Electrochemical Reduction of 1-Aryl-4-nitroazoles
|
|
|
- Kelley Fletcher
- 6 years ago
- Views:
Transcription
1 Polish J.Chem., 74, (2000) Electrochemical Reduction of 1-Aryl-4-nitroazoles by A. Tallec 1, R. azard 1, J. Suwiñski 2* and P. Wagner 2 1 Laboratoire d Electrochimie, Université de Rennes 1, Campus de Beaulieu, Rennes Cedex, France 2 Institute of Organic Chemistry and Technology, Silesian University of Technology, Krzywoustego 4, Gliwice, Poland, * suwinski@polsl.gliwice.pl (Received March 20th, 2000) Electrochemical reduction of the nitro group in 1-aryl-4-nitroazoles occurs in slightly acidic medium as a typical four-electron process leading to hydroxylamine group formation. The forming 1-aryl-4-hydroxylaminoazoles undergo further reactions. Stability of the hydroxylamines depends on the heteroring (imidazole or pyrazole) and on C-substituents. More stable derivatives condense with the nitroso intermediates to yield azoxycompounds. Less stable hydroxylamines undergo rearrangements, azole ring reduction and decomposition. Some products of these transformations, following the electrochemical reduction, were isolated and their structures were fully characterized by standard methods. Key words: nitroazoles, electroreduction, mechanisms Reduction of nitroazoles is important because of their activity in biological systems. Moreover, metabolic reduction of the nitro group is responsible for the toxicity of nitro compounds [1]. Many studies have reported on radiosensitizing efficiency of nitroazoles in cancer therapy related to their high reduction potentials [2]. It is known that nitroazoles follow the general pattern of other aromatic nitro compounds reduction. Therefore, the nitro group is reduced to the amino group via the nitroso and hydroxylamine groups. Electrochemical (polarographic or voltammetric) reduction of the nitro group occurs in slightly acidic medium as a four-electron process, leading to the hydroxylamine group formation (wave 1). A possible condensation of the nitroso and hydroxylamino derivatives yields azoxycompounds that can be further reduced. Moreover, in an acidic medium the protonated hydroxylamino group can undergo a two-electron reduction, leading to a formation of the amino group (wave 2) [3]. Several hydroxylamino- and aminoazoles are unstable and decompose in the reaction media to ring opening products [4]. Thus, products of 4-nitroazoles electrochemical reduction have been isolated in a few cases only. umerous papers concern the electroreduction of 1-alkyl-4-nitroazoles [5], none of them describing the electrochemical behavior of 4-nitroazoles with -aryl substituent yet. This paper intends to fill this gap. For a long time 1-aryl-4-nitroazoles, particularly 1-phenyl-4-nitroimidazoles have not been available. Recently, we have * Author for correspondence
2 1178 A. Tallec et al. elaborated a very efficient method for their synthesis. It involves a treatment of 1,4-dinitroimidazoles with aniline or its C-substituted derivatives in aqueous-methanol solution at room temperature [6]. Some 1-aryl-4-nitroimidazoles have been tested as radiosensitizers of hypoxic tumor cells, showing a higher efficiency as compared with the corresponding 1-alkyl derivatives [7]. Since biological toxicity and radiosensitizing efficiency of nitro compounds involve their reduction and depend on their reduction potentials, we have studied the polarographic and voltammetric behaviors of some 4-nitro-1-phenylazoles. EXPERIMETAL Synthesis of 4-nitroazoles: Starting 1-aryl-4-nitroimidazoles 1a c were obtained according to [6]. Properties of the compounds were in accordance with those reported there. Synthesis of 5-cyano-2-methyl-4-nitro and of 5-carbamoyl-2-methyl-4-nitro (1d e) we also described earlier [11]. 1-Benzyl-4-nitroimidazole (1f, m.p C) we obtained by alkylation of 4(5)-nitroimidazole with benzyl bromide using a known method [13]. 1-enyl-4-nitropyrazole (1f, m.p C) was prepared according to [6]. Polarographic and voltammetric measurements: Polarograms were recorded at a dropping mercury electrode (dropping time = 2 s) using a three-electrode PAR 362 potentiostat coupled with a XY Kipp & Zonen recorder. The starting material concentration in a mixture of aqueous supporting electrolyte and ethanol (1:1) was 10 3 mole/dm 3. The compositions of three supporting electrolytes were: sulfuric acid (0.5 mole/dm 3 ), acetate buffer (acetic acid 0.5 mole/dm 3, sodium acetate 0.5 mole/dm 3 )or hydrochloric acid (3 mole/dm 3 ). Voltammetric curves were recorded at a Metrohm MDE with the same apparatus as above at a scan rate of 0.2 or 0.5 Vs 1. Preparative electrolyses: Preparative electrolyses were carried out at a mercury pool electrode in the cell described in [15]. The working potential was imposed by a Tacussel PRT potentiostat, while the reference electrode was a saturated calomel electrode (SCE). An amount of electricity was measured with Tacussel IG5 coulometer. For a typical run, the starting material (10 2 mole) was dissolved in 120 cm 3 of the catolyte (aqueous supporting electrolyte and ethanol 1:1); electrolysis was then performed under continuous nitrogen flow. The post-reaction mixtures were neutralized with a saturated sodium bicarbonate solution. Ethanol was evaporated then under diminished pressure at ca. 50 C. Possible precipitations were collected and the filtrate extracted three times with methylene chloride. The extracts were dried by anhydrous magnesium sulfate(vi) and the solvent was evaporated under diminished pressure at 50 C. The residue and precipitations were combined and recrystallized from suitable solvents as indicated below. Characteristic of the isolated products: 4-Imino-1-phenyl-5-imidazolidinone yield 66%; m.p. (ethanol-water) C; 1 MR (ppm, chloroform-d 1 ): (m, 5, Ar), 5.46 (br. s, 2, 2 ), 5.27 (s, 2, C 2 ); 13 C MR (ppm, chloroform-d 1 ): , , , , , , 69.12; RMS 70 ev for C O requires: , found: ; MS 70 ev (m/e, %) M + : 175 (49.52), 119 (2.50), 104 (5.14), 77 (9.90), 56 (100.00). 4-Imino-1-benzyl-5-imidazolidinone yield 54%; m.p. (ethanol-water) C; 1 MR (ppm, chloroform-d 1 ): (m, 5, Ar), 5.17 (br. s, 2, 2 ), 4.58 (s, 2, C 2 ), 4.50 (s, 2, C 2 ); 13 C MR (ppm, chloroform-d 1 ): , , , , , , 68.38, 46.06; MS 70 ev (m/e, %) M + : 189 (90.9), 106 (46.4), 91 (100.0), 57 (78.3); Found for C O: C 63.70; 5.72, 22, requires: 13C 63.48, 5.86, ,4 -Azoxy-5,5 -dicyano-2,2 -dimethyl-1,1 -diphenyl-imidazole yield 93%; m.p. (ethanol) 265 C dec.; 1 MR (ppm, chloroform-d 1 ): (m, 10, Ar), 1.93 (s, 6, C 3 ); MS 70 ev (m/e, %) M + : 408 (11.7), 364 (44.9), 363 (72.9), 223 (46.9), 155 (25.6), 118 (67.2), 77 (100.0), 52 (30.0); Found for C O: C 64.70, 3.95, 27, requires: 44C 64.70, 3.95, Cyano-4-hydroxylamino-2-methyl yield 40%; m.p. (methanol-water) 152 C dec.; 1 MR (ppm, DMSO-d 6 ): 8.43 (s, 2, O), (m, 5, Ar), 1.67 (s, 3, C 3 );
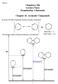
3 Electrochemical reduction of 1-aryl-4-nitroazoles 1179 MS 70eV (m/e, %) M + : 214 (32.8), 198 (90.5), 156 (67.7), 129 (37.0), 77 (100.0), 52 (58.2); Found for C O: C 61.97, 4.63, 26, requires: 28C 61.67, 4.71, Imino-2-methyl-1-phenylimidazolo[4,5-d]isoxazolinone yield 4.3%; m.p. (ethanol) C; 1 MR (ppm, DMSO-d 6 ): (br. s, 1, ), (s, 1, ), (m, 5, Ar), 2.28 (s, 3, C 3 ); 13 C MR (ppm, DMSO-d 6 ): , , , , , , , 20.84; Found for C O: C 61.61, 4.84, 26.15, requires: C 61.67, 4.71, Amino-5-chloro-1-phenylpyrazole yield 23%; m.p. (petroleum ether) C; 1 MR (ppm, chloroform-d 1 ): (m, 6, Ar + C); RMS 70 ev for C Cl requires: , found: ; MS 70 ev (m/e, %) M + : 193 (99.06), 166 (10.12), 131 (25.91), 104 (100.00), 77 (63.39), 51 (27.30). 4,4 -Azoxy-1,1 -diphenylpyrazole yield 60% [10]. 4,4 -Azoxy-5,5 -dicyano-1,1,3,3 -tetramethylpyrazole yield 67% [10]. 4-Acetylamino-5-hydroxy-1-phenylpyrazole yield 27%; m.p. (ethanol-water) C; 1 MR (ppm, DMSO-d 6 ): (br. s, 1, ), 9.92 (br. s, 1, O), (m, 6, Ar + C), 2.04 (s, 3, C 3 ); 13 C MR (ppm, DMSO-d 6 ): , , , , , 22.20; RMS 70 ev for C O 2 requires: found: ; MS 70 ev (m/e, %) M + : 217 (100.00), 199 (32.81), 175 (55.15), 120 (37.42), 77 (64.90), 43 (64.67). RESULTS AD DISCUSSIO Electrochemical investigations: Polarographic four-electron reduction potentials (E 1/2, wave 1) for the compounds mentioned were measured in the acetate buffer of p = 4.8 (Table). The reduction potentials measured are rather unexpectedly low, considering high toxicity and sensibilizing efficiency of the compounds. Moreover, E 1/2 for 1-benzyl-4-nitroimidazole is much lower ( = 0.12 V) than that of 4-nitro-. As it should be expected, a reduction potential increases with the increase in electron withdrawing character of substituents. Electron releasing effect of the methyl group on the reduction potential is stronger at C5 (1c) than at C2 (1b) position. We have not found E 1/2 polarographic potentials for the corresponding 1-methyl-4-nitroazoles in the acetate buffer of p = 4.8. Possible extrapolation of E 1/2 values, measured under different conditions, could have affected the comparison between 1-alkyl and 1-aryl derivatives. Cyclic voltammetric experiments have proved that 1-aryl-4-nitroimidazoles (1a e), in the acetate buffer - ethanol solution, are reduced indeed to the corresponding hydroxylamines (2a e) (Scheme 1, Figure). R' O 2 R Scheme 1 +4e O R' O 1 2 a: R=R =; b: R=C 3,R =; c: R=, R =C 3 ; d: R=C 3,R =C; e: R=C 3,R =CO 2 R
4 1180 A. Tallec et al. Table. Polarographic E 1/2 potentials for the four-electron reduction of 4-nitro-1-phenylazoles and of 1-benzyl-4-nitroimidazole in the acetate buffer-ethanol solution of p = 4.8. o. 4-nitroazole E 1/2 [V] o. 4-nitroazole E 1/2 [V] 1a 4-nitro d 5-cyano-2-methyl nitro 1b 2-methyl-4-nitro e 5-carbamoyl-2-methyl-4-nitro c 5-methyl-4-nitro f 1-benzyl-4-nitroimidazole nitro-1-phenylpyrazole 0.53 Figure. Cyclic voltammograms recorded for 10 3 M solutions of 1d, 1a, 1b, 1c in 0.5 M acetate buffer + EtO 1:1 by volume. Sweep scan: 200 mvs 1. owever, stability of 2 depends on C-substituents at the imidazole ring and decreases in the order 2d > 2a, 2f > 2b > 2c (see Fig.). Then, the most stable hydroxylamine is 2d, containing the electron withdrawing cyano group at C5 (the reversible system nitroso hydroxylamine is clearly observed at 0.2 Vs 1 ), while the least stable one is 2c (slight appearance of the nitroso hydroxylamine system only at 0.5 Vs 1 ). Electrochemical reduction of 4-nitroimidazoles: Chemical reduction of 1-aryl- -4-nitroimidazoles can be achieved in few ways: by hydrogenation on a metal catalyst [8], by a treatment with iron in carboxylic acid [9] or by the hydride reduction, e.g. with sodium borohydride [10]. In the first two reactions unstable 4-amino-1-arylimidazoles can be isolated only as -acyl derivatives. 4-Amino-5-cyano-2-methyl-
5 Electrochemical reduction of 1-aryl-4-nitroazoles 1181 Scheme 2 C O C 3 [O] C O C 3 2d 5d - 2 O C O C 3 7d 3 C C C O 6d C 3 can be separated as a free base [11]. The hydride reduction leads to 1-aryl-4-oximinoimidazolines; thus, the imidazole ring also undergoes reduction. In contrast to that, the hydride reduction of 1-aryl-4-nitropyrazoles yields azoxycompounds [8]. Those results show that 4-amino- and 4-hydroxylaminoimidazoles, not containing electron-withdrawing substituents at C5, are less stable than isomeric pyrazoles. Therefore, it is not surprising that our attempts of preparative electrochemical reduction of 4-nitros were only partly successful, while 4-nitro-1-phenylpyrazole (3) gave a corresponding azoxycompound in a good yield. Electroreduction of 1d in acetic buffer leads to the relatively stable hydroxylamine 2d; the latter, when left in solution under nitrogen for twelve hours, gives, besides some starting material, a mixture of the azoxy derivative 6d and the iminoisoxazolinone 7d (Scheme 2). The compound 7d probably arises from an intramolecular addition of the -hydroxy group to the neighbouring cyano substituent. The electroreduction of 1e leads to the corresponding hydroxylamine 2e, stable only when kept under nitrogen; nevertheless, it can be reduced at more negative potential into the corresponding amine already obtained by chemical reduction [11]. Moderately stable hydroxylamine 2a prepared in the acetate buffer underwent Gattermann-Bamberger rearrangement to yield 66% of 4-imino-1-phenyl-5- -imidazolidinone (8a). We observed a similar behavior for 2f obtained from 1f and rearranging to 1-benzyl-4-imino-5-imidazolidinone in 54% yield. Thus, the presence of an aryl substituent at 1 is not critical for stability of 4-hydroxylaminoimidazoles since 1-benzyl- and 1-phenyl- derivatives gave products of the same type (Scheme 3). At least, as expected from voltammetric investigations, no significant results have been obtained from electroreduction of compounds 1b and 1c.
6 1182 A. Tallec et al. Scheme 3 rear. O O O 2a 2 8a We also carried out the electrochemical reduction of 4-nitros in dilute sulfuric or hydrochloric acid. It led to the imidazole ring decomposition products with formation of aniline. Electrochemical reduction of 4-nitro-1-phenylpyrazole: Compound 3 was reduced in the acetate buffer to the corresponding hydroxylamine 4 that, when partly oxidized to the nitroso derivative, gave 1,1 -diphenyl-4,4 -azoxypyrazole in 60% yield. ydroxylamine 4, when left overnight in the reaction medium, underwent a partial transformation into 4-acetylamino-5-hydroxypyrazole (9). The mechanism of the transformation is not clear. A similar transformation has been reported for a benzene derivative [12]. One can assume that the acetate anion attacks a protonated molecule of 4 at C5. The attack is accompanied by a departure of the water molecule from the protonated hydroxylamino group. An unstable non-aromatic intermediate transforms into the final product through tautomerization and intramolecular transfer of the acetyl group from the oxygen to the exocyclic imine nitrogen atom (Scheme 4). Scheme 4 +4e + 4 O 2 3 O 4 2 O 3 CCOO 3 COO - 2 O O 2 O- 3 CO 9 4-itro-1-phenylpyrazole (3) reduced in 3 M hydrochloric acid yields 23% of 4-amino-5-chloro-1-phenylpyrazole (10) as the only isolated product. The formation of 10 can be rationalized as follows. A nucleophilic attack of the chloride anion on the protonated hydroxylamine 4, accompanied by departure of a molecule of water, leads to the formation of a non-aromatic intermediate. Tautomerization of the latter yields 10 (Scheme 5). The transformations of 4 in the acetate buffer and in hydrochloric acid seem to be alike in principle, though they yield different products.
7 Electrochemical reduction of 1-aryl-4-nitroazoles 1183 Scheme 5 Cl - 2 O Cl Cl 2 O 2 10 COCLUSIOS Concluding the results obtained we can say that a course of the four-electron electrochemical reduction of 4-nitroazoles in slightly acidic solution does not depend on a ring (imidazole or pyrazole) and on substituents at it. These structural factors affect only the stability of the corresponding hydroxylamines and therefore the structures of the products. This conclusion agrees with conclusions drawn by us from the results of chemical reduction of nitroazoles. REFERECES 1. oss M.B, Panicucci R., McClelland. R.A and Rauth A.M., Biochem.armacol., 37, 2585 (1988). 2. Adams G.E., Flockhart I.R., Smithen C.E.,Stratford I., Wardman P. and Watts M.E., Radiat. Res., 67, 9 (1976). 3. Lund. and Baizer M., Organic Electrochemistry, Marcel Dekker., Y, Katritzky A.R., Rees C.W. and Scriven E.F.V., Comprehensive eterocyclic Chemistry, Pergamon, Exeter, Dumanovic D., Jovanovic J., Suznjevic D., Erceg M. and Zuman P., Electroanalysis, 4, 871 (1992), and references cited therein. 6. Salwiñska E. and Suwiñski J., Polish J. Chem., 64, 813 (1990). 7. Suwiñski J., Szczepankiewicz W. and Wide³ M., Archiv. arm. (Weinheim, Germany), 325, 949 (1990). 8. Walczak K. and Suwiñski J., Polish J. Chem., 67, 691 (1993). 9. Suwiñski J. and Wagner P., unpublished results. 10. Suwiñski J., Wagner P. and olt E.M., Tetrahedron, 52, 9541 (1996). 11. Suwiñski J., Walczak K. and Wagner P., Polish J. Appl. Chem., 38, 499 (1994). 12. Le Guyeder M., Bull. Soc. Chim. Fr., 1867 (1966). 13. Cosar C., Crisan C., orclois R., Jacob R.M., Robert J., Tchelitcheff S. and Vaupre R., Arzn. Forsch., 16, 23 (1966). 14. Finer I.L. and urloch R.J., J. Chem. Soc., 3021 (1957). 15. Moinet C. and Peltier D., Bull. Soc. Chim. Fr., 690 (1969).
Chapter 23 Phenols CH. 23. Nomenclature. The OH group takes precedence as the parent phenol.
 CH. 23 Chapter 23 Phenols Nomenclature The OH group takes precedence as the parent phenol. Carboxyl and acyl groups take precedence over the OH group. The OH group is a strong electron-donating group through
CH. 23 Chapter 23 Phenols Nomenclature The OH group takes precedence as the parent phenol. Carboxyl and acyl groups take precedence over the OH group. The OH group is a strong electron-donating group through
Chapter 17. Reactions of Aromatic Compounds
 Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Although benzene s pi electrons are in a stable aromatic system, they are available to attack a strong electrophile to give
Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Although benzene s pi electrons are in a stable aromatic system, they are available to attack a strong electrophile to give
Organic Chemistry II KEY March 27, 2013
 rganic Chemistry II KEY March 27, 2013 Exam 2: VERSI C 1. Rank the dienophiles from most reactive to least reactive in the Diels Alder reaction (most>least) E I II III IV > II > III > IV b) III > I > II
rganic Chemistry II KEY March 27, 2013 Exam 2: VERSI C 1. Rank the dienophiles from most reactive to least reactive in the Diels Alder reaction (most>least) E I II III IV > II > III > IV b) III > I > II
Organic Chemistry II KEY March 27, Which of the following reaction intermediates will form the fastest in the reaction below?
 rganic Chemistry II KEY March 27, 2013 Exam 2: VERSI D 1. Which of the following reaction intermediates will form the fastest in the reaction below? C 1 equiv a 2 a) IV b) III c) II d) II & III e) I I.
rganic Chemistry II KEY March 27, 2013 Exam 2: VERSI D 1. Which of the following reaction intermediates will form the fastest in the reaction below? C 1 equiv a 2 a) IV b) III c) II d) II & III e) I I.
Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
12/27/2010. Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
N_HW1 N_HW1. 1. What is the purpose of the H 2 O in this sequence?
 N_HW1 N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. 1. What is the purpose of the H 2 O in this
N_HW1 N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. 1. What is the purpose of the H 2 O in this
18.8 Oxidation. Oxidation by silver ion requires an alkaline medium
 18.8 Oxidation Oxidation by silver ion requires an alkaline medium Test for detecting aldehydes Tollens reagent to prevent precipitation of the insoluble silver oxide, a complexing agent is added: ammonia
18.8 Oxidation Oxidation by silver ion requires an alkaline medium Test for detecting aldehydes Tollens reagent to prevent precipitation of the insoluble silver oxide, a complexing agent is added: ammonia
Suggested solutions for Chapter 29
 s for Chapter 29 29 PRBLEM 1 or each of the following reactions (a) state what kind of substitution is suggested and (b) suggest what product might be formed if monosubstitution occured. Br 2 3 2 S 4 S
s for Chapter 29 29 PRBLEM 1 or each of the following reactions (a) state what kind of substitution is suggested and (b) suggest what product might be formed if monosubstitution occured. Br 2 3 2 S 4 S
Chapter 22 Amines. Nomenclature Amines are classified according to the degree of substitution at nitrogen.
 CH. 22 Chapter 22 Amines Amines are very important in biological chemistry. Most of the bases in biological acid-base reactions are amines. They are also very important nucleophiles in biochemical reactions.
CH. 22 Chapter 22 Amines Amines are very important in biological chemistry. Most of the bases in biological acid-base reactions are amines. They are also very important nucleophiles in biochemical reactions.
Chapter 19: Amines. Introduction
 Chapter 19: Amines Chap 19 HW: (be able to name amines); 37, 39, 41, 42, 44, 46, 47, 48, 53-55, 57, 58 Introduction Organic derivatives of ammonia. Many are biologically active. Chap 19: Amines Slide 19-2
Chapter 19: Amines Chap 19 HW: (be able to name amines); 37, 39, 41, 42, 44, 46, 47, 48, 53-55, 57, 58 Introduction Organic derivatives of ammonia. Many are biologically active. Chap 19: Amines Slide 19-2
Aromatic Compounds II
 2302272 Org Chem II Part I Lecture 2 Aromatic Compounds II Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 17 in Organic Chemistry, 8 th Edition, L.
2302272 Org Chem II Part I Lecture 2 Aromatic Compounds II Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 17 in Organic Chemistry, 8 th Edition, L.
molecules ISSN
 Molecules 2005, 10, 1318 1324 molecules ISS 1420-3049 http://www.mdpi.org Synthesis of ew Potentially Bioactive Bicyclic 2-Pyridones Maxime D. Crozet 1, Pascal George 2, Michel P. Crozet 1 and Patrice
Molecules 2005, 10, 1318 1324 molecules ISS 1420-3049 http://www.mdpi.org Synthesis of ew Potentially Bioactive Bicyclic 2-Pyridones Maxime D. Crozet 1, Pascal George 2, Michel P. Crozet 1 and Patrice
Tautomerism in Alkyl and -OH Derivatives of Heterocycles containing Two Heteroatoms
 Tautomerism in Alkyl and - Derivatives of eterocycles containing Two eteroatoms Colette Jermann Master in chemistry - Year eterocyclic Chemistry Coursework 1 Summary Type of tautomerism S Isoxazol Isothiazole
Tautomerism in Alkyl and - Derivatives of eterocycles containing Two eteroatoms Colette Jermann Master in chemistry - Year eterocyclic Chemistry Coursework 1 Summary Type of tautomerism S Isoxazol Isothiazole
Chapter 15. Reactions of Aromatic Compounds. Electrophilic Aromatic Substitution on Arenes. The first step is the slow, rate-determining step
 Electrophilic Aromatic Substitution on Arenes Chapter 15 Reactions of Aromatic Compounds The characteristic reaction of aromatic rings is substitution initiated by an electrophile halogenation nitration
Electrophilic Aromatic Substitution on Arenes Chapter 15 Reactions of Aromatic Compounds The characteristic reaction of aromatic rings is substitution initiated by an electrophile halogenation nitration
Synthesis and Characterization of Some New Aminoimidazoles
 Asian Journal of Chemistry Vol. 19, o. 7 (2007), 4963-4968 Synthesis and Characterization of Some ew Aminoimidazoles A. YAYAZADE* and M. AGI Department of Chemistry, University of Guilan, P.O. Box 1914,
Asian Journal of Chemistry Vol. 19, o. 7 (2007), 4963-4968 Synthesis and Characterization of Some ew Aminoimidazoles A. YAYAZADE* and M. AGI Department of Chemistry, University of Guilan, P.O. Box 1914,
Electropolymerization of cobalto(5,10,15-tris(4-aminophenyl)- 20-phenylporphyrin) for electrochemical detection of antioxidant-antipyrine
 Supplementary material Electropolymerization of cobalto(5,10,15-tris(4-aminophenyl)- 20-phenylporphyrin) for electrochemical detection of antioxidant-antipyrine Sambandam Anandan* a, Arumugam Manivel a,
Supplementary material Electropolymerization of cobalto(5,10,15-tris(4-aminophenyl)- 20-phenylporphyrin) for electrochemical detection of antioxidant-antipyrine Sambandam Anandan* a, Arumugam Manivel a,
Chapter 12. Reactions of Arenes: Electrophilic Aromatic Substitution. Class Notes. A. The method by which substituted benzenes are synthesized
 Chapter 12 Reactions of Arenes: Electrophilic Aromatic Substitution Chapter 12 suggested problems: 22, 23, 26, 27, 32, 33 Class Notes I. Electrophilic aromatic substitution reactions A. The method by which
Chapter 12 Reactions of Arenes: Electrophilic Aromatic Substitution Chapter 12 suggested problems: 22, 23, 26, 27, 32, 33 Class Notes I. Electrophilic aromatic substitution reactions A. The method by which
Aldehydes and Ketones Reactions. Dr. Sapna Gupta
 Aldehydes and Ketones Reactions Dr. Sapna Gupta Reactions of Aldehydes and Ketones Nucleophilic Addition A strong nucleophile attacks the carbonyl carbon, forming an alkoxide ion that is then protonated.
Aldehydes and Ketones Reactions Dr. Sapna Gupta Reactions of Aldehydes and Ketones Nucleophilic Addition A strong nucleophile attacks the carbonyl carbon, forming an alkoxide ion that is then protonated.
Synthesis of Nitriles a. dehydration of 1 amides using POCl 3 : b. SN2 reaction of cyanide ion on halides:
 I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
Chapter 17 Reactions of Aromatic Compounds
 rganic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice all Electrophilic
rganic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice all Electrophilic
H 2 SO 4 Ar-NO 2 + H2O
 Phenyl group: Shorthand for phenyl: Ph, C 6 5,. An aryl group is an aromatic group: phenyl, substituted phenyl, or other aromatic group. Shorthand: Ar Generalized electrophilic aromatic substitution: E
Phenyl group: Shorthand for phenyl: Ph, C 6 5,. An aryl group is an aromatic group: phenyl, substituted phenyl, or other aromatic group. Shorthand: Ar Generalized electrophilic aromatic substitution: E
CHAPTER 16 - CHEMISTRY OF BENZENE: ELECTROPHILIC AROMATIC SUBSTITUTION
 CAPTR 16 - CMISTRY F BNZN: LCTRPILIC ARMATIC SUBSTITUTIN As stated in the previous chapter, benzene and other aromatic rings do not undergo electrophilic addition reactions of the simple alkenes but rather
CAPTR 16 - CMISTRY F BNZN: LCTRPILIC ARMATIC SUBSTITUTIN As stated in the previous chapter, benzene and other aromatic rings do not undergo electrophilic addition reactions of the simple alkenes but rather
Lecture Topics: I. Electrophilic Aromatic Substitution (EAS)
 Reactions of Aromatic Compounds Reading: Wade chapter 17, sections 17-1- 17-15 Study Problems: 17-44, 17-46, 17-47, 17-48, 17-51, 17-52, 17-53, 17-59, 17-61 Key Concepts and Skills: Predict and propose
Reactions of Aromatic Compounds Reading: Wade chapter 17, sections 17-1- 17-15 Study Problems: 17-44, 17-46, 17-47, 17-48, 17-51, 17-52, 17-53, 17-59, 17-61 Key Concepts and Skills: Predict and propose
Organic Chemistry, 7 L. G. Wade, Jr. Chapter , Prentice Hall
 Organic Chemistry, 7 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds 2010, Prentice Hall Electrophilic Aromatic Substitution Although h benzene s pi electrons are in a stable aromatic
Organic Chemistry, 7 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds 2010, Prentice Hall Electrophilic Aromatic Substitution Although h benzene s pi electrons are in a stable aromatic
Chapter 20. Amines. Nomenclature for amines. Aryl amines
 Nomenclature for amines Chapter 20 Common names are widely used, named as alkylamines Systematic (IUPAC) nomenclature replaces the -e of the corresponding parent alkane with -amine Amines Simple secondary
Nomenclature for amines Chapter 20 Common names are widely used, named as alkylamines Systematic (IUPAC) nomenclature replaces the -e of the corresponding parent alkane with -amine Amines Simple secondary
Chapter 17 Reactions of Aromatic Compounds. Electrophilic Aromatic Substitution
 Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Electrophile substitutes for a hydrogen on the benzene ring. Chapter 17: Aromatics 2-Reactions Slide 17-2 1 Mechanism Step
Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Electrophile substitutes for a hydrogen on the benzene ring. Chapter 17: Aromatics 2-Reactions Slide 17-2 1 Mechanism Step
HW #3: 14.26, 14.28, 14.30, 14.32, 14.36, 14.42, 14.46, 14.52, 14.56, Alcohols, Ethers and Thiols
 Chemistry 131 Lecture 8: Alcohols, Ethers and Sulfur Analogs: Structure, Nomenclature, Physical Properties, and Chemical Reactivity Chapter 14 in McMurry, Ballantine, et. al. 7 th edition HW #3: 14.26,
Chemistry 131 Lecture 8: Alcohols, Ethers and Sulfur Analogs: Structure, Nomenclature, Physical Properties, and Chemical Reactivity Chapter 14 in McMurry, Ballantine, et. al. 7 th edition HW #3: 14.26,
Synthesis of N-(3(5)-Aryl-1,2,4-triazol-5(3)-yl)-N -carbethoxythioureas and Their Tautomerism in DMSO Solution
 ynthesis of -(3(5)-Aryl-1,2,4-triazol-5(3)-yl)- -carbethoxythioureas and Their Tautomerism in DMO olution Anton V. Dolzhenko, riday Bera and Wai Keung Chui Department of Pharmacy, Faculty of cience, ational
ynthesis of -(3(5)-Aryl-1,2,4-triazol-5(3)-yl)- -carbethoxythioureas and Their Tautomerism in DMO olution Anton V. Dolzhenko, riday Bera and Wai Keung Chui Department of Pharmacy, Faculty of cience, ational
Lecture 15. More Carbonyl Chemistry. Alcohols React with Aldehydes and Ketones in two steps first O R'OH, H + OR" 2R"OH R + H 2 O OR" 3/8/16
 Lecture 15 More Carbonyl Chemistry R" R C + R' 2R" R C R" R' + 2 March 8, 2016 Alcohols React with Aldehydes and Ketones in two steps first R', + R R 1 emiacetal reacts further in acid to yield an acetal
Lecture 15 More Carbonyl Chemistry R" R C + R' 2R" R C R" R' + 2 March 8, 2016 Alcohols React with Aldehydes and Ketones in two steps first R', + R R 1 emiacetal reacts further in acid to yield an acetal
Chem 263 March 7, 2006
 Chem 263 March 7, 2006 Aldehydes and Ketones Aldehydes and ketones contain a carbonyl group, in which the carbon atom is doubly bonded to an oxygen atom. The carbonyl group is highly polarized, with a
Chem 263 March 7, 2006 Aldehydes and Ketones Aldehydes and ketones contain a carbonyl group, in which the carbon atom is doubly bonded to an oxygen atom. The carbonyl group is highly polarized, with a
CHAPTER-5 CYCLIC VOLTAMETRIC STUDIES OF NOVEL INDOLE ANALOGUES PREPARED IN THE PRESENT STUDY
 CHAPTER-5 CYCLIC VOLTAMETRIC STUDIES OF NOVEL INDOLE ANALOGUES PREPARED IN THE PRESENT STUDY Page No. 175-187 5.1 Introduction 5.2 Theoretical 5.3 Experimental 5.4 References 5. 1 Introduction Electrochemical
CHAPTER-5 CYCLIC VOLTAMETRIC STUDIES OF NOVEL INDOLE ANALOGUES PREPARED IN THE PRESENT STUDY Page No. 175-187 5.1 Introduction 5.2 Theoretical 5.3 Experimental 5.4 References 5. 1 Introduction Electrochemical
Chapter 10: Carboxylic Acids and Their Derivatives
 Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
Chapter 8. Acidity, Basicity and pk a
 Chapter 8 Acidity, Basicity and pk a p182 In this reaction water is acting as a base, according to our definition above, by accepting a proton from HCl which in turn is acting as an acid by donating a
Chapter 8 Acidity, Basicity and pk a p182 In this reaction water is acting as a base, according to our definition above, by accepting a proton from HCl which in turn is acting as an acid by donating a
11/26/ Polycyclic Aromatic Compounds. Polycyclic Aromatic Compounds. Polycyclic Aromatic Compounds
 9.5 Polycyclic Aromatic Compounds The general concept of aromaticity can be extended to include polycyclic aromatic compounds Benzo[a]pyrene is one of the cancer-causing substances found in tobacco smoke
9.5 Polycyclic Aromatic Compounds The general concept of aromaticity can be extended to include polycyclic aromatic compounds Benzo[a]pyrene is one of the cancer-causing substances found in tobacco smoke
Chapter 7: Alcohols, Phenols and Thiols
 Chapter 7: Alcohols, Phenols and Thiols 45 -Alcohols have the general formula R-OH and are characterized by the presence of a hydroxyl group, -OH. -Phenols have a hydroxyl group attached directly to an
Chapter 7: Alcohols, Phenols and Thiols 45 -Alcohols have the general formula R-OH and are characterized by the presence of a hydroxyl group, -OH. -Phenols have a hydroxyl group attached directly to an
896 Chapter 21 Amines H H N R R R N R H R N R H O H R 3 N CH 2 NH 2 NHCH 3
 896 Chapter 21 Amines 21.20 Summary Section 21.1 Section 21.2 Section 21.3 Section 21.4 Alkylamines are compounds of the type shown, where R, R, and R are alkyl groups. ne or more of these groups is an
896 Chapter 21 Amines 21.20 Summary Section 21.1 Section 21.2 Section 21.3 Section 21.4 Alkylamines are compounds of the type shown, where R, R, and R are alkyl groups. ne or more of these groups is an
Synthesis of Amines Amine Alkylation by SN2 reaction II. Reductive Amination
 Synthesis of Amines I. Amine Alkylation by SN2 reaction Amines can be alkylated in SN2 fashion by alkyl halides; primary halides are best for this purpose. This is not a practical reaction for formation
Synthesis of Amines I. Amine Alkylation by SN2 reaction Amines can be alkylated in SN2 fashion by alkyl halides; primary halides are best for this purpose. This is not a practical reaction for formation
Alcohols, Ethers, & Epoxides
 Alcohols, Ethers, & Epoxides Alcohols Structure and Bonding Enols and Phenols Compounds having a hydroxy group on a sp 2 hybridized carbon enols and phenols undergo different reactions than alcohols. Chapter
Alcohols, Ethers, & Epoxides Alcohols Structure and Bonding Enols and Phenols Compounds having a hydroxy group on a sp 2 hybridized carbon enols and phenols undergo different reactions than alcohols. Chapter
CARBONYL COMPOUNDS: OXIDATION-REDUCTION REACTION
 CARBONYL COMPOUNDS: OXIDATION-REDUCTION REACTION Introduction Several functional groups contain the carbonyl group Carbonyl groups can be converted into alcohols by various reactions Structure of the Carbonyl
CARBONYL COMPOUNDS: OXIDATION-REDUCTION REACTION Introduction Several functional groups contain the carbonyl group Carbonyl groups can be converted into alcohols by various reactions Structure of the Carbonyl
Aryl Halides. Structure
 Aryl Halides Structure Aryl halides are compounds containing halogen attached directly to an aromatic ring. They have the general formula ArX, where Ar is phenyl, substituted phenyl. X= F,Cl,Br,I An aryl
Aryl Halides Structure Aryl halides are compounds containing halogen attached directly to an aromatic ring. They have the general formula ArX, where Ar is phenyl, substituted phenyl. X= F,Cl,Br,I An aryl
Benzenes & Aromatic Compounds
 Benzenes & Aromatic Compounds 1 Structure of Benzene H H C C C H C 6 H 6 H C C C H H A cyclic conjugate molecule Benzene is a colourless odourless liquid, boiling at 80 o C and melting at 5 o C. It is
Benzenes & Aromatic Compounds 1 Structure of Benzene H H C C C H C 6 H 6 H C C C H H A cyclic conjugate molecule Benzene is a colourless odourless liquid, boiling at 80 o C and melting at 5 o C. It is
Chem 263 Oct. 4, 2016
 Chem 263 ct. 4, 2016 ow to determine position and reactivity: Examples The strongest donating group wins: 2 3 2 S 4 + 3 2 2 S 4 2 2 + 2 2 3 2 S 4 2 2 2 2,4,6-trinitrophenol picric acid This reactivity
Chem 263 ct. 4, 2016 ow to determine position and reactivity: Examples The strongest donating group wins: 2 3 2 S 4 + 3 2 2 S 4 2 2 + 2 2 3 2 S 4 2 2 2 2,4,6-trinitrophenol picric acid This reactivity
Topic 9. Aldehydes & Ketones
 Chemistry 2213a Fall 2012 Western University Topic 9. Aldehydes & Ketones A. Structure and Nomenclature The carbonyl group is present in aldehydes and ketones and is the most important group in bio-organic
Chemistry 2213a Fall 2012 Western University Topic 9. Aldehydes & Ketones A. Structure and Nomenclature The carbonyl group is present in aldehydes and ketones and is the most important group in bio-organic
Class XII: Chemistry Chapter 13: Amines Top concepts
 Class XII: Chemistry Chapter 13: Amines Top concepts 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification
Class XII: Chemistry Chapter 13: Amines Top concepts 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification
Properties of Amines
 Properties of Amines 1. Boiling Point and Water Solubility It is instructive to compare the boiling points and water solubility of amines with those of corresponding alcohols and ethers. The dominant factor
Properties of Amines 1. Boiling Point and Water Solubility It is instructive to compare the boiling points and water solubility of amines with those of corresponding alcohols and ethers. The dominant factor
Supplementary Materials
 Supplementary Materials ORTHOGOALLY POSITIOED DIAMIO PYRROLE- AD IMIDAZOLE- COTAIIG POLYAMIDES: SYTHESIS OF 1-(3-SUBSTITUTED-PROPYL)-4- ITROPYRROLE-2-CARBOXYLIC ACID AD 1-(3-CHLOROPROPYL)-4- ITROIMIDAZOLE-2-CARBOXYLIC
Supplementary Materials ORTHOGOALLY POSITIOED DIAMIO PYRROLE- AD IMIDAZOLE- COTAIIG POLYAMIDES: SYTHESIS OF 1-(3-SUBSTITUTED-PROPYL)-4- ITROPYRROLE-2-CARBOXYLIC ACID AD 1-(3-CHLOROPROPYL)-4- ITROIMIDAZOLE-2-CARBOXYLIC
Chapter 5. Aromatic Compounds
 Chapter 5. Aromatic Compounds 5.1 Structure of Benzene: The Kekule Proposal Mid-1800s, benzene was known to have the molecular formula C 6 6. Benzene reacts with 2 in the presence of iron to give substitution
Chapter 5. Aromatic Compounds 5.1 Structure of Benzene: The Kekule Proposal Mid-1800s, benzene was known to have the molecular formula C 6 6. Benzene reacts with 2 in the presence of iron to give substitution
Chapter 21. Organic Chemistry 4 th Edition Paula Yurkanis Bruice. More About Amines. Heterocyclic Compounds
 Organic Chemistry 4 th Edition Paula Yurkanis Bruice Chapter 21 More About Amines. Heterocyclic Compounds Irene Lee Case Western Reserve University Cleveland, OH 2004, Prentice Hall Amines An amine is
Organic Chemistry 4 th Edition Paula Yurkanis Bruice Chapter 21 More About Amines. Heterocyclic Compounds Irene Lee Case Western Reserve University Cleveland, OH 2004, Prentice Hall Amines An amine is
CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher. Quiz # 4. Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m.
 CHEM 347 Quiz # 4 Spring 2014 Page 1 of 9 CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher Quiz # 4 Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m. Student Name (Printed)
CHEM 347 Quiz # 4 Spring 2014 Page 1 of 9 CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher Quiz # 4 Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m. Student Name (Printed)
Reaction of Sodium Azide with 1,1 Dicyclohexenyl Diepoxides
 Reaction of Sodium Azide with 1,1 Dicyclohexenyl Diepoxides Darvich, Mohammad Raouf * + ; Olyai, Abolfazl and Shabanikia, Akbar Department of Chemistry, Faculty of Science, University of Tehran, Tehran,
Reaction of Sodium Azide with 1,1 Dicyclohexenyl Diepoxides Darvich, Mohammad Raouf * + ; Olyai, Abolfazl and Shabanikia, Akbar Department of Chemistry, Faculty of Science, University of Tehran, Tehran,
Preparation of Substituted Methyl o-nitrophenyl Sulfides
 Molecules 00, 7, 7-17 molecules I 140-3049 http://www.mdpi.org Preparation of ubstituted Methyl o-itrophenyl ulfides Katerina Dudova, Frantisek Castek, Vladimir Machacek * and Petr imunek Department of
Molecules 00, 7, 7-17 molecules I 140-3049 http://www.mdpi.org Preparation of ubstituted Methyl o-itrophenyl ulfides Katerina Dudova, Frantisek Castek, Vladimir Machacek * and Petr imunek Department of
Advanced Analytical Chemistry Lecture 19. Chem 4631
 Advanced Analytical Chemistry Lecture 19 Chem 4631 Organic Electrochemistry is a multidisciplinary science overlapping the fields of organic chemistry, biochemistry, physical chemistry and electrochemistry.
Advanced Analytical Chemistry Lecture 19 Chem 4631 Organic Electrochemistry is a multidisciplinary science overlapping the fields of organic chemistry, biochemistry, physical chemistry and electrochemistry.
Honors Cup Synthetic Proposal
 onors Cup Synthetic Proposal Section: 270-V Group Members: Azhar Carim, Ian Cross, Albert Tang Title: Synthesis of indigo from -(2-bromoethyl)-2-nitrobenzamide Introduction: Indigo has been used as a dye
onors Cup Synthetic Proposal Section: 270-V Group Members: Azhar Carim, Ian Cross, Albert Tang Title: Synthesis of indigo from -(2-bromoethyl)-2-nitrobenzamide Introduction: Indigo has been used as a dye
Chapter 9 Aldehydes and Ketones Excluded Sections:
 Chapter 9 Aldehydes and Ketones Excluded Sections: 9.14-9.19 Aldehydes and ketones are found in many fragrant odors of many fruits, fine perfumes, hormones etc. some examples are listed below. Aldehydes
Chapter 9 Aldehydes and Ketones Excluded Sections: 9.14-9.19 Aldehydes and ketones are found in many fragrant odors of many fruits, fine perfumes, hormones etc. some examples are listed below. Aldehydes
Chapter 16: Aromatic Compounds
 Chamras Chemistry 106 Lecture otes xamination 2 Materials Chapter 16: Aromatic Compounds Benzene, the Most Commonly Known Aromatic Compound: The aromatic nature of benzene stabilizes it 36 kcal.mol 1.
Chamras Chemistry 106 Lecture otes xamination 2 Materials Chapter 16: Aromatic Compounds Benzene, the Most Commonly Known Aromatic Compound: The aromatic nature of benzene stabilizes it 36 kcal.mol 1.
Carboxylic Acids and Nitriles
 Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Dr. Munther Abdujaleel M.A. Assist. Prof. Org. Chem.
 Dr. Munther Abdujaleel M.A. Assist. Prof. Org. Chem. Amines An amine, derivatives of ammonia NH 3 has the general formula RNH 2, R 2 NH, or R 3 N, where R is any alkyl or aryl group. For example: Adernalin
Dr. Munther Abdujaleel M.A. Assist. Prof. Org. Chem. Amines An amine, derivatives of ammonia NH 3 has the general formula RNH 2, R 2 NH, or R 3 N, where R is any alkyl or aryl group. For example: Adernalin
ζ ε δ γ β α α β γ δ ε ζ
 hem 263 Nov 17, 2016 eactions at the α-arbon The alpha carbon is the carbon adjacent to the carbonyl carbon. Beta is the next one, followed by gamma, delta, epsilon, and so on. 2 ε 2 δ 2 γ 2 2 β α The
hem 263 Nov 17, 2016 eactions at the α-arbon The alpha carbon is the carbon adjacent to the carbonyl carbon. Beta is the next one, followed by gamma, delta, epsilon, and so on. 2 ε 2 δ 2 γ 2 2 β α The
The now-banned diet drug fen-phen is a mixture of two synthetic substituted benzene: fenfluramine and phentermine.
 The now-banned diet drug fen-phen is a mixture of two synthetic substituted benzene: fenfluramine and phentermine. Chemists have synthesized compounds with structures similar to adrenaline, producing amphetamine.
The now-banned diet drug fen-phen is a mixture of two synthetic substituted benzene: fenfluramine and phentermine. Chemists have synthesized compounds with structures similar to adrenaline, producing amphetamine.
Supplementary Information
 Supplementary Information C aryl -C alkyl bond formation from Cu(ClO 4 ) 2 -mediated oxidative cross coupling reaction between arenes and alkyllithium reagents through structurally well-defined Ar-Cu(III)
Supplementary Information C aryl -C alkyl bond formation from Cu(ClO 4 ) 2 -mediated oxidative cross coupling reaction between arenes and alkyllithium reagents through structurally well-defined Ar-Cu(III)
Name/CG: 2012 Term 2 Organic Chemistry Revision (Session II) Deductive Question
 Name/G: 2012 Term 2 rganic hemistry Revision (Session II) Deductive Question 1(a) A yellow liquid A, 7 7 N 2, reacts with alkaline potassium manganate (VII) and on acidification gives a yellow solid B,
Name/G: 2012 Term 2 rganic hemistry Revision (Session II) Deductive Question 1(a) A yellow liquid A, 7 7 N 2, reacts with alkaline potassium manganate (VII) and on acidification gives a yellow solid B,
All Classes of Organic Compounds
 Amines All Classes of Organic Compounds ydrocarbons Functionalized ydrocarbons F,Cl,Br O,S, Alkanes Alkenes Alkynes Aromatics alides -O- -S- -- Alcohols Phenols Ethers Thiols Dissulfides Amines O C O C
Amines All Classes of Organic Compounds ydrocarbons Functionalized ydrocarbons F,Cl,Br O,S, Alkanes Alkenes Alkynes Aromatics alides -O- -S- -- Alcohols Phenols Ethers Thiols Dissulfides Amines O C O C
Module No and Title. PAPER No: 5 ; TITLE : Organic Chemistry-II MODULE No: 25 ; TITLE: S E 1 reactions
 Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 25; S E 1 reactions CHE_P5_M25 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. S E 1 reactions 3.1
Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 25; S E 1 reactions CHE_P5_M25 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. S E 1 reactions 3.1
Nitro compounds are named by writing the word nitro before the name of the parent compound.
 Nitro compounds are an important class of organic compounds which may be regarded as derived from hydrocarbons by the replacement of one or more hydrogen atoms by nitro (NO₂) groups. Nitro arenes(i.e.
Nitro compounds are an important class of organic compounds which may be regarded as derived from hydrocarbons by the replacement of one or more hydrogen atoms by nitro (NO₂) groups. Nitro arenes(i.e.
Histidine as a catalyst in organic synthesis: A facile in situ synthesis of α, N-diarylnitrones
 Proc. Indian Acad. Sci. (Chem. Sci.), Vol. 113, o. 4, August 2001, pp 291 296 Indian Academy of Sciences istidine as a catalyst in organic synthesis: A facile in situ synthesis of α, -diarylnitrones 1.
Proc. Indian Acad. Sci. (Chem. Sci.), Vol. 113, o. 4, August 2001, pp 291 296 Indian Academy of Sciences istidine as a catalyst in organic synthesis: A facile in situ synthesis of α, -diarylnitrones 1.
CHEM Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W
 CHEM 2425. Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W Short Answer Exhibit 16-1 MATCH a structure or term from the following list with each description below. Place
CHEM 2425. Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W Short Answer Exhibit 16-1 MATCH a structure or term from the following list with each description below. Place
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group
 Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
R. EL-SAYED, ABDALLAH A. MOHAMED and E.M. MABROUK*
 Material Science Research India Vol. 7(2), 339-346 (2010) DC-polarography and cyclic voltammetric studies of some mono and bis azo compounds derived from aromatic primary amines and 2,3-dihydroxynaphthalene
Material Science Research India Vol. 7(2), 339-346 (2010) DC-polarography and cyclic voltammetric studies of some mono and bis azo compounds derived from aromatic primary amines and 2,3-dihydroxynaphthalene
Electronic Supplementary Information
 Electronic Supplementary Information 1. Reagents All the L-amino acids and L-glutathione (reduced form) were purchased from Sangon Biotch. o. (Shanghai, hina); Methyl orange, potassium tetrachloropalladate(ii),
Electronic Supplementary Information 1. Reagents All the L-amino acids and L-glutathione (reduced form) were purchased from Sangon Biotch. o. (Shanghai, hina); Methyl orange, potassium tetrachloropalladate(ii),
1. What is the major organic product obtained from the following sequence of reactions?
 CH320 N N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. Carefully record your answers on the Scantron
CH320 N N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. Carefully record your answers on the Scantron
Phenols, Ethers, and Organic Sulfur Compound
 Phenols, Ethers, and rganic Sulfur Compound Phenols - Structure General Structure - A hydroxy () group attached directly to an aromatic ring: Phenol α-naphthol β-naphthol Note: C2 is not a phenol. Phenols
Phenols, Ethers, and rganic Sulfur Compound Phenols - Structure General Structure - A hydroxy () group attached directly to an aromatic ring: Phenol α-naphthol β-naphthol Note: C2 is not a phenol. Phenols
Experiment 3: Preparation of Lidocaine
 Experiment 3: Preparation of Lidocaine This two-step synthesis involves the following conversion: 2,6-dimethylaniline α- chloro-2, 6-dimethylacetanilide Lidocaine. This synthetic scheme is shown in equation
Experiment 3: Preparation of Lidocaine This two-step synthesis involves the following conversion: 2,6-dimethylaniline α- chloro-2, 6-dimethylacetanilide Lidocaine. This synthetic scheme is shown in equation
Supplementary Note 1 : Chemical synthesis of (E/Z)-4,8-dimethylnona-2,7-dien-4-ol (4)
 Supplementary Note 1 : Chemical synthesis of (E/Z)-4,8-dimethylnona-2,7-dien-4-ol (4) A solution of propenyl magnesium bromide in THF (17.5 mmol) under nitrogen atmosphere was cooled in an ice bath and
Supplementary Note 1 : Chemical synthesis of (E/Z)-4,8-dimethylnona-2,7-dien-4-ol (4) A solution of propenyl magnesium bromide in THF (17.5 mmol) under nitrogen atmosphere was cooled in an ice bath and
ACID-BASE EXTRACTION
 ACID-BASE EXTRACTION An acid-base extraction is a type of liquid-liquid extraction. It typically involves different solubility levels in water and an organic solvent. The organic solvent may be any carbon-based
ACID-BASE EXTRACTION An acid-base extraction is a type of liquid-liquid extraction. It typically involves different solubility levels in water and an organic solvent. The organic solvent may be any carbon-based
(2) After dissolving a solid in a solvent at high temperature, the solution is not filtered.
 Name Key 216 W13-Exam No. 1 Page 2 I. (10 points) The goal of recrystallization is to obtain purified material with a maximized recovery. For each of the following cases, indicate as to which of the two
Name Key 216 W13-Exam No. 1 Page 2 I. (10 points) The goal of recrystallization is to obtain purified material with a maximized recovery. For each of the following cases, indicate as to which of the two
Chem 263 Notes March 2, 2006
 Chem 263 Notes March 2, 2006 Average for the midterm is 102.5 / 150 (approx. 68%). Preparation of Aldehydes and Ketones There are several methods to prepare aldehydes and ketones. We will only deal with
Chem 263 Notes March 2, 2006 Average for the midterm is 102.5 / 150 (approx. 68%). Preparation of Aldehydes and Ketones There are several methods to prepare aldehydes and ketones. We will only deal with
10.1 Acids and Bases in Aqueous Solution
 10.1 Acids and Bases in Aqueous Solution Arrhenius Definition of Acids and Bases An acid is a substance that gives hydrogen ions, H +, when dissolved in water. In fact, H + reacts with water and produces
10.1 Acids and Bases in Aqueous Solution Arrhenius Definition of Acids and Bases An acid is a substance that gives hydrogen ions, H +, when dissolved in water. In fact, H + reacts with water and produces
Review Experiments Formation of Polymers Reduction of Vanillin
 Review Experiments Formation of Polymers What is a polymer? What is polymerization? What is the difference between an addition polymerization and a condensation polymerization? Which type of polymerization
Review Experiments Formation of Polymers What is a polymer? What is polymerization? What is the difference between an addition polymerization and a condensation polymerization? Which type of polymerization
Chemistry 283g Experiment 4
 Chemistry 283g xperiment 4 XPRIMNT 4: lectrophilic Aromatic Substitution: A Friedel-Craft Acylation Reaction Relevant sections in the text: Fox & Whitesell, 3 rd d. Chapter 11, especially pg. 524-526,
Chemistry 283g xperiment 4 XPRIMNT 4: lectrophilic Aromatic Substitution: A Friedel-Craft Acylation Reaction Relevant sections in the text: Fox & Whitesell, 3 rd d. Chapter 11, especially pg. 524-526,
CONDENSATION OF N1TROMETHANE AND NITROETHANE WITH ETHYL MALEATE AND FUMARATE IN THE PRESENCE OF POTASSIUM FLUORIDE*
 CONDENSATION OF N1TROMETHANE AND NITROETHANE WITH ETHYL MALEATE AND FUMARATE IN THE PRESENCE OF POTASSIUM FLUORIDE* A. OSTASZYNSKI, J. WIELGAT and T. URBANSKI Institute of Organic Synthesis, Polish Academy
CONDENSATION OF N1TROMETHANE AND NITROETHANE WITH ETHYL MALEATE AND FUMARATE IN THE PRESENCE OF POTASSIUM FLUORIDE* A. OSTASZYNSKI, J. WIELGAT and T. URBANSKI Institute of Organic Synthesis, Polish Academy
Chapter 25: The Chemistry of Life: Organic and Biological Chemistry
 Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
SURVEY ON ARYL COMPOUNDS
 Journal of Plastic and Polymer Technology (JPPT) Vol. 1, Issue 1, Jun 2015, 111-132 TJPRC Pvt. Ltd SURVEY ON ARYL COMPOUNDS NAGHAM MAHMOOD ALJAMALI Organic Chemistry, Department of Chemistry, College of
Journal of Plastic and Polymer Technology (JPPT) Vol. 1, Issue 1, Jun 2015, 111-132 TJPRC Pvt. Ltd SURVEY ON ARYL COMPOUNDS NAGHAM MAHMOOD ALJAMALI Organic Chemistry, Department of Chemistry, College of
Q.1 Draw structures for all amines of molecular formula C 4 H 11 N. Classify them as primary, secondary or tertiary amines.
 1 AMIES Structure ontain the 2 group. lassification primary (1 ) amines secondary (2 ) amines tertiary (3 ) amines quarternary (4 ) ammonium salts + 1 2 3 4 Aliphatic Aromatic methylamine, ethylamine,
1 AMIES Structure ontain the 2 group. lassification primary (1 ) amines secondary (2 ) amines tertiary (3 ) amines quarternary (4 ) ammonium salts + 1 2 3 4 Aliphatic Aromatic methylamine, ethylamine,
Multistep Synthesis of 5-isopropyl-1,3-cyclohexanedione
 Multistep Synthesis of 5-isopropyl-1,3-cyclohexanedione The purpose of this experiment was to synthesize 5-isopropyl-1,3-cyclohexanedione from commercially available compounds. To do this, acetone and
Multistep Synthesis of 5-isopropyl-1,3-cyclohexanedione The purpose of this experiment was to synthesize 5-isopropyl-1,3-cyclohexanedione from commercially available compounds. To do this, acetone and
18. Arene Diazonium Ion Reactions
 18. Arene Diazonium Ion Reactions A. Introduction In the previous laboratory experiment, you explored the functionalization of benzene via electrophilic aromatic substitution reactions. In these reactions,
18. Arene Diazonium Ion Reactions A. Introduction In the previous laboratory experiment, you explored the functionalization of benzene via electrophilic aromatic substitution reactions. In these reactions,
16. Chemistry of Benzene: Electrophilic Aromatic Substitution. Based on McMurry s Organic Chemistry, 7 th edition
 16. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 7 th edition Substitution Reactions of Benzene and Its Derivatives Benzene is aromatic: a cyclic conjugated
16. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 7 th edition Substitution Reactions of Benzene and Its Derivatives Benzene is aromatic: a cyclic conjugated
AMINES. 3. Secondary When two hydrogen atoms are replaced by two alkyl or aryl groups.
 AMINES Amine may be regarded as derivative of ammonia formed by replacement of one or more hydrogen atoms by corresponding number of alkyl or aryl group CLASSIFICATION 1. Ammonia 2. Primary amine 3. Secondary
AMINES Amine may be regarded as derivative of ammonia formed by replacement of one or more hydrogen atoms by corresponding number of alkyl or aryl group CLASSIFICATION 1. Ammonia 2. Primary amine 3. Secondary
Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution
 John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution Paul D. Adams University of Arkansas Substitution Reactions of Benzene and Its Derivatives
John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution Paul D. Adams University of Arkansas Substitution Reactions of Benzene and Its Derivatives
Supporting Information
 1 Supporting Information Gold-catalyzed Synthesis of 3-arylindoles via Annulation of Nitrosoarenes and Alkynes Siva Murru, August A. Gallo and Radhey S. Srivastava* Department of Chemistry, University
1 Supporting Information Gold-catalyzed Synthesis of 3-arylindoles via Annulation of Nitrosoarenes and Alkynes Siva Murru, August A. Gallo and Radhey S. Srivastava* Department of Chemistry, University
Alkylamines are compounds of the type shown, where R, R, and R are alkyl groups. One or more of these groups is an aryl group in arylamines.
 22.20 Summary 955 22.20 SUMMARY Section 22.1 Alkylamines are compounds of the type shown, where R, R, and R are alkyl groups. ne or more of these groups is an aryl group in s. R R R R amine R Secondary
22.20 Summary 955 22.20 SUMMARY Section 22.1 Alkylamines are compounds of the type shown, where R, R, and R are alkyl groups. ne or more of these groups is an aryl group in s. R R R R amine R Secondary
Synthesis of new stable pseudobases
 Synthesis of new stable pseudobases Zsuzsanna Riedl *, György Hajós, András Messmer, and Orsolya Egyed Institute of Chemistry, Chemical Research Center, Hungarian Academy of Sciences, H-1525 Budapest,
Synthesis of new stable pseudobases Zsuzsanna Riedl *, György Hajós, András Messmer, and Orsolya Egyed Institute of Chemistry, Chemical Research Center, Hungarian Academy of Sciences, H-1525 Budapest,
Supporting Information
 Supporting Information Antonio Palumbo Piccionello, Annalisa Guarcello, Silvestre Buscemi, icolò Vivona, and Andrea Pace* Dipartimento di Chimica Organica "E. Paternò", Università degli Studi di Palermo,
Supporting Information Antonio Palumbo Piccionello, Annalisa Guarcello, Silvestre Buscemi, icolò Vivona, and Andrea Pace* Dipartimento di Chimica Organica "E. Paternò", Università degli Studi di Palermo,
Chapter 13 Reactions of Arenes Electrophilic Aromatic Substitution
 . 13 hapter 13 eactions of Arenes lectrophilic Aromatic ubstitution lectrophiles add to aromatic rings in a fashion somewhat similar to the addition of electrophiles to alkenes. ecall: 3 4 Y 1 4 2 1 δ
. 13 hapter 13 eactions of Arenes lectrophilic Aromatic ubstitution lectrophiles add to aromatic rings in a fashion somewhat similar to the addition of electrophiles to alkenes. ecall: 3 4 Y 1 4 2 1 δ
Chapter 12 Alcohols from Carbonyl Compounds: Oxidation-Reduction and Organometallic Compounds
 Chapter 12 Alcohols from Carbonyl Compounds: Oxidation-Reduction and Organometallic Compounds Introduction Several functional groups contain the carbonyl group Carbonyl groups can be converted into alcohols
Chapter 12 Alcohols from Carbonyl Compounds: Oxidation-Reduction and Organometallic Compounds Introduction Several functional groups contain the carbonyl group Carbonyl groups can be converted into alcohols
Supplementary data. Department of Chemistry, Guru Ghasidas Vishwavidyalaya, Bilaspur , Chhattisgarh, India.
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Supplementary data On-water Facile Synthesis of poly-substituted 6-arylamino pyridines and
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Supplementary data On-water Facile Synthesis of poly-substituted 6-arylamino pyridines and
REACTIONS OF AROMATIC COMPOUNDS
 A STUDENT SHOULD BE ABLE TO: REACTIONS OF AROMATIC COMPOUNDS 1. Predict the product(s) of Electrophilic aromatic substitution (EAS): halogenation, sulfonation, nitration, Friedel- Crafts alkylation and
A STUDENT SHOULD BE ABLE TO: REACTIONS OF AROMATIC COMPOUNDS 1. Predict the product(s) of Electrophilic aromatic substitution (EAS): halogenation, sulfonation, nitration, Friedel- Crafts alkylation and
Reducing Agents. Linda M. Sweeting 1998
 Reducing Agents Linda M. Sweeting 1998 Reduction is defined in chemistry as loss of oxygen, gain of hydrogen or gain of electrons; the gain of electrons enables you to calculate an oxidation state. Hydride
Reducing Agents Linda M. Sweeting 1998 Reduction is defined in chemistry as loss of oxygen, gain of hydrogen or gain of electrons; the gain of electrons enables you to calculate an oxidation state. Hydride
An Aldol Condensation to synthesize Chalcones. By: Blake Burger FFR#3
 An Aldol Condensation to synthesize Chalcones By: Blake Burger FFR#3 Burger, 2 Introduction A chalcone is a molecule formed by two core functional groups: an aromatic ketone and an enone. Chalcones are
An Aldol Condensation to synthesize Chalcones By: Blake Burger FFR#3 Burger, 2 Introduction A chalcone is a molecule formed by two core functional groups: an aromatic ketone and an enone. Chalcones are
Benzylamine reacts with nitrous acid to form unstable diazonium salt, which in turn gives alcohol with the evolution of nitrogen gas.
 Benzylamine reacts with nitrous acid to form unstable diazonium salt, which in turn gives alcohol with the evolution of nitrogen gas. On the other hand, aniline reacts with HNO2 at a low temperature to
Benzylamine reacts with nitrous acid to form unstable diazonium salt, which in turn gives alcohol with the evolution of nitrogen gas. On the other hand, aniline reacts with HNO2 at a low temperature to
