Laser Physics. Introduction to Laser Essentials Lecture 4. The Interaction of Electromagnetic Radiation with Matter 28/10/1431.
|
|
|
- Amy Hunter
- 6 years ago
- Views:
Transcription
1 Laser Physics Introduction to Laser Essentials Lecture The Interaction of Electromagnetic Radiation with Matter 2 1
2 Emission and Absorption of Radiation نعلم أن الذرة تكتسب طاقة وتفقدھا بصورة مستمرة وإن انتقال الطاقة إلى الذرة يتم بواسطة طريقتين ھما: Collisions with other atoms, and the transfer of kinetic energy as a result of the collision. This kinetic energy is transferred into internal energy of the atom. Absorption and emission of electromagnetic radiation وحيث أن عملية الليزر تعتمد على انتقال الطاقة من خالل امتصاص Absorption اإلشعاع الكھرومغناطيسي ثم تكبيره وانبعاثه emission على شكل شعاع ليزر لذا سندرس ظاھرة االمتصاص واالنبعاث. 3 Emission and Absorption of Radiation The interactions between electromagnetic radiation and matter cause changes in the energy states of the electrons in matter. Electrons can be transferred from one energy level to another, while absorbing or emitting a certain amount of energy. This amount of energy is equal to the energy difference between these two energy levels (E 2 -E 1 ). When this energy is absorbed or emitted in a form of electromagnetic radiation, the energy difference between these two energy levels (E 2 -E 1 ) determines uniquely the frequency (ν) of the electromagnetic radiation: 4 2
3 Example The visible spectrum wavelength range is: [μm] ( [nm]). The wavelength of the violet light is the shortest, andthe wavelength of the red light is the longest. Calculate: a) What is the frequency range of the visible spectrum. b) What is the amount of the photon s energy associated with the violet light, compared to the photon energy of the red light. 5 Solution: The frequency of violet light: The frequency of red light: The difference in frequencies: 6 3
4 The energy of a violet photon: The energy of a red photon: The difference in energies between the violet photon and the red photon is: 2.15*10-19 [J] This example shows how much more energy the violet photon have compared to the red photon. 7 Question Calculate in units of Nanometer, the wavelength of light emitted by the transition from energy level E3 to energy level E2 in which: E1 = 0 [ev] E2 = 1.1 [ev] E3 = 3.5 [ev] 8 4
5 Emission and Absorption of Radiation Every system in nature "prefers" to be in the lowest energy state. This state is called the Ground state. When energy is applied to a system, The atoms in the material are excited, andraised to a higher energy level. (The terms "excited atoms", "excited states", and "excited electrons" are used here with no distinction) These electrons will remain in the excited state for a certain period of time, and then will return to lower energy states while emitting energy in the exact amount of the difference between the energy levels ( E). If this energy is transmitted as electromagnetic energy, it is called photon. 9 Spontaneous Emission The emission of the individual photon is random, being done individually by each excited atom, with no relation to photons emitted by other atoms. When photons are randomly emitted from different atoms at different times, the process is called Spontaneous Emission. Since this emission is independent of external influence, there is no preferred direction for different photons, and there is no phase relation between photons emitted by different atoms. Spontaneous emission is one of a family of processes, called relaxation processes, by which the excited atoms return to equilibrium (ground state). This "classic" explanation assumes that the specific frequencies emitted by an excited atom are the same as the characteristic frequencies of the atom, which means that the emission spectrum is identical to the absorption spectrum. 10 5
6 Three possible processes between photons and atoms Stimulated Absorption Spontaneous Emission Stimulated Emission 11 Photon Absorption Photon Absorption: A photon with frequency 12 hits an atom at rest (left), and excites it to higher energy level (E 2 ) while the photon is absorbed. 12 6
7 Spontaneous Emission Spontaneous emission of a photon: An atom in an excited state (left) emits a photon with frequency 12 and goes to a lower energy level (E 1 ). 13 Stimulated Emission Stimulated emission of a photon: A photon with frequency 12 hit an excited atom (left), and cause emission of two photons with frequency 12 while the atom goes to a lower energy level (E 1 ). 14 7
8 Stimulated Absorption We saw that the process of photon absorption by the atom is a process of raising the atom (electron) from a lower energy level into a higher energy level (excited state), by an amount of energy which is equivalent to the energy of the absorbed photon. Stimulated Emission The incoming photon is an electromagnetic field which is oscillating in time and space. This field forces the excited atom to oscillate with the same frequency and phase as the applied force, which means that the atom can not oscillate freely, but is forced to oscillate coherently with the incoming photon 15 Remember that two photons with the same wavelength (frequency) have the same energy: E = h = hc/ The incoming photon does not change at all as a result of the stimulated emission process. As a result of the stimulated emission process, we have two identical photons created from one photon and one excited state. Thus we have amplification in the sense that the number of photons has increased. 16 8
9 Average Lifetime Atoms stay in an excited level only for a short time (about 10-8 [sec]), and then they return to a lower energy level by spontaneous emission. Every energy level has a characteristic average lifetime, which is the average time the electron exists in the excited state before making a spontaneous transition. Thus, this is the time in which the excited atoms returned to a lower energy level. According to the quantum theory, the transition from one energy level to another is described by statistical probability. The probability of transition from higher energy level to a lower one is inversely proportional to the lifetime of the higher energy level. 17 When the transition probability is low for a specific transition, the lifetime of this energy level is longer (about 10-3 sec), and this level becomes a "meta-stable" level. In this meta-stable level a large population of atoms can assembled. As we shall see, this level can be a candidate for lasing process. When the population number of a higher energy level is bigger than the population number of a lower energy level, a condition of "population inversion" isestablished. If a population inversion exists between two energy levels, the probability is high that an incoming photon will stimulate an excited atom to return to a lower state, while emitting another photon of light. The probability for this process depend on the match between the energy of the incoming photon and the energy difference between these two levels 18 9
PHYS 172: Modern Mechanics Fall 2009
 PHYS 172: Modern Mechanics Fall 2009 Lecture 14 Energy Quantization Read 7.1 7.9 Reading Question: Ch. 7, Secs 1-5 A simple model for the hydrogen atom treats the electron as a particle in circular orbit
PHYS 172: Modern Mechanics Fall 2009 Lecture 14 Energy Quantization Read 7.1 7.9 Reading Question: Ch. 7, Secs 1-5 A simple model for the hydrogen atom treats the electron as a particle in circular orbit
Higher -o-o-o- Past Paper questions o-o-o- 3.4 Spectra
 Higher -o-o-o- Past Paper questions 1991-2010 -o-o-o- 3.4 Spectra 1992 Q37 The diagram below shows the energy levels for the hydrogen atom. (a) Between which two energy levels would an electron transition
Higher -o-o-o- Past Paper questions 1991-2010 -o-o-o- 3.4 Spectra 1992 Q37 The diagram below shows the energy levels for the hydrogen atom. (a) Between which two energy levels would an electron transition
Laserphysik. Prof. Yong Lei & Dr. Yang Xu. Fachgebiet Angewandte Nanophysik, Institut für Physik
 Laserphysik Prof. Yong Lei & Dr. Yang Xu Fachgebiet Angewandte Nanophysik, Institut für Physik Contact: yong.lei@tu-ilmenau.de; yang.xu@tu-ilmenau.de Office: Heisenbergbau V 202, Unterpörlitzer Straße
Laserphysik Prof. Yong Lei & Dr. Yang Xu Fachgebiet Angewandte Nanophysik, Institut für Physik Contact: yong.lei@tu-ilmenau.de; yang.xu@tu-ilmenau.de Office: Heisenbergbau V 202, Unterpörlitzer Straße
LASER. Light Amplification by Stimulated Emission of Radiation
 LASER Light Amplification by Stimulated Emission of Radiation Laser Fundamentals The light emitted from a laser is monochromatic, that is, it is of one color/wavelength. In contrast, ordinary white light
LASER Light Amplification by Stimulated Emission of Radiation Laser Fundamentals The light emitted from a laser is monochromatic, that is, it is of one color/wavelength. In contrast, ordinary white light
Chapter 28 Assignment Solutions
 Chapter 28 Assignment Solutions Page 770 #23-26, 29-30, 43-48, 55 23) Complete the following concept map using these terms: energy levels, fixed electron radii, Bohr model, photon emission and absorption,
Chapter 28 Assignment Solutions Page 770 #23-26, 29-30, 43-48, 55 23) Complete the following concept map using these terms: energy levels, fixed electron radii, Bohr model, photon emission and absorption,
Introduction to Electromagnetic Radiation and Radiative Transfer
 Introduction to Electromagnetic Radiation and Radiative Transfer Temperature Dice Results Visible light, infrared (IR), ultraviolet (UV), X-rays, γ-rays, microwaves, and radio are all forms of electromagnetic
Introduction to Electromagnetic Radiation and Radiative Transfer Temperature Dice Results Visible light, infrared (IR), ultraviolet (UV), X-rays, γ-rays, microwaves, and radio are all forms of electromagnetic
PH300 Spring Homework 06
 PH300 Spring 2011 Homework 06 Total Points: 30 1. (1 Point) Each week you should review both your answers and the solutions for the previous week's homework to make sure that you understand all the questions
PH300 Spring 2011 Homework 06 Total Points: 30 1. (1 Point) Each week you should review both your answers and the solutions for the previous week's homework to make sure that you understand all the questions
1) Introduction 2) Photo electric effect 3) Dual nature of matter 4) Bohr s atom model 5) LASERS
 1) Introduction 2) Photo electric effect 3) Dual nature of matter 4) Bohr s atom model 5) LASERS 1. Introduction Types of electron emission, Dunnington s method, different types of spectra, Fraunhoffer
1) Introduction 2) Photo electric effect 3) Dual nature of matter 4) Bohr s atom model 5) LASERS 1. Introduction Types of electron emission, Dunnington s method, different types of spectra, Fraunhoffer
What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light
 What are Lasers? What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light emitted in a directed beam Light is coherenent
What are Lasers? What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light emitted in a directed beam Light is coherenent
The Sine Wave. You commonly see waves in the environment. Light Sound Electricity Ocean waves
 The Sine Wave Mathematically, a function that represents a smooth oscillation For example, if we drew the motion of how the weight bobs on the spring to the weight we would draw out a sine wave. The Sine
The Sine Wave Mathematically, a function that represents a smooth oscillation For example, if we drew the motion of how the weight bobs on the spring to the weight we would draw out a sine wave. The Sine
External (differential) quantum efficiency Number of additional photons emitted / number of additional electrons injected
 Semiconductor Lasers Comparison with LEDs The light emitted by a laser is generally more directional, more intense and has a narrower frequency distribution than light from an LED. The external efficiency
Semiconductor Lasers Comparison with LEDs The light emitted by a laser is generally more directional, more intense and has a narrower frequency distribution than light from an LED. The external efficiency
22. Lasers. Stimulated Emission: Gain. Population Inversion. Rate equation analysis. Two-level, three-level, and four-level systems
 . Lasers Stimulated Emission: Gain Population Inversion Rate equation analysis Two-level, three-level, and four-level systems What is a laser? LASER: Light Amplification by Stimulated Emission of Radiation
. Lasers Stimulated Emission: Gain Population Inversion Rate equation analysis Two-level, three-level, and four-level systems What is a laser? LASER: Light Amplification by Stimulated Emission of Radiation
The ELECTRON: Wave Particle Duality. chapter 4
 The ELECTRON: Wave Particle Duality chapter 4 What do we know about light? Before 1900 s scientists thought light behaved as a wave. This belief changed when it was discovered that light also has particle
The ELECTRON: Wave Particle Duality chapter 4 What do we know about light? Before 1900 s scientists thought light behaved as a wave. This belief changed when it was discovered that light also has particle
General Chemistry by Ebbing and Gammon, 8th Edition
 Chem 1045 General Chemistry by Ebbing and Gammon, 8th Edition George W.J. Kenney, Jr Last Update: 26-Mar-2009 Chapter 7: Quantum Theory of the Atom These Notes are to SUPPLIMENT the Text, They do NOT Replace
Chem 1045 General Chemistry by Ebbing and Gammon, 8th Edition George W.J. Kenney, Jr Last Update: 26-Mar-2009 Chapter 7: Quantum Theory of the Atom These Notes are to SUPPLIMENT the Text, They do NOT Replace
Atomic Spectroscopy and Energy Levels
 Activity 18 Atomic Spectroscopy and Energy Levels Why? The emission of light by the hydrogen atom and other atoms played a key role in helping scientists to understand the electronic structure of atoms.
Activity 18 Atomic Spectroscopy and Energy Levels Why? The emission of light by the hydrogen atom and other atoms played a key role in helping scientists to understand the electronic structure of atoms.
What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light
 What are Lasers? What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light emitted in a directed beam Light is coherenent
What are Lasers? What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light emitted in a directed beam Light is coherenent
Chapter 13. Phys 322 Lecture 34. Modern optics
 Chapter 13 Phys 3 Lecture 34 Modern optics Blackbodies and Lasers* Blackbodies Stimulated Emission Gain and Inversion The Laser Four-level System Threshold Some lasers Pump Fast decay Laser Fast decay
Chapter 13 Phys 3 Lecture 34 Modern optics Blackbodies and Lasers* Blackbodies Stimulated Emission Gain and Inversion The Laser Four-level System Threshold Some lasers Pump Fast decay Laser Fast decay
What Makes a Laser Light Amplification by Stimulated Emission of Radiation Main Requirements of the Laser Laser Gain Medium (provides the light
 What Makes a Laser Light Amplification by Stimulated Emission of Radiation Main Requirements of the Laser Laser Gain Medium (provides the light amplification) Optical Resonator Cavity (greatly increase
What Makes a Laser Light Amplification by Stimulated Emission of Radiation Main Requirements of the Laser Laser Gain Medium (provides the light amplification) Optical Resonator Cavity (greatly increase
FIBER OPTICS. Prof. R.K. Shevgaonkar. Department of Electrical Engineering. Indian Institute of Technology, Bombay. Lecture: 15. Optical Sources-LASER
 FIBER OPTICS Prof. R.K. Shevgaonkar Department of Electrical Engineering Indian Institute of Technology, Bombay Lecture: 15 Optical Sources-LASER Fiber Optics, Prof. R.K. Shevgaonkar, Dept. of Electrical
FIBER OPTICS Prof. R.K. Shevgaonkar Department of Electrical Engineering Indian Institute of Technology, Bombay Lecture: 15 Optical Sources-LASER Fiber Optics, Prof. R.K. Shevgaonkar, Dept. of Electrical
A fluorescent tube is filled with mercury vapour at low pressure. After mercury atoms have been excited they emit photons.
 Q1.(a) A fluorescent tube is filled with mercury vapour at low pressure. After mercury atoms have been excited they emit photons. In which part of the electromagnetic spectrum are these photons? What is
Q1.(a) A fluorescent tube is filled with mercury vapour at low pressure. After mercury atoms have been excited they emit photons. In which part of the electromagnetic spectrum are these photons? What is
I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited
 NCCS 1.1.2 & 1.1.3 I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited state I will describe how an electron
NCCS 1.1.2 & 1.1.3 I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited state I will describe how an electron
Write the electron configuration for Chromium (Cr):
 Write the electron configuration for Chromium (Cr): Energy level Aufbau Principle Atomic orbital Quantum Hund s Rule Atomic number Electron Configuration Whole number Pauli Exlcusion Principle Quantum
Write the electron configuration for Chromium (Cr): Energy level Aufbau Principle Atomic orbital Quantum Hund s Rule Atomic number Electron Configuration Whole number Pauli Exlcusion Principle Quantum
The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of hydrogen atom.
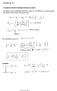 Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Chapter 39. Particles Behaving as Waves
 Chapter 39 Particles Behaving as Waves 39.1 Electron Waves Light has a dual nature. Light exhibits both wave and particle characteristics. Louis de Broglie postulated in 1924 that if nature is symmetric,
Chapter 39 Particles Behaving as Waves 39.1 Electron Waves Light has a dual nature. Light exhibits both wave and particle characteristics. Louis de Broglie postulated in 1924 that if nature is symmetric,
Waves & Oscillations
 Physics 42200 Waves & Oscillations Lecture 32 Electromagnetic Waves Spring 2016 Semester Matthew Jones Electromagnetism Geometric optics overlooks the wave nature of light. Light inconsistent with longitudinal
Physics 42200 Waves & Oscillations Lecture 32 Electromagnetic Waves Spring 2016 Semester Matthew Jones Electromagnetism Geometric optics overlooks the wave nature of light. Light inconsistent with longitudinal
NOTES: 5.3 Light and Atomic Spectra (more Quantum Mechanics!)
 NOTES: 5.3 Light and Atomic Spectra (more Quantum Mechanics!) Light WAVE or PARTICLE? Electromagnetic Radiation Electromagnetic radiation includes: -radio waves -microwaves -infrared waves -visible light
NOTES: 5.3 Light and Atomic Spectra (more Quantum Mechanics!) Light WAVE or PARTICLE? Electromagnetic Radiation Electromagnetic radiation includes: -radio waves -microwaves -infrared waves -visible light
Advanced Optical Communications Prof. R. K. Shevgaonkar Department of Electrical Engineering Indian Institute of Technology, Bombay
 Advanced Optical Communications Prof. R. K. Shevgaonkar Department of Electrical Engineering Indian Institute of Technology, Bombay Lecture No. # 15 Laser - I In the last lecture, we discussed various
Advanced Optical Communications Prof. R. K. Shevgaonkar Department of Electrical Engineering Indian Institute of Technology, Bombay Lecture No. # 15 Laser - I In the last lecture, we discussed various
Chemistry Instrumental Analysis Lecture 5. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 5 Light Amplification by Stimulated Emission of Radiation High Intensities Narrow Bandwidths Coherent Outputs Applications CD/DVD Readers Fiber Optics Spectroscopy
Chemistry 4631 Instrumental Analysis Lecture 5 Light Amplification by Stimulated Emission of Radiation High Intensities Narrow Bandwidths Coherent Outputs Applications CD/DVD Readers Fiber Optics Spectroscopy
Chapters 31 Atomic Physics
 Chapters 31 Atomic Physics 1 Overview of Chapter 31 Early Models of the Atom The Spectrum of Atomic Hydrogen Bohr s Model of the Hydrogen Atom de Broglie Waves and the Bohr Model The Quantum Mechanical
Chapters 31 Atomic Physics 1 Overview of Chapter 31 Early Models of the Atom The Spectrum of Atomic Hydrogen Bohr s Model of the Hydrogen Atom de Broglie Waves and the Bohr Model The Quantum Mechanical
Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE Brooks/Cole - Thomson
 Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE 1 7.1 The Nature of Light 2 Most subatomic particles behave as PARTICLES and obey the physics of waves. Light is a type of electromagnetic radiation Light consists
Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE 1 7.1 The Nature of Light 2 Most subatomic particles behave as PARTICLES and obey the physics of waves. Light is a type of electromagnetic radiation Light consists
Laser Types Two main types depending on time operation Continuous Wave (CW) Pulsed operation Pulsed is easier, CW more useful
 What Makes a Laser Light Amplification by Stimulated Emission of Radiation Main Requirements of the Laser Laser Gain Medium (provides the light amplification) Optical Resonator Cavity (greatly increase
What Makes a Laser Light Amplification by Stimulated Emission of Radiation Main Requirements of the Laser Laser Gain Medium (provides the light amplification) Optical Resonator Cavity (greatly increase
Waves & Oscillations
 Physics 42200 Waves & Oscillations Lecture 25 Propagation of Light Spring 2013 Semester Matthew Jones Midterm Exam: Date: Wednesday, March 6 th Time: 8:00 10:00 pm Room: PHYS 203 Material: French, chapters
Physics 42200 Waves & Oscillations Lecture 25 Propagation of Light Spring 2013 Semester Matthew Jones Midterm Exam: Date: Wednesday, March 6 th Time: 8:00 10:00 pm Room: PHYS 203 Material: French, chapters
Quick Review. 1. Kinetic Molecular Theory. 2. Average kinetic energy and average velocity. 3. Graham s Law of Effusion. 4. Real Gas Behavior.
 Quick Review 1. Kinetic Molecular Theory. 2. Average kinetic energy and average velocity. 3. Graham s Law of Effusion. 4. Real Gas Behavior. Emission spectra Every element has a unique emission spectrum
Quick Review 1. Kinetic Molecular Theory. 2. Average kinetic energy and average velocity. 3. Graham s Law of Effusion. 4. Real Gas Behavior. Emission spectra Every element has a unique emission spectrum
Student Exploration: Bohr Model of Hydrogen
 Name: Date: Student Exploration: Bohr Model of Hydrogen Vocabulary: absorption spectrum, Bohr model, electron volt, emission spectrum, energy level, ionization energy, laser, orbital, photon [Note to teachers
Name: Date: Student Exploration: Bohr Model of Hydrogen Vocabulary: absorption spectrum, Bohr model, electron volt, emission spectrum, energy level, ionization energy, laser, orbital, photon [Note to teachers
LASER. Light Amplification by Stimulated Emission of Radiation
 LASER Light Amplification by Stimulated Emission of Radiation Energy Level, Definitions The valence band is the highest filled band The conduction band is the next higher empty band The energy gap has
LASER Light Amplification by Stimulated Emission of Radiation Energy Level, Definitions The valence band is the highest filled band The conduction band is the next higher empty band The energy gap has
LASER. Challenging MCQ questions by The Physics Cafe. Compiled and selected by The Physics Cafe
 LSER hallenging MQ questions by The Physics afe ompiled and selected by The Physics afe www.thephysicsafe.com www.pmc.sg 1 laser point creates a spot on a screen as it reflects 70% of the light striking
LSER hallenging MQ questions by The Physics afe ompiled and selected by The Physics afe www.thephysicsafe.com www.pmc.sg 1 laser point creates a spot on a screen as it reflects 70% of the light striking
A. 0.7 ev B. 1.1 ev C. 1.4 ev D. 1.8 ev E. 2.1 ev F. 3.2 ev G. Something else
 A molecule has the energy levels shown in the diagram at the right. We begin with a large number of these molecules in their ground states. We want to raise a lot of these molecules to the state labeled
A molecule has the energy levels shown in the diagram at the right. We begin with a large number of these molecules in their ground states. We want to raise a lot of these molecules to the state labeled
We have already discussed what color is.
 The Atom The Electrons in the Atom Reading Assignment: Read the entire chapter. Homework: see the web site for homework. http://web.fccj.org/~smilczan/psc/homework7_11.htm Electrons are the glue that hold
The Atom The Electrons in the Atom Reading Assignment: Read the entire chapter. Homework: see the web site for homework. http://web.fccj.org/~smilczan/psc/homework7_11.htm Electrons are the glue that hold
MODERN OPTICS. P47 Optics: Unit 9
 MODERN OPTICS P47 Optics: Unit 9 Course Outline Unit 1: Electromagnetic Waves Unit 2: Interaction with Matter Unit 3: Geometric Optics Unit 4: Superposition of Waves Unit 5: Polarization Unit 6: Interference
MODERN OPTICS P47 Optics: Unit 9 Course Outline Unit 1: Electromagnetic Waves Unit 2: Interaction with Matter Unit 3: Geometric Optics Unit 4: Superposition of Waves Unit 5: Polarization Unit 6: Interference
Molecular spectroscopy
 Molecular spectroscopy Origin of spectral lines = absorption, emission and scattering of a photon when the energy of a molecule changes: rad( ) M M * rad( ' ) ' v' 0 0 absorption( ) emission ( ) scattering
Molecular spectroscopy Origin of spectral lines = absorption, emission and scattering of a photon when the energy of a molecule changes: rad( ) M M * rad( ' ) ' v' 0 0 absorption( ) emission ( ) scattering
Unit-2 LASER. Syllabus: Properties of lasers, types of lasers, derivation of Einstein A & B Coefficients, Working He-Ne and Ruby lasers.
 Unit-2 LASER Syllabus: Properties of lasers, types of lasers, derivation of Einstein A & B Coefficients, Working He-Ne and Ruby lasers. Page 1 LASER: The word LASER is acronym for light amplification by
Unit-2 LASER Syllabus: Properties of lasers, types of lasers, derivation of Einstein A & B Coefficients, Working He-Ne and Ruby lasers. Page 1 LASER: The word LASER is acronym for light amplification by
Complete nomenclature for electron orbitals
 Complete nomenclature for electron orbitals Bohr s model worked but it lacked a satisfactory reason why. De Broglie suggested that all particles have a wave nature. u l=h/p Enter de Broglie again It was
Complete nomenclature for electron orbitals Bohr s model worked but it lacked a satisfactory reason why. De Broglie suggested that all particles have a wave nature. u l=h/p Enter de Broglie again It was
A system of two lenses is achromatic when the separation between them is
 L e c t u r e 1 5 1 Eyepieces Single eye lens in a telescope / microscope produces spherical and chromatic aberrations. The field of view is also narrow. The eye lens is replaced by a system of lenses
L e c t u r e 1 5 1 Eyepieces Single eye lens in a telescope / microscope produces spherical and chromatic aberrations. The field of view is also narrow. The eye lens is replaced by a system of lenses
Theory of optically thin emission line spectroscopy
 Theory of optically thin emission line spectroscopy 1 Important definitions In general the spectrum of a source consists of a continuum and several line components. Processes which give raise to the continuous
Theory of optically thin emission line spectroscopy 1 Important definitions In general the spectrum of a source consists of a continuum and several line components. Processes which give raise to the continuous
Particle nature of light & Quantization
 Particle nature of light & Quantization A quantity is quantized if its possible values are limited to a discrete set. An example from classical physics is the allowed frequencies of standing waves on a
Particle nature of light & Quantization A quantity is quantized if its possible values are limited to a discrete set. An example from classical physics is the allowed frequencies of standing waves on a
Introduction to Quantum Mechanics Prof. Manoj Kumar Harbola Department of Physics Indian Institute of Technology, Kanpur
 Introduction to Quantum Mechanics Prof. Manoj Kumar Harbola Department of Physics Indian Institute of Technology, Kanpur Lecture - 06 Stimulated emission and amplification of light in a LASER (Refer Slide
Introduction to Quantum Mechanics Prof. Manoj Kumar Harbola Department of Physics Indian Institute of Technology, Kanpur Lecture - 06 Stimulated emission and amplification of light in a LASER (Refer Slide
Einstein. Quantum Physics at a glance. Planck s Hypothesis (blackbody radiation) (ultraviolet catastrophe) Quantized Energy
 Quantum Physics at a glance Quantum Physics deals with the study of light and particles at atomic and smaller levels. Planck s Hypothesis (blackbody radiation) (ultraviolet catastrophe) Quantized Energy
Quantum Physics at a glance Quantum Physics deals with the study of light and particles at atomic and smaller levels. Planck s Hypothesis (blackbody radiation) (ultraviolet catastrophe) Quantized Energy
General Considerations 1
 General Considerations 1 Absorption or emission of electromagnetic radiation results in a permanent energy transfer from the emitting object or to the absorbing medium. This permanent energy transfer can
General Considerations 1 Absorption or emission of electromagnetic radiation results in a permanent energy transfer from the emitting object or to the absorbing medium. This permanent energy transfer can
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy
 Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
Lecture 3: Light absorbance
 Lecture 3: Light absorbance Perturbation Response 1 Light in Chemistry Light Response 0-3 Absorbance spectrum of benzene 2 Absorption Visible Light in Chemistry S 2 S 1 Fluorescence http://www.microscopyu.com
Lecture 3: Light absorbance Perturbation Response 1 Light in Chemistry Light Response 0-3 Absorbance spectrum of benzene 2 Absorption Visible Light in Chemistry S 2 S 1 Fluorescence http://www.microscopyu.com
Laser Physics. Introduction to Laser Essentials Lecture 3. LASER Properties 28/10/1431. Physics Academy
 Laser Physics Introduction to Laser Essentials Lecture 3 www.hazemsakeek.com www.physicsacademy.org LASER Properties LASER Light Amplification Stimulated Emission Radiation 2 1 Properties of LASER شعاع
Laser Physics Introduction to Laser Essentials Lecture 3 www.hazemsakeek.com www.physicsacademy.org LASER Properties LASER Light Amplification Stimulated Emission Radiation 2 1 Properties of LASER شعاع
Phys 2310 Fri. Dec. 12, 2014 Today s Topics. Begin Chapter 13: Lasers Reading for Next Time
 Phys 2310 Fri. Dec. 12, 2014 Today s Topics Begin Chapter 13: Lasers Reading for Next Time 1 Reading this Week By Fri.: Ch. 13 (13.1, 13.3) Lasers, Holography 2 Homework this Week No Homework this chapter.
Phys 2310 Fri. Dec. 12, 2014 Today s Topics Begin Chapter 13: Lasers Reading for Next Time 1 Reading this Week By Fri.: Ch. 13 (13.1, 13.3) Lasers, Holography 2 Homework this Week No Homework this chapter.
Atomic Physics Worksheet. between 4000 and 5000 Angstroms ( nanometers): between 6500 and 7500 Angstroms ( nanometers):
 Atomic Physics Worksheet 1. Which of the gas samples shows an emission line with a wavelength between 4000 and 5000 Angstroms (400-500 nanometers): between 6500 and 7500 Angstroms (650-750 nanometers):
Atomic Physics Worksheet 1. Which of the gas samples shows an emission line with a wavelength between 4000 and 5000 Angstroms (400-500 nanometers): between 6500 and 7500 Angstroms (650-750 nanometers):
LASERS. Dr D. Arun Kumar Assistant Professor Department of Physical Sciences Bannari Amman Institute of Technology Sathyamangalam
 LASERS Dr D. Arun Kumar Assistant Professor Department of Physical Sciences Bannari Amman Institute of Technology Sathyamangalam General Objective To understand the principle, characteristics and types
LASERS Dr D. Arun Kumar Assistant Professor Department of Physical Sciences Bannari Amman Institute of Technology Sathyamangalam General Objective To understand the principle, characteristics and types
Lecture 15: Time-Dependent QM & Tunneling Review and Examples, Ammonia Maser
 ecture 15: Time-Dependent QM & Tunneling Review and Examples, Ammonia Maser ψ(x,t=0) 2 U(x) 0 x ψ(x,t 0 ) 2 x U 0 0 E x 0 x ecture 15, p.1 Special (Optional) ecture Quantum Information One of the most
ecture 15: Time-Dependent QM & Tunneling Review and Examples, Ammonia Maser ψ(x,t=0) 2 U(x) 0 x ψ(x,t 0 ) 2 x U 0 0 E x 0 x ecture 15, p.1 Special (Optional) ecture Quantum Information One of the most
Chem 321 Lecture 18 - Spectrophotometry 10/31/13
 Student Learning Objectives Chem 321 Lecture 18 - Spectrophotometry 10/31/13 In the lab you will use spectrophotometric techniques to determine the amount of iron, calcium and magnesium in unknowns. Although
Student Learning Objectives Chem 321 Lecture 18 - Spectrophotometry 10/31/13 In the lab you will use spectrophotometric techniques to determine the amount of iron, calcium and magnesium in unknowns. Although
PhET Light Emission and Lasers (27 points available x 2/3 = 18 points max score)
 IB PHYSICS Name: Period: Date: DEVIL PHYSICS BADDEST CLASS ON CAMPUS GETTING STARTED: PhET Light Emission and Lasers (27 points available x 2/3 = 18 points max score) Go to https://phet.colorado.edu/ Click
IB PHYSICS Name: Period: Date: DEVIL PHYSICS BADDEST CLASS ON CAMPUS GETTING STARTED: PhET Light Emission and Lasers (27 points available x 2/3 = 18 points max score) Go to https://phet.colorado.edu/ Click
Chapter 9: Quantization of Light
 Chapter 9: Quantization of Light Max Planck started the revolution of quantum theory by challenging the classical physics and the classical wave theory of light. He proposed the concept of quantization
Chapter 9: Quantization of Light Max Planck started the revolution of quantum theory by challenging the classical physics and the classical wave theory of light. He proposed the concept of quantization
Phys 322 Lecture 34. Chapter 13. Modern optics. Note: 10 points will be given for attendance today and for the rest of the semester.
 Chapter 13 Phys 322 Lecture 34 Modern optics Note: 10 points will be given for attendance today and for the rest of the semester. Presentation schedule Name Topic Date Alip, Abylaikhan lasers Nov. 30th
Chapter 13 Phys 322 Lecture 34 Modern optics Note: 10 points will be given for attendance today and for the rest of the semester. Presentation schedule Name Topic Date Alip, Abylaikhan lasers Nov. 30th
Information in Radio Waves
 Summative Assessment: Natural Sources of Radio Performance expectation: Develop and use a model of two objects interacting through electric or magnetic fields to illustrate the forces between objects and
Summative Assessment: Natural Sources of Radio Performance expectation: Develop and use a model of two objects interacting through electric or magnetic fields to illustrate the forces between objects and
The Structure of the Atom
 CHAPTER 5 The Structure of the Atom 5.4 Light and Spectroscopy 460 370 BC 1808 1870 1897 1910 1925 Today Democritus Atomism Dalton Modern atomic theory Crookes Cathode rays Thomson Discovery of the electron
CHAPTER 5 The Structure of the Atom 5.4 Light and Spectroscopy 460 370 BC 1808 1870 1897 1910 1925 Today Democritus Atomism Dalton Modern atomic theory Crookes Cathode rays Thomson Discovery of the electron
Light Emission.
 Light Emission www.physics.sfasu.edu/friedfeld/ch29lec.ppt Radio waves are produced by electrons moving up and down an antenna. Visible light is produced by electrons changing energy states in an atom.
Light Emission www.physics.sfasu.edu/friedfeld/ch29lec.ppt Radio waves are produced by electrons moving up and down an antenna. Visible light is produced by electrons changing energy states in an atom.
Earlier we learned that hot, opaque objects produce continuous spectra of radiation of different wavelengths.
 Section7: The Bohr Atom Earlier we learned that hot, opaque objects produce continuous spectra of radiation of different wavelengths. Continuous Spectrum Everyone has seen the spectrum produced when white
Section7: The Bohr Atom Earlier we learned that hot, opaque objects produce continuous spectra of radiation of different wavelengths. Continuous Spectrum Everyone has seen the spectrum produced when white
Lecture 0. NC State University
 Chemistry 736 Lecture 0 Overview NC State University Overview of Spectroscopy Electronic states and energies Transitions between states Absorption and emission Electronic spectroscopy Instrumentation Concepts
Chemistry 736 Lecture 0 Overview NC State University Overview of Spectroscopy Electronic states and energies Transitions between states Absorption and emission Electronic spectroscopy Instrumentation Concepts
where n = (an integer) =
 5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
Bohr. Electronic Structure. Spectroscope. Spectroscope
 Bohr Electronic Structure Bohr proposed that the atom has only certain allowable energy states. Spectroscope Using a device called a it was found that gaseous elements emitted electromagnetic radiation
Bohr Electronic Structure Bohr proposed that the atom has only certain allowable energy states. Spectroscope Using a device called a it was found that gaseous elements emitted electromagnetic radiation
Chemistry 795T. Lecture 7. Electromagnetic Spectrum Black body Radiation. NC State University
 Chemistry 795T Lecture 7 Electromagnetic Spectrum Black body Radiation NC State University Black body Radiation An ideal emitter of radiation is called a black body. Observation: that peak of the energy
Chemistry 795T Lecture 7 Electromagnetic Spectrum Black body Radiation NC State University Black body Radiation An ideal emitter of radiation is called a black body. Observation: that peak of the energy
Chemistry 795T. Black body Radiation. The wavelength and the frequency. The electromagnetic spectrum. Lecture 7
 Chemistry 795T Lecture 7 Electromagnetic Spectrum Black body Radiation NC State University Black body Radiation An ideal emitter of radiation is called a black body. Observation: that peak of the energy
Chemistry 795T Lecture 7 Electromagnetic Spectrum Black body Radiation NC State University Black body Radiation An ideal emitter of radiation is called a black body. Observation: that peak of the energy
ATMO/OPTI 656b Spring 2009
 Nomenclature and Definition of Radiation Quantities The various Radiation Quantities are defined in Table 2-1. Keeping them straight is difficult and the meanings may vary from textbook to textbook. I
Nomenclature and Definition of Radiation Quantities The various Radiation Quantities are defined in Table 2-1. Keeping them straight is difficult and the meanings may vary from textbook to textbook. I
Lasers & Holography. Ulrich Heintz Brown University. 4/5/2016 Ulrich Heintz - PHYS 1560 Lecture 10 1
 Lasers & Holography Ulrich Heintz Brown University 4/5/2016 Ulrich Heintz - PHYS 1560 Lecture 10 1 Lecture schedule Date Topic Thu, Jan 28 Introductory meeting Tue, Feb 2 Safety training Thu, Feb 4 Lab
Lasers & Holography Ulrich Heintz Brown University 4/5/2016 Ulrich Heintz - PHYS 1560 Lecture 10 1 Lecture schedule Date Topic Thu, Jan 28 Introductory meeting Tue, Feb 2 Safety training Thu, Feb 4 Lab
Stimulated Emission. Electrons can absorb photons from medium. Accelerated electrons emit light to return their ground state
 Lecture 15 Stimulated Emission Devices- Lasers Stimulated emission and light amplification Einstein coefficients Optical fiber amplifiers Gas laser and He-Ne Laser The output spectrum of a gas laser Laser
Lecture 15 Stimulated Emission Devices- Lasers Stimulated emission and light amplification Einstein coefficients Optical fiber amplifiers Gas laser and He-Ne Laser The output spectrum of a gas laser Laser
CHEMISTRY - TRO 4E CH.7 - THE QUANTUM-MECHANICAL MODEL OF THE ATOM
 !! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
!! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
Laser Physics 168 Chapter 1 Introductory concepts. Nayer Eradat SJSU Spring 2012
 Laser Physics 168 Chapter 1 Introductory concepts Nayer Eradat SJSU Spring 2012 Material to be covered IntroducCon to many concepts that will be covered Three major elements of a laser 1. An accve material
Laser Physics 168 Chapter 1 Introductory concepts Nayer Eradat SJSU Spring 2012 Material to be covered IntroducCon to many concepts that will be covered Three major elements of a laser 1. An accve material
ATOMIC PHYSICS. history/cosmology/tools/ tools-spectroscopy.htm CHAPTER 9 - FROM SPECTROSCOPY TO ATOMS
 ATOMIC PHYSICS http://www.aip.org/ history/cosmology/tools/ tools-spectroscopy.htm CHAPTER 9 - FROM SPECTROSCOPY TO ATOMS What We Will Study Basics of electromagnetic radiation - The AC generator, again
ATOMIC PHYSICS http://www.aip.org/ history/cosmology/tools/ tools-spectroscopy.htm CHAPTER 9 - FROM SPECTROSCOPY TO ATOMS What We Will Study Basics of electromagnetic radiation - The AC generator, again
5.111 Lecture Summary #5 Friday, September 12, 2014
 5.111 Lecture Summary #5 Friday, September 12, 2014 Readings for today: Section 1.3 Atomic Spectra, Section 1.7 up to equation 9b Wavefunctions and Energy Levels, Section 1.8 The Principle Quantum Number.
5.111 Lecture Summary #5 Friday, September 12, 2014 Readings for today: Section 1.3 Atomic Spectra, Section 1.7 up to equation 9b Wavefunctions and Energy Levels, Section 1.8 The Principle Quantum Number.
Hint: unit of energy transferred is equal to: hν = hc
 CH 0 Fall 08 Discussion # Chapter 8 Your name: TF s name Discussion Day/Time: Things you should know when you leave Discussion today: Atomic (matter) Emission and absorption of light. Energy Conservation
CH 0 Fall 08 Discussion # Chapter 8 Your name: TF s name Discussion Day/Time: Things you should know when you leave Discussion today: Atomic (matter) Emission and absorption of light. Energy Conservation
PhysicsAndMathsTutor.com 1
 Q1. When a clean metal surface in a vacuum is irradiated with ultraviolet radiation of a certain frequency, electrons are emitted from the metal. (a) Explain why the kinetic energy of the emitted electrons
Q1. When a clean metal surface in a vacuum is irradiated with ultraviolet radiation of a certain frequency, electrons are emitted from the metal. (a) Explain why the kinetic energy of the emitted electrons
Modern Physics- Introduction. L 35 Modern Physics [1] ATOMS and classical physics. Newton s Laws have flaws! accelerated charges radiate energy
![Modern Physics- Introduction. L 35 Modern Physics [1] ATOMS and classical physics. Newton s Laws have flaws! accelerated charges radiate energy Modern Physics- Introduction. L 35 Modern Physics [1] ATOMS and classical physics. Newton s Laws have flaws! accelerated charges radiate energy](/thumbs/87/96907347.jpg) L 35 Modern Physics [1] Introduction- quantum physics Particles of light PHOTONS The photoelectric effect Photocells & intrusion detection devices The Bohr atom emission & absorption of radiation LASERS
L 35 Modern Physics [1] Introduction- quantum physics Particles of light PHOTONS The photoelectric effect Photocells & intrusion detection devices The Bohr atom emission & absorption of radiation LASERS
[2] (b) An electron is accelerated from rest through a potential difference of 300 V.
![[2] (b) An electron is accelerated from rest through a potential difference of 300 V. [2] (b) An electron is accelerated from rest through a potential difference of 300 V.](/thumbs/89/98791036.jpg) 1 (a) In atomic physics electron energies are often stated in electronvolts (ev) Define the electronvolt. State its value in joule.. [2] (b) An electron is accelerated from rest through a potential difference
1 (a) In atomic physics electron energies are often stated in electronvolts (ev) Define the electronvolt. State its value in joule.. [2] (b) An electron is accelerated from rest through a potential difference
Chapter 3. Electromagnetic Theory, Photons. and Light. Lecture 7
 Lecture 7 Chapter 3 Electromagnetic Theory, Photons. and Light Sources of light Emission of light by atoms The electromagnetic spectrum see supplementary material posted on the course website Electric
Lecture 7 Chapter 3 Electromagnetic Theory, Photons. and Light Sources of light Emission of light by atoms The electromagnetic spectrum see supplementary material posted on the course website Electric
X-Rays from Atoms. These are called K α X-rays See table 29.1 for the energy of K α X-rays produced by some elements. Section 29.3
 X-Rays from Atoms The highest photon energy available in a hydrogen atom is in the ultraviolet part of the electromagnetic spectrum Other atoms can emit much more energetic photons larger Z, more electric
X-Rays from Atoms The highest photon energy available in a hydrogen atom is in the ultraviolet part of the electromagnetic spectrum Other atoms can emit much more energetic photons larger Z, more electric
OPTICAL GAIN AND LASERS
 OPTICAL GAIN AND LASERS 01-02-1 BY DAVID ROCKWELL DIRECTOR, RESEARCH & DEVELOPMENT fsona COMMUNICATIONS MARCH 6, 2001 OUTLINE 01-02-2 I. DEFINITIONS, BASIC CONCEPTS II. III. IV. OPTICAL GAIN AND ABSORPTION
OPTICAL GAIN AND LASERS 01-02-1 BY DAVID ROCKWELL DIRECTOR, RESEARCH & DEVELOPMENT fsona COMMUNICATIONS MARCH 6, 2001 OUTLINE 01-02-2 I. DEFINITIONS, BASIC CONCEPTS II. III. IV. OPTICAL GAIN AND ABSORPTION
Atoms, Molecules and Solids. From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation of the wave function:
 Essay outline and Ref to main article due next Wed. HW 9: M Chap 5: Exercise 4 M Chap 7: Question A M Chap 8: Question A From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation
Essay outline and Ref to main article due next Wed. HW 9: M Chap 5: Exercise 4 M Chap 7: Question A M Chap 8: Question A From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation
Modern Physics. Unit 6: Hydrogen Atom - Radiation Lecture 6.5: Optical Absorption. Ron Reifenberger Professor of Physics Purdue University
 Modern Physics Unit 6: Hydrogen tom - Radiation Lecture 6.5: Optical bsorption Ron Reifenberger Professor of Physics Purdue University 1 We now have a simple quantum model for how light is emitted. How
Modern Physics Unit 6: Hydrogen tom - Radiation Lecture 6.5: Optical bsorption Ron Reifenberger Professor of Physics Purdue University 1 We now have a simple quantum model for how light is emitted. How
LASERS AGAIN? Phys 1020, Day 17: Questions? LASERS: Next Up: Cameras and optics Eyes to web: Final Project Info
 LASERS AGAIN? Phys 1020, Day 17: Questions? LASERS: 14.3 Next Up: Cameras and optics Eyes to web: Final Project Info 1 Group Exercise Your pennies will simulate a two state atom; tails = ground state,
LASERS AGAIN? Phys 1020, Day 17: Questions? LASERS: 14.3 Next Up: Cameras and optics Eyes to web: Final Project Info 1 Group Exercise Your pennies will simulate a two state atom; tails = ground state,
Materialwissenschaft und Nanotechnologie. Introduction to Lasers
 Materialwissenschaft und Nanotechnologie Introduction to Lasers Dr. Andrés Lasagni Lehrstuhl für Funktionswerkstoffe Sommersemester 007 1-Introduction to LASER Contents: Light sources LASER definition
Materialwissenschaft und Nanotechnologie Introduction to Lasers Dr. Andrés Lasagni Lehrstuhl für Funktionswerkstoffe Sommersemester 007 1-Introduction to LASER Contents: Light sources LASER definition
Classification of spectroscopic methods
 Introduction Spectroscopy is the study of the interaction between the electromagnetic radiation and the matter. Spectrophotometry is the measurement of these interactions i.e. the measurement of the intensity
Introduction Spectroscopy is the study of the interaction between the electromagnetic radiation and the matter. Spectrophotometry is the measurement of these interactions i.e. the measurement of the intensity
Energy and the Quantum Theory
 Energy and the Quantum Theory Light electrons are understood by comparing them to light 1. radiant energy 2. travels through space 3. makes you feel warm Light has properties of waves and particles Amplitude:
Energy and the Quantum Theory Light electrons are understood by comparing them to light 1. radiant energy 2. travels through space 3. makes you feel warm Light has properties of waves and particles Amplitude:
Physics 107: Ideas of Modern Physics
 Physics 107: Ideas of Modern Physics Exam 3 Nov. 30, 2005 Name ID # Section # On the Scantron sheet, 1) Fill in your name 2) Fill in your student ID # (not your social security #) 3) Fill in your section
Physics 107: Ideas of Modern Physics Exam 3 Nov. 30, 2005 Name ID # Section # On the Scantron sheet, 1) Fill in your name 2) Fill in your student ID # (not your social security #) 3) Fill in your section
Chapter 5 Electrons In Atoms
 Chapter 5 Electrons In Atoms 5.1 Revising the Atomic Model 5.2 Electron Arrangement in Atoms 5.3 Atomic Emission Spectra and the Quantum Mechanical Model 1 Copyright Pearson Education, Inc., or its affiliates.
Chapter 5 Electrons In Atoms 5.1 Revising the Atomic Model 5.2 Electron Arrangement in Atoms 5.3 Atomic Emission Spectra and the Quantum Mechanical Model 1 Copyright Pearson Education, Inc., or its affiliates.
UNIT : QUANTUM THEORY AND THE ATOM
 Name St.No. Date(YY/MM/DD) / / Section UNIT 102-10: QUANTUM THEORY AND THE ATOM OBJECTIVES Atomic Spectra for Hydrogen, Mercury and Neon. 1. To observe various atomic spectra with a diffraction grating
Name St.No. Date(YY/MM/DD) / / Section UNIT 102-10: QUANTUM THEORY AND THE ATOM OBJECTIVES Atomic Spectra for Hydrogen, Mercury and Neon. 1. To observe various atomic spectra with a diffraction grating
Chemistry Instrumental Analysis Lecture 2. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 2 Electromagnetic Radiation Can be described by means of a classical sinusoidal wave model. Oscillating electric and magnetic field. (Wave model) wavelength,
Chemistry 4631 Instrumental Analysis Lecture 2 Electromagnetic Radiation Can be described by means of a classical sinusoidal wave model. Oscillating electric and magnetic field. (Wave model) wavelength,
The Photoelectric Effect
 The Photoelectric Effect Lenard s experiment The photon model Light as photons Einstein s explanation of the photoelectric effect Photon energy Electron volts Electron energy 1 Lenard s experiment Philipp
The Photoelectric Effect Lenard s experiment The photon model Light as photons Einstein s explanation of the photoelectric effect Photon energy Electron volts Electron energy 1 Lenard s experiment Philipp
hf = E 1 - E 2 hc = E 1 - E 2 λ FXA 2008 Candidates should be able to : EMISSION LINE SPECTRA
 1 Candidates should be able to : EMISSION LINE SPECTRA Explain how spectral lines are evidence for the existence of discrete energy levels in isolated atoms (i.e. in a gas discharge lamp). Describe the
1 Candidates should be able to : EMISSION LINE SPECTRA Explain how spectral lines are evidence for the existence of discrete energy levels in isolated atoms (i.e. in a gas discharge lamp). Describe the
9. RADIATIVE PROCESSES I -- ATOMS AND LIGHT
 QM P453 F95 (Zorn) Atoms in the Radiation Field, I page 9.1 9. RADIATIVE PROCESSES I -- ATOMS AND LIGHT We have discussed the field as a system and described it in terms of excitations of its normal modes.
QM P453 F95 (Zorn) Atoms in the Radiation Field, I page 9.1 9. RADIATIVE PROCESSES I -- ATOMS AND LIGHT We have discussed the field as a system and described it in terms of excitations of its normal modes.
Physics and the Quantum Mechanical Model
 chemistry 1 of 38 Mechanical Model Neon advertising signs are formed from glass tubes bent in various shapes. An electric current passing through the gas in each glass tube makes the gas glow with its
chemistry 1 of 38 Mechanical Model Neon advertising signs are formed from glass tubes bent in various shapes. An electric current passing through the gas in each glass tube makes the gas glow with its
Wavelength λ Velocity v. Electric Field Strength Amplitude A. Time t or Distance x time for 1 λ to pass fixed point. # of λ passing per s ν= 1 p
 Introduction to Spectroscopy (Chapter 6) Electromagnetic radiation (wave) description: Wavelength λ Velocity v Electric Field Strength 0 Amplitude A Time t or Distance x Period p Frequency ν time for 1
Introduction to Spectroscopy (Chapter 6) Electromagnetic radiation (wave) description: Wavelength λ Velocity v Electric Field Strength 0 Amplitude A Time t or Distance x Period p Frequency ν time for 1
Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell?
 AP Chemistry Ms. Ye Name Date Block Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell? 1 st shell 2 nd shell
AP Chemistry Ms. Ye Name Date Block Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell? 1 st shell 2 nd shell
Observation of Atomic Spectra
 Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
c = λν 10/23/13 What gives gas-filled lights their colors? Chapter 5 Electrons In Atoms
 CHEMISTRY & YOU What gives gas-filled lights their colors? Chapter 5 Electrons In Atoms 5.1 Revising the Atomic Model 5. Electron Arrangement in Atoms 5.3 Atomic and the Quantum Mechanical Model An electric
CHEMISTRY & YOU What gives gas-filled lights their colors? Chapter 5 Electrons In Atoms 5.1 Revising the Atomic Model 5. Electron Arrangement in Atoms 5.3 Atomic and the Quantum Mechanical Model An electric
