MOLECULAR DYNAMICS SIMULATIONS OF IONIC COPOLYMERS
|
|
|
- Ralf Barrett
- 5 years ago
- Views:
Transcription
1 COMPUTATIONAL METHODS IN SCIENCE AND TECHNOLOGY 6, (2000) MOLECULAR DYNAMICS SIMULATIONS OF IONIC COPOLYMERS MICHAŁ BANASZAK Macromolecular Physics Laboratory Institute of Physics, Adam Mickiewicz University ul. Umultowska 85, Poznań, Poland Abstract: Molecular Dynamics (MD) studies of ionic diblock copolymers are reported. The symmetrical diblocks are shown to exhibit a microphase separation transition (MST) similar to that of neutral diblocks. The MST was investigated by measuring thermodynamic, structural and dynamic properties: density, simulation box dimensions, structure factor, anisotropy in structure factor and directional diffusion constants. The slow cooling of the system from a high-temperature disordered phase to low-temperature ordered microphase separated phase was achieved in both temperature controlled ('NVT') MD, and temperature and pressure controlled ('NPT') MD. INTRODUCTION Ionic polymers (polyelectrolytes, ionomers) are of considerable current interest [1,2]. In those systems one ionic species, e.g., anion, is chemically bonded to the polymer chain either to a pendant group or to the backbone chain; the cation (counterion), on the other hand, is not chemically bonded to the chain and free to move if its thermal energy can overcome the electrostatic forces. The polymer chains can bestow a rubbery mechanical response whilst the counterions are more mobile and provide the electrical conductivity. The microarchitecture of those copolymer-like materials is complex, and also controlls the counterion mobility. In this article we present results of molecular dynamics (MD) simulations of symmetric diblock copolymers in which one block is an ionic polymer and one neutral, and both blocks have nearly the same size. The symmetric diblocks offer also a good model system for comparison with neutral (non-ionic) systems since the neutral diblock copolymers are fairly well understood theoretically and have been extensively studied experimentally and in simulations [3-26]. Unlike many previous simulations, which are Monte Carlo (MC) lattice simulations, our study employs a continuum space molecular dynamics method which is efficient at high densities, can easier be implemented to simulate the constant pressure systems, and finally it is better in calculating both the structural and dynamical properties, for example diffusion constant. It should be noted that while ionic polymer dilute solutions are fairly well understood, this is not the case for ionic polymer melts. We expect, however, a great deal of both excluded volume interaction screening and electrostatic interaction screening [25]. Since our main interest is in universal properties of the ionic polymer, we employ a coarsegrained model. This means that we are going to ignore the valence angle potential and the torsional potential, and keep only the rigid bonds along the chain, short range repulsive interaction and the electrostatic interactions. In our simulations we use molecular dynamics with Berendsen loose coupling, either only to thermostat ('NVT'), or both to barostat and thermostat ('NPT') in order to explore the
2 16 Molecular Dynamics Simulations of Ionic Copolymer microphase separation in ionic nearly symmetric diblock copolymers. While neutral diblock copolymers have been given much attention, in simulations theoretical studies and experimental studies, the ionic copolymers enjoyed less attention because of their complexity. By comparing the microphase behaviour of both ionic and neutral (non-ionic) systems we aim at showing both similarities and differences between them. Since the counterion transport properties in microphase-separated ionic copolymers are of great practical interest, we want to probe the relationship between counterion diffusion and microstructure. Finally by gaining insight into the symmetric diblock case, we lay foundations for our future studies, i.e., simulations of asymmetric ionic diblocks, ionic multiblocks or ionic branched copolymers such as NAFION. Neutral diblock copolymers can self-assemble into a variety of ordered microstructures. This self-assembly or ordering takes place as those copolymers are cooled down from a disordered high temperature phase to low temperature ordered phase. This process is called order-disorder transition (ODT) or microphase separation transition (MST), and is characterized by a temperature which marks the onset of long range order T ODT. It is well established experimentally that the microphase (ordered phase) can look like an array of bcc spheres, hexagonally packed cylinders, or spatially ordered layers. These microphases are often referred to as the "classical" microstructures. Close to the ODT the interface between the microdomains is diffused, and interfaces can fill up most of the space. This segregation regime is called weak segregation limit (WSL). By contrast, when the interfaces between microdomains are sharp and highly localized the system is in strong segregation limit (SSL). The diblock copolymer microstructure in the strong segregation regime is largely determined by volumetric considerations. The diblocks with roughly the same volume of both species form layers (lamellae), while diblock with significant (more than about 80% by volume) excess of one species form ordered spheres (micelles). The diblocks with intermediate volume fractions usually form hexagonally packed cylinders. Apart from those 'classical' microstructures, there have been discovered 'non-classical' bicontinuous microstructures such as ordered bicontinuous double diamond (OBDD) structure space group), gyroid bicontinuous structure space group) and hexagonally perforated layers. There is still some controversy about the existence and thermodynamic stability of those 'non-classical' microstructures. The experimental phase diagram of a real diblock copolymer can be found, for example in reference [10], Theories of neutral diblock copolymers have been successfully advanced over the past 20 years [6, 11, 22 and 23], both in the strong and weak segregation limits. In the weak segregation regime Leibler's mean-field Random Phase Approximation (RPA) approach [6] was particularly useful. This theory predicts a phase diagram in Flory interaction parameter, χ, multiplied by degree of polymerization, N, and diblock composition. When one goes beyond the mean-field approximation, and fluctuations are taken into account in the one-loop approximation [11], then the phase is more complex, but the RPA approach is still very useful in qualitative description of diblock copolymers. In the strong segregation limit there have been many selfconsistent numerical calculations initiated by Helfand and coworkers [23], and also almost analytical calculation presented first by Semenov [22].
3 M. Banaszak 17 For ionic copolymer system, the theoretical work done by Marko and Rabin [8] is particularly relevant for our study. They looked at microphase-separation properties of diblock copolymers composed of a neutral polymers joined to an ionic block. For a symmetric diblock, they predict a broad weak segregation region due to a heavy entropic penalty for counterions localization in strongly segregated micro domains. The work by Nyrkova et al. [3] is also interesting because it predicts a new limit for microphase separated ionic copolymers, the super strong segregation limit (SSSL). Their theory is applicable to highly asymmetric multiblock copolymers which form micelles. The chains in those micelles become fully extented in this limit. Most of the computer simulations of diblock copolymers are lattice simulations. The first three-dimensional Monte Carlo (MC) lattice simulations of block copolymer melts have been performed by Binder and Fried for both symmetric [13] and asymmetric [12] case. These simulations have been extensive yielding numerous significant results and ideas. Other relevant MC studies include the work by Mattice and coworkers [19], Weyersberg and Vilgis [14], and Freire and coworkers [15]. The current MC studies of diblocks, however, are limited by being performed on a lattice. Moreover the relation between MC "time" and physical time is open to debate. We therefore think that off-lattice MD simulations give both better insight and better representation of physical reality. The recent study of Grest et al. [16] reports an MD off-lattice simulation of a non-ionic diblock copolymer. This study is also interesting because it shows the advantage of simulating under constant pressure. This advantage is the freedom of the system to change the shape and size of the simulation box, and thus to achieve its equilibrium periodicity and microstructure. The ionic conductivity can in principle be determined directly by MD or NEMD but accurate measurements are difficult. We have instead assumed that the proportionality between conductivity and self-diffusion is a reasonable assumption for our systems. Consequently we have concentrated attention on measurements of cation diffusion coefficients which are expressed as the reduced mean square displacements, without dividing them by the corresponding time. The reduced density, ρ* = σ 3 ρ (where a is the diameter of the bead), is initially set to 0.5 which is closer to the semidilute regime than to a melt. This low bead density was chosen in order to speed up the equilibration. Our model system consists of twenty 31-mer diblocks and 160 cations, total 780 atoms, confined in a cubic box yielding the desired density. However, some simulations were carried out using 160 chains and 1280 cations. It was necessary to restrict the chain lengths due to very short structural relaxation times in these systems. Although our samples are not in the entangled regime, chains are sufficiently long to differentiate between the chain motions and cation motions. Finally it should be noted that some of the results presented in this paper has been already reported elsewhere [25,26], MODEL The basic model we apply is a linear freely joined Kramers chain of bead monomers, each corresponding to the size scale of the persistence length of a real polymer. Pairs of neighboring
4 18 Molecular Dynamics Simulations of Ionic Copolymer beads are connected by links of identical length. Our diblock copolymer, shown schematically in Fig. 1, consists of a neutral block of 15 beads, and the ionic block of 16 beads; in the latter block every second bead carries a negative elementary charge. Eight positively charged beads, i.e., cations, are included to maintain electroneutrality. Volumetricly this makes the system slightly asymmetric in favor of ionic block. Fig. 1. Schematic picture of the ionic copolymer chain. We have used a simplified interaction potential which incorporates explicit coulomb contributions superimposed on universal excluded volume interactions. Long range ion interactions are calculated by the Ewald method. The excluded volume effect is modelled using the purely repulsive "WCA" modification of the Lennard-Jones (LJ) potential between interactions centres representing neutral monomers, anionic monomers and "free" cations: Here r ij is the separation of monomers i and j, and σ are the usual LJ parameters which have the same magnitude for all beads. The link length between beads was set equal to σ. Including coulomb interactions between the charges the total interaction potential can therefore be written as follows: where the q's are charge numbers and e R is the dielectric constant. We can write this a more useful form; where E ij σ is the electrostatic potential at an ion separation of σ. Since the unlike charge interaction provides the only cohesive energy in the model we define E _σ as a measure of the depth of this potential where The actual location of the minimum of the potential energy between unlike charged beads depends on the ratio (E σ /ε), from now on referred to as γ. By simple analytical calculations it can be shown that if γ is chosen to be 24, then this minimum is located at σ, and the depth of the minimum is ((γ - l)/γ) E σ which differs only by about 2.5% from E σ. For a given value of σ temperature is thus reported as T* = kt/ E σ, density as p* = Nσ 3 /V, pressure as P* = Pσ 3 / E σ, and time as t* = t (E σ /(mσ 2 )) 1/2, where m is the mass of the bead. The optimized time step Δt* was found to be We should note that the significance of the
5 M. Banaszak 19 reduced temperature for instance is rather different than in the ordinary LJ system due to extensive mutual screening of the ion interactions in the melt. In our simulations we measured those properties which are the most useful in determining the ODT and the characterization of cation transport properties. First of all we measured the partial structure factors, defined as follows: where N is the total number of atoms, and is the number of atoms of type α, for example cations, and the summation over label i runs through all the atoms of type α. In disordered isotropic phase the structure factor can be spherically averaged since all spatial directions are equivalent. However, as the ODT is approached we expect the spherical symmetry to be broken, and accompanied by the rapid growth of the peak in the structure at a characteristic wavelength, k*; indicates the periodicity of the self-assembling lamellar microphase. In this study, we show the reduced mean square displacement of cations or z, as the measure of directional diffusion. We also measured the normalized square end-to-end distance of polymer chain, since it increases substantially in the course of ODT. Similarly we report the normalized chain end-to-distance of the ionic block, and the neutral block, RESULTS First we report 'NVT' results. In Fig. 2 we show the temperature dependence of four properties which clearly indicate the onset of microphase separation in the ionic copolymer. These results were obtained by cooling an N = 20 sample initially prepared as an isotropic disordered melt and equilibrated for 10 5 steps at T* = which is more than twice the temperature of the ODT. The cooling to T* = was accomplished incrementally in 14 stages; at each stage the system was equilibrated for 5 x 10 4 time steps followed by a sampling period of 5 x 10 4 steps. At temperatures between and ~0.04 the melt samples were homogeneous and the appearance of a strong nearest neighbour peak in the unlike charge radial distribution functions was an evidence for the existence of counterion condensation on the ionic blocks. Visualization of sample structures revealed that below T* ~ 0.04 the structure became an isotropic and a lamella morphology could be distinguished (see Fig. 3) as would be predicted for a near symmetric block copolymer. In periodic systems the sample spontaneously adopts a layer structure which is commensurate with the periodic boundaries of the simulation cell. The negative and positive ion partial structure factors are indistinguishable for the ordered microphase and in our simulations the peaks in these structure factors show that the lamella periodicity for the N = 20 system is L/2
6 20 Molecular Dynamics Simulations of Ionic Copolymer ( 11.6 a ) and the symmetry of the system is broken in the 100 direction whilst for the N = 160 sample the periodicity is, the broken symmetry direction being perpendicular to the 110 direction of the cubic cell. Due to condensed counterions the volume composition of ionic blocks is 0.62 and not 0.5 and this slight imbalance may be the cause of the perforations in the Fig. 2. Inverse temperature dependence of properties showing the onset of microphase separation on cooling the smaller (N = 20) sample: (a) the anisotropy invariant Q 1 of the directional structure factor S(k), evaluated for a spherical shell with 8 > kl/π > 2, (b) pressure components normal and tangential to eventual lamella plane of the microphase, (c) mean squared displacements for counterions normal and tangential to eventual lamella plane of the microphase after 5 x 10 4 time steps, (d) chain mean squared end-to-end distance, showing chain expansion through the ODT.
7 M. Banaszak 21 neutral layers seen in Fig. 3. Although there is experimental evidence for this kind of 'nonclassical' structure in neutral copolymers [19], we cannot be sure at this stage whether it is truly stable or whether it is a consequence of the boundary restrictions. Fig. 3. Instantaneous snapshot of the microphase structure in the 160 chain system at a temperature of T* = 0.024; 8 neighbouring periodic cells are shown to emphasize the perforated lamella morphology. The anisotropy of the structure factor [12] shows clearly the onset of structural order for wave vectors corresponding to the low-k peak in the structure factor. The principal moments of the angular distribution of structure factor, S(k), are ordered such that λ 1, λ 3. Symmetry breakdown accompanying ODT can then be characterized by two invariants, Q 1 and Q 2. λ 2 and for lamella symmetry is predicted to be significantly different from zero. It is shown in Fig. 2a that as the sample is cooled gradually increases and starts to fluctuate. It then changes rapidly to a value of about 0.5, suggesting that the ODT is at about T* = 0.043, while is close to zero, as expected. At high temperature in the disordered phase the diagonal components of the pressure tensor, are all equal within the statistical error (Fig 2a), but at about T* = 0.048, the pressure in the direction of symmetry breakdown starts to increase due to the onset of lamella ordering. At T* = the sample is in the microphase-separated regime and there is a significant difference in the tensor components normal and parallel to the direction of lamella ordering.
8 22 Molecular Dynamics Simulations of Ionic Copolymer There is a significant increase of the copolymer square end-to-end distance as the system is cooled through the ODT and the two dissimilar blocks become separated in space, as shown in Fig. 2(d). Similar effects have been noted in simulations of non-ionic copolymers [16]. There are departures from Gaussian ideality even in the initial (high-temperature) regime but this chain expansion is largely due to fact that the system is in the semidilute regime. In contrast to the case of the overall dimensions the ionic blocks show remarkably little extension over the whole temperature range and in the case of the ionic blocks we attribute this to efficient electrostatic screening of charged monomer repulsions by condensed counterions. Screening of this kind have also recently been observed in simulations of isolated ionic chains [5] for coulomb interaction strengths above the critical value for Manning condensation. In that case the collapse was interpreted in terms of dipole interactions between ion pairs [20]. In the ionic copolymer melt the electrostatic screening is even more effective and the ionic structure below the ODT is comparable with that of a molten salt [21], A quantity of considerable interest in these ionic systems is the counterion diffusion constant which is related to the ionic conductivity. As the system undergoes separation the counterions distribute themselves in the alternate ionic layers and consequently the counterion mobility in a direction normal to the layers decreases rapidly at the ODT as shown in Fig 2c. In the direction parallel to the lamella planes the diffusional mobility remains high over the whole temperature range studied. The mean squared displacements of the charged monomers were found to be an order of magnitude smaller than the counterions at long times and this decoupling of the diffusional motions is another important consequence of electrostatic screening. The high counterion mobility in the region of the ODT can also be related to the low value of the glass transition temperature for the ionic blocks. In the case of homopolymers with the same composition as the ionic blocks studied here but at much higher density of 0.93 the glass transition appears at T*~0.014 [18]; for the lower density used here one expects the glass transition to be at an even lower temperature. Now we turn our attention to 'NPT' results. These results were obtained by cooling an N = 20 sample initially prepared as an isotropic disordered melt at high temperature. The cooling to T* was accomplished incrementally in 14 stages; at each stage the system equilibration was followed by a sampling period. As we cooled the system, we monitored its properties. In particular, we looked at the square end-to-end distance of the polymer chains, which is expected to increase as we lower the temperature with more dramatic expansion in the vicinity of ODT. We also measured the square end-to-end distances for both blocks, neutral and charged. The results indicated an increase of copolymer square end-to-end distance as the system is cooled [26]. We also measured box dimensions and density, Figure 4 (a and b). At high-temperature disordered phase all diagonal components, L x, L y, L z are equal within the statistical error, but at about T* = 0.07, which is close to the ODT, the dimension in the direction of symmetry breakdown starts to increase, suggesting a force driving the ODT. At T* = 0.024, which con es-
9 M. Banaszak 23 Fig. 4. Temperature dependence of (a) density and (b) box dimensions in the 'NPT' cooling process. Fig. 5. Snapshots from the 'NPT' simulation (from upper left to lower right): T* = 0.098, 0.073, and
10 24 Molecular Dynamics Simulations of Ionic Copolymer ponds yields a well microphase-separated sample, there is a significant difference in box dimensions. Similarly the density increases as the system is cooled. Snapshots from the simulations (shown in Figure 5) visualize the changes of the simulation box dimensions as the system is cooled. CONCLUSIONS We have presented a generic mesoscopic model for simulation studies which can be used effectively to study microphase separation in ionic copolymers. Ion condensation screens the intensity and range of the electrostatic interactions and for the near symmetric block copolymer studied here this leads to microphase separation which has very similar characteristics to that observed in purely neutral copolymers. It also results in decoupled diffusion of counterions. The dimensions of the microphase in the 'NVT' cooling are distorted by boundary conditions but such effects were reduced by using controlled pressure 'NPT' cooling. Acknowledgement This research was supported by the Polish Committee for Scientific Research (grant no. KBN 8T11F 01214). References Eisenberg, A.; King,M. Ion-Containing Polymers; Academic Press New York 1977Gouin, J-P.; Bosse, F.; Nguyen, D.; Williams, C. E.; Eisenberg, A., Macromolecules, 26, 7250 (1993) Nyrkova, I. A.; Khokhlov, A. R.; Doi M., Macromolecules, 26, 3601(1993) Semenov, A. N.; Nyrkova, I. A.; Khokhlov, A. R., in Ionomers: Characterization, Theory and Applications, Ed. S. Schlick, CRC Press, 1996 Winkler, R. G.; Gold, M; Reineker, P.; Phys. Rev. Lett. 80, 3731(1998) Leibler, L., Macromolecules, 13, 1602(1980) Gonzales-Mozuelos, P.; Olvera de la Cruz, M.; J.Chem.Phys. 100, 507 (1994) Marko, J. F; Rabin, Y.; Macromolecules, 25, 1503(1992) Bates, F. S.; Fredrickson, G. H.; Ann. Rev. Phys. Chem., 41, 525(1990) Khandpur, A. K.; Forster, S.; Bates, F. S.; Hamley, I. W.; Ryan, A. J.; Bras, W.; Almdal, K.; Moitensen, K., Macromolecules, 28, 8796(1995) Fredrickson, G. H.; Helfand, E; J. Chem. Phys. 87, 697(1987) Fried, H.; Binder, K., J. Chem. Phys., 94, 8349(1991) Binder, K.; Fried, H., Macromolecules, 26, 6878(1993) Weyersberg, A.; Vilgis, T. A., Phys. Rev. E, 48, 377(1993) Molina, L. A.; Freire, J. J., Macromolecules, 28, 2705(1995) Grest, G. S.; Lacasse, M.-D.; Kremer, K.; Gupta, A. M., J. Chem. Phys., 105, 10583(1996) Groot, R. D.; Madden, T. J; J. Chem. Phys., 108, 8349(1998) C. Wong and J. H. R. Clarke, submitted for publication Matsen, M. W.; Bates, F. S.; Macromolecules, 29, 1091(1996) Khokhlov, A. R.; Kramarenko, E. Y., Macromolecules 29, 681(1996) Clarke, J. H. R.; Smith, W.; Woodcock, L.V.; J. Chem. Phys. 84,2290 (1986) Semenov, A. N.; Sov. Phys, JETP, 61, 733(1985) Helfand, E.; Wassermann, Z.R.; Macromolecules, 9, 879(1976); 11, 960(1978); 13, 994(1980) Banaszak, M. and Clarke. J. H. R., unpublished data Banaszak, M. and Clarke. J. H. R Phys. Rev. E60, 5753(1999) Banaszak, M. and Clarke. J. H. R., submitted for publication
Computer simulation of copolymer phase behavior
 JOURNAL OF CHEMICAL PHYSICS VOLUME 117, NUMBER 22 8 DECEMBER 2002 Computer simulation of copolymer phase behavior Andrew J. Schultz, Carol K. Hall, a) and Jan Genzer Department of Chemical Engineering,
JOURNAL OF CHEMICAL PHYSICS VOLUME 117, NUMBER 22 8 DECEMBER 2002 Computer simulation of copolymer phase behavior Andrew J. Schultz, Carol K. Hall, a) and Jan Genzer Department of Chemical Engineering,
Chapter 2. Block copolymers. a b c
 Chapter 2 Block copolymers In this thesis, the lamellar orientation in thin films of a symmetric diblock copolymer polystyrene-polymethylmethacylate P(S-b-MMA) under competing effects of surface interactions
Chapter 2 Block copolymers In this thesis, the lamellar orientation in thin films of a symmetric diblock copolymer polystyrene-polymethylmethacylate P(S-b-MMA) under competing effects of surface interactions
arxiv: v1 [cond-mat.soft] 11 Oct 2014
![arxiv: v1 [cond-mat.soft] 11 Oct 2014 arxiv: v1 [cond-mat.soft] 11 Oct 2014](/thumbs/72/67445951.jpg) Phase diagram of diblock copolymer melt in dimension d=5 M. Dziecielski, 1 K. Lewandowski, 1 and M. Banaszak 1, 1 Faculty of Physics, A. Mickiewicz University ul. Umultowska 85, 61-614 Poznan, Poland (Dated:
Phase diagram of diblock copolymer melt in dimension d=5 M. Dziecielski, 1 K. Lewandowski, 1 and M. Banaszak 1, 1 Faculty of Physics, A. Mickiewicz University ul. Umultowska 85, 61-614 Poznan, Poland (Dated:
My path: BS, Rose-Hulman PhD, UIUC Postdoc, Sandia. Map from Wikimedia Commons
 CoarseGrained Modeling of Ionomers and SaltDoped Block Copolymers Lisa Hall H.C. Slip Slider Assistant Professor William G. Lowrie Department of Chemical and Biomolecular Engineering The Ohio State University
CoarseGrained Modeling of Ionomers and SaltDoped Block Copolymers Lisa Hall H.C. Slip Slider Assistant Professor William G. Lowrie Department of Chemical and Biomolecular Engineering The Ohio State University
Block copolymer microstructures in the intermediate-segregation regime
 Block copolymer microstructures in the intermediate-segregation regime M. W. Matsen a) and F. S. Bates Department of Chemical Engineering and Materials Science, University of Minnesota, Minneapolis, Minnesota
Block copolymer microstructures in the intermediate-segregation regime M. W. Matsen a) and F. S. Bates Department of Chemical Engineering and Materials Science, University of Minnesota, Minneapolis, Minnesota
Chap. 2. Polymers Introduction. - Polymers: synthetic materials <--> natural materials
 Chap. 2. Polymers 2.1. Introduction - Polymers: synthetic materials natural materials no gas phase, not simple liquid (much more viscous), not perfectly crystalline, etc 2.3. Polymer Chain Conformation
Chap. 2. Polymers 2.1. Introduction - Polymers: synthetic materials natural materials no gas phase, not simple liquid (much more viscous), not perfectly crystalline, etc 2.3. Polymer Chain Conformation
Introduction to molecular dynamics
 1 Introduction to molecular dynamics Yves Lansac Université François Rabelais, Tours, France Visiting MSE, GIST for the summer Molecular Simulation 2 Molecular simulation is a computational experiment.
1 Introduction to molecular dynamics Yves Lansac Université François Rabelais, Tours, France Visiting MSE, GIST for the summer Molecular Simulation 2 Molecular simulation is a computational experiment.
Molecular Dynamics Simulations of Polyampholyte-Polyelectrolyte Complexes in Solutions
 5300 Macromolecules 2005, 38, 5300-5312 Molecular Dynamics Simulations of Polyampholyte-Polyelectrolyte Complexes in Solutions Junhwan Jeon and Andrey V. Dobrynin* Polymer Program, Institute of Materials
5300 Macromolecules 2005, 38, 5300-5312 Molecular Dynamics Simulations of Polyampholyte-Polyelectrolyte Complexes in Solutions Junhwan Jeon and Andrey V. Dobrynin* Polymer Program, Institute of Materials
PHYSICAL REVIEW E 69,
 Morphology and phase diagram of complex block copolymers: ABC linear triblock copolymers Ping Tang, Feng Qiu,* Hongdong Zhang, and Yuliang Yang Department of Macromolecular Science, The Key Laboratory
Morphology and phase diagram of complex block copolymers: ABC linear triblock copolymers Ping Tang, Feng Qiu,* Hongdong Zhang, and Yuliang Yang Department of Macromolecular Science, The Key Laboratory
Supplementary Information for Atomistic Simulation of Spinodal Phase Separation Preceding Polymer Crystallization
 Supplementary Information for Atomistic Simulation of Spinodal Phase Separation Preceding Polymer Crystallization Richard H. Gee * Naida Lacevic and Laurence E. Fried University of California Lawrence
Supplementary Information for Atomistic Simulation of Spinodal Phase Separation Preceding Polymer Crystallization Richard H. Gee * Naida Lacevic and Laurence E. Fried University of California Lawrence
Simulation studies of self-assembly of end-tethered nanorods in solution and role of rod aspect ratio and tether length
 THE JOURNAL OF CHEMICAL PHYSICS 125, 184903 2006 Simulation studies of self-assembly of end-tethered nanorods in solution and role of rod aspect ratio and tether length Mark A. Horsch and Zhenli Zhang
THE JOURNAL OF CHEMICAL PHYSICS 125, 184903 2006 Simulation studies of self-assembly of end-tethered nanorods in solution and role of rod aspect ratio and tether length Mark A. Horsch and Zhenli Zhang
Systematic Coarse-Graining and Concurrent Multiresolution Simulation of Molecular Liquids
 Systematic Coarse-Graining and Concurrent Multiresolution Simulation of Molecular Liquids Cameron F. Abrams Department of Chemical and Biological Engineering Drexel University Philadelphia, PA USA 9 June
Systematic Coarse-Graining and Concurrent Multiresolution Simulation of Molecular Liquids Cameron F. Abrams Department of Chemical and Biological Engineering Drexel University Philadelphia, PA USA 9 June
Hyeyoung Shin a, Tod A. Pascal ab, William A. Goddard III abc*, and Hyungjun Kim a* Korea
 The Scaled Effective Solvent Method for Predicting the Equilibrium Ensemble of Structures with Analysis of Thermodynamic Properties of Amorphous Polyethylene Glycol-Water Mixtures Hyeyoung Shin a, Tod
The Scaled Effective Solvent Method for Predicting the Equilibrium Ensemble of Structures with Analysis of Thermodynamic Properties of Amorphous Polyethylene Glycol-Water Mixtures Hyeyoung Shin a, Tod
Pre-yield non-affine fluctuations and a hidden critical point in strained crystals
 Supplementary Information for: Pre-yield non-affine fluctuations and a hidden critical point in strained crystals Tamoghna Das, a,b Saswati Ganguly, b Surajit Sengupta c and Madan Rao d a Collective Interactions
Supplementary Information for: Pre-yield non-affine fluctuations and a hidden critical point in strained crystals Tamoghna Das, a,b Saswati Ganguly, b Surajit Sengupta c and Madan Rao d a Collective Interactions
- intermolecular forces forces that exist between molecules
 Chapter 11: Intermolecular Forces, Liquids, and Solids - intermolecular forces forces that exist between molecules 11.1 A Molecular Comparison of Liquids and Solids - gases - average kinetic energy of
Chapter 11: Intermolecular Forces, Liquids, and Solids - intermolecular forces forces that exist between molecules 11.1 A Molecular Comparison of Liquids and Solids - gases - average kinetic energy of
Spherical phases with tunable steric interactions formed in
 pubs.acs.org/macroletters σ Phase Formed in Conformationally Asymmetric AB-Type Block Copolymers Nan Xie, Weihua Li,*, Feng Qiu, and An-Chang Shi State Key Laboratory of Molecular Engineering of Polymers,
pubs.acs.org/macroletters σ Phase Formed in Conformationally Asymmetric AB-Type Block Copolymers Nan Xie, Weihua Li,*, Feng Qiu, and An-Chang Shi State Key Laboratory of Molecular Engineering of Polymers,
Dynamic lattice liquid (DLL) model in computer simulation of the structure and dynamics of polymer condensed systems
 e-polymers 2012, no. 079 http://www.e-polymers.org ISSN 1618-7229 Dynamic lattice liquid (DLL) model in computer simulation of the structure and dynamics of polymer condensed systems Anna Blim *, Tomasz
e-polymers 2012, no. 079 http://www.e-polymers.org ISSN 1618-7229 Dynamic lattice liquid (DLL) model in computer simulation of the structure and dynamics of polymer condensed systems Anna Blim *, Tomasz
CHAPTER 2 INTERATOMIC FORCES. atoms together in a solid?
 CHAPTER 2 INTERATOMIC FORCES What kind of force holds the atoms together in a solid? Interatomic Binding All of the mechanisms which cause bonding between the atoms derive from electrostatic interaction
CHAPTER 2 INTERATOMIC FORCES What kind of force holds the atoms together in a solid? Interatomic Binding All of the mechanisms which cause bonding between the atoms derive from electrostatic interaction
Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers
 Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers Hong-ming Ding 1 & Yu-qiang Ma 1,2, 1 National Laboratory of Solid State Microstructures and Department
Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers Hong-ming Ding 1 & Yu-qiang Ma 1,2, 1 National Laboratory of Solid State Microstructures and Department
Monte Carlo Simulation of Ferroelectric Domain Structure: Electrostatic and Elastic Strain Energy Contributions
 Monte Carlo Simulation of Ferroelectric Domain Structure: Electrostatic and Elastic Strain Energy Contributions B.G. Potter, Jr., B.A. Tuttle, and V. Tikare Sandia National Laboratories Albuquerque, NM
Monte Carlo Simulation of Ferroelectric Domain Structure: Electrostatic and Elastic Strain Energy Contributions B.G. Potter, Jr., B.A. Tuttle, and V. Tikare Sandia National Laboratories Albuquerque, NM
Chapter 2 Polymer Physics Concentrated Solutions and Melts
 Chapter 2 Polymer Physics Concentrated Solutions and Melts Chapter 1 discussed the statistical thermodynamics of an isolated polymer chain in a solvent. The conformation of an isolated polymer coil in
Chapter 2 Polymer Physics Concentrated Solutions and Melts Chapter 1 discussed the statistical thermodynamics of an isolated polymer chain in a solvent. The conformation of an isolated polymer coil in
Directed Assembly of Functionalized Nanoparticles with Amphiphilic Diblock Copolymers. Contents
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Electronic Supplementary Information for Directed Assembly of Functionalized Nanoparticles
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Electronic Supplementary Information for Directed Assembly of Functionalized Nanoparticles
On the Dynamics and Disentanglement in Thin and Two-Dimensional Polymer Films
 J. Phys. IV France 1 (006) Pr1-1 c EDP Sciences, Les Ulis On the Dynamics and Disentanglement in Thin and Two-Dimensional Polymer Films H. Meyer, T. Kreer, A. Cavallo, J. P. Wittmer and J. Baschnagel 1
J. Phys. IV France 1 (006) Pr1-1 c EDP Sciences, Les Ulis On the Dynamics and Disentanglement in Thin and Two-Dimensional Polymer Films H. Meyer, T. Kreer, A. Cavallo, J. P. Wittmer and J. Baschnagel 1
CHEM Principles of Chemistry II Chapter 10 - Liquids and Solids
 CHEM 1212 - Principles of Chemistry II Chapter 10 - Liquids and Solids 10.1 Intermolecular Forces recall intramolecular (within the molecule) bonding whereby atoms can form stable units called molecules
CHEM 1212 - Principles of Chemistry II Chapter 10 - Liquids and Solids 10.1 Intermolecular Forces recall intramolecular (within the molecule) bonding whereby atoms can form stable units called molecules
Aqueous solutions. Solubility of different compounds in water
 Aqueous solutions Solubility of different compounds in water The dissolution of molecules into water (in any solvent actually) causes a volume change of the solution; the size of this volume change is
Aqueous solutions Solubility of different compounds in water The dissolution of molecules into water (in any solvent actually) causes a volume change of the solution; the size of this volume change is
Potentials, periodicity
 Potentials, periodicity Lecture 2 1/23/18 1 Survey responses 2 Topic requests DFT (10), Molecular dynamics (7), Monte Carlo (5) Machine Learning (4), High-throughput, Databases (4) NEB, phonons, Non-equilibrium
Potentials, periodicity Lecture 2 1/23/18 1 Survey responses 2 Topic requests DFT (10), Molecular dynamics (7), Monte Carlo (5) Machine Learning (4), High-throughput, Databases (4) NEB, phonons, Non-equilibrium
High-Resolution Implementation of Self-Consistent Field Theory
 High-Resolution Implementation of Self-Consistent Field Theory Eric W. Cochran Chemical and Biological Engineering Iowa State University Carlos Garcia-Cervera Department of Mathematics University of California
High-Resolution Implementation of Self-Consistent Field Theory Eric W. Cochran Chemical and Biological Engineering Iowa State University Carlos Garcia-Cervera Department of Mathematics University of California
Structuring of hydrophobic and hydrophilic polymers at interfaces Stephen Donaldson ChE 210D Final Project Abstract
 Structuring of hydrophobic and hydrophilic polymers at interfaces Stephen Donaldson ChE 210D Final Project Abstract In this work, a simplified Lennard-Jones (LJ) sphere model is used to simulate the aggregation,
Structuring of hydrophobic and hydrophilic polymers at interfaces Stephen Donaldson ChE 210D Final Project Abstract In this work, a simplified Lennard-Jones (LJ) sphere model is used to simulate the aggregation,
Ionic Bonding. Example: Atomic Radius: Na (r = 0.192nm) Cl (r = 0.099nm) Ionic Radius : Na (r = 0.095nm) Cl (r = 0.181nm)
 Ionic Bonding Ion: an atom or molecule that gains or loses electrons (acquires an electrical charge). Atoms form cations (+charge), when they lose electrons, or anions (- charge), when they gain electrons.
Ionic Bonding Ion: an atom or molecule that gains or loses electrons (acquires an electrical charge). Atoms form cations (+charge), when they lose electrons, or anions (- charge), when they gain electrons.
The effects of Lowe Andersen temperature controlling method on the polymer properties in mesoscopic simulations
 THE JOURNAL OF CHEMICAL PHYSICS 22, 04907 2005 The effects of Lowe Andersen temperature controlling method on the polymer properties in mesoscopic simulations Li-Jun Chen, Zhong-Yuan Lu, a Hu-Jun Qian,
THE JOURNAL OF CHEMICAL PHYSICS 22, 04907 2005 The effects of Lowe Andersen temperature controlling method on the polymer properties in mesoscopic simulations Li-Jun Chen, Zhong-Yuan Lu, a Hu-Jun Qian,
Chapter 10: Liquids and Solids
 Chapter 10: Liquids and Solids Chapter 10: Liquids and Solids *Liquids and solids show many similarities and are strikingly different from their gaseous state. 10.1 Intermolecular Forces Intermolecular
Chapter 10: Liquids and Solids Chapter 10: Liquids and Solids *Liquids and solids show many similarities and are strikingly different from their gaseous state. 10.1 Intermolecular Forces Intermolecular
Monica Olvera de la Cruz Northwestern University Department of Materials Science and Engineering 2220 Campus Drive Evanston, IL 60202
 Introduction to Biomaterials and Biomolecular Self Assembly Monica Olvera de la Cruz orthwestern University Department of Materials Science and Engineering 0 Campus Drive Evanston, IL 600 Outline Introduction
Introduction to Biomaterials and Biomolecular Self Assembly Monica Olvera de la Cruz orthwestern University Department of Materials Science and Engineering 0 Campus Drive Evanston, IL 600 Outline Introduction
 Title Theory of solutions in the energy r of the molecular flexibility Author(s) Matubayasi, N; Nakahara, M Citation JOURNAL OF CHEMICAL PHYSICS (2003), 9702 Issue Date 2003-11-08 URL http://hdl.handle.net/2433/50354
Title Theory of solutions in the energy r of the molecular flexibility Author(s) Matubayasi, N; Nakahara, M Citation JOURNAL OF CHEMICAL PHYSICS (2003), 9702 Issue Date 2003-11-08 URL http://hdl.handle.net/2433/50354
Microphase separation in multiblock copolymer melts Smirnova, YG; ten Brinke, G; Erukhimovich, IY; Smirnova, Yuliya G.; Erukhimovich, Igor Ya.
 University of Groningen Microphase separation in multiblock copolymer melts Smirnova, YG; ten Brinke, G; Erukhimovich, IY; Smirnova, Yuliya G.; Erukhimovich, Igor Ya. Published in: Journal of Chemical
University of Groningen Microphase separation in multiblock copolymer melts Smirnova, YG; ten Brinke, G; Erukhimovich, IY; Smirnova, Yuliya G.; Erukhimovich, Igor Ya. Published in: Journal of Chemical
Coarse-Grained Models!
 Coarse-Grained Models! Large and complex molecules (e.g. long polymers) can not be simulated on the all-atom level! Requires coarse-graining of the model! Coarse-grained models are usually also particles
Coarse-Grained Models! Large and complex molecules (e.g. long polymers) can not be simulated on the all-atom level! Requires coarse-graining of the model! Coarse-grained models are usually also particles
Physical Chemistry of Polymers (4)
 Physical Chemistry of Polymers (4) Dr. Z. Maghsoud CONCENTRATED SOLUTIONS, PHASE SEPARATION BEHAVIOR, AND DIFFUSION A wide range of modern research as well as a variety of engineering applications exist
Physical Chemistry of Polymers (4) Dr. Z. Maghsoud CONCENTRATED SOLUTIONS, PHASE SEPARATION BEHAVIOR, AND DIFFUSION A wide range of modern research as well as a variety of engineering applications exist
Proteins polymer molecules, folded in complex structures. Konstantin Popov Department of Biochemistry and Biophysics
 Proteins polymer molecules, folded in complex structures Konstantin Popov Department of Biochemistry and Biophysics Outline General aspects of polymer theory Size and persistent length of ideal linear
Proteins polymer molecules, folded in complex structures Konstantin Popov Department of Biochemistry and Biophysics Outline General aspects of polymer theory Size and persistent length of ideal linear
Self Organization. Order. Homogeneous state. Structurally ordered state. Structurally ordered state. Order. Disorder
 Muthukumar, M., Ober, C.K. and Thomas, E.L., "Competing Interactions and Levels of Ordering in Self-Organizing Materials," Science, 277, 1225-1237 (1997). Self Organization Homogeneous state Order Disorder
Muthukumar, M., Ober, C.K. and Thomas, E.L., "Competing Interactions and Levels of Ordering in Self-Organizing Materials," Science, 277, 1225-1237 (1997). Self Organization Homogeneous state Order Disorder
arxiv: v1 [cond-mat.stat-mech] 11 Oct 2014
![arxiv: v1 [cond-mat.stat-mech] 11 Oct 2014 arxiv: v1 [cond-mat.stat-mech] 11 Oct 2014](/thumbs/88/115855721.jpg) Heuristic Monte Carlo Method Applied to Cooperative Motion Algorithm for Binary Lattice Fluid P. Knycha la 1, and M. Banaszak 2 1 Chair of Computer Science, The President Stanislaw arxiv:1410.3015v1 [cond-mat.stat-mech]
Heuristic Monte Carlo Method Applied to Cooperative Motion Algorithm for Binary Lattice Fluid P. Knycha la 1, and M. Banaszak 2 1 Chair of Computer Science, The President Stanislaw arxiv:1410.3015v1 [cond-mat.stat-mech]
On the size and shape of self-assembled micelles
 On the size and shape of self-assembled micelles Peter H. Nelson, Gregory C. Rutledge, and T. Alan Hatton a) Department of Chemical Engineering, Massachusetts Institute of Technology, Cambridge, Massachusetts
On the size and shape of self-assembled micelles Peter H. Nelson, Gregory C. Rutledge, and T. Alan Hatton a) Department of Chemical Engineering, Massachusetts Institute of Technology, Cambridge, Massachusetts
EXAM I COURSE TFY4310 MOLECULAR BIOPHYSICS December Suggested resolution
 page 1 of 7 EXAM I COURSE TFY4310 MOLECULAR BIOPHYSICS December 2013 Suggested resolution Exercise 1. [total: 25 p] a) [t: 5 p] Describe the bonding [1.5 p] and the molecular orbitals [1.5 p] of the ethylene
page 1 of 7 EXAM I COURSE TFY4310 MOLECULAR BIOPHYSICS December 2013 Suggested resolution Exercise 1. [total: 25 p] a) [t: 5 p] Describe the bonding [1.5 p] and the molecular orbitals [1.5 p] of the ethylene
Example: Uniaxial Deformation. With Axi-symmetric sample cross-section dl l 0 l x. α x since α x α y α z = 1 Rewriting ΔS α ) explicitly in terms of α
 Eample: Uniaial Deformation y α With Ai-symmetric sample cross-section l dl l 0 l, d Deform along, α = α = l0 l0 = α, α y = α z = Poisson contraction in lateral directions α since α α y α z = Rewriting
Eample: Uniaial Deformation y α With Ai-symmetric sample cross-section l dl l 0 l, d Deform along, α = α = l0 l0 = α, α y = α z = Poisson contraction in lateral directions α since α α y α z = Rewriting
File ISM02. Dynamics of Soft Matter
 File ISM02 Dynamics of Soft Matter 1 Modes of dynamics Quantum Dynamics t: fs-ps, x: 0.1 nm (seldom of importance for soft matter) Molecular Dynamics t: ps µs, x: 1 10 nm Brownian Dynamics t: ns>ps, x:
File ISM02 Dynamics of Soft Matter 1 Modes of dynamics Quantum Dynamics t: fs-ps, x: 0.1 nm (seldom of importance for soft matter) Molecular Dynamics t: ps µs, x: 1 10 nm Brownian Dynamics t: ns>ps, x:
CHARGED POLYMERS THE STORY SO FAR
 CHARGED POLYMERS THE STORY SO FAR Andrey V Dobrynin Institute of Materials Science &Department of Physics University of Connecticut What are polyelectrolytes? Poly(styrene sulfonate) CH-CH 2 SO Na Poly(methacrylic
CHARGED POLYMERS THE STORY SO FAR Andrey V Dobrynin Institute of Materials Science &Department of Physics University of Connecticut What are polyelectrolytes? Poly(styrene sulfonate) CH-CH 2 SO Na Poly(methacrylic
CARBON 2004 Providence, Rhode Island. Adsorption of Flexible n-butane and n-hexane on Graphitized Thermal Carbon Black and in Slit Pores
 CARBON Providence, Rhode Island Adsorption of Flexible n-butane and n-hexane on Graphitized Thermal Carbon Black and in Slit Pores D. D. Do* and H. D. Do, University of Queensland, St. Lucia, Qld 7, Australia
CARBON Providence, Rhode Island Adsorption of Flexible n-butane and n-hexane on Graphitized Thermal Carbon Black and in Slit Pores D. D. Do* and H. D. Do, University of Queensland, St. Lucia, Qld 7, Australia
Coil to Globule Transition: This follows Giant Molecules by Alexander Yu. Grosberg and Alexei R. Khokhlov (1997).
 Coil to Globule Transition: This follows Giant Molecules by Alexander Yu. Grosberg and Alexei R. Khokhlov (1997). The Flory Krigbaum expression for the free energy of a self-avoiding chain is given by,
Coil to Globule Transition: This follows Giant Molecules by Alexander Yu. Grosberg and Alexei R. Khokhlov (1997). The Flory Krigbaum expression for the free energy of a self-avoiding chain is given by,
Kinetics of layer hopping in a diblock copolymer lamellar phase
 Eur. Phys. J. E 27, 407 411 (2008) DOI 10.1140/epje/i2008-10402-8 THE EUROPEAN PHYSICAL JOURNAL E Kinetics of layer hopping in a diblock copolymer lamellar phase A.B. Croll 1,M.W.Matsen 2, A.-C. Shi 1,
Eur. Phys. J. E 27, 407 411 (2008) DOI 10.1140/epje/i2008-10402-8 THE EUROPEAN PHYSICAL JOURNAL E Kinetics of layer hopping in a diblock copolymer lamellar phase A.B. Croll 1,M.W.Matsen 2, A.-C. Shi 1,
PHASE TRANSITIONS IN SOFT MATTER SYSTEMS
 OUTLINE: Topic D. PHASE TRANSITIONS IN SOFT MATTER SYSTEMS Definition of a phase Classification of phase transitions Thermodynamics of mixing (gases, polymers, etc.) Mean-field approaches in the spirit
OUTLINE: Topic D. PHASE TRANSITIONS IN SOFT MATTER SYSTEMS Definition of a phase Classification of phase transitions Thermodynamics of mixing (gases, polymers, etc.) Mean-field approaches in the spirit
Applicable Simulation Methods for Directed Self-Assembly -Advantages and Disadvantages of These Methods
 Review Applicable Simulation Methods for Directed Self-Assembly -Advantages and Disadvantages of These Methods Hiroshi Morita Journal of Photopolymer Science and Technology Volume 26, Number 6 (2013) 801
Review Applicable Simulation Methods for Directed Self-Assembly -Advantages and Disadvantages of These Methods Hiroshi Morita Journal of Photopolymer Science and Technology Volume 26, Number 6 (2013) 801
Computer simulation methods (2) Dr. Vania Calandrini
 Computer simulation methods (2) Dr. Vania Calandrini in the previous lecture: time average versus ensemble average MC versus MD simulations equipartition theorem (=> computing T) virial theorem (=> computing
Computer simulation methods (2) Dr. Vania Calandrini in the previous lecture: time average versus ensemble average MC versus MD simulations equipartition theorem (=> computing T) virial theorem (=> computing
Final Morphology of Complex Materials
 120314 Final Morphology of Complex Materials 1) Proteins are the prototypical model for hierarchy. a) Give the generic chemical structure for an amino acid and a protein molecule (a tripeptide). b) Label
120314 Final Morphology of Complex Materials 1) Proteins are the prototypical model for hierarchy. a) Give the generic chemical structure for an amino acid and a protein molecule (a tripeptide). b) Label
V = 2ze 2 n. . a. i=1
 IITS: Statistical Physics in Biology Assignment # 3 KU Leuven 5/29/2013 Coulomb Interactions & Polymers 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions
IITS: Statistical Physics in Biology Assignment # 3 KU Leuven 5/29/2013 Coulomb Interactions & Polymers 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions
MONTE CARLO DYNAMICS OF DIAMOND-LATTICE MULTICHAIN SYSTEMS
 241 MONTE CARLO DYNAMICS OF DIAMOND-LATTICE MULTICHAIN SYSTEMS Andrzej Kolinski,* Jeffrey Skolnick t and Robert Yaris Department of Chemistry, Washington University, St. Louis, MO 63130 ABSTRACT We present
241 MONTE CARLO DYNAMICS OF DIAMOND-LATTICE MULTICHAIN SYSTEMS Andrzej Kolinski,* Jeffrey Skolnick t and Robert Yaris Department of Chemistry, Washington University, St. Louis, MO 63130 ABSTRACT We present
ICCP Project 2 - Advanced Monte Carlo Methods Choose one of the three options below
 ICCP Project 2 - Advanced Monte Carlo Methods Choose one of the three options below Introduction In statistical physics Monte Carlo methods are considered to have started in the Manhattan project (1940
ICCP Project 2 - Advanced Monte Carlo Methods Choose one of the three options below Introduction In statistical physics Monte Carlo methods are considered to have started in the Manhattan project (1940
2 Structure. 2.1 Coulomb interactions
 2 Structure 2.1 Coulomb interactions While the information needed for reproduction of living systems is chiefly maintained in the sequence of macromolecules, any practical use of this information must
2 Structure 2.1 Coulomb interactions While the information needed for reproduction of living systems is chiefly maintained in the sequence of macromolecules, any practical use of this information must
Atoms & Their Interactions
 Lecture 2 Atoms & Their Interactions Si: the heart of electronic materials Intel, 300mm Si wafer, 200 μm thick and 48-core CPU ( cloud computing on a chip ) Twin Creeks Technologies, San Jose, Si wafer,
Lecture 2 Atoms & Their Interactions Si: the heart of electronic materials Intel, 300mm Si wafer, 200 μm thick and 48-core CPU ( cloud computing on a chip ) Twin Creeks Technologies, San Jose, Si wafer,
arxiv:cond-mat/ v1 2 Feb 94
 cond-mat/9402010 Properties and Origins of Protein Secondary Structure Nicholas D. Socci (1), William S. Bialek (2), and José Nelson Onuchic (1) (1) Department of Physics, University of California at San
cond-mat/9402010 Properties and Origins of Protein Secondary Structure Nicholas D. Socci (1), William S. Bialek (2), and José Nelson Onuchic (1) (1) Department of Physics, University of California at San
Orientation and Phase Behavior of Block Copolymers in External Electric Fields
 Orientation and Phase Behavior of Block Copolymers in External Electric Fields DISSERTATION zur Erlangung des akademischen Grades eines Doktors der Naturwissenschaften - Dr. rer. nat. - der Fakultät Biologie,
Orientation and Phase Behavior of Block Copolymers in External Electric Fields DISSERTATION zur Erlangung des akademischen Grades eines Doktors der Naturwissenschaften - Dr. rer. nat. - der Fakultät Biologie,
 Title Super- and subcritical hydration of Thermodynamics of hydration Author(s) Matubayasi, N; Nakahara, M Citation JOURNAL OF CHEMICAL PHYSICS (2000), 8109 Issue Date 2000-05-08 URL http://hdl.handle.net/2433/50350
Title Super- and subcritical hydration of Thermodynamics of hydration Author(s) Matubayasi, N; Nakahara, M Citation JOURNAL OF CHEMICAL PHYSICS (2000), 8109 Issue Date 2000-05-08 URL http://hdl.handle.net/2433/50350
Critical Temperature - the temperature above which the liquid state of a substance no longer exists regardless of the pressure.
 Critical Temperature - the temperature above which the liquid state of a substance no longer exists regardless of the pressure. Critical Pressure - the vapor pressure at the critical temperature. Properties
Critical Temperature - the temperature above which the liquid state of a substance no longer exists regardless of the pressure. Critical Pressure - the vapor pressure at the critical temperature. Properties
Chapter 11. Intermolecular Forces and Liquids & Solids
 Chapter 11 Intermolecular Forces and Liquids & Solids The Kinetic Molecular Theory of Liquids & Solids Gases vs. Liquids & Solids difference is distance between molecules Liquids Molecules close together;
Chapter 11 Intermolecular Forces and Liquids & Solids The Kinetic Molecular Theory of Liquids & Solids Gases vs. Liquids & Solids difference is distance between molecules Liquids Molecules close together;
Equilibrium sampling of self-associating polymer solutions: A parallel selective tempering approach
 THE JOURNAL OF CHEMICAL PHYSICS 123, 124912 2005 Equilibrium sampling of self-associating polymer solutions: A parallel selective tempering approach Chakravarthy Ayyagari, a Dmitry Bedrov, and Grant D.
THE JOURNAL OF CHEMICAL PHYSICS 123, 124912 2005 Equilibrium sampling of self-associating polymer solutions: A parallel selective tempering approach Chakravarthy Ayyagari, a Dmitry Bedrov, and Grant D.
Lecture 6 - Bonding in Crystals
 Lecture 6 onding in Crystals inding in Crystals (Kittel Ch. 3) inding of atoms to form crystals A crystal is a repeated array of atoms Why do they form? What are characteristic bonding mechanisms? How
Lecture 6 onding in Crystals inding in Crystals (Kittel Ch. 3) inding of atoms to form crystals A crystal is a repeated array of atoms Why do they form? What are characteristic bonding mechanisms? How
8.592J HST.452J: Statistical Physics in Biology
 Assignment # 4 8.592J HST.452J: Statistical Physics in Biology Coulomb Interactions 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions scales as Q 2 E c.
Assignment # 4 8.592J HST.452J: Statistical Physics in Biology Coulomb Interactions 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions scales as Q 2 E c.
Structure, dynamics and heterogeneity: solid-state NMR of polymers. Jeremy Titman, School of Chemistry, University of Nottingham
 Structure, dynamics and heterogeneity: solid-state NMR of polymers Jeremy Titman, School of Chemistry, University of Nottingham Structure, dynamics and heterogeneity Structure Dynamics conformation, tacticity,
Structure, dynamics and heterogeneity: solid-state NMR of polymers Jeremy Titman, School of Chemistry, University of Nottingham Structure, dynamics and heterogeneity Structure Dynamics conformation, tacticity,
Chapter 10. Liquids and Solids
 Chapter 10 Liquids and Solids Chapter 10 Table of Contents 10.1 Intermolecular Forces 10.2 The Liquid State 10.3 An Introduction to Structures and Types of Solids 10.4 Structure and Bonding in Metals 10.5
Chapter 10 Liquids and Solids Chapter 10 Table of Contents 10.1 Intermolecular Forces 10.2 The Liquid State 10.3 An Introduction to Structures and Types of Solids 10.4 Structure and Bonding in Metals 10.5
Fluctuations in polymer blends
 Fluctuations in polymer blends Dominik Düchs and Friederike Schmid Fakultät für Physik, Universität Bielefeld, Postfach 100131, 33501 Bielefeld E-mail: {schmid, duechs}@physik.uni-bielefeld.de We have
Fluctuations in polymer blends Dominik Düchs and Friederike Schmid Fakultät für Physik, Universität Bielefeld, Postfach 100131, 33501 Bielefeld E-mail: {schmid, duechs}@physik.uni-bielefeld.de We have
ON ALGORITHMS FOR BROWNIAN DYNAMICS COMPUTER SIMULATIONS
 COMPUTATIONAL METHODS IN SCIENCE AND TECHNOLOGY 4,35-42 (1998) ON ALGORITHMS FOR BROWNIAN DYNAMICS COMPUTER SIMULATIONS ARKADIUSZ C. BRAŃKA Institute of Molecular Physics, Polish Academy of Sciences, Smoluchowskiego
COMPUTATIONAL METHODS IN SCIENCE AND TECHNOLOGY 4,35-42 (1998) ON ALGORITHMS FOR BROWNIAN DYNAMICS COMPUTER SIMULATIONS ARKADIUSZ C. BRAŃKA Institute of Molecular Physics, Polish Academy of Sciences, Smoluchowskiego
Fluctuation effects in block copolymer melts
 JOURAL OF CHEMICAL PHYSICS VOLUME 118, UMBER 16 22 APRIL 2003 Fluctuation effects in block copolymer melts O.. Vassiliev a) and M. W. Matsen b) Polymer Science Centre, University of Reading, Whiteknights,
JOURAL OF CHEMICAL PHYSICS VOLUME 118, UMBER 16 22 APRIL 2003 Fluctuation effects in block copolymer melts O.. Vassiliev a) and M. W. Matsen b) Polymer Science Centre, University of Reading, Whiteknights,
Chapter 21. Electric Fields
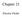 Chapter 21 Electric Fields The Origin of Electricity The electrical nature of matter is inherent in the atoms of all substances. An atom consists of a small relatively massive nucleus that contains particles
Chapter 21 Electric Fields The Origin of Electricity The electrical nature of matter is inherent in the atoms of all substances. An atom consists of a small relatively massive nucleus that contains particles
Structural properties of low-density liquid alkali metals
 PRAMANA c Indian Academy of Sciences Vol. 65, No. 6 journal of December 2005 physics pp. 1085 1096 Structural properties of low-density liquid alkali metals A AKANDE 1, G A ADEBAYO 1,2 and O AKINLADE 2
PRAMANA c Indian Academy of Sciences Vol. 65, No. 6 journal of December 2005 physics pp. 1085 1096 Structural properties of low-density liquid alkali metals A AKANDE 1, G A ADEBAYO 1,2 and O AKINLADE 2
Gas-liquid phase separation in oppositely charged colloids: stability and interfacial tension
 7 Gas-liquid phase separation in oppositely charged colloids: stability and interfacial tension We study the phase behaviour and the interfacial tension of the screened Coulomb (Yukawa) restricted primitive
7 Gas-liquid phase separation in oppositely charged colloids: stability and interfacial tension We study the phase behaviour and the interfacial tension of the screened Coulomb (Yukawa) restricted primitive
Phase-separation dynamics of a ternary mixture coupled with reversible chemical reaction
 JOURNAL OF CHEMICAL PHYSICS VOLUME 6, NUMBER 4 JANUARY 00 Phase-separation dynamics of a ternary mixture coupled with reversible chemical reaction Chaohui Tong and Yuliang Yang a) Department of Macromolecular
JOURNAL OF CHEMICAL PHYSICS VOLUME 6, NUMBER 4 JANUARY 00 Phase-separation dynamics of a ternary mixture coupled with reversible chemical reaction Chaohui Tong and Yuliang Yang a) Department of Macromolecular
Using Molecular Dynamics to Compute Properties CHEM 430
 Using Molecular Dynamics to Compute Properties CHEM 43 Heat Capacity and Energy Fluctuations Running an MD Simulation Equilibration Phase Before data-collection and results can be analyzed the system
Using Molecular Dynamics to Compute Properties CHEM 43 Heat Capacity and Energy Fluctuations Running an MD Simulation Equilibration Phase Before data-collection and results can be analyzed the system
Molecular dynamics study of isobaric and isochoric glass transitions in a model amorphous polymer
 JOURNAL OF CHEMICAL PHYSICS VOLUME 110, NUMBER 14 8 APRIL 1999 Molecular dynamics study of isobaric and isochoric glass transitions in a model amorphous polymer Liu Yang, a) David J. Srolovitz, and Albert
JOURNAL OF CHEMICAL PHYSICS VOLUME 110, NUMBER 14 8 APRIL 1999 Molecular dynamics study of isobaric and isochoric glass transitions in a model amorphous polymer Liu Yang, a) David J. Srolovitz, and Albert
Hydrogels in charged solvents
 Hydrogels in charged solvents Peter Košovan, Christian Holm, Tobias Richter 27. May 2013 Institute for Computational Physics Pfaffenwaldring 27 D-70569 Stuttgart Germany Tobias Richter (ICP, Stuttgart)
Hydrogels in charged solvents Peter Košovan, Christian Holm, Tobias Richter 27. May 2013 Institute for Computational Physics Pfaffenwaldring 27 D-70569 Stuttgart Germany Tobias Richter (ICP, Stuttgart)
A Molecular Dynamics Simulation of a Homogeneous Organic-Inorganic Hybrid Silica Membrane
 A Molecular Dynamics Simulation of a Homogeneous Organic-Inorganic Hybrid Silica Membrane Supplementary Information: Simulation Procedure and Physical Property Analysis Simulation Procedure The molecular
A Molecular Dynamics Simulation of a Homogeneous Organic-Inorganic Hybrid Silica Membrane Supplementary Information: Simulation Procedure and Physical Property Analysis Simulation Procedure The molecular
Materials 218/UCSB: Class III Cohesion in solids van der Waals, ionic, covalent, metallic
 Materials 218/UCSB: Class III Cohesion in solids van der Waals, ionic, covalent, metallic Ram Seshadri (seshadri@mrl.ucsb.edu) Introduction There are four forces in nature. The strong and the weak interactions
Materials 218/UCSB: Class III Cohesion in solids van der Waals, ionic, covalent, metallic Ram Seshadri (seshadri@mrl.ucsb.edu) Introduction There are four forces in nature. The strong and the weak interactions
Random isotropic structures and possible glass transitions in diblock copolymer melts
 33 Chapter 3 Random isotropic structures and possible glass transitions in diblock copolymer melts This chapter is reprinted from our paper, C.-Z. Zhang and Z.-G. Wang Phys. Rev. E 73, 031804 (006). Copyright
33 Chapter 3 Random isotropic structures and possible glass transitions in diblock copolymer melts This chapter is reprinted from our paper, C.-Z. Zhang and Z.-G. Wang Phys. Rev. E 73, 031804 (006). Copyright
What is Classical Molecular Dynamics?
 What is Classical Molecular Dynamics? Simulation of explicit particles (atoms, ions,... ) Particles interact via relatively simple analytical potential functions Newton s equations of motion are integrated
What is Classical Molecular Dynamics? Simulation of explicit particles (atoms, ions,... ) Particles interact via relatively simple analytical potential functions Newton s equations of motion are integrated
φ(z) Application of SCF to Surfaces and Interfaces (abridged from notes by D.J. Irvine)
 Application of SCF to Surfaces and Interfaces (abridged from notes by D.J. Irvine) Edwards continuum field theory reviewed above is just one flavor of selfconsistent mean field theory, but all mean field
Application of SCF to Surfaces and Interfaces (abridged from notes by D.J. Irvine) Edwards continuum field theory reviewed above is just one flavor of selfconsistent mean field theory, but all mean field
Hierarchy in Block Copolymer Morphology (Web report) MANGESH CHAMPHEKAR (Materials Science and Engg.)
 Hierarchy in Block Copolymer Morphology (Web report) By MANGESH CHAMPHEKAR (Materials Science and Engg.) ABSTRACT In the recent years, the study of block copolymers has received special attention from
Hierarchy in Block Copolymer Morphology (Web report) By MANGESH CHAMPHEKAR (Materials Science and Engg.) ABSTRACT In the recent years, the study of block copolymers has received special attention from
Coarse-grained molecular dynamics study of block copolymer/nanoparticle composites under elongational flow
 THE JOURNAL OF CHEMICAL PHYSICS 131, 214904 2009 Coarse-grained molecular dynamics study of block copolymer/nanoparticle composites under elongational flow Vibha Kalra and Yong Lak Joo a School of Chemical
THE JOURNAL OF CHEMICAL PHYSICS 131, 214904 2009 Coarse-grained molecular dynamics study of block copolymer/nanoparticle composites under elongational flow Vibha Kalra and Yong Lak Joo a School of Chemical
Chapter 5: Molecular Scale Models for Macroscopic Dynamic Response. Fluctuation-Dissipation Theorem:
 G. R. Strobl, Chapter 6 "The Physics of Polymers, 2'nd Ed." Springer, NY, (1997). R. B. Bird, R. C. Armstrong, O. Hassager, "Dynamics of Polymeric Liquids", Vol. 2, John Wiley and Sons (1977). M. Doi,
G. R. Strobl, Chapter 6 "The Physics of Polymers, 2'nd Ed." Springer, NY, (1997). R. B. Bird, R. C. Armstrong, O. Hassager, "Dynamics of Polymeric Liquids", Vol. 2, John Wiley and Sons (1977). M. Doi,
Class XII Chapter 1 Electric Charges And Fields Physics
 Class XII Chapter 1 Electric Charges And Fields Physics Question 1.1: What is the force between two small charged spheres having charges of 2 10 7 C and 3 10 7 C placed 30 cm apart in air? Answer: Repulsive
Class XII Chapter 1 Electric Charges And Fields Physics Question 1.1: What is the force between two small charged spheres having charges of 2 10 7 C and 3 10 7 C placed 30 cm apart in air? Answer: Repulsive
Molecular Dynamics Simulations of Glass Formation and Crystallization in Binary Liquid Metals
 Citation & Copyright (to be inserted by the publisher ) Molecular Dynamics Simulations of Glass Formation and Crystallization in Binary Liquid Metals Hyon-Jee Lee 1,2, Tahir Cagin 2, William A. Goddard
Citation & Copyright (to be inserted by the publisher ) Molecular Dynamics Simulations of Glass Formation and Crystallization in Binary Liquid Metals Hyon-Jee Lee 1,2, Tahir Cagin 2, William A. Goddard
Scientific Computing II
 Scientific Computing II Molecular Dynamics Simulation Michael Bader SCCS Summer Term 2015 Molecular Dynamics Simulation, Summer Term 2015 1 Continuum Mechanics for Fluid Mechanics? Molecular Dynamics the
Scientific Computing II Molecular Dynamics Simulation Michael Bader SCCS Summer Term 2015 Molecular Dynamics Simulation, Summer Term 2015 1 Continuum Mechanics for Fluid Mechanics? Molecular Dynamics the
Spins and spin-orbit coupling in semiconductors, metals, and nanostructures
 B. Halperin Spin lecture 1 Spins and spin-orbit coupling in semiconductors, metals, and nanostructures Behavior of non-equilibrium spin populations. Spin relaxation and spin transport. How does one produce
B. Halperin Spin lecture 1 Spins and spin-orbit coupling in semiconductors, metals, and nanostructures Behavior of non-equilibrium spin populations. Spin relaxation and spin transport. How does one produce
201. The Nature o f the Metallic Bond. III
 No. 8] 913 201. The Nature o f the Metallic Bond. III Atomic Interactions in Alloys. I By San-ichiro MIZUSHIMA, M.J.A., and Isao Ichishima Tokyo Research Institute, Yawata Iron and Steel Co. Ltd., Ida,
No. 8] 913 201. The Nature o f the Metallic Bond. III Atomic Interactions in Alloys. I By San-ichiro MIZUSHIMA, M.J.A., and Isao Ichishima Tokyo Research Institute, Yawata Iron and Steel Co. Ltd., Ida,
Computer Simulation of Liquid Crystal Polymer Systems with Complex Topology
 Computer Simulation of Liquid Crystal Polymer Systems with Complex Topology Anna Zarembo Laboratory of Polymer Chemistry Department of Chemistry University of Helsinki Finland Academic Dissertation To
Computer Simulation of Liquid Crystal Polymer Systems with Complex Topology Anna Zarembo Laboratory of Polymer Chemistry Department of Chemistry University of Helsinki Finland Academic Dissertation To
Dissipative particle dynamics: Bridging the gap between atomistic and mesoscopic simulation
 Dissipative particle dynamics: Bridging the gap between atomistic and mesoscopic simulation Robert D. Groot and Patrick B. Warren Unilever Research Port Sunlight Laboratory, Quarry Road East, Bebington,
Dissipative particle dynamics: Bridging the gap between atomistic and mesoscopic simulation Robert D. Groot and Patrick B. Warren Unilever Research Port Sunlight Laboratory, Quarry Road East, Bebington,
Linear Melt Rheology and Small-Angle X-ray Scattering of AB Diblocks vs A 2 B 2 Four Arm Star Block Copolymers
 Macromolecules 2000, 33, 8399-8414 8399 Linear Melt Rheology and Small-Angle X-ray Scattering of AB Diblocks vs A 2 B 2 Four Arm Star Block Copolymers D. M. A. Buzza,*,, A. H. Fzea,, J. B. Allgaier, R.
Macromolecules 2000, 33, 8399-8414 8399 Linear Melt Rheology and Small-Angle X-ray Scattering of AB Diblocks vs A 2 B 2 Four Arm Star Block Copolymers D. M. A. Buzza,*,, A. H. Fzea,, J. B. Allgaier, R.
SIMULATIONAL ANALYSIS OF GLASSES PREPARED VIA DIFFERENT INTERATOMIC POTENTIALS
 Journal of Optoelectronics and Advanced Materials Vol. 7, No. 4, August 2005, p. 1915-1922 SIMULATIONAL ANALYSIS OF GLASSES PREPARED VIA DIFFERENT INTERATOMIC POTENTIALS Y. Sano, J. K ga, F. Yonezawa Department
Journal of Optoelectronics and Advanced Materials Vol. 7, No. 4, August 2005, p. 1915-1922 SIMULATIONAL ANALYSIS OF GLASSES PREPARED VIA DIFFERENT INTERATOMIC POTENTIALS Y. Sano, J. K ga, F. Yonezawa Department
Atoms, electrons and Solids
 Atoms, electrons and Solids Shell model of an atom negative electron orbiting a positive nucleus QM tells that to minimize total energy the electrons fill up shells. Each orbit in a shell has a specific
Atoms, electrons and Solids Shell model of an atom negative electron orbiting a positive nucleus QM tells that to minimize total energy the electrons fill up shells. Each orbit in a shell has a specific
Multiple time step Monte Carlo
 JOURNAL OF CHEMICAL PHYSICS VOLUME 117, NUMBER 18 8 NOVEMBER 2002 Multiple time step Monte Carlo Balázs Hetényi a) Department of Chemistry, Princeton University, Princeton, NJ 08544 and Department of Chemistry
JOURNAL OF CHEMICAL PHYSICS VOLUME 117, NUMBER 18 8 NOVEMBER 2002 Multiple time step Monte Carlo Balázs Hetényi a) Department of Chemistry, Princeton University, Princeton, NJ 08544 and Department of Chemistry
Amorphous Polymers: Polymer Conformation Laboratory 1: Module 1
 D E P A R T M E N T O F M A T E R I A L S S C I E N C E A N D E N G I N E E R I N G M A S S A C H U S E T T S I N S T I T U T E O F T E C H N O L O G Y 3.014 Materials Laboratory Fall 2008 Amorphous Polymers:
D E P A R T M E N T O F M A T E R I A L S S C I E N C E A N D E N G I N E E R I N G M A S S A C H U S E T T S I N S T I T U T E O F T E C H N O L O G Y 3.014 Materials Laboratory Fall 2008 Amorphous Polymers:
Depletion Attraction of Sheet-like Ion Aggregates in Low-Dielectric Ionomer Melts Keran Lu, 1 Janna K. Maranas, 1 1, a)
 Sample Title Depletion Attraction of Sheet-like Ion Aggregates in Low-Dielectric Ionomer Melts Keran Lu, 1 Janna K. Maranas, 1 1, a) and Scott T. Milner The Pennsylvania State University 12 Fenske Laboratory,
Sample Title Depletion Attraction of Sheet-like Ion Aggregates in Low-Dielectric Ionomer Melts Keran Lu, 1 Janna K. Maranas, 1 1, a) and Scott T. Milner The Pennsylvania State University 12 Fenske Laboratory,
Chapter 6: The Rouse Model. The Bead (friction factor) and Spring (Gaussian entropy) Molecular Model:
 G. R. Strobl, Chapter 6 "The Physics of Polymers, 2'nd Ed." Springer, NY, (1997). R. B. Bird, R. C. Armstrong, O. Hassager, "Dynamics of Polymeric Liquids", Vol. 2, John Wiley and Sons (1977). M. Doi,
G. R. Strobl, Chapter 6 "The Physics of Polymers, 2'nd Ed." Springer, NY, (1997). R. B. Bird, R. C. Armstrong, O. Hassager, "Dynamics of Polymeric Liquids", Vol. 2, John Wiley and Sons (1977). M. Doi,
The dynamics of order order phase separation
 IOP PUBLISHING JOURNAL OF PHYSICS: CONDENSED MATTER J. Phys.: Condens. Matter 0 (008) 155107 (10pp) doi:10.1088/0953-8984/0/15/155107 The dynamics of order order phase separation K Yamada and S Komura
IOP PUBLISHING JOURNAL OF PHYSICS: CONDENSED MATTER J. Phys.: Condens. Matter 0 (008) 155107 (10pp) doi:10.1088/0953-8984/0/15/155107 The dynamics of order order phase separation K Yamada and S Komura
