Hypermetallic molecules
|
|
|
- Cathleen Wiggins
- 5 years ago
- Views:
Transcription
1 1 Hypermetallic molecules (Picture courtesy of Python M)
2 2 An ab initio study of the alkaline earth oxides BeOBe, MgOMg, CaOCa, SrOSr, BaOBa, and RaORa. Bojana Ostojić (Belgrade), Per Jensen (Wuppertal), Peter Schwerdtfeger (Massey University, New Zealand), Phil Bunker (NRC, Ottawa). Hypermetallic since they have one more metal atom than they should
3 3 PERIODIC TABLE OF ELEMENTS [He]2s 2 [Ne]3s 2 [Ar]4s 2 Chemistry 1.0.1: M 2+ O 2- is formed! Why bonding to another M? [Rn]7s 2 MOM?
4 4 PERIODIC TABLE OF ELEMENTS [He]2s 2 [Ne]3s 2 Chemistry 1.0.1: M 2+ O 2- is formed! Why bonding to another M? [Ar]4s 2 Chemistry 1.0.2: Because MO is closer to M + O - [Rn]7s 2 MOM OK!
5 5 Apart from BeOBe, little known experimentally. Our interest in these molecules is three-fold: To make precise ab initio calculations on them in order to understand the electronic structure, To predict the IR and electronic spectra and to compare with the experimental spectra, To calculate Singlet-Triplet splittings and S-T interaction strengths in order to test these molecules as candidates for high precision spectral measurements aimed at looking for a time-variation in M p /m e and fine structure constant.
6 6 The ab initio slide Please direct questions to Molecule Method Basis set BeOBe CASSCF MRCI cc-pcvqz MgOMg FV-CASSCF MRCISD cc-pcvqz CaOCa FV-CASSCF MRCISD+RS2C cc-pwcvqz-dk SrOSr CASSCF MRCISD+RS2C Sadlej pvtz and Stuttgart relativistic small-core effective core potential for Sr BaOBa and RaORa CASSCF MRCI cc-pcvqz and Stuttgart small-core relativistic effective core potential ECP46MDF and ECP78MDF for Ba and Ra Nuclear-motion calculations and spectral simulations with MORBID
7 7 BeOBe, MgOMg, CaOCa, SrOSr, BaOBa, and RaORa. We find that all these MOM molecules have 1 + A linear Σg ground electronic state and a fairly low lying 3 + linear Σu first excited electronic state. S-T spin-orbit coupling too small for them to be good candidates for use in measuring the time dependence of the fundamental parameters.
8 8 BeOBe bending potential curves
9 9 BeOBe exp Experiment: Merritt, Bondybey and Heaven, JPC A113, (2009)
10 10 BeOBe Singlet spectrum Triplet spectrum At 300 K. All bands very weak
11 11 MgOMg ~ T e (a) = 671 cm -1 Diatomic 24 Mg 16 O cm -1
12 12 MgOMg 420 nm 3.7 D 310 nm 4.0 D 360 nm 4.1 D 340 nm 2.9 D
13 13 MgOMg From Discussion:
14 14 MgOMg A 1 + Σ u E 1 Π u X X 420 nm 310 nm b 3 Π g c 3 Π g a a 360 nm 340 nm
15 15 CaOCa Also think that spectrum shows linear CaCaO molecule
16 16 CaCaO (linear) Ca Ca O bent 40 Ca+ 16 O 3 44 Ca+ 16 O 3 CaO O , , Ca 16 O υ = cm -1 in gas phase υ = 707 cm -1 in N 2 matrix. 44 Ca+55% 18 O , 461 Our results for CaOCa in the X 1 + Σ g state: 44 Ca+55% 18 O 2 In solid N 2 at 17 K 720 υ 1 υ 2 υ
17 17 SrOSr singlettriplet interaction S-T interaction? ΔE ~ 2 cm -1 What is S-T matrix element? even J/odd J even N/odd N
18 18 SrOSr ST matrix element = = 0.01 cm -1 a ~ 0 0 ~ X 7 1 ΔE ~ 2 cm -1 Thus energy shift ~ (0.01) 2 /2 cm -1 = cm -1
19 19 MOM equilibrium bond length S-T splitting
20 20 BaOBa bending potential energy curves Ba 0.5+ O - Ba nm 910 nm Predicted emission wavelengths
21 21 BaOBa Gee et al. The cluster isolated chemical reaction technique. West and Poland JCP 66, 2139 (1977) Triplet Emission ~700 nm Singlet Emission ~900 nm
22 22 Spring has arrived in Wuppertal! Thank you for your attention!
FACULTY OF MATHEMATICS AND NATURAL SCIENCES PHYSICAL AND THEORETICAL CHEMISTRY. (Picture courtesy of Python M)
 1 (Picture courtesy of Python M) 2 Theoretical spectra of CaOCa in the two lowest electronic states B. Ostojić, University of Belgrade P. R. Bunker, Massey University, Auckland P. Schwerdtfeger, Massey
1 (Picture courtesy of Python M) 2 Theoretical spectra of CaOCa in the two lowest electronic states B. Ostojić, University of Belgrade P. R. Bunker, Massey University, Auckland P. Schwerdtfeger, Massey
5 questions, 3 points each, 15 points total possible. 26 Fe Cu Ni Co Pd Ag Ru 101.
 Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Speed of light c = m/s. x n e a x d x = 1. 2 n+1 a n π a. He Li Ne Na Ar K Ni 58.
 Physical Chemistry II Test Name: KEY CHEM 464 Spring 18 Chapters 7-11 Average = 1. / 16 6 questions worth a total of 16 points Planck's constant h = 6.63 1-34 J s Speed of light c = 3. 1 8 m/s ħ = h π
Physical Chemistry II Test Name: KEY CHEM 464 Spring 18 Chapters 7-11 Average = 1. / 16 6 questions worth a total of 16 points Planck's constant h = 6.63 1-34 J s Speed of light c = 3. 1 8 m/s ħ = h π
Investigation of Spectroscopic Properties and Spin-Orbit Splitting in the X 2 Π and A 2 Π Electronic States of the SO + Cation
 Int. J. Mol. Sci. 2012, 13, 8189-8209; doi:10.3390/ijms13078189 Article OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Investigation of Spectroscopic Properties
Int. J. Mol. Sci. 2012, 13, 8189-8209; doi:10.3390/ijms13078189 Article OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Investigation of Spectroscopic Properties
Calculation of excitation energies for heavy-element systems
 Calculation of excitation energies for heavy-element systems Stefan Knecht ETH Zürich, Laboratorium für Physikalische Chemie, Switzerland http://www.reiher.ethz.ch/people/knechste stefan.knecht@phys.chem.ethz.ch
Calculation of excitation energies for heavy-element systems Stefan Knecht ETH Zürich, Laboratorium für Physikalische Chemie, Switzerland http://www.reiher.ethz.ch/people/knechste stefan.knecht@phys.chem.ethz.ch
Calculations of band structures
 Chemistry and Physics at Albany Planning for the Future Calculations of band structures using wave-function based correlation methods Elke Pahl Centre of Theoretical Chemistry and Physics Institute of
Chemistry and Physics at Albany Planning for the Future Calculations of band structures using wave-function based correlation methods Elke Pahl Centre of Theoretical Chemistry and Physics Institute of
CHAPTER 13 Molecular Spectroscopy 2: Electronic Transitions
 CHAPTER 13 Molecular Spectroscopy 2: Electronic Transitions I. General Features of Electronic spectroscopy. A. Visible and ultraviolet photons excite electronic state transitions. ε photon = 120 to 1200
CHAPTER 13 Molecular Spectroscopy 2: Electronic Transitions I. General Features of Electronic spectroscopy. A. Visible and ultraviolet photons excite electronic state transitions. ε photon = 120 to 1200
Relativistic and correlated calculations on the ground, excited, and ionized states of iodine
 Relativistic and correlated calculations on the ground, excited, and ionized states of iodine W. A. de Jong, L. Visscher, a) and W. C. Nieuwpoort Laboratory for Chemical Physics and Materials Science Centre,
Relativistic and correlated calculations on the ground, excited, and ionized states of iodine W. A. de Jong, L. Visscher, a) and W. C. Nieuwpoort Laboratory for Chemical Physics and Materials Science Centre,
... but using electron configurations to describe how aluminum bromide forms is a bit cumbersome! Can we simplify the picture a bit?
 193... but using electron configurations to describe how aluminum bromide forms is a bit cumbersome! Can we simplify the picture a bit? LEWIS NOTATION / ELECTRON-DOT NOTATION - Lewis notation represents
193... but using electron configurations to describe how aluminum bromide forms is a bit cumbersome! Can we simplify the picture a bit? LEWIS NOTATION / ELECTRON-DOT NOTATION - Lewis notation represents
Chemistry 213 Practical Spectroscopy
 Chemistry 213 Practical Spectroscopy Dave Berg djberg@uvic.ca Elliott 314 A course in determining structure by spectroscopic methods Different types of spectroscopy afford different information about molecules
Chemistry 213 Practical Spectroscopy Dave Berg djberg@uvic.ca Elliott 314 A course in determining structure by spectroscopic methods Different types of spectroscopy afford different information about molecules
Diamagnetism and Paramagnetism in Atoms and Molecules
 Diamagnetism and Paramagnetism in Atoms and Molecules Trygve Helgaker Alex Borgoo, Maria Dimitrova, Jürgen Gauss, Florian Hampe, Christof Holzer, Wim Klopper, Trond Saue, Peter Schwerdtfeger, Stella Stopkowicz,
Diamagnetism and Paramagnetism in Atoms and Molecules Trygve Helgaker Alex Borgoo, Maria Dimitrova, Jürgen Gauss, Florian Hampe, Christof Holzer, Wim Klopper, Trond Saue, Peter Schwerdtfeger, Stella Stopkowicz,
Topic 3: Periodicity OBJECTIVES FOR TODAY: Fall in love with the Periodic Table, Interpret trends in atomic radii, ionic radii, ionization energies &
 Topic 3: Periodicity OBJECTIVES FOR TODAY: Fall in love with the Periodic Table, Interpret trends in atomic radii, ionic radii, ionization energies & electronegativity The Periodic Table What is the periodic
Topic 3: Periodicity OBJECTIVES FOR TODAY: Fall in love with the Periodic Table, Interpret trends in atomic radii, ionic radii, ionization energies & electronegativity The Periodic Table What is the periodic
Theoretical study of spectroscopic parameters of alkali -Al and alkaline earth-al dimers
 Theor Chem Account (2008) 121:165 172 DOI 10.1007/s00214-008-0460-5 REGULAR ARTICLE Theoretical study of spectroscopic parameters of alkali -Al and alkaline earth-al dimers Jianchuan Wang Deming Zhai Fei
Theor Chem Account (2008) 121:165 172 DOI 10.1007/s00214-008-0460-5 REGULAR ARTICLE Theoretical study of spectroscopic parameters of alkali -Al and alkaline earth-al dimers Jianchuan Wang Deming Zhai Fei
Physical Chemistry Lab II CHEM 4644 Spring 2011 Final Exam 5 questions at 3 points each equals 15 total points possible.
 Physical Chemistry Lab II Name: KEY CHEM 4644 Spring 2011 Final Exam 5 questions at 3 points each equals 15 total points possible. Constants: c = 3.00 10 8 m/s h = 6.63 10-34 J s 1 Hartree = 4.36 10-18
Physical Chemistry Lab II Name: KEY CHEM 4644 Spring 2011 Final Exam 5 questions at 3 points each equals 15 total points possible. Constants: c = 3.00 10 8 m/s h = 6.63 10-34 J s 1 Hartree = 4.36 10-18
Atomic Structure and Atomic Spectra
 Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
ATMO 551a Fall Resonant Electromagnetic (EM) Interactions in Planetary atmospheres. Electron transition between different electron orbits
 Resonant Electromagnetic (EM) Interactions in Planetary atmospheres There are three classes of energy states that interact with EM radiation that we are interested in to understand how light (EM radiation)
Resonant Electromagnetic (EM) Interactions in Planetary atmospheres There are three classes of energy states that interact with EM radiation that we are interested in to understand how light (EM radiation)
Quantum chemistry and vibrational spectra
 Chapter 3 Quantum chemistry and vibrational spectra This chapter presents the quantum chemical results for the systems studied in this work, FHF (Section 3.) and OHF (Section 3.3). These triatomic anions
Chapter 3 Quantum chemistry and vibrational spectra This chapter presents the quantum chemical results for the systems studied in this work, FHF (Section 3.) and OHF (Section 3.3). These triatomic anions
5.80 Small-Molecule Spectroscopy and Dynamics
 MIT OpenCourseWare http://ocw.mit.edu 5.80 Small-Molecule Spectroscopy and Dynamics Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.80 Lecture
MIT OpenCourseWare http://ocw.mit.edu 5.80 Small-Molecule Spectroscopy and Dynamics Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.80 Lecture
Lecture 6: Physical Methods II. UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy
 Lecture 6: Physical Methods II UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy Physical Methods used in bioinorganic chemistry X ray crystallography X ray absorption (XAS)
Lecture 6: Physical Methods II UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy Physical Methods used in bioinorganic chemistry X ray crystallography X ray absorption (XAS)
PAPER No. : 8 (PHYSICAL SPECTROSCOPY) MODULE NO. : 23 (NORMAL MODES AND IRREDUCIBLE REPRESENTATIONS FOR POLYATOMIC MOLECULES)
 Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 23/ Normal modes and irreducible representations for polyatomic molecules CHE_P8_M23 TABLE OF CONTENTS 1. Learning
Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 23/ Normal modes and irreducible representations for polyatomic molecules CHE_P8_M23 TABLE OF CONTENTS 1. Learning
CHEM 130 Exp. 8: Molecular Models
 CHEM 130 Exp. 8: Molecular Models In this lab, we will learn and practice predicting molecular structures from molecular formulas. The Periodic Table of the Elements IA 1 H IIA IIIA IVA VA VIA VIIA 3 5
CHEM 130 Exp. 8: Molecular Models In this lab, we will learn and practice predicting molecular structures from molecular formulas. The Periodic Table of the Elements IA 1 H IIA IIIA IVA VA VIA VIIA 3 5
CHEMISTRY 110 EXAM 1 SEPTEMBER 20, 2010 FORM A
 CHEMISTRY 110 EXAM 1 SEPTEMBER 20, 2010 FORM A 1. What are the correct numbers of protons, neutrons and electrons in a 39 K + ion? p n e A. 20 19 18 B. 20 19 19 C. 19 20 18 D. 19 20 19 E. 20 19 20 2. Which
CHEMISTRY 110 EXAM 1 SEPTEMBER 20, 2010 FORM A 1. What are the correct numbers of protons, neutrons and electrons in a 39 K + ion? p n e A. 20 19 18 B. 20 19 19 C. 19 20 18 D. 19 20 19 E. 20 19 20 2. Which
"What Do I Remember From Introductory Chemistry?" - A Problem Set
 1 "What Do I Remember From Introductory Chemistry?" - A Problem Set These problems review a portion of the material from introductory chemistry that you should be very familiar with. 1. The electron configuration
1 "What Do I Remember From Introductory Chemistry?" - A Problem Set These problems review a portion of the material from introductory chemistry that you should be very familiar with. 1. The electron configuration
Chemistry 543--Final Exam--Keiderling May 5, pm SES
 Chemistry 543--Final Exam--Keiderling May 5,1992 -- 1-5pm -- 174 SES Please answer all questions in the answer book provided. Make sure your name is clearly indicated and that the answers are clearly numbered,
Chemistry 543--Final Exam--Keiderling May 5,1992 -- 1-5pm -- 174 SES Please answer all questions in the answer book provided. Make sure your name is clearly indicated and that the answers are clearly numbered,
Lecture 9 Electronic Spectroscopy
 Lecture 9 Electronic Spectroscopy Molecular Orbital Theory: A Review - LCAO approximaton & AO overlap - Variation Principle & Secular Determinant - Homonuclear Diatomic MOs - Energy Levels, Bond Order
Lecture 9 Electronic Spectroscopy Molecular Orbital Theory: A Review - LCAO approximaton & AO overlap - Variation Principle & Secular Determinant - Homonuclear Diatomic MOs - Energy Levels, Bond Order
Spectroscopy of the Low Lying States of CaO +
 Spectroscopy of the Low Lying States of CaO + 7 nd Interna+onal Symposium on Molecular Spectroscopy FE 09 July 3, 017 Robert A. VanGundy, Michael C. Heaven CaO + Mo8va8on Laser Cooling and Ion Trapping
Spectroscopy of the Low Lying States of CaO + 7 nd Interna+onal Symposium on Molecular Spectroscopy FE 09 July 3, 017 Robert A. VanGundy, Michael C. Heaven CaO + Mo8va8on Laser Cooling and Ion Trapping
Experiment 2 - NMR Spectroscopy
 Experiment 2 - NMR Spectroscopy OBJECTIVE to understand the important role of nuclear magnetic resonance spectroscopy in the study of the structures of organic compounds to develop an understanding of
Experiment 2 - NMR Spectroscopy OBJECTIVE to understand the important role of nuclear magnetic resonance spectroscopy in the study of the structures of organic compounds to develop an understanding of
- A CHEMICAL BOND is a strong attractive force between the atoms in a compound. attractive forces between oppositely charged ions
 191 CHEMICAL BONDS - A CHEMICAL BOND is a strong attractive force between the atoms in a compound. 3 TYPES OF CHEMICAL BOND Ionic bonds attractive forces between oppositely charged ions sodium chloride
191 CHEMICAL BONDS - A CHEMICAL BOND is a strong attractive force between the atoms in a compound. 3 TYPES OF CHEMICAL BOND Ionic bonds attractive forces between oppositely charged ions sodium chloride
- Light has properties of WAVES such as DIFFRACTION (it bends around small obstructions).
 170 LIGHT wavelength Diffraction frequency = wavelengths / time = - Light has properties of WAVES such as DIFFRACTION (it bends around small obstructions). - Einstein noted that viewing light as a particle
170 LIGHT wavelength Diffraction frequency = wavelengths / time = - Light has properties of WAVES such as DIFFRACTION (it bends around small obstructions). - Einstein noted that viewing light as a particle
Lecture 6 - spectroscopy
 Lecture 6 - spectroscopy 1 Light Electromagnetic radiation can be thought of as either a wave or as a particle (particle/wave duality). For scattering of light by particles, air, and surfaces, wave theory
Lecture 6 - spectroscopy 1 Light Electromagnetic radiation can be thought of as either a wave or as a particle (particle/wave duality). For scattering of light by particles, air, and surfaces, wave theory
(FIRST) IONIZATION ENERGY
 181 (FIRST) IONIZATION ENERGY - The amount of energy required to remove a single electron from the outer shell of an atom. - Relates to reactivity for metals. The easier it is to remove an electron, the
181 (FIRST) IONIZATION ENERGY - The amount of energy required to remove a single electron from the outer shell of an atom. - Relates to reactivity for metals. The easier it is to remove an electron, the
NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY
 NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY A STUDENT SHOULD BE ABLE TO: 1. Identify and explain the processes involved in proton ( 1 H) and carbon-13 ( 13 C) nuclear magnetic resonance
NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY A STUDENT SHOULD BE ABLE TO: 1. Identify and explain the processes involved in proton ( 1 H) and carbon-13 ( 13 C) nuclear magnetic resonance
Born-Oppenheimer Approximation
 Born-Oppenheimer Approximation Adiabatic Assumption: Nuclei move so much more slowly than electron that the electrons that the electrons are assumed to be obtained if the nuclear kinetic energy is ignored,
Born-Oppenheimer Approximation Adiabatic Assumption: Nuclei move so much more slowly than electron that the electrons that the electrons are assumed to be obtained if the nuclear kinetic energy is ignored,
Electronic Spectra of Complexes
 Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
In the fourth problem set, you derived the MO diagrams for two complexes containing Cr-Cr bonds:
 Problem 1 (2 points) Part 1 a. Consider the following V III complexes: V(H2O)6 3+, VF6 3-, and VCl6 3-. The table below contains the energies corresponding to the two lowest spin-allowed d-d transitions
Problem 1 (2 points) Part 1 a. Consider the following V III complexes: V(H2O)6 3+, VF6 3-, and VCl6 3-. The table below contains the energies corresponding to the two lowest spin-allowed d-d transitions
Theoretical determination of the heat of formation of methylene
 Theoretical determination of the heat of formation of methylene Nikos L. Doltsinis and Peter J. Knowles School of Chemistry, University of Birmingham, Edgbaston, Birmingham B5 2TT, United Kingdom The heat
Theoretical determination of the heat of formation of methylene Nikos L. Doltsinis and Peter J. Knowles School of Chemistry, University of Birmingham, Edgbaston, Birmingham B5 2TT, United Kingdom The heat
1. Predict the structure of the molecules given by the following spectral data: a Mass spectrum:m + = 116
 Additional Problems for practice.. Predict the structure of the molecules given by the following spectral data: a Mass spectrum:m + = IR: weak absorption at 9 cm - medium absorption at cm - NMR 7 3 3 C
Additional Problems for practice.. Predict the structure of the molecules given by the following spectral data: a Mass spectrum:m + = IR: weak absorption at 9 cm - medium absorption at cm - NMR 7 3 3 C
Chapter 10: Modern Atomic Theory and the Periodic Table. How does atomic structure relate to the periodic table? 10.1 Electromagnetic Radiation
 Chapter 10: Modern Atomic Theory and the Periodic Table How does atomic structure relate to the periodic table? 10.1 Electromagnetic Radiation Electromagnetic (EM) radiation is a form of energy that exhibits
Chapter 10: Modern Atomic Theory and the Periodic Table How does atomic structure relate to the periodic table? 10.1 Electromagnetic Radiation Electromagnetic (EM) radiation is a form of energy that exhibits
Lec20 Fri 3mar17
 564-17 Lec20 Fri 3mar17 [PDF]GAUSSIAN 09W TUTORIAL www.molcalx.com.cn/wp-content/uploads/2015/01/gaussian09w_tutorial.pdf by A Tomberg - Cited by 8 - Related articles GAUSSIAN 09W TUTORIAL. AN INTRODUCTION
564-17 Lec20 Fri 3mar17 [PDF]GAUSSIAN 09W TUTORIAL www.molcalx.com.cn/wp-content/uploads/2015/01/gaussian09w_tutorial.pdf by A Tomberg - Cited by 8 - Related articles GAUSSIAN 09W TUTORIAL. AN INTRODUCTION
A.P. Chemistry Practice Test - Ch. 7, Atomic Structure and Periodicity
 A.P. Chemistry Practice Test - Ch. 7, Atomic Structure and Periodicity 1) Ham radio operators often broadcast on the 6-meter band. The frequency of this electromagnetic radiation is MHz. A) 50 B) 20 C)
A.P. Chemistry Practice Test - Ch. 7, Atomic Structure and Periodicity 1) Ham radio operators often broadcast on the 6-meter band. The frequency of this electromagnetic radiation is MHz. A) 50 B) 20 C)
- Some properties of elements can be related to their positions on the periodic table.
 179 PERIODIC TRENDS - Some properties of elements can be related to their positions on the periodic table. ATOMIC RADIUS - The distance between the nucleus of the atoms and the outermost shell of the electron
179 PERIODIC TRENDS - Some properties of elements can be related to their positions on the periodic table. ATOMIC RADIUS - The distance between the nucleus of the atoms and the outermost shell of the electron
Lectures Spectroscopy. Fall 2012
 Lectures 19-20 Spectroscopy Fall 2012 1 spectroscopic principles (Chem 1M/1N exps. #6 and #11) 4 1 spectroscopic excitations ( E = h = hc/ ; = c ) (nm) (sec -1 ) radiation technique molecular excitation
Lectures 19-20 Spectroscopy Fall 2012 1 spectroscopic principles (Chem 1M/1N exps. #6 and #11) 4 1 spectroscopic excitations ( E = h = hc/ ; = c ) (nm) (sec -1 ) radiation technique molecular excitation
Lectures Spectroscopy. Fall 2012
 Lectures 19-20 Spectroscopy Fall 2012 1 spectroscopic principles (Chem 1M/1N exps. #6 and #11) 4 spectroscopic excitations ( E = h = hc/ ; = c ) (nm) (sec -1 ) radiation technique molecular excitation
Lectures 19-20 Spectroscopy Fall 2012 1 spectroscopic principles (Chem 1M/1N exps. #6 and #11) 4 spectroscopic excitations ( E = h = hc/ ; = c ) (nm) (sec -1 ) radiation technique molecular excitation
Exercise 1: Structure and dipole moment of a small molecule
 Introduction to computational chemistry Exercise 1: Structure and dipole moment of a small molecule Vesa Hänninen 1 Introduction In this exercise the equilibrium structure and the dipole moment of a small
Introduction to computational chemistry Exercise 1: Structure and dipole moment of a small molecule Vesa Hänninen 1 Introduction In this exercise the equilibrium structure and the dipole moment of a small
CHEMISTRY - BROWN 13E CH.7 - PERIODIC PROPERTIES OF THE ELEMENTS
 !! www.clutchprep.com CONCEPT: EFFECTIVE NUCLEAR CHARGE & SLATER S RULES When looking at any particular electron within an atom it experiences two major forces. A(n) force from the nucleus and a(n) force
!! www.clutchprep.com CONCEPT: EFFECTIVE NUCLEAR CHARGE & SLATER S RULES When looking at any particular electron within an atom it experiences two major forces. A(n) force from the nucleus and a(n) force
PHYS 450 Spring semester Lecture 08: Optical Spectroscopy and Spectral Lines. Ron Reifenberger Birck Nanotechnology Center Purdue University
 /4/01 PHYS 450 Spring semester 01 Lecture 08: Optical Spectroscopy and Spectral Lines Ron Reifenberger Birck Nanotechnology Center Purdue University Lecture 08 1 Roadmap: Where We ve Been and Where We
/4/01 PHYS 450 Spring semester 01 Lecture 08: Optical Spectroscopy and Spectral Lines Ron Reifenberger Birck Nanotechnology Center Purdue University Lecture 08 1 Roadmap: Where We ve Been and Where We
Guide to the Extended Step-Pyramid Periodic Table
 Guide to the Extended Step-Pyramid Periodic Table William B. Jensen Department of Chemistry University of Cincinnati Cincinnati, OH 452201-0172 The extended step-pyramid table recognizes that elements
Guide to the Extended Step-Pyramid Periodic Table William B. Jensen Department of Chemistry University of Cincinnati Cincinnati, OH 452201-0172 The extended step-pyramid table recognizes that elements
Elements and the Periodic Table
 CHAPTER 6 Elements and the Periodic Table 6.2 Properties of Groups of Elements There are millions and millions of different kinds of matter (compounds) composed of the same 92 elements. These elements
CHAPTER 6 Elements and the Periodic Table 6.2 Properties of Groups of Elements There are millions and millions of different kinds of matter (compounds) composed of the same 92 elements. These elements
Molecular Magnetism. Magnetic Resonance Parameters. Trygve Helgaker
 Molecular Magnetism Magnetic Resonance Parameters Trygve Helgaker Centre for Theoretical and Computational Chemistry (CTCC), Department of Chemistry, University of Oslo, Norway Laboratoire de Chimie Théorique,
Molecular Magnetism Magnetic Resonance Parameters Trygve Helgaker Centre for Theoretical and Computational Chemistry (CTCC), Department of Chemistry, University of Oslo, Norway Laboratoire de Chimie Théorique,
MODERN ATOMIC THEORY AND THE PERIODIC TABLE
 C10 04/19/2013 13:34:14 Page 114 CHAPTER 10 MODERN ATOMIC THEORY AND THE PERIODIC TABLE SOLUTIONS TO REVIEW QUESTIONS 1. Wavelength is defined as the distance between consecutive peaks in a wave. It is
C10 04/19/2013 13:34:14 Page 114 CHAPTER 10 MODERN ATOMIC THEORY AND THE PERIODIC TABLE SOLUTIONS TO REVIEW QUESTIONS 1. Wavelength is defined as the distance between consecutive peaks in a wave. It is
Performance of Hartree-Fock and Correlated Methods
 Chemistry 460 Fall 2017 Dr. Jean M. Standard December 4, 2017 Performance of Hartree-Fock and Correlated Methods Hartree-Fock Methods Hartree-Fock methods generally yield optimized geomtries and molecular
Chemistry 460 Fall 2017 Dr. Jean M. Standard December 4, 2017 Performance of Hartree-Fock and Correlated Methods Hartree-Fock Methods Hartree-Fock methods generally yield optimized geomtries and molecular
NPTEL/IITM. Molecular Spectroscopy Lectures 1 & 2. Prof.K. Mangala Sunder Page 1 of 15. Topics. Part I : Introductory concepts Topics
 Molecular Spectroscopy Lectures 1 & 2 Part I : Introductory concepts Topics Why spectroscopy? Introduction to electromagnetic radiation Interaction of radiation with matter What are spectra? Beer-Lambert
Molecular Spectroscopy Lectures 1 & 2 Part I : Introductory concepts Topics Why spectroscopy? Introduction to electromagnetic radiation Interaction of radiation with matter What are spectra? Beer-Lambert
Large Amplitude Bending Motion: Computational Molecular Spectroscopy and Experiments of Transition-Metal Containing Isocyanide and Cyanides.
 WG13 63 rd Columbus 2008/Jun/18 Large Amplitude Bending Motion: Computational Molecular Spectroscopy and Experiments of Transition-Metal Containing Isocyanide and Cyanides. Tsuneo HIRANO, a,b Rei OKUDA,
WG13 63 rd Columbus 2008/Jun/18 Large Amplitude Bending Motion: Computational Molecular Spectroscopy and Experiments of Transition-Metal Containing Isocyanide and Cyanides. Tsuneo HIRANO, a,b Rei OKUDA,
Electrons. Unit H Chapter 6
 Electrons Unit H Chapter 6 1 Electrons were discovered by 1. Dalton 2. Lavoisier 3. Proust 4. Mendeleev 6. Rutherford 7. Bohr 8. Schrodinger 9. Dirac 5. Thomson 2 Electrons were discovered by 1. Dalton
Electrons Unit H Chapter 6 1 Electrons were discovered by 1. Dalton 2. Lavoisier 3. Proust 4. Mendeleev 6. Rutherford 7. Bohr 8. Schrodinger 9. Dirac 5. Thomson 2 Electrons were discovered by 1. Dalton
Accurate multireference configuration interaction calculations on the lowest 1 and 3 electronic states of C 2,CN, BN, and BO
 Accurate multireference configuration interaction calculations on the lowest 1 and 3 electronic states of C 2,CN, BN, and BO Kirk A. Peterson a) Department of Chemistry, Washington State University and
Accurate multireference configuration interaction calculations on the lowest 1 and 3 electronic states of C 2,CN, BN, and BO Kirk A. Peterson a) Department of Chemistry, Washington State University and
How many grams of sodium metal is required to completely react with 2545 grams of chlorine gas?
 146 EXAMPLE PROBLEM: How many grams of sodium metal is required to completely react with 2545 grams of chlorine gas? 1 - Convert 2545 grams of chlorine gas to moles. Use formula weight. 2 - Convert moles
146 EXAMPLE PROBLEM: How many grams of sodium metal is required to completely react with 2545 grams of chlorine gas? 1 - Convert 2545 grams of chlorine gas to moles. Use formula weight. 2 - Convert moles
Chapter 8 Problem Solutions
 Chapter 8 Problem Solutions 1. The energy needed to detach the electron from a hydrogen atom is 13.6 ev, but the energy needed to detach an electron from a hydrogen molecule is 15.7 ev. Why do you think
Chapter 8 Problem Solutions 1. The energy needed to detach the electron from a hydrogen atom is 13.6 ev, but the energy needed to detach an electron from a hydrogen molecule is 15.7 ev. Why do you think
b) For this ground state, obtain all possible J values and order them from lowest to highest in energy.
 Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
b) For this ground state, obtain all possible J values and order them from lowest to highest in energy.
 Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
- Why are phase labels required? Because phase changes either absorb or release energy. ... what does this mean?
 157 SINCE the enthalpy change does NOT depend on path, this means that we can use standard values for enthalpy to predict the heat change in reactions that we have not tested in a calorimeter. THERMOCHEMICAL
157 SINCE the enthalpy change does NOT depend on path, this means that we can use standard values for enthalpy to predict the heat change in reactions that we have not tested in a calorimeter. THERMOCHEMICAL
Chemistry 1A, Spring 2007 Midterm Exam 2 March 5, 2007 (90 min, closed book)
 Chemistry 1A, Spring 2007 Midterm Exam 2 March 5, 2007 (90 min, closed book) Name: KEY SID: TA Name: 1.) Write your name on every page of this exam. 2.) This exam has 40 multiple choice questions. Fill
Chemistry 1A, Spring 2007 Midterm Exam 2 March 5, 2007 (90 min, closed book) Name: KEY SID: TA Name: 1.) Write your name on every page of this exam. 2.) This exam has 40 multiple choice questions. Fill
Inorganic Chemistry with Doc M. Day 19. Transition Metals Complexes IV: Spectroscopy
 Inorganic Chemistry with Doc M. Day 19. Transition Metals Complexes IV: Spectroscopy Topics: 1. The visible spectrum and the d-orbitals 3. Octahedral fields 2. Term symbols and the method of microstates
Inorganic Chemistry with Doc M. Day 19. Transition Metals Complexes IV: Spectroscopy Topics: 1. The visible spectrum and the d-orbitals 3. Octahedral fields 2. Term symbols and the method of microstates
- Some properties of elements can be related to their positions on the periodic table.
 180 PERIODIC TRENDS - Some properties of elements can be related to their positions on the periodic table. ATOMIC RADIUS - The distance between the nucleus of the atoms and the outermost shell of the electron
180 PERIODIC TRENDS - Some properties of elements can be related to their positions on the periodic table. ATOMIC RADIUS - The distance between the nucleus of the atoms and the outermost shell of the electron
Name: Date: Atomic Structure 2017 Mrs. Mannion Version 1
 Name: Atomic Structure 2017 1. The mass of a proton is approximately equal to the mass of A) a beta particle B) an electron C) an alpha particle D) a neutron 2. What are the characteristics of a neutron?
Name: Atomic Structure 2017 1. The mass of a proton is approximately equal to the mass of A) a beta particle B) an electron C) an alpha particle D) a neutron 2. What are the characteristics of a neutron?
The Quantum Mechanical Atom
 The Quantum Mechanical Atom CHAPTER 8 Chemistry: The Molecular Nature of Matter, 6 th edition By Jesperson, Brady, & Hyslop CHAPTER 8: Quantum Mechanical Atom Learning Objectives q Light as Waves, Wavelength
The Quantum Mechanical Atom CHAPTER 8 Chemistry: The Molecular Nature of Matter, 6 th edition By Jesperson, Brady, & Hyslop CHAPTER 8: Quantum Mechanical Atom Learning Objectives q Light as Waves, Wavelength
Group Members: Your Name In Class Exercise #6. Photon A. Energy B
 Group Members: Your Name In Class Exercise #6 Shell Structure of Atoms Part II Photoelectron Spectroscopy Photoelectron spectroscopy is closely related to the photoelectric effect. When high energy photons
Group Members: Your Name In Class Exercise #6 Shell Structure of Atoms Part II Photoelectron Spectroscopy Photoelectron spectroscopy is closely related to the photoelectric effect. When high energy photons
QUANTUM CHEMISTRY FOR TRANSITION METALS
 QUANTUM CHEMISTRY FOR TRANSITION METALS Outline I Introduction II Correlation Static correlation effects MC methods DFT III Relativity Generalities From 4 to 1 components Effective core potential Outline
QUANTUM CHEMISTRY FOR TRANSITION METALS Outline I Introduction II Correlation Static correlation effects MC methods DFT III Relativity Generalities From 4 to 1 components Effective core potential Outline
1. What is the phenomenon that occurs when certain metals emit electrons when illuminated by particular wavelengths of light? a.
 CHEMISTRY 123-07 Midterm #3 solution key December 02, 2010 Statistics: Average: 77 p (77%); Highest: 100 p (100%); Lowest: 33 p (33%) Number of students performing at or above average: 54 (52%) Number
CHEMISTRY 123-07 Midterm #3 solution key December 02, 2010 Statistics: Average: 77 p (77%); Highest: 100 p (100%); Lowest: 33 p (33%) Number of students performing at or above average: 54 (52%) Number
Assumed knowledge. Chemistry 2. Learning outcomes. Electronic spectroscopy of polyatomic molecules. Franck-Condon Principle (reprise)
 Chemistry 2 Lecture 11 Electronic spectroscopy of polyatomic molecules Assumed knowledge For bound excited states, transitions to the individual vibrational levels of the excited state are observed with
Chemistry 2 Lecture 11 Electronic spectroscopy of polyatomic molecules Assumed knowledge For bound excited states, transitions to the individual vibrational levels of the excited state are observed with
Electronic Structure and Dipole Moment Calculations of the Electronic States of the Molecule ZnS
 Modern Applied Science; Vol. 12, No. 3; 2018 ISSN 1913-1844 E-ISSN 1913-1852 Published by Canadian Center of Science and Education Electronic Structure and Dipole Moment Calculations of the Electronic
Modern Applied Science; Vol. 12, No. 3; 2018 ISSN 1913-1844 E-ISSN 1913-1852 Published by Canadian Center of Science and Education Electronic Structure and Dipole Moment Calculations of the Electronic
Glow-Sticks are Forbidden. Zachary G Wolfe. Department of Physics and Physical Oceanography. University of North Carolina Wilmington
 Glow-Sticks are Forbidden Zachary G Wolfe Department of Physics and Physical Oceanography University of North Carolina Wilmington Abstract: Physics students learn early that certain electronic transitions
Glow-Sticks are Forbidden Zachary G Wolfe Department of Physics and Physical Oceanography University of North Carolina Wilmington Abstract: Physics students learn early that certain electronic transitions
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which one of the following is not one of the postulates of Dalton's atomic theory? A)
Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which one of the following is not one of the postulates of Dalton's atomic theory? A)
Same idea for polyatomics, keep track of identical atom e.g. NH 3 consider only valence electrons F(2s,2p) H(1s)
 XIII 63 Polyatomic bonding -09 -mod, Notes (13) Engel 16-17 Balance: nuclear repulsion, positive e-n attraction, neg. united atom AO ε i applies to all bonding, just more nuclei repulsion biggest at low
XIII 63 Polyatomic bonding -09 -mod, Notes (13) Engel 16-17 Balance: nuclear repulsion, positive e-n attraction, neg. united atom AO ε i applies to all bonding, just more nuclei repulsion biggest at low
16.1 Molecular Vibrations
 16.1 Molecular Vibrations molecular degrees of freedom are used to predict the number of vibrational modes vibrations occur as coordinated movement among many nuclei the harmonic oscillator approximation
16.1 Molecular Vibrations molecular degrees of freedom are used to predict the number of vibrational modes vibrations occur as coordinated movement among many nuclei the harmonic oscillator approximation
( R) MOLECULES: VALENCE BOND DESCRIPTION VALENCE BOND APPROXIMATION METHOD. Example: H 2. at some fixed R, repeat for many different values
 5.6 004 Lecture #9 page MOLECULES: VLENCE OND DESCRIPTION VLENCE OND PPROXIMTION METHOD Example: H H = + + r r r r r R Determine E ( R) at some fixed R, repeat for many different values Electronic Schrodinger
5.6 004 Lecture #9 page MOLECULES: VLENCE OND DESCRIPTION VLENCE OND PPROXIMTION METHOD Example: H H = + + r r r r r R Determine E ( R) at some fixed R, repeat for many different values Electronic Schrodinger
- Some properties of elements can be related to their positions on the periodic table.
 179 PERIODIC TRENDS - Some properties of elements can be related to their positions on the periodic table. ATOMIC RADIUS - The distance between the nucleus of the atoms and the outermost shell of the electron
179 PERIODIC TRENDS - Some properties of elements can be related to their positions on the periodic table. ATOMIC RADIUS - The distance between the nucleus of the atoms and the outermost shell of the electron
How many grams of sodium metal is required to completely react with 2545 grams of chlorine gas?
 EXAMPLE PROBLEM: How many grams of sodium metal is required to completely react with 2545 grams of chlorine gas? 1 - Convert 2545 grams of chlorine to moles chlorine using formula weight 2 - Convert moles
EXAMPLE PROBLEM: How many grams of sodium metal is required to completely react with 2545 grams of chlorine gas? 1 - Convert 2545 grams of chlorine to moles chlorine using formula weight 2 - Convert moles
Bonds in molecules are formed by the interactions between electrons.
 CHEM 2060 Lecture 6: Electrostatic Interactions L6-1 PART TWO: Electrostatic Interactions In the first section of this course, we were more concerned with structural aspects of molecules. In this section
CHEM 2060 Lecture 6: Electrostatic Interactions L6-1 PART TWO: Electrostatic Interactions In the first section of this course, we were more concerned with structural aspects of molecules. In this section
How we describe bonding is affected strongly by how we describe where/how electrons are held by atoms in molecules.
 CHEM 2060 Lecture 8: Atomic Configurations L8-1 Electronic Configurations How we describe bonding is affected strongly by how we describe where/how electrons are held by atoms in molecules. We know that
CHEM 2060 Lecture 8: Atomic Configurations L8-1 Electronic Configurations How we describe bonding is affected strongly by how we describe where/how electrons are held by atoms in molecules. We know that
Inorganic Chemistry with Doc M. Fall Semester, 2011 Day 19. Transition Metals Complexes IV: Spectroscopy
 Inorganic Chemistry with Doc M. Fall Semester, 011 Day 19. Transition Metals Complexes IV: Spectroscopy Name(s): lement: Topics: 1. The visible spectrum and the d-orbitals 3. Octahedral fields. Term symbols
Inorganic Chemistry with Doc M. Fall Semester, 011 Day 19. Transition Metals Complexes IV: Spectroscopy Name(s): lement: Topics: 1. The visible spectrum and the d-orbitals 3. Octahedral fields. Term symbols
The electronic structure of solids. Charge transport in solids
 The electronic structure of solids We need a picture of the electronic structure of solid that we can use to explain experimental observations and make predictions Why is diamond an insulator? Why is sodium
The electronic structure of solids We need a picture of the electronic structure of solid that we can use to explain experimental observations and make predictions Why is diamond an insulator? Why is sodium
Midterm Exam I. CHEM 181: Introduction to Chemical Principles September 24, 2015 Key
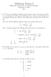 Midterm Exam I CHEM 8: Introduction to Chemical Principles September 24, 205 Key. A Li 2+ ion in an unknown, highly excited electronic state, first emits a photon at a wavelength of 4.36 µm, and following
Midterm Exam I CHEM 8: Introduction to Chemical Principles September 24, 205 Key. A Li 2+ ion in an unknown, highly excited electronic state, first emits a photon at a wavelength of 4.36 µm, and following
- Some properties of elements can be related to their positions on the periodic table.
 186 PERIODIC TRENDS - Some properties of elements can be related to their positions on the periodic table. ATOMIC RADIUS - The distance between the nucleus of the atoms and the outermost shell of the electron
186 PERIODIC TRENDS - Some properties of elements can be related to their positions on the periodic table. ATOMIC RADIUS - The distance between the nucleus of the atoms and the outermost shell of the electron
The Periodic Table. Periodic Properties. Can you explain this graph? Valence Electrons. Valence Electrons. Paramagnetism
 Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Excited State Processes
 Excited State Processes Photophysics Fluorescence (singlet state emission) Phosphorescence (triplet state emission) Internal conversion (transition to singlet gr. state) Intersystem crossing (transition
Excited State Processes Photophysics Fluorescence (singlet state emission) Phosphorescence (triplet state emission) Internal conversion (transition to singlet gr. state) Intersystem crossing (transition
4. NMR spectra. Interpreting NMR spectra. Low-resolution NMR spectra. There are two kinds: Low-resolution NMR spectra. High-resolution NMR spectra
 1 Interpreting NMR spectra There are two kinds: Low-resolution NMR spectra High-resolution NMR spectra In both cases the horizontal scale is labelled in terms of chemical shift, δ, and increases from right
1 Interpreting NMR spectra There are two kinds: Low-resolution NMR spectra High-resolution NMR spectra In both cases the horizontal scale is labelled in terms of chemical shift, δ, and increases from right
Unit 1, Lesson 01: Summary of Atomic Structure so far
 Unit 1, Lesson 01: Summary of Atomic Structure so far Atoms are made of sub-atomic particles: Protons: found in the nucleus, charge of 1+, mass of 1 amu (u) Neutrons: found in nucleus, no charge, mass
Unit 1, Lesson 01: Summary of Atomic Structure so far Atoms are made of sub-atomic particles: Protons: found in the nucleus, charge of 1+, mass of 1 amu (u) Neutrons: found in nucleus, no charge, mass
Chemistry 21b Final Examination
 Chemistry 21b Final Examination Out: 11 March 2011 Due: 16 March 2011, 5 pm This is an open book examination, and so you may use McQuarrie or Harris and Bertolucci along with the posted Lecture Notes and
Chemistry 21b Final Examination Out: 11 March 2011 Due: 16 March 2011, 5 pm This is an open book examination, and so you may use McQuarrie or Harris and Bertolucci along with the posted Lecture Notes and
Trends in the Periodic Table
 Trends in the Periodic Table OBJECTIVES FOR TODAY: Fall in love with the Periodic Table, Interpret group and period trends in atomic radii, ionization energies and electronegativity The Periodic Table
Trends in the Periodic Table OBJECTIVES FOR TODAY: Fall in love with the Periodic Table, Interpret group and period trends in atomic radii, ionization energies and electronegativity The Periodic Table
THEORY OF MOLECULE. A molecule consists of two or more atoms with certain distances between them
 THEORY OF MOLECULE A molecule consists of two or more atoms with certain distances between them through interaction of outer electrons. Distances are determined by sum of all forces between the atoms.
THEORY OF MOLECULE A molecule consists of two or more atoms with certain distances between them through interaction of outer electrons. Distances are determined by sum of all forces between the atoms.
Chemistry B11 Chapter 3 Atoms
 Chapter 3 Atoms Element: is a substance that consists of identical atoms (hydrogen, oxygen, and Iron). 116 elements are known (88 occur in nature and chemist have made the others in the lab). Compound:
Chapter 3 Atoms Element: is a substance that consists of identical atoms (hydrogen, oxygen, and Iron). 116 elements are known (88 occur in nature and chemist have made the others in the lab). Compound:
Putting a spin on time dependent electronic structure theory! Joshua Goings! General Exam! Thursday, May 14, 10:00am CHB 339
 Putting a spin on time dependent electronic structure theory Joshua Goings General Exam Thursday, May 14, 10:00am CHB 339 Thank you to General Exam Committee Xiaosong Li (chair) Jim Pfaendtner (GSR) Matthew
Putting a spin on time dependent electronic structure theory Joshua Goings General Exam Thursday, May 14, 10:00am CHB 339 Thank you to General Exam Committee Xiaosong Li (chair) Jim Pfaendtner (GSR) Matthew
(Picture courtesy of Python M)
 (Picture courtesy of Python M) Where is Wuppertal? The Wuppertal monorail The general scheme of things (at least in this lecture.) Potential energy surface(s) Dipole moment surface(s)... in general obtained
(Picture courtesy of Python M) Where is Wuppertal? The Wuppertal monorail The general scheme of things (at least in this lecture.) Potential energy surface(s) Dipole moment surface(s)... in general obtained
Computational Material Science Part II. Ito Chao ( ) Institute of Chemistry Academia Sinica
 Computational Material Science Part II Ito Chao ( ) Institute of Chemistry Academia Sinica Ab Initio Implementations of Hartree-Fock Molecular Orbital Theory Fundamental assumption of HF theory: each electron
Computational Material Science Part II Ito Chao ( ) Institute of Chemistry Academia Sinica Ab Initio Implementations of Hartree-Fock Molecular Orbital Theory Fundamental assumption of HF theory: each electron
Problems with the Wave Theory of Light (Photoelectric Effect)
 CHEM101 NOTES Properties of Light Found that the wave theory could not work for some experiments e.g. the photovoltaic effect This is because the classic EM view of light could not account for some of
CHEM101 NOTES Properties of Light Found that the wave theory could not work for some experiments e.g. the photovoltaic effect This is because the classic EM view of light could not account for some of
Chemistry 218 Spring Molecular Structure
 Chemistry 218 Spring 2015-2016 Molecular Structure R. Sultan COURSE SYLLABUS Email: rsultan@aub.edu.lb Homepage: http://staff.aub.edu.lb/~rsultan/ Lectures: 12:30-13:45 T, Th. 101 Chemistry Textbook: P.
Chemistry 218 Spring 2015-2016 Molecular Structure R. Sultan COURSE SYLLABUS Email: rsultan@aub.edu.lb Homepage: http://staff.aub.edu.lb/~rsultan/ Lectures: 12:30-13:45 T, Th. 101 Chemistry Textbook: P.
NAME: FIRST EXAMINATION
 1 Chemistry 64 Winter 1994 NAME: FIRST EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
1 Chemistry 64 Winter 1994 NAME: FIRST EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
Organic Chemistry: CHEM2322
 Conjugated Systems Organic Chemistry: We met in Chem 2321 unsaturated bonds as either a C=C bond or C C bond. If these unsaturated bonds are well separated then they react independently however if there
Conjugated Systems Organic Chemistry: We met in Chem 2321 unsaturated bonds as either a C=C bond or C C bond. If these unsaturated bonds are well separated then they react independently however if there
Using Web-Based Computations in Organic Chemistry
 10/30/2017 1 Using Web-Based Computations in Organic Chemistry John Keller UAF Department of Chemistry & Biochemistry The UAF WebMO site Practical aspects of computational chemistry theory and nomenclature
10/30/2017 1 Using Web-Based Computations in Organic Chemistry John Keller UAF Department of Chemistry & Biochemistry The UAF WebMO site Practical aspects of computational chemistry theory and nomenclature
indicating the configuration they correspond to and predict their relative energy.
 Problem 1 (1 point) Three center four electron (3c/4e) bonds were introduced in class. John F. Berry (Dalton Trans. 2012, 41, 700-713) discusses the effect of the larger density of states for the 3c/4e
Problem 1 (1 point) Three center four electron (3c/4e) bonds were introduced in class. John F. Berry (Dalton Trans. 2012, 41, 700-713) discusses the effect of the larger density of states for the 3c/4e
