Thermodynamics Test Clio Invitational January 26, 2013
|
|
|
- Christal Hancock
- 6 years ago
- Views:
Transcription
1 Thermodynamics Test Clio Invitational January 26, 2013 School Name: Team Number: Variables specified: s = specific heat C = heat capacity H f = heat of fusion H v = heat of vaporization Given information: Substance s (J/g C) Carbon (graphite) Granite Iron Lead Silver Water (ice) 2.06 Water (steam) 2.02 Water (liquid) 4.18 Substance H f (J/g) H v (J/g) Water Answer the following questions to the best of your ability using the information provided. Put your school name and team number on BOTH your test booklet AND answer key. Please fill in ALL of your answers on the answer key to be graded.
2 1. The heat capacity of a 6 kg bar of metal is 2.01 kj/ C. What is the specific heat of the metal in units cal/g C? a b c d How many calories are required to raise the temperature of g of water by 100 C? a x 10 3 b x 10 4 c d Water was heated in a pot on a stove from 55 F to 212 F. Calculate the final temperature of the water in Kelvin. a. 70 b. 100 c. 13 d The specific heat of 50 g of copper is J/g C. What is the specific heat of 175 g of copper in J/g C? a b c d How many Calories are in 900 kj? a x 10 2 b. 215 x 10 3 c. 215 d x You were given a 15 g sample of an unknown substance at 85 C and added it to a Styrofoam cup holding 400 g of water at 20 C. The final system temperature was 24 C. Which unknown substance were you given? a. Substance A: s = 7.31 J/g C b. Substance B: s = 0.36 J/g C c. Substance C: s = 4.18 J/g C d. Substance D: s = 5.25 J/g C 7. If 7 g of water at 2 C is mixed with 14 g of water at 21 C, what is the final temperature of the mixture? a. 10 C
3 b. 15 C c. 20 C d. 25 C 8. Suppose object A has two times the specific heat and two times the mass of object B. If the same amount of heat is applied to both objects, how will the temperature change of A be related to the temperature change of B? a. 2 T A = T B b. T A = T B c. T A = 4 T B d. 4 T A = T B 9. The quantity of heat that will raise the temperature of 1 g of a substance by 1 C is: a. Heat capacity b. Specific heat c. Molar heat capacity d. Work 10. If a system performs 60 J of work and receives 30 J of heat, what is the E for this change? a. 30 J b. 90 J c. -90 J d. -30 J Use the following information for questions A 15 g mass of metal was heated to 95 C and dumped into 100 g of water at 30 C. The temperature of the water increased to 32 C. 11. How many joules did the water absorb? a. 945 b. 836 c d How many joules did the metal lose? a. 945 b. 836 c d What is the heat capacity of the metal in J/ C? a b
4 c d What is the specific heat of the metal in J/g C? a b c d What is the meaning of a positive value for heat? a. Substance experienced a fall in temperature. b. The mass of the substance was very large. c. The heat was gained by the substance from the surroundings. d. The specific heat of the substance was very small. 16. What is the SI unit for energy? a. kilocalorie b. calorie c. joule d. kilojoule 17. What equation does the First Law of Thermodynamics define? a. q = ms T b. U = q + w c. H = H products - H reactants d. T = T f - T i 18. How are heat capacity and specific heat related? a. C = ms T b. C = s/ T c. C = ms d. C = m T 19. A container holding 5.00 kg of water experienced a decrease in temperature from C to C. How much energy has left the water in kj? a b c d For questions 20-23: The temperature of kg of water was raised from 216K to 240K. How much energy was transferred into the water in: 20. joules?
5 a b c d kilojoules? a b c d calories? a b c d kilocalories? a b c d For an exothermic reaction, H is: a. Positive b. Negative c. Not enough information 25. Calculate the molar heat capacity of silver in J/mol C. a b c d A gallon bottle of water with a temperature of 90 C is packaged with a gallon bottle of water at a temperature of 40 C. If no heat escapes to the surroundings, what will the final temperature of each bottle of water be at equilibrium? a. 50 C b. 55 C c. 60 C d. 65 C 27. What is the symbol for heat? a. w
6 b. E c. Q d. C 28. How much heat (in J) is released by 35 g of water if its temperature is decreased from 90 C to 20 C? a b c d True or false: at the site of a state change on a heat curve, heat is applied to the system, but the temperature does not increase. a. true b. false 30. Calculate the energy needed to heat 3 g of ice at -10 C to steam at 150 C. a J b J c J d J 31. Water boils at 100 C with a molar heat of vaporization of kj. What is the entropy change when: H2O (g) H2O (l) at 100 C? a. insufficient information. b J/K c J/K d J/K 32. Which of the following describes a system that CANNOT be spontaneous? a. ΔH is positive and ΔS is negative. b. ΔH is positive and ΔS is positive. c. ΔH is negative and ΔS is negative. d. ΔH is negative and ΔS is positive. 33. Given the following thermochemical data: N 2 O 4 (g) 2NO 2 (g) ΔH = kj 2NO (g) + O 2 (g) 2NO 2 (g) ΔH = kj Determine the heat of reaction for: 2NO (g) + O 2 (g) N 2 O 4 (g) a kj
7 b kj c kj d kj 34. If 100mL of water at 50 C are put over a low flame for two minutes a. the average velocity of the particles will increase. b. the temperature will increase. c. the total heat energy of the sample will increase. d. all of the above will occur. 35. Which of the following does not represent an increase in entropy? a. a gas changing into a liquid b. a solid changing into a liquid c. a solute dissolving in water d. water being heated 36. Which of the following is not involved in the calculation of heat absorbed as a substance melts? a. change in temperature b. heat of fusion of the substance c. mass of the substance 37. A process that absorbs heat from its surroundings is said to be a. endothermic b. exothermic c. thermochemical d. none of the above 38. Given the equation 2Mg (s) + O2 (g) 2MgO (s) kj, which of the following is true? a. H = kj b. H = kj c. the reaction is endothermic d. the system is drawing heat from the surroundings 39. Given the equation mentioned in the above question, how much heat would be involved in the formation of 4.0 mol of MgO (s)? a kj b. 180 kj c. 36 kj d. 72 kj 40. A metal weighing 50.0 g absorbs J of heat when its temperature increases by C. What is the specific heat of the metal in J/g C?
8 a b c d In which of the following states does a given substance have the highest entropy? a. gas b. liquid c. solid d. the entropy for these states are all the same 42. In which of these systems is the entropy decreasing? a. a gas condensing to a liquid b. a solid decomposing into a liquid and a gas c. salt dissolving in water d. snow melting 43. The characteristics that make a reaction most likely be spontaneous are a. - H, - S b. - H, + S c. + H, - S d. + H, + S 44. A reaction is most likely to be spontaneous when during the course of a reaction: a. the energy content and the entropy decreases b. the energy content and the entropy increase c. the energy content decrease and the entropy increases d. the energy content increase and the entropy decreases 45. Doing which of the following generally increases the entropy of a substance? a. Freezing it b. Dissolving it in water c. Condensing it d. All of the above 46. How much heat is released when 108 g of water at 0 C freezes to ice at 0 C? a kj b kj c. 649 kj d kj 47. The temperature of an 8.0 g sample of metal changed from 25 C to 50 C when it absorbed 420 J of heat. What is the specific heat of the metal (in J/g C)? a. 130
9 b. 2.1 c d At room temperature, a reaction is not spontaneous. At an elevated temperature, however, the reaction is spontaneous. Which of the following describes the system? ΔS ΔH a. positive positive b. positive negative c. negative positive d. negative negative 49. Two water-based solutions are mixed and a reaction occurs. The temperature of the water-based solutions increases from 25 C to 27 C. Given that the total mass of the water is 75 grams, what quantity of heat was generated in this reaction to cause the temperature change? a. 125 J b. 314 J c J d. 627 J 50. Convert 476 K to Fahrenheit. a. 203 F b. 397 F c. 365 F d. 95 F
CHAPTER 17 Thermochemistry
 CHAPTER 17 Thermochemistry Thermochemistry The study of the heat changes that occur during chemical reactions and physical changes of state. Chemical Change: new substances created during chemical reaction
CHAPTER 17 Thermochemistry Thermochemistry The study of the heat changes that occur during chemical reactions and physical changes of state. Chemical Change: new substances created during chemical reaction
Chemistry Heat Review. Heat: Temperature: Enthalpy: Calorimetry: Activation energy:
 Chemistry Heat Review Name Date Vocabulary Heat: Temperature: Enthalpy: Calorimetry: Activation energy: Formulas Heat of phase change Heat for temperature increase Heat of reaction Endothermic/Exothermic
Chemistry Heat Review Name Date Vocabulary Heat: Temperature: Enthalpy: Calorimetry: Activation energy: Formulas Heat of phase change Heat for temperature increase Heat of reaction Endothermic/Exothermic
Topic 5: Energetics. Heat & Calorimetry. Thursday, March 22, 2012
 Topic 5: Energetics Heat & Calorimetry 1 Heat is energy that is transferred from one object to another due to a difference in temperature Temperature is a measure of the average kinetic energy of a body
Topic 5: Energetics Heat & Calorimetry 1 Heat is energy that is transferred from one object to another due to a difference in temperature Temperature is a measure of the average kinetic energy of a body
Ch. 17 Thermochemistry
 Ch. 17 Thermochemistry 17.1 The Flow of Energy Energy Transformations Thermochemistry: study of energy changes in chemical reactions and changes in state Chemical potential energy: energy stored in bonds
Ch. 17 Thermochemistry 17.1 The Flow of Energy Energy Transformations Thermochemistry: study of energy changes in chemical reactions and changes in state Chemical potential energy: energy stored in bonds
I. The Nature of Energy A. Energy
 I. The Nature of Energy A. Energy is the ability to do work or produce heat. It exists in 2 forms: 1. Potential energy is energy due to the composition or position of an object. 2. Kinetic energy is energy
I. The Nature of Energy A. Energy is the ability to do work or produce heat. It exists in 2 forms: 1. Potential energy is energy due to the composition or position of an object. 2. Kinetic energy is energy
Unit 7 Kinetics and Thermodynamics
 17.1 The Flow of Energy Heat and Work Unit 7 Kinetics and Thermodynamics I. Energy Transformations A. Temperature 1. A measure of the average kinetic energy of the particles in a sample of matter B. Heat
17.1 The Flow of Energy Heat and Work Unit 7 Kinetics and Thermodynamics I. Energy Transformations A. Temperature 1. A measure of the average kinetic energy of the particles in a sample of matter B. Heat
Name: Thermochemistry. Practice Test C. General Chemistry Honors Chemistry
 Name: Thermochemistry C Practice Test C General Chemistry Honors Chemistry 1 Objective 1: Use the relationship between mass, specific heat, and temperature change to calculate the heat flow during a chemical
Name: Thermochemistry C Practice Test C General Chemistry Honors Chemistry 1 Objective 1: Use the relationship between mass, specific heat, and temperature change to calculate the heat flow during a chemical
Thermodynamics C Test
 Northern Regional: January 19 th, 2019 Thermodynamics C Test Name(s): Team Name: School Name: Team Number: Rank: Score: Science Olympiad North Florida Regional at the University of Florida Thermodynamics
Northern Regional: January 19 th, 2019 Thermodynamics C Test Name(s): Team Name: School Name: Team Number: Rank: Score: Science Olympiad North Florida Regional at the University of Florida Thermodynamics
Energy and Energy Calculations Test Provide the correct answer as a word, phrase or sentence. (3 points each) 1) Define Matter.
 Provide the correct answer as a word, phrase or sentence. (3 points each) 1) Define Matter. 2) What is ENERGY? 3) Give an example of an endothermic process. 4) Give an example of an exothermic process.
Provide the correct answer as a word, phrase or sentence. (3 points each) 1) Define Matter. 2) What is ENERGY? 3) Give an example of an endothermic process. 4) Give an example of an exothermic process.
Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy
 Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes
 Thermochemistry Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes heat flows from high to low (hot cool) endothermic reactions: absorb energy
Thermochemistry Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes heat flows from high to low (hot cool) endothermic reactions: absorb energy
Name: Class: Date: ID: A
 Name: Class: _ Date: _ ID: A Chpter 17 review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of these phase changes is an endothermic process? a.
Name: Class: _ Date: _ ID: A Chpter 17 review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of these phase changes is an endothermic process? a.
Heat. Heat Terminology 04/12/2017. System Definitions. System Definitions
 System Definitions Heat Physical Science 20 Ms. Hayduk Heat Terminology System: the part of the universe being studied (big Earth, or small one atom) Surroundings: the part of the universe outside the
System Definitions Heat Physical Science 20 Ms. Hayduk Heat Terminology System: the part of the universe being studied (big Earth, or small one atom) Surroundings: the part of the universe outside the
Temperature and Its Measurement
 Temperature and Its Measurement When the physical properties are no longer changing, the objects are said to be in thermal equilibrium. Two or more objects in thermal equilibrium have the same temperature.
Temperature and Its Measurement When the physical properties are no longer changing, the objects are said to be in thermal equilibrium. Two or more objects in thermal equilibrium have the same temperature.
Thermochemistry. Energy and Chemical Change
 Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
CHAPTER 17: THERMOCHEMISTRY. Mrs. Brayfield
 CHAPTER 17: THERMOCHEMISTRY Mrs. Brayfield REVIEW What is the law of conservation of energy? It states that energy cannot be created or destroyed So the energy of any process is the same THERMOCHEMISTRY
CHAPTER 17: THERMOCHEMISTRY Mrs. Brayfield REVIEW What is the law of conservation of energy? It states that energy cannot be created or destroyed So the energy of any process is the same THERMOCHEMISTRY
Name Class Date. As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings.
 Name Class Date Thermochemistry 17.1 The Flow of Energy As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings. Process Cause Effect endothermic
Name Class Date Thermochemistry 17.1 The Flow of Energy As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings. Process Cause Effect endothermic
Chapter 11. Thermochemistry. 1. Let s begin by previewing the chapter (Page 292). 2. We will partner read Pages
 Chapter 11 Thermochemistry 1. Let s begin by previewing the chapter (Page 292). 2. We will partner read Pages 293-94 The Flow of energy - heat Thermochemistry concerned with the heat changes that occur
Chapter 11 Thermochemistry 1. Let s begin by previewing the chapter (Page 292). 2. We will partner read Pages 293-94 The Flow of energy - heat Thermochemistry concerned with the heat changes that occur
Matter and Energy Review Packet
 Name Date Matter and Energy Review Packet 1. A compound differs from a mixture in that a compound always has a (1) homogeneous composition (2) maximum of two components (3) minimum of three components
Name Date Matter and Energy Review Packet 1. A compound differs from a mixture in that a compound always has a (1) homogeneous composition (2) maximum of two components (3) minimum of three components
Name: General Chemistry Chapter 11 Thermochemistry- Heat and Chemical Change
 Name: General Chemistry Chapter 11 Thermochemistry- Heat and Chemical Change Notepack 1 Section 11.1: The Flow of Energy Heat (Pages 293 299) 1. Define the following terms: a. Thermochemistry b. Energy
Name: General Chemistry Chapter 11 Thermochemistry- Heat and Chemical Change Notepack 1 Section 11.1: The Flow of Energy Heat (Pages 293 299) 1. Define the following terms: a. Thermochemistry b. Energy
Selected Questions on Chapter 5 Thermochemistry
 Selected Questions on Chapter 5 Thermochemistry Circle the correct answer: 1) At what velocity (m/s) must a 20.0 g object be moving in order to possess a kinetic energy of 1.00 J? A) 1.00 B) 100 10 2 C)
Selected Questions on Chapter 5 Thermochemistry Circle the correct answer: 1) At what velocity (m/s) must a 20.0 g object be moving in order to possess a kinetic energy of 1.00 J? A) 1.00 B) 100 10 2 C)
Thermochemistry. Energy and Chemical Change
 Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
Thermochemistry: Energy Flow and Chemical Reactions
 Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Chapter 5 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Ch. 14 Notes ENERGY AND CHEMICAL CHANGE NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics.
 Ch. 14 Notes ENERGY AND CHEMICAL CHANGE NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Energy the capacity to do work or produce heat A. two basic types of
Ch. 14 Notes ENERGY AND CHEMICAL CHANGE NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Energy the capacity to do work or produce heat A. two basic types of
Name Date Class THE FLOW OF ENERGY HEAT AND WORK
 17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
Name Energy Test period Date
 Name Energy Test period Date 1. The temperature 30. K expressed in degrees Celsius is 1) 243ºC 2) 243ºC 3) 303ºC 4) 303ºC 2. The potential energy diagram for a chemical reaction is shown below. 4. A sample
Name Energy Test period Date 1. The temperature 30. K expressed in degrees Celsius is 1) 243ºC 2) 243ºC 3) 303ºC 4) 303ºC 2. The potential energy diagram for a chemical reaction is shown below. 4. A sample
Name Date Class SECTION 16.1 PROPERTIES OF SOLUTIONS
 SOLUTIONS Practice Problems In your notebook, solve the following problems. SECTION 16.1 PROPERTIES OF SOLUTIONS 1. The solubility of CO 2 in water at 1.22 atm is 0.54 g/l. What is the solubility of carbon
SOLUTIONS Practice Problems In your notebook, solve the following problems. SECTION 16.1 PROPERTIES OF SOLUTIONS 1. The solubility of CO 2 in water at 1.22 atm is 0.54 g/l. What is the solubility of carbon
Energy, Heat and Temperature. Introduction
 Energy, Heat and Temperature Introduction 3 basic types of energy: Potential (possibility of doing work because of composition or position) Kinetic (moving objects doing work) Radiant (energy transferred
Energy, Heat and Temperature Introduction 3 basic types of energy: Potential (possibility of doing work because of composition or position) Kinetic (moving objects doing work) Radiant (energy transferred
Thermodynamics- Chapter 19 Schedule and Notes
 Thermodynamics- Chapter 19 Schedule and Notes Date Topics Video cast DUE Assignment during class time One Review of thermodynamics ONE and TWO Review of thermo Wksheet Two 19.1-4; state function THREE
Thermodynamics- Chapter 19 Schedule and Notes Date Topics Video cast DUE Assignment during class time One Review of thermodynamics ONE and TWO Review of thermo Wksheet Two 19.1-4; state function THREE
Unit 6: Energy. Aim: What is Energy? Energy: Energy is required to bring about changes in matter (atoms, ions, or molecules).
 Name: Date: Unit 6: Energy Aim: What is Energy? Energy: Energy is required to bring about changes in matter (atoms, ions, or molecules). Physical Changes Chemical Changes Example: Example: Energy is measured
Name: Date: Unit 6: Energy Aim: What is Energy? Energy: Energy is required to bring about changes in matter (atoms, ions, or molecules). Physical Changes Chemical Changes Example: Example: Energy is measured
Ch 100: Fundamentals for Chemistry
 Ch 100: Fundamentals for Chemistry Chapter 4: Properties of Matter Lecture Notes Physical & Chemical Properties Physical Properties are the characteristics of matter that can be changed without changing
Ch 100: Fundamentals for Chemistry Chapter 4: Properties of Matter Lecture Notes Physical & Chemical Properties Physical Properties are the characteristics of matter that can be changed without changing
Chapter 11. Thermochemistry: Heat & Chemical Change
 Chapter 11 Thermochemistry: Heat & Chemical Change The Flow of Energy Thermochemistry: Study of heat changes that occur during physical processes and chemical reactions Energy Energy is the capacity to
Chapter 11 Thermochemistry: Heat & Chemical Change The Flow of Energy Thermochemistry: Study of heat changes that occur during physical processes and chemical reactions Energy Energy is the capacity to
Unit 15 Energy and Thermochemistry Notes
 Name KEY Period CRHS Academic Chemistry Unit 15 Energy and Thermochemistry Notes Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
Name KEY Period CRHS Academic Chemistry Unit 15 Energy and Thermochemistry Notes Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
EDULABZ INTERNATIONAL. Heat ASSIGNMENT
 Heat ASSIGNMENT 1. Fill in the blank spaces by choosing the correct words from the list given below : List : substance, thermal capacity, mass, latent, heat, cold, constant, water, J C 1, fusion, hot.
Heat ASSIGNMENT 1. Fill in the blank spaces by choosing the correct words from the list given below : List : substance, thermal capacity, mass, latent, heat, cold, constant, water, J C 1, fusion, hot.
Supplemental Activities. Module: Thermodynamics. Section: Second Law of Thermodynamics Key
 Supplemental Activities Module: Thermodynamics Section: Second Law of Thermodynamics Key Spontaneity ACTIVITY 1 The purpose of this activity is to practice your understanding of the concept of spontaneous
Supplemental Activities Module: Thermodynamics Section: Second Law of Thermodynamics Key Spontaneity ACTIVITY 1 The purpose of this activity is to practice your understanding of the concept of spontaneous
Chapter 17 Thermochemistry
 Chapter 17 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Chapter 17 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Slide 1 / Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy. a only b only c only a and c b and c
 Slide 1 / 84 1 Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy A B C D E a only b only c only a and c b and c Slide 2 / 84 2 The internal energy of a system
Slide 1 / 84 1 Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy A B C D E a only b only c only a and c b and c Slide 2 / 84 2 The internal energy of a system
THERMOCHEMISTRY. This section explains the relationship between energy and heat, and distinguishes between heat capacity and specific heat.
 I Name _ Date _ Class _ THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY-HEAT AND WORK (pages 505-510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
I Name _ Date _ Class _ THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY-HEAT AND WORK (pages 505-510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
Name Chemistry / / SOL Questions Chapter 9 For each of the following, fill in the correct answer on the BLUE side of the scantron.
 Name Chemistry / / SOL Questions Chapter 9 For each of the following, fill in the correct answer on the BLUE side of the scantron. 1. Which number on the graph to the right represents the effect of the
Name Chemistry / / SOL Questions Chapter 9 For each of the following, fill in the correct answer on the BLUE side of the scantron. 1. Which number on the graph to the right represents the effect of the
Topic 05 Energetics : Heat Change. IB Chemistry T05D01
 Topic 05 Energetics 5.1-5.2: Heat Change IB Chemistry T05D01 5.1 Exothermic and endothermic reactions - 1 hour 5.1.1 Define the terms exothermic reaction, endothermic reaction and standard enthalpy change
Topic 05 Energetics 5.1-5.2: Heat Change IB Chemistry T05D01 5.1 Exothermic and endothermic reactions - 1 hour 5.1.1 Define the terms exothermic reaction, endothermic reaction and standard enthalpy change
A). Yes. B). No. Q15 Is it possible for a solid metal ball to float in mercury?
 Q15 Is it possible for a solid metal ball to float in mercury? A). Yes. B). No. The upward force is the weight of liquid displaced and the downward force is the weight of the ball. If the density of the
Q15 Is it possible for a solid metal ball to float in mercury? A). Yes. B). No. The upward force is the weight of liquid displaced and the downward force is the weight of the ball. If the density of the
Unit 15 Energy and Thermochemistry Notes
 Name Period CRHS Academic Chemistry Unit 15 Energy and Thermochemistry Notes Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
Name Period CRHS Academic Chemistry Unit 15 Energy and Thermochemistry Notes Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
Class work on Calorimetry. January 11 and 12, 2011
 Class work on Calorimetry January 11 and 12, 2011 Name 1. The number of calories needed to raise the temperature of 100 grams of water 10 degrees Celsius is the same as the number of calories needed to
Class work on Calorimetry January 11 and 12, 2011 Name 1. The number of calories needed to raise the temperature of 100 grams of water 10 degrees Celsius is the same as the number of calories needed to
2. State the direction of heat transfer between the surroundings and the water in the bottle from 7 a.m. to 3 p.m.
 Base your answers to questions 1 through 3 on the information below. A student investigated heat transfer using a bottle of water. The student placed the bottle in a room at 20.5 C. The student measured
Base your answers to questions 1 through 3 on the information below. A student investigated heat transfer using a bottle of water. The student placed the bottle in a room at 20.5 C. The student measured
Energy and Chemical Change
 Energy and Chemical Change Section 16.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Energy and Chemical Change Section 16.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Name Date Class THERMOCHEMISTRY
 Name Date Class 17 THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY HEAT AND WORK (pages 505 510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
Name Date Class 17 THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY HEAT AND WORK (pages 505 510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
Chapter 2 Heat, Temperature and the First Law of Thermodynamics
 Chapter 2 Heat, Temperature and the First Law of Thermodynamics 2.1. Temperature and the Zeroth Law of Thermodynamics 2.2. Thermal Expansion 2.3. Heat and the Absorption of Heat by Solids and Liquids 2.4.
Chapter 2 Heat, Temperature and the First Law of Thermodynamics 2.1. Temperature and the Zeroth Law of Thermodynamics 2.2. Thermal Expansion 2.3. Heat and the Absorption of Heat by Solids and Liquids 2.4.
Q=mcDt. Chemistry 30 notes review of specific heat capacity. Thermodynamics the movement of thermal energy
 Chemistry 30 notes review of specific heat capacity Thermodynamics the movement of thermal energy o Open system: both matter and energy are allowed to enter or leave the system. o Closed system: energy
Chemistry 30 notes review of specific heat capacity Thermodynamics the movement of thermal energy o Open system: both matter and energy are allowed to enter or leave the system. o Closed system: energy
Chemistry Grade : 11 Term-3/Final Exam Revision Sheet
 Chemistry Grade : 11 Term-3/Final Exam Revision Sheet Exam Date: Tuesday 12/6/2018 CCS:Chem.6a,6b,6c,6d,6e,6f,7a,7b,7d,7c,7e,7f,1g Chapter(12):Solutions Sections:1,2,3 Textbook pages 378 to 408 Chapter(16):Reaction
Chemistry Grade : 11 Term-3/Final Exam Revision Sheet Exam Date: Tuesday 12/6/2018 CCS:Chem.6a,6b,6c,6d,6e,6f,7a,7b,7d,7c,7e,7f,1g Chapter(12):Solutions Sections:1,2,3 Textbook pages 378 to 408 Chapter(16):Reaction
Thermochemistry. The study of energy changes that occur during chemical reactions and changes in state.
 Energy Thermochemistry The study of energy changes that occur during chemical reactions and changes in state. The Nature of Energy Energy - the ability to do work or produce heat Energy is stored in the
Energy Thermochemistry The study of energy changes that occur during chemical reactions and changes in state. The Nature of Energy Energy - the ability to do work or produce heat Energy is stored in the
0. Graphite is thermodynamically less stable that diamond under standard conditions. 1. True 2. False
 0. Graphite is thermodynamically less stable that diamond under standard conditions. 1. True 2. False 1. Which statement would be the best interpretation of the First Law of Thermodynamics? 1. The total
0. Graphite is thermodynamically less stable that diamond under standard conditions. 1. True 2. False 1. Which statement would be the best interpretation of the First Law of Thermodynamics? 1. The total
Energy Ability to produce change or do work. First Law of Thermodynamics. Heat (q) Quantity of thermal energy
 THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
Chapter 17: Energy and Kinetics
 Pages 510-547 S K K Chapter 17: Energy and Kinetics Thermochemistry: Causes of change in systems Kinetics: Rate of reaction progress (speed) Heat, Energy, and Temperature changes S J J Heat vs Temperature
Pages 510-547 S K K Chapter 17: Energy and Kinetics Thermochemistry: Causes of change in systems Kinetics: Rate of reaction progress (speed) Heat, Energy, and Temperature changes S J J Heat vs Temperature
THERMOCHEMISTRY & DEFINITIONS
 THERMOCHEMISTRY & DEFINITIONS Thermochemistry is the study of the study of relationships between chemistry and energy. All chemical changes and many physical changes involve exchange of energy with the
THERMOCHEMISTRY & DEFINITIONS Thermochemistry is the study of the study of relationships between chemistry and energy. All chemical changes and many physical changes involve exchange of energy with the
Chapter 15 Energy and Chemical Change
 Chapter 15 Energy and Chemical Change Chemical reactions usually absorb or release energy. Section 1: Energy Section 2: Heat Section 3: Thermochemical Equations Section 4: Calculating Enthalpy Change Section
Chapter 15 Energy and Chemical Change Chemical reactions usually absorb or release energy. Section 1: Energy Section 2: Heat Section 3: Thermochemical Equations Section 4: Calculating Enthalpy Change Section
Energy. Different types of energy exist (heat, potential, kinetic, chemical, nuclear etc.)
 Change in Energy Energy Different types of energy exist (heat, potential, kinetic, chemical, nuclear etc.) Heat - the energy transferred between objects that are at different temperatures. Unit of heat
Change in Energy Energy Different types of energy exist (heat, potential, kinetic, chemical, nuclear etc.) Heat - the energy transferred between objects that are at different temperatures. Unit of heat
Thermodynamics B Test
 Northern Regional: January 19 th, 2019 Thermodynamics B Test Name(s): Team Name: School Name: Team Number: Rank: Score: Science Olympiad North Florida Regional at the University of Florida Thermodynamics
Northern Regional: January 19 th, 2019 Thermodynamics B Test Name(s): Team Name: School Name: Team Number: Rank: Score: Science Olympiad North Florida Regional at the University of Florida Thermodynamics
2. If the volume of a container holding a gas is reduced, what will happen to the presure within the container?
 1. Which gas law states that the volume of a fixed mass of a gas is directly proportional to its Kelvin temperature if the pressure is kept constant? A. Boyle s law B. Charles law C. Dalton s law D. Gay-Lussac
1. Which gas law states that the volume of a fixed mass of a gas is directly proportional to its Kelvin temperature if the pressure is kept constant? A. Boyle s law B. Charles law C. Dalton s law D. Gay-Lussac
CP CHEMISTRY STUDY GUIDE The Kinetic Theory of Matter (Chapters 10 and 14)
 Unit 9 CP CHEMISTRY STUDY GUIDE The Kinetic Theory of Matter (Chapters 10 and 14) Unit Goals ( During this unit I will ) 1. Explain the behavior of matter in terms of the relationships between temperature,
Unit 9 CP CHEMISTRY STUDY GUIDE The Kinetic Theory of Matter (Chapters 10 and 14) Unit Goals ( During this unit I will ) 1. Explain the behavior of matter in terms of the relationships between temperature,
Thermochemistry: Heat and Chemical Change
 Thermochemistry: Heat and Chemical Change 1 Heat or Thermal Energy (q) Heat is a form of energy Is heat the same as temperature? Heat flows between two objects at different temperatures. Hot Cold 2 Chemical
Thermochemistry: Heat and Chemical Change 1 Heat or Thermal Energy (q) Heat is a form of energy Is heat the same as temperature? Heat flows between two objects at different temperatures. Hot Cold 2 Chemical
Energy Ability to produce change or do work. First Law of Thermodynamics. Heat (q) Quantity of thermal energy
 THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
Chapter 10 Lecture Notes: Thermodynamics
 Chapter 10 Lecture Notes: Thermodynamics During this unit of study, we will cover three main areas. A lot of this information is NOT included in your text book, which is a shame. Therefore, the notes you
Chapter 10 Lecture Notes: Thermodynamics During this unit of study, we will cover three main areas. A lot of this information is NOT included in your text book, which is a shame. Therefore, the notes you
3. When the external pressure is kpa torr, water will boil at what temperature? a C b C c. 100 C d. 18 C
 Chemistry EOC Review 5: Physical Behavior of Matter 1. Which gas is monatomic at STP? a. chlorine b. fluorine c. neon d. nitrogen 2. What Kelvin temperature is equal to 25 C? a. 248 K b. 298 K c. 100 K
Chemistry EOC Review 5: Physical Behavior of Matter 1. Which gas is monatomic at STP? a. chlorine b. fluorine c. neon d. nitrogen 2. What Kelvin temperature is equal to 25 C? a. 248 K b. 298 K c. 100 K
Calculate the mass of L of oxygen gas at 25.0 C and 1.18 atm pressure.
 148 Calculate the mass of 22650 L of oxygen gas at 25.0 C and 1.18 atm pressure. 1 - Convert the volume of oxygen gas to moles using IDEAL GAS EQUATION 2 - Convert moles oxygen gas to mass using formula
148 Calculate the mass of 22650 L of oxygen gas at 25.0 C and 1.18 atm pressure. 1 - Convert the volume of oxygen gas to moles using IDEAL GAS EQUATION 2 - Convert moles oxygen gas to mass using formula
11B, 11E Temperature and heat are related but not identical.
 Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes
 Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes Students will be expected to: Compare the molar enthalpies of several combustion reactions involving organic compounds.
Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes Students will be expected to: Compare the molar enthalpies of several combustion reactions involving organic compounds.
Exercises Temperature (pages ) 1. Define temperature. 2. Explain how a common liquid thermometer works.
 Exercises 21.1 Temperature (pages 407 408) 1. Define temperature. 2. Explain how a common liquid thermometer works. Match each number with the corresponding description. Temperature Description 3. 273
Exercises 21.1 Temperature (pages 407 408) 1. Define temperature. 2. Explain how a common liquid thermometer works. Match each number with the corresponding description. Temperature Description 3. 273
Ch. 6 Enthalpy Changes
 Ch. 6 Enthalpy Changes Energy: The capacity to do work. In Physics, there are 2 main types of energy Kinetic (energy of motion) = ½ mv 2 Potential (energy of position due to gravity)= mgh In Chemistry,
Ch. 6 Enthalpy Changes Energy: The capacity to do work. In Physics, there are 2 main types of energy Kinetic (energy of motion) = ½ mv 2 Potential (energy of position due to gravity)= mgh In Chemistry,
Energy Transformations
 Thermochemistry Energy Transformations Thermochemistry - concerned with heat changes that occur during chemical reactions Energy - capacity for doing work or supplying heat weightless, odorless, tasteless
Thermochemistry Energy Transformations Thermochemistry - concerned with heat changes that occur during chemical reactions Energy - capacity for doing work or supplying heat weightless, odorless, tasteless
0 o K is called absolute zero. Water Freezes: 273 o K Water Boils: 373 o K
 Part I Notes Temperature and Heat The terms at the right all mean the same thing. The heat energy of a substance is the sum of the kinetic and potential energies of all of the atoms and molecules in the
Part I Notes Temperature and Heat The terms at the right all mean the same thing. The heat energy of a substance is the sum of the kinetic and potential energies of all of the atoms and molecules in the
This reaction is ENDOTHERMIC. Energy is being transferred from the room/flask/etc. (the SURROUNDINGS) to the reaction itself (the SYSTEM).
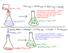 151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
CHEMISTRY 109 #25 - REVIEW
 CHEMISTRY 109 Help Sheet #25 - REVIEW Chapter 4 (Part I); Sections 4.1-4.6; Ch. 9, Section 9.4a-9.4c (pg 387) ** Review the appropriate topics for your lecture section ** Prepared by Dr. Tony Jacob http://www.chem.wisc.edu/areas/clc
CHEMISTRY 109 Help Sheet #25 - REVIEW Chapter 4 (Part I); Sections 4.1-4.6; Ch. 9, Section 9.4a-9.4c (pg 387) ** Review the appropriate topics for your lecture section ** Prepared by Dr. Tony Jacob http://www.chem.wisc.edu/areas/clc
Name: Regents Chemistry: Mr. Palermo. Student Version. Notes: Unit 6A Heat
 Name: Regents Chemistry: Mr. Palermo Student Version Notes: Unit 6A Heat Name: KEY IDEAS Heat is a transfer of energy (usually thermal energy) from a body of higher temperature to a body of lower temperature.
Name: Regents Chemistry: Mr. Palermo Student Version Notes: Unit 6A Heat Name: KEY IDEAS Heat is a transfer of energy (usually thermal energy) from a body of higher temperature to a body of lower temperature.
1. Thermo = the that happen in a chemical reaction. 4. You must ADD energy to melt solids into liquids example:
 ThermoChem Notes 1. Thermo = the that happen in a chemical reaction. 2. When heat is given off it is an reaction. 3. Sometimes energy is absorbed in order for the reaction happen; this is called an reaction.
ThermoChem Notes 1. Thermo = the that happen in a chemical reaction. 2. When heat is given off it is an reaction. 3. Sometimes energy is absorbed in order for the reaction happen; this is called an reaction.
UNIT 15 - Reaction Energy & Reaction Kinetics. I. Thermochemistry: study of heat in chemical reactions and phase changes
 I. Thermochemistry: study of heat in chemical reactions and phase changes II. A. Heat equation (change in temperature): Q = m. C. p T 1. Q = heat (unit is Joules) 2. m = mass (unit is grams) 3. C p = specific
I. Thermochemistry: study of heat in chemical reactions and phase changes II. A. Heat equation (change in temperature): Q = m. C. p T 1. Q = heat (unit is Joules) 2. m = mass (unit is grams) 3. C p = specific
Section 9: Thermodynamics and Energy
 Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
Thermochemistry HW. PSI Chemistry
 Thermochemistry HW PSI Chemistry Name Energy 1) Objects can possess energy as: (a) endothermic energy (b) potential energy A) a only B) b only C) c only D) a and c E) b and c (c) kinetic energy 2) The
Thermochemistry HW PSI Chemistry Name Energy 1) Objects can possess energy as: (a) endothermic energy (b) potential energy A) a only B) b only C) c only D) a and c E) b and c (c) kinetic energy 2) The
Chapter 5. Thermochemistry
 Chapter 5 Thermochemistry Energy Thermodynamics Study of the relationship between heat, work, and other forms of energy Thermochemistry A branch of thermodynamics Focuses on the study of heat given off
Chapter 5 Thermochemistry Energy Thermodynamics Study of the relationship between heat, work, and other forms of energy Thermochemistry A branch of thermodynamics Focuses on the study of heat given off
solid IMF>liquid IMF>gas IMF Draw a diagram to represent the 3 common states of matter of a given substance: solid liquid gas
 Thermochemistry Part 1 Notes States of Matter and Intermolecular Forces (IMF) Chemistry HP At the end of this unit, students should be able to: Describe the various states of matter in terms of kinetic
Thermochemistry Part 1 Notes States of Matter and Intermolecular Forces (IMF) Chemistry HP At the end of this unit, students should be able to: Describe the various states of matter in terms of kinetic
Chemistry Lab Fairfax High School Invitational January 7, Team Number: High School: Team Members Names:
 Chemistry Lab Fairfax High School Invitational January 7, 2017 Team Number: High School: Team Members Names: Reference Values: Gas Constant, R = 8.314 J mol -1 K -1 Gas Constant, R = 0.08206 L atm mol
Chemistry Lab Fairfax High School Invitational January 7, 2017 Team Number: High School: Team Members Names: Reference Values: Gas Constant, R = 8.314 J mol -1 K -1 Gas Constant, R = 0.08206 L atm mol
Chapter 5 Principles of Chemical Reactivity: Energy and Chemical Reactions
 Chapter 5 Principles of Chemical Reactivity: Energy and Chemical Reactions Jeffrey Mack California State University, Sacramento Energy & Chemistry Questions that need to be addressed: How do we measure
Chapter 5 Principles of Chemical Reactivity: Energy and Chemical Reactions Jeffrey Mack California State University, Sacramento Energy & Chemistry Questions that need to be addressed: How do we measure
Energy and Chemical Change
 Energy and Chemical Change Section 15.1 Energy Section 15.2 Heat Section 15.3 Thermochemical Equations Section 15.4 Calculating Enthalpy Change Section 15.5 Reaction Spontaneity Click a hyperlink or folder
Energy and Chemical Change Section 15.1 Energy Section 15.2 Heat Section 15.3 Thermochemical Equations Section 15.4 Calculating Enthalpy Change Section 15.5 Reaction Spontaneity Click a hyperlink or folder
Lecture 10. What is energy? Professor Hicks Inorganic Chemistry (CHE151) Ability to do work. Work means moving something against a force
 Lecture 10 Professor Hicks Inorganic Chemistry (CHE151) Ability to do work What is energy? Work means moving something against a force Energy thought of as an imaginary liquid that gets moved from one
Lecture 10 Professor Hicks Inorganic Chemistry (CHE151) Ability to do work What is energy? Work means moving something against a force Energy thought of as an imaginary liquid that gets moved from one
Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions
 Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions Terms, definitions, topics Joule, calorie (Re-read p 57-58) Thermochemistry Exothermic reaction Endothermic reaction
Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions Terms, definitions, topics Joule, calorie (Re-read p 57-58) Thermochemistry Exothermic reaction Endothermic reaction
Thermochemistry. Energy (and Thermochemistry) World of Chemistry Chapter 10. Energy. Energy
 Thermochemistry Thermodynamics is the science of the relationship between heat and other forms of energy. (and Thermochemistry) World of Chemistry Chapter 10 is defined as the ability to do work or produce
Thermochemistry Thermodynamics is the science of the relationship between heat and other forms of energy. (and Thermochemistry) World of Chemistry Chapter 10 is defined as the ability to do work or produce
Name: Regents Chemistry: Mr. Palermo. Notes: Unit 7 Heat.
 Name: Regents Chemistry: Mr. Palermo Notes: Unit 7 Heat 1 Name: KEY IDEAS Heat is a transfer of energy (usually thermal energy) from a body of higher temperature to a body of lower temperature. Thermal
Name: Regents Chemistry: Mr. Palermo Notes: Unit 7 Heat 1 Name: KEY IDEAS Heat is a transfer of energy (usually thermal energy) from a body of higher temperature to a body of lower temperature. Thermal
Chapter 8 Thermochemistry: Chemical Energy
 Chapter 8 Thermochemistry: Chemical Energy 國防醫學院生化學科王明芳老師 2011-11-8 & 2011-11-15 Chapter 8/1 Energy and Its Conservation Conservation of Energy Law: Energy cannot be created or destroyed; it can only be
Chapter 8 Thermochemistry: Chemical Energy 國防醫學院生化學科王明芳老師 2011-11-8 & 2011-11-15 Chapter 8/1 Energy and Its Conservation Conservation of Energy Law: Energy cannot be created or destroyed; it can only be
Name Chemistry / / Understanding Phase Changes
 Name Chemistry / / Understanding Phase Changes As a piece of ice is exposed to a warmer environment, it begins to absorb heat. The heat causes the solid molecules to vibrate faster. Eventually, the ice
Name Chemistry / / Understanding Phase Changes As a piece of ice is exposed to a warmer environment, it begins to absorb heat. The heat causes the solid molecules to vibrate faster. Eventually, the ice
CP Chapter 17 Thermochemistry
 CP Chapter 17 Thermochemistry Thermochemistry Thermochemistry is the study of energy that occur during chemical reactions and phase changes (changes of state) The Nature of Energy Energy is the ability
CP Chapter 17 Thermochemistry Thermochemistry Thermochemistry is the study of energy that occur during chemical reactions and phase changes (changes of state) The Nature of Energy Energy is the ability
Chemistry Chapter 16. Reaction Energy
 Chemistry Reaction Energy Section 16.1.I Thermochemistry Objectives Define temperature and state the units in which it is measured. Define heat and state its units. Perform specific-heat calculations.
Chemistry Reaction Energy Section 16.1.I Thermochemistry Objectives Define temperature and state the units in which it is measured. Define heat and state its units. Perform specific-heat calculations.
Advanced Chemistry Practice Problems
 Thermodynamics: Review of Thermochemistry 1. Question: What is the sign of DH for an exothermic reaction? An endothermic reaction? Answer: ΔH is negative for an exothermic reaction and positive for an
Thermodynamics: Review of Thermochemistry 1. Question: What is the sign of DH for an exothermic reaction? An endothermic reaction? Answer: ΔH is negative for an exothermic reaction and positive for an
Energy and Chemical Change
 Energy and Chemical Change Reviewing Vocabulary Match the definition in Column A with the term in Column B. h e d p c f a r m t j i s l u k n q g o Column A 1. The ability to do work or produce heat 2.
Energy and Chemical Change Reviewing Vocabulary Match the definition in Column A with the term in Column B. h e d p c f a r m t j i s l u k n q g o Column A 1. The ability to do work or produce heat 2.
Energy Heat Work Heat Capacity Enthalpy
 Energy Heat Work Heat Capacity Enthalpy 1 Prof. Zvi C. Koren 20.07.2010 Thermodynamics vs. Kinetics Thermodynamics Thermo = Thermo + Dynamics E (Note: Absolute E can never be determined by humans!) Can
Energy Heat Work Heat Capacity Enthalpy 1 Prof. Zvi C. Koren 20.07.2010 Thermodynamics vs. Kinetics Thermodynamics Thermo = Thermo + Dynamics E (Note: Absolute E can never be determined by humans!) Can
Chapter 11 Thermochemistry Heat and Chemical Change
 Chemistry/ PEP Name: Date: Chapter 11 Thermochemistry Heat and Chemical Change Chapter 11:1 35, 57, 60, 61, 71 Section 11.1 The Flow of Energy - Heat 1. When 435 of heat is added to 3.4 g of olive oil
Chemistry/ PEP Name: Date: Chapter 11 Thermochemistry Heat and Chemical Change Chapter 11:1 35, 57, 60, 61, 71 Section 11.1 The Flow of Energy - Heat 1. When 435 of heat is added to 3.4 g of olive oil
AlCl3(aq) + 3 NaOH(aq) Al(OH)3(s) + 3 NaCl(aq)
 1. Under which conditions does a real gas behave most like an ideal gas? A) at low temperatures and high pressures B) at low temperatures and low pressures C) at high temperatures and high pressures D)
1. Under which conditions does a real gas behave most like an ideal gas? A) at low temperatures and high pressures B) at low temperatures and low pressures C) at high temperatures and high pressures D)
= (25.0 g)(0.137 J/g C)[61.2 C - (-31.4 C)] = 317 J (= kj)
![= (25.0 g)(0.137 J/g C)[61.2 C - (-31.4 C)] = 317 J (= kj) = (25.0 g)(0.137 J/g C)[61.2 C - (-31.4 C)] = 317 J (= kj)](/thumbs/89/101004910.jpg) CHEM 101A ARMSTRONG SOLUTIONS TO TOPIC D PROBLEMS 1) For all problems involving energy, you may give your answer in either joules or kilojoules, unless the problem specifies a unit. (In general, though,
CHEM 101A ARMSTRONG SOLUTIONS TO TOPIC D PROBLEMS 1) For all problems involving energy, you may give your answer in either joules or kilojoules, unless the problem specifies a unit. (In general, though,
Chemical Reactions Chapter 17 Study Guide (Unit 10)
 Chemical Reactions Chapter 17 Study Guide (Unit 10) Name: Hr: Understand and be able to explain all of the key concepts. Define and understand all of the survival words Memorize the names and symbols for
Chemical Reactions Chapter 17 Study Guide (Unit 10) Name: Hr: Understand and be able to explain all of the key concepts. Define and understand all of the survival words Memorize the names and symbols for
17.4 Calculating Heats Essential Understanding Heats of reaction can be calculated when it is difficult or
 17.4 Calculating Heats of Reaction Essential Understanding Heats of reaction can be calculated when it is difficult or impossible to measure them directly. Lesson Summary Hess s Law Hess s law provides
17.4 Calculating Heats of Reaction Essential Understanding Heats of reaction can be calculated when it is difficult or impossible to measure them directly. Lesson Summary Hess s Law Hess s law provides
Chapter 3: Matter and Energy
 Chapter 3: Matter and Energy Convert between Fahrenheit, Celsius, and Kelvin temperature scales. Relate energy, temperature change, and heat capacity. The atoms and molecules that compose matter are in
Chapter 3: Matter and Energy Convert between Fahrenheit, Celsius, and Kelvin temperature scales. Relate energy, temperature change, and heat capacity. The atoms and molecules that compose matter are in
