Previewer Tools Show All In View: Hide All Description Atomic Structure
|
|
|
- Lorin Griffith
- 5 years ago
- Views:
Transcription
1 Assignment Previewer Chapter 7 (397929) About this Assignment Previewer Tools Show All In View: Hide All Close this window Hidden: Assignment Score Mark Help/Hints Key Solution Show New Randomization Reload from Editor Description Atomic Structure Details Instructions Atomic Structure 1. KT6 7.P.005. [489845] Show Details Green light has a wavelength of nm. What is the energy, in joules, of one photon of green light? J/photon What is the energy, in joules, of 1.0 mol of photons of green light? J/mol 2. KT6 7.P.006. [467162] Show Details Violet light has a wavelength of about 409 nm. What is its frequency? s -1 Calculate the energy of one photon of violet light. J What is the energy of 1.1 mol of violet photons? kj Compare the energy of photons of violet light with those of red light. Which is more energetic? nmlkj violet light nmlkj red light 3. KT6 7.P.012. [467500] Show Details You are an engineer a switch that works by the photoelectric effect. The metal you wish to use in your device requires J/atom to remove an electron. Will the switch work if the light falling on the metal has a wavelength of 540 nm or greater? nmlkj yes nmlkj no Why or why not? nmlkj The device will work because wavelengths less than 540 nm cannot be used for device operation. nmlkj The device will not work because the atmosphere will filter out wavelengths less than 540 nm. nmlkj The device will work because wavelengths greater than 540 nm can operate the device. 4. KT6 7.P.013. [467352] Show Details The most prominent line in the spectrum of mercury is at nm. Other lines are located at nm, nm, nm, and nm. (a) Which of these lines represents the most energetic light? nmlkj nm nmlkj nm nmlkj nm 1 of 5 8/29/2006 1:50 PM
2 nmlkj nm nmlkj nm (b) What is the frequency of the most prominent line? s -1 What is the energy of one photon with this wavelength? J/photon (c) Are any of these lines found in the spectrum of mercury shown in Figure 7.9? nmlkj yes nmlkj no Which ones? gfedc nm gfedc nm gfedc nm gfedc nm gfedc nm gfedc None of these. What color or colors are these lines? gfedc violet gfedc blue gfedc red gfedc orange gfedc yellow gfedc green gfedc None of these. Figure KT6 7.P.017. [467332] Show Details Consider only transitions involving the following energy levels for the hydrogen atom (where the energy level spacing below are not to scale). n = 5 n = 4 n = 3 n = 2 n = 1 (a) How many emission lines are possible, considering only these five quantum levels? lines (b) Photons of the highest frequency are emitted in a transition from the level with n = to a 2 of 5 8/29/2006 1:50 PM
3 level with the n =. (c) The emission line having the longest wavelength corresponds to a transition from the level with n = to a level with the n =. 6. KT6 7.P.019. [467435] Show Details The energy emitted when an electron moves from a higher energy state to a lower energy state in any atom can be observed as electromagnetic radiation. Figure 7.9 Figure 7.12 (a) Which involves the emission of less energy in the H atom, an electron moving from n = 4 to n = 2 or an electron moving from n = 3 to n = 2? nmlkj Both emit the same energy. nmlkj n = 3 to n = 2 nmlkj n = 4 to n = 2 nmlkj Energy is never emitted. (b) Which involves the emission of more energy in the H atom, an electron moving from n = 4 to n = 1 or an electron changing from n = 5 to n = 2? Explain. nmlkj n = 5 to n = 2, because the energy level are progressively widening at higher levels. nmlkj Both these jumps are not associated with energy emission. 3 of 5 8/29/2006 1:50 PM
4 nmlkj Both have the same energy level as the jump is three orbits. nmlkj n = 4 to n = 1, because the energy levels are progressively closer at higher levels. 7. KT6 7.P.025. [467434] Show Details Calculate the wavelength, in nanometers, associated with a g golf ball moving at 30. m/s (about 67 mph). nm How fast must the ball travel to have a wavelength of 4.5 m/s 10-3 nm? 8. KT6 7.P.028. [489799] Show Details Answer the following questions. (a) When n = 4, = 2, and m = -1, to what orbital type does this refer? (Type the orbital label using the format 1s for 1s.) (b) How many orbitals occur in the n = 3 electron shell? How many subshells? What are the letter labels of the subshells? nmlkj s nmlkj s, p nmlkj s, p, d nmlkj s, p, d, f nmlkj s, p, d, f, g (c) If a subshell is labeled f, how many orbitals occur in the subshell? What are the values of m? nmlkj 0 nmlkj 0, 1 nmlkj 0, 1, 2 nmlkj 0, 1, 2, 3 9. KT6 7.P.035. [489872] Show Details What is the maximum number of orbitals that can be identified by each of the following sets of quantum numbers? (0 is a possible answer.) (a) n = 7, = 5 (b) n = 2, = 1, m = +3 (c) n = 4, = 2 (d) n = 6, = 3, m = KT6 7.P.054. [489811] Show Details 4 of 5 8/29/2006 1:50 PM
5 Radiation in the ultraviolet region of the electromagnetic spectrum is quite energetic. It is this radiation that causes dyes to fade and your skin to burn. If you are bombarded with 1.00 mol of photons with a wavelength of 284 nm, what amount of energy, in kilojoules per mole of photons, are you being subjected to? kj/mol Submit for Testing 5 of 5 8/29/2006 1:50 PM
Atomic Structure. Standing Waves x10 8 m/s. (or Hz or 1/s) λ Node
 Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
2) The number of cycles that pass through a stationary point is called A) wavelength. B) amplitude. C) frequency. D) area. E) median.
 Chemistry Structure and Properties 2nd Edition Tro Test Bank Full Download: http://testbanklive.com/download/chemistry-structure-and-properties-2nd-edition-tro-test-bank/ Chemistry: Structure & Properties,
Chemistry Structure and Properties 2nd Edition Tro Test Bank Full Download: http://testbanklive.com/download/chemistry-structure-and-properties-2nd-edition-tro-test-bank/ Chemistry: Structure & Properties,
Atomic Structure 11/21/2011
 Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
CHEM Chapter 6. Basic Quantum Chemistry (Homework). WL36
 CHEM 1411. Chapter 6. Basic Quantum Chemistry (Homework). WL36 1. The Bohr model of the hydrogen atom found its greatest support in experimental work on the photoelectric effect. A) True B) False 2. A
CHEM 1411. Chapter 6. Basic Quantum Chemistry (Homework). WL36 1. The Bohr model of the hydrogen atom found its greatest support in experimental work on the photoelectric effect. A) True B) False 2. A
Atomic Spectroscopy. Objectives
 Atomic Spectroscopy Name Objectives explain the difference between emission and absorption spectra calculate the energy of orbits in the Bohr model of hydrogen calculate E for energy transitions in the
Atomic Spectroscopy Name Objectives explain the difference between emission and absorption spectra calculate the energy of orbits in the Bohr model of hydrogen calculate E for energy transitions in the
WEEK 2: 4 SEP THRU 10 SEP; LECTURES 4-6
 Learning Objectives Energy: Light as energy Describe the wave nature of light, wavelength, and frequency using the equation c = λν What is meant by the particle nature of light? Calculate the energy of
Learning Objectives Energy: Light as energy Describe the wave nature of light, wavelength, and frequency using the equation c = λν What is meant by the particle nature of light? Calculate the energy of
Chem 110 Practice Midterm 2014
 Name Chem 110 Practice Midterm 2014 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following statements is true? 1) A) Two electrons
Name Chem 110 Practice Midterm 2014 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following statements is true? 1) A) Two electrons
Accounts for certain objects being colored. Used in medicine (examples?) Allows us to learn about structure of the atom
 1.1 Interaction of Light and Matter Accounts for certain objects being colored Used in medicine (examples?) 1.2 Wavelike Properties of Light Wavelength, : peak to peak distance Amplitude: height of the
1.1 Interaction of Light and Matter Accounts for certain objects being colored Used in medicine (examples?) 1.2 Wavelike Properties of Light Wavelength, : peak to peak distance Amplitude: height of the
I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited
 NCCS 1.1.2 & 1.1.3 I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited state I will describe how an electron
NCCS 1.1.2 & 1.1.3 I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited state I will describe how an electron
The ELECTRON: Wave Particle Duality. chapter 4
 The ELECTRON: Wave Particle Duality chapter 4 What do we know about light? Before 1900 s scientists thought light behaved as a wave. This belief changed when it was discovered that light also has particle
The ELECTRON: Wave Particle Duality chapter 4 What do we know about light? Before 1900 s scientists thought light behaved as a wave. This belief changed when it was discovered that light also has particle
THE ATOMIC SPECTRUM OF HYDROGEN
 THE ATOMIC SPECTRUM OF HYDROGEN When atoms are excited, either in an electric discharge or with heat, they tend to give off light. The light is emitted only at certain wavelengths that are characteristic
THE ATOMIC SPECTRUM OF HYDROGEN When atoms are excited, either in an electric discharge or with heat, they tend to give off light. The light is emitted only at certain wavelengths that are characteristic
Chapter 2 Atoms and Elements. Electromagnetic Radiation. Electromagnetic Spectrum. Electron Energy Levels. 2.6 Electron Energy Levels
 Chapter 2 Atoms and Elements Electromagnetic Radiation 2.6 Electron Energy Levels Electromagnetic radiation Consists of energy particles called photons that travel as waves. Includes low energy particles
Chapter 2 Atoms and Elements Electromagnetic Radiation 2.6 Electron Energy Levels Electromagnetic radiation Consists of energy particles called photons that travel as waves. Includes low energy particles
Properties of Light and Atomic Structure. Chapter 7. So Where are the Electrons? Electronic Structure of Atoms. The Wave Nature of Light!
 Properties of Light and Atomic Structure Chapter 7 So Where are the Electrons? We know where the protons and neutrons are Nuclear structure of atoms (Chapter 2) The interaction of light and matter helps
Properties of Light and Atomic Structure Chapter 7 So Where are the Electrons? We know where the protons and neutrons are Nuclear structure of atoms (Chapter 2) The interaction of light and matter helps
Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell?
 AP Chemistry Ms. Ye Name Date Block Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell? 1 st shell 2 nd shell
AP Chemistry Ms. Ye Name Date Block Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell? 1 st shell 2 nd shell
Test #3 Last Name First Name Zumdahl, Chapters 6 and 7 November 2, 2004
 Form H Chemistry 1441-023 Name (please print) Test #3 Last Name First Name Zumdahl, Chapters 6 and 7 November 2, 2004 Instructions: 1. This exam consists of 25 questions. 2. No scratch paper is allowed.
Form H Chemistry 1441-023 Name (please print) Test #3 Last Name First Name Zumdahl, Chapters 6 and 7 November 2, 2004 Instructions: 1. This exam consists of 25 questions. 2. No scratch paper is allowed.
Homework 04 - Electromagnetic Radiation
 HW04 - Electromagnetic Radiation This is a preview of the published version of the quiz Started: Jul 7 at 9:43am Quiz Instructions Homework 04 - Electromagnetic Radiation Question 1-7 What is the frequency
HW04 - Electromagnetic Radiation This is a preview of the published version of the quiz Started: Jul 7 at 9:43am Quiz Instructions Homework 04 - Electromagnetic Radiation Question 1-7 What is the frequency
CHE 105 Fall 2016 Exam 3
 CHE 105 Fall 2016 Exam 3 Your Name: Your ID: Question #: 1 Samples of CO2(g), C6H6(g), Cl2(g) and C3H8(g) are placed in separate glass vessels (only one gas to a vessel) at the same temperature and pressure.
CHE 105 Fall 2016 Exam 3 Your Name: Your ID: Question #: 1 Samples of CO2(g), C6H6(g), Cl2(g) and C3H8(g) are placed in separate glass vessels (only one gas to a vessel) at the same temperature and pressure.
Atomic Spectroscopy and Energy Levels
 Activity 18 Atomic Spectroscopy and Energy Levels Why? The emission of light by the hydrogen atom and other atoms played a key role in helping scientists to understand the electronic structure of atoms.
Activity 18 Atomic Spectroscopy and Energy Levels Why? The emission of light by the hydrogen atom and other atoms played a key role in helping scientists to understand the electronic structure of atoms.
Hint: unit of energy transferred is equal to: hν = hc
 CH 0 Fall 08 Discussion # Chapter 8 Your name: TF s name Discussion Day/Time: Things you should know when you leave Discussion today: Atomic (matter) Emission and absorption of light. Energy Conservation
CH 0 Fall 08 Discussion # Chapter 8 Your name: TF s name Discussion Day/Time: Things you should know when you leave Discussion today: Atomic (matter) Emission and absorption of light. Energy Conservation
PHYS General Physics II Lab The Balmer Series for Hydrogen Source. c = speed of light = 3 x 10 8 m/s
 PHYS 1040 - General Physics II Lab The Balmer Series for Hydrogen Source Purpose: The purpose of this experiment is to analyze the emission of light from a hydrogen source and measure and the wavelengths
PHYS 1040 - General Physics II Lab The Balmer Series for Hydrogen Source Purpose: The purpose of this experiment is to analyze the emission of light from a hydrogen source and measure and the wavelengths
The Sine Wave. You commonly see waves in the environment. Light Sound Electricity Ocean waves
 The Sine Wave Mathematically, a function that represents a smooth oscillation For example, if we drew the motion of how the weight bobs on the spring to the weight we would draw out a sine wave. The Sine
The Sine Wave Mathematically, a function that represents a smooth oscillation For example, if we drew the motion of how the weight bobs on the spring to the weight we would draw out a sine wave. The Sine
 Lower Frequency Higher Frequency Although the wave model of light explains many aspects of the behavior of light, several observations of electromagnetic radiation are not explained by this model: 1. The
Lower Frequency Higher Frequency Although the wave model of light explains many aspects of the behavior of light, several observations of electromagnetic radiation are not explained by this model: 1. The
Wavelength (λ)- Frequency (ν)- Which of the following has a higher frequency?
 Name: Unit 5- Light and Energy Electromagnetic Spectrum Notes Electromagnetic radiation is a form of energy that emits wave-like behavior as it travels through space. Amplitude (a)- Wavelength (λ)- Which
Name: Unit 5- Light and Energy Electromagnetic Spectrum Notes Electromagnetic radiation is a form of energy that emits wave-like behavior as it travels through space. Amplitude (a)- Wavelength (λ)- Which
Electrons, Energy, & the Electromagnetic Spectrum Notes
 Electrons, Energy, & the Electromagnetic Spectrum Notes Bohr Model Diagram Interpretation What form of EM radiation is released when an electron in a hydrogen atom falls from the 5 th energy level to the
Electrons, Energy, & the Electromagnetic Spectrum Notes Bohr Model Diagram Interpretation What form of EM radiation is released when an electron in a hydrogen atom falls from the 5 th energy level to the
Electron Energy and Light
 Why? Electron Energy and Light How does light reveal the behavior of electrons in an atom? From fireworks to stars, the color of light is useful in finding out what s in matter. The emission of light by
Why? Electron Energy and Light How does light reveal the behavior of electrons in an atom? From fireworks to stars, the color of light is useful in finding out what s in matter. The emission of light by
Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE Brooks/Cole - Thomson
 Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE 1 7.1 The Nature of Light 2 Most subatomic particles behave as PARTICLES and obey the physics of waves. Light is a type of electromagnetic radiation Light consists
Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE 1 7.1 The Nature of Light 2 Most subatomic particles behave as PARTICLES and obey the physics of waves. Light is a type of electromagnetic radiation Light consists
CHEMISTRY 1031 EXAM III QUESTIONS FOR THE REVIEW WORKSHOP (You will need a periodic table. Useful information is on page 4. Answers are on page 5.
 CHEMISTRY 1031 EXAM III QUESTIONS FOR THE REVIEW WORKSHOP (You will need a periodic table. Useful information is on page 4. Answers are on page 5.) 1 1. Calculate the minimum energy required to decompose
CHEMISTRY 1031 EXAM III QUESTIONS FOR THE REVIEW WORKSHOP (You will need a periodic table. Useful information is on page 4. Answers are on page 5.) 1 1. Calculate the minimum energy required to decompose
Chp 6: Atomic Structure
 Chp 6: Atomic Structure 1. Electromagnetic Radiation 2. Light Energy 3. Line Spectra & the Bohr Model 4. Electron & Wave-Particle Duality 5. Quantum Chemistry & Wave Mechanics 6. Atomic Orbitals Overview
Chp 6: Atomic Structure 1. Electromagnetic Radiation 2. Light Energy 3. Line Spectra & the Bohr Model 4. Electron & Wave-Particle Duality 5. Quantum Chemistry & Wave Mechanics 6. Atomic Orbitals Overview
2008 Brooks/Cole 2. Frequency (Hz)
 Electromagnetic Radiation and Matter Oscillating electric and magnetic fields. Magnetic field Electric field Chapter 7: Electron Configurations and the Periodic Table Traveling wave moves through space
Electromagnetic Radiation and Matter Oscillating electric and magnetic fields. Magnetic field Electric field Chapter 7: Electron Configurations and the Periodic Table Traveling wave moves through space
Exam 2. Remember to refer to the Periodic Table handout that is separate from this exam copy.
 001 version last name first name signature McCord CH301 unique: 49885 TTh 9:30 am - 11 am Exam 2 Oct 15, 2018 Monday 7:30-9:00 PM A - Mi in BUR 106 Mo - Z in JES A121A Remember to refer to the Periodic
001 version last name first name signature McCord CH301 unique: 49885 TTh 9:30 am - 11 am Exam 2 Oct 15, 2018 Monday 7:30-9:00 PM A - Mi in BUR 106 Mo - Z in JES A121A Remember to refer to the Periodic
Electronic Structure and the Periodic Table. Unit 6 Honors Chemistry
 Electronic Structure and the Periodic Table Unit 6 Honors Chemistry Wave Theory of Light James Clerk Maxwell Electromagnetic waves a form of energy that exhibits wavelike behavior as it travels through
Electronic Structure and the Periodic Table Unit 6 Honors Chemistry Wave Theory of Light James Clerk Maxwell Electromagnetic waves a form of energy that exhibits wavelike behavior as it travels through
Electron Energy and Light
 Why? Electron Energy and Light How does light reveal the behavior of electrons in an atom? From fireworks to stars, the color of light is useful in finding out what s in matter. The emission of light by
Why? Electron Energy and Light How does light reveal the behavior of electrons in an atom? From fireworks to stars, the color of light is useful in finding out what s in matter. The emission of light by
Key Equations. Determining the smallest change in an atom's energy.
 ATOMIC STRUCTURE AND PERIODICITY Matter and Energy Key Equations λν = c ΔE = hν Relating speed of a wave to its wavelength and frequency. Determining the smallest change in an atom's energy. H( λ =R n
ATOMIC STRUCTURE AND PERIODICITY Matter and Energy Key Equations λν = c ΔE = hν Relating speed of a wave to its wavelength and frequency. Determining the smallest change in an atom's energy. H( λ =R n
LIGHT AND THE QUANTUM MODEL
 LIGHT AND THE QUANTUM MODEL WAVES Wavelength ( ) - length of one complete wave Frequency ( ) - # of waves that pass a point during a certain time period hertz (Hz) = 1/s Amplitude (A) - distance from the
LIGHT AND THE QUANTUM MODEL WAVES Wavelength ( ) - length of one complete wave Frequency ( ) - # of waves that pass a point during a certain time period hertz (Hz) = 1/s Amplitude (A) - distance from the
Honors Unit 6 Notes - Atomic Structure
 Name: Honors Unit 6 Notes - Atomic Structure Objectives: 1. Students will have a general understanding of the wave nature of light and the interrelationship between frequency, wavelength, and speed of
Name: Honors Unit 6 Notes - Atomic Structure Objectives: 1. Students will have a general understanding of the wave nature of light and the interrelationship between frequency, wavelength, and speed of
NOTES: 5.3 Light and Atomic Spectra (more Quantum Mechanics!)
 NOTES: 5.3 Light and Atomic Spectra (more Quantum Mechanics!) Light WAVE or PARTICLE? Electromagnetic Radiation Electromagnetic radiation includes: -radio waves -microwaves -infrared waves -visible light
NOTES: 5.3 Light and Atomic Spectra (more Quantum Mechanics!) Light WAVE or PARTICLE? Electromagnetic Radiation Electromagnetic radiation includes: -radio waves -microwaves -infrared waves -visible light
CDO CP Chemistry Unit 5
 1. Of the three particles; protons, neutrons, and electrons, which one(s) are responsible for most of the mass of an atom? a) the protons only b) the electrons only c) the neutrons only d) the protons
1. Of the three particles; protons, neutrons, and electrons, which one(s) are responsible for most of the mass of an atom? a) the protons only b) the electrons only c) the neutrons only d) the protons
Question: Can we use our simple shell model of the atom to make some predictions?
 During Class Invention Question: Can we use our simple shell model of the atom to make some predictions? 1. Describe the nature of the interaction between protons and electrons in an atom? Consider using
During Class Invention Question: Can we use our simple shell model of the atom to make some predictions? 1. Describe the nature of the interaction between protons and electrons in an atom? Consider using
Unit 7. Atomic Structure
 Unit 7. Atomic Structure Upon successful completion of this unit, the students should be able to: 7.1 List the eight regions of the electromagnetic spectrum in the designated order and perform calculations
Unit 7. Atomic Structure Upon successful completion of this unit, the students should be able to: 7.1 List the eight regions of the electromagnetic spectrum in the designated order and perform calculations
An element Is a substance that cannot be split into simpler substance. It is composed of discrete particles called atoms.
 digitalteachers.co.ug Atomic structure Inorganic chemistry deals with the physical and chemical properties of the elements of the the periodic table. An element Is a substance that cannot be split into
digitalteachers.co.ug Atomic structure Inorganic chemistry deals with the physical and chemical properties of the elements of the the periodic table. An element Is a substance that cannot be split into
CSUS Department of Chemistry Experiment 9 Chem. 1A
 CSUS Department of Chemistry xperiment 9 Chem. 1A xp. 9 PR-Lab ASSIGNMNT Name: Lab Section (1) Use equation (2) [see the discussion on the next page] to calculate the energies of the ten lowest states
CSUS Department of Chemistry xperiment 9 Chem. 1A xp. 9 PR-Lab ASSIGNMNT Name: Lab Section (1) Use equation (2) [see the discussion on the next page] to calculate the energies of the ten lowest states
University Chemistry
 University Chemistry 1 Electromagnetic Radiation Electromagnetic radiation has some of the properties of both a particle and a wave. Particles: have a definite mass and they occupy space. Wave have no
University Chemistry 1 Electromagnetic Radiation Electromagnetic radiation has some of the properties of both a particle and a wave. Particles: have a definite mass and they occupy space. Wave have no
Laboratory Atomic Emission Spectrum
 Laboratory Atomic Emission Spectrum Pre-Lab Questions: Answer the following questions in complete sentences by reading through the Overview and Background sections below. 1. What is the purpose of the
Laboratory Atomic Emission Spectrum Pre-Lab Questions: Answer the following questions in complete sentences by reading through the Overview and Background sections below. 1. What is the purpose of the
Chapter 6. Quantum Theory and the Electronic Structure of Atoms Part 1
 Chapter 6 Quantum Theory and the Electronic Structure of Atoms Part 1 The nature of light Quantum theory Topics Bohr s theory of the hydrogen atom Wave properties of matter Quantum mechanics Quantum numbers
Chapter 6 Quantum Theory and the Electronic Structure of Atoms Part 1 The nature of light Quantum theory Topics Bohr s theory of the hydrogen atom Wave properties of matter Quantum mechanics Quantum numbers
Quantum Theory and the Electronic Structure of Atoms
 Quantum Theory and the Electronic Structure of Atoms Chapter 7 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Properties of Waves Wavelength ( ) is the distance
Quantum Theory and the Electronic Structure of Atoms Chapter 7 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Properties of Waves Wavelength ( ) is the distance
CHM 111 Unit 7 Sample Questions
 Name: Class: Date: As you work these problems, consider and explain: A. What type of question is it? B. How do you know what type of question it is? C. What information are you looking for? D. What information
Name: Class: Date: As you work these problems, consider and explain: A. What type of question is it? B. How do you know what type of question it is? C. What information are you looking for? D. What information
Sample Exercise 6.1 Concepts of Wavelength and Frequency
 Sample Exercise 6.1 Concepts of Wavelength and Frequency Two electromagnetic waves are represented in the margin. (a) Which wave has the higher frequency? (b) If one wave represents visible light and the
Sample Exercise 6.1 Concepts of Wavelength and Frequency Two electromagnetic waves are represented in the margin. (a) Which wave has the higher frequency? (b) If one wave represents visible light and the
Atomic Structure and Periodicity
 Atomic Structure and Periodicity Atoms and isotopes: Isotopes-#p + same for all but mass number is different b/c of # n o Average atomic mass is weighted average of all the isotopes for an element Average
Atomic Structure and Periodicity Atoms and isotopes: Isotopes-#p + same for all but mass number is different b/c of # n o Average atomic mass is weighted average of all the isotopes for an element Average
Atomic Structure. 1. For a hydrogen atom which electron transition requires the largest amount of energy?
 Atomic Structure 1. For a hydrogen atom which electron transition requires the largest amount of energy? A. n = 4 to n = 10 B. n = 3 to n = 2 C. n = 3 to n = 4 D. n = 1 to n = 3 E. n = 2 to n = 4 2. Which
Atomic Structure 1. For a hydrogen atom which electron transition requires the largest amount of energy? A. n = 4 to n = 10 B. n = 3 to n = 2 C. n = 3 to n = 4 D. n = 1 to n = 3 E. n = 2 to n = 4 2. Which
Chapter 7. The Quantum Mechanical Model of the Atom
 Chapter 7 The Quantum Mechanical Model of the Atom The Nature of Light:Its Wave Nature Light is a form of electromagnetic radiation composed of perpendicular oscillating waves, one for the electric field
Chapter 7 The Quantum Mechanical Model of the Atom The Nature of Light:Its Wave Nature Light is a form of electromagnetic radiation composed of perpendicular oscillating waves, one for the electric field
Academic Chemistry Chapter 5 Electrons in Atoms Notes
 Academic Chemistry Chapter 5 Electrons in Atoms Notes Name Date ATOMIC MODEL See Atomic Model Timeline worksheet for specifics. ELECTRONS Quantum Mechanical (QM) Model- This is the currently accepted model
Academic Chemistry Chapter 5 Electrons in Atoms Notes Name Date ATOMIC MODEL See Atomic Model Timeline worksheet for specifics. ELECTRONS Quantum Mechanical (QM) Model- This is the currently accepted model
CHEMISTRY - TRO 4E CH.7 - THE QUANTUM-MECHANICAL MODEL OF THE ATOM
 !! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
!! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
U N I T T E S T P R A C T I C E
 South Pasadena AP Chemistry Name 8 Atomic Theory Period Date U N I T T E S T P R A C T I C E Part 1 Multiple Choice You should allocate 25 minutes to finish this portion of the test. No calculator should
South Pasadena AP Chemistry Name 8 Atomic Theory Period Date U N I T T E S T P R A C T I C E Part 1 Multiple Choice You should allocate 25 minutes to finish this portion of the test. No calculator should
Chapter 7 Introduction to Spectroscopy
 Name AP CHEM / / Chapter 7 Introduction to Spectroscopy Spectroscopy is the study of the interaction of radiant energy and matter. The electromagnetic spectrum diagram is shown below to use a reference.
Name AP CHEM / / Chapter 7 Introduction to Spectroscopy Spectroscopy is the study of the interaction of radiant energy and matter. The electromagnetic spectrum diagram is shown below to use a reference.
Observation of Atomic Spectra
 Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
Chapter 5 Multiple Choice Questions
 Chapter 5 Multiple Choice Questions 1. What is the wavelength of radiation that has a frequency of 2.10 1014 s 1? A. 6.30 1022 m B. 7.00 102 nm C. 7.00 105 m D. 1.43 10 6 m E. 3.00 108 m 2. Calculate the
Chapter 5 Multiple Choice Questions 1. What is the wavelength of radiation that has a frequency of 2.10 1014 s 1? A. 6.30 1022 m B. 7.00 102 nm C. 7.00 105 m D. 1.43 10 6 m E. 3.00 108 m 2. Calculate the
Chemistry 111 Dr. Kevin Moore
 Chemistry 111 Dr. Kevin Moore Black Body Radiation Heated objects emit radiation based on its temperature Higher temperatures produce higher frequencies PhotoElectric Effect Light on a clean metal surface
Chemistry 111 Dr. Kevin Moore Black Body Radiation Heated objects emit radiation based on its temperature Higher temperatures produce higher frequencies PhotoElectric Effect Light on a clean metal surface
EM SPECTRUM, WAVELENGTH, FREQUENCY, AND ENERGY WORKSHEET
 EM SPECTRUM, WAVELENGTH, FREQUENCY, AND ENERGY WORKSHEET 1.) Look at the EM spectrum below to answer this question. As you move across the visible light spectrum from red to violet (A) Does the wavelength
EM SPECTRUM, WAVELENGTH, FREQUENCY, AND ENERGY WORKSHEET 1.) Look at the EM spectrum below to answer this question. As you move across the visible light spectrum from red to violet (A) Does the wavelength
Write the electron configuration for Chromium (Cr):
 Write the electron configuration for Chromium (Cr): Energy level Aufbau Principle Atomic orbital Quantum Hund s Rule Atomic number Electron Configuration Whole number Pauli Exlcusion Principle Quantum
Write the electron configuration for Chromium (Cr): Energy level Aufbau Principle Atomic orbital Quantum Hund s Rule Atomic number Electron Configuration Whole number Pauli Exlcusion Principle Quantum
AP Chapter 6 Study Questions
 Class: Date: AP Chapter 6 Study Questions True/False Indicate whether the statement is true or false. 1. The wavelength of radio waves can be longer than a football field. 2. Black body radiation is the
Class: Date: AP Chapter 6 Study Questions True/False Indicate whether the statement is true or false. 1. The wavelength of radio waves can be longer than a football field. 2. Black body radiation is the
Electrons, Energy, & the Electromagnetic Spectrum Notes Simplified, 2-D Bohr Model: Figure 2. Figure 3 UNIT 4 - ELECTRONS & ELECTRON ARRANGEMENT
 Electrons, Energy, & the Electromagnetic Spectrum Notes Simplified, 2-D Bohr Model: Figure 1 UNIT 4 - ELECTRONS & ELECTRON ARRANGEMENT Figure 2 Figure 3 The energy is released as electromagnetic radiation.
Electrons, Energy, & the Electromagnetic Spectrum Notes Simplified, 2-D Bohr Model: Figure 1 UNIT 4 - ELECTRONS & ELECTRON ARRANGEMENT Figure 2 Figure 3 The energy is released as electromagnetic radiation.
Unit 3. Chapter 4 Electrons in the Atom. Niels Bohr s Model. Recall the Evolution of the Atom. Bohr s planetary model
 Unit 3 Chapter 4 Electrons in the Atom Electrons in the Atom (Chapter 4) & The Periodic Table/Trends (Chapter 5) Niels Bohr s Model Recall the Evolution of the Atom He had a question: Why don t the electrons
Unit 3 Chapter 4 Electrons in the Atom Electrons in the Atom (Chapter 4) & The Periodic Table/Trends (Chapter 5) Niels Bohr s Model Recall the Evolution of the Atom He had a question: Why don t the electrons
Ch 7 Quantum Theory of the Atom (light and atomic structure)
 Ch 7 Quantum Theory of the Atom (light and atomic structure) Electromagnetic Radiation - Electromagnetic radiation consists of oscillations in electric and magnetic fields. The oscillations can be described
Ch 7 Quantum Theory of the Atom (light and atomic structure) Electromagnetic Radiation - Electromagnetic radiation consists of oscillations in electric and magnetic fields. The oscillations can be described
Chemistry 400 Note: Problem #21 is a separate 10-point assignment! I. Multiple Choice
 Chemistry 400 Homework 7, Chapter 7 Note: Problem #21 is a separate 10-point assignment! I. Multiple Choice 1. Which graph best describes the radial wavefunction of a 2s orbital? (A) (B) (C) (D) *2. The
Chemistry 400 Homework 7, Chapter 7 Note: Problem #21 is a separate 10-point assignment! I. Multiple Choice 1. Which graph best describes the radial wavefunction of a 2s orbital? (A) (B) (C) (D) *2. The
ATOMIC STRUCTURE ELECTRON CONFIGURATION 10/13/15 PROJECT DATE. Tuesday, October 13, 15
 PROJECT DATE ATOMIC STRUCTURE ELECTRON CONFIGURATION 10/13/15 Agenda Begin Topic 2.2 Electron Configuration Homework: Energy of waves calculations Due tomorrow! Yes - tomorrow Atomic Structure The nuclear
PROJECT DATE ATOMIC STRUCTURE ELECTRON CONFIGURATION 10/13/15 Agenda Begin Topic 2.2 Electron Configuration Homework: Energy of waves calculations Due tomorrow! Yes - tomorrow Atomic Structure The nuclear
Energy and the Quantum Theory
 Energy and the Quantum Theory Light electrons are understood by comparing them to light 1. radiant energy 2. travels through space 3. makes you feel warm Light has properties of waves and particles Amplitude:
Energy and the Quantum Theory Light electrons are understood by comparing them to light 1. radiant energy 2. travels through space 3. makes you feel warm Light has properties of waves and particles Amplitude:
2) The energy of a photon of light is proportional to its frequency and proportional to its wavelength.
 Advanced Chemistry Chapter 13 Review Name Per Show all work Wave Properties 1) Which one of the following is correct? A) ν + λ = c B) ν λ = c C) ν = cλ D) λ = c ν E) νλ = c 2) The energy of a photon of
Advanced Chemistry Chapter 13 Review Name Per Show all work Wave Properties 1) Which one of the following is correct? A) ν + λ = c B) ν λ = c C) ν = cλ D) λ = c ν E) νλ = c 2) The energy of a photon of
Name Class Date. Chapter: Arrangement of Electrons in Atoms
 Assessment Chapter Test A Chapter: Arrangement of Electrons in Atoms In the space provided, write the letter of the term that best completes each sentence or best answers each question. 1. Which of the
Assessment Chapter Test A Chapter: Arrangement of Electrons in Atoms In the space provided, write the letter of the term that best completes each sentence or best answers each question. 1. Which of the
Provide a short and specific definition in YOUR OWN WORDS. Do not use the definition from the book. Electromagnetic Radiation
 Name: Provide a short and specific definition in YOUR OWN WORDS. Do not use the definition from the book Additional Notes: Electromagnetic Radiation Electromagnetic Spectrum Wavelength Frequency Photoelectric
Name: Provide a short and specific definition in YOUR OWN WORDS. Do not use the definition from the book Additional Notes: Electromagnetic Radiation Electromagnetic Spectrum Wavelength Frequency Photoelectric
Table of Contents Electrons in Atoms > Light and Quantized Energy > Quantum Theory and the Atom > Electron Configuration
 Electrons in Atoms October 20, 2014 Table of Contents Electrons in Atoms > Light and Quantized Energy > Quantum Theory and the Atom > Electron Configuration 1 Electromagnetic Spectrum Electromagnetic radiation
Electrons in Atoms October 20, 2014 Table of Contents Electrons in Atoms > Light and Quantized Energy > Quantum Theory and the Atom > Electron Configuration 1 Electromagnetic Spectrum Electromagnetic radiation
2 Examiner SECTION A. Answer all the questions in the spaces provided.
 2 SECTION A Answer all the questions in the spaces provided. 1. An isotope of magnesium, 27 Mg, is used to detect leaks in water pipes. (a) It decays by β emission with a half life of 9 5 minutes. (i)
2 SECTION A Answer all the questions in the spaces provided. 1. An isotope of magnesium, 27 Mg, is used to detect leaks in water pipes. (a) It decays by β emission with a half life of 9 5 minutes. (i)
Color. 3. Why are the color labels in the table above plural (i.e., Reds rather than Red )?
 NS D3 Electron Energy and Light Name From fireworks to stars, the color of light is useful in finding out what s in matter. The emission of light by hydrogen and other atoms has played a key role in understanding
NS D3 Electron Energy and Light Name From fireworks to stars, the color of light is useful in finding out what s in matter. The emission of light by hydrogen and other atoms has played a key role in understanding
Periodicity and the Electronic Structure of Atoms 國防醫學院生化學科王明芳老師
 Periodicity and the Electronic Structure of Atoms 國防醫學院生化學科王明芳老師 2018-10-2 1 2 Light and the Electromagnetic Spectrum Electromagnetic energy ( light ) is characterized by wavelength, frequency, and amplitude.
Periodicity and the Electronic Structure of Atoms 國防醫學院生化學科王明芳老師 2018-10-2 1 2 Light and the Electromagnetic Spectrum Electromagnetic energy ( light ) is characterized by wavelength, frequency, and amplitude.
Atomic Structure and the Periodic Table
 Atomic Structure and the Periodic Table The electronic structure of an atom determines its characteristics Studying atoms by analyzing light emissions/absorptions Spectroscopy: analysis of light emitted
Atomic Structure and the Periodic Table The electronic structure of an atom determines its characteristics Studying atoms by analyzing light emissions/absorptions Spectroscopy: analysis of light emitted
Background: The Electromagnetic Spectrum
 Background: The Electromagnetic Spectrum Wavelength (λ) in meters wavelength decreasing 10 4 10 2 10 0 10-2 10-4 10-6 10-8 10-10 10-12 10-14 microwaves ultraviolet Gamma rays Radio waves AM 10 4 Shortwave
Background: The Electromagnetic Spectrum Wavelength (λ) in meters wavelength decreasing 10 4 10 2 10 0 10-2 10-4 10-6 10-8 10-10 10-12 10-14 microwaves ultraviolet Gamma rays Radio waves AM 10 4 Shortwave
Chemistry 1A, Fall 2004 Midterm Exam I, Version 1 Sept 21, 2004 (90 min, closed book)
 Chemistry 1A, Fall 2004 Midterm Exam I, Version 1 Sept 21, 2004 (90 min, closed book) Name: SID: Identification Sticker TA Name: Write your name on every page of this exam. This exam has 40 multiple choice
Chemistry 1A, Fall 2004 Midterm Exam I, Version 1 Sept 21, 2004 (90 min, closed book) Name: SID: Identification Sticker TA Name: Write your name on every page of this exam. This exam has 40 multiple choice
General Chemistry by Ebbing and Gammon, 8th Edition
 Chem 1045 General Chemistry by Ebbing and Gammon, 8th Edition George W.J. Kenney, Jr Last Update: 26-Mar-2009 Chapter 7: Quantum Theory of the Atom These Notes are to SUPPLIMENT the Text, They do NOT Replace
Chem 1045 General Chemistry by Ebbing and Gammon, 8th Edition George W.J. Kenney, Jr Last Update: 26-Mar-2009 Chapter 7: Quantum Theory of the Atom These Notes are to SUPPLIMENT the Text, They do NOT Replace
Chapter 7 Quantum Theory and the Electronic Structure of Atoms
 Chapter 7 Quantum Theory and the Electronic Structure of Atoms 1- T/F 1. The Bohr model of the hydrogen atom found its greatest support in experimental work on the photoelectric effect. (a) True (b) False
Chapter 7 Quantum Theory and the Electronic Structure of Atoms 1- T/F 1. The Bohr model of the hydrogen atom found its greatest support in experimental work on the photoelectric effect. (a) True (b) False
1 Electrons are emitted from a metal surface when it is illuminated with suitable electromagnetic radiation. ...[1]
![1 Electrons are emitted from a metal surface when it is illuminated with suitable electromagnetic radiation. ...[1] 1 Electrons are emitted from a metal surface when it is illuminated with suitable electromagnetic radiation. ...[1]](/thumbs/81/83978040.jpg) 1 Electrons are emitted from a metal surface when it is illuminated with suitable electromagnetic radiation. 1 (a) (b) Name the effect described above....[1] The variation with frequency f of the maximum
1 Electrons are emitted from a metal surface when it is illuminated with suitable electromagnetic radiation. 1 (a) (b) Name the effect described above....[1] The variation with frequency f of the maximum
Atomic Term Symbols and Energy Splitting. λ=5890 Å
 Chemistry 362 Spring 2018 Dr. Jean M. Standard April 18, 2018 Atomic Term Symbols and Energy Splitting 1. Atomic Term Symbols and the Sodium D-Line The sodium D-line is responsible for the familiar orange
Chemistry 362 Spring 2018 Dr. Jean M. Standard April 18, 2018 Atomic Term Symbols and Energy Splitting 1. Atomic Term Symbols and the Sodium D-Line The sodium D-line is responsible for the familiar orange
Chapter 6 Electronic Structure of Atoms. 許富銀 ( Hsu Fu-Yin)
 Chapter 6 Electronic Structure of Atoms 許富銀 ( Hsu Fu-Yin) 1 The Wave Nature of Light The light we see with our eyes, visible light, is one type of electromagnetic radiation. electromagnetic radiation carries
Chapter 6 Electronic Structure of Atoms 許富銀 ( Hsu Fu-Yin) 1 The Wave Nature of Light The light we see with our eyes, visible light, is one type of electromagnetic radiation. electromagnetic radiation carries
Atomic Theory: Spectroscopy and Flame Tests
 Atomic Theory: Spectroscopy and Flame Tests Introduction Light energy is also known as electromagnetic (EM) radiation. The light that we observe with our eyes, visible light, is just a small portion of
Atomic Theory: Spectroscopy and Flame Tests Introduction Light energy is also known as electromagnetic (EM) radiation. The light that we observe with our eyes, visible light, is just a small portion of
Unit 1. Electronic Structure page 1
 Unit 1 Electronic Structure Section 1. Learning Outcomes Practice Questions Answers Electronic Structure Electromagnetic spectrum / calculations Electron configuration / Periodic Table Electronic Structure
Unit 1 Electronic Structure Section 1. Learning Outcomes Practice Questions Answers Electronic Structure Electromagnetic spectrum / calculations Electron configuration / Periodic Table Electronic Structure
Ch 9 Electrons in Atoms & the Periodic Table Study Sheet Acc. Chemistry SCANTRON. Name /99. 3) Light is a type of matter. 3)
 Ch 9 Electrons in Atoms & the Periodic Table Study Sheet Acc. Chemistry SCANTRON Name /99 TRUE/FALSE. Write 'T' if the statement is true and 'F' if the statement is false. 1) When the elements are arranged
Ch 9 Electrons in Atoms & the Periodic Table Study Sheet Acc. Chemistry SCANTRON Name /99 TRUE/FALSE. Write 'T' if the statement is true and 'F' if the statement is false. 1) When the elements are arranged
Lesmahagow High School AHChemistry Inorganic and Physical Chemistry Lesmahagow High School CfE Advanced Higher Chemistry
 Lesmahagow High School CfE Advanced Higher Chemistry Unit 1 Inorganic and Physical Chemistry Electromagnetic Radiation and Atomic Spectra 1 Electromagnetic Radiation Radiation such as light, microwaves,
Lesmahagow High School CfE Advanced Higher Chemistry Unit 1 Inorganic and Physical Chemistry Electromagnetic Radiation and Atomic Spectra 1 Electromagnetic Radiation Radiation such as light, microwaves,
Lecture 11 Atomic Structure
 Lecture 11 Atomic Structure Earlier in the semester, you read about the discoveries that lead to the proposal of the nuclear atom, an atom of atomic number Z, composed of a positively charged nucleus surrounded
Lecture 11 Atomic Structure Earlier in the semester, you read about the discoveries that lead to the proposal of the nuclear atom, an atom of atomic number Z, composed of a positively charged nucleus surrounded
Chemistry 1A Midterm Exam 1 February 12, Potentially Useful Information. Violet Blue Green Yellow Orange Red Wavelength (nm)
 Chemistry 1A Midterm Exam 1 February 12, 2013 Professor Pines 5 pages total Student Name: Student ID#: Potentially Useful Information Violet Blue Green Yellow Orange Red 400 500 600 700 Wavelength (nm)
Chemistry 1A Midterm Exam 1 February 12, 2013 Professor Pines 5 pages total Student Name: Student ID#: Potentially Useful Information Violet Blue Green Yellow Orange Red 400 500 600 700 Wavelength (nm)
Unit 3: Electron configuration and periodicity
 Unit 3: Electron configuration and periodicity Group 1 BOHR MODELS Group 18 H Group 2 Group 13 Group 14 Group 15 Group 16 Group 17 He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca His theory couldn t
Unit 3: Electron configuration and periodicity Group 1 BOHR MODELS Group 18 H Group 2 Group 13 Group 14 Group 15 Group 16 Group 17 He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca His theory couldn t
Chemistry 1A, Fall 2003 Midterm 1 Sept 16, 2003 (90 min, closed book)
 Name: SID: TA Name: Chemistry 1A, Fall 2003 Midterm 1 Sept 16, 2003 (90 min, closed book) This exam has 38 multiple choice questions. Fill in the Scantron form AND circle your answer on the exam. Each
Name: SID: TA Name: Chemistry 1A, Fall 2003 Midterm 1 Sept 16, 2003 (90 min, closed book) This exam has 38 multiple choice questions. Fill in the Scantron form AND circle your answer on the exam. Each
Chapter 6 Electronic Structure of Atoms
 Chapter 6. Electronic Structure of Atoms NOTE: Review your notes from Honors or regular Chemistry for the sequence of atomic models and the evidence that allowed scientists to change the model. If you
Chapter 6. Electronic Structure of Atoms NOTE: Review your notes from Honors or regular Chemistry for the sequence of atomic models and the evidence that allowed scientists to change the model. If you
29:006 FINAL EXAM FRIDAY MAY 11 3:00 5:00 PM IN LR1 VAN
 L 33 Modern Physics [1] 29:006 FINAL EXAM FRIDAY MAY 11 3:00 5:00 PM IN LR1 VAN Introduction- quantum physics Particles of light PHOTONS The photoelectric effect Photocells & intrusion detection devices
L 33 Modern Physics [1] 29:006 FINAL EXAM FRIDAY MAY 11 3:00 5:00 PM IN LR1 VAN Introduction- quantum physics Particles of light PHOTONS The photoelectric effect Photocells & intrusion detection devices
What are the isotopes of hydrogen? H, 2 H, and 3 H
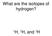 What are the isotopes of hydrogen? 1 H, 2 H, and 3 H The average mass of a boron atom is 10.81. Assuming you were able to isolate only one boron atom, the chance that you would randomly get one with a
What are the isotopes of hydrogen? 1 H, 2 H, and 3 H The average mass of a boron atom is 10.81. Assuming you were able to isolate only one boron atom, the chance that you would randomly get one with a
The Bohr Model of the Atom
 Unit 4: The Bohr Model of the Atom Properties of light Before the 1900 s, light was thought to behave only as a wave. Light is a type of electromagnetic radiation - a form of energy that exhibits wave
Unit 4: The Bohr Model of the Atom Properties of light Before the 1900 s, light was thought to behave only as a wave. Light is a type of electromagnetic radiation - a form of energy that exhibits wave
The Quantum Mechanical Atom
 The Quantum Mechanical Atom CHAPTER 7 Chemistry: The Molecular Nature of Matter, 6 th edition By Jesperson, Brady, & Hyslop CHAPTER 8: Quantum Mechanical Atom Learning Objectives q Light as Waves, Wavelength
The Quantum Mechanical Atom CHAPTER 7 Chemistry: The Molecular Nature of Matter, 6 th edition By Jesperson, Brady, & Hyslop CHAPTER 8: Quantum Mechanical Atom Learning Objectives q Light as Waves, Wavelength
CHEMISTRY - ZUMDAHL 8E CH.7 - ATOMIC STRUCTURE & PERIODICITY.
 !! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
!! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
Chapter 6. Electronic Structure of Atoms
 Chapter 6. Electronic Structure of Atoms NOTE: Review your notes from Honors or regular Chemistry for the sequence of atomic models and the evidence that allowed scientists to change the model. If you
Chapter 6. Electronic Structure of Atoms NOTE: Review your notes from Honors or regular Chemistry for the sequence of atomic models and the evidence that allowed scientists to change the model. If you
Chapter 7. The Quantum Mechanical Model of the Atom
 Chapter 7 The Quantum Mechanical Model of the Atom Quantum Mechanics The Behavior of the Very Small Electrons are incredibly small. Electron behavior determines much of the behavior of atoms. Directly
Chapter 7 The Quantum Mechanical Model of the Atom Quantum Mechanics The Behavior of the Very Small Electrons are incredibly small. Electron behavior determines much of the behavior of atoms. Directly
Electromagnetic Radiation. is a form of energy that exhibits wavelike behavior as it travels through space.
 Electromagnetic Radiation is a form of energy that exhibits wavelike behavior as it travels through space. What are the 7 forms of electromagnetic radiation, in order of INCREASING wavelength? gamma rays
Electromagnetic Radiation is a form of energy that exhibits wavelike behavior as it travels through space. What are the 7 forms of electromagnetic radiation, in order of INCREASING wavelength? gamma rays
Physics and the Quantum Mechanical Model
 chemistry 1 of 38 Mechanical Model Neon advertising signs are formed from glass tubes bent in various shapes. An electric current passing through the gas in each glass tube makes the gas glow with its
chemistry 1 of 38 Mechanical Model Neon advertising signs are formed from glass tubes bent in various shapes. An electric current passing through the gas in each glass tube makes the gas glow with its
Einstein. Quantum Physics at a glance. Planck s Hypothesis (blackbody radiation) (ultraviolet catastrophe) Quantized Energy
 Quantum Physics at a glance Quantum Physics deals with the study of light and particles at atomic and smaller levels. Planck s Hypothesis (blackbody radiation) (ultraviolet catastrophe) Quantized Energy
Quantum Physics at a glance Quantum Physics deals with the study of light and particles at atomic and smaller levels. Planck s Hypothesis (blackbody radiation) (ultraviolet catastrophe) Quantized Energy
