MCM-41 Ordered Mesoporous Molecular Sieves Synthesis and Characterization
|
|
|
- Estella Chapman
- 6 years ago
- Views:
Transcription
1 Materials Research, Vol. 2, No. 3, , MCM-41 Ordered Mesoporous Molecular Sieves Synthesis and Characterization Rogério A.A. Melo a, Marcus V. Giotto a, João Rocha b, Ernesto A. Urquieta-González a a Departamento de Engenharia Química, Universidade Federal de São Carlos, C.P. 676, São Carlos - SP, Brazil b Departamento de Química, Universidade de Aveiro, 3810 Aveiro, Portugal Received: August 15, 1998; Revised: March 30, 1999 The aim of this work was to study the hydrothermal synthesis of Si and SiAlMCM-41 performed under both autogenic pressure and refluxing conditions. XRD data showed that the MCM-41 phase may be formed by both processes and that the synthesized material in the presence of Al and/or under reflux presents the hexagonally arrangement of less ordered mesopores. However, as verified by XRD and physisorption data, the order was improved with higher synthesis times. 29 Si and 1 H - 29 Si C/P MAS NMR spectra showed that a great part of the Si atoms exists as silanol groups which originate resonance peaks at -110, -100 and -91 ppm. The presence of Al atoms may generate Si(3Si, Al) and Si(2Si, 2Al) environments which might be contributing to resonance peaks at -100 and -91 ppm. The 27 Al MAS NMR spectrum of the as synthesized AlSiMCM-41 showed a resonance peak of tetrahedral framework aluminum close to 53 ppm and two others, one close to 14 ppm attributed to Al(H2O)6 +3 species and the other a weak signal close to 32 ppm attributed to pentacoordinated Al. 27 Al MAS NMR spectra of the calcined sample showed a peak at 0 ppm corresponding to an hexacoordinated extra-framework aluminum formed during calcination. Keywords: MCM-41, synthesis, characterization 1. Introduction Molecular sieves with a zeolite structure and pore diameter < 15 Å exhibit a shape selectivity that enables them to be used as adsorbents or catalysts in a variety of processes. However, reactions involving bulky molecules require structures with channel diameters at mesopore scale. In the early nineties 1,2, scientists from Mobil Oil Corporation synthesized ordered mesoporous materials of the M41S type, family to which MCM-41 belongs. This material possesses a porous system consisting of hexagonally arranged channels with diameters varying from 15 to 100 Å. MCM-41 has attracted the attention of scientists due to its elevated specific surface area, high thermal and hydrothermal stability, possibility of controlling its pore size and its hydrophobicity and acidity. These characteristics have made MCM-41 a promising material as catalyst and/or support 3 and to be used in industrial processes of adsorption 4, ion exchange 5 and environmental control 6. The scope of the current work was to investigate the hydrothermal synthesis of SiMCM-41 and AlSiMCM-41 performed under both autogenic pressure and refluxing conditions. The solids obtained were characterized by XRD, SEM, nitrogen physisorption TGA/DTG, 29 Si MAS NMR; 1 H - 29 Si CP/MAS NMR and 27 Al MAS NMR. 2. Materials and Methods 2.1. Synthesis of MCM-41 The molar compositions 1,2,7 and synthesis times used are listed in Table 1. The used precursors consisted basically of amorphous silica (Aerosil-200/Degussa); sodium trisilicate (Aldrich); tetramethylammonium hydroxide (Aldrich); sodium aluminate (Riedel De Haën); cetyltrimethylammonium bromide (Merck) and distilled water. Part of the reaction mixture was placed in teflon cups inside of steel autoclaves being subsequently submitted to hydrothermal treatment under autogenic pressure in a oven at 100 C. The remaining part was treated at the same tem-
2 174 Melo et al. Materials Research Table 1. Molar compositions and synthesis time. Samples Composition Reflux Y(h) Aut Pres Y(h) SiMCM-41 SiO 2:0.07Na 2O:0.27CTMA:0.14TMAOH:100H 2O 24; ; 288 AlSiMCM-41 SiO 2:0.025Al 2O 3:0.07Na 2O:0.27CTMA:0.14TMAOH:100H 2O 24; ; 288 perature under refluxing conditions. After the synthesis the solids were separated from the reaction mixture through centrifugation, washed and dried at 110 C. Removal of the organic material occurred by calcination at 550 C under nitrogen flow (100 cm 3 /min g) for 1 h and subsequently under synthetic air flow (100 cm 3 /min g) for 6 h. The samples were designated as SiMCM-41(X/Y) and Al- SiMCM-41(X/Y), where X = P presents the samples obtained under autogenic pressure and X = R under refluxing conditions; Y corresponds to the synthesis time in hours Characterization X-ray powder diffraction patterns were recorded on a Siemens D500 diffractometer using a monochromated CuΚ α radiation in the angular range from 1 to 10 (2θ); the 2θ scanning speed was 0.6 /min. The scanning electron micro-analysis was performed on a Zeiss DSM 960 Scanning Electron Microscope operated at 30 kv. Thermogravimetric analysis was carried out on a Thermal Analyst 2100 TA Instruments equipment in the temperature range from 30 to 900 C (heating rate 3 C/min) in O 2 flow (30 cm 3 /min). Nitrogen physisorption measurement at -196 C was conducted on a NOVA 1200 TGA Quantachrome Corporation analyzer. Solid state 29 Si MAS NMR spectra were recorded on a Varian 400 MHz spectrometer at 79.5 MHz with 50 pulses (4.3 µs) and 5 khz spinning rates. The recycle delay was 100 s. The 1 H - 29 Si C/P MAS NMR spectrum was recorded with 4.5 µs 1 H (90 ) pulses with a contact time of 5 ms and 5 s of recycle delay. The 27 Al MAS NMR spectra were recorded on a Bruker MSL 400P spectrometer operating at MHz with 0.6 s (π/18) pulses and a recycle delay of 0.8 s. 3. Results and Discussion 3.1. X-ray powder diffraction XRD analysis of the as synthesized samples indicated that the mesoporous MCM-41 molecular sieve may be obtained by both processes and allowed identification of the peaks relative to the (100), (110), (200) and (210) planes corresponding to an hexagonal structure with symmetry P6 for the reflection planes (hk0), indicating that there are no planes for l 0 1,2. The values of the parameter a o of the hexagonal pore arrangement for different samples are summarized in Table 2. These values are seen to be very representative compared to those presented by other authors who used the same surfactant 8. The parameter a o is also seen to show a tendency to increase with synthesis time for both processes, which has been attributed to the increase in the wall thickness of the mesopores 9. Figure 1 shows X-ray diffraction patterns for samples SiMCM-41 and AlSiMCM-41 synthesized in 48 h. The diffraction peak related to the (100) plane is seen to be more Figure 1. XRD of the as synthesized: a) SiMCM-41, and b) AlSiMCM-41 samples.
3 Vol. 2, No. 3, 1999 MCM-41 Ordered Mesoporous Molecular Sieves Synthesis 175 Table 2. Parameter a0 for SiMCM-41 and AlSiMCM-41 samples. Samples a 0 (Å) SiMCM-41(R/24) SiMCM-41(R/108) SiMCM-41(P/48) SiMCM-41(P/288) AlSiMCM-41(R/24) AlSiMCM-41(R/108) AlSiMCM-41(P/48) AlSiMCM-41(P/288) intense for samples obtained under autogenic pressure when compared to those obtained under reflux, which can be interpreted as an indication of higher ordering of the mesopore channels 10. The materials synthesized in the presence of aluminum exhibit broader and slightly less intense diffraction peaks, indicating that within 48 h synthesis, the hexagonal arrangement of the mesopores in these materials is less ordered compared to SiMCM Nitrogen physisorption The adsorption/desorption isotherms of N 2 (Fig. 2) and the values of specific surface area (S BET ), mean diameter of the mesopores (d mp ), total volume of pores (V t ) and wall thickness (ε) obtained for calcined SiMCM-41 (Table 3) are in agreement with those published by other authors 11. Curves in Fig. 2 and data from Table 3 show an increase in the adsorption capacity of N 2 with time for the SiMCM-41 samples synthesized under reflux, indicating an improvement in the order of the mesopore phase, already observed by the increase in the XRD peak intensity (Fig. 3). However, as it has been observed from the data in Fig. 1, the MCM-41 phase is formed more readily under autogenic pressure, showing a small dependence with synthesis time, as it can be seen in Fig. 4 and from the data in Table 3. Figure 2. Adsorption/desorption isotherms of N2 for calcined SiMCM-41 samples TG/DTG analysis Thermogravimetric analysis of the as synthesized Al- SiMCM-41(P/288) sample presented a DTG curve typical of MCM-41 (Fig. 5). The as synthesized material presents approximately 50 wt% of the MCM-41 phase. The remaining part corresponds to organic material whose mass loss is related to three exothermal stages: (a) between C: decomposition of the surfactant 2 ; (b) between C: breaking of the hydrocarbon chain, and (c) between C: combustion of the surfactant and water loss associated with condensation of silanol groups 12. Figure 3. XRD of the as synthesized SiMCM-41(R/Y) samples.
4 176 Melo et al. Materials Research Table 3. Textural characteristics of the calcined SiMCM-41 samples. Sample Average diameter of the mesopores d mp (Å) Wall thickness ε* (Å) Area S BET (m 2 /g) Total pore volume (V t) (cm 3 /g) SiMCM-41(R/24) SiMCM-41(R/108) SiMCM-41(P/48) SiMCM-41(P/288) ε = a0-dmp. Figure 4. XRD of the as synthesized SiMCM-41(P/Y) samples. Figure 5. TGA-DTG analysis of the as synthesized AlSiMCM-41 (P/288) sample NMR spectra Figure Si MAS NMR spectra of the AlSiMCM-41 (R/108) samples. The 29 Si MAS NMR spectra of the as synthesized and calcined AlSiMCM-41(R/108) samples, shown in Fig. 6 bears much resemblance to that of amorphous silica, indicating that the local arrangement of the Si-O-Si bonds at the pore walls is irregular and that the angle between these bonds vary over a wide range. The 1 H - 29 Si CP/MAS NMR spectrum (Fig. 7) of the calcined sample, shows that, similar to amorphous silica, part of the Si atoms exist as silanol groups which originate resonance peaks at -110, -100 and -91 ppm. These shifts have been attributed to the Si(4SiO), Si(OSi) 3 OH and Si(OSi) 2 (OH) 2 environments. Chemical analysis of this sample supplied a ratio Si/Al = 9. The presence of Al atoms in the MCM-41 network may generate Si(3Si, Al) and Si(2Si, 2Al) environments, which might be contributing to the resonance peak intensities at -100 and -91 ppm 13. The 27 Al MAS NMR spectra of the as synthesized and calcined AlSiMCM-41(R/108) samples (Fig. 8), present a resonance peak close to 53 ppm which is attributed to structural tetrahedral coordinated aluminum. The as synthesized sample shows one peak close to 14 ppm and a weak signal at 32 ppm, which have been attributed to hexacoordinated aluminum, possibly forming Al(H 2 O) 6 3+ species within the pores and pentacoordinated species, respectively. These resonances were not detected in the calcinated sample 13. For the calcined sample the peak
5 Vol. 2, No. 3, 1999 MCM-41 Ordered Mesoporous Molecular Sieves Synthesis 177 Figure 7. 1 H- 29 Si CP/MAS NMR spectrum of the AlSiMCM-41 (R/108) sample. around 0 ppm is representative of hexacoordinated aluminum belonging to extra-structural species formed during calcination 13,14. Figure Al MAS RMN spectra of the AlSiMCM-41(R/108) samples SEM micrographs Figure 9 presents typical micrographs of the calcined SiMCM-41(R/108) and SiMCM-41(P/48) samples. Formation of the SiMCM-41 phase at hydrothermal conditions under autogenic pressure is seen to result, in shorter time, in a solid consisting of small agglomerates (Fig. 9a). The SiMCM-41 obtained under autogenic pressure, synthesized under reflux conditions (Fig. 9b) presents agglomerates, similar to those obtained under autogenic pressure, and worm-like particles; this morphology practically did not change with synthesis time. The AlSiMCM-41 samples exhibit agglomerates with sizes between 3 and 200 µm, independent of the used synthesis method (Fig. 10). These samples did not show a tendency to form particles with a particular morphology. 4. Conclusion XRD analysis of SiMCM-41 and AlSiMCM-41 samples showed that the MCM-41 phase may be formed by both processes and that the synthesized material in the presence of aluminum and/or under refluxing conditions Figure 9. SEM micrographs of the SiMCM-41 samples. (a) Si MCM-41 (p/48), (b) Si MCM-41 (R/108).
6 178 Melo et al. Materials Research Figure 10. SEM micrographs of the AlSiMCM-41 samples. (a) AlSi MCM-41 (R/24), (b) AlSi MCM-41 (P/288). presents a hexagonal arrangement of less ordered mesopores. However, in the latter case, as verified by XRD and physisorption data, improvement in order was observed with higher synthesis times. 29 Si MAS NMR and 1 H - 29 Si C/P MAS NMR spectra showed that a great part of the Si atoms exist as silanol groups which originate resonance peaks at -110, -100 and -91 ppm. The presence of Al atoms may generate Si(3Si,Al) and Si(2Si, 2Al) environments which might be contributing to resonance peaks at -100 and -91 ppm. The 27 Al MAS NMR spectra of the as synthesized AlSiMCM-41(R/108) confirm a strong resonance peak of tetrahedral framework aluminum close to 53 ppm and two others resonances, one close to 14 ppm attributed to Al(H 2 O) 6 +3 species within the pores and the other, a weak signal close to 32 ppm attributed to pentacoordinated aluminum species. 27 Al MAS NMR spectra of the calcined sample showed the peak at 53 ppm and another at 0 ppm corresponding to an hexacoordinated extra-framework aluminum species formed during the calcination process. Acknowledgments The authors thankfully acknowledge the financial support for this study provided by FAPESP (grant n. 97/0317-0) and for the Doctor s scholarship granted by CAPES to Rogério A.A. de Melo. References 1. Kresge, C.T.; Leonowicz, M.E.; Roth, W.J.; Vartuli, J.C.; Beck, J. S. Nature, n. 359, p. 710, Beck, J.S.; Vartuli, J.C.; Leonowicz, M.E.; Kresge, C.T.; Schmitt, K.D.; Chu, C.T.W.; Olson, D.H.; Sheppard, E.W.; McCullen, S.B.; Higgins, J.B.; Schlenker, J.L. J. Am. Chem. Soc., n. 114, p , Corma, A.; Martínez, A.; Martínez-Soria, V.; Montón, J.B. J. Catal., n. 153, p. 25, Rathousky, J.; Zukai, A.; Franke, O.; Schulz-Ekloff, G. J. Chem. Soc., Faraday Trans., n. 91, p. 937, Kim, M.J.; Kwak, J.H.; Shinae, J.; Shinae, Ryong, R. J. Phys. Chem., n. 99, p , Feng, X.; Fryxell, G.E.; Wang, L. -Q.; Kilm. A.Y.; Liu, J.; Kemner, K.M. Science, n. 276, p. 923, Monnier, A.; Schüth, F.; Huo, Q.; Kumar, D.; Margolese, D.; Maxwel, R.S.; Stucky, G.D.; Krishnamurty, M.; Petroff, P.; Firouzi, A.; Janicke, M.; Chmelka, B.F. Science., n. 261, p. 1299, Carvalho, W.A.; Varaldo, P.B.; Wallau, M. Zeolites, n. 18, p. 408, Cheng. C. -F.; Zhou, W.; et al. J. Am. Chem. Soc., Faraday Trans., n. 93, p. 359, Beck, J.S.; Vartuli, J.C. Current Opinion in Solid State and Mat. Science, n. 1, p. 76, Cui, J.; Yue, J.; Sun, Y.H.; Dong, W.Y.; Gao, Z. Progress in Zeolite and Microporous Materials, Chon, H.; Ihm, H.K.; Uh, Y.S., eds., Stud. Surf. Sci. Catal., Elsevier, The Netherlans, v. 105, p. 687, Montes, A.; Cosenza, E.; Giannetto, G.; Urquieta, E.; Melo, R.A.; Gnep, N.S.; Guisnet, M. Mesoporous Molecular Sieves, Bonneviot, L.; Béland, F.; Danumah, C.; Giasson, S.; Kaliaguine, S., eds., Stud. Surf.
7 Vol. 2, No. 3, 1999 MCM-41 Ordered Mesoporous Molecular Sieves Synthesis 179 Sci. Catal., Elsevier, The Netherlans, v. 117, p. 237, Rocha, J.; Liepold, A.; Roos, K.; Reschetilowski, W.; Esculcas, A.P.; Philippou, A.; Anderson, M.W. J. Chem. Soc., Faraday Trans., v. 92, n. 22, p. 4623, Luan, Z.; Cheng, C.-F.; Zhou, W.; Klinowski, J. J. Phys. Chem., v. 99, n. 1018, 1995.
Quantitative measurement of a mixture of mesophases cubic MCM-48 and hexagonal MCM-41 by 13C CP/MAS NMR
 University Technology Malaysia From the SelectedWorks of Hadi Nur May, 2004 Quantitative measurement of a mixture of mesophases cubic MCM-48 and hexagonal MCM-41 by 13C CP/MAS NMR Hadi Nur, University
University Technology Malaysia From the SelectedWorks of Hadi Nur May, 2004 Quantitative measurement of a mixture of mesophases cubic MCM-48 and hexagonal MCM-41 by 13C CP/MAS NMR Hadi Nur, University
Synthesis, Characterization and Catalytic Activity of MCM-41 Catalyst for Nitration of Phenol
 http://www.e-journals.in Chemical Science Transactions DOI:10.7598/cst2015.952 2015, 4(2), 438-442 RESEARCH ARTICLE Synthesis, Characterization and Catalytic Activity of MCM-41 Catalyst for Nitration of
http://www.e-journals.in Chemical Science Transactions DOI:10.7598/cst2015.952 2015, 4(2), 438-442 RESEARCH ARTICLE Synthesis, Characterization and Catalytic Activity of MCM-41 Catalyst for Nitration of
sensors ISSN by MDPI
 Sensors 2006, 6, 318-323 sensors ISSN 1424-8220 2006 by MDPI http://www.mdpi.org/sensors Gas Sensing Properties of Ordered Mesoporous SnO 2 Thorsten Wagner 1,2,*, Claus-Dieter Kohl 1, Michael Fröba 2 and
Sensors 2006, 6, 318-323 sensors ISSN 1424-8220 2006 by MDPI http://www.mdpi.org/sensors Gas Sensing Properties of Ordered Mesoporous SnO 2 Thorsten Wagner 1,2,*, Claus-Dieter Kohl 1, Michael Fröba 2 and
Supporting Information
 Supporting Information Wiley-VCH 2007 69451 Weinheim, Germany Efficient Bifunctional Nanocatalysts by Simple Postgrafting of Spatially-Isolated Catalytic Groups on Mesoporous Materials By Krishna K. Sharma
Supporting Information Wiley-VCH 2007 69451 Weinheim, Germany Efficient Bifunctional Nanocatalysts by Simple Postgrafting of Spatially-Isolated Catalytic Groups on Mesoporous Materials By Krishna K. Sharma
Characterization of Highly Ordered MCM-41 Silicas Using X-ray Diffraction and Nitrogen Adsorption
 Langmuir 1999, 15, 5279-5284 5279 Characterization of Highly Ordered MCM-41 Silicas Using X-ray Diffraction and Nitrogen Adsorption Michal Kruk and Mietek Jaroniec* Department of Chemistry, Kent State
Langmuir 1999, 15, 5279-5284 5279 Characterization of Highly Ordered MCM-41 Silicas Using X-ray Diffraction and Nitrogen Adsorption Michal Kruk and Mietek Jaroniec* Department of Chemistry, Kent State
Synthesis of Mesoporous ZSM-5 Zeolite Crystals by Conventional Hydrothermal Treatment
 Synthesis of Mesoporous ZSM-5 Zeolite Crystals by Conventional Hydrothermal Treatment Ming Zhou,* Ali A. Rownaghi, and Jonas Hedlund,* *Chemical Technology, Luleå University of Technology, SE-971 87 Luleå,
Synthesis of Mesoporous ZSM-5 Zeolite Crystals by Conventional Hydrothermal Treatment Ming Zhou,* Ali A. Rownaghi, and Jonas Hedlund,* *Chemical Technology, Luleå University of Technology, SE-971 87 Luleå,
Supporting Information. for. Delamination of Layered Zeolite Precursors under. Mild Conditions: Synthesis of UCB-1 via
 Supporting Information for Delamination of Layered Zeolite Precursors under Mild Conditions: Synthesis of UCB-1 via Fluoride/Chloride Anion-Promoted Exfoliation Isao Ogino, Michael M. Nigra, Son-Jong Hwang,
Supporting Information for Delamination of Layered Zeolite Precursors under Mild Conditions: Synthesis of UCB-1 via Fluoride/Chloride Anion-Promoted Exfoliation Isao Ogino, Michael M. Nigra, Son-Jong Hwang,
Hydrothermal stability of MCM-48 improved by postsynthesis restructuring in salt solution
 Microporous and Mesoporous Materials 41 (2000) 119±127 www.elsevier.nl/locate/micromeso Hydrothermal stability of MCM-48 improved by postsynthesis restructuring in salt solution Shinae Jun a, Ji Man Kim
Microporous and Mesoporous Materials 41 (2000) 119±127 www.elsevier.nl/locate/micromeso Hydrothermal stability of MCM-48 improved by postsynthesis restructuring in salt solution Shinae Jun a, Ji Man Kim
PREPARATION OF MCM-48 MESOPOROUS MOLECULAR SIEVE INFLUENCE OF PREPARATION CONDITIONS ON THE STRUCTURAL PROPERTIES
 Digest Journal of Nanomaterials and Biostructures Vol. 11, No. 1, January - March 2016, p. 271-276 PREPARATION OF MCM-48 MESOPOROUS MOLECULAR SIEVE INFLUENCE OF PREPARATION CONDITIONS ON THE STRUCTURAL
Digest Journal of Nanomaterials and Biostructures Vol. 11, No. 1, January - March 2016, p. 271-276 PREPARATION OF MCM-48 MESOPOROUS MOLECULAR SIEVE INFLUENCE OF PREPARATION CONDITIONS ON THE STRUCTURAL
Optimization of Synthetic Parameters for Mesoporous Molecular Sieve MCM-41 Using Surfactant CTACl
 Synthetic Parameters for MCM-41 Using CTACl 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. Photophysik. Photo. Chem. 1950, 45, 222. (d) Adams, R.; Elslager, E. F.; Young, T. E. J. Am. Chem. Soc. 1953,
Synthetic Parameters for MCM-41 Using CTACl 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. Photophysik. Photo. Chem. 1950, 45, 222. (d) Adams, R.; Elslager, E. F.; Young, T. E. J. Am. Chem. Soc. 1953,
Temperature Effect on Pore Structure of Nanostructured Zeolite Particles Synthesized by Aerosol Spray Method
 Temperature Effect on Pore Structure of Nanostructured Zeolite Particles Synthesized by Aerosol Spray Method Yu-Chih Lin 1 and Hsunling Bai 2 1. Department of Environmental Engineering and Health, Yuanpei
Temperature Effect on Pore Structure of Nanostructured Zeolite Particles Synthesized by Aerosol Spray Method Yu-Chih Lin 1 and Hsunling Bai 2 1. Department of Environmental Engineering and Health, Yuanpei
Electronic Supplementary Information (ESI) Efficient synthesis of the Cu-SSZ-39 catalyst for DeNOx applications
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information (ESI) Efficient synthesis of the Cu-SSZ-39 catalyst for DeNOx
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information (ESI) Efficient synthesis of the Cu-SSZ-39 catalyst for DeNOx
Min Bum Park, Sang Hyun Ahn, Nak Ho Ahn and Suk Bong Hong*
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 S1 Supplementary Information for Charge density mismatch synthesis of MEI- and BPH-type zeolites
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 S1 Supplementary Information for Charge density mismatch synthesis of MEI- and BPH-type zeolites
Control of Wall Thickness and Extraordinarily High Hydrothermal Stability of Nanoporous MCM-41 Silica
 Journal of the Chinese Chemical Society, 2007, 54, 35-40 35 Control of Wall Thickness and Extraordinarily High Hydrothermal Stability of Nanoporous MCM-41 Silica Chi-Feng Cheng* ( ), Shu-Hsien Chou ( ),
Journal of the Chinese Chemical Society, 2007, 54, 35-40 35 Control of Wall Thickness and Extraordinarily High Hydrothermal Stability of Nanoporous MCM-41 Silica Chi-Feng Cheng* ( ), Shu-Hsien Chou ( ),
Julien Schmitt, postdoc in the Physical Chemistry department. Internship 2010: Study of the SAXS scattering pattern of mesoporous materials
 Before starting Julien Schmitt, postdoc in the Physical Chemistry department Internship 2010: Study of the SAXS scattering pattern of mesoporous materials PhD 2011-2014: Self-assembly mechanism of mesoporous
Before starting Julien Schmitt, postdoc in the Physical Chemistry department Internship 2010: Study of the SAXS scattering pattern of mesoporous materials PhD 2011-2014: Self-assembly mechanism of mesoporous
Synthesis of uniform hollow silica spheres with ordered mesoporous shells in a CO 2 induced nanoemulsion. Supporting Information
 Synthesis of uniform hollow silica spheres with ordered mesoporous shells in a CO 2 induced nanoemulsion Yueju Zhao, Jianling Zhang, Wei Li, Chaoxing Zhang, Buxing Han* Beijing National Laboratory for
Synthesis of uniform hollow silica spheres with ordered mesoporous shells in a CO 2 induced nanoemulsion Yueju Zhao, Jianling Zhang, Wei Li, Chaoxing Zhang, Buxing Han* Beijing National Laboratory for
Homogeneous vs Heterogeneous Catalysts
 Homogeneous vs Heterogeneous Catalysts Homogeneous Liquid phase Low temperature High selectivity High diffusivity Easy heat transfer Difficult catalyst separation Expensive recycling Well-defined active
Homogeneous vs Heterogeneous Catalysts Homogeneous Liquid phase Low temperature High selectivity High diffusivity Easy heat transfer Difficult catalyst separation Expensive recycling Well-defined active
NANOSILICEOUS MATRIX FOR DRUG INCAPSULATION
 NANOSILICEOUS MATRIX FOR DRUG INCAPSULATION L. G. HANU 1, A. M. HANU 1, E. POPOVICI 1, G. BURTICĂ 2, D. TIMPU 3 1 Faculty of Chemistry, Univ. Al. I. Cuza, Iaºi 2 University Politehnica, Timiºoara 3 P.
NANOSILICEOUS MATRIX FOR DRUG INCAPSULATION L. G. HANU 1, A. M. HANU 1, E. POPOVICI 1, G. BURTICĂ 2, D. TIMPU 3 1 Faculty of Chemistry, Univ. Al. I. Cuza, Iaºi 2 University Politehnica, Timiºoara 3 P.
Electronic Supplementary Information
 Electronic Supplementary Information Designed Copper-amine Complex as an Efficient Template for One-pot Synthesis of Cu-SSZ-13 Zeolite with Excellent Activity for Selective Catalytic Reduction of NOx by
Electronic Supplementary Information Designed Copper-amine Complex as an Efficient Template for One-pot Synthesis of Cu-SSZ-13 Zeolite with Excellent Activity for Selective Catalytic Reduction of NOx by
Synthesis and Characterization of Magnesium Substituted Aluminophosphate Molecular Sieves with AEL Structure
 Journal of Natural Gas Chemistry 13(2004)231 237 Synthesis and Characterization of Magnesium Substituted Aluminophosphate Molecular Sieves with AEL Structure Benjing Xu, Ling Qian, Xinmei Liu, Chunmin
Journal of Natural Gas Chemistry 13(2004)231 237 Synthesis and Characterization of Magnesium Substituted Aluminophosphate Molecular Sieves with AEL Structure Benjing Xu, Ling Qian, Xinmei Liu, Chunmin
Supporting Information. Nanoscale Kirkendall Growth of Silicalite-1 Zeolite Mesocrystals with. Controlled Mesoporosity and Size
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Supporting Information Nanoscale Kirkendall Growth of Silicalite-1 Zeolite Mesocrystals with Controlled
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Supporting Information Nanoscale Kirkendall Growth of Silicalite-1 Zeolite Mesocrystals with Controlled
Amine-impregnated silica monolith with a hierarchical pore structure: enhancement of CO 2 capture capacity
 1 Electronic Supplementary Information (ESI) Amine-impregnated silica monolith with a hierarchical pore structure: enhancement of CO 2 capture capacity for Chao Chen, Seung-Tae Yang, Wha-Seung Ahn* and
1 Electronic Supplementary Information (ESI) Amine-impregnated silica monolith with a hierarchical pore structure: enhancement of CO 2 capture capacity for Chao Chen, Seung-Tae Yang, Wha-Seung Ahn* and
Rearrangement of allyl phenyl ether over Al-MCM-41
 Journal of Catalysis 229 (2005) 105 113 www.elsevier.com/locate/jcat Rearrangement of allyl phenyl ether over Al-MCM-41 Nevin T. Mathew, S. Khaire, S. Mayadevi, R. Jha, S. Sivasanker National Chemical
Journal of Catalysis 229 (2005) 105 113 www.elsevier.com/locate/jcat Rearrangement of allyl phenyl ether over Al-MCM-41 Nevin T. Mathew, S. Khaire, S. Mayadevi, R. Jha, S. Sivasanker National Chemical
Dry-gel conversion synthesis of Cr-MIL-101 aided by grinding: High surface area high yield synthesis with minimum purification
 Electronic Supporting Informations (ESI): Dry-gel conversion synthesis of Cr-MIL-101 aided by grinding: High surface area high yield synthesis with minimum purification Jun Kim, Yu-Ri Lee and Wha-Seung
Electronic Supporting Informations (ESI): Dry-gel conversion synthesis of Cr-MIL-101 aided by grinding: High surface area high yield synthesis with minimum purification Jun Kim, Yu-Ri Lee and Wha-Seung
Synthesis and Alkylation of Phenol over Periodic Mesoporous Organosilicas Functionalized with Sulfonic Acid Group
 Zeolitic Materials, Vol. 3, No. 1, December 2002, 20-28 Synthesis and Alkylation of Phenol over Periodic Mesoporous Organosilicas Functionalized with Sulfonic Acid Group Xingdong Yuan, Hyung Ik Lee, Jin
Zeolitic Materials, Vol. 3, No. 1, December 2002, 20-28 Synthesis and Alkylation of Phenol over Periodic Mesoporous Organosilicas Functionalized with Sulfonic Acid Group Xingdong Yuan, Hyung Ik Lee, Jin
Modification of Surface and Structural Properties of Ordered Mesoporous Silicates
 Adsorption 5, 39 45 (1999) c 1999 Kluwer Academic Publishers. Manufactured in The Netherlands. Modification of Surface and Structural Properties of Ordered Mesoporous Silicates MIETEK JARONIEC, MICHAL
Adsorption 5, 39 45 (1999) c 1999 Kluwer Academic Publishers. Manufactured in The Netherlands. Modification of Surface and Structural Properties of Ordered Mesoporous Silicates MIETEK JARONIEC, MICHAL
Application of Mesoporous MCM-48 and SBA-15 Materials for the Separation of Biochemicals Dissolved in Aqueous Solution
 Korean J. Chem. Eng., 21(1), 246-251 (2004) Application of Mesoporous MCM-48 and SBA-15 Materials for the Separation of Biochemicals Dissolved in Aqueous Solution Jae Wook Lee, Dae Lim Cho*, Wang Geun
Korean J. Chem. Eng., 21(1), 246-251 (2004) Application of Mesoporous MCM-48 and SBA-15 Materials for the Separation of Biochemicals Dissolved in Aqueous Solution Jae Wook Lee, Dae Lim Cho*, Wang Geun
Electronic Supplementary Information (ESI)
 Electronic Supplementary Information (ESI) Cu-SSZ-39, an active and hydrothermally stable catalyst for the selective catalytic reduction of NOx Manuel Moliner, *a Cristina Franch, a Eduardo Palomares,
Electronic Supplementary Information (ESI) Cu-SSZ-39, an active and hydrothermally stable catalyst for the selective catalytic reduction of NOx Manuel Moliner, *a Cristina Franch, a Eduardo Palomares,
Supporting Information
 Supporting Information Tetraethylammonium hydroxide (35 wt.%) (TEAOH), aluminum powder, fumed silica, zinc acetate dehydrate, Ludox HS-40, pure silica MCM-41, and tetraethylammonium fluoride were purchased
Supporting Information Tetraethylammonium hydroxide (35 wt.%) (TEAOH), aluminum powder, fumed silica, zinc acetate dehydrate, Ludox HS-40, pure silica MCM-41, and tetraethylammonium fluoride were purchased
Synthesis of Mesoporous Silica in Acidic Condition by Solvent Evaporation Method
 Korean J. Chem. Eng., 16(3), 331-337 (1999) Synthesis of Mesoporous Silica in Acidic Condition by Solvent Evaporation Method Hyun-Seog Roh, Jong-San Chang and Sang-Eon Park Industrial Catalysis Research
Korean J. Chem. Eng., 16(3), 331-337 (1999) Synthesis of Mesoporous Silica in Acidic Condition by Solvent Evaporation Method Hyun-Seog Roh, Jong-San Chang and Sang-Eon Park Industrial Catalysis Research
Zeolite Nanoclusters Coated onto the Mesopore Walls of SBA-15
 Published on Web 10/15/2004 Zeolite Nanoclusters Coated onto the Mesopore Walls of SBA-15 Trong-On Do, Andrei Nossov, Marie-Anne Springuel-Huet, Celine Schneider, Jeremy L. Bretherton, Colin A. Fyfe, and
Published on Web 10/15/2004 Zeolite Nanoclusters Coated onto the Mesopore Walls of SBA-15 Trong-On Do, Andrei Nossov, Marie-Anne Springuel-Huet, Celine Schneider, Jeremy L. Bretherton, Colin A. Fyfe, and
Supplementary Information. Simple Quaternary Ammonium Cations-Templated Syntheses
 Supplementary Information Simple Quaternary Ammonium Cations-Templated Syntheses of Extra-Large Pore Germanosilicate Zeolites Risheng Bai, Qiming Sun, Ning Wang, Yongcun Zou, Guanqi Guo, Sara Iborra, Avelino
Supplementary Information Simple Quaternary Ammonium Cations-Templated Syntheses of Extra-Large Pore Germanosilicate Zeolites Risheng Bai, Qiming Sun, Ning Wang, Yongcun Zou, Guanqi Guo, Sara Iborra, Avelino
Supporting Information For
 Supporting Information For Heteroatom-Substituted Delaminated Zeolites as Solid Lewis-Acid Catalysts Xiaoying Ouyang 1, Son-Jong Hwang 2, Dan Xie 3, Thomas Rea 3, Stacey I. Zones*,1,3, Alexander Katz*,1
Supporting Information For Heteroatom-Substituted Delaminated Zeolites as Solid Lewis-Acid Catalysts Xiaoying Ouyang 1, Son-Jong Hwang 2, Dan Xie 3, Thomas Rea 3, Stacey I. Zones*,1,3, Alexander Katz*,1
A triazine-based covalent organic polymer for efficient CO 2 adsorption
 Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 Supporting Information A triazine-based covalent organic polymer for efficient CO
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 Supporting Information A triazine-based covalent organic polymer for efficient CO
supports Lenka Matejova 1, Satu Pitkäaho 2 *, Satu Ojala 2, Tomas Cajthaml 3, Riitta L. Keiski 2 and Olga Solcova 1
 66 Oxidation of dichloromethane over Pt and Au impregnated on extracted and calcined TiO 2 supports Lenka Matejova 1, Satu Pitkäaho 2 *, Satu Ojala 2, Tomas Cajthaml 3, Riitta L. Keiski 2 and Olga Solcova
66 Oxidation of dichloromethane over Pt and Au impregnated on extracted and calcined TiO 2 supports Lenka Matejova 1, Satu Pitkäaho 2 *, Satu Ojala 2, Tomas Cajthaml 3, Riitta L. Keiski 2 and Olga Solcova
Preparation and characterization of poly(styrenemethacrylic acid)/mcm-41 core/shell nanocomposite microspheres
 e-polymers 2009, no. 121 http://www.e-polymers.org ISSN 1618-7229 Preparation and characterization of poly(styrenemethacrylic acid)/mcm-41 core/shell nanocomposite microspheres Yansheng Zhao, * Xingji
e-polymers 2009, no. 121 http://www.e-polymers.org ISSN 1618-7229 Preparation and characterization of poly(styrenemethacrylic acid)/mcm-41 core/shell nanocomposite microspheres Yansheng Zhao, * Xingji
Synthesis and Characterization of Nanoporous Carbon Materials; The Effect of Surfactant Concentrations and Salts
 ISSN: 0973-4945; CODEN ECJHAO E- Chemistry http://www.e-journals.net 2011, 8(1), 196-200 Synthesis and Characterization of Nanoporous Carbon Materials; The Effect of Surfactant Concentrations and Salts
ISSN: 0973-4945; CODEN ECJHAO E- Chemistry http://www.e-journals.net 2011, 8(1), 196-200 Synthesis and Characterization of Nanoporous Carbon Materials; The Effect of Surfactant Concentrations and Salts
PREPARATION, CHARACTERIZATION AND OPTICAL PROPERTIES OF MCM-41/RHODAMINE B. Yue-Xiang Liu and Qing-Zhou Zhai *
 , 145-150. ISSN 1011-3924 Printed in Ethiopia 2009 Chemical Society of Ethiopia SHORT COMMUNICATION PREPARATION, CHARACTERIZATION AND OPTICAL PROPERTIES OF MCM-41/RHODAMINE B Yue-Xiang Liu and Qing-Zhou
, 145-150. ISSN 1011-3924 Printed in Ethiopia 2009 Chemical Society of Ethiopia SHORT COMMUNICATION PREPARATION, CHARACTERIZATION AND OPTICAL PROPERTIES OF MCM-41/RHODAMINE B Yue-Xiang Liu and Qing-Zhou
Template-Free Synthesis of Beta Zeolite Membranes on Porous α-al 2 O 3 Supports
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information for Template-Free Synthesis of Beta Zeolite Membranes on Porous
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information for Template-Free Synthesis of Beta Zeolite Membranes on Porous
MESOPOROUS MOLECULAR SIEVE MCM-41 SYNTHESIS FROM FLUORIDE MEDIA
 Brazilian Journal of Chemical Engineering ISSN 14-6632 Printed in Brazil www.abeq.org.br/bjche Vol. 28, No. 4, pp. 649-658, October - December, 211 MESOPOROUS MOLECULAR SIEVE MCM-41 SYNTHESIS FROM FLUORIDE
Brazilian Journal of Chemical Engineering ISSN 14-6632 Printed in Brazil www.abeq.org.br/bjche Vol. 28, No. 4, pp. 649-658, October - December, 211 MESOPOROUS MOLECULAR SIEVE MCM-41 SYNTHESIS FROM FLUORIDE
Al-containing MCM-41 type materials prepared by different synthesis methods: Hydrothermal stability and catalytic properties
 Al-containing MCM-41 type materials prepared by different synthesis methods: Hydrothermal stability and catalytic properties J.P. Lourenço *1, A. Fernandes 2, C. Henriques 2, M.F. Ribeiro 2 1 Universidade
Al-containing MCM-41 type materials prepared by different synthesis methods: Hydrothermal stability and catalytic properties J.P. Lourenço *1, A. Fernandes 2, C. Henriques 2, M.F. Ribeiro 2 1 Universidade
Control of Pore Sizes in Mesoporous Silica Templated by Liquid Crystals in Block Copolymer-Cosurfactant-Water Systems
 5304 Langmuir 2000, 16, 5304-5310 Control of Pore Sizes in Mesoporous Silica Templated by Liquid Crystals in Block Copolymer-Cosurfactant-Water Systems Pingyun Feng,*, Xianhui Bu, and David J. Pine*, Departments
5304 Langmuir 2000, 16, 5304-5310 Control of Pore Sizes in Mesoporous Silica Templated by Liquid Crystals in Block Copolymer-Cosurfactant-Water Systems Pingyun Feng,*, Xianhui Bu, and David J. Pine*, Departments
Synthesis of mesoporous aluminosilicates with low Si/Al ratios using a single-source molecular precursor under acidic conditions
 J Porous Mater (2006) 13: 187 193 DOI 10.1007/s10934-006-8003-8 Synthesis of mesoporous aluminosilicates with low Si/Al ratios using a single-source molecular precursor under acidic conditions Ying Li
J Porous Mater (2006) 13: 187 193 DOI 10.1007/s10934-006-8003-8 Synthesis of mesoporous aluminosilicates with low Si/Al ratios using a single-source molecular precursor under acidic conditions Ying Li
Tailor-Made Nanostructured Ion Selective MCM-48 Membranes
 Mat. Res. Soc. Symp. Proc. Vol. 775 2003 Materials Research Society P1.4.1 Tailor-Made Nanostructured Ion Selective MCM-48 Membranes Sankhanilay Roy Chowdhury, Riaan Schmuhl, Klaas Keizer, Albert van den
Mat. Res. Soc. Symp. Proc. Vol. 775 2003 Materials Research Society P1.4.1 Tailor-Made Nanostructured Ion Selective MCM-48 Membranes Sankhanilay Roy Chowdhury, Riaan Schmuhl, Klaas Keizer, Albert van den
Supplementary Information
 Supplementary Information Fabrication of Novel Rattle-Type Magnetic Mesoporous carbon Microspheres for Removal of Microcystins Xinghua Zhang and Long Jiang* Beijing National Laboratory for Molecular Science
Supplementary Information Fabrication of Novel Rattle-Type Magnetic Mesoporous carbon Microspheres for Removal of Microcystins Xinghua Zhang and Long Jiang* Beijing National Laboratory for Molecular Science
Having a High Mg/Al Molar Ratio
 SUPPORTING INFORMATION High-Temperature CO 2 Sorption on Hydrotalcite Having a High Mg/Al Molar Ratio Suji Kim, Sang Goo Jeon, and Ki Bong Lee*, Department of Chemical and Biological Engineering, Korea
SUPPORTING INFORMATION High-Temperature CO 2 Sorption on Hydrotalcite Having a High Mg/Al Molar Ratio Suji Kim, Sang Goo Jeon, and Ki Bong Lee*, Department of Chemical and Biological Engineering, Korea
Supplementary information
 Supplementary information Supplementary Information for Exceptional Ammonia Uptake by a Covalent Organic Framework Christian J. Doonan, David J. Tranchemontagne,T. Grant Glover, Joseph R. Hunt, Omar M.
Supplementary information Supplementary Information for Exceptional Ammonia Uptake by a Covalent Organic Framework Christian J. Doonan, David J. Tranchemontagne,T. Grant Glover, Joseph R. Hunt, Omar M.
Supplementary Information
 Supplementary Information 1. Thermodynamic data The isomerization of glucose into fructose using can be represented as: The equilibrium constant K eq and equilibrium conversion were calculated as follows:
Supplementary Information 1. Thermodynamic data The isomerization of glucose into fructose using can be represented as: The equilibrium constant K eq and equilibrium conversion were calculated as follows:
Diels-Alder Cycloaddition of Cyclopentadiene with Ethylacrylate Catalyzed by Mesoporous Al-MCM-48 and Al-MCM-41 Catalysts
 Diels-Alder Reaction over Al-MCM-48 Catalyst Bull. Korean Chem. Soc. 2008, Vol. 29, No. 10 1993 Diels-Alder Cycloaddition of Cyclopentadiene with Ethylacrylate Catalyzed by Mesoporous Al-MCM-48 and Al-MCM-41
Diels-Alder Reaction over Al-MCM-48 Catalyst Bull. Korean Chem. Soc. 2008, Vol. 29, No. 10 1993 Diels-Alder Cycloaddition of Cyclopentadiene with Ethylacrylate Catalyzed by Mesoporous Al-MCM-48 and Al-MCM-41
Wei Zhou and Dehua He*
 Lengthen alkyl spacer to increase SBA-15-anchored Rh-P complex activities in 1-octene hydroformylation Wei Zhou and Dehua He* Innovative Catalysis Program, Key Lab of Organoelectronics & Molecular Engineering
Lengthen alkyl spacer to increase SBA-15-anchored Rh-P complex activities in 1-octene hydroformylation Wei Zhou and Dehua He* Innovative Catalysis Program, Key Lab of Organoelectronics & Molecular Engineering
Thermal Conductivity of Covalent Organic Frameworks as a Function of Their Pore Size
 Thermal Conductivity of Covalent Organic Frameworks as a Function of Their Pore Size Sunny K. S. Freitas a ; Raquel S. Borges b ; Claudia Merlini c, Guilherme M. O. Barra d, Pierre M. Esteves a, * a. Instituto
Thermal Conductivity of Covalent Organic Frameworks as a Function of Their Pore Size Sunny K. S. Freitas a ; Raquel S. Borges b ; Claudia Merlini c, Guilherme M. O. Barra d, Pierre M. Esteves a, * a. Instituto
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Controllable integration of ultrasmall noble metal nanoparticles
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Controllable integration of ultrasmall noble metal nanoparticles
Supporting Information. CdS/mesoporous ZnS core/shell particles for efficient and stable photocatalytic hydrogen evolution under visible light
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2014 Supporting Information CdS/mesoporous ZnS core/shell particles for efficient
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2014 Supporting Information CdS/mesoporous ZnS core/shell particles for efficient
Aviation Fuel Production from Lipids by a Single-Step Route using
 Aviation Fuel Production from Lipids by a Single-Step Route using Hierarchical Mesoporous Zeolites Deepak Verma, Rohit Kumar, Bharat S. Rana, Anil K. Sinha* CSIR-Indian Institute of Petroleum, Dehradun-2485,
Aviation Fuel Production from Lipids by a Single-Step Route using Hierarchical Mesoporous Zeolites Deepak Verma, Rohit Kumar, Bharat S. Rana, Anil K. Sinha* CSIR-Indian Institute of Petroleum, Dehradun-2485,
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Engineering Growth Defects: A New Route Towards Hierarchical
Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Engineering Growth Defects: A New Route Towards Hierarchical
Supplementary Information. for. Zeolites ZSM-25 and PST-20: Selective Carbon Dioxide Adsorbents at High. Pressures
 S1 Supplementary Information for Zeolites ZSM-25 and PST-20: Selective Carbon Dioxide Adsorbents at High Pressures Jung Gi Min, K. Christian Kemp, and Suk Bong Hong* Center for Ordered Nanoporous Materials
S1 Supplementary Information for Zeolites ZSM-25 and PST-20: Selective Carbon Dioxide Adsorbents at High Pressures Jung Gi Min, K. Christian Kemp, and Suk Bong Hong* Center for Ordered Nanoporous Materials
Shape Assisted Fabrication of Fluorescent Cages of Squarate based Metal-Organic Coordination Frameworks
 Supporting Information Shape Assisted Fabrication of Fluorescent Cages of Squarate based Metal-Organic Coordination Frameworks Kolleboyina Jayaramulu, a Katla Sai Krishna, a Subi J. George, b Muthuswamy
Supporting Information Shape Assisted Fabrication of Fluorescent Cages of Squarate based Metal-Organic Coordination Frameworks Kolleboyina Jayaramulu, a Katla Sai Krishna, a Subi J. George, b Muthuswamy
Supplementary Figure S1: Solution NMR spectra for D-glucose with a 4:1 sugar:sb molar ratio in H 2 O. a) 13 C NMR of D-(1-13 C)glucose, b) 13 C NMR
 a c b d Supplementary Figure S1: Solution NMR spectra for D-glucose with a 4:1 sugar:sb molar ratio in H 2 O. a) 13 C NMR of D-(1-13 C)glucose, b) 13 C NMR of D-glucose, c) 11 B NMR, d) 1 H NMR a c b d
a c b d Supplementary Figure S1: Solution NMR spectra for D-glucose with a 4:1 sugar:sb molar ratio in H 2 O. a) 13 C NMR of D-(1-13 C)glucose, b) 13 C NMR of D-glucose, c) 11 B NMR, d) 1 H NMR a c b d
Supporting Information
 upporting Information ynthesis of Thiol-functionalized Mesoporpous ilica Material (M) To synthesize the thiol-functionalized mesoporous silica (M), we modified the welldeveloped cetyltrimethylammonium
upporting Information ynthesis of Thiol-functionalized Mesoporpous ilica Material (M) To synthesize the thiol-functionalized mesoporous silica (M), we modified the welldeveloped cetyltrimethylammonium
Constructing covalent organic frameworks in water via dynamic covalent bonding
 Supporting information IUCrJ Volume 3 (2016) Supporting information for article: Constructing covalent organic frameworks in water via dynamic covalent bonding Jayshri Thote, Harshitha Barike Aiyappa,
Supporting information IUCrJ Volume 3 (2016) Supporting information for article: Constructing covalent organic frameworks in water via dynamic covalent bonding Jayshri Thote, Harshitha Barike Aiyappa,
Photodecomposition of Water Catalyzed by Zr- and Ti-MCM-41
 ESO 25(6) #7759 Energy Sources, 25:591 596, 2003 Copyright 2003 Taylor & Francis 0090-8312/03 $12.00 +.00 DOI: 10.1080/00908310390195651 Photodecomposition of Water Catalyzed by Zr- and Ti-MCM-41 S.-H.
ESO 25(6) #7759 Energy Sources, 25:591 596, 2003 Copyright 2003 Taylor & Francis 0090-8312/03 $12.00 +.00 DOI: 10.1080/00908310390195651 Photodecomposition of Water Catalyzed by Zr- and Ti-MCM-41 S.-H.
Facile synthesis of polymer and carbon spheres decorated with highly dispersed metal nanoparticles
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 1 Facile synthesis of polymer and carbon spheres decorated with highly dispersed metal nanoparticles
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 1 Facile synthesis of polymer and carbon spheres decorated with highly dispersed metal nanoparticles
Environment Protection Engineering. ADSORPTIVE PERFORMANCE OF MCM-41 TOWARDS Hg(II) IN WATER. ADSORPTION AND DESORPTION STUDIES
 Environment Protection Engineering Vol. 42 2016 No. 1 DOI: 10.5277/epe160101 QING ZHOU ZHAI 1, WEI-HUA HU 1, XIAO-YU GUO 1 ADSORPTIVE PERFORMANCE OF MCM-41 TOWARDS Hg(II) IN WATER. ADSORPTION AND DESORPTION
Environment Protection Engineering Vol. 42 2016 No. 1 DOI: 10.5277/epe160101 QING ZHOU ZHAI 1, WEI-HUA HU 1, XIAO-YU GUO 1 ADSORPTIVE PERFORMANCE OF MCM-41 TOWARDS Hg(II) IN WATER. ADSORPTION AND DESORPTION
Silica-Pillared H-kenyaites: Interlamellar Base Catalyzed-Reaction of Tetraethylorthosilicate in Water Suspension
 Silica-Pillared Molecular Sieves from Kenyaite Bull. Korean Chem. Soc. 1999, Vol. 20, No. 1 69 Silica-Pillared H-kenyaites: Interlamellar Base Catalyzed-Reaction of Tetraethylorthosilicate in Water Suspension
Silica-Pillared Molecular Sieves from Kenyaite Bull. Korean Chem. Soc. 1999, Vol. 20, No. 1 69 Silica-Pillared H-kenyaites: Interlamellar Base Catalyzed-Reaction of Tetraethylorthosilicate in Water Suspension
Supporting Information. SBA-15 Supported Fe, Ni, Fe-Ni Bimetallic Catalysts for Wet Oxidation of Bisphenol-A
 Wet Oxidation of Bisphenol A by Metal Supported SBA-15 Bull. Korean Chem. Soc. 2014, Vol. 35, No. 12 1 http://dx.doi.org/10.5012/bkcs.2014.35.12.xxx Supporting Information SBA-15 Supported Fe, Ni, Fe-Ni
Wet Oxidation of Bisphenol A by Metal Supported SBA-15 Bull. Korean Chem. Soc. 2014, Vol. 35, No. 12 1 http://dx.doi.org/10.5012/bkcs.2014.35.12.xxx Supporting Information SBA-15 Supported Fe, Ni, Fe-Ni
Synthesis of a Zeolite Column with a Monolithic Microhoneycomb Structure Using the Ice Template Method
 Synthesis of a Zeolite Column with a Monolithic Microhoneycomb Structure Using the Ice Template Method Shin R. Mukai, Shinya Murata, Kazufusa Onodera and Izumi Yamada *1 Graduate School of Engineering,
Synthesis of a Zeolite Column with a Monolithic Microhoneycomb Structure Using the Ice Template Method Shin R. Mukai, Shinya Murata, Kazufusa Onodera and Izumi Yamada *1 Graduate School of Engineering,
Mesostructured Zeolite Y - High Hydrothermal Stability and Superior FCC Catalytic Performance
 Supporting Information Mesostructured Zeolite Y - High Hydrothermal Stability and Superior FCC Catalytic Performance Javier García-Martínez,* Marvin Johnson, Julia Valla, Kunhao Li, and Jackie Y. Ying*
Supporting Information Mesostructured Zeolite Y - High Hydrothermal Stability and Superior FCC Catalytic Performance Javier García-Martínez,* Marvin Johnson, Julia Valla, Kunhao Li, and Jackie Y. Ying*
Sodium silicate as source of silica for synthesis of mesoporous SBA-15
 IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Sodium silicate as source of silica for synthesis of mesoporous SBA-15 To cite this article: Norhasyimi Rahmat et al 2016 IOP
IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Sodium silicate as source of silica for synthesis of mesoporous SBA-15 To cite this article: Norhasyimi Rahmat et al 2016 IOP
Supporting Information. Microwindows on the Shell
 Supporting Information Organic-inorganic Hybrid Hollow Nanospheres with Microwindows on the Shell Jian Liu,, Qihua Yang,*, Lei Zhang, Hengquan Yang, Jinsuo Gao,, and Can Li*, State Key Laboratory of Catalysis,
Supporting Information Organic-inorganic Hybrid Hollow Nanospheres with Microwindows on the Shell Jian Liu,, Qihua Yang,*, Lei Zhang, Hengquan Yang, Jinsuo Gao,, and Can Li*, State Key Laboratory of Catalysis,
Surface Modification of PTMS Particles with Organosilanes: TEOS-, VTMS-, and MTMS-Modified Particles
 Journal of Sol-Gel Science and Technology 31, 117 121, 2004 c 2004 Kluwer Academic Publishers. Manufactured in The United States. Surface Modification of PTMS Particles with Organosilanes: TEOS-, VTMS-,
Journal of Sol-Gel Science and Technology 31, 117 121, 2004 c 2004 Kluwer Academic Publishers. Manufactured in The United States. Surface Modification of PTMS Particles with Organosilanes: TEOS-, VTMS-,
Synthesis of ordered microporous carbons via template technique
 Synthesis of ordered microporous carbons via template technique Zhou Ying, Yao Qimei, Qiu Jieshan *, Guo Hongchen, Sun Zongwei Carbon Research Laboratory, Center for Nano Materials and Science, School
Synthesis of ordered microporous carbons via template technique Zhou Ying, Yao Qimei, Qiu Jieshan *, Guo Hongchen, Sun Zongwei Carbon Research Laboratory, Center for Nano Materials and Science, School
Chlorohydrination of Allyl Chloride with HCl and H 2 O 2 to Produce. Dichloropropanols Catalyzed by Hollow TS-1 Zeolite
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 216 Chlorohydrination of Allyl Chloride with and 2 O 2 to Produce Dichloropropanols Catalyzed
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 216 Chlorohydrination of Allyl Chloride with and 2 O 2 to Produce Dichloropropanols Catalyzed
Eco-Compatible Continuous Halogenation of Aromatics
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Eco-Compatible Continuous Halogenation of Aromatics P. Losch, a J. F. Kolb, a A. Astafan,
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Eco-Compatible Continuous Halogenation of Aromatics P. Losch, a J. F. Kolb, a A. Astafan,
Supporting information
 Supporting information Hierarchical Macro-meso-microporous ZSM-5 Zeolite Hollow Fibers With Highly Efficient Catalytic Cracking Capability Jia Liu, a Guiyuan Jiang,* a Ying Liu, a Jiancheng Di, b Yajun
Supporting information Hierarchical Macro-meso-microporous ZSM-5 Zeolite Hollow Fibers With Highly Efficient Catalytic Cracking Capability Jia Liu, a Guiyuan Jiang,* a Ying Liu, a Jiancheng Di, b Yajun
Fe(III)-salen encapsulated Al-MCM-41 as a catalyst in the polymerisation of bisphenol-a
 University Technology Malaysia From the SelectedWorks of Hadi Nur February, 2005 Fe(III)-salen encapsulated Al-MCM-41 as a catalyst in the polymerisation of bisphenol-a Halimaton Hamdan, University Technology
University Technology Malaysia From the SelectedWorks of Hadi Nur February, 2005 Fe(III)-salen encapsulated Al-MCM-41 as a catalyst in the polymerisation of bisphenol-a Halimaton Hamdan, University Technology
Utilization of Rice Husk Ash Silica in Controlled Releasing Application
 Journal of Metals, Materials and Minerals, Vol.9 No.2 pp.6-65, 29 Utilization of Rice Husk Ash Silica in Controlled Releasing Application Piyawan PRAWINGWONG, Chaiyan CHAIYA 2, Prasert REUBROYCHAROEN 3
Journal of Metals, Materials and Minerals, Vol.9 No.2 pp.6-65, 29 Utilization of Rice Husk Ash Silica in Controlled Releasing Application Piyawan PRAWINGWONG, Chaiyan CHAIYA 2, Prasert REUBROYCHAROEN 3
Supporting Information
 Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2018 Supporting Information 2-Methylimidazole-Assisted Synthesis of Two-Dimensional MOF-5 Catalyst
Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2018 Supporting Information 2-Methylimidazole-Assisted Synthesis of Two-Dimensional MOF-5 Catalyst
STUDY ON SORPTION OF SOME TOXIC AND HEAVY IONS IN DILUTE SOLUTIONS BY CLINOPTILOLITE
 J. Sci. I. R. Iran Vol. 12, No. 3, Summer 2001 STUDY ON SORPTION OF SOME TOXIC AND HEAVY IONS IN DILUTE SOLUTIONS BY CLINOPTILOLITE F. Farzaneh *, Z. Shivapour, N. Khosravi Talebi and J. Sayyad Mashhoor
J. Sci. I. R. Iran Vol. 12, No. 3, Summer 2001 STUDY ON SORPTION OF SOME TOXIC AND HEAVY IONS IN DILUTE SOLUTIONS BY CLINOPTILOLITE F. Farzaneh *, Z. Shivapour, N. Khosravi Talebi and J. Sayyad Mashhoor
Rapid, Efficient Phase Pure Synthesis of Ca 2 AlNO 3 Layered Double Hydroxide
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting Information Rapid, Efficient Phase Pure Synthesis of Ca 2 AlNO
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting Information Rapid, Efficient Phase Pure Synthesis of Ca 2 AlNO
Mesolamellar composite of TiN and CTAB using fluoride ion bridge: synthesis, mechanism & characterization
 Vol.2, No.5, 464-468 (2010) http://dx.doi.org/10.4236/ns.2010.25057 Natural Science Mesolamellar composite of TiN and CTAB using fluoride ion bridge: synthesis, mechanism & characterization Tumbavanam
Vol.2, No.5, 464-468 (2010) http://dx.doi.org/10.4236/ns.2010.25057 Natural Science Mesolamellar composite of TiN and CTAB using fluoride ion bridge: synthesis, mechanism & characterization Tumbavanam
EFFECT OF AGING TIME TOWARD CRYSTALLINITY OF PRODUCTS IN SYNTHESIS OF MESOPOROUS SILICATES MCM-41
 310 EFFECT OF AGING TIME TOWARD CRYSTALLINITY OF PRODUCTS IN SYNTHESIS OF MESOPOROUS SILICATES MCM-41 Suyanta 1,2,*, Narsito 2, Endang Tri Wahyuni 2, Triyono 2, and Sutarno 2 1 Postgraduate Student of
310 EFFECT OF AGING TIME TOWARD CRYSTALLINITY OF PRODUCTS IN SYNTHESIS OF MESOPOROUS SILICATES MCM-41 Suyanta 1,2,*, Narsito 2, Endang Tri Wahyuni 2, Triyono 2, and Sutarno 2 1 Postgraduate Student of
Utilization of Diatomite as Silica Source for the Synthesis of Ordered Mesoporous Silica Materials
 Utilization of Diatomite as Silica Source for the Synthesis of Ordered Mesoporous Silica Materials Jirapa Kraisit, Chulalongkorn University, Thailand Chutimon Satirapipathkul, Chulalongkorn University,
Utilization of Diatomite as Silica Source for the Synthesis of Ordered Mesoporous Silica Materials Jirapa Kraisit, Chulalongkorn University, Thailand Chutimon Satirapipathkul, Chulalongkorn University,
Room Temperature Hydrogen Generation from Hydrous Hydrazine for Chemical Hydrogen Storage
 (Supporting Information) Room Temperature Hydrogen Generation from Hydrous Hydrazine for Chemical Hydrogen Storage Sanjay Kumar Singh, Xin-Bo Zhang, and Qiang Xu* National Institute of Advanced Industrial
(Supporting Information) Room Temperature Hydrogen Generation from Hydrous Hydrazine for Chemical Hydrogen Storage Sanjay Kumar Singh, Xin-Bo Zhang, and Qiang Xu* National Institute of Advanced Industrial
Surface modification of as-synthesized lamellar mesostructured silica obtained by liquid crystal templating
 PAPER Surface modification of as-synthesized lamellar mesostructured silica obtained by liquid crystal templating C. Henrist,* ab C. Vogels, c A. Rulmont a and R. Cloots ab NJC www.rsc.org/njc a SUPRATECS,
PAPER Surface modification of as-synthesized lamellar mesostructured silica obtained by liquid crystal templating C. Henrist,* ab C. Vogels, c A. Rulmont a and R. Cloots ab NJC www.rsc.org/njc a SUPRATECS,
Microstructured Porous Silica Obtained via Colloidal Crystal Templates
 Paper No. 203e Microstructured Porous Silica Obtained via Colloidal Crystal Templates O. D. Velev, T. A. Jede, R. F. Lobo and A. M. Lenhoff Department of Chemical Engineering, University of Delaware, Newark
Paper No. 203e Microstructured Porous Silica Obtained via Colloidal Crystal Templates O. D. Velev, T. A. Jede, R. F. Lobo and A. M. Lenhoff Department of Chemical Engineering, University of Delaware, Newark
Supporting Information. for. Synthesis and Catalytic Behavior of Ferrierite Zeolite Nanoneedles
 Supporting Information for Synthesis and Catalytic Behavior of Ferrierite Zeolite Nanoneedles Yoorim Lee, Min Bum Park, Pyung Soon Kim, Aurélie Vicente, Christian Fernandez, In-Sik Nam, and Suk Bong Hong,*
Supporting Information for Synthesis and Catalytic Behavior of Ferrierite Zeolite Nanoneedles Yoorim Lee, Min Bum Park, Pyung Soon Kim, Aurélie Vicente, Christian Fernandez, In-Sik Nam, and Suk Bong Hong,*
PRODUCTION OF MCM-41 FROM RICE HUSK
 18 TH INTERNATIONAL CONFERENCE ON COMPOSITE MATERIALS PRODUCTION OF MCM-41 FROM RICE HUSK K.N. Hui 1, J.Y. Lee 1, W. Guo 1, K.S. Hui 2,* 1 Department of Materials Science and Engineering, Pusan National
18 TH INTERNATIONAL CONFERENCE ON COMPOSITE MATERIALS PRODUCTION OF MCM-41 FROM RICE HUSK K.N. Hui 1, J.Y. Lee 1, W. Guo 1, K.S. Hui 2,* 1 Department of Materials Science and Engineering, Pusan National
Study on Mesoporous Al-SBA-15 with Enhanced Acidity and Hydrothermal Stability for Heavy Oil Hydrocracking Conversion
 American Journal of Materials Synthesis and Processing 2018; 3(3): 39-46 http://www.sciencepublishinggroup.com/j/ajmsp doi: 10.11648/j.XXXX.2018XXXX.XX ISSN: 2575-2154 (Print); ISSN: 2575-1530 (Online)
American Journal of Materials Synthesis and Processing 2018; 3(3): 39-46 http://www.sciencepublishinggroup.com/j/ajmsp doi: 10.11648/j.XXXX.2018XXXX.XX ISSN: 2575-2154 (Print); ISSN: 2575-1530 (Online)
CONVERSION OF ETHANOL TO OLEFINS OVER HZSM-5 CATALYSTS
 CONVERSION OF ETHANOL TO OLEFINS OVER HZSM-5 CATALYSTS P. R. S. SALBEGO *1, G. S. N. M. CENTENARO 1, R. KLAIC 1, N. I. CANABARRO 1, T. K. MULLER 1, M. A. MAZUTTI 1, E. L. FOLETTO 1, S. L. JAHN 1 1 Universidade
CONVERSION OF ETHANOL TO OLEFINS OVER HZSM-5 CATALYSTS P. R. S. SALBEGO *1, G. S. N. M. CENTENARO 1, R. KLAIC 1, N. I. CANABARRO 1, T. K. MULLER 1, M. A. MAZUTTI 1, E. L. FOLETTO 1, S. L. JAHN 1 1 Universidade
Supporting Information
 Supporting Information Insights into CO 2 and CH 4 Adsorption by Pristine and Aromatic Amine- Modified Periodic Mesoporous Phenylene-Silicas Mirtha A. O. Lourenço, a Christophe Siquet, b,* João Santos,
Supporting Information Insights into CO 2 and CH 4 Adsorption by Pristine and Aromatic Amine- Modified Periodic Mesoporous Phenylene-Silicas Mirtha A. O. Lourenço, a Christophe Siquet, b,* João Santos,
Supporting Information
 Supporting Information Wu et al. 10.1073/pnas.1323989111 SI Materials and Methods Synthesis of Starting Materials. MCM-41 was synthesized hydrothermally according to the method reported by Gallis and Landry
Supporting Information Wu et al. 10.1073/pnas.1323989111 SI Materials and Methods Synthesis of Starting Materials. MCM-41 was synthesized hydrothermally according to the method reported by Gallis and Landry
Catalytic activity of the beta zeolite with enhanced textural properties in the Friedel-Crafts acylation of aromatic compounds
 Zeolites and Related Materials: Trends, Targets and Challenges Proceedings of 4 th International FEZA Conference A. Gedeon, P. Massiani and F. Babboneau (Editors) 28 Elsevier B.V. All rights reserved.
Zeolites and Related Materials: Trends, Targets and Challenges Proceedings of 4 th International FEZA Conference A. Gedeon, P. Massiani and F. Babboneau (Editors) 28 Elsevier B.V. All rights reserved.
Supporting Information
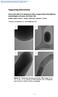 Supporting Information Interaction effects of mesoporous silica nanoparticles with different morphologies on human red blood cells Madhura Joglekar, Robert A. Roggers, Yannan Zhao, and Brian G. Trewyn
Supporting Information Interaction effects of mesoporous silica nanoparticles with different morphologies on human red blood cells Madhura Joglekar, Robert A. Roggers, Yannan Zhao, and Brian G. Trewyn
Adsorption effects in aluminophosphate molecular sieves studied by double-rotation NMR
 Catalysis Letters 15 (1992) 65-73 65 Adsorption effects in aluminophosphate molecular sieves studied by double-rotation NMR R. Jelinek, B.F. Chmelka 1, y. Wu 2, M.E. Davis 3, J.G. Ulan 4, R. Gronsky 5
Catalysis Letters 15 (1992) 65-73 65 Adsorption effects in aluminophosphate molecular sieves studied by double-rotation NMR R. Jelinek, B.F. Chmelka 1, y. Wu 2, M.E. Davis 3, J.G. Ulan 4, R. Gronsky 5
CHAPTER 4. SYNTHESIS, CHARACTERIZATION OF TiO 2 NANOTUBES AND THEIR APPLICATION IN DYE SENSITIZED SOLAR CELL
 93 CHAPTER 4 SYNTHESIS, CHARACTERIZATION OF TiO 2 NANOTUBES AND THEIR APPLICATION IN DYE SENSITIZED SOLAR CELL 4.1 INTRODUCTION TiO 2 -derived nanotubes are expected to be applicable for several applications,
93 CHAPTER 4 SYNTHESIS, CHARACTERIZATION OF TiO 2 NANOTUBES AND THEIR APPLICATION IN DYE SENSITIZED SOLAR CELL 4.1 INTRODUCTION TiO 2 -derived nanotubes are expected to be applicable for several applications,
Energy Procedia 00 (2011) SHC J. Jänchen a, H. Stach b, a*
 Energy Procedia 00 (2011) 000 000 Energy Procedia www.elsevier.com/locate/procedia SHC 2012 Adsorption properties of porous materials for solar thermal energy storage and heat pump applications J. Jänchen
Energy Procedia 00 (2011) 000 000 Energy Procedia www.elsevier.com/locate/procedia SHC 2012 Adsorption properties of porous materials for solar thermal energy storage and heat pump applications J. Jänchen
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 214 Supporting Information Lei Liu, ab Yijie Xia, b Jie Zhang* b a) China Center for Modernization
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 214 Supporting Information Lei Liu, ab Yijie Xia, b Jie Zhang* b a) China Center for Modernization
Very low temperature CO oxidation over colloidally deposited gold nanoparticles on Mg(OH) 2 and MgO
 Supporing Information Very low temperature CO oxidation over colloidally deposited gold nanoparticles on Mg(OH) 2 and MgO Chun-Jiang Jia, Yong Liu, Hans Bongard, Ferdi Schüth* Max-Planck-Institut für Kohlenforschung,
Supporing Information Very low temperature CO oxidation over colloidally deposited gold nanoparticles on Mg(OH) 2 and MgO Chun-Jiang Jia, Yong Liu, Hans Bongard, Ferdi Schüth* Max-Planck-Institut für Kohlenforschung,
Characterization of Hexagonal and Lamellar Mesoporous Silicas, Aluminosilicates and Gallosilicates by Small-Angle X-ray Scatteringt
 1065 J. Appl. Cryst. (1997). 30, 1065-1074 Characterization of Hexagonal and Lamellar Mesoporous Silicas, Aluminosilicates and Gallosilicates by Small-Angle X-ray Scatteringt Guy VAN DEN BOSSCHE, a* ROGER
1065 J. Appl. Cryst. (1997). 30, 1065-1074 Characterization of Hexagonal and Lamellar Mesoporous Silicas, Aluminosilicates and Gallosilicates by Small-Angle X-ray Scatteringt Guy VAN DEN BOSSCHE, a* ROGER
Synthesis of highly b-oriented zeolite MFI films by suppressing. twin crystal growth during the secondary growth
 Supplementary Information Synthesis of highly b-oriented zeolite MFI films by suppressing twin crystal growth during the secondary growth Xianming Li, a Yong Peng, a Zhengbao Wang,* a and Yushan Yan a,
Supplementary Information Synthesis of highly b-oriented zeolite MFI films by suppressing twin crystal growth during the secondary growth Xianming Li, a Yong Peng, a Zhengbao Wang,* a and Yushan Yan a,
