Photoabsorption and Photoionization Studies of the Amino Acid Proline in the VUV Region
|
|
|
- Cleopatra Phillips
- 5 years ago
- Views:
Transcription
1 940 Brazilian Journal of Physics, vol. 35, no. 4A, December, 2005 Photoabsorption and Photoionization Studies of the Amino Acid Proline in the VUV Region L. H. Coutinho, M. G. P. Homem, R. L. Cavasso-Filho,R.R.T. Marinho, A. F. Lago, G. G. B. de Souza, and A. Naves de Brito Laboratório Nacional de Luz Síncrotron (LNLS), Box 6192, , Campinas-SP Brazil Instituto de Física, Universidade de Brasília, Box 4455, , Brasília-DF Brazil Instituto de Química, Universidade Federal do Rio de Janeiro, Cidade Universitária, Ilha do Fundão, , Rio de Janeiro-RJ Brazil Received on 11 January, 2005 Ionic fragmentation of the sublimated amino acid DL-proline has been studied using time-of-flight mass spectrometry and synchrotron radiation. Total ion yield and mass spectra were recorded in the 13 to 21.6 ev energy range. Partial ion yields have been calculated for the produced fragments and the results analyzed in a comparative way. Mass spectrum of proline previously obtained at ev using photons from a discharge lamp (He I), was used as reference in the comparison to the synchrotron radiation based spectra. The loss of the COOH fragment represents the most probable dissociation pathway following the photoionization of DLproline in the valence region. These are the first results of total and partial ion yields spectra for this molecule in its gas phase in the valence region using time-of-flight spectrometry. I. INTRODUCTION In recent years, there has been an increasing interest in the spectroscopic study of amino acids due to the fundamental importance of these molecules, which are building blocks of proteins. Knowledge on the valence shell electronic structure of amino acids is necessary for understanding the details of their photo-interaction and reactivity. In addition, valence photoionization studies provide a means for understanding the photo-stability, as well as the pathways leading to the photofragmentation products of these molecules following Vacuum Ultraviolet (VUV) excitation or ionization. Such information is of great importance for several areas, for instance, astrophysics and photochemistry, since amino acids have been found in the interstellar medium and meteoritic materials [1 3]. Only a few papers devoted to the experimental study of the amino acids in the VUV regions have been found in the literature. Powis et al. [4] reported on the valence and C 1s core level photoelectron spectra and quantum calculations of gaseous alanine and threonine. Jochims et al. [5] reported an extensive photoionization study in the 6-22 ev energy range, using quadrupole mass spectrometry, of five amino acids: glycine, α-alanine, β-alanine, α-aminoisobutyric acid and α- valine. The Time-Of-Flight (TOF) spectra of the sublimated alpha amino acids L-alanine, L-proline, L-valine and glycine, have been recently reported by Lago et al. [6], from photodissociation induced by He I (21.21 ev) photons. This previous results for proline [6], using a He I lamp and a similar spectrometer to the one used in this work, have also been used here for sake of comparison. Present address: Department of Chemistry, University of North Carolina, Chapel Hill, NC, , USA In this paper we report the dissociative photoionization of the amino acid DL-proline in the gas phase, by using Time-Of-Flight Mass Spectrometry (TOF-MS). Photons from a synchrotron radiation source and a discharge lamp (He I) were used as ionizing agents. The resulting photoionization products as well as fragmentation pathways leading to those species have been presented and discussed. Total Ion Yield (TIY) and Partial Ion Yield (PIY) spectra have been recorded in the ev energy range. Mass spectra have been also recorded as a function of photon energy. The resulting decay channels were analyzed in a comparative way. The spectra are, in general, similar to those obtained from electron impact (70 ev) experiments [7]. Within our knowledge, no similar TOF-MS data is available in the literature. II. EXPERIMENT The experimental set-up used has been described in detail in Ref. [8]. The ions produced by photoionization and photodissociation were detected using a TOF spectrometer schematically shown in Fig. 1. The mass spectra are recorded by the PhotoElectron PhotoIon COincidence technique (PEPICO). TIY and PIY spectra were obtained by scanning the photon energy and recording the corresponding ions produced in the photoionization process. In the PEPICO spectra, the relative areas of the peaks reflect the relative abundance of each species and the width is proportional to the Kinetic Energy Release (KER) during the photofragmentation process. The TOF spectrometer was mounted on a chamber that can rotate with respect to the polarization vector of the exciting beam, and was kept in the pseudo magic angle of 54.7 o during the measurements. At this angle, possible angular effects (discrimination) due the angular distribution of the photoelectron are avoided since this distribution is independent of the asymmetry parameter beta [8, 9].
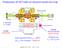
2 L. H. Coutinho et al. 941 Relative Yields (arb b. units) FIG. 1: Schematic drawing of the TOF mass spectrometer used for the measurements. The sublimated amino acid enters the ionization region by a needle placed between two plates that accelerate the ions to the flight tube and MicroChannel Plates (MCP) detectors on the right side, and the electrons to the MCP detectors on the left side. The arrival of an electron in the detector initiates a clock that marks the flight-time of the correlated ion. Energy (ev) FIG. 2: Total ion yield and partial ion yield of the produced fragments after photoionization of DL-proline. Above 21.6 ev the intensities drop due to the effect of the harmonic filter. The vacuum chamber base pressure is usually maintained at about 10 9 mbar. The photons were provided by the Toroidal Grating Monochromator beamline (TGM) [10] from the LNLS storage ring, located in Campinas-SP, Brazil. This beamline provides linearly polarized radiation from a bending magnet and operates in the range from 12 to 300 ev. The photon intensity was recorded by means of a light sensitive diode. In order to minimize contributions from higher order harmonics in the photon beam, a mixed metallic/gas filter has been added to the beamline. It basically consists in filling a certain region of the beamline with a rare gas (neon), which is isolated from the rest of the vacuum system by two thin metallic foils [11]. Therefore, contributions from higher-order light from the monochromator were negligible in the energy range studied. When using this filter we are able to perform measurements at energies up to 21.6 ev, the absorption edge of Ne. The DL-proline sample was purchased from Sigma- Aldrich in the form of crystalline powder with minimum purity of 99%, and was used without any further purification. Due to the low vapor pressure of this compound, it was necessary to use a heated source for sublimation. More details on the sublimation procedure are available in Ref. [6]. The pressure in the chamber was kept in the 10 7 mbar range during the experiments. In order to confirm the absence of decomposition of the samples upon heating, we have performed a Liquid Chromatographic-Mass Spectrometry (LC-MS) experiment after the sublimation procedure. The LC-MS results confirmed that no degradation occurred before sublimation of the sample in the temperature range used during our measurements. III. RESULTS We recorded several PEPICO spectra using synchrotron radiation, to analyse both total and partial ion yields. The spectral range from 13 to 22 ev was covered, with points being collected every 0.2 ev. From this set of spectra we were able to construct the graph shown in Fig. 2, where for each photon energy we plot the relative production of the main fragments (PIY) and the TIY. The yields are normalized at their maximum values. The pressure in the experimental chamber and the intensity of the photon beam were used to normalize the data. From these curves we can see that the production of ions increases as the photon energy increases. Besides, a higher slope between 16 and 18 ev can also be observed. From the TIY curve we can distinguish three structures around the energies 14.4 ev, 17.0 ev and 19.2 ev. Above 21.6 ev the intensities drop due to the effect of the harmonic filter. The values of the PIY obtained for DL-proline as function of the photon energy are also shown in table 1. It is important to compare both Fig. 2 and table 1 when analyzing the results. As an example let us take OH + : in the graph it shows a large increase on its production above 18 ev, but if we look on the table we observe that even at ev its production is only about 1% of total. The PIY curves on the graph should not be quantitatively compared one to the other, since their intensities have been normalized at their maximum values, and the table should be used to have a better understanding of the results. To complement the results above we took PEPICO spectra on the top of the main features observed in the TIY spectra, and the results are presented in Figs. 3 and 4. From table 1 the main fragments resulting from the photofragmentation of DL-proline are C 4 H 8 N + and C 2 H 3 O +, which correspond to approximately 90% of all detected fragments in the lower part of the spectra, decreasing to about 60% at ev. The parent ion presented a very low intensity, suggesting an efficient molecular bond rupture and instability of this ion in the time scale of the experiment (the flight-time of this ion in our TOF spectrometer was approximately 4.2 µs). The most intense peak in all the PEPICO spectra shown in Figs. 3 and 4 is attributed to C 4 H 8 N +, which corresponds to the loss of a neutral fragment COOH, which is in agreement with results previously obtained for L-proline by the He I photoionization [6].
3 942 Brazilian Journal of Physics, vol. 35, no. 4A, December, 2005 FIG. 3: Mass spectra of DL-proline at 14.2, 14.8, 15.3 and 16.6 ev photon energies. FIG. 4: Mass spectra of DL-proline at 18.2, 19.0, 19.3 and ev photon energies. We also point out the presence of ions formed from rearrangement reactions, such as OH +,H 2 O + and CO +. However, the presence of water contamination in the peaks at =17 and 18 can not be discarded. The formation of fragments such as OH + and CO + may result from the fast dissociation of the COOH + ion, which was absent from the mass spectra. The CO + fragment presents a dramatic increase in its production with the increasing of the photon energy when compared to the other fragments, as can be observed both from the table 1 and from Figs. 3 and 4. The spectrum of L-proline at ev (He I) shown in Fig. 5 was taken from Lago et al. [6]. In this figure it is possible to identify a higher number of ionic fragments than in our present results for DL-proline, taken with synchrotron radiation. Also the relative productions of the main fragments are different. One hypothesis to explain this divergence is the occurrence of mass discrimination in the detector used in our spectrometer. The efficiency detection of the microchannel plates is a function of the kinetic energy of the fragments [8]. In this sense, the heavier masses would have smaller detection FIG. 5: Mass spectra of L-proline taken from Lago et al. [6] using a He I lamp.
4 L. H. Coutinho et al. 943 TABLE I: PIY in % for the amino acid DL-proline after photoionization for several photon energies. Fragment Photon Energy (ev) H OH H 2 O HCN C 2 H + 3 CO CH 2 O NO + C 2 NH C 3 H + 5 C 2 NH C 2 H 2 O + C 2 H 3 O C 2 H 4 O CHO C 4 H 6 N C 4 H 7 N C 4 H 8 N C 2 HNO PIY He I lamp / PIY SR efficiency. To try to confirm this hypothesis we constructed the graph in Fig. 6, where we compare the relative ionic productions for two spectra taken at ev, one from Ref. [6], using the He I lamp, and the other using synchrotron radiation. Care was taken to add together all unresolved peaks in a group of lines thus eliminating spurious fluctuations coming from the different resolution between the synchrotron radiation and He I spectra. Furthermore, the peaks H 2 O +,OH + and H + were also removed from analysis since we clearly detect signs of a small water contribution. This contribution changed in the He I and synchrotron radiation spectra. From this graph we can observe that the ratio between the partial productions of the two spectra (the one with the discharge lamp and the one with synchrotron radiation) increases for heavier masses. This could support our hypothesis of mass discrimination stated above. In summary, time-of-flight mass spectrometry has been employed to the study of the natural amino acid DL-proline using synchrotron radiation as the ionization source. Total ion yield and mass spectra were recorded from 13 to 21.6 ev, using a metallic/gas filter coupled to the beamline in order to reduce the contribution from higher order harmonics contamination. Fragmentation products were identified and the relative production among them for a few photon energies was obtained. The loss of the COOH fragment represents the most probable dissociation pathway following the photoionization of DL-proline in the valence region FIG. 6: The graph shows the ratio between the partial ion productions for the observed fragments using He I lamp and synchrotron radiation (SR) at ev. The point at =42 shows the contribution of the peaks at =41, 42 and 43 in the mass spectra in order to minimize the effects due the different mass resolution between the spectra obtained with He I and synchrotron radiation. The peaks at =17, 18 was not included due the different water contamination in spectra. IV. ACKNOWLEDGMENTS The authors wish to acknowledge the staff of the LNLS and support from the Brazilian agencies CAPES, CNPq, FAPERJ, FAP-DF and FAPESP. [1] A. Brack (Ed.), The Molecular Origins of Life, Cambridge University Press, UK (1998). [2] L. E. Snyder, Origins Life Evol. Biosphere 27, 115 (1997). [3] M. H. Engel and B. Nagy, Nature (London) 296, 837 (1982). [4] I. Powis, E. E. Rennie, U. Hergenhahn, O. Kugeler, and R. Bussy-Socrate, J. Phys. Chem. A 107, 25 (2003). [5] H.-W. Jochims, M. Schwell, J-L. Chotin, M. Clemino, F. Dulieu, H. Baumgartel, and S. Leach, Chem. Phys. 298, 279 (2004). [6] A. F. Lago, L. H. Coutinho, R. R. T. Marinho, A. Naves de Brito, and G. G. B. de Souza, Chem. Phys. 307, 9 (2004). [7] NIST, Chemistry webbook ( [8] F. Burmeister, L. H. Coutinho, R. R. T. Marinho, M. G. P. Homem, M. A. A. de Morais, A. Mocellin, O. Bjorneholm, S. L. Sorensen, P. T. Fonseca, J. G. Pacheco, and A. Naves de Brito. Submitted to J. Elect. Spect. Rel. Phen. (2004).
5 944 Brazilian Journal of Physics, vol. 35, no. 4A, December, 2005 [9] A. Mocellin. R. R. T. Marinho, L. H. Coutinho, F. Burmeister, K. Wiesner, and A. Naves de Brito, Chem. Phys. 289, 163 (2003). [10] P. T. Fonseca, J. G. Pacheco, E. D. Samogin, and A. R. B. de Castro, Rev. Sci. Instrum. 63, 1256 (1992). [11] R. L. Cavasso-Filho, M. G. P. Homem, R. Landers, and A. Naves de Brito. J. Elect. Spect. Rel. Phen , 1125 (2005).
Excitation and ionic fragmentation of gas-phase biomolecules using electrons and synchrotron radiation
 Journal of Physics: Conference Series Excitation and ionic fragmentation of gas-phase biomolecules using electrons and synchrotron radiation To cite this article: G G B de Souza et al 2007 J. Phys.: Conf.
Journal of Physics: Conference Series Excitation and ionic fragmentation of gas-phase biomolecules using electrons and synchrotron radiation To cite this article: G G B de Souza et al 2007 J. Phys.: Conf.
Photoelectron Spectroscopy using High Order Harmonic Generation
 Photoelectron Spectroscopy using High Order Harmonic Generation Alana Ogata Yamanouchi Lab, University of Tokyo ABSTRACT The analysis of photochemical processes has been previously limited by the short
Photoelectron Spectroscopy using High Order Harmonic Generation Alana Ogata Yamanouchi Lab, University of Tokyo ABSTRACT The analysis of photochemical processes has been previously limited by the short
Vibrational effect on the fragmentation dynamics of the C K-shell excited CF 2 CH 2
 Vibrational effect on the fragmentation dynamics of the C K-shell excited CF 2 CH 2 K. Okada a, Y. Yamana a, T. Ibuki b, A. Fujii c, S. Nagaoka c, K. Tabayashi a, Y. Shimada b, Y. Morishita d, Y. Tamenori
Vibrational effect on the fragmentation dynamics of the C K-shell excited CF 2 CH 2 K. Okada a, Y. Yamana a, T. Ibuki b, A. Fujii c, S. Nagaoka c, K. Tabayashi a, Y. Shimada b, Y. Morishita d, Y. Tamenori
2. Separate the ions based on their mass to charge (m/e) ratio. 3. Measure the relative abundance of the ions that are produced
 I. Mass spectrometry: capable of providing both quantitative and qualitative information about samples as small as 100 pg (!) and with molar masses in the 10 4-10 5 kdalton range A. The mass spectrometer
I. Mass spectrometry: capable of providing both quantitative and qualitative information about samples as small as 100 pg (!) and with molar masses in the 10 4-10 5 kdalton range A. The mass spectrometer
Laser Dissociation of Protonated PAHs
 100 Chapter 5 Laser Dissociation of Protonated PAHs 5.1 Experiments The photodissociation experiments were performed with protonated PAHs using different laser sources. The calculations from Chapter 3
100 Chapter 5 Laser Dissociation of Protonated PAHs 5.1 Experiments The photodissociation experiments were performed with protonated PAHs using different laser sources. The calculations from Chapter 3
X-ray induced fragmentation of size-selected salt clusterions stored in an ion trap
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 1 X-ray induced fragmentation of size-selected salt clusterions stored in an ion trap Mauritz
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 1 X-ray induced fragmentation of size-selected salt clusterions stored in an ion trap Mauritz
Instrumental Analysis. Mass Spectrometry. Lecturer:! Somsak Sirichai
 303351 Instrumental Analysis Mass Spectrometry Lecturer:! Somsak Sirichai Mass Spectrometry What is Mass spectrometry (MS)? An analytic method that employs ionization and mass analysis of compounds in
303351 Instrumental Analysis Mass Spectrometry Lecturer:! Somsak Sirichai Mass Spectrometry What is Mass spectrometry (MS)? An analytic method that employs ionization and mass analysis of compounds in
arxiv: v1 [physics.chem-ph] 29 Jul 2010
![arxiv: v1 [physics.chem-ph] 29 Jul 2010 arxiv: v1 [physics.chem-ph] 29 Jul 2010](/thumbs/88/116577559.jpg) Dissociative Electron Attachment to Polyatomic Molecules - V : Formic Acid and n-propyl Amine N. Bhargava Ram 1, and E. Krishnakumar 1, 1 Tata Institute of Fundamental Research, Mumbai 400005, India Abstract
Dissociative Electron Attachment to Polyatomic Molecules - V : Formic Acid and n-propyl Amine N. Bhargava Ram 1, and E. Krishnakumar 1, 1 Tata Institute of Fundamental Research, Mumbai 400005, India Abstract
Fundamentals of Mass Spectrometry. Fundamentals of Mass Spectrometry. Learning Objective. Proteomics
 Mass spectrometry (MS) is the technique for protein identification and analysis by production of charged molecular species in vacuum, and their separation by magnetic and electric fields based on mass
Mass spectrometry (MS) is the technique for protein identification and analysis by production of charged molecular species in vacuum, and their separation by magnetic and electric fields based on mass
Propose a structure for an alcohol, C4H10O, that has the following
 Propose a structure for an alcohol, C4H10O, that has the following 13CNMR spectral data: Broadband _ decoupled 13CNMR: 19.0, 31.7, 69.5 б DEPT _90: 31.7 б DEPT _ 135: positive peak at 19.0 & 31.7 б, negative
Propose a structure for an alcohol, C4H10O, that has the following 13CNMR spectral data: Broadband _ decoupled 13CNMR: 19.0, 31.7, 69.5 б DEPT _90: 31.7 б DEPT _ 135: positive peak at 19.0 & 31.7 б, negative
CEE 772: Instrumental Methods in Environmental Analysis
 Updated: 10 December 2014 Print version CEE 772: Instrumental Methods in Environmental Analysis Lecture #21 Mass Spectrometry: Mass Filters & Spectrometers (Skoog, Chapt. 20, pp.511-524) (Harris, Chapt.
Updated: 10 December 2014 Print version CEE 772: Instrumental Methods in Environmental Analysis Lecture #21 Mass Spectrometry: Mass Filters & Spectrometers (Skoog, Chapt. 20, pp.511-524) (Harris, Chapt.
Interpretation of Organic Spectra. Chem 4361/8361
 Interpretation of Organic Spectra Chem 4361/8361 Characteristics of Common Spectrometric Methods H-1 C-13 MS IR/RAMAN UV-VIS ORD/CD X- RAY Radiation type RF RF Not relevant IR UV to visible UV to visible
Interpretation of Organic Spectra Chem 4361/8361 Characteristics of Common Spectrometric Methods H-1 C-13 MS IR/RAMAN UV-VIS ORD/CD X- RAY Radiation type RF RF Not relevant IR UV to visible UV to visible
Harris: Quantitative Chemical Analysis, Eight Edition
 Harris: Quantitative Chemical Analysis, Eight Edition CHAPTER 21: MASS SPECTROMETRY CHAPTER 21: Opener 21.0 Mass Spectrometry Mass Spectrometry provides information about 1) The elemental composition of
Harris: Quantitative Chemical Analysis, Eight Edition CHAPTER 21: MASS SPECTROMETRY CHAPTER 21: Opener 21.0 Mass Spectrometry Mass Spectrometry provides information about 1) The elemental composition of
MS/MS .LQGVRI0606([SHULPHQWV
 0DVV6SHFWURPHWHUV Tandem Mass Spectrometry (MS/MS) :KDWLV0606" Mass spectrometers are commonly combined with separation devices such as gas chromatographs (GC) and liquid chromatographs (LC). The GC or
0DVV6SHFWURPHWHUV Tandem Mass Spectrometry (MS/MS) :KDWLV0606" Mass spectrometers are commonly combined with separation devices such as gas chromatographs (GC) and liquid chromatographs (LC). The GC or
Abstract... I. Acknowledgements... III. Table of Content... V. List of Tables... VIII. List of Figures... IX
 Abstract... I Acknowledgements... III Table of Content... V List of Tables... VIII List of Figures... IX Chapter One IR-VUV Photoionization Spectroscopy 1.1 Introduction... 1 1.2 Vacuum-Ultraviolet-Ionization
Abstract... I Acknowledgements... III Table of Content... V List of Tables... VIII List of Figures... IX Chapter One IR-VUV Photoionization Spectroscopy 1.1 Introduction... 1 1.2 Vacuum-Ultraviolet-Ionization
Lecture 8: Mass Spectrometry
 intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene experiment CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can we get from MS spectrum?
intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene experiment CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can we get from MS spectrum?
CEE 772 Lecture #27 12/10/2014. CEE 772: Instrumental Methods in Environmental Analysis
 Updated: 10 December 2014 Print version CEE 772: Instrumental Methods in Environmental Analysis Lecture #21 Mass Spectrometry: Mass Filters & Spectrometers (Skoog, Chapt. 20, pp.511 524) (Harris, Chapt.
Updated: 10 December 2014 Print version CEE 772: Instrumental Methods in Environmental Analysis Lecture #21 Mass Spectrometry: Mass Filters & Spectrometers (Skoog, Chapt. 20, pp.511 524) (Harris, Chapt.
Lecture 8: Mass Spectrometry
 intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can be obtained from a MS spectrum?
intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can be obtained from a MS spectrum?
Paolo Tosi Dipartimento di Fisica - Università di Trento
 Paolo Tosi Dipartimento di Fisica - Università di Trento Email : paolo.tosi@unitn.it Chemical synthesis in acetonitrile containing discharges. Insights from photoionization experiments with synchrotron
Paolo Tosi Dipartimento di Fisica - Università di Trento Email : paolo.tosi@unitn.it Chemical synthesis in acetonitrile containing discharges. Insights from photoionization experiments with synchrotron
A velocity map imaging spectrometer for electron-ion and ion-ion coincidence. experiments with synchrotron radiation
 A velocity map imaging spectrometer for electron-ion and ion-ion coincidence experiments with synchrotron radiation D. Rolles 1,2, Z. D. Pešić 1,2, M. Perri 1,2, R. C. Bilodeau 1,2, G. D. Ackerman 2, B.
A velocity map imaging spectrometer for electron-ion and ion-ion coincidence experiments with synchrotron radiation D. Rolles 1,2, Z. D. Pešić 1,2, M. Perri 1,2, R. C. Bilodeau 1,2, G. D. Ackerman 2, B.
Upper limits for the circular dichroism for the C 1s and O 1s core excitation of methyl oxirane
 Upper limits for the circular dichroism for the C 1s and O 1s core excitation of methyl oxirane G. Prümper, T. Lischke, H. Fukuzawa, K. Ueda Institute of Multidisciplinary Research for Advanced Materials,
Upper limits for the circular dichroism for the C 1s and O 1s core excitation of methyl oxirane G. Prümper, T. Lischke, H. Fukuzawa, K. Ueda Institute of Multidisciplinary Research for Advanced Materials,
Gas phase molecular physics experiments in synchrotron sources of second and third generation
 Gas phase molecular physics experiments in synchrotron sources of second and third generation Wednesday 24 Nov 2010 at 13:10 (00h30') This talk will present an overview of gasphase experiments of molecular
Gas phase molecular physics experiments in synchrotron sources of second and third generation Wednesday 24 Nov 2010 at 13:10 (00h30') This talk will present an overview of gasphase experiments of molecular
Welcome to Organic Chemistry II
 Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
Time of Flight Mass Spectroscopy and Velocity Map Imaging
 Time of Flight Mass Spectroscopy and Velocity Map Imaging Geet Ghanshyam January 26, 2013 Velocity map imaging (VMI) is used to study the dynamics of various dissociative electron attachment (DEA) processes
Time of Flight Mass Spectroscopy and Velocity Map Imaging Geet Ghanshyam January 26, 2013 Velocity map imaging (VMI) is used to study the dynamics of various dissociative electron attachment (DEA) processes
PHOTOELECTRON SPECTROSCOPY OF RADICALS
 PHOTOELECTRON SPECTROSCOPY OF RADICALS Shibu E S (CY07D011) Uday Baskar Rao (CY06D028) Jasmine Maria Joseph (CY07D010) Goutam Kumar Kole (CY05C008) Puran Kumar De (CY05C013) 1 PHOTOELECTRON SPECTROSCOPY
PHOTOELECTRON SPECTROSCOPY OF RADICALS Shibu E S (CY07D011) Uday Baskar Rao (CY06D028) Jasmine Maria Joseph (CY07D010) Goutam Kumar Kole (CY05C008) Puran Kumar De (CY05C013) 1 PHOTOELECTRON SPECTROSCOPY
Electron spectroscopy Lecture Kai M. Siegbahn ( ) Nobel Price 1981 High resolution Electron Spectroscopy
 Electron spectroscopy Lecture 1-21 Kai M. Siegbahn (1918 - ) Nobel Price 1981 High resolution Electron Spectroscopy 653: Electron Spectroscopy urse structure cture 1. Introduction to electron spectroscopies
Electron spectroscopy Lecture 1-21 Kai M. Siegbahn (1918 - ) Nobel Price 1981 High resolution Electron Spectroscopy 653: Electron Spectroscopy urse structure cture 1. Introduction to electron spectroscopies
Photodissociation of organic molecules in star-forming regions. II. Acetic acid ABSTRACT
 A&A 449, 1289 1296 (2006) DOI: 10.1051/0004-6361:20053927 c ESO 2006 Astronomy & Astrophysics Photodissociation of organic molecules in star-forming regions II. Acetic acid S. Pilling 1,2,A.C.F.Santos
A&A 449, 1289 1296 (2006) DOI: 10.1051/0004-6361:20053927 c ESO 2006 Astronomy & Astrophysics Photodissociation of organic molecules in star-forming regions II. Acetic acid S. Pilling 1,2,A.C.F.Santos
high temp ( K) Chapter 20: Atomic Spectroscopy
 high temp (2000-6000K) Chapter 20: Atomic Spectroscopy 20-1. An Overview Most compounds Atoms in gas phase high temp (2000-6000K) (AES) (AAS) (AFS) sample Mass-to-charge (ICP-MS) Atomic Absorption experiment
high temp (2000-6000K) Chapter 20: Atomic Spectroscopy 20-1. An Overview Most compounds Atoms in gas phase high temp (2000-6000K) (AES) (AAS) (AFS) sample Mass-to-charge (ICP-MS) Atomic Absorption experiment
Chemistry 311: Topic 3 - Mass Spectrometry
 Mass Spectroscopy: A technique used to measure the mass-to-charge ratio of molecules and atoms. Often characteristic ions produced by an induced unimolecular dissociation of a molecule are measured. These
Mass Spectroscopy: A technique used to measure the mass-to-charge ratio of molecules and atoms. Often characteristic ions produced by an induced unimolecular dissociation of a molecule are measured. These
3 Use of Mass Spectra to Obtain Structural Information
 3 Use of Mass Spectra to Obtain Structural Information 1 Mass Spectrometry One of the most sensitive and versatile analytical tools More sensitive than other spectroscopic methods (e.g. IR spectroscopy)
3 Use of Mass Spectra to Obtain Structural Information 1 Mass Spectrometry One of the most sensitive and versatile analytical tools More sensitive than other spectroscopic methods (e.g. IR spectroscopy)
Lecture 14 Organic Chemistry 1
 CHEM 232 Organic Chemistry I at Chicago Lecture 14 Organic Chemistry 1 Professor Duncan Wardrop February 25, 2010 1 CHEM 232 Organic Chemistry I at Chicago Mass Spectrometry Sections: 13.24-13.25 2 Spectroscopy
CHEM 232 Organic Chemistry I at Chicago Lecture 14 Organic Chemistry 1 Professor Duncan Wardrop February 25, 2010 1 CHEM 232 Organic Chemistry I at Chicago Mass Spectrometry Sections: 13.24-13.25 2 Spectroscopy
Hydrogen Abstraction/Acetylene Addition Revealed
 Hydrogen Abstraction/Acetylene Addition Revealed Dorian S. N. Parker, Ralf I. Kaiser,* Tyler P. Troy, and Musahid Ahmed* University of Hawaii at Manoa, USA Lawrence Berkeley National Laboratory, USA Angew.
Hydrogen Abstraction/Acetylene Addition Revealed Dorian S. N. Parker, Ralf I. Kaiser,* Tyler P. Troy, and Musahid Ahmed* University of Hawaii at Manoa, USA Lawrence Berkeley National Laboratory, USA Angew.
An introduction to X- ray photoelectron spectroscopy
 An introduction to X- ray photoelectron spectroscopy X-ray photoelectron spectroscopy belongs to a broad class of spectroscopic techniques, collectively called, electron spectroscopy. In general terms,
An introduction to X- ray photoelectron spectroscopy X-ray photoelectron spectroscopy belongs to a broad class of spectroscopic techniques, collectively called, electron spectroscopy. In general terms,
Because light behaves like a wave, we can describe it in one of two ways by its wavelength or by its frequency.
 Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
Hydrogenation of solid hydrogen cyanide HCN and methanimine CH 2 NH at low temperature
 Hydrogenation of solid hydrogen cyanide HCN and methanimine CH 2 NH at low temperature P. Theule, F. Borget, F. Mispelaer, G. Danger, F. Duvernay, J. C. Guillemin, and T. Chiavassa 5 534,A64 (2011) By
Hydrogenation of solid hydrogen cyanide HCN and methanimine CH 2 NH at low temperature P. Theule, F. Borget, F. Mispelaer, G. Danger, F. Duvernay, J. C. Guillemin, and T. Chiavassa 5 534,A64 (2011) By
Photofragmentation of valence states of CF2X2 + (X=H,Cl,Br) studied by threshold photoelectron - photoion coincidence spectroscopy
 Photofragmentation of valence states of CF2X2 (X=H,Cl,Br) studied by threshold photoelectron - photoion coincidence spectroscopy D.P. Seccombe, R.P. Tuckett and B.O. Fisher J. Chem. Phys., (2001) 114,
Photofragmentation of valence states of CF2X2 (X=H,Cl,Br) studied by threshold photoelectron - photoion coincidence spectroscopy D.P. Seccombe, R.P. Tuckett and B.O. Fisher J. Chem. Phys., (2001) 114,
Extrel Application Note
 Extrel Application Note Real-Time Plasma Monitoring and Detection of Trace H 2 O and HF Species in an Argon Based Plasma Jian Wei, 575 Epsilon Drive, Pittsburgh, PA 15238. (Presented at the 191st Electrochemical
Extrel Application Note Real-Time Plasma Monitoring and Detection of Trace H 2 O and HF Species in an Argon Based Plasma Jian Wei, 575 Epsilon Drive, Pittsburgh, PA 15238. (Presented at the 191st Electrochemical
Spectroscopy, Energetics,, and Reaction Dynamics of Neutrals and Ions by High-Resolution. Photoionization and Photoelectron Methods
 Spectroscopy, Energetics,, and Reaction Dynamics of Neutrals and Ions by High-Resolution Photoionization and Photoelectron Methods Cheuk-Yiu Ng Department of Chemistry University of California, Davis Davis,
Spectroscopy, Energetics,, and Reaction Dynamics of Neutrals and Ions by High-Resolution Photoionization and Photoelectron Methods Cheuk-Yiu Ng Department of Chemistry University of California, Davis Davis,
Single photon ionization of hydrogen bonded clusters with a soft x-ray laser: HCOOH x and HCOOH y H 2 O z
 THE JOURNAL OF CHEMICAL PHYSICS 126, 244301 2007 Single photon ionization of hydrogen bonded clusters with a soft x-ray laser: HCOOH x and HCOOH y H 2 O z S. Heinbuch Department of Electrical and Computer
THE JOURNAL OF CHEMICAL PHYSICS 126, 244301 2007 Single photon ionization of hydrogen bonded clusters with a soft x-ray laser: HCOOH x and HCOOH y H 2 O z S. Heinbuch Department of Electrical and Computer
TANDEM MASS SPECTROSCOPY
 TANDEM MASS SPECTROSCOPY 1 MASS SPECTROMETER TYPES OF MASS SPECTROMETER PRINCIPLE TANDEM MASS SPECTROMETER INSTRUMENTATION QUADRAPOLE MASS ANALYZER TRIPLE QUADRAPOLE MASS ANALYZER TIME OF FLIGHT MASS ANALYSER
TANDEM MASS SPECTROSCOPY 1 MASS SPECTROMETER TYPES OF MASS SPECTROMETER PRINCIPLE TANDEM MASS SPECTROMETER INSTRUMENTATION QUADRAPOLE MASS ANALYZER TRIPLE QUADRAPOLE MASS ANALYZER TIME OF FLIGHT MASS ANALYSER
Association of H + with H 2 at Low Temperatures
 WDS'11 Proceedings of Contributed Papers, Part II, 175 179, 011. ISBN 978--7378-185-9 MATFYZPRESS Association of with at Low Temperatures I. Zymak, P. Jusko, S. Roučka, D. Mulin, R. Plašil, and J. Glosík
WDS'11 Proceedings of Contributed Papers, Part II, 175 179, 011. ISBN 978--7378-185-9 MATFYZPRESS Association of with at Low Temperatures I. Zymak, P. Jusko, S. Roučka, D. Mulin, R. Plašil, and J. Glosík
Mass Spectrometry. What is Mass Spectrometry?
 Mass Spectrometry What is Mass Spectrometry? Mass Spectrometry (MS): The generation of gaseous ions from a sample, separation of these ions by mass-to-charge ratio, and measurement of relative abundance
Mass Spectrometry What is Mass Spectrometry? Mass Spectrometry (MS): The generation of gaseous ions from a sample, separation of these ions by mass-to-charge ratio, and measurement of relative abundance
MS Goals and Applications. MS Goals and Applications
 MS Goals and Applications 3 Several variations on a theme, three common steps Form gas-phase ions choice of ionization method depends on sample identity and information required Separate ions on basis
MS Goals and Applications 3 Several variations on a theme, three common steps Form gas-phase ions choice of ionization method depends on sample identity and information required Separate ions on basis
Secondary Ion Mass Spectrometry (SIMS)
 CHEM53200: Lecture 10 Secondary Ion Mass Spectrometry (SIMS) Major reference: Surface Analysis Edited by J. C. Vickerman (1997). 1 Primary particles may be: Secondary particles can be e s, neutral species
CHEM53200: Lecture 10 Secondary Ion Mass Spectrometry (SIMS) Major reference: Surface Analysis Edited by J. C. Vickerman (1997). 1 Primary particles may be: Secondary particles can be e s, neutral species
Chemistry Instrumental Analysis Lecture 34. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 34 From molecular to elemental analysis there are three major techniques used for elemental analysis: Optical spectrometry Mass spectrometry X-ray spectrometry
Chemistry 4631 Instrumental Analysis Lecture 34 From molecular to elemental analysis there are three major techniques used for elemental analysis: Optical spectrometry Mass spectrometry X-ray spectrometry
X-Rays, Electrons and Lithography: Fundamental Processes in Molecular Radiation Chemistry
 X-Rays, Electrons and Lithography: Fundamental Processes in Molecular Radiation Chemistry D. Frank Ogletree Molecular Foundry, Berkeley Lab Berkeley CA USA Our Berkeley Lab Team EUV Lithography and Pattern
X-Rays, Electrons and Lithography: Fundamental Processes in Molecular Radiation Chemistry D. Frank Ogletree Molecular Foundry, Berkeley Lab Berkeley CA USA Our Berkeley Lab Team EUV Lithography and Pattern
Vacuum-Ultraviolet-Excited and CH 2 Cl 2 /H 2 O-Amplified Ionization- Coupled Mass Spectrometry for Oxygenated Organics Analysis
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 Supporting Information for Vacuum-Ultraviolet-Excited and CH 2 Cl 2 /H 2 O-Amplified Ionization- Coupled Mass Spectrometry
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 Supporting Information for Vacuum-Ultraviolet-Excited and CH 2 Cl 2 /H 2 O-Amplified Ionization- Coupled Mass Spectrometry
DPP06 Meeting of The American Physical Society. Production of negative ion plasmas using perfluoromethylcyclohexane (C 7 F 14 )
 1 POSTER JP1.00100 [Bull. APS 51, 165 (2006)] DPP06 Meeting of The American Physical Society Production of negative ion plasmas using perfluoromethylcyclohexane (C 7 F 14 ) Su-Hyun Kim, Robert Merlino,
1 POSTER JP1.00100 [Bull. APS 51, 165 (2006)] DPP06 Meeting of The American Physical Society Production of negative ion plasmas using perfluoromethylcyclohexane (C 7 F 14 ) Su-Hyun Kim, Robert Merlino,
386 Brazilian Journal of Physics, vol. 30, no. 2, June, 2000 Atomic Transitions for the Doubly Ionized Argon Spectrum, Ar III 1 F. R. T. Luna, 2 F. Br
 386 Brazilian Journal of Physics, vol. 30, no. 2, June, 2000 Atomic Transitions for the Doubly Ionized Argon Spectrum, Ar III 1 F. R. T. Luna, 2 F. Bredice, 3 G. H. Cavalcanti, and 1;4 A. G. Trigueiros,
386 Brazilian Journal of Physics, vol. 30, no. 2, June, 2000 Atomic Transitions for the Doubly Ionized Argon Spectrum, Ar III 1 F. R. T. Luna, 2 F. Bredice, 3 G. H. Cavalcanti, and 1;4 A. G. Trigueiros,
Magic numbers in transition metal Fe, Ti, Zr, Nb, and Ta clusters observed by time-of-flight mass spectrometry
 JOURNAL OF CHEMICAL PHYSICS VOLUME 111, NUMBER 1 1 JULY 1999 Magic numbers in transition metal Fe, Ti, Zr, Nb, and Ta clusters observed by time-of-flight mass spectrometry Masaki Sakurai, Koji Watanabe,
JOURNAL OF CHEMICAL PHYSICS VOLUME 111, NUMBER 1 1 JULY 1999 Magic numbers in transition metal Fe, Ti, Zr, Nb, and Ta clusters observed by time-of-flight mass spectrometry Masaki Sakurai, Koji Watanabe,
MASS SPECTROMETRY. Topics
 MASS SPECTROMETRY MALDI-TOF AND ESI-MS Topics Principle of Mass Spectrometry MALDI-TOF Determination of Mw of Proteins Structural Information by MS: Primary Sequence of a Protein 1 A. Principles Ionization:
MASS SPECTROMETRY MALDI-TOF AND ESI-MS Topics Principle of Mass Spectrometry MALDI-TOF Determination of Mw of Proteins Structural Information by MS: Primary Sequence of a Protein 1 A. Principles Ionization:
M M e M M H M M H. Ion Sources
 Ion Sources Overview of Various Ion Sources After introducing samples into a mass spectrometer, the next important step is the conversion of neutral molecules or compounds to gas phase ions. The ions could
Ion Sources Overview of Various Ion Sources After introducing samples into a mass spectrometer, the next important step is the conversion of neutral molecules or compounds to gas phase ions. The ions could
Photodissociation Dynamics of 4-Aminobenzonitrile + (Water) n Clusters
 4-Aminobenzonitrile Water Clusters Bull. Korean Chem. Soc. 2008, Vol. 29, No. 11 2109 Photodissociation Dynamics of 4-Aminobenzonitrile + (Water) n Clusters Mi Ae Lee, Sang Hwan Nam, Hye Sun Park, Nu Ri
4-Aminobenzonitrile Water Clusters Bull. Korean Chem. Soc. 2008, Vol. 29, No. 11 2109 Photodissociation Dynamics of 4-Aminobenzonitrile + (Water) n Clusters Mi Ae Lee, Sang Hwan Nam, Hye Sun Park, Nu Ri
RECOMMENDATIONS FOR NOMENCLATURE OF MASS SPECTROMETRY
 international UNION OF PURE AND APPLIED CHEMISTRY ANALYTICAL CHEMISTRY DIVISION COMMISSION ON ANALYTICAL NOMENCLATURE RECOMMENDATIONS FOR NOMENCLATURE OF MASS SPECTROMETRY RULES APPROVED 1973 LONDON BUTTER
international UNION OF PURE AND APPLIED CHEMISTRY ANALYTICAL CHEMISTRY DIVISION COMMISSION ON ANALYTICAL NOMENCLATURE RECOMMENDATIONS FOR NOMENCLATURE OF MASS SPECTROMETRY RULES APPROVED 1973 LONDON BUTTER
Ultraviolet-Visible and Infrared Spectrophotometry
 Ultraviolet-Visible and Infrared Spectrophotometry Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451
Ultraviolet-Visible and Infrared Spectrophotometry Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451
Ion induced dissociation of acetylene - butterflies. C.P. Safvan IUAC, New Delhi
 Ion induced dissociation of acetylene - butterflies C.P. Safvan IUAC, New Delhi Ar 8+ + (HCI) (ABC) (ABC) q+ A x+ + B y+ + C z+ where q = x + y + z Molecular structure bond angle, rearrangement Kinematics
Ion induced dissociation of acetylene - butterflies C.P. Safvan IUAC, New Delhi Ar 8+ + (HCI) (ABC) (ABC) q+ A x+ + B y+ + C z+ where q = x + y + z Molecular structure bond angle, rearrangement Kinematics
ARAPUCA light trap for large liquid argon time projection chambers
 ARAPUCA light trap for large liquid argon time projection chambers Author affiliation: Centro Brasileiro de Pesquisas Físicas (CBPF), R. Dr. Xavier Sigaud, 150 - Urca, Rio de Janeiro-RJ, 22290-180, Brazil
ARAPUCA light trap for large liquid argon time projection chambers Author affiliation: Centro Brasileiro de Pesquisas Físicas (CBPF), R. Dr. Xavier Sigaud, 150 - Urca, Rio de Janeiro-RJ, 22290-180, Brazil
Physical Chemistry Chemical Physics
 / Journal Homepage / Table of Contents for this issue PAPER www.rsc.org/pccp Physical Chemistry Chemical Physics Conformation specific and charge directed reactivity of radical cation intermediates of
/ Journal Homepage / Table of Contents for this issue PAPER www.rsc.org/pccp Physical Chemistry Chemical Physics Conformation specific and charge directed reactivity of radical cation intermediates of
Dissociative Photoionization of sulfenylcarbonyl -C(O)S- compounds induced by synchrotron radiation
 INVITED LECTURES REVISTA MEXICANA DE FÍSICA S 56 (2) 24 29 JUNIO 2010 Dissociative Photoionization of sulfenylcarbonyl -C(O)S- compounds induced by synchrotron radiation M. Geronés a, M.F. Erben a, E.
INVITED LECTURES REVISTA MEXICANA DE FÍSICA S 56 (2) 24 29 JUNIO 2010 Dissociative Photoionization of sulfenylcarbonyl -C(O)S- compounds induced by synchrotron radiation M. Geronés a, M.F. Erben a, E.
IR Spectroscopy and physicochemical effects on astrophysical ices produced by energetic ions collisions
 IR Spectroscopy and physicochemical effects on astrophysical ices produced by energetic ions collisions Ana Lucia de Barros Physics Department Centro Federal de Educação Tecnológica Celso Suckow da Fonseca
IR Spectroscopy and physicochemical effects on astrophysical ices produced by energetic ions collisions Ana Lucia de Barros Physics Department Centro Federal de Educação Tecnológica Celso Suckow da Fonseca
FINE PATTERN ETCHING OF SILICON USING SR-ASSISTED IONIZATION OF CF4 GAS
 Technical Paper Journal of Photopolymer Science and Technology Volume 6, Number 4(1993) 617-624 1993TAPJ FINE PATTERN ETCHING OF SILICON USING SR-ASSISTED IONIZATION OF CF4 GAS YASUO NARA, YosHIHto SUGITA,
Technical Paper Journal of Photopolymer Science and Technology Volume 6, Number 4(1993) 617-624 1993TAPJ FINE PATTERN ETCHING OF SILICON USING SR-ASSISTED IONIZATION OF CF4 GAS YASUO NARA, YosHIHto SUGITA,
Studies on the photodissociation and symmetry of SO 2 D
 JOURNAL OF CHEMICAL PHYSICS VOLUME 118, NUMBER 20 22 MAY 2003 Studies on the photodissociation and symmetry of SO 2 D Limin Zhang, a) Zhong Wang, Jiang Li, Feng Wang, Shilin Liu, Shuqin Yu, and Xingxiao
JOURNAL OF CHEMICAL PHYSICS VOLUME 118, NUMBER 20 22 MAY 2003 Studies on the photodissociation and symmetry of SO 2 D Limin Zhang, a) Zhong Wang, Jiang Li, Feng Wang, Shilin Liu, Shuqin Yu, and Xingxiao
Production of HCI with an electron beam ion trap
 Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
X-Ray Photoelectron Spectroscopy (XPS)
 X-Ray Photoelectron Spectroscopy (XPS) Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Fulmer 261A Electron Spectroscopy for Chemical Analysis (ESCA) The basic principle of the photoelectric effect
X-Ray Photoelectron Spectroscopy (XPS) Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Fulmer 261A Electron Spectroscopy for Chemical Analysis (ESCA) The basic principle of the photoelectric effect
An ion source performs the following two functions:
 Ionization The Ion Source An ion source performs the following two functions: 1) converts sample atoms or molecules to ionized particles (ions) in the gas phase (sometimes the task of introducing the atoms
Ionization The Ion Source An ion source performs the following two functions: 1) converts sample atoms or molecules to ionized particles (ions) in the gas phase (sometimes the task of introducing the atoms
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY
 ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
Chapter 5. Mass spectrometry
 ionization and fragmentation Chapter 5. Mass spectrometry which fragmentations? mass and frequency, m/z and count rate Reading: Pavia Chapters 3 and 4 Don t need 3.3 B-D, 3.4 B-D Use the text to clarify
ionization and fragmentation Chapter 5. Mass spectrometry which fragmentations? mass and frequency, m/z and count rate Reading: Pavia Chapters 3 and 4 Don t need 3.3 B-D, 3.4 B-D Use the text to clarify
LIQUID CHROMATOGRAPHY-MASS SPECTROMETRY (LC/MS) Presented by: Dr. T. Nageswara Rao M.Pharm PhD KTPC
 LIQUID CHROMATOGRAPHY-MASS SPECTROMETRY (LC/MS) Presented by: Dr. T. Nageswara Rao M.Pharm PhD KTPC INTRODUCTION Principle: LC/MS is a technique that combines physical separation capabilities of liquid
LIQUID CHROMATOGRAPHY-MASS SPECTROMETRY (LC/MS) Presented by: Dr. T. Nageswara Rao M.Pharm PhD KTPC INTRODUCTION Principle: LC/MS is a technique that combines physical separation capabilities of liquid
Mass Spectrometry. Introduction EI-MS and CI-MS Molecular mass & formulas Principles of fragmentation Fragmentation patterns Isotopic effects
 Mass Spectrometry Introduction EI-MS and CI-MS Molecular mass & formulas Principles of fragmentation Fragmentation patterns Isotopic effects 1 Introduction to MS Mass spectrometry is the method of analysis
Mass Spectrometry Introduction EI-MS and CI-MS Molecular mass & formulas Principles of fragmentation Fragmentation patterns Isotopic effects 1 Introduction to MS Mass spectrometry is the method of analysis
ELECTRON IMPACT IONIZATION OF HELIUM [(e,2e) & (e,3e)] INVESTIGATED WITH COLD TARGET RECOIL-ION MOMENTUM SPECTROSCOPY
![ELECTRON IMPACT IONIZATION OF HELIUM [(e,2e) & (e,3e)] INVESTIGATED WITH COLD TARGET RECOIL-ION MOMENTUM SPECTROSCOPY ELECTRON IMPACT IONIZATION OF HELIUM [(e,2e) & (e,3e)] INVESTIGATED WITH COLD TARGET RECOIL-ION MOMENTUM SPECTROSCOPY](/thumbs/71/65949100.jpg) ELECTRON IMPACT IONIZATION OF HELIUM [(e,2e) & (e,3e)] INVESTIGATED WITH COLD TARGET RECOIL-ION MOMENTUM SPECTROSCOPY E. Erturk, 1 L. Spielberger, 1 M. Achler, 1 L. Schmidt, 1 R. Dorner, 1 Th. Weber, 1
ELECTRON IMPACT IONIZATION OF HELIUM [(e,2e) & (e,3e)] INVESTIGATED WITH COLD TARGET RECOIL-ION MOMENTUM SPECTROSCOPY E. Erturk, 1 L. Spielberger, 1 M. Achler, 1 L. Schmidt, 1 R. Dorner, 1 Th. Weber, 1
Spectroscopic Investigation of Polycyclic Aromatic Hydrocarbons Trapped in Liquid Helium Clusters
 Spectroscopic Investigation of Polycyclic Aromatic Hydrocarbons Trapped in Liquid Helium Clusters Friedrich Huisken and Serge Krasnokutski Max-Planck-Institut für Strömungsforschung, Bunsenstr. 10, D-37073
Spectroscopic Investigation of Polycyclic Aromatic Hydrocarbons Trapped in Liquid Helium Clusters Friedrich Huisken and Serge Krasnokutski Max-Planck-Institut für Strömungsforschung, Bunsenstr. 10, D-37073
Investigation of Coherent Emission from the NSLS VUV Ring
 SPIE Accelerator Based Infrared Sources and Spectroscopic Applications Proc. 3775, 88 94 (1999) Investigation of Coherent Emission from the NSLS VUV Ring G.L. Carr, R.P.S.M. Lobo, J.D. LaVeigne, D.H. Reitze,
SPIE Accelerator Based Infrared Sources and Spectroscopic Applications Proc. 3775, 88 94 (1999) Investigation of Coherent Emission from the NSLS VUV Ring G.L. Carr, R.P.S.M. Lobo, J.D. LaVeigne, D.H. Reitze,
PHYS 3446 Lecture #12
 PHYS 3446 Lecture #12 Wednesday, Oct. 18, 2006 Dr. 1. Particle Detection Ionization Detectors MWPC Scintillation Counters Time of Flight 1 Announcements Next LPCC Workshop Preparation work Each group to
PHYS 3446 Lecture #12 Wednesday, Oct. 18, 2006 Dr. 1. Particle Detection Ionization Detectors MWPC Scintillation Counters Time of Flight 1 Announcements Next LPCC Workshop Preparation work Each group to
Towards using molecular ions as qubits: Femtosecond control of molecular fragmentation with multiple knobs
 PRAMANA c Indian Academy of Sciences Vol. 75, No. 6 journal of December 2010 physics pp. 1065 1069 Towards using molecular ions as qubits: Femtosecond control of molecular fragmentation with multiple knobs
PRAMANA c Indian Academy of Sciences Vol. 75, No. 6 journal of December 2010 physics pp. 1065 1069 Towards using molecular ions as qubits: Femtosecond control of molecular fragmentation with multiple knobs
Lecture 15: Introduction to mass spectrometry-i
 Lecture 15: Introduction to mass spectrometry-i Mass spectrometry (MS) is an analytical technique that measures the mass/charge ratio of charged particles in vacuum. Mass spectrometry can determine masse/charge
Lecture 15: Introduction to mass spectrometry-i Mass spectrometry (MS) is an analytical technique that measures the mass/charge ratio of charged particles in vacuum. Mass spectrometry can determine masse/charge
(2) Read each statement carefully and pick the one that is incorrect in its information.
 Organic Chemistry - Problem Drill 17: IR and Mass Spectra No. 1 of 10 1. Which statement about infrared spectroscopy is incorrect? (A) IR spectroscopy is a method of structure determination based on the
Organic Chemistry - Problem Drill 17: IR and Mass Spectra No. 1 of 10 1. Which statement about infrared spectroscopy is incorrect? (A) IR spectroscopy is a method of structure determination based on the
Mass Analyzers. Principles of the three most common types magnetic sector, quadrupole and time of flight - will be discussed herein.
 Mass Analyzers After the production of ions in ion sources, the next critical step in mass spectrometry is to separate these gas phase ions according to their mass-to-charge ratio (m/z). Ions are extracted
Mass Analyzers After the production of ions in ion sources, the next critical step in mass spectrometry is to separate these gas phase ions according to their mass-to-charge ratio (m/z). Ions are extracted
Ch. 9 Atomic Absorption & Atomic Fluorescence Spectrometry
 Ch. 9 Atomic Absorption & Atomic Fluorescence Spectrometry 9.1 9A. Atomization Most fundamental for both techniques. Typical types 1. flame - burner type 2. Electrothermal graphite furnace 3. Specialized
Ch. 9 Atomic Absorption & Atomic Fluorescence Spectrometry 9.1 9A. Atomization Most fundamental for both techniques. Typical types 1. flame - burner type 2. Electrothermal graphite furnace 3. Specialized
DISSOCIATIVE IONIZATION OF CARBON DISULPHIDE IN THE GAS PHASE. HEAT OF FORMATION OF THE CS RADICAL
 DISSOCIATIVE IONIZATION OF CARBON DISULPHIDE IN THE GAS PHASE. HEAT OF FORMATION OF THE CS RADICAL M.-J. HUBIN-FRANSKIN 1, R. LOCHT and J. KATIHABWA Institut de Chimie, Université de Liège, Sart Tilman
DISSOCIATIVE IONIZATION OF CARBON DISULPHIDE IN THE GAS PHASE. HEAT OF FORMATION OF THE CS RADICAL M.-J. HUBIN-FRANSKIN 1, R. LOCHT and J. KATIHABWA Institut de Chimie, Université de Liège, Sart Tilman
1 Electrons are emitted from a metal surface when it is illuminated with suitable electromagnetic radiation. ...[1]
![1 Electrons are emitted from a metal surface when it is illuminated with suitable electromagnetic radiation. ...[1] 1 Electrons are emitted from a metal surface when it is illuminated with suitable electromagnetic radiation. ...[1]](/thumbs/81/83978040.jpg) 1 Electrons are emitted from a metal surface when it is illuminated with suitable electromagnetic radiation. 1 (a) (b) Name the effect described above....[1] The variation with frequency f of the maximum
1 Electrons are emitted from a metal surface when it is illuminated with suitable electromagnetic radiation. 1 (a) (b) Name the effect described above....[1] The variation with frequency f of the maximum
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY
 ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
Energy Spectroscopy. Ex.: Fe/MgO
 Energy Spectroscopy Spectroscopy gives access to the electronic properties (and thus chemistry, magnetism,..) of the investigated system with thickness dependence Ex.: Fe/MgO Fe O Mg Control of the oxidation
Energy Spectroscopy Spectroscopy gives access to the electronic properties (and thus chemistry, magnetism,..) of the investigated system with thickness dependence Ex.: Fe/MgO Fe O Mg Control of the oxidation
CHEM 241 UNIT 5: PART A DETERMINATION OF ORGANIC STRUCTURES BY SPECTROSCOPIC METHODS [MASS SPECTROMETRY]
![CHEM 241 UNIT 5: PART A DETERMINATION OF ORGANIC STRUCTURES BY SPECTROSCOPIC METHODS [MASS SPECTROMETRY] CHEM 241 UNIT 5: PART A DETERMINATION OF ORGANIC STRUCTURES BY SPECTROSCOPIC METHODS [MASS SPECTROMETRY]](/thumbs/83/88348834.jpg) CHEM 241 UNIT 5: PART A DETERMINATION OF ORGANIC STRUCTURES BY SPECTROSCOPIC METHODS [MASS SPECTROMETRY] 1 Introduction Outline Mass spectrometry (MS) 2 INTRODUCTION The analysis of the outcome of a reaction
CHEM 241 UNIT 5: PART A DETERMINATION OF ORGANIC STRUCTURES BY SPECTROSCOPIC METHODS [MASS SPECTROMETRY] 1 Introduction Outline Mass spectrometry (MS) 2 INTRODUCTION The analysis of the outcome of a reaction
(Refer Slide Time 00:09) (Refer Slide Time 00:13)
 (Refer Slide Time 00:09) Mass Spectrometry Based Proteomics Professor Sanjeeva Srivastava Department of Biosciences and Bioengineering Indian Institute of Technology, Bombay Mod 02 Lecture Number 09 (Refer
(Refer Slide Time 00:09) Mass Spectrometry Based Proteomics Professor Sanjeeva Srivastava Department of Biosciences and Bioengineering Indian Institute of Technology, Bombay Mod 02 Lecture Number 09 (Refer
PHOTOELECTRON SPECTROSCOPY IN AIR (PESA)
 PHOTOELECTRON SPECTROSCOPY IN AIR (PESA) LEADERS IN GAS DETECTION Since 1977 Model AC-3 Features: Atmospheric pressure operation (unique in the world) Estimate work function, ionization potential, density
PHOTOELECTRON SPECTROSCOPY IN AIR (PESA) LEADERS IN GAS DETECTION Since 1977 Model AC-3 Features: Atmospheric pressure operation (unique in the world) Estimate work function, ionization potential, density
= : K A
 Atoms and Nuclei. State two limitations of JJ Thomson s model of atom. 2. Write the SI unit for activity of a radioactive substance. 3. What observations led JJ Thomson to conclusion that all atoms have
Atoms and Nuclei. State two limitations of JJ Thomson s model of atom. 2. Write the SI unit for activity of a radioactive substance. 3. What observations led JJ Thomson to conclusion that all atoms have
CHEM*3440. X-Ray Energies. Bremsstrahlung Radiation. X-ray Line Spectra. Chemical Instrumentation. X-Ray Spectroscopy. Topic 13
 X-Ray Energies very short wavelength radiation 0.1Å to 10 nm (100 Å) CHEM*3440 Chemical Instrumentation Topic 13 X-Ray Spectroscopy Visible - Ultraviolet (UV) - Vacuum UV (VUV) - Extreme UV (XUV) - Soft
X-Ray Energies very short wavelength radiation 0.1Å to 10 nm (100 Å) CHEM*3440 Chemical Instrumentation Topic 13 X-Ray Spectroscopy Visible - Ultraviolet (UV) - Vacuum UV (VUV) - Extreme UV (XUV) - Soft
MS Goals and Applications. MS Goals and Applications
 MS Goals and Applications 1 Several variations on a theme, three common steps Form gas-phase ions choice of ionization method depends on sample identity and information required Separate ions on basis
MS Goals and Applications 1 Several variations on a theme, three common steps Form gas-phase ions choice of ionization method depends on sample identity and information required Separate ions on basis
Atomic Emission Spectroscopy
 Atomic Emission Spectroscopy Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451 Saudi Arabia Building:
Atomic Emission Spectroscopy Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451 Saudi Arabia Building:
An Investigation of Benzene Using Ultrafast Laser Spectroscopy. Ryan Barnett. The Ohio State University
 An Investigation of Benzene Using Ultrafast Laser Spectroscopy Ryan Barnett The Ohio State University NSF/REU/OSU Advisor: Linn Van Woerkom Introduction Molecular spectroscopy has been used throughout
An Investigation of Benzene Using Ultrafast Laser Spectroscopy Ryan Barnett The Ohio State University NSF/REU/OSU Advisor: Linn Van Woerkom Introduction Molecular spectroscopy has been used throughout
Application of Surface Analysis for Root Cause Failure Analysis
 Application of Surface Analysis for Root Cause Failure Analysis David A. Cole Evans Analytical Group East Windsor, NJ Specialists in Materials Characterization Outline Introduction X-Ray Photoelectron
Application of Surface Analysis for Root Cause Failure Analysis David A. Cole Evans Analytical Group East Windsor, NJ Specialists in Materials Characterization Outline Introduction X-Ray Photoelectron
Characterization of the operation of RITs with iodine
 Characterization of the operation of RITs with iodine IEPC-2017-368 Presented at the 35th International Electric Propulsion Conference Georgia Institute of Technology Atlanta, Georgia USA Waldemar Gärtner
Characterization of the operation of RITs with iodine IEPC-2017-368 Presented at the 35th International Electric Propulsion Conference Georgia Institute of Technology Atlanta, Georgia USA Waldemar Gärtner
Current Research and Future Plans
 Current Research and Future Plans Bhas Bapat PRL, Ahmedbad At IISER Pune, Jan 2014 Bhas Bapat (PRL, Ahmedbad) Current Research and Future Plans At IISER Pune, Jan 2014 1 / 21 Contents Broad Areas Dissociative
Current Research and Future Plans Bhas Bapat PRL, Ahmedbad At IISER Pune, Jan 2014 Bhas Bapat (PRL, Ahmedbad) Current Research and Future Plans At IISER Pune, Jan 2014 1 / 21 Contents Broad Areas Dissociative
Mass Spectrometry and Proteomics - Lecture 2 - Matthias Trost Newcastle University
 Mass Spectrometry and Proteomics - Lecture 2 - Matthias Trost Newcastle University matthias.trost@ncl.ac.uk Previously: Resolution and other basics MALDI Electrospray 40 Lecture 2 Mass analysers Detectors
Mass Spectrometry and Proteomics - Lecture 2 - Matthias Trost Newcastle University matthias.trost@ncl.ac.uk Previously: Resolution and other basics MALDI Electrospray 40 Lecture 2 Mass analysers Detectors
Electronic and vibrational spectra of aniline benzene heterodimer and aniline homo-dimer ions
 Electronic and vibrational spectra of aniline benzene heterodimer and aniline homo-dimer ions Kazuhiko Ohashi a,*, Yoshiya Inokuchi b, Hironobu Izutsu a, Kazuyuki Hino a, Norifumi Yamamoto a, Nobuyuki
Electronic and vibrational spectra of aniline benzene heterodimer and aniline homo-dimer ions Kazuhiko Ohashi a,*, Yoshiya Inokuchi b, Hironobu Izutsu a, Kazuyuki Hino a, Norifumi Yamamoto a, Nobuyuki
Chapter 37 Early Quantum Theory and Models of the Atom. Copyright 2009 Pearson Education, Inc.
 Chapter 37 Early Quantum Theory and Models of the Atom Planck s Quantum Hypothesis; Blackbody Radiation Photon Theory of Light and the Photoelectric Effect Energy, Mass, and Momentum of a Photon Compton
Chapter 37 Early Quantum Theory and Models of the Atom Planck s Quantum Hypothesis; Blackbody Radiation Photon Theory of Light and the Photoelectric Effect Energy, Mass, and Momentum of a Photon Compton
Time of Flight Mass Spectrometry of Ions Generated by Molecules in Intense Laser Fields
 Time of Flight Mass Spectrometry of Ions Generated by Molecules in Intense Laser Fields Mingtong Han 5 th August, 013 Abstract Photoionization of acetylene, dimethylacetylene and methylacetylene in an
Time of Flight Mass Spectrometry of Ions Generated by Molecules in Intense Laser Fields Mingtong Han 5 th August, 013 Abstract Photoionization of acetylene, dimethylacetylene and methylacetylene in an
AMO at FLASH. FELs provide unique opportunities and challenges for AMO physics. due to essentially three reasons:
 Experience at FLASH AMO at FLASH FELs provide unique opportunities and challenges for AMO physics due to essentially three reasons: AMO at FLASH 1. huge integrated flux dilute samples Highly charged ions
Experience at FLASH AMO at FLASH FELs provide unique opportunities and challenges for AMO physics due to essentially three reasons: AMO at FLASH 1. huge integrated flux dilute samples Highly charged ions
Introduction to Synchrotron Radiation
 Introduction to Synchrotron Radiation Frederico Alves Lima Centro Nacional de Pesquisa em Energia e Materiais - CNPEM Laboratório Nacional de Luz Síncrotron - LNLS International School on Laser-Beam Interactions
Introduction to Synchrotron Radiation Frederico Alves Lima Centro Nacional de Pesquisa em Energia e Materiais - CNPEM Laboratório Nacional de Luz Síncrotron - LNLS International School on Laser-Beam Interactions
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K. Sharma Department of Biotechnology Indian Institute of Technology, Roorkee
 Analytical Technologies in Biotechnology Prof. Dr. Ashwani K. Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 6 Spectroscopic Techniques Lecture - 6 Atomic Spectroscopy
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K. Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 6 Spectroscopic Techniques Lecture - 6 Atomic Spectroscopy
