Final report Results and evaluation of key comparison CCM.P-K12.1 for very low helium flow rates (leak rates)
|
|
|
- Allyson McDowell
- 5 years ago
- Views:
Transcription
1 Final report Results and evaluation of key comparison CCM.P-K1.1 for very low helium flow rates (leak rates) J. Šetina 1, M. Vičar, D. Pražák 1 Institute of Metals and Technology (IMT), Lepi pot 11, 1000 Ljubljana, Slovenia Czech Metrological Institute (CMI), Okružní 31, Brno, 63800, Czech Republic Version 1:
2 CONTENT 1. INTRODUCTION TRANSFER STANDARDS AND QUANTITY TO BE DETERMINED PARTICIPATING LABORATORIES AND THEIR MEASUREMENT SYSTEMS CMI IMT CHRONOLOGY AND MEASUREMENT PROCEDURE UNCERTAINTIES OF REFERENCE STANDARDS RESULTS OF THE PILOT LABORATORY (IMT) TEMPERATURE COEFFICIENT OF TRANSFER STANDARD LEAK RESULTS OF IMT (PILOT) RESULTS OF CMI TEMPERATURE COEFFICIENT OF TRANSFER STANDARD LEAKS RESULTS OF CMI TIME-DEPENDENT BEHAVIOUR OF TRANSFER STANDARD CALCULATION OF REFERENCE VALUE AND DEGREE OF EQUIVALENCE DEGREE OF EQUIVALENCE OF IMT AND CMI LINKING TO THE REFERENCE VALUE OF THE CCM KEY COMPARISON DISCUSSION AND CONCLUSIONS REFERENCES... 19
3 1. Introduction The Czech Metrology Institute (CMI) took part and failed to prove equivalence in the Key Comparison CCM.P-K1 of helium leak artefacts (leak rates) into vacuum [1]. CMI used a method of comparison of He flow from the constant pressure variable volume flowmeters and from the transfer standard. A quadrupole mass spectrometer (QMS) Prisma QMA 00 served as a comparator. The failure was caused by an atmospheric leak through a valve. Manipulation with this leaking valve significantly changed the concentrations of atmospheric gases in the chamber. He signal depends on the partial pressures of other present gases. Nitrogen and oxygen levels stabilised quickly, but water remained unstable even after 0 min. As a solution (except changing the valve), CMI vacuum laboratory lowered the emission current of QMS from nominal ma to ( ) ma. The dependence of the He signal on the residual atmosphere is weaker by the lower emission currents. Hence it has a better short-time stability to serve as a He flow-rates comparator. Further, a lower partial pressure of water is reached by opening a by-pass parallel to the orifice. Both precautions increase a relative noise of the He signal. Hence it is necessary to use the longer integration times (cca 5 s for the lowest leak rates). However, the resultant total uncertainty is lower than originally. The Institute of Metals and Technology (IMT), a successful participant in CCM.P-K1, volunteered to serve as pilot and link in a following bilateral comparison of IMT and CMI that obtained designation CCM.P-K1.1 in June 01. It was decided to perform the comparison with a glass permeation helium leak artefact at nominally mol/s ( mbar L s -1 ) at 3 C. The comparison measurements were performed in October 01.. Transfer standards and quantity to be determined The helium permeation leak Alcatel FV4300, serial No. FC served as the transfer standard. Its nominal leak rate at 0 C by time of purchase in 007 equalled mol/s ( mbar L s -1 ) and its nominal temperature coefficient equals +3 %/K. The leak was equipped with a KF16 flange and a valve. The valve was left open during transportation to minimize the accumulation of helium gas downstream of the permeation leak element. This kept the density of helium in the permeating part 3
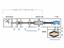
4 roughly constant during transportation. This was important in order to have acceptable time constants for reaching equilibrium after installation in a laboratory. For transport, the connecting flange was equipped by a protective plastic cover leaving it more or less open to atmosphere. The measurand determined by each laboratory was the molar flow rate of the helium atoms flowing out of the transfer standard leak at the time of calibration: q t test leak at 3.0 C, (1) where is the number of moles of helium exiting out of the leak in the time t. This quantity depends on the temperature of the leak artefact. For this reason the target temperature was (3.0 0.) C. This could not be always followed, but all the values taken at a temperature different from 3.0 C were recalculated to values which would have been measured at 3.0 C exactly. This was done by applying a measured temperature coefficient of the leak. Since the flow from the transfer standard leaks was permanent and the gas reservoir limited, q could also be time dependent. But this effect could be neglected thanks to the short time needed for this comparison. The molar flow was not measured directly, but the known energetic throughputs qpv [] at some thermodynamic temperature T were converted from qpv into q by: q q pv RT, where R = Pa m³ mol -1 K -1 is the universal gas constant. () 3. Participating laboratories and their measurement systems The only participants in this comparison were IMT - Slovenia (pilot) and CMI - Czech Republic. Both their laboratory standards took part in CCM.P-K1 and were considered as primary [1] CMI The CMI used a comparison method with a constant pressure flowmeter. A quadrupole mass spectrometer (QMS, Balzers Prisma) installed at the calibration chamber of a continuous expansion system [3] to [7] served as indicator to compare the helium gas 4
5 flows from the flow meter (signal on QMS: I) and from the transfer standard leak (signal on QMS: I1), which were intermittently admitted into the calibration chamber, see Fig. 1. Figure 1 Schematic of CMI set-up The calibrated leak was connected to an auxiliary turbomolecular pump (backed by a membrane pump) in the periods when it was not connected to the calibration chamber. The linearity of the QMS was checked in the relevant range of this comparison. The non-linearity was found to be insignificant. Nevertheless the gas flow from the flowmeter was set by the sapphire valve so to give a partial pressure signal as close as possible to the signal caused by the transfer standard. The molar flow rate from the leak under calibration is given by q I 1 1, leak q,fm I I I 1, (3) where I1, are offset corrected readings. The offset reading is obtained when there is no helium flow onto the QMS. 5
6 No thermal bath was utilised to stabilize the temperature of the transfer standards. Instead, they were thermally insulated by the means of a foam wrap. Its temperature was measured by two sensors Pt1000 fixed to its reservoir container and the mean of their indications was taken into account. The temperature of the transfer standard remained stable within ±0.1 C. 3.. IMT The IMT has developed a primary helium leak [8] based on a glass permeation element, a reservoir with adjustable helium gas pressure and a calibration facility for in-situ measurement of generated helium gas flow by a pressure rise method using spinning rotor gauge, see Fig.. IMT primary reference leak He SRG 1 CH1 UUT QMS1 SRG CH NEG ORIFICE BAG QMS TURBO Figure The IMT leak calibration/comparison system 6
7 The fill pressure in the reservoir can be varied from 100 Pa to 1 MPa, to generate flows from mol/s to mol/s with the glass permeation element at room temperature. Compared to the description in [8] some improvements were made, e.g. a temperature shroud made of Al with a possibility to heat the helium reservoir with the glass permeation element to 140 C, Ti/Ta getter was replaced with ST 1 NEG from SAES Getters. This primary helium leak is connected to a leak comparator apparatus shown in Fig.. The He partial pressure in the comparator vacuum chamber (CH) is measured with a quadrupole mass spectrometer (QMS1). The He gas pressure in the reservoir of the primary leak is adjusted to produce nearly the same He signal as the unit under test (UUT). The volume of the comparator vacuum chamber was also determined. This enables direct primary measurement of He gas flow from UUT (the transfer standard) by pressure rise method in CH (nominal volume 7 L) using SRG (traceable to PTB). The purity of accumulated helium gas can be checked by another quadrupole mass spectrometer (QMS). This method is generally used for He flows from mol/s to mol/s, and was applied in this comparison. 4. Chronology and measurement procedure In order to determine the stability of the transfer standard, it was decided that two measurement series will be made by CMI, prior and after the measurements at IMT. Tab. 1 presents the actual chronology of the calibrations. Each laboratory was required to perform 6 measurements on the first calibration day and to repeat this series on the second day. Table 1 Chronology of measurements. CMI1 and CMI mean the 1 st and nd calibration sequence carried out by CMI. Laboratory Measurement CMI and IMT and CMI and
8 5. Uncertainties of reference standards The Type B uncertainty of IMT was 0.49 % (for k = 1) and of CMI from 1.5 to 1.54 % (for k = 1). Type A uncertainties will show up in the scatter of data at repeat measurements, and were calculated using methods described in Sec. 6 and Results of the pilot laboratory (IMT) 6.1. Temperature coefficient of transfer standard leak To correct the data obtained at different temperatures to a common reference temperature, the temperature coefficients of the flow rate from the leak artefact had to be determined. It was done from the comparison data plus some extra preliminary measurements. A linear least square fit was applied to the data to determine the slope as relative temperature coefficient T of the flow rates: : q q 3C T T %/K. (4) To correct the measured leak rates to a common temperature of 3 C, the following formula was applied: C q T 1 T q, j 3, j j T j. Herein q,,j is the leak rate as determined by participant j for the temperature Tj in kelvins at the time of calibration. (5) 6.. Results of IMT (pilot) The results of the pilot are summarized in Tab.. In each row the calibration date and time, the mean temperature of the leak, the mean measured leak rate q() at this temperature, the uncertainty of the used standard uet (k = 1) as the main part of the Type B uncertainty and the molar flow rate q(3 C) calculated by Eq. (5) are given. After each day the mean and the standard deviation of the mean ua of, q() and q(3 C) are stated. The value of ua was calculated by u A 1 N( N 1) N q, l q l1. (6) 8
9 Table Results of the pilot laboratory (IMT). Date Time q () uet (k=1) uet (k=1) q (3 C) HH:MM C mol/s mol/s % mol/s : E E E : E E E : E E E : E E E : E E E : E E E-11 mean E E-11 ua E-14.E : E E E : E E E : E E E : E E E : E E E : E E E-11 mean e e-11 ua E E-14 Room temperature was (3.5 ± 1.0) C on the 1 st day and (.5 ± 1.0) C on the nd day. Leak temperature measurement uncertainty equals 0.10 C for k = 1. As one can see from Tab., the temperatures of the leak varied from.69 C to 3.88 C. Hence we decided to consider also the uncertainty of the temperature correction. Another point to consider is, whether the values obtained at the two calibration days belong to the same parent distribution. We assume that the effects contributing to the Type B uncertainties are no different at (more or less successive) calibrations days within the same sequence. Therefore we consider the values measured on different days to be compatible, if the absolute difference between the mean values is smaller than two standard deviations of the difference: q, q,1 ua,1 ua,. (7) Herein q,1 and q, are the mean values determined on their respective calibration days with ua,1, ua, being their respective sample standard deviations. 9
10 Figure 3 The results of IMT. There are shown q (3 C), their means and twice standard deviations of these means. The time axis is out of scale, showing only ordinality and grouping into calibration days. From Tab. one can calculate that the difference of mean values on different days in a sequence ( mol/s) agreed within two standard deviations of the difference ( mol/s). Hence the mean and the standard deviation of the mean of all the data at the two measurement days can be taken for further evaluation. The results of the pilot taken together are summarized in Tab. 3. In each row the mean temperature of the leak, the mean corrected leak rate q(3 C), the temperature correction factor tc and its uncertainty T T j 10
11 u tc u u 1 T T T for (k = 1), the uncertainty of the used standard uet (k = 1) and Type B uncertainty calculated as u B et utc tc u q are given. Table 3 Results of the pilot laboratory elaborated together. q (3 C) tc utc (k=1) uet (k=1) ub C mol/s - - mol/s mol/s E E E E E E E E E E E E E E E E E E E E E E E E E E E E E E E E E E E E-13 The resultant mean value of the pilot equals mol/s with ua calculated by (6) equal to mol/s and maximal ub equal to mol/s. The combined uncertainty u (dominated by ub) was calculated by u. (8) u A ub The effective number of degrees of freedom is cca 19000, so the expanded uncertainty can be calculated using k =. Hence the result of the pilot is (.583 ± 0.09) mol/s. 7. Results of CMI 7.1. Temperature coefficient of transfer standard leaks Also CMI determined the value of the temperature coefficient of the transfer standard. It was done from the comparison data plus some extra preliminary measurements. A linear least square fit was applied to the data to determine the slope as relative temperature coefficient T of the flow rates: q T %/K. (9) q 3C T 11
12 This value was used to correct the leak rates measured by CMI to a common temperature of 3 C following formula (5). 7.. Results of CMI Table 4 Results of CMI. Date Time q() uet (k=1) uet (k=1) q(3 C) HH:MM C mol/s mol/s % mol/s : E E E : E E E : E E E : E E E : E E E : E E E-11 mean e e-11 st.dev.of mean E E : E E E : E E E : E E E : E E E : E E E : E E E-11 mean e e-11 st.dev.of mean E E : E E E : E E ,64E : E E E : E E E : E E E : E E E-11 mean.93.57e e-11 st.dev.of mean E E : E E E : E E E : E E E : E E E : E E E : E E E-11 mean E E-11 st.dev.of mean E-13 1.E-13 The results of CMI are summarized in Tab. 4. The meaning of the symbols is the same as for Tab. 3. Room temperature was always between.6 C and 3.0 C with uncertainty 0.5 C. Leak temperature measurement uncertainty equals 0.10 C for k = 1. As one can see 1
13 from Tab. 4, the temperatures of the leak varied from.88 C to 3.05 C. Hence we decided to consider also the uncertainty of the temperature correction Time-dependent behaviour of transfer standard The physical principle of the reference leak (permeation of He from fixed reservoir) gives permanent decay of He gas flow with time. However the decay rate is usually sufficiently small so it is practically insignificant in a short period of time (two weeks), providing there is no additional loss of He from reservoir due to a leaky reservoir. Nominal decay rate at room temperature as given by the manufacturer for our transfer leak is % per year. In two weeks period, which was overall duration of the comparison, the decay would be only 0.08 %, which is practically immeasurable. Linear regression of the CMI measurements that were done at the beginning and at the end of the comparison gives the decay rate of 0.07 %/d, but the R-square of the regression is only 0.1 (figure 4). Figure 4 Time dependence of leak rate of the transfer standard. The scatter of the measured data is significantly larger than possible drift in two weeks period, therefore we decided to use for the calculation of the equivalence of two 13
14 laboratories the mean value of two CMI measurement series and neglect the drift in two weeks period. In addition the measurements of the pilot lab IMT (also shown in figure 4) were done almost in the middle of the time interval between the two measurements of CMI, so the mean value of the two measurements of CMI practically cancels out the effect of linear drift of transfer standard in present comparison. Another point to consider is, whether the values obtained at the four calibration days belong to the same parent distribution. We assume that the effects contributing to the Type B uncertainties are no different during two weeks. Therefore we consider the values measured on different days to be compatible, if condition (7) is fulfilled. Figure 5 The results of IMT (red) and CMI (blue). There are shown q (3 C), their means and double standard deviations of these means. The time axis is out of scale, showing only ordinality and grouping into calibration days. 14
15 From Tab. 4 and Fig. 5 one can see that the difference of mean values obtained on two successive days at CMI ( mol/s and mol/s) agreed within two standard deviations of the differences ( mol/s and mol/s). Moreover, this is also valid for the 1 st and the 3 rd day, the 1 st and the 4 th day, the nd and the 3 rd day and the nd and the 4 th day, when we get the differences ( mol/s, mol/s, mol/s and mol/s) agreed within double standard deviations of the differences ( mol/s, mol/s, mol/s and mol/s). Hence the mean and the standard deviation of the mean of all the data at the four measurement days can be taken for further evaluation. The results of CMI taken together are summarized in Tab. 5. In each row the mean temperature of the leak, the mean corrected leak rate q(3 C), the temperature correction factor tc and its uncertainty for (k = 1), the uncertainty T T j of the used standard uet (k = 1) and Type B uncertainty calculated as u B et utc tc u q are given. Table 5 Results of CMI elaborated together. u et (k=1) q (3 C) tc u tc (k=1) u B C mol/s mol/s - - mol/s E-13.6E E E-13.68E E E E E E-13.56E E E-13.63E E E E E E E E E E E E-13.64E E E E E E-13.58E E E-13.60E E E E E E-13.64E E E E E E E E E E E E E E E-13.56E E E-13.63E E E E E E E E E E E E E E-13 15
16 The resultant mean value of the participating lab (CMI) equals mol/s with ua calculated by (6) equal to mol/s and maximal ub equal to mol/s. The combined uncertainty u (dominated by ub) was calculated by (8). The effective number of degrees of freedom is cca 18000, so the expanded uncertainty can be calculated using k =. Hence the result is (.5884 ± 0.08) mol/s. 8. Calculation of reference value and degree of equivalence The reference value of this comparison can be calculated as q rv 1 qimt qcmi (10) and its uncertainty as U rv 1 U IMT UCMI. (11) Hence q (.5858 ± 0.044) mol/s. rv Now we can get the differences from the reference value D j q j q rv. (1) Hence D mol/s and D mol/s. IMT CMI The uncertainties of these differences are U Dj rv j U U. (13) Hence and U D IMT = mol/s U D CMI = mol/s. Now we can determine the degrees of equivalence E n D U j Dj Hence E 0.05 nimt. (14) 16
17 and E ncmi 9. Degree of equivalence of IMT and CMI The pair-wise degree of equivalence of IMT and CMI can be calculated as q U IMT IMT q CMI U CMI (15) 10. Linking to the reference value of the CCM key comparison The reference value of K1 was qrv_k1 = mol/s [1]. Because a different transfer standard was used in CCM.P-K1.1, a leak rate qrv_k1.1 = mol/s is significantly different from qrv_k1. Ratio of both reference values is: RK1/K1.1 = qrv_k1 / qrv_k1.1 = (16) Difference of IMT from the reference value of the comparison CCM.P-K1 was K1-IMT = mol/s and its associated expanded uncertainty for (k = ) U(K1-IMT) = mol/s [1]. Difference of IMT from the reference value of comparison CCM.P-K1.1 was K1.1-IMT = mol/s and its expanded uncertainty U(K1.1-IMT) = mol/s. Difference of CMI from the reference value of comparison CCM.P-K1.1 was K1.1-CMI = mol/s and its expanded uncertainty U(K1.1-CMI) = mol/s. In CCM.P-K1.1, IMT used the same primary system and the same measurement method as in CCM.P-K1. Relative uncertainties of IMT system at mol/s and at mol/s are the same. Relative uncertainties of CMI system are also the same at mol/s and at mol/s. To link K1 and K1.1, the reference value qrv_k1.1, and all associated differences and uncertainties of both labs in K1.1 have to be corrected by multiplying with RK1/K
18 This correction conserves the relative differences and relative uncertainties. Hence we get the following values. qrv_k1.1_c = mol/s RK1/K1.1 = mol/s, U(qrv_K1.1_C) = mol/s RK1/K1.1 = mol/s, K1.1-IMT_C = mol/s RK1/K1.1 = mol/s, U(K1.1-IMT_C) = mol/s RK1/K1.1 = mol/s, K1.1-CMI_C = mol/s RK1/K1.1 = mol/s, U(K1.1-CMI_C) = mol/s RK1/K1.1 = mol/s. Now the link to CCM.P-K1 can be determined. Difference of the CMI from the reference value of CCM.P-K1 is: K1-CMI = K1-IMT K1.1-IMT_C + K1.1-CMI_C = = ( ) mol/s = mol/s. (17) Uncertainty of the difference of CMI from the reference value of CCM.P-K1 is: U(K1-CMI) = U(K1.1-CMI_C) = mol/s. And finaly the equivalence of CMI to the CCM.P-K1 reference value is: X K1-CMI U = 0.1. K1-CMI It can be easily seen that CMI is also equivalent with the reference value of key comparison CCM.P-K Discussion and conclusions CMI proved equivalence both with IMT and with the reference value of the key comparison CCM.P-K1. 18
19 1. References [1] Jousten K Arai K Becker U Bodnar O Boineau F Fedchak J A Gorobey V Wu J Mari D Mohan P Setina J Toman B Vičar M Yu H Y. 013 Metrologia 50 Tech. Suppl doi: / /50/1a/07001 [] Peggs GN 1976 Vacuum 6 31 [3] Peksa L Řepa P Gronych T Tesař J Pražák D 004 Vacuum [4] Tesar J Prazak D Stanek F Repa P Peksa L 007 Vacuum [5] Gronych T Peksa L Řepa P Wild J Tesař J Pražák D Krajíček Z 008 Metrologia [6] Peksa L Gronych T Řepa P Tesař J Vičar M Pražák D Krajíček Z Wild J. 008 Metrologia , [7] Peksa L et al 008 J. Phys.: Conf. Ser (4pp) doi: / /100/9/09009 [8] Erjavec B Šetina J 003 XVII IMEKO World Congress, Dubrovnik, Croatia Zagreb: HMD - Croatian Metrology Society,
AE 3051, Lab #16. Investigation of the Ideal Gas State Equation. By: George P. Burdell. Group E3
 AE 3051, Lab #16 Investigation of the Ideal Gas State Equation By: George P. Burdell Group E3 Summer Semester 000 Abstract The validity of the ideal gas equation of state was experimentally tested for
AE 3051, Lab #16 Investigation of the Ideal Gas State Equation By: George P. Burdell Group E3 Summer Semester 000 Abstract The validity of the ideal gas equation of state was experimentally tested for
Review of investigation of influence of operational parameters on metrological characteristics of QMS within EMRP IND12 project
 Review of investigation of influence of operational parameters on metrological characteristics of QMS within EMRP IND12 project Janez Setina Institute of Metals and Technology, (IMT), Slovenia 1 Some operational
Review of investigation of influence of operational parameters on metrological characteristics of QMS within EMRP IND12 project Janez Setina Institute of Metals and Technology, (IMT), Slovenia 1 Some operational
Final Report August 2010
 Bilateral Comparison of 100 pf Capacitance Standards (ongoing BIPM key comparison BIPM.EM-K14.b) between the CMI, Czech Republic and the BIPM, January-July 2009 J. Streit** and N. Fletcher* *Bureau International
Bilateral Comparison of 100 pf Capacitance Standards (ongoing BIPM key comparison BIPM.EM-K14.b) between the CMI, Czech Republic and the BIPM, January-July 2009 J. Streit** and N. Fletcher* *Bureau International
Lecture 4. Ultrahigh Vacuum Science and Technology
 Lecture 4 Ultrahigh Vacuum Science and Technology Why do we need UHV? 1 Atmosphere = 760 torr; 1 torr = 133 Pa; N ~ 2.5 10 19 molecules/cm 3 Hertz-Knudsen equation p ZW 1/ 2 ( 2mk T) At p = 10-6 Torr it
Lecture 4 Ultrahigh Vacuum Science and Technology Why do we need UHV? 1 Atmosphere = 760 torr; 1 torr = 133 Pa; N ~ 2.5 10 19 molecules/cm 3 Hertz-Knudsen equation p ZW 1/ 2 ( 2mk T) At p = 10-6 Torr it
Determination of Minimum Detectable Partial Pressure (MDPP) of QMS and its Uncertainty
 Workshop on Towards more reliable partial and outgassing rate measurements Berlin, Germany, January 30 - February 1, 2017 Determination of Minimum Detectable Partial Pressure (MDPP) of QMS and its Uncertainty
Workshop on Towards more reliable partial and outgassing rate measurements Berlin, Germany, January 30 - February 1, 2017 Determination of Minimum Detectable Partial Pressure (MDPP) of QMS and its Uncertainty
Nanoholes for leak metrology
 Vacuum Metrology for Industry Nanoholes for leak metrology Università Degli Studi di Genova, Italy OUTLINE INTRODUCTION FABRICATION OF NANOHOLES GEOMETRICAL CHARACTERIZATION LEAK DEVICES RESULTS: PTB INRIM
Vacuum Metrology for Industry Nanoholes for leak metrology Università Degli Studi di Genova, Italy OUTLINE INTRODUCTION FABRICATION OF NANOHOLES GEOMETRICAL CHARACTERIZATION LEAK DEVICES RESULTS: PTB INRIM
Final Report on the Torque Key Comparison CCM.T-K2 Measurand Torque: 0 kn m, 10 kn m, 20 kn m Dirk Röske 1), Koji Ogushi 2)
 Final Report on the Torque Key Comparison CCM.T-K Measurand Torque: kn m, 1 kn m, kn m Dirk Röske 1), Koji Ogushi ) 1) ) Dirk RÖSKE Physikalisch-Technische Bundesanstalt Department 1. Solid Mechanics orking
Final Report on the Torque Key Comparison CCM.T-K Measurand Torque: kn m, 1 kn m, kn m Dirk Röske 1), Koji Ogushi ) 1) ) Dirk RÖSKE Physikalisch-Technische Bundesanstalt Department 1. Solid Mechanics orking
PTB S 16.5 MN HYDRAULIC AMPLIFICATION MACHINE AFTER MODERNIZATION
 IMEKO 22 nd TC3, 15 th TC5 and 3 rd TC22 International Conferences 3 to 5 February, 2014, Cape Town, Republic of South Africa PTB S 16.5 MN HYDRAULIC AMPLIFICATION MACHINE AFTER MODERNIZATION R. Kumme
IMEKO 22 nd TC3, 15 th TC5 and 3 rd TC22 International Conferences 3 to 5 February, 2014, Cape Town, Republic of South Africa PTB S 16.5 MN HYDRAULIC AMPLIFICATION MACHINE AFTER MODERNIZATION R. Kumme
Deuterium Gas Analysis by Residual Gas Analyzer
 Journal of Physics: Conference Series Deuterium Gas Analysis by Residual Gas Analyzer To cite this article: B K Das et al 2012 J. Phys.: Conf. Ser. 390 012009 View the article online for updates and enhancements.
Journal of Physics: Conference Series Deuterium Gas Analysis by Residual Gas Analyzer To cite this article: B K Das et al 2012 J. Phys.: Conf. Ser. 390 012009 View the article online for updates and enhancements.
TYPICAL PRESSURE MEASUREMENT UNCERTAINTY DEFINED BY AN FPG8601 FORCE BALANCED PISTON GAUGE
 TYPICAL PRESSURE MEASUREMENT UNCERTAINTY DEFINED BY AN FPG8601 FORCE BALANCED PISTON GAUGE Michael Bair and Pierre Delajoud 2002 DEC 10 Revised 2004 MAR 26 FORWARD FPG8601 is a pressure standard designed
TYPICAL PRESSURE MEASUREMENT UNCERTAINTY DEFINED BY AN FPG8601 FORCE BALANCED PISTON GAUGE Michael Bair and Pierre Delajoud 2002 DEC 10 Revised 2004 MAR 26 FORWARD FPG8601 is a pressure standard designed
Preparation Primary Gas Reference Material (PGRM) by using NIMT Facilities
 Preparation Primary Gas Reference Material (PGRM) by using NIMT Facilities By Ms.Ratirat Sinweeruthai June 9-11, 2010 Tsukuba, Japan 8 th APMP/TCQM Gas CRM Workshop Overview Objective of Study EMU of Weighing
Preparation Primary Gas Reference Material (PGRM) by using NIMT Facilities By Ms.Ratirat Sinweeruthai June 9-11, 2010 Tsukuba, Japan 8 th APMP/TCQM Gas CRM Workshop Overview Objective of Study EMU of Weighing
PREDICTION OF THE OUTPUT VOLTAGE OF DC VOLTAGE STANDARDS
 XIX IMEKO World Congress Fundamental and Applied Metrology September 6 11, 009, Lisbon, ortugal REDICTION OF THE OUTUT VOLTAGE OF DC VOLTAGE STANDARDS Damir Ilić, Alan Šala, Ivan Leniček Faculty of Electrical
XIX IMEKO World Congress Fundamental and Applied Metrology September 6 11, 009, Lisbon, ortugal REDICTION OF THE OUTUT VOLTAGE OF DC VOLTAGE STANDARDS Damir Ilić, Alan Šala, Ivan Leniček Faculty of Electrical
High pressure comparisons between seven European National Laboratories
 J4/2007 High pressure comparisons between seven European National Laboratories Range 50 MPa to 500 MPa Report on EUROMET Project 881 Markku Rantanen, Sari Semenoja, Måns Ackerholm, Antoine Condereys, Zdenek
J4/2007 High pressure comparisons between seven European National Laboratories Range 50 MPa to 500 MPa Report on EUROMET Project 881 Markku Rantanen, Sari Semenoja, Måns Ackerholm, Antoine Condereys, Zdenek
Calibration of capacitance diaphragm gauges with 1333 Pa full scale by direct comparison to resonant silicon gauge and static expansion system
 ACTA IMEKO June 2014, Volume 3, Number 2, 48 53 www.imeko.org Calibration of capacitance diaphragm gauges with 1333 Pa full scale by direct comparison to resonant silicon gauge and static expansion system
ACTA IMEKO June 2014, Volume 3, Number 2, 48 53 www.imeko.org Calibration of capacitance diaphragm gauges with 1333 Pa full scale by direct comparison to resonant silicon gauge and static expansion system
The MMs are 4 KRAL screw meters, normally permanently installed in the BEV test rig as secondary working standards.
 Report: Intercomparison of calibration of master meters (screw meters) installed in a test rig, by two independent test methods (by BEV-standard capacity tanks and by CMI- Brooks piston prover) Euromet
Report: Intercomparison of calibration of master meters (screw meters) installed in a test rig, by two independent test methods (by BEV-standard capacity tanks and by CMI- Brooks piston prover) Euromet
Final Report on Comparison of Hydraulic Pressure Balance Effective Area Determination in the Range up to 80 MPa
 EURAMET - THE EUROPEAN ASSOCIATION OF NATIONAL METROLOGY INSTITUTES Comparison Identifier: EURAMET 1414 (Registered in KCDB as EURAMET.M.P-S18) Final Report on Comparison of Hydraulic Pressure Balance
EURAMET - THE EUROPEAN ASSOCIATION OF NATIONAL METROLOGY INSTITUTES Comparison Identifier: EURAMET 1414 (Registered in KCDB as EURAMET.M.P-S18) Final Report on Comparison of Hydraulic Pressure Balance
EVALUATION OF ADSORPTION EFFECT BY USING SILICON SPHERE AND SILICON SURFACE ARTEFACTS
 XVIII IMEKO WORLD CONGRESS Metrology for a Sustainable Development September, 17 22, 2006, Rio de Janeiro, Brazil EVALUATION OF ADSORPTION EFFECT BY USING SILICON SPHERE AND SILICON SURFACE ARTEFACTS Jin
XVIII IMEKO WORLD CONGRESS Metrology for a Sustainable Development September, 17 22, 2006, Rio de Janeiro, Brazil EVALUATION OF ADSORPTION EFFECT BY USING SILICON SPHERE AND SILICON SURFACE ARTEFACTS Jin
Physics 4C Chapter 19: The Kinetic Theory of Gases
 Physics 4C Chapter 19: The Kinetic Theory of Gases Whether you think you can or think you can t, you re usually right. Henry Ford The only thing in life that is achieved without effort is failure. Source
Physics 4C Chapter 19: The Kinetic Theory of Gases Whether you think you can or think you can t, you re usually right. Henry Ford The only thing in life that is achieved without effort is failure. Source
If the dividing wall were allowed to move, which of the following statements would not be true about its equilibrium position?
 PHYS 213 Exams Database Midterm (A) A block slides across a rough surface, eventually coming to a stop. 1) What happens to the block's internal thermal energy and entropy? a. and both stay the same b.
PHYS 213 Exams Database Midterm (A) A block slides across a rough surface, eventually coming to a stop. 1) What happens to the block's internal thermal energy and entropy? a. and both stay the same b.
Thermodynamics. Joule-Thomson effect Ideal and Real Gases. What you need: Complete Equipment Set, Manual on CD-ROM included
 Ideal Real Gases Thermodynamics 30600 What you can learn about Real gas Intrinsic energy Gay-Lussac theory Throttling Van der Waals equation Van der Waals force Inverse Inversion temperature Principle:
Ideal Real Gases Thermodynamics 30600 What you can learn about Real gas Intrinsic energy Gay-Lussac theory Throttling Van der Waals equation Van der Waals force Inverse Inversion temperature Principle:
Interferometric determination of thermal expansion coefficient of piston/cylinder unit preliminary investigation
 Journal of Physics: Conference Series PAPER OPEN ACCESS Interferometric determination of thermal expansion coefficient of piston/cylinder unit preliminary investigation To cite this article: L Grgec Bermanec
Journal of Physics: Conference Series PAPER OPEN ACCESS Interferometric determination of thermal expansion coefficient of piston/cylinder unit preliminary investigation To cite this article: L Grgec Bermanec
PRIMARY STANDARD AND TRACEABILITY CHAIN FOR MICROFLOW OF LIQUIDS
 PRIMARY STANDARD AND TRACEABILITY CHAIN FOR MICROFLOW OF LIQUIDS Miroslava Benkova 1 and Ivan Mikulecky 1 Czech Institute of Metrology, Okruzni 31, Brno, Czech Republic Slovak Institute of Metrology, Karloveska
PRIMARY STANDARD AND TRACEABILITY CHAIN FOR MICROFLOW OF LIQUIDS Miroslava Benkova 1 and Ivan Mikulecky 1 Czech Institute of Metrology, Okruzni 31, Brno, Czech Republic Slovak Institute of Metrology, Karloveska
A Method for Verifying Traceability in Effective Area for High Pressure Oil Piston-Cylinders
 A Method for Verifying Traceability in Effective Area for High Pressure Oil Piston-Cylinders Abstract Speaker/Author: Michael Bair Fluke Calibration 4765 East Beautiful Lane Phoenix, AZ 85042 Ph: 602 773
A Method for Verifying Traceability in Effective Area for High Pressure Oil Piston-Cylinders Abstract Speaker/Author: Michael Bair Fluke Calibration 4765 East Beautiful Lane Phoenix, AZ 85042 Ph: 602 773
Standard Practices for Air Speed Calibration Testing
 Standard Practices for Air Speed Calibration Testing Rachael V. Coquilla Bryza Wind Lab, Fairfield, California Air speed calibration is a test process where the output from a wind measuring instrument
Standard Practices for Air Speed Calibration Testing Rachael V. Coquilla Bryza Wind Lab, Fairfield, California Air speed calibration is a test process where the output from a wind measuring instrument
Speed Distribution at CONSTANT Temperature is given by the Maxwell Boltzmann Speed Distribution
 Temperature ~ Average KE of each particle Particles have different speeds Gas Particles are in constant RANDOM motion Average KE of each particle is: 3/2 kt Pressure is due to momentum transfer Speed Distribution
Temperature ~ Average KE of each particle Particles have different speeds Gas Particles are in constant RANDOM motion Average KE of each particle is: 3/2 kt Pressure is due to momentum transfer Speed Distribution
EURAMET.M.P-S9 / EURAMET 1170, LOOP2 Comparison in the negative gauge pressure range -950 to 0 hpa FINAL REPORT
 EURAMET.M.P-S9 / EURAMET 1170, LOOP Comparison in the negative gauge pressure range -950 to 0 hpa FINAL REPORT P. Otal 1, F. Boineau 1, N. Medina, D. Pražák 3, C. Wüthrich 4, S. Saxholm 5, W. Sabuga 6,
EURAMET.M.P-S9 / EURAMET 1170, LOOP Comparison in the negative gauge pressure range -950 to 0 hpa FINAL REPORT P. Otal 1, F. Boineau 1, N. Medina, D. Pražák 3, C. Wüthrich 4, S. Saxholm 5, W. Sabuga 6,
Water Vapour Effects in Mass Measurement
 Water Vapour Effects in Mass Measurement N.-E. Khélifa To cite this version: N.-E. Khélifa. Water Vapour Effects in Mass Measurement. Measurement. Water Vapour Effects in Mass Measurement, May 2007, Smolenice,
Water Vapour Effects in Mass Measurement N.-E. Khélifa To cite this version: N.-E. Khélifa. Water Vapour Effects in Mass Measurement. Measurement. Water Vapour Effects in Mass Measurement, May 2007, Smolenice,
DEVELOPMENT AND APPLICATION OF AN APPROACH TO ASSESS THE GLOBAL THERMAL EFFICIENCY OF A THERMAL ELECTRIC POWER PLANT
 DEVELOPMENT AND APPLICATION OF AN APPROACH TO ASSESS THE GLOBAL THERMAL EFFICIENCY OF A THERMAL ELECTRIC POWER PLANT Carlos Lueska, carlos@lueska.com.br CHP Consultoria de Energia S/C, São Paulo, SP, Brazil
DEVELOPMENT AND APPLICATION OF AN APPROACH TO ASSESS THE GLOBAL THERMAL EFFICIENCY OF A THERMAL ELECTRIC POWER PLANT Carlos Lueska, carlos@lueska.com.br CHP Consultoria de Energia S/C, São Paulo, SP, Brazil
I. Yang, C. H. Song, Y.-G. Kim & K. S. Gam
 Cryostat for Fixed-Point Calibration of Capsule-Type SPRTs I. Yang, C. H. Song, Y.-G. Kim & K. S. Gam International Journal of Thermophysics Journal of Thermophysical Properties and Thermophysics and Its
Cryostat for Fixed-Point Calibration of Capsule-Type SPRTs I. Yang, C. H. Song, Y.-G. Kim & K. S. Gam International Journal of Thermophysics Journal of Thermophysical Properties and Thermophysics and Its
Lab O2. Variation of Atmospheric Pressure with Altitude. We all live near the bottom of an ocean of air. At sea level, the weight of the air
 O2.1 Lab O2. Variation of Atmospheric Pressure with Altitude Introduction In this lab you will investigate the ideal gas law. By moving a plunger in a sealed container of air, you will find the variation
O2.1 Lab O2. Variation of Atmospheric Pressure with Altitude Introduction In this lab you will investigate the ideal gas law. By moving a plunger in a sealed container of air, you will find the variation
Compressible Gas Flow
 Compressible Gas Flow by Elizabeth Adolph Submitted to Dr. C. Grant Willson CHE53M Department of Chemical Engineering The University of Texas at Austin Fall 008 Compressible Gas Flow Abstract In this lab,
Compressible Gas Flow by Elizabeth Adolph Submitted to Dr. C. Grant Willson CHE53M Department of Chemical Engineering The University of Texas at Austin Fall 008 Compressible Gas Flow Abstract In this lab,
MASS DETERMINATION OF SILICON SPHERES USED FOR THE AVOGADRO PROJECT
 MASS DETERMINATION OF SILICON SPHERES USED FOR THE AVOGADRO PROJECT Michael Borys, Michael Gläser, Michael Mecke Physikalisch-Technische Bundesanstalt (PTB), Germany ABSTRACT Spheres made of a silicon
MASS DETERMINATION OF SILICON SPHERES USED FOR THE AVOGADRO PROJECT Michael Borys, Michael Gläser, Michael Mecke Physikalisch-Technische Bundesanstalt (PTB), Germany ABSTRACT Spheres made of a silicon
High pressure comparison among seven European national laboratories
 IMEKO 20 th TC3, 3 rd TC16 and 1 st TC22 International Conference Cultivating metrological knowledge 27 th to 30 th November, 2007. Merida, Mexico. High pressure comparison among seven European national
IMEKO 20 th TC3, 3 rd TC16 and 1 st TC22 International Conference Cultivating metrological knowledge 27 th to 30 th November, 2007. Merida, Mexico. High pressure comparison among seven European national
Physics 231 Topic 12: Temperature, Thermal Expansion, and Ideal Gases Alex Brown Nov
 Physics 231 Topic 12: Temperature, Thermal Expansion, and Ideal Gases Alex Brown Nov 18-23 2015 MSU Physics 231 Fall 2015 1 homework 3 rd midterm final Thursday 8-10 pm makeup Friday final 9-11 am MSU
Physics 231 Topic 12: Temperature, Thermal Expansion, and Ideal Gases Alex Brown Nov 18-23 2015 MSU Physics 231 Fall 2015 1 homework 3 rd midterm final Thursday 8-10 pm makeup Friday final 9-11 am MSU
FINAL REPORT. Key comparison EUROMET.M.P-K1.a. Euromet project 442 A. Pressure range: 0.1 Pa 1000 Pa. December 2004
 FINAL REPORT Key comparison EUROMET.M.P-K1.a Euromet proect 44 A Pressure range: 0.1 Pa 1000 Pa Anita Calcatelli 1, Fredrik Arrhen, Mercede Bergoglio 1, John Greenwood 3, Rifat Kangi 4, Karl Jousten 5,
FINAL REPORT Key comparison EUROMET.M.P-K1.a Euromet proect 44 A Pressure range: 0.1 Pa 1000 Pa Anita Calcatelli 1, Fredrik Arrhen, Mercede Bergoglio 1, John Greenwood 3, Rifat Kangi 4, Karl Jousten 5,
Downloaded from
 Chapter 13 (Kinetic Theory) Q1. A cubic vessel (with face horizontal + vertical) contains an ideal gas at NTP. The vessel is being carried by a rocket which is moving at a speed of500 ms in vertical direction.
Chapter 13 (Kinetic Theory) Q1. A cubic vessel (with face horizontal + vertical) contains an ideal gas at NTP. The vessel is being carried by a rocket which is moving at a speed of500 ms in vertical direction.
Key comparison BIPM.RI(I)-K7 of the air-kerma standards of the CMI, Czech Republic and the BIPM in mammography x-rays
 Key comparison BIPM.RI(I)-K7 of the air-kerma standards of the CMI, Czech Republic and the BIPM in mammography x-rays C Kessler 1, D T Burns 1, P Roger 1, V Sochor 2 1 Bureau International des Poids et
Key comparison BIPM.RI(I)-K7 of the air-kerma standards of the CMI, Czech Republic and the BIPM in mammography x-rays C Kessler 1, D T Burns 1, P Roger 1, V Sochor 2 1 Bureau International des Poids et
Ideal Gas Law and Absolute Zero
 Experiment IX Ideal Gas Law and Absolute Zero I. Purpose The purpose of this lab is to examine the relationship between the pressure, volume and temperature of air in a closed chamber. To do this, you
Experiment IX Ideal Gas Law and Absolute Zero I. Purpose The purpose of this lab is to examine the relationship between the pressure, volume and temperature of air in a closed chamber. To do this, you
Vacuum techniques (down to 1 K)
 Vacuum techniques (down to 1 K) For isolation (deep Knudsen regime) liquid helium dewar / inner vacuum jacket Leak testing at level 10-11 Pa m3/s (10-10 mbar l/s) liquid helium dewar & transfer syphon
Vacuum techniques (down to 1 K) For isolation (deep Knudsen regime) liquid helium dewar / inner vacuum jacket Leak testing at level 10-11 Pa m3/s (10-10 mbar l/s) liquid helium dewar & transfer syphon
A calibration facility for automatic weather stations
 METEOROLOGICAL APPLICATIONS Meteorol. Appl. 22: 842 846 (2015) Published online 16 November 2015 in Wiley Online Library (wileyonlinelibrary.com) DOI: 10.1002/met.1514 A calibration facility for automatic
METEOROLOGICAL APPLICATIONS Meteorol. Appl. 22: 842 846 (2015) Published online 16 November 2015 in Wiley Online Library (wileyonlinelibrary.com) DOI: 10.1002/met.1514 A calibration facility for automatic
METHODS TO CONFIRM THE MEASUREMENT CAPABILITY OF THE FORCE STANDARD MACHINES AFTER REINSTALLATION
 IMEKO 22 nd TC3, 12 th TC5 and 3 rd TC22 International Conferences 3 to 5 February, 2014, Cape Town, Republic of South Africa METHODS TO CONFIRM THE MEASUREMENT CAPABILITY OF THE FORCE STANDARD MACHINES
IMEKO 22 nd TC3, 12 th TC5 and 3 rd TC22 International Conferences 3 to 5 February, 2014, Cape Town, Republic of South Africa METHODS TO CONFIRM THE MEASUREMENT CAPABILITY OF THE FORCE STANDARD MACHINES
He II Heat transfer through a Corrugated Tube - Test Report
 He II Heat transfer through a Corrugated Tube - Test Report Authors: Ch. Darve, Y. Huang, T. Nicol, T. Peterson Keywords: LHC inner triplet, heat exchanger, He II heat transfer, Kapitza resistance. Abstract
He II Heat transfer through a Corrugated Tube - Test Report Authors: Ch. Darve, Y. Huang, T. Nicol, T. Peterson Keywords: LHC inner triplet, heat exchanger, He II heat transfer, Kapitza resistance. Abstract
Chapter 17. Temperature. Dr. Armen Kocharian
 Chapter 17 Temperature Dr. Armen Kocharian Temperature We associate the concept of temperature with how hot or cold an objects feels Our senses provide us with a qualitative indication of temperature Our
Chapter 17 Temperature Dr. Armen Kocharian Temperature We associate the concept of temperature with how hot or cold an objects feels Our senses provide us with a qualitative indication of temperature Our
Continued Work toward XHV for the Jefferson Lab Polarized Electron Source
 Continued Work toward XHV for the Jefferson Lab Polarized Electron Source Marcy Stutzman, Philip Adderley, Matt Poelker Thomas Jefferson National Accelerator Facility Newport News, VA 23601 Thomas Jefferson
Continued Work toward XHV for the Jefferson Lab Polarized Electron Source Marcy Stutzman, Philip Adderley, Matt Poelker Thomas Jefferson National Accelerator Facility Newport News, VA 23601 Thomas Jefferson
Chapter 11 Ideal gases
 OCR (A) specifications: 5.4.10c,d,e,i,j,k Chapter 11 Ideal gases Worksheet Worked examples Practical: Determining absolute zero of temperature from the pressure law End-of-chapter test Marking scheme:
OCR (A) specifications: 5.4.10c,d,e,i,j,k Chapter 11 Ideal gases Worksheet Worked examples Practical: Determining absolute zero of temperature from the pressure law End-of-chapter test Marking scheme:
Temperature and Length. Pieter Greeff October 2013
 Temperature and Length Pieter Greeff October 2013 Temperature and Length: Main Content Temperature Effects on Length and Form Measurements Motivation and ISO 1 CTE Uncertainty Temperature Measurement GBI
Temperature and Length Pieter Greeff October 2013 Temperature and Length: Main Content Temperature Effects on Length and Form Measurements Motivation and ISO 1 CTE Uncertainty Temperature Measurement GBI
CHAPTER 16 A MACROSCOPIC DESCRIPTION OF MATTER
 CHAPTER 16 A MACROSCOPIC DESCRIPTION OF MATTER This brief chapter provides an introduction to thermodynamics. The goal is to use phenomenological descriptions of the microscopic details of matter in order
CHAPTER 16 A MACROSCOPIC DESCRIPTION OF MATTER This brief chapter provides an introduction to thermodynamics. The goal is to use phenomenological descriptions of the microscopic details of matter in order
Uncertainty Evaluation of the New Setup for Measurement of Water-Vapor Permeation Rate by a Dew-Point Sensor
 Int J Thermophys (2012) 33:1595 1605 DOI 10.1007/s10765-012-1213-1 Uncertainty Evaluation of the New Setup for Measurement of Water-Vapor Permeation Rate by a Dew-Point Sensor D. Hudoklin J. Šetina J.
Int J Thermophys (2012) 33:1595 1605 DOI 10.1007/s10765-012-1213-1 Uncertainty Evaluation of the New Setup for Measurement of Water-Vapor Permeation Rate by a Dew-Point Sensor D. Hudoklin J. Šetina J.
Ideal Gas Law Experiment
 rev 05/2018 Ideal Gas Law Experiment Equipment List Qty Item Part number 1 Ideal Gas Law Apparatus TD-8596A 1 Pressure Sensor Absolute CI-6532A 1 Analog Adaptor Introduction The purpose of this lab is
rev 05/2018 Ideal Gas Law Experiment Equipment List Qty Item Part number 1 Ideal Gas Law Apparatus TD-8596A 1 Pressure Sensor Absolute CI-6532A 1 Analog Adaptor Introduction The purpose of this lab is
Experimental Methods and Analysis
 Chapter 3 28 Experimental Methods and Analysis 1. Equipment The fundamental basis of quantitative adsorption analysis is the measurement of excess adsorption isotherms. Each isotherm comprises a series
Chapter 3 28 Experimental Methods and Analysis 1. Equipment The fundamental basis of quantitative adsorption analysis is the measurement of excess adsorption isotherms. Each isotherm comprises a series
Demonstrating Competency and Equivalency of Two Commercial SPRT Calibration Facilities
 Demonstrating Competency and Equivalency of Two Commercial SPRT Calibration Facilities T. J. Wiandt 1,2 1 Fluke Corporation, Hart Scientific Division, American Fork, Utah United States. 2 E-mail: tom.wiandt@hartscientific.com
Demonstrating Competency and Equivalency of Two Commercial SPRT Calibration Facilities T. J. Wiandt 1,2 1 Fluke Corporation, Hart Scientific Division, American Fork, Utah United States. 2 E-mail: tom.wiandt@hartscientific.com
Final Report on Key Comparison EUROMET.M.P-K3.a in the Gauge Pressure Range 50 kpa to 1000 kpa. May 2005
 Final Report on Key Comparion EUROMET.M.P-K3.a in the Gauge Preure Range 50 kpa to 1000 kpa Pierre OTAL 1, Jo VERBEEK 2, Ditmar STEINDL 3, Jiri TESAR 4, Vladimir N. GOROBEI 5, Carmen MATILLA VINCENTE 6,
Final Report on Key Comparion EUROMET.M.P-K3.a in the Gauge Preure Range 50 kpa to 1000 kpa Pierre OTAL 1, Jo VERBEEK 2, Ditmar STEINDL 3, Jiri TESAR 4, Vladimir N. GOROBEI 5, Carmen MATILLA VINCENTE 6,
Final report Comparison Identifier: APMP.SIM.M.P-K1c Other designation: NIST-NPLI/Pressure/2
 Final report Comparison Identifier: APMP.SIM.M.P-K1c Other designation: NIST-NPLI/Pressure/2 Bilateral comparison between NIST (USA) and NPLI (India) in the pneumatic pressure region 0.4 MPa to 4.0 MPa
Final report Comparison Identifier: APMP.SIM.M.P-K1c Other designation: NIST-NPLI/Pressure/2 Bilateral comparison between NIST (USA) and NPLI (India) in the pneumatic pressure region 0.4 MPa to 4.0 MPa
Thermodynamic Properties of Low-GWP Refrigerant for Centrifugal Chiller
 Purdue University Purdue e-pubs International Refrigeration and Air Conditioning Conference School of Mechanical Engineering 2016 Thermodynamic Properties of Low-GWP Refrigerant for Centrifugal Chiller
Purdue University Purdue e-pubs International Refrigeration and Air Conditioning Conference School of Mechanical Engineering 2016 Thermodynamic Properties of Low-GWP Refrigerant for Centrifugal Chiller
A 3 Vapor pressure of volatile liquids
 Versuchsanleitungen zum Praktikum Physikalische Chemie für Anfänger 1 A 3 Vapor pressure of volatile liquids Purpose The purpose of this experiment is to determine the vaporization enthalpy, entropy and
Versuchsanleitungen zum Praktikum Physikalische Chemie für Anfänger 1 A 3 Vapor pressure of volatile liquids Purpose The purpose of this experiment is to determine the vaporization enthalpy, entropy and
gskin Application Note: Calorimetry
 1 / 7 gskin Application Note: Calorimetry Direct and precise calorimetric measurements with the gskin Sensor The gskin Heat Flux Sensor enables direct and highly precise measurements of reaction enthalpies
1 / 7 gskin Application Note: Calorimetry Direct and precise calorimetric measurements with the gskin Sensor The gskin Heat Flux Sensor enables direct and highly precise measurements of reaction enthalpies
Typical pressure measurement uncertainty defined by an FPG8601 Force Balanced Piston Gauge
 Typical pressure measurement uncertainty defined by an FPG8601 Force Balanced Piston Gauge 2090TN05 Revised 2004 March 26 Technical Note FPG8601 is a pressure standard designed to cover the range from
Typical pressure measurement uncertainty defined by an FPG8601 Force Balanced Piston Gauge 2090TN05 Revised 2004 March 26 Technical Note FPG8601 is a pressure standard designed to cover the range from
CHARACTERIZATION OF LPM'S 1-T DEW POINT GENERATOR
 XVIII IMEKO WORLD CONGRESS Metrology for a Sustainable Development September, 17 22, 2006, Rio de Janeiro, Brazil CHARACTERIZATION OF LPM'S 1-T DEW POINT GENERATOR Davor Zvizdic, Tomislav Stasic, Lovorka
XVIII IMEKO WORLD CONGRESS Metrology for a Sustainable Development September, 17 22, 2006, Rio de Janeiro, Brazil CHARACTERIZATION OF LPM'S 1-T DEW POINT GENERATOR Davor Zvizdic, Tomislav Stasic, Lovorka
Earlier Lecture. In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide.
 41 1 Earlier Lecture In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide. Silicon diodes have negligible i 2 R losses. Cernox RTDs offer high response
41 1 Earlier Lecture In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide. Silicon diodes have negligible i 2 R losses. Cernox RTDs offer high response
EXPERIMENT 3. HEAT-CAPACITY RATIOS FOR GASES
 EXERIMENT 3. HEAT-CAACITY RATIOS FOR GASES The ratio Cp/Cv of the heat capacity of a gas at constant pressure to that at constant volume will be determined by either the method of adiabatic expansion.
EXERIMENT 3. HEAT-CAACITY RATIOS FOR GASES The ratio Cp/Cv of the heat capacity of a gas at constant pressure to that at constant volume will be determined by either the method of adiabatic expansion.
Force Key Comparison CCM.F-K1.a and CCM.F-K1.b 5 kn and 10 kn. Aimo Pusa MIKES Finland
 Force Key Comparison CCM.F-K1.a and CCM.F-K1.b 5 kn and 10 kn Aimo Pusa 09.02.2009 MIKES Finland 2 Content Page Content 3 Foreword 4 Chapter 1 1.1 General 6 1.2 Characteristics of the transducers 8 1.3
Force Key Comparison CCM.F-K1.a and CCM.F-K1.b 5 kn and 10 kn Aimo Pusa 09.02.2009 MIKES Finland 2 Content Page Content 3 Foreword 4 Chapter 1 1.1 General 6 1.2 Characteristics of the transducers 8 1.3
Review of Anemometer Calibration Standards
 Review of Anemometer Calibration Standards Rachael V. Coquilla rvcoquilla@otechwind.com Otech Engineering, Inc., Davis, CA Anemometer calibration defines a relationship between the measured signals from
Review of Anemometer Calibration Standards Rachael V. Coquilla rvcoquilla@otechwind.com Otech Engineering, Inc., Davis, CA Anemometer calibration defines a relationship between the measured signals from
Introduction to Blackbody Sources
 Introduction to s This section contains dedicated blackbody sources for low uncertainty calibration of infrared thermometers. A range of portable primary blackbody sources combine high emissivity with
Introduction to s This section contains dedicated blackbody sources for low uncertainty calibration of infrared thermometers. A range of portable primary blackbody sources combine high emissivity with
Final Report on Key Comparison EURAMET.M.P-K13 in the Range 50 MPa to 500 MPa of Hydraulic Gauge Pressure
 Final Report on Key Comparison EURAMET.M.P-K13 in the Range 50 MPa to 500 MPa of Hydraulic Gauge Pressure I. Kocas 1, W. Sabuga 2, M. Bergoglio 3, A. Eltaweel 4, C. Korasie 5, P. Farar 6, J. Setina 7,
Final Report on Key Comparison EURAMET.M.P-K13 in the Range 50 MPa to 500 MPa of Hydraulic Gauge Pressure I. Kocas 1, W. Sabuga 2, M. Bergoglio 3, A. Eltaweel 4, C. Korasie 5, P. Farar 6, J. Setina 7,
Certification of a High Capacity Force Machine for Testing of Load Cells According to OIML R60
 Certification of a High Capacity Force Machine for Testing of Load Cells According to OIML R60 Bernd Meißner Physikalisch-Technische Bundesanstalt Weighing Instruments Laboratory Braunschweig Abstract
Certification of a High Capacity Force Machine for Testing of Load Cells According to OIML R60 Bernd Meißner Physikalisch-Technische Bundesanstalt Weighing Instruments Laboratory Braunschweig Abstract
Thermodynamics. Atoms are in constant motion, which increases with temperature.
 Thermodynamics SOME DEFINITIONS: THERMO related to heat DYNAMICS the study of motion SYSTEM an object or set of objects ENVIRONMENT the rest of the universe MICROSCOPIC at an atomic or molecular level
Thermodynamics SOME DEFINITIONS: THERMO related to heat DYNAMICS the study of motion SYSTEM an object or set of objects ENVIRONMENT the rest of the universe MICROSCOPIC at an atomic or molecular level
Vacuum. Kai Schwarzwälder, Institut für Physik Universität Basel October 6 th 2006
 Physics,, Technology and Techniques of the Vacuum Kai Schwarzwälder, Institut für Physik Universität Basel October 6 th 2006 Outline Introduction and basics Defintion of Vacuum Vacuum A vacuum is a volume
Physics,, Technology and Techniques of the Vacuum Kai Schwarzwälder, Institut für Physik Universität Basel October 6 th 2006 Outline Introduction and basics Defintion of Vacuum Vacuum A vacuum is a volume
Gas Thermometer and Absolute Zero
 Chapter 1 Gas Thermometer and Absolute Zero Name: Lab Partner: Section: 1.1 Purpose Construct a temperature scale and determine absolute zero temperature (the temperature at which molecular motion ceases).
Chapter 1 Gas Thermometer and Absolute Zero Name: Lab Partner: Section: 1.1 Purpose Construct a temperature scale and determine absolute zero temperature (the temperature at which molecular motion ceases).
Temperature and Length
 Temperature and Length Speaker / Author: Pieter Greeff NMISA Private Bag X34, Lynnwood Ridge, Pretoria, 0040, RSA e-mail: pgreeff@nmisa.org Phone: 012 841 2299 Abstract The environment, especially temperature,
Temperature and Length Speaker / Author: Pieter Greeff NMISA Private Bag X34, Lynnwood Ridge, Pretoria, 0040, RSA e-mail: pgreeff@nmisa.org Phone: 012 841 2299 Abstract The environment, especially temperature,
Chapter Four. Experimental
 Chapter Four 4.1 Materials N,N-Diethyl monoethanolamine (purity 98%) used in all experiments was purchased from Spectrochem Pvt. Ltd., Mumbai. N-Ethyl monoethanolamine, N-(- aminoethyl)ethanolamine, diethanolamine,
Chapter Four 4.1 Materials N,N-Diethyl monoethanolamine (purity 98%) used in all experiments was purchased from Spectrochem Pvt. Ltd., Mumbai. N-Ethyl monoethanolamine, N-(- aminoethyl)ethanolamine, diethanolamine,
K n. III. Gas flow. 1. The nature of the gas : Knudsen s number. 2. Relative flow : Reynold s number R = ( dimensionless )
 III. Gas flow. The nature of the gas : Knudsen s number K n λ d 2. Relative flow : U ρ d η U : stream velocity ρ : mass density Reynold s number R ( dimensionless ) 3. Flow regions - turbulent : R > 2200
III. Gas flow. The nature of the gas : Knudsen s number K n λ d 2. Relative flow : U ρ d η U : stream velocity ρ : mass density Reynold s number R ( dimensionless ) 3. Flow regions - turbulent : R > 2200
VACUUM PUMPING METHODS
 VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
Automated volume measurement for weihts using acoustic volumeter
 IMEKO 20th TC3, 3rd TC16 and 1st TC22 International Conference Cultivating metrological knowledge 27th to 30th November, 2007. Merida, Mexico. Automated volume measurement for weihts using acoustic volumeter
IMEKO 20th TC3, 3rd TC16 and 1st TC22 International Conference Cultivating metrological knowledge 27th to 30th November, 2007. Merida, Mexico. Automated volume measurement for weihts using acoustic volumeter
Experiment B6 Thermal Properties of Materials Procedure
 Experiment B6 Thermal Properties of Materials Procedure Deliverables: Checked lab notebook, Brief technical memo Overview In this lab, you will examine the thermal properties of various materials commonly
Experiment B6 Thermal Properties of Materials Procedure Deliverables: Checked lab notebook, Brief technical memo Overview In this lab, you will examine the thermal properties of various materials commonly
A thermodynamic system is taken from an initial state X along the path XYZX as shown in the PV-diagram.
 AP Physics Multiple Choice Practice Thermodynamics 1. The maximum efficiency of a heat engine that operates between temperatures of 1500 K in the firing chamber and 600 K in the exhaust chamber is most
AP Physics Multiple Choice Practice Thermodynamics 1. The maximum efficiency of a heat engine that operates between temperatures of 1500 K in the firing chamber and 600 K in the exhaust chamber is most
VSMOW Triple Point of Water Cells: Borosilicate versus Fused- Quartz
 VSMOW Triple Point of Water Cells: Borosilicate versus Fused- Quartz M. Zhao 1,3 and G. F. Strouse 2 1 Fluke Corporation, Hart Scientific Division, American Fork, Utah 84003, U.S.A. 2 National Institute
VSMOW Triple Point of Water Cells: Borosilicate versus Fused- Quartz M. Zhao 1,3 and G. F. Strouse 2 1 Fluke Corporation, Hart Scientific Division, American Fork, Utah 84003, U.S.A. 2 National Institute
 Real Gases 1. The value of compressibility factor for one mole of a gas under critical states is 1) 3/8 2) 2/3 3) 8/27 4) 27/8 2. an der Waal s equation for one mole of CO2 gas at low pressure will be
Real Gases 1. The value of compressibility factor for one mole of a gas under critical states is 1) 3/8 2) 2/3 3) 8/27 4) 27/8 2. an der Waal s equation for one mole of CO2 gas at low pressure will be
(2) The volume of molecules is negligible in comparison to the volume of gas. (3) Molecules of a gas moves randomly in all direction.
 9.1 Kinetic Theory of Gases : Assumption (1) The molecules of a gas are identical, spherical and perfectly elastic point masses. (2) The volume of molecules is negligible in comparison to the volume of
9.1 Kinetic Theory of Gases : Assumption (1) The molecules of a gas are identical, spherical and perfectly elastic point masses. (2) The volume of molecules is negligible in comparison to the volume of
INTRODUCTION Strained Silicon Monochromator Magnesium Housing Windows for Monochromator Shutter and Collimator Fission Detector HOPG Monochromator
 Design for a Four-Blade Neutron Interferometer INTRODUCTION Strained Silicon Monochromator The neutron beam used for this interferometer is separated from the NIST reactor's main beam using a strained
Design for a Four-Blade Neutron Interferometer INTRODUCTION Strained Silicon Monochromator The neutron beam used for this interferometer is separated from the NIST reactor's main beam using a strained
Gas Volumes and the Ideal Gas Law
 Section 3, 9B s Gases react in whole-number ratios. Equal volumes of gases under the same conditions contain equal numbers of molecules. All gases have a volume of 22.4 L under standard conditions. In
Section 3, 9B s Gases react in whole-number ratios. Equal volumes of gases under the same conditions contain equal numbers of molecules. All gases have a volume of 22.4 L under standard conditions. In
Intercomparison of Thermal Expansion Measurements. EUROMET Project 275. Final Report
 Eidgenössisches Amt für Messwesen Office fédéral de métrologie Ufficio federale di metrologia Swiss Federal Office of Metrology Intercomparison of Thermal Expansion Measurements EUROMET Project 75 Final
Eidgenössisches Amt für Messwesen Office fédéral de métrologie Ufficio federale di metrologia Swiss Federal Office of Metrology Intercomparison of Thermal Expansion Measurements EUROMET Project 75 Final
Calibration capabilities at PTB for radiation thermometry, quantitative thermography and emissivity
 14 th Quantitative InfraRed Thermography Conference Calibration capabilities at PTB for radiation thermometry, quantitative thermography and emissivity by I. Müller*, A. Adibekyan*, B. Gutschwager*, E.
14 th Quantitative InfraRed Thermography Conference Calibration capabilities at PTB for radiation thermometry, quantitative thermography and emissivity by I. Müller*, A. Adibekyan*, B. Gutschwager*, E.
COMPLETION AND MEASUREMENT UNCERTAINTY BUDGET OF THE MULTI-COMPONENT MEASURING DEVISE FOR FORCE UP TO 1 MN AND TORQUE UP TO 2 KN M
 XXI IMEKO World Congress Measurement in Research and Industry August 30 September 4, 2015, Prague, Czech Republic COMPLETION AND MEASUREMENT UNCERTAINTY BUDGET OF THE MULTI-COMPONENT MEASURING DEVISE FOR
XXI IMEKO World Congress Measurement in Research and Industry August 30 September 4, 2015, Prague, Czech Republic COMPLETION AND MEASUREMENT UNCERTAINTY BUDGET OF THE MULTI-COMPONENT MEASURING DEVISE FOR
A comparison of primary platinum-iridium kilogram mass standards among eighteen European NMIs
 IMEKO 20 th TC3, 3 rd TC16 and 1 st TC22 International Conference Cultivating metrological knowledge 27 th to 30 th November, 2007. Merida, Mexico. A comparison of primary platinum-iridium kilogram mass
IMEKO 20 th TC3, 3 rd TC16 and 1 st TC22 International Conference Cultivating metrological knowledge 27 th to 30 th November, 2007. Merida, Mexico. A comparison of primary platinum-iridium kilogram mass
Application of the Multi-current Transient Hot-Wire Technique for Absolute Measurements of the Thermal Conductivity of Glycols
 International Journal of Thermophysics, Vol. 26, No. 3, May 2005 ( 2005) DOI: 10.1007/s10765-005-5568-4 Application of the Multi-current Transient Hot-Wire Technique for Absolute Measurements of the Thermal
International Journal of Thermophysics, Vol. 26, No. 3, May 2005 ( 2005) DOI: 10.1007/s10765-005-5568-4 Application of the Multi-current Transient Hot-Wire Technique for Absolute Measurements of the Thermal
Theory (NOTE: This theory is the same that we covered before in Experiment 11on the Ideal Gas model)
 Experiment 12 CHARLES LAW Objectives 1. To set up a model of thermal machine, 2. To put to work the model to verify Charles law, 3. To describe and explain Charles law Theory (NOTE: This theory is the
Experiment 12 CHARLES LAW Objectives 1. To set up a model of thermal machine, 2. To put to work the model to verify Charles law, 3. To describe and explain Charles law Theory (NOTE: This theory is the
Thermal Treatment of Stainless Steel towards the Zero Outgassing Rate
 Thermal Treatment of Stainless Steel towards the Zero Outgassing Rate Vincenc Nemanic, Institute of Surface Engineering and Optoelectronics, Teslova 30, 1000 Ljubljana, Slovenia vincenc.nemanic@guest.arnes.si
Thermal Treatment of Stainless Steel towards the Zero Outgassing Rate Vincenc Nemanic, Institute of Surface Engineering and Optoelectronics, Teslova 30, 1000 Ljubljana, Slovenia vincenc.nemanic@guest.arnes.si
Absolute differential cross sections for electron elastic scattering and vibrational excitation in nitrogen in the angular range from 120 to 180
 J. Phys. B: At. Mol. Opt. Phys. 33 (2000) L527 L532. Printed in the UK PII: S0953-4075(00)50902-X LETTER TO THE EDITOR Absolute differential cross sections for electron elastic scattering and vibrational
J. Phys. B: At. Mol. Opt. Phys. 33 (2000) L527 L532. Printed in the UK PII: S0953-4075(00)50902-X LETTER TO THE EDITOR Absolute differential cross sections for electron elastic scattering and vibrational
Gas Volumes and the Ideal Gas Law
 SECTION 11.3 Gas Volumes and the Ideal Gas Law Section 2 presented laws that describe the relationship between the pressure, temperature, and volume of a gas. The volume of a gas is also related to the
SECTION 11.3 Gas Volumes and the Ideal Gas Law Section 2 presented laws that describe the relationship between the pressure, temperature, and volume of a gas. The volume of a gas is also related to the
Commissioning of the Beta Secondary Standard (BSS2)
 Commissioning of the Beta Secondary Standard (BSS2) Speaker / Author: R.W. Thoka* Co-author: S. Jozela* * National Metrology Institute of South Africa (NMISA), Private Bag X 34, Lynnwood Ridge, Pretoria,
Commissioning of the Beta Secondary Standard (BSS2) Speaker / Author: R.W. Thoka* Co-author: S. Jozela* * National Metrology Institute of South Africa (NMISA), Private Bag X 34, Lynnwood Ridge, Pretoria,
Rocket propulsion Prof. K. Ramamurthi Department of Mechanical Engineering Indian Institute of Technology, Madras. Lecture 09 Theory of Nozzles
 Rocket propulsion Prof. K. Ramamurthi Department of Mechanical Engineering Indian Institute of Technology, Madras Lecture 09 Theory of Nozzles (Refer Slide Time: 00:14) Good morning. We will develop the
Rocket propulsion Prof. K. Ramamurthi Department of Mechanical Engineering Indian Institute of Technology, Madras Lecture 09 Theory of Nozzles (Refer Slide Time: 00:14) Good morning. We will develop the
Evaluation of the Effect of Relative Humidity of Air on the Coefficients of Critical Flow Venturi Nozzles
 Evaluation of the Effect of Relative Humidity of Air on the Coefficients of Critical Flow Venturi Nozzles K. Chahine and M. Ballico National Measurement Institute, Australia P O Box 264, Lindfield, NSW
Evaluation of the Effect of Relative Humidity of Air on the Coefficients of Critical Flow Venturi Nozzles K. Chahine and M. Ballico National Measurement Institute, Australia P O Box 264, Lindfield, NSW
Anomalous production of gaseous 4 He at the inside of DScathode during D 2 O-electrolysis
 Arata, Y. and Y.C. Zhang, Anomalous production of gaseous 4 He at the inside of 'DS cathode' during D 2 O- electrolysis. Proc. Jpn. Acad., Ser. B, 1999. 75: p. 281. Anomalous production of gaseous 4 He
Arata, Y. and Y.C. Zhang, Anomalous production of gaseous 4 He at the inside of 'DS cathode' during D 2 O- electrolysis. Proc. Jpn. Acad., Ser. B, 1999. 75: p. 281. Anomalous production of gaseous 4 He
Revision Guide for Chapter 13
 Matter: very simple Revision Guide for Chapter Contents Student s Checklist Revision Notes Ideal gas... Ideal gas laws... Assumptions of kinetic theory of gases... 5 Internal energy... 6 Specific thermal
Matter: very simple Revision Guide for Chapter Contents Student s Checklist Revision Notes Ideal gas... Ideal gas laws... Assumptions of kinetic theory of gases... 5 Internal energy... 6 Specific thermal
I. TEMPERATURE DEPENDENCE OF VAPOUR PRESSURE. THEORETICAL INTRODUCTION.
 25 DETERMINATION OF THE VAPOUR PRESSURE OF A PURE LIQUID SUBSTANCE BY THE SIMPLE STATIC METHOD AS A FUNCTION OF TEMPERATURE. DETERMINATION OF THE ENTHALPY CHANGE OF EVAPORATION. The necessary theoretical
25 DETERMINATION OF THE VAPOUR PRESSURE OF A PURE LIQUID SUBSTANCE BY THE SIMPLE STATIC METHOD AS A FUNCTION OF TEMPERATURE. DETERMINATION OF THE ENTHALPY CHANGE OF EVAPORATION. The necessary theoretical
Comparison of V and 10 V DC Voltage References
 Comparison of 1.018 V and 10 V DC Voltage References Technical protocol for BIPM.EM-K11.a and.b comparisons 1. Introduction The Mutual Recognition Arrangement among National Metrology Institutes (NMIs)
Comparison of 1.018 V and 10 V DC Voltage References Technical protocol for BIPM.EM-K11.a and.b comparisons 1. Introduction The Mutual Recognition Arrangement among National Metrology Institutes (NMIs)
Final Results of Bilateral Comparison between NIST and PTB for Flows of High Pressure Natural Gas
 Final Results of Bilateral Comparison between NIST and for Flows of High Pressure Natural Gas B. Mickan (, Germany, Bodo.Mickan@ptb.de) H. Toebben (pigsar TM, Germany) A. Johnson (NIST,USA, Aaron.Johnnson@nist.gov)
Final Results of Bilateral Comparison between NIST and for Flows of High Pressure Natural Gas B. Mickan (, Germany, Bodo.Mickan@ptb.de) H. Toebben (pigsar TM, Germany) A. Johnson (NIST,USA, Aaron.Johnnson@nist.gov)
CBE 450 Chemical Reactor Fundamentals Fall, 2009 Homework Assignment #1
 CBE 45 Chemical Reactor Fundamentals Fall, 9 omework Assignment #1 1. Review of Mass Balances of Reacting Systems atural gas (assume pure methane) is being burned in air (assume.79 mol% and.1 mol% O ).
CBE 45 Chemical Reactor Fundamentals Fall, 9 omework Assignment #1 1. Review of Mass Balances of Reacting Systems atural gas (assume pure methane) is being burned in air (assume.79 mol% and.1 mol% O ).
North Sea Flow Measurement Workshop
 North Sea Flow Measurement Workshop October 22, 2018 - October 24, 2018 Ardoe House Hotel, Blairs, Aberdeen United Kingdom P. Alberto Giuliano Albo, Simona Lago and Giuseppe Cavuoto Total net energy transferred
North Sea Flow Measurement Workshop October 22, 2018 - October 24, 2018 Ardoe House Hotel, Blairs, Aberdeen United Kingdom P. Alberto Giuliano Albo, Simona Lago and Giuseppe Cavuoto Total net energy transferred
A TEST OF THE PRECIPITATION AMOUNT AND INTENSITY MEASUREMENTS WITH THE OTT PLUVIO
 A TEST OF THE PRECIPITATION AMOUNT AND INTENSITY MEASUREMENTS WITH THE OTT PLUVIO Wiel M.F. Wauben, Instrumental Department, Royal Netherlands Meteorological Institute (KNMI) P.O. Box 201, 3730 AE De Bilt,
A TEST OF THE PRECIPITATION AMOUNT AND INTENSITY MEASUREMENTS WITH THE OTT PLUVIO Wiel M.F. Wauben, Instrumental Department, Royal Netherlands Meteorological Institute (KNMI) P.O. Box 201, 3730 AE De Bilt,
