17.2 Thermochemical Equations
|
|
|
- Bertina Barker
- 5 years ago
- Views:
Transcription
1 17.2. Thermochemical Equations Thermochemical Equations Lesson Objectives Define enthalpy, and know the conditions under which the enthalpy change in a reaction is equal to the heat absorbed or released. Describe the principles behind calorimetry, and be able to calculate the heat absorbed or released during a process that occurs in a calorimeter. Write and solve problems with thermochemical equations. Lesson Vocabulary calorimeter calorimetry enthalpy heat of reaction thermochemical equation Check Your Understanding Recalling Prior Knowledge How is specific heat used in calculating the heat absorbed or released in a process? What relationships are shown in a balanced chemical equation? Heat is either absorbed or released during chemical reactions. In this lesson, you will learn about how heat changes are measured in the lab and how quantities of heat are represented in a chemical equation. Enthalpy Heat changes in chemical reactions are most often measured in the laboratory under conditions in which the reacting system is open to the atmosphere. In that case, the system is at a constant pressure. Enthalpy (H) is the heat content of a system at constant pressure. Chemists routinely measure changes in the enthalpy of a chemical system as reactants are converted into products. The heat that is absorbed or released by a reaction at constant pressure is the same as the enthalpy change, which is given the symbol H. Unless otherwise specified, all reactions in this text are assumed to take place at constant pressure. Therefore, heat and enthalpy change will be used interchangeably, and q = H. 516
2 Chapter 17. Thermochemistry Calorimetry Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical reaction or physical process. A calorimeter is an insulated container that is used to measure heat changes. The reactions that can most easily be analyzed in a calorimetry experiment involve only liquids or aqueous solutions. A frequently used and inexpensive calorimeter is a set of nested foam cups fitted with a lid to limit the heat exchange between the liquid in the cup and the air in the surroundings ( Figure 17.5). In a typical calorimetry experiment, specific volumes of the reactants are dispensed into separate containers, and the temperature of each is measured. They are then mixed into the calorimeter, which starts the reaction. The reactant mixture is stirred until the reaction is complete, and the temperature of the reaction is continuously monitored. FIGURE 17.5 In a simple constant-pressure calorimeter, the temperature of a waterbased reaction is monitored as the reaction takes place. The substances dissolved in the water are the system, and the water itself is the surroundings. The key to all calorimetry experiments is the assumption that there is no heat exchange between the insulated calorimeter and the room. Consider the case of a reaction taking place between aqueous reactants. The water in which the solids have been dissolved is the surroundings, while the dissolved substances are the system. The temperature change that is measured is the temperature change that is occurring in the surroundings. If the temperature of the water increases as the reaction occurs, the reaction is exothermic. Heat was released by the system into the surrounding water. An endothermic reaction absorbs heat from the surroundings, so the temperature of the water decreases as heat leaves the surroundings to enter the system. The temperature change of the water is measured in the experiment, and the specific heat of water can be used to calculate the heat absorbed by the surroundings (q surr ). q surr = m c p T In this equation, m is the mass of the water, c p is the specific heat of the water, and T is T f T i. The heat absorbed by the surroundings is equal, but opposite in sign, to the heat released by the system. Because the heat change is determined at constant pressure, the heat released by the system (q sys ) is equal to the enthalpy change ( H). q sys = H = -q surr = -(m c p T) The sign of H is positive for an endothermic reaction and negative for an exothermic reaction. Sample Problem 17.2: Calorimetry and Enthalpy Changes In an experiment, 25.0 ml of 1.00 M HCl at 25.0 C is added to 25.0 ml of 1.00 M NaOH at 25.0 C in a foam cup calorimeter. As the reaction occurs, the temperature of the solution rises to 32.0 C. Calculate the enthalpy change 517
3 17.2. Thermochemical Equations ( H) in kj for this reaction. Assume the densities of the solutions are 1.00 g/ml and that their specific heats are the same as that of pure water. Step 1: List the known quantities and plan the problem. Known c p = 4.18 J/g C V f inal = 25.0 ml ml = 50.0 ml T = 32.0 C 25.0 C = 7.0 Density = 1.00 g/ml Unknown H =? kj The volume and density can be used to find the mass of the solution after mixing. Then, calculate the change in enthalpy by using H = q sys = q surr = (m c p T). Step 2: Solve. m = 50.0 ml 1.00 g 1 ml = 50.0 g H = (m c p T) = -(50.0 g 4.18 J/g C 7.0 C) = J = -1.5 kj Step 3: Think about the result. The enthalpy change is negative because the reaction releases heat to the surroundings, causing the temperature of the water to rise. Practice Problems 1. A rock is heated and dropped into a foam cup calorimeter containing 35.0 ml of water at 21.4 C. The temperature of the water rises to 24.8 C. How many joules of heat were released by the rock? 2. A g sample of an unknown metal is heated to C. This hot metal is then placed into a foam cup calorimeter containing 50.0 g of water at C. The water and metal come to a final temperature of C. Calculate the specific heat of the metal, and use the table above ( Table 17.1) to identify it. Heats of Reaction When methane gas is combusted, heat is released, making the reaction exothermic. Specifically, the combustion of 1 mol of methane releases kilojoules of heat energy. This information can be shown as part of the balanced equation. CH 4 (g) + 2O 2 (g) CO 2 (g) + 2H 2 O(l) kj The equation tells us that 1 mol of methane combines with 2 mol of oxygen to produce 1 mol of carbon dioxide and 2 mol of water. In the process, kj is released, so it is written as a product of the reaction. A thermochemical equation is a chemical equation that includes the enthalpy change of the reaction. The process in the above thermochemical equation can be shown visually below ( Figure 17.6 (A)). 518
4 Chapter 17. Thermochemistry FIGURE 17.6 (A) As reactants are converted to products in an exothermic reaction, enthalpy is released into the surroundings. The enthalpy change of the reaction is negative. (B) As reactants are converted to products in an endothermic reaction, enthalpy is absorbed from the surroundings. The enthalpy change of the reaction is positive. In the combustion of methane example, the enthalpy change is negative because heat is being released by the system. Therefore, the overall enthalpy of the system decreases. The heat of reaction is the enthalpy change for a chemical reaction. In the case above, the heat of reaction is kj. The thermochemical reaction can also be written in this way: CH 4 (g) + 2O 2 (g) CO 2 (g) + 2H 2 O(l) H = kj Heats of reaction are typically measured in kilojoules. It is important to include the physical states of the reactants and products in a thermochemical equation, because the value of H depends on those states. Endothermic reactions absorb energy from the surroundings as the reaction occurs. When 1 mol of calcium carbonate decomposes into 1 mol of calcium oxide and 1 mol of carbon dioxide, kj of heat is absorbed. This process is shown visually above ( Figure 17.6 (B)). When heat is absorbed during a reaction, it can be written as a reactant. The thermochemical reaction is shown below. CaCO 3 (s) kj CaO(s) + CO 2 (g) The reaction is endothermic, so the sign of the enthalpy change is positive. CaCO 3 (s) CaO(s) + CO 2 (g) H = kj Stoichiometry and Thermochemical Equations Chemistry problems that involve enthalpy changes can be solved by techniques similar to stoichiometry problems. Refer again to the combustion reaction of methane. Since the reaction of 1 mol of methane releases kj, the reaction of 2 mol of methane would release kj = 1781 kj. The reaction of 0.5 mol of methane would release kj / 2 = kj. As with other stoichiometry problems, the moles of a reactant or product can be linked to mass or volume. Sample Problem 17.3: Calculating Enthalpy Changes Sulfur dioxide gas reacts with oxygen to form sulfur trioxide in an exothermic reaction according to the following thermochemical equation. 2SO 2 (g) + O 2 (g) 2SO 3 (g) kj Calculate the enthalpy change that occurs when 58.0 g of sulfur dioxide is reacted with excess oxygen. Step 1: List the known quantities and plan the problem. 519
5 17.2. Thermochemical Equations Known mass of SO 2 = 58.0 g molar mass of SO 2 = g/mol H = 198 kj for the reaction of 2 mol SO 2 Unknown H =? kj The calculation requires two steps. First, the mass of SO 2 is converted to moles. Then, mol SO 2 is multiplied by the conversion factor ( 198 kj/2 mol SO 2 ). Step 2: Solve. H = 58.0 g SO 2 1 mol SO kj = 89.6 kj g SO 2 2 mol SO 2 Step 3: Think about your result. The mass of sulfur dioxide is slightly less than 1 mol. Since 198 kj is released for every 2 mol of SO 2 that reacts, the heat released when about 1 mol reacts is one half of 198. The 89.6 kj is slightly less than half of 198. The sign of H is negative because the reaction is exothermic. Practice Problem 3. Given the reaction below for the decomposition of mercury(ii) oxide: (a) What is H when mol of HgO fully reacts? (b) A certain reaction produces 28.4 g of Hg. How much heat was absorbed in this reaction? (c) What volume of oxygen gas at STP is produced in a reaction that absorbs 432 kj of heat? Lesson Summary Enthalpy is the heat content of a system. When a chemical reaction or physical process occurs at constant pressure, the heat absorbed or released by the system is equal to the enthalpy change of the system. A calorimeter is an insulated device used in the laboratory to measure the enthalpy change during a reaction. Thermochemical equations show the heat that is either absorbed or released during a reaction. The enthalpy change ( H) for a reaction can be used as a conversion factor in solving problems. Lesson Review Questions Reviewing Concepts What experimental condition is required for the heat change in a reaction to be numerically equal to the enthalpy change ( H)?
6 Chapter 17. Thermochemistry 2. Why is a foam cup used as a calorimeter rather than a glass beaker? 3. What are some possible sources of error that would be present in an experiment where a foam cup is used as a calorimeter? 4. When 1 mol of nitrogen gas reacts with 3 mol of hydrogen gas, 2 mol of ammonia gas is produced and 92.6 kj of heat is released. Write the thermochemical equation. Problems 5. Given the following reaction for the formation of water from hydrogen and oxygen gases: What is the H for the following reactions? a. H 2 (g) + 1/2O 2 (g) H 2 O(l) b. 2H 2 O(l) 2H 2 (g) + O 2 (g) ml of M HCl is mixed with 100. ml of M NaOH in a foam cup calorimeter. The initial temperatures of both solutions are C. After the reaction occurs, the temperature rises to C. Calculate the enthalpy change for the reaction. Assume the densities of the solutions are 1.00 g/ml and the specific heat is 4.18 J/g C. 7. An ice cube is dropped into a foam cup calorimeter containing 95.0 ml of water at 20.0 C. The temperature drops to 13.7 C as the ice cube melts. How much total heat (in kj) was released by the water into the ice cube? g of lead is heated to 155 C and then placed into a foam cup calorimeter containing 70.0 ml of water at 15.2 C. Assuming no heat loss, calculate the final temperature of the water and lead. 9. Given the balanced equation below for the highly exothermic reaction of aluminum with oxygen to form aluminum oxide: a. What is the value of H when 1.00 mol of aluminum reacts? b. What is the value of H when 5.23 g of aluminum reacts? c. What is the value of H when 6.17 L of oxygen gas at STP reacts with excess aluminum? d. A certain reaction releases 6881 kj of heat. What mass of aluminum oxide was produced in the reaction? 10. Propane gas combusts according to the following equation L of propane at atm is combusted at 513. C. How much heat is released in the reaction? Assume the propane is an ideal gas. Further Reading / Supplemental Links Enthalpy, The Thermochemical Equation, Points to Consider Enthalpy changes always accompany changes in state. What are the heat of fusion and heat of vaporization, and how can they be used to solve problems? What is the heat of solution? 521
5/14/14. How can you measure the amount of heat released when a match burns?
 CHEMISTRY & YOU Chapter 7 Thermochemistry How can you measure the amount of heat released when a match burns? 7. The Flow of Energy 7.3 Heat in Changes of State 7.4 Calculating Heats of Reaction Remember:
CHEMISTRY & YOU Chapter 7 Thermochemistry How can you measure the amount of heat released when a match burns? 7. The Flow of Energy 7.3 Heat in Changes of State 7.4 Calculating Heats of Reaction Remember:
Name Class Date. As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings.
 Name Class Date Thermochemistry 17.1 The Flow of Energy As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings. Process Cause Effect endothermic
Name Class Date Thermochemistry 17.1 The Flow of Energy As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings. Process Cause Effect endothermic
Name: Thermochemistry. Practice Test C. General Chemistry Honors Chemistry
 Name: Thermochemistry C Practice Test C General Chemistry Honors Chemistry 1 Objective 1: Use the relationship between mass, specific heat, and temperature change to calculate the heat flow during a chemical
Name: Thermochemistry C Practice Test C General Chemistry Honors Chemistry 1 Objective 1: Use the relationship between mass, specific heat, and temperature change to calculate the heat flow during a chemical
Thermochemistry. Energy. 1st Law of Thermodynamics. Enthalpy / Calorimetry. Enthalpy of Formation
 THERMOCHEMISTRY Thermochemistry Energy 1st Law of Thermodynamics Enthalpy / Calorimetry Hess' Law Enthalpy of Formation The Nature of Energy Kinetic Energy and Potential Energy Kinetic energy is the energy
THERMOCHEMISTRY Thermochemistry Energy 1st Law of Thermodynamics Enthalpy / Calorimetry Hess' Law Enthalpy of Formation The Nature of Energy Kinetic Energy and Potential Energy Kinetic energy is the energy
17.4 Calculating Heats Essential Understanding Heats of reaction can be calculated when it is difficult or
 17.4 Calculating Heats of Reaction Essential Understanding Heats of reaction can be calculated when it is difficult or impossible to measure them directly. Lesson Summary Hess s Law Hess s law provides
17.4 Calculating Heats of Reaction Essential Understanding Heats of reaction can be calculated when it is difficult or impossible to measure them directly. Lesson Summary Hess s Law Hess s law provides
Name: General Chemistry Chapter 11 Thermochemistry- Heat and Chemical Change
 Name: General Chemistry Chapter 11 Thermochemistry- Heat and Chemical Change Notepack 1 Section 11.1: The Flow of Energy Heat (Pages 293 299) 1. Define the following terms: a. Thermochemistry b. Energy
Name: General Chemistry Chapter 11 Thermochemistry- Heat and Chemical Change Notepack 1 Section 11.1: The Flow of Energy Heat (Pages 293 299) 1. Define the following terms: a. Thermochemistry b. Energy
Name Date Class THE FLOW OF ENERGY HEAT AND WORK
 17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
Measuring and Expressing Enthalpy Changes. Copyright Pearson Prentice Hall. Measuring and Expressing Enthalpy Changes. Calorimetry
 Measuring and Expressing Enthalpy Changes A burning match releases heat to its surroundings in all directions. How much heat does this exothermic reaction release? You will learn to measure heat flow in
Measuring and Expressing Enthalpy Changes A burning match releases heat to its surroundings in all directions. How much heat does this exothermic reaction release? You will learn to measure heat flow in
Section 9: Thermodynamics and Energy
 Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
Name Date Class SECTION 16.1 PROPERTIES OF SOLUTIONS
 SOLUTIONS Practice Problems In your notebook, solve the following problems. SECTION 16.1 PROPERTIES OF SOLUTIONS 1. The solubility of CO 2 in water at 1.22 atm is 0.54 g/l. What is the solubility of carbon
SOLUTIONS Practice Problems In your notebook, solve the following problems. SECTION 16.1 PROPERTIES OF SOLUTIONS 1. The solubility of CO 2 in water at 1.22 atm is 0.54 g/l. What is the solubility of carbon
8.6 The Thermodynamic Standard State
 8.6 The Thermodynamic Standard State The value of H reported for a reaction depends on the number of moles of reactants...or how much matter is contained in the system C 3 H 8 (g) + 5O 2 (g) > 3CO 2 (g)
8.6 The Thermodynamic Standard State The value of H reported for a reaction depends on the number of moles of reactants...or how much matter is contained in the system C 3 H 8 (g) + 5O 2 (g) > 3CO 2 (g)
Chemistry Chapter 16. Reaction Energy
 Chemistry Reaction Energy Section 16.1.I Thermochemistry Objectives Define temperature and state the units in which it is measured. Define heat and state its units. Perform specific-heat calculations.
Chemistry Reaction Energy Section 16.1.I Thermochemistry Objectives Define temperature and state the units in which it is measured. Define heat and state its units. Perform specific-heat calculations.
Thermochemistry (chapter 5)
 Thermochemistry (chapter 5) Basic Definitions: Thermochemistry = the study of the energy changes that accompany physical and chemical changes of matter. Energy is defined as the ability to do work or the
Thermochemistry (chapter 5) Basic Definitions: Thermochemistry = the study of the energy changes that accompany physical and chemical changes of matter. Energy is defined as the ability to do work or the
Chapter 8. Thermochemistry 강의개요. 8.1 Principles of Heat Flow. 2) Magnitude of Heat Flow. 1) State Properties. Basic concepts : study of heat flow
 강의개요 Basic concepts : study of heat flow Chapter 8 Thermochemistry Calorimetry : experimental measurement of the magnitude and direction of heat flow Thermochemical Equations Copyright 2005 연세대학교이학계열일반화학및실험
강의개요 Basic concepts : study of heat flow Chapter 8 Thermochemistry Calorimetry : experimental measurement of the magnitude and direction of heat flow Thermochemical Equations Copyright 2005 연세대학교이학계열일반화학및실험
Thermochemistry. Energy (and Thermochemistry) World of Chemistry Chapter 10. Energy. Energy
 Thermochemistry Thermodynamics is the science of the relationship between heat and other forms of energy. (and Thermochemistry) World of Chemistry Chapter 10 is defined as the ability to do work or produce
Thermochemistry Thermodynamics is the science of the relationship between heat and other forms of energy. (and Thermochemistry) World of Chemistry Chapter 10 is defined as the ability to do work or produce
AP Chapter 6: Thermochemistry Name
 AP Chapter 6: Thermochemistry Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. AP Chapter 6: Thermochemistry 2 Warm-Ups (Show your work for credit)
AP Chapter 6: Thermochemistry Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. AP Chapter 6: Thermochemistry 2 Warm-Ups (Show your work for credit)
3.2 Calorimetry and Enthalpy
 3.2 Calorimetry and Enthalpy Heat Capacity Specific heat capacity (c) is the quantity of thermal energy required to raise the temperature of 1 g of a substance by 1 C. The SI units for specific heat capacity
3.2 Calorimetry and Enthalpy Heat Capacity Specific heat capacity (c) is the quantity of thermal energy required to raise the temperature of 1 g of a substance by 1 C. The SI units for specific heat capacity
Chemistry Slide 1 of 33
 Chemistry 17.2 1 of 33 17.2 Measuring and Expressing Enthalpy Changes A burning match releases heat to its surroundings in all directions. How much heat does this exothermic reaction release? You will
Chemistry 17.2 1 of 33 17.2 Measuring and Expressing Enthalpy Changes A burning match releases heat to its surroundings in all directions. How much heat does this exothermic reaction release? You will
Ch. 17 Thermochemistry
 Ch. 17 Thermochemistry 17.1 The Flow of Energy Energy Transformations Thermochemistry: study of energy changes in chemical reactions and changes in state Chemical potential energy: energy stored in bonds
Ch. 17 Thermochemistry 17.1 The Flow of Energy Energy Transformations Thermochemistry: study of energy changes in chemical reactions and changes in state Chemical potential energy: energy stored in bonds
Thermochemistry: Energy Flow and Chemical Reactions
 Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
Chapter 11 Thermochemistry Heat and Chemical Change
 Chemistry/ PEP Name: Date: Chapter 11 Thermochemistry Heat and Chemical Change Chapter 11:1 35, 57, 60, 61, 71 Section 11.1 The Flow of Energy - Heat 1. When 435 of heat is added to 3.4 g of olive oil
Chemistry/ PEP Name: Date: Chapter 11 Thermochemistry Heat and Chemical Change Chapter 11:1 35, 57, 60, 61, 71 Section 11.1 The Flow of Energy - Heat 1. When 435 of heat is added to 3.4 g of olive oil
11B, 11E Temperature and heat are related but not identical.
 Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Chapter 5 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Thermochemistry: Heat and Chemical Change
 Thermochemistry: Heat and Chemical Change 1 Heat or Thermal Energy (q) Heat is a form of energy Is heat the same as temperature? Heat flows between two objects at different temperatures. Hot Cold 2 Chemical
Thermochemistry: Heat and Chemical Change 1 Heat or Thermal Energy (q) Heat is a form of energy Is heat the same as temperature? Heat flows between two objects at different temperatures. Hot Cold 2 Chemical
Thermochemistry Enthalpy & Hess Law. Packet #35
 Thermochemistry Enthalpy & Hess Law Packet #35 Introduction I Thermochemistry, is the branch of chemistry that, investigates the amount of energy that is gained or lost during a chemical reaction. Introduction
Thermochemistry Enthalpy & Hess Law Packet #35 Introduction I Thermochemistry, is the branch of chemistry that, investigates the amount of energy that is gained or lost during a chemical reaction. Introduction
Thermochemistry. Mr.V
 Thermochemistry Mr.V Introduction to Energy changes System Surroundings Exothermic Endothermic Internal energy Enthalpy Definitions System A specified part of the universe which is under investigation
Thermochemistry Mr.V Introduction to Energy changes System Surroundings Exothermic Endothermic Internal energy Enthalpy Definitions System A specified part of the universe which is under investigation
Name Date Class THERMOCHEMISTRY
 Name Date Class 17 THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY HEAT AND WORK (pages 505 510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
Name Date Class 17 THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY HEAT AND WORK (pages 505 510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes
 Thermochemistry Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes heat flows from high to low (hot cool) endothermic reactions: absorb energy
Thermochemistry Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes heat flows from high to low (hot cool) endothermic reactions: absorb energy
I. The Nature of Energy A. Energy
 I. The Nature of Energy A. Energy is the ability to do work or produce heat. It exists in 2 forms: 1. Potential energy is energy due to the composition or position of an object. 2. Kinetic energy is energy
I. The Nature of Energy A. Energy is the ability to do work or produce heat. It exists in 2 forms: 1. Potential energy is energy due to the composition or position of an object. 2. Kinetic energy is energy
Chapter 17 Thermochemistry
 Chapter 17 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Chapter 17 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Energy Transformations
 Thermochemistry Energy Transformations Thermochemistry - concerned with heat changes that occur during chemical reactions Energy - capacity for doing work or supplying heat weightless, odorless, tasteless
Thermochemistry Energy Transformations Thermochemistry - concerned with heat changes that occur during chemical reactions Energy - capacity for doing work or supplying heat weightless, odorless, tasteless
Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat.
 CHEM134- F18 Dr. Al- Qaisi Chapter 06: Thermodynamics Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat. Energy is anything that has the capacity
CHEM134- F18 Dr. Al- Qaisi Chapter 06: Thermodynamics Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat. Energy is anything that has the capacity
The following gas laws describes an ideal gas, where
 Alief ISD Chemistry STAAR Review Reporting Category 4: Gases and Thermochemistry C.9.A Describe and calculate the relations between volume, pressure, number of moles, and temperature for an ideal gas as
Alief ISD Chemistry STAAR Review Reporting Category 4: Gases and Thermochemistry C.9.A Describe and calculate the relations between volume, pressure, number of moles, and temperature for an ideal gas as
Ch 9 Practice Problems
 Ch 9 Practice Problems 1. One mole of an ideal gas is expanded from a volume of 1.50 L to a volume of 10.18 L against a constant external pressure of 1.03 atm. Calculate the work. (1 L atm = 101.3 J) A)
Ch 9 Practice Problems 1. One mole of an ideal gas is expanded from a volume of 1.50 L to a volume of 10.18 L against a constant external pressure of 1.03 atm. Calculate the work. (1 L atm = 101.3 J) A)
Thermochemistry (chapter 5)
 Thermochemistry (chapter 5) Is the study of the energy changes that accompany physical and chemical changes. Energy is defined as the ability to do work or the capacity to produce change. The forms of
Thermochemistry (chapter 5) Is the study of the energy changes that accompany physical and chemical changes. Energy is defined as the ability to do work or the capacity to produce change. The forms of
Thermochemistry. The study of the ENERGY CHANGES that accompany changes in matter. 3 Ways: Monday, February 3, 2014
 Thermochemistry The study of the ENERGY CHANGES that accompany changes in matter 3 Ways: 1 Thermodynamics FIRST LAW OF THERMODYNAMICS the total amount of energy in the universe is constant (conservation
Thermochemistry The study of the ENERGY CHANGES that accompany changes in matter 3 Ways: 1 Thermodynamics FIRST LAW OF THERMODYNAMICS the total amount of energy in the universe is constant (conservation
Enthalpies of Reaction
 Enthalpies of Reaction Enthalpy is an extensive property Magnitude of H is directly related to the amount of reactant used up in a process. CH 4 (g) + 2O 2 (g) CO 2 (g) + 2H 2 O(l) H = 890 kj 2CH 4 (g)
Enthalpies of Reaction Enthalpy is an extensive property Magnitude of H is directly related to the amount of reactant used up in a process. CH 4 (g) + 2O 2 (g) CO 2 (g) + 2H 2 O(l) H = 890 kj 2CH 4 (g)
Introduction to Thermochemistry. Thermochemistry Unit. Definition. Terminology. Terminology. Terminology 07/04/2016. Chemistry 30
 Thermochemistry Unit Introduction to Thermochemistry Chemistry 30 Definition Thermochemistry is the branch of chemistry concerned with the heat produced and used in chemical reactions. Most of thermochemistry
Thermochemistry Unit Introduction to Thermochemistry Chemistry 30 Definition Thermochemistry is the branch of chemistry concerned with the heat produced and used in chemical reactions. Most of thermochemistry
Learning Check. How much heat, q, is required to raise the temperature of 1000 kg of iron and 1000 kg of water from 25 C to 75 C?
 Learning Check q = c * m * ΔT How much heat, q, is required to raise the temperature of 1000 kg of iron and 1000 kg of water from 25 C to 75 C? (c water =4.184 J/ C g, c iron =0.450 J/ C g) q Fe = 0.450
Learning Check q = c * m * ΔT How much heat, q, is required to raise the temperature of 1000 kg of iron and 1000 kg of water from 25 C to 75 C? (c water =4.184 J/ C g, c iron =0.450 J/ C g) q Fe = 0.450
Section 1 - Thermochemistry
 Reaction Energy Section 1 - Thermochemistry Virtually every chemical reaction is accompanied by a change in energy. Chemical reactions usually absorb or release energy as heat. You learned in Chapter 12
Reaction Energy Section 1 - Thermochemistry Virtually every chemical reaction is accompanied by a change in energy. Chemical reactions usually absorb or release energy as heat. You learned in Chapter 12
THERMOCHEMISTRY CHAPTER 11
 THERMOCHEMISTRY CHAPTER 11 ENERGY AND HEAT nthermochemistry: The study of the energy changes that accompany chemical reactions and changes in the physical states of matter. ENERGY AND HEAT nwork: Energy
THERMOCHEMISTRY CHAPTER 11 ENERGY AND HEAT nthermochemistry: The study of the energy changes that accompany chemical reactions and changes in the physical states of matter. ENERGY AND HEAT nwork: Energy
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Energy -Very much a chemistry topic Every chemical change has an accompanying change of. Combustion of fossil fuels The discharging a battery Metabolism of foods If we are to
Chapter 5 Thermochemistry Energy -Very much a chemistry topic Every chemical change has an accompanying change of. Combustion of fossil fuels The discharging a battery Metabolism of foods If we are to
Chapter 6. Thermochemistry
 Chapter 6. Thermochemistry 1 1. Terms to Know: thermodynamics thermochemistry energy kinetic energy potential energy heat heat vs. temperature work work of expanding gases work of expanding gases under
Chapter 6. Thermochemistry 1 1. Terms to Know: thermodynamics thermochemistry energy kinetic energy potential energy heat heat vs. temperature work work of expanding gases work of expanding gases under
Thermochemistry. Chapter 6. Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
 Thermochemistry Chapter 6 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Energy is the capacity to do work. Radiant energy comes from the sun and is earth s
Thermochemistry Chapter 6 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Energy is the capacity to do work. Radiant energy comes from the sun and is earth s
Chemistry 30: Thermochemistry. Practice Problems
 Name: Period: Chemistry 30: Thermochemistry Practice Problems Date: Heat and Temperature 1. Pretend you are doing a scientific study on the planet Earth. a. Name three things in the system you are studying.
Name: Period: Chemistry 30: Thermochemistry Practice Problems Date: Heat and Temperature 1. Pretend you are doing a scientific study on the planet Earth. a. Name three things in the system you are studying.
Chapter 5 Practice Multiple Choice & Free
 Name Response 1. A system has an increase in internal energy, E, of 40 kj. If 20 kj of work, w, is done on the system, what is the heat change, q? a) +60 kj d) -20 kj b) +40 kj e) -60 kj c) +20 kj 2. Which
Name Response 1. A system has an increase in internal energy, E, of 40 kj. If 20 kj of work, w, is done on the system, what is the heat change, q? a) +60 kj d) -20 kj b) +40 kj e) -60 kj c) +20 kj 2. Which
Chapter 8. Thermochemistry
 Chapter 8 Thermochemistry Copyright 2001 by Harcourt, Inc. All rights reserved. Requests for permission to make copies of any part of the work should be mailed to the following address: Permissions Department,
Chapter 8 Thermochemistry Copyright 2001 by Harcourt, Inc. All rights reserved. Requests for permission to make copies of any part of the work should be mailed to the following address: Permissions Department,
Energetics. These processes involve energy exchanges between the reacting system and its surroundings.
 Energetics Chemical reactions involve: the breaking of bonds between atoms the making of new bonds between atoms These processes involve energy exchanges between the reacting system and its surroundings.
Energetics Chemical reactions involve: the breaking of bonds between atoms the making of new bonds between atoms These processes involve energy exchanges between the reacting system and its surroundings.
CHAPTER 17 Thermochemistry
 CHAPTER 17 Thermochemistry Thermochemistry The study of the heat changes that occur during chemical reactions and physical changes of state. Chemical Change: new substances created during chemical reaction
CHAPTER 17 Thermochemistry Thermochemistry The study of the heat changes that occur during chemical reactions and physical changes of state. Chemical Change: new substances created during chemical reaction
IB Chemistry Solutions Gasses and Energy
 Solutions A solution is a homogeneous mixture it looks like one substance. An aqueous solution will be a clear mixture with only one visible phase. Be careful with the definitions of clear and colourless.
Solutions A solution is a homogeneous mixture it looks like one substance. An aqueous solution will be a clear mixture with only one visible phase. Be careful with the definitions of clear and colourless.
Chapter 5 Principles of Chemical Reactivity: Energy and Chemical Reactions
 Chapter 5 Principles of Chemical Reactivity: Energy and Chemical Reactions Jeffrey Mack California State University, Sacramento Energy & Chemistry Questions that need to be addressed: How do we measure
Chapter 5 Principles of Chemical Reactivity: Energy and Chemical Reactions Jeffrey Mack California State University, Sacramento Energy & Chemistry Questions that need to be addressed: How do we measure
This reaction is ENDOTHERMIC. Energy is being transferred from the room/flask/etc. (the SURROUNDINGS) to the reaction itself (the SYSTEM).
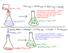 151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
Energy and Chemical Change
 Energy and Chemical Change Section 16.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Energy and Chemical Change Section 16.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Energy Relationships in Chemical Reactions
 Energy Relationships in Chemical Reactions What is heat? What is a state function? What is enthalpy? Is enthalpy a state function? What does this mean? How can we calculate this? How are the methods the
Energy Relationships in Chemical Reactions What is heat? What is a state function? What is enthalpy? Is enthalpy a state function? What does this mean? How can we calculate this? How are the methods the
Chapter 5. Thermochemistry
 Chapter 5 Thermochemistry Dr. A. Al-Saadi 1 Preview Introduction to thermochemistry: Potential energy and kinetic energy. Chemical energy. Internal energy, work and heat. Exothermic vs. endothermic reactions.
Chapter 5 Thermochemistry Dr. A. Al-Saadi 1 Preview Introduction to thermochemistry: Potential energy and kinetic energy. Chemical energy. Internal energy, work and heat. Exothermic vs. endothermic reactions.
Selected Questions on Chapter 5 Thermochemistry
 Selected Questions on Chapter 5 Thermochemistry Circle the correct answer: 1) At what velocity (m/s) must a 20.0 g object be moving in order to possess a kinetic energy of 1.00 J? A) 1.00 B) 100 10 2 C)
Selected Questions on Chapter 5 Thermochemistry Circle the correct answer: 1) At what velocity (m/s) must a 20.0 g object be moving in order to possess a kinetic energy of 1.00 J? A) 1.00 B) 100 10 2 C)
1.4 Enthalpy. What is chemical energy?
 1.4 Enthalpy What is chemical energy? Chemical energy is a form of potential energy which is stored in chemical bonds. Chemical bonds are the attractive forces that bind atoms together. As a reaction takes
1.4 Enthalpy What is chemical energy? Chemical energy is a form of potential energy which is stored in chemical bonds. Chemical bonds are the attractive forces that bind atoms together. As a reaction takes
Chemical Energetics. First Law of thermodynamics: Energy can be neither created nor destroyed but It can be converted from one form to another.
 Chemical Energetics First Law of thermodynamics: Energy can be neither created nor destroyed but It can be converted from one form to another. All chemical reactions are accompanied by some form of energy
Chemical Energetics First Law of thermodynamics: Energy can be neither created nor destroyed but It can be converted from one form to another. All chemical reactions are accompanied by some form of energy
Chapter 5 Thermochemistry. 許富銀 ( Hsu Fu-Yin)
 Chapter 5 Thermochemistry 許富銀 ( Hsu Fu-Yin) 1 Thermodynamics The study of energy and its transformations is known as thermodynamics The relationships between chemical reactions and energy changes that
Chapter 5 Thermochemistry 許富銀 ( Hsu Fu-Yin) 1 Thermodynamics The study of energy and its transformations is known as thermodynamics The relationships between chemical reactions and energy changes that
Chapter 3. Thermochemistry: Energy Flow and Chemical Change. 5.1 Forms of Energy and Their Interconversion
 Chapter 3 Thermochemistry: Energy Flow and Chemical Change 5.1 Forms of Energy and Their Interconversion 5.2 Enthalpy: Chemical Change at Constant Pressure 5.3 Calorimetry: Measuring the Heat of a Chemical
Chapter 3 Thermochemistry: Energy Flow and Chemical Change 5.1 Forms of Energy and Their Interconversion 5.2 Enthalpy: Chemical Change at Constant Pressure 5.3 Calorimetry: Measuring the Heat of a Chemical
The Nature of Energy. Chapter Six: Kinetic vs. Potential Energy. Energy and Work. Temperature vs. Heat
 The Nature of Energy Chapter Six: THERMOCHEMISTRY Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationship between chemical reactions and energy changes
The Nature of Energy Chapter Six: THERMOCHEMISTRY Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationship between chemical reactions and energy changes
DETERMINING AND USING H
 DETERMINING AND USING H INTRODUCTION CHANGES IN CHEMISTRY Chemistry is the science that studies matter and the changes it undergoes. Changes are divided into two categories: physical and chemical. During
DETERMINING AND USING H INTRODUCTION CHANGES IN CHEMISTRY Chemistry is the science that studies matter and the changes it undergoes. Changes are divided into two categories: physical and chemical. During
Energy Changes in Reactions p
 Energy Changes in Reactions p.126 210 Heat vs. temperature: Heat is a form of energy, it is transferred from one system to another Temperature is an indication of the intensity of heat, it measures the
Energy Changes in Reactions p.126 210 Heat vs. temperature: Heat is a form of energy, it is transferred from one system to another Temperature is an indication of the intensity of heat, it measures the
June Which is a closed system? (A) burning candle (B) halogen lightbulb (C) hot water in a sink (D) ripening banana
 June 2005 28. Which is a closed system? burning candle halogen lightbulb hot water in a sink ripening banana 29. Which involves the greatest energy change? chemical reaction nuclear reaction phase change
June 2005 28. Which is a closed system? burning candle halogen lightbulb hot water in a sink ripening banana 29. Which involves the greatest energy change? chemical reaction nuclear reaction phase change
Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions
 Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions Terms, definitions, topics Joule, calorie (Re-read p 57-58) Thermochemistry Exothermic reaction Endothermic reaction
Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions Terms, definitions, topics Joule, calorie (Re-read p 57-58) Thermochemistry Exothermic reaction Endothermic reaction
Thermochemistry. Section The flow of energy
 Thermochemistry Section 17.1 - The flow of energy What is Energy? Energy is the capacity for doing work or supplying heat Energy does not have mass or volume, and it can only be detected because of its
Thermochemistry Section 17.1 - The flow of energy What is Energy? Energy is the capacity for doing work or supplying heat Energy does not have mass or volume, and it can only be detected because of its
Slide 1 / Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy. a only b only c only a and c b and c
 Slide 1 / 84 1 Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy A B C D E a only b only c only a and c b and c Slide 2 / 84 2 The internal energy of a system
Slide 1 / 84 1 Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy A B C D E a only b only c only a and c b and c Slide 2 / 84 2 The internal energy of a system
Thermochemistry Notes
 Name: Thermochemistry Notes I. Thermochemistry deals with the changes in energy that accompany a chemical reaction. Energy is measured in a quantity called enthalpy, represented as H. The change in energy
Name: Thermochemistry Notes I. Thermochemistry deals with the changes in energy that accompany a chemical reaction. Energy is measured in a quantity called enthalpy, represented as H. The change in energy
Energy Ability to produce change or do work. First Law of Thermodynamics. Heat (q) Quantity of thermal energy
 THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
Energy and Chemical Change
 Energy and Chemical Change Section 15.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Energy and Chemical Change Section 15.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Chapter 8 Thermochemistry: Chemical Energy. Chemical Thermodynamics
 Chapter 8 Thermochemistry: Chemical Energy Chapter 8 1 Chemical Thermodynamics Chemical Thermodynamics is the study of the energetics of a chemical reaction. Thermodynamics deals with the absorption or
Chapter 8 Thermochemistry: Chemical Energy Chapter 8 1 Chemical Thermodynamics Chemical Thermodynamics is the study of the energetics of a chemical reaction. Thermodynamics deals with the absorption or
Unit 5 A3: Energy changes in industry
 1. ENTHALPY CHANGES Unit 5 A3: Energy changes in industry 1.1 Introduction to enthalpy and enthalpy changes 2 1.2 Enthalpy profile diagrams 2 1.3 Activation energy 3 1.4 Standard conditions 5 1.5 Standard
1. ENTHALPY CHANGES Unit 5 A3: Energy changes in industry 1.1 Introduction to enthalpy and enthalpy changes 2 1.2 Enthalpy profile diagrams 2 1.3 Activation energy 3 1.4 Standard conditions 5 1.5 Standard
3. What is the energy (in J/photon) associated with a photon with a wavelength of 245 nm?
 I. (40 points) A. (10 points) The following 3 questions are not related. There will be no partial credit given but you must show all work. (You MUST write units for your numerical answers. Answers without
I. (40 points) A. (10 points) The following 3 questions are not related. There will be no partial credit given but you must show all work. (You MUST write units for your numerical answers. Answers without
Heat. Heat Terminology 04/12/2017. System Definitions. System Definitions
 System Definitions Heat Physical Science 20 Ms. Hayduk Heat Terminology System: the part of the universe being studied (big Earth, or small one atom) Surroundings: the part of the universe outside the
System Definitions Heat Physical Science 20 Ms. Hayduk Heat Terminology System: the part of the universe being studied (big Earth, or small one atom) Surroundings: the part of the universe outside the
Energy Changes in Chemical Reactions
 Chapter Energy Changes in Chemical Reactions 10.1 Energy and Energy Changes 10.2 Introduction to Thermodynamics States and State Functions The First Law of Thermodynamics Work and Heat 10.3 Enthalpy Reactions
Chapter Energy Changes in Chemical Reactions 10.1 Energy and Energy Changes 10.2 Introduction to Thermodynamics States and State Functions The First Law of Thermodynamics Work and Heat 10.3 Enthalpy Reactions
Thermochemistry. Questions to ponder. Because 4/20/14. an ice-cube? an ice-cube? Part 2: Calorimetry. But I KNOW. Q=mc T, but T=0
 Thermochemistry Part 2: Calorimetry p p If you leave your keys and your chemistry book sitting in the sun on a hot summer day, which one is hotter? Why is there a difference in temperature between the
Thermochemistry Part 2: Calorimetry p p If you leave your keys and your chemistry book sitting in the sun on a hot summer day, which one is hotter? Why is there a difference in temperature between the
Enthalpy. Enthalpy. Enthalpy. Enthalpy. E = q + w. Internal Energy at Constant Volume SYSTEM. heat transfer in (endothermic), +q
 heat transfer in (endothermic), +q heat transfer out (exothermic), -q SYSTEM E = q + w w transfer in (+w) w transfer out (-w) Internal Energy at Constant Volume E = KE + PE ΔE = q + w Because most systems,
heat transfer in (endothermic), +q heat transfer out (exothermic), -q SYSTEM E = q + w w transfer in (+w) w transfer out (-w) Internal Energy at Constant Volume E = KE + PE ΔE = q + w Because most systems,
Thermodynamics- Chapter 19 Schedule and Notes
 Thermodynamics- Chapter 19 Schedule and Notes Date Topics Video cast DUE Assignment during class time One Review of thermodynamics ONE and TWO Review of thermo Wksheet Two 19.1-4; state function THREE
Thermodynamics- Chapter 19 Schedule and Notes Date Topics Video cast DUE Assignment during class time One Review of thermodynamics ONE and TWO Review of thermo Wksheet Two 19.1-4; state function THREE
Name: Class: Date: ID: A
 Name: Class: _ Date: _ ID: A Chpter 17 review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of these phase changes is an endothermic process? a.
Name: Class: _ Date: _ ID: A Chpter 17 review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of these phase changes is an endothermic process? a.
Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy
 Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Chapter 6: Thermochemistry
 Chapter 6: Thermochemistry Section 6.1: Introduction to Thermochemistry Thermochemistry refers to the study of heat flow or heat energy in a chemical reaction. In a study of Thermochemistry the chemical
Chapter 6: Thermochemistry Section 6.1: Introduction to Thermochemistry Thermochemistry refers to the study of heat flow or heat energy in a chemical reaction. In a study of Thermochemistry the chemical
CHEM 1105 S10 March 11 & 14, 2014
 CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
Thermochemistry. Chapter 6. Concept Check 6.1. Concept Check 6.2. Solution
 Chapter 6 Thermochemistry Concept Check 6.1 A solar-powered water pump has photovoltaic cells on protruding top panels. These cells collect energy from sunlight, storing it momentarily in a battery, which
Chapter 6 Thermochemistry Concept Check 6.1 A solar-powered water pump has photovoltaic cells on protruding top panels. These cells collect energy from sunlight, storing it momentarily in a battery, which
Calorimetry: Heat of Solution
 Calorimetry: Heat of Solution When a substance undergoes a change in temperature, the quantity of heat lost or gained can be calculated using the following relationship: (heat) = m s T (1) The specific
Calorimetry: Heat of Solution When a substance undergoes a change in temperature, the quantity of heat lost or gained can be calculated using the following relationship: (heat) = m s T (1) The specific
Chapter 6 Problems: 9, 19, 24, 25, 26, 27, 31-33, 37, 39, 43, 45, 47, 48, 53, 55, 57, 59, 65, 67, 73, 78-82, 85, 89, 93
 Chapter 6 Problems: 9, 19, 24, 25, 26, 27, 31-33, 37, 39, 43, 45, 47, 48, 53, 55, 57, 59, 65, 67, 73, 78-82, 85, 89, 93 Chapter 6 Thermochemistry The study of chemical reactions and the energy changes
Chapter 6 Problems: 9, 19, 24, 25, 26, 27, 31-33, 37, 39, 43, 45, 47, 48, 53, 55, 57, 59, 65, 67, 73, 78-82, 85, 89, 93 Chapter 6 Thermochemistry The study of chemical reactions and the energy changes
Energy Changes in Chemical Reactions
 Unit 3 Energetics Unit 3-1 Section 3.1 Energy Changes in Chemical Reactions ( 1 ) Conservation of energy An object which is capable of doing work is said to possess energy. There are many forms of energy:
Unit 3 Energetics Unit 3-1 Section 3.1 Energy Changes in Chemical Reactions ( 1 ) Conservation of energy An object which is capable of doing work is said to possess energy. There are many forms of energy:
Thermochemistry HW. PSI Chemistry
 Thermochemistry HW PSI Chemistry Name Energy 1) Objects can possess energy as: (a) endothermic energy (b) potential energy A) a only B) b only C) c only D) a and c E) b and c (c) kinetic energy 2) The
Thermochemistry HW PSI Chemistry Name Energy 1) Objects can possess energy as: (a) endothermic energy (b) potential energy A) a only B) b only C) c only D) a and c E) b and c (c) kinetic energy 2) The
ENTHALPY, INTERNAL ENERGY, AND CHEMICAL REACTIONS: AN OUTLINE FOR CHEM 101A
 ENTHALPY, INTERNAL ENERGY, AND CHEMICAL REACTIONS: AN OUTLINE FOR CHEM 101A PART 1: KEY TERMS AND SYMBOLS IN THERMOCHEMISTRY System and surroundings When we talk about any kind of change, such as a chemical
ENTHALPY, INTERNAL ENERGY, AND CHEMICAL REACTIONS: AN OUTLINE FOR CHEM 101A PART 1: KEY TERMS AND SYMBOLS IN THERMOCHEMISTRY System and surroundings When we talk about any kind of change, such as a chemical
Quantities in Chemical Reactions
 Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Gas Laws. Bonding. Solutions M= moles solute Mass %= mass solute x 100. Acids and Bases. Thermochemistry q = mc T
 Name Period Teacher Practice Test: OTHS Academic Chemistry Spring Semester 2017 The exam will have 100 multiple choice questions (1 point each) Formula sheet (see below) and Periodic table will be provided
Name Period Teacher Practice Test: OTHS Academic Chemistry Spring Semester 2017 The exam will have 100 multiple choice questions (1 point each) Formula sheet (see below) and Periodic table will be provided
Thermochemistry-Part 1
 Brad Collins Thermochemistry-Part 1 Chapter 7 Thermochemistry Thermodynamics: The study of energy Thermochemistry: The study of energy in chemical reactions Energy: The capacity to do work Work = force
Brad Collins Thermochemistry-Part 1 Chapter 7 Thermochemistry Thermodynamics: The study of energy Thermochemistry: The study of energy in chemical reactions Energy: The capacity to do work Work = force
Energy Ability to produce change or do work. First Law of Thermodynamics. Heat (q) Quantity of thermal energy
 THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
Enthalpy and Internal Energy
 Enthalpy and Internal Energy H or ΔH is used to symbolize enthalpy. The mathematical expression of the First Law of Thermodynamics is: ΔE = q + w, where ΔE is the change in internal energy, q is heat and
Enthalpy and Internal Energy H or ΔH is used to symbolize enthalpy. The mathematical expression of the First Law of Thermodynamics is: ΔE = q + w, where ΔE is the change in internal energy, q is heat and
Chapter 5 THERMO. THERMO chemistry. 5.4 Enthalpy of Reactions 5.5 Calorimetry 5.6 Hess s Law 5.7 Enthalpies of Formation
 Chapter 5 THERMO THERMO chemistry 5.4 Enthalpy of Reactions 5.5 Calorimetry 5.6 Hess s Law 5.7 Enthalpies of Formation Chemical Equations 1 st WRITE the Chemical Equation 2 nd BALANCE the Chemical Equation
Chapter 5 THERMO THERMO chemistry 5.4 Enthalpy of Reactions 5.5 Calorimetry 5.6 Hess s Law 5.7 Enthalpies of Formation Chemical Equations 1 st WRITE the Chemical Equation 2 nd BALANCE the Chemical Equation
Chemistry Grade : 11 Term-3/Final Exam Revision Sheet
 Chemistry Grade : 11 Term-3/Final Exam Revision Sheet Exam Date: Tuesday 12/6/2018 CCS:Chem.6a,6b,6c,6d,6e,6f,7a,7b,7d,7c,7e,7f,1g Chapter(12):Solutions Sections:1,2,3 Textbook pages 378 to 408 Chapter(16):Reaction
Chemistry Grade : 11 Term-3/Final Exam Revision Sheet Exam Date: Tuesday 12/6/2018 CCS:Chem.6a,6b,6c,6d,6e,6f,7a,7b,7d,7c,7e,7f,1g Chapter(12):Solutions Sections:1,2,3 Textbook pages 378 to 408 Chapter(16):Reaction
Energetics. Topic
 Energetics Topic 5.1 5.2 Topic 5.1 Exothermic and Endothermic Reactions?? total energy of the universe is a constant if a system loses energy, it must be gained by the surroundings, and vice versa Enthalpy
Energetics Topic 5.1 5.2 Topic 5.1 Exothermic and Endothermic Reactions?? total energy of the universe is a constant if a system loses energy, it must be gained by the surroundings, and vice versa Enthalpy
CHEMISTRY - TRO 4E CH.6 - THERMOCHEMISTRY.
 !! www.clutchprep.com CONCEPT: ENERGY CHANGES AND ENERGY CONSERVATION is the branch of physical science concerned with heat and its transformations to and from other forms of energy. is the branch of chemistry
!! www.clutchprep.com CONCEPT: ENERGY CHANGES AND ENERGY CONSERVATION is the branch of physical science concerned with heat and its transformations to and from other forms of energy. is the branch of chemistry
CHAPTER 17: THERMOCHEMISTRY. Mrs. Brayfield
 CHAPTER 17: THERMOCHEMISTRY Mrs. Brayfield REVIEW What is the law of conservation of energy? It states that energy cannot be created or destroyed So the energy of any process is the same THERMOCHEMISTRY
CHAPTER 17: THERMOCHEMISTRY Mrs. Brayfield REVIEW What is the law of conservation of energy? It states that energy cannot be created or destroyed So the energy of any process is the same THERMOCHEMISTRY
The Nature of Energy Energy is the ability to do work or produce Heat, q or Q, is ; flows due to temperature differences (always to )
 CP Chapter 17 Thermochemistry 2014-2015 Thermochemistry Thermochemistry is the study of energy that occur during chemical and physical changes (changes of state) The Nature of Energy Energy is the ability
CP Chapter 17 Thermochemistry 2014-2015 Thermochemistry Thermochemistry is the study of energy that occur during chemical and physical changes (changes of state) The Nature of Energy Energy is the ability
2. Calculate the heat change in joules when 45.2 g of steam at C condenses to water at the same temperature.!
 Chem L3 Worksheet:Thermochemistry: Chapter 11 Page 1 Water Heating Cooling curve Problems ( Stairstep ): pp. 292-325, 549-556 1. How much heat is released when 65.0 g of steam cools from 150.0 C to 130.0
Chem L3 Worksheet:Thermochemistry: Chapter 11 Page 1 Water Heating Cooling curve Problems ( Stairstep ): pp. 292-325, 549-556 1. How much heat is released when 65.0 g of steam cools from 150.0 C to 130.0
