The general solution of Schrödinger equation in three dimensions (if V does not depend on time) are solutions of time-independent Schrödinger equation
|
|
|
- Leslie Owen
- 5 years ago
- Views:
Transcription
1 Lecture 27st Page 1 Lecture 27 L27.P1 Review Schrödinger equation The general solution of Schrödinger equation in three dimensions (if V does not depend on time) is where functions are solutions of time-independent Schrödinger equation If potential V is spherically symmetric, i.e. only depends on distance to the origin r, then the separable solutions are spherical harmonics where and are solutions of radial equation Hydrogen-like atom energy levels:
2 Lecture 27st Page 2 L27.P2 Identical particles Bosons and fermions In classical mechanics, you can always identify which particle is which. In quantum mechanics, you simply can't say which electron is which as you can not put any labels on them to tell them apart. There are two possible ways to deal with indistinguishable particles, i.e. to construct two-particle wave function from single particle wave functions and that is non committal to which particle is in which state: Symmetric Antisymmetric Therefore, quantum mechanics allows for two kinds of identical particles: bosons (for the "+" sign) and fermions (for the "-" sign). N-particle states are constructed in the same way, antisymmteric state for fermions (which can be easily written as Slater determinant) and symmetric state for bosons; the normalization factor is. In our non-relativistic quantum mechanics we accept the following statement as an axiom: All particles with integer spin are bosons, all particles with half-integer spin are fermions. Note that it is total wave function that has to be antisymmetric. Therefore, for example if spatial wave function for the electrons is symmetric, then the corresponding spin state has to be antisymmteric. Note: make sure that you can add angular momenta and know what are singlet and triplet states. From the above, two identical fermions can not occupy the same state. It is called Pauli exclusion principle.
3 Lecture 27st Page 3 Perturbation theory L27.P3 General formalism of the problem: Suppose that we solved the time-independent Schrödinger equation for some potential and obtained a complete set of orthonormal eigenfunctions and corresponding eigenvalues. This is the problem that we completely understand and know solutions for. We mark all these solutions and the Hamiltonian with " " label. Now we slightly perturb the potential: The problem of the perturbation theory is to find eigenvalues and eigenfunctions of the perturbed potential, i.e. to solve approximately the following equation: using the known solutions of the problem Nondegenerate perturbation theory We expand our solution as follows in terms of perturbation H' The first-order correction to the energy is given by: First-order correction to the wave function is given by Note that as long as m n, the denominator can not be zero as long as energy levels are nondegenerate. If the energy levels are degenerate, we need degenerate perturbation theory ( consider later).
4 Lecture 27st Page 4 The second-order correction to the energy is L27.P4 Degenerate perturbation theory Suppose now that the states energy. are degenerate, i.e. have the same How to calculate first-order energy correction E 1? In the case of n-fold degeneracy, E 1 are eigenvalues of n x n matrix Variational method The variational principle let you get an upper bound for the ground state energy when you can not directly solve the Schrödinger's equation. How does it work? (1) Pick any normalized function. (2) The ground state energy Egs is 3) Some choices of the trial function will get your Egs that is close to actual value. If you picked a function with a parameter, minimize the resulting expression for. Substitute resulting value of the parameter into to get lowest upper bound on Egs.
5 Lecture 27st Page 5 L27.P5 WKB approximation This method allows to obtain approximate solutions to the time-independent Schrödinger equation in one dimension and is particularly useful in calculating tunneling rates through potential barriers and bound state energies. "Classical" region, E > V General solution is the combination of these two. If E < V (non-classical region), then p is imaginary but we can still write Transmission probability: over non-classical region Formulas above are derived with the assumption that the potential is slowly varying in comparison with the wavelength λ (or 1/κ for E<V). This is not the case for the turning points, where "classical" region connects with "non classical" region. In this case WKB approximation breaks down. Note: make sure that you know how to apply connection formulas to derive quantization conditions 0 for potential with two vertical walls at x1 and x2 for potential with one vertical wall at x1 for potential with no vertical walls. turning points
6 Lecture 27st Page 6 Scattering L27.P6 Differential (scattering) cross-section: Total cross section is defined as the integral of D( ) over all solid angles: Quantum scattering theory Our problem: incident plane wave traveling in Z direction encounters a scattering potential that produces outgoing spherical wave: Particle energy Therefore, the solutions of the Schrödinger equation have the general form: scattering amplitude
7 Lecture 27st Page 7 Partial wave analysis L27.P7 For spherically symmetric potential, the scattering amplitude may be calculated using partial wave analysis formula The partial wave amplitudes are found by solving the Schrödinger equation for the area where V 0 and using the boundary conditions. Substituting the expression for the partial wave scattering amplitude (1) into the formula for the differential cross section and integrating over all solid angles yields simple expression for the total scattering cross-section: Scattering amplitude in Born approximation. As before, the differential and total cross sections are given by (1) Low-energy scattering (2) Spherically symmetric potential.
8 Lecture 27st Page 8 L27.P8 Notes on the exam preparation & exam taking: 1. Make sure that you know, understand and can use all formulas and concepts from this lecture. 2. Make sure that you can solve on your own and without looking into any notes any problem done in class in Lectures or from homeworks (if integrals are complicated, use Maple, Matematica, etc.) 3. During exam, look through all the problems first. Start with the one you know best and the one that is shortest to write a solution for. 4. Make sure that you read the problem very carefully and understand what is being asked. If you are unsure, ask me. 5. To save time, make sure you are not repeating the same calculations. For example, if you need to do several similar integrals, make sure that you are not redoing the ones you have already done. If you are out of time and you have not finished, write an outline of what you would do to finish the problem if you had time.
9 Lecture 27st Page 9 P1 Practice final Problem 1 Three imaginary "spinless" fermions are confined into a one-dimensional box of length L. The confinement potential is V = We assume that there is no interaction between the fermions. What is the ground state? Find its energy and write the corresponding wave function(s). Is it degenerate?
10 Lecture 27st Page 10 Problem 2 Consider a quantum system with just three linearly independent states. Suppose the Hamiltonian, in matrix form is where c is a constant, c «1. Calculate first-order and second-order corrections to the energies.
11 Lecture 27st Page 11 P2
12 Lecture 27st Page 12 Problem 3 A particle of mass m is in a one-dimensional potential given by for x 0, where K is a positive constant, and with infinite potential barrier at x=0. Using the trial wave function make the best estimate of the ground state energy.
13 Lecture 27st Page 13 Problem 4 Use WKB approximation to find the energy levels of "half-harmonic oscillator": otherwise Solution
14 Lecture 27st Page 14 Problem 5. Use low-energy scattering in Born approximation to calculate differential and total cross sections for low-energy soft-sphere scattering,
15 Lecture 27st Page 15 Possibly useful formulas Solutions for one-dimensional infinite square well of width a:
16 Lecture 27st Page 16 Connection formulas when potential slopes upward: If the potential slopes downward, the connection formulas are:
The general solution of Schrödinger equation in three dimensions (if V does not depend on time) are solutions of time-independent Schrödinger equation
 Lecture 17 Page 1 Lecture 17 L17.P1 Review Schrödinger equation The general solution of Schrödinger equation in three dimensions (if V does not depend on time) is where functions are solutions of time-independent
Lecture 17 Page 1 Lecture 17 L17.P1 Review Schrödinger equation The general solution of Schrödinger equation in three dimensions (if V does not depend on time) is where functions are solutions of time-independent
Lecture #1. Review. Postulates of quantum mechanics (1-3) Postulate 1
 L1.P1 Lecture #1 Review Postulates of quantum mechanics (1-3) Postulate 1 The state of a system at any instant of time may be represented by a wave function which is continuous and differentiable. Specifically,
L1.P1 Lecture #1 Review Postulates of quantum mechanics (1-3) Postulate 1 The state of a system at any instant of time may be represented by a wave function which is continuous and differentiable. Specifically,
Potential energy, from Coulomb's law. Potential is spherically symmetric. Therefore, solutions must have form
 Lecture 6 Page 1 Atoms L6.P1 Review of hydrogen atom Heavy proton (put at the origin), charge e and much lighter electron, charge -e. Potential energy, from Coulomb's law Potential is spherically symmetric.
Lecture 6 Page 1 Atoms L6.P1 Review of hydrogen atom Heavy proton (put at the origin), charge e and much lighter electron, charge -e. Potential energy, from Coulomb's law Potential is spherically symmetric.
Quantum Physics III (8.06) Spring 2007 FINAL EXAMINATION Monday May 21, 9:00 am You have 3 hours.
 Quantum Physics III (8.06) Spring 2007 FINAL EXAMINATION Monday May 21, 9:00 am You have 3 hours. There are 10 problems, totalling 180 points. Do all problems. Answer all problems in the white books provided.
Quantum Physics III (8.06) Spring 2007 FINAL EXAMINATION Monday May 21, 9:00 am You have 3 hours. There are 10 problems, totalling 180 points. Do all problems. Answer all problems in the white books provided.
Chapter 4 (Lecture 6-7) Schrodinger equation for some simple systems Table: List of various one dimensional potentials System Physical correspondence
 V, E, Chapter (Lecture 6-7) Schrodinger equation for some simple systems Table: List of various one dimensional potentials System Physical correspondence Potential Total Energies and Probability density
V, E, Chapter (Lecture 6-7) Schrodinger equation for some simple systems Table: List of various one dimensional potentials System Physical correspondence Potential Total Energies and Probability density
Quantum Physics II (8.05) Fall 2002 Outline
 Quantum Physics II (8.05) Fall 2002 Outline 1. General structure of quantum mechanics. 8.04 was based primarily on wave mechanics. We review that foundation with the intent to build a more formal basis
Quantum Physics II (8.05) Fall 2002 Outline 1. General structure of quantum mechanics. 8.04 was based primarily on wave mechanics. We review that foundation with the intent to build a more formal basis
Degeneracy & in particular to Hydrogen atom
 Degeneracy & in particular to Hydrogen atom In quantum mechanics, an energy level is said to be degenerate if it corresponds to two or more different measurable states of a quantum system. Conversely,
Degeneracy & in particular to Hydrogen atom In quantum mechanics, an energy level is said to be degenerate if it corresponds to two or more different measurable states of a quantum system. Conversely,
Chemistry 3502/4502. Final Exam Part I. May 14, 2005
 Advocacy chit Chemistry 350/450 Final Exam Part I May 4, 005. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle
Advocacy chit Chemistry 350/450 Final Exam Part I May 4, 005. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle
Quantum Physics II (8.05) Fall 2002 Assignment 12 and Study Aid
 Quantum Physics II (8.05) Fall 2002 Assignment 12 and Study Aid Announcement This handout includes 9 problems. The first 5 are the problem set due. The last 4 cover material from the final few lectures
Quantum Physics II (8.05) Fall 2002 Assignment 12 and Study Aid Announcement This handout includes 9 problems. The first 5 are the problem set due. The last 4 cover material from the final few lectures
Introduction to Quantum Mechanics PVK - Solutions. Nicolas Lanzetti
 Introduction to Quantum Mechanics PVK - Solutions Nicolas Lanzetti lnicolas@student.ethz.ch 1 Contents 1 The Wave Function and the Schrödinger Equation 3 1.1 Quick Checks......................................
Introduction to Quantum Mechanics PVK - Solutions Nicolas Lanzetti lnicolas@student.ethz.ch 1 Contents 1 The Wave Function and the Schrödinger Equation 3 1.1 Quick Checks......................................
CHM Physical Chemistry II Chapter 9 - Supplementary Material. 1. Constuction of orbitals from the spherical harmonics
 CHM 3411 - Physical Chemistry II Chapter 9 - Supplementary Material 1. Constuction of orbitals from the spherical harmonics The wavefunctions that are solutions to the time independent Schrodinger equation
CHM 3411 - Physical Chemistry II Chapter 9 - Supplementary Material 1. Constuction of orbitals from the spherical harmonics The wavefunctions that are solutions to the time independent Schrodinger equation
Quantum Mechanics: Fundamentals
 Kurt Gottfried Tung-Mow Yan Quantum Mechanics: Fundamentals Second Edition With 75 Figures Springer Preface vii Fundamental Concepts 1 1.1 Complementarity and Uncertainty 1 (a) Complementarity 2 (b) The
Kurt Gottfried Tung-Mow Yan Quantum Mechanics: Fundamentals Second Edition With 75 Figures Springer Preface vii Fundamental Concepts 1 1.1 Complementarity and Uncertainty 1 (a) Complementarity 2 (b) The
Lecture 5. Hartree-Fock Theory. WS2010/11: Introduction to Nuclear and Particle Physics
 Lecture 5 Hartree-Fock Theory WS2010/11: Introduction to Nuclear and Particle Physics Particle-number representation: General formalism The simplest starting point for a many-body state is a system of
Lecture 5 Hartree-Fock Theory WS2010/11: Introduction to Nuclear and Particle Physics Particle-number representation: General formalism The simplest starting point for a many-body state is a system of
Brief review of Quantum Mechanics (QM)
 Brief review of Quantum Mechanics (QM) Note: This is a collection of several formulae and facts that we will use throughout the course. It is by no means a complete discussion of QM, nor will I attempt
Brief review of Quantum Mechanics (QM) Note: This is a collection of several formulae and facts that we will use throughout the course. It is by no means a complete discussion of QM, nor will I attempt
A. F. J. Levi 1 EE539: Engineering Quantum Mechanics. Fall 2017.
 A. F. J. Levi 1 Engineering Quantum Mechanics. Fall 2017. TTh 9.00 a.m. 10.50 a.m., VHE 210. Web site: http://alevi.usc.edu Web site: http://classes.usc.edu/term-20173/classes/ee EE539: Abstract and Prerequisites
A. F. J. Levi 1 Engineering Quantum Mechanics. Fall 2017. TTh 9.00 a.m. 10.50 a.m., VHE 210. Web site: http://alevi.usc.edu Web site: http://classes.usc.edu/term-20173/classes/ee EE539: Abstract and Prerequisites
Time part of the equation can be separated by substituting independent equation
 Lecture 9 Schrödinger Equation in 3D and Angular Momentum Operator In this section we will construct 3D Schrödinger equation and we give some simple examples. In this course we will consider problems where
Lecture 9 Schrödinger Equation in 3D and Angular Momentum Operator In this section we will construct 3D Schrödinger equation and we give some simple examples. In this course we will consider problems where
QUANTUM MECHANICS. Franz Schwabl. Translated by Ronald Kates. ff Springer
 Franz Schwabl QUANTUM MECHANICS Translated by Ronald Kates Second Revised Edition With 122Figures, 16Tables, Numerous Worked Examples, and 126 Problems ff Springer Contents 1. Historical and Experimental
Franz Schwabl QUANTUM MECHANICS Translated by Ronald Kates Second Revised Edition With 122Figures, 16Tables, Numerous Worked Examples, and 126 Problems ff Springer Contents 1. Historical and Experimental
Chem 442 Review for Exam 2. Exact separation of the Hamiltonian of a hydrogenic atom into center-of-mass (3D) and relative (3D) components.
 Chem 44 Review for Exam Hydrogenic atoms: The Coulomb energy between two point charges Ze and e: V r Ze r Exact separation of the Hamiltonian of a hydrogenic atom into center-of-mass (3D) and relative
Chem 44 Review for Exam Hydrogenic atoms: The Coulomb energy between two point charges Ze and e: V r Ze r Exact separation of the Hamiltonian of a hydrogenic atom into center-of-mass (3D) and relative
Final Exam. Tuesday, May 8, Starting at 8:30 a.m., Hoyt Hall.
 Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. Summary of Chapter 38 In Quantum Mechanics particles are represented by wave functions Ψ. The absolute square of the wave function Ψ 2
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. Summary of Chapter 38 In Quantum Mechanics particles are represented by wave functions Ψ. The absolute square of the wave function Ψ 2
LECTURES ON QUANTUM MECHANICS
 LECTURES ON QUANTUM MECHANICS GORDON BAYM Unitsersity of Illinois A II I' Advanced Bock Progrant A Member of the Perseus Books Group CONTENTS Preface v Chapter 1 Photon Polarization 1 Transformation of
LECTURES ON QUANTUM MECHANICS GORDON BAYM Unitsersity of Illinois A II I' Advanced Bock Progrant A Member of the Perseus Books Group CONTENTS Preface v Chapter 1 Photon Polarization 1 Transformation of
Chemistry 3502/4502. Exam III. All Hallows Eve/Samhain, ) This is a multiple choice exam. Circle the correct answer.
 B Chemistry 3502/4502 Exam III All Hallows Eve/Samhain, 2003 1) This is a multiple choice exam. Circle the correct answer. 2) There is one correct answer to every problem. There is no partial credit. 3)
B Chemistry 3502/4502 Exam III All Hallows Eve/Samhain, 2003 1) This is a multiple choice exam. Circle the correct answer. 2) There is one correct answer to every problem. There is no partial credit. 3)
Atomic Structure and Atomic Spectra
 Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
Basic Physical Chemistry Lecture 2. Keisuke Goda Summer Semester 2015
 Basic Physical Chemistry Lecture 2 Keisuke Goda Summer Semester 2015 Lecture schedule Since we only have three lectures, let s focus on a few important topics of quantum chemistry and structural chemistry
Basic Physical Chemistry Lecture 2 Keisuke Goda Summer Semester 2015 Lecture schedule Since we only have three lectures, let s focus on a few important topics of quantum chemistry and structural chemistry
For example, in one dimension if we had two particles in a one-dimensional infinite potential well described by the following two wave functions.
 Identical particles In classical physics one can label particles in such a way as to leave the dynamics unaltered or follow the trajectory of the particles say by making a movie with a fast camera. Thus
Identical particles In classical physics one can label particles in such a way as to leave the dynamics unaltered or follow the trajectory of the particles say by making a movie with a fast camera. Thus
QUANTUM MECHANICS SECOND EDITION G. ARULDHAS
 QUANTUM MECHANICS SECOND EDITION G. ARULDHAS Formerly, Professor and Head of Physics and Dean, Faculty of Science University of Kerala New Delhi-110001 2009 QUANTUM MECHANICS, 2nd Ed. G. Aruldhas 2009
QUANTUM MECHANICS SECOND EDITION G. ARULDHAS Formerly, Professor and Head of Physics and Dean, Faculty of Science University of Kerala New Delhi-110001 2009 QUANTUM MECHANICS, 2nd Ed. G. Aruldhas 2009
Chemistry 3502/4502. Final Exam Part I. May 14, 2005
 Chemistry 3502/4502 Final Exam Part I May 14, 2005 1. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle (e) The
Chemistry 3502/4502 Final Exam Part I May 14, 2005 1. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle (e) The
Intermission: Let s review the essentials of the Helium Atom
 PHYS3022 Applied Quantum Mechanics Problem Set 4 Due Date: 6 March 2018 (Tuesday) T+2 = 8 March 2018 All problem sets should be handed in not later than 5pm on the due date. Drop your assignments in the
PHYS3022 Applied Quantum Mechanics Problem Set 4 Due Date: 6 March 2018 (Tuesday) T+2 = 8 March 2018 All problem sets should be handed in not later than 5pm on the due date. Drop your assignments in the
PHYSICS 721/821 - Spring Semester ODU. Graduate Quantum Mechanics II Midterm Exam - Solution
 PHYSICS 72/82 - Spring Semester 2 - ODU Graduate Quantum Mechanics II Midterm Exam - Solution Problem ) An electron (mass 5, ev/c 2 ) is in a one-dimensional potential well as sketched to the right (the
PHYSICS 72/82 - Spring Semester 2 - ODU Graduate Quantum Mechanics II Midterm Exam - Solution Problem ) An electron (mass 5, ev/c 2 ) is in a one-dimensional potential well as sketched to the right (the
CHAPTER 6 Quantum Mechanics II
 CHAPTER 6 Quantum Mechanics II 6.1 6.2 6.3 6.4 6.5 6.6 6.7 The Schrödinger Wave Equation Expectation Values Infinite Square-Well Potential Finite Square-Well Potential Three-Dimensional Infinite-Potential
CHAPTER 6 Quantum Mechanics II 6.1 6.2 6.3 6.4 6.5 6.6 6.7 The Schrödinger Wave Equation Expectation Values Infinite Square-Well Potential Finite Square-Well Potential Three-Dimensional Infinite-Potential
Spring /2/ pts 1 point per minute
 Physics 519 MIDTERM Name: Spring 014 6//14 80 pts 1 point per minute Exam procedures. Please write your name above. Please sit away from other students. If you have a question about the exam, please ask.
Physics 519 MIDTERM Name: Spring 014 6//14 80 pts 1 point per minute Exam procedures. Please write your name above. Please sit away from other students. If you have a question about the exam, please ask.
Next topic: Quantum Field Theories for Quantum Many-Particle Systems; or "Second Quantization"
 Next topic: Quantum Field Theories for Quantum Many-Particle Systems; or "Second Quantization" Outline 1 Bosons and Fermions 2 N-particle wave functions ("first quantization" 3 The method of quantized
Next topic: Quantum Field Theories for Quantum Many-Particle Systems; or "Second Quantization" Outline 1 Bosons and Fermions 2 N-particle wave functions ("first quantization" 3 The method of quantized
Solution Set of Homework # 6 Monday, December 12, Textbook: Claude Cohen Tannoudji, Bernard Diu and Franck Laloë, Second Volume
 Department of Physics Quantum II, 570 Temple University Instructor: Z.-E. Meziani Solution Set of Homework # 6 Monday, December, 06 Textbook: Claude Cohen Tannoudji, Bernard Diu and Franck Laloë, Second
Department of Physics Quantum II, 570 Temple University Instructor: Z.-E. Meziani Solution Set of Homework # 6 Monday, December, 06 Textbook: Claude Cohen Tannoudji, Bernard Diu and Franck Laloë, Second
Quantum Mechanics Solutions. λ i λ j v j v j v i v i.
 Quantum Mechanics Solutions 1. (a) If H has an orthonormal basis consisting of the eigenvectors { v i } of A with eigenvalues λ i C, then A can be written in terms of its spectral decomposition as A =
Quantum Mechanics Solutions 1. (a) If H has an orthonormal basis consisting of the eigenvectors { v i } of A with eigenvalues λ i C, then A can be written in terms of its spectral decomposition as A =
Lecture 14 The Free Electron Gas: Density of States
 Lecture 4 The Free Electron Gas: Density of States Today:. Spin.. Fermionic nature of electrons. 3. Understanding the properties of metals: the free electron model and the role of Pauli s exclusion principle.
Lecture 4 The Free Electron Gas: Density of States Today:. Spin.. Fermionic nature of electrons. 3. Understanding the properties of metals: the free electron model and the role of Pauli s exclusion principle.
Problem 1: Step Potential (10 points)
 Problem 1: Step Potential (10 points) 1 Consider the potential V (x). V (x) = { 0, x 0 V, x > 0 A particle of mass m and kinetic energy E approaches the step from x < 0. a) Write the solution to Schrodinger
Problem 1: Step Potential (10 points) 1 Consider the potential V (x). V (x) = { 0, x 0 V, x > 0 A particle of mass m and kinetic energy E approaches the step from x < 0. a) Write the solution to Schrodinger
Atkins & de Paula: Atkins Physical Chemistry 9e Checklist of key ideas. Chapter 8: Quantum Theory: Techniques and Applications
 Atkins & de Paula: Atkins Physical Chemistry 9e Checklist of key ideas Chapter 8: Quantum Theory: Techniques and Applications TRANSLATIONAL MOTION wavefunction of free particle, ψ k = Ae ikx + Be ikx,
Atkins & de Paula: Atkins Physical Chemistry 9e Checklist of key ideas Chapter 8: Quantum Theory: Techniques and Applications TRANSLATIONAL MOTION wavefunction of free particle, ψ k = Ae ikx + Be ikx,
UGC ACADEMY LEADING INSTITUE FOR CSIR-JRF/NET, GATE & JAM PH 05 PHYSICAL SCIENCE TEST SERIES # 1. Quantum, Statistical & Thermal Physics
 UGC ACADEMY LEADING INSTITUE FOR CSIR-JRF/NET, GATE & JAM BOOKLET CODE SUBJECT CODE PH 05 PHYSICAL SCIENCE TEST SERIES # Quantum, Statistical & Thermal Physics Timing: 3: H M.M: 00 Instructions. This test
UGC ACADEMY LEADING INSTITUE FOR CSIR-JRF/NET, GATE & JAM BOOKLET CODE SUBJECT CODE PH 05 PHYSICAL SCIENCE TEST SERIES # Quantum, Statistical & Thermal Physics Timing: 3: H M.M: 00 Instructions. This test
Chem 467 Supplement to Lecture 19 Hydrogen Atom, Atomic Orbitals
 Chem 467 Supplement to Lecture 19 Hydrogen Atom, Atomic Orbitals Pre-Quantum Atomic Structure The existence of atoms and molecules had long been theorized, but never rigorously proven until the late 19
Chem 467 Supplement to Lecture 19 Hydrogen Atom, Atomic Orbitals Pre-Quantum Atomic Structure The existence of atoms and molecules had long been theorized, but never rigorously proven until the late 19
ECE 487 Lecture 5 : Foundations of Quantum Mechanics IV Class Outline:
 ECE 487 Lecture 5 : Foundations of Quantum Mechanics IV Class Outline: Linearly Varying Potential Triangular Potential Well Time-Dependent Schrödinger Equation Things you should know when you leave Key
ECE 487 Lecture 5 : Foundations of Quantum Mechanics IV Class Outline: Linearly Varying Potential Triangular Potential Well Time-Dependent Schrödinger Equation Things you should know when you leave Key
Lecture 6 Scattering theory Partial Wave Analysis. SS2011: Introduction to Nuclear and Particle Physics, Part 2 2
 Lecture 6 Scattering theory Partial Wave Analysis SS2011: Introduction to Nuclear and Particle Physics, Part 2 2 1 The Born approximation for the differential cross section is valid if the interaction
Lecture 6 Scattering theory Partial Wave Analysis SS2011: Introduction to Nuclear and Particle Physics, Part 2 2 1 The Born approximation for the differential cross section is valid if the interaction
PHY4604 Introduction to Quantum Mechanics Fall 2004 Final Exam SOLUTIONS December 17, 2004, 7:30 a.m.- 9:30 a.m.
 PHY464 Introduction to Quantum Mechanics Fall 4 Final Eam SOLUTIONS December 7, 4, 7:3 a.m.- 9:3 a.m. No other materials allowed. If you can t do one part of a problem, solve subsequent parts in terms
PHY464 Introduction to Quantum Mechanics Fall 4 Final Eam SOLUTIONS December 7, 4, 7:3 a.m.- 9:3 a.m. No other materials allowed. If you can t do one part of a problem, solve subsequent parts in terms
Lecture 4 Quantum mechanics in more than one-dimension
 Lecture 4 Quantum mechanics in more than one-dimension Background Previously, we have addressed quantum mechanics of 1d systems and explored bound and unbound (scattering) states. Although general concepts
Lecture 4 Quantum mechanics in more than one-dimension Background Previously, we have addressed quantum mechanics of 1d systems and explored bound and unbound (scattering) states. Although general concepts
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer. Lecture 20, March 8, 2006
 Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer Lecture 20, March 8, 2006 Solved Homework We determined that the two coefficients in our two-gaussian
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer Lecture 20, March 8, 2006 Solved Homework We determined that the two coefficients in our two-gaussian
Probability and Normalization
 Probability and Normalization Although we don t know exactly where the particle might be inside the box, we know that it has to be in the box. This means that, ψ ( x) dx = 1 (normalization condition) L
Probability and Normalization Although we don t know exactly where the particle might be inside the box, we know that it has to be in the box. This means that, ψ ( x) dx = 1 (normalization condition) L
Chemistry 3502/4502. Exam III. March 28, ) Circle the correct answer on multiple-choice problems.
 A Chemistry 352/452 Exam III March 28, 25 1) Circle the correct answer on multiple-choice problems. 2) There is one correct answer to every multiple-choice problem. There is no partial credit. On the short-answer
A Chemistry 352/452 Exam III March 28, 25 1) Circle the correct answer on multiple-choice problems. 2) There is one correct answer to every multiple-choice problem. There is no partial credit. On the short-answer
Chem 452 Mega Practice Exam 1
 Last Name: First Name: PSU ID #: Chem 45 Mega Practice Exam 1 Cover Sheet Closed Book, Notes, and NO Calculator The exam will consist of approximately 5 similar questions worth 4 points each. This mega-exam
Last Name: First Name: PSU ID #: Chem 45 Mega Practice Exam 1 Cover Sheet Closed Book, Notes, and NO Calculator The exam will consist of approximately 5 similar questions worth 4 points each. This mega-exam
PHYSICS PH.D. COMPREHENSIVE EXAM 2006
 PHYSICS PH.D. COMPREHENSIVE EXAM 2006 (1) In construction work, a practical means of establishing a vertical reference line is the use of a plumb line a mass hanging in equilibrium from a long vertical
PHYSICS PH.D. COMPREHENSIVE EXAM 2006 (1) In construction work, a practical means of establishing a vertical reference line is the use of a plumb line a mass hanging in equilibrium from a long vertical
Lecture Notes. Quantum Theory. Prof. Maximilian Kreuzer. Institute for Theoretical Physics Vienna University of Technology. covering the contents of
 Lecture Notes Quantum Theory by Prof. Maximilian Kreuzer Institute for Theoretical Physics Vienna University of Technology covering the contents of 136.019 Quantentheorie I and 136.027 Quantentheorie II
Lecture Notes Quantum Theory by Prof. Maximilian Kreuzer Institute for Theoretical Physics Vienna University of Technology covering the contents of 136.019 Quantentheorie I and 136.027 Quantentheorie II
Identical Particles in Quantum Mechanics
 Identical Particles in Quantum Mechanics Chapter 20 P. J. Grandinetti Chem. 4300 Nov 17, 2017 P. J. Grandinetti (Chem. 4300) Identical Particles in Quantum Mechanics Nov 17, 2017 1 / 20 Wolfgang Pauli
Identical Particles in Quantum Mechanics Chapter 20 P. J. Grandinetti Chem. 4300 Nov 17, 2017 P. J. Grandinetti (Chem. 4300) Identical Particles in Quantum Mechanics Nov 17, 2017 1 / 20 Wolfgang Pauli
Energy Level Energy Level Diagrams for Diagrams for Simple Hydrogen Model
 Quantum Mechanics and Atomic Physics Lecture 20: Real Hydrogen Atom /Identical particles http://www.physics.rutgers.edu/ugrad/361 physics edu/ugrad/361 Prof. Sean Oh Last time Hydrogen atom: electron in
Quantum Mechanics and Atomic Physics Lecture 20: Real Hydrogen Atom /Identical particles http://www.physics.rutgers.edu/ugrad/361 physics edu/ugrad/361 Prof. Sean Oh Last time Hydrogen atom: electron in
Chapter 9: Multi- Electron Atoms Ground States and X- ray Excitation
 Chapter 9: Multi- Electron Atoms Ground States and X- ray Excitation Up to now we have considered one-electron atoms. Almost all atoms are multiple-electron atoms and their description is more complicated
Chapter 9: Multi- Electron Atoms Ground States and X- ray Excitation Up to now we have considered one-electron atoms. Almost all atoms are multiple-electron atoms and their description is more complicated
Lecture #13 1. Incorporating a vector potential into the Hamiltonian 2. Spin postulates 3. Description of spin states 4. Identical particles in
 Lecture #3. Incorporating a vector potential into the Hamiltonian. Spin postulates 3. Description of spin states 4. Identical particles in classical and QM 5. Exchange degeneracy - the fundamental problem
Lecture #3. Incorporating a vector potential into the Hamiltonian. Spin postulates 3. Description of spin states 4. Identical particles in classical and QM 5. Exchange degeneracy - the fundamental problem
3.024 Electrical, Optical, and Magnetic Properties of Materials Spring 2012 Recitation 3 Notes
 3.024 Electrical, Optical, and Magnetic Properties of Materials Spring 2012 Outline 1. Schr dinger: Eigenfunction Problems & Operator Properties 2. Piecewise Function/Continuity Review -Scattering from
3.024 Electrical, Optical, and Magnetic Properties of Materials Spring 2012 Outline 1. Schr dinger: Eigenfunction Problems & Operator Properties 2. Piecewise Function/Continuity Review -Scattering from
PH 451/551 Quantum Mechanics Capstone Winter 201x
 These are the questions from the W7 exam presented as practice problems. The equation sheet is PH 45/55 Quantum Mechanics Capstone Winter x TOTAL POINTS: xx Weniger 6, time There are xx questions, for
These are the questions from the W7 exam presented as practice problems. The equation sheet is PH 45/55 Quantum Mechanics Capstone Winter x TOTAL POINTS: xx Weniger 6, time There are xx questions, for
Quantum mechanics of many-fermion systems
 Quantum mechanics of many-fermion systems Kouichi Hagino Tohoku University, Sendai, Japan 1. Identical particles: Fermions and Bosons 2. Simple examples: systems with two identical particles 3. Pauli principle
Quantum mechanics of many-fermion systems Kouichi Hagino Tohoku University, Sendai, Japan 1. Identical particles: Fermions and Bosons 2. Simple examples: systems with two identical particles 3. Pauli principle
Quantum Mechanics. p " The Uncertainty Principle places fundamental limits on our measurements :
 Student Selected Module 2005/2006 (SSM-0032) 17 th November 2005 Quantum Mechanics Outline : Review of Previous Lecture. Single Particle Wavefunctions. Time-Independent Schrödinger equation. Particle in
Student Selected Module 2005/2006 (SSM-0032) 17 th November 2005 Quantum Mechanics Outline : Review of Previous Lecture. Single Particle Wavefunctions. Time-Independent Schrödinger equation. Particle in
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer. Lecture 22, March 20, 2006
 Chem 350/450 Physical Chemistry II Quantum Mechanics 3 Credits Spring Semester 006 Christopher J. Cramer Lecture, March 0, 006 Some material in this lecture has been adapted from Cramer, C. J. Essentials
Chem 350/450 Physical Chemistry II Quantum Mechanics 3 Credits Spring Semester 006 Christopher J. Cramer Lecture, March 0, 006 Some material in this lecture has been adapted from Cramer, C. J. Essentials
P. W. Atkins and R. S. Friedman. Molecular Quantum Mechanics THIRD EDITION
 P. W. Atkins and R. S. Friedman Molecular Quantum Mechanics THIRD EDITION Oxford New York Tokyo OXFORD UNIVERSITY PRESS 1997 Introduction and orientation 1 Black-body radiation 1 Heat capacities 2 The
P. W. Atkins and R. S. Friedman Molecular Quantum Mechanics THIRD EDITION Oxford New York Tokyo OXFORD UNIVERSITY PRESS 1997 Introduction and orientation 1 Black-body radiation 1 Heat capacities 2 The
Structure of diatomic molecules
 Structure of diatomic molecules January 8, 00 1 Nature of molecules; energies of molecular motions Molecules are of course atoms that are held together by shared valence electrons. That is, most of each
Structure of diatomic molecules January 8, 00 1 Nature of molecules; energies of molecular motions Molecules are of course atoms that are held together by shared valence electrons. That is, most of each
J10M.1 - Rod on a Rail (M93M.2)
 Part I - Mechanics J10M.1 - Rod on a Rail (M93M.2) J10M.1 - Rod on a Rail (M93M.2) s α l θ g z x A uniform rod of length l and mass m moves in the x-z plane. One end of the rod is suspended from a straight
Part I - Mechanics J10M.1 - Rod on a Rail (M93M.2) J10M.1 - Rod on a Rail (M93M.2) s α l θ g z x A uniform rod of length l and mass m moves in the x-z plane. One end of the rod is suspended from a straight
Comparing and Improving Quark Models for the Triply Bottom Baryon Spectrum
 Comparing and Improving Quark Models for the Triply Bottom Baryon Spectrum A thesis submitted in partial fulfillment of the requirements for the degree of Bachelor of Science degree in Physics from the
Comparing and Improving Quark Models for the Triply Bottom Baryon Spectrum A thesis submitted in partial fulfillment of the requirements for the degree of Bachelor of Science degree in Physics from the
which implies that we can take solutions which are simultaneous eigen functions of
 Module 1 : Quantum Mechanics Chapter 6 : Quantum mechanics in 3-D Quantum mechanics in 3-D For most physical systems, the dynamics is in 3-D. The solutions to the general 3-d problem are quite complicated,
Module 1 : Quantum Mechanics Chapter 6 : Quantum mechanics in 3-D Quantum mechanics in 3-D For most physical systems, the dynamics is in 3-D. The solutions to the general 3-d problem are quite complicated,
Quantum Physics in the Nanoworld
 Hans Lüth Quantum Physics in the Nanoworld Schrödinger's Cat and the Dwarfs 4) Springer Contents 1 Introduction 1 1.1 General and Historical Remarks 1 1.2 Importance for Science and Technology 3 1.3 Philosophical
Hans Lüth Quantum Physics in the Nanoworld Schrödinger's Cat and the Dwarfs 4) Springer Contents 1 Introduction 1 1.1 General and Historical Remarks 1 1.2 Importance for Science and Technology 3 1.3 Philosophical
6. Qualitative Solutions of the TISE
 6. Qualitative Solutions of the TISE Copyright c 2015 2016, Daniel V. Schroeder Our goal for the next few lessons is to solve the time-independent Schrödinger equation (TISE) for a variety of one-dimensional
6. Qualitative Solutions of the TISE Copyright c 2015 2016, Daniel V. Schroeder Our goal for the next few lessons is to solve the time-independent Schrödinger equation (TISE) for a variety of one-dimensional
CONTENTS. vii. CHAPTER 2 Operators 15
 CHAPTER 1 Why Quantum Mechanics? 1 1.1 Newtonian Mechanics and Classical Electromagnetism 1 (a) Newtonian Mechanics 1 (b) Electromagnetism 2 1.2 Black Body Radiation 3 1.3 The Heat Capacity of Solids and
CHAPTER 1 Why Quantum Mechanics? 1 1.1 Newtonian Mechanics and Classical Electromagnetism 1 (a) Newtonian Mechanics 1 (b) Electromagnetism 2 1.2 Black Body Radiation 3 1.3 The Heat Capacity of Solids and
Physics 342 Lecture 30. Solids. Lecture 30. Physics 342 Quantum Mechanics I
 Physics 342 Lecture 30 Solids Lecture 30 Physics 342 Quantum Mechanics I Friday, April 18th, 2008 We can consider simple models of solids these highlight some special techniques. 30.1 An Electron in a
Physics 342 Lecture 30 Solids Lecture 30 Physics 342 Quantum Mechanics I Friday, April 18th, 2008 We can consider simple models of solids these highlight some special techniques. 30.1 An Electron in a
Total Angular Momentum for Hydrogen
 Physics 4 Lecture 7 Total Angular Momentum for Hydrogen Lecture 7 Physics 4 Quantum Mechanics I Friday, April th, 008 We have the Hydrogen Hamiltonian for central potential φ(r), we can write: H r = p
Physics 4 Lecture 7 Total Angular Momentum for Hydrogen Lecture 7 Physics 4 Quantum Mechanics I Friday, April th, 008 We have the Hydrogen Hamiltonian for central potential φ(r), we can write: H r = p
only two orbitals, and therefore only two combinations to worry about, but things get
 131 Lecture 1 It is fairly easy to write down an antisymmetric wavefunction for helium since there are only two orbitals, and therefore only two combinations to worry about, but things get complicated
131 Lecture 1 It is fairly easy to write down an antisymmetric wavefunction for helium since there are only two orbitals, and therefore only two combinations to worry about, but things get complicated
The Postulates of Quantum Mechanics Common operators in QM: Potential Energy. Often depends on position operator: Kinetic Energy 1-D case: 3-D case
 The Postulates of Quantum Mechanics Common operators in QM: Potential Energy Often depends on position operator: Kinetic Energy 1-D case: 3-D case Time Total energy = Hamiltonian To find out about the
The Postulates of Quantum Mechanics Common operators in QM: Potential Energy Often depends on position operator: Kinetic Energy 1-D case: 3-D case Time Total energy = Hamiltonian To find out about the
( ) dσ 1 dσ 2 + α * 2
 Chemistry 36 Dr. Jean M. Standard Problem Set Solutions. The spin up and spin down eigenfunctions for each electron in a many-electron system are normalized and orthogonal as given by the relations, α
Chemistry 36 Dr. Jean M. Standard Problem Set Solutions. The spin up and spin down eigenfunctions for each electron in a many-electron system are normalized and orthogonal as given by the relations, α
Multielectron Atoms.
 Multielectron Atoms. Chem 639. Spectroscopy. Spring 00 S.Smirnov Atomic Units Mass m e 1 9.109 10 31 kg Charge e 1.60 10 19 C Angular momentum 1 1.055 10 34 J s Permittivity 4 0 1 1.113 10 10 C J 1 m 1
Multielectron Atoms. Chem 639. Spectroscopy. Spring 00 S.Smirnov Atomic Units Mass m e 1 9.109 10 31 kg Charge e 1.60 10 19 C Angular momentum 1 1.055 10 34 J s Permittivity 4 0 1 1.113 10 10 C J 1 m 1
Basic Quantum Mechanics
 Frederick Lanni 10feb'12 Basic Quantum Mechanics Part I. Where Schrodinger's equation comes from. A. Planck's quantum hypothesis, formulated in 1900, was that exchange of energy between an electromagnetic
Frederick Lanni 10feb'12 Basic Quantum Mechanics Part I. Where Schrodinger's equation comes from. A. Planck's quantum hypothesis, formulated in 1900, was that exchange of energy between an electromagnetic
C. Show your answer in part B agrees with your answer in part A in the limit that the constant c 0.
 Problem #1 A. A projectile of mass m is shot vertically in the gravitational field. Its initial velocity is v o. Assuming there is no air resistance, how high does m go? B. Now assume the projectile is
Problem #1 A. A projectile of mass m is shot vertically in the gravitational field. Its initial velocity is v o. Assuming there is no air resistance, how high does m go? B. Now assume the projectile is
Problem 1: Spin 1 2. particles (10 points)
 Problem 1: Spin 1 particles 1 points 1 Consider a system made up of spin 1/ particles. If one measures the spin of the particles, one can only measure spin up or spin down. The general spin state of a
Problem 1: Spin 1 particles 1 points 1 Consider a system made up of spin 1/ particles. If one measures the spin of the particles, one can only measure spin up or spin down. The general spin state of a
L11.P1 Lecture 11. Quantum statistical mechanics: summary
 Lecture 11 Page 1 L11.P1 Lecture 11 Quantum statistical mechanics: summary At absolute zero temperature, a physical system occupies the lowest possible energy configuration. When the temperature increases,
Lecture 11 Page 1 L11.P1 Lecture 11 Quantum statistical mechanics: summary At absolute zero temperature, a physical system occupies the lowest possible energy configuration. When the temperature increases,
e L 2m e the Bohr magneton
 e L μl = L = μb 2m with : μ B e e 2m e the Bohr magneton Classical interation of magnetic moment and B field: (Young and Freedman, Ch. 27) E = potential energy = μ i B = μbcosθ τ = torque = μ B, perpendicular
e L μl = L = μb 2m with : μ B e e 2m e the Bohr magneton Classical interation of magnetic moment and B field: (Young and Freedman, Ch. 27) E = potential energy = μ i B = μbcosθ τ = torque = μ B, perpendicular
E = 2 (E 1)+ 2 (4E 1) +1 (9E 1) =19E 1
 Quantum Mechanics and Atomic Physics Lecture 22: Multi-electron Atoms http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Last Time Multi-electron atoms and Pauli s exclusion principle Electrons
Quantum Mechanics and Atomic Physics Lecture 22: Multi-electron Atoms http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Last Time Multi-electron atoms and Pauli s exclusion principle Electrons
Lecture 01. Introduction to Elementary Particle Physics
 Introduction to Elementary Particle Physics Particle Astrophysics Particle physics Fundamental constituents of nature Most basic building blocks Describe all particles and interactions Shortest length
Introduction to Elementary Particle Physics Particle Astrophysics Particle physics Fundamental constituents of nature Most basic building blocks Describe all particles and interactions Shortest length
3. Quantum Mechanics in 3D
 3. Quantum Mechanics in 3D 3.1 Introduction Last time, we derived the time dependent Schrödinger equation, starting from three basic postulates: 1) The time evolution of a state can be expressed as a unitary
3. Quantum Mechanics in 3D 3.1 Introduction Last time, we derived the time dependent Schrödinger equation, starting from three basic postulates: 1) The time evolution of a state can be expressed as a unitary
d 3 r d 3 vf( r, v) = N (2) = CV C = n where n N/V is the total number of molecules per unit volume. Hence e βmv2 /2 d 3 rd 3 v (5)
 LECTURE 12 Maxwell Velocity Distribution Suppose we have a dilute gas of molecules, each with mass m. If the gas is dilute enough, we can ignore the interactions between the molecules and the energy will
LECTURE 12 Maxwell Velocity Distribution Suppose we have a dilute gas of molecules, each with mass m. If the gas is dilute enough, we can ignore the interactions between the molecules and the energy will
QUANTUM MECHANICS Intro to Basic Features
 PCES 4.21 QUANTUM MECHANICS Intro to Basic Features 1. QUANTUM INTERFERENCE & QUANTUM PATHS Rather than explain the rules of quantum mechanics as they were devised, we first look at a more modern formulation
PCES 4.21 QUANTUM MECHANICS Intro to Basic Features 1. QUANTUM INTERFERENCE & QUANTUM PATHS Rather than explain the rules of quantum mechanics as they were devised, we first look at a more modern formulation
Practical Quantum Mechanics
 Siegfried Flügge Practical Quantum Mechanics With 78 Figures Springer-Verlag Berlin Heidelberg New York London Paris Tokyo Hong Kong Barcelona Budapest Contents Volume I I. General Concepts 1. Law of probability
Siegfried Flügge Practical Quantum Mechanics With 78 Figures Springer-Verlag Berlin Heidelberg New York London Paris Tokyo Hong Kong Barcelona Budapest Contents Volume I I. General Concepts 1. Law of probability
Solutions Final exam 633
 Solutions Final exam 633 S.J. van Enk (Dated: June 9, 2008) (1) [25 points] You have a source that produces pairs of spin-1/2 particles. With probability p they are in the singlet state, ( )/ 2, and with
Solutions Final exam 633 S.J. van Enk (Dated: June 9, 2008) (1) [25 points] You have a source that produces pairs of spin-1/2 particles. With probability p they are in the singlet state, ( )/ 2, and with
The Hydrogen Atom. Dr. Sabry El-Taher 1. e 4. U U r
 The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
6.1 Nondegenerate Perturbation Theory
 6.1 Nondegenerate Perturbation Theory Analytic solutions to the Schrödinger equation have not been found for many interesting systems. Fortunately, it is often possible to find expressions which are analytic
6.1 Nondegenerate Perturbation Theory Analytic solutions to the Schrödinger equation have not been found for many interesting systems. Fortunately, it is often possible to find expressions which are analytic
LSU Dept. of Physics and Astronomy Qualifying Exam Quantum Mechanics Question Bank (07/2017) =!2 π 2 a cos π x
 LSU Dept. of Physics and Astronomy Qualifying Exam Quantum Mechanics Question Bank (7/17) 1. For a particle trapped in the potential V(x) = for a x a and V(x) = otherwise, the ground state energy and eigenfunction
LSU Dept. of Physics and Astronomy Qualifying Exam Quantum Mechanics Question Bank (7/17) 1. For a particle trapped in the potential V(x) = for a x a and V(x) = otherwise, the ground state energy and eigenfunction
Quantum Mechanics II
 Quantum Mechanics II Prof. Boris Altshuler April, 20 Lecture 2. Scattering Theory Reviewed Remember what we have covered so far for scattering theory. We separated the Hamiltonian similar to the way in
Quantum Mechanics II Prof. Boris Altshuler April, 20 Lecture 2. Scattering Theory Reviewed Remember what we have covered so far for scattering theory. We separated the Hamiltonian similar to the way in
Electrons in a weak periodic potential
 Electrons in a weak periodic potential Assumptions: 1. Static defect-free lattice perfectly periodic potential. 2. Weak potential perturbative effect on the free electron states. Perfect periodicity of
Electrons in a weak periodic potential Assumptions: 1. Static defect-free lattice perfectly periodic potential. 2. Weak potential perturbative effect on the free electron states. Perfect periodicity of
Atoms, Molecules and Solids. From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation of the wave function:
 Essay outline and Ref to main article due next Wed. HW 9: M Chap 5: Exercise 4 M Chap 7: Question A M Chap 8: Question A From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation
Essay outline and Ref to main article due next Wed. HW 9: M Chap 5: Exercise 4 M Chap 7: Question A M Chap 8: Question A From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation
Applications of Quantum Theory to Some Simple Systems
 Applications of Quantum Theory to Some Simple Systems Arbitrariness in the value of total energy. We will use classical mechanics, and for simplicity of the discussion, consider a particle of mass m moving
Applications of Quantum Theory to Some Simple Systems Arbitrariness in the value of total energy. We will use classical mechanics, and for simplicity of the discussion, consider a particle of mass m moving
STONY BROOK UNIVERSITY DEPARTMENT OF PHYSICS AND ASTRONOMY. Comprehensive Examination. Classical Mechanics. August 25, 2014
 STONY BROOK UNIVERSITY DEPARTMENT OF PHYSICS AND ASTRONOMY Comprehensive Examination Classical Mechanics August 25, 2014 General Instructions: Three problems are given. If you take this exam as a placement
STONY BROOK UNIVERSITY DEPARTMENT OF PHYSICS AND ASTRONOMY Comprehensive Examination Classical Mechanics August 25, 2014 General Instructions: Three problems are given. If you take this exam as a placement
Atomic Structure. Chapter 8
 Atomic Structure Chapter 8 Overview To understand atomic structure requires understanding a special aspect of the electron - spin and its related magnetism - and properties of a collection of identical
Atomic Structure Chapter 8 Overview To understand atomic structure requires understanding a special aspect of the electron - spin and its related magnetism - and properties of a collection of identical
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall Duration: 2h 30m
 Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. ------------------- Duration: 2h 30m Chapter 39 Quantum Mechanics of Atoms Units of Chapter 39 39-1 Quantum-Mechanical View of Atoms 39-2
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. ------------------- Duration: 2h 30m Chapter 39 Quantum Mechanics of Atoms Units of Chapter 39 39-1 Quantum-Mechanical View of Atoms 39-2
University of Illinois at Chicago Department of Physics. Quantum Mechanics Qualifying Examination. January 7, 2013 (Tuesday) 9:00 am - 12:00 noon
 University of Illinois at Chicago Department of Physics Quantum Mechanics Qualifying Examination January 7, 13 Tuesday 9: am - 1: noon Full credit can be achieved from completely correct answers to 4 questions
University of Illinois at Chicago Department of Physics Quantum Mechanics Qualifying Examination January 7, 13 Tuesday 9: am - 1: noon Full credit can be achieved from completely correct answers to 4 questions
UNIVERSITY OF MARYLAND Department of Physics College Park, Maryland. PHYSICS Ph.D. QUALIFYING EXAMINATION PART II
 UNIVERSITY OF MARYLAND Department of Physics College Park, Maryland PHYSICS Ph.D. QUALIFYING EXAMINATION PART II January 20, 2017 9:00 a.m. 1:00 p.m. Do any four problems. Each problem is worth 25 points.
UNIVERSITY OF MARYLAND Department of Physics College Park, Maryland PHYSICS Ph.D. QUALIFYING EXAMINATION PART II January 20, 2017 9:00 a.m. 1:00 p.m. Do any four problems. Each problem is worth 25 points.
Chemistry 881 Lecture Topics Fall 2001
 Chemistry 881 Lecture Topics Fall 2001 Texts PHYSICAL CHEMISTRY A Molecular Approach McQuarrie and Simon MATHEMATICS for PHYSICAL CHEMISTRY, Mortimer i. Mathematics Review (M, Chapters 1,2,3 & 4; M&S,
Chemistry 881 Lecture Topics Fall 2001 Texts PHYSICAL CHEMISTRY A Molecular Approach McQuarrie and Simon MATHEMATICS for PHYSICAL CHEMISTRY, Mortimer i. Mathematics Review (M, Chapters 1,2,3 & 4; M&S,
Quantum Mechanics Solutions
 Quantum Mechanics Solutions (a (i f A and B are Hermitian, since (AB = B A = BA, operator AB is Hermitian if and only if A and B commute So, we know that [A,B] = 0, which means that the Hilbert space H
Quantum Mechanics Solutions (a (i f A and B are Hermitian, since (AB = B A = BA, operator AB is Hermitian if and only if A and B commute So, we know that [A,B] = 0, which means that the Hilbert space H
Paradigms in Physics: Quantum Mechanics
 Paradigms in Physics: Quantum Mechanics David H. McIntyre Corinne A. Manogue Janet Tate Oregon State University 23 November 2010 Copyright 2010 by David H. McIntyre, Corinne A. Manogue, Janet Tate CONTENTS
Paradigms in Physics: Quantum Mechanics David H. McIntyre Corinne A. Manogue Janet Tate Oregon State University 23 November 2010 Copyright 2010 by David H. McIntyre, Corinne A. Manogue, Janet Tate CONTENTS
The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of hydrogen atom.
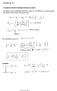 Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Solution of Second Midterm Examination Thursday November 09, 2017
 Department of Physics Quantum Mechanics II, Physics 570 Temple University Instructor: Z.-E. Meziani Solution of Second Midterm Examination Thursday November 09, 017 Problem 1. (10pts Consider a system
Department of Physics Quantum Mechanics II, Physics 570 Temple University Instructor: Z.-E. Meziani Solution of Second Midterm Examination Thursday November 09, 017 Problem 1. (10pts Consider a system
