A coupled field and modelling study on aerosol - cloud interaction
|
|
|
- Aileen Patrick
- 5 years ago
- Views:
Transcription
1 A coupled field and modelling study on aerosol - cloud interaction (AFO 2000 projects FEBUKO (FKZ 07ATF01) and MODMEP (FKZ 07ATF40)) Hartmut Herrmann*, Ralf Wolke* and the FEBUKO/MODMEP-team** *: Leibniz-Institut für Troposphärenforschung, Permoserstr. 15, D Leipzig herrmann@tropos.de; wolke@tropos.de; **:Leibniz-Institut für Troposphärenforschung, Leipzig; Brandenburgische Technische Universität Cottbus; Max-Planck-Institut für Meteorologie, Hamburg; Johann Wolfgang Goethe-Universität Frankfurt and Technische Universität Darmstadt Introduction The FEBUKO and MODMEP projects within AFO 2000 will improve the understanding of tropospheric multiphase processes and especially the interaction of aerosols and clouds with an emphasis on organic particle constituents. Field experiments on aerosol and cloud chemistry and physics, model development and model application are combined to investigate chemical and physical transformation of particles within a cloud passage. A complex experimental data set was provided by two field campaigns. In MODMEP the development is directed towards a cloud module which combines a complex multiphase chemistry with detailed microphysics. The description of both is given with high size resolution of the drop spectrum. The influence of simplifications within single components and the kind of their coupling on the simulation results is investigated for different tropospheric situations. Furthermore, techniques are provided and tested which allow an effective implementation of multiphase processes in multidimensional cloud and chemistrytransport models. Figure 1: Investigation area The FEBUKO experiments at the three research sites in the Thüringer Wald (Goldlauter, Schmücke and Gehlberg) were carried out in the autumn of 2001 and 2002, respectively, to characterize the aerosol and cloud water with respect to their chemical composition and their physical properties (Figure 1). The speciation of organic components was one of the most important tasks for the different size classes of the particle phase and in cloud water. The air masses encountered in the experimental region are of anthropogenically influenced origin and have then been exposed to biogenic emissions on their way from the Rhein-Main area to the Thüringer Wald. Experimental At the two valley measurement sites (Goldlauter (GL) upwind site, 605 m asl, and Gehlberg (GB) downwind site, 732 m asl) and at the mountain site (Schmücke (SM), 937 m asl) a complex set of meteorological parameters, trace gas components, cloud parameters and particle characteristics was determined. The sampling of particles using filter samplers and impactors for the chemical constitution measurements was carried out at the valley stations together with measurements of particle number concentrations, size distributions and hygroscopic properties. On top of a 20 m research tower a variety of different cloud water samplers were operated on the Schmücke. The interstitial aerosol and the cloud droplet residues were collected and physically characterized by mean of a counterflow virtual impactor (CVI) and an inlet to interstitial particles (INT) as well.
2 The flow connection between the three stations is an important prerequisite for the whole experiment. Hence, special tracer experiments, measurements of number and size distribution of particles as well as trace gas concentrations were performed. In addition, the synoptic experimental conditions were carefully investigated and flow simulations for the complex terrain area are currently in progress. Results and discussion In both campaigns in 2001 and 2002 a total set of 14 cloud events was identified for intensive measurements. The most important conditions of (i) a connected flow, (ii) an orographic cloud and (iii) good data coverage were best fulfilled during three events. The results within the present contribution refer to a 15 hours measurement period on 26/27-October 2001 (22:00 to 13:00 UTC). On the modelling side, at the time of writing of this contribution simulations were made with an air parcel travelling from Goldlauter to the top of the mountain into the orographic cloud. The goal of the simulations was to investigate the multiphase chemistry occurring in orographic clouds and to understand the interaction of particle phase and gas phase of atmospheric trace constituents due to phase transfer and chemical transformation. Simulation results have been compared with cloud water measurements in order to interpret the experimental data and for validation of the multiphase chemistry model. Physical aerosol characterization Particle number concentration Particle number size distributions were measured in GL and GB using a Differential Mobility Particle Sizer (DMPS) system. These measurements are complementary to the interstitial (INT) and Counter Flow Virtual Impactor (CVI) number size distribution measurements on the mountain station Schmücke (SM). In Figure 2, mean number size distributions at all sites are plotted for 27 th of October 2001, 0:00 to 13:00 UTC. The black and green curves show the number size distribution measured in GL and GB while the red one represents the interstitial aerosol at the top of Schmücke in cloud. The peak number concentrations of the Aitken modes at GB and Schmücke are adjusted by a factor of 1.13 to the peak concentration at GL to recognize also small deviations. By this adjustment, the rising edge in the small particle size range Figure 2: Particle number size distribution at Goldlauter (GL), between 25 and 60 nm of both Gehlberg (GB) and Schmücke (SM) on Oct. 27, The black and curves are becoming identical not green curves show the mean size distribution at GL and GB, only qualitatively but also respectively, whereas the red and light blue curves represent the quantitatively. The mean number interstitial (INT) and CVI size distributions. The size distributions of size distributions between GL and SM INT and GB have been normalized the level of GL by a factor of Finally the dark blue curve shows the deviation between the GL GB do not show a significant and SM INT size distribution. difference. Mean number size distributions of the droplet residues (light blue) and interstitial particles (red) are shown. They have been measured in-cloud at SM during the cloud event in the size range between 25 and 900 nm by two DMPS systems attached to the CVI and INT inlet system. Particles larger than a certain size are expected to become activated, which is demonstrated by the deviation of both distributions above 70 nm. Figure 2 shows this difference (dark blue) and represents the indirect
3 determination of the CCN number size distribution. This curve agrees well with the direct CVI measurement, which confirms that the phase partitioning due to the droplet activation is successfully observed during the connected flow between GL and SM. Chemical aerosol characterization In order to gain a deeper insight into budgets and conversions of specific organic compounds in tropospheric clouds and to have a realistic data set for the model initialisation, one of the aims of FEBUKO is a comprehensive organic speciation at all three measuring sites in addition to standard particle and cloudwater chemical analysis. Organics are usually trace compounds in different atmospheric phases which make their determination a challenging task. A variety of sampling devices was applied, some of them exclusively for organic speciation. With impactors, filters and spray collectors aerosol particles were sampled. Sorbent-filled cartridges were used for gas-phase measurements and four different types of cloud water samplers collected the liquid phase on the mountain. Most of the instruments give time-resolved information about organic concentration levels with a sampling time interval of 2 hours. Complementary to this the impactors allow insights into the size distribution of condensed organic material before and after cloud processing. After the campaign the samples were analysed using analytical equipment like gas chromatography/mass spectrometry (GC/MS), high performance liquid chromatography/mass spectrometry (HPLC/MS) or capillary electrophoresis (CE). With these field measurement equipment it was possible to create an extensive pool of data for dozens of organic substances in their respective most relevant atmospheric phases. Aliphatic and aromatic aldehydes and ketones as well as other volatile organic compounds (VOCs) like short-chain alkanes, alkenes, and volatile aromatics could be identified and quantified in the gas phase on a time-resolved basis. Depending on their polarity some of them can be scavenged by the cloud and therefore they were measured also in the liquid phase. The possible photooxidation products of carbonyls and VOCs such as higher oxygenated species like monocarboxylic acids in the gas phase and in cloud water and dicarboxylic acids in the particle phase and in cloud water were analysed in a time-resolved manner. Size distributions before and after the cloud passage as well as the corresponding liquid-phase concentrations are as well available for lower vapour pressure compounds such as long-chain alkanes, alcohols, sugars, fatty acids, dicarboxylic acids, and several biogenic compounds such as pinonaldehyde, squalene, and others. Bulk concentrations from filters for interstitial particles allow us to calculate directly the in-cloud scavenging for some of these compounds. More details of the organic analysis can be found under The complex data set obtained in the FEBUKO field experiments is of huge importance for the MODMEP model initialisation and validation. Besides, we can interpret it by comparing, correlating and balancing concentrations of selected species for different atmospheric phases, different times and different particle sizes. In combination with the model results we may now be able to achieve a better understanding of phase partitioning and chemical multiphase processes of tropospheric organic material. Model initialization The model developed within the course of the MODMEP project is initialized by means of the particle composition based on measurements with a five stage Berner impactor at the upwind site GL as described before. In the case of the cloud event on the 27 th of October 2001, most of the mass was found on the third impactor stage, which collects aerosol particles with a diameter between 0.42 µm and 1.2 µm. Nitrate, sulfate, ammonium, organic and elementary carbon are the main components of the aerosol. In order to correct for losses encountered by the Berner impactor in the case of chloride and nitrate, data obtained with the Steam Jet sampler were used and distributed over the impactor stages according to the relative size distribution of each of these species derived with the Berner impactor. The alkaline and earth alkaline metal initial concentrations, i.e. sodium, magnesium, calcium and potassium, are taken from the Berner impactor. Other metal ion concentrations, i.e. manganese, iron, copper, aluminium and zinc, are initialised according to measurements made with a High- Volume-Sierra-Anderson (PM 10 ) sampler. These measure total contents are distributed over the
4 impactor stages as the mass determined by the impactors. The initialization of the dicarboxylic acids is based on impactor and spray collector measurements. To calculate the water mass of Goldlauter, aerosol growth factors were used. The used growth factors were measured during the ACE2 campaign, and are in a good agreement with the measured growth factors by a HTDMA analyzer within the FEBUKO field campaign. In the gas phase NO, NO 2, O 3, and SO 2 were measured every 5 seconds with commercial instruments. For the measurement of HNO 2 and HNO 3 the wet effluent diffusion denuder technique was used. The measured organic species were implemented as stand-alone species or were lumped together into groups as used in the regional atmospheric chemistry mechanism RACM. Implemented stand-alone species are: formaldehyde, ethane, ethene, formic acid, glyoxal, methylglyoxal, and isoprene. Measured lumped gas phase species considered are aldehydes, ketones, unsaturated monoaldehydes, acetic acid and higher acids. For example 19 measured aldehydes were summed up to the lumped species ALD. For some unmeasured species initial concentrations were adapted from the CAPRAM standard scenario urban due to a reasonable agreement between measurements and the abovementioned scenario. Tracer experiments were carried out within the FEBUKO field campaign in order to verify the existence of a connected flow and to determine the transport time between the three sampling stations. Based on the experiments a realistic transport time between Goldlauter and Schmücke of minutes resulted, depending on meteorological conditions. For the initial wind speed, data from the measurement site of the German weather service station located in Meiningen was used. For the cloud event on the 27 th of October 2001 at 9.00 UTC an initial wind speed of 4 m s -1 was applied, which led to a simulation time of about 11 minutes from the downwind site to the summit. For the simulations described here, a constant size-independent deposition velocity of 10-3 s -1 was considered. Coupled time-integration of detailed microphysics and complex multiphase chemistry in a size resolved cloud box model To better understand the interaction, effects and evolution of the different physico-chemical processes taking place in the atmosphere their modelling requires a detailed description of all transformations with equal rigor. The most recently available models focus either on detailed microphysics or complex multiphase chemistry. In the framework of the joint AFO2000-project MODMEP the air parcel model SPACCIM ( SPectral Aerosol Cloud Chemistry Interaction Model ) was developed for the description of cloud processes by coupling complex multiphase chemistry and detailed microphysics. The description of both process groups is given for a size-resolved particle/drop spectrum. Either the movement of the air parcel can follow a predefined trajectory (e.g., simulated by the regional model system LM-MUSCAT) or the vertical velocity is calculated for a buoyant parcel in prescribed environmental conditions. Entrainment and detrainment processes are included in a parametrized form. The model allows a detailed description of the transformation of gases and particles shortly before cloud formation, during the cloud life time and shortly after cloud evaporation. Treatment of multiphase processes The governing equations are the mass balance equations for a size-resolved particle/drop spectrum in a box model. The spectrum is described using a sectional approach. The prognostic microphysical variables for each bin are water mass, total and soluble particulate mass as well as particle number. The model considers growth/shrinking and impaction of aerosol particles as well as nucleation, condensation/evaporation and collision-coalescence of drops as most important physical processes. One important feature of the model is the description of the water phase transfer feedback on water vapour and air temperature (latent heat release) which is done in an iterative way to avoid overestimation of super-saturation (and, therefore, nucleation) especially at cloud base. The model accounts for the fact that larger aerosol particles do not have enough time to reach their equilibrium size at high relative humidities near 100% due to the faster changing environment. The spectral multiphase chemistry model considers two types of chemical reactions: Forward reaction types in gas and aqueous phase including photolysis and temperature dependent reactions,
5 Equilibrium reaction types, considered as forward and backward reaction. The phase transfer is parametrized by the Schwartz approach with the hypothesis of well mixed droplets. The applied mechanism (including phase transfer and kinetic constants) is given as an input file. Therefore, a high flexibility concerning changes in the chemical mechanism is ensured, and the replacement of the entire reaction system is also possible (Wolke et al., 2001). The performance of the model was shown for simple chemical test mechanisms (with only inorganic chemistry) as well as for very complex mechanisms of the CAPRAM family particle phase organic chemistry in a varying degree of complexity dependent of the CAPRAM version. Coupling scheme and time integration The model variables can be grouped into microphysical variables, chemical variables and mass fluxes between different particle/droplet classes caused by microphysical exchange processes (e.g., by aggregation, break up, condensation). These mass fluxes are schematically shown in Figure 3. When two particles coagulate, for instance, their masses are added to the resulting particle class assuming internally mixed aerosol in each class. The feedback of changes in the chemical composition by gas scavenging and chemical reactions on microphysical processes (e.g., water condensation growth rates via changes in surface tension and the Raoult term) is also implemented. The activation of droplets is explicitly described in the microphysical model. Figure 3: Schematic representation of mass transport between several sections due to microphysical processes. The red hatches part represents the mass fraction of one selected species in the corresponding section. The time integration scheme for solving the multiphase problem is based on the multistep implicit method (Backward Differentiation Formula) widely used for the integration of stiff differential equations. In our approach, the resulting linear sparse systems are solved exploiting the properties of the Jacobian (sparsity, block structure, different types of coupling). An approximate matrix factorization to decouple the microphysical transport terms and the other parts of the Jacobian is possible. This is seen as an operator splitting at the linear algebra level and yields a computing gain (Wolke and Knoth, 2002). In addition to the higher order BDF scheme, the non-autonomous second order Rosenbrock method is implemented, presenting a low computing effort and showing a satisfactory accuracy. The model SPACCIM is evaluated with data from the FEBUKO field campaign and other case studies from literature. It offers several parameter setups allowing sensitivity studies to reach optimal runs for each studied case. Microphysical model results Figure 4 shows the cloud liquid water content (LWC, left) and drop number concentration (right) on Schmücke for the event 26/27 th of October The model calculates the LWC adiabatically which leads to an overestimation of about 25% compared to the findings of the three different measurements carried out. The time dependent structures are represented rather well. In contrast to the LWC drop number depends on transport time from Goldlauter to Schmücke. Two model cases (red line: slow transport, green line: fast transport) are compared with measurement data. Drop number highly depends on the vertical velocity and, therefore, super-saturation especially at cloud base. The faster the transport and the higher the vertical velocity, the higher is the super-saturation and the more drops are activated.
6 Figure 4: Cloud liquid water content (LWC, left) and drop number concentration (right) on Schmücke for /27 High LWC corresponds to a low cloud base and a rather gentle terrain (compare Figure 1) connected with weak updrafts leading to the lowest drop numbers in the model (2-4 UTC, 7-9 UTC). The opposite is true for low LWC: steep orography with high velocities leads to (unrealistically) higher drop numbers in the model (4 UTC, 6 UTC, UTC). These strong variations in the modelled drop numbers are not reflected by the experimental data where drop numbers are rather constant between 400 and 600 cm -3. Generally, drop numbers fit very well with the experimental findings for LWCs above 0.4 g m -3, corresponding to a rather low cloud base. For lower LWCs the model seems to overestimate the vertical velocity and, therefore, super-saturation and activation. Simulations with the coupled chemistry microphysics model SPACCIM and CAPRAM By means of SPACCIM, simulations of the hill cap cloud passage experiment taken place on 27 th of October 2001 were carried out. Calculations with an air parcel following a trajectory from the upwind site to the summit station Schmücke were performed. The simulation results have been compared to experimental data from the summit station. The applied explicit aqueous phase radical mechanisms consist of CAPRAM 2.3 (Herrmann et al., 2000) and CAPRAM 2.4 (MODAC-mechanism, Ervens et al., 2003). The gas phase chemistry is described by the regional atmospheric mechanism RACM (Stockwell et al., 1997). Phase transfer processes are treated by means of the resistance model of Schwartz considering Henry s equilibrium, gas phase diffusion and mass accommodation. For the simulations a highly resolved particle spectrum is considered. A total number of 64 size bins ( µm < d < µm) is considered, where multiphase chemistry occurs in droplets where the LWC exceeds 10-9 g m -3. In the near future a feedback from the chemistry to the microphysics will also be implemented. For the initial wind speed data from the measurement site of the German weather service station located in Meiningen was used. For the cloud event on 27 th of October 2001 at 9:00 UTC an initial wind speed of 4 m s -1 was applied, which led to a simulation time of about 11 minutes from the upwind station to the summit. For the simulations a constant deposition velocity of 10-3 s -1 was considered. Simulation results For the respective cloud event on 27 th of October 2001 the exact time for the simulations was fixed at 9:00 UTC due to a good agreement between the microphysical model and the measurement with regards to the total LWC and number concentration. At the upwind station a LWC of about 10-4 g m -3 exists. At the summit station the total LWC will reach a value of g m -3. At the end of the simulation, at the top of the mountain most of the LWC will be in the droplets with a diameter
7 Figure 5: Size and time dependent evolution of the LWC between the luff and the summit station, comparison between measurement and simulation result at Schmücke between 8 and 16 µm. The maximum of the LWC distribution, as can be seen from Figure 5, occurs at droplets with a diameter of circa 12 µm. In Figure 5 also the LWC spectra measured by an FSSP (Forward Scattering Spectrometer Probe) is plotted. It can be seen that the measured spectrum of the orographic cloud is broader than the calculated spectra. This discrepancy could be caused by a too broad spectra produced by the FSSP a too narrow spectrum resulted from the microphysical model or a combination of both. Activation of the aerosol particles will occur after a simulation time of about 300 s. In Figure 6 the size-resolved sulfate concentration is represented. The concentration profile represents the sum of sulfate concentration over the size bins with a diameter bigger than 3.5 µm. At the summit the concentration was measured with a cloud water bulk collector. The collector had sampled cloud droplets with a diameter bigger than 3.5 µm. At the summit the calculated SO 4 2- concentration reached a value of mol l -1, HSO 4 - a value of mol l -1 and H 2 SO 4 a value of mol l -1. As can be seen from the plot there is a good agreement between the calculated and measured value. As expected, due to uptake of liquid water the concentration decreases in time. At the summit, in the small droplets with less water content there will be a high sulfate concentration. In the small interstitial aerosol particles the sulfate concentration reaches values of about mol l -1. In the size range where most of the liquid water can be found a sulfate concentration of circa mol l -1 occurs. Analyzing the sources of sulfate, it can be said that sulfur oxidation proceeds mainly via the oxidation of bisulfite by hydrogen peroxide, i.e. - - HSO 3 + H 2 O 2 HSO 4 + H 2 O (R-1) Before actually reaching cloud conditions or supersaturation and hence during the activation 2- Figure 6: Size-resolved SO 4 concentration profile, comparison between measurement and simulation result at the summit, the red point representing the experimentally determined value process, in the small droplets sulfur oxidation via aqueous phase radical chemistry involving transition metal ions plays a role and contributes to S(IV) to S(VI) conversion. At the final stage of this complex radical pathway sulfate is formed by the oxidation of Fe 2+ by the so-called sulfate radical anion SO 4 - : SO Fe 2+ SO Fe 3+ (R-2) Sulfate production will be controlled by the SO 2 uptake from the gas phase, process significantly influenced by the ph value. The model exactly reproduces the measured H + concentration of mol l -1 at Schmücke. A further detailed analysis concerning the transformation of inorganic species, e.g. nitrate, chloride, the speciation of transition metal ions, e.g. iron, copper, manganese, and the aqueous oxidation of organic compounds, e.g. acetaldehyde, glyoxal, oxalate, is ongoing.
8 Summary and outlook A coupled version of detailed microphysics and a complex multiphase chemistry in a box model is realized, allowing a high flexibility concerning the use of chemical mechanisms and the simulation of case studies like the FEBUKO field experiment. In the simulations of the latter experiments using SPACCIM and CAPRAM for some species there is a quite good agreement between the measurements and model results, with deviations below a factor of two, e.g., NO 3 -, SO 4 2-, K +, HCOOH, CH 3 COOH. For other species, e.g. Fe(II)-Fe(III), HCHO, bigger differences were encountered. It has to be noted that some of the negative deviations are due to the fact that for the current model results the input aerosol size distribution is only considered for up to Dp = 900 nm. A course mode treatment is under development and may lead to better agreement for species such as most of the cations. For a better description of organic chemistry, simulations with the CAPRAM 2.5 mechanism will be conducted. The CAPRAM 2.5 mechanism consisting of the CAPRAM 2.4 (MODAC mechanism) reduced, coupled to the CAPRAM organic extension considers the oxidation of organic compounds with up to four carbon atoms. Also in the near future simulations with an air parcel travelling from the upwind site, passing through a hill cap cloud, and then transported to the downwind site will be carried out to better chemical modifications of the particle ensemble after a single cloud process under real field conditions. The SPACCIM model development initiated after MODMEP will continue to further refine the available model framework. The CAPRAM mechanism will be continuously updated and last, but not least, the Schmücke hill-capped cloud experimental site is ready for further focussed ground based aerosol-cloud-interaction experiments with emphasis on organics under real conditions and hence complementary to more specialized laboratory experiments. Acknowledgements The work was supported by the Bundesministerium für Bildung und Forschung (BMBF). We thank the Umweltbundesamt (UBA) and the Deutscher Wetterdienst (DWD Offenbach) for good cooperation and support. We thank all the colleagues that participated in the FEBUKO experiments and for the realization of the numerous analytical measurements: H. Bachmann, B. Gerlach, S. Haferkorn, N. Heim, A. Kappe, A. Thomas. References S. Schwartz, "Mass transport consideration pertinent to aqueous phase reactions of gases in liquid water clouds." Chemistry of Multiphase Atmospheric System. W. Jaeschke. Berlin, Springer, 1986, W. Stockwell, Kirchner, F., Kuhn, M., and Seefeld, S., 'A new mechanism for regional atmospheric chemistry modeling.' in Journal of Geophysical Research JGR, 102, 1997, H. Herrmann, Ervens, B., Jacobi, H.-W., Wolke, R., Nowacki, P., and Zellner R., 'Capram2.3: A chemical aqueous phase radical mechanism for tropospheric chemistry.' in Journal of Atmospheric Chemistry, 38, 2000, H. Herrmann: MODAC-Model Development for Tropospheric Aerosol and Cloud Chemistry. Final report to Project ENVA-CT , 2000, CEC Brussels. R. Wolke and O. Knoth, 'Time-integration of multiphase chemistry in size-resolved cloud models', in Applied Numerical Mathematics, 42, 2002, B. Ervens, G. V. Buxton, G. A. Salmon, J. Williams, M. Bydder, F. Dentener, C. George, P. Mirabel and H. Herrmann, 'CAPRAM2.4 (MODAC mechanism): An extended and condensed tropospheric aqueous phase mechanism and its application', in Journal of Geophysical Research JGR, 108, 2002, 4426.
CAPRAM MODELLING OF THE PHYSICO-CHEMICAL CLOUD PROCESSING OF TROPOSPHERIC AEROSOLS
 CAPRAM MODELLING OF THE PHYSICO-CHEMICAL CLOUD PROCESSING OF TROPOSPHERIC AEROSOLS A. Tilgner 1 *, R. Wolke 1 and H. Herrmann 1 1 Leibniz-Institut für Troposphärenforschung, Permoserstr. 15, D-04318 Leipzig,
CAPRAM MODELLING OF THE PHYSICO-CHEMICAL CLOUD PROCESSING OF TROPOSPHERIC AEROSOLS A. Tilgner 1 *, R. Wolke 1 and H. Herrmann 1 1 Leibniz-Institut für Troposphärenforschung, Permoserstr. 15, D-04318 Leipzig,
Tropospheric Aqueous Phase Chemistry Laboratory and Modelling Studies
 Tropospheric Aqueous Phase Chemistry Laboratory and Modelling Studies Hartmut Herrmann, B. Ervens, J. Hesper and F. Wicktor Institut für Troposphärenforschung Permoserstr.15, 04318 Leipzig Overview Results
Tropospheric Aqueous Phase Chemistry Laboratory and Modelling Studies Hartmut Herrmann, B. Ervens, J. Hesper and F. Wicktor Institut für Troposphärenforschung Permoserstr.15, 04318 Leipzig Overview Results
Gas, Cloudwater, and Rain Hydrogen Peroxide and Methylhydroperoxide Measurements in RICO
 Gas, Cloudwater, and Rain Hydrogen Peroxide and Methylhydroperoxide Measurements in RICO Brian G. Heikes, Center for Atmospheric Chemical Studies, Graduate School of Oceanography, University of Rhode Island
Gas, Cloudwater, and Rain Hydrogen Peroxide and Methylhydroperoxide Measurements in RICO Brian G. Heikes, Center for Atmospheric Chemical Studies, Graduate School of Oceanography, University of Rhode Island
INTERACTIONS OF AEROSOLS AND GASES WITH CLOUDS AND PRECIPITATION IN THE ONLINE-COUPLED REGIONAL CHEMISTRY-TRANSPORT MODEL COSMO-ART
 INTERACTIONS OF AEROSOLS AND GASES WITH CLOUDS AND PRECIPITATION IN THE ONLINE-COUPLED REGIONAL CHEMISTRY-TRANSPORT MODEL COSMO-ART Christoph Knote* and Dominik Brunner Laboratory for Air Pollution / Environmental
INTERACTIONS OF AEROSOLS AND GASES WITH CLOUDS AND PRECIPITATION IN THE ONLINE-COUPLED REGIONAL CHEMISTRY-TRANSPORT MODEL COSMO-ART Christoph Knote* and Dominik Brunner Laboratory for Air Pollution / Environmental
Chapter Eight: Conclusions and Future Work
 2004 PhD Thesis 202 Chapter Eight: Conclusions and Future Work 8.1 Conclusions The Aerodyne aerosol mass spectrometer is capable of providing quantitative information on the chemical composition of the
2004 PhD Thesis 202 Chapter Eight: Conclusions and Future Work 8.1 Conclusions The Aerodyne aerosol mass spectrometer is capable of providing quantitative information on the chemical composition of the
Implementation of modules for wet and dry deposition into the ECMWF Integrated Forecast System
 Implementation of modules for wet and dry deposition into the ECMWF Integrated Forecast System Johannes Flemming (ECMWF), Vincent Huijnen (KNMI) and Luke Jones (ECMWF) Deliverable D G-RG 4.6 1 Abstract
Implementation of modules for wet and dry deposition into the ECMWF Integrated Forecast System Johannes Flemming (ECMWF), Vincent Huijnen (KNMI) and Luke Jones (ECMWF) Deliverable D G-RG 4.6 1 Abstract
Influence of Biogenic VOCs on Photooxidant Formation: Simulation Experiments in EUPHORE and Comparison with Model Calculations
 Introduction Influence of Biogenic VOCs on Photooxidant Formation: Simulation Experiments in EUPHORE and Comparison with Model Calculations Fraunhofer Institut Atmosphärische Umweltforschung, IFU Kreuzeckbahnstr.
Introduction Influence of Biogenic VOCs on Photooxidant Formation: Simulation Experiments in EUPHORE and Comparison with Model Calculations Fraunhofer Institut Atmosphärische Umweltforschung, IFU Kreuzeckbahnstr.
Abstract. 1 Introduction
 The influence of cloud chemistry on the budget of photo-oxidants G. Mauersberger Brandenberg Technical University Cottbus, Rudower Chausse 5, D-12489 Berlin, Germany Abstract The RADM II gas-phase system
The influence of cloud chemistry on the budget of photo-oxidants G. Mauersberger Brandenberg Technical University Cottbus, Rudower Chausse 5, D-12489 Berlin, Germany Abstract The RADM II gas-phase system
PRECIPITATION PROCESSES
 PRECIPITATION PROCESSES Loknath Adhikari This summary deals with the mechanisms of warm rain processes and tries to summarize the factors affecting the rapid growth of hydrometeors in clouds from (sub)
PRECIPITATION PROCESSES Loknath Adhikari This summary deals with the mechanisms of warm rain processes and tries to summarize the factors affecting the rapid growth of hydrometeors in clouds from (sub)
CHAPTER 8. AEROSOLS 8.1 SOURCES AND SINKS OF AEROSOLS
 1 CHAPTER 8 AEROSOLS Aerosols in the atmosphere have several important environmental effects They are a respiratory health hazard at the high concentrations found in urban environments They scatter and
1 CHAPTER 8 AEROSOLS Aerosols in the atmosphere have several important environmental effects They are a respiratory health hazard at the high concentrations found in urban environments They scatter and
ABSTRACT INTRODUCTION
 The role of clouds in transformation and removal of air pollutants D. Moller, G. Mauersberger Department for Air Chemistry, Fraunhofer Institut for Atmospheric Environmental Research, D-1199 Berlin, Germany
The role of clouds in transformation and removal of air pollutants D. Moller, G. Mauersberger Department for Air Chemistry, Fraunhofer Institut for Atmospheric Environmental Research, D-1199 Berlin, Germany
Review of the SAPRC-16 Chemical Mechanism and Comparison with the Regional Atmospheric Chemistry Mechanism, Version-2
 VOC O 3 NO NO NO 2 O 3 NO 2 Review of the SAPRC-16 Chemical Mechanism and Comparison with the Regional Atmospheric Chemistry Mechanism, Version-2 NO 2 William R. Stockwell a, Emily Saunders b, Rosa Fitzgerald
VOC O 3 NO NO NO 2 O 3 NO 2 Review of the SAPRC-16 Chemical Mechanism and Comparison with the Regional Atmospheric Chemistry Mechanism, Version-2 NO 2 William R. Stockwell a, Emily Saunders b, Rosa Fitzgerald
Supplement of Modeling organic aerosol composition at the puy de Dôme mountain (France) for two contrasted air masses with the WRF-Chem model
 Supplement of Atmos. Chem. Phys. Discuss., 1, 19 1, 01 http://www.atmos-chem-phys-discuss.net/1/19/01/ doi:10.19/acpd-1-19-01-supplement Author(s) 01. CC Attribution.0 License. Supplement of Modeling organic
Supplement of Atmos. Chem. Phys. Discuss., 1, 19 1, 01 http://www.atmos-chem-phys-discuss.net/1/19/01/ doi:10.19/acpd-1-19-01-supplement Author(s) 01. CC Attribution.0 License. Supplement of Modeling organic
Lab 4 Major Anions In Atmospheric Aerosol Particles
 Georgia Institute of Technology School of Earth and Atmospheric Sciences EAS 4641 Spring 2008 Lab 4 Major Anions In Atmospheric Aerosol Particles Purpose of Lab 4: This experiment will involve determining
Georgia Institute of Technology School of Earth and Atmospheric Sciences EAS 4641 Spring 2008 Lab 4 Major Anions In Atmospheric Aerosol Particles Purpose of Lab 4: This experiment will involve determining
Aqueous-Phase Chemistry in TM4-ECPL: SOA Formation via Cloud Processes Stelios Myriokefalitakis 1 Kostas Tsigaridis 2,3 Maria Kanakidou 1
 TM meeting, Heraklion, June 2010 Aqueous-Phase Chemistry in TM4-ECPL: SOA Formation via Cloud Processes Stelios Myriokefalitakis 1 Kostas Tsigaridis 2,3 Maria Kanakidou 1 1 Environmental Chemical Processes
TM meeting, Heraklion, June 2010 Aqueous-Phase Chemistry in TM4-ECPL: SOA Formation via Cloud Processes Stelios Myriokefalitakis 1 Kostas Tsigaridis 2,3 Maria Kanakidou 1 1 Environmental Chemical Processes
Aerosol modeling with WRF/Chem
 Aerosol modeling with WRF/Chem Jan Kazil University of Colorado / NOAA Earth System Research Laboratory WRF/Chem Tutorial, 3 August 2015 (WRF/Chem 3.7) Part I - Introduction Overview of... Aerosol Aerosol
Aerosol modeling with WRF/Chem Jan Kazil University of Colorado / NOAA Earth System Research Laboratory WRF/Chem Tutorial, 3 August 2015 (WRF/Chem 3.7) Part I - Introduction Overview of... Aerosol Aerosol
pinene (at 2 and 50 Torr) and β-pinene (at 200 Torr) with OH have been determined in varied conditions.
 ABSTRACT The composition of the troposphere is strongly affected by biogenic and anthropogenic emissions of chemical compounds, including hydrocarbons, carbon monoxide, and the nitrogen oxides. The emissions
ABSTRACT The composition of the troposphere is strongly affected by biogenic and anthropogenic emissions of chemical compounds, including hydrocarbons, carbon monoxide, and the nitrogen oxides. The emissions
Aerosol-Cloud-Radiation Interactions in Atmospheric Forecast Models
 Aerosol-Cloud-Radiation Interactions in Atmospheric Forecast Models John H. Seinfeld, Principal Investigator California Institute of Technology 1200 E. California Blvd., M/C 210-41 Pasadena, CA 91125 phone:
Aerosol-Cloud-Radiation Interactions in Atmospheric Forecast Models John H. Seinfeld, Principal Investigator California Institute of Technology 1200 E. California Blvd., M/C 210-41 Pasadena, CA 91125 phone:
CAPRAM 2.4 (MODAC mechanism): An extended and condensed tropospheric aqueous phase mechanism and its application
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. D14, 4426, doi:10.1029/2002jd002202, 2003 CAPRAM 2.4 (MODAC mechanism): An extended and condensed tropospheric aqueous phase mechanism and its application
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. D14, 4426, doi:10.1029/2002jd002202, 2003 CAPRAM 2.4 (MODAC mechanism): An extended and condensed tropospheric aqueous phase mechanism and its application
Parameterization of the nitric acid effect on CCN activation
 Atmos. Chem. Phys., 5, 879 885, 25 SRef-ID: 168-7324/acp/25-5-879 European Geosciences Union Atmospheric Chemistry and Physics Parameterization of the nitric acid effect on CCN activation S. Romakkaniemi,
Atmos. Chem. Phys., 5, 879 885, 25 SRef-ID: 168-7324/acp/25-5-879 European Geosciences Union Atmospheric Chemistry and Physics Parameterization of the nitric acid effect on CCN activation S. Romakkaniemi,
Separating Cloud Forming Nuclei from Interstitial Aerosol
 Chapter 16 Separating Cloud Forming Nuclei from Interstitial Aerosol Gourihar Kulkarni Additional information is available at the end of the chapter http://dx.doi.org/10.5772/50589 1. Introduction Our
Chapter 16 Separating Cloud Forming Nuclei from Interstitial Aerosol Gourihar Kulkarni Additional information is available at the end of the chapter http://dx.doi.org/10.5772/50589 1. Introduction Our
Quantification of Icelandic dust export: proposal of a combined measurement and modeling experiment
 Quantification of Icelandic dust export: proposal of a combined measurement and modeling experiment Konrad Kandler, Stephan Weinbruch, Kerstin Schepanski Technische Universität Darmstadt, Applied Geosciences
Quantification of Icelandic dust export: proposal of a combined measurement and modeling experiment Konrad Kandler, Stephan Weinbruch, Kerstin Schepanski Technische Universität Darmstadt, Applied Geosciences
Name of research institute or organization: Technische Universität Darmstadt, Institut für Angewandte Geowissenschaften, Umweltmineralogie
 Name of research institute or organization: Technische Universität Darmstadt, Institut für Angewandte Geowissenschaften, Umweltmineralogie Title of project: In-situ environmental scanning electron microscopic
Name of research institute or organization: Technische Universität Darmstadt, Institut für Angewandte Geowissenschaften, Umweltmineralogie Title of project: In-situ environmental scanning electron microscopic
Updated Dust-Iron Dissolution Mechanism: Effects Of Organic Acids, Photolysis, and Dust Mineralogy
 Updated Dust-Iron Dissolution Mechanism: Effects Of Organic Acids, Photolysis, and Dust Mineralogy Nicholas Meskhidze & Matthew Johnson First International Workshop on the Long Range Transport and Impacts
Updated Dust-Iron Dissolution Mechanism: Effects Of Organic Acids, Photolysis, and Dust Mineralogy Nicholas Meskhidze & Matthew Johnson First International Workshop on the Long Range Transport and Impacts
Tropospheric OH chemistry
 Tropospheric OH chemistry CO Oxidation mechanism: CO + OH CO 2 + H, H + O 2 + M HO 2 + M, HO 2 + NO OH + NO 2 NO 2 + hν (+O 2 ) NO + O 3 Initiation step Propagation Net: CO + 2 O 2 CO 2 + O 3 HO 2 + HO
Tropospheric OH chemistry CO Oxidation mechanism: CO + OH CO 2 + H, H + O 2 + M HO 2 + M, HO 2 + NO OH + NO 2 NO 2 + hν (+O 2 ) NO + O 3 Initiation step Propagation Net: CO + 2 O 2 CO 2 + O 3 HO 2 + HO
2005 UPDATES TO THE CARBON BOND MECHANISM: CB05
 2005 UPDATES TO THE CARBON BOND MECHANISM: CB05 Gary Z. Whitten, Smog Reyes Greg Yarwood, ENVIRON smogreyes@yahoo.com International Conference on Chemical Mechanisms December 6, 2006 Ackowledgements EPA:
2005 UPDATES TO THE CARBON BOND MECHANISM: CB05 Gary Z. Whitten, Smog Reyes Greg Yarwood, ENVIRON smogreyes@yahoo.com International Conference on Chemical Mechanisms December 6, 2006 Ackowledgements EPA:
A cloud microphysics model including trace gas condensation and sulfate chemistry
 BOREAL ENVIRONMENT RESEARCH 8: 43 424 ISSN 239-695 Helsinki December 23 23 A cloud microphysics model including trace gas condensation and sulfate chemistry Harri Kokkola ), Sami Romakkaniemi ), Markku
BOREAL ENVIRONMENT RESEARCH 8: 43 424 ISSN 239-695 Helsinki December 23 23 A cloud microphysics model including trace gas condensation and sulfate chemistry Harri Kokkola ), Sami Romakkaniemi ), Markku
Implications of Sulfate Aerosols on Clouds, Precipitation and Hydrological Cycle
 Implications of Sulfate Aerosols on Clouds, Precipitation and Hydrological Cycle Source: Sulfate aerosols are produced by chemical reactions in the atmosphere from gaseous precursors (with the exception
Implications of Sulfate Aerosols on Clouds, Precipitation and Hydrological Cycle Source: Sulfate aerosols are produced by chemical reactions in the atmosphere from gaseous precursors (with the exception
Response to Referee 2
 Response to Referee 2 S. Metzger et al. 10 August 2018 We thank the referee for the manuscript review. Please find our pointby-point reply below. Accordingly, the revised MS will include clarifications.
Response to Referee 2 S. Metzger et al. 10 August 2018 We thank the referee for the manuscript review. Please find our pointby-point reply below. Accordingly, the revised MS will include clarifications.
Modeling of cloud microphysics: from simple concepts to sophisticated parameterizations. Part I: warm-rain microphysics
 Modeling of cloud microphysics: from simple concepts to sophisticated parameterizations. Part I: warm-rain microphysics Wojciech Grabowski National Center for Atmospheric Research, Boulder, Colorado parameterization
Modeling of cloud microphysics: from simple concepts to sophisticated parameterizations. Part I: warm-rain microphysics Wojciech Grabowski National Center for Atmospheric Research, Boulder, Colorado parameterization
Aerosol Dynamics. Antti Lauri NetFAM Summer School Zelenogorsk, 9 July 2008
 Aerosol Dynamics Antti Lauri NetFAM Summer School Zelenogorsk, 9 July 2008 Department of Physics, Division of Atmospheric Sciences and Geophysics, University of Helsinki Aerosol Dynamics: What? A way to
Aerosol Dynamics Antti Lauri NetFAM Summer School Zelenogorsk, 9 July 2008 Department of Physics, Division of Atmospheric Sciences and Geophysics, University of Helsinki Aerosol Dynamics: What? A way to
ICSE Board Class IX Chemistry Paper 3 Solution
 ICSE Board Class IX Chemistry Paper 3 Solution SECTION I Answer 1 i. The number of electrons, that atom can lose, gain or share during a chemical reaction is called its valency. ii. Solute: A solute is
ICSE Board Class IX Chemistry Paper 3 Solution SECTION I Answer 1 i. The number of electrons, that atom can lose, gain or share during a chemical reaction is called its valency. ii. Solute: A solute is
Supporting information
 Supporting information Chemistry of urban grime: Inorganic ion composition of grime vs. particles in Leipzig, Germany Alyson M. Baergen, 1 Sarah A. Styler, Dominik van Pinxteren, Konrad Müller, Hartmut
Supporting information Chemistry of urban grime: Inorganic ion composition of grime vs. particles in Leipzig, Germany Alyson M. Baergen, 1 Sarah A. Styler, Dominik van Pinxteren, Konrad Müller, Hartmut
The Atmospheric Chemistry and Physics of Ammonia
 The Atmospheric Chemistry and Physics of Ammonia Russell Dickerson Dept. Meteorology, The University of Maryland Presented at the National Atmospheric Deposition Program Ammonia Workshop October 23, 2003
The Atmospheric Chemistry and Physics of Ammonia Russell Dickerson Dept. Meteorology, The University of Maryland Presented at the National Atmospheric Deposition Program Ammonia Workshop October 23, 2003
AEROSOL-CLOUD INTERACTIONS AND PRECIPITATION IN A GLOBAL SCALE. SAHEL Conference April 2007 CILSS Ouagadougou, Burkina Faso
 AEROSOL-CLOUD INTERACTIONS AND PRECIPITATION IN A GLOBAL SCALE SAHEL Conference 2007 2-6 April 2007 CILSS Ouagadougou, Burkina Faso The aerosol/precipitation connection Aerosol environment has changed
AEROSOL-CLOUD INTERACTIONS AND PRECIPITATION IN A GLOBAL SCALE SAHEL Conference 2007 2-6 April 2007 CILSS Ouagadougou, Burkina Faso The aerosol/precipitation connection Aerosol environment has changed
Workshop on Atmospheric Deposition: Processesand Environmental Impacts May 2012
 2339-14 Workshop on Atmospheric Deposition: Processesand Environmental Impacts 21-25 May 2012 Processes in wet deposition: In-cloud and below cloud scavenging Karine Sellegri Laboratoire de Meteorologie
2339-14 Workshop on Atmospheric Deposition: Processesand Environmental Impacts 21-25 May 2012 Processes in wet deposition: In-cloud and below cloud scavenging Karine Sellegri Laboratoire de Meteorologie
Diesel soot aging in urban plumes within hours under cold dark and humid conditions
 Supporting information for Diesel soot aging in urban plumes within hours under cold dark and humid conditions A. C. Eriksson 1,2*, C. Wittbom 1, P. Roldin 1,3, M. Sporre 4, E. Öström 1,5, P. Nilsson 2,
Supporting information for Diesel soot aging in urban plumes within hours under cold dark and humid conditions A. C. Eriksson 1,2*, C. Wittbom 1, P. Roldin 1,3, M. Sporre 4, E. Öström 1,5, P. Nilsson 2,
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 111, D23S45, doi: /2005jd006880, 2006
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 111,, doi:10.1029/2005jd006880, 2006 Oxalic acid in clear and cloudy atmospheres: Analysis of data from International Consortium for Atmospheric Research on Transport
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 111,, doi:10.1029/2005jd006880, 2006 Oxalic acid in clear and cloudy atmospheres: Analysis of data from International Consortium for Atmospheric Research on Transport
Collision and Coalescence 3/3/2010. ATS 351 Lab 7 Precipitation. Droplet Growth by Collision and Coalescence. March 7, 2006
 ATS 351 Lab 7 Precipitation March 7, 2006 Droplet Growth by Collision and Coalescence Growth by condensation alone takes too long ( 15 C -) Occurs in clouds with tops warmer than 5 F Greater the speed
ATS 351 Lab 7 Precipitation March 7, 2006 Droplet Growth by Collision and Coalescence Growth by condensation alone takes too long ( 15 C -) Occurs in clouds with tops warmer than 5 F Greater the speed
Precipitation Processes METR σ is the surface tension, ρ l is the water density, R v is the Gas constant for water vapor, T is the air
 Precipitation Processes METR 2011 Introduction In order to grow things on earth, they need water. The way that the earth naturally irrigates is through snowfall and rainfall. Therefore, it is important
Precipitation Processes METR 2011 Introduction In order to grow things on earth, they need water. The way that the earth naturally irrigates is through snowfall and rainfall. Therefore, it is important
Continuous measurement of airborne particles and gases
 Continuous measurement of airborne particles and gases Jeff Collett and Taehyoung Lee Atmospheric Science Department Colorado State University Funding: USDA/AES and NPS Outline Why measure particles and
Continuous measurement of airborne particles and gases Jeff Collett and Taehyoung Lee Atmospheric Science Department Colorado State University Funding: USDA/AES and NPS Outline Why measure particles and
Importance of vertical velocity variations in the cloud droplet nucleation process of marine stratus clouds
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 110,, doi:10.1029/2004jd004922, 2005 Importance of vertical velocity variations in the cloud droplet nucleation process of marine stratus clouds Yiran Peng, 1,2 Ulrike
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 110,, doi:10.1029/2004jd004922, 2005 Importance of vertical velocity variations in the cloud droplet nucleation process of marine stratus clouds Yiran Peng, 1,2 Ulrike
model to data comparison over Europe for year 2001
 Model Model by model to for year 2001 Model to CEREA (joint laboratory ENPC & EdF) Friday 15 September 2006 Model Air quality modeling (PAM project, PRIMEQUAL-PREDIT). Transboundary pollution, transfer
Model Model by model to for year 2001 Model to CEREA (joint laboratory ENPC & EdF) Friday 15 September 2006 Model Air quality modeling (PAM project, PRIMEQUAL-PREDIT). Transboundary pollution, transfer
Appendix 1. Periodic Table and Atomic Structure. History of the idea of elements.
 Appendix 1 Detailed list of additions and deletions This appendix provides a detailed list of additions and deletions compared with the former (1983) Leaving Certificate Chemistry syllabus. Completely
Appendix 1 Detailed list of additions and deletions This appendix provides a detailed list of additions and deletions compared with the former (1983) Leaving Certificate Chemistry syllabus. Completely
Atmospheric chemistry Acidification
 Atmospheric chemistry Acidification Presented by Pontus Roldin Most material from Erik Swietlicki Avd. för Kärnfysik Fysiska institutionen Lunds universitet Acidification 1 Acidification Sulphur- and nitrogen-containing
Atmospheric chemistry Acidification Presented by Pontus Roldin Most material from Erik Swietlicki Avd. för Kärnfysik Fysiska institutionen Lunds universitet Acidification 1 Acidification Sulphur- and nitrogen-containing
Air Monitoring. Semi-continuous determination of ambient air quality
 Air Monitoring Semi-continuous determination of ambient air quality The Particle Into Liquid Sampler a simple solution for the determination of ions in aerosol particles 02 Combustion of fossil fuels for
Air Monitoring Semi-continuous determination of ambient air quality The Particle Into Liquid Sampler a simple solution for the determination of ions in aerosol particles 02 Combustion of fossil fuels for
Insights Into Atmospheric Organic Aerosols Using An Aerosol Mass Spectrometer
 Insights Into Atmospheric Organic Aerosols Using An Aerosol Mass Spectrometer A thesis submitted to the University of Manchester Institute of Science and Technology for the degree of Doctor of Philosophy
Insights Into Atmospheric Organic Aerosols Using An Aerosol Mass Spectrometer A thesis submitted to the University of Manchester Institute of Science and Technology for the degree of Doctor of Philosophy
Aerosols AP sizes AP types Sources Sinks Amount and lifetime Aerosol radiative effects. Aerosols. Trude Storelvmo Aerosols 1 / 21
 Aerosols Trude Storelvmo Aerosols 1 / 21 Aerosols: Definition Definition of an aerosol: disperse system with air as carrier gas and a solid or liquid or a mixture of both as disperse phases. Aerosol particles
Aerosols Trude Storelvmo Aerosols 1 / 21 Aerosols: Definition Definition of an aerosol: disperse system with air as carrier gas and a solid or liquid or a mixture of both as disperse phases. Aerosol particles
Exam 2: Cloud Physics April 16, 2008 Physical Meteorology Questions 1-10 are worth 5 points each. Questions are worth 10 points each.
 Exam : Cloud Physics April, 8 Physical Meteorology 344 Name Questions - are worth 5 points each. Questions -5 are worth points each.. Rank the concentrations of the following from lowest () to highest
Exam : Cloud Physics April, 8 Physical Meteorology 344 Name Questions - are worth 5 points each. Questions -5 are worth points each.. Rank the concentrations of the following from lowest () to highest
Aerosol Basics: Definitions, size distributions, structure
 Aerosol Basics: Definitions, size distributions, structure Antti Lauri NetFAM Summer School Zelenogorsk, 9 July 2008 Department of Physics, Division of Atmospheric Sciences and Geophysics, University of
Aerosol Basics: Definitions, size distributions, structure Antti Lauri NetFAM Summer School Zelenogorsk, 9 July 2008 Department of Physics, Division of Atmospheric Sciences and Geophysics, University of
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION DOI: 10.1038/NGEO1809 Cloud droplet number enhanced by co-condensation of organic vapours supplementary information David Topping, Paul Connolly and Gordon McFiggans S1. Model
SUPPLEMENTARY INFORMATION DOI: 10.1038/NGEO1809 Cloud droplet number enhanced by co-condensation of organic vapours supplementary information David Topping, Paul Connolly and Gordon McFiggans S1. Model
Name of research institute or organization: Technische Universität Darmstadt, Institut für Angewandte Geowissenschaften, Umweltmineralogie
 Name of research institute or organization: Technische Universität Darmstadt, Institut für Angewandte Geowissenschaften, Umweltmineralogie Title of project: Single particle analysis of ice nuclei and interstitial
Name of research institute or organization: Technische Universität Darmstadt, Institut für Angewandte Geowissenschaften, Umweltmineralogie Title of project: Single particle analysis of ice nuclei and interstitial
On the importance of aqueous-phase chemistry on the oxidative capacity of the troposphere: A 3-dimensional global modeling study
 C O M E C A P 2 0 1 4 e - b o o k o f p r o c e e d i n g s v o l. 2 P a g e 282 On the importance of aqueous-phase chemistry on the oxidative capacity of the troposphere: A 3-dimensional global modeling
C O M E C A P 2 0 1 4 e - b o o k o f p r o c e e d i n g s v o l. 2 P a g e 282 On the importance of aqueous-phase chemistry on the oxidative capacity of the troposphere: A 3-dimensional global modeling
Chapter 25: The Chemistry of Life: Organic and Biological Chemistry
 Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Conditions of the Simulations
 Conditions of the Simulations Location: 44.4N, 73.9W, z = 1.5 km, Whiteface Mountain (WFM) summit Start time: 1730 (5:30 pm) local time September 17, 2016 End time: 1500 (3 pm) local time September 18,
Conditions of the Simulations Location: 44.4N, 73.9W, z = 1.5 km, Whiteface Mountain (WFM) summit Start time: 1730 (5:30 pm) local time September 17, 2016 End time: 1500 (3 pm) local time September 18,
The Sensitivity of Global Nucleation, CCN and Climate to SO2 and Criegee-Intermediate Chemistry
 The Sensitivity of Global Nucleation, CCN and Climate to SO2 and Criegee-Intermediate Chemistry Jeff Pierce, Mat Evans, Cat Scott, Steve D'Andrea, Delphine Farmer, Erik Swietlicki and Dom Spracklen Pierce,
The Sensitivity of Global Nucleation, CCN and Climate to SO2 and Criegee-Intermediate Chemistry Jeff Pierce, Mat Evans, Cat Scott, Steve D'Andrea, Delphine Farmer, Erik Swietlicki and Dom Spracklen Pierce,
A hierarchy of one- and two-moment microphysical parameterizations in the COSMO model
 Deutscher Wetterdienst GB Forschung und Entwicklung A hierarchy of one- and two-moment microphysical parameterizations in the COSMO model Axel Seifert German Weather Service Offenbach, Germany Ulrich Blahak
Deutscher Wetterdienst GB Forschung und Entwicklung A hierarchy of one- and two-moment microphysical parameterizations in the COSMO model Axel Seifert German Weather Service Offenbach, Germany Ulrich Blahak
Unit 4: Chemical Changes (Higher Content)
 Metals react with oxygen to produce metal oxides. E.g. Copper + Oxygen > Copper Oxide The reactions are oxidation reactions because the metals gain oxygen. Reactivity of Metals Metal Extraction Metals
Metals react with oxygen to produce metal oxides. E.g. Copper + Oxygen > Copper Oxide The reactions are oxidation reactions because the metals gain oxygen. Reactivity of Metals Metal Extraction Metals
Duncan Axisa*, Amit Teller, Roelof Bruintjes, Dan Breed, Roelof Burger National Center for Atmospheric Research (NCAR), Boulder CO USA
 J12.1 AEROSOL - CLOUD INTERACTIONS OVER ISTANBUL, TURKEY AND CENTRAL SAUDI ARABIA Duncan Axisa*, Amit Teller, Roelof Bruintjes, Dan Breed, Roelof Burger National Center for Atmospheric Research (NCAR),
J12.1 AEROSOL - CLOUD INTERACTIONS OVER ISTANBUL, TURKEY AND CENTRAL SAUDI ARABIA Duncan Axisa*, Amit Teller, Roelof Bruintjes, Dan Breed, Roelof Burger National Center for Atmospheric Research (NCAR),
EXPERIMENTS. Testing products of combustion: Reducing Copper(III) Oxide to Copper. Page 4
 APPARATUS Page 2 APPARATUS Page 3 Reducing Copper(III) Oxide to Copper EXPERIMENTS Page 4 Testing products of combustion: EXPERIMENTS Showing that oxygen and water is needed for rusting iron Page 5 Showing
APPARATUS Page 2 APPARATUS Page 3 Reducing Copper(III) Oxide to Copper EXPERIMENTS Page 4 Testing products of combustion: EXPERIMENTS Showing that oxygen and water is needed for rusting iron Page 5 Showing
Warm Cloud Processes. Some definitions. Two ways to make big drops: Effects of cloud condensation nuclei
 Warm Cloud Processes Dr. Christopher M. Godfrey University of North Carolina at Asheville Warm clouds lie completely below the 0 isotherm 0 o C Some definitions Liquid water content (LWC) Amount of liquid
Warm Cloud Processes Dr. Christopher M. Godfrey University of North Carolina at Asheville Warm clouds lie completely below the 0 isotherm 0 o C Some definitions Liquid water content (LWC) Amount of liquid
ATOC 3500/CHEM 3152 Week 9, March 8, 2016
 ATOC 3500/CHEM 3152 Week 9, March 8, 2016 Hand back Midterm Exams (average = 84) Interaction of atmospheric constituents with light Haze and Visibility Aerosol formation processes (more detail) Haze and
ATOC 3500/CHEM 3152 Week 9, March 8, 2016 Hand back Midterm Exams (average = 84) Interaction of atmospheric constituents with light Haze and Visibility Aerosol formation processes (more detail) Haze and
Review Topic 8: Phases of Matter and Mixtures
 Name: Score: 24 / 24 points (100%) Review Topic 8: Phases of Matter and Mixtures Multiple Choice Identify the choice that best completes the statement or answers the question. C 1. Soda water is a solution
Name: Score: 24 / 24 points (100%) Review Topic 8: Phases of Matter and Mixtures Multiple Choice Identify the choice that best completes the statement or answers the question. C 1. Soda water is a solution
A New Mechanism for Regional Atmospheric Chemistry Modelling. A contribution to subproject CMD. W.R. Stockwell', F Kirchner^ M. Kuhn' and S.
 A New Mechanism for Regional Atmospheric Chemistry Modelling A contribution to subproject CMD W.R. Stockwell', F Kirchner^ M. Kuhn' and S. Seefeld* *Fraunhofer Institute for Atmospheric Environmental Research
A New Mechanism for Regional Atmospheric Chemistry Modelling A contribution to subproject CMD W.R. Stockwell', F Kirchner^ M. Kuhn' and S. Seefeld* *Fraunhofer Institute for Atmospheric Environmental Research
Structure-activity relationships for the development of MCM/GECKOA mechanisms
 Atmospheric Chemical Mechanisms, Davis, December 2018 Structure-activity relationships for the development of MCM/GECKOA mechanisms Acknowledgement: Bernard Aumont Richard Valorso, Marie Camredon LISA
Atmospheric Chemical Mechanisms, Davis, December 2018 Structure-activity relationships for the development of MCM/GECKOA mechanisms Acknowledgement: Bernard Aumont Richard Valorso, Marie Camredon LISA
Introduction to aerosol modeling with WRF/Chem
 Introduction to aerosol modeling with WRF/Chem Jan Kazil University of Colorado/National Oceanic and Atmospheric Administration Outline 1. Aerosols in the atmosphere: Sources Processes Interaction with
Introduction to aerosol modeling with WRF/Chem Jan Kazil University of Colorado/National Oceanic and Atmospheric Administration Outline 1. Aerosols in the atmosphere: Sources Processes Interaction with
VOCALS REx: Aerosol Physics at the Ocean Surface On the NOAA RV Ronald H. Brown October, November 2008
 VOCALS REx: Aerosol Physics at the Ocean Surface On the NOAA RV Ronald H. Brown October, November 8 Catherine Hoyle, NOAA PMEL Derek Coffman, NOAA PMEL Tim Bates, NOAA PMEL Trish Quinn, NOAA PMEL Lelia
VOCALS REx: Aerosol Physics at the Ocean Surface On the NOAA RV Ronald H. Brown October, November 8 Catherine Hoyle, NOAA PMEL Derek Coffman, NOAA PMEL Tim Bates, NOAA PMEL Trish Quinn, NOAA PMEL Lelia
ICSE Board. Class X Chemistry. Board Paper Time: 1½ hrs Total Marks: 80
 ICSE Board Class X Chemistry Board Paper 2013 Time: 1½ hrs Total Marks: 80 General Instructions: 1. Answers to this paper must be written on the paper provided separately. 2. You will NOT be allowed to
ICSE Board Class X Chemistry Board Paper 2013 Time: 1½ hrs Total Marks: 80 General Instructions: 1. Answers to this paper must be written on the paper provided separately. 2. You will NOT be allowed to
PALM - Cloud Physics. Contents. PALM group. last update: Monday 21 st September, 2015
 PALM - Cloud Physics PALM group Institute of Meteorology and Climatology, Leibniz Universität Hannover last update: Monday 21 st September, 2015 PALM group PALM Seminar 1 / 16 Contents Motivation Approach
PALM - Cloud Physics PALM group Institute of Meteorology and Climatology, Leibniz Universität Hannover last update: Monday 21 st September, 2015 PALM group PALM Seminar 1 / 16 Contents Motivation Approach
Covalent (sharing of electron pairs) Ionic ( electrostatic attraction between oppositely charged ions)
 Covalent (sharing of electron pairs) Ionic ( electrostatic attraction between oppositely charged ions) Metallic (electrostatic attraction between + metal ions and delocalised electrons) Group 1 ions 1+
Covalent (sharing of electron pairs) Ionic ( electrostatic attraction between oppositely charged ions) Metallic (electrostatic attraction between + metal ions and delocalised electrons) Group 1 ions 1+
Experimental Methods for the Detection of Atmospheric Trace Gases
 Experimental Methods for the Detection of Atmospheric Trace Gases Andreas Hofzumahaus Forschungszentrum Jülich, IEK-8 Literature: D.E. Heard, Analytical Techniques for Atmospheric Measurement, Blackwell
Experimental Methods for the Detection of Atmospheric Trace Gases Andreas Hofzumahaus Forschungszentrum Jülich, IEK-8 Literature: D.E. Heard, Analytical Techniques for Atmospheric Measurement, Blackwell
Influence of Organic-Containing Aerosols on Marine Boundary Layer Processes
 DISTRIBUTION STATEMENT A. Approved for public release; distribution is unlimited. Influence of Organic-Containing Aerosols on Marine Boundary Layer Processes John H. Seinfeld California Institute of Technology,
DISTRIBUTION STATEMENT A. Approved for public release; distribution is unlimited. Influence of Organic-Containing Aerosols on Marine Boundary Layer Processes John H. Seinfeld California Institute of Technology,
High-Speed Gas and Headspace Analysis for the Process-Line and Laboratory: SIFT- MS IFPAC 2017
 High-Speed Gas and Headspace Analysis for the Process-Line and Laboratory: SIFT- MS IFPAC 2017 Y.J. Mange D.B. Milligan V.S. Langford B.J. Prince M. Perkins C. Anderson T. Wilks Who is using Syft Technologies
High-Speed Gas and Headspace Analysis for the Process-Line and Laboratory: SIFT- MS IFPAC 2017 Y.J. Mange D.B. Milligan V.S. Langford B.J. Prince M. Perkins C. Anderson T. Wilks Who is using Syft Technologies
representing in-cloud oxidation of sulfur in a particle-based cloud-microphysics scheme
 representing incloud oxidation of sulfur in a particlebased cloudmicrophysics scheme Anna Jaruga, Hanna Paw lowska, Sylwester Arabas Institute of Geophysics Faculty of Physics, University of Warsaw 8 th
representing incloud oxidation of sulfur in a particlebased cloudmicrophysics scheme Anna Jaruga, Hanna Paw lowska, Sylwester Arabas Institute of Geophysics Faculty of Physics, University of Warsaw 8 th
Solution Formation. Copyright Houghton Mifflin Company.All rights reserved. Presentation of Lecture Outlines, 12 2
 Solutions Solution Formation A solution is a homogeneous mixture of two or more substances, consisting of ions or molecules. (See Animation: Solution Equilibrium). A colloid, although it also appears to
Solutions Solution Formation A solution is a homogeneous mixture of two or more substances, consisting of ions or molecules. (See Animation: Solution Equilibrium). A colloid, although it also appears to
Frank Drewnick & Johannes Schneider & Silke S. Hings & Nele Hock & Kevin Noone & Admir Targino & Silke Weimer & Stephan Borrmann
 DOI 10.1007/s10874-006-9036-8 Measurement of Ambient, Interstitial, and Residual Aerosol Particles on a Mountaintop Site in Central Sweden using an Aerosol Mass Spectrometer and a CVI Frank Drewnick &
DOI 10.1007/s10874-006-9036-8 Measurement of Ambient, Interstitial, and Residual Aerosol Particles on a Mountaintop Site in Central Sweden using an Aerosol Mass Spectrometer and a CVI Frank Drewnick &
ATMOSPHERIC SCIENCE-ATS (ATS)
 Atmospheric Science-ATS (ATS) 1 ATMOSPHERIC SCIENCE-ATS (ATS) Courses ATS 150 Science of Global Climate Change Credits: 3 (3-0-0) Physical basis of climate change. Energy budget of the earth, the greenhouse
Atmospheric Science-ATS (ATS) 1 ATMOSPHERIC SCIENCE-ATS (ATS) Courses ATS 150 Science of Global Climate Change Credits: 3 (3-0-0) Physical basis of climate change. Energy budget of the earth, the greenhouse
Deutscher Wetterdienst
 Deutscher Wetterdienst Modelling the Volcanic Ash Episode: Experiences with COSMO-ART Detlev Majewski (FE1) Bernhard Vogel, Heike Vogel (KIT) Thomas Hanisch, Jochen Förstner (FE13), Ulrich Pflüger (FE15)
Deutscher Wetterdienst Modelling the Volcanic Ash Episode: Experiences with COSMO-ART Detlev Majewski (FE1) Bernhard Vogel, Heike Vogel (KIT) Thomas Hanisch, Jochen Förstner (FE13), Ulrich Pflüger (FE15)
Science 1.5 AS Demonstrate understanding of aspects of acids and bases WORKBOOK. Working to Excellence
 Science 1.5 AS 90944 Demonstrate understanding of aspects of acids and bases WORKBOOK Working to Excellence CONTENTS 1. Writing Excellence answers to Ion Formation questions 2. Writing Excellence answers
Science 1.5 AS 90944 Demonstrate understanding of aspects of acids and bases WORKBOOK Working to Excellence CONTENTS 1. Writing Excellence answers to Ion Formation questions 2. Writing Excellence answers
Physical Processes & Issues
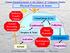 Physical Processes & Issues Radiative Transfer Climate VIS IR Cloud Drops & Ice Aerosol Processing Air quality Condensation Droplets & Xtals Cloud Dynamics Collection Aerosol Activation Hydrological Cycle
Physical Processes & Issues Radiative Transfer Climate VIS IR Cloud Drops & Ice Aerosol Processing Air quality Condensation Droplets & Xtals Cloud Dynamics Collection Aerosol Activation Hydrological Cycle
Precipitation Processes
 Precipitation Processes Dave Rahn Precipitation formation processes may be classified into two categories. These are cold and warm processes, where cold processes can only occur below 0 C and warm processes
Precipitation Processes Dave Rahn Precipitation formation processes may be classified into two categories. These are cold and warm processes, where cold processes can only occur below 0 C and warm processes
Urban background aerosols: Negative correlations of particle modes and fragmentation mechanism
 Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L11811, doi:10.1029/2006gl029109, 2007 Urban background aerosols: Negative correlations of particle modes and fragmentation mechanism
Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L11811, doi:10.1029/2006gl029109, 2007 Urban background aerosols: Negative correlations of particle modes and fragmentation mechanism
Summary of the cloud chemistry modeling intercomparison: Photochemical box model simulation
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. D7, 4214, doi:10.1029/2002jd002673, 2003 Summary of the cloud chemistry modeling intercomparison: Photochemical box model simulation M. C. Barth, 1 S. Sillman,
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. D7, 4214, doi:10.1029/2002jd002673, 2003 Summary of the cloud chemistry modeling intercomparison: Photochemical box model simulation M. C. Barth, 1 S. Sillman,
Parametrizing cloud and precipitation in today s NWP and climate models. Richard Forbes
 Parametrizing cloud and precipitation in today s NWP and climate models Richard Forbes (ECMWF) with thanks to Peter Bechtold and Martin Köhler RMetS National Meeting on Clouds and Precipitation, 16 Nov
Parametrizing cloud and precipitation in today s NWP and climate models Richard Forbes (ECMWF) with thanks to Peter Bechtold and Martin Köhler RMetS National Meeting on Clouds and Precipitation, 16 Nov
YEAR 10- Chemistry Term 1 plan
 YEAR 10- Chemistry Term 1 plan 2016-2017 Week Topic Learning outcomes 1 1. The particulate nature of matter State the distinguishing properties of solids, liquids and gases. Describe the structure of solids,
YEAR 10- Chemistry Term 1 plan 2016-2017 Week Topic Learning outcomes 1 1. The particulate nature of matter State the distinguishing properties of solids, liquids and gases. Describe the structure of solids,
Direct radiative forcing due to aerosols in Asia during March 2002
 Direct radiative forcing due to aerosols in Asia during March 2002 Soon-Ung Park, Jae-In Jeong* Center for Atmospheric and Environmental Modeling *School of Earth and Environmental Sciences, Seoul National
Direct radiative forcing due to aerosols in Asia during March 2002 Soon-Ung Park, Jae-In Jeong* Center for Atmospheric and Environmental Modeling *School of Earth and Environmental Sciences, Seoul National
CURRENT STATUS OF THE DEVELOPMENT AND EVALUATION OF AN UPDATED DETAILED MECHANISM FOR VOC OXIDATION
 CURRENT STATUS OF THE DEVELOPMENT AND EVALUATION OF AN UPDATED DETAILED MECHANISM FOR VOC OXIDATION William P. L. Carter Statewide Air Pollution Research Center and College of Engineering, Center for Environmental
CURRENT STATUS OF THE DEVELOPMENT AND EVALUATION OF AN UPDATED DETAILED MECHANISM FOR VOC OXIDATION William P. L. Carter Statewide Air Pollution Research Center and College of Engineering, Center for Environmental
CHAPTER 1. MEASURES OF ATMOSPHERIC COMPOSITION
 1 CHAPTER 1. MEASURES OF ATMOSPHERIC COMPOSITION The objective of atmospheric chemistry is to understand the factors that control the concentrations of chemical species in the atmosphere. In this book
1 CHAPTER 1. MEASURES OF ATMOSPHERIC COMPOSITION The objective of atmospheric chemistry is to understand the factors that control the concentrations of chemical species in the atmosphere. In this book
Organic Compounds - Formation Fate and Impact on Troposphere
 Organic Compounds - Formation Fate and Impact on Troposphere i.gensch@fz-juelich.de 2 / 20 Organic Compounds - Formation Fate and Impact on Troposphere i.gensch@fz-juelich.de 2 / 20 Definitions VOC: organic
Organic Compounds - Formation Fate and Impact on Troposphere i.gensch@fz-juelich.de 2 / 20 Organic Compounds - Formation Fate and Impact on Troposphere i.gensch@fz-juelich.de 2 / 20 Definitions VOC: organic
Preview from Notesale.co.uk Page 1 of 61
 Chemistry IGCSE Section A: Principles of Chemistry Chapter 1: States of Matter 1) Name the three states of matter and each one s particles (3). Solid (6): -Example: Ice. -Diagram of particles: -Spacing
Chemistry IGCSE Section A: Principles of Chemistry Chapter 1: States of Matter 1) Name the three states of matter and each one s particles (3). Solid (6): -Example: Ice. -Diagram of particles: -Spacing
A REVIEW OF OUR UNDERSTANDING OF THE AEROSOL CLOUD INTERACTION FROM THE PERSPECTIVE OF A BIN RESOLVED CLOUD SCALE MODELLING
 JP3.4 A REVIEW OF OUR UNDERSTANDING OF THE AEROSOL CLOUD INTERACTION FROM THE PERSPECTIVE OF A BIN RESOLVED CLOUD SCALE MODELLING Andrea I. Flossmann and W. Wobrock Clermont University, Aubière, France
JP3.4 A REVIEW OF OUR UNDERSTANDING OF THE AEROSOL CLOUD INTERACTION FROM THE PERSPECTIVE OF A BIN RESOLVED CLOUD SCALE MODELLING Andrea I. Flossmann and W. Wobrock Clermont University, Aubière, France
CHEMISTRY. Time : 3 Hrs. Max. Marks : 70
 CHEMISTRY Time : 3 Hrs. Max. Marks : 70 General instructions 1. All questions are compulsory. 2. Q.no. 1-5 are very short answer questions & carry 1 marks each Section-A. 3. Q.no. 6-12 are short answer
CHEMISTRY Time : 3 Hrs. Max. Marks : 70 General instructions 1. All questions are compulsory. 2. Q.no. 1-5 are very short answer questions & carry 1 marks each Section-A. 3. Q.no. 6-12 are short answer
The role of sea ice algae in Arctic marine ecosystems and oceanic emissions of dimethylsulfide. Hakase Hayashida Candidacy Exam August 19, 2015
 The role of sea ice algae in Arctic marine ecosystems and oceanic emissions of dimethylsulfide Hakase Hayashida Candidacy Exam August 19, 2015 Introduction: Role of sea ice algae in Arctic marine ecosystems
The role of sea ice algae in Arctic marine ecosystems and oceanic emissions of dimethylsulfide Hakase Hayashida Candidacy Exam August 19, 2015 Introduction: Role of sea ice algae in Arctic marine ecosystems
Slides partly by Antti Lauri and Hannele Korhonen. Liquid or solid particles suspended in a carrier gas Described by their
 Atmospheric Aerosols Slides partly by Antti Lauri and Hannele Korhonen Aerosol particles Liquid or solid particles suspended in a carrier gas Described by their Size Concentration - Number - Surface -
Atmospheric Aerosols Slides partly by Antti Lauri and Hannele Korhonen Aerosol particles Liquid or solid particles suspended in a carrier gas Described by their Size Concentration - Number - Surface -
Putting Secondary Organic Aerosol in Global Models. Issues and Approaches
 Putting Secondary Organic Aerosol in Global Models (and FRP s) Issues and Approaches Dean Hegg (University of Washington) Walter Sessions (NRL Monterey) Outline of Talk 1. The necessity of dealing with
Putting Secondary Organic Aerosol in Global Models (and FRP s) Issues and Approaches Dean Hegg (University of Washington) Walter Sessions (NRL Monterey) Outline of Talk 1. The necessity of dealing with
Role of atmospheric aerosol concentration on deep convective precipitation: Cloud-resolving model simulations
 Role of atmospheric aerosol concentration on deep convective precipitation: Cloud-resolving model simulations Wei-Kuo Tao,1 Xiaowen Li,1,2 Alexander Khain,3 Toshihisa Matsui,1,2 Stephen Lang,4 and Joanne
Role of atmospheric aerosol concentration on deep convective precipitation: Cloud-resolving model simulations Wei-Kuo Tao,1 Xiaowen Li,1,2 Alexander Khain,3 Toshihisa Matsui,1,2 Stephen Lang,4 and Joanne
MAHESH TUTORIALS I.C.S.E.
 MAHESH TUTORIALS I.C.S.E. GRADE - X (2017-2018) Exam No. : MT/ICSE/SEMI PRELIM - I-SET -A 008 Sulphuric acid, Ammonia, Analytical Chemistry, Organic Chemistry HCl, Nitric acid, Metallurgy Chemistry SCIENCE
MAHESH TUTORIALS I.C.S.E. GRADE - X (2017-2018) Exam No. : MT/ICSE/SEMI PRELIM - I-SET -A 008 Sulphuric acid, Ammonia, Analytical Chemistry, Organic Chemistry HCl, Nitric acid, Metallurgy Chemistry SCIENCE
IARA - GoAmazon 2014
 IARA - GoAmazon 2014 Activities related to Aerosol, Cloud, Precipitation, and Radiation Interactions and Dynamics of Convective Cloud Systems (ACRIDICON) and CHUVA Project Luiz.Machado@cptec.inpe.br CHUVA
IARA - GoAmazon 2014 Activities related to Aerosol, Cloud, Precipitation, and Radiation Interactions and Dynamics of Convective Cloud Systems (ACRIDICON) and CHUVA Project Luiz.Machado@cptec.inpe.br CHUVA
