HYDROGEN SPECTRUM. Figure 1 shows the energy level scheme for the hydrogen atom as calculated from equation. Figure 1 Figure 2
|
|
|
- Maximillian Anderson
- 5 years ago
- Views:
Transcription
1 15 Jul 04 Hydrogen.1 HYDROGEN SPECTRUM In this experiment the wavelengths of the visible emission lines of hydrogen (Balmer series) will be measured and compared to the values predicted by Bohr s quantum theory. Theory: According to the quantum theory of Niels Bohr (1913), the hydrogen atom can only exist in discrete energy states whose energies are given by: E n 4 me = 8ε h 1 n o (1) where e and m are the electron charge and mass, respectively; ε o is the dielectric permittivity of free space; h is Planck s constant; and n is the quantum number (n = 1, 2, 3,...). (1). Figure 1 shows the energy level scheme for the hydrogen atom as calculated from equation Figure 1 Figure 2 Under normal conditions, the hydrogen atom remains in its ground state (n = 1). It is possible to populate the various excited states (n > 1) by heating, electric discharge, irradiation, etc. The following decay from an excited state (n i ) to a lower-lying excited state or the ground state (n f ) is
2 15 Jul 04 Hydrogen.2 accompanied by the emission of electromagnetic radiation (a photon). The frequency of this radiation is determined by Bohr s second postulate: E = hν where E is the energy difference between the two hydrogen atom states. Using equation (1), for a transition from the initial energy state n i to the final energy state n f : E = hν = E i E f 4 me hν = 8ε h 1 n 4 me 8ε h 1 n o i o f 4 me 1 1 hν = ε h n n o f i Now ν = c/λ, so 4 1 me 1 1 = λ ε h c n n o f i = R λ 2 2 n n where R = m 1 (the Rydberg constant). f i All transitions which end at the same energy level, called final state (n f ), form a series. The various series are named in honour of their discoverers. n f = 1 is the Lyman series, n f = 2 is the Balmer series, n f = 3 is the Paschen series, n f = 4 is the Bracket series, and n f = 5 is the Pfundt series (see Figure 2). The first four transitions of the Balmer series of hydrogen can be observed as a line emission spectrum in the visible region. They are traditionally named H α (red, n i = 3), H β (blue-green, n i = 4), H γ (violet, n i = 5), and H δ (violet, n i = 6). The H δ line is of very low intensity and so the lines originating in higher energy states are of even lower intensity. Apparatus: The apparatus used to observe the spectrum consists of a spectrometer and a diffraction grating. A diffraction grating is a piece of transparent material with very fine, closely-spaced rulings or grooves cut or moulded into it.
3 15 Jul 04 Hydrogen.3 The grating is extremely fragile and must not be touched. When a monochromatic beam of light is incident on the grating, an interference pattern is formed as the beam passes through each of the grooves. The pattern consists of sharp bright lines whose angular positions are determined by mλ = d sinθ provided the grating is perpendicular to the incident light. m is the order of the line (m = 0, ±1, ±2,...); λ is the wavelength; d is the spacing between the rulings on the grating; and θ is the deviation angle of the transmitted beam. If the incident beam is composed of light of different wavelengths, these wavelengths will be separated into distinct lines. Figure 3 shows the spectrum formed by light of different wavelengths passing through a grating. Figure 3 The hydrogen source is an electric discharge tube filled with water vapour. The discharge is confined in a capillary tube 1 mm in diameter and 50 mm long, thus providing a high intensity line source. The tube is powered by 5000 V so use extreme caution when near the power supply and tube. Figure 4 shows the arrangement of the grating spectrometer.
4 15 Jul 04 Hydrogen.4 Figure 4 Note the locknuts and fine adjustment screws on the spectrometer. Note that the vernier scale yields measurements in decimal degrees to an accuracy of 0.1. A mercury light source is provided for calibration of the grating spectrometer. Procedure and Experiment: 1. Determine the controls which allow locking and unlocking of the grating table, locking and unlocking of the spectrometer table, and fine and coarse positioning of the telescope. 2. Practise reading the vernier scale until you are confident of your measurements. 3. Place the collimator slit of the spectrometer about 0.5 cm from the mercury light source. Remember, do not touch the diffraction grating. 4. For the grating equation, mλ = d sinθ, to be applicable, the grating must be perpendicular to the incident light. a) Set the spectrometer to view the slit directly as shown in Figure 5a. b) Unlock the spectrometer table and set it to read 0, then lock it. c) Unlock the telescope and swing it clockwise through 90 ; lock it. d) Unlock the grating table and rotate the grating so as to view the reflected image of the slit. This places the grating at 45 to the incident light at shown in Figure 5b. Lock the grating table. e) Unlock the spectrometer table and swing it through 135 so that the grating is perpendicular to the incident light and on the side furthest from the slit. See Figure 5c. f) Lock the spectrometer table and unlock the telescope. You are now ready to make measurements.
5 15 Jul 04 Hydrogen.5 Figure 5 5. The grating constant d is determined by using light of known wavelength, measuring the angle of diffraction, and using mλ = d sinθ. 6. In first order, measure the angular locations, φ 1 and φ 2, of the green line of mercury on each side of the incident direction. See Figure 6. θ = φ φ Figure 6
6 15 Jul 04 Hydrogen.6 Measure the locations of the violet 1, blue 3, and one of the yellow lines of mercury as well. Using the wavelength values given in the following table, calculate a d value for each line, then average your results to obtain the grating constant value that will be used for the remainder of the experiment. Mercury Spectrum Colour Wavelength (nm) Violet Violet Blue Blue Blue Blue-Green Blue-Green Green Green-Yellow Yellow Yellow Orange Orange Red Red Relative Intensity 7. Replace the mercury source with the hydrogen source. Measure the angular positions on each side of the incident direction, and calculate the wavelengths, in as many orders as possible for the H α, H β, H γ, and H δ hydrogen lines. Other lines may appear due to impurities in the source but these will be easily distinguishable from the hydrogen lines. The H α (red), H β (bluegreen), and H γ (violet) lines should be measurable in at least two orders, while the H δ (faint violet) will likely only be visible in first order. To facilitate observation of the spectral lines, turn off the room and hall lights and drape the black cloth over the spectrometer. 8. Compare the average experimental wavelength values for the H α, H β, H γ, and H δ lines with those calculated theoretically from the Bohr equation.
HYDROGEN SPECTRUM = 2
 MP6 OBJECT 3 HYDROGEN SPECTRUM MP6. The object o this experiment is to observe some o the lines in the emission spectrum o hydrogen, and to compare their experimentally determined wavelengths with those
MP6 OBJECT 3 HYDROGEN SPECTRUM MP6. The object o this experiment is to observe some o the lines in the emission spectrum o hydrogen, and to compare their experimentally determined wavelengths with those
Any first year text, sections on atomic structure, spectral lines and spectrometers
 Physics 33 Experiment 5 Atomic Spectra References Any first year text, sections on atomic structure, spectral lines and spectrometers Any modern physics text, eg F.K. Richtmeyer, E.H. Kennard and J.N.
Physics 33 Experiment 5 Atomic Spectra References Any first year text, sections on atomic structure, spectral lines and spectrometers Any modern physics text, eg F.K. Richtmeyer, E.H. Kennard and J.N.
Physics 23 Fall 1998 Lab 4 - The Hydrogen Spectrum
 Physics 3 Fall 998 Lab 4 - The Hydrogen Spectrum Theory In the late 800's, it was known that when a gas is excited by means of an electric discharge and the light emitted is viewed through a diffraction
Physics 3 Fall 998 Lab 4 - The Hydrogen Spectrum Theory In the late 800's, it was known that when a gas is excited by means of an electric discharge and the light emitted is viewed through a diffraction
The Quantum Model of the Hydrogen Atom
 Physics 109 Science 1 Experiment 1 1 The Quantum Model of the Hydrogen Atom In this experiment you will use a spectrometer to determine the wavelengths of the visible lines of atomic hydrogen. The goal
Physics 109 Science 1 Experiment 1 1 The Quantum Model of the Hydrogen Atom In this experiment you will use a spectrometer to determine the wavelengths of the visible lines of atomic hydrogen. The goal
Lab 5: Spectroscopy & the Hydrogen Atom Phy248 Spring 2009
 Lab 5: Spectroscopy & the Hydrogen Atom Phy248 Spring 2009 Name Section Return this spreadsheet to your TA that will use it to score your lab. To receive full credit you must use complete sentences and
Lab 5: Spectroscopy & the Hydrogen Atom Phy248 Spring 2009 Name Section Return this spreadsheet to your TA that will use it to score your lab. To receive full credit you must use complete sentences and
EXPERIMENT 12 THE GRATING SPECTROMETER AND ATOMIC SPECTRA
 OBJECTIVES Learn the theory of the grating spectrometer Observe the spectrum of mercury and hydrogen Measure the grating constant of a diffraction grating Measure the Rydberg Constant EXPERIMENT THE GRATING
OBJECTIVES Learn the theory of the grating spectrometer Observe the spectrum of mercury and hydrogen Measure the grating constant of a diffraction grating Measure the Rydberg Constant EXPERIMENT THE GRATING
Ph 3455/MSE 3255 Experiment 2: Atomic Spectra
 Ph 3455/MSE 3255 Experiment 2: Atomic Spectra Background Reading: Tipler, Llewellyn pp. 163-165 Apparatus: Spectrometer, sodium lamp, hydrogen lamp, mercury lamp, diffraction grating, watchmaker eyeglass,
Ph 3455/MSE 3255 Experiment 2: Atomic Spectra Background Reading: Tipler, Llewellyn pp. 163-165 Apparatus: Spectrometer, sodium lamp, hydrogen lamp, mercury lamp, diffraction grating, watchmaker eyeglass,
Laboratory #29: Spectrometer
 INDIANA UNIVERSITY, DEPARTMENT OF PHYSICS, P309 LABORATORY Laboratory #29: Spectrometer Goal: Learn to adjust an optical spectrometer, use a transmission grating to measure known spectral lines of mercury,
INDIANA UNIVERSITY, DEPARTMENT OF PHYSICS, P309 LABORATORY Laboratory #29: Spectrometer Goal: Learn to adjust an optical spectrometer, use a transmission grating to measure known spectral lines of mercury,
The Emission Spectra of Light
 The Emission Spectra of Light Objectives: Theory: 1.... measured the wavelength limits of the color bands in the visible spectrum, 2.... measured the wavelengths of the emission lines of the hydrogen Balmer
The Emission Spectra of Light Objectives: Theory: 1.... measured the wavelength limits of the color bands in the visible spectrum, 2.... measured the wavelengths of the emission lines of the hydrogen Balmer
PC1144 Physics IV. Atomic Spectra
 PC1144 Physics IV Atomic Spectra 1 Objectives Investigate how well the visible light wavelengths of hydrogen predicted by the Bohr theory agree with experimental values. Determine an experimental value
PC1144 Physics IV Atomic Spectra 1 Objectives Investigate how well the visible light wavelengths of hydrogen predicted by the Bohr theory agree with experimental values. Determine an experimental value
ATOMIC SPECTRA. To identify elements through their emission spectra. Apparatus: spectrometer, spectral tubes, power supply, incandescent lamp.
 ATOMIC SPECTRA Objective: To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify elements through
ATOMIC SPECTRA Objective: To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify elements through
THE DIFFRACTION GRATING SPECTROMETER
 Purpose Theory THE DIFFRACTION GRATING SPECTROMETER a. To study diffraction of light using a diffraction grating spectrometer b. To measure the wavelengths of certain lines in the spectrum of the mercury
Purpose Theory THE DIFFRACTION GRATING SPECTROMETER a. To study diffraction of light using a diffraction grating spectrometer b. To measure the wavelengths of certain lines in the spectrum of the mercury
Observation of Atomic Spectra
 Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
Lab 10: Spectroscopy & the Hydrogen Atom Phy208 Fall 2008
 Lab 10: Spectroscopy & the Hydrogen Atom Phy208 Fall 2008 Name Section This sheet is the lab document your TA will use to score your lab. It is to be turned in at the end of lab. To receive full credit
Lab 10: Spectroscopy & the Hydrogen Atom Phy208 Fall 2008 Name Section This sheet is the lab document your TA will use to score your lab. It is to be turned in at the end of lab. To receive full credit
Diffraction of light by a grating
 (ta initials) first name (print) last name (print) brock id (ab17cd) (lab date) Experiment 5 Diffraction of light by a grating In this Experiment you will learn the geometical analysis of a diffraction
(ta initials) first name (print) last name (print) brock id (ab17cd) (lab date) Experiment 5 Diffraction of light by a grating In this Experiment you will learn the geometical analysis of a diffraction
PHYS General Physics II Lab The Balmer Series for Hydrogen Source. c = speed of light = 3 x 10 8 m/s
 PHYS 1040 - General Physics II Lab The Balmer Series for Hydrogen Source Purpose: The purpose of this experiment is to analyze the emission of light from a hydrogen source and measure and the wavelengths
PHYS 1040 - General Physics II Lab The Balmer Series for Hydrogen Source Purpose: The purpose of this experiment is to analyze the emission of light from a hydrogen source and measure and the wavelengths
Lab report 30 EXPERIMENT 4. REFRACTION OF LIGHT
 30 EXPERIMENT 4. REFRACTION OF LIGHT Lab report Go to your course homepage on Sakai (Resources, Lab templates) to access the online lab report worksheet for this experiment. The worksheet has to be completed
30 EXPERIMENT 4. REFRACTION OF LIGHT Lab report Go to your course homepage on Sakai (Resources, Lab templates) to access the online lab report worksheet for this experiment. The worksheet has to be completed
ATOMIC SPECTRA. Objective:
 1 ATOMIC SPECTRA Objective: To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify an unknown
1 ATOMIC SPECTRA Objective: To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify an unknown
The Grating Spectrometer and Atomic Spectra
 PHY 192 Grating Spectrometer Spring 2012 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating
PHY 192 Grating Spectrometer Spring 2012 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating
Atomic emission spectra experiment
 Atomic emission spectra experiment Contents 1 Overview 1 2 Equipment 1 3 Measuring the grating spacing using the sodium D-lines 4 4 Measurement of hydrogen lines and the Rydberg Constant 5 5 Measurement
Atomic emission spectra experiment Contents 1 Overview 1 2 Equipment 1 3 Measuring the grating spacing using the sodium D-lines 4 4 Measurement of hydrogen lines and the Rydberg Constant 5 5 Measurement
high energy state for the electron in the atom low energy state for the electron in the atom
 Atomic Spectra Objectives The objectives of this experiment are to: 1) Build and calibrate a simple spectroscope capable of measuring wavelengths of visible light. 2) Measure several wavelengths of light
Atomic Spectra Objectives The objectives of this experiment are to: 1) Build and calibrate a simple spectroscope capable of measuring wavelengths of visible light. 2) Measure several wavelengths of light
Experiment 7: Spectrum of the Hydrogen Atom
 Experiment 7: Spectrum of the Hydrogen Nate Saffold nas2173@columbia.edu Office Hour: Mondays, 5:30-6:30PM INTRO TO EXPERIMENTAL PHYS-LAB 1493/1494/2699 Introduction The physics behind: The spectrum of
Experiment 7: Spectrum of the Hydrogen Nate Saffold nas2173@columbia.edu Office Hour: Mondays, 5:30-6:30PM INTRO TO EXPERIMENTAL PHYS-LAB 1493/1494/2699 Introduction The physics behind: The spectrum of
Atomic Spectra. d sin θ = mλ (1)
 Atomic Spectra Objectives: To measure the wavelengths of visible light emitted by atomic hydrogen and verify that the measured wavelengths obey the empirical Rydberg formula. To observe emission spectra
Atomic Spectra Objectives: To measure the wavelengths of visible light emitted by atomic hydrogen and verify that the measured wavelengths obey the empirical Rydberg formula. To observe emission spectra
The Grating Spectrometer and Atomic Spectra
 PHY 192 Grating Spectrometer 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating was introduced.
PHY 192 Grating Spectrometer 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating was introduced.
Pre-lab Quiz/PHYS 224. Your name Lab section
 Pre-lab Quiz/PHYS 224 THE DIFFRACTION GRATING AND THE OPTICAL SPECTRUM Your name Lab section 1. What are the goals of this experiment? 2. If the period of a diffraction grating is d = 1,000 nm, where the
Pre-lab Quiz/PHYS 224 THE DIFFRACTION GRATING AND THE OPTICAL SPECTRUM Your name Lab section 1. What are the goals of this experiment? 2. If the period of a diffraction grating is d = 1,000 nm, where the
Atomic Spectra. Eric Reichwein David Steinberg Department of Physics University of California, Santa Cruz. August 30, 2012
 Atomic Spectra Eric Reichwein David Steinberg Department of Physics University of California, Santa Cruz August 30, 0 Abstract To observe helium spectral lines we used a spectrometer. From a table of known
Atomic Spectra Eric Reichwein David Steinberg Department of Physics University of California, Santa Cruz August 30, 0 Abstract To observe helium spectral lines we used a spectrometer. From a table of known
Spectrum of Hydrogen. Physics 227 Lab
 Introduction In today's lab you will be dealing with an area of physics called quantum mechanics. The only quantum mechanical idea that you will be using today is that electrons in an atom can exist only
Introduction In today's lab you will be dealing with an area of physics called quantum mechanics. The only quantum mechanical idea that you will be using today is that electrons in an atom can exist only
University of Massachusetts, Amherst
 PHYSICS 286: Modern Physics Laboratory SPRING 2010 (A. Dinsmore and K. Kumar) Feb 2009 Experiment 4: THE FRANCK HERTZ EXPERIMENT Electronic Excitations of a Gas, and Evidence for the Quantization of Atomic
PHYSICS 286: Modern Physics Laboratory SPRING 2010 (A. Dinsmore and K. Kumar) Feb 2009 Experiment 4: THE FRANCK HERTZ EXPERIMENT Electronic Excitations of a Gas, and Evidence for the Quantization of Atomic
Experiment 24: Spectroscopy
 Experiment 24: Spectroscopy Figure 24.1: Spectroscopy EQUIPMENT High Voltage Power Supply Incandescent Light Source (3) Gas Discharge Tubes: 1. Helium 2. Hydrogen 3. Unknown Element Spectrometer Felt (1)
Experiment 24: Spectroscopy Figure 24.1: Spectroscopy EQUIPMENT High Voltage Power Supply Incandescent Light Source (3) Gas Discharge Tubes: 1. Helium 2. Hydrogen 3. Unknown Element Spectrometer Felt (1)
Atomic Spectra 1. Name Date Partners ATOMIC SPECTRA
 Atomic Spectra 1 Name Date Partners ATOMIC SPECTRA Sodium Mercury Lithium Hydrogen Atomic line spectra are characteristic for every element. These are emission spectra (without color). OBJECTIVES Review
Atomic Spectra 1 Name Date Partners ATOMIC SPECTRA Sodium Mercury Lithium Hydrogen Atomic line spectra are characteristic for every element. These are emission spectra (without color). OBJECTIVES Review
Physics 24, Spring 2007 Lab 2 - Complex Spectra
 Physics 24, Spring 2007 Lab 2 - Complex Spectra Theory The optical spectra of isolated atoms consist of discrete, unequally spaced lines. This fact could not be understood on the basis of classical atomic
Physics 24, Spring 2007 Lab 2 - Complex Spectra Theory The optical spectra of isolated atoms consist of discrete, unequally spaced lines. This fact could not be understood on the basis of classical atomic
THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY
 THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY Warning: The mercury spectral lamps emit UV radiation. Do not stare into the lamp. Avoid exposure where possible. Introduction
THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY Warning: The mercury spectral lamps emit UV radiation. Do not stare into the lamp. Avoid exposure where possible. Introduction
EXPERIMENT 14. The Atomic Spectrum of Hydrogen
 Name: Laboratory Section: Laboratory Section Date: Partners Names: Grade: Last Revised on March 18, 2003 EXPERIMENT 14 The Atomic Spectrum of Hydrogen 0. Pre-Laboratory Work [2 pts] 1. You will be using
Name: Laboratory Section: Laboratory Section Date: Partners Names: Grade: Last Revised on March 18, 2003 EXPERIMENT 14 The Atomic Spectrum of Hydrogen 0. Pre-Laboratory Work [2 pts] 1. You will be using
Atomic Spectra & Electron Energy Levels
 CHM151LL: ATOMIC SPECTRA & ELECTRON ENERGY LEVELS 1 Atomic Spectra & Electron Energy Levels OBJECTIVES: To measure the wavelength of visible light emitted by excited atoms to calculate the energy of that
CHM151LL: ATOMIC SPECTRA & ELECTRON ENERGY LEVELS 1 Atomic Spectra & Electron Energy Levels OBJECTIVES: To measure the wavelength of visible light emitted by excited atoms to calculate the energy of that
Chapter 8. Spectroscopy. 8.1 Purpose. 8.2 Introduction
 Chapter 8 Spectroscopy 8.1 Purpose In the experiment atomic spectra will be investigated. The spectra of three know materials will be observed. The composition of an unknown material will be determined.
Chapter 8 Spectroscopy 8.1 Purpose In the experiment atomic spectra will be investigated. The spectra of three know materials will be observed. The composition of an unknown material will be determined.
Rydberg constant from atomic spectra of gases
 Page 1 of 8 Rydberg constant from atomic spectra of gases Objective - Calibrating a prism spectrometer to convert the scale readings in wavelengths of spectral lines. - Observing the Balmer series of atomic
Page 1 of 8 Rydberg constant from atomic spectra of gases Objective - Calibrating a prism spectrometer to convert the scale readings in wavelengths of spectral lines. - Observing the Balmer series of atomic
Diffraction Gratings, Atomic Spectra. Prof. Shawhan (substituting for Prof. Hall) November 14, 2016
 Diffraction Gratings, Atomic Spectra Prof. Shawhan (substituting for Prof. Hall) November 14, 2016 1 Increase number of slits: 2 Visual Comparisons 3 4 8 2 Diffraction Grating Note: despite the name, this
Diffraction Gratings, Atomic Spectra Prof. Shawhan (substituting for Prof. Hall) November 14, 2016 1 Increase number of slits: 2 Visual Comparisons 3 4 8 2 Diffraction Grating Note: despite the name, this
Introduction. Procedure and Data
 Introduction The spectrum is the entire range over which some measurable property of a physical system of phenomenon can vary. Systems that have spectrums include sound frequency, electromagnetic radiation
Introduction The spectrum is the entire range over which some measurable property of a physical system of phenomenon can vary. Systems that have spectrums include sound frequency, electromagnetic radiation
Emission Spectroscopy
 Objectives Emission Spectroscopy Observe spectral lines from a hydrogen gas discharge tube Determine the initial and final energy levels for the electronic transitions associated with the visible portion
Objectives Emission Spectroscopy Observe spectral lines from a hydrogen gas discharge tube Determine the initial and final energy levels for the electronic transitions associated with the visible portion
Stellar Astrophysics: The Interaction of Light and Matter
 Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of hydrogen atom.
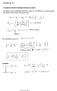 Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
LAB 12 ATOMIC SPECTRA
 217 Name Date Partners LAB 12 ATOMIC SPECTRA OBJECTIVES Review the quantum nature of light and how light photons are produced in atoms. Learn to use an optical spectrometer to measure light wavelengths.
217 Name Date Partners LAB 12 ATOMIC SPECTRA OBJECTIVES Review the quantum nature of light and how light photons are produced in atoms. Learn to use an optical spectrometer to measure light wavelengths.
PHYSICS 122/124 Lab EXPERIMENT NO. 9 ATOMIC SPECTRA
 PHYSICS 1/14 Lab EXPERIMENT NO. 9 ATOMIC SPECTRA The purpose of this laboratory is to study energy levels of the Hydrogen atom by observing the spectrum of emitted light when Hydrogen atoms make transitions
PHYSICS 1/14 Lab EXPERIMENT NO. 9 ATOMIC SPECTRA The purpose of this laboratory is to study energy levels of the Hydrogen atom by observing the spectrum of emitted light when Hydrogen atoms make transitions
The Hydrogen Spectrum
 1 The Hydrogen Spectrum PHYS 1301 F98 Prof. T.E. Coan Last edit: 6 Aug 98 Introduction In last week's laboratory experiment on diffraction, you should have noticed that the light from the mercury discharge
1 The Hydrogen Spectrum PHYS 1301 F98 Prof. T.E. Coan Last edit: 6 Aug 98 Introduction In last week's laboratory experiment on diffraction, you should have noticed that the light from the mercury discharge
DISPERSION OF A GLASS PRISM
 PH2 page 1 DISPERSION OF A GLASS PRISM OBJECTIVE The objective of this experiment is to analyze the emission spectrum of helium and to analyze the dispersion of a glass prism by measuring the index of
PH2 page 1 DISPERSION OF A GLASS PRISM OBJECTIVE The objective of this experiment is to analyze the emission spectrum of helium and to analyze the dispersion of a glass prism by measuring the index of
Emission Spectrum of Atomic Gases. Prelab Questions
 Emission Spectrum of Atomic Gases Prelab Questions Before this coming to this lab, please review your text for the physics of the spectrum of visible light and of diffraction grating spectrometer.. Which
Emission Spectrum of Atomic Gases Prelab Questions Before this coming to this lab, please review your text for the physics of the spectrum of visible light and of diffraction grating spectrometer.. Which
Energy levels and atomic structures lectures chapter one
 Structure of Atom An atom is the smallest constituent unit of ordinary matter that has the properties of a element. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are
Structure of Atom An atom is the smallest constituent unit of ordinary matter that has the properties of a element. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are
Atomic Emission Spectra
 Atomic Emission Spectra Objectives The objectives of this laboratory are as follows: To build and calibrate a simple meter-stick spectroscope that is capable of measuring wavelengths of visible light.
Atomic Emission Spectra Objectives The objectives of this laboratory are as follows: To build and calibrate a simple meter-stick spectroscope that is capable of measuring wavelengths of visible light.
Determination of Cauchy s Contants
 8. Determination of Cauchy s Contants 8.1 Objective: To determine Cauchy s Constants using a prism and spectrometer. Apparatus: Glass prism, spectrometer and mercury vapour lamp. 8. Theory: The wavelength
8. Determination of Cauchy s Contants 8.1 Objective: To determine Cauchy s Constants using a prism and spectrometer. Apparatus: Glass prism, spectrometer and mercury vapour lamp. 8. Theory: The wavelength
EXPERIMENT 17: Atomic Emission
 EXPERIMENT 17: Atomic Emission PURPOSE: To construct an energy level diagram of the hydrogen atom To identify an element from its line spectrum. PRINCIPLES: White light, such as emitted by the sun or an
EXPERIMENT 17: Atomic Emission PURPOSE: To construct an energy level diagram of the hydrogen atom To identify an element from its line spectrum. PRINCIPLES: White light, such as emitted by the sun or an
Quantum Physics Objective: Apparatus:
 1 Quantum Physics Objective: 1. To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify an unknown
1 Quantum Physics Objective: 1. To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify an unknown
Chapter 37 Early Quantum Theory and Models of the Atom
 Chapter 37 Early Quantum Theory and Models of the Atom Units of Chapter 37 37-7 Wave Nature of Matter 37-8 Electron Microscopes 37-9 Early Models of the Atom 37-10 Atomic Spectra: Key to the Structure
Chapter 37 Early Quantum Theory and Models of the Atom Units of Chapter 37 37-7 Wave Nature of Matter 37-8 Electron Microscopes 37-9 Early Models of the Atom 37-10 Atomic Spectra: Key to the Structure
NORTHERN ILLINOIS UNIVERSITY PHYSICS DEPARTMENT. Physics 211 E&M and Quantum Physics Spring Lab #9: Diffraction Spectroscopy
 NORTHERN ILLINOIS UNIVERSITY PHYSICS DEPARTMENT Physics 211 E&M and Quantum Physics Spring 2018 Lab #9: Diffraction Spectroscopy Lab Writeup Due: Mon/Wed/Thu/Fri, April 30/ May 2/3/4, 2018 Background All
NORTHERN ILLINOIS UNIVERSITY PHYSICS DEPARTMENT Physics 211 E&M and Quantum Physics Spring 2018 Lab #9: Diffraction Spectroscopy Lab Writeup Due: Mon/Wed/Thu/Fri, April 30/ May 2/3/4, 2018 Background All
To determine the wavelengths of light emitted by a mercury vapour lamp by using a diffraction grating.
 12. Diffraction grating OBJECT To determine the wavelengths of light emitted by a mercury vapour lamp by using a diffraction grating. INTRODUCTION: Consider a light beam transmitted through an aperture
12. Diffraction grating OBJECT To determine the wavelengths of light emitted by a mercury vapour lamp by using a diffraction grating. INTRODUCTION: Consider a light beam transmitted through an aperture
Using the Spectrometer
 Using the Spectrometer Introduction When an atom is stimulated it can respond by emitting a spectrum of light. The spectrum comprises discrete wavelengths whose values are characteristic of the particular
Using the Spectrometer Introduction When an atom is stimulated it can respond by emitting a spectrum of light. The spectrum comprises discrete wavelengths whose values are characteristic of the particular
10.1 Introduction: The Bohr Hydrogen Atom
 Chapter 10 Experiment 8: The Spectral Nature of Light 10.1 Introduction: The Bohr Hydrogen Atom Historical Aside The 19 th century was a golden age of science. We could explain everything we observed.
Chapter 10 Experiment 8: The Spectral Nature of Light 10.1 Introduction: The Bohr Hydrogen Atom Historical Aside The 19 th century was a golden age of science. We could explain everything we observed.
Quantum Theory of the Atom
 The Wave Nature of Light Quantum Theory of the Atom Electromagnetic radiation carries energy = radiant energy some forms are visible light, x rays, and radio waves Wavelength ( λ) is the distance between
The Wave Nature of Light Quantum Theory of the Atom Electromagnetic radiation carries energy = radiant energy some forms are visible light, x rays, and radio waves Wavelength ( λ) is the distance between
Visible Spectrometer
 Visible Spectrometer Experiment VIS University of Florida Department of Physics PHY4803L Advanced Physics Laboratory Objective The Balmer spectral lines from a hydrogen discharge lamp are observed with
Visible Spectrometer Experiment VIS University of Florida Department of Physics PHY4803L Advanced Physics Laboratory Objective The Balmer spectral lines from a hydrogen discharge lamp are observed with
Atomic Spectroscopy. Absorption and Emission Spectra. Lodovico Lappetito. SpettroscopiaAtomica - 15/07/2015 Pag. 1
 Atomic Spectroscopy Absorption and Emission Spectra Lodovico Lappetito SpettroscopiaAtomica - 15/07/2015 Pag. 1 Table of Contents Atomic Spectra... 3 Diffraction Grating Spectrometer... 4 Spectral Lamps...
Atomic Spectroscopy Absorption and Emission Spectra Lodovico Lappetito SpettroscopiaAtomica - 15/07/2015 Pag. 1 Table of Contents Atomic Spectra... 3 Diffraction Grating Spectrometer... 4 Spectral Lamps...
UNIVERSITY OF CALIFORNIA - SANTA CRUZ DEPARTMENT OF PHYSICS PHYS 133 PROFESSOR: SHER. Atomic Spectra. Benjamin Stahl
 UNIVERSITY OF CALIFORNIA - SANTA CRUZ DEPARTMENT OF PHYSICS PHYS 133 PROFESSOR: SHER Atomic Spectra Benjamin Stahl Lab Partners: Aaron Lopez & Dillon Teal April 2, 2014 Abstract As an introduction to spectroscopy,
UNIVERSITY OF CALIFORNIA - SANTA CRUZ DEPARTMENT OF PHYSICS PHYS 133 PROFESSOR: SHER Atomic Spectra Benjamin Stahl Lab Partners: Aaron Lopez & Dillon Teal April 2, 2014 Abstract As an introduction to spectroscopy,
Name Date: Course number: MAKE SURE TA & TI STAMPS EVERY PAGE BEFORE YOU START EXPERIMENT 14. The Atomic Spectrum of Hydrogen
 Laboratory Section: Last Revised on September 21, 2016 Partners Names: Grade: EXPERIMENT 14 The Atomic Spectrum of Hydrogen 0. Pre-Laboratory Work [2 pts] 1. You will be using a diffraction grating in
Laboratory Section: Last Revised on September 21, 2016 Partners Names: Grade: EXPERIMENT 14 The Atomic Spectrum of Hydrogen 0. Pre-Laboratory Work [2 pts] 1. You will be using a diffraction grating in
THE ATOMIC SPECTRUM OF HYDROGEN
 THE ATOMIC SPECTRUM OF HYDROGEN When atoms are excited, either in an electric discharge or with heat, they tend to give off light. The light is emitted only at certain wavelengths that are characteristic
THE ATOMIC SPECTRUM OF HYDROGEN When atoms are excited, either in an electric discharge or with heat, they tend to give off light. The light is emitted only at certain wavelengths that are characteristic
I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited
 NCCS 1.1.2 & 1.1.3 I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited state I will describe how an electron
NCCS 1.1.2 & 1.1.3 I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited state I will describe how an electron
E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 FRAUNHOFER DIFFRACTION
 E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 FRAUNHOFER DIFFRACTION References for Fraunhofer Diffraction 1. Jenkins and White Fundamentals of Optics. Chapters on Fraunhofer diffraction and
E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 FRAUNHOFER DIFFRACTION References for Fraunhofer Diffraction 1. Jenkins and White Fundamentals of Optics. Chapters on Fraunhofer diffraction and
where n = (an integer) =
 5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
Yellow. Strontium red white. green. yellow violet. green. red. Chapter 4. Arrangement of Electrons in Atoms. Table of Contents
 Chapter 4 Arrangement of Electrons in Atoms Table of Contents Section 1 Section 2 Section 3 The Development of a New Atomic Model The Quantum Model of the Atom Electron Configurations Sodium Yellow Strontium
Chapter 4 Arrangement of Electrons in Atoms Table of Contents Section 1 Section 2 Section 3 The Development of a New Atomic Model The Quantum Model of the Atom Electron Configurations Sodium Yellow Strontium
Optical Spectroscopy and Atomic Structure. PHYS 0219 Optical Spectroscopy and Atomic Structure 1
 Optical Spectroscopy and Atomic Structure PHYS 0219 Optical Spectroscopy and Atomic Structure 1 Optical Spectroscopy and Atomic Structure This experiment has four parts: 1. Spectroscope Setup - Your lab
Optical Spectroscopy and Atomic Structure PHYS 0219 Optical Spectroscopy and Atomic Structure 1 Optical Spectroscopy and Atomic Structure This experiment has four parts: 1. Spectroscope Setup - Your lab
Chapter 6 Electronic structure of atoms
 Chapter 6 Electronic structure of atoms light photons spectra Heisenberg s uncertainty principle atomic orbitals electron configurations the periodic table 6.1 The wave nature of light Visible light is
Chapter 6 Electronic structure of atoms light photons spectra Heisenberg s uncertainty principle atomic orbitals electron configurations the periodic table 6.1 The wave nature of light Visible light is
Note: Common units for visible light wavelengths are the Angstrom (Å) and the nanometer (nm).
 Modern Physics Laboratory Spectra and Spectrometers, Balmer Spectrum of Hydrogen In this experiment, we display continuous and discrete emission spectra and explore the use of several types of spectrometers.
Modern Physics Laboratory Spectra and Spectrometers, Balmer Spectrum of Hydrogen In this experiment, we display continuous and discrete emission spectra and explore the use of several types of spectrometers.
EMISSION SPECTROSCOPY
 IFM The Department of Physics, Chemistry and Biology LAB 57 EMISSION SPECTROSCOPY NAME PERSONAL NUMBER DATE APPROVED I. OBJECTIVES - Understand the principle of atomic emission spectra. - Know how to acquire
IFM The Department of Physics, Chemistry and Biology LAB 57 EMISSION SPECTROSCOPY NAME PERSONAL NUMBER DATE APPROVED I. OBJECTIVES - Understand the principle of atomic emission spectra. - Know how to acquire
EXPERIMENT 12 THE WAVELENGTH OF LIGHT; THE DIFFRACTION GRATING
 EXPERIMENT 12 THE WAVELENGTH OF LIGHT; THE DIFFRACTION GRATING INTRODUCTION: One of the most fascinating chapters in the history of physics has been the search for an understanding of the true nature of
EXPERIMENT 12 THE WAVELENGTH OF LIGHT; THE DIFFRACTION GRATING INTRODUCTION: One of the most fascinating chapters in the history of physics has been the search for an understanding of the true nature of
E n = n h ν. The oscillators must absorb or emit energy in discrete multiples of the fundamental quantum of energy given by.
 Planck s s Radiation Law Planck made two modifications to the classical theory The oscillators (of electromagnetic origin) can only have certain discrete energies determined by E n = n h ν with n is an
Planck s s Radiation Law Planck made two modifications to the classical theory The oscillators (of electromagnetic origin) can only have certain discrete energies determined by E n = n h ν with n is an
Atomic Structure. Standing Waves x10 8 m/s. (or Hz or 1/s) λ Node
 Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
Physics 1CL OPTICAL SPECTROSCOPY Spring 2010
 Introduction In this lab, you will use a diffraction grating to split up light into the various colors which make up the different wavelengths of the visible electromagnetic spectrum. You will assemble
Introduction In this lab, you will use a diffraction grating to split up light into the various colors which make up the different wavelengths of the visible electromagnetic spectrum. You will assemble
Dispersion of Glass Introduction Apparatus Theory
 Dispersion of Glass Introduction This experiment will develop skills in aligning and using a spectrometer to measure dispersion of glass, and choosing a suitable fit for data and plotting the resulting
Dispersion of Glass Introduction This experiment will develop skills in aligning and using a spectrometer to measure dispersion of glass, and choosing a suitable fit for data and plotting the resulting
Atomic Spectra HISTORY AND THEORY
 Atomic Spectra HISTORY AND THEORY When atoms of a gas are excited (by high voltage, for instance) they will give off light. Each element (in fact, each isotope) gives off a characteristic atomic spectrum,
Atomic Spectra HISTORY AND THEORY When atoms of a gas are excited (by high voltage, for instance) they will give off light. Each element (in fact, each isotope) gives off a characteristic atomic spectrum,
The Photoelectric Effect
 Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Chapter 4. Dispersion of Glass. 4.1 Introduction. 4.2 Apparatus
 Chapter 4 Dispersion of Glass 4.1 Introduction This experiment will develop skills in choosing a suitable fit for data and plotting the resulting curve. Curve fitting will count for a big chunk of the
Chapter 4 Dispersion of Glass 4.1 Introduction This experiment will develop skills in choosing a suitable fit for data and plotting the resulting curve. Curve fitting will count for a big chunk of the
Visible Spectrometer
 Visible Spectrometer Experiment VIS University of Florida Department of Physics PHY4803L Advanced Physics Laboratory Objective The Balmer spectral lines from a hydrogen discharge lamp are observed with
Visible Spectrometer Experiment VIS University of Florida Department of Physics PHY4803L Advanced Physics Laboratory Objective The Balmer spectral lines from a hydrogen discharge lamp are observed with
Atomic Structure and the Periodic Table
 Atomic Structure and the Periodic Table The electronic structure of an atom determines its characteristics Studying atoms by analyzing light emissions/absorptions Spectroscopy: analysis of light emitted
Atomic Structure and the Periodic Table The electronic structure of an atom determines its characteristics Studying atoms by analyzing light emissions/absorptions Spectroscopy: analysis of light emitted
The Spectrophotometer and Atomic Spectra of Hydrogen Physics 246
 The Spectrophotometer and Atomic Spectra of Hydrogen Physics 46 Introduction: When heated sufficiently, most elements emit light. With a spectrometer, the emitted light can be broken down into its various
The Spectrophotometer and Atomic Spectra of Hydrogen Physics 46 Introduction: When heated sufficiently, most elements emit light. With a spectrometer, the emitted light can be broken down into its various
10. Wavelength measurement using prism spectroscopy
 Spk 0. Wavelength measurement using prism spectroscopy 0. Introduction The study of emitted spectra of electromagnetic waves by excited atoms makes for one of the most important methods to investigate
Spk 0. Wavelength measurement using prism spectroscopy 0. Introduction The study of emitted spectra of electromagnetic waves by excited atoms makes for one of the most important methods to investigate
EXPERIMENT 5:Determination of the refractive index (µ) of the material of a prism using sprectometer
 EXPERIMENT 5:Determination of the refractive index (µ) of the material of a prism using sprectometer Debangshu Mukherjee B.Sc Physics,1st year Chennai Mathematical Institute 17.10.008 1 Aim of Experiment
EXPERIMENT 5:Determination of the refractive index (µ) of the material of a prism using sprectometer Debangshu Mukherjee B.Sc Physics,1st year Chennai Mathematical Institute 17.10.008 1 Aim of Experiment
is the minimum stopping potential for which the current between the plates reduces to zero.
 Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
Chemistry 212 ATOMIC SPECTROSCOPY
 Chemistry 212 ATOMIC SPECTROSCOPY The emission and absorption of light energy of particular wavelengths by atoms and molecules is a common phenomenon. The emissions/absorptions are characteristic for each
Chemistry 212 ATOMIC SPECTROSCOPY The emission and absorption of light energy of particular wavelengths by atoms and molecules is a common phenomenon. The emissions/absorptions are characteristic for each
Problems with the atomic model?
 Modern Atomic Theory- Electronic Structure of Atoms DR HNIMIR-CH7 Where should (-) electrons be found? Problems with the atomic model? First, a Little About Electromagnetic Radiation- Waves Another Look
Modern Atomic Theory- Electronic Structure of Atoms DR HNIMIR-CH7 Where should (-) electrons be found? Problems with the atomic model? First, a Little About Electromagnetic Radiation- Waves Another Look
UNIT : QUANTUM THEORY AND THE ATOM
 Name St.No. Date(YY/MM/DD) / / Section UNIT 102-10: QUANTUM THEORY AND THE ATOM OBJECTIVES Atomic Spectra for Hydrogen, Mercury and Neon. 1. To observe various atomic spectra with a diffraction grating
Name St.No. Date(YY/MM/DD) / / Section UNIT 102-10: QUANTUM THEORY AND THE ATOM OBJECTIVES Atomic Spectra for Hydrogen, Mercury and Neon. 1. To observe various atomic spectra with a diffraction grating
Dispersion of light by a prism
 Dispersion of light by a prism Aim: (i) To calculate refractive index µ of a prism for various wavelengths (λ) of Hg and to find dispersive power of the material of the prism (ii) To plot µ-/λ curve and
Dispersion of light by a prism Aim: (i) To calculate refractive index µ of a prism for various wavelengths (λ) of Hg and to find dispersive power of the material of the prism (ii) To plot µ-/λ curve and
Lecture 0. NC State University
 Chemistry 736 Lecture 0 Overview NC State University Overview of Spectroscopy Electronic states and energies Transitions between states Absorption and emission Electronic spectroscopy Instrumentation Concepts
Chemistry 736 Lecture 0 Overview NC State University Overview of Spectroscopy Electronic states and energies Transitions between states Absorption and emission Electronic spectroscopy Instrumentation Concepts
Electronic structure of atoms
 Chapter 1 Electronic structure of atoms light photons spectra Heisenberg s uncertainty principle atomic orbitals electron configurations the periodic table 1.1 The wave nature of light Much of our understanding
Chapter 1 Electronic structure of atoms light photons spectra Heisenberg s uncertainty principle atomic orbitals electron configurations the periodic table 1.1 The wave nature of light Much of our understanding
APAS Laboratory { PAGE } Spectroscopy SPECTROSCOPY
 SPECTROSCOPY SYNOPSIS: In this lab you will eplore different types of emission spectra, calibrate a spectrometer using the spectrum of a known element, and use your calibration to identify an unknown element.
SPECTROSCOPY SYNOPSIS: In this lab you will eplore different types of emission spectra, calibrate a spectrometer using the spectrum of a known element, and use your calibration to identify an unknown element.
Higher -o-o-o- Past Paper questions o-o-o- 3.4 Spectra
 Higher -o-o-o- Past Paper questions 1991-2010 -o-o-o- 3.4 Spectra 1992 Q37 The diagram below shows the energy levels for the hydrogen atom. (a) Between which two energy levels would an electron transition
Higher -o-o-o- Past Paper questions 1991-2010 -o-o-o- 3.4 Spectra 1992 Q37 The diagram below shows the energy levels for the hydrogen atom. (a) Between which two energy levels would an electron transition
DIFFRACTION GRATING. OBJECTIVE: To use the diffraction grating in the formation of spectra and in the measurement of wavelengths.
 DIFFRACTION GRATING OBJECTIVE: To use the diffraction grating in the formation of spectra and in the measurement of wavelengths. THEORY: The operation of the grating is depicted in Fig. 1 on page Lens
DIFFRACTION GRATING OBJECTIVE: To use the diffraction grating in the formation of spectra and in the measurement of wavelengths. THEORY: The operation of the grating is depicted in Fig. 1 on page Lens
Unit 2 - Particles and Waves - Part 2
 WAVE-PARTICLE DUALITY Unit - Particles and Waves - Part 8. The photoelectric effect and wave particle duality Photoelectric effect as evidence for the particulate nature of light. Photons of sufficient
WAVE-PARTICLE DUALITY Unit - Particles and Waves - Part 8. The photoelectric effect and wave particle duality Photoelectric effect as evidence for the particulate nature of light. Photons of sufficient
Atomic spectra of one and two-electron systems
 Atomic spectra of one and two-electron systems Key Words Term symbol, Selection rule, Fine structure, Atomic spectra, Sodium D-line, Hund s rules, Russell-Saunders coupling, j-j coupling, Spin-orbit coupling,
Atomic spectra of one and two-electron systems Key Words Term symbol, Selection rule, Fine structure, Atomic spectra, Sodium D-line, Hund s rules, Russell-Saunders coupling, j-j coupling, Spin-orbit coupling,
ONE-ELECTRON AND TWO-ELECTRON SPECTRA
 ONE-ELECTRON AND TWO-ELECTRON SPECTRA (A) FINE STRUCTURE AND ONE-ELECTRON SPECTRUM PRINCIPLE AND TASK The well-known spectral lines of He are used for calibrating the diffraction spectrometer. The wavelengths
ONE-ELECTRON AND TWO-ELECTRON SPECTRA (A) FINE STRUCTURE AND ONE-ELECTRON SPECTRUM PRINCIPLE AND TASK The well-known spectral lines of He are used for calibrating the diffraction spectrometer. The wavelengths
EXPERIMENT NO. 6. OBJECT: To determine the wavelength of any three lines of mercury light by diffraction grating in1 st and 2 nd order spectrum.
 EXPEIMENT NO. 6 OBJECT: To determine the wavelength of any three lines of mercury light by diffraction grating in1 st and 2 nd order spectrum. APPAATUS: A diffraction grating, Spectrometer, spirit level,
EXPEIMENT NO. 6 OBJECT: To determine the wavelength of any three lines of mercury light by diffraction grating in1 st and 2 nd order spectrum. APPAATUS: A diffraction grating, Spectrometer, spirit level,
Chapter 37 Early Quantum Theory and Models of the Atom. Copyright 2009 Pearson Education, Inc.
 Chapter 37 Early Quantum Theory and Models of the Atom Planck s Quantum Hypothesis; Blackbody Radiation Photon Theory of Light and the Photoelectric Effect Energy, Mass, and Momentum of a Photon Compton
Chapter 37 Early Quantum Theory and Models of the Atom Planck s Quantum Hypothesis; Blackbody Radiation Photon Theory of Light and the Photoelectric Effect Energy, Mass, and Momentum of a Photon Compton
X-RAY SPECTRA. Theory:
 12 Oct 18 X-ray.1 X-RAY SPECTRA In this experiment, a number of measurements involving x-rays will be made. The spectrum of x-rays emitted from a molybdenum target will be measured, and the experimental
12 Oct 18 X-ray.1 X-RAY SPECTRA In this experiment, a number of measurements involving x-rays will be made. The spectrum of x-rays emitted from a molybdenum target will be measured, and the experimental
Lecture 11 Atomic Structure
 Lecture 11 Atomic Structure Earlier in the semester, you read about the discoveries that lead to the proposal of the nuclear atom, an atom of atomic number Z, composed of a positively charged nucleus surrounded
Lecture 11 Atomic Structure Earlier in the semester, you read about the discoveries that lead to the proposal of the nuclear atom, an atom of atomic number Z, composed of a positively charged nucleus surrounded
