"Undissolved ionic compounds":
|
|
|
- Cecilia Chapman
- 6 years ago
- Views:
Transcription
1 115 "Undissolved ionic compounds": How can I tell if an ionic compound dissolves in water? - consult experimental data: "solubility rules"! A few of the "rules"... - Compounds that contain a Group IA cation (or ammonium) are soluble - Nitrates and acetates are soluble - Carbonates, phosphates, and hydroxides tend to be insoluble... or see the web site for a solubility chart. #8 - hydroxides generally insoiluble, except Group IA, ammonium, calcium strontium, barium Conclusion: iron(iii) hydroxide is insoluble. #2 - acetates are soluble, no common exceptions. Conclusion: calcium acetate is soluble. #3 - Iodides usually dissolve, exceptions are silver, mercury, lead Conclusion: silver(i) iodide is INSOLUBLE #5 - Most carbonates are insoluble Conclusion - barium carbonate is insoluble.
2 116 Exchange Chemistry - Three kinds of exchange chemistry. PRECIPITATION Formation of IONIC SOLID ACID/BASE or NEUTRALIZATION Formation of MOLECULES! GAS FORMATION (formation of unstable molecules) SOME (but not all) reactions that form gases are examples of exchange chemistry. Just because you mix together two ionic compounds does NOT mean that a reaction will occur. You need a DRIVING FORCE for a reaction.
3 117 PRECIPITATION REACTIONS - driving force is the formation of an insoluble ionic compound. ions: This reaction is driven by the formation of SOLID magnesium phosphate When you're trying to complete a precipitation reaction: Write the IONS that form when the reactants are dissolved. Make NEW compounds by pairing up cations with anions. Don't forget that the positive and negative charges must balance each other out! Use the solubility rules to determine the PHASE of each new compound - solid or aqueous. Balance the overall equation.
4 118 NO REACTION! ions: "exchange"... dissolves in water... dissolves in water So, no solid forms here. All possible combinations of these four ions result in compounds that dissolve readily in water. NO CHANGE, therefore NO DRIVING FORCE, and NO REACTION We will learn about other driving forces than the formation of solid, but these driving forces do not apply to this reaction
5 119 ACID/BASE REACTIONS (also called NEUTRALIZATION REACTIONS) - There are several stable molecules that may be formed in double replacement reactions, but the most common is WATER! - Double replacement reactions that form water are also called "neutralizations" acid base salt ionic compound * To make water ( ), you need a source of hydrogen ion ( ) and hydroxide ion ( )... assumes you're reacting STRONG acid with STRONG base! This is the NET IONIC EQUATION for many neutralizations
6 120 ACIDS - compounds that release hydrogen ion (H ), when dissolved in water. Properties of acids: - Corrosive: React with most metals to give off hydrogen gas - Cause chemical burns on contact - Taste sour (like citrus - citric acid!) - Changes litmus indicator to RED BASES - Substances that release hydroxide ion (OH ) when dissolved in water Properties of bases: - Caustic: Attack and dissolve organic matter (think lye, which is NaOH) - Cause skin/eye damage on contact - Taste bitter - changes litmus indicator to BLUE Due to the dissolving action of base on your skin, bases will feel "slippery". The base ITSELF is not particularly slippery, but what's left of your skin IS!
7 121 ACID/BASE or NEUTRALIZATION reactions continued - the driving force of these reactions is the formation of water molecules. Net ionic equation From the acid From the base ions: The formation of WATER MOLECULES drives this reaction! - How can this reaction be detected? - ph detector (indicator paper, etc.) - do the products have similar chemical properties to the reactants? - release of heat!... formation of water is usually accompanied by a release of heat
8 122 GAS FORMATION / OTHER MOLECULES - There are a few other molecules that can be made with exchange-type chemistry. - Most of these molecules are unstable and can break apart to form gases. - Formation of a weak acid: - The formation of ANY weak acid in an exchange-type reaction can be a driving force. - Some weak acids are unstable and can break apart into gas molecules. Gas bubbles can leave solution!... but how would you form carbonic acid in an exchange-type reaction? Formation of carbonic acid drives the reaction... BUT...
9 but when we mix sulfuric acid and sodium bicarbonate, we observe BUBBLES. We need to write an equation that agrees with our observations. We know that carbonic acid decomposes, so we go ahead and put that into our equation. Other molecules of interest: sulfurous acid - React an ACID with a SULFITE hydrogen sulfide (gas) - React an ACID with a SULFIDE
- electrolytes: substances that dissolve in water to form charge-carrying solutions
 111 Electrolytes and Ionic Theory - electrolytes: substances that dissolve in water to form charge-carrying solutions * Electrolytes form ions in solution - (ions that are mobile are able to carry charge!).
111 Electrolytes and Ionic Theory - electrolytes: substances that dissolve in water to form charge-carrying solutions * Electrolytes form ions in solution - (ions that are mobile are able to carry charge!).
- Double replacement reactions that form water are also called "neutralizations"
 119 ACID/BASE REACTIONS (also called NEUTRALIZATION REACTIONS) - There are several stable molecules that may be formed in double replacement reactions, but the most common is WATER! - Double replacement
119 ACID/BASE REACTIONS (also called NEUTRALIZATION REACTIONS) - There are several stable molecules that may be formed in double replacement reactions, but the most common is WATER! - Double replacement
97 MOLECULAR AND IONIC EQUATIONS
 97 MOLECULAR AND IONIC EQUATIONS - Ions that show up IN THE SAME FORM on the left and right sides of a chemical equation are called SPECTATOR IONS. If we rewrite an ionic equation to leave out the spectator
97 MOLECULAR AND IONIC EQUATIONS - Ions that show up IN THE SAME FORM on the left and right sides of a chemical equation are called SPECTATOR IONS. If we rewrite an ionic equation to leave out the spectator
- Double replacement reactions that form water are also called "neutralizations"
 101 ACID/BASE REACTIONS (also called NEUTRALIZATION REACTIONS) - There are several stable molecules that may be formed in double replacement reactions, but the most common is WATER! - Double replacement
101 ACID/BASE REACTIONS (also called NEUTRALIZATION REACTIONS) - There are several stable molecules that may be formed in double replacement reactions, but the most common is WATER! - Double replacement
acrylonitrile Calculate how many grams of acrylonitrile could be obtained from 651 kg of propylene, assuming there is excess NO present.
 propylene acrylonitrile Calculate how many grams of acrylonitrile could be obtained from 651 kg of propylene, assuming there is excess NO present. 1 - Change the mass of propylene to moles propylene (formula
propylene acrylonitrile Calculate how many grams of acrylonitrile could be obtained from 651 kg of propylene, assuming there is excess NO present. 1 - Change the mass of propylene to moles propylene (formula
How many milliliters of 6.00M hydrochloric acid is needed to completely react with 25.0 g of sodium carbonate?
 102 Example: How many milliliters of 6.00M hydrochloric acid is needed to completely react with 25.0 g of sodium carbonate? 1) Convert 25.0 grams sodium carbonate to moles. Use FORMULA WEIGHT. 2) Convert
102 Example: How many milliliters of 6.00M hydrochloric acid is needed to completely react with 25.0 g of sodium carbonate? 1) Convert 25.0 grams sodium carbonate to moles. Use FORMULA WEIGHT. 2) Convert
- Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS
 63 POLYATOMIC IONS - Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS - Polyatomic ions form ionic compounds in the same way that single-element ions
63 POLYATOMIC IONS - Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS - Polyatomic ions form ionic compounds in the same way that single-element ions
DOUBLE DISPLACEMENT REACTIONS. Double your pleasure, double your fun
 DOUBLE DISPLACEMENT REACTIONS Double your pleasure, double your fun Industrial processes produce unwanted by-products. Dissolved toxic metal ions-copper, mercury, and cadmium-are common leftovers in the
DOUBLE DISPLACEMENT REACTIONS Double your pleasure, double your fun Industrial processes produce unwanted by-products. Dissolved toxic metal ions-copper, mercury, and cadmium-are common leftovers in the
DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME
 DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The name of an ionic compound is made of the names of the CATION and ANION in the compound. - To get the FORMULA, you must figure out the SMALLEST
DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The name of an ionic compound is made of the names of the CATION and ANION in the compound. - To get the FORMULA, you must figure out the SMALLEST
- Use the STEM NAME of the element, then add "-ide" suffix
 65 ANIONS 2 kinds Main-group nonmetals - Use the STEM NAME of the element, then add "-ide" suffix N : "nitride" ion P : "phosphide ion" O : "oxide ion" F : "fluoride ion" Polyatomic ions - Memorize list.(see
65 ANIONS 2 kinds Main-group nonmetals - Use the STEM NAME of the element, then add "-ide" suffix N : "nitride" ion P : "phosphide ion" O : "oxide ion" F : "fluoride ion" Polyatomic ions - Memorize list.(see
- Use the STEM NAME of the element, then add "-ide" suffix
 65 ANIONS 2 kinds Main-group nonmetals - Use the STEM NAME of the element, then add "-ide" suffix N : "nitride" ion P : "phosphide ion" O : "oxide ion" F : "fluoride ion" Polyatomic ions - Memorize list.(see
65 ANIONS 2 kinds Main-group nonmetals - Use the STEM NAME of the element, then add "-ide" suffix N : "nitride" ion P : "phosphide ion" O : "oxide ion" F : "fluoride ion" Polyatomic ions - Memorize list.(see
Name CHEMISTRY / / Oxide Reactions & Net Ionic Reactions
 Name CHEMISTRY / / Oxide Reactions & Net Ionic Reactions The first type of reactions we will look at today are reactions between an oxide (a compound with oxygen as its anion) and water. There are two
Name CHEMISTRY / / Oxide Reactions & Net Ionic Reactions The first type of reactions we will look at today are reactions between an oxide (a compound with oxygen as its anion) and water. There are two
Unit 5 Lesson 2 Acids, Bases, and Salts. Copyright Houghton Mifflin Harcourt Publishing Company
 Donations Accepted What are acids and bases? Acids and bases are chemicals that increase the number of ions present in a water solution when they dissolve. Lemon juice and vinegar both contain acid. Shampoo
Donations Accepted What are acids and bases? Acids and bases are chemicals that increase the number of ions present in a water solution when they dissolve. Lemon juice and vinegar both contain acid. Shampoo
NAMING IONIC COMPOUNDS
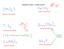 NAMING IONIC COMPOUNDS iron(ii) carbonate ammonium sulfide titanium (IV) sulfide barium phosphate Spelling matters! calcium nitrate sodium phosphide DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE
NAMING IONIC COMPOUNDS iron(ii) carbonate ammonium sulfide titanium (IV) sulfide barium phosphate Spelling matters! calcium nitrate sodium phosphide DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE
Name HONORS CHEMISTRY / / Oxide Reactions & Net Ionic Reactions
 Name HONORS CHEMISTRY / / Oxide Reactions & Net Ionic Reactions The first type of reactions we will look at today are reactions between an oxide (a compound with oxygen as its anion) and water. There are
Name HONORS CHEMISTRY / / Oxide Reactions & Net Ionic Reactions The first type of reactions we will look at today are reactions between an oxide (a compound with oxygen as its anion) and water. There are
NAMING IONIC COMPOUNDS. ammonium sulfide. iron(ii) carbonate. barium phosphate. titanium(iv) sulfide. Spelling matters!
 67 NAMING IONIC COMPOUNDS ammonium sulfide iron(ii) carbonate titanium(iv) sulfide barium phosphate Spelling matters! barium phosphide 68 DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The
67 NAMING IONIC COMPOUNDS ammonium sulfide iron(ii) carbonate titanium(iv) sulfide barium phosphate Spelling matters! barium phosphide 68 DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The
Identification of White Powders
 CSI Chemistry Activity 4 Identification of White Powders GOALS In this activity you will: Create and use a flowchart to identify an unknown entity. Identify an unknown ionic compound based on an understanding
CSI Chemistry Activity 4 Identification of White Powders GOALS In this activity you will: Create and use a flowchart to identify an unknown entity. Identify an unknown ionic compound based on an understanding
Solubility Rules and Net Ionic Equations
 Solubility Rules and Net Ionic Equations Why? Solubility of a salt depends upon the type of ions in the salt. Some salts are soluble in water and others are not. When two soluble salts are mixed together
Solubility Rules and Net Ionic Equations Why? Solubility of a salt depends upon the type of ions in the salt. Some salts are soluble in water and others are not. When two soluble salts are mixed together
Part 01 - Notes: Reactions & Classification
 Objectives: Identify, define, and explain: combination reaction, synthesis reaction, decomposition reaction, single replacement reaction, double replacement reaction, combustion reaction, rapid oxidation,
Objectives: Identify, define, and explain: combination reaction, synthesis reaction, decomposition reaction, single replacement reaction, double replacement reaction, combustion reaction, rapid oxidation,
- Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS
 63 POLYATOMIC IONS - Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS - Polyatomic ions form ionic compounds in the same way that single-element ions
63 POLYATOMIC IONS - Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS - Polyatomic ions form ionic compounds in the same way that single-element ions
Understand what acids and alkalis are, and where they are found.
 Lesson Aims- Understand what acids and alkalis are, and where they are found. Test a range of household products with litmus indicator to see whether they are acidic or alkaline. Found in citrus fruit
Lesson Aims- Understand what acids and alkalis are, and where they are found. Test a range of household products with litmus indicator to see whether they are acidic or alkaline. Found in citrus fruit
 CHEMISTRY 20 Simple Composition (Formation) and Simple Decomposition Reactions! (sc aka f) elements combine to form one single product! (sd) reactant breaks down to form pure elements from the periodic
CHEMISTRY 20 Simple Composition (Formation) and Simple Decomposition Reactions! (sc aka f) elements combine to form one single product! (sd) reactant breaks down to form pure elements from the periodic
Chemical Reactions and Equations
 Chemical Reactions and Equations 5-1 5.1 What is a Chemical Reaction? A chemical reaction is a chemical change. A chemical reaction occurs when one or more substances is converted into one or more new
Chemical Reactions and Equations 5-1 5.1 What is a Chemical Reaction? A chemical reaction is a chemical change. A chemical reaction occurs when one or more substances is converted into one or more new
POGIL EXERCISE 12 How Can You Predict Chemical Reactions?
 RUN TIME: 60 MIN POGIL 12 Page 1 of 7 POGIL EXERCISE 12 How Can You Predict Chemical Reactions? Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder
RUN TIME: 60 MIN POGIL 12 Page 1 of 7 POGIL EXERCISE 12 How Can You Predict Chemical Reactions? Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder
Unit 12: Acids & Bases. Aim: What are the definitions and properties of an acid and a base? Properties of an Acid. Taste Sour.
 Unit 12: Acids & Bases Aim: What are the definitions and properties of an acid and a base? Mar 23 12:08 PM Properties of an Acid 3. Are electrolytes. (Dissociate and conduct electricity when aq) 2. Turns
Unit 12: Acids & Bases Aim: What are the definitions and properties of an acid and a base? Mar 23 12:08 PM Properties of an Acid 3. Are electrolytes. (Dissociate and conduct electricity when aq) 2. Turns
Net Ionic Equations. Making Sense of Chemical Reactions
 Making Sense of Chemical Reactions Now that you have mastered writing balanced chemical equations it is time to take a deeper look at what is really taking place chemically in each reaction. There are
Making Sense of Chemical Reactions Now that you have mastered writing balanced chemical equations it is time to take a deeper look at what is really taking place chemically in each reaction. There are
Name Honors Chemistry / /
 Name Honors Chemistry / / Redox Reactions Rules for Assigning Oxidation Numbers Oxidation state of: Charge Examples Neutral monoatomic or molecular elements 0 Na(s), Cl 2 (g), S 8 (s), O 2 (g), Hg(l) Fluorine
Name Honors Chemistry / / Redox Reactions Rules for Assigning Oxidation Numbers Oxidation state of: Charge Examples Neutral monoatomic or molecular elements 0 Na(s), Cl 2 (g), S 8 (s), O 2 (g), Hg(l) Fluorine
Net Ionic Reactions. The reaction between strong acids and strong bases is one example:
 Net Ionic Reactions Model 1 Net Ionic Reactions. Net ionic reactions are frequently used when strong electrolytes react in solution to form nonelectrolytes or weak electrolytes. These equations let you
Net Ionic Reactions Model 1 Net Ionic Reactions. Net ionic reactions are frequently used when strong electrolytes react in solution to form nonelectrolytes or weak electrolytes. These equations let you
Chemistry and Reactions Year 9 Extension Science. 1 GZ Science Resources 2014
 Chemistry and Reactions Year 9 Extension Science 1 GZ Science Resources 2014 1a Reactants join together to form new products during chemical reactions The atoms present in the reactants rearrange themselves
Chemistry and Reactions Year 9 Extension Science 1 GZ Science Resources 2014 1a Reactants join together to form new products during chemical reactions The atoms present in the reactants rearrange themselves
ed. Brad Collins Aqueous Chemistry Chapter 5 Some images copyright The McGraw-Hill Companies, Inc. Sunday, August 18, 13
 ed. Brad Collins Aqueous Chemistry Chapter 5 Some images copyright The McGraw-Hill Companies, Inc. A solution is a homogenous mixture of 2 or more substances at the molecular level The solute(s) is(are)
ed. Brad Collins Aqueous Chemistry Chapter 5 Some images copyright The McGraw-Hill Companies, Inc. A solution is a homogenous mixture of 2 or more substances at the molecular level The solute(s) is(are)
DETERMINING IONIC FORMULAS strontium oxide
 69 sodium sulfate DETERMINING IONIC FORMULAS strontium oxide tin(ii) phosphate chromium(iii) nitrate barium hydroxide titanium(iv) chloride When adding a subscript to a polyatomic ion, put the polyatomic
69 sodium sulfate DETERMINING IONIC FORMULAS strontium oxide tin(ii) phosphate chromium(iii) nitrate barium hydroxide titanium(iv) chloride When adding a subscript to a polyatomic ion, put the polyatomic
Classifying Substances
 Classifying Substances There are other ways you can use to classify compounds. For example, you can classify some compounds as acids or bases. You are very familiar with acids and bases because you see
Classifying Substances There are other ways you can use to classify compounds. For example, you can classify some compounds as acids or bases. You are very familiar with acids and bases because you see
What Do You Think? Investigate GOALS
 Cool Chemistry Show Activity 4 Chemical Equations GOALS In this activity you will: Represent chemical changes using word equations and chemical equations. Distinguish between different classes of chemical
Cool Chemistry Show Activity 4 Chemical Equations GOALS In this activity you will: Represent chemical changes using word equations and chemical equations. Distinguish between different classes of chemical
NCEA Chemistry 2.2 Identify Ions AS 91162
 NCEA Chemistry 2.2 Identify Ions AS 91162 What is this NCEA Achievement Standard? When a student achieves a standard, they gain a number of credits. Students must achieve a certain number of credits to
NCEA Chemistry 2.2 Identify Ions AS 91162 What is this NCEA Achievement Standard? When a student achieves a standard, they gain a number of credits. Students must achieve a certain number of credits to
chapter 14: ions in aqueous solutions
 chapter 14: ions in aqueous solutions Dissociation When a compound that is made of ions dissolves in water, the ions separate from one another. This is called dissociation. NaCl(s) Na + (aq) + Cl - (aq)
chapter 14: ions in aqueous solutions Dissociation When a compound that is made of ions dissolves in water, the ions separate from one another. This is called dissociation. NaCl(s) Na + (aq) + Cl - (aq)
Santa Monica College Chemistry 11
 Types of Reactions Objectives The objectives of this laboratory are as follows: To perform several types of simple chemical reactions, To become familiar with some common observable signs of chemical reactions,
Types of Reactions Objectives The objectives of this laboratory are as follows: To perform several types of simple chemical reactions, To become familiar with some common observable signs of chemical reactions,
Acids Bases and Salts Acid
 Acids Bases and Salts Acid ph less than 7.0 Sour taste Electrolyte Names of Acids Binary acids Contain only 2 elements Begin with hydro; end with ic Ternary acids Ex: H 2 S = hydrosulfuric Contain a polyatomic
Acids Bases and Salts Acid ph less than 7.0 Sour taste Electrolyte Names of Acids Binary acids Contain only 2 elements Begin with hydro; end with ic Ternary acids Ex: H 2 S = hydrosulfuric Contain a polyatomic
Double replacement reactions
 1. Learn to predict Double replacement reaction If and when a reaction occurs, what are the products? 2. Learn to write Double replacement reaction: (i) Balanced chemical reaction (ii) Net ionicreaction
1. Learn to predict Double replacement reaction If and when a reaction occurs, what are the products? 2. Learn to write Double replacement reaction: (i) Balanced chemical reaction (ii) Net ionicreaction
NET IONIC REACTIONS in AQUEOUS SOLUTIONS AB + CD AD + CB
 NET IONIC REACTIONS in AQUEOUS SOLUTIONS Double replacements are among the most common of the simple chemical reactions. Consider the hypothetical reaction: AB + CD AD + CB where AB exists as A + and B
NET IONIC REACTIONS in AQUEOUS SOLUTIONS Double replacements are among the most common of the simple chemical reactions. Consider the hypothetical reaction: AB + CD AD + CB where AB exists as A + and B
INTRODUCTORY CHEMISTRY Concepts and Critical Thinking Seventh Edition by Charles H. Corwin
 Lecture INTRODUCTORY CHEMISTRY Concepts and Critical Thinking Seventh Edition by Charles H. Corwin Acids and Bases Properties of Acids An acid is any substance that releases hydrogen ions, H +, into water.
Lecture INTRODUCTORY CHEMISTRY Concepts and Critical Thinking Seventh Edition by Charles H. Corwin Acids and Bases Properties of Acids An acid is any substance that releases hydrogen ions, H +, into water.
Announcements. There are 3-classes of chemical reactions that occur in aqueous solution.
 Announcements Exam 1 Results: Mean: 71% Range: 39.5%-93.5% Median: 72% Other Bio-LS Class Mean 72% Please read Chapter 4 and complete problems. Please see me for help. There are 3-classes of chemical reactions
Announcements Exam 1 Results: Mean: 71% Range: 39.5%-93.5% Median: 72% Other Bio-LS Class Mean 72% Please read Chapter 4 and complete problems. Please see me for help. There are 3-classes of chemical reactions
I. Properties of Aqueous Solutions A) Electrolytes and Non-Electrolytes B) Predicting Solubility* II. Reactions of Ionic Compounds in Solution*
 Chapter 5 Reactions in Aqueous Solutions Titrations Kick Acid!!! 1 I. Properties of Aqueous Solutions A) Electrolytes and Non-Electrolytes B) Predicting Solubility* II. Reactions of Ionic Compounds in
Chapter 5 Reactions in Aqueous Solutions Titrations Kick Acid!!! 1 I. Properties of Aqueous Solutions A) Electrolytes and Non-Electrolytes B) Predicting Solubility* II. Reactions of Ionic Compounds in
Experiment 8 - Double Displacement Reactions
 Experiment 8 - Double Displacement Reactions A double displacement reaction involves two ionic compounds that are dissolved in water. In a double displacement reaction, it appears as though the ions are
Experiment 8 - Double Displacement Reactions A double displacement reaction involves two ionic compounds that are dissolved in water. In a double displacement reaction, it appears as though the ions are
Chapter 14: Acids and Bases
 Chapter 14: Acids and Bases Properties of Acids and Bases What is an acid? Some examples of common items containing acids: Vinegar contains acetic acid; lemons and citrus fruits contain citric acid; many
Chapter 14: Acids and Bases Properties of Acids and Bases What is an acid? Some examples of common items containing acids: Vinegar contains acetic acid; lemons and citrus fruits contain citric acid; many
Intro to Reactions/ Balancing Equations
 Intro to Reactions/ Balancing Equations Chemical Reactions Chemical reactions involve change. Evidence of a chemical reaction could include the following Evolution of heat, light, and/or sound Production
Intro to Reactions/ Balancing Equations Chemical Reactions Chemical reactions involve change. Evidence of a chemical reaction could include the following Evolution of heat, light, and/or sound Production
NET IONIC EQUATIONS. Electrolyte Behavior
 NET IONIC EQUATIONS Net ionic equations are useful in that they show only those chemical species directly participating in a chemical reaction. They are thus simpler than the overall equation, and help
NET IONIC EQUATIONS Net ionic equations are useful in that they show only those chemical species directly participating in a chemical reaction. They are thus simpler than the overall equation, and help
More reaction types. combustions and acid/base neutralizations
 More reaction types combustions and acid/base neutralizations Combustion reactions C x H y + O 2(g) CO 2(g) + H 2 O (l) + E If the hydrocarbon contains nitrogen as well C x H y N z + O 2(g) CO 2(g) + H
More reaction types combustions and acid/base neutralizations Combustion reactions C x H y + O 2(g) CO 2(g) + H 2 O (l) + E If the hydrocarbon contains nitrogen as well C x H y N z + O 2(g) CO 2(g) + H
Chapter 3: Solution Chemistry (For best results when printing these notes, use the pdf version of this file)
 Chapter 3: Solution Chemistry (For best results when printing these notes, use the pdf version of this file) Section 3.1: Solubility Rules (For Ionic Compounds in Water) Section 3.1.1: Introduction Solubility
Chapter 3: Solution Chemistry (For best results when printing these notes, use the pdf version of this file) Section 3.1: Solubility Rules (For Ionic Compounds in Water) Section 3.1.1: Introduction Solubility
Reaction Types and Chemical Equations
 Cool Chemistry Show Section 4 Reaction Types and Chemical Equations What Do You See? Learning Outcomes In this section you will Represent chemical changes using word equations and chemical equations. Distinguish
Cool Chemistry Show Section 4 Reaction Types and Chemical Equations What Do You See? Learning Outcomes In this section you will Represent chemical changes using word equations and chemical equations. Distinguish
Ch. 8 Chemical Reactions
 Ch. 8 Chemical Reactions Intro to Reactions I II III IV V Signs of a Chemical Reaction Evolution of heat and light Formation of a gas Formation of a precipitate Color change Law of Conservation of Mass
Ch. 8 Chemical Reactions Intro to Reactions I II III IV V Signs of a Chemical Reaction Evolution of heat and light Formation of a gas Formation of a precipitate Color change Law of Conservation of Mass
Properties of Compounds
 Chapter 6. Properties of Compounds Comparing properties of elements and compounds Compounds are formed when elements combine together in fixed proportions. The compound formed will often have properties
Chapter 6. Properties of Compounds Comparing properties of elements and compounds Compounds are formed when elements combine together in fixed proportions. The compound formed will often have properties
Types of Chemical Reactions
 Types of Chemical Reactions Objectives Perform and observe the results of a variety of chemical reactions. Become familiar with the observable signs of chemical reactions. Identify the products formed
Types of Chemical Reactions Objectives Perform and observe the results of a variety of chemical reactions. Become familiar with the observable signs of chemical reactions. Identify the products formed
Chemical Reactions. Chemical Reactions Chemical reactions have a standard format when written:
 0.3.notebook A chemical property is a behaviour that occurs when substances change to create a new substance. When a new substance is created, a chemical change has occurred. New colour Evidence of chemical
0.3.notebook A chemical property is a behaviour that occurs when substances change to create a new substance. When a new substance is created, a chemical change has occurred. New colour Evidence of chemical
11.3 Reactions in Aqueous Solution. Chapter 11 Chemical Reactions Reactions in Aqueous Solution
 Chapter 11 Chemical Reactions 11.1 Describing Chemical Reactions 11.2 Types of Chemical Reactions 11.3 Reactions in Aqueous Solution 1 CHEMISTRY & YOU How did soda straws get into limestone caves? These
Chapter 11 Chemical Reactions 11.1 Describing Chemical Reactions 11.2 Types of Chemical Reactions 11.3 Reactions in Aqueous Solution 1 CHEMISTRY & YOU How did soda straws get into limestone caves? These
CHEM 103 Acid-Base Reactions
 CHEM 103 Acid-Base Reactions Lecture Notes March 2, 2006 Prof. Sevian 1 Chem 103 Please sit with your groups today. We will be doing a group problem at the end of class. 2 2005 H. Sevian 1 Agenda Recap:
CHEM 103 Acid-Base Reactions Lecture Notes March 2, 2006 Prof. Sevian 1 Chem 103 Please sit with your groups today. We will be doing a group problem at the end of class. 2 2005 H. Sevian 1 Agenda Recap:
Combustion Reactions (another example of redox) Combustion
 Combustion Reactions (another example of redox) Combustion is the burning of a fuel by rapid oxidation with oxygen in air. - General reaction for carbon based fuels is: Balancing Combustion Reactions Step
Combustion Reactions (another example of redox) Combustion is the burning of a fuel by rapid oxidation with oxygen in air. - General reaction for carbon based fuels is: Balancing Combustion Reactions Step
Identification of ions and gases Assignment
 Name: ( ) ate: lass: Identification of ions and gases ssignment 1. n excess of sodium hydroxide is added to an aqueous solution of salt X and boiled. mmonia gas is only given off after aluminium foil is
Name: ( ) ate: lass: Identification of ions and gases ssignment 1. n excess of sodium hydroxide is added to an aqueous solution of salt X and boiled. mmonia gas is only given off after aluminium foil is
1 A solution contains barium ions and silver ions and one type of anion. What could the anion be? chloride only. nitrate only.
 For more awesome resources, visit us at www.savemyexams.co.uk/ Identification of Ions and Gases Question Paper 2 Level IGSE Subject hemistry (0620/0971) Exam oard ambridge International Examinations (IE)
For more awesome resources, visit us at www.savemyexams.co.uk/ Identification of Ions and Gases Question Paper 2 Level IGSE Subject hemistry (0620/0971) Exam oard ambridge International Examinations (IE)
CHM Electrolytes and the Ionic Theory (r14) Charles Taylor 1/5
 CHM 110 - Electrolytes and the Ionic Theory (r14) - 2014 Charles Taylor 1/5 Introduction In 1884, Arrhenius proposed that some substances broke up when dissolved in water to form freely moving ions. We've
CHM 110 - Electrolytes and the Ionic Theory (r14) - 2014 Charles Taylor 1/5 Introduction In 1884, Arrhenius proposed that some substances broke up when dissolved in water to form freely moving ions. We've
flame test acidified silver nitrate solution added
 1 The results of two tests on solid X are shown. test aqueous sodium hydroxide added acidified silver nitrate added observation green precipitate formed yellow precipitate formed What is X? copper(ii)
1 The results of two tests on solid X are shown. test aqueous sodium hydroxide added acidified silver nitrate added observation green precipitate formed yellow precipitate formed What is X? copper(ii)
Equations. Chemical Reactions #1
 Equations Chemical Reactions #1 equations show the complete chemical formulas. Does not indicate ionic character equation shows all ions. Actually how the particles exist in the solution Steps for Writing
Equations Chemical Reactions #1 equations show the complete chemical formulas. Does not indicate ionic character equation shows all ions. Actually how the particles exist in the solution Steps for Writing
Reactions in Aqueous Solutions
 Reactions in Aqueous Solutions 1 Chapter 4 General Properties of Aqueous Solutions (4.1) Precipitation Reactions (4.2) Acid-Base Reactions (4.3) Oxidation-Reduction Reactions (4.4) Concentration of Solutions
Reactions in Aqueous Solutions 1 Chapter 4 General Properties of Aqueous Solutions (4.1) Precipitation Reactions (4.2) Acid-Base Reactions (4.3) Oxidation-Reduction Reactions (4.4) Concentration of Solutions
DETERMINING IONIC FORMULAS strontium oxide
 69 sodium sulfate DETERMINING IONIC FORMULAS strontium oxide tin(ii) phosphate chromium(iii) nitrate barium hydroxide titanium(iv) chloride Note: Be careful when adding subscripts to HYDROXIDE, CYANIDE,
69 sodium sulfate DETERMINING IONIC FORMULAS strontium oxide tin(ii) phosphate chromium(iii) nitrate barium hydroxide titanium(iv) chloride Note: Be careful when adding subscripts to HYDROXIDE, CYANIDE,
elemental state. There are two different possibilities: DESCRIPTION 1. One cation (+ ion) replaces another. 2. One anion (- ion) replaces another.
 CHEMICAL TYPES HANDOUT In these reactions, a free element reacts with a compound to form another compound and release one of the elements of the original compound in the elemental state. There are two
CHEMICAL TYPES HANDOUT In these reactions, a free element reacts with a compound to form another compound and release one of the elements of the original compound in the elemental state. There are two
Reactions in Aqueous Solutions
 Copyright 2004 by houghton Mifflin Company. Reactions in Aqueous Solutions Chapter 7 All rights reserved. 1 7.1 Predicting if a Rxn Will Occur When chemicals are mixed and one of these driving forces can
Copyright 2004 by houghton Mifflin Company. Reactions in Aqueous Solutions Chapter 7 All rights reserved. 1 7.1 Predicting if a Rxn Will Occur When chemicals are mixed and one of these driving forces can
Various Types of Reactions
 Various Types of Reactions Matthew Park Outline: 1. Synthesis / Replacement / Decomposition Reactions 2. Precipitation Reactions 3. Acid-Base Reactions 4. Summary: Metathesis Reactions NOTE: Not all of
Various Types of Reactions Matthew Park Outline: 1. Synthesis / Replacement / Decomposition Reactions 2. Precipitation Reactions 3. Acid-Base Reactions 4. Summary: Metathesis Reactions NOTE: Not all of
Types of Chemical Reactions
 Types of Chemical Reactions There are five types of chemical reactions: 1. Formation (combination) 2. Decomposition 3. Single Displacement 4. Double Displacement 5. Combustion 1 Formation (Combination)
Types of Chemical Reactions There are five types of chemical reactions: 1. Formation (combination) 2. Decomposition 3. Single Displacement 4. Double Displacement 5. Combustion 1 Formation (Combination)
Show your work for all questions; answer all parts of all questions. No work = no credit.
 Quiz: Ch 3 & 4 Name: Version M (32 pts) September 16, 2005 AP Chem Period: 1 2 3 4 Show your work for all questions; answer all parts of all questions. No work = no credit. 1. (10 pts) A compound contains
Quiz: Ch 3 & 4 Name: Version M (32 pts) September 16, 2005 AP Chem Period: 1 2 3 4 Show your work for all questions; answer all parts of all questions. No work = no credit. 1. (10 pts) A compound contains
ie) HCl (aq) H + (aq) + Cl - (aq) *Like all equations, dissociation equations are written in balanced form
 Acids and Bases Acids - substances which dissolve in water to form H + ions in solution ie) HCl (aq) H + (aq) + Cl - (aq) *Like all equations, dissociation equations are written in balanced form a) contain
Acids and Bases Acids - substances which dissolve in water to form H + ions in solution ie) HCl (aq) H + (aq) + Cl - (aq) *Like all equations, dissociation equations are written in balanced form a) contain
Types of Chemical Reactions
 Types of Chemical Reactions 1) Combination (Synthesis) Reaction 2) Decomposition 3) Single Replacement 4) Double Replacement 5) Combustion 6) Oxidation-Reduction (Redox) Combination (Synthesis) Reactions
Types of Chemical Reactions 1) Combination (Synthesis) Reaction 2) Decomposition 3) Single Replacement 4) Double Replacement 5) Combustion 6) Oxidation-Reduction (Redox) Combination (Synthesis) Reactions
Identification of Ions and Gases
 Identification of Ions and Gases Question Paper 1 Level IGSE Subject hemistry (0620/0971) Exam oard ambridge International Examinations (IE) Topic cids, bases and salts Sub-Topic Identification of ions
Identification of Ions and Gases Question Paper 1 Level IGSE Subject hemistry (0620/0971) Exam oard ambridge International Examinations (IE) Topic cids, bases and salts Sub-Topic Identification of ions
Ch 7 Chemical Reactions Study Guide Accelerated Chemistry SCANTRON
 Ch 7 Chemical Reactions Study Guide Accelerated Chemistry SCANTRON Name /80 TRUE/FALSE. Write 'T' if the statement is true and 'F' if the statement is false. Correct the False statments by changing the
Ch 7 Chemical Reactions Study Guide Accelerated Chemistry SCANTRON Name /80 TRUE/FALSE. Write 'T' if the statement is true and 'F' if the statement is false. Correct the False statments by changing the
Session 8: LECTURE OUTLINE (SECTIONS I1 I4 pp F61 F67)
 Session 8: LECTURE OUTLINE (SECTIONS I1 I4 pp F61 F67) I. Elecrolytes a. Soluble substances b. Insoluble substances c. Electrolytes d. Non-Electrolytes e. Ions and electrical conductivity f. Strong and
Session 8: LECTURE OUTLINE (SECTIONS I1 I4 pp F61 F67) I. Elecrolytes a. Soluble substances b. Insoluble substances c. Electrolytes d. Non-Electrolytes e. Ions and electrical conductivity f. Strong and
2. What characteristic of water makes it the universal solvent? Nonpolar large molecules long-chain hydrocarbon molecules polar
 PS Chemistry Chapter 22 & 23 Review Test Date Chapter 22 Suggestions for Studying Section 1 Know that a solution is made up of a solute and solvent. Be able to provide an example of a solute and a solvent.
PS Chemistry Chapter 22 & 23 Review Test Date Chapter 22 Suggestions for Studying Section 1 Know that a solution is made up of a solute and solvent. Be able to provide an example of a solute and a solvent.
4.5: Acids and Bases. SCH3U: Solutions and Solubility. Properties of Pure and Aqueous Substances. Arrhenius Theory. Acid:
 4.5: Acids and Bases Properties of Pure and Aqueous Substances Arrhenius Theory Acid: Acids are sour tasting and corrosive. They react readily with active metals such as Zn, Ca, Mg and carbonate-based
4.5: Acids and Bases Properties of Pure and Aqueous Substances Arrhenius Theory Acid: Acids are sour tasting and corrosive. They react readily with active metals such as Zn, Ca, Mg and carbonate-based
3/27/2015. So the question that arises is, how can you tell the difference between an ionic solution and a solution containing a molecular acid?
 A neat thing about chemistry is that there are exceptions to most rules. Previously, we learned that ionic compounds form electrolytic solutions but molecular compounds do not form electrolytic solutions.
A neat thing about chemistry is that there are exceptions to most rules. Previously, we learned that ionic compounds form electrolytic solutions but molecular compounds do not form electrolytic solutions.
The Chemistry of Acids and Bases
 The Chemistry of Acids and Bases 1 Acid and Bases 2 Acid and Bases 3 Acid and Bases 4 Acids 5 Have a sour taste. Vinegar is a solution of acetic acid. Citrus fruits contain citric acid. React with certain
The Chemistry of Acids and Bases 1 Acid and Bases 2 Acid and Bases 3 Acid and Bases 4 Acids 5 Have a sour taste. Vinegar is a solution of acetic acid. Citrus fruits contain citric acid. React with certain
Chapter 4 Reactions in Aqueous Solution
 Chapter 4 Reactions in Aqueous Solution Homework Chapter 4 11, 15, 21, 23, 27, 29, 35, 41, 45, 47, 51, 55, 57, 61, 63, 73, 75, 81, 85 1 2 Chapter Objectives Solution To understand the nature of ionic substances
Chapter 4 Reactions in Aqueous Solution Homework Chapter 4 11, 15, 21, 23, 27, 29, 35, 41, 45, 47, 51, 55, 57, 61, 63, 73, 75, 81, 85 1 2 Chapter Objectives Solution To understand the nature of ionic substances
Acids and Bases. April 10, Honors Acid and Bases Intro.notebook. Q: What does it mean for a reaction not to go to completion or equilibrium?
 Acids and Bases Unit objectives Q: What makes a solution acidic or basic? Q: What about an acid/base makes it acidic/basic? Q: How does and acid/base produce H+/OH In other words: What are the ways in
Acids and Bases Unit objectives Q: What makes a solution acidic or basic? Q: What about an acid/base makes it acidic/basic? Q: How does and acid/base produce H+/OH In other words: What are the ways in
TYPES OF CHEMICAL REACTIONS
 TYPES OF CHEMICAL REACTIONS http://www.youtube.com/watch?v=te4668aarck&feature=related The Five Major Class of Chemical Reaction http://www.youtube.com/watch?v=i HHvx1VC_8 Jan 2 8:07 PM 1 Nov 19 8:48 AM
TYPES OF CHEMICAL REACTIONS http://www.youtube.com/watch?v=te4668aarck&feature=related The Five Major Class of Chemical Reaction http://www.youtube.com/watch?v=i HHvx1VC_8 Jan 2 8:07 PM 1 Nov 19 8:48 AM
Experiments. EXPERIMENT 5 Aqueous Reactions Safety Introduction
 QC Chemistry 113.1 Laboratory Manual Version 3, Spring 2010 Experiments EXPERIMENT 5 Aqueous Reactions 5.1. Safety Aqueous solutions containing ions of transition metals should never be poured down the
QC Chemistry 113.1 Laboratory Manual Version 3, Spring 2010 Experiments EXPERIMENT 5 Aqueous Reactions 5.1. Safety Aqueous solutions containing ions of transition metals should never be poured down the
Acids and Bases. Chapters 20 and 21
 Acids and Bases Chapters 20 and 21 Acid and Bases Have a sour taste. Vinegar is a solution of acetic acid. Citrus fruits contain citric acid. React with certain metals to produce hydrogen gas. React with
Acids and Bases Chapters 20 and 21 Acid and Bases Have a sour taste. Vinegar is a solution of acetic acid. Citrus fruits contain citric acid. React with certain metals to produce hydrogen gas. React with
Science 10. Unit 2: Chemistry. Book 5: Acid -Base Chemistry & the ph Scale. Block: Name:
 Science 10 Unit 2: Chemistry Book 5: Acid -Base Chemistry & the ph Scale Name: Block: 1 Classifying Substances There are other ways you can use to classify compounds. For example, you can classify some
Science 10 Unit 2: Chemistry Book 5: Acid -Base Chemistry & the ph Scale Name: Block: 1 Classifying Substances There are other ways you can use to classify compounds. For example, you can classify some
Chapter 5 Classification and Balancing of Chemical Reactions
 Chapter 5 Classification and Balancing of Chemical Reactions 5.1 Chemical Equations Chemical equations describe chemical reactions. - As words: hydrogen plus oxygen combine to form water - As a chemical
Chapter 5 Classification and Balancing of Chemical Reactions 5.1 Chemical Equations Chemical equations describe chemical reactions. - As words: hydrogen plus oxygen combine to form water - As a chemical
11/3/09. Aqueous Solubility of Compounds. Aqueous Solubility of Ionic Compounds. Aqueous Solubility of Ionic Compounds
 Aqueous Solubility of Compounds Not all compounds dissolve in water. Solubility varies from compound to compound. Chapter 5: Chemical Reactions Soluble ionic compounds dissociate. Ions are solvated Most
Aqueous Solubility of Compounds Not all compounds dissolve in water. Solubility varies from compound to compound. Chapter 5: Chemical Reactions Soluble ionic compounds dissociate. Ions are solvated Most
Solubility Reactions. objectives
 Solubility Reactions objectives (#4 2) How do chemicals undergo a solubility reaction? (#4 2a) A student shall be able to identify if a reaction is a solubility reaction? (#4 2b) Students should be able
Solubility Reactions objectives (#4 2) How do chemicals undergo a solubility reaction? (#4 2a) A student shall be able to identify if a reaction is a solubility reaction? (#4 2b) Students should be able
Lesson #7: Introduction to Acids and Bases
 Lesson #7: Introduction to Acids and Bases Acid (As Defined by Arrhenius) Properties of Acids In 1887 Swedish Chemist Svante Arrhenius defined an Acid as: A Compound (Ionic or Molecular) that dissociates
Lesson #7: Introduction to Acids and Bases Acid (As Defined by Arrhenius) Properties of Acids In 1887 Swedish Chemist Svante Arrhenius defined an Acid as: A Compound (Ionic or Molecular) that dissociates
9/24/09 Chem 111 Experiment #7 Solutions and Reactions Brown, LeMay, and Bursten Chapter
 Chem 111 Experiment #7 Solutions and Reactions Brown, LeMay, and Bursten Chapter 4.1-4.4 KEY VOCABULARY: 1. Ionic compound a compound composed of cations (+) and anions (-). Many ionic compounds dissociate
Chem 111 Experiment #7 Solutions and Reactions Brown, LeMay, and Bursten Chapter 4.1-4.4 KEY VOCABULARY: 1. Ionic compound a compound composed of cations (+) and anions (-). Many ionic compounds dissociate
Acids and Bases. Acids
 1 Acids and Bases Acids Although some acids can burn and are dangerous to handle, most acids in foods are safe to eat. What acids have in common, however, is that they contain at least one hydrogen atom
1 Acids and Bases Acids Although some acids can burn and are dangerous to handle, most acids in foods are safe to eat. What acids have in common, however, is that they contain at least one hydrogen atom
CHEMICAL REACTIONS. The process by which one or more substances are changed into one or more different substances
 CHEMICAL REACTIONS The process by which one or more substances are changed into one or more different substances Equations Reactions are represented by a chemical equation Reactants Products Must have
CHEMICAL REACTIONS The process by which one or more substances are changed into one or more different substances Equations Reactions are represented by a chemical equation Reactants Products Must have
Preliminary Chemistry
 Name: Preliminary Chemistry Lesson 6 Water In Theory. This booklet is your best friend. Success is Contagious. Synergy Chemistry. 0466 342 939 garyzhanghsc@gmaiil.com www.hscsynergyeducation.weebly.com
Name: Preliminary Chemistry Lesson 6 Water In Theory. This booklet is your best friend. Success is Contagious. Synergy Chemistry. 0466 342 939 garyzhanghsc@gmaiil.com www.hscsynergyeducation.weebly.com
You try: 2) HC 7H 6O 2 3) N 2O 5. 5) HClO 4. 7) Rb 2C 2O 4 8) H 3PO 4 9) AgI 10) Sr(OH) 2. What kind of compound is it? NON ELECTROLYTE (NE)
 Solubility: Solubility is the measure of how much of a solute will dissolve in a solvent. In general chemistry, we usually talk about water as the solvent, so we are talking about what compounds will dissolve
Solubility: Solubility is the measure of how much of a solute will dissolve in a solvent. In general chemistry, we usually talk about water as the solvent, so we are talking about what compounds will dissolve
CHEMICAL REACTIONS. There are three ways we write chemical equations. 1. Molecular Equations 2. Full Ionic Equations 3. Net Ionic Equations
 CHEMICAL REACTIONS Reactants: Zn + I 2 Product: Zn I 2 Unit 2 Chemical Reactions The unit 2 exam will cover material from multiple chapters. You are responsible for the following from your text on exam
CHEMICAL REACTIONS Reactants: Zn + I 2 Product: Zn I 2 Unit 2 Chemical Reactions The unit 2 exam will cover material from multiple chapters. You are responsible for the following from your text on exam
Chapter 15 - Acids and Bases Fundamental Concepts
 Chapter 15 - Acids and Bases Fundamental Concepts Acids and Bases: Basic Definitions Properties of Acids Sour Taste React with active metals (Al, Zn, Fe) to yield H 2 gas: Corrosive React with carbonates
Chapter 15 - Acids and Bases Fundamental Concepts Acids and Bases: Basic Definitions Properties of Acids Sour Taste React with active metals (Al, Zn, Fe) to yield H 2 gas: Corrosive React with carbonates
Chapter 8 Acids, Bases, and Acid-Base Reactions. An Introduction to Chemistry by Mark Bishop
 Chapter 8 Acids, Bases, and Acid-Base Reactions An Introduction to Chemistry by Mark Bishop Chapter Map Arrhenius Base Definitions A base is a substance that generates OH when added to water. A basic solution
Chapter 8 Acids, Bases, and Acid-Base Reactions An Introduction to Chemistry by Mark Bishop Chapter Map Arrhenius Base Definitions A base is a substance that generates OH when added to water. A basic solution
Unit 8 Chemical Reactions- Funsheets
 Part A- Balancing Equations and Types of Reactions Balance AND identify the following reactions: Unit 8 Chemical Reactions- Funsheets 1) Mg + Zn(NO 3) 2 Zn Mg(NO 3) 2 2) Ba + AgNO 3 Ag + Ba(NO 3) 2 3)
Part A- Balancing Equations and Types of Reactions Balance AND identify the following reactions: Unit 8 Chemical Reactions- Funsheets 1) Mg + Zn(NO 3) 2 Zn Mg(NO 3) 2 2) Ba + AgNO 3 Ag + Ba(NO 3) 2 3)
Chapter 6. Acids, Bases, and Acid-Base Reactions
 Chapter 6 Acids, Bases, and Acid-Base Reactions Chapter Map Arrhenius Acid Definition Anacid is a substance that generates hydronium ions, H 3 O + (often described as H + ), when added to water. An acidic
Chapter 6 Acids, Bases, and Acid-Base Reactions Chapter Map Arrhenius Acid Definition Anacid is a substance that generates hydronium ions, H 3 O + (often described as H + ), when added to water. An acidic
SCHOOL YEAR CH- 13 IONS IN AQUEOUS SOLUTIONS AND COLLIGATIVE PROPERTIES SUBJECT: CHEMISTRY GRADE : 11 TEST A
 SCHOOL YEAR 2017-18 NAME: CH- 13 IONS IN AQUEOUS SOLUTIONS AND COLLIGATIVE PROPERTIES SUBJECT: CHEMISTRY GRADE : 11 TEST A Choose the best answer from the options that follow each question. 1. A solute
SCHOOL YEAR 2017-18 NAME: CH- 13 IONS IN AQUEOUS SOLUTIONS AND COLLIGATIVE PROPERTIES SUBJECT: CHEMISTRY GRADE : 11 TEST A Choose the best answer from the options that follow each question. 1. A solute
Acids and Bases. Feb 28 4:40 PM
 Acids and Bases H O s O Cl H O O H H N H Na O H H Feb 28 4:40 PM Properties of Acids 1. Taste sour 2. Conduct electrical current 3. Liberate H 2 gas when reacted with a metal. 4. Cause certain dyes to
Acids and Bases H O s O Cl H O O H H N H Na O H H Feb 28 4:40 PM Properties of Acids 1. Taste sour 2. Conduct electrical current 3. Liberate H 2 gas when reacted with a metal. 4. Cause certain dyes to
Solubility Rules for Ionic Compounds Arrhenius Acid Base Theory
 Chapter 4 Reactions in Aqueous Solutions Ionic compounds dissociate in water yielding electrolyte solutions. H 2 O NaCl(s) Na + (aq) + Cl - (aq) The ions are hydrated by water. Strong & Weak Electrolytes
Chapter 4 Reactions in Aqueous Solutions Ionic compounds dissociate in water yielding electrolyte solutions. H 2 O NaCl(s) Na + (aq) + Cl - (aq) The ions are hydrated by water. Strong & Weak Electrolytes
