PHY320 Class Test Topic 1 Elemental Abundances All questions are worth 1 mark unless otherwise stated
|
|
|
- Lesley O’Brien’
- 5 years ago
- Views:
Transcription
1 Topic 1 Elemental Abundances 1. What is the origin of the Earth s atmosphere? 2. Name the 2 distinct topographical regions on the Moon. 3. In the model of chemical affinities which class of elements forms much of the Earth s mantle? 4. What evidence is there to support the idea that the Moon was once in a molten state? 5. What type of meteorite is most abundantly found on Earth? 6. What is a chondrule? 7. In the study of the Earth s structure using seismic information which of the 2 types of body wave is able to pass through all materials? 8. It is widely believed that the Russian dissident Alexander Litvinenko was poisoned using Polonium-210, an alpha emitter. It is estimated that he ingested 10µgrams of the substance in a London sushi bar on 1 st November Given that the half-life of Polonium-210 is 138 days estimate how much of the 10 µgrams of Polonium-210 was present in his body at the time of his death on 23 rd November (3 marks) Topic 2 Spectral Lines k = 1.38 x J K -1 = 8.62 x 10-5 ev K -1 m H = x kg = MeV/c 2 9. Why are spectral lines in the UV region not detected by an observer on Earth? 10. Which theory is responsible for the existence of natural broadening?
2 11. Why are Lithium and Beryllium more common on Earth than in the Sun? 12. Of the two star populations I and II which has the highest heavy element content? 13. Using the graph in the lecture notes estimate the peak emission wavelength (in Å or nm) of a star with the surface temperature of 7000K. 14. The Wien Law gives the wavelength at which the intensity radiated by a black body at a particular temperature is a maximum. It can be written as: λ max T = x 10-3 m K Use this relationship to estimate the temperatures equivalent to the colours violet (420 nm), green (530 nm), and red (650 nm) (2 marks) 15. Calculate the FWHM of the thermal Doppler broadening of the hydrogen-alpha spectral line (wavelength=6563å) due to the Maxwellian velocity distribution of the hydrogen if its temperature is 6000K. (3 marks) Topic 3 Primordial Nucleosynthesis Boltzmann s constant, k B = 1.38 x J.K -1 = 8.62 x 10-5 ev.k -1 Mass of a proton, m p = x kg = MeV.c -2 Mass of a neutron, m n = x kg = MeV.c -2 1 parsec (pc) = 3.08 x m 16. Give 2 pieces of observational evidence for the Big Bang. 17. What is the mean photon energy at a temperature of K? 18. List 4 elements that were formed during primordial nucleosynthesis. 19. What is the recessional velocity of a galaxy with redshift z=0.183? 20. What is the current temperature of the Cosmic Microwave Background? 21. Using the upper limit of the Hubble constant from the lecture notes calculate the age of the Universe in years. 22. Place the following 6 events in the order that they occur after the Big Bang (4 marks)
3 a. Formation of hadrons (baryons and mesons) b. Formation of galaxies c. Neutrinos decouple from matter d. Photons decouple, origin of CMB e. All fundamental forces are unified f. Primordial nucleosynthesis starts Topic 4 Thermonuclear Fusion 23. What is the source of thermal energy that enables nuclei to undergo fusion in stars? 24. What process hinders nuclear fusion? 25. Which reaction dominates the timescale in the pp chain? 26. What is meant by photo-disintegration? 27. Why does the synthesis of elements in thermonuclear fusion stop at Iron? 28. Why is there high nitrogen abundance during the CNO cycle? 29. What is the final evolutionary stage of a star of mass less than 1.4 solar masses? 30. From the details of the ppi chain in the lecture notes calculate the net release of energy per proton in the formation of one 4 He nucleus. Include the energy from the annihilation of the positrons. (show your working, 3 marks) Topic 5 Neutron Capture 31. From the portion of the chart of nuclides overleaf, or otherwise, write X down 3 isotopes in the form ZA N 32. Where does the local approximation equality break down? 33. What are typical fluxes for the r- and s- process to occur?
4 34. Write out the following nuclear reactions and their effects on a nucleus with Z protons and A nucleons. (4 marks) For example: (n, γ) is equivalent to: (A,Z) + n (A+1,Z) + γ (a) (p,γ) (b) (α,γ) (c) inverse beta-decay (d) (d,p) (d=deuteron) 35. What mechanism terminates the r-process in the mass region 230 < A < 270? 36. What is the line of beta stability? 37. Why do s-process neutrons tend to have a Maxwell-Boltzmann type distribution? 38. Draw the path of the s-process on the part of the chart of nuclides, starting at 56 Fe and ending at 64 Cu (Shaded boxes with a percentage value, (e.g %) are isotopes that are stable against β decay, other boxes with half-lives (e.g d) are isotopes that do beta decay). (2 marks) Topic 6 - Supernovae
5 n = amu 4 He = amu 12 C = amu 16 O = amu 1 amu = MeV/c 2 = 1.66x10-27 kg 39. What is the key feature that distinguishes SN of types I and II? 40. The following reaction takes place in a Red Giant, calculate its Q-value. 12 C + 4 He 16 O + n 41. Is the above reaction exothermic or endothermic? 42. Write down the reaction for the photodissociation of Iron in the core of a star prior to a type II supernova. 43. Under what circumstances might a hydrogen/helium flash (nova) occur? 44. How have type Ia SN been used recently in cosmology? 45. What objects are formed from the core remnant of a type II SN? 46. Explain the lack of H emission and absorption lines in type I SN. 47. What reaction governs the late light curve in most SN? What is the critical timescale here? (2 marks) 48. What is the key method of transportation of energy out of a SN explosion? What experimental evidence do we have to support this? (2 marks) Topic 7 Cosmic Rays 49. What is a primary cosmic ray? 50. Write down the decay of an anti-muon (positively charged muon) into a positron and neutrinos. 51. Name 2 possible sources of the highest energy cosmic rays.
6 52. What is fluorescence? 53. Why is it difficult to pinpoint the sources of cosmic rays? 54. Do recent results from AUGER confirm or reject the GZK hypothesis? 55. An electron travels with velocity 2.5x10 8 m.s -1 through a medium of refractive index 1.3. Using the relationship in your notes, what is the angle between the electron and the resulting Cerenkov photons? (show your working, 3 marks). 56. A detector records 2500 cosmic rays per hour at 1 GeV, how many would it detect per hour at 20 GeV assuming the detector is 100% efficient at both energies (assume an E -2.6 energy dependence of the cosmic ray spectrum)? (2 marks) 57. What is the principal decay product of cosmic rays as seen at ground level? Topic 8 Galactic Chemical Evolution 58. The return fraction R represents the fraction of a star that is returned to the ISM. What are the 2 main ways in which this happens? (2 marks 59. What is meant by metallicity? 60. Why are so-called dredge-up events important? 61. What is a primary element? 62. In the so-called Simple GCE model it is assumed that the system is isolated what does this mean? 63. What is the problem in assuming that the initial conditions of a system is a gas containing primordial nucleosynthesis abundances?
IB Test. Astrophysics HL. Name_solution / a) Describe what is meant by a nebula [1]
![IB Test. Astrophysics HL. Name_solution / a) Describe what is meant by a nebula [1] IB Test. Astrophysics HL. Name_solution / a) Describe what is meant by a nebula [1]](/thumbs/73/69495220.jpg) IB Test Astrophysics HL Name_solution / 47 1. a) Describe what is meant by a nebula [1] an intergalactic cloud of gas and dust where all stars begin to form b) Explain how the Jeans criterion applies to
IB Test Astrophysics HL Name_solution / 47 1. a) Describe what is meant by a nebula [1] an intergalactic cloud of gas and dust where all stars begin to form b) Explain how the Jeans criterion applies to
Astrophysical Nucleosynthesis
 R. D. Gehrz ASTRO 2001, Fall Semester 2018 1 RDG The Chemical Evolution of the Universe 2RDG 1 The Stellar Evolution Cycle 3 RDG a v a v X X V = v a + v X 4 RDG reaction rate r n n s cm ax a X r r ( E)
R. D. Gehrz ASTRO 2001, Fall Semester 2018 1 RDG The Chemical Evolution of the Universe 2RDG 1 The Stellar Evolution Cycle 3 RDG a v a v X X V = v a + v X 4 RDG reaction rate r n n s cm ax a X r r ( E)
Neutron-to-proton ratio
 Neutron-to-proton ratio After one second, the Universe had cooled to 10 13 K. The Universe was filled with protons, neutrons, electrons, and neutrinos. The temperature was high enough that they interconverted
Neutron-to-proton ratio After one second, the Universe had cooled to 10 13 K. The Universe was filled with protons, neutrons, electrons, and neutrinos. The temperature was high enough that they interconverted
Nuclear Physics and Nuclear Reactions
 Slide 1 / 33 Nuclear Physics and Nuclear Reactions The Nucleus Slide 2 / 33 Proton: The charge on a proton is +1.6x10-19 C. The mass of a proton is 1.6726x10-27 kg. Neutron: The neutron is neutral. The
Slide 1 / 33 Nuclear Physics and Nuclear Reactions The Nucleus Slide 2 / 33 Proton: The charge on a proton is +1.6x10-19 C. The mass of a proton is 1.6726x10-27 kg. Neutron: The neutron is neutral. The
Alta Chemistry CHAPTER 25. Nuclear Chemistry: Radiation, Radioactivity & its Applications
 CHAPTER 25 Nuclear Chemistry: Radiation, Radioactivity & its Applications Nuclear Chemistry Nuclear Chemistry deals with changes in the nucleus The nucleus of an atom contains Protons Positively Charged
CHAPTER 25 Nuclear Chemistry: Radiation, Radioactivity & its Applications Nuclear Chemistry Nuclear Chemistry deals with changes in the nucleus The nucleus of an atom contains Protons Positively Charged
Lecture 19 Nuclear Astrophysics. Baryons, Dark Matter, Dark Energy. Experimental Nuclear Physics PHYS 741
 Lecture 19 Nuclear Astrophysics Baryons, Dark Matter, Dark Energy Experimental Nuclear Physics PHYS 741 heeger@wisc.edu References and Figures from: - Haxton, Nuclear Astrophysics - Basdevant, Fundamentals
Lecture 19 Nuclear Astrophysics Baryons, Dark Matter, Dark Energy Experimental Nuclear Physics PHYS 741 heeger@wisc.edu References and Figures from: - Haxton, Nuclear Astrophysics - Basdevant, Fundamentals
CHAPTER 7 TEST REVIEW
 IB PHYSICS Name: Period: Date: # Marks: 94 Raw Score: IB Curve: DEVIL PHYSICS BADDEST CLASS ON CAMPUS CHAPTER 7 TEST REVIEW 1. An alpha particle is accelerated through a potential difference of 10 kv.
IB PHYSICS Name: Period: Date: # Marks: 94 Raw Score: IB Curve: DEVIL PHYSICS BADDEST CLASS ON CAMPUS CHAPTER 7 TEST REVIEW 1. An alpha particle is accelerated through a potential difference of 10 kv.
Interactions. Laws. Evolution
 Lecture Origin of the Elements MODEL: Origin of the Elements or Nucleosynthesis Fundamental Particles quarks, gluons, leptons, photons, neutrinos + Basic Forces gravity, electromagnetic, nuclear Interactions
Lecture Origin of the Elements MODEL: Origin of the Elements or Nucleosynthesis Fundamental Particles quarks, gluons, leptons, photons, neutrinos + Basic Forces gravity, electromagnetic, nuclear Interactions
NJCTL.org 2015 AP Physics 2 Nuclear Physics
 AP Physics 2 Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
AP Physics 2 Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
John Ellison University of California, Riverside. Quarknet 2008 at UCR
 Overview of Particle Physics John Ellison University of California, Riverside Quarknet 2008 at UCR 1 Particle Physics What is it? Study of the elementary constituents of matter And the fundamental forces
Overview of Particle Physics John Ellison University of California, Riverside Quarknet 2008 at UCR 1 Particle Physics What is it? Study of the elementary constituents of matter And the fundamental forces
Topic 7. Relevance to the course
 Topic 7 Cosmic Rays Relevance to the course Need to go back to the elemental abundance curve Isotopes of certain low A elements such as Li, Be and B have larger abundances on Earth than you would expect
Topic 7 Cosmic Rays Relevance to the course Need to go back to the elemental abundance curve Isotopes of certain low A elements such as Li, Be and B have larger abundances on Earth than you would expect
Lecture PowerPoints. Chapter 33 Physics: Principles with Applications, 7 th edition Giancoli
 Lecture PowerPoints Chapter 33 Physics: Principles with Applications, 7 th edition Giancoli This work is protected by United States copyright laws and is provided solely for the use of instructors in teaching
Lecture PowerPoints Chapter 33 Physics: Principles with Applications, 7 th edition Giancoli This work is protected by United States copyright laws and is provided solely for the use of instructors in teaching
Nuclear Astrophysics - I
 Nuclear Astrophysics - I Carl Brune Ohio University, Athens Ohio Exotic Beam Summer School 2016 July 20, 2016 Astrophysics and Cosmology Observations Underlying Physics Electromagnetic Spectrum: radio,
Nuclear Astrophysics - I Carl Brune Ohio University, Athens Ohio Exotic Beam Summer School 2016 July 20, 2016 Astrophysics and Cosmology Observations Underlying Physics Electromagnetic Spectrum: radio,
Isotropy and Homogeneity
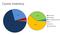 Cosmic inventory Isotropy and Homogeneity On large scales the Universe is isotropic (looks the same in all directions) and homogeneity (the same average density at all locations. This is determined from
Cosmic inventory Isotropy and Homogeneity On large scales the Universe is isotropic (looks the same in all directions) and homogeneity (the same average density at all locations. This is determined from
ORIGIN OF THE ELEMENETS
 VISUAL PHYSICS ONLINE ORIGIN OF THE ELEMENETS Watch Video: The Origin of the Elements The ordinary matter in our universe (known as baryonic matter) is made up of 94 naturally occurring elements. It is
VISUAL PHYSICS ONLINE ORIGIN OF THE ELEMENETS Watch Video: The Origin of the Elements The ordinary matter in our universe (known as baryonic matter) is made up of 94 naturally occurring elements. It is
Nuclear Physics Questions. 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of?
 Nuclear Physics Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
Nuclear Physics Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
99 Years from Discovery : What is our current picture on Cosmic Rays? #6 How cosmic rays travel to Earth? Presented by Nahee Park
 99 Years from Discovery : What is our current picture on Cosmic Rays? #6 How cosmic rays travel to Earth? Presented by Nahee Park #5 How do Cosmic Rays gain their energy? I. Acceleration mechanism of CR
99 Years from Discovery : What is our current picture on Cosmic Rays? #6 How cosmic rays travel to Earth? Presented by Nahee Park #5 How do Cosmic Rays gain their energy? I. Acceleration mechanism of CR
Nuclear Physics Part 2A: Radioactive Decays
 Nuclear Physics Part 2A: Radioactive Decays Last modified: 23/10/2018 Links What is a Decay? Alpha Decay Definition Q-value Example Not Every Alpha Decay is Possible Beta Decay β rays are electrons Anti-particles
Nuclear Physics Part 2A: Radioactive Decays Last modified: 23/10/2018 Links What is a Decay? Alpha Decay Definition Q-value Example Not Every Alpha Decay is Possible Beta Decay β rays are electrons Anti-particles
Fundamental Stellar Parameters. Radiative Transfer. Stellar Atmospheres. Equations of Stellar Structure
 Fundamental Stellar Parameters Radiative Transfer Stellar Atmospheres Equations of Stellar Structure Nuclear Reactions in Stellar Interiors Binding Energy Coulomb Barrier Penetration Hydrogen Burning Reactions
Fundamental Stellar Parameters Radiative Transfer Stellar Atmospheres Equations of Stellar Structure Nuclear Reactions in Stellar Interiors Binding Energy Coulomb Barrier Penetration Hydrogen Burning Reactions
Unity in the Whole Structure
 Cosmology II Unity in the Whole Structure How is it possible by any methods of observation yet known to the astronomer to learn anything about the universe as a whole? It is possible only because the universe,
Cosmology II Unity in the Whole Structure How is it possible by any methods of observation yet known to the astronomer to learn anything about the universe as a whole? It is possible only because the universe,
The origin of the light elements in the early Universe
 1 HUBERT REEVES* The origin of the light elements in the early Universe Shortly after World War II, George Gamov and his collaborators (Alpher et al. 148) considered the possibility that all chemical elements
1 HUBERT REEVES* The origin of the light elements in the early Universe Shortly after World War II, George Gamov and his collaborators (Alpher et al. 148) considered the possibility that all chemical elements
In the Beginning. After about three minutes the temperature had cooled even further, so that neutrons were able to combine with 1 H to form 2 H;
 In the Beginning Obviously, before we can have any geochemistry we need some elements to react with one another. The most commonly held scientific view for the origin of the universe is the "Big Bang"
In the Beginning Obviously, before we can have any geochemistry we need some elements to react with one another. The most commonly held scientific view for the origin of the universe is the "Big Bang"
H 1. Nuclear Physics. Nuclear Physics. 1. Parts of Atom. A. Nuclear Structure. 2b. Nomenclature. 2. Isotopes. AstroPhysics Notes
 AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia Nuclear Physics A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions Updated: 0Feb07 Rough draft A. Nuclear Structure. Parts of Atom. Parts of
AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia Nuclear Physics A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions Updated: 0Feb07 Rough draft A. Nuclear Structure. Parts of Atom. Parts of
Nucleosíntesis primordial
 Tema 5 Nucleosíntesis primordial Asignatura de Física Nuclear Curso académico 2009/2010 Universidad de Santiago de Compostela Big Bang cosmology 1.1 The Universe today The present state of the Universe
Tema 5 Nucleosíntesis primordial Asignatura de Física Nuclear Curso académico 2009/2010 Universidad de Santiago de Compostela Big Bang cosmology 1.1 The Universe today The present state of the Universe
The Early Universe and the Big Bang
 The Early Universe and the Big Bang Class 24 Prof J. Kenney June 28, 2018 Final Exam: Friday June 29 at 2-5pm in Watson A48 What the Final Exam will emphasize: Classroom lectures 10-24 (starting FRI June
The Early Universe and the Big Bang Class 24 Prof J. Kenney June 28, 2018 Final Exam: Friday June 29 at 2-5pm in Watson A48 What the Final Exam will emphasize: Classroom lectures 10-24 (starting FRI June
= : K A
 Atoms and Nuclei. State two limitations of JJ Thomson s model of atom. 2. Write the SI unit for activity of a radioactive substance. 3. What observations led JJ Thomson to conclusion that all atoms have
Atoms and Nuclei. State two limitations of JJ Thomson s model of atom. 2. Write the SI unit for activity of a radioactive substance. 3. What observations led JJ Thomson to conclusion that all atoms have
32 IONIZING RADIATION, NUCLEAR ENERGY, AND ELEMENTARY PARTICLES
 32 IONIZING RADIATION, NUCLEAR ENERGY, AND ELEMENTARY PARTICLES 32.1 Biological Effects of Ionizing Radiation γ-rays (high-energy photons) can penetrate almost anything, but do comparatively little damage.
32 IONIZING RADIATION, NUCLEAR ENERGY, AND ELEMENTARY PARTICLES 32.1 Biological Effects of Ionizing Radiation γ-rays (high-energy photons) can penetrate almost anything, but do comparatively little damage.
H 1. Nuclear Physics. Nuclear Physics. 1. Parts of Atom. 2. Isotopes. AstroPhysics Notes. Dr. Bill Pezzaglia. Rough draft. A.
 AstroPhysics Notes Tom Lehrer: Elements Dr. Bill Pezzaglia Nuclear Physics Updated: 0Feb Rough draft Nuclear Physics A. Nuclear Structure A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions. Parts
AstroPhysics Notes Tom Lehrer: Elements Dr. Bill Pezzaglia Nuclear Physics Updated: 0Feb Rough draft Nuclear Physics A. Nuclear Structure A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions. Parts
Cosmic Rays in the Galaxy
 1, Over View Cosmic Rays in the Galaxy Discovery : Legendary baloon flight of Victor Hess Observation of Cosmic Rays : Satellite, Balloon (Direct), Air shower (Indirect) Energy Spectrum of Cosmic Rays
1, Over View Cosmic Rays in the Galaxy Discovery : Legendary baloon flight of Victor Hess Observation of Cosmic Rays : Satellite, Balloon (Direct), Air shower (Indirect) Energy Spectrum of Cosmic Rays
BIG BANG SUMMARY NOTES
 BIG BANG SUMMARY NOTES BIG BANG THEORY Studies of red-shifts of distant galaxies show that the universe is expanding. This and other observations has led to the Big Bang Theory The Big Bang Theory claims
BIG BANG SUMMARY NOTES BIG BANG THEORY Studies of red-shifts of distant galaxies show that the universe is expanding. This and other observations has led to the Big Bang Theory The Big Bang Theory claims
Physics 3204 UNIT 3 Test Matter Energy Interface
 Physics 3204 UNIT 3 Test Matter Energy Interface 2005 2006 Time: 60 minutes Total Value: 33 Marks Formulae and Constants v = f λ E = hf h f = E k + W 0 E = m c 2 p = h λ 1 A= A T 0 2 t 1 2 E k = ½ mv 2
Physics 3204 UNIT 3 Test Matter Energy Interface 2005 2006 Time: 60 minutes Total Value: 33 Marks Formulae and Constants v = f λ E = hf h f = E k + W 0 E = m c 2 p = h λ 1 A= A T 0 2 t 1 2 E k = ½ mv 2
Nucleosynthesis. W. F. McDonough 1. Neutrino Science, Tohoku University, Sendai , Japan. (Dated: Tuesday 24 th April, 2018)
 Nucleosynthesis W. F. McDonough 1 1 Department of Earth Sciences and Research Center for Neutrino Science, Tohoku University, Sendai 980-8578, Japan (Dated: Tuesday 24 th April, 2018) Goals Where were
Nucleosynthesis W. F. McDonough 1 1 Department of Earth Sciences and Research Center for Neutrino Science, Tohoku University, Sendai 980-8578, Japan (Dated: Tuesday 24 th April, 2018) Goals Where were
ASTR 1120 General Astronomy: Stars & Galaxies. OUR Universe: Accelerating Universe
 ASTR 1120 General Astronomy: Stars & Galaxies FINAL: Saturday, Dec 12th, 7:30pm, HERE ALTERNATE FINAL: Monday, Dec 7th, 5:30pm in Muenzinger E131 Last OBSERVING session, Tue, Dec.8th, 7pm Please check
ASTR 1120 General Astronomy: Stars & Galaxies FINAL: Saturday, Dec 12th, 7:30pm, HERE ALTERNATE FINAL: Monday, Dec 7th, 5:30pm in Muenzinger E131 Last OBSERVING session, Tue, Dec.8th, 7pm Please check
Astrochemistry. Special course in astronomy 53855, 5 op. Jorma Harju, Julien Montillaud, Olli Sipilä. Department of Physics
 Astrochemistry Special course in astronomy 53855, 5 op Jorma Harju, Julien Montillaud, Olli Sipilä Department of Physics Spring 2013, Fridays 12:15-13:45, Lecture room D117 Course web page http://www.courses.physics.helsinki.fi/astro/astrokemia
Astrochemistry Special course in astronomy 53855, 5 op Jorma Harju, Julien Montillaud, Olli Sipilä Department of Physics Spring 2013, Fridays 12:15-13:45, Lecture room D117 Course web page http://www.courses.physics.helsinki.fi/astro/astrokemia
Unity in the Whole Structure Evolution of the Universe from 13 to 4 Billion Years Ago
 Unity in the Whole Structure Evolution of the Universe from 13 to 4 Billion Years Ago Prof. Dr. Harold Geller hgeller@gmu.edu http://physics.gmu.edu/~hgeller/ Department of Physics and Astronomy George
Unity in the Whole Structure Evolution of the Universe from 13 to 4 Billion Years Ago Prof. Dr. Harold Geller hgeller@gmu.edu http://physics.gmu.edu/~hgeller/ Department of Physics and Astronomy George
1) Radioactive Decay, Nucleosynthesis, and Basic Geochronology
 1) Radioactive Decay, Nucleosynthesis, and Basic Geochronology Reading (all from White s Notes) Lecture 1: Introduction And Physics Of The Nucleus: Skim Lecture 1: Radioactive Decay- Read all Lecture 3:
1) Radioactive Decay, Nucleosynthesis, and Basic Geochronology Reading (all from White s Notes) Lecture 1: Introduction And Physics Of The Nucleus: Skim Lecture 1: Radioactive Decay- Read all Lecture 3:
Primordial (Big Bang) Nucleosynthesis
 Primordial (Big Bang) Nucleosynthesis H Li Be Which elements? He METALS - 1942: Gamow suggests a Big Bang origin of the elements. - 1948: Alpher, Bethe & Gamow: all elements are synthesized minutes after
Primordial (Big Bang) Nucleosynthesis H Li Be Which elements? He METALS - 1942: Gamow suggests a Big Bang origin of the elements. - 1948: Alpher, Bethe & Gamow: all elements are synthesized minutes after
Plasma Universe. The origin of CMB
 Plasma Universe As we go back in time, temperature goes up. T=2.73(1+z) K At z~1100, T~3000 K About the same temperature as M-dwarfs Ionization of hydrogen atoms H + photon! p + e - Inverse process: recombination
Plasma Universe As we go back in time, temperature goes up. T=2.73(1+z) K At z~1100, T~3000 K About the same temperature as M-dwarfs Ionization of hydrogen atoms H + photon! p + e - Inverse process: recombination
1 (a) Explain what is meant by a white dwarf when describing the evolution of a star [1]
![1 (a) Explain what is meant by a white dwarf when describing the evolution of a star [1] 1 (a) Explain what is meant by a white dwarf when describing the evolution of a star [1]](/thumbs/79/79642472.jpg) 1 (a) Explain what is meant by a white dwarf when describing the evolution of a star.... [1] (b) Antares is a red giant and one of the brightest stars in the night sky. The parallax angle for this star
1 (a) Explain what is meant by a white dwarf when describing the evolution of a star.... [1] (b) Antares is a red giant and one of the brightest stars in the night sky. The parallax angle for this star
Thursday, April 23, 15. Nuclear Physics
 Nuclear Physics Some Properties of Nuclei! All nuclei are composed of protons and neutrons! Exception is ordinary hydrogen with just a proton! The atomic number, Z, equals the number of protons in the
Nuclear Physics Some Properties of Nuclei! All nuclei are composed of protons and neutrons! Exception is ordinary hydrogen with just a proton! The atomic number, Z, equals the number of protons in the
Brief Introduction to Cosmology
 Brief Introduction to Cosmology Matias Zaldarriaga Harvard University August 2006 Basic Questions in Cosmology: How does the Universe evolve? What is the universe made off? How is matter distributed? How
Brief Introduction to Cosmology Matias Zaldarriaga Harvard University August 2006 Basic Questions in Cosmology: How does the Universe evolve? What is the universe made off? How is matter distributed? How
Lecture 3: Big Bang Nucleosynthesis The First Three Minutes
 Lecture 3: Big Bang Nucleosynthesis The First Three Minutes Last time: particle anti-particle soup --> quark soup --> neutron-proton soup p / n ratio at onset of 2 D formation Today: Form 2 D and 4 He
Lecture 3: Big Bang Nucleosynthesis The First Three Minutes Last time: particle anti-particle soup --> quark soup --> neutron-proton soup p / n ratio at onset of 2 D formation Today: Form 2 D and 4 He
Brock University. Test 1, February, 2017 Number of pages: 9 Course: ASTR 1P02 Number of Students: 480 Date of Examination: February 6, 2017
 Brock University Test 1, February, 2017 Number of pages: 9 Course: ASTR 1P02 Number of Students: 480 Date of Examination: February 6, 2017 Number of hours: 50 min Time of Examination: 18:00 18:50 Instructor:
Brock University Test 1, February, 2017 Number of pages: 9 Course: ASTR 1P02 Number of Students: 480 Date of Examination: February 6, 2017 Number of hours: 50 min Time of Examination: 18:00 18:50 Instructor:
Stars and their properties: (Chapters 11 and 12)
 Stars and their properties: (Chapters 11 and 12) To classify stars we determine the following properties for stars: 1. Distance : Needed to determine how much energy stars produce and radiate away by using
Stars and their properties: (Chapters 11 and 12) To classify stars we determine the following properties for stars: 1. Distance : Needed to determine how much energy stars produce and radiate away by using
Radioactivity is the emission of high energy released when the of atoms change. Radioactivity can be or.
 Chapter 19 1 RADIOACTIVITY Radioactivity is the emission of high energy released when the of atoms change. Radioactivity can be or. TYPES OF RADIATION OR EMITTED ENERGY IN NUCLEAR CHANGES Radiation is
Chapter 19 1 RADIOACTIVITY Radioactivity is the emission of high energy released when the of atoms change. Radioactivity can be or. TYPES OF RADIATION OR EMITTED ENERGY IN NUCLEAR CHANGES Radiation is
Stellar processes, nucleosynthesis OUTLINE
 Stellar processes, nucleosynthesis OUTLINE Reading this week: White 313-326 and 421-464 Today 1. Stellar processes 2. Nucleosynthesis Powerpoint credit: Using significant parts of a WHOI ppt 1 Question
Stellar processes, nucleosynthesis OUTLINE Reading this week: White 313-326 and 421-464 Today 1. Stellar processes 2. Nucleosynthesis Powerpoint credit: Using significant parts of a WHOI ppt 1 Question
lepton era BBN high T neutrinos in equilibrium reaction proceeds if GF=Fermi constant LHS falls with T more rapidly than RHS!
 lepton era BBN high T neutrinos in equilibrium GF=Fermi constant reaction proceeds if LHS falls with T more rapidly than RHS! PAIR ANNIHILATION t=10 s t=0.2 s neutrinos decouple We cannot measure the
lepton era BBN high T neutrinos in equilibrium GF=Fermi constant reaction proceeds if LHS falls with T more rapidly than RHS! PAIR ANNIHILATION t=10 s t=0.2 s neutrinos decouple We cannot measure the
Nuclear Physics and Radioactivity
 Nuclear Physics and Radioactivity Structure and Properties of the Nucleus Nucleus is made of protons and neutrons Proton has positive charge: Neutron is electrically neutral: Neutrons and protons are collectively
Nuclear Physics and Radioactivity Structure and Properties of the Nucleus Nucleus is made of protons and neutrons Proton has positive charge: Neutron is electrically neutral: Neutrons and protons are collectively
Chapter 22. Preview. Objectives Properties of the Nucleus Nuclear Stability Binding Energy Sample Problem. Section 1 The Nucleus
 Section 1 The Nucleus Preview Objectives Properties of the Nucleus Nuclear Stability Binding Energy Sample Problem Section 1 The Nucleus Objectives Identify the properties of the nucleus of an atom. Explain
Section 1 The Nucleus Preview Objectives Properties of the Nucleus Nuclear Stability Binding Energy Sample Problem Section 1 The Nucleus Objectives Identify the properties of the nucleus of an atom. Explain
Today in Astronomy 142
 Today in Astronomy 142! Elementary particles and their interactions, nuclei, and energy generation in stars.! Nuclear fusion reactions in stars TT Cygni: Carbon Star Credit: H. Olofsson (Stockholm Obs.)
Today in Astronomy 142! Elementary particles and their interactions, nuclei, and energy generation in stars.! Nuclear fusion reactions in stars TT Cygni: Carbon Star Credit: H. Olofsson (Stockholm Obs.)
Astronomy 1504 Section 10 Final Exam Version 1 May 6, 1999
 Astronomy 1504 Section 10 Final Exam Version 1 May 6, 1999 Reminder: When I write these questions, I believe that there is one one correct answer. The questions consist of all parts a e. Read the entire
Astronomy 1504 Section 10 Final Exam Version 1 May 6, 1999 Reminder: When I write these questions, I believe that there is one one correct answer. The questions consist of all parts a e. Read the entire
Brock University. Test 1, January, 2015 Number of pages: 9 Course: ASTR 1P02 Number of Students: 500 Date of Examination: January 29, 2015
 Brock University Test 1, January, 2015 Number of pages: 9 Course: ASTR 1P02 Number of Students: 500 Date of Examination: January 29, 2015 Number of hours: 50 min Time of Examination: 18:00 15:50 Instructor:
Brock University Test 1, January, 2015 Number of pages: 9 Course: ASTR 1P02 Number of Students: 500 Date of Examination: January 29, 2015 Number of hours: 50 min Time of Examination: 18:00 15:50 Instructor:
Nuclear Physics. PHY232 Remco Zegers Room W109 cyclotron building.
 Nuclear Physics PHY232 Remco Zegers zegers@nscl.msu.edu Room W109 cyclotron building http://www.nscl.msu.edu/~zegers/phy232.html Periodic table of elements We saw that the periodic table of elements can
Nuclear Physics PHY232 Remco Zegers zegers@nscl.msu.edu Room W109 cyclotron building http://www.nscl.msu.edu/~zegers/phy232.html Periodic table of elements We saw that the periodic table of elements can
Part I: The Dawn of Time
 Part I: The Dawn of Time Topics within Part I. 1. Origins of the Universe: from the Infinite to the Subatomic. 2. Planets and Meteorites: Neighbors in Space 3. Birth of the Earth and Moon: a Coupled System
Part I: The Dawn of Time Topics within Part I. 1. Origins of the Universe: from the Infinite to the Subatomic. 2. Planets and Meteorites: Neighbors in Space 3. Birth of the Earth and Moon: a Coupled System
UNIT VIII ATOMS AND NUCLEI
 UNIT VIII ATOMS AND NUCLEI Weightage Marks : 06 Alpha-particles scattering experiment, Rutherford s model of atom, Bohr Model, energy levels, Hydrogen spectrum. Composition and size of Nucleus, atomic
UNIT VIII ATOMS AND NUCLEI Weightage Marks : 06 Alpha-particles scattering experiment, Rutherford s model of atom, Bohr Model, energy levels, Hydrogen spectrum. Composition and size of Nucleus, atomic
MockTime.com. Ans: (b) Q6. Curie is a unit of [1989] (a) energy of gamma-rays (b) half-life (c) radioactivity (d) intensity of gamma-rays Ans: (c)
![MockTime.com. Ans: (b) Q6. Curie is a unit of [1989] (a) energy of gamma-rays (b) half-life (c) radioactivity (d) intensity of gamma-rays Ans: (c) MockTime.com. Ans: (b) Q6. Curie is a unit of [1989] (a) energy of gamma-rays (b) half-life (c) radioactivity (d) intensity of gamma-rays Ans: (c)](/thumbs/95/125098222.jpg) Chapter Nuclei Q1. A radioactive sample with a half life of 1 month has the label: Activity = 2 micro curies on 1 8 1991. What would be its activity two months earlier? [1988] 1.0 micro curie 0.5 micro
Chapter Nuclei Q1. A radioactive sample with a half life of 1 month has the label: Activity = 2 micro curies on 1 8 1991. What would be its activity two months earlier? [1988] 1.0 micro curie 0.5 micro
Basic Nuclear Theory. Lecture 1 The Atom and Nuclear Stability
 Basic Nuclear Theory Lecture 1 The Atom and Nuclear Stability Introduction Nuclear power is made possible by energy emitted from either nuclear fission or nuclear fusion. Current nuclear power plants utilize
Basic Nuclear Theory Lecture 1 The Atom and Nuclear Stability Introduction Nuclear power is made possible by energy emitted from either nuclear fission or nuclear fusion. Current nuclear power plants utilize
Lecture notes 8: Nuclear reactions in solar/stellar interiors
 Lecture notes 8: Nuclear reactions in solar/stellar interiors Atomic Nuclei We will henceforth often write protons 1 1p as 1 1H to underline that hydrogen, deuterium and tritium are chemically similar.
Lecture notes 8: Nuclear reactions in solar/stellar interiors Atomic Nuclei We will henceforth often write protons 1 1p as 1 1H to underline that hydrogen, deuterium and tritium are chemically similar.
PHYS 420: Astrophysics & Cosmology
 PHYS 420: Astrophysics & Cosmology Dr Richard H. Cyburt Assistant Professor of Physics My office: 402c in the Science Building My phone: (304) 384-6006 My email: rcyburt@concord.edu My webpage: www.concord.edu/rcyburt
PHYS 420: Astrophysics & Cosmology Dr Richard H. Cyburt Assistant Professor of Physics My office: 402c in the Science Building My phone: (304) 384-6006 My email: rcyburt@concord.edu My webpage: www.concord.edu/rcyburt
Card #1/28. Card #2/28. Science Revision P2. Science Revision P2. Science Revision P2. Card #4/28. Topic: F = ma. Topic: Resultant Forces
 Card #1/28 Card #2/28 Topic: Resultant Forces Topic: F = ma Topic: Distance-TIme Graphs Card #3/28 Card #4/28 Topic: Velocity-Time Graphs Card #2/28 Card #1/28 Card #4/28 Card #3/28 Card #5/28 Card #6/28
Card #1/28 Card #2/28 Topic: Resultant Forces Topic: F = ma Topic: Distance-TIme Graphs Card #3/28 Card #4/28 Topic: Velocity-Time Graphs Card #2/28 Card #1/28 Card #4/28 Card #3/28 Card #5/28 Card #6/28
Topics Covered in Chapter. Light and Other Electromagnetic Radiation. A Subatomic Interlude II. A Subatomic Interlude. A Subatomic Interlude III
 Light and Other Electromagnetic Radiation Topics Covered in Chapter 1.Structure of Atoms 2.Origins of Electromagnetic Radiation 3.Objects with Different Temperature and their Electromagnetic Radiation
Light and Other Electromagnetic Radiation Topics Covered in Chapter 1.Structure of Atoms 2.Origins of Electromagnetic Radiation 3.Objects with Different Temperature and their Electromagnetic Radiation
Light and Other Electromagnetic Radiation
 Light and Other Electromagnetic Radiation 1 Topics Covered in Chapter 1.Structure of Atoms 2.Origins of Electromagnetic Radiation 3.Objects with Different Temperature and their Electromagnetic Radiation
Light and Other Electromagnetic Radiation 1 Topics Covered in Chapter 1.Structure of Atoms 2.Origins of Electromagnetic Radiation 3.Objects with Different Temperature and their Electromagnetic Radiation
Chapter 28 Lecture. Nuclear Physics Pearson Education, Inc.
 Chapter 28 Lecture Nuclear Physics Nuclear Physics How are new elements created? What are the natural sources of ionizing radiation? How does carbon dating work? Be sure you know how to: Use the right-hand
Chapter 28 Lecture Nuclear Physics Nuclear Physics How are new elements created? What are the natural sources of ionizing radiation? How does carbon dating work? Be sure you know how to: Use the right-hand
Earlier in time, all the matter must have been squeezed more tightly together and a lot hotter AT R=0 have the Big Bang
 Re-cap from last lecture Discovery of the CMB- logic From Hubble s observations, we know the Universe is expanding This can be understood theoretically in terms of solutions of GR equations Earlier in
Re-cap from last lecture Discovery of the CMB- logic From Hubble s observations, we know the Universe is expanding This can be understood theoretically in terms of solutions of GR equations Earlier in
DEVIL PHYSICS THE BADDEST CLASS ON CAMPUS IB PHYSICS
 DEVIL PHYSICS THE BADDEST CLASS ON CAMPUS IB PHYSICS TSOKOS OPTION E-6 GALAXIES Introductory Video: The Big Bang Theory Objectives Understand the Hubble classification scheme of galaxies and describe the
DEVIL PHYSICS THE BADDEST CLASS ON CAMPUS IB PHYSICS TSOKOS OPTION E-6 GALAXIES Introductory Video: The Big Bang Theory Objectives Understand the Hubble classification scheme of galaxies and describe the
Review Questions for the new topics that will be on the Final Exam
 Review Questions for the new topics that will be on the Final Exam Be sure to review the lecture-tutorials and the material we covered on the first three exams. How does speed differ from velocity? Give
Review Questions for the new topics that will be on the Final Exam Be sure to review the lecture-tutorials and the material we covered on the first three exams. How does speed differ from velocity? Give
The slides with white background you need to know. The slides with blue background just have some cool information.
 The slides with white background you need to know. The slides with blue background just have some cool information. The Big Bang cosmology the study of the origin, properties, processes, and evolution
The slides with white background you need to know. The slides with blue background just have some cool information. The Big Bang cosmology the study of the origin, properties, processes, and evolution
Lecture Outlines Chapter 32. Physics, 3 rd Edition James S. Walker
 Lecture Outlines Chapter 32 Physics, 3 rd Edition James S. Walker 2007 Pearson Prentice Hall This work is protected by United States copyright laws and is provided solely for the use of instructors in
Lecture Outlines Chapter 32 Physics, 3 rd Edition James S. Walker 2007 Pearson Prentice Hall This work is protected by United States copyright laws and is provided solely for the use of instructors in
How to Build a Habitable Planet Summary. Chapter 1 The Setting
 How to Build a Habitable Planet Summary Chapter 1 The Setting The universe as we know it began about 15 billion years ago with an explosion that is called the big bang. There is no record of any prior
How to Build a Habitable Planet Summary Chapter 1 The Setting The universe as we know it began about 15 billion years ago with an explosion that is called the big bang. There is no record of any prior
Phys102 Lecture 29, 30, 31 Nuclear Physics and Radioactivity
 Phys10 Lecture 9, 30, 31 Nuclear Physics and Radioactivity Key Points Structure and Properties of the Nucleus Alpha, Beta and Gamma Decays References 30-1,,3,4,5,6,7. Atomic Structure Nitrogen (N) Atom
Phys10 Lecture 9, 30, 31 Nuclear Physics and Radioactivity Key Points Structure and Properties of the Nucleus Alpha, Beta and Gamma Decays References 30-1,,3,4,5,6,7. Atomic Structure Nitrogen (N) Atom
The role of supernovae in the origins of life
 Origin of Life The role of supernovae in the origins of life Ben Maybee Haybridge High School and Sixth Form, Worcestershire, UK. E-mail: maybeebw08@haybridge.worcs.sch.uk DOI: 10.4103/0974-6102.92199
Origin of Life The role of supernovae in the origins of life Ben Maybee Haybridge High School and Sixth Form, Worcestershire, UK. E-mail: maybeebw08@haybridge.worcs.sch.uk DOI: 10.4103/0974-6102.92199
Cosmology. Thermal history of the universe Primordial nucleosynthesis WIMPs as dark matter Recombination Horizon problem Flatness problem Inflation
 Cosmology Thermal history of the universe Primordial nucleosynthesis WIMPs as dark matter Recombination Horizon problem Flatness problem Inflation Energy density versus scale factor z=1/a-1 Early times,
Cosmology Thermal history of the universe Primordial nucleosynthesis WIMPs as dark matter Recombination Horizon problem Flatness problem Inflation Energy density versus scale factor z=1/a-1 Early times,
The Big Bang Theory was first proposed in the late 1920 s. This singularity was incredibly dense and hot.
 The Big Bang Theory was first proposed in the late 1920 s. It states that there was an infinitely small, infinitely dense point that contained everything that is the universe. This singularity was incredibly
The Big Bang Theory was first proposed in the late 1920 s. It states that there was an infinitely small, infinitely dense point that contained everything that is the universe. This singularity was incredibly
Particles involved proton neutron electron positron gamma ray 1
 TOPIC : Nuclear and radiation chemistry Nuclide - an atom with a particular mass number and atomic number Isotopes - nuclides with the same atomic number (Z) but different mass numbers (A) Notation A Element
TOPIC : Nuclear and radiation chemistry Nuclide - an atom with a particular mass number and atomic number Isotopes - nuclides with the same atomic number (Z) but different mass numbers (A) Notation A Element
Chapter 12: Nuclear Reaction
 Chapter 12: Nuclear Reaction A nuclear reaction occurs when a nucleus is unstable or is being bombarded by a nuclear particle. The product of a nuclear reaction is a new nuclide with an emission of a nuclear
Chapter 12: Nuclear Reaction A nuclear reaction occurs when a nucleus is unstable or is being bombarded by a nuclear particle. The product of a nuclear reaction is a new nuclide with an emission of a nuclear
Questions on Universe
 Questions on Universe 1. The Doppler shift may be used in the study of distant galaxies. Explain what is meant by a Doppler shift and how it is used to deduce the motion of distant galaxies. You may be
Questions on Universe 1. The Doppler shift may be used in the study of distant galaxies. Explain what is meant by a Doppler shift and how it is used to deduce the motion of distant galaxies. You may be
Stellar Structure. Observationally, we can determine: Can we explain all these observations?
 Stellar Structure Observationally, we can determine: Flux Mass Distance Luminosity Temperature Radius Spectral Type Composition Can we explain all these observations? Stellar Structure Plan: Use our general
Stellar Structure Observationally, we can determine: Flux Mass Distance Luminosity Temperature Radius Spectral Type Composition Can we explain all these observations? Stellar Structure Plan: Use our general
MAJOR NUCLEAR BURNING STAGES
 MAJOR NUCLEAR BURNING STAGES The Coulomb barrier is higher for heavier nuclei with high charge: The first reactions to occur are those involving light nuclei -- Starting from hydrogen burning, helium burning
MAJOR NUCLEAR BURNING STAGES The Coulomb barrier is higher for heavier nuclei with high charge: The first reactions to occur are those involving light nuclei -- Starting from hydrogen burning, helium burning
Radioactivity. Radioactivity
 The Law of Radioactive Decay. 72 The law of radioactive decay. It turns out that the probability per unit time for any radioactive nucleus to decay is a constant, called the decay constant, lambda, ".
The Law of Radioactive Decay. 72 The law of radioactive decay. It turns out that the probability per unit time for any radioactive nucleus to decay is a constant, called the decay constant, lambda, ".
What do the nuclei of different molybdenum isotopes have in common?
 Q1.(a) There are many isotopes of the element molybdenum (Mo). What do the nuclei of different molybdenum isotopes have in common? The isotope molybdenum-99 is produced inside some nuclear power stations
Q1.(a) There are many isotopes of the element molybdenum (Mo). What do the nuclei of different molybdenum isotopes have in common? The isotope molybdenum-99 is produced inside some nuclear power stations
Nuclear Physics. Radioactivity. # protons = # neutrons. Strong Nuclear Force. Checkpoint 4/17/2013. A Z Nucleus = Protons+ Neutrons
 Marie Curie 1867-1934 Radioactivity Spontaneous emission of radiation from the nucleus of an unstable isotope. Antoine Henri Becquerel 1852-1908 Wilhelm Roentgen 1845-1923 Nuclear Physics A Z Nucleus =
Marie Curie 1867-1934 Radioactivity Spontaneous emission of radiation from the nucleus of an unstable isotope. Antoine Henri Becquerel 1852-1908 Wilhelm Roentgen 1845-1923 Nuclear Physics A Z Nucleus =
Lecture 2: The First Second origin of neutrons and protons
 Lecture 2: The First Second origin of neutrons and protons Hot Big Bang Expanding and cooling Soup of free particles + anti-particles Symmetry breaking Soup of free quarks Quarks confined into neutrons
Lecture 2: The First Second origin of neutrons and protons Hot Big Bang Expanding and cooling Soup of free particles + anti-particles Symmetry breaking Soup of free quarks Quarks confined into neutrons
Name Final Exam December 14, 2016
 Name Final Exam December 14, 016 This test consists of five parts. Please note that in parts II through V, you can skip one question of those offered. Part I: Multiple Choice (mixed new and review questions)
Name Final Exam December 14, 016 This test consists of five parts. Please note that in parts II through V, you can skip one question of those offered. Part I: Multiple Choice (mixed new and review questions)
[1] (c) Some fruits, such as bananas, are naturally radioactive because they contain the unstable isotope of potassium-40 ( K.
![[1] (c) Some fruits, such as bananas, are naturally radioactive because they contain the unstable isotope of potassium-40 ( K. [1] (c) Some fruits, such as bananas, are naturally radioactive because they contain the unstable isotope of potassium-40 ( K.](/thumbs/82/85827188.jpg) (a) State, with a reason, whether or not protons and neutrons are fundamental particles....... [] (b) State two fundamental particles that can be classified as leptons.... [] (c) Some fruits, such as bananas,
(a) State, with a reason, whether or not protons and neutrons are fundamental particles....... [] (b) State two fundamental particles that can be classified as leptons.... [] (c) Some fruits, such as bananas,
13 Synthesis of heavier elements. introduc)on to Astrophysics, C. Bertulani, Texas A&M-Commerce 1
 13 Synthesis of heavier elements introduc)on to Astrophysics, C. Bertulani, Texas A&M-Commerce 1 The triple α Reaction When hydrogen fusion ends, the core of a star collapses and the temperature can reach
13 Synthesis of heavier elements introduc)on to Astrophysics, C. Bertulani, Texas A&M-Commerce 1 The triple α Reaction When hydrogen fusion ends, the core of a star collapses and the temperature can reach
Conceptos generales de astrofísica
 Tema 14 Conceptos generales de astrofísica Asignatura de Física Nuclear Curso académico 2009/2010 Universidad de Santiago de Compostela 1 1. Nuclear Astrophysic s domain Nuclear Astrophysics is a relatively
Tema 14 Conceptos generales de astrofísica Asignatura de Física Nuclear Curso académico 2009/2010 Universidad de Santiago de Compostela 1 1. Nuclear Astrophysic s domain Nuclear Astrophysics is a relatively
u d Fig. 6.1 (i) Identify the anti-proton from the table of particles shown in Fig [1]
![u d Fig. 6.1 (i) Identify the anti-proton from the table of particles shown in Fig [1] u d Fig. 6.1 (i) Identify the anti-proton from the table of particles shown in Fig [1]](/thumbs/79/80122691.jpg) 1 (a) Fig. 6.1 shows the quark composition of some particles. proton neutron A B u u d u d d u d u u u u d Fig. 6.1 (i) Identify the anti-proton from the table of particles shown in Fig. 6.1. (ii) State
1 (a) Fig. 6.1 shows the quark composition of some particles. proton neutron A B u u d u d d u d u u u u d Fig. 6.1 (i) Identify the anti-proton from the table of particles shown in Fig. 6.1. (ii) State
Guiding Questions. The Deaths of Stars. Pathways of Stellar Evolution GOOD TO KNOW. Low-mass stars go through two distinct red-giant stages
 The Deaths of Stars 1 Guiding Questions 1. What kinds of nuclear reactions occur within a star like the Sun as it ages? 2. Where did the carbon atoms in our bodies come from? 3. What is a planetary nebula,
The Deaths of Stars 1 Guiding Questions 1. What kinds of nuclear reactions occur within a star like the Sun as it ages? 2. Where did the carbon atoms in our bodies come from? 3. What is a planetary nebula,
The Deaths of Stars 1
 The Deaths of Stars 1 Guiding Questions 1. What kinds of nuclear reactions occur within a star like the Sun as it ages? 2. Where did the carbon atoms in our bodies come from? 3. What is a planetary nebula,
The Deaths of Stars 1 Guiding Questions 1. What kinds of nuclear reactions occur within a star like the Sun as it ages? 2. Where did the carbon atoms in our bodies come from? 3. What is a planetary nebula,
Lecture 3: Big Bang Nucleosynthesis
 Lecture 3: Big Bang Nucleosynthesis Last time: particle anti-particle soup --> quark soup --> neutron-proton soup. Today: Form 2 D and 4 He Form heavier nuclei? Discuss primordial abundances X p, Y p,
Lecture 3: Big Bang Nucleosynthesis Last time: particle anti-particle soup --> quark soup --> neutron-proton soup. Today: Form 2 D and 4 He Form heavier nuclei? Discuss primordial abundances X p, Y p,
Binding Energy. Bởi: OpenStaxCollege
 Binding Energy Bởi: OpenStaxCollege The more tightly bound a system is, the stronger the forces that hold it together and the greater the energy required to pull it apart. We can therefore learn about
Binding Energy Bởi: OpenStaxCollege The more tightly bound a system is, the stronger the forces that hold it together and the greater the energy required to pull it apart. We can therefore learn about
Lecture 33 Chapter 22, Sections 1-2 Nuclear Stability and Decay. Energy Barriers Types of Decay Nuclear Decay Kinetics
 Lecture 33 Chapter 22, Sections -2 Nuclear Stability and Decay Energy Barriers Types of Decay Nuclear Decay Kinetics Nuclear Chemistry Nuclei Review Nucleons: protons and neutrons Atomic number number
Lecture 33 Chapter 22, Sections -2 Nuclear Stability and Decay Energy Barriers Types of Decay Nuclear Decay Kinetics Nuclear Chemistry Nuclei Review Nucleons: protons and neutrons Atomic number number
Stellar Evolution: The Deaths of Stars. Guiding Questions. Pathways of Stellar Evolution. Chapter Twenty-Two
 Stellar Evolution: The Deaths of Stars Chapter Twenty-Two Guiding Questions 1. What kinds of nuclear reactions occur within a star like the Sun as it ages? 2. Where did the carbon atoms in our bodies come
Stellar Evolution: The Deaths of Stars Chapter Twenty-Two Guiding Questions 1. What kinds of nuclear reactions occur within a star like the Sun as it ages? 2. Where did the carbon atoms in our bodies come
T7-1 [255 marks] The graph shows the relationship between binding energy per nucleon and nucleon number. In which region are nuclei most stable?
![T7-1 [255 marks] The graph shows the relationship between binding energy per nucleon and nucleon number. In which region are nuclei most stable? T7-1 [255 marks] The graph shows the relationship between binding energy per nucleon and nucleon number. In which region are nuclei most stable?](/thumbs/77/75398334.jpg) T7-1 [255 marks] 1. In the Geiger Marsden experiment alpha particles were directed at a thin gold foil. Which of the following shows how the majority of the alpha particles behaved after reaching the foil?
T7-1 [255 marks] 1. In the Geiger Marsden experiment alpha particles were directed at a thin gold foil. Which of the following shows how the majority of the alpha particles behaved after reaching the foil?
HNRS 227 Lecture 18 October 2007 Chapter 12. Stars, Galaxies and the Universe presented by Dr. Geller
 HNRS 227 Lecture 18 October 2007 Chapter 12 Stars, Galaxies and the Universe presented by Dr. Geller Recall from Chapters 1-11 Units of length, mass, density, time, and metric system The Scientific Method
HNRS 227 Lecture 18 October 2007 Chapter 12 Stars, Galaxies and the Universe presented by Dr. Geller Recall from Chapters 1-11 Units of length, mass, density, time, and metric system The Scientific Method
Guiding Questions. The Deaths of Stars. Pathways of Stellar Evolution GOOD TO KNOW. Low-mass stars go through two distinct red-giant stages
 The Deaths of Stars Guiding Questions 1. What kinds of nuclear reactions occur within a star like the Sun as it ages? 2. Where did the carbon atoms in our bodies come from? 3. What is a planetary nebula,
The Deaths of Stars Guiding Questions 1. What kinds of nuclear reactions occur within a star like the Sun as it ages? 2. Where did the carbon atoms in our bodies come from? 3. What is a planetary nebula,
Complete the table by ticking one box in each row to identify the appropriate isotope. The first row has been completed for you.
 Q1.The table below contains five statements that refer to isotopes and some radium isotopes. Ra Ra Ra Ra Isotope with the smallest mass number Isotope with most neutrons in nucleus Isotope with nucleus
Q1.The table below contains five statements that refer to isotopes and some radium isotopes. Ra Ra Ra Ra Isotope with the smallest mass number Isotope with most neutrons in nucleus Isotope with nucleus
LIFE CYCLE OF A STAR
 LIFE CYCLE OF A STAR First stage = Protostar PROTOSTAR Cloud of gas and dust many light-years across Gravity tries to pull the materials together Eventually, at the center of the ball of dust and gas,
LIFE CYCLE OF A STAR First stage = Protostar PROTOSTAR Cloud of gas and dust many light-years across Gravity tries to pull the materials together Eventually, at the center of the ball of dust and gas,
1. This question is about the Rutherford model of the atom.
 1. This question is about the Rutherford model of the atom. (a) Most alpha particles used to bombard a thin gold foil pass through the foil without a significant change in direction. A few alpha particles
1. This question is about the Rutherford model of the atom. (a) Most alpha particles used to bombard a thin gold foil pass through the foil without a significant change in direction. A few alpha particles
Lecture 3: Big Bang Nucleosynthesis The First Three Minutes Last time:
 Lecture 3: Big Bang Nucleosynthesis The First Three Minutes Last time: particle anti-particle soup --> quark soup --> neutron-proton soup p / n ratio at onset of 2 D formation Today: Form 2 D and 4 He
Lecture 3: Big Bang Nucleosynthesis The First Three Minutes Last time: particle anti-particle soup --> quark soup --> neutron-proton soup p / n ratio at onset of 2 D formation Today: Form 2 D and 4 He
