n(λ) = c/v(λ). Figure 1: Dispersion curves for some common optical glass types.
|
|
|
- Rolf Mosley
- 5 years ago
- Views:
Transcription
1 Physics 2310 Lab 2: The Dispersion of Optical Glass Dr. Michael Pierce (Univ. of Wyoming) Based on a lab by Dr. M. Kruger (Univ. of Missouri, Kansas City) Purpose: The purpose of this lab is to introduce students to the dispersion of light in optical materials. That is, to the dependence of the index of refraction upon wavelength. This property must be considered during the design of optical instruments. Theoretical Basis: We all know that a prism disperses light due to the change in the index of refraction with wavelength. As a result the refracted angle of light through a prism depends on wavelength. Specifically: n(λ) = c/v(λ). No optical system is perfect and so a designer must carefully weigh cost vs. complexity in designing a lens or system of lenses for a particular application. This naturally results in compromises in performance. One example is the residual color seen around the images produced by your binoculars, riflescope, or telescope. In this case the designer choses different types of glass for the lenses in order to balance and partially cancel the residual color that results from the dispersion of light. A plot of the refractive indices of some representative materials vs. wavelength is shown here: Figure 1: Dispersion curves for some common optical glass types.
2 Note that the index is generally higher at shorter wavelengths and so a prism refracts (bends) blue light more than red. It is also apparent that some materials disperse light more (flint) or less (crown) than others. Cauchy modeled the dispersion of optical glass as a simple power series in wavelength (λ). Specifically, he used: n (!) = A + B! + C 2! 4 but our data are probably only sufficient to determine the first two terms. That is: n (!) = A + B! 2 Another measure of the dispersion that is much more useful in the design of optical instruments is the Abbe number. Abbe defined the dispersion of an optical glass in terms of the indices measured at certain wavelengths. Specifically as: V d = n d!1 n F! n C where d, F, and C designate the Fraunhoffer lines seen in the spectrum of the Sun. The F and C lines are from Hydrogen and the d line is the Sodium doublet. However, a strong Helium line is found at nearly the same wavelength and, since Helium also has several other lines we could use to measure n(λ), we will simply use the yellow Helium line for n d instead. Note that the Abbe number is a measure of the dispersion (dn/dλ) from blue to red scaled by the index of refraction in the yellow. We will discuss the Abbe number in more detail when we discuss the design of achromatic lenses. Minimum Deviation Angle for a Prism. At a given (fixed) wavelength a prism will refract light through a range of angles depending on the angle of the light incident on the prism. In fact there is an angle of minimum deviation (δ m ) as a prism is rotated. This condition, derived in lecture, is satisfied in the symmetric case shown below. Specifically, when θ 1 = θ 2, and θ 2 = θ 1 and thus θ 2 = α/2 and θ 1 = θ 2 + δ m /2 = (α + δ m )/2.
3 Figure 2: Geometry for an equilateral prism orientated for minimum deviation. When combined with Snell s law we can derive a relationship between the index of refraction of the prism and the angle of minimum deviation (see the lecture). Namely: n(!) = sin" 1 sin" 2 =! sin 1 2 [# +$ $ m (!)] " # % & ' sin! * ), ( 2 + So, this relationship can be used to compute the index of refraction at any wavelength given the angle of minimum deviation measured for that wavelength. (1) Procedure. Begin by reviewing the equipment on the laboratory optical bench. Take a picture with your smart phone or draw a quick sketch of the apparatus and label each component or make notes on the picture on the following page.
4 Figure 3: Components of the prism spectrograph. a) The light source is a high voltage tube that ionizes a gas (Hydrogen) by stripping it of its electron. Upon recombination the electron cascades to its lowest level releasing photons corresponding to each of its allowed energy. This results in a series of discrete spectral lines or colors. b) The collimator consists of a slit at the focus of a lens that collimates the light like a flashlight beam so that all light rays will enter the prism at the same angle. c) The prism and rotational stage. The prism is centered on a silver rotational stage so that careful rotation of the stage will rotate the prism and change the angle of incidence. The dispersed light from the prism emerges at different angles according to its wavelength and this is the deviation angle. d) The telescope and eyepiece. The telescope focuses the dispersed light onto a crosshair and the eyepiece acts as a magnifying glass to enlarge the image and crosshair. The telescope can be moved to center the crosshair on any particular spectral line.
5 (3) Verifying the Spectroscopic Alignment: In order to make measurements we will first need to set the protractor angle at a sensible for making measurements. Begin by: a) Removing the prism and block from the smaller, silver rotational stage. b) Loosening the two clamps on the lower part of the device, below the larger black rotational stage. This allows both the large rotational stage and the telescope to move. c) Turn on the spectral lamp and move the telescope back and forth until you can see and image of the slit through which the spectral lamp is shining. It should look like a vertical line. If it is out of focus carefully rotate the eyepiece of the telescope back and forth while pushing it in and out until the slit image is in focus. When the line is close to the crosshair clamp the telescope by tightening the upper clamp. Use the fine adjustment knob to center the crosshair on the slit image. d) Now rotate the large black stage until the inner vernier scales read 0-deg. and 180-deg. Use the little magnifiers to verify this. Clamp the lowest clamp to set the stage in place. Don t unclamp or move this the rest of the lab. e) Now rotate the telescope until the outer scales read about 25-deg and 205- deg. The outer two measurement scales are offset by about 180-deg so when you take measurements be sure and subtract 180-deg. from the one that is now reading about 205-deg. Don t forget to do this. Clamp the telescope in place. We will use the fine adjustment screws from now on. f) Finally have your TA put the prism in place on the silver stage. This is a little tricky so it s best that they do it. Verify that you can see the spectral emission lines through the telescope. Be sure that the uppermost silver stage is unclamped since you will need to be able to rotate it. (4) Measurement of the Angle of Minimum Deviation: Before you begin any measurements log into a computer and open an Excel spreadsheet. Alternatively, make a table by hand. Make a column for wavelengths and another three for two angle measurements and their average. Be sure that you are subtracting 180-deg. from the one side that reads about 205-deg or have Excel do it for you. Don t forget to do this. For each spectrum tube you use refer to the Appendix for a picture of its spectrum and a table of its spectral line wavelengths. Enter these wavelengths into the appropriate column of your data table. You should see the emission line spectrum of hydrogen (see Appendix). When you look through the telescope you should be able to see the spectral lines. Now we are ready to start taking measurements. a) Slowly rotate the silver stage back and forth and you will notice that there is a position where as the prism table is rotated in one direction a
6 b) c) spectral line will actually move in one direction, stop and begin moving in the opposite direction. The angle at which this happens is called the angle of minimum deviation. Assuming you are using the Hydrogen tube watch the red line until it just stops moving right before it reverses direction. Stop rotating the silver stage. Loosen the telescope clamp and look through the telescope as you move it until the crosshair is aligned with the spectral line. Clamp it an move the fine adjust knob if you like. Slowly rotate the prism back and forth to be sure that it is set and the angle of minimum deviation. Adjust the telescope with the fine adjust if necessary. When you are satisfied record both vernier readings (see Appendix I). Don t forget to subtract 180-deg from the largest reading. Repeat the measurement by have each person in your group do this two times, averaging the results. Figure 4: Top view of the prism spectrograph. d) Repeat the above measurements for all of the lines. Turn off the lamp power supply and wait for the hydrogen source to cool. Replace the hydrogen source with the helium source, turn it on and repeat the measurements for all of the lines that you can see.
7 (5) Data Analysis. 1) Tabulate λ, and n for all of the lines. Use Excel to insert a column after your wavelengths and compute 1/λ 2. Also be sure to add a column to the right in which you compute the average of your vernier readings. Now add another column to the right in which you compute the angle of minimum deviation (see previous section) and another in which you solve the equation on page 3 for n. Be sure to convert your angles to radians or Excel will do something unexpected! 2) Make a plot of n vs. λ. Be sure to label your axes. 3) Using the first two terms of Cauchy s equation, determine A and B from your data. Plot n vs 1/λ 2 and fit the best-fit trend line on the same graph. That is, the best fit to the equation: n(λ) = A + B/λ 2. Note that the equation will be linear and thus the values of A and B are easy to determine. Comment on your fit. 4) Using equation from part 3) and your value of B, determine dn/dλ for λ=400, 500 and 600 nm. 5) The Abbe number is very useful for characterizing the dispersion of optical glass. What are the Abbe numbers for the prism glass? Before you leave the lab make sure that you have your lab partner s so that the data can be shared within your lab group. Summary: In this lab we ve measured the minimum deviation angle for a prism at a variety of wavelengths. The data have been plotted and analyzed using Excel in order to fit these data to the Cauchy model. We have also used these data to calculate the Abbe number for the prism glass. We will see later in lecture how the Abbe number is used in the design of Achromatic (color corrected) lenses. Appendix I: Reading verniers Verniers are read using both the lower and upper scale, the lower scale being in degrees and the upper scale in minutes. Find the largest number on the lower scale that is to the left of the 0 of the upper scale. For the photograph below, this is degrees. Then find where the lines on the upper scale overlap those of the lower scale and read the number off of the upper scale. For the photograph, this is 10 minutes. Thus, this vernier reads: 184 o.5 and 10
8 Figure A1: The Vernier Scale. The prism spectrometer has 2 verniers. There would be a 180 o difference in their readings if the system worked perfectly. This is not the case, so we will record the readings from both verniers, and in the end average our results using Excel.
9 Appendix II: Emission Spectral Lines The hydrogen lines are at: Wavelength (nm) Name Color Relative Intensity Fraunhofer C Red Fraunhofer F Blue Green Violet Violet 15 The helium lines are at: Wavelength (nm) Name Color Relative Intensity Red Red Yellow Lt. Green Dk. Green Blue Green Blue Violet 225 The Hg lines are at: Wavelength (nm) Name Color I-line ultraviolet (UVA) H-line violet G-line blue green yellow-orange The Ne lines are at: Wavelength (nm) Color yellow yellow-orange orange orange orange red red very red very red The relative intensities are approximations. You may use the line spectra below to help you identify the wavelength of each lines. Please note that for the helium spectrum, the reddest line is missing from the following figure.
10 Figure AII-1: Spectral Line Atlas.
11
DISPERSION OF A GLASS PRISM
 PH2 page 1 DISPERSION OF A GLASS PRISM OBJECTIVE The objective of this experiment is to analyze the emission spectrum of helium and to analyze the dispersion of a glass prism by measuring the index of
PH2 page 1 DISPERSION OF A GLASS PRISM OBJECTIVE The objective of this experiment is to analyze the emission spectrum of helium and to analyze the dispersion of a glass prism by measuring the index of
Chapter 4. Dispersion of Glass. 4.1 Introduction. 4.2 Apparatus
 Chapter 4 Dispersion of Glass 4.1 Introduction This experiment will develop skills in choosing a suitable fit for data and plotting the resulting curve. Curve fitting will count for a big chunk of the
Chapter 4 Dispersion of Glass 4.1 Introduction This experiment will develop skills in choosing a suitable fit for data and plotting the resulting curve. Curve fitting will count for a big chunk of the
Ph 3455/MSE 3255 Experiment 2: Atomic Spectra
 Ph 3455/MSE 3255 Experiment 2: Atomic Spectra Background Reading: Tipler, Llewellyn pp. 163-165 Apparatus: Spectrometer, sodium lamp, hydrogen lamp, mercury lamp, diffraction grating, watchmaker eyeglass,
Ph 3455/MSE 3255 Experiment 2: Atomic Spectra Background Reading: Tipler, Llewellyn pp. 163-165 Apparatus: Spectrometer, sodium lamp, hydrogen lamp, mercury lamp, diffraction grating, watchmaker eyeglass,
Lab 5: Spectroscopy & the Hydrogen Atom Phy248 Spring 2009
 Lab 5: Spectroscopy & the Hydrogen Atom Phy248 Spring 2009 Name Section Return this spreadsheet to your TA that will use it to score your lab. To receive full credit you must use complete sentences and
Lab 5: Spectroscopy & the Hydrogen Atom Phy248 Spring 2009 Name Section Return this spreadsheet to your TA that will use it to score your lab. To receive full credit you must use complete sentences and
Lab 10: Spectroscopy & the Hydrogen Atom Phy208 Fall 2008
 Lab 10: Spectroscopy & the Hydrogen Atom Phy208 Fall 2008 Name Section This sheet is the lab document your TA will use to score your lab. It is to be turned in at the end of lab. To receive full credit
Lab 10: Spectroscopy & the Hydrogen Atom Phy208 Fall 2008 Name Section This sheet is the lab document your TA will use to score your lab. It is to be turned in at the end of lab. To receive full credit
The Emission Spectra of Light
 The Emission Spectra of Light Objectives: Theory: 1.... measured the wavelength limits of the color bands in the visible spectrum, 2.... measured the wavelengths of the emission lines of the hydrogen Balmer
The Emission Spectra of Light Objectives: Theory: 1.... measured the wavelength limits of the color bands in the visible spectrum, 2.... measured the wavelengths of the emission lines of the hydrogen Balmer
Dispersion of Glass Introduction Apparatus Theory
 Dispersion of Glass Introduction This experiment will develop skills in aligning and using a spectrometer to measure dispersion of glass, and choosing a suitable fit for data and plotting the resulting
Dispersion of Glass Introduction This experiment will develop skills in aligning and using a spectrometer to measure dispersion of glass, and choosing a suitable fit for data and plotting the resulting
Science Lab I Properties of Light
 Art & Science of Light Fall 2007 Science Lab I Properties of Light Prepared by: Dr. Dharshi Bopegedera 1 Using the Filtergraph (15 minutes) 1. Turn on the filtergraph, place a card on it and look at the
Art & Science of Light Fall 2007 Science Lab I Properties of Light Prepared by: Dr. Dharshi Bopegedera 1 Using the Filtergraph (15 minutes) 1. Turn on the filtergraph, place a card on it and look at the
THE DIFFRACTION GRATING SPECTROMETER
 Purpose Theory THE DIFFRACTION GRATING SPECTROMETER a. To study diffraction of light using a diffraction grating spectrometer b. To measure the wavelengths of certain lines in the spectrum of the mercury
Purpose Theory THE DIFFRACTION GRATING SPECTROMETER a. To study diffraction of light using a diffraction grating spectrometer b. To measure the wavelengths of certain lines in the spectrum of the mercury
Atomic Spectra HISTORY AND THEORY
 Atomic Spectra HISTORY AND THEORY When atoms of a gas are excited (by high voltage, for instance) they will give off light. Each element (in fact, each isotope) gives off a characteristic atomic spectrum,
Atomic Spectra HISTORY AND THEORY When atoms of a gas are excited (by high voltage, for instance) they will give off light. Each element (in fact, each isotope) gives off a characteristic atomic spectrum,
Laboratory #29: Spectrometer
 INDIANA UNIVERSITY, DEPARTMENT OF PHYSICS, P309 LABORATORY Laboratory #29: Spectrometer Goal: Learn to adjust an optical spectrometer, use a transmission grating to measure known spectral lines of mercury,
INDIANA UNIVERSITY, DEPARTMENT OF PHYSICS, P309 LABORATORY Laboratory #29: Spectrometer Goal: Learn to adjust an optical spectrometer, use a transmission grating to measure known spectral lines of mercury,
LAB 10: OPTICAL MATERIALS AND DISPERSION I
 OPTI 202L - Geometrical and Instrumental Optics Lab LAB 10: OPTICAL MATERIALS AND DISPERSION I 10-1 Measuring the refractive index of a material is one of the most fundamental optical measurements, and
OPTI 202L - Geometrical and Instrumental Optics Lab LAB 10: OPTICAL MATERIALS AND DISPERSION I 10-1 Measuring the refractive index of a material is one of the most fundamental optical measurements, and
EXPERIMENT 14. The Atomic Spectrum of Hydrogen
 Name: Laboratory Section: Laboratory Section Date: Partners Names: Grade: Last Revised on March 18, 2003 EXPERIMENT 14 The Atomic Spectrum of Hydrogen 0. Pre-Laboratory Work [2 pts] 1. You will be using
Name: Laboratory Section: Laboratory Section Date: Partners Names: Grade: Last Revised on March 18, 2003 EXPERIMENT 14 The Atomic Spectrum of Hydrogen 0. Pre-Laboratory Work [2 pts] 1. You will be using
EXPERIMENT 12 THE GRATING SPECTROMETER AND ATOMIC SPECTRA
 OBJECTIVES Learn the theory of the grating spectrometer Observe the spectrum of mercury and hydrogen Measure the grating constant of a diffraction grating Measure the Rydberg Constant EXPERIMENT THE GRATING
OBJECTIVES Learn the theory of the grating spectrometer Observe the spectrum of mercury and hydrogen Measure the grating constant of a diffraction grating Measure the Rydberg Constant EXPERIMENT THE GRATING
Experiment #5: Cauchy s Formula
 Experiment #5: Cauchy s Formula Carl Adams October 14, 2011 1 Purpose This experiment is a continuation of Experiment #4. It is assumed you have an aligned spectrometer. 2 Safety/Protocol 1. The gas discharge
Experiment #5: Cauchy s Formula Carl Adams October 14, 2011 1 Purpose This experiment is a continuation of Experiment #4. It is assumed you have an aligned spectrometer. 2 Safety/Protocol 1. The gas discharge
Name Date: Course number: MAKE SURE TA & TI STAMPS EVERY PAGE BEFORE YOU START EXPERIMENT 14. The Atomic Spectrum of Hydrogen
 Laboratory Section: Last Revised on September 21, 2016 Partners Names: Grade: EXPERIMENT 14 The Atomic Spectrum of Hydrogen 0. Pre-Laboratory Work [2 pts] 1. You will be using a diffraction grating in
Laboratory Section: Last Revised on September 21, 2016 Partners Names: Grade: EXPERIMENT 14 The Atomic Spectrum of Hydrogen 0. Pre-Laboratory Work [2 pts] 1. You will be using a diffraction grating in
Optics. Measuring the line spectra of inert gases and metal vapors using a prism spectrometer. LD Physics Leaflets P
 Optics Spectrometer Prism spectrometer LD Physics Leaflets P5.7.1.1 Measuring the line spectra of inert gases and metal vapors using a prism spectrometer Objects of the experiment Adjusting the prism spectrometer.
Optics Spectrometer Prism spectrometer LD Physics Leaflets P5.7.1.1 Measuring the line spectra of inert gases and metal vapors using a prism spectrometer Objects of the experiment Adjusting the prism spectrometer.
E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 FRAUNHOFER DIFFRACTION
 E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 FRAUNHOFER DIFFRACTION References for Fraunhofer Diffraction 1. Jenkins and White Fundamentals of Optics. Chapters on Fraunhofer diffraction and
E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 FRAUNHOFER DIFFRACTION References for Fraunhofer Diffraction 1. Jenkins and White Fundamentals of Optics. Chapters on Fraunhofer diffraction and
Rydberg constant from atomic spectra of gases
 Page 1 of 8 Rydberg constant from atomic spectra of gases Objective - Calibrating a prism spectrometer to convert the scale readings in wavelengths of spectral lines. - Observing the Balmer series of atomic
Page 1 of 8 Rydberg constant from atomic spectra of gases Objective - Calibrating a prism spectrometer to convert the scale readings in wavelengths of spectral lines. - Observing the Balmer series of atomic
Atomic spectra of one and two-electron systems
 Atomic spectra of one and two-electron systems Key Words Term symbol, Selection rule, Fine structure, Atomic spectra, Sodium D-line, Hund s rules, Russell-Saunders coupling, j-j coupling, Spin-orbit coupling,
Atomic spectra of one and two-electron systems Key Words Term symbol, Selection rule, Fine structure, Atomic spectra, Sodium D-line, Hund s rules, Russell-Saunders coupling, j-j coupling, Spin-orbit coupling,
Dispersion of light by a prism
 Dispersion of light by a prism Aim: (i) To calculate refractive index µ of a prism for various wavelengths (λ) of Hg and to find dispersive power of the material of the prism (ii) To plot µ-/λ curve and
Dispersion of light by a prism Aim: (i) To calculate refractive index µ of a prism for various wavelengths (λ) of Hg and to find dispersive power of the material of the prism (ii) To plot µ-/λ curve and
Any first year text, sections on atomic structure, spectral lines and spectrometers
 Physics 33 Experiment 5 Atomic Spectra References Any first year text, sections on atomic structure, spectral lines and spectrometers Any modern physics text, eg F.K. Richtmeyer, E.H. Kennard and J.N.
Physics 33 Experiment 5 Atomic Spectra References Any first year text, sections on atomic structure, spectral lines and spectrometers Any modern physics text, eg F.K. Richtmeyer, E.H. Kennard and J.N.
NORTHERN ILLINOIS UNIVERSITY PHYSICS DEPARTMENT. Physics 211 E&M and Quantum Physics Spring Lab #9: Diffraction Spectroscopy
 NORTHERN ILLINOIS UNIVERSITY PHYSICS DEPARTMENT Physics 211 E&M and Quantum Physics Spring 2018 Lab #9: Diffraction Spectroscopy Lab Writeup Due: Mon/Wed/Thu/Fri, April 30/ May 2/3/4, 2018 Background All
NORTHERN ILLINOIS UNIVERSITY PHYSICS DEPARTMENT Physics 211 E&M and Quantum Physics Spring 2018 Lab #9: Diffraction Spectroscopy Lab Writeup Due: Mon/Wed/Thu/Fri, April 30/ May 2/3/4, 2018 Background All
The Quantum Model of the Hydrogen Atom
 Physics 109 Science 1 Experiment 1 1 The Quantum Model of the Hydrogen Atom In this experiment you will use a spectrometer to determine the wavelengths of the visible lines of atomic hydrogen. The goal
Physics 109 Science 1 Experiment 1 1 The Quantum Model of the Hydrogen Atom In this experiment you will use a spectrometer to determine the wavelengths of the visible lines of atomic hydrogen. The goal
Physics 23 Fall 1998 Lab 4 - The Hydrogen Spectrum
 Physics 3 Fall 998 Lab 4 - The Hydrogen Spectrum Theory In the late 800's, it was known that when a gas is excited by means of an electric discharge and the light emitted is viewed through a diffraction
Physics 3 Fall 998 Lab 4 - The Hydrogen Spectrum Theory In the late 800's, it was known that when a gas is excited by means of an electric discharge and the light emitted is viewed through a diffraction
Pre-lab Quiz/PHYS 224. Your name Lab section
 Pre-lab Quiz/PHYS 224 THE DIFFRACTION GRATING AND THE OPTICAL SPECTRUM Your name Lab section 1. What are the goals of this experiment? 2. If the period of a diffraction grating is d = 1,000 nm, where the
Pre-lab Quiz/PHYS 224 THE DIFFRACTION GRATING AND THE OPTICAL SPECTRUM Your name Lab section 1. What are the goals of this experiment? 2. If the period of a diffraction grating is d = 1,000 nm, where the
high energy state for the electron in the atom low energy state for the electron in the atom
 Atomic Spectra Objectives The objectives of this experiment are to: 1) Build and calibrate a simple spectroscope capable of measuring wavelengths of visible light. 2) Measure several wavelengths of light
Atomic Spectra Objectives The objectives of this experiment are to: 1) Build and calibrate a simple spectroscope capable of measuring wavelengths of visible light. 2) Measure several wavelengths of light
Atomic Emission Spectra
 Atomic Emission Spectra Objectives The objectives of this laboratory are as follows: To build and calibrate a simple meter-stick spectroscope that is capable of measuring wavelengths of visible light.
Atomic Emission Spectra Objectives The objectives of this laboratory are as follows: To build and calibrate a simple meter-stick spectroscope that is capable of measuring wavelengths of visible light.
The Grating Spectrometer and Atomic Spectra
 PHY 192 Grating Spectrometer 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating was introduced.
PHY 192 Grating Spectrometer 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating was introduced.
APAS Laboratory { PAGE } Spectroscopy SPECTROSCOPY
 SPECTROSCOPY SYNOPSIS: In this lab you will eplore different types of emission spectra, calibrate a spectrometer using the spectrum of a known element, and use your calibration to identify an unknown element.
SPECTROSCOPY SYNOPSIS: In this lab you will eplore different types of emission spectra, calibrate a spectrometer using the spectrum of a known element, and use your calibration to identify an unknown element.
PHYS General Physics II Lab The Balmer Series for Hydrogen Source. c = speed of light = 3 x 10 8 m/s
 PHYS 1040 - General Physics II Lab The Balmer Series for Hydrogen Source Purpose: The purpose of this experiment is to analyze the emission of light from a hydrogen source and measure and the wavelengths
PHYS 1040 - General Physics II Lab The Balmer Series for Hydrogen Source Purpose: The purpose of this experiment is to analyze the emission of light from a hydrogen source and measure and the wavelengths
Physics 1CL OPTICAL SPECTROSCOPY Spring 2010
 Introduction In this lab, you will use a diffraction grating to split up light into the various colors which make up the different wavelengths of the visible electromagnetic spectrum. You will assemble
Introduction In this lab, you will use a diffraction grating to split up light into the various colors which make up the different wavelengths of the visible electromagnetic spectrum. You will assemble
Atomic Spectra. d sin θ = mλ (1)
 Atomic Spectra Objectives: To measure the wavelengths of visible light emitted by atomic hydrogen and verify that the measured wavelengths obey the empirical Rydberg formula. To observe emission spectra
Atomic Spectra Objectives: To measure the wavelengths of visible light emitted by atomic hydrogen and verify that the measured wavelengths obey the empirical Rydberg formula. To observe emission spectra
AS 101: Day Lab #2 Summer Spectroscopy
 Spectroscopy Goals To see light dispersed into its constituent colors To study how temperature, light intensity, and light color are related To see spectral lines from different elements in emission and
Spectroscopy Goals To see light dispersed into its constituent colors To study how temperature, light intensity, and light color are related To see spectral lines from different elements in emission and
Speed of Light in Glass
 Experiment (1) Speed of Light in Glass Objective:- This experiment is used to determine the speed of propagation of light waves in glass. Apparatus:- Prism, spectrometer, Halogen lamp source. Theory:-
Experiment (1) Speed of Light in Glass Objective:- This experiment is used to determine the speed of propagation of light waves in glass. Apparatus:- Prism, spectrometer, Halogen lamp source. Theory:-
Experiment #4: Optical Spectrometer and the Prism Deviation
 Experiment #4: Optical Spectrometer and the Prism Deviation Carl Adams October 2, 2011 1 Purpose In the first part of this lab you will set up and become familiar with an optical spectrometer. In the second
Experiment #4: Optical Spectrometer and the Prism Deviation Carl Adams October 2, 2011 1 Purpose In the first part of this lab you will set up and become familiar with an optical spectrometer. In the second
Instruction Manual and Experiment Guide for the PASCO scientific Model SP E 2/96 STUDENT SPECTROMETER. Copyright January 1991 $7.
 Instruction Manual and Experiment Guide for the PASCO scientific Model SP-9268 012-02135E 2/96 STUDENT SPECTROMETER Copyright January 1991 $7.50 012-02135E Spectrometer Table of Contents Section Page
Instruction Manual and Experiment Guide for the PASCO scientific Model SP-9268 012-02135E 2/96 STUDENT SPECTROMETER Copyright January 1991 $7.50 012-02135E Spectrometer Table of Contents Section Page
Physics 1C OPTICAL SPECTROSCOPY Rev. 2-AH. Introduction
 Introduction In this lab you will use a diffraction grating to split up light into its various colors (like a rainbow). You will assemble a spectrometer, incorporating the diffraction grating. A spectrometer
Introduction In this lab you will use a diffraction grating to split up light into its various colors (like a rainbow). You will assemble a spectrometer, incorporating the diffraction grating. A spectrometer
Atomic emission spectra experiment
 Atomic emission spectra experiment Contents 1 Overview 1 2 Equipment 1 3 Measuring the grating spacing using the sodium D-lines 4 4 Measurement of hydrogen lines and the Rydberg Constant 5 5 Measurement
Atomic emission spectra experiment Contents 1 Overview 1 2 Equipment 1 3 Measuring the grating spacing using the sodium D-lines 4 4 Measurement of hydrogen lines and the Rydberg Constant 5 5 Measurement
DAY LABORATORY EXERCISE: SPECTROSCOPY
 AS101 - Day Laboratory: Spectroscopy Page 1 DAY LABORATORY EXERCISE: SPECTROSCOPY Goals: To see light dispersed into its constituent colors To study how temperature, light intensity, and light color are
AS101 - Day Laboratory: Spectroscopy Page 1 DAY LABORATORY EXERCISE: SPECTROSCOPY Goals: To see light dispersed into its constituent colors To study how temperature, light intensity, and light color are
Physics 24, Spring 2007 Lab 2 - Complex Spectra
 Physics 24, Spring 2007 Lab 2 - Complex Spectra Theory The optical spectra of isolated atoms consist of discrete, unequally spaced lines. This fact could not be understood on the basis of classical atomic
Physics 24, Spring 2007 Lab 2 - Complex Spectra Theory The optical spectra of isolated atoms consist of discrete, unequally spaced lines. This fact could not be understood on the basis of classical atomic
Observation of Atomic Spectra
 Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
You Are the Spectrometer! A Look Inside Astronomy's Essential Instrument (Robert B. Friedman & Matthew K. Sharp)
 You Are the Spectrometer! A Look Inside Astronomy's Essential Instrument (Robert B. Friedman & Matthew K. Sharp) Introduction Astronomy is a unique science because unlike many of the other sciences, the
You Are the Spectrometer! A Look Inside Astronomy's Essential Instrument (Robert B. Friedman & Matthew K. Sharp) Introduction Astronomy is a unique science because unlike many of the other sciences, the
The Grating Spectrometer and Atomic Spectra
 PHY 192 Grating Spectrometer Spring 2012 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating
PHY 192 Grating Spectrometer Spring 2012 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating
10. Wavelength measurement using prism spectroscopy
 Spk 0. Wavelength measurement using prism spectroscopy 0. Introduction The study of emitted spectra of electromagnetic waves by excited atoms makes for one of the most important methods to investigate
Spk 0. Wavelength measurement using prism spectroscopy 0. Introduction The study of emitted spectra of electromagnetic waves by excited atoms makes for one of the most important methods to investigate
Experiment 24: Spectroscopy
 Experiment 24: Spectroscopy Figure 24.1: Spectroscopy EQUIPMENT High Voltage Power Supply Incandescent Light Source (3) Gas Discharge Tubes: 1. Helium 2. Hydrogen 3. Unknown Element Spectrometer Felt (1)
Experiment 24: Spectroscopy Figure 24.1: Spectroscopy EQUIPMENT High Voltage Power Supply Incandescent Light Source (3) Gas Discharge Tubes: 1. Helium 2. Hydrogen 3. Unknown Element Spectrometer Felt (1)
Protokoll. Grundpraktikum II - Optical Spectroscopy
 Protokoll Grundpraktikum II - Optical Spectroscopy 1 Elaboration 1.1 Optical Spectroscopy Student: Hauke Rasch, Martin Borchert Tutor: Madsen Date: 22.October2014 This experiment is about the fundamental
Protokoll Grundpraktikum II - Optical Spectroscopy 1 Elaboration 1.1 Optical Spectroscopy Student: Hauke Rasch, Martin Borchert Tutor: Madsen Date: 22.October2014 This experiment is about the fundamental
ATOMIC SPECTRA. Objective:
 1 ATOMIC SPECTRA Objective: To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify an unknown
1 ATOMIC SPECTRA Objective: To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify an unknown
Determination of Cauchy s Contants
 8. Determination of Cauchy s Contants 8.1 Objective: To determine Cauchy s Constants using a prism and spectrometer. Apparatus: Glass prism, spectrometer and mercury vapour lamp. 8. Theory: The wavelength
8. Determination of Cauchy s Contants 8.1 Objective: To determine Cauchy s Constants using a prism and spectrometer. Apparatus: Glass prism, spectrometer and mercury vapour lamp. 8. Theory: The wavelength
Experiment 9. Emission Spectra. measure the emission spectrum of a source of light using the digital spectrometer.
 Experiment 9 Emission Spectra 9.1 Objectives By the end of this experiment, you will be able to: measure the emission spectrum of a source of light using the digital spectrometer. find the wavelength of
Experiment 9 Emission Spectra 9.1 Objectives By the end of this experiment, you will be able to: measure the emission spectrum of a source of light using the digital spectrometer. find the wavelength of
University of Massachusetts, Amherst
 PHYSICS 286: Modern Physics Laboratory SPRING 2010 (A. Dinsmore and K. Kumar) Feb 2009 Experiment 4: THE FRANCK HERTZ EXPERIMENT Electronic Excitations of a Gas, and Evidence for the Quantization of Atomic
PHYSICS 286: Modern Physics Laboratory SPRING 2010 (A. Dinsmore and K. Kumar) Feb 2009 Experiment 4: THE FRANCK HERTZ EXPERIMENT Electronic Excitations of a Gas, and Evidence for the Quantization of Atomic
Spectrum of Hydrogen. Physics 227 Lab
 Introduction In today's lab you will be dealing with an area of physics called quantum mechanics. The only quantum mechanical idea that you will be using today is that electrons in an atom can exist only
Introduction In today's lab you will be dealing with an area of physics called quantum mechanics. The only quantum mechanical idea that you will be using today is that electrons in an atom can exist only
LAB 12 ATOMIC SPECTRA
 217 Name Date Partners LAB 12 ATOMIC SPECTRA OBJECTIVES Review the quantum nature of light and how light photons are produced in atoms. Learn to use an optical spectrometer to measure light wavelengths.
217 Name Date Partners LAB 12 ATOMIC SPECTRA OBJECTIVES Review the quantum nature of light and how light photons are produced in atoms. Learn to use an optical spectrometer to measure light wavelengths.
Atomic Spectra 1. Name Date Partners ATOMIC SPECTRA
 Atomic Spectra 1 Name Date Partners ATOMIC SPECTRA Sodium Mercury Lithium Hydrogen Atomic line spectra are characteristic for every element. These are emission spectra (without color). OBJECTIVES Review
Atomic Spectra 1 Name Date Partners ATOMIC SPECTRA Sodium Mercury Lithium Hydrogen Atomic line spectra are characteristic for every element. These are emission spectra (without color). OBJECTIVES Review
Dispersion and resolving power of the prism and grating spectroscope (Item No.: P )
 Dispersion and resolving power of the prism and grating spectroscope (Item No.: P2210300) Curricular Relevance Area of Expertise: Physics Education Level: University Topic: Light and Optics Subtopic: Diffraction
Dispersion and resolving power of the prism and grating spectroscope (Item No.: P2210300) Curricular Relevance Area of Expertise: Physics Education Level: University Topic: Light and Optics Subtopic: Diffraction
DISPERSION AND SPECTRA CHAPTER 20
 CHAPTER 20 DISPERSION AND SPECTRA 20.1 DISPERSION As mentioned earlier, the refractive index of a material depends slightly on the wavelength of light. The relation between the two may be approximately
CHAPTER 20 DISPERSION AND SPECTRA 20.1 DISPERSION As mentioned earlier, the refractive index of a material depends slightly on the wavelength of light. The relation between the two may be approximately
ATOMIC SPECTRA. To identify elements through their emission spectra. Apparatus: spectrometer, spectral tubes, power supply, incandescent lamp.
 ATOMIC SPECTRA Objective: To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify elements through
ATOMIC SPECTRA Objective: To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify elements through
Note: Common units for visible light wavelengths are the Angstrom (Å) and the nanometer (nm).
 Modern Physics Laboratory Spectra and Spectrometers, Balmer Spectrum of Hydrogen In this experiment, we display continuous and discrete emission spectra and explore the use of several types of spectrometers.
Modern Physics Laboratory Spectra and Spectrometers, Balmer Spectrum of Hydrogen In this experiment, we display continuous and discrete emission spectra and explore the use of several types of spectrometers.
Emission Spectrum of Atomic Gases. Prelab Questions
 Emission Spectrum of Atomic Gases Prelab Questions Before this coming to this lab, please review your text for the physics of the spectrum of visible light and of diffraction grating spectrometer.. Which
Emission Spectrum of Atomic Gases Prelab Questions Before this coming to this lab, please review your text for the physics of the spectrum of visible light and of diffraction grating spectrometer.. Which
Operating Instructions Spectro-Goniometer Student. 1 Functional Elements. 2 Safety Precautions. Figure 1: Spectro-Goniometer Student
 Operating Instructions Spectro-Goniometer Student 1 Functional Elements Figure 1: Spectro-Goniometer Student 1. Adjustable entrance slit, holding screw for slit cover 2. Lock ring fixing entrance slit
Operating Instructions Spectro-Goniometer Student 1 Functional Elements Figure 1: Spectro-Goniometer Student 1. Adjustable entrance slit, holding screw for slit cover 2. Lock ring fixing entrance slit
Laboratory Atomic Emission Spectrum
 Laboratory Atomic Emission Spectrum Pre-Lab Questions: Answer the following questions in complete sentences by reading through the Overview and Background sections below. 1. What is the purpose of the
Laboratory Atomic Emission Spectrum Pre-Lab Questions: Answer the following questions in complete sentences by reading through the Overview and Background sections below. 1. What is the purpose of the
Using the Spectrometer
 Using the Spectrometer Introduction When an atom is stimulated it can respond by emitting a spectrum of light. The spectrum comprises discrete wavelengths whose values are characteristic of the particular
Using the Spectrometer Introduction When an atom is stimulated it can respond by emitting a spectrum of light. The spectrum comprises discrete wavelengths whose values are characteristic of the particular
Assignment 3 Due September 27, 2010
 Assignment 3 Due September 27, 2010 Text readings Stops section 5.3 Dispersing and Reflecting Prisms [sections 5.5.1 and 5.5.2] Optical systems section 5.7 Lens Aberrations [section 6.3] Be careful about
Assignment 3 Due September 27, 2010 Text readings Stops section 5.3 Dispersing and Reflecting Prisms [sections 5.5.1 and 5.5.2] Optical systems section 5.7 Lens Aberrations [section 6.3] Be careful about
To determine the wavelengths of light emitted by a mercury vapour lamp by using a diffraction grating.
 12. Diffraction grating OBJECT To determine the wavelengths of light emitted by a mercury vapour lamp by using a diffraction grating. INTRODUCTION: Consider a light beam transmitted through an aperture
12. Diffraction grating OBJECT To determine the wavelengths of light emitted by a mercury vapour lamp by using a diffraction grating. INTRODUCTION: Consider a light beam transmitted through an aperture
EXPERIMENT 09 OBSERVATION OF SPECTRA
 EXPERIMENT 09 OBSERVATION OF SPECTRA INTRODUCTION: In physics, as in very other area of study, one of the most valuable questions a student can learn to ask is, How do they know that? Thus, when you read
EXPERIMENT 09 OBSERVATION OF SPECTRA INTRODUCTION: In physics, as in very other area of study, one of the most valuable questions a student can learn to ask is, How do they know that? Thus, when you read
Atomic Spectroscopy. Objectives
 Atomic Spectroscopy Name Objectives explain the difference between emission and absorption spectra calculate the energy of orbits in the Bohr model of hydrogen calculate E for energy transitions in the
Atomic Spectroscopy Name Objectives explain the difference between emission and absorption spectra calculate the energy of orbits in the Bohr model of hydrogen calculate E for energy transitions in the
ACTIVITY 1. Exploring Light from Gases
 Name: WAVES of matter Class: Visual Quantum Mechanics ACTIVITY 1 Exploring Light from Gases Goal We will view the colors of light which are emitted by different gases. From these patterns of light we gain
Name: WAVES of matter Class: Visual Quantum Mechanics ACTIVITY 1 Exploring Light from Gases Goal We will view the colors of light which are emitted by different gases. From these patterns of light we gain
How Do We Get Light from Matter: The Origin of Emission
 1 How Do We Get Light from Matter: The Origin of Emission Lines ORGANIZATION Pre-Lab: Origins of Lines Mode: inquiry, groups of 2 Grading: lab notes and post-lab questions Safety: no special requirements
1 How Do We Get Light from Matter: The Origin of Emission Lines ORGANIZATION Pre-Lab: Origins of Lines Mode: inquiry, groups of 2 Grading: lab notes and post-lab questions Safety: no special requirements
IO.5 Elliptically Polarized Light
 1. Purpose: IO.5 Elliptically Polarized Light Analyze elliptically polarized light; determine the orientation of the vibration ellipse and the ratio of its semi-axes. 2. Apparatus: Gaertner Scientific
1. Purpose: IO.5 Elliptically Polarized Light Analyze elliptically polarized light; determine the orientation of the vibration ellipse and the ratio of its semi-axes. 2. Apparatus: Gaertner Scientific
Lab report 30 EXPERIMENT 4. REFRACTION OF LIGHT
 30 EXPERIMENT 4. REFRACTION OF LIGHT Lab report Go to your course homepage on Sakai (Resources, Lab templates) to access the online lab report worksheet for this experiment. The worksheet has to be completed
30 EXPERIMENT 4. REFRACTION OF LIGHT Lab report Go to your course homepage on Sakai (Resources, Lab templates) to access the online lab report worksheet for this experiment. The worksheet has to be completed
Physics Lab #2: Spectroscopy
 Physics 10263 Lab #2: Spectroscopy Introduction This lab is meant to serve as an introduction to the science of spectroscopy. In this lab, we ll learn about how emission and absorption works, and we ll
Physics 10263 Lab #2: Spectroscopy Introduction This lab is meant to serve as an introduction to the science of spectroscopy. In this lab, we ll learn about how emission and absorption works, and we ll
Practical 1P4 Energy Levels and Band Gaps
 Practical 1P4 Energy Levels and Band Gaps What you should learn from this practical Science This practical illustrates some of the points from the lecture course on Elementary Quantum Mechanics and Bonding
Practical 1P4 Energy Levels and Band Gaps What you should learn from this practical Science This practical illustrates some of the points from the lecture course on Elementary Quantum Mechanics and Bonding
Visible Spectrometer
 Visible Spectrometer Experiment VIS University of Florida Department of Physics PHY4803L Advanced Physics Laboratory Objective The Balmer spectral lines from a hydrogen discharge lamp are observed with
Visible Spectrometer Experiment VIS University of Florida Department of Physics PHY4803L Advanced Physics Laboratory Objective The Balmer spectral lines from a hydrogen discharge lamp are observed with
Obtain an optical "bench" setup (there should be three sliding mounts on the calibrated horizontal bar. The setup is shown in the diagram below.
 Astronomy 100 Name(s): Exercise 4: Telescopes and spectroscopy Once the various focal issues are resolved, magnification of a small image is a significant consideration for a telescope. Though a planet
Astronomy 100 Name(s): Exercise 4: Telescopes and spectroscopy Once the various focal issues are resolved, magnification of a small image is a significant consideration for a telescope. Though a planet
Practical 1P4 Energy Levels and Band Gaps
 Practical 1P4 Energy Levels and Band Gaps What you should learn from this practical Science This practical illustrates some of the points from the lecture course on Elementary Quantum Mechanics and Bonding
Practical 1P4 Energy Levels and Band Gaps What you should learn from this practical Science This practical illustrates some of the points from the lecture course on Elementary Quantum Mechanics and Bonding
Experiment 7: Spectrum of the Hydrogen Atom
 Experiment 7: Spectrum of the Hydrogen Nate Saffold nas2173@columbia.edu Office Hour: Mondays, 5:30-6:30PM INTRO TO EXPERIMENTAL PHYS-LAB 1493/1494/2699 Introduction The physics behind: The spectrum of
Experiment 7: Spectrum of the Hydrogen Nate Saffold nas2173@columbia.edu Office Hour: Mondays, 5:30-6:30PM INTRO TO EXPERIMENTAL PHYS-LAB 1493/1494/2699 Introduction The physics behind: The spectrum of
PHYSICS 122/124 Lab EXPERIMENT NO. 9 ATOMIC SPECTRA
 PHYSICS 1/14 Lab EXPERIMENT NO. 9 ATOMIC SPECTRA The purpose of this laboratory is to study energy levels of the Hydrogen atom by observing the spectrum of emitted light when Hydrogen atoms make transitions
PHYSICS 1/14 Lab EXPERIMENT NO. 9 ATOMIC SPECTRA The purpose of this laboratory is to study energy levels of the Hydrogen atom by observing the spectrum of emitted light when Hydrogen atoms make transitions
UNIVERSITY OF CALIFORNIA - SANTA CRUZ DEPARTMENT OF PHYSICS PHYS 133 PROFESSOR: SHER. Atomic Spectra. Benjamin Stahl
 UNIVERSITY OF CALIFORNIA - SANTA CRUZ DEPARTMENT OF PHYSICS PHYS 133 PROFESSOR: SHER Atomic Spectra Benjamin Stahl Lab Partners: Aaron Lopez & Dillon Teal April 2, 2014 Abstract As an introduction to spectroscopy,
UNIVERSITY OF CALIFORNIA - SANTA CRUZ DEPARTMENT OF PHYSICS PHYS 133 PROFESSOR: SHER Atomic Spectra Benjamin Stahl Lab Partners: Aaron Lopez & Dillon Teal April 2, 2014 Abstract As an introduction to spectroscopy,
EXPERIMENT 5:Determination of the refractive index (µ) of the material of a prism using sprectometer
 EXPERIMENT 5:Determination of the refractive index (µ) of the material of a prism using sprectometer Debangshu Mukherjee B.Sc Physics,1st year Chennai Mathematical Institute 17.10.008 1 Aim of Experiment
EXPERIMENT 5:Determination of the refractive index (µ) of the material of a prism using sprectometer Debangshu Mukherjee B.Sc Physics,1st year Chennai Mathematical Institute 17.10.008 1 Aim of Experiment
Experiment #9. Atomic Emission Spectroscopy
 Introduction Experiment #9. Atomic Emission Spectroscopy Spectroscopy is the study of the interaction of light with matter. This interaction can be in the form of the absorption or the emission of electromagnetic
Introduction Experiment #9. Atomic Emission Spectroscopy Spectroscopy is the study of the interaction of light with matter. This interaction can be in the form of the absorption or the emission of electromagnetic
INTRODUCTION TO THE TELESCOPE
 INTRODUCTION TO THE TELESCOPE What will you learn in this Lab? For a few of the labs this semester, you will be using an 8-inch Celestron telescope to take observations. This lab will introduce you to
INTRODUCTION TO THE TELESCOPE What will you learn in this Lab? For a few of the labs this semester, you will be using an 8-inch Celestron telescope to take observations. This lab will introduce you to
PHYS 450 Fall semester Lecture 05: Dispersion and the Prism Spectrometer. Ron Reifenberger Birck Nanotechnology Center Purdue University
 PHYS 450 Fall semester 06 Lecture 05: Dispersion and the Prism Spectrometer Ron Reifenberger Birck Nanotechnology Center Purdue University Lecture 05 Prisms Dispersion of Light n n As early as the 3th
PHYS 450 Fall semester 06 Lecture 05: Dispersion and the Prism Spectrometer Ron Reifenberger Birck Nanotechnology Center Purdue University Lecture 05 Prisms Dispersion of Light n n As early as the 3th
Spectroscopy of Various Light Sources: The Interactions between Light and Matter ASTR 170B1, Spring 2017, Lab #2. 1 Introduction.
 Spectroscopy of Various Light Sources: The Interactions between Light and Matter ASTR 170B1, Spring 2017, Lab #2 DUE IN CLASS ON Thursday Sept 28! You CAN work in a group of 2, but you need only turn in
Spectroscopy of Various Light Sources: The Interactions between Light and Matter ASTR 170B1, Spring 2017, Lab #2 DUE IN CLASS ON Thursday Sept 28! You CAN work in a group of 2, but you need only turn in
Visible Spectrometer
 Visible Spectrometer Experiment VIS University of Florida Department of Physics PHY4803L Advanced Physics Laboratory Objective The Balmer spectral lines from a hydrogen discharge lamp are observed with
Visible Spectrometer Experiment VIS University of Florida Department of Physics PHY4803L Advanced Physics Laboratory Objective The Balmer spectral lines from a hydrogen discharge lamp are observed with
Blackbody Radiation EX-9920 ScienceWorkshop Page 1 of 8. Blackbody Radiation
 Blackbody Radiation EX-9920 ScienceWorkshop Page 1 of 8 EQUIPMENT Blackbody Radiation INCLUDED: 1 Prism Spectrophotometer Kit OS-8544 1 Optics Bench (60 cm) OS-8541 1 Spectrophotometer Accessory Kit OS-8537
Blackbody Radiation EX-9920 ScienceWorkshop Page 1 of 8 EQUIPMENT Blackbody Radiation INCLUDED: 1 Prism Spectrophotometer Kit OS-8544 1 Optics Bench (60 cm) OS-8541 1 Spectrophotometer Accessory Kit OS-8537
PC1144 Physics IV. Atomic Spectra
 PC1144 Physics IV Atomic Spectra 1 Objectives Investigate how well the visible light wavelengths of hydrogen predicted by the Bohr theory agree with experimental values. Determine an experimental value
PC1144 Physics IV Atomic Spectra 1 Objectives Investigate how well the visible light wavelengths of hydrogen predicted by the Bohr theory agree with experimental values. Determine an experimental value
ONE-ELECTRON AND TWO-ELECTRON SPECTRA
 ONE-ELECTRON AND TWO-ELECTRON SPECTRA (A) FINE STRUCTURE AND ONE-ELECTRON SPECTRUM PRINCIPLE AND TASK The well-known spectral lines of He are used for calibrating the diffraction spectrometer. The wavelengths
ONE-ELECTRON AND TWO-ELECTRON SPECTRA (A) FINE STRUCTURE AND ONE-ELECTRON SPECTRUM PRINCIPLE AND TASK The well-known spectral lines of He are used for calibrating the diffraction spectrometer. The wavelengths
Introduction to Telescopes Pre-lab
 AST 114 Spring 2005 Introduction to Telescopes Introduction to Telescopes Pre-lab 1. Read through the "Parts of a Telescope" section of the lab. You may be asked to point out the different parts of the
AST 114 Spring 2005 Introduction to Telescopes Introduction to Telescopes Pre-lab 1. Read through the "Parts of a Telescope" section of the lab. You may be asked to point out the different parts of the
Fig. 8.1 illustrates the three measurements. air medium A. ray 1. air medium A. ray 2. air medium A. ray 3. Fig For Examiner s Use
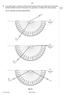 9 9 9 14 8 In an optics lesson, a Physics student traces the paths of three s of light near the boundary between medium A and. The student uses a protractor to measure the various angles. Fig. 8.1 illustrates
9 9 9 14 8 In an optics lesson, a Physics student traces the paths of three s of light near the boundary between medium A and. The student uses a protractor to measure the various angles. Fig. 8.1 illustrates
Since focal length = focal power
 RAY OPTICS PREVIOUS EAMCET BITS (ENGINEERING ). The two lenses of an achromatic doublet should have : [EAMCET 009 E] ) equal powers ) equal dispersive powers ) equal ratio of their power and dispersive
RAY OPTICS PREVIOUS EAMCET BITS (ENGINEERING ). The two lenses of an achromatic doublet should have : [EAMCET 009 E] ) equal powers ) equal dispersive powers ) equal ratio of their power and dispersive
OPTICAL INSTRUMENTS VERY SHORT ANSWER QUESTIONS
 OPTICAL INSTRUMENTS VERY SHORT ANSWER QUESTIONS Q-1. The difference in the focal lengths of the two lenses is larger in which case microscope or telescope? Q-2. What is the formula for angular magnification
OPTICAL INSTRUMENTS VERY SHORT ANSWER QUESTIONS Q-1. The difference in the focal lengths of the two lenses is larger in which case microscope or telescope? Q-2. What is the formula for angular magnification
PHYSICS 116 SPECTROSCOPY: DETERMINATION OF THE WAVELENGTH OF LIGHT
 Name Date Lab Time Lab TA PHYSICS 116 SPECTROSCOPY: DETERMINATION OF THE WAVELENGTH OF LIGHT I. PURPOSE To use a diffraction grating to investigate the spectra produced by several unknown gas discharge
Name Date Lab Time Lab TA PHYSICS 116 SPECTROSCOPY: DETERMINATION OF THE WAVELENGTH OF LIGHT I. PURPOSE To use a diffraction grating to investigate the spectra produced by several unknown gas discharge
EXPERIMENT 12 THE WAVELENGTH OF LIGHT; THE DIFFRACTION GRATING
 EXPERIMENT 12 THE WAVELENGTH OF LIGHT; THE DIFFRACTION GRATING INTRODUCTION: One of the most fascinating chapters in the history of physics has been the search for an understanding of the true nature of
EXPERIMENT 12 THE WAVELENGTH OF LIGHT; THE DIFFRACTION GRATING INTRODUCTION: One of the most fascinating chapters in the history of physics has been the search for an understanding of the true nature of
where c m s (1)
 General Physics Experiment 6 Spectrum of Hydrogen s Emission Lines Objectives: < To determine wave lengths of the bright emission lines of hydrogen. < To test the relationship between wavelength and energy
General Physics Experiment 6 Spectrum of Hydrogen s Emission Lines Objectives: < To determine wave lengths of the bright emission lines of hydrogen. < To test the relationship between wavelength and energy
Modern Physics Laboratory MP2 Blackbody Radiation
 Purpose MP2 Blackbody Radiation In this experiment, you will investigate the spectrum of the blackbody radiation and its dependence on the temperature of the body. Equipment and components Tungsten light
Purpose MP2 Blackbody Radiation In this experiment, you will investigate the spectrum of the blackbody radiation and its dependence on the temperature of the body. Equipment and components Tungsten light
Exp. P-6 Blackbody Radiation
 Exp. P-6 Blackbody Radiation Updated Feb 2019 by A. Azelis, M. Hohlmann Equipment Prism Spectrophotometrer Kit Optics Bench (60 cm) Spectrophotometer Accessory Kit Aperture Bracket High Sensitivity Light
Exp. P-6 Blackbody Radiation Updated Feb 2019 by A. Azelis, M. Hohlmann Equipment Prism Spectrophotometrer Kit Optics Bench (60 cm) Spectrophotometer Accessory Kit Aperture Bracket High Sensitivity Light
PAPER 338 OPTICAL AND INFRARED ASTRONOMICAL TELESCOPES AND INSTRUMENTS
 MATHEMATICAL TRIPOS Part III Monday, 12 June, 2017 1:30 pm to 3:30 pm PAPER 338 OPTICAL AND INFRARED ASTRONOMICAL TELESCOPES AND INSTRUMENTS Attempt no more than TWO questions. There are THREE questions
MATHEMATICAL TRIPOS Part III Monday, 12 June, 2017 1:30 pm to 3:30 pm PAPER 338 OPTICAL AND INFRARED ASTRONOMICAL TELESCOPES AND INSTRUMENTS Attempt no more than TWO questions. There are THREE questions
Atomic Theory: Spectroscopy and Flame Tests
 Atomic Theory: Spectroscopy and Flame Tests Introduction Light energy is also known as electromagnetic (EM) radiation. The light that we observe with our eyes, visible light, is just a small portion of
Atomic Theory: Spectroscopy and Flame Tests Introduction Light energy is also known as electromagnetic (EM) radiation. The light that we observe with our eyes, visible light, is just a small portion of
Physics 231 Lab 8 & 9
 Physics 231 Lab 8 & 9 Atomic Spectra and Energy & Momentum for a multi-particle system (you) Name: KEY Partner: Equipment: Force Plate, Motion Sensor mounted on high rod, hydrogen emission tubes, hand-held
Physics 231 Lab 8 & 9 Atomic Spectra and Energy & Momentum for a multi-particle system (you) Name: KEY Partner: Equipment: Force Plate, Motion Sensor mounted on high rod, hydrogen emission tubes, hand-held
