CHEM 330. Final Exam December 8, 2010 ANSWERS. This a closed-notes, closed-book exam. The use of molecular models is allowed
|
|
|
- Cuthbert Park
- 5 years ago
- Views:
Transcription
1 CEM 330 Final Exam December 8, 2010 Your name: ASWERS This a closed-notes, closed-book exam The use of molecular models is allowed This exam contains 10 pages Time: 2h 30 min 1. / / / / / / / / 40 TTAL / 250 = / 100 This exam counts for 45% of your CEM 330 final grade
2 Chem 330 final exam p. 2 of (20 pts.) Indicate the approximate pka for the dissociation of the in boldface in the substances listed below approx. pka = Et tert-u Et Et approx. pka = CF 3 S (20 pts.) Write a chemical equation to show an example of (do not write mechanisms just the reactions): a. A cross-claisen reaction that works well under conditions of reversible enolate formation: CEt C 3 CEt a, cat. Et CEt b. An Evans aldol reaction that occurs between stereochemically matched reactants: C + u u n c. A nucleophilic addition to a chiral aldehyde that occurs in accord with the Felkin-Ahn reactivity model: C + TMS Et 2 F 3 d. A nucleophilic addition to a chiral aldehyde that occurs in accord with the Cram-Felkin reactivity model: TMS C + Et 2 F 3
3 Chem 330 final exam p. 3 of 10 e. An Evans aldol reaction that occurs between stereochemically mismatched reactants: C + u n u 3. (20 pts.) Write accurate mechanisms for the following known reactions: a. 150 C CEt EtC + C 3 C EtC Alder EtC Diels- retro- Diels- Alder EtC C b. clearly a aylis- illman reaction: C catalyst A C Et catalyst A = May be rendered as while writing the mechanism C C C ± + C C +
4 Chem 330 final exam p. 4 of (30 pts.) riefly rationalize the stereochemical outcome of the following reactions and write clear diagrams to illustrate your arguments: a. The major product of the Mukaiyama aldol reaction of A with is compound C if F 3 is used as the promoter, but compound D if TiCl 4 is used instead: F 3 C TMS TiCl4 + C A D Product C forms in accord with the Felkin-Anh model from the aldehyde-f 3 complex i : F 3 i F 3 TMS Re-face attack (Felkin- Anh) F 3 TMS C Product D forms in accord with the Felkin-Anh chelated model from the aldehyde-ticl 4 complex ii : TiCl 4 ii 2 Cl 4 Ti 2 TMS Si-face attack (chelated Felkin- Anh) 2 Cl 4 Ti TMS D b. In the presence of F 3, the Danishefsky diene undergoes a Diels-Alder-like reaction with aldehyde E, acting now as a dienophile, to give F as the major product: TMS C F 3 E F the regiochemical the diene attacks ( ) course of the reaction (+) ( ) the C= group in is in accod with (+) accord with the atomic polarization: R (+) Cram-Felkin TMS ( ) reactivity model: TMS F 3 the reaction occurs with exo topology due to the lack of secondary orbital interactions: TMS SI's!!! So: TMS exo-topology (less steric compression) prevails TMS TMS Si-face attack (Cram- Felkin) F
5 Chem 330 final exam p. 5 of (40 pts.) Complete the following diagrams by writing all the missing reagents / intermediate products in the appropriate boxes. Important: (i) aqueous workups are understood; (ii) compounds a. must be drawn with the correct configuration of all stereogenic centers. CEt n CEt C 2 2 EtC CEt a, cat. Et heat, then mild 3 + EtC (rg. Lett. 2010, 12, 5146) b. Cy 2 -Cl Et 3, then a(ac) 3 n C TS n TS (rg. Lett. 2010, 12, 5530) n TS c. TES- EtC DIAL Py S 3 DMS then Et 3 TES- C A u u u 2 Tf and Et 3 then:
6 Chem 330 final exam p. 6 of TIPSTf Et 3 A + TES 2. TAF TIPS C 3. Py S 3 DMS then Et 3 (rg. Lett. 2006, 8, 2791) 6. (40 pts.) Predict the structure of the major product expected from the following reactions. otes: (i) it is not necessary to draw mechanisms; (ii) aqueous workups at appropriate stages are understood. a. EtC CEt 1. a, cat. Et then C 2 =C-C 2 r 2. Aq. ar, DMS, reflux 1. SCl 2 2. ldrum's acid, Et 3 b. C 3., reflux 4. excess a, I I c. 1. Li, liq. 3 then 2. C 3 a
7 Chem 330 final exam p. 7 of 10 d. 1. LDA, TF 2. -C e Tf Et 3 2. C f. Et 1. LDA, TF 2. -TS C TS Et g. C excess C aq. a h. 1. LDA, TF / MPA Et 2. C Et
8 Chem 330 final exam p. 8 of (40 pts.) Complete the following equations by indicating all the reagents that are necessary to effect the transformations shown. Provide your answers as a numbered list of reagents, in the correct order, written over/under the reaction arrows. ote: aqueous workups are understood and are not to be included in your answers. a. 1. TMS-I, TMS , then Zn / + 3. Et, + 4. tert-uk b. TS 1. aq. Li 4. tert-uk 2. CDI 5. (Et) 2 C= 3. -C=C(Et)Li (from EtCEt + LDA) TS Et c. 1. LDA, then Ser 4. tert-uk 2. MCPA, then heat 5. (Et) 2 C= 3. 2 CuLi, then a, cat. I (C 2 ) 5 I, TMEDA Et CEt d. Li TS 1. LDA, TF CEt TS TS C Et 78 C 2. TSCl, Et 3 4. Py S 3, DMS, then Et 3 3. DIAL 5. 3 P=C 2 1. TMSTf, Et 3 e. 2. Cl, TiCl 4
9 Chem 330 final exam p. 9 of (40 pts.) Propose a method to achieve the enantioselective synthesis of the molecules shown below starting with the suggested building blocks. e careful about protecting groups and configurations of stereocenters. Assume the availability of all needed reagents, auxiliaries, etc. Present your answer as a clear flowchart. It is not necessary to draw mechanisms or to indicate aqueous workups. a. TS TS starting with: TS I 1. tert-uli 2. (u 3 P) 2 CuI 3. CEt TS DIAL Et TS CEt TS DIAL (Cram- Felkin) Et Li LDA TF 78 C CEt TS TS TS TPAP M TS 1. DIAL 2. Py S 3, DMS then Et 3 TS TS TS TSCl, Et 3 u u + TS 3 P=C C u 2 Tf, Et 3
10 Chem 330 final exam p. 10 of 10 b. C starting with: TS 1. aq. Li 2. TAF TS Cy 2 Tf TS 3 P=CCEt S Et 3 2 Cy 2 n CEt TS TS Py S 3 DMS then Et 3 n LDA TS DIAL TS TS DIAL Li n TS Pr 3 r alternative answers may be acceptable appy olidays!
CHEM 330. Final Exam December 11, 2007 A N S W E R S. This a closed-notes, closed-book exam. The use of molecular models is allowed
 CEM 330 Final Exam December 11, 2007 Your name: A S W E R S This a closed-notes, closed-book exam The use of molecular models is allowed This exam contains 12 pages Time: 2h 30 min 1. / 20 / 20 3. / 30
CEM 330 Final Exam December 11, 2007 Your name: A S W E R S This a closed-notes, closed-book exam The use of molecular models is allowed This exam contains 12 pages Time: 2h 30 min 1. / 20 / 20 3. / 30
CHEM 330. Final Exam December 5, 2014 ANSWERS. This a closed-notes, closed-book exam. The use of molecular models is allowed
 CEM 330 Final Exam December 5, 2014 Your name: ASWERS This a closed-notes, closed-book exam The use of molecular models is allowed This exam consists of 12 pages Time: 2h 30 min 1. / 30 2. / 30 3. / 30
CEM 330 Final Exam December 5, 2014 Your name: ASWERS This a closed-notes, closed-book exam The use of molecular models is allowed This exam consists of 12 pages Time: 2h 30 min 1. / 30 2. / 30 3. / 30
CHEM 330. Final Exam December 11, This a closed-notes, closed-book exam. The use of molecular models is allowed. This exam contains 12 pages
 CEM 330 Final Exam December 11, 2007 Your name: This a closed-notes, closed-book exam The use of molecular models is allowed This exam contains 12 pages Time: 2h 30 min 1. / 20 2. / 20 3. / 30 4. / 40
CEM 330 Final Exam December 11, 2007 Your name: This a closed-notes, closed-book exam The use of molecular models is allowed This exam contains 12 pages Time: 2h 30 min 1. / 20 2. / 20 3. / 30 4. / 40
CHEM 203. Final Exam December 12, This a closed-notes, closed-book exam. You may use your set of molecular models. This test contains 11 pages
 CEM 203 Final Exam December 12, 2012 Your name: This a closed-notes, closed-book exam You may use your set of molecular models This test contains 11 pages Time: 2h 30 min 1. / 20 2. / 24 3. / 26 4. / 30
CEM 203 Final Exam December 12, 2012 Your name: This a closed-notes, closed-book exam You may use your set of molecular models This test contains 11 pages Time: 2h 30 min 1. / 20 2. / 24 3. / 26 4. / 30
CHEM 203. Final Exam December 16, This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Final Exam December 16, 2014 Your name: This a closed-notes, closed-book exam You may use your set of molecular models This test consists of 10 pages Time: 2h 30 min 1. / 20 2. / 20 3. / 30 4.
CEM 203 Final Exam December 16, 2014 Your name: This a closed-notes, closed-book exam You may use your set of molecular models This test consists of 10 pages Time: 2h 30 min 1. / 20 2. / 20 3. / 30 4.
CHEM 203. Final Exam December 15, 2010 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
CHEM 203. Midterm Exam 2 November 12, 2013 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Midterm Exam 2 November 12, 2013 ANSWERS Your name: This a closed-notes, closed-book exam You may use your set of molecular models This document contains 7 pages Time: 1h 30 min 1. / 10 2. / 15
CEM 203 Midterm Exam 2 November 12, 2013 ANSWERS Your name: This a closed-notes, closed-book exam You may use your set of molecular models This document contains 7 pages Time: 1h 30 min 1. / 10 2. / 15
CHEM 203. Midterm Exam 2 November 13, 2014 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Midterm Exam 2 ovember 13, 2014 Your name: ASWERS This a closed-notes, closed-book exam You may use your set of molecular models This document contains 7 pages Time: 1h 30 min 1. / 10 2. / 15 3.
CEM 203 Midterm Exam 2 ovember 13, 2014 Your name: ASWERS This a closed-notes, closed-book exam You may use your set of molecular models This document contains 7 pages Time: 1h 30 min 1. / 10 2. / 15 3.
CHEM 203. Final Exam December 18, This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Final Exam December 18, 2013 Your name: This a closed-notes, closed-book exam You may use your set of molecular models This test consists of 10 pages Time: 2h 30 min 1. / 20 2. / 20 3. / 30 4.
CEM 203 Final Exam December 18, 2013 Your name: This a closed-notes, closed-book exam You may use your set of molecular models This test consists of 10 pages Time: 2h 30 min 1. / 20 2. / 20 3. / 30 4.
CHEM 203. Final Exam December 18, 2013 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Your name: Final Exam December 18, 2013 ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test consists of 10 pages Time: 2h 30 min 1. / 20 2. / 20 3.
CEM 203 Your name: Final Exam December 18, 2013 ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test consists of 10 pages Time: 2h 30 min 1. / 20 2. / 20 3.
Name: CHEM 633: Advanced Organic Chem: Physical Problem Set 5 Due 11/10/17
 ame: CEM 633: Advanced rganic Chem: ysical Problem Set 5 Due 11/10/17 Please do not look up references until after you turn in the problem set unless otherwise noted. For the following problems, please
ame: CEM 633: Advanced rganic Chem: ysical Problem Set 5 Due 11/10/17 Please do not look up references until after you turn in the problem set unless otherwise noted. For the following problems, please
CHEM 203. Midterm Exam 1 October 31, 2008 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Midterm Exam 1 ctober 31, 2008 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This exam contains 8 pages Time: 1h 30 min 1. / 15 2. / 16 3. /
CEM 203 Midterm Exam 1 ctober 31, 2008 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This exam contains 8 pages Time: 1h 30 min 1. / 15 2. / 16 3. /
(5) a. b. (6) N H CH 3 N H O H O (7) CH 3 O (8) OCH 3 2. explanation:
 ame Key 15 W1-Exam o. Page I. (16 points) For each of the following pairs of compounds, predict which compound is more acidic. Compare the two underlined s for each pair and circle the compound that is
ame Key 15 W1-Exam o. Page I. (16 points) For each of the following pairs of compounds, predict which compound is more acidic. Compare the two underlined s for each pair and circle the compound that is
Chem 22 Final Exam Practice
 Chem 22 Final Exam Practice Questions taken from regular tests given during the previous semesters. Only one answer is correct unless the question says otherwise. The questions are somewhat scrambled with
Chem 22 Final Exam Practice Questions taken from regular tests given during the previous semesters. Only one answer is correct unless the question says otherwise. The questions are somewhat scrambled with
Final Exam /415 ( CHEM 6352 Fall %) Name
 Final Exam /415 ( CEM 6352 Fall 2011 %) Name Dec. 15 th, 2011 8:00-12:00 You may NT use any references or aids to complete the following with the exception of a chemical model set and the scrap paper that
Final Exam /415 ( CEM 6352 Fall 2011 %) Name Dec. 15 th, 2011 8:00-12:00 You may NT use any references or aids to complete the following with the exception of a chemical model set and the scrap paper that
O + k 2. H(D) Ar. MeO H(D) rate-determining. step?
 ame: CEM 633: Advanced rganic Chem: ysical Problem Set 6 (Due Thurs, 12/8/16) Please do not look up references until after you turn in the problem set unless otherwise noted. For the following problems,
ame: CEM 633: Advanced rganic Chem: ysical Problem Set 6 (Due Thurs, 12/8/16) Please do not look up references until after you turn in the problem set unless otherwise noted. For the following problems,
CEM 852 Exam-2 April 2, 2016
 CM 852 xam-2 April 2, 2016 This exam consists of 6 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 100. In answering your questions, please
CM 852 xam-2 April 2, 2016 This exam consists of 6 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 100. In answering your questions, please
Name: CHEM 633: Advanced Organic Chemistry: Physical Final Exam. Please answer the following questions clearly and concisely.
 Name: 1 CEM 633: Advanced rganic Chemistry: Physical Final Exam Please answer the following questions clearly and concisely. You may write your answers in the space provided and/or on additional pages.
Name: 1 CEM 633: Advanced rganic Chemistry: Physical Final Exam Please answer the following questions clearly and concisely. You may write your answers in the space provided and/or on additional pages.
CEM 852 Exam LDA, THF, 0 C, 15 min; then
 CEM 85 Exam- April, 005 This exam consists of 5 pages. Please write ALL your answers in the answer books. Please write legibly and draw all structures clearly. Good luck. 1. Provide examples of the following
CEM 85 Exam- April, 005 This exam consists of 5 pages. Please write ALL your answers in the answer books. Please write legibly and draw all structures clearly. Good luck. 1. Provide examples of the following
California State Polytechnic University, Pomona Chem 315. Exam Points 1. Nomenclature (1) 30
 hem 315 Final Exam Fall, 2007 Beauchamp ame: Topic Total Points Exam Points 1. omenclature (1) 30 redit 2. Relative rder of Reactivity 20 3. Reactions Page, using reactions learned thus far (30) 30 4.
hem 315 Final Exam Fall, 2007 Beauchamp ame: Topic Total Points Exam Points 1. omenclature (1) 30 redit 2. Relative rder of Reactivity 20 3. Reactions Page, using reactions learned thus far (30) 30 4.
2.222 Practice Problems 2003
 2.222 Practice Problems 2003 Set #1 1. Provide the missing starting compound(s), reagent/solvent, or product to correctly complete each of the following. Most people in the class have not done this type
2.222 Practice Problems 2003 Set #1 1. Provide the missing starting compound(s), reagent/solvent, or product to correctly complete each of the following. Most people in the class have not done this type
CEM 852 Exam-1 February 20, 2016
 EM 852 Exam-1 February 20, 2016 This exam consists of 5 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 100. In answering your questions,
EM 852 Exam-1 February 20, 2016 This exam consists of 5 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 100. In answering your questions,
CEM 852 Exam-2 April 11, 2015
 CEM 852 Exam-2 April 11, 2015 This exam consists of 6 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 100. In answering your questions,
CEM 852 Exam-2 April 11, 2015 This exam consists of 6 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 100. In answering your questions,
CEM 852 Final Exam. May 6, 2010
 CEM 852 Final Exam May 6, 2010 This exam consists of 7 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 150. n answering your questions,
CEM 852 Final Exam May 6, 2010 This exam consists of 7 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 150. n answering your questions,
Asymmetric Catalysis by Lewis Acids and Amines
 Asymmetric Catalysis by Lewis Acids and Amines Asymmetric Lewis acid catalysis - Chiral (bisooxazoline) copper (II) complexes - Monodentate Lewis acids: the formyl -bond Amine catalysed reactions Asymmetric
Asymmetric Catalysis by Lewis Acids and Amines Asymmetric Lewis acid catalysis - Chiral (bisooxazoline) copper (II) complexes - Monodentate Lewis acids: the formyl -bond Amine catalysed reactions Asymmetric
Final Exam April 30, 2014
 Bennett Department of Chemistry Chemistry 234 Final Exam April 30, 2014 ame: This exam is a closed book, closed notes. Calculators and a molecular model set are allowed. You must show your work in order
Bennett Department of Chemistry Chemistry 234 Final Exam April 30, 2014 ame: This exam is a closed book, closed notes. Calculators and a molecular model set are allowed. You must show your work in order
Stereoselective reactions of enolates
 1 Stereoselective reactions of enolates Chiral auxiliaries are frequently used to allow diastereoselective enolate reactions Possibly the most extensively studied are the Evan s oxazolidinones These are
1 Stereoselective reactions of enolates Chiral auxiliaries are frequently used to allow diastereoselective enolate reactions Possibly the most extensively studied are the Evan s oxazolidinones These are
O H HO H. !-D-galactopyranose
 ame Key W06-Exam o. Page I. ( points) A disaccharide is cleaved by a β-glycosidase, an enzyme that specifically hydrolyzes a β- glycosidic linkage. When the disaccharide is treated with excess dimethyl
ame Key W06-Exam o. Page I. ( points) A disaccharide is cleaved by a β-glycosidase, an enzyme that specifically hydrolyzes a β- glycosidic linkage. When the disaccharide is treated with excess dimethyl
CHEMISTRY 332 FINAL EXAM. December 14, I. (18 points) II. (48 points) III. (50 points) IV. (40 points) V. (26 points) VI.
 Seat No. Name (please print and circle your last name) CEMISTRY 332 FINAL EXAM December 14, 2005 I. (18 points) II. (48 points) III. (50 points) IV. (40 points) V. (26 points) VI. (18 points) TTAL (200
Seat No. Name (please print and circle your last name) CEMISTRY 332 FINAL EXAM December 14, 2005 I. (18 points) II. (48 points) III. (50 points) IV. (40 points) V. (26 points) VI. (18 points) TTAL (200
Stereoselective reactions of the carbonyl group
 1 Stereoselective reactions of the carbonyl group We have seen many examples of substrate control in nucleophilic addition to the carbonyl group (Felkin-Ahn & chelation control) If molecule does not contain
1 Stereoselective reactions of the carbonyl group We have seen many examples of substrate control in nucleophilic addition to the carbonyl group (Felkin-Ahn & chelation control) If molecule does not contain
Initials: 1. Chem 633: Advanced Organic Chemistry 2016 Final Exam
 Initials: 1 ame: Chem 633: Advanced rganic Chemistry 2016 Final Exam This exam is closed note, closed book. Please answer the following questions clearly and concisely. In general, use pictures and less
Initials: 1 ame: Chem 633: Advanced rganic Chemistry 2016 Final Exam This exam is closed note, closed book. Please answer the following questions clearly and concisely. In general, use pictures and less
Name: CHEM 633/634 Problem Set 1: Review Due Tues, Aug 29, 2017 (First Lecture!)
 ame: CEM 633/634 Problem Set 1: Review Due Tues, Aug 29, 2017 (First Lecture!) Please print this problem set. Answers must be in the spaces or boxes provided to receive full credit. You may work in groups,
ame: CEM 633/634 Problem Set 1: Review Due Tues, Aug 29, 2017 (First Lecture!) Please print this problem set. Answers must be in the spaces or boxes provided to receive full credit. You may work in groups,
Chemistry 35 Exam 2 Answers - April 9, 2007
 Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
CEM 852 Final Exam. May 5, 2011
 CEM 852 Final Exam May 5, 2011 This exam consists of 8 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 150. In answering your questions,
CEM 852 Final Exam May 5, 2011 This exam consists of 8 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 150. In answering your questions,
CHEM 330. Problem set Indicate the polarity of the starred carbon atoms in the following molecules:
 CEM 330 Problem set 1 1. Indicate the polarity of the starred carbon atoms in the following molecules: (+) Br ( ) (+) ( ) 2. Identify appropriate synthons and suitable reagents for the formation of the
CEM 330 Problem set 1 1. Indicate the polarity of the starred carbon atoms in the following molecules: (+) Br ( ) (+) ( ) 2. Identify appropriate synthons and suitable reagents for the formation of the
Stereoselective Organic Synthesis
 Stereoselective rganic Synthesis Prabhat Arya Professor and Leader, Chemical Biology Program Dean, Academic Affairs, Institute of Life Sciences (An Associate Institute of University of yderabad Supported
Stereoselective rganic Synthesis Prabhat Arya Professor and Leader, Chemical Biology Program Dean, Academic Affairs, Institute of Life Sciences (An Associate Institute of University of yderabad Supported
Midterm Exam 1. Chem 3B, Fall 2016 Thursday, September 29, :00 9:00 pm. Name. Student ID
 Midterm Exam 1 Chem 3B, Fall 2016 Thursday, September 29, 2016 7:00 9:00 pm ame Student ID You have 120 minutes to complete this exam. Please provide all answers in the space provided. Work drawn in the
Midterm Exam 1 Chem 3B, Fall 2016 Thursday, September 29, 2016 7:00 9:00 pm ame Student ID You have 120 minutes to complete this exam. Please provide all answers in the space provided. Work drawn in the
Stereoselective Organic Synthesis
 Stereoselective rganic Synthesis Prabhat Arya Professor and Leader, Chemical Biology Program Dean, Academic Affairs, Institute of Life Sciences (An Associate Institute of University of yderabad Supported
Stereoselective rganic Synthesis Prabhat Arya Professor and Leader, Chemical Biology Program Dean, Academic Affairs, Institute of Life Sciences (An Associate Institute of University of yderabad Supported
Suggested solutions for Chapter 34
 s for Chapter 34 34 PRBLEM 1 Predict the structure of the product of this Diels- Alder reaction. C 2 +? 3 Si Can you deal with a moderately complicated Diels- Alder? The diene is electron- rich and will
s for Chapter 34 34 PRBLEM 1 Predict the structure of the product of this Diels- Alder reaction. C 2 +? 3 Si Can you deal with a moderately complicated Diels- Alder? The diene is electron- rich and will
Only five of the molecules below may be prepared as the sole product of allylic halogenation of the respective alkene. Circle those five.
 Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 6 Answers Question 1. nly five of the molecules below may be prepared as the sole product of allylic halogenation of the respective alkene. Circle those
Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 6 Answers Question 1. nly five of the molecules below may be prepared as the sole product of allylic halogenation of the respective alkene. Circle those
Answers To Chapter 7 Problems.
 Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
Chem 112A: Final Exam
 Chem 112A: Final Exam December 14th, 2011 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
Chem 112A: Final Exam December 14th, 2011 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
CHEM 8A Summer Student ID # Organic Chemistry FINAL EXAM (400 points)
 CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry FINAL EXAM (400 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the
CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry FINAL EXAM (400 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the
Lecture Notes Chem 51B S. King I. Conjugation
 Lecture Notes Chem 51B S. King Chapter 16 Conjugation, Resonance, and Dienes I. Conjugation Conjugation occurs whenever p-orbitals can overlap on three or more adjacent atoms. Conjugated systems are more
Lecture Notes Chem 51B S. King Chapter 16 Conjugation, Resonance, and Dienes I. Conjugation Conjugation occurs whenever p-orbitals can overlap on three or more adjacent atoms. Conjugated systems are more
California State Polytechnic University, Pomona. Exam Points 1. Nomenclature (1) 25
 hem 316 Final Spring, 2004 Beauchamp ame: Topic Total Points Exam Points 1. omenclature (1) 25 redit Tautomers (acidic or base conditions) 20 3. Reactions Page (10 x 3 = 30) 30 4. Aromatic Mechanism and
hem 316 Final Spring, 2004 Beauchamp ame: Topic Total Points Exam Points 1. omenclature (1) 25 redit Tautomers (acidic or base conditions) 20 3. Reactions Page (10 x 3 = 30) 30 4. Aromatic Mechanism and
Chem 112A: Final Exam
 Chem 112A: Final Exam December 15th, 2010 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
Chem 112A: Final Exam December 15th, 2010 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
CHEM 232 Exam Two April 5, 2010
 CEM 232 Exam Two April 5, 2010 Name (-1 pt) TA Name (-1 pt) UIN (-1 pt) Question 1 [Qualitative anking, 40 points] A significant amount of time has been devoted to describing the physical and chemical
CEM 232 Exam Two April 5, 2010 Name (-1 pt) TA Name (-1 pt) UIN (-1 pt) Question 1 [Qualitative anking, 40 points] A significant amount of time has been devoted to describing the physical and chemical
Exam 4. Name CHEM 210. PBr 3. HBr. 1) NaH 2) Br H 2 SO 4. 1) NaOCH 3 2) dil. H 3 O + O CH 3 OH, H 2 SO 4. 1) LDA, -78 o C.
 Exam 4 Name CEM 210 1. (48 pts) Complete the equations for the following reactions. Show all organic products if two or more products form, indicate the major product. Indicate proper stereochemistry where
Exam 4 Name CEM 210 1. (48 pts) Complete the equations for the following reactions. Show all organic products if two or more products form, indicate the major product. Indicate proper stereochemistry where
[3,3]-Sigmatropic rearrangements
![[3,3]-Sigmatropic rearrangements [3,3]-Sigmatropic rearrangements](/thumbs/75/71504906.jpg) 1 [3,3]-Sigmatropic rearrangements heat R 1 R 3 R 1 R 3 R 1 R 3 A class of pericyclic reactions whose stereochemical outcome is governed by the geometric requirements of the cyclic transition state Reactions
1 [3,3]-Sigmatropic rearrangements heat R 1 R 3 R 1 R 3 R 1 R 3 A class of pericyclic reactions whose stereochemical outcome is governed by the geometric requirements of the cyclic transition state Reactions
The aldol reaction with metal enolates proceeds by a chair-like, pericyclic process: favored. disfavored. favored. disfavored
 The aldol reaction with metal enolates proceeds by a chair-like, pericyclic process: Z-enolates: M 2 M 2 syn 2 C 2 favored 2 M 2 anti disfavored E-enolates: M 2 2 C 3 C 3 C 2 favored 2 M M disfavored In
The aldol reaction with metal enolates proceeds by a chair-like, pericyclic process: Z-enolates: M 2 M 2 syn 2 C 2 favored 2 M 2 anti disfavored E-enolates: M 2 2 C 3 C 3 C 2 favored 2 M M disfavored In
Guideline 5: Tactical Bonds
 Guideline 5: Tactical Bonds f the molecule contains more than one functional group and hence there is a choice of which bonds to disconnect, how do you decide? Practice, but here are a few rough guidelines
Guideline 5: Tactical Bonds f the molecule contains more than one functional group and hence there is a choice of which bonds to disconnect, how do you decide? Practice, but here are a few rough guidelines
CHEMISTRY 850 Exam I Saturday, October 20, 2012 NAME (Print)
 CEMISTRY 850 Exam I Saturday, ctober 20, 2012 AME (Print) Question Max Points Score 1 12 2 8 3 12 4 10 5 14 6 10 7 12 8 24 9 16 10 8 11 8 12 14 13 16 14 36 BUS 8 Exam Total 1 1) Draw the detailed arrow
CEMISTRY 850 Exam I Saturday, ctober 20, 2012 AME (Print) Question Max Points Score 1 12 2 8 3 12 4 10 5 14 6 10 7 12 8 24 9 16 10 8 11 8 12 14 13 16 14 36 BUS 8 Exam Total 1 1) Draw the detailed arrow
OChem2 Course Pack. Practice Problems by Chapter. Practice Exams
 Chem2 Course Pack Practice Problems by Chapter Practice Exams Chemistry 3720 Problem Sets Chemistry 3720 Ch. 13-19 Synthesis Problems These problems are typical of those that will be on the upcoming exams
Chem2 Course Pack Practice Problems by Chapter Practice Exams Chemistry 3720 Problem Sets Chemistry 3720 Ch. 13-19 Synthesis Problems These problems are typical of those that will be on the upcoming exams
Also note here that the product is always a six membered ring with a double bond in it.
 Diels Alder Class 1 March 3, 2011 I want to talk in some detail over the next few classes about Diels Alder reactions. I am sure that most of you have heard of Diels Alder reactions before, but we will
Diels Alder Class 1 March 3, 2011 I want to talk in some detail over the next few classes about Diels Alder reactions. I am sure that most of you have heard of Diels Alder reactions before, but we will
THE UNIVERSITY OF CALGARY FACULTY OF SCIENCE FINAL EXAMINATION CHEMISTRY 350
 Page 1 of 16 TE UNIVERSITY F CALGARY FACULTY F SCIENCE FINAL EXAMINATIN CEMISTRY 350 April, 1997 Time: 3 ours PLEASE WRITE YUR NAME, STUDENT I.D. NUMBER AND SECTIN NUMBER (01 for MWF lectures and 02 for
Page 1 of 16 TE UNIVERSITY F CALGARY FACULTY F SCIENCE FINAL EXAMINATIN CEMISTRY 350 April, 1997 Time: 3 ours PLEASE WRITE YUR NAME, STUDENT I.D. NUMBER AND SECTIN NUMBER (01 for MWF lectures and 02 for
Stereoselective reactions of enolates: auxiliaries
 1 Stereoselective reactions of enolates: auxiliaries Chiral auxiliaries are frequently used to allow diastereoselective enolate reactions Possibly the most extensively studied are the Evan s oxazolidinones
1 Stereoselective reactions of enolates: auxiliaries Chiral auxiliaries are frequently used to allow diastereoselective enolate reactions Possibly the most extensively studied are the Evan s oxazolidinones
O N N. electrons in ring
 ame I. ( points) Page 1 A. The compound shown below is a commonly prescribed antifungal drug. It belongs to a category of compounds known as triazoles. Analyze the geometry and other properties for various
ame I. ( points) Page 1 A. The compound shown below is a commonly prescribed antifungal drug. It belongs to a category of compounds known as triazoles. Analyze the geometry and other properties for various
Total synthesis of Spongistatin
 Literature Semminar 1. Introduction: Total synthesis of Spongistatin Chen Zhihua (M2) Isolation: Pettit et al. J. rg. Chem. 1993, 58, 1302. Kitagawa et al. Tetrahedron Lett. 1993, 34, 1993. Fusetani et
Literature Semminar 1. Introduction: Total synthesis of Spongistatin Chen Zhihua (M2) Isolation: Pettit et al. J. rg. Chem. 1993, 58, 1302. Kitagawa et al. Tetrahedron Lett. 1993, 34, 1993. Fusetani et
Final Exam /340 ( CHEM 6352 Fall %) Name
 Final Exam /340 ( CEM 6352 Fall 2012 %) Name Dec. 15 th, 2012 9:30-13:45 You may NT use any references or aids to complete the following with the exception of a chemical model set and the scrap paper that
Final Exam /340 ( CEM 6352 Fall 2012 %) Name Dec. 15 th, 2012 9:30-13:45 You may NT use any references or aids to complete the following with the exception of a chemical model set and the scrap paper that
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320
 Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
UCSC, Binder. CHEM 8B Organic Chemistry II EXAM 2A, Winter 2018 (200 points)
 UCSC, Binder Name CEM 8B rganic Chemistry II EXAM 2A, Winter 2018 (200 points) Use your knowledge of organic chemistry conventions to answer the questions in the proper manner. Be sure to read all questions
UCSC, Binder Name CEM 8B rganic Chemistry II EXAM 2A, Winter 2018 (200 points) Use your knowledge of organic chemistry conventions to answer the questions in the proper manner. Be sure to read all questions
Exam 3 Professor R. Hoenigman
 I pledge to uphold the CU onor Code: CEM 3331-100 Spring 2008 Exam 3 Professor R. oenigman igh = 102 Low = 15 Average = 68 Signature ame (printed) Last four digits of your student ID number Recitation
I pledge to uphold the CU onor Code: CEM 3331-100 Spring 2008 Exam 3 Professor R. oenigman igh = 102 Low = 15 Average = 68 Signature ame (printed) Last four digits of your student ID number Recitation
Synthetic Developments Towards the Preparation of Erythromycin and Erythronolide Derivatives
 ynthetic Developments Towards the Preparation of Erythromycin and Erythronolide Derivatives Russell C. mith Denmark Group Meeting 8-9-2005 most extensive project in organic synthesis this phenomenon is
ynthetic Developments Towards the Preparation of Erythromycin and Erythronolide Derivatives Russell C. mith Denmark Group Meeting 8-9-2005 most extensive project in organic synthesis this phenomenon is
Chiral Catalyst II. Palladium Catalysed Allylic Displacement ( -allyl complexes) 1. L n Pd(0) 2. Nuc
 Chiral Catalyst II ast lecture we looked at asymmetric catalysis for oxidation and reduction Many other organic transformations, this has led to much investigation Today we will look at some others...
Chiral Catalyst II ast lecture we looked at asymmetric catalysis for oxidation and reduction Many other organic transformations, this has led to much investigation Today we will look at some others...
Diels-Alder Reaction
 Diels-Alder Reaction Method for synthesis of 6-membered ring ne-step, concerted reaction Termed [4+2] cycloaddition reaction where 4 and 2 electrons react. + Diels-Alder Reaction Discovered by. Diels and
Diels-Alder Reaction Method for synthesis of 6-membered ring ne-step, concerted reaction Termed [4+2] cycloaddition reaction where 4 and 2 electrons react. + Diels-Alder Reaction Discovered by. Diels and
Partial Periodic Table
 CEM 3311, Fall 2011 Professor Walba Third our Exam November 17, 2011 scores: 1) 20 2) 20 3) 20 4) 20 5) 20 100 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder tudent, I have neither
CEM 3311, Fall 2011 Professor Walba Third our Exam November 17, 2011 scores: 1) 20 2) 20 3) 20 4) 20 5) 20 100 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder tudent, I have neither
Chem 2600 Final Exam 2007, April 21st, Question One (10 marks)
 hem 2600 Final Exam 2007, April 21st, 9:00 am to 12:00 am You are permitted the use of a model kit; data sheets of pkas, pi Mos, periodic table and NMR/IR tables are provided by your instructor. No other
hem 2600 Final Exam 2007, April 21st, 9:00 am to 12:00 am You are permitted the use of a model kit; data sheets of pkas, pi Mos, periodic table and NMR/IR tables are provided by your instructor. No other
R or S? oxidation #: hybridization:
 Remember Lone pair-assisted ionization. ame 15 F07-Exam o. 1 Page I. (0 points) The following compounds are those used in our study on the enzymatic transformation of cholesterol to pregnenolone. 1) Designate
Remember Lone pair-assisted ionization. ame 15 F07-Exam o. 1 Page I. (0 points) The following compounds are those used in our study on the enzymatic transformation of cholesterol to pregnenolone. 1) Designate
Exam #1. Chemistry 334. Principles of Organic Chemistry II. Thursday October 5, 2006
 Exam #1 Chemistry 334 Principles of rganic Chemistry II Thursday ctober 5, 2006 ame: KEY. The exam is worth a total of 100 points; there are six questions. Please show all work to receive full credit for
Exam #1 Chemistry 334 Principles of rganic Chemistry II Thursday ctober 5, 2006 ame: KEY. The exam is worth a total of 100 points; there are six questions. Please show all work to receive full credit for
Chemistry 234 Practice Exam 3. The Periodic Table
 ame: Last First MI Chemistry 234 Practice Exam 3 Instructions: The first 15 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron sheet.
ame: Last First MI Chemistry 234 Practice Exam 3 Instructions: The first 15 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron sheet.
Suggested solutions for Chapter 41
 s for Chapter 41 41 PBLEM 1 Explain how this synthesis of amino acids, starting with natural proline, works. Explain the stereoselectivity of each step after the first. C 2 C 2 3 CF 3 C 2 2 Pd 2 C 2 +
s for Chapter 41 41 PBLEM 1 Explain how this synthesis of amino acids, starting with natural proline, works. Explain the stereoselectivity of each step after the first. C 2 C 2 3 CF 3 C 2 2 Pd 2 C 2 +
Cyclooctatetraene (2R)-2-phenylbutane benzyl chloride cycloocta-1,3,5,7-tetraene
 CM238-02 S05 Practice Material for xam 1 This is a compilation from relevant problems from last spring s exam 1&2. This is too long! 1. (4 pts) Draw 2 of the following 3: Cross one out or graded in order.
CM238-02 S05 Practice Material for xam 1 This is a compilation from relevant problems from last spring s exam 1&2. This is too long! 1. (4 pts) Draw 2 of the following 3: Cross one out or graded in order.
Exam 2 - Key Unless otherwise indicated, clearly indicate stereochemistry. You may use molecular models.
 Exam 2 - Key Unless otherwise indicated, clearly indicate stereochemistry. You may use molecular models. 1. (16 pts) For each of the following hypothetical equilibration reactions 1 below, write the pk
Exam 2 - Key Unless otherwise indicated, clearly indicate stereochemistry. You may use molecular models. 1. (16 pts) For each of the following hypothetical equilibration reactions 1 below, write the pk
MgBr 17 H 16 N 2 O 3 HO N. no partial. 4 each; can be either order -2 if steps unnumbered 1) CH 3 OH/TsOH. 1) NaH 2) CH 3 I
 I. (43 points) ame Page 1.05.215EP1 omplete the following reaction schemes as necessary. Sequential experimental steps should be numbered appropriately! omplete structures should be shown. (a) J 2005 70
I. (43 points) ame Page 1.05.215EP1 omplete the following reaction schemes as necessary. Sequential experimental steps should be numbered appropriately! omplete structures should be shown. (a) J 2005 70
Tips for taking exams in 852
 Comprehensive Tactical Methods in rganic Synthesis W. D. Wulff 1) Know the relative reactivity of carbonyl compounds Tips for taking exams in 852 Cl > > ' > > ' N2 eg: 'Mg Et ' 1equiv. 1equiv. ' ' Et 50%
Comprehensive Tactical Methods in rganic Synthesis W. D. Wulff 1) Know the relative reactivity of carbonyl compounds Tips for taking exams in 852 Cl > > ' > > ' N2 eg: 'Mg Et ' 1equiv. 1equiv. ' ' Et 50%
Aldehydes and Ketones : Aldol Reactions
 Aldehydes and Ketones : Aldol Reactions The Acidity of the a Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons a to carbonyls are unusually acidic The resulting anion is stabilized by
Aldehydes and Ketones : Aldol Reactions The Acidity of the a Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons a to carbonyls are unusually acidic The resulting anion is stabilized by
Dr. Steven Pedersen February 27, Chemistry 3B. Midterm 1
 r. Steven Pedersen February 7, 017 Student name: ASWER KEY Chemistry 3B Midterm 1 Student I: (Also include your SI in the top left corner of each page) Student signature: Problem 1 Problem Problem 3 Problem
r. Steven Pedersen February 7, 017 Student name: ASWER KEY Chemistry 3B Midterm 1 Student I: (Also include your SI in the top left corner of each page) Student signature: Problem 1 Problem Problem 3 Problem
MODULE CODE: CHEM10003 ORGANIC CHEMISTRY 4 EXAM
 School of Science and Sport Physical Sciences, Chemistry Paisley Campus Session 2014-15 Trimester 2 MDULE CDE: CHEM10003 RGANIC CHEMISTRY 4 EXAM Date: 15th May 2015 Time: 10.00am 12.00pm Attempt THREE
School of Science and Sport Physical Sciences, Chemistry Paisley Campus Session 2014-15 Trimester 2 MDULE CDE: CHEM10003 RGANIC CHEMISTRY 4 EXAM Date: 15th May 2015 Time: 10.00am 12.00pm Attempt THREE
CuI CuI eage lic R tal ome rgan gbr ommon
 Common rganometallic eagents Li Et 2 Li Mg Et 2 Li alkyllithium rignard Mg Mg Li Zn TF ZnCl 2 TF dialkylzinc Zn 2 2 Zn Li CuI TF ganocuprate CuI 2 2 CuI common electrophile pairings ' Cl ' '' ' ' ' ' '
Common rganometallic eagents Li Et 2 Li Mg Et 2 Li alkyllithium rignard Mg Mg Li Zn TF ZnCl 2 TF dialkylzinc Zn 2 2 Zn Li CuI TF ganocuprate CuI 2 2 CuI common electrophile pairings ' Cl ' '' ' ' ' ' '
Strategies for Stereocontrolled Synthesis
 Chemistry. Synthetic rganic Chemistry II Lecture 3 March, 2007 Rick L. Danheiser Massachusetts Institute of Technology! Thermodynamic Control Strategies! Kinetic Control Strategies! Strategies for the
Chemistry. Synthetic rganic Chemistry II Lecture 3 March, 2007 Rick L. Danheiser Massachusetts Institute of Technology! Thermodynamic Control Strategies! Kinetic Control Strategies! Strategies for the
Chemistry Organic Chemistry II: Reactivity and Synthesis ANSWERS FOR FINAL EXAM Winter 2004
 ASWER KEY Page 1 of 18 Chemistry 2.222 rganic Chemistry II: Reactivity and Synthesis ASWERS FR FIAL EXAM Winter 2004 Paper umber 631 Thursday April 22, 2004 1:30 4:30 pm University Centre Rm 210-224 Students
ASWER KEY Page 1 of 18 Chemistry 2.222 rganic Chemistry II: Reactivity and Synthesis ASWERS FR FIAL EXAM Winter 2004 Paper umber 631 Thursday April 22, 2004 1:30 4:30 pm University Centre Rm 210-224 Students
VINBLASTINE. H MeO 2 C MeO. OAc. CO 2 Me. Me H
 VIBLATIE 2 C 1 C 2 Ac a 3: catharanthine C 2 Ac C 2 2: ( )-vindoline xidation 5' 2 C 3' 16' 20' Ac C 2 1: (+)-vinblastine b 4 C 2 TFAA, -50 C Polonovski fragmentation 6' 5' 16' C 2 5 TFA 4' 3' 15' 16'
VIBLATIE 2 C 1 C 2 Ac a 3: catharanthine C 2 Ac C 2 2: ( )-vindoline xidation 5' 2 C 3' 16' 20' Ac C 2 1: (+)-vinblastine b 4 C 2 TFAA, -50 C Polonovski fragmentation 6' 5' 16' C 2 5 TFA 4' 3' 15' 16'
Structure at the isoelectric point: The predominant form of this tripeptide at ph 1 has a net charge of: (circle one)
 ame Key 21 F07-Final exam Page 2 I. (1 points) Using the pka information given on page 12, match the following isoelectric point (pi) values (3.08, 6.00, and 10.76) to their corresponding amino acids by
ame Key 21 F07-Final exam Page 2 I. (1 points) Using the pka information given on page 12, match the following isoelectric point (pi) values (3.08, 6.00, and 10.76) to their corresponding amino acids by
Chem 342 Organic Chemistry II Final Exam 13 May 2009
 hem 342 rganic hemistry II Final Exam 13 May 2009 KEY Please read through each question carefully and answer in the spaces provided. A good strategy is to go through the test and answer all the questions
hem 342 rganic hemistry II Final Exam 13 May 2009 KEY Please read through each question carefully and answer in the spaces provided. A good strategy is to go through the test and answer all the questions
Chem 14D Final Exam. Spring 2010 / Prof. Neil Garg. Thursday, June 10, :30 AM 2:30 PM
 General Instructions: Chem 14D Final Exam Spring 2010 / Prof. eil Garg This is a standard closed-note exam. Thursday, June 10, 2010 11:30 AM 2:30 PM Chem 14D Final Exam P.1 Please read each question carefully
General Instructions: Chem 14D Final Exam Spring 2010 / Prof. eil Garg This is a standard closed-note exam. Thursday, June 10, 2010 11:30 AM 2:30 PM Chem 14D Final Exam P.1 Please read each question carefully
2.0 h; 240 points please print clearly Dr. Kathleen Nolta Signature. Problem Points Score GSI I 42 II 35 III 36 IV 46 V 42 VI 39 Total 240
 hemistry 10 irst ame inal Examination Last ame.0 h; 0 points please print clearly Dr. Kathleen olta ignature UM ID # Problem Points core GI I II 5 III IV V VI 9 Total 0 Precision in drawing counts. heck
hemistry 10 irst ame inal Examination Last ame.0 h; 0 points please print clearly Dr. Kathleen olta ignature UM ID # Problem Points core GI I II 5 III IV V VI 9 Total 0 Precision in drawing counts. heck
Renaud Group Exercise Set
 Renaud Group Exercise Set Prepared by ick Tappin 08/07/16 Spectroscopy 1. Deduce the structures for compounds A, B, and C C 6 10 3 A IR: 1745 and 1720 cm -1 13 C-MR: δ 208, 172, 51, 37, 32, and 27 ab 4
Renaud Group Exercise Set Prepared by ick Tappin 08/07/16 Spectroscopy 1. Deduce the structures for compounds A, B, and C C 6 10 3 A IR: 1745 and 1720 cm -1 13 C-MR: δ 208, 172, 51, 37, 32, and 27 ab 4
Radical Reactions. Radical Stability!!! bond dissociation energies X Y X + Y. bond BDE (kcal/mol) bond BDE (kcal/mol) CH 3 CH 3 CH 2 95 O H R 2 C H
 adical eactions adical Stability!!! bond dissociation energies X Y X Y bond BDE (kcal/mol) bond BDE (kcal/mol) C 3 104 108 C 3 C 2 98 110 95 2 C 102 (-) 93 (C-) 92 C 3 C 3 36 89 85 C 3 C 3 80 adical eactions
adical eactions adical Stability!!! bond dissociation energies X Y X Y bond BDE (kcal/mol) bond BDE (kcal/mol) C 3 104 108 C 3 C 2 98 110 95 2 C 102 (-) 93 (C-) 92 C 3 C 3 36 89 85 C 3 C 3 80 adical eactions
THE UNIVERSITY OF CALGARY FACULTY OF SCIENCE FINAL EXAMINATION CHEMISTRY 353
 TE UNIVERSITY F CALGARY FACULTY F SCIENCE FINAL EXAMINATIN CEMISTRY 353 April 29 th, 2002 Time: 3 ours PLEASE WRITE YUR NAME, STUDENT I.D. NUMBER AND SECTIN NUMBER (01 for MWF lectures and 02 for TR lectures)
TE UNIVERSITY F CALGARY FACULTY F SCIENCE FINAL EXAMINATIN CEMISTRY 353 April 29 th, 2002 Time: 3 ours PLEASE WRITE YUR NAME, STUDENT I.D. NUMBER AND SECTIN NUMBER (01 for MWF lectures and 02 for TR lectures)
Organic Chemistry I (Chem340), Spring Final Exam
 rganic Chemistry I (Chem340), pring 2005 Final Exam This is a closed-book exam. No aid is to be given to or received from another person. Model set and calculator may be used, but cannot be shared. Please
rganic Chemistry I (Chem340), pring 2005 Final Exam This is a closed-book exam. No aid is to be given to or received from another person. Model set and calculator may be used, but cannot be shared. Please
Partial Periodic Table
 Easily Legible Printed Name: CHEM 3351 (100), Fall 2016 Professor Walba Second Hour Exam October 18, 2016 CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither
Easily Legible Printed Name: CHEM 3351 (100), Fall 2016 Professor Walba Second Hour Exam October 18, 2016 CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither
Note: You must have your answers written in pen if you want a regrade!!!!
 NAME (Print): SIGNATURE: Chemistry 310N Dr. Brent Iverson 2nd Midterm March 27, 2008 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long,
NAME (Print): SIGNATURE: Chemistry 310N Dr. Brent Iverson 2nd Midterm March 27, 2008 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long,
CHEMISTRY 332 FALL 08 EXAM II October 22-23, 2008
 First Three Letters of Last Name NAME Network ID CHEMISTRY 332 FALL 08 EXAM II October 22-23, 2008 The following materials are permissible during the exam: molecular model kits, course notes (printed,
First Three Letters of Last Name NAME Network ID CHEMISTRY 332 FALL 08 EXAM II October 22-23, 2008 The following materials are permissible during the exam: molecular model kits, course notes (printed,
Midterm #2 Chem 3A - Fall 2013 Nov. 12, :00 8:45 pm. Name SID
 Midterm #2 Chem 3A - Fall 2013 Nov. 12, 2013 7:00 8:45 pm Name SID Including the title page, there should be 6 total questions spread over 8 pages (printed on both sides). Please provide all answers in
Midterm #2 Chem 3A - Fall 2013 Nov. 12, 2013 7:00 8:45 pm Name SID Including the title page, there should be 6 total questions spread over 8 pages (printed on both sides). Please provide all answers in
CHAPTER 24 HW: CARBONYL CONDENSATIONS
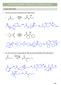 CAPTER 24 W: CARBNYL CNDENSATINS ALDL REACTIN 1. Draw the curved arrow mechanism for each aldol reaction. Na 2 product Na Et prod. 2. Give the curved arrow mechanism for this aldol reaction (and dehydration
CAPTER 24 W: CARBNYL CNDENSATINS ALDL REACTIN 1. Draw the curved arrow mechanism for each aldol reaction. Na 2 product Na Et prod. 2. Give the curved arrow mechanism for this aldol reaction (and dehydration
Section Practice Exam II Solutions
 Paul Bracher Chem 30 Section 7 Section Practice Exam II s Whether problems old r problems new, You d better practice, r you ll fail exam II. -- Anonymous TF Problem 1 a) Rank the following series of electrophiles
Paul Bracher Chem 30 Section 7 Section Practice Exam II s Whether problems old r problems new, You d better practice, r you ll fail exam II. -- Anonymous TF Problem 1 a) Rank the following series of electrophiles
CHEMISTRY 335, A01 SYNTHETIC METHODS IN ORGANIC CHEMISTRY FINAL EXAM APRIL 22, 2009 NAME: KEY STUDENT ID:
 UIVESITY VICTIA Chem 335 A01: Page 1 CEMISTY 335, A01 SYTETIC METDS I GAIC CEMISTY IAL EXAM APIL 22, 2009 AME: KEY STUDET ID: ISTUCT: ASE TTAL MAKS = 70 DUATI: 3 US (9:00 AM 12:00 PM) TIS EXAMIATI PAPE
UIVESITY VICTIA Chem 335 A01: Page 1 CEMISTY 335, A01 SYTETIC METDS I GAIC CEMISTY IAL EXAM APIL 22, 2009 AME: KEY STUDET ID: ISTUCT: ASE TTAL MAKS = 70 DUATI: 3 US (9:00 AM 12:00 PM) TIS EXAMIATI PAPE
First 2-Hour Exam. By printing your name below, you pledge that
 Chem 3331 Fall 2004 Exam # 1 1 Name First 2-our Exam By printing your name below, you pledge that "n my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized
Chem 3331 Fall 2004 Exam # 1 1 Name First 2-our Exam By printing your name below, you pledge that "n my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized
FOURTH EXAMINATION. (1)..20 pts... (2)..24 pts... (3)..18 pts... (4)..12 pts... (5)..12 pts... (6).. 6 pts... (7).. 8 pts... Bonus 4 pts...
 Name: CHEM 331 FURTH EXAMINATIN All answers should be written on the exam in the spaces provided. Clearly indicate your answers in the spaces provided; if I have to guess as to what or where your answer
Name: CHEM 331 FURTH EXAMINATIN All answers should be written on the exam in the spaces provided. Clearly indicate your answers in the spaces provided; if I have to guess as to what or where your answer
c. Oxidizing agent shown here oxidizes 2º alcohols to ketones and 1º alcohols to carboxylic acids. 3º alcohols DO NOT REACT.
 Exam 1 (Ch 17 and Review of CEM 331) Answer Key: 1. ne-step Questions: You need to know reagents for reagent arrows and to be able to draw products. I know a lot of them seem to look alike its your job
Exam 1 (Ch 17 and Review of CEM 331) Answer Key: 1. ne-step Questions: You need to know reagents for reagent arrows and to be able to draw products. I know a lot of them seem to look alike its your job
