1. 2. Differentiate states of matter. Arrangement of Particles
|
|
|
- Jerome Shepherd
- 5 years ago
- Views:
Transcription
1 Lesson 2 Predict three facts that will be discussed in Lesson 2 after reading the headings. Record your predictions in your Science Journal. Definition: Define physical property, and give two examples Differentiate states of matter. State Arrangement of Particles Motion of Particles Solid Liquid Gas Contrast three size-dependent physical properties. Circle the measurement that changes with location. Property Description Copyright McGraw-Hill Education. 14 Foundations of Chemistry
2 CHAPTER 1 Foundations of Chemistry LESSON 2 What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement or a D if you disagree. After you ve read this lesson, reread the statements to see if you have changed your mind. Key Concepts What are some physical properties of matter? How are physical properties used to separate mixtures? Before Statement After 3. The weight of a material never changes, regardless of where it is. 4. Boiling is one method used to separate parts of a mixture. Copyright McGraw-Hill Education. As you read in Lesson 1, the arrangement of atoms determines whether matter is a substance or a mixture. The arrangement of atoms also determines the properties of different types of matter. Each element and compound has a unique set of properties. When substances mix together and form mixtures, the properties of the substances that make up the mixture are still present. You can observe some properties of matter. For example, you can see that gold is shiny. You can measure other properties of matter. For example, you can measure the mass of a sample of iron. Think about how you might describe the substances and mixtures in and around a flowing mountain stream. Could you describe some of the matter as a solid or a liquid? If you picked up a handful of pebbles from the bottom of the stream, why would the water leave your hand but not the pebbles? Could you describe the mass of the various rocks in the stream? Each of these questions asks about the physical properties of matter. A physical property is a characteristic of matter that you can observe or measure without changing the identity of the matter. There are many types of physical properties. You will read about some types of physical properties in this lesson. Identify the Main Ideas Write a phrase beside each paragraph that summarizes the main point of the paragraph. Use the phrases to review the lesson. REVIEW VOCABULARY property a characteristic used to describe something 1. Observe Record two physical properties of the matter around you. Foundations of Chemistry 15
3 Make a three-tab book to record what you learn about different states of matter. Solid Liquid Gas 2. Consider How does water vapor in the air change state below the freezing point? Reading Check 3. Contrast How do solids, liquids, and gases differ? States of Matter How do aluminum, water, and air differ? Recall that aluminum is an element, water is a compound, and air is a mixture. How else do these three types of matter differ? At room temperature, aluminum is a solid, water is a liquid, and air is a gas. Solids, liquids, and gases are called states of matter. The state of matter is a physical property of matter. Substances and mixtures can be solids, liquids, or gases. For example, water in the ocean is a liquid, but water in an iceberg is a solid. In addition, water vapor in the air above the ocean is a gas. The particles (atoms or groups of atoms) that make up all matter move constantly and attract each other. Your pencil is made up of trillions of moving particles. Every solid, liquid, and gas is made up of moving particles that attract one another. The state of matter depends on how close together the particles are and how fast they move. The particles in a solid are very close together. They can move only by vibrating back and forth. This is why solids cannot easily change shape. The particles in a liquid are slightly farther apart than in a solid. Therefore, the particles can move past one another. This is why you can pour a liquid. The particles in a gas are farther apart. They move quickly and spread out to fill their container. Size-Dependent Properties State is only one of many physical properties of matter. Some physical properties, such as mass and volume, depend on the size or the amount of matter. Measurements of these properties vary depending on how much matter is in a sample. Mass Imagine holding a small dumbbell in one hand and a larger one in your other hand. What do you notice? The larger dumbbell seems heavier. The larger dumbbell has more mass than the smaller one. Mass is the amount of matter in an object. Mass is a sizedependent property of a given substance because its value depends on the size of a sample. Copyright McGraw-Hill Education. 16 Foundations of Chemistry
4 Mass and Weight An object s mass and weight are not the same. Mass is an amount of matter in something. Weight is the pull of gravity on that matter. Weight changes with location, but mass does not. Suppose a dumbbell is on the Moon. The dumbbell would have the same mass on the Moon that it has on Earth. However, the Moon s gravity is much less than Earth s gravity. As a result, the dumbbell would weigh less on the Moon than on Earth. Volume Another physical property that depends on the size or amount of a substance is volume. A unit often used to measure volume is the milliliter (ml). Volume is the amount of space something takes up. Suppose a full bottle of water contains 400 ml of water. If you pour exactly half of the water out, the bottle contains half of the original volume, or 200 ml, of water. Size-Independent Properties Some physical properties of a substance do not depend on the amount of matter present. These properties are the same for small samples and large samples. They are called sizeindependent properties. The table below and on the next page describes several physical properties of matter. The table provides examples of how physical properties can be used to separate mixtures. Notice that conductivity, boiling and melting points, state, density, solubility, and magnetism are size-independent properties. Melting Point and Boiling Point The temperature at which a substance changes from a solid to a liquid is its melting point. The temperature at which a substance changes from a liquid to a gas is its boiling point. Different substances have different boiling points and melting points. For example, the boiling point for water is 100 C at sea level. The boiling point does not change for different volumes of water. 4. Analyze Does an astronaut have more mass on Earth than in space? Why or why not? Math Skills When you compare two numbers by division, you are using a ratio. Density can be written as a ratio of mass and volume. What is the density of a substance if a 5-mL sample has a mass of 25 g? a. Set up a ratio. Mass Volume = 25 g 5 ml b. Divide the numerator by the denominator to get the mass (in g) of 1 ml. 25 g 5 ml = 5 g 1 ml c. The density is 5 g/ml. 5. Use Ratios A sample of wood has a mass of 12 g and a volume of 16 ml. What is the density of the wood? of Matter Copyright McGraw-Hill Education. Property Mass Conductivity Volume Size-dependent or size-independent Description of property How the property is used to separate a mixture (example) size-dependent size-independent size-dependent the amount of matter in an object Mass typically is not used to separate a mixture. the ability of matter to conduct, or carry along, electricity or heat Conductivity typically is not used to separate a mixture. the amount of space something occupies Volume could be used to separate mixtures whose parts can be separated by filtration. Foundations of Chemistry 17
5 Key Concept Check 6. Name What are five different physical properties of matter? Interpreting Tables 7. Consider How might you separate a mixture of iron filings and salt? Density Imagine holding a bowling ball in one hand and a foam ball of the same size in the other. The bowling ball seems heavier because the density of the material that makes up the bowling ball is greater than the density of foam. Density is the mass per unit volume of a substance. Like melting point and boiling point, density is a size-independent property. Conductivity Another property that is independent of the sample size is conductivity. Electrical conductivity is the ability of matter to conduct, or carry along, an electric current. Copper often is used for electrical wiring because it has high electrical conductivity. Thermal conductivity is the ability of a material to conduct thermal energy. Metals tend to have high electrical and thermal conductivity. Stainless steel, for example, often is used to make cooking pots because of its high thermal conductivity. However, the handles on the pan probably are made out of wood, plastic, or some other substance that has low thermal conductivity. Solubility Have you ever made lemonade by stirring a powdered drink mix into water? As you stir, the powder mixes evenly in the water. In other words, the powder dissolves in the water. What would happen if you tried to dissolve sand in water? No matter how much you stir, the sand does not dissolve. Solubility is the ability of one substance to dissolve in another. The drink powder is soluble in water, but sand is not. The table below explains how physical properties such as conductivity and solubility can be used to identify objects and separate mixtures. of Matter Boiling/Melting Points State of Matter Density Solubility Magnetism size-independent size-dependent size-independent size-dependent size-independent the temperature at which a material changes state whether something is a solid, a liquid, or a gas the amount of mass per unit of volume the ability of one substance to dissolve in another attractive force for some metals, especially iron Copyright McGraw-Hill Education. Each part of a mixture will boil or melt at a different temperature. A liquid can be poured off a solid. Objects with greater density sink in objects with less density. Dissolve a soluble material to separate it from a material with less solubility. Use a magnet to attract iron shavings from a mixture of metals. 18 Foundations of Chemistry
6 Separating Mixtures In Lesson 1, you read about different types of mixtures. Recall that the substances that make up mixtures are not held together by chemical bonds. When substances form a mixture, the properties of the individual substances do not change. You can separate the individual substances out of most mixtures by using differences in their physical properties. For example, when salt and water form a solution, the salt and the water do not lose any of their individual properties. Therefore, you can separate the salt from the water by using differences in their physical properties. Water has a lower boiling point than salt. When you boil salt water, the water evaporates, and the salt remains. You cannot use physical properties to separate a compound into the elements it contains. The atoms that make up a compound are bonded together and cannot be separated by physical means. For example, you cannot separate the hydrogen atoms from the oxygen atoms in water by boiling water. Key Concept Check 8. Explain How are physical properties used to separate mixtures? Copyright McGraw-Hill Education. Foundations of Chemistry 19
7 Mini Glossary density: the mass per unit volume of a substance mass: the amount of matter in an object physical property: a characteristic of matter that you can observe or measure without changing the identity of the matter solubility: the ability of one substance to dissolve in another 1. Review the terms and their definitions in the Mini Glossary. Write a sentence that explains how mass and weight are different. 2. Physical properties of matter can be classified as size-dependent or size-independent. Define each classification, and give at least two examples of each. size-dependent size-independent definition: definition: examples: examples: 3. If you mix sand and water in a container, the sand will sink to the bottom. Explain why. What do you think Reread the statements at the beginning of the lesson. Fill in the After column with an A if you agree with the statement or a D if you disagree. Did you change your mind? ConnectED Log on to ConnectED.mcgraw-hill.com and access your textbook to find this lesson s resources. END OF LESSON Copyright McGraw-Hill Education. 20 Foundations of Chemistry
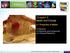
8 Lesson 2 (continued) Describe four size-independent physical properties. Melting and boiling point: Density: Size-Independent Properties Conductivity: Solubility: Identify and describe three physical properties that can be used to separate mixtures. Property How it can be used to separate a mixture Copyright McGraw-Hill Education. Synthesize It Look again at the picture of the man panning for gold on the first page of Lesson 2. Another method he could use to separate the mixture would be to sift the sediment through a screen. What physical property affects how sifting separates a mixture? Would this be as effective for finding gold as panning? Foundations of Chemistry 21
Foundations of Chemistry
 Foundations of Chemistry Physical Properties Physical Properties As you read in Lesson 1, the arrangement of atoms determines whether matter is a substance or a mixture. The arrangement of atoms also determines
Foundations of Chemistry Physical Properties Physical Properties As you read in Lesson 1, the arrangement of atoms determines whether matter is a substance or a mixture. The arrangement of atoms also determines
Matter: Properties and Changes
 CHAPTER 12 Matter: Properties and Changes Matter and Its Properties What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column
CHAPTER 12 Matter: Properties and Changes Matter and Its Properties What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column
Foundations of Chemistry
 Name Foundations of Chemistry What is matter, and how does it change? Date Before You Read Before you read the chapter, think about what you know about matter and how it changes Record three things that
Name Foundations of Chemistry What is matter, and how does it change? Date Before You Read Before you read the chapter, think about what you know about matter and how it changes Record three things that
Foundations of Chemistry
 Foundations of Chemistry Physical Changes Key Concepts How can a change in energy affect the state of matter? What happens when something dissolves? What is meant by conservation of mass? What do you think?
Foundations of Chemistry Physical Changes Key Concepts How can a change in energy affect the state of matter? What happens when something dissolves? What is meant by conservation of mass? What do you think?
Elements and Chemical Bonds
 CHAPTER 11 LESSON 3 Elements and Chemical Bonds Ionic and Metallic Bonds Key Concepts What is an ionic compound? How do metallic bonds differ from covalent and ionic bonds? What do you think? Read the
CHAPTER 11 LESSON 3 Elements and Chemical Bonds Ionic and Metallic Bonds Key Concepts What is an ionic compound? How do metallic bonds differ from covalent and ionic bonds? What do you think? Read the
States of Matter. Changes in State
 CHAPTER 8 States of Matter LESSON 2 Changes in State What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
CHAPTER 8 States of Matter LESSON 2 Changes in State What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
Elements and Chemical Bonds
 CAPTER 11 LESSN 2 Elements and Chemical Bonds Compounds, Chemical Formulas, and Covalent Bonds Key Concepts ow do elements differ from the compounds they form? What are some common properties of a covalent
CAPTER 11 LESSN 2 Elements and Chemical Bonds Compounds, Chemical Formulas, and Covalent Bonds Key Concepts ow do elements differ from the compounds they form? What are some common properties of a covalent
Before Statement After
 Thermal Energy Thermal Energy, Temperature, and Heat What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
Thermal Energy Thermal Energy, Temperature, and Heat What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
States of Matter. Solids, Liquids, and Gases
 States of Matter Solids, Liquids, and Gases What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
States of Matter Solids, Liquids, and Gases What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
Matter and Atoms. The Structure of Atoms
 CHAPTER 11 Matter and Atoms LESSON 2 The Structure of Atoms What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you
CHAPTER 11 Matter and Atoms LESSON 2 The Structure of Atoms What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you
Matter: Properties and Changes
 Matter: Properties and Changes Matter and Its Changes What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree
Matter: Properties and Changes Matter and Its Changes What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree
Matter and Atoms. Substances and Mixtures
 Matter and Atoms Substances and Mixtures What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
Matter and Atoms Substances and Mixtures What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
Minerals and Rocks. Minerals
 Minerals and Rocks Minerals What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement or a D if
Minerals and Rocks Minerals What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement or a D if
Ch. 7 Foundations of Chemistry
 Ch. 7 Foundations of Chemistry Classifying Matter Matter is anything that has mass and takes up space. Atom - smallest particle of matter 2 Main Classifications of Matter Substance Mixtures Substances
Ch. 7 Foundations of Chemistry Classifying Matter Matter is anything that has mass and takes up space. Atom - smallest particle of matter 2 Main Classifications of Matter Substance Mixtures Substances
Its Properties. Its Changes
 Chapter Introduction Lesson 1 Lesson 2 Chapter Wrap-Up Matter and Its Properties Matter and Its Changes What gives a substance its unique identity? What do you think? Before you begin, decide if you agree
Chapter Introduction Lesson 1 Lesson 2 Chapter Wrap-Up Matter and Its Properties Matter and Its Changes What gives a substance its unique identity? What do you think? Before you begin, decide if you agree
Ms. Harris Lesson Plans Week of 10/9-10/13/ th grade Science
 Ms. Harris Lesson Plans Week of 10/9-10/13/2017 8 th grade Science 4 th & 5 th hours Planning 1 st hour MONDAY: 1. Ls.1 Science Notebook for BW. Due Friday. 2. Go over Test TUESDAY: 1. Ch.1-1 Outline 2.
Ms. Harris Lesson Plans Week of 10/9-10/13/2017 8 th grade Science 4 th & 5 th hours Planning 1 st hour MONDAY: 1. Ls.1 Science Notebook for BW. Due Friday. 2. Go over Test TUESDAY: 1. Ch.1-1 Outline 2.
Properties and Structure of Matter
 Properties and Structure of Matter Chapter 10 You can use a spider map to organize the main ideas and supporting details of a topic such as properties of matter. Look at the example shown below. The central
Properties and Structure of Matter Chapter 10 You can use a spider map to organize the main ideas and supporting details of a topic such as properties of matter. Look at the example shown below. The central
States of Matter. What physical changes and energy changes occur as matter goes from one state to another?
 Name States of Matter Date What physical changes and energy changes occur as matter goes from one state to another? Before You Read Before you read the chapter, think about what you know about states of
Name States of Matter Date What physical changes and energy changes occur as matter goes from one state to another? Before You Read Before you read the chapter, think about what you know about states of
Lesson 3 Mixtures and Solutions
 Lesson 3 Mixtures and Solutions Who This lesson is designed for fifth graders ranging in age from nine to eleven. What This lesson is part of the Physical Science Unit of Instruction and based on California
Lesson 3 Mixtures and Solutions Who This lesson is designed for fifth graders ranging in age from nine to eleven. What This lesson is part of the Physical Science Unit of Instruction and based on California
Volume. measures how much space matter takes up. solubility. The amount of mass for an object is called. matter
 measures how much space matter takes up. Volume is the resistance of an object to sink, and it depends on. The ability of a substance to dissolve into another substance is called. The amount of mass for
measures how much space matter takes up. Volume is the resistance of an object to sink, and it depends on. The ability of a substance to dissolve into another substance is called. The amount of mass for
Mixtures, Solubility, and Acid/Base Solutions
 CHAPTER 9 Mixtures, Solubility, and Acid/Base Solutions Substances and Mixtures What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before
CHAPTER 9 Mixtures, Solubility, and Acid/Base Solutions Substances and Mixtures What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before
Chemical Reactions and Equations
 Chemical Reactions and Equations Changes and Chemical Reactions Key Concepts Why do chemical reactions always involve a change in? What is the difference between an endothermic reaction and an exothermic
Chemical Reactions and Equations Changes and Chemical Reactions Key Concepts Why do chemical reactions always involve a change in? What is the difference between an endothermic reaction and an exothermic
3 Mixtures. How do mixtures differ from elements and compounds? How can mixtures be separated? What are solutions, and how are they characterized?
 CHAPTER 5 3 Mixtures SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: How do mixtures differ from elements and compounds?
CHAPTER 5 3 Mixtures SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: How do mixtures differ from elements and compounds?
Mixtures, Solubility, and Acid/Base Solutions
 Mixtures, Solubility, and Acid/Base Solutions Properties of Solutions Key Concepts Why do some substances dissolve in water and others do not? How do concentration and solubility differ? How can the solubility
Mixtures, Solubility, and Acid/Base Solutions Properties of Solutions Key Concepts Why do some substances dissolve in water and others do not? How do concentration and solubility differ? How can the solubility
Making Sense of Matter Study Guide. Matter is anything that takes up space and has mass.
 Making Sense of Matter Study Guide Matter is all around us everything is made of matter. Matter is anything that takes up space and has mass. We can classify objects by their physical properties. Physical
Making Sense of Matter Study Guide Matter is all around us everything is made of matter. Matter is anything that takes up space and has mass. We can classify objects by their physical properties. Physical
Magnetism. Magnets and Magnetic Fields S N
 Magnetism Magnets and Magnetic Fields What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
Magnetism Magnets and Magnetic Fields What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
Matter and Its Changes
 Lesson 2 Matter and Its Changes 6.NS.3, 6.NS.5, 6.NS.6, 6.NS.7, 6.NS.10, 6.1.1, 6.1.2, 6.1.3 Skim or scan the heading, boldfaced words, and pictures in the lesson. Identify or predict three facts you will
Lesson 2 Matter and Its Changes 6.NS.3, 6.NS.5, 6.NS.6, 6.NS.7, 6.NS.10, 6.1.1, 6.1.2, 6.1.3 Skim or scan the heading, boldfaced words, and pictures in the lesson. Identify or predict three facts you will
Name Class Date. How do mixtures differ from elements and compounds? How can mixtures be separated? What are solutions?
 CHAPTER 3 3 Mixtures SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: How do mixtures differ from elements and compounds?
CHAPTER 3 3 Mixtures SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: How do mixtures differ from elements and compounds?
Elements and Chemical Bonds
 Elements and Chemical Bonds Electrons and Energy Levels What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree
Elements and Chemical Bonds Electrons and Energy Levels What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree
Lesson 1 Matter and Its Properties
 Lesson 1 Student Labs and Activities Page Launch Lab 8 Content Vocabulary 9 Lesson Outline 10 MiniLab 12 Content Practice A 13 Content Practice B 14 Math Skills 15 School to Home 16 Key Concept Builders
Lesson 1 Student Labs and Activities Page Launch Lab 8 Content Vocabulary 9 Lesson Outline 10 MiniLab 12 Content Practice A 13 Content Practice B 14 Math Skills 15 School to Home 16 Key Concept Builders
Motion and Forces. Forces
 CHAPTER 8 Motion and LESSON 3 What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement or a D
CHAPTER 8 Motion and LESSON 3 What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement or a D
6 th Grade Introduction to Chemistry
 Lesson 1 (Describing Matter) 6 th Grade Introduction to Chemistry Matter anything that has mass and takes up space All the stuff in the natural world is matter. Chapter 1: Introduction to Matter Chemistry
Lesson 1 (Describing Matter) 6 th Grade Introduction to Chemistry Matter anything that has mass and takes up space All the stuff in the natural world is matter. Chapter 1: Introduction to Matter Chemistry
2 Properties of Matter
 CHAPTER 2 2 Properties of Matter SECTION Matter KEY IDEAS As you read this section, keep these questions in mind: Why are color, volume, and density physical properties? Why are flammability and reactivity
CHAPTER 2 2 Properties of Matter SECTION Matter KEY IDEAS As you read this section, keep these questions in mind: Why are color, volume, and density physical properties? Why are flammability and reactivity
Name: 1. Which of the following is probably true about 300 ml of sand and 300 ml of water?
 Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
1 Classifying Matter KEY IDEAS READING TOOLBOX MATTER AND CHEMISTRY. Matter. As you read this section, keep these questions in mind: Name Class Date
 CHAPTER 2 1 Classifying Matter SECTION Matter KEY IDEAS As you read this section, keep these questions in mind: How can matter be classified? Why are elements and compounds considered pure substances?
CHAPTER 2 1 Classifying Matter SECTION Matter KEY IDEAS As you read this section, keep these questions in mind: How can matter be classified? Why are elements and compounds considered pure substances?
Elements, Compounds, and Mixtures. Matter: Properties and Changes
 Elements, Compounds, and Mixtures Matter: Properties and Changes Warm up Observe the pictures and use the following terms to name them: element, compound, or mixture. Explain your answer. Warm up answers
Elements, Compounds, and Mixtures Matter: Properties and Changes Warm up Observe the pictures and use the following terms to name them: element, compound, or mixture. Explain your answer. Warm up answers
Describing Mo tion. Position and Motion
 Describing Mo tion Position and Motion What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
Describing Mo tion Position and Motion What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
Density: The property that compares an object s mass to its volume. Mass is the measure of the amount of matter that makes up an object.
 Science Chapter 6: Matter Study Guide Lesson One: Properties of Matter A property is a characteristic of an object. You can identify properties of matter using your senses. Color, Size, Shape, Texture,
Science Chapter 6: Matter Study Guide Lesson One: Properties of Matter A property is a characteristic of an object. You can identify properties of matter using your senses. Color, Size, Shape, Texture,
The Laws of Motion. Gravity and Friction
 The Laws of Motion Gravity and Friction What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
The Laws of Motion Gravity and Friction What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
Matter Questions. Pure Substances Questions. 1. How is matter classified? 2. What is a pure substance? Give an example of a pure substance.
 Unit 6 ~ Learning Guide Name: Instructions: Using a pencil, complete the following notes as you work through the related lessons. Show ALL work as is explained in the lessons. You are required to have
Unit 6 ~ Learning Guide Name: Instructions: Using a pencil, complete the following notes as you work through the related lessons. Show ALL work as is explained in the lessons. You are required to have
Earth s Atmosphere. Energy Transfer in the Atmosphere. 3. All the energy from the Sun reaches Earth s surface.
 CHAPTER 11 LESSON 2 Earth s Atmosphere Energy Transfer in the Atmosphere Key Concepts How does energy transfer from the Sun to Earth and to the atmosphere? How are air circulation patterns within the atmosphere
CHAPTER 11 LESSON 2 Earth s Atmosphere Energy Transfer in the Atmosphere Key Concepts How does energy transfer from the Sun to Earth and to the atmosphere? How are air circulation patterns within the atmosphere
Name Class Date. How do mixtures differ from elements and compounds? How can mixtures be separated? What are solutions?
 CHAPTER 4 3 Mixtures SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: How do mixtures differ from elements and compounds?
CHAPTER 4 3 Mixtures SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: How do mixtures differ from elements and compounds?
Physical Property. Critical Thinking
 CHAPTER 1 2 Physical Properties SECTION The Properties of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What are physical properties of matter? What
CHAPTER 1 2 Physical Properties SECTION The Properties of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What are physical properties of matter? What
Thermal Energy. Thermal Energy Transfers
 Thermal Energy Thermal Energy Transfers Key Concepts What is the effect of having a small specific heat? What happens to a material when it is heated? In what ways can thermal energy be transferred? What
Thermal Energy Thermal Energy Transfers Key Concepts What is the effect of having a small specific heat? What happens to a material when it is heated? In what ways can thermal energy be transferred? What
Section 2: Properties of Matter
 Section 2: A physical property can be observed without changing the identity of the material; a chemical property can be observed when one or more new substances are formed. K What I Know W What I Want
Section 2: A physical property can be observed without changing the identity of the material; a chemical property can be observed when one or more new substances are formed. K What I Know W What I Want
Separating Mixtures. Name: Class:
 Separating Mixtures Name: Class: Directions: Match the words with the big ideas by writing the letter beside the idea. Use the same words in the short article below. Use key words and phrases from the
Separating Mixtures Name: Class: Directions: Match the words with the big ideas by writing the letter beside the idea. Use the same words in the short article below. Use key words and phrases from the
Copyright 2015 Edmentum - All rights reserved.
 Study Island Copyright 2015 Edmentum - All rights reserved. Generation Date: 03/16/2015 Generated By: Kristina Brown 1. Edgar is stranded on an island surrounded by an ocean of salt water. He is thirsty,
Study Island Copyright 2015 Edmentum - All rights reserved. Generation Date: 03/16/2015 Generated By: Kristina Brown 1. Edgar is stranded on an island surrounded by an ocean of salt water. He is thirsty,
The ability of a substance to be rolled. into wire The physical form of matter (solid, liquid, or gas)
 CHAPTER 2 2 Physical Properties SECTION The Properties of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What are physical properties of matter? What
CHAPTER 2 2 Physical Properties SECTION The Properties of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What are physical properties of matter? What
Lesson 1.2 Classifying Matter
 Lesson 1.2 Classifying Matter Vocabulary element atom chemical bond mixture molecule compound chemical formula What is Matter Made Of? What is matter? Why is one kind of matter different from another kind
Lesson 1.2 Classifying Matter Vocabulary element atom chemical bond mixture molecule compound chemical formula What is Matter Made Of? What is matter? Why is one kind of matter different from another kind
Matter and Its Properties. Unit 2
 Matter and Its Properties Unit 2 Lesson 1: Physical & Chemical Properties & Changes Unit 2: Matter and Its Properties Section 1: Physical Properties & Change Lesson 1: Physical & Chemical Properties &
Matter and Its Properties Unit 2 Lesson 1: Physical & Chemical Properties & Changes Unit 2: Matter and Its Properties Section 1: Physical Properties & Change Lesson 1: Physical & Chemical Properties &
The Laws of Motion. Newton s Second Law
 The Laws of Motion Newton s Second Law Key Concepts What is Newton s second law of motion? How does centripetal force affect circular motion? What do you think? Read the two statements below and decide
The Laws of Motion Newton s Second Law Key Concepts What is Newton s second law of motion? How does centripetal force affect circular motion? What do you think? Read the two statements below and decide
Name Class Date. Chapter Use each of the following terms in a separate sentence: physical property and physical change.
 Skills Worksheet Section Review Chapter 2-2 Physical Properties USING KEY TERMS 1. Use each of the following terms in a separate sentence: physical property and physical change. UNDERSTANDING KEY IDEAS
Skills Worksheet Section Review Chapter 2-2 Physical Properties USING KEY TERMS 1. Use each of the following terms in a separate sentence: physical property and physical change. UNDERSTANDING KEY IDEAS
Earth s Ocean Waters
 Earth s Ocean Waters BigIdeas Nearly three-quarters of Earth is covered by water, the majority of which is saltwater found in the ocean. Water has many unique properties that shape our planet and life
Earth s Ocean Waters BigIdeas Nearly three-quarters of Earth is covered by water, the majority of which is saltwater found in the ocean. Water has many unique properties that shape our planet and life
How many of you like trail mix?
 Essential Questions What is a mixture? What is a solution? What is the difference between a mixture and a solution? How can materials in a mixture or solution be separated? How many of you like trail mix?
Essential Questions What is a mixture? What is a solution? What is the difference between a mixture and a solution? How can materials in a mixture or solution be separated? How many of you like trail mix?
Elements and Chemical Bonds
 Name Elements and Chemical Bonds How do elements join together to form chemical compounds? Before You Read Before you read the chapter, think about what you know about elements and chemical bonds Record
Name Elements and Chemical Bonds How do elements join together to form chemical compounds? Before You Read Before you read the chapter, think about what you know about elements and chemical bonds Record
2 How Substances Dissolve
 CHAPTER 8 SECTION Solutions 2 How Substances Dissolve KEY IDEAS As you read this section, keep these questions in mind: Why is water called the universal solvent? How do substances dissolve? Why Do Substances
CHAPTER 8 SECTION Solutions 2 How Substances Dissolve KEY IDEAS As you read this section, keep these questions in mind: Why is water called the universal solvent? How do substances dissolve? Why Do Substances
Before Statement After
 CHAPTER 3 Minerals LESSON 1 What is a mineral? What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the
CHAPTER 3 Minerals LESSON 1 What is a mineral? What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the
Motion, Forces, and Newton s Laws
 Motion, Forces, and Newton s Laws Describing Motion What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
Motion, Forces, and Newton s Laws Describing Motion What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
Stars and Galaxies. Evolution of Stars
 Stars and Galaxies Evolution of Stars What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
Stars and Galaxies Evolution of Stars What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
Motion and Forces. Describing Motion
 CHAPTER Motion and Forces LESSON 1 Describing Motion What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
CHAPTER Motion and Forces LESSON 1 Describing Motion What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
Classifying Matter. What is matter?
 What do ice cream, root beer, and carbon dioxide gas have in common? Not only do these ingredients combine to make a good treat on a hot, summer day, but they are also made of matter. Matter can be found
What do ice cream, root beer, and carbon dioxide gas have in common? Not only do these ingredients combine to make a good treat on a hot, summer day, but they are also made of matter. Matter can be found
After you read this section, you should be able to answer these questions:
 CHAPTER 3 12 SECTION Properties of Matter Physical Properties California Science Standards 8.7.c, 8.8.a, 8.8.b, 8.8.d BEFORE YOU READ After you read this section, you should be able to answer these questions:
CHAPTER 3 12 SECTION Properties of Matter Physical Properties California Science Standards 8.7.c, 8.8.a, 8.8.b, 8.8.d BEFORE YOU READ After you read this section, you should be able to answer these questions:
Lesson 2 Changes in State
 Lesson 2 Changes in State Student Labs and Activities Page Launch Lab 25 Content Vocabulary 26 Lesson Outline 27 MiniLab 29 Content Practice A 30 Content Practice B 31 Language Arts Support 32 School to
Lesson 2 Changes in State Student Labs and Activities Page Launch Lab 25 Content Vocabulary 26 Lesson Outline 27 MiniLab 29 Content Practice A 30 Content Practice B 31 Language Arts Support 32 School to
Earth s Structure. Earth s Interior. 3. Earth s interior is made of distinct layers.
 Earth s Structure Earth s Interior Key Concepts What are the interior layers of Earth? What evidence indicates that Earth has a solid inner core and an outer liquid core? What do you think? Read the two
Earth s Structure Earth s Interior Key Concepts What are the interior layers of Earth? What evidence indicates that Earth has a solid inner core and an outer liquid core? What do you think? Read the two
2 How Substances Dissolve
 CHAPTER 8 SECTION Solutions 2 How Substances Dissolve KEY IDEAS As you read this section, keep these questions in mind: Why is water called the universal solvent? How do substances dissolve? Why Do Substances
CHAPTER 8 SECTION Solutions 2 How Substances Dissolve KEY IDEAS As you read this section, keep these questions in mind: Why is water called the universal solvent? How do substances dissolve? Why Do Substances
Matter: Properties & Change
 Matter: Properties & Change Essential Vocabulary 6.P.2.1 Recognize that all matter is made up of atoms and atoms of the same element are all alike, but are different from the atoms of other elements. 6.P.2.2
Matter: Properties & Change Essential Vocabulary 6.P.2.1 Recognize that all matter is made up of atoms and atoms of the same element are all alike, but are different from the atoms of other elements. 6.P.2.2
2.1 Properties of Matter > Chapter 2 Matter and Change. 2.1 Properties of Matter. 2.2 Mixtures 2.3 Elements and Compounds 2.4 Chemical Reactions
 21 Properties of Matter > 1 Copyright Pearson Education, Inc, or its affiliates All Rights Reserved Chapter 2 Matter and Change 21 Properties of Matter 22 Mixtures 23 Elements and Compounds 24 Chemical
21 Properties of Matter > 1 Copyright Pearson Education, Inc, or its affiliates All Rights Reserved Chapter 2 Matter and Change 21 Properties of Matter 22 Mixtures 23 Elements and Compounds 24 Chemical
Matter and Change. Monday Tuesday Wednesday Thursday Friday. Textbook Activity. Meet in B122. Hw: pg 7 Hw: pg 4. Matter Review
 Matter and Change Monday Tuesday Wednesday Thursday Friday SEPTEMBER 1 2 3 4 5 NO SCHOOL Matter & Change Classifying Matter Textbook Activity Meet in B122 Physical and Chemical Properties and Changes Indiana
Matter and Change Monday Tuesday Wednesday Thursday Friday SEPTEMBER 1 2 3 4 5 NO SCHOOL Matter & Change Classifying Matter Textbook Activity Meet in B122 Physical and Chemical Properties and Changes Indiana
SCI-5 Deane_Units1_2_SOL_Practice_Test Exam not valid for Paper Pencil Test Sessions
 SCI-5 Deane_Units1_2_SOL_Practice_Test Exam not valid for Paper Pencil Test Sessions [Exam ID:1PEFE2 1 Directions: Type your answer in the box. Use a whole number. What is the mass of the rock shown? g
SCI-5 Deane_Units1_2_SOL_Practice_Test Exam not valid for Paper Pencil Test Sessions [Exam ID:1PEFE2 1 Directions: Type your answer in the box. Use a whole number. What is the mass of the rock shown? g
ANSWER KEY. Introduction to Matter
 Chapter Project Worksheet 1 1. The object to be massed is placed on one side of the balance. Objects with known masses are placed on the other side of the arm until the balance arm is level. 2. Sample
Chapter Project Worksheet 1 1. The object to be massed is placed on one side of the balance. Objects with known masses are placed on the other side of the arm until the balance arm is level. 2. Sample
EXTRA CREDIT PAGES K
 Mixtures (13K) EXTRA CREDIT PAGES 13-14 K Elements and compounds are pure substances, but most of the materials you see every day are not. Instead, they are mixtures. A mixture is made of two or more substances
Mixtures (13K) EXTRA CREDIT PAGES 13-14 K Elements and compounds are pure substances, but most of the materials you see every day are not. Instead, they are mixtures. A mixture is made of two or more substances
Electric Charge and Electric Forces
 CHAPTER 19 Electricity LESSON 1 Electric Charge and Electric Forces What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column
CHAPTER 19 Electricity LESSON 1 Electric Charge and Electric Forces What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column
Physical and Chemical Properties
 Chapter 17 Section 17.1 Physical or Chemical Properties Physical and Chemical Properties Terms: Physical Property Physical Change Chemical Property Chemical Change Law of Conservation of Mass Physical
Chapter 17 Section 17.1 Physical or Chemical Properties Physical and Chemical Properties Terms: Physical Property Physical Change Chemical Property Chemical Change Law of Conservation of Mass Physical
Lesson 9: States of Matter
 Lesson 9: States of Matter Do Now 6O, 6S 11.8.18 Take out HW 6.14 to be checked. Copy info into CJ keep CJ out and open on desk throughout class. On Do Now Page #5, copy and answer: 1. If you use a magnet
Lesson 9: States of Matter Do Now 6O, 6S 11.8.18 Take out HW 6.14 to be checked. Copy info into CJ keep CJ out and open on desk throughout class. On Do Now Page #5, copy and answer: 1. If you use a magnet
ì<(sk$m)=bdjehh< +^-Ä-U-Ä-U
 Genre Comprehension Skill Text Features Science Content Nonfiction Predict Labels Captions Diagrams Glossary Matter Scott Foresman Science 5.11 ì
Genre Comprehension Skill Text Features Science Content Nonfiction Predict Labels Captions Diagrams Glossary Matter Scott Foresman Science 5.11 ì
S8P All of the substances on the periodic table are classified as elements because they
 S8P1-2 1. Putting sand and salt together makes A. a compound. B. an element. C. a mixture. D. a solution. 2. All of the substances on the periodic table are classified as elements because they A. are pure
S8P1-2 1. Putting sand and salt together makes A. a compound. B. an element. C. a mixture. D. a solution. 2. All of the substances on the periodic table are classified as elements because they A. are pure
Chapter 7: Kinetic Molecular Theory. 7.1 States of Matter
 Chapter 7: Kinetic Molecular Theory 7.1 States of Matter 7.1 KMT and Changes in State Matter: anything with mass and volume Mass: quantity of matter that a substance or object contains (g or kg) Volume:
Chapter 7: Kinetic Molecular Theory 7.1 States of Matter 7.1 KMT and Changes in State Matter: anything with mass and volume Mass: quantity of matter that a substance or object contains (g or kg) Volume:
Unsaved Test, Version: 1 1
 Name: Key Concepts Select the term that best completes the statement. A. beam balance B. graduated cylinder C. scale D. matter E. mass F. newton G. volume H. height I. weight J. kilogram 1. Anything that
Name: Key Concepts Select the term that best completes the statement. A. beam balance B. graduated cylinder C. scale D. matter E. mass F. newton G. volume H. height I. weight J. kilogram 1. Anything that
1 Development of the Atomic Theory
 CHAPTER 4 1 Development of the Atomic Theory SECTION Introduction to Atoms BEFORE YOU READ After you read this section, you should be able to answer these questions: What is the atomic theory? How has
CHAPTER 4 1 Development of the Atomic Theory SECTION Introduction to Atoms BEFORE YOU READ After you read this section, you should be able to answer these questions: What is the atomic theory? How has
Big Idea: Matter is descried by its properties and may undergo changes
 Big Idea: Matter is descried by its properties and may undergo changes Unit 1 Lesson 4 Pure Substances and Mixtures Essential Question: How do pure substances and mixtures compare? Copyright Houghton Mifflin
Big Idea: Matter is descried by its properties and may undergo changes Unit 1 Lesson 4 Pure Substances and Mixtures Essential Question: How do pure substances and mixtures compare? Copyright Houghton Mifflin
1 Elements. TAKE A LOOK 2. Identify Look at the illustration, and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 5 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 5 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
mixtures reflect What properties could you use to separate the ingredients in these mixtures?
 reflect Every day, we interact with many different kinds of matter. We look at it, feel it, taste it, and even breathe it. Sometimes different types of matter are combined. For example, a salad might have
reflect Every day, we interact with many different kinds of matter. We look at it, feel it, taste it, and even breathe it. Sometimes different types of matter are combined. For example, a salad might have
ì<(sk$m)=bdijag< +^-Ä-U-Ä-U
 Genre Comprehension Skill Text Features Science Content Nonfiction Compare and Contrast Captions Labels Matter Chart Glossary Scott Foresman Science 4.11 ì
Genre Comprehension Skill Text Features Science Content Nonfiction Compare and Contrast Captions Labels Matter Chart Glossary Scott Foresman Science 4.11 ì
Matter and Change. Introductory Concepts for Physical Science or Chemistry
 Matter and Change Introductory Concepts for Physical Science or Chemistry Properties of Matter Matter Anything that has mass and occupies space VOLUME WEIGHT RELIABILITY How does one describe the quantities
Matter and Change Introductory Concepts for Physical Science or Chemistry Properties of Matter Matter Anything that has mass and occupies space VOLUME WEIGHT RELIABILITY How does one describe the quantities
Matter and Its Properties
 Section 2 4A, 4B, 4C, 4D Main Ideas Atoms are the building blocks of matter. All substances have characteristic properties. Matter can be a pure substance or a mixture. 4A differentiate between physical
Section 2 4A, 4B, 4C, 4D Main Ideas Atoms are the building blocks of matter. All substances have characteristic properties. Matter can be a pure substance or a mixture. 4A differentiate between physical
Lesson 02: Physical Properties of Matter. 01 Matter
 Chemistry 11, Physical Properties, Unit 02 1 Lesson 02: Physical Properties of Matter 01 Matter Almost everything in the universe is made of matter matter has volume matter has mass matter is made up of
Chemistry 11, Physical Properties, Unit 02 1 Lesson 02: Physical Properties of Matter 01 Matter Almost everything in the universe is made of matter matter has volume matter has mass matter is made up of
Elements and Chemical Bonds. Chapter 11
 Elements and Chemical Bonds Chapter 11 Essential Question How does understanding periodic trends allow us to predict properties of different elements? Vocabulary Ionic bond Covalent bond Compounds, Chemical
Elements and Chemical Bonds Chapter 11 Essential Question How does understanding periodic trends allow us to predict properties of different elements? Vocabulary Ionic bond Covalent bond Compounds, Chemical
Matter, mass, and volume are related.
 Suppose you had all of the parts needed to make an at home aquarium: a tank, water, rocks, plants, and some fish. To put the aquarium together, you would need to arrange the rocks and plants in the tank.
Suppose you had all of the parts needed to make an at home aquarium: a tank, water, rocks, plants, and some fish. To put the aquarium together, you would need to arrange the rocks and plants in the tank.
Objectives. Inertia. Is air matter? Is Light matter? Chapter 2. Chapter 2. Table of Contents. Chapter 2. Chapter 2. Section 1 What Is Matter?
 The Properties of Matter Section 1 What Is Matter? Table of Contents Section 1 What Is Matter? Section 2 Physical Properties Section 3 Chemical Properties Objectives Describe the two properties of all
The Properties of Matter Section 1 What Is Matter? Table of Contents Section 1 What Is Matter? Section 2 Physical Properties Section 3 Chemical Properties Objectives Describe the two properties of all
Atoms and molecules are in motion and have energy
 Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Earth s Changing Surface
 Earth s Changing Surface Earthquakes and Volcanoes Key Concepts What causes earthquakes? What causes volcanoes to form? How do earthquakes and volcanoes change Earth s surface? What do you think? Read
Earth s Changing Surface Earthquakes and Volcanoes Key Concepts What causes earthquakes? What causes volcanoes to form? How do earthquakes and volcanoes change Earth s surface? What do you think? Read
Earth s Atmosphere. Describing Earth s Atmosphere
 CHAPTER 4 Earth s Atmosphere LESSON 1 Describing Earth s Atmosphere What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column
CHAPTER 4 Earth s Atmosphere LESSON 1 Describing Earth s Atmosphere What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column
Name Class Date. What is a change of state? What happens during a change of state? What can happen when a substance loses or gains energy?
 CHAPTER 2 3 Changes of State SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a change of state? What happens during a change
CHAPTER 2 3 Changes of State SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a change of state? What happens during a change
IES LAURETUM SCIENCE NAME. MIXTURES, ELEMENTS AND COMPOUNDS
 IES LAURETUM SCIENCE NAME. MIXTURES, ELEMENTS AND COMPOUNDS 1 CONTENTS 1. INTRODUCING MIXTURES 2. CLASSIFYING MIXTURES 3. SEPARATING MIXTURES 4. WHAT ARE SOLUTIONS? 5. PURE SUBSTANCES: ELEMENTS AND COMPOUNDS
IES LAURETUM SCIENCE NAME. MIXTURES, ELEMENTS AND COMPOUNDS 1 CONTENTS 1. INTRODUCING MIXTURES 2. CLASSIFYING MIXTURES 3. SEPARATING MIXTURES 4. WHAT ARE SOLUTIONS? 5. PURE SUBSTANCES: ELEMENTS AND COMPOUNDS
Lockerbie Academy. S1 Science. Elements, Compounds and Mixtures Homework Booklet
 Lockerbie Academy S Science Elements, Compounds and Mixtures Homework Booklet Homework From Lesson - Research and Design Researching Use the internet or books from a library to research the element below
Lockerbie Academy S Science Elements, Compounds and Mixtures Homework Booklet Homework From Lesson - Research and Design Researching Use the internet or books from a library to research the element below
Stars and Galaxies. The Sun and Other Stars
 CHAPTER 22 Stars and Galaxies LESSON 2 The Sun and Other Stars What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you
CHAPTER 22 Stars and Galaxies LESSON 2 The Sun and Other Stars What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you
Solids (cont.) Describe the movement of particles in a solid and the forces between them.
 Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
States of Matter. Solid. Liquid. Gas Plasma
 States of Matter Solid Liquid Gas Plasma Your turn Write four (4) examples each for Solids, Liquids, and Gases. Try to use examples you have personally been in contact with How they relate Based on what
States of Matter Solid Liquid Gas Plasma Your turn Write four (4) examples each for Solids, Liquids, and Gases. Try to use examples you have personally been in contact with How they relate Based on what
Name Date Class MATTER AND CHANGE. SECTION 2.1 PROPERTIES OF MATTER (pages 39 42)
 2 MATTER AND CHANGE SECTION 2.1 PROPERTIES OF MATTER (pages 39 42) This section helps you distinguish extensive from intensive properties and identify substances by their properties. It teaches you how
2 MATTER AND CHANGE SECTION 2.1 PROPERTIES OF MATTER (pages 39 42) This section helps you distinguish extensive from intensive properties and identify substances by their properties. It teaches you how
STUDENT NAME DATE ID GRADE 5 SCIENCE
 STUDENT NAME DATE ID GRADE 5 SCIENCE Administered December 2005 DIRECTIONS Read each question and choose the best answer. Be sure to mark all of your answers. SAMPLE A Objects That Objects That Do Conduct
STUDENT NAME DATE ID GRADE 5 SCIENCE Administered December 2005 DIRECTIONS Read each question and choose the best answer. Be sure to mark all of your answers. SAMPLE A Objects That Objects That Do Conduct
