Setting The motor that rotates the sample about an axis normal to the diffraction plane is called (or ).
|
|
|
- Wesley Robbins
- 6 years ago
- Views:
Transcription
1 X-Ray Diffraction X-ray diffraction geometry A simple X-ray diffraction (XRD) experiment might be set up as shown below. We need a parallel X-ray source, which is usually an X-ray tube in a fixed position with optics to both collimate the beam and filter out only a narrow range of X-ray wavelengths. The X-ray beam diffracts off a sample, and the diffracted intensity is measured with a detector on the end of an arm that can rotate about the sample. The plane of diffraction contains the incident (wavevector k k) and diffracted (wavevector k ) X-ray beams. We must be able to rotate the sample so that the RLV is interest is in the diffraction plane. Often, we want to examine the diffraction from planes parallel to the substrate, i.e., having their g vectors oriented along the substrate normal. In that case, as we scan through some range of with the detector, we must be simultaneously changing the sample orientation w.r.t. the incident beam to maintain k k g. It is certainly possible to rotate the source, instead of the sample, but the source has high-voltage cables and water lines, whereas the sample is often small, so a fixed source is more common. Setting The motor that rotates the sample about an axis normal to the diffraction plane is called (or ). It has an especially important role for films. We usually mount the substrate so that its plane is parallel to the beam axis and normal to the diffraction plane when 0. Then, if want to scan along the normal to the substrate plane, we set
2 In this case, and are basically synonymous. X-ray diffraction pattern from nanoparticles Powder diffraction is a way to analyze a material by grinding it up into many small particles that are randomly oriented. But we may refer to the diffraction pattern from an collection of nanoparticles, or even a polycrystalline material, as a powder pattern. Powder patterns are simple to analyze, because there is no dependence on specimen orientation; every orientation is represented in the data. So standard patterns are recorded for comparison to any arbitrary test sample. An example of a powder pattern from PbTe nanoparticles is shown below. There are a few things to notice. The most important is the positions of the peaks. We can identify their Miller indices once we have found the correct standard (vertical lines) for comparison. But some attention should be given to the relative peak heights. It is very uncommon to be able to quantitatively explain the absolute peak heights, but the relative heights should be a fairly close match. This is especially true for real powders, when the particle size is larger than a few microns. When comparing nanoparticles to a powder standard ( as in the example above) we should allow some differences due to the particular particle shapes. Another feature we should notice above is the peak widths. This sample contained very small particles, only a few nm in diameter, which causes a very broad peaks, more than - in. For a large-grained powder, the peaks may be quite narrow. In that case, the peak widths are mainly determined by the XRD instrument, rather than the sample. Powder diffraction standards The most common database of powder diffraction standards is managed by the Joint Committee on powder Diffraction Standards (JCPDS), which is associated with the International Union of
3 3 Crystallography. Each particular standard is saved in a powder diffraction file (PDF). These standard files are interchangeably called JCPDS files or PDFs. A portion of an example is shown below: The file is assigned a unique number, contains a name for the material, and some information about how the data was acquired. Then there is information about the physical properties, including the crystallographic space group and unit-cell dimensions. There are then some comments about the origins of the specimen. Last is a listing of the peak positions and/or d spacings, relative intensities (in %), and Miller indices. Two conventions for scattering vector In the discussion of electron diffraction, the lengths of reciprocal-space vectors were always the inverse of a corresponding distance, such as a wavelength or d-spacing. In XRD, it is more common to include a factor of when defining wavevector. The difference is whether the is written explicitly in the exponent of a complex exponential, or whether it is included in the wavenumber.
4 4 The point is simply that I may slip between the two conventions inadvertently. Please bear with me. Integrated intensity factors for powder There is more than one way to express the intensity of a diffraction peak. The number of counts per unit area per unit time is the absolute intensity. What we actually measure is the total number over some finite area of detector per unit time. The total number per unit time integrated over the full range of the peak in is called the integrated intensity. The integrated intensity in powder diffraction is proportional to a number of factors that we will list later. The peak can have various shapes. Assuming all of the peak shapes are the same (with just different parameters) we could express the integrated intensity as: I I int max θ where θ is the width in θ. Peak fitting One way to extract parameters such as maximum intensity I max and width θ is by least-squares curve fitting. This requires an expected curve shape the depends on a few parameters, which can be varied until the squared difference between the data and fit function at each point is minimized. In practice, each data point is usually weighted by its uncertainty. It is sometimes preferable to plot the data as a funciton of q, instead of. The centroid is q 0 and the width is q. Some common functions to use are listed below: Gaussian q q0 fq 0, q q exp ln q, //gaussian Lorentzian fq 0, q q, lortenzian q q0 q
5 5 Pearson7 f 0,, q / m q q0 q q q m m, pearson-7 We have arranged these in each case so that the width parameter q precisely the full width at half maximum (FWHM) of the peak, which is a standard metric. Note that the peaks are all normalized for maximum value of unity, but the the integrated intensities are not generally the same. The gaussian shape is used throughout statistics and physics. It is often called a normal distribution, and represents the probability distribution when a quantity has a definite mean. The lorentzian shape is unusual because it has no mean or standard deviation. Nonetheless, it does have a well-defined centroid (which corresponds to the median) and a FWHM. The pearson-7 shape is a purely mathematical construct that makes it easy to vary between gaussian and lorentzian shapes. Notice the extra parameter, m. If m=, we get the lorentzian shape. If m, we get the gaussian shape. There is no such correspondence for m, but we don't always care if the peak assume any particular, theoretical shape, especially if all we need to know is the peak position. Normalization In these forms, these peaks are normalized w.r.t. I max, not integrated intensity. But the areas under the curves are not all the same.
6 6 Another important lineshape The voigt function is defined as the convolution of a gaussian and a lorentzian centered at the same point q, i.e.: 0 Voigt Gaussian Lorentzian fq, qq fq 0 q q f q0 q q 0,, It turns out that, since the gaussian and lorentzian are normalized, the voigt will be, too. The centroid q 0 doesn't change, but, because of the lorentzian component, the voigt has no well-defined mean or standard deviation. There is no simple expression for the FWHM q as a functions of q and q, but we know q will be larger than both q and q. Sometime people use a pseudovoigt function, which is taken as a linear combination of a gaussian and a lorentzian, i.e.: Pseudovoigt Gaussian Lorentzian fq q q fq 0 q q f q0 q q 0,,, This is definitely easier to analyze, though the physical basis is shaky. We know the centroid remains q 0, and if q q q, the FWHM will remain q. But the pseudovoigt really only approximates a voigt for certain combinations of q, q, and. X-ray sources We know that ionization results in characteristic X-ray emission, while acceleration of electrons results in bremsstrahlung X rays. When we expose a target to an electron beam, both contributions will be present in the spectrum. There are two K lines and one K line. For X-rays sources, we usually want to use the intensity of the K lines.
7 7 The most common type of X-ray source in laboratories is an X-ray tube. Inside the tube is a stationary metal target (usually Cu). When the source is on, the target is exposed to a beam of electrons from a filament, usually with around 40 KV accelerating potential. There are windows in the tube from which the X ray beam escapes and is directed onto a specimen. The scattered electron intensity is measured by a detector. Usually, there is a monochromator somewhere between the source and detector to remove most of the continuous bremsstrahlung spectrum, as well as the K. But it is also possible to use a thin metal filter, such as Ni, which has an X-ray absorption spectrum that blocks the K and other unwanted X-ray energies. It is difficult to filter out the K, so it is often present in the resulting diffraction patterns. The diffraction plane can be horizontal or vertical (or something else), but there needs to be freedom to move the specimen and detector w.r.t the incident beam. There are a variety of ways to do that.
8 8 Common X-ray sources By far, the most common X-ray target used is Cu, with the alignment set so that the radiation used is Cu- K, but Mo- K is not too uncommon. target: Cu label transition E (KeV) (nm) relative intensity K p3 K p s s mean (nm) 0.54 target: Mo label transition E (KeV) (nm) relative intensity K p3 K p s s mean (nm) The relative intensities of K and K are fixed at : by the multiplicities of the initial states involved. Because they are so close in wavelength, it is common to take a weighted sum as a reasonable estimation of the wavelength, when both are present. Recall that, as photons, the energy-wavelength relationship is given by: 40 ev nm.4 KeV nm E There are other targets available too, such as Co and Ag. K peak doublets Using K radiation, it is unusual to completely filter out the K, so one is left with a doublet of closely spaced peaks. These are evident at high scattering angles. If there is no filtering of the K, we know that the relative intensity is precisely IK IK.0. These two peaks in can be thought of as one peak in q that correspond to a single lattice spacing. The values are related by 4sin 4sin q d So the two peaks should not be fit independently, as the relative positions and relative intensities are fixed.
9 9 Fitting the K doublet I would recommend fitting the peaks with something of the form sin sin I A f q 0, q,... A f q0, q,... b0 b b Essentially, the argument of the fitting functions is q sin, where a different wavelength is used for each peak. But the centroids and FWHMs in q are the same, and amplitudes are fixed at :. Lorentz-polarization factor For a powder pattern, using unpolarized X rays, a variety of intensity contributions related to the scattering geometry should be considered. These are often combined into a single term, called the Lorentz-polarization factor cos θb LθBcosθB cos θ B sinθ sin θ sin θ sinθ B B B B All of the diffracted intensities will be weighted by this factor. But there are other factors, too. There is a multiplicity factor m for powders, because symmetry equivalent planes will superpose in the diffracted intensity. This is the number of equivalent permutations of Miller indices that correspond to a particular reflection. For example, a cubic crystal has 8 types of {} planes and types of {0} planes. Since the orientation of a powder is completely random, any of these permutations can contribute to a particular peak. Powder X-ray diffraction is almost completely kinematical diffraction, so the intensities vary as structure-factor squared. We also saw this in electron diffraction, we just evaluate it a little differently for X rays, compared to electrons. I m F Lθ Mθ, T B B We also have included the Lorentz-polarization factor and the Debye-Waller, or thermal, factors M θ B, T, which accounts for the damping of scattering power for large-angle reflections due to crystal vibrations.
10 0 Particle-size broadening We have not consider the peak width careful yet. It can be very broad for small nanoparticles. This is related to the geometric effect we discussed in TEM, in which the distribution of diffraction intensity in reciprocal-space is related to the fourier transform of the particle shape. Let's say the scattering vector is qk k g+s, where s is the excitation error and g is an RLV. For a radial scan, we can say s g a. Note that this is completely different from TEM, where we assumed s g. Write q sin, so Now dq cos d cos sq The intensity is a gaussian w.r.t. So cos sa s a a I I e max cos N a Now, let's say our particle size is L Na. Then the FWHM is I max I e max Lcos From this, we can estimate the width of the peak in : ln 0.94 Lcos Lcos Sometimes, this is just written K L cos where K. This is called the Scherrer equation.
X-ray diffraction geometry
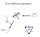 X-ray diffraction geometry Setting controls sample orientation in the diffraction plane. most important for single-crystal diffraction For any poly- (or nano-) crystalline specimen, we usually set: 1 X-ray
X-ray diffraction geometry Setting controls sample orientation in the diffraction plane. most important for single-crystal diffraction For any poly- (or nano-) crystalline specimen, we usually set: 1 X-ray
X-rays. X-ray Radiography - absorption is a function of Z and density. X-ray crystallography. X-ray spectrometry
 X-rays Wilhelm K. Roentgen (1845-1923) NP in Physics 1901 X-ray Radiography - absorption is a function of Z and density X-ray crystallography X-ray spectrometry X-rays Cu K α E = 8.05 kev λ = 1.541 Å Interaction
X-rays Wilhelm K. Roentgen (1845-1923) NP in Physics 1901 X-ray Radiography - absorption is a function of Z and density X-ray crystallography X-ray spectrometry X-rays Cu K α E = 8.05 kev λ = 1.541 Å Interaction
General theory of diffraction
 General theory of diffraction X-rays scatter off the charge density (r), neutrons scatter off the spin density. Coherent scattering (diffraction) creates the Fourier transform of (r) from real to reciprocal
General theory of diffraction X-rays scatter off the charge density (r), neutrons scatter off the spin density. Coherent scattering (diffraction) creates the Fourier transform of (r) from real to reciprocal
AP5301/ Name the major parts of an optical microscope and state their functions.
 Review Problems on Optical Microscopy AP5301/8301-2015 1. Name the major parts of an optical microscope and state their functions. 2. Compare the focal lengths of two glass converging lenses, one with
Review Problems on Optical Microscopy AP5301/8301-2015 1. Name the major parts of an optical microscope and state their functions. 2. Compare the focal lengths of two glass converging lenses, one with
X-ray diffraction is a non-invasive method for determining many types of
 Chapter X-ray Diffraction.1 Introduction X-ray diffraction is a non-invasive method for determining many types of structural features in both crystalline and amorphous materials. In the case of single
Chapter X-ray Diffraction.1 Introduction X-ray diffraction is a non-invasive method for determining many types of structural features in both crystalline and amorphous materials. In the case of single
Lecture 23 X-Ray & UV Techniques
 Lecture 23 X-Ray & UV Techniques Schroder: Chapter 11.3 1/50 Announcements Homework 6/6: Will be online on later today. Due Wednesday June 6th at 10:00am. I will return it at the final exam (14 th June).
Lecture 23 X-Ray & UV Techniques Schroder: Chapter 11.3 1/50 Announcements Homework 6/6: Will be online on later today. Due Wednesday June 6th at 10:00am. I will return it at the final exam (14 th June).
print first name print last name print student id grade
 print first name print last name print student id grade Experiment 2 X-ray fluorescence X-ray fluorescence (XRF) and X-ray diffraction (XRD) may be used to determine the constituent elements and the crystalline
print first name print last name print student id grade Experiment 2 X-ray fluorescence X-ray fluorescence (XRF) and X-ray diffraction (XRD) may be used to determine the constituent elements and the crystalline
X-RAY SPECTRA. Theory:
 12 Oct 18 X-ray.1 X-RAY SPECTRA In this experiment, a number of measurements involving x-rays will be made. The spectrum of x-rays emitted from a molybdenum target will be measured, and the experimental
12 Oct 18 X-ray.1 X-RAY SPECTRA In this experiment, a number of measurements involving x-rays will be made. The spectrum of x-rays emitted from a molybdenum target will be measured, and the experimental
Roger Johnson Structure and Dynamics: X-ray Diffraction Lecture 6
 6.1. Summary In this Lecture we cover the theory of x-ray diffraction, which gives direct information about the atomic structure of crystals. In these experiments, the wavelength of the incident beam must
6.1. Summary In this Lecture we cover the theory of x-ray diffraction, which gives direct information about the atomic structure of crystals. In these experiments, the wavelength of the incident beam must
X-ray practical: Crystallography
 X-ray practical: Crystallography Aim: To familiarise oneself with the operation of Tex-X-Ometer spectrometer and to use it to determine the lattice spacing in NaCl and LiF single crystals. Background:
X-ray practical: Crystallography Aim: To familiarise oneself with the operation of Tex-X-Ometer spectrometer and to use it to determine the lattice spacing in NaCl and LiF single crystals. Background:
Structural characterization. Part 1
 Structural characterization Part 1 Experimental methods X-ray diffraction Electron diffraction Neutron diffraction Light diffraction EXAFS-Extended X- ray absorption fine structure XANES-X-ray absorption
Structural characterization Part 1 Experimental methods X-ray diffraction Electron diffraction Neutron diffraction Light diffraction EXAFS-Extended X- ray absorption fine structure XANES-X-ray absorption
LAB 01 X-RAY EMISSION & ABSORPTION
 LAB 0 X-RAY EMISSION & ABSORPTION REPORT BY: TEAM MEMBER NAME: Ashley Tsai LAB SECTION No. 05 GROUP 2 EXPERIMENT DATE: Feb., 204 SUBMISSION DATE: Feb. 8, 204 Page of 3 ABSTRACT The goal of this experiment
LAB 0 X-RAY EMISSION & ABSORPTION REPORT BY: TEAM MEMBER NAME: Ashley Tsai LAB SECTION No. 05 GROUP 2 EXPERIMENT DATE: Feb., 204 SUBMISSION DATE: Feb. 8, 204 Page of 3 ABSTRACT The goal of this experiment
Structure of Surfaces
 Structure of Surfaces C Stepped surface Interference of two waves Bragg s law Path difference = AB+BC =2dsin ( =glancing angle) If, n =2dsin, constructive interference Ex) in a cubic lattice of unit cell
Structure of Surfaces C Stepped surface Interference of two waves Bragg s law Path difference = AB+BC =2dsin ( =glancing angle) If, n =2dsin, constructive interference Ex) in a cubic lattice of unit cell
Part 1: What is XAFS? What does it tell us? The EXAFS equation. Part 2: Basic steps in the analysis Quick overview of typical analysis
 Introduction to XAFS Part 1: What is XAFS? What does it tell us? The EXAFS equation Part 2: Basic steps in the analysis Quick overview of typical analysis Tomorrow Measurement methods and examples The
Introduction to XAFS Part 1: What is XAFS? What does it tell us? The EXAFS equation Part 2: Basic steps in the analysis Quick overview of typical analysis Tomorrow Measurement methods and examples The
3.012 Fund of Mat Sci: Structure Lecture 18
 3.012 Fund of Mat Sci: Structure Lecture 18 X-RAYS AT WORK An X-ray diffraction image for the protein myoglobin. Source: Wikipedia. Model of helical domains in myoglobin. Image courtesy of Magnus Manske
3.012 Fund of Mat Sci: Structure Lecture 18 X-RAYS AT WORK An X-ray diffraction image for the protein myoglobin. Source: Wikipedia. Model of helical domains in myoglobin. Image courtesy of Magnus Manske
X-ray, Neutron and e-beam scattering
 X-ray, Neutron and e-beam scattering Introduction Why scattering? Diffraction basics Neutrons and x-rays Techniques Direct and reciprocal space Single crystals Powders CaFe 2 As 2 an example What is the
X-ray, Neutron and e-beam scattering Introduction Why scattering? Diffraction basics Neutrons and x-rays Techniques Direct and reciprocal space Single crystals Powders CaFe 2 As 2 an example What is the
Elastic and Inelastic Scattering in Electron Diffraction and Imaging
 Elastic and Inelastic Scattering in Electron Diffraction and Imaging Contents Introduction Symbols and definitions Part A Diffraction and imaging of elastically scattered electrons Chapter 1. Basic kinematical
Elastic and Inelastic Scattering in Electron Diffraction and Imaging Contents Introduction Symbols and definitions Part A Diffraction and imaging of elastically scattered electrons Chapter 1. Basic kinematical
Swanning about in Reciprocal Space. Kenneth, what is the wavevector?
 Swanning about in Reciprocal Space or, Kenneth, what is the wavevector? Stanford Synchrotron Radiation Laboratory Principles The relationship between the reciprocal lattice vector and the wave vector is
Swanning about in Reciprocal Space or, Kenneth, what is the wavevector? Stanford Synchrotron Radiation Laboratory Principles The relationship between the reciprocal lattice vector and the wave vector is
V 11: Electron Diffraction
 Martin-Luther-University Halle-Wittenberg Institute of Physics Advanced Practical Lab Course V 11: Electron Diffraction An electron beam conditioned by an electron optical system is diffracted by a polycrystalline,
Martin-Luther-University Halle-Wittenberg Institute of Physics Advanced Practical Lab Course V 11: Electron Diffraction An electron beam conditioned by an electron optical system is diffracted by a polycrystalline,
Rajesh Prasad Department of Applied Mechanics Indian Institute of Technology New Delhi
 TEQIP WORKSHOP ON HIGH RESOLUTION X-RAY AND ELECTRON DIFFRACTION, FEB 01, 2016, IIT-K. Introduction to x-ray diffraction Peak Positions and Intensities Rajesh Prasad Department of Applied Mechanics Indian
TEQIP WORKSHOP ON HIGH RESOLUTION X-RAY AND ELECTRON DIFFRACTION, FEB 01, 2016, IIT-K. Introduction to x-ray diffraction Peak Positions and Intensities Rajesh Prasad Department of Applied Mechanics Indian
X-ray Spectroscopy. Danny Bennett and Maeve Madigan. October 12, 2015
 X-ray Spectroscopy Danny Bennett and Maeve Madigan October 12, 2015 Abstract Various X-ray spectra were obtained, and their properties were investigated. The characteristic peaks were identified for a
X-ray Spectroscopy Danny Bennett and Maeve Madigan October 12, 2015 Abstract Various X-ray spectra were obtained, and their properties were investigated. The characteristic peaks were identified for a
Transmission Electron Microscopy
 L. Reimer H. Kohl Transmission Electron Microscopy Physics of Image Formation Fifth Edition el Springer Contents 1 Introduction... 1 1.1 Transmission Electron Microscopy... 1 1.1.1 Conventional Transmission
L. Reimer H. Kohl Transmission Electron Microscopy Physics of Image Formation Fifth Edition el Springer Contents 1 Introduction... 1 1.1 Transmission Electron Microscopy... 1 1.1.1 Conventional Transmission
Imaging Methods: Scanning Force Microscopy (SFM / AFM)
 Imaging Methods: Scanning Force Microscopy (SFM / AFM) The atomic force microscope (AFM) probes the surface of a sample with a sharp tip, a couple of microns long and often less than 100 Å in diameter.
Imaging Methods: Scanning Force Microscopy (SFM / AFM) The atomic force microscope (AFM) probes the surface of a sample with a sharp tip, a couple of microns long and often less than 100 Å in diameter.
Röntgenpraktikum. M. Oehzelt. (based on the diploma thesis of T. Haber [1])
![Röntgenpraktikum. M. Oehzelt. (based on the diploma thesis of T. Haber [1]) Röntgenpraktikum. M. Oehzelt. (based on the diploma thesis of T. Haber [1])](/thumbs/91/106501237.jpg) Röntgenpraktikum M. Oehzelt (based on the diploma thesis of T. Haber [1]) October 21, 2004 Contents 1 Fundamentals 2 1.1 X-Ray Radiation......................... 2 1.1.1 Bremsstrahlung......................
Röntgenpraktikum M. Oehzelt (based on the diploma thesis of T. Haber [1]) October 21, 2004 Contents 1 Fundamentals 2 1.1 X-Ray Radiation......................... 2 1.1.1 Bremsstrahlung......................
X-Ray Emission and Absorption
 X-Ray Emission and Absorption Author: Mike Nill Alex Bryant February 6, 20 Abstract X-rays were produced by two bench-top diffractometers using a copper target. Various nickel filters were placed in front
X-Ray Emission and Absorption Author: Mike Nill Alex Bryant February 6, 20 Abstract X-rays were produced by two bench-top diffractometers using a copper target. Various nickel filters were placed in front
X-Rays Edited 2/19/18 by DGH & Stephen Albright
 X-Rays Edited 2/19/18 by DGH & Stephen Albright PURPOSE OF EXPERIMENT: To investigate the production, diffraction and absorption of x-rays. REFERENCES: Tipler, 3-6, 4-4; Enge, Wehr and Richards, Chapter
X-Rays Edited 2/19/18 by DGH & Stephen Albright PURPOSE OF EXPERIMENT: To investigate the production, diffraction and absorption of x-rays. REFERENCES: Tipler, 3-6, 4-4; Enge, Wehr and Richards, Chapter
Chapter 2. X-ray X. Diffraction and Reciprocal Lattice. Scattering from Lattices
 Chapter. X-ray X Diffraction and Reciprocal Lattice Diffraction of waves by crystals Reciprocal Lattice Diffraction of X-rays Powder diffraction Single crystal X-ray diffraction Scattering from Lattices
Chapter. X-ray X Diffraction and Reciprocal Lattice Diffraction of waves by crystals Reciprocal Lattice Diffraction of X-rays Powder diffraction Single crystal X-ray diffraction Scattering from Lattices
Surface Sensitivity & Surface Specificity
 Surface Sensitivity & Surface Specificity The problems of sensitivity and detection limits are common to all forms of spectroscopy. In its simplest form, the question of sensitivity boils down to whether
Surface Sensitivity & Surface Specificity The problems of sensitivity and detection limits are common to all forms of spectroscopy. In its simplest form, the question of sensitivity boils down to whether
Andrew D. Kent. 1 Introduction. p 1
 Compton Effect Andrew D. Kent Introduction One of the most important experiments in the early days of quantum mechanics (93) studied the interaction between light and matter; it determined the change in
Compton Effect Andrew D. Kent Introduction One of the most important experiments in the early days of quantum mechanics (93) studied the interaction between light and matter; it determined the change in
High-Resolution. Transmission. Electron Microscopy
 Part 4 High-Resolution Transmission Electron Microscopy 186 Significance high-resolution transmission electron microscopy (HRTEM): resolve object details smaller than 1nm (10 9 m) image the interior of
Part 4 High-Resolution Transmission Electron Microscopy 186 Significance high-resolution transmission electron microscopy (HRTEM): resolve object details smaller than 1nm (10 9 m) image the interior of
Structure Report for J. Reibenspies
 X-ray Diffraction Laboratory Center for Chemical Characterization and Analysis Department of Chemistry Texas A & M University Structure Report for J. Reibenspies Project Name: Sucrose Date: January 29,
X-ray Diffraction Laboratory Center for Chemical Characterization and Analysis Department of Chemistry Texas A & M University Structure Report for J. Reibenspies Project Name: Sucrose Date: January 29,
UNIVERSITY OF SURREY DEPARTMENT OF PHYSICS. Level 1: Experiment 2F THE ABSORPTION, DIFFRACTION AND EMISSION OF X- RAY RADIATION
 UNIVERSITY OF SURREY DEPARTMENT OF PHYSICS Level 1: Experiment 2F THE ABSORPTION, DIFFRACTION AND EMISSION OF X- RAY RADIATION 1 AIMS 1.1 Physics These experiments are intended to give some experience
UNIVERSITY OF SURREY DEPARTMENT OF PHYSICS Level 1: Experiment 2F THE ABSORPTION, DIFFRACTION AND EMISSION OF X- RAY RADIATION 1 AIMS 1.1 Physics These experiments are intended to give some experience
Keble College - Hilary 2012 Section VI: Condensed matter physics Tutorial 2 - Lattices and scattering
 Tomi Johnson Keble College - Hilary 2012 Section VI: Condensed matter physics Tutorial 2 - Lattices and scattering Please leave your work in the Clarendon laboratory s J pigeon hole by 5pm on Monday of
Tomi Johnson Keble College - Hilary 2012 Section VI: Condensed matter physics Tutorial 2 - Lattices and scattering Please leave your work in the Clarendon laboratory s J pigeon hole by 5pm on Monday of
Transmission Electron Microscopy and Diffractometry of Materials
 Brent Fultz James Howe Transmission Electron Microscopy and Diffractometry of Materials Fourth Edition ~Springer 1 1 Diffraction and the X-Ray Powder Diffractometer 1 1.1 Diffraction... 1 1.1.1 Introduction
Brent Fultz James Howe Transmission Electron Microscopy and Diffractometry of Materials Fourth Edition ~Springer 1 1 Diffraction and the X-Ray Powder Diffractometer 1 1.1 Diffraction... 1 1.1.1 Introduction
Muffin-tin potentials in EXAFS analysis
 J. Synchrotron Rad. (5)., doi:.7/s6577555 Supporting information Volume (5) Supporting information for article: Muffin-tin potentials in EXAFS analysis B. Ravel Supplemental materials: Muffin tin potentials
J. Synchrotron Rad. (5)., doi:.7/s6577555 Supporting information Volume (5) Supporting information for article: Muffin-tin potentials in EXAFS analysis B. Ravel Supplemental materials: Muffin tin potentials
Good Diffraction Practice Webinar Series
 Good Diffraction Practice Webinar Series High Resolution X-ray Diffractometry (1) Mar 24, 2011 www.bruker-webinars.com Welcome Heiko Ress Global Marketing Manager Bruker AXS Inc. Madison, Wisconsin, USA
Good Diffraction Practice Webinar Series High Resolution X-ray Diffractometry (1) Mar 24, 2011 www.bruker-webinars.com Welcome Heiko Ress Global Marketing Manager Bruker AXS Inc. Madison, Wisconsin, USA
Scattering Techniques and Geometries How to choose a beamline. Christopher J. Tassone
 Scattering Techniques and Geometries How to choose a beamline Christopher J. Tassone Why Care About Geometries? How do you decide which beamline you want to use? Questions you should be asking Do I want
Scattering Techniques and Geometries How to choose a beamline Christopher J. Tassone Why Care About Geometries? How do you decide which beamline you want to use? Questions you should be asking Do I want
Particles and Waves Particles Waves
 Particles and Waves Particles Discrete and occupy space Exist in only one location at a time Position and velocity can be determined with infinite accuracy Interact by collisions, scattering. Waves Extended,
Particles and Waves Particles Discrete and occupy space Exist in only one location at a time Position and velocity can be determined with infinite accuracy Interact by collisions, scattering. Waves Extended,
NEW CORRECTION PROCEDURE FOR X-RAY SPECTROSCOPIC FLUORESCENCE DATA: SIMULATIONS AND EXPERIMENT
 Copyright JCPDS - International Centre for Diffraction Data 2005, Advances in X-ray Analysis, Volume 48. 266 NEW CORRECTION PROCEDURE FOR X-RAY SPECTROSCOPIC FLUORESCENCE DATA: SIMULATIONS AND EXPERIMENT
Copyright JCPDS - International Centre for Diffraction Data 2005, Advances in X-ray Analysis, Volume 48. 266 NEW CORRECTION PROCEDURE FOR X-RAY SPECTROSCOPIC FLUORESCENCE DATA: SIMULATIONS AND EXPERIMENT
Chemistry Instrumental Analysis Lecture 19 Chapter 12. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 19 Chapter 12 There are three major techniques used for elemental analysis: Optical spectrometry Mass spectrometry X-ray spectrometry X-ray Techniques include:
Chemistry 4631 Instrumental Analysis Lecture 19 Chapter 12 There are three major techniques used for elemental analysis: Optical spectrometry Mass spectrometry X-ray spectrometry X-ray Techniques include:
Data collection Strategy. Apurva Mehta
 Data collection Strategy Apurva Mehta Outline Before.. Resolution, Aberrations and detectors During.. What is the scientific question? How will probing the structure help? Is there an alternative method?
Data collection Strategy Apurva Mehta Outline Before.. Resolution, Aberrations and detectors During.. What is the scientific question? How will probing the structure help? Is there an alternative method?
THE IMPORTANCE OF THE SPECIMEN DISPLACEMENT CORRECTION IN RIETVELD PATTERN FITTING WITH SYMMETRIC REFLECTION-OPTICS DIFFRACTION DATA
 Copyright(c)JCPDS-International Centre for Diffraction Data 2001,Advances in X-ray Analysis,Vol.44 96 THE IMPORTANCE OF THE SPECIMEN DISPLACEMENT CORRECTION IN RIETVELD PATTERN FITTING WITH SYMMETRIC REFLECTION-OPTICS
Copyright(c)JCPDS-International Centre for Diffraction Data 2001,Advances in X-ray Analysis,Vol.44 96 THE IMPORTANCE OF THE SPECIMEN DISPLACEMENT CORRECTION IN RIETVELD PATTERN FITTING WITH SYMMETRIC REFLECTION-OPTICS
Photon Physics. Week 4 26/02/2013
 Photon Physics Week 4 6//13 1 Classical atom-field interaction Lorentz oscillator: Classical electron oscillator with frequency ω and damping constant γ Eqn of motion: Final result: Classical atom-field
Photon Physics Week 4 6//13 1 Classical atom-field interaction Lorentz oscillator: Classical electron oscillator with frequency ω and damping constant γ Eqn of motion: Final result: Classical atom-field
Crystal Structure SOLID STATE PHYSICS. Lecture 5. A.H. Harker. thelecture thenextlecture. Physics and Astronomy UCL
 Crystal Structure thelecture thenextlecture SOLID STATE PHYSICS Lecture 5 A.H. Harker Physics and Astronomy UCL Structure & Diffraction Crystal Diffraction (continued) 2.4 Experimental Methods Notes: examples
Crystal Structure thelecture thenextlecture SOLID STATE PHYSICS Lecture 5 A.H. Harker Physics and Astronomy UCL Structure & Diffraction Crystal Diffraction (continued) 2.4 Experimental Methods Notes: examples
Silver Thin Film Characterization
 Silver Thin Film Characterization.1 Introduction Thin films of Ag layered structures, typically less than a micron in thickness, are tailored to achieve desired functional properties. Typical characterization
Silver Thin Film Characterization.1 Introduction Thin films of Ag layered structures, typically less than a micron in thickness, are tailored to achieve desired functional properties. Typical characterization
4. Inelastic Scattering
 1 4. Inelastic Scattering Some inelastic scattering processes A vast range of inelastic scattering processes can occur during illumination of a specimen with a highenergy electron beam. In principle, many
1 4. Inelastic Scattering Some inelastic scattering processes A vast range of inelastic scattering processes can occur during illumination of a specimen with a highenergy electron beam. In principle, many
Scattering Lecture. February 24, 2014
 Scattering Lecture February 24, 2014 Structure Determination by Scattering Waves of radiation scattered by different objects interfere to give rise to an observable pattern! The wavelength needs to close
Scattering Lecture February 24, 2014 Structure Determination by Scattering Waves of radiation scattered by different objects interfere to give rise to an observable pattern! The wavelength needs to close
Detection of X-Rays. Solid state detectors Proportional counters Microcalorimeters Detector characteristics
 Detection of X-Rays Solid state detectors Proportional counters Microcalorimeters Detector characteristics Solid State X-ray Detectors X-ray interacts in material to produce photoelectrons which are collected
Detection of X-Rays Solid state detectors Proportional counters Microcalorimeters Detector characteristics Solid State X-ray Detectors X-ray interacts in material to produce photoelectrons which are collected
An Introduction to Diffraction and Scattering. School of Chemistry The University of Sydney
 An Introduction to Diffraction and Scattering Brendan J. Kennedy School of Chemistry The University of Sydney 1) Strong forces 2) Weak forces Types of Forces 3) Electromagnetic forces 4) Gravity Types
An Introduction to Diffraction and Scattering Brendan J. Kennedy School of Chemistry The University of Sydney 1) Strong forces 2) Weak forces Types of Forces 3) Electromagnetic forces 4) Gravity Types
(2) A two-dimensional solid has an electron energy band of the form, . [1]
![(2) A two-dimensional solid has an electron energy band of the form, . [1] (2) A two-dimensional solid has an electron energy band of the form, . [1]](/thumbs/96/128751828.jpg) (1) The figure shows a two-dimensional periodic lattice, containing A atoms (white) and B atoms (black). The section of lattice shown makes a 3a 4a rectangle, as shown (measured from A atom to A atom).
(1) The figure shows a two-dimensional periodic lattice, containing A atoms (white) and B atoms (black). The section of lattice shown makes a 3a 4a rectangle, as shown (measured from A atom to A atom).
TEP Examination of the structure of NaCl monocrystals with different orientations
 Examination of the structure of NaCl TEP Related topics Characteristic X-radiation, energy levels, crystal structures, reciprocal lattices, Miller indices, atomic form factor, structure factor, and Bragg
Examination of the structure of NaCl TEP Related topics Characteristic X-radiation, energy levels, crystal structures, reciprocal lattices, Miller indices, atomic form factor, structure factor, and Bragg
Notes on x-ray scattering - M. Le Tacon, B. Keimer (06/2015)
 Notes on x-ray scattering - M. Le Tacon, B. Keimer (06/2015) Interaction of x-ray with matter: - Photoelectric absorption - Elastic (coherent) scattering (Thomson Scattering) - Inelastic (incoherent) scattering
Notes on x-ray scattering - M. Le Tacon, B. Keimer (06/2015) Interaction of x-ray with matter: - Photoelectric absorption - Elastic (coherent) scattering (Thomson Scattering) - Inelastic (incoherent) scattering
Quantum Condensed Matter Physics Lecture 5
 Quantum Condensed Matter Physics Lecture 5 detector sample X-ray source monochromator David Ritchie http://www.sp.phy.cam.ac.uk/drp2/home QCMP Lent/Easter 2019 5.1 Quantum Condensed Matter Physics 1. Classical
Quantum Condensed Matter Physics Lecture 5 detector sample X-ray source monochromator David Ritchie http://www.sp.phy.cam.ac.uk/drp2/home QCMP Lent/Easter 2019 5.1 Quantum Condensed Matter Physics 1. Classical
Absorption of X-rays
 Absorption of X-rays TEP Related topics Bremsstrahlung, characteristic X-radiation, Bragg scattering, law of absorption, mass absorption coefficient, absorption edges, half-value thickness, photoelectric
Absorption of X-rays TEP Related topics Bremsstrahlung, characteristic X-radiation, Bragg scattering, law of absorption, mass absorption coefficient, absorption edges, half-value thickness, photoelectric
CHARACTERIZATION of NANOMATERIALS KHP
 CHARACTERIZATION of NANOMATERIALS Overview of the most common nanocharacterization techniques MAIN CHARACTERIZATION TECHNIQUES: 1.Transmission Electron Microscope (TEM) 2. Scanning Electron Microscope
CHARACTERIZATION of NANOMATERIALS Overview of the most common nanocharacterization techniques MAIN CHARACTERIZATION TECHNIQUES: 1.Transmission Electron Microscope (TEM) 2. Scanning Electron Microscope
Bragg reflection :determining the lattice constants of monocrystals
 Bragg reflection :determining the lattice constants of monocrystals Objectives: 1-Investagating Bragg reflection at Nacl monocrystal -determinig the lattice constant a 0 of NaCl. Theory: Bragg's law of
Bragg reflection :determining the lattice constants of monocrystals Objectives: 1-Investagating Bragg reflection at Nacl monocrystal -determinig the lattice constant a 0 of NaCl. Theory: Bragg's law of
de Broglie Waves h p de Broglie argued Light exhibits both wave and particle properties
 de Broglie argued de Broglie Waves Light exhibits both wave and particle properties Wave interference, diffraction Particle photoelectric effect, Compton effect Then matter (particles) should exhibit both
de Broglie argued de Broglie Waves Light exhibits both wave and particle properties Wave interference, diffraction Particle photoelectric effect, Compton effect Then matter (particles) should exhibit both
Crystals, X-rays and Proteins
 Crystals, X-rays and Proteins Comprehensive Protein Crystallography Dennis Sherwood MA (Hons), MPhil, PhD Jon Cooper BA (Hons), PhD OXFORD UNIVERSITY PRESS Contents List of symbols xiv PART I FUNDAMENTALS
Crystals, X-rays and Proteins Comprehensive Protein Crystallography Dennis Sherwood MA (Hons), MPhil, PhD Jon Cooper BA (Hons), PhD OXFORD UNIVERSITY PRESS Contents List of symbols xiv PART I FUNDAMENTALS
CHAPTER 3. OPTICAL STUDIES ON SnS NANOPARTICLES
 42 CHAPTER 3 OPTICAL STUDIES ON SnS NANOPARTICLES 3.1 INTRODUCTION In recent years, considerable interest has been shown on semiconducting nanostructures owing to their enhanced optical and electrical
42 CHAPTER 3 OPTICAL STUDIES ON SnS NANOPARTICLES 3.1 INTRODUCTION In recent years, considerable interest has been shown on semiconducting nanostructures owing to their enhanced optical and electrical
Electron Diffraction
 Exp-3-Electron Diffraction.doc (TJR) Physics Department, University of Windsor Introduction 64-311 Laboratory Experiment 3 Electron Diffraction In 1924 de Broglie predicted that the wavelength of matter
Exp-3-Electron Diffraction.doc (TJR) Physics Department, University of Windsor Introduction 64-311 Laboratory Experiment 3 Electron Diffraction In 1924 de Broglie predicted that the wavelength of matter
X-ray uorescence, X-ray powder diraction and Raman spectrosopy
 X-ray uorescence, X-ray powder diraction and Raman spectrosopy Wubulikasimu Yibulayin, Lovro Pavleti, Kuerbannisa Muhetaer 12.05.2016. Abstract In this report we report about the experiments that were
X-ray uorescence, X-ray powder diraction and Raman spectrosopy Wubulikasimu Yibulayin, Lovro Pavleti, Kuerbannisa Muhetaer 12.05.2016. Abstract In this report we report about the experiments that were
E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051
 E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 38, 45 ELECTRON DIFFRACTION Introduction This experiment, using a cathode ray tube, permits the first hand observation that electrons, which are usually though
E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 38, 45 ELECTRON DIFFRACTION Introduction This experiment, using a cathode ray tube, permits the first hand observation that electrons, which are usually though
Structure analysis: Electron diffraction LEED TEM RHEED
 Structure analysis: Electron diffraction LEED: Low Energy Electron Diffraction SPA-LEED: Spot Profile Analysis Low Energy Electron diffraction RHEED: Reflection High Energy Electron Diffraction TEM: Transmission
Structure analysis: Electron diffraction LEED: Low Energy Electron Diffraction SPA-LEED: Spot Profile Analysis Low Energy Electron diffraction RHEED: Reflection High Energy Electron Diffraction TEM: Transmission
X-RAY SCATTERING AND MOSELEY S LAW. OBJECTIVE: To investigate Moseley s law using X-ray absorption and to observe X- ray scattering.
 X-RAY SCATTERING AND MOSELEY S LAW OBJECTIVE: To investigate Moseley s law using X-ray absorption and to observe X- ray scattering. READING: Krane, Section 8.5. BACKGROUND: In 1913, Henry Moseley measured
X-RAY SCATTERING AND MOSELEY S LAW OBJECTIVE: To investigate Moseley s law using X-ray absorption and to observe X- ray scattering. READING: Krane, Section 8.5. BACKGROUND: In 1913, Henry Moseley measured
Crystal Structure Determination II
 Crystal Structure Determination II Dr. Falak Sher Pakistan Institute of Engineering and Applied Sciences 09/10/2010 Diffraction Intensities The integrated intensity, I (hkl) (peak area) of each powder
Crystal Structure Determination II Dr. Falak Sher Pakistan Institute of Engineering and Applied Sciences 09/10/2010 Diffraction Intensities The integrated intensity, I (hkl) (peak area) of each powder
Lecture 10. Lidar Effective Cross-Section vs. Convolution
 Lecture 10. Lidar Effective Cross-Section vs. Convolution q Introduction q Convolution in Lineshape Determination -- Voigt Lineshape (Lorentzian Gaussian) q Effective Cross Section for Single Isotope --
Lecture 10. Lidar Effective Cross-Section vs. Convolution q Introduction q Convolution in Lineshape Determination -- Voigt Lineshape (Lorentzian Gaussian) q Effective Cross Section for Single Isotope --
3.012 Structure An Introduction to X-ray Diffraction
 3.012 Structure An Introduction to X-ray Diffraction This handout summarizes some topics that are important for understanding x-ray diffraction. The following references provide a thorough explanation
3.012 Structure An Introduction to X-ray Diffraction This handout summarizes some topics that are important for understanding x-ray diffraction. The following references provide a thorough explanation
Detecting high energy photons. Interactions of photons with matter Properties of detectors (with examples)
 Detecting high energy photons Interactions of photons with matter Properties of detectors (with examples) Interactions of high energy photons with matter Cross section/attenution length/optical depth Photoelectric
Detecting high energy photons Interactions of photons with matter Properties of detectors (with examples) Interactions of high energy photons with matter Cross section/attenution length/optical depth Photoelectric
X Rays & Crystals. Characterizing Mineral Chemistry & Structure. J.D. Price
 X Rays & Crystals Characterizing Mineral Chemistry & Structure J.D. Price Light - electromagnetic spectrum Wave behavior vs. particle behavior If atoms are on the 10-10 m scale, we need to use sufficiently
X Rays & Crystals Characterizing Mineral Chemistry & Structure J.D. Price Light - electromagnetic spectrum Wave behavior vs. particle behavior If atoms are on the 10-10 m scale, we need to use sufficiently
Title of file for HTML: Supplementary Information Description: Supplementary Figures and Supplementary References
 Title of file for HTML: Supplementary Information Description: Supplementary Figures and Supplementary References Supplementary Figure 1. SEM images of perovskite single-crystal patterned thin film with
Title of file for HTML: Supplementary Information Description: Supplementary Figures and Supplementary References Supplementary Figure 1. SEM images of perovskite single-crystal patterned thin film with
5.8 Auger Electron Spectroscopy (AES)
 5.8 Auger Electron Spectroscopy (AES) 5.8.1 The Auger Process X-ray and high energy electron bombardment of atom can create core hole Core hole will eventually decay via either (i) photon emission (x-ray
5.8 Auger Electron Spectroscopy (AES) 5.8.1 The Auger Process X-ray and high energy electron bombardment of atom can create core hole Core hole will eventually decay via either (i) photon emission (x-ray
MSE405 Microstructure Characterization XRD-1 Lab X-ray diffraction in crystallography
 X-ray diraction in crystallography I. Goals Crystallography is the science that studies the structure (and structure-derived properties) o crystals. Among its many tools, X-ray diraction (XRD) has had
X-ray diraction in crystallography I. Goals Crystallography is the science that studies the structure (and structure-derived properties) o crystals. Among its many tools, X-ray diraction (XRD) has had
Neutron and x-ray spectroscopy
 Neutron and x-ray spectroscopy B. Keimer Max-Planck-Institute for Solid State Research outline 1. self-contained introduction neutron scattering and spectroscopy x-ray scattering and spectroscopy 2. application
Neutron and x-ray spectroscopy B. Keimer Max-Planck-Institute for Solid State Research outline 1. self-contained introduction neutron scattering and spectroscopy x-ray scattering and spectroscopy 2. application
Electron Microscopy I
 Characterization of Catalysts and Surfaces Characterization Techniques in Heterogeneous Catalysis Electron Microscopy I Introduction Properties of electrons Electron-matter interactions and their applications
Characterization of Catalysts and Surfaces Characterization Techniques in Heterogeneous Catalysis Electron Microscopy I Introduction Properties of electrons Electron-matter interactions and their applications
X-ray Diffraction. Interaction of Waves Reciprocal Lattice and Diffraction X-ray Scattering by Atoms The Integrated Intensity
 X-ray Diraction Interaction o Waves Reciprocal Lattice and Diraction X-ray Scattering by Atoms The Integrated Intensity Basic Principles o Interaction o Waves Periodic waves characteristic: Frequency :
X-ray Diraction Interaction o Waves Reciprocal Lattice and Diraction X-ray Scattering by Atoms The Integrated Intensity Basic Principles o Interaction o Waves Periodic waves characteristic: Frequency :
4. Other diffraction techniques
 4. Other diffraction techniques 4.1 Reflection High Energy Electron Diffraction (RHEED) Setup: - Grazing-incidence high energy electron beam (3-5 kev: MEED,
4. Other diffraction techniques 4.1 Reflection High Energy Electron Diffraction (RHEED) Setup: - Grazing-incidence high energy electron beam (3-5 kev: MEED,
Electron Diffraction
 Electron iffraction o moving electrons display wave nature? To answer this question you will direct a beam of electrons through a thin layer of carbon and analyze the resulting pattern. Theory Louis de
Electron iffraction o moving electrons display wave nature? To answer this question you will direct a beam of electrons through a thin layer of carbon and analyze the resulting pattern. Theory Louis de
Diffractometer. Geometry Optics Detectors
 Diffractometer Geometry Optics Detectors Diffractometers Debye Scherrer Camera V.K. Pecharsky and P.Y. Zavalij Fundamentals of Powder Diffraction and Structural Characterization of Materials. Diffractometers
Diffractometer Geometry Optics Detectors Diffractometers Debye Scherrer Camera V.K. Pecharsky and P.Y. Zavalij Fundamentals of Powder Diffraction and Structural Characterization of Materials. Diffractometers
2D XRD Imaging by Projection-Type X-Ray Microscope
 0/25 National Institute for Materials Science,Tsukuba, Japan 2D XRD Imaging by Projection-Type X-Ray Microscope 1. Introduction - What s projection-type X-ray microscope? 2. Examples for inhomogeneous/patterned
0/25 National Institute for Materials Science,Tsukuba, Japan 2D XRD Imaging by Projection-Type X-Ray Microscope 1. Introduction - What s projection-type X-ray microscope? 2. Examples for inhomogeneous/patterned
Summary Chapter 2: Wave diffraction and the reciprocal lattice.
 Summary Chapter : Wave diffraction and the reciprocal lattice. In chapter we discussed crystal diffraction and introduced the reciprocal lattice. Since crystal have a translation symmetry as discussed
Summary Chapter : Wave diffraction and the reciprocal lattice. In chapter we discussed crystal diffraction and introduced the reciprocal lattice. Since crystal have a translation symmetry as discussed
Supplementary Information
 Supplementary Information Bismuth Sulfide Nanoflowers for Detection of X-rays in the Mammographic Energy Range Shruti Nambiar a,b, Ernest K. Osei a,c,d, John T.W.Yeow *,a,b a Department of Systems Design
Supplementary Information Bismuth Sulfide Nanoflowers for Detection of X-rays in the Mammographic Energy Range Shruti Nambiar a,b, Ernest K. Osei a,c,d, John T.W.Yeow *,a,b a Department of Systems Design
Probing Atomic Crystals: Bragg Diffraction
 1 Probing Atomic Crystals: Bragg Diffraction OBJECTIVE: To learn how scientists probe the structure of solids, using a scaled-up version of X-ray Diffraction. APPARATUS: Steel ball "crystal", microwave
1 Probing Atomic Crystals: Bragg Diffraction OBJECTIVE: To learn how scientists probe the structure of solids, using a scaled-up version of X-ray Diffraction. APPARATUS: Steel ball "crystal", microwave
Scattering and Diffraction
 Scattering and Diffraction Adventures in k-space, part 1 Lenson Pellouchoud SLAC / SSL XSD summer school 7/16/018 lenson@slac.stanford.edu Outline Elastic Scattering eview / overview and terminology Form
Scattering and Diffraction Adventures in k-space, part 1 Lenson Pellouchoud SLAC / SSL XSD summer school 7/16/018 lenson@slac.stanford.edu Outline Elastic Scattering eview / overview and terminology Form
Characteristic X-rays of molybdenum
 Characteristic X-rays of molybdenum TEP Related Topics X-ray tubes, bremsstrahlung, characteristic X-radiation, energy levels, crystal structures, lattice constant, absorption of X-rays, absorption edges,
Characteristic X-rays of molybdenum TEP Related Topics X-ray tubes, bremsstrahlung, characteristic X-radiation, energy levels, crystal structures, lattice constant, absorption of X-rays, absorption edges,
Atomic and nuclear physics
 Atomic and nuclear physics X-ray physics Attenuation of x-rays LEYBOLD Physics Leaflets P6.3.2.2 Investigating the wavelength dependency of the coefficient of attenuation Objects of the experiment To measure
Atomic and nuclear physics X-ray physics Attenuation of x-rays LEYBOLD Physics Leaflets P6.3.2.2 Investigating the wavelength dependency of the coefficient of attenuation Objects of the experiment To measure
Revision Guide. Chapter 7 Quantum Behaviour
 Revision Guide Chapter 7 Quantum Behaviour Contents CONTENTS... 2 REVISION CHECKLIST... 3 REVISION NOTES... 4 QUANTUM BEHAVIOUR... 4 Random arrival of photons... 4 Photoelectric effect... 5 PHASE AN PHASORS...
Revision Guide Chapter 7 Quantum Behaviour Contents CONTENTS... 2 REVISION CHECKLIST... 3 REVISION NOTES... 4 QUANTUM BEHAVIOUR... 4 Random arrival of photons... 4 Photoelectric effect... 5 PHASE AN PHASORS...
Handout 7 Reciprocal Space
 Handout 7 Reciprocal Space Useful concepts for the analysis of diffraction data http://homepages.utoledo.edu/clind/ Concepts versus reality Reflection from lattice planes is just a concept that helps us
Handout 7 Reciprocal Space Useful concepts for the analysis of diffraction data http://homepages.utoledo.edu/clind/ Concepts versus reality Reflection from lattice planes is just a concept that helps us
X-ray absorption. 4. Prove that / = f(z 3.12 ) applies.
 Related topics Bremsstrahlung, characteristic radiation, Bragg scattering, law of absorption, mass absorption coefficient, absorption edge, half-value thickness, photoelectric effect, Compton scattering,
Related topics Bremsstrahlung, characteristic radiation, Bragg scattering, law of absorption, mass absorption coefficient, absorption edge, half-value thickness, photoelectric effect, Compton scattering,
SOLID STATE 18. Reciprocal Space
 SOLID STATE 8 Reciprocal Space Wave vectors and the concept of K-space can simplify the explanation of several properties of the solid state. They will be introduced to provide more information on diffraction
SOLID STATE 8 Reciprocal Space Wave vectors and the concept of K-space can simplify the explanation of several properties of the solid state. They will be introduced to provide more information on diffraction
A. 0.7 ev B. 1.1 ev C. 1.4 ev D. 1.8 ev E. 2.1 ev F. 3.2 ev G. Something else
 A molecule has the energy levels shown in the diagram at the right. We begin with a large number of these molecules in their ground states. We want to raise a lot of these molecules to the state labeled
A molecule has the energy levels shown in the diagram at the right. We begin with a large number of these molecules in their ground states. We want to raise a lot of these molecules to the state labeled
DIFFRACTION PHYSICS THIRD REVISED EDITION JOHN M. COWLEY. Regents' Professor enzeritus Arizona State University
 DIFFRACTION PHYSICS THIRD REVISED EDITION JOHN M. COWLEY Regents' Professor enzeritus Arizona State University 1995 ELSEVIER Amsterdam Lausanne New York Oxford Shannon Tokyo CONTENTS Preface to the first
DIFFRACTION PHYSICS THIRD REVISED EDITION JOHN M. COWLEY Regents' Professor enzeritus Arizona State University 1995 ELSEVIER Amsterdam Lausanne New York Oxford Shannon Tokyo CONTENTS Preface to the first
Geology 777 Monte Carlo Exercise I
 Geology 777 Monte Carlo Exercise I Purpose The goal of this exercise is to get you to think like an electron... to start to think about where electrons from the stream of high energy electrons go when
Geology 777 Monte Carlo Exercise I Purpose The goal of this exercise is to get you to think like an electron... to start to think about where electrons from the stream of high energy electrons go when
Some Topics in Optics
 Some Topics in Optics The HeNe LASER The index of refraction and dispersion Interference The Michelson Interferometer Diffraction Wavemeter Fabry-Pérot Etalon and Interferometer The Helium Neon LASER A
Some Topics in Optics The HeNe LASER The index of refraction and dispersion Interference The Michelson Interferometer Diffraction Wavemeter Fabry-Pérot Etalon and Interferometer The Helium Neon LASER A
Supplementary Materials for
 wwwsciencemagorg/cgi/content/full/scienceaaa3035/dc1 Supplementary Materials for Spatially structured photons that travel in free space slower than the speed of light Daniel Giovannini, Jacquiline Romero,
wwwsciencemagorg/cgi/content/full/scienceaaa3035/dc1 Supplementary Materials for Spatially structured photons that travel in free space slower than the speed of light Daniel Giovannini, Jacquiline Romero,
Data Collection. Overview. Methods. Counter Methods. Crystal Quality with -Scans
 Data Collection Overview with a unit cell, possible space group and computer reference frame (orientation matrix); the location of diffracted x-rays can be calculated (h k l) and intercepted by something
Data Collection Overview with a unit cell, possible space group and computer reference frame (orientation matrix); the location of diffracted x-rays can be calculated (h k l) and intercepted by something
and another with a peak frequency ω 2
 Physics Qualifying Examination Part I 7-Minute Questions September 13, 2014 1. A sealed container is divided into two volumes by a moveable piston. There are N A molecules on one side and N B molecules
Physics Qualifying Examination Part I 7-Minute Questions September 13, 2014 1. A sealed container is divided into two volumes by a moveable piston. There are N A molecules on one side and N B molecules
CHEM*3440. X-Ray Energies. Bremsstrahlung Radiation. X-ray Line Spectra. Chemical Instrumentation. X-Ray Spectroscopy. Topic 13
 X-Ray Energies very short wavelength radiation 0.1Å to 10 nm (100 Å) CHEM*3440 Chemical Instrumentation Topic 13 X-Ray Spectroscopy Visible - Ultraviolet (UV) - Vacuum UV (VUV) - Extreme UV (XUV) - Soft
X-Ray Energies very short wavelength radiation 0.1Å to 10 nm (100 Å) CHEM*3440 Chemical Instrumentation Topic 13 X-Ray Spectroscopy Visible - Ultraviolet (UV) - Vacuum UV (VUV) - Extreme UV (XUV) - Soft
3.17 Strukturanalyse mit Röntgenstrahlen nach Debye- Scherrer
 3.17 Strukturanalyse mit Röntgenstrahlen nach Debye- Scherrer Ausarbeitung (engl.) Fortgeschrittenenpraktikum an der TU Darmstadt Versuch durchgeführt von: Mussie Beian, Jan Schupp, Florian Wetzel Versuchsdatum:
3.17 Strukturanalyse mit Röntgenstrahlen nach Debye- Scherrer Ausarbeitung (engl.) Fortgeschrittenenpraktikum an der TU Darmstadt Versuch durchgeführt von: Mussie Beian, Jan Schupp, Florian Wetzel Versuchsdatum:
Resolution: maximum limit of diffraction (asymmetric)
 Resolution: maximum limit of diffraction (asymmetric) crystal Y X-ray source 2θ X direct beam tan 2θ = Y X d = resolution 2d sinθ = λ detector 1 Unit Cell: two vectors in plane of image c* Observe: b*
Resolution: maximum limit of diffraction (asymmetric) crystal Y X-ray source 2θ X direct beam tan 2θ = Y X d = resolution 2d sinθ = λ detector 1 Unit Cell: two vectors in plane of image c* Observe: b*
Lab Manual: Determination of Planck s constant with x-rays
 Lab Manual: Determination of Planck s constant with x-rays 1. Purpose: To obtain a better understanding on the production of X-rays, the bremsstrahlung radiation and the characteristic radiation of a Molybdenum
Lab Manual: Determination of Planck s constant with x-rays 1. Purpose: To obtain a better understanding on the production of X-rays, the bremsstrahlung radiation and the characteristic radiation of a Molybdenum
