and catalyst and state, in general terms, how a catalyst works. State
Direct Catalytic Conversion of Methane
 Direct Catalytic Conversion of Methane Xiulian Pan, Zengjian An, Ding Ma, Yide Xu, Xinhe Bao State Key Laboratory of Catlaysis, Dalian Institute of Chemical Physics, Chinese Academy of Sciences www.fruit.dicp.ac.cn
Direct Catalytic Conversion of Methane Xiulian Pan, Zengjian An, Ding Ma, Yide Xu, Xinhe Bao State Key Laboratory of Catlaysis, Dalian Institute of Chemical Physics, Chinese Academy of Sciences www.fruit.dicp.ac.cn
ONE POT SYNTHESIS OF NITRILES FROM ALDEHYDES AND HYDROXYLAMINE HYDROCHLORIDE USING SODIUM SULPHATE IN DMF UNDER REFLUX CONDITION
 WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Dinanath et al. Volume 3, Issue 1, 592-596. Research Article ISSN 2278 4357 ONE POT SYNTHESIS OF NITRILES FROM ALDEHYDES AND HYDROXYLAMINE HYDROCHLORIDE
WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Dinanath et al. Volume 3, Issue 1, 592-596. Research Article ISSN 2278 4357 ONE POT SYNTHESIS OF NITRILES FROM ALDEHYDES AND HYDROXYLAMINE HYDROCHLORIDE
nsaturated Hydrocarbons: Alkenes, Cycloalkenes and Dienes
 240 Chem nsaturated Hydrocarbons: Alkenes, Cycloalkenes and Dienes 1 Chapter 3 Alkenes or Olefines Crabon-Carbon double bond C n H 2n Hybridization in Alkenes: 1.34 A 2 Nomenclature of Alkenes and Cycloalkenes
240 Chem nsaturated Hydrocarbons: Alkenes, Cycloalkenes and Dienes 1 Chapter 3 Alkenes or Olefines Crabon-Carbon double bond C n H 2n Hybridization in Alkenes: 1.34 A 2 Nomenclature of Alkenes and Cycloalkenes
Coupling Alkane Dehydrogenation with Hydrogenation Reactions on Cation-Exchanged Zeolites
 Studies in Surface Science and Catalysis 130 A. Corma, F.V. Melo, S.Mendioroz and J.L.G. Fierro (Editors) 9 2000 Elsevier Science B.V. All rights reserved. 899 Coupling Alkane Dehydrogenation with Hydrogenation
Studies in Surface Science and Catalysis 130 A. Corma, F.V. Melo, S.Mendioroz and J.L.G. Fierro (Editors) 9 2000 Elsevier Science B.V. All rights reserved. 899 Coupling Alkane Dehydrogenation with Hydrogenation
Name: CHEM 633: Advanced Organic Chemistry: Physical Final Exam. Please answer the following questions clearly and concisely.
 Name: 1 CEM 633: Advanced rganic Chemistry: Physical Final Exam Please answer the following questions clearly and concisely. You may write your answers in the space provided and/or on additional pages.
Name: 1 CEM 633: Advanced rganic Chemistry: Physical Final Exam Please answer the following questions clearly and concisely. You may write your answers in the space provided and/or on additional pages.
SURVEY ON ARYL COMPOUNDS
 Journal of Plastic and Polymer Technology (JPPT) Vol. 1, Issue 1, Jun 2015, 111-132 TJPRC Pvt. Ltd SURVEY ON ARYL COMPOUNDS NAGHAM MAHMOOD ALJAMALI Organic Chemistry, Department of Chemistry, College of
Journal of Plastic and Polymer Technology (JPPT) Vol. 1, Issue 1, Jun 2015, 111-132 TJPRC Pvt. Ltd SURVEY ON ARYL COMPOUNDS NAGHAM MAHMOOD ALJAMALI Organic Chemistry, Department of Chemistry, College of
THE DIELS-ALDER REACTION
 22.6 TE DIELS-ALDER REATIN 977 2 Both overlaps are bonding. ± 2 ± 2 2 M of the diene LUM of the alkene ( 2 ) (*) The [ + 2] cycloaddition is allowed by a thermal pathway. Both overlaps are bonding, so
22.6 TE DIELS-ALDER REATIN 977 2 Both overlaps are bonding. ± 2 ± 2 2 M of the diene LUM of the alkene ( 2 ) (*) The [ + 2] cycloaddition is allowed by a thermal pathway. Both overlaps are bonding, so
*Assignments could be reversed. *
 Name Key 5 W-Exam No. Page I. (6 points) Identify the indicated pairs of hydrogens in each of the following compounds as (i) homotopic, (ii) enantiotopic, or (iii) diastereotopic s. Write the answers as
Name Key 5 W-Exam No. Page I. (6 points) Identify the indicated pairs of hydrogens in each of the following compounds as (i) homotopic, (ii) enantiotopic, or (iii) diastereotopic s. Write the answers as
Metal-catalyzed asymmetric hetero-diels-alder reactions of unactivated dienes with glyoxylates
 Pure & Appl. Chern., Vol. 70, No. 5, pp. 11 17-1 122, 1998. Printed in Great Britain. (0 1998 IUPAC Metal-catalyzed asymmetric hetero-diels-alder reactions of unactivated dienes with glyoxylates Mogens
Pure & Appl. Chern., Vol. 70, No. 5, pp. 11 17-1 122, 1998. Printed in Great Britain. (0 1998 IUPAC Metal-catalyzed asymmetric hetero-diels-alder reactions of unactivated dienes with glyoxylates Mogens
Rational Development of Solid Lewis Acid Catalysts for Biomass Conversion
 University of Massachusetts Amherst ScholarWorks@UMass Amherst Doctoral Dissertations Dissertations and Theses 2016 Rational Development of Solid Lewis Acid Catalysts for Biomass Conversion Chun-Chih Chang
University of Massachusetts Amherst ScholarWorks@UMass Amherst Doctoral Dissertations Dissertations and Theses 2016 Rational Development of Solid Lewis Acid Catalysts for Biomass Conversion Chun-Chih Chang
25 Zeolites as Catalysts for the Synthesis of Fine Chemicals
 775 25 Zeolites as Catalysts for the Synthesis of Fine Chemicals Maria J. Climent, Avelino Corma, and Sara Iborra 25.1 Introduction Zeolites are crystalline microporous materials that are widely used in
775 25 Zeolites as Catalysts for the Synthesis of Fine Chemicals Maria J. Climent, Avelino Corma, and Sara Iborra 25.1 Introduction Zeolites are crystalline microporous materials that are widely used in
Diels Alder Cycloaddition Strategy for Kinetic Resolution of Chiral Pyrazolidinones
 Iowa State University From the SelectedWorks of Levi M. Stanley August, 2009 Diels Alder Cycloaddition Strategy for Kinetic Resolution of Chiral Pyrazolidinones Mukund P. Sibi Keisuke Kawashima Levi M.
Iowa State University From the SelectedWorks of Levi M. Stanley August, 2009 Diels Alder Cycloaddition Strategy for Kinetic Resolution of Chiral Pyrazolidinones Mukund P. Sibi Keisuke Kawashima Levi M.
C(sp)-C(sp 3 ) Bond Formation through Cu-Catalyzed Cross-Coupling of N-Tosylhydrazones and Trialkylsilylethyne
 Supporting Information for C(sp)-C(sp 3 ) Bond Formation through Cu-Catalyzed Cross-Coupling of N-Tosylhydrazones and Trialkylsilylethyne Fei Ye, Xiaoshen Ma, Qing Xiao, Huan Li, Yan Zhang, Jianbo Wang*,,
Supporting Information for C(sp)-C(sp 3 ) Bond Formation through Cu-Catalyzed Cross-Coupling of N-Tosylhydrazones and Trialkylsilylethyne Fei Ye, Xiaoshen Ma, Qing Xiao, Huan Li, Yan Zhang, Jianbo Wang*,,
Answers To Chapter 7 Problems.
 Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
Copper(I)/TF-Biphamphos Catalyzed Asymmetric Nitroso. Diels-Alders Reaction
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Copper(I)/TF-Biphamphos Catalyzed Asymmetric Nitroso Diels-Alders
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Copper(I)/TF-Biphamphos Catalyzed Asymmetric Nitroso Diels-Alders
Highly active and reusable catalyst from Fe-Mg-hydrotalcite anionic clay for Friedel Crafts type benzylation reactions
 J. Chem. Sci., Vol. 117, No. 6, November 2005, pp. 635 639. Indian Academy of Sciences. Highly active and reusable catalyst from Fe-Mg-hydrotalcite anionic clay for Friedel Crafts type benzylation reactions
J. Chem. Sci., Vol. 117, No. 6, November 2005, pp. 635 639. Indian Academy of Sciences. Highly active and reusable catalyst from Fe-Mg-hydrotalcite anionic clay for Friedel Crafts type benzylation reactions
SECTION 8. Aromaticity Antiaromaticity. Electrocyclic and Diels-Alder Reaction. Sigmatropic H and Alkyl Shift.
 SECTION 8 Aromaticity Antiaromaticity Electrocyclic and Diels-Alder Reaction. Sigmatropic H and Alkyl Shift. (2014) 1 Aromaticity Antiromaticity Electrocyclic Reaction 2 Cyclobutadiene and Benzene Based
SECTION 8 Aromaticity Antiaromaticity Electrocyclic and Diels-Alder Reaction. Sigmatropic H and Alkyl Shift. (2014) 1 Aromaticity Antiromaticity Electrocyclic Reaction 2 Cyclobutadiene and Benzene Based
Acetylation of aromatics over acid zeolites: Seeking a viable alternative to Friedel Crafts catalysts*
 Pure Appl. Chem., Vol. 79, No. 11, pp. 1833 1838, 2007. doi:10.1351/pac200779111833 2007 IUPAC Acetylation of aromatics over acid zeolites: Seeking a viable alternative to Friedel Crafts catalysts* Matteo
Pure Appl. Chem., Vol. 79, No. 11, pp. 1833 1838, 2007. doi:10.1351/pac200779111833 2007 IUPAC Acetylation of aromatics over acid zeolites: Seeking a viable alternative to Friedel Crafts catalysts* Matteo
CHAPTER 4: CATALYTIC PROPERTIES OF ZSM-5 ZEOLITES AND CUBIC MESOPOROUS MATERIALS
 102 CHAPTER 4: CATALYTIC PROPERTIES OF ZSM-5 ZEOLITES AND CUBIC MESOPOROUS MATERIALS Chapter summary The role of heterogeneous catalysts in organic reactions is included in this chapter. Two organic reactions,
102 CHAPTER 4: CATALYTIC PROPERTIES OF ZSM-5 ZEOLITES AND CUBIC MESOPOROUS MATERIALS Chapter summary The role of heterogeneous catalysts in organic reactions is included in this chapter. Two organic reactions,
Chem 22 Final Exam Practice
 Chem 22 Final Exam Practice Questions taken from regular tests given during the previous semesters. Only one answer is correct unless the question says otherwise. The questions are somewhat scrambled with
Chem 22 Final Exam Practice Questions taken from regular tests given during the previous semesters. Only one answer is correct unless the question says otherwise. The questions are somewhat scrambled with
Molecular Orbitals of Ethene
 Molecular Orbitals of Ethene 1 Molecular Orbital Analysis of Ethene Dimerisation the reaction is said to be a "symmetry forbidden" interestingly, this reaction is rare and very slow! Molecular Orbitals
Molecular Orbitals of Ethene 1 Molecular Orbital Analysis of Ethene Dimerisation the reaction is said to be a "symmetry forbidden" interestingly, this reaction is rare and very slow! Molecular Orbitals
Chemistry Organic Chemistry II: Reactivity and Synthesis FINAL EXAM Winter 2003
 Name: Student No: Page 1 of 13 Chemistry 2.222 rganic Chemistry II: Reactivity and Synthesis FINAL EXAM Winter 2003 Paper Number 546 Thursday April 24, 2003 6:00 9:00 pm Frank Kennedy own Gym Students
Name: Student No: Page 1 of 13 Chemistry 2.222 rganic Chemistry II: Reactivity and Synthesis FINAL EXAM Winter 2003 Paper Number 546 Thursday April 24, 2003 6:00 9:00 pm Frank Kennedy own Gym Students
Catalyzed N-acylation of carbamates and oxazolidinones by Heteropolyacids (HPAs)
 Catalyzed -acylation of carbamates and oxazolidinones by eteropolyacids (PAs) Ali Gharib 1,2 *, Manouchehr Jahangir 1, Mina Roshani 1 1 Department of Chemistry, Islamic Azad University, Mashhad, IRA 2
Catalyzed -acylation of carbamates and oxazolidinones by eteropolyacids (PAs) Ali Gharib 1,2 *, Manouchehr Jahangir 1, Mina Roshani 1 1 Department of Chemistry, Islamic Azad University, Mashhad, IRA 2
Organic Chemistry: CHEM2322
 Conjugated Systems Organic Chemistry: We met in Chem 2321 unsaturated bonds as either a C=C bond or C C bond. If these unsaturated bonds are well separated then they react independently however if there
Conjugated Systems Organic Chemistry: We met in Chem 2321 unsaturated bonds as either a C=C bond or C C bond. If these unsaturated bonds are well separated then they react independently however if there
CHEM Exam 1 February 11, 2016 Constants and Equations: R = 8.31 J/mol-K. Beer-Lambert Law: A log bc. Michaelis-Menten Equation: v0 M
 CHEM 1423 - Exam 1 February 11, 2016 Constants and Equations: R = 8.31 J/mol-K Io Beer-Lambert Law: A log bc I Vm[ S] Michaelis-Menten Equation: v0 K [ S] M CHEM 1423 - Exam 1 February 11, 2016 Name (60)
CHEM 1423 - Exam 1 February 11, 2016 Constants and Equations: R = 8.31 J/mol-K Io Beer-Lambert Law: A log bc I Vm[ S] Michaelis-Menten Equation: v0 K [ S] M CHEM 1423 - Exam 1 February 11, 2016 Name (60)
Supporting Information
 Supporting Information Cobalt(II)-Catalyzed Acyloxylation of C- Bonds in Aromatic Amides with Carboxylic Acids Rina Ueno, Satoko atsui, and aoto Chatani* Department of Applied Chemistry, Faculty of Engineering,
Supporting Information Cobalt(II)-Catalyzed Acyloxylation of C- Bonds in Aromatic Amides with Carboxylic Acids Rina Ueno, Satoko atsui, and aoto Chatani* Department of Applied Chemistry, Faculty of Engineering,
2.222 Practice Problems 2003
 2.222 Practice Problems 2003 Set #1 1. Provide the missing starting compound(s), reagent/solvent, or product to correctly complete each of the following. Most people in the class have not done this type
2.222 Practice Problems 2003 Set #1 1. Provide the missing starting compound(s), reagent/solvent, or product to correctly complete each of the following. Most people in the class have not done this type
The Effect of Solvent on a Lewis Acid Catalyzed Diels-Alder Reaction, Using Computed and Experimental Kinetic Isotope Effects
 The Effect of Solvent on a Lewis Acid Catalyzed Diels-Alder Reaction, Using Computed and Experimental Kinetic Isotope Effects Orlando Acevedo and Jeffrey D. Evanseck Org. Lett., 5 (5), 649-652, 2003 Presented
The Effect of Solvent on a Lewis Acid Catalyzed Diels-Alder Reaction, Using Computed and Experimental Kinetic Isotope Effects Orlando Acevedo and Jeffrey D. Evanseck Org. Lett., 5 (5), 649-652, 2003 Presented
F. Matloubi Moghaddam 1;, M. Akhlaghi 1, L. Hojabri 1 and M.G. Dekamin 2. Abstract. INTRODUCTION
 Transactions C: Chemistry and Chemical Engineering Vol. 16, No. 2, pp. 81{88 c Sharif University of Technology, December 2009 A New Eco-Friendly and Ecient Mesoporous Solid Acid Catalyst for the Alkylation
Transactions C: Chemistry and Chemical Engineering Vol. 16, No. 2, pp. 81{88 c Sharif University of Technology, December 2009 A New Eco-Friendly and Ecient Mesoporous Solid Acid Catalyst for the Alkylation
General Chemistry II FINAL EXAMINATION
 General Chemistry II 141 FINAL EXAMINATIN PART I ( points each) CHEM 141 (Fall 009) Version AF09 Multiple choice - scantron. Please D NT write or mark on this paper. Write your correct answer on 1. Which
General Chemistry II 141 FINAL EXAMINATIN PART I ( points each) CHEM 141 (Fall 009) Version AF09 Multiple choice - scantron. Please D NT write or mark on this paper. Write your correct answer on 1. Which
DAMIETTA UNIVERSITY CHEM-405: PERICYCLIC REACTIONS LECTURE
 DAMIETTA UNIVERSITY CHEM-405: PERICYCLIC REACTIONS LECTURE 8 Dr Ali El-Agamey 1 LEARNING OUTCOMES LECTURE 8 (1) The Woodward-Hoffmann rules for [1,n] sigmatropic rearrangements -[1,2] cationic shift -[1,2]
DAMIETTA UNIVERSITY CHEM-405: PERICYCLIC REACTIONS LECTURE 8 Dr Ali El-Agamey 1 LEARNING OUTCOMES LECTURE 8 (1) The Woodward-Hoffmann rules for [1,n] sigmatropic rearrangements -[1,2] cationic shift -[1,2]
Tautomerism and Keto Enol Equilibrium
 Tautomerism and Keto Enol Equilibrium Enols & enolates are important nucleophiles in organic & biochemistry. Keto-Enol Equilibrium: Tautomerisation can be catalyzed by either acids or bases. Relative stability
Tautomerism and Keto Enol Equilibrium Enols & enolates are important nucleophiles in organic & biochemistry. Keto-Enol Equilibrium: Tautomerisation can be catalyzed by either acids or bases. Relative stability
