Evidence of cross-hybridization artifact in expressed sequence tags (ESTs) on cdna microarrays
|
|
|
- Pamela Boone
- 5 years ago
- Views:
Transcription
1 E -b (EST) DA D H, Sb D P Rb O D M F L W P S, R S C G, b, Ab W -b - DA DA - (T). T (T), (T), (EST) DA RA. A b DA EST - T b -. O (T) b -b RA. T b DA b b b EST (T). I, DA (T) DA b. K: DA M, E-T S, EST, C-b, P(T), P(A),. ITRODUCTIO S DA,,, /. T ( ), ( ). M, b. A, b () b C, H, Sb, P, O D, F, W, S, S, G,. C M U, D S U Pb, S Pb H, H G U Pb, S M, D C M U, D P I H M C, U W F
2 () b. T, b, (.., ). O, : b b,,, b b b. I b. S, DA (EST) (T). W -. RESULTS D. W DA, - DA U Pb (P. b F. b ) - (C/C) b S M Db b (B. ). W. A, b b. P (T) b b. W b RA (T). I (P.) % b b T. A (EST). I, % b (T). U b, (T). E. I b,. A (b), ( ). O, (T). O b b, (T). T b, -b, RA. E (T). W (T). T (), (), (). I,, : (T). I b (T), T b,. I, W - DA. T CBI b AI T b C.
3 -, (T) (-) (-); b. O, -, b. E b (T). T b (T), b (T). F C =,,..., b : V = C V (X ) C V (X ) C (T) V (X ) (.., ). W V b b C,, T-, (,, C V, ). T,, b C b (T). F - DA, b (T). H, -, b b (T). E b (T). W b b (T) b. E b, C ( b T, ). F, (,,, b ). F, b (T). T, - DA, b ( P./F. ), (T) ( ). T b, T- b b b. T. T b. I, b b b b b ( ) (T), b. T : H : F, (T). H : I. P-. Tb -. A α =.,, H, DA DA. - α =. -. T DA (T). T b E. H, -.
4 - DA b (T) b (T) -. TZD b b Tb : E - - DA, - DA, -(C/C) DISCUSSIO E. E (EST) (- b) DA b. T, RA,, (.., ), / (.., ). T RA -b (T). T DA,, -, b b [B]. B b, EST (%)[Wb & L], b. EST. W DA, RA -A. T - (T) b GB. T EST - (T) b -b. S DA b b b b- () DA, b b DA A T b EST DA (T). T b. O b, b, - b -- b. H, b. W b b -b. S DA b-, -b b b -A (T). I,, b b -A DA. T b EST b,. W (T), CBI GB,. T-. A - DA b -. I -, -b b ( ) b,. O -.
5 H, - EST (T), b -b. L -b b. T b. T, (.., C C) b, -- b (T). I. E EST b. A -b b. W b DA. I, - EST (T). S. B, -b DA, b AOVA,,,. F,, b b. I F & D (), b b b. METHODS. TZD- T DA RA TL - (TZD)(P,. b ). C T (I C., Cb, CA) RA. RA /. DA b RA. T RA./L (T) C. R X - b (I C., Cb, CA).L (U/µL SS II RT; I C.),.µ DTT (.M),.L TP (ATP,GTP,TTP M), µ P CTP ( C/, C/) R (U/, P C., M, WI) C. E b b Q S G- S C (R D C., I, I) C b. DA (GF, GF M; R G, Cb, CA), DA,. T b L Mb (R G), µl P-A (./L, R G), µl C- H DA ( C,./L, I C.) C. T b b b b C. A b, C L X (SSC) b % S (SDS) C.X SSC b % SDS. P-b PI (M D, S, CA) S (M D). S IQ (M D).
6 . Cb T DA b (MCA) (SMC) b (SAH) b (CSF) RA (F. b ). O, b b b b,,. SAH CSF - b MCA SMC. T MCA SMC RA DA. U b. RA T (L T). T RA S b RA. DA b b b. Hb b µ RA. RA.µ/µ C. R X - b (I C., Cb, CA) (U/µ SS II RT; I C.), µ/ L - - (- - P-b, µc ), R (U/, P C., M, WI),.M DTT C. E b b Q S G- S C (R D C., I, I) C. S (GF, GF M; R G, Cb, CA). T b Mb (R G, Cb, CA), µ P-A (./, R G, Cb, C), µ C- H DA ( C,.µ/µ, I C., Cb, CA) C. T b b b b C. A b, C X S (SSC) b % S (SDS) C.X SSC b % SDS. T PI (M D, S, CA) b G GF. O, IQ (M D, S, CA). F TE b C. Hb b, b G PI.. B Pb b b S M Db (://.S.EDU/MA/SMD/). B.. b b b - C/C [B]. M b b (://..//.).
7 . S T. T b b b b b b. H. T H : F, (T). H : I. P-. T - b : S : F b β b -T : (M,.., M ) (T) =,,..., ). S : P (M,.., M ) b b (M τ(),.., M τ() ) -T. D. T B b : β b b =,..., B. S : T - b b b b : = B b=,..,b I(β b > β b ). A. F (),, b. ACKOWLEDGMETS T J K J R. T b ASA CC-. REFERECES. B J.C.,.. S b. P A S. (), - ().. B M.F., L G., S M.B. b:. G R. () - ().. D, A. C., H, D. V. B M A, Cb U P ().. F, J., D, S. A b, # ( ).. S, M., DA M: A P A, O U P ().. Wb, T.G., L, D., B: A P G A G P, S E. A D. B W-I, ().. WEB SITE REFERECES H b. τ,...,.
8 . S M Db ://-.S.EDU/MA/SMD/. B. M b ://-..//.. C B I, E ://.b.../e/ b b b b b b F : T ( b ) TZD-.
9 IDETIFIERS best I: EST :. GB A: AI GB : CLOE IFO C I: IMAGE: ( ) S: IMAGE C, LLL DA : DA SEQUECE TTTTTTTTTTTTTTTACAGGGAAACAGCATTTTTAATGTTTTATTTT TCACTTGTGAAAAATATATATAATATATCTTCCACATACAGAAGAGTC CTGCAGCTTGAGTCAGAGGAAGCTGAAAGAAAGGCACATACAGGGA GCAGATCTTTCCATACAGTTTTCAGTTAAACCAGCATTCAGGGCACA GCAAGTGACAACAAAAGCCCAGGCTGCCTGTGCACACGACTCTGAA GAGAAACATCACAGACAACCCTTAGGTCTACCATGAATGGTTTCTAT TTAATAGACCTATCAGACCACCCGGGAACAAATTGATACCTGAACAA AGACACACCAGGGCAAGACAAAT F : E EST b CBI GB b. T (T). TZD ()..... T (b).... T () T F : () M (T). (T); (b) P, V, b b C. (T),, =,,,..; () b TZD-, b C
10 C. B ().... T (b)... T () T F : T F, b b. Cb ().... T (b)... T () T F : T F, b -.
Supporting Information. Organocatalytic Synthesis of 4-Aryl-1,2,3,4-tetrahydropyridines from. Morita-Baylis-Hillman Carbonates through a One-Pot
 Supporting Information Organocatalytic Synthesis of 4-Aryl-1,2,3,4-tetrahydropyridines from Morita-Baylis-Hillman Carbonates through a One-Pot Three-Component Cyclization Jian Wei, Yuntong Li, Cheng Tao,
Supporting Information Organocatalytic Synthesis of 4-Aryl-1,2,3,4-tetrahydropyridines from Morita-Baylis-Hillman Carbonates through a One-Pot Three-Component Cyclization Jian Wei, Yuntong Li, Cheng Tao,
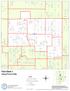
Flint Ward 1 !( 1. Voting Precinct Map «13 «15 «10. Legend. Precinct Number. Precinct Boundaries. Streets. Hydrography. Date: 1/18/2017.
 T c K Tb K p b b F H O N F H ff ff p V H G F Y 2 Rx G J J Kcbc T H F H p p p p H Hb Gc O R IOOO RTENT V Jc K Ox F Hb Ox F G x R b 6 1 R F R b c j p G E F 1 R p bb G H Gc Y Hb 2 F 3 4 b R K K V R H p Rbb
T c K Tb K p b b F H O N F H ff ff p V H G F Y 2 Rx G J J Kcbc T H F H p p p p H Hb Gc O R IOOO RTENT V Jc K Ox F Hb Ox F G x R b 6 1 R F R b c j p G E F 1 R p bb G H Gc Y Hb 2 F 3 4 b R K K V R H p Rbb
RESOURCE, SUPPORT, AND DEVELOPMENT, INC
 RESOURCE, SUPPORT, AND DEVELOPMENT, INC P w b B, H, Lww, R L, M A Pb RSD, I S 2006 Vm 4 BOARD OF DIRECTORS P P E K V-P B R S L B-Sw L T N Ew A DB D S ADMINISTRATIVE TEAM CEO R M Hm R D J Sz Ex S E Lm F
RESOURCE, SUPPORT, AND DEVELOPMENT, INC P w b B, H, Lww, R L, M A Pb RSD, I S 2006 Vm 4 BOARD OF DIRECTORS P P E K V-P B R S L B-Sw L T N Ew A DB D S ADMINISTRATIVE TEAM CEO R M Hm R D J Sz Ex S E Lm F
Help us transform the Central Suburbs bus network Tell us what you think
 Hv F p p 1 p 2 p 3 b pp cg p f vc pg 5 6 Cc f vc f pg 3 T v: Fp fbc f pg 8, fbc f AT.gv.z/NN c f AT.gv.z/NN? C v ( pg 4) c (09) 366 6400. Hp f C Sbb b T Fbc p f 1 Ocb 10 Dcb 2015 vg pbc p f Ac f fg Ac
Hv F p p 1 p 2 p 3 b pp cg p f vc pg 5 6 Cc f vc f pg 3 T v: Fp fbc f pg 8, fbc f AT.gv.z/NN c f AT.gv.z/NN? C v ( pg 4) c (09) 366 6400. Hp f C Sbb b T Fbc p f 1 Ocb 10 Dcb 2015 vg pbc p f Ac f fg Ac
Stackable Storage & Dispensing Systems
 Sb Sg & Dg S VE OMOVE OMOVE OMOVE OMO GC GC GC G NDS NDS NDS NDS ND -w v b q 0 0 0 07 MN. 80 S 0 M b g g w b. g w v: w g g b w W b g v S b. b v v b q. j b q g! g j N - O M O V E BE OF CONENS: SCKBE NKS...
Sb Sg & Dg S VE OMOVE OMOVE OMOVE OMO GC GC GC G NDS NDS NDS NDS ND -w v b q 0 0 0 07 MN. 80 S 0 M b g g w b. g w v: w g g b w W b g v S b. b v v b q. j b q g! g j N - O M O V E BE OF CONENS: SCKBE NKS...
Inside! Your Impact...p 5 How You Raised Awareness...p 9 The Bigger Picture...p 14
 Ii! Y I... 5 H Y Ri A... 9 T Bi Pi... 14 W W v B, W W Gi f b i T. i fii) Fi F v (211 iq M, f i D F fii i i i. i, xii W. b 7 201 i f k ik f xiv f i T v 2017 i i i i i, i i i x ff fi fi-v i fi x M Fi W.
Ii! Y I... 5 H Y Ri A... 9 T Bi Pi... 14 W W v B, W W Gi f b i T. i fii) Fi F v (211 iq M, f i D F fii i i i. i, xii W. b 7 201 i f k ik f xiv f i T v 2017 i i i i i, i i i x ff fi fi-v i fi x M Fi W.
Executive Committee and Officers ( )
 Gifted and Talented International V o l u m e 2 4, N u m b e r 2, D e c e m b e r, 2 0 0 9. G i f t e d a n d T a l e n t e d I n t e r n a t i o n a2 l 4 ( 2), D e c e m b e r, 2 0 0 9. 1 T h e W o r
Gifted and Talented International V o l u m e 2 4, N u m b e r 2, D e c e m b e r, 2 0 0 9. G i f t e d a n d T a l e n t e d I n t e r n a t i o n a2 l 4 ( 2), D e c e m b e r, 2 0 0 9. 1 T h e W o r
CHM 101 PRACTICE TEST 1 Page 1 of 4
 CHM 101 PRACTICE TEST 1 Page 1 of 4 Please show calculations (stuffed equations) on all mathematical problems!! On the actual test, "naked answers, with no work shown, will receive no credit even if correct.
CHM 101 PRACTICE TEST 1 Page 1 of 4 Please show calculations (stuffed equations) on all mathematical problems!! On the actual test, "naked answers, with no work shown, will receive no credit even if correct.
to Highbury via Massey University, Constellation Station, Smales Farm Station, Akoranga Station and Northcote
 b v Nc, g, F, C Uv Hgb p (p 4030) F g (p 4063) F (p 3353) L D (p 3848) 6.00 6.0 6.2 6.20 6.30 6.45 6.50 6.30 6.40 6.42 6.50 7.00 7.5 7.20 7.00 7.0 7.2 7.20 7.30 7.45 7.50 7.30 7.40 7.42 7.50 8.00 8.5 8.20
b v Nc, g, F, C Uv Hgb p (p 4030) F g (p 4063) F (p 3353) L D (p 3848) 6.00 6.0 6.2 6.20 6.30 6.45 6.50 6.30 6.40 6.42 6.50 7.00 7.5 7.20 7.00 7.0 7.2 7.20 7.30 7.45 7.50 7.30 7.40 7.42 7.50 8.00 8.5 8.20
The Periodic Table of Elements
 The Periodic Table of Elements 8 Uuo Uus Uuh (9) Uup (88) Uuq (89) Uut (8) Uub (8) Rg () 0 Ds (9) 09 Mt (8) 08 Hs (9) 0 h () 0 Sg () 0 Db () 0 Rf () 0 Lr () 88 Ra () 8 Fr () 8 Rn () 8 At (0) 8 Po (09)
The Periodic Table of Elements 8 Uuo Uus Uuh (9) Uup (88) Uuq (89) Uut (8) Uub (8) Rg () 0 Ds (9) 09 Mt (8) 08 Hs (9) 0 h () 0 Sg () 0 Db () 0 Rf () 0 Lr () 88 Ra () 8 Fr () 8 Rn () 8 At (0) 8 Po (09)
[ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34.
![[ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34. [ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34.](/thumbs/74/69911105.jpg) .. - 2-2009 [661.87.+661.88]:543.4(075.8) 35.20:34.2373-60..,..,..,..,.. -60 : /... ;. 2-. : -, 2008. 134. ISBN 5-98298-299-7 -., -,,. - «,, -, -», - 550800,, 240600 «-», -. [661.87.+661.88]:543.4(075.8)
.. - 2-2009 [661.87.+661.88]:543.4(075.8) 35.20:34.2373-60..,..,..,..,.. -60 : /... ;. 2-. : -, 2008. 134. ISBN 5-98298-299-7 -., -,,. - «,, -, -», - 550800,, 240600 «-», -. [661.87.+661.88]:543.4(075.8)
Last 4 Digits of USC ID:
 Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
Advanced Placement. Chemistry. Integrated Rates
 Advanced Placement Chemistry Integrated Rates 204 47.90 9.22 78.49 (26) 50.94 92.9 80.95 (262) 52.00 93.94 83.85 (263) 54.938 (98) 86.2 (262) 55.85 0. 90.2 (265) 58.93 02.9 92.2 (266) H Li Na K Rb Cs Fr
Advanced Placement Chemistry Integrated Rates 204 47.90 9.22 78.49 (26) 50.94 92.9 80.95 (262) 52.00 93.94 83.85 (263) 54.938 (98) 86.2 (262) 55.85 0. 90.2 (265) 58.93 02.9 92.2 (266) H Li Na K Rb Cs Fr
University of Toronto. Final Exam
 University of Toronto Final Exam Date - Dec 16, 013 Duration:.5 hrs ECE331 Electronic Circuits Lecturer - D. Johns ANSWER QUESTIONS ON THESE SHEETS USING BACKS IF NECESSARY 1. Equation sheet is on last
University of Toronto Final Exam Date - Dec 16, 013 Duration:.5 hrs ECE331 Electronic Circuits Lecturer - D. Johns ANSWER QUESTIONS ON THESE SHEETS USING BACKS IF NECESSARY 1. Equation sheet is on last
American Cakery. ... made by erlenbacher
 A C P O D W P P- C / 1050 NEW C-Cff -C S 1700 1 7/6 105 NEW Bb-C S 1900 1 7/6 5 10519 NEW C-B-C S 1950 1 7/6 5 1056 NEW Rb-G-C S 1700 1 7/6 6 1055 NEW R B 1100 16 1/1 7 1051 NEW C B 750 1 1/1 10 101051
A C P O D W P P- C / 1050 NEW C-Cff -C S 1700 1 7/6 105 NEW Bb-C S 1900 1 7/6 5 10519 NEW C-B-C S 1950 1 7/6 5 1056 NEW Rb-G-C S 1700 1 7/6 6 1055 NEW R B 1100 16 1/1 7 1051 NEW C B 750 1 1/1 10 101051
4 8 N v btr 20, 20 th r l f ff nt f l t. r t pl n f r th n tr t n f h h v lr d b n r d t, rd n t h h th t b t f l rd n t f th rld ll b n tr t d n R th
 n r t d n 20 2 :24 T P bl D n, l d t z d http:.h th tr t. r pd l 4 8 N v btr 20, 20 th r l f ff nt f l t. r t pl n f r th n tr t n f h h v lr d b n r d t, rd n t h h th t b t f l rd n t f th rld ll b n
n r t d n 20 2 :24 T P bl D n, l d t z d http:.h th tr t. r pd l 4 8 N v btr 20, 20 th r l f ff nt f l t. r t pl n f r th n tr t n f h h v lr d b n r d t, rd n t h h th t b t f l rd n t f th rld ll b n
August 30, 2013 Volume 85 Issue 2 echo.snu.edu 6612 NW 42nd St. Bethany, OK (405)
 B S B OKC w TE Ag 30, 2013 V 85 I 2 d 6612 NW 42d S B, OK 73008 (405) 491-6382 R f v j xd P b K Rb K Rb, Ed--f E, v d f bdg vg f d d b d d f Of, b f g j, d Bd j g w d d g, SNU d g- j w fw T w d vd bdg
B S B OKC w TE Ag 30, 2013 V 85 I 2 d 6612 NW 42d S B, OK 73008 (405) 491-6382 R f v j xd P b K Rb K Rb, Ed--f E, v d f bdg vg f d d b d d f Of, b f g j, d Bd j g w d d g, SNU d g- j w fw T w d vd bdg
CHEM 10113, Quiz 5 October 26, 2011
 CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
(please print) (1) (18) H IIA IIIA IVA VA VIA VIIA He (2) (13) (14) (15) (16) (17)
 CHEM 10113, Quiz 3 September 28, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 3 September 28, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
2014 CANADIAN SURF LIFESAVING CHAMPIONSHIPS PROGRAM
 2014 CANAIAN URF LIFEAVING CHAMPIONHIP PROGRAM Lv Lv vy b v, w,, w Lv y w k Lv z by I Oy C (IOC) Cw G F T IOC z I L v F (IL) w v v IL N Mb Oz v b v T Lv y v by v C I Lv W C z I L v F Cw Lv C z Ry L v y
2014 CANAIAN URF LIFEAVING CHAMPIONHIP PROGRAM Lv Lv vy b v, w,, w Lv y w k Lv z by I Oy C (IOC) Cw G F T IOC z I L v F (IL) w v v IL N Mb Oz v b v T Lv y v by v C I Lv W C z I L v F Cw Lv C z Ry L v y
BRAUER Clamp Cross Reference BRAUER# Equal/Similar DESTACO # CARR-LANE # STANDARD.VERTICAL.CLAMPS V75/1B E 201-B CL-151-VTC N/A 201-TB N/A V75/2B E
 BRAUER Clamp Cross Reference BRAUER# Equal/Similar DESTACO # CARR-LANE # STANDARD.VERTICAL.CLAMPS V75/1B E 201-B CL-151-VTC 201-TB V75/2B E 201 CL-150-VTC 201-T CL-152-VTC V100/1B V100/1C V100/2B V100/2C
BRAUER Clamp Cross Reference BRAUER# Equal/Similar DESTACO # CARR-LANE # STANDARD.VERTICAL.CLAMPS V75/1B E 201-B CL-151-VTC 201-TB V75/2B E 201 CL-150-VTC 201-T CL-152-VTC V100/1B V100/1C V100/2B V100/2C
PROOF/ÉPREUVE ISO INTERNATIONAL STANDARD. Space environment (natural and artificial) Galactic cosmic ray model
 INTERNATIONAL STANDARD ISO 15390 First edition 2004-##-## Space environment (natural and artificial) Galactic cosmic ray model Environnement spatial (naturel et artificiel) Modèle de rayonnement cosmique
INTERNATIONAL STANDARD ISO 15390 First edition 2004-##-## Space environment (natural and artificial) Galactic cosmic ray model Environnement spatial (naturel et artificiel) Modèle de rayonnement cosmique
End-to-end Performance Diagnosis
 E-- Pf Dg [IEEE 802.16 M P Tp (Rv. 0)] D Nb: IEEE 802.16-12-0370-00-S D Sb: 2012-05-15 S: P Kpy V: - Gg I f Tgy E-: p AT.g. A GA * R: S f p b by IEEE 802.16 Mgy Sy Gp
E-- Pf Dg [IEEE 802.16 M P Tp (Rv. 0)] D Nb: IEEE 802.16-12-0370-00-S D Sb: 2012-05-15 S: P Kpy V: - Gg I f Tgy E-: p AT.g. A GA * R: S f p b by IEEE 802.16 Mgy Sy Gp
0# E % D 0 D - C AB
 5-70,- 393 %& 44 03& / / %0& / / 405 4 90//7-90/8/3 ) /7 0% 0 - @AB 5? 07 5 >0< 98 % =< < ; 98 07 &? % B % - G %0A 0@ % F0 % 08 403 08 M3 @ K0 J? F0 4< - G @ I 0 QR 4 @ 8 >5 5 % 08 OF0 80P 0O 0N 0@ 80SP
5-70,- 393 %& 44 03& / / %0& / / 405 4 90//7-90/8/3 ) /7 0% 0 - @AB 5? 07 5 >0< 98 % =< < ; 98 07 &? % B % - G %0A 0@ % F0 % 08 403 08 M3 @ K0 J? F0 4< - G @ I 0 QR 4 @ 8 >5 5 % 08 OF0 80P 0O 0N 0@ 80SP
CMSC 313 Lecture 17 Postulates & Theorems of Boolean Algebra Semiconductors CMOS Logic Gates
 CMSC 313 Lecture 17 Postulates & Theorems of Boolean Algebra Semiconductors CMOS Logic Gates UMBC, CMSC313, Richard Chang Last Time Overview of second half of this course Logic gates &
CMSC 313 Lecture 17 Postulates & Theorems of Boolean Algebra Semiconductors CMOS Logic Gates UMBC, CMSC313, Richard Chang Last Time Overview of second half of this course Logic gates &
ORBITAL DIAGRAM - A graphical representation of the quantum number "map" of electrons around an atom.
 178 (MAGNETIC) SPIN QUANTUM NUMBER: "spin down" or "spin up" - An ORBITAL (region with fixed "n", "l" and "ml" values) can hold TWO electrons. ORBITAL DIAGRAM - A graphical representation of the quantum
178 (MAGNETIC) SPIN QUANTUM NUMBER: "spin down" or "spin up" - An ORBITAL (region with fixed "n", "l" and "ml" values) can hold TWO electrons. ORBITAL DIAGRAM - A graphical representation of the quantum
FINAL EXAM April 26, 2004
 CM 1045 (11:15 am Lecture) Dr. Light FINAL EXAM April 26, 2004 Name (please print) Check your recitation section: Sec. 21 5:30-6:20 pm (Popovic) Sec. 24 3:30-4:20 pm (Giunta) Sec. 22 6:30-7:20 pm (Popovic)
CM 1045 (11:15 am Lecture) Dr. Light FINAL EXAM April 26, 2004 Name (please print) Check your recitation section: Sec. 21 5:30-6:20 pm (Popovic) Sec. 24 3:30-4:20 pm (Giunta) Sec. 22 6:30-7:20 pm (Popovic)
POLYTECHNIC OF NAMIBIA
 POLYTECHNIC OF NAMIBIA DEPARTMENT OF HEALTH SCIENCES BACHELOR OF ENVIRONMENTAL HEALTH SCIENCES HEALTH SCIENCE CHEMISTRY (HSC 511S) NQF level 5 SECOND OPPORTUNITY EXAMINATION November 2014 TIME: MARKS:
POLYTECHNIC OF NAMIBIA DEPARTMENT OF HEALTH SCIENCES BACHELOR OF ENVIRONMENTAL HEALTH SCIENCES HEALTH SCIENCE CHEMISTRY (HSC 511S) NQF level 5 SECOND OPPORTUNITY EXAMINATION November 2014 TIME: MARKS:
GENERAL PRINCIPLES OF CHEMISTRY - CHEM110 TEST 3
 School of Chemistry, University of KwaZulu-Natal, Westville Campus GENERAL PRINCIPLES CHEMISTRY - CHEM110 TEST 3 Date: WEDNESDAY, 5 May 2010 Total marks: 25 Time: 18h00 18h45 Examiners: Mrs H Govender
School of Chemistry, University of KwaZulu-Natal, Westville Campus GENERAL PRINCIPLES CHEMISTRY - CHEM110 TEST 3 Date: WEDNESDAY, 5 May 2010 Total marks: 25 Time: 18h00 18h45 Examiners: Mrs H Govender
Made the FIRST periodic table
 Made the FIRST periodic table 1869 Mendeleev organized the periodic table based on the similar properties and relativities of certain elements Later, Henri Moseley organized the elements by increasing
Made the FIRST periodic table 1869 Mendeleev organized the periodic table based on the similar properties and relativities of certain elements Later, Henri Moseley organized the elements by increasing
02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr
 Chemistry 05 B First Letter of PLEASE PRINT YOUR NAME IN BLOCK LETTERS Exam last Name Name: 02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr Lab TA s Name: Question Points Score Grader 2 2 9 3 9 4 2
Chemistry 05 B First Letter of PLEASE PRINT YOUR NAME IN BLOCK LETTERS Exam last Name Name: 02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr Lab TA s Name: Question Points Score Grader 2 2 9 3 9 4 2
CHEM 108 (Spring-2008) Exam. 3 (105 pts)
 CHEM 08 (Spring-008) Exam. (05 pts) Name: --------------------------------------------------------------------------, CLID # -------------------------------- LAST NAME, First (Circle the alphabet segment
CHEM 08 (Spring-008) Exam. (05 pts) Name: --------------------------------------------------------------------------, CLID # -------------------------------- LAST NAME, First (Circle the alphabet segment
35H MPa Hydraulic Cylinder 3.5 MPa Hydraulic Cylinder 35H-3
 - - - - ff ff - - - - - - B B BB f f f f f f f 6 96 f f f f f f f 6 f LF LZ f 6 MM f 9 P D RR DD M6 M6 M6 M. M. M. M. M. SL. E 6 6 9 ZB Z EE RC/ RC/ RC/ RC/ RC/ ZM 6 F FP 6 K KK M. M. M. M. M M M M f f
- - - - ff ff - - - - - - B B BB f f f f f f f 6 96 f f f f f f f 6 f LF LZ f 6 MM f 9 P D RR DD M6 M6 M6 M. M. M. M. M. SL. E 6 6 9 ZB Z EE RC/ RC/ RC/ RC/ RC/ ZM 6 F FP 6 K KK M. M. M. M. M M M M f f
CHEM 251 (Fall-2003) Final Exam (100 pts)
 CEM 251 (Fall-2003) Final Exam (100 pts) Name: -------------------------------------------------------------------------------, SSN -------------------------------- LAST NAME, First (Circle the alphabet
CEM 251 (Fall-2003) Final Exam (100 pts) Name: -------------------------------------------------------------------------------, SSN -------------------------------- LAST NAME, First (Circle the alphabet
Appendix B Impact Table - Proposed Routes
 PSC REF#:360505 Impact Table - Proposed s Table B-1 Table B-2 Table B-3 Table B-4 Table B-5 Impact Summaries-Hill Valley Substation Impact Summaries-Mississippi River Routing Area Impact Summaries- Western
PSC REF#:360505 Impact Table - Proposed s Table B-1 Table B-2 Table B-3 Table B-4 Table B-5 Impact Summaries-Hill Valley Substation Impact Summaries-Mississippi River Routing Area Impact Summaries- Western
SYSTEM OF CIRCLES If d is the distance between the centers of two intersecting circles with radii r 1, r 2 and θ is the
 SYSTEM OF CIRCLES Theorem: If d is the distance between the centers of two intersecting circles with radii r 1, r 2 and θ is the 2 2 2 d r1 r2 angle between the circles then cos θ =. 2r r 1 2 Proof: Let
SYSTEM OF CIRCLES Theorem: If d is the distance between the centers of two intersecting circles with radii r 1, r 2 and θ is the 2 2 2 d r1 r2 angle between the circles then cos θ =. 2r r 1 2 Proof: Let
I M P O R T A N T S A F E T Y I N S T R U C T I O N S W h e n u s i n g t h i s e l e c t r o n i c d e v i c e, b a s i c p r e c a u t i o n s s h o
 I M P O R T A N T S A F E T Y I N S T R U C T I O N S W h e n u s i n g t h i s e l e c t r o n i c d e v i c e, b a s i c p r e c a u t i o n s s h o u l d a l w a y s b e t a k e n, i n c l u d f o l
I M P O R T A N T S A F E T Y I N S T R U C T I O N S W h e n u s i n g t h i s e l e c t r o n i c d e v i c e, b a s i c p r e c a u t i o n s s h o u l d a l w a y s b e t a k e n, i n c l u d f o l
K. 27 Co. 28 Ni. 29 Cu Rb. 46 Pd. 45 Rh. 47 Ag Cs Ir. 78 Pt.
 1 IA 1 H Hydrogen 1.01 Atomic number Element symbol Element name Atomic mass VIIIA 1 H 1.01 IIA IIIA IVA VA VIA VIIA 2 He 4.00 Metalloids 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 O 16.00 9 F
1 IA 1 H Hydrogen 1.01 Atomic number Element symbol Element name Atomic mass VIIIA 1 H 1.01 IIA IIIA IVA VA VIA VIIA 2 He 4.00 Metalloids 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 O 16.00 9 F
Part 2. Multiple choice (use answer card). 90 pts. total. 3 pts. each.
 1 Exam I CHEM 1303.001 Name (print legibly) Seat no. On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Part 1. Nomenclature. 10 pts. total. 2 pts. each. Fill in
1 Exam I CHEM 1303.001 Name (print legibly) Seat no. On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Part 1. Nomenclature. 10 pts. total. 2 pts. each. Fill in
Chemistry 2 Exam Roane State Academic Festival. Name (print neatly) School
 Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
Radiometric Dating (tap anywhere)
 Radiometric Dating (tap anywhere) Protons Neutrons Electrons Elements on the periodic table are STABLE Elements can have radioactive versions of itself called ISOTOPES!! Page 1 in your ESRT has your list!
Radiometric Dating (tap anywhere) Protons Neutrons Electrons Elements on the periodic table are STABLE Elements can have radioactive versions of itself called ISOTOPES!! Page 1 in your ESRT has your list!
If anything confuses you or is not clear, raise your hand and ask!
 CHM 1045 Dr. Light s Section December 10, 2002 FINAL EXAM Name (please print) Recitation Section Meeting Time This exam consists of six pages. Make sure you have one of each. Print your name at the top
CHM 1045 Dr. Light s Section December 10, 2002 FINAL EXAM Name (please print) Recitation Section Meeting Time This exam consists of six pages. Make sure you have one of each. Print your name at the top
HANDOUT SET GENERAL CHEMISTRY II
 HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
The Periodic Table. Periodic Properties. Can you explain this graph? Valence Electrons. Valence Electrons. Paramagnetism
 Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
1 Introduction JARRETT WALKER + ASSOCIATES. SEPTA Philadelphia Bus Network Choices Report
 Ici AETT AE + AIATE Ppi N ic p i ic p? Ti p i ii pfc f b ii i f Ppi, i c f i i ic p,. T f i p i pi bc f fc ii c pi ici ff i b. T p i c ic p bc i cic ci. If f i ic iip, i f i ppc c b, i i p., iip i f i,
Ici AETT AE + AIATE Ppi N ic p i ic p? Ti p i ii pfc f b ii i f Ppi, i c f i i ic p,. T f i p i pi bc f fc ii c pi ici ff i b. T p i c ic p bc i cic ci. If f i ic iip, i f i ppc c b, i i p., iip i f i,
Overload Relays. SIRIUS 3RU1 Thermal Overload Relays. 3RU11 for standard applications. 5/46 Siemens LV 1 AO 2011
 SIRIUS 3RU1 Thermal Overview 1 2 7 3 4 6 "Increased safety" type of EEx e according to ATEX directive 94/9/EC The 3RU11 thermal overload relays are suitable for the overload of explosion-proof motors with
SIRIUS 3RU1 Thermal Overview 1 2 7 3 4 6 "Increased safety" type of EEx e according to ATEX directive 94/9/EC The 3RU11 thermal overload relays are suitable for the overload of explosion-proof motors with
Atoms and the Periodic Table
 Atoms and the Periodic Table Parts of the Atom Proton Found in the nucleus Number of protons defines the element Charge +1, mass 1 Parts of the Atom Neutron Found in the nucleus Stabilizes the nucleus
Atoms and the Periodic Table Parts of the Atom Proton Found in the nucleus Number of protons defines the element Charge +1, mass 1 Parts of the Atom Neutron Found in the nucleus Stabilizes the nucleus
5 questions, 3 points each, 15 points total possible. 26 Fe Cu Ni Co Pd Ag Ru 101.
 Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
A L A BA M A L A W R E V IE W
 A L A BA M A L A W R E V IE W Volume 52 Fall 2000 Number 1 B E F O R E D I S A B I L I T Y C I V I L R I G HT S : C I V I L W A R P E N S I O N S A N D TH E P O L I T I C S O F D I S A B I L I T Y I N
A L A BA M A L A W R E V IE W Volume 52 Fall 2000 Number 1 B E F O R E D I S A B I L I T Y C I V I L R I G HT S : C I V I L W A R P E N S I O N S A N D TH E P O L I T I C S O F D I S A B I L I T Y I N
CHEM 172 EXAMINATION 2. February 12, Dr. Kimberly M. Broekemeier NAME: l = 2r l = 8 1/2 r l = (4/3 1/2 )r. h = 6.
 EM 17 EXAMINATION February 1, 009 Dr. Kimberly M. Broekemeier NAME: P 1 1 P1 R T T1 ln = - ( - ) l = r l = 8 1/ r l = (4/3 1/ )r onstants: c = 3.00 X 10 8 m/s h = 6.63 X 10-34 J x s R = 0.0806 L x atm/mol
EM 17 EXAMINATION February 1, 009 Dr. Kimberly M. Broekemeier NAME: P 1 1 P1 R T T1 ln = - ( - ) l = r l = 8 1/ r l = (4/3 1/ )r onstants: c = 3.00 X 10 8 m/s h = 6.63 X 10-34 J x s R = 0.0806 L x atm/mol
Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
8. Relax and do well.
 CHEM 15 Exam II John II. Gelder March 4, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table, a solubility
CHEM 15 Exam II John II. Gelder March 4, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table, a solubility
- A polar molecule has an uneven distribution of electron density, making it have ends (poles) that are slightly charged.
 14 POLARITY and shape: - A polar molecule has an uneven distribution of electron density, making it have ends (poles) that are slightly charged. POLARITY influences several easily observable properties.
14 POLARITY and shape: - A polar molecule has an uneven distribution of electron density, making it have ends (poles) that are slightly charged. POLARITY influences several easily observable properties.
Guide to the Extended Step-Pyramid Periodic Table
 Guide to the Extended Step-Pyramid Periodic Table William B. Jensen Department of Chemistry University of Cincinnati Cincinnati, OH 452201-0172 The extended step-pyramid table recognizes that elements
Guide to the Extended Step-Pyramid Periodic Table William B. Jensen Department of Chemistry University of Cincinnati Cincinnati, OH 452201-0172 The extended step-pyramid table recognizes that elements
Solutions and Ions. Pure Substances
 Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
Faculty of Natural and Agricultural Sciences Chemistry Department. Semester Test 1. Analytical Chemistry CMY 283. Time: 120 min Marks: 100 Pages: 6
 Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 120 min
Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 120 min
Page Input. Shield EURO 3-STA GND. 470pF Z1B. (+20 db) 10K % C38 LINK LEVEL. To Sheet 3 GND. 120Hz. Page Level R11 15K. 7kHz GND .
 Ducker Depth 0 0 db R K R0 K SSM S Ducker Depth 00 To Sheet ZA C 0. R.K R 0K DUCK To Sheet Page Input S PT Phantom Power J EURO STA R 00 L T L T C 0/ C 0pF R0 0 R 0 C 00/0 SA Mic/Line PT C 00/0 R K R.K
Ducker Depth 0 0 db R K R0 K SSM S Ducker Depth 00 To Sheet ZA C 0. R.K R 0K DUCK To Sheet Page Input S PT Phantom Power J EURO STA R 00 L T L T C 0/ C 0pF R0 0 R 0 C 00/0 SA Mic/Line PT C 00/0 R K R.K
CP 52 Page In & Zone Sensitivity
 db SSM S R.0K R0.00K To Sheet ZA ON OFF C 0. R.K R 0.0K DUCK To Sheet Page Input Shield R 00 S PP Phantom Power 0/V L T C 0PF C L 0PF T EURO POS R0 0 R 0 R.0K 00/0V R Mic/Line.K R.K R.0K R 00 R. R0A KRD
db SSM S R.0K R0.00K To Sheet ZA ON OFF C 0. R.K R 0.0K DUCK To Sheet Page Input Shield R 00 S PP Phantom Power 0/V L T C 0PF C L 0PF T EURO POS R0 0 R 0 R.0K 00/0V R Mic/Line.K R.K R.0K R 00 R. R0A KRD
-"l" also contributes ENERGY. Higher values for "l" mean the electron has higher energy.
 175 - Giving the four parameters will uniquely identify an electron around an atom. No two electrons in the same atom can share all four. These parameters are called QUANTUM NUMBERS. PRINCIPAL QUANTUM
175 - Giving the four parameters will uniquely identify an electron around an atom. No two electrons in the same atom can share all four. These parameters are called QUANTUM NUMBERS. PRINCIPAL QUANTUM
4 4 N v b r t, 20 xpr n f th ll f th p p l t n p pr d. H ndr d nd th nd f t v L th n n f th pr v n f V ln, r dn nd l r thr n nt pr n, h r th ff r d nd
 n r t d n 20 20 0 : 0 T P bl D n, l d t z d http:.h th tr t. r pd l 4 4 N v b r t, 20 xpr n f th ll f th p p l t n p pr d. H ndr d nd th nd f t v L th n n f th pr v n f V ln, r dn nd l r thr n nt pr n,
n r t d n 20 20 0 : 0 T P bl D n, l d t z d http:.h th tr t. r pd l 4 4 N v b r t, 20 xpr n f th ll f th p p l t n p pr d. H ndr d nd th nd f t v L th n n f th pr v n f V ln, r dn nd l r thr n nt pr n,
Element Cube Project (x2)
 Element Cube Project (x2) Background: As a class, we will construct a three dimensional periodic table by each student selecting two elements in which you will need to create an element cube. Helpful Links
Element Cube Project (x2) Background: As a class, we will construct a three dimensional periodic table by each student selecting two elements in which you will need to create an element cube. Helpful Links
Whitney Grummon. She kick started a fire in my soul Teaching me a tool to cleanse my mind That ll last a life time. That s how I will remember
 W Gmm S kk f m T m m m T f m T I mmb N m p f p f f G L A f b k, b k v M k b p:, bb m, m f m, v. A b m, f mm mm f v b G p. S m m z pp pv pm f, k mk, f v M. I m, I m, fm k p x. S f 45 m m CMS, I p mf,. B
W Gmm S kk f m T m m m T f m T I mmb N m p f p f f G L A f b k, b k v M k b p:, bb m, m f m, v. A b m, f mm mm f v b G p. S m m z pp pv pm f, k mk, f v M. I m, I m, fm k p x. S f 45 m m CMS, I p mf,. B
 lectures accompanying the book: Solid State Physics: An Introduction, by Philip ofmann (2nd edition 2015, ISBN-10: 3527412824, ISBN-13: 978-3527412822, Wiley-VC Berlin. www.philiphofmann.net 1 Bonds between
lectures accompanying the book: Solid State Physics: An Introduction, by Philip ofmann (2nd edition 2015, ISBN-10: 3527412824, ISBN-13: 978-3527412822, Wiley-VC Berlin. www.philiphofmann.net 1 Bonds between
Chemistry 431 Practice Final Exam Fall Hours
 Chemistry 431 Practice Final Exam Fall 2018 3 Hours R =8.3144 J mol 1 K 1 R=.0821 L atm mol 1 K 1 R=.08314 L bar mol 1 K 1 k=1.381 10 23 J molecule 1 K 1 h=6.626 10 34 Js N A = 6.022 10 23 molecules mol
Chemistry 431 Practice Final Exam Fall 2018 3 Hours R =8.3144 J mol 1 K 1 R=.0821 L atm mol 1 K 1 R=.08314 L bar mol 1 K 1 k=1.381 10 23 J molecule 1 K 1 h=6.626 10 34 Js N A = 6.022 10 23 molecules mol
9/20/2017. Elements are Pure Substances that cannot be broken down into simpler substances by chemical change (contain Only One Type of Atom)
 CAPTER 6: TE PERIODIC TABLE Elements are Pure Substances that cannot be broken down into simpler substances by chemical change (contain Only One Type of Atom) The Periodic Table (Mendeleev) In 1872, Dmitri
CAPTER 6: TE PERIODIC TABLE Elements are Pure Substances that cannot be broken down into simpler substances by chemical change (contain Only One Type of Atom) The Periodic Table (Mendeleev) In 1872, Dmitri
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
VIIIA H PREDICTING CHARGE
 58 IA PREDICTING CHARGE VIIIA H IIA IIIA IVA VA VIA VIIA You can reliably determine the charge using our method for Groups IA, IIA, IIIB, Aluminum, and the Group VA, VIA, and VIIA NONMETALS Li Be B C N
58 IA PREDICTING CHARGE VIIIA H IIA IIIA IVA VA VIA VIIA You can reliably determine the charge using our method for Groups IA, IIA, IIIB, Aluminum, and the Group VA, VIA, and VIIA NONMETALS Li Be B C N
Physical Chemistry I CHEM 4641 Final Exam 13 questions, 30 points
 Physical Chemistry I CHEM 4641 Final Exam 13 questions, 30 points Name: KEY Gas constant: R = 8.314 J mol -1 K -1 = 0.008314 kj mol -1 K -1. Boltzmann constant k = 1.381 10-23 J/K = 0.6950 cm -1 /K h =
Physical Chemistry I CHEM 4641 Final Exam 13 questions, 30 points Name: KEY Gas constant: R = 8.314 J mol -1 K -1 = 0.008314 kj mol -1 K -1. Boltzmann constant k = 1.381 10-23 J/K = 0.6950 cm -1 /K h =
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Practice Final Exam CH 201
 Practice Final Exam CH 201 Name: (please print) Student I D : Instructions - Read Carefully 1. Please show your work, and put your final answers in the spaces provided. 2. Point values for each question
Practice Final Exam CH 201 Name: (please print) Student I D : Instructions - Read Carefully 1. Please show your work, and put your final answers in the spaces provided. 2. Point values for each question
26th Feb To 16th Apr 2017
 ST EUPHRASIA SYRO-MALABAR PARISH ADELAIDE NORTH PARISH TEAM Rv F Bj J Cck [P P] M J & J M 26 Fb T 16 A 2017 B V1 I 1 S f L D 16 A 2017 W c c b f x f L f v, f g c g g g f [Kkk] j f f J P K MASS TIMES [Cc
ST EUPHRASIA SYRO-MALABAR PARISH ADELAIDE NORTH PARISH TEAM Rv F Bj J Cck [P P] M J & J M 26 Fb T 16 A 2017 B V1 I 1 S f L D 16 A 2017 W c c b f x f L f v, f g c g g g f [Kkk] j f f J P K MASS TIMES [Cc
ARE YOU PREPARED FOR A TSUNAMI?
 Eq g g? g! b, gb bg K f g pc T c fc E YOU EE FO TUNI? Y pp c b ffc f Kāp I b f p : g g f p f gg b pp f gc g f g f g p p Kāp c c - Y f j 8000 pp Kāp c z. If q pp c Kāp, c. I p p g b f f g g f c qc c f g.
Eq g g? g! b, gb bg K f g pc T c fc E YOU EE FO TUNI? Y pp c b ffc f Kāp I b f p : g g f p f gg b pp f gc g f g f g p p Kāp c c - Y f j 8000 pp Kāp c z. If q pp c Kāp, c. I p p g b f f g g f c qc c f g.
Circle the letters only. NO ANSWERS in the Columns!
 Chemistry 1304.001 Name (please print) Exam 5 (100 points) April 18, 2018 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Chemistry 1304.001 Name (please print) Exam 5 (100 points) April 18, 2018 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
CHEM 10123/10125, Exam 2
 CHEM 10123/10125, Exam 2 March 7, 2012 (50 minutes) Name (please print) Please box your answers, and remember that significant figures, phases (for chemical equations), and units do count! 1. (13 points)
CHEM 10123/10125, Exam 2 March 7, 2012 (50 minutes) Name (please print) Please box your answers, and remember that significant figures, phases (for chemical equations), and units do count! 1. (13 points)
A system of matrix equations and a linear matrix equation over arbitrary regular rings with identity
 Linear Algebra and its Applications 384 2004) 43 54 www.elsevier.com/locate/laa A system of matrix equations and a linear matrix equation over arbitrary regular rings with identity Qing-Wen Wang Department
Linear Algebra and its Applications 384 2004) 43 54 www.elsevier.com/locate/laa A system of matrix equations and a linear matrix equation over arbitrary regular rings with identity Qing-Wen Wang Department
o C *$ go ! b», S AT? g (i * ^ fc fa fa U - S 8 += C fl o.2h 2 fl 'fl O ' 0> fl l-h cvo *, &! 5 a o3 a; O g 02 QJ 01 fls g! r«'-fl O fl s- ccco
 > p >>>> ft^. 2 Tble f Generl rdnes. t^-t - +«0 -P k*ph? -- i t t i S i-h l -H i-h -d. *- e Stf H2 t s - ^ d - 'Ct? "fi p= + V t r & ^ C d Si d n. M. s - W ^ m» H ft ^.2. S'Sll-pl e Cl h /~v S s, -P s'l
> p >>>> ft^. 2 Tble f Generl rdnes. t^-t - +«0 -P k*ph? -- i t t i S i-h l -H i-h -d. *- e Stf H2 t s - ^ d - 'Ct? "fi p= + V t r & ^ C d Si d n. M. s - W ^ m» H ft ^.2. S'Sll-pl e Cl h /~v S s, -P s'l
ers The Extraordinary Boogie and Swing Festival MUNICH GERMANY 9
 W!!! C, E p, C J 95 RT H v D L T BI 10 N P f B 70 5 z V D AY b T Ex B Fv 28.02. - 04.03. MUNICH 1 GERMANY 9 R O C KT H AT W I N G. C O M WING ENT F O LD DLY PRE R WO PROU 14 5 DAY HT G I N UIC M E LIV
W!!! C, E p, C J 95 RT H v D L T BI 10 N P f B 70 5 z V D AY b T Ex B Fv 28.02. - 04.03. MUNICH 1 GERMANY 9 R O C KT H AT W I N G. C O M WING ENT F O LD DLY PRE R WO PROU 14 5 DAY HT G I N UIC M E LIV
8. Relax and do well.
 CHEM 1014 Exam I John I. Gelder September 16, 1999 Name TA's Name Lab Section Please sign your name below to give permission to post your course scores on homework, laboratories and exams. If you do not
CHEM 1014 Exam I John I. Gelder September 16, 1999 Name TA's Name Lab Section Please sign your name below to give permission to post your course scores on homework, laboratories and exams. If you do not
Part Two. Southern Florida s Early Native People
 19 www.m.. 2002 F Mm N H, G, F P Tw S F E N P 20 2002 F Mm N H, G, F www.m.. P b P O b gg g c. I c m c Iq Bx Mm. S F E N P T m S F E N P Iq Bx, Mm wb www.m... W w F w? T g m w c w F 500 BCE c C ( m g -cc),
19 www.m.. 2002 F Mm N H, G, F P Tw S F E N P 20 2002 F Mm N H, G, F www.m.. P b P O b gg g c. I c m c Iq Bx Mm. S F E N P T m S F E N P Iq Bx, Mm wb www.m... W w F w? T g m w c w F 500 BCE c C ( m g -cc),
WRITING AN IONIC FORMULA
 WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example: If
WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example: If
8. Relax and do well.
 CHEM 1225 Exam III John III. Gelder April 8, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table and
CHEM 1225 Exam III John III. Gelder April 8, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table and
8. Relax and do well.
 CHEM 1314.03 Exam I John I. Gelder September 25, 1997 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
CHEM 1314.03 Exam I John I. Gelder September 25, 1997 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
Example: If a simple ionic compound is made of these two ions, what is its formula? In the final formula, don't write the charges on the ions!
 88 WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example:
88 WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example:
Future Self-Guides. E,.?, :0-..-.,0 Q., 5...q ',D5', 4,] 1-}., d-'.4.., _. ZoltAn Dbrnyei Introduction. u u rt 5,4) ,-,4, a. a aci,, u 4.
![Future Self-Guides. E,.?, :0-..-.,0 Q., 5...q ',D5', 4,] 1-}., d-'.4.., _. ZoltAn Dbrnyei Introduction. u u rt 5,4) ,-,4, a. a aci,, u 4. Future Self-Guides. E,.?, :0-..-.,0 Q., 5...q ',D5', 4,] 1-}., d-'.4.., _. ZoltAn Dbrnyei Introduction. u u rt 5,4) ,-,4, a. a aci,, u 4.](/thumbs/92/109036622.jpg) te SelfGi ZltAn Dbnyei Intdtin ; ) Q) 4 t? ) t _ 4 73 y S _ E _ p p 4 t t 4) 1_ ::_ J 1 `i () L VI O I4 " " 1 D 4 L e Q) 1 k) QJ 7 j ZS _Le t 1 ej!2 i1 L 77 7 G (4) 4 6 t (1 ;7 bb F) t f; n (i M Q) 7S
te SelfGi ZltAn Dbnyei Intdtin ; ) Q) 4 t? ) t _ 4 73 y S _ E _ p p 4 t t 4) 1_ ::_ J 1 `i () L VI O I4 " " 1 D 4 L e Q) 1 k) QJ 7 j ZS _Le t 1 ej!2 i1 L 77 7 G (4) 4 6 t (1 ;7 bb F) t f; n (i M Q) 7S
TRU Chemistry Contest Chemistry 12 May 21, 2003 Time: 90 minutes
 TRU Chemistry Contest Chemistry 12 May 21, 2003 Time: 90 minutes Last Name First name School Teacher Please follow the instructions below. We will send your teacher a report on your performance. Top performers
TRU Chemistry Contest Chemistry 12 May 21, 2003 Time: 90 minutes Last Name First name School Teacher Please follow the instructions below. We will send your teacher a report on your performance. Top performers
3.2. Built Environment: Land Use and Transportation
 Affc, gfc Ipc, g p, g, pc., f A c c q q f qc b c f pjc p., ffc b c f A cp A 2, bc b f c f p fc. 3.2. : L U Tp 3.2.1. L U T c p xg UGA Kp C. I p cg F I f A pc f cg. T fc f pc c f, cg c, cpb. I cb pc c f
Affc, gfc Ipc, g p, g, pc., f A c c q q f qc b c f pjc p., ffc b c f A cp A 2, bc b f c f p fc. 3.2. : L U Tp 3.2.1. L U T c p xg UGA Kp C. I p cg F I f A pc f cg. T fc f pc c f, cg c, cpb. I cb pc c f
CHEM 107 (Spring-2004) Exam 2 (100 pts)
 CHEM 107 (Spring-2004) Exam 2 (100 pts) Name: ------------------------------------------------------------------------, SSN -------------------------------- LAST NAME, First (Circle the alphabet segment
CHEM 107 (Spring-2004) Exam 2 (100 pts) Name: ------------------------------------------------------------------------, SSN -------------------------------- LAST NAME, First (Circle the alphabet segment
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
PERIODIC TABLE OF THE ELEMENTS
 Useful Constants and equations: K = o C + 273 Avogadro's number = 6.022 x 10 23 d = density = mass/volume R H = 2.178 x 10-18 J c = E = h = hc/ h = 6.626 x 10-34 J s c = 2.998 x 10 8 m/s E n = -R H Z 2
Useful Constants and equations: K = o C + 273 Avogadro's number = 6.022 x 10 23 d = density = mass/volume R H = 2.178 x 10-18 J c = E = h = hc/ h = 6.626 x 10-34 J s c = 2.998 x 10 8 m/s E n = -R H Z 2
CHEMICAL COMPOUNDS MOLECULAR COMPOUNDS
 48 CHEMICAL COMPOUNDS - Dalton's theory does not mention this, but there is more than one way for atoms to come together to make chemical compounds! - There are TWO common kinds of chemical compound, classified
48 CHEMICAL COMPOUNDS - Dalton's theory does not mention this, but there is more than one way for atoms to come together to make chemical compounds! - There are TWO common kinds of chemical compound, classified
VIIIA H PREDICTING CHARGE
 58 IA PREDICTING CHARGE VIIIA H IIA IIIA IVA VA VIA VIIA You can reliably determine the charge using our method for Groups IA, IIA, IIIB, Aluminum, and the Group VA, VIA, and VIIA NONMETALS Li Be B C N
58 IA PREDICTING CHARGE VIIIA H IIA IIIA IVA VA VIA VIIA You can reliably determine the charge using our method for Groups IA, IIA, IIIB, Aluminum, and the Group VA, VIA, and VIIA NONMETALS Li Be B C N
8. Relax and do well.
 CHEM 1515 Exam II John II. Gelder October 14, 1993 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last two pages include a periodic table, a
CHEM 1515 Exam II John II. Gelder October 14, 1993 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last two pages include a periodic table, a
Faculty of Natural and Agricultural Sciences Chemistry Department. Semester Test 1 MEMO. Analytical Chemistry CMY 283
 Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 MEMO Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 90
Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 MEMO Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 90
CHEM 107 (Spring-2005) Exam 3 (100 pts)
 CHEM 107 (Spring-2005) Exam 3 (100 pts) Name: ------------------------------------------------------------------------, Clid # ------------------------------ LAST NAME, First (Circle the alphabet segment
CHEM 107 (Spring-2005) Exam 3 (100 pts) Name: ------------------------------------------------------------------------, Clid # ------------------------------ LAST NAME, First (Circle the alphabet segment
Coolsicles. Chicken Tenders. Hearty Beef Stew
 Cck T M B Cck P Cc Hy B Sw D Ec, Ec pp y y cc y w C F K TM c p b y by N Fz R F Ac (NFRA) pp w cc pc Y M Ip (YMI). T p y cc w $50 pp y c ( ). C F K TM p c b cc, p c, b pyc cvy. I w y wk b pv w v w pc w
Cck T M B Cck P Cc Hy B Sw D Ec, Ec pp y y cc y w C F K TM c p b y by N Fz R F Ac (NFRA) pp w cc pc Y M Ip (YMI). T p y cc w $50 pp y c ( ). C F K TM p c b cc, p c, b pyc cvy. I w y wk b pv w v w pc w
Reporting Category 1: Matter and Energy
 Name: Science Teacher: Reporting Category 1: Matter and Energy Atoms Fill in the missing information to summarize what you know about atomic structure. Name of Subatomic Particle Location within the Atom
Name: Science Teacher: Reporting Category 1: Matter and Energy Atoms Fill in the missing information to summarize what you know about atomic structure. Name of Subatomic Particle Location within the Atom
single-layer transition metal dichalcogenides MC2
 single-layer transition metal dichalcogenides MC2 Period 1 1 H 18 He 2 Group 1 2 Li Be Group 13 14 15 16 17 18 B C N O F Ne 3 4 Na K Mg Ca Group 3 4 5 6 7 8 9 10 11 12 Sc Ti V Cr Mn Fe Co Ni Cu Zn Al Ga
single-layer transition metal dichalcogenides MC2 Period 1 1 H 18 He 2 Group 1 2 Li Be Group 13 14 15 16 17 18 B C N O F Ne 3 4 Na K Mg Ca Group 3 4 5 6 7 8 9 10 11 12 Sc Ti V Cr Mn Fe Co Ni Cu Zn Al Ga
Errata of CMOS Analog Circuit Design 2 nd Edition By Phillip E. Allen and Douglas R. Holberg
 Errata 2 nd Ed. (5/22/2) Page Errata of CMOS Analog Circuit Design 2 nd Edition By Phillip E. Allen and Douglas R. Holberg Page Errata 82 Line 4 after figure 3.2-3, CISW CJSW 88 Line between Eqs. (3.3-2)
Errata 2 nd Ed. (5/22/2) Page Errata of CMOS Analog Circuit Design 2 nd Edition By Phillip E. Allen and Douglas R. Holberg Page Errata 82 Line 4 after figure 3.2-3, CISW CJSW 88 Line between Eqs. (3.3-2)
8. Relax and do well.
 CHEM 1314 3;30 pm Theory Exam III John III. Gelder November 13, 2002 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last page include a periodic
CHEM 1314 3;30 pm Theory Exam III John III. Gelder November 13, 2002 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last page include a periodic
Mathematical Logics. 12. Soundness and Completeness of tableaux reasoning in first order logic. Luciano Serafini
 12. Soundness and Completeness of tableaux reasoning in first order logic Fondazione Bruno Kessler, Trento, Italy November 14, 2013 Example of tableaux Example Consider the following formulas: (a) xyz(p(x,
12. Soundness and Completeness of tableaux reasoning in first order logic Fondazione Bruno Kessler, Trento, Italy November 14, 2013 Example of tableaux Example Consider the following formulas: (a) xyz(p(x,
