The octagon method for finding exceptional points, and application to hydrogen-like systems in parallel electric and magnetic fields
|
|
|
- Daniela Andrews
- 5 years ago
- Views:
Transcription
1 Institute of Theoretical Physics, University of Stuttgart, in collaboration with M. Feldmaier, F. Schweiner, J. Main, and H. Cartarius The octagon method for finding exceptional points, and application to hydrogen-like systems in parallel electric and magnetic fields Günter Wunner Institute of Theoretical Physics, University of Stuttgart, in collaboration with M. Feldmaier, F. Schweiner, J. Main, and H. Cartarius Prague, 6 June 2016
2 The coworkers M. Feldmaier F. Schweiner J. Main H. Cartarius Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
3 A feature of open quantum systems: Resonances exhibit avoided crossings on the real energy axis when the physical parameters of the systems are varied: example: hydrogen atom in parallel electric and magnetic fields Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
4 A feature of open quantum systems: Resonances exhibit avoided crossings on the real energy axis when the physical parameters of the systems are varied: example: hydrogen atom in parallel electric and magnetic fields Indicative of an exceptional point in the complex energy plane. Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
5 A feature of open quantum systems: Resonances exhibit avoided crossings on the real energy axis when the physical parameters of the systems are varied: example: hydrogen atom in parallel electric and magnetic fields Indicative of an exceptional point in the complex energy plane. How to find the exceptional point? Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
6 ] k 2 ) + ([A 2 (I ) 2m I2 + B Motivation ] {k1, k 2 } + B 3 2 (I 1S h2 + I 2 S h1 ) 2m 0 ( I 1 S h1 1 )] ) k 2 3 IS 1 h + c.p. 2m 0 ) + c.p. H so (1) Experiments on excitons in Cu 2 O in the group of D. Fröhlich and M. Beyer at the University of Dortmund tron mass m 0 and spin-orbit coupling ). (2) ], the energy shift -band is set to zero e spin matrices of he Pauli matrices. are defined as in,, band structure of Cu 2 O FIG. 1. (Color online) Band structure of Cu 2O [16]. Due to the spin-orbit coupling (2) the valence band splits into a lower lying fourfold-degenerated band ( ) Γ + 8 of and a higher lying twofold-degenerated band ( ) Γ + 7. The lowest lying conduction band has Γ + 6 symmetry. Depending on the bands involved, one distinguishes between the yellow, green, blue and violet Excitons: electron-hole pairs in semiconductors - hydrogen-like systems Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
7 Motivation ARCH LETTER Experiments on excitons in Cu 2 O in the group of D. Fröhlich and M. Beyer: Optical density 1.0 a n = 3 b n = n = 5 n = 6 Tsumeb mine Namibia c n = 12 n = 13 d n = 22 n = 23 n = 24 n = mm 30 mm 16 mm Photon energy (ev) 1 High-resolution absorption spectra of yellow P excitons T. Kazimierczuk in Cu 2 O. et al. crystal Giantfrom Rydberg which excitons samples in theofcopper different oxidesize Cuand 2 O. crystal Nature orientation (2014) w tra are measured with a single-frequency laser on a natural sample of c, A large crystal and a thin crystal mounted strain-free in a brass ho ss 34 mm at 1.2 Günter K. Peaks Wunner correspond (ITP1) to resonances Rydberg withsystems different in parallel electric d, Wavefunction and magnetic of fields the P exciton withprague, n To June visualize 2016 the 5 / giant 20 e
8 Rydberg excitons in magnetic and electric fields Reference field strengths: e B 0 m = 2E Ryd, ea Bohr F 0 = 2E Ryd Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
9 Rydberg excitons in magnetic and electric fields Reference field strengths: e B 0 m = 2E Ryd, ea Bohr F 0 = 2E Ryd H Cu 2 O E Ryd 13.6 ev ev a Bohr nm 1.04 nm B T T F V/cm V/cm Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
10 Rydberg systems in parallel electr. and magnet. fields Hydrogen atom: H hyd = p2 e2 1 2m 0 4πε 0 r + e B L z + e2 B 2 ( x 2 + y 2) + e F z 2m 0 8m 0 m p /m m p can be taken as approx. infinite Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
11 Rydberg systems in parallel electr. and magnet. fields Hydrogen atom: H hyd = p2 e2 1 2m 0 4πε 0 r + e B L z + e2 B 2 ( x 2 + y 2) + e F z 2m 0 8m 0 m p /m m p can be taken as approx. infinite Rydberg excitons: H ex = p2 2µ e2 1 4πε 0 ε r r + e B 2µ m h m e L z + e2 B 2 ( x 2 + y 2) + e F z m h + m e 8µ m e = 0.99m 0, m h = 0.62m 0 both masses are important, µ = 0.38m 0 Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
12 Rydberg systems in parallel electr. and magnet. fields H ex = p2 2µ e2 1 4πε 0 ε r r + e B 2µ m h m e L z + e2 B 2 ( x 2 + y 2) + e F z m h + m e 8µ parallel fields: angular momentum is conserved with respect to z-axis (L z m) paramagnetic term: B-dependent shift of zero point energy H = H H P Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
13 Rydberg systems in parallel electr. and magnet. fields H ex = p2 2µ e2 1 4πε 0 ε r r + e B 2µ m h m e L z + e2 B 2 ( x 2 + y 2) + e F z m h + m e 8µ parallel fields: angular momentum is conserved with respect to z-axis (L z m) paramagnetic term: B-dependent shift of zero point energy H = H H P in dimensionless units: lengths in a Bohr, energies in 2E Ryd, γ = B/B 0 and f = F/F 0 Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
14 Rydberg systems in parallel electr. and magnet. fields H ex = p2 2µ e2 1 4πε 0 ε r r + e B 2µ m h m e L z + e2 B 2 ( x 2 + y 2) + e F z m h + m e 8µ parallel fields: angular momentum is conserved with respect to z-axis (L z m) paramagnetic term: B-dependent shift of zero point energy H = H H P in dimensionless units: lengths in a Bohr, energies in 2E Ryd, γ = B/B 0 and f = F/F 0 Hamiltonian of hydrogen-like systems in parallel fields H = 1 2 p 2 1 r γ2 ( x 2 + y 2) + f z Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
15 In electric fields: bound states resonances Coulomb potential: V (x) const x bound state x Coulomb-Stark potential: V (x) const x + f x resonance x resonances : non-stationary or quasi-bound states Ψ(r, 0) in position space time evolution: Ψ(r, t) = e i E t Ψ(r, 0) complex energy E = E r i Γ 2 resonances with complex energy eigenvalues can be calculated by solving the Schrödinger equation using the complex rotation method Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
16 Finding exceptional points: Octagon method close to an EP: describe two related states by 2d matrix M kl = a (0) kl + a (γ) kl (γ γ 0) + a (f) kl (f f 0), γ 0, f 0 : initial guesses for the eigenvalues E 1 and E 2 of M we set: κ E 1 + E 2 = tr(m) = A + B (γ γ 0) + C (f f 0), η (E 1 E 2) 2 = tr 2 (M) 4 det(m) = D + E (γ γ 0) + F (f f 0) + G (γ γ 0) 2 + H (γ γ 0) (f f 0) + I (f f 0) 2, Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
17 Finding exceptional points: Octagon method close to an EP: describe two related states by 2d matrix M kl = a (0) kl + a (γ) kl (γ γ 0) + a (f) kl (f f 0), γ 0, f 0 : initial guesses for the eigenvalues E 1 and E 2 of M we set: A = κ 0 κ E 1 + E 2 = tr(m) = A + B (γ γ 0) + C (f f 0), η (E 1 E 2) 2 = tr 2 (M) 4 det(m) = D + E (γ γ 0) + F (f f 0) B = C = κ 3 κ 7 2h f D = η 0 E = η 1 η 5 2h γ G = η 1+η 5 2η 0 2h 2 γ H = η 2 η 4 +η 6 η 8 2h γ h f F = I = + G (γ γ 0) 2 + H (γ γ 0) (f f 0) + I (f f 0) 2, κ1 κ5 2h γ η3 η7 2h f η3 + η7 2η0 2h 2 f f γ h f h γ 2 8 Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
18 Octagon method: iterative algorithm estimate the new position (γ EP, f EP ) of EP: 0 = η (E 1 E 2 ) 2 = D + E x + F y + G x 2 + H x y + I y 2 with x (γ EP γ 0 ) and y (f EP f 0 ). Choose the correct one of four (complex) solutions. Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
19 Octagon method: iterative algorithm estimate the new position (γ EP, f EP ) of EP: 0 = η (E 1 E 2 ) 2 = D + E x + F y + G x 2 + H x y + I y 2 with x (γ EP γ 0 ) and y (f EP f 0 ). Choose the correct one of four (complex) solutions. Iterative algorithm to find position (γ EP, f EP ) of EP position estimate γ (n) EP, f (n) EP in step n is taken as the centre point of a new octagon in step n + 1 γ (n+1) 0 = γ (n) EP ; f (n+1) 0 = f (n) EP ; γ EP = lim n γ(n) 0 f EP = lim f (n) n 0 Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
20 Octagon method: example b) E E EP,i E EP,i 1 E 1,i E 2,i iterations i γ/f = 80 levels labelled by principal quantum numbers n for γ, f 0 initial parameters: γ 0 = f 0 = Re(E) = E EP,i = (E 1,i + E 2,i )/2 est. position of EP with the OM: γ EP = f EP = Re(E EP ) = Im(E EP ) = Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
21 Octagon method: verification of the EP found Im(E) [10 6 ] (7) Wη=0 = 0 η=0 = 1 W (9) Re(E) [10 3 ] Two possibilities to verify EP: 1 solve Eq. (2) step-by-step while encircling the EP high numerical effort 2 use Eqs. (3) with the known coefficients of the OM to check exchange behaviour of the resonances numerically very cheap H = 1 2 p 2 1 r γ2 ( x 2 + y 2) + f z (2) κ E 1 + E 2 = A + B (γ γ 0) + C (f f 0) η (E 1 E 2) 2 = D + E (γ γ 0) + F (f f 0) + G (γ γ 0) 2 (3) +H (γ γ 0) (f f 0) + I (f f 0) 2 Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
22 Results: more EPs a) 125 γep/fep c) 10.0 fep [10 4 ] Re(E) [10 2 ] Re(E) [10 2 ] b) 10.0 γep [10 2 ] d) 10.0 γep [10 2 ] Re(E) [10 2 ] f EP [10 4 ] γ/f = 120 EPs found (in material independent units) Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
23 Results: comparison hydrogen atom vs. Cu 2 O Hydrogen atom: reduced mass: µ m 0 dielectric constant: ε r = 1 Cuprous oxide: reduced mass: µ = 0.38m 0 dielectric constant ε r = 7.50 B [T] F [ V cm Hydrogen atom V ] Er [ev] Ei [mev] B [T] F [ cm Cu 2O ] Er [mev] Ei [µev] Energies for Cu 2 O need to be corrected by E gap = ev Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
24 Conclusion and outlook Octagon method: an improved method for finding EPs: converges to precise position of EP in parameter space and yields its precise complex energy only initial parameters of an avoided crossing are needed verification of the estimated EP without further time-consuming quantum-mechanical calculations Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
25 Conclusion and outlook Octagon method: an improved method for finding EPs: converges to precise position of EP in parameter space and yields its precise complex energy only initial parameters of an avoided crossing are needed verification of the estimated EP without further time-consuming quantum-mechanical calculations Many EPs in Rydberg systems in parallel external fields: appropriate units: results hold both for the hydrogen atom and for Rydberg excitons in Cu 2O many EPs in Cu 2O are in an experimentally accessible regime below 10 T experimental verification of our theoretical predictions: measurement of photo absorption spectra analysis by means of harmonic inversion Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
26 Conclusion and outlook Octagon method: an improved method for finding EPs: converges to precise position of EP in parameter space and yields its precise complex energy only initial parameters of an avoided crossing are needed verification of the estimated EP without further time-consuming quantum-mechanical calculations Many EPs in Rydberg systems in parallel external fields: appropriate units: results hold both for the hydrogen atom and for Rydberg excitons in Cu 2O many EPs in Cu 2O are in an experimentally accessible regime below 10 T experimental verification of our theoretical predictions: measurement of photo absorption spectra analysis by means of harmonic inversion Further information and more details M. Feldmaier, J. Main, F. Schweiner, H. Cartarius and G. Wunner. Rydberg systems in parallel electric and magnetic fields: an improved method for finding exceptional points arxiv: , in press (2016) Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
27 Complex rotation method Im(E) Im(E) exposed resonances Re(E) Re(E) bound states continuous states bound states 2θ continuous states without complex rotation complex rotation r r e iθ bound states with real energy remain unaffected by complex rotation continuum states are rotated by 2θ into the complex plane for large enough θ resonances are exposed. Positions of the resonances are independent of rotation angle. Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
28 Computation of eigenstates Introduce dilated semiparabolic coordinates: µ = µ r b = 1 r + z, b ν = ν r b = 1 b r z, ϕ = arctan ( y x b b exp(iθ) induces the complex rotation of the real coordinates (µ r, ν r, ϕ). Schrödinger equation in dilated semiparabolic coordinates: ( 2H 0 4b ) 4 b8 γ 2 (µ 4 ν 2 + µ 2 ν 4 ) + b 6 f (µ 4 ν 4 ) Ψ = λ (µ 2 + ν 2 ) Ψ, λ = 1 + 2b 4 E : the generalised eigenvalue H 0 = H µ + H ν sum of two 2d harmonic oscillators: H ρ = ( 12 ρ + 12 ) ρ2, ρ = 1 ρ ρ ρ ρ m2, ρ {µ, ν} ρ2 ), Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
29 Computation of eigenstates Set up the Hamiltonian matrix in the product basis of the eigenstates of the 2d harmonic oscillator Ψ Nµ,N ν,m(µ, ν, ϕ) = N µ, N ν, m = N µ, m N ν, m N µ! N ν! (N µ + m )! (N ν + m )! 2 π f N µ,m(µ) f Nν,m(ν) e im ϕ, f Nρ,m(ρ) = e ρ2 2 ρ m L m N ρ (ρ 2 ) for ρ {µ, ν}, Wave function of a state with (complex) energy E i Ψ i (µ, ν, ϕ) = c i,nµ,n ν,m Ψ Nµ,N ν,m(µ, ν, ϕ), N µ,n ν,m with the expansion coefficients c i,nµ,n ν,m of the associated eigenvector obtained from the diagonalization of the Hamiltonian matrix in the oscillator basis. Günter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
30 Motivation Rydberg excitons of Cu2 O with a magnetic field added: Energy (ev) Magnetic Field (T) et al. Quantum chaos of up Rydberg excitons. Preprint (2016) Figure 1: Transmission spectrum of the Cu2 O yellowaßmann series measured in magnetic fields to 7 T starting from the state with principal quantum number n=4. The red dashed line marks the band gap at ev. Gu nter Wunner (ITP1) Rydberg systems in parallel electric and magnetic fields Prague, 6 June / 20
arxiv: v3 [quant-ph] 19 May 2016
![arxiv: v3 [quant-ph] 19 May 2016 arxiv: v3 [quant-ph] 19 May 2016](/thumbs/95/126121244.jpg) arxiv:1602.00909v3 [quant-ph] 19 May 2016 Rydberg systems in parallel electric and magnetic fields: an improved method for finding exceptional points Matthias Feldmaier, Jörg Main, Frank Schweiner, Holger
arxiv:1602.00909v3 [quant-ph] 19 May 2016 Rydberg systems in parallel electric and magnetic fields: an improved method for finding exceptional points Matthias Feldmaier, Jörg Main, Frank Schweiner, Holger
arxiv: v2 [quant-ph] 11 Feb 2016
![arxiv: v2 [quant-ph] 11 Feb 2016 arxiv: v2 [quant-ph] 11 Feb 2016](/thumbs/91/106423511.jpg) arxiv:1602.00909v2 [quant-ph] 11 Feb 2016 Rydberg systems in parallel electric and magnetic fields: an improved method for finding exceptional points Matthias Feldmaier, Jörg Main, Frank Schweiner, Holger
arxiv:1602.00909v2 [quant-ph] 11 Feb 2016 Rydberg systems in parallel electric and magnetic fields: an improved method for finding exceptional points Matthias Feldmaier, Jörg Main, Frank Schweiner, Holger
2.4. Quantum Mechanical description of hydrogen atom
 2.4. Quantum Mechanical description of hydrogen atom Atomic units Quantity Atomic unit SI Conversion Ang. mom. h [J s] h = 1, 05459 10 34 Js Mass m e [kg] m e = 9, 1094 10 31 kg Charge e [C] e = 1, 6022
2.4. Quantum Mechanical description of hydrogen atom Atomic units Quantity Atomic unit SI Conversion Ang. mom. h [J s] h = 1, 05459 10 34 Js Mass m e [kg] m e = 9, 1094 10 31 kg Charge e [C] e = 1, 6022
Electronic Structure of Atoms. Chapter 6
 Electronic Structure of Atoms Chapter 6 Electronic Structure of Atoms 1. The Wave Nature of Light All waves have: a) characteristic wavelength, λ b) amplitude, A Electronic Structure of Atoms 1. The Wave
Electronic Structure of Atoms Chapter 6 Electronic Structure of Atoms 1. The Wave Nature of Light All waves have: a) characteristic wavelength, λ b) amplitude, A Electronic Structure of Atoms 1. The Wave
Quantum Mechanics & Atomic Structure (Chapter 11)
 Quantum Mechanics & Atomic Structure (Chapter 11) Quantum mechanics: Microscopic theory of light & matter at molecular scale and smaller. Atoms and radiation (light) have both wave-like and particlelike
Quantum Mechanics & Atomic Structure (Chapter 11) Quantum mechanics: Microscopic theory of light & matter at molecular scale and smaller. Atoms and radiation (light) have both wave-like and particlelike
Physics of Semiconductors (Problems for report)
 Physics of Semiconductors (Problems for report) Shingo Katsumoto Institute for Solid State Physics, University of Tokyo July, 0 Choose two from the following eight problems and solve them. I. Fundamentals
Physics of Semiconductors (Problems for report) Shingo Katsumoto Institute for Solid State Physics, University of Tokyo July, 0 Choose two from the following eight problems and solve them. I. Fundamentals
where n = (an integer) =
 5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
Chemistry 120A 2nd Midterm. 1. (36 pts) For this question, recall the energy levels of the Hydrogenic Hamiltonian (1-electron):
 April 6th, 24 Chemistry 2A 2nd Midterm. (36 pts) For this question, recall the energy levels of the Hydrogenic Hamiltonian (-electron): E n = m e Z 2 e 4 /2 2 n 2 = E Z 2 /n 2, n =, 2, 3,... where Ze is
April 6th, 24 Chemistry 2A 2nd Midterm. (36 pts) For this question, recall the energy levels of the Hydrogenic Hamiltonian (-electron): E n = m e Z 2 e 4 /2 2 n 2 = E Z 2 /n 2, n =, 2, 3,... where Ze is
The 3 dimensional Schrödinger Equation
 Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
PHY4604 Introduction to Quantum Mechanics Fall 2004 Final Exam SOLUTIONS December 17, 2004, 7:30 a.m.- 9:30 a.m.
 PHY464 Introduction to Quantum Mechanics Fall 4 Final Eam SOLUTIONS December 7, 4, 7:3 a.m.- 9:3 a.m. No other materials allowed. If you can t do one part of a problem, solve subsequent parts in terms
PHY464 Introduction to Quantum Mechanics Fall 4 Final Eam SOLUTIONS December 7, 4, 7:3 a.m.- 9:3 a.m. No other materials allowed. If you can t do one part of a problem, solve subsequent parts in terms
Lecture contents. Burstein shift Excitons Interband transitions in quantum wells Quantum confined Stark effect. NNSE 618 Lecture #15
 1 Lecture contents Burstein shift Excitons Interband transitions in quantum wells Quantum confined Stark effect Absorption edges in semiconductors Offset corresponds to bandgap Abs. coefficient is orders
1 Lecture contents Burstein shift Excitons Interband transitions in quantum wells Quantum confined Stark effect Absorption edges in semiconductors Offset corresponds to bandgap Abs. coefficient is orders
( ( ; R H = 109,677 cm -1
 CHAPTER 9 Atomic Structure and Spectra I. The Hydrogenic Atoms (one electron species). H, He +1, Li 2+, A. Clues from Line Spectra. Reminder: fundamental equations of spectroscopy: ε Photon = hν relation
CHAPTER 9 Atomic Structure and Spectra I. The Hydrogenic Atoms (one electron species). H, He +1, Li 2+, A. Clues from Line Spectra. Reminder: fundamental equations of spectroscopy: ε Photon = hν relation
Outline Spherical symmetry Free particle Coulomb problem Keywords and References. Central potentials. Sourendu Gupta. TIFR, Mumbai, India
 Central potentials Sourendu Gupta TIFR, Mumbai, India Quantum Mechanics 1 2013 3 October, 2013 Outline 1 Outline 2 Rotationally invariant potentials 3 The free particle 4 The Coulomb problem 5 Keywords
Central potentials Sourendu Gupta TIFR, Mumbai, India Quantum Mechanics 1 2013 3 October, 2013 Outline 1 Outline 2 Rotationally invariant potentials 3 The free particle 4 The Coulomb problem 5 Keywords
Large quasiparticles known as Rydberg excitons have been detected in a natural crystal of
 Solid-state physics An historic experiment redesigned Large quasiparticles known as Rydberg excitons have been detected in a natural crystal of copper oxide found at the Tsumeb mine in Namibia. The result
Solid-state physics An historic experiment redesigned Large quasiparticles known as Rydberg excitons have been detected in a natural crystal of copper oxide found at the Tsumeb mine in Namibia. The result
Problem 1: A 3-D Spherical Well(10 Points)
 Problem : A 3-D Spherical Well( Points) For this problem, consider a particle of mass m in a three-dimensional spherical potential well, V (r), given as, V = r a/2 V = W r > a/2. with W >. All of the following
Problem : A 3-D Spherical Well( Points) For this problem, consider a particle of mass m in a three-dimensional spherical potential well, V (r), given as, V = r a/2 V = W r > a/2. with W >. All of the following
Lecture 4 Quantum mechanics in more than one-dimension
 Lecture 4 Quantum mechanics in more than one-dimension Background Previously, we have addressed quantum mechanics of 1d systems and explored bound and unbound (scattering) states. Although general concepts
Lecture 4 Quantum mechanics in more than one-dimension Background Previously, we have addressed quantum mechanics of 1d systems and explored bound and unbound (scattering) states. Although general concepts
CONTENTS. vii. CHAPTER 2 Operators 15
 CHAPTER 1 Why Quantum Mechanics? 1 1.1 Newtonian Mechanics and Classical Electromagnetism 1 (a) Newtonian Mechanics 1 (b) Electromagnetism 2 1.2 Black Body Radiation 3 1.3 The Heat Capacity of Solids and
CHAPTER 1 Why Quantum Mechanics? 1 1.1 Newtonian Mechanics and Classical Electromagnetism 1 (a) Newtonian Mechanics 1 (b) Electromagnetism 2 1.2 Black Body Radiation 3 1.3 The Heat Capacity of Solids and
ψ s a ˆn a s b ˆn b ψ Hint: Because the state is spherically symmetric the answer can depend only on the angle between the two directions.
 1. Quantum Mechanics (Fall 2004) Two spin-half particles are in a state with total spin zero. Let ˆn a and ˆn b be unit vectors in two arbitrary directions. Calculate the expectation value of the product
1. Quantum Mechanics (Fall 2004) Two spin-half particles are in a state with total spin zero. Let ˆn a and ˆn b be unit vectors in two arbitrary directions. Calculate the expectation value of the product
Quantum Physics in the Nanoworld
 Hans Lüth Quantum Physics in the Nanoworld Schrödinger's Cat and the Dwarfs 4) Springer Contents 1 Introduction 1 1.1 General and Historical Remarks 1 1.2 Importance for Science and Technology 3 1.3 Philosophical
Hans Lüth Quantum Physics in the Nanoworld Schrödinger's Cat and the Dwarfs 4) Springer Contents 1 Introduction 1 1.1 General and Historical Remarks 1 1.2 Importance for Science and Technology 3 1.3 Philosophical
Magneto-Excitons in Semiconductor Quantum Rings
 phys. stat. sol. (a) 190, No. 3, 781 785 (2002) Magneto-Excitons in Semiconductor Quantum Rings I. Galbraith 1 ), F. J. Braid, and R. J. Warburton Department of Physics, Heriot-Watt University, Edinburgh,
phys. stat. sol. (a) 190, No. 3, 781 785 (2002) Magneto-Excitons in Semiconductor Quantum Rings I. Galbraith 1 ), F. J. Braid, and R. J. Warburton Department of Physics, Heriot-Watt University, Edinburgh,
Lecture #13 1. Incorporating a vector potential into the Hamiltonian 2. Spin postulates 3. Description of spin states 4. Identical particles in
 Lecture #3. Incorporating a vector potential into the Hamiltonian. Spin postulates 3. Description of spin states 4. Identical particles in classical and QM 5. Exchange degeneracy - the fundamental problem
Lecture #3. Incorporating a vector potential into the Hamiltonian. Spin postulates 3. Description of spin states 4. Identical particles in classical and QM 5. Exchange degeneracy - the fundamental problem
The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of hydrogen atom.
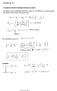 Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
26 Group Theory Basics
 26 Group Theory Basics 1. Reference: Group Theory and Quantum Mechanics by Michael Tinkham. 2. We said earlier that we will go looking for the set of operators that commute with the molecular Hamiltonian.
26 Group Theory Basics 1. Reference: Group Theory and Quantum Mechanics by Michael Tinkham. 2. We said earlier that we will go looking for the set of operators that commute with the molecular Hamiltonian.
Lecture 10. Central potential
 Lecture 10 Central potential 89 90 LECTURE 10. CENTRAL POTENTIAL 10.1 Introduction We are now ready to study a generic class of three-dimensional physical systems. They are the systems that have a central
Lecture 10 Central potential 89 90 LECTURE 10. CENTRAL POTENTIAL 10.1 Introduction We are now ready to study a generic class of three-dimensional physical systems. They are the systems that have a central
The Hydrogen Atom. Chapter 18. P. J. Grandinetti. Nov 6, Chem P. J. Grandinetti (Chem. 4300) The Hydrogen Atom Nov 6, / 41
 The Hydrogen Atom Chapter 18 P. J. Grandinetti Chem. 4300 Nov 6, 2017 P. J. Grandinetti (Chem. 4300) The Hydrogen Atom Nov 6, 2017 1 / 41 The Hydrogen Atom Hydrogen atom is simplest atomic system where
The Hydrogen Atom Chapter 18 P. J. Grandinetti Chem. 4300 Nov 6, 2017 P. J. Grandinetti (Chem. 4300) The Hydrogen Atom Nov 6, 2017 1 / 41 The Hydrogen Atom Hydrogen atom is simplest atomic system where
Sommerfeld (1920) noted energy levels of Li deduced from spectroscopy looked like H, with slight adjustment of principal quantum number:
 Spin. Historical Spectroscopy of Alkali atoms First expt. to suggest need for electron spin: observation of splitting of expected spectral lines for alkali atoms: i.e. expect one line based on analogy
Spin. Historical Spectroscopy of Alkali atoms First expt. to suggest need for electron spin: observation of splitting of expected spectral lines for alkali atoms: i.e. expect one line based on analogy
Chapter 9: Multi- Electron Atoms Ground States and X- ray Excitation
 Chapter 9: Multi- Electron Atoms Ground States and X- ray Excitation Up to now we have considered one-electron atoms. Almost all atoms are multiple-electron atoms and their description is more complicated
Chapter 9: Multi- Electron Atoms Ground States and X- ray Excitation Up to now we have considered one-electron atoms. Almost all atoms are multiple-electron atoms and their description is more complicated
e L 2m e the Bohr magneton
 e L μl = L = μb 2m with : μ B e e 2m e the Bohr magneton Classical interation of magnetic moment and B field: (Young and Freedman, Ch. 27) E = potential energy = μ i B = μbcosθ τ = torque = μ B, perpendicular
e L μl = L = μb 2m with : μ B e e 2m e the Bohr magneton Classical interation of magnetic moment and B field: (Young and Freedman, Ch. 27) E = potential energy = μ i B = μbcosθ τ = torque = μ B, perpendicular
ECE440 Nanoelectronics. Lecture 07 Atomic Orbitals
 ECE44 Nanoelectronics Lecture 7 Atomic Orbitals Atoms and atomic orbitals It is instructive to compare the simple model of a spherically symmetrical potential for r R V ( r) for r R and the simplest hydrogen
ECE44 Nanoelectronics Lecture 7 Atomic Orbitals Atoms and atomic orbitals It is instructive to compare the simple model of a spherically symmetrical potential for r R V ( r) for r R and the simplest hydrogen
Magnetism in low dimensions from first principles. Atomic magnetism. Gustav Bihlmayer. Gustav Bihlmayer
 IFF 10 p. 1 Magnetism in low dimensions from first principles Atomic magnetism Gustav Bihlmayer Institut für Festkörperforschung, Quantum Theory of Materials Gustav Bihlmayer Institut für Festkörperforschung
IFF 10 p. 1 Magnetism in low dimensions from first principles Atomic magnetism Gustav Bihlmayer Institut für Festkörperforschung, Quantum Theory of Materials Gustav Bihlmayer Institut für Festkörperforschung
Physics of Condensed Matter I
 Physics of Condensed Matter I 1100-4INZ`PC Faculty of Physics UW Jacek.Szczytko@fuw.edu.pl Summary of the lecture 1. Quantum mechanics 2. Optical transitions 3. Lasers 4. Optics 5. Molecules 6. Properties
Physics of Condensed Matter I 1100-4INZ`PC Faculty of Physics UW Jacek.Szczytko@fuw.edu.pl Summary of the lecture 1. Quantum mechanics 2. Optical transitions 3. Lasers 4. Optics 5. Molecules 6. Properties
Problem 1: Spin 1 2. particles (10 points)
 Problem 1: Spin 1 particles 1 points 1 Consider a system made up of spin 1/ particles. If one measures the spin of the particles, one can only measure spin up or spin down. The general spin state of a
Problem 1: Spin 1 particles 1 points 1 Consider a system made up of spin 1/ particles. If one measures the spin of the particles, one can only measure spin up or spin down. The general spin state of a
Quantum Physics III (8.06) Spring 2007 FINAL EXAMINATION Monday May 21, 9:00 am You have 3 hours.
 Quantum Physics III (8.06) Spring 2007 FINAL EXAMINATION Monday May 21, 9:00 am You have 3 hours. There are 10 problems, totalling 180 points. Do all problems. Answer all problems in the white books provided.
Quantum Physics III (8.06) Spring 2007 FINAL EXAMINATION Monday May 21, 9:00 am You have 3 hours. There are 10 problems, totalling 180 points. Do all problems. Answer all problems in the white books provided.
Qualifying Exam. Aug Part II. Please use blank paper for your work do not write on problems sheets!
 Qualifying Exam Aug. 2015 Part II Please use blank paper for your work do not write on problems sheets! Solve only one problem from each of the four sections Mechanics, Quantum Mechanics, Statistical Physics
Qualifying Exam Aug. 2015 Part II Please use blank paper for your work do not write on problems sheets! Solve only one problem from each of the four sections Mechanics, Quantum Mechanics, Statistical Physics
Potential energy, from Coulomb's law. Potential is spherically symmetric. Therefore, solutions must have form
 Lecture 6 Page 1 Atoms L6.P1 Review of hydrogen atom Heavy proton (put at the origin), charge e and much lighter electron, charge -e. Potential energy, from Coulomb's law Potential is spherically symmetric.
Lecture 6 Page 1 Atoms L6.P1 Review of hydrogen atom Heavy proton (put at the origin), charge e and much lighter electron, charge -e. Potential energy, from Coulomb's law Potential is spherically symmetric.
Lecture 3: Optical Properties of Insulators, Semiconductors, and Metals. 5 nm
 Metals Lecture 3: Optical Properties of Insulators, Semiconductors, and Metals 5 nm Course Info Next Week (Sept. 5 and 7) no classes First H/W is due Sept. 1 The Previous Lecture Origin frequency dependence
Metals Lecture 3: Optical Properties of Insulators, Semiconductors, and Metals 5 nm Course Info Next Week (Sept. 5 and 7) no classes First H/W is due Sept. 1 The Previous Lecture Origin frequency dependence
THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY
 THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY Warning: The mercury spectral lamps emit UV radiation. Do not stare into the lamp. Avoid exposure where possible. Introduction
THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY Warning: The mercury spectral lamps emit UV radiation. Do not stare into the lamp. Avoid exposure where possible. Introduction
Tight-Binding Model of Electronic Structures
 Tight-Binding Model of Electronic Structures Consider a collection of N atoms. The electronic structure of this system refers to its electronic wave function and the description of how it is related to
Tight-Binding Model of Electronic Structures Consider a collection of N atoms. The electronic structure of this system refers to its electronic wave function and the description of how it is related to
Atomic Structure and Atomic Spectra
 Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
Minimal Update of Solid State Physics
 Minimal Update of Solid State Physics It is expected that participants are acquainted with basics of solid state physics. Therefore here we will refresh only those aspects, which are absolutely necessary
Minimal Update of Solid State Physics It is expected that participants are acquainted with basics of solid state physics. Therefore here we will refresh only those aspects, which are absolutely necessary
14. Structure of Nuclei
 14. Structure of Nuclei Particle and Nuclear Physics Dr. Tina Potter Dr. Tina Potter 14. Structure of Nuclei 1 In this section... Magic Numbers The Nuclear Shell Model Excited States Dr. Tina Potter 14.
14. Structure of Nuclei Particle and Nuclear Physics Dr. Tina Potter Dr. Tina Potter 14. Structure of Nuclei 1 In this section... Magic Numbers The Nuclear Shell Model Excited States Dr. Tina Potter 14.
G : Quantum Mechanics II
 G5.666: Quantum Mechanics II Notes for Lecture 5 I. REPRESENTING STATES IN THE FULL HILBERT SPACE Given a representation of the states that span the spin Hilbert space, we now need to consider the problem
G5.666: Quantum Mechanics II Notes for Lecture 5 I. REPRESENTING STATES IN THE FULL HILBERT SPACE Given a representation of the states that span the spin Hilbert space, we now need to consider the problem
Time-dependent density functional theory
 Time-dependent density functional theory E.K.U. Gross Max-Planck Institute for Microstructure Physics OUTLINE LECTURE I Phenomena to be described by TDDFT Some generalities on functional theories LECTURE
Time-dependent density functional theory E.K.U. Gross Max-Planck Institute for Microstructure Physics OUTLINE LECTURE I Phenomena to be described by TDDFT Some generalities on functional theories LECTURE
PHYS 3313 Section 001 Lecture # 22
 PHYS 3313 Section 001 Lecture # 22 Dr. Barry Spurlock Simple Harmonic Oscillator Barriers and Tunneling Alpha Particle Decay Schrodinger Equation on Hydrogen Atom Solutions for Schrodinger Equation for
PHYS 3313 Section 001 Lecture # 22 Dr. Barry Spurlock Simple Harmonic Oscillator Barriers and Tunneling Alpha Particle Decay Schrodinger Equation on Hydrogen Atom Solutions for Schrodinger Equation for
Introduction to Quantum Mechanics PVK - Solutions. Nicolas Lanzetti
 Introduction to Quantum Mechanics PVK - Solutions Nicolas Lanzetti lnicolas@student.ethz.ch 1 Contents 1 The Wave Function and the Schrödinger Equation 3 1.1 Quick Checks......................................
Introduction to Quantum Mechanics PVK - Solutions Nicolas Lanzetti lnicolas@student.ethz.ch 1 Contents 1 The Wave Function and the Schrödinger Equation 3 1.1 Quick Checks......................................
Chm 331 Fall 2015, Exercise Set 4 NMR Review Problems
 Chm 331 Fall 015, Exercise Set 4 NMR Review Problems Mr. Linck Version.0. Compiled December 1, 015 at 11:04:44 4.1 Diagonal Matrix Elements for the nmr H 0 Find the diagonal matrix elements for H 0 (the
Chm 331 Fall 015, Exercise Set 4 NMR Review Problems Mr. Linck Version.0. Compiled December 1, 015 at 11:04:44 4.1 Diagonal Matrix Elements for the nmr H 0 Find the diagonal matrix elements for H 0 (the
Line spectrum (contd.) Bohr s Planetary Atom
 Line spectrum (contd.) Hydrogen shows lines in the visible region of the spectrum (red, blue-green, blue and violet). The wavelengths of these lines can be calculated by an equation proposed by J. J. Balmer:
Line spectrum (contd.) Hydrogen shows lines in the visible region of the spectrum (red, blue-green, blue and violet). The wavelengths of these lines can be calculated by an equation proposed by J. J. Balmer:
SECOND PUBLIC EXAMINATION. Honour School of Physics Part C: 4 Year Course. Honour School of Physics and Philosophy Part C C3: CONDENSED MATTER PHYSICS
 A11046W1 SECOND PUBLIC EXAMINATION Honour School of Physics Part C: 4 Year Course Honour School of Physics and Philosophy Part C C3: CONDENSED MATTER PHYSICS TRINITY TERM 2015 Wednesday, 17 June, 2.30
A11046W1 SECOND PUBLIC EXAMINATION Honour School of Physics Part C: 4 Year Course Honour School of Physics and Philosophy Part C C3: CONDENSED MATTER PHYSICS TRINITY TERM 2015 Wednesday, 17 June, 2.30
Phys 622 Problems Chapter 5
 1 Phys 622 Problems Chapter 5 Problem 1 The correct basis set of perturbation theory Consider the relativistic correction to the electron-nucleus interaction H LS = α L S, also known as the spin-orbit
1 Phys 622 Problems Chapter 5 Problem 1 The correct basis set of perturbation theory Consider the relativistic correction to the electron-nucleus interaction H LS = α L S, also known as the spin-orbit
The Hydrogen Atom. Dr. Sabry El-Taher 1. e 4. U U r
 The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
μ (vector) = magnetic dipole moment (not to be confused with the permeability μ). Magnetism Electromagnetic Fields in a Solid
 Magnetism Electromagnetic Fields in a Solid SI units cgs (Gaussian) units Total magnetic field: B = μ 0 (H + M) = μ μ 0 H B = H + 4π M = μ H Total electric field: E = 1/ε 0 (D P) = 1/εε 0 D E = D 4π P
Magnetism Electromagnetic Fields in a Solid SI units cgs (Gaussian) units Total magnetic field: B = μ 0 (H + M) = μ μ 0 H B = H + 4π M = μ H Total electric field: E = 1/ε 0 (D P) = 1/εε 0 D E = D 4π P
Preliminary Quantum Questions
 Preliminary Quantum Questions Thomas Ouldridge October 01 1. Certain quantities that appear in the theory of hydrogen have wider application in atomic physics: the Bohr radius a 0, the Rydberg constant
Preliminary Quantum Questions Thomas Ouldridge October 01 1. Certain quantities that appear in the theory of hydrogen have wider application in atomic physics: the Bohr radius a 0, the Rydberg constant
PH 451/551 Quantum Mechanics Capstone Winter 201x
 These are the questions from the W7 exam presented as practice problems. The equation sheet is PH 45/55 Quantum Mechanics Capstone Winter x TOTAL POINTS: xx Weniger 6, time There are xx questions, for
These are the questions from the W7 exam presented as practice problems. The equation sheet is PH 45/55 Quantum Mechanics Capstone Winter x TOTAL POINTS: xx Weniger 6, time There are xx questions, for
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall Duration: 2h 30m
 Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. ------------------- Duration: 2h 30m Chapter 39 Quantum Mechanics of Atoms Units of Chapter 39 39-1 Quantum-Mechanical View of Atoms 39-2
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. ------------------- Duration: 2h 30m Chapter 39 Quantum Mechanics of Atoms Units of Chapter 39 39-1 Quantum-Mechanical View of Atoms 39-2
The Quantum Heisenberg Ferromagnet
 The Quantum Heisenberg Ferromagnet Soon after Schrödinger discovered the wave equation of quantum mechanics, Heisenberg and Dirac developed the first successful quantum theory of ferromagnetism W. Heisenberg,
The Quantum Heisenberg Ferromagnet Soon after Schrödinger discovered the wave equation of quantum mechanics, Heisenberg and Dirac developed the first successful quantum theory of ferromagnetism W. Heisenberg,
PHYS852 Quantum Mechanics II, Spring 2010 HOMEWORK ASSIGNMENT 8: Solutions. Topics covered: hydrogen fine structure
 PHYS85 Quantum Mechanics II, Spring HOMEWORK ASSIGNMENT 8: Solutions Topics covered: hydrogen fine structure. [ pts] Let the Hamiltonian H depend on the parameter λ, so that H = H(λ). The eigenstates and
PHYS85 Quantum Mechanics II, Spring HOMEWORK ASSIGNMENT 8: Solutions Topics covered: hydrogen fine structure. [ pts] Let the Hamiltonian H depend on the parameter λ, so that H = H(λ). The eigenstates and
Final Exam. Tuesday, May 8, Starting at 8:30 a.m., Hoyt Hall.
 Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. Summary of Chapter 38 In Quantum Mechanics particles are represented by wave functions Ψ. The absolute square of the wave function Ψ 2
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. Summary of Chapter 38 In Quantum Mechanics particles are represented by wave functions Ψ. The absolute square of the wave function Ψ 2
Electron States of Diatomic Molecules
 IISER Pune March 2018 Hamiltonian for a Diatomic Molecule The hamiltonian for a diatomic molecule can be considered to be made up of three terms Ĥ = ˆT N + ˆT el + ˆV where ˆT N is the kinetic energy operator
IISER Pune March 2018 Hamiltonian for a Diatomic Molecule The hamiltonian for a diatomic molecule can be considered to be made up of three terms Ĥ = ˆT N + ˆT el + ˆV where ˆT N is the kinetic energy operator
The Central Force Problem: Hydrogen Atom
 The Central Force Problem: Hydrogen Atom B. Ramachandran Separation of Variables The Schrödinger equation for an atomic system with Z protons in the nucleus and one electron outside is h µ Ze ψ = Eψ, r
The Central Force Problem: Hydrogen Atom B. Ramachandran Separation of Variables The Schrödinger equation for an atomic system with Z protons in the nucleus and one electron outside is h µ Ze ψ = Eψ, r
Lecture 5. Hartree-Fock Theory. WS2010/11: Introduction to Nuclear and Particle Physics
 Lecture 5 Hartree-Fock Theory WS2010/11: Introduction to Nuclear and Particle Physics Particle-number representation: General formalism The simplest starting point for a many-body state is a system of
Lecture 5 Hartree-Fock Theory WS2010/11: Introduction to Nuclear and Particle Physics Particle-number representation: General formalism The simplest starting point for a many-body state is a system of
Chemistry 3502/4502. Final Exam Part I. May 14, 2005
 Chemistry 3502/4502 Final Exam Part I May 14, 2005 1. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle (e) The
Chemistry 3502/4502 Final Exam Part I May 14, 2005 1. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle (e) The
Terms to Know. 10.Angular quantum number 11.Magnetic quantum number 12.Spin quantum number
 Terms to Know 1. Photon 2. Atomic emission spectrum 3. Ground state 4. Atomic orbital 5. Aufbau principle 6. Pauli exclusion principle 7. Hunds rule 8. Electron configuration 9. Principle quantum number
Terms to Know 1. Photon 2. Atomic emission spectrum 3. Ground state 4. Atomic orbital 5. Aufbau principle 6. Pauli exclusion principle 7. Hunds rule 8. Electron configuration 9. Principle quantum number
Chemistry 3502/4502. Final Exam Part I. May 14, 2005
 Advocacy chit Chemistry 350/450 Final Exam Part I May 4, 005. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle
Advocacy chit Chemistry 350/450 Final Exam Part I May 4, 005. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle
( ). Expanding the square and keeping in mind that
 One-electron atom in a Magnetic Field When the atom is in a magnetic field the magnetic moment of the electron due to its orbital motion and its spin interacts with the field and the Schrodinger Hamiltonian
One-electron atom in a Magnetic Field When the atom is in a magnetic field the magnetic moment of the electron due to its orbital motion and its spin interacts with the field and the Schrodinger Hamiltonian
Chapter 2 Approximation Methods Can be Used When Exact Solutions to the Schrödinger Equation Can Not be Found.
 Chapter 2 Approximation Methods Can be Used When Exact Solutions to the Schrödinger Equation Can Not be Found. In applying quantum mechanics to 'real' chemical problems, one is usually faced with a Schrödinger
Chapter 2 Approximation Methods Can be Used When Exact Solutions to the Schrödinger Equation Can Not be Found. In applying quantum mechanics to 'real' chemical problems, one is usually faced with a Schrödinger
Degeneracy & in particular to Hydrogen atom
 Degeneracy & in particular to Hydrogen atom In quantum mechanics, an energy level is said to be degenerate if it corresponds to two or more different measurable states of a quantum system. Conversely,
Degeneracy & in particular to Hydrogen atom In quantum mechanics, an energy level is said to be degenerate if it corresponds to two or more different measurable states of a quantum system. Conversely,
Rotations in Quantum Mechanics
 Rotations in Quantum Mechanics We have seen that physical transformations are represented in quantum mechanics by unitary operators acting on the Hilbert space. In this section, we ll think about the specific
Rotations in Quantum Mechanics We have seen that physical transformations are represented in quantum mechanics by unitary operators acting on the Hilbert space. In this section, we ll think about the specific
 Math Questions for the 2011 PhD Qualifier Exam 1. Evaluate the following definite integral 3" 4 where! ( x) is the Dirac! - function. # " 4 [ ( )] dx x 2! cos x 2. Consider the differential equation dx
Math Questions for the 2011 PhD Qualifier Exam 1. Evaluate the following definite integral 3" 4 where! ( x) is the Dirac! - function. # " 4 [ ( )] dx x 2! cos x 2. Consider the differential equation dx
Mathematical Tripos Part IB Michaelmas Term Example Sheet 1. Values of some physical constants are given on the supplementary sheet
 Mathematical Tripos Part IB Michaelmas Term 2015 Quantum Mechanics Dr. J.M. Evans Example Sheet 1 Values of some physical constants are given on the supplementary sheet 1. Whenasampleofpotassiumisilluminatedwithlightofwavelength3
Mathematical Tripos Part IB Michaelmas Term 2015 Quantum Mechanics Dr. J.M. Evans Example Sheet 1 Values of some physical constants are given on the supplementary sheet 1. Whenasampleofpotassiumisilluminatedwithlightofwavelength3
Review of Optical Properties of Materials
 Review of Optical Properties of Materials Review of optics Absorption in semiconductors: qualitative discussion Derivation of Optical Absorption Coefficient in Direct Semiconductors Photons When dealing
Review of Optical Properties of Materials Review of optics Absorption in semiconductors: qualitative discussion Derivation of Optical Absorption Coefficient in Direct Semiconductors Photons When dealing
3: Many electrons. Orbital symmetries. l =2 1. m l
 3: Many electrons Orbital symmetries Atomic orbitals are labelled according to the principal quantum number, n, and the orbital angular momentum quantum number, l. Electrons in a diatomic molecule experience
3: Many electrons Orbital symmetries Atomic orbitals are labelled according to the principal quantum number, n, and the orbital angular momentum quantum number, l. Electrons in a diatomic molecule experience
Electronic structure of correlated electron systems. Lecture 2
 Electronic structure of correlated electron systems Lecture 2 Band Structure approach vs atomic Band structure Delocalized Bloch states Fill up states with electrons starting from the lowest energy No
Electronic structure of correlated electron systems Lecture 2 Band Structure approach vs atomic Band structure Delocalized Bloch states Fill up states with electrons starting from the lowest energy No
The Postulates of Quantum Mechanics Common operators in QM: Potential Energy. Often depends on position operator: Kinetic Energy 1-D case: 3-D case
 The Postulates of Quantum Mechanics Common operators in QM: Potential Energy Often depends on position operator: Kinetic Energy 1-D case: 3-D case Time Total energy = Hamiltonian To find out about the
The Postulates of Quantum Mechanics Common operators in QM: Potential Energy Often depends on position operator: Kinetic Energy 1-D case: 3-D case Time Total energy = Hamiltonian To find out about the
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/3/7/e1700704/dc1 Supplementary Materials for Giant Rashba splitting in 2D organic-inorganic halide perovskites measured by transient spectroscopies Yaxin Zhai,
advances.sciencemag.org/cgi/content/full/3/7/e1700704/dc1 Supplementary Materials for Giant Rashba splitting in 2D organic-inorganic halide perovskites measured by transient spectroscopies Yaxin Zhai,
Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model
 Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model Laser operation Simplified energy conversion processes in a laser medium:
Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model Laser operation Simplified energy conversion processes in a laser medium:
Preliminary Examination - Day 1 Thursday, August 9, 2018
 UNL - Department of Physics and Astronomy Preliminary Examination - Day Thursday, August 9, 8 This test covers the topics of Thermodynamics and Statistical Mechanics (Topic ) and Quantum Mechanics (Topic
UNL - Department of Physics and Astronomy Preliminary Examination - Day Thursday, August 9, 8 This test covers the topics of Thermodynamics and Statistical Mechanics (Topic ) and Quantum Mechanics (Topic
Electron Arrangement - Part 1
 Brad Collins Electron Arrangement - Part 1 Chapter 8 Some images Copyright The McGraw-Hill Companies, Inc. Properties of Waves Wavelength (λ) is the distance between identical points on successive waves.
Brad Collins Electron Arrangement - Part 1 Chapter 8 Some images Copyright The McGraw-Hill Companies, Inc. Properties of Waves Wavelength (λ) is the distance between identical points on successive waves.
1 Supplementary Figure
 Supplementary Figure Tunneling conductance ns.5..5..5 a n =... B = T B = T. - -5 - -5 5 Sample bias mv E n mev 5-5 - -5 5-5 - -5 4 n 8 4 8 nb / T / b T T 9T 8T 7T 6T 5T 4T Figure S: Landau-level spectra
Supplementary Figure Tunneling conductance ns.5..5..5 a n =... B = T B = T. - -5 - -5 5 Sample bias mv E n mev 5-5 - -5 5-5 - -5 4 n 8 4 8 nb / T / b T T 9T 8T 7T 6T 5T 4T Figure S: Landau-level spectra
arxiv: v2 [cond-mat.mtrl-sci] 17 Jul 2018
![arxiv: v2 [cond-mat.mtrl-sci] 17 Jul 2018 arxiv: v2 [cond-mat.mtrl-sci] 17 Jul 2018](/thumbs/92/107902302.jpg) Magneto-Stark-effect of yellow excitons in cuprous oxide Patric Rommel, Frank Schweiner, and Jörg Main Institut für Theoretische Physik 1, Universität Stuttgart, 70550 Stuttgart, Germany Julian Heckötter,
Magneto-Stark-effect of yellow excitons in cuprous oxide Patric Rommel, Frank Schweiner, and Jörg Main Institut für Theoretische Physik 1, Universität Stuttgart, 70550 Stuttgart, Germany Julian Heckötter,
Quantum Theory of Angular Momentum and Atomic Structure
 Quantum Theory of Angular Momentum and Atomic Structure VBS/MRC Angular Momentum 0 Motivation...the questions Whence the periodic table? Concepts in Materials Science I VBS/MRC Angular Momentum 1 Motivation...the
Quantum Theory of Angular Momentum and Atomic Structure VBS/MRC Angular Momentum 0 Motivation...the questions Whence the periodic table? Concepts in Materials Science I VBS/MRC Angular Momentum 1 Motivation...the
WEEK 2: 4 SEP THRU 10 SEP; LECTURES 4-6
 Learning Objectives Energy: Light as energy Describe the wave nature of light, wavelength, and frequency using the equation c = λν What is meant by the particle nature of light? Calculate the energy of
Learning Objectives Energy: Light as energy Describe the wave nature of light, wavelength, and frequency using the equation c = λν What is meant by the particle nature of light? Calculate the energy of
Problems and Multiple Choice Questions
 Problems and Multiple Choice Questions 1. A momentum operator in one dimension is 2. A position operator in 3 dimensions is 3. A kinetic energy operator in 1 dimension is 4. If two operator commute, a)
Problems and Multiple Choice Questions 1. A momentum operator in one dimension is 2. A position operator in 3 dimensions is 3. A kinetic energy operator in 1 dimension is 4. If two operator commute, a)
Physics 828 Problem Set 7 Due Wednesday 02/24/2010
 Physics 88 Problem Set 7 Due Wednesday /4/ 7)a)Consider the proton to be a uniformly charged sphere of radius f m Determine the correction to the s ground state energy 4 points) This is a standard problem
Physics 88 Problem Set 7 Due Wednesday /4/ 7)a)Consider the proton to be a uniformly charged sphere of radius f m Determine the correction to the s ground state energy 4 points) This is a standard problem
LECTURES ON QUANTUM MECHANICS
 LECTURES ON QUANTUM MECHANICS GORDON BAYM Unitsersity of Illinois A II I' Advanced Bock Progrant A Member of the Perseus Books Group CONTENTS Preface v Chapter 1 Photon Polarization 1 Transformation of
LECTURES ON QUANTUM MECHANICS GORDON BAYM Unitsersity of Illinois A II I' Advanced Bock Progrant A Member of the Perseus Books Group CONTENTS Preface v Chapter 1 Photon Polarization 1 Transformation of
Electron Interactions and Nanotube Fluorescence Spectroscopy C.L. Kane & E.J. Mele
 Electron Interactions and Nanotube Fluorescence Spectroscopy C.L. Kane & E.J. Mele Large radius theory of optical transitions in semiconducting nanotubes derived from low energy theory of graphene Phys.
Electron Interactions and Nanotube Fluorescence Spectroscopy C.L. Kane & E.J. Mele Large radius theory of optical transitions in semiconducting nanotubes derived from low energy theory of graphene Phys.
sin[( t 2 Home Problem Set #1 Due : September 10 (Wed), 2008
 Home Problem Set #1 Due : September 10 (Wed), 008 1. Answer the following questions related to the wave-particle duality. (a) When an electron (mass m) is moving with the velocity of υ, what is the wave
Home Problem Set #1 Due : September 10 (Wed), 008 1. Answer the following questions related to the wave-particle duality. (a) When an electron (mass m) is moving with the velocity of υ, what is the wave
Electron Configurations
 Ch08 Electron Configurations We now understand the orbital structure of atoms. Next we explore how electrons filling that structure change it. version 1.5 Nick DeMello, PhD. 2007-2016 2 Ch08 Putting Electrons
Ch08 Electron Configurations We now understand the orbital structure of atoms. Next we explore how electrons filling that structure change it. version 1.5 Nick DeMello, PhD. 2007-2016 2 Ch08 Putting Electrons
Atkins & de Paula: Atkins Physical Chemistry 9e Checklist of key ideas. Chapter 8: Quantum Theory: Techniques and Applications
 Atkins & de Paula: Atkins Physical Chemistry 9e Checklist of key ideas Chapter 8: Quantum Theory: Techniques and Applications TRANSLATIONAL MOTION wavefunction of free particle, ψ k = Ae ikx + Be ikx,
Atkins & de Paula: Atkins Physical Chemistry 9e Checklist of key ideas Chapter 8: Quantum Theory: Techniques and Applications TRANSLATIONAL MOTION wavefunction of free particle, ψ k = Ae ikx + Be ikx,
Optical Properties of Lattice Vibrations
 Optical Properties of Lattice Vibrations For a collection of classical charged Simple Harmonic Oscillators, the dielectric function is given by: Where N i is the number of oscillators with frequency ω
Optical Properties of Lattice Vibrations For a collection of classical charged Simple Harmonic Oscillators, the dielectric function is given by: Where N i is the number of oscillators with frequency ω
Quantum Mechanics Solutions
 Quantum Mechanics Solutions (a (i f A and B are Hermitian, since (AB = B A = BA, operator AB is Hermitian if and only if A and B commute So, we know that [A,B] = 0, which means that the Hilbert space H
Quantum Mechanics Solutions (a (i f A and B are Hermitian, since (AB = B A = BA, operator AB is Hermitian if and only if A and B commute So, we know that [A,B] = 0, which means that the Hilbert space H
The Overhauser Instability
 The Overhauser Instability Zoltán Radnai and Richard Needs TCM Group ESDG Talk 14th February 2007 Typeset by FoilTEX Introduction Hartree-Fock theory and Homogeneous Electron Gas Noncollinear spins and
The Overhauser Instability Zoltán Radnai and Richard Needs TCM Group ESDG Talk 14th February 2007 Typeset by FoilTEX Introduction Hartree-Fock theory and Homogeneous Electron Gas Noncollinear spins and
We now turn to our first quantum mechanical problems that represent real, as
 84 Lectures 16-17 We now turn to our first quantum mechanical problems that represent real, as opposed to idealized, systems. These problems are the structures of atoms. We will begin first with hydrogen-like
84 Lectures 16-17 We now turn to our first quantum mechanical problems that represent real, as opposed to idealized, systems. These problems are the structures of atoms. We will begin first with hydrogen-like
IV. Electronic Spectroscopy, Angular Momentum, and Magnetic Resonance
 IV. Electronic Spectroscopy, Angular Momentum, and Magnetic Resonance The foundation of electronic spectroscopy is the exact solution of the time-independent Schrodinger equation for the hydrogen atom.
IV. Electronic Spectroscopy, Angular Momentum, and Magnetic Resonance The foundation of electronic spectroscopy is the exact solution of the time-independent Schrodinger equation for the hydrogen atom.
One-electron Atom. (in spherical coordinates), where Y lm. are spherical harmonics, we arrive at the following Schrödinger equation:
 One-electron Atom The atomic orbitals of hydrogen-like atoms are solutions to the Schrödinger equation in a spherically symmetric potential. In this case, the potential term is the potential given by Coulomb's
One-electron Atom The atomic orbitals of hydrogen-like atoms are solutions to the Schrödinger equation in a spherically symmetric potential. In this case, the potential term is the potential given by Coulomb's
20 The Hydrogen Atom. Ze2 r R (20.1) H( r, R) = h2 2m 2 r h2 2M 2 R
 20 The Hydrogen Atom 1. We want to solve the time independent Schrödinger Equation for the hydrogen atom. 2. There are two particles in the system, an electron and a nucleus, and so we can write the Hamiltonian
20 The Hydrogen Atom 1. We want to solve the time independent Schrödinger Equation for the hydrogen atom. 2. There are two particles in the system, an electron and a nucleus, and so we can write the Hamiltonian
Exciton spectroscopy
 Lehrstuhl Werkstoffe der Elektrotechnik Exciton spectroscopy in wide bandgap semiconductors Lehrstuhl Werkstoffe der Elektrotechnik (WW6), Universität Erlangen-Nürnberg, Martensstr. 7, 91058 Erlangen Vortrag
Lehrstuhl Werkstoffe der Elektrotechnik Exciton spectroscopy in wide bandgap semiconductors Lehrstuhl Werkstoffe der Elektrotechnik (WW6), Universität Erlangen-Nürnberg, Martensstr. 7, 91058 Erlangen Vortrag
Molecules in Magnetic Fields
 Molecules in Magnetic Fields Trygve Helgaker Hylleraas Centre, Department of Chemistry, University of Oslo, Norway and Centre for Advanced Study at the Norwegian Academy of Science and Letters, Oslo, Norway
Molecules in Magnetic Fields Trygve Helgaker Hylleraas Centre, Department of Chemistry, University of Oslo, Norway and Centre for Advanced Study at the Norwegian Academy of Science and Letters, Oslo, Norway
0 belonging to the unperturbed Hamiltonian H 0 are known
 Time Independent Perturbation Theory D Perturbation theory is used in two qualitatively different contexts in quantum chemistry. It allows one to estimate (because perturbation theory is usually employed
Time Independent Perturbation Theory D Perturbation theory is used in two qualitatively different contexts in quantum chemistry. It allows one to estimate (because perturbation theory is usually employed
atoms and light. Chapter Goal: To understand the structure and properties of atoms.
 Quantum mechanics provides us with an understanding of atomic structure and atomic properties. Lasers are one of the most important applications of the quantummechanical properties of atoms and light.
Quantum mechanics provides us with an understanding of atomic structure and atomic properties. Lasers are one of the most important applications of the quantummechanical properties of atoms and light.
Lecture 4 Quantum mechanics in more than one-dimension
 Lecture 4 Quantum mechanics in more than one-dimension Background Previously, we have addressed quantum mechanics of 1d systems and explored bound and unbound (scattering) states. Although general concepts
Lecture 4 Quantum mechanics in more than one-dimension Background Previously, we have addressed quantum mechanics of 1d systems and explored bound and unbound (scattering) states. Although general concepts
