Quantum Mechanics for Scientists and Engineers. David Miller
|
|
|
- Darleen Sharp
- 6 years ago
- Views:
Transcription
1 Quantum Mechanics for Scientists and Engineers David Miller
2 The particle in a box
3 The particle in a box Linearity and normalization
4 Linearity and Schrödinger s equation We see that Schrödinger s equation is linear m V r E The wavefunction appears only in first order there are no second or higher order terms such as or 3 So, if is a solution, so also is a this just corresponds to multiplying both sides by the constant a
5 Normalization of the wavefunction Born postulated the probability Pr of finding a particle near a point r is r Specifically let us define P r as a probability density For some very small (infinitesimal) volume d 3 r around r the probability of finding the particle in that volume is P r d r 3
6 Normalization of the wavefunction The sum of all such probabilities should be 1 So 3 Prd r 1 r Can we choose so that we can use r as the probability density not just proportional to probability density? Unless we have been lucky our solution to Schrödinger s equation did not give a r so that 3 d r r 1
7 Normalization of the wavefunction Generally, this integral would give some other real positive number which we could write as 1/ a where a is some (possibly complex) number That is, 3 1 r d r a But we know that if r is a solution of Schrödinger s equation so also is a r
8 Normalization of the wavefunction So if we use the solution N a instead of then 3 N r d r 1 and we can use N r as the probability density, i.e., N r r P N r would then be called a normalized wavefunction
9 Normalization of the wavefunction So, to summarize normalization we take the solution we have obtained from Schrödinger s wave equation we integrate r to get a number we call 1/ a then we obtain the normalized wavefunction a for which N 3 N r d r 1 and we can use N r as the probability density
10 Technical notes on normalization Note that normalization only sets the magnitude of a not the phase we are free to choose any phase for a or indeed for the original solution a phase factor expi is just another number by which we can multiply the solution and still have a solution
11 Technical notes on normalization If we think of space as infinite functions like sin kx,, and exp ikr cannot be normalized in this way Technically, their squared modulus is not Lebesgue integrable They are not L functions This difficulty is mathematical, not physical It is caused by over-idealizing the mathematics to get functions that are simple to use cos kz
12 Technical notes on normalization There are work-arounds for this difficulty 1 - only work with finite volumes in actual problems this is the most common solution - use normalization to a delta function introduces another infinity to compensate for the first one This can be done but we will try to avoid it
13
14 The particle in a box Solving for the particle in a box
15 Particle in a box We consider a particle of mass m with a spatially-varying potential V(z) in the z direction so we have a Schrödinger equation z d V z z E z m dz where E is the energy of the particle and (z) is the wavefunction
16 Particle in a box Suppose the potential energy is a simple rectangular potential well thickness L z Potential energy is constant inside we choose V 0 there rising to infinity at the walls i.e., at z 0 and z Lz We will sometimes call this an infinite or infinitely deep (potential) well Energy V 0 z 0 L z z L z
17 Particle in a box Because these potentials at z 0 and at z L z are infinitely high but the particle s energy E is presumably finite we presume there is no possibility of finding the particle outside i.e., for z 0 or z Lz so the wavefunction is 0 there so should be 0 at the walls Energy V 0 z 0 L z z L z
18 Particle in a box With these choices inside the well the Schrödinger equation d z V z z E z m dz becomes d z E z m dz with the boundary conditions and 0 0 L z 0 Energy V 0 z 0 L z z L z
19 Particle in a box The general solution to the equation d z E z m dz is of the form z Asin kz Bcos kz where A and B are constants and k me/ The boundary condition 0 0 means B 0 because cos 0 1 Energy V 0 z 0 L z z L z
20 Particle in a box With now z Asin kz and the condition L z 0 kz must be a multiple of, i.e., k me/ n / Lz where n is an integer k Since, therefore, E m the solutions are n z n nz Ansin with En L z m Lz Energy n 3 n n 1 E 3 E E 1
21 Particle in a box We restrict n to positive integers n 1,, for the following reasons Since sinasina for any real number a the wavefunctions with negative n are the same as those with positive n within an arbitrary factor, here -1 the wavefunction for n 0 is trivial the wavefunction is 0 everywhere Energy n n 3 n n 1 z E 3 E E 1 n z Ansin Lz
22 Particle in a box We can normalize the wavefunctions L z n z L z An sin dz An 0 Lz To have this integral equal 1 choose An / Lz Note A n can be complex All such solutions are arbitrary within a unit complex factor Energy n 3 n n 1 E 3 E E 1 Conventionally, we choose A n real for simplicity in writing
23 Particle in a box E n n m Lz n 3 E 3 n z n z sin Lz Lz Energy n E n 1,, 0 n 1 E 1 L z
24
25 The particle in a box Nature of particle-in-a-box solutions
26 Eigenvalues and eigenfunctions Solutions with a specific set of allowed values of a parameter (here energy) eigenvalues and with a particular function associated with each such value eigenfunctions can be called eigensolutions n E n z n m Lz n z sin Lz Lz n 1,,
27 Eigenvalues and eigenfunctions Here since the parameter is an energy we can call the eigenvalues eigenenergies and we can refer to the eigenfunctions as the energy eigenfunctions n E n z n m Lz n z sin Lz Lz n 1,,
28 Degeneracy Note in some problems it can be possible to have more than one eigenfunction with a given eigenvalue a phenomenon known as degeneracy The number of such states with the same eigenvalue is called the degeneracy of that state
29 Parity of wavefunctions Note these eigenfunctions have definite symmetry n 3 the n 1 function is the mirror image on the left of what it is on the right n such a function has even parity or is said to be an even function The n 3 eigenfunction is also even n 1
30 Parity of wavefunctions The n eigenfunction is an inverted image the value at any point on the right of the center is exactly minus the value at the mirror image point on the left of the center Such a function has odd parity or is said to be an odd function n 3 n n 1
31 Parity of wavefunctions For this symmetric well problem the functions alternate between being even and odd and all the solutions are either even or odd i.e., all the solutions have a definite parity Such definite parity is common in symmetric problems it is mathematically very helpful n 3 n n 1
32 Quantum confinement This particle-in-a-box behavior is very different from the classical case 1 there is only a discrete set of possible values for the energy there is a minimum possible energy for the particle corresponding to n 1 here E 1 / m / Lz sometimes called a zero-point energy Energy 0 n 3 n n 1 L z E 3 E E 1
33 Quantum confinement 3 - the particle is not uniformly distributed over the box, and its distribution is different for n 3 different energies It is almost never found very near to the walls of the box n the probability obeys a standing wave pattern 0 n 1 Energy E 3 E E 1 L z
34 Quantum confinement In the lowest state ( n 1 ), it is most likely to be found near the center of the box In higher states, n 3 E 3 there are points inside the box where the particle will never be found Energy 0 n n 1 E E 1 L z
35 Quantum confinement Note that each successively higher energy state has one more zero in the eigenfunction This is very common behavior in quantum mechanics Energy 0 n 3 n n 1 E 3 E E 1 L z
36 Energies in quantum mechanics In quantum mechanical calculations we can always use Joules as units of energy but these are rather large A very convenient energy unit which also has a simple physical significance 19 is the electron-volt (ev) J the energy change of an electron in moving through an electrostatic potential change of 1V Energy in ev = energy in Joules/e 19 e electronic charge C (Coulombs)
37 Orders of magnitude E.g., confine an electron in a 5 Å (0.5 nm) thick box The first allowed level for the electron is o E m / / J1.5 ev The separation between the first and second allowed energies ( E ) E1 3E1 is 4.5eV which is a characteristic size of major energy separations between levels in an atom
38
Quantum Mechanics for Scientists and Engineers. David Miller
 Quantum Mechanics for Scientists and Engineers David Miller Particles in potential wells The finite potential well Insert video here (split screen) Finite potential well Lesson 7 Particles in potential
Quantum Mechanics for Scientists and Engineers David Miller Particles in potential wells The finite potential well Insert video here (split screen) Finite potential well Lesson 7 Particles in potential
2.2 Schrödinger s wave equation
 2.2 Schrödinger s wave equation Slides: Video 2.2.1 Schrödinger wave equation introduction Text reference: Quantum Mechanics for Scientists and Engineers Section Chapter 2 introduction Schrödinger s wave
2.2 Schrödinger s wave equation Slides: Video 2.2.1 Schrödinger wave equation introduction Text reference: Quantum Mechanics for Scientists and Engineers Section Chapter 2 introduction Schrödinger s wave
Applied Nuclear Physics (Fall 2006) Lecture 3 (9/13/06) Bound States in One Dimensional Systems Particle in a Square Well
 22.101 Applied Nuclear Physics (Fall 2006) Lecture 3 (9/13/06) Bound States in One Dimensional Systems Particle in a Square Well References - R. L. Liboff, Introductory Quantum Mechanics (Holden Day, New
22.101 Applied Nuclear Physics (Fall 2006) Lecture 3 (9/13/06) Bound States in One Dimensional Systems Particle in a Square Well References - R. L. Liboff, Introductory Quantum Mechanics (Holden Day, New
df(x) dx = h(x) Chemistry 4531 Mathematical Preliminaries Spring 2009 I. A Primer on Differential Equations Order of differential equation
 Chemistry 4531 Mathematical Preliminaries Spring 009 I. A Primer on Differential Equations Order of differential equation Linearity of differential equation Partial vs. Ordinary Differential Equations
Chemistry 4531 Mathematical Preliminaries Spring 009 I. A Primer on Differential Equations Order of differential equation Linearity of differential equation Partial vs. Ordinary Differential Equations
Particle in one-dimensional box
 Particle in the box Particle in one-dimensional box V(x) -a 0 a +~ An example of a situation in which only bound states exist in a quantum system. We consider the stationary states of a particle confined
Particle in the box Particle in one-dimensional box V(x) -a 0 a +~ An example of a situation in which only bound states exist in a quantum system. We consider the stationary states of a particle confined
An operator is a transformation that takes a function as an input and produces another function (usually).
 Formalism of Quantum Mechanics Operators Engel 3.2 An operator is a transformation that takes a function as an input and produces another function (usually). Example: In QM, most operators are linear:
Formalism of Quantum Mechanics Operators Engel 3.2 An operator is a transformation that takes a function as an input and produces another function (usually). Example: In QM, most operators are linear:
Quantum Mechanics for Scientists and Engineers. David Miller
 Quantum Mechanics for Scientists and Engineers David Miller Time evolution of superpositions Time evolution of superpositions Superposition for a particle in a box Superposition for a particle in a box
Quantum Mechanics for Scientists and Engineers David Miller Time evolution of superpositions Time evolution of superpositions Superposition for a particle in a box Superposition for a particle in a box
4.1 Time evolution of superpositions. Slides: Video Introduction to time evolution of superpositions
 4.1 Time evolution of superpositions Slides: Video 4.1.1 Introduction to time evolution of superpositions Time evolution of superpositions Quantum mechanics for scientists and engineers David Miller 4.1
4.1 Time evolution of superpositions Slides: Video 4.1.1 Introduction to time evolution of superpositions Time evolution of superpositions Quantum mechanics for scientists and engineers David Miller 4.1
= X = X ( ~) } ( ) ( ) On the other hand, when the Hamiltonian acts on ( ) one finds that
 6. A general normalized solution to Schrödinger s equation of motion for a particle moving in a time-independent potential is of the form ( ) = P } where the and () are, respectively, eigenvalues and normalized
6. A general normalized solution to Schrödinger s equation of motion for a particle moving in a time-independent potential is of the form ( ) = P } where the and () are, respectively, eigenvalues and normalized
ECE 487 Lecture 5 : Foundations of Quantum Mechanics IV Class Outline:
 ECE 487 Lecture 5 : Foundations of Quantum Mechanics IV Class Outline: Linearly Varying Potential Triangular Potential Well Time-Dependent Schrödinger Equation Things you should know when you leave Key
ECE 487 Lecture 5 : Foundations of Quantum Mechanics IV Class Outline: Linearly Varying Potential Triangular Potential Well Time-Dependent Schrödinger Equation Things you should know when you leave Key
Lecture #5: Begin Quantum Mechanics: Free Particle and Particle in a 1D Box
 561 Fall 017 Lecture #5 page 1 Last time: Lecture #5: Begin Quantum Mechanics: Free Particle and Particle in a 1D Box 1-D Wave equation u x = 1 u v t * u(x,t): displacements as function of x,t * nd -order:
561 Fall 017 Lecture #5 page 1 Last time: Lecture #5: Begin Quantum Mechanics: Free Particle and Particle in a 1D Box 1-D Wave equation u x = 1 u v t * u(x,t): displacements as function of x,t * nd -order:
1 Expansion in orthogonal functions
 Notes "orthogonal" January 9 Expansion in orthogonal functions To obtain a more useful form of the Green s function, we ll want to expand in orthogonal functions that are (relatively) easy to integrate.
Notes "orthogonal" January 9 Expansion in orthogonal functions To obtain a more useful form of the Green s function, we ll want to expand in orthogonal functions that are (relatively) easy to integrate.
5.1 Uncertainty principle and particle current
 5.1 Uncertainty principle and particle current Slides: Video 5.1.1 Momentum, position, and the uncertainty principle Text reference: Quantum Mechanics for Scientists and Engineers Sections 3.1 3.13 Uncertainty
5.1 Uncertainty principle and particle current Slides: Video 5.1.1 Momentum, position, and the uncertainty principle Text reference: Quantum Mechanics for Scientists and Engineers Sections 3.1 3.13 Uncertainty
df(x) = h(x) dx Chemistry 4531 Mathematical Preliminaries Spring 2009 I. A Primer on Differential Equations Order of differential equation
 Chemistry 4531 Mathematical Preliminaries Spring 009 I. A Primer on Differential Equations Order of differential equation Linearity of differential equation Partial vs. Ordinary Differential Equations
Chemistry 4531 Mathematical Preliminaries Spring 009 I. A Primer on Differential Equations Order of differential equation Linearity of differential equation Partial vs. Ordinary Differential Equations
Quantum Physics Lecture 8
 Quantum Physics ecture 8 Steady state Schroedinger Equation (SSSE): eigenvalue & eigenfunction particle in a box re-visited Wavefunctions and energy states normalisation probability density Expectation
Quantum Physics ecture 8 Steady state Schroedinger Equation (SSSE): eigenvalue & eigenfunction particle in a box re-visited Wavefunctions and energy states normalisation probability density Expectation
The Hydrogen Atom. Dr. Sabry El-Taher 1. e 4. U U r
 The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
Page 404. Lecture 22: Simple Harmonic Oscillator: Energy Basis Date Given: 2008/11/19 Date Revised: 2008/11/19
 Page 404 Lecture : Simple Harmonic Oscillator: Energy Basis Date Given: 008/11/19 Date Revised: 008/11/19 Coordinate Basis Section 6. The One-Dimensional Simple Harmonic Oscillator: Coordinate Basis Page
Page 404 Lecture : Simple Harmonic Oscillator: Energy Basis Date Given: 008/11/19 Date Revised: 008/11/19 Coordinate Basis Section 6. The One-Dimensional Simple Harmonic Oscillator: Coordinate Basis Page
Applications of Quantum Theory to Some Simple Systems
 Applications of Quantum Theory to Some Simple Systems Arbitrariness in the value of total energy. We will use classical mechanics, and for simplicity of the discussion, consider a particle of mass m moving
Applications of Quantum Theory to Some Simple Systems Arbitrariness in the value of total energy. We will use classical mechanics, and for simplicity of the discussion, consider a particle of mass m moving
Homework Problem Set 3 Solutions
 Chemistry 460 Dr. Jean M. Standard Homework Problem Set 3 Solutions 1. See Section 2-5 of Lowe and Peterson for more information about this example. When a particle experiences zero potential energy over
Chemistry 460 Dr. Jean M. Standard Homework Problem Set 3 Solutions 1. See Section 2-5 of Lowe and Peterson for more information about this example. When a particle experiences zero potential energy over
The Sommerfeld Polynomial Method: Harmonic Oscillator Example
 Chemistry 460 Fall 2017 Dr. Jean M. Standard October 2, 2017 The Sommerfeld Polynomial Method: Harmonic Oscillator Example Scaling the Harmonic Oscillator Equation Recall the basic definitions of the harmonic
Chemistry 460 Fall 2017 Dr. Jean M. Standard October 2, 2017 The Sommerfeld Polynomial Method: Harmonic Oscillator Example Scaling the Harmonic Oscillator Equation Recall the basic definitions of the harmonic
Total Angular Momentum for Hydrogen
 Physics 4 Lecture 7 Total Angular Momentum for Hydrogen Lecture 7 Physics 4 Quantum Mechanics I Friday, April th, 008 We have the Hydrogen Hamiltonian for central potential φ(r), we can write: H r = p
Physics 4 Lecture 7 Total Angular Momentum for Hydrogen Lecture 7 Physics 4 Quantum Mechanics I Friday, April th, 008 We have the Hydrogen Hamiltonian for central potential φ(r), we can write: H r = p
Quantum Mechanics for Scientists and Engineers. David Miller
 Quantum Mechanics for Scientists and Engineers David Miller Wavepackets Wavepackets Group velocity Group velocity Consider two waves at different frequencies 1 and 2 and suppose that the wave velocity
Quantum Mechanics for Scientists and Engineers David Miller Wavepackets Wavepackets Group velocity Group velocity Consider two waves at different frequencies 1 and 2 and suppose that the wave velocity
Physics 43 Chapter 41 Homework #11 Key
 Physics 43 Chapter 4 Homework # Key π sin. A particle in an infinitely deep square well has a wave function given by ( ) for and zero otherwise. Determine the epectation value of. Determine the probability
Physics 43 Chapter 4 Homework # Key π sin. A particle in an infinitely deep square well has a wave function given by ( ) for and zero otherwise. Determine the epectation value of. Determine the probability
ECE 487 Lecture 6 : Time-Dependent Quantum Mechanics I Class Outline:
 ECE 487 Lecture 6 : Time-Dependent Quantum Mechanics I Class Outline: Time-Dependent Schrödinger Equation Solutions to thetime-dependent Schrödinger Equation Expansion of Energy Eigenstates Things you
ECE 487 Lecture 6 : Time-Dependent Quantum Mechanics I Class Outline: Time-Dependent Schrödinger Equation Solutions to thetime-dependent Schrödinger Equation Expansion of Energy Eigenstates Things you
Problems and Multiple Choice Questions
 Problems and Multiple Choice Questions 1. A momentum operator in one dimension is 2. A position operator in 3 dimensions is 3. A kinetic energy operator in 1 dimension is 4. If two operator commute, a)
Problems and Multiple Choice Questions 1. A momentum operator in one dimension is 2. A position operator in 3 dimensions is 3. A kinetic energy operator in 1 dimension is 4. If two operator commute, a)
Postulates and Theorems of Quantum Mechanics
 Postulates and Theorems of Quantum Mechanics Literally, a postulate is something taen as self-evident or assumed without proof as a basis for reasoning. It is simply is Postulate 1: State of a physical
Postulates and Theorems of Quantum Mechanics Literally, a postulate is something taen as self-evident or assumed without proof as a basis for reasoning. It is simply is Postulate 1: State of a physical
Sample Quantum Chemistry Exam 1 Solutions
 Chemistry 46 Fall 217 Dr Jean M Standard September 27, 217 Name SAMPE EXAM Sample Quantum Chemistry Exam 1 Solutions 1 (24 points Answer the following questions by selecting the correct answer from the
Chemistry 46 Fall 217 Dr Jean M Standard September 27, 217 Name SAMPE EXAM Sample Quantum Chemistry Exam 1 Solutions 1 (24 points Answer the following questions by selecting the correct answer from the
Quantum Mechanics: Particles in Potentials
 Quantum Mechanics: Particles in Potentials 3 april 2010 I. Applications of the Postulates of Quantum Mechanics Now that some of the machinery of quantum mechanics has been assembled, one can begin to apply
Quantum Mechanics: Particles in Potentials 3 april 2010 I. Applications of the Postulates of Quantum Mechanics Now that some of the machinery of quantum mechanics has been assembled, one can begin to apply
1 Commutators (10 pts)
 Final Exam Solutions 37A Fall 0 I. Siddiqi / E. Dodds Commutators 0 pts) ) Consider the operator  = Ĵx Ĵ y + ĴyĴx where J i represents the total angular momentum in the ith direction. a) Express both
Final Exam Solutions 37A Fall 0 I. Siddiqi / E. Dodds Commutators 0 pts) ) Consider the operator  = Ĵx Ĵ y + ĴyĴx where J i represents the total angular momentum in the ith direction. a) Express both
Lecture 6. Four postulates of quantum mechanics. The eigenvalue equation. Momentum and energy operators. Dirac delta function. Expectation values
 Lecture 6 Four postulates of quantum mechanics The eigenvalue equation Momentum and energy operators Dirac delta function Expectation values Objectives Learn about eigenvalue equations and operators. Learn
Lecture 6 Four postulates of quantum mechanics The eigenvalue equation Momentum and energy operators Dirac delta function Expectation values Objectives Learn about eigenvalue equations and operators. Learn
Chapter 4 (Lecture 6-7) Schrodinger equation for some simple systems Table: List of various one dimensional potentials System Physical correspondence
 V, E, Chapter (Lecture 6-7) Schrodinger equation for some simple systems Table: List of various one dimensional potentials System Physical correspondence Potential Total Energies and Probability density
V, E, Chapter (Lecture 6-7) Schrodinger equation for some simple systems Table: List of various one dimensional potentials System Physical correspondence Potential Total Energies and Probability density
Quantum Mechanics for Scientists and Engineers. David Miller
 Quantum Mechanics for Scientists and Engineers David Miller Background mathematics 5 Sum, factorial and product notations Summation notation If we want to add a set of numbers a 1, a 2, a 3, and a 4, we
Quantum Mechanics for Scientists and Engineers David Miller Background mathematics 5 Sum, factorial and product notations Summation notation If we want to add a set of numbers a 1, a 2, a 3, and a 4, we
2. As we shall see, we choose to write in terms of σ x because ( X ) 2 = σ 2 x.
 Section 5.1 Simple One-Dimensional Problems: The Free Particle Page 9 The Free Particle Gaussian Wave Packets The Gaussian wave packet initial state is one of the few states for which both the { x } and
Section 5.1 Simple One-Dimensional Problems: The Free Particle Page 9 The Free Particle Gaussian Wave Packets The Gaussian wave packet initial state is one of the few states for which both the { x } and
Quantum Physics II (8.05) Fall 2002 Assignment 11
 Quantum Physics II (8.05) Fall 00 Assignment 11 Readings Most of the reading needed for this problem set was already given on Problem Set 9. The new readings are: Phase shifts are discussed in Cohen-Tannoudji
Quantum Physics II (8.05) Fall 00 Assignment 11 Readings Most of the reading needed for this problem set was already given on Problem Set 9. The new readings are: Phase shifts are discussed in Cohen-Tannoudji
In chapter 3, when we discussed metallic bonding, the primary attribute was that electrons are delocalized.
 Lecture 15: free electron gas Up to this point we have not discussed electrons explicitly, but electrons are the first offenders for most useful phenomena in materials. When we distinguish between metals,
Lecture 15: free electron gas Up to this point we have not discussed electrons explicitly, but electrons are the first offenders for most useful phenomena in materials. When we distinguish between metals,
The Schrodinger Wave Equation (Engel 2.4) In QM, the behavior of a particle is described by its wave function Ψ(x,t) which we get by solving:
 When do we use Quantum Mechanics? (Engel 2.1) Basically, when λ is close in magnitude to the dimensions of the problem, and to the degree that the system has a discrete energy spectrum The Schrodinger
When do we use Quantum Mechanics? (Engel 2.1) Basically, when λ is close in magnitude to the dimensions of the problem, and to the degree that the system has a discrete energy spectrum The Schrodinger
UNIVERSITY OF SURREY FACULTY OF ENGINEERING AND PHYSICAL SCIENCES DEPARTMENT OF PHYSICS. BSc and MPhys Undergraduate Programmes in Physics LEVEL HE2
 Phys/Level /1/9/Semester, 009-10 (1 handout) UNIVERSITY OF SURREY FACULTY OF ENGINEERING AND PHYSICAL SCIENCES DEPARTMENT OF PHYSICS BSc and MPhys Undergraduate Programmes in Physics LEVEL HE PAPER 1 MATHEMATICAL,
Phys/Level /1/9/Semester, 009-10 (1 handout) UNIVERSITY OF SURREY FACULTY OF ENGINEERING AND PHYSICAL SCIENCES DEPARTMENT OF PHYSICS BSc and MPhys Undergraduate Programmes in Physics LEVEL HE PAPER 1 MATHEMATICAL,
TP computing lab - Integrating the 1D stationary Schrödinger eq
 TP computing lab - Integrating the 1D stationary Schrödinger equation September 21, 2010 The stationary 1D Schrödinger equation The time-independent (stationary) Schrödinger equation is given by Eψ(x)
TP computing lab - Integrating the 1D stationary Schrödinger equation September 21, 2010 The stationary 1D Schrödinger equation The time-independent (stationary) Schrödinger equation is given by Eψ(x)
CHAPTER 6 Quantum Mechanics II
 CHAPTER 6 Quantum Mechanics II 6.1 6.2 6.3 6.4 6.5 6.6 6.7 The Schrödinger Wave Equation Expectation Values Infinite Square-Well Potential Finite Square-Well Potential Three-Dimensional Infinite-Potential
CHAPTER 6 Quantum Mechanics II 6.1 6.2 6.3 6.4 6.5 6.6 6.7 The Schrödinger Wave Equation Expectation Values Infinite Square-Well Potential Finite Square-Well Potential Three-Dimensional Infinite-Potential
Ae ikx Be ikx. Quantum theory: techniques and applications
 Quantum theory: techniques and applications There exist three basic modes of motion: translation, vibration, and rotation. All three play an important role in chemistry because they are ways in which molecules
Quantum theory: techniques and applications There exist three basic modes of motion: translation, vibration, and rotation. All three play an important role in chemistry because they are ways in which molecules
8.1 The hydrogen atom solutions
 8.1 The hydrogen atom solutions Slides: Video 8.1.1 Separating for the radial equation Text reference: Quantum Mechanics for Scientists and Engineers Section 10.4 (up to Solution of the hydrogen radial
8.1 The hydrogen atom solutions Slides: Video 8.1.1 Separating for the radial equation Text reference: Quantum Mechanics for Scientists and Engineers Section 10.4 (up to Solution of the hydrogen radial
Quantum Mechanics Solutions
 Quantum Mechanics Solutions (a (i f A and B are Hermitian, since (AB = B A = BA, operator AB is Hermitian if and only if A and B commute So, we know that [A,B] = 0, which means that the Hilbert space H
Quantum Mechanics Solutions (a (i f A and B are Hermitian, since (AB = B A = BA, operator AB is Hermitian if and only if A and B commute So, we know that [A,B] = 0, which means that the Hilbert space H
Quantum Mechanics-I Prof. Dr. S. Lakshmi Bala Department of Physics Indian Institute of Technology, Madras. Lecture - 21 Square-Integrable Functions
 Quantum Mechanics-I Prof. Dr. S. Lakshmi Bala Department of Physics Indian Institute of Technology, Madras Lecture - 21 Square-Integrable Functions (Refer Slide Time: 00:06) (Refer Slide Time: 00:14) We
Quantum Mechanics-I Prof. Dr. S. Lakshmi Bala Department of Physics Indian Institute of Technology, Madras Lecture - 21 Square-Integrable Functions (Refer Slide Time: 00:06) (Refer Slide Time: 00:14) We
Solution 01. Sut 25268
 Solution. Since this is an estimate, more than one solution is possible depending on the approximations made. One solution is given The object of this section is to estimate the the size of an atom. While
Solution. Since this is an estimate, more than one solution is possible depending on the approximations made. One solution is given The object of this section is to estimate the the size of an atom. While
UNIVERSITY OF MARYLAND Department of Physics College Park, Maryland. PHYSICS Ph.D. QUALIFYING EXAMINATION PART II
 UNIVERSITY OF MARYLAND Department of Physics College Park, Maryland PHYSICS Ph.D. QUALIFYING EXAMINATION PART II August 23, 208 9:00 a.m. :00 p.m. Do any four problems. Each problem is worth 25 points.
UNIVERSITY OF MARYLAND Department of Physics College Park, Maryland PHYSICS Ph.D. QUALIFYING EXAMINATION PART II August 23, 208 9:00 a.m. :00 p.m. Do any four problems. Each problem is worth 25 points.
One-electron Atom. (in spherical coordinates), where Y lm. are spherical harmonics, we arrive at the following Schrödinger equation:
 One-electron Atom The atomic orbitals of hydrogen-like atoms are solutions to the Schrödinger equation in a spherically symmetric potential. In this case, the potential term is the potential given by Coulomb's
One-electron Atom The atomic orbitals of hydrogen-like atoms are solutions to the Schrödinger equation in a spherically symmetric potential. In this case, the potential term is the potential given by Coulomb's
2. The Schrödinger equation for one-particle problems. 5. Atoms and the periodic table of chemical elements
 1 Historical introduction The Schrödinger equation for one-particle problems 3 Mathematical tools for quantum chemistry 4 The postulates of quantum mechanics 5 Atoms and the periodic table of chemical
1 Historical introduction The Schrödinger equation for one-particle problems 3 Mathematical tools for quantum chemistry 4 The postulates of quantum mechanics 5 Atoms and the periodic table of chemical
Lecture 13: Electromagnetic Theory Professor D. K. Ghosh, Physics Department, I.I.T., Bombay. Poisson s and Laplace s Equations
 Poisson s and Laplace s Equations Lecture 13: Electromagnetic Theory Professor D. K. Ghosh, Physics Department, I.I.T., Bombay We will spend some time in looking at the mathematical foundations of electrostatics.
Poisson s and Laplace s Equations Lecture 13: Electromagnetic Theory Professor D. K. Ghosh, Physics Department, I.I.T., Bombay We will spend some time in looking at the mathematical foundations of electrostatics.
Lecture 10: The Schrödinger Equation Lecture 10, p 1
 Lecture 10: The Schrödinger Equation Lecture 10, p 1 Overview Probability distributions Schrödinger s Equation Particle in a Bo Matter waves in an infinite square well Quantized energy levels y() U= n=1
Lecture 10: The Schrödinger Equation Lecture 10, p 1 Overview Probability distributions Schrödinger s Equation Particle in a Bo Matter waves in an infinite square well Quantized energy levels y() U= n=1
Opinions on quantum mechanics. CHAPTER 6 Quantum Mechanics II. 6.1: The Schrödinger Wave Equation. Normalization and Probability
 CHAPTER 6 Quantum Mechanics II 6.1 The Schrödinger Wave Equation 6. Expectation Values 6.3 Infinite Square-Well Potential 6.4 Finite Square-Well Potential 6.5 Three-Dimensional Infinite- 6.6 Simple Harmonic
CHAPTER 6 Quantum Mechanics II 6.1 The Schrödinger Wave Equation 6. Expectation Values 6.3 Infinite Square-Well Potential 6.4 Finite Square-Well Potential 6.5 Three-Dimensional Infinite- 6.6 Simple Harmonic
Tutorial for PhET Sim Quantum Bound States
 Tutorial for PhET Sim Quantum Bound States J. Mathias Weber * JILA and Department of Chemistry & Biochemistry, University of Colorado at Boulder With this PhET Sim, we will explore the properties of some
Tutorial for PhET Sim Quantum Bound States J. Mathias Weber * JILA and Department of Chemistry & Biochemistry, University of Colorado at Boulder With this PhET Sim, we will explore the properties of some
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer. Lecture 7, February 1, 2006
 Chem 350/450 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 006 Christopher J. Cramer ecture 7, February 1, 006 Solved Homework We are given that A is a Hermitian operator such that
Chem 350/450 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 006 Christopher J. Cramer ecture 7, February 1, 006 Solved Homework We are given that A is a Hermitian operator such that
The Schrodinger Equation and Postulates Common operators in QM: Potential Energy. Often depends on position operator: Kinetic Energy 1-D case:
 The Schrodinger Equation and Postulates Common operators in QM: Potential Energy Often depends on position operator: Kinetic Energy 1-D case: 3-D case Time Total energy = Hamiltonian To find out about
The Schrodinger Equation and Postulates Common operators in QM: Potential Energy Often depends on position operator: Kinetic Energy 1-D case: 3-D case Time Total energy = Hamiltonian To find out about
Generators for Continuous Coordinate Transformations
 Page 636 Lecture 37: Coordinate Transformations: Continuous Passive Coordinate Transformations Active Coordinate Transformations Date Revised: 2009/01/28 Date Given: 2009/01/26 Generators for Continuous
Page 636 Lecture 37: Coordinate Transformations: Continuous Passive Coordinate Transformations Active Coordinate Transformations Date Revised: 2009/01/28 Date Given: 2009/01/26 Generators for Continuous
Electrons in a periodic potential
 Chapter 3 Electrons in a periodic potential 3.1 Bloch s theorem. We consider in this chapter electrons under the influence of a static, periodic potential V (x), i.e. such that it fulfills V (x) = V (x
Chapter 3 Electrons in a periodic potential 3.1 Bloch s theorem. We consider in this chapter electrons under the influence of a static, periodic potential V (x), i.e. such that it fulfills V (x) = V (x
7.3 The hydrogen atom
 7.3 The hydrogen atom Slides: Video 7.3.1 Multiple particle wavefunctions Text reference: Quantum Mechanics for Scientists and Engineers Chapter 10 introduction and Section 10.1 The hydrogen atom Multiple
7.3 The hydrogen atom Slides: Video 7.3.1 Multiple particle wavefunctions Text reference: Quantum Mechanics for Scientists and Engineers Chapter 10 introduction and Section 10.1 The hydrogen atom Multiple
General Physical Chemistry II
 General Physical Chemistry II Lecture 3 Aleksey Kocherzhenko September 2, 2014" Last time " The time-independent Schrödinger equation" Erwin Schrödinger " ~ 2 2m d 2 (x) dx 2 The wavefunction:" (x) The
General Physical Chemistry II Lecture 3 Aleksey Kocherzhenko September 2, 2014" Last time " The time-independent Schrödinger equation" Erwin Schrödinger " ~ 2 2m d 2 (x) dx 2 The wavefunction:" (x) The
SCATTERING. V 0 x a. V V x a. d Asin kx 2m dx. 2mE k. Acos x or Asin. A cosh x or Asinh
 SCATTERING Consider a particle of mass m moving along the x axis with energy E. It encounters a potential given by V0 x a VV x a 0 In the region x > a we have with d E Asinkx m dx In the region x < a we
SCATTERING Consider a particle of mass m moving along the x axis with energy E. It encounters a potential given by V0 x a VV x a 0 In the region x > a we have with d E Asinkx m dx In the region x < a we
CHAPTER 6 Quantum Mechanics II
 CHAPTER 6 Quantum Mechanics II 6.1 The Schrödinger Wave Equation 6.2 Expectation Values 6.3 Infinite Square-Well Potential 6.4 Finite Square-Well Potential 6.5 Three-Dimensional Infinite-Potential Well
CHAPTER 6 Quantum Mechanics II 6.1 The Schrödinger Wave Equation 6.2 Expectation Values 6.3 Infinite Square-Well Potential 6.4 Finite Square-Well Potential 6.5 Three-Dimensional Infinite-Potential Well
C/CS/Phys C191 Particle-in-a-box, Spin 10/02/08 Fall 2008 Lecture 11
 C/CS/Phys C191 Particle-in-a-box, Spin 10/0/08 Fall 008 Lecture 11 Last time we saw that the time dependent Schr. eqn. can be decomposed into two equations, one in time (t) and one in space (x): space
C/CS/Phys C191 Particle-in-a-box, Spin 10/0/08 Fall 008 Lecture 11 Last time we saw that the time dependent Schr. eqn. can be decomposed into two equations, one in time (t) and one in space (x): space
Notes on wavefunctions IV: the Schrödinger equation in a potential and energy eigenstates.
 Notes on wavefunctions IV: the Schrödinger equation in a potential and energy eigenstates. We have now seen that the wavefunction for a free electron changes with time according to the Schrödinger Equation
Notes on wavefunctions IV: the Schrödinger equation in a potential and energy eigenstates. We have now seen that the wavefunction for a free electron changes with time according to the Schrödinger Equation
The free electron gas: Sommerfeld model
 The free electron gas: Sommerfeld model Valence electrons: delocalized gas of free electrons (selectrons of alkalies) which is not perturbed by the ion cores (ionic radius of Na + equals 0.97 Å, internuclear
The free electron gas: Sommerfeld model Valence electrons: delocalized gas of free electrons (selectrons of alkalies) which is not perturbed by the ion cores (ionic radius of Na + equals 0.97 Å, internuclear
Lecture-XXVI. Time-Independent Schrodinger Equation
 Lecture-XXVI Time-Independent Schrodinger Equation Time Independent Schrodinger Equation: The time-dependent Schrodinger equation: Assume that V is independent of time t. In that case the Schrodinger equation
Lecture-XXVI Time-Independent Schrodinger Equation Time Independent Schrodinger Equation: The time-dependent Schrodinger equation: Assume that V is independent of time t. In that case the Schrodinger equation
If electrons moved in simple orbits, p and x could be determined, but this violates the Heisenberg Uncertainty Principle.
 CHEM 2060 Lecture 18: Particle in a Box L18-1 Atomic Orbitals If electrons moved in simple orbits, p and x could be determined, but this violates the Heisenberg Uncertainty Principle. We can only talk
CHEM 2060 Lecture 18: Particle in a Box L18-1 Atomic Orbitals If electrons moved in simple orbits, p and x could be determined, but this violates the Heisenberg Uncertainty Principle. We can only talk
atoms and light. Chapter Goal: To understand the structure and properties of atoms.
 Quantum mechanics provides us with an understanding of atomic structure and atomic properties. Lasers are one of the most important applications of the quantummechanical properties of atoms and light.
Quantum mechanics provides us with an understanding of atomic structure and atomic properties. Lasers are one of the most important applications of the quantummechanical properties of atoms and light.
This is known as charge quantization. Neutral particles, like neutron and photon have zero charge.
 Module 2: Electrostatics Lecture 6: Quantization of Charge Objectives In this lecture you will learn the following Quantization Of Charge and its measurement Coulomb's Law of force between electric charge
Module 2: Electrostatics Lecture 6: Quantization of Charge Objectives In this lecture you will learn the following Quantization Of Charge and its measurement Coulomb's Law of force between electric charge
Lecture 10: Solving the Time-Independent Schrödinger Equation. 1 Stationary States 1. 2 Solving for Energy Eigenstates 3
 Contents Lecture 1: Solving the Time-Independent Schrödinger Equation B. Zwiebach March 14, 16 1 Stationary States 1 Solving for Energy Eigenstates 3 3 Free particle on a circle. 6 1 Stationary States
Contents Lecture 1: Solving the Time-Independent Schrödinger Equation B. Zwiebach March 14, 16 1 Stationary States 1 Solving for Energy Eigenstates 3 3 Free particle on a circle. 6 1 Stationary States
PHYS 502 Lecture 8: Legendre Functions. Dr. Vasileios Lempesis
 PHYS 502 Lecture 8: Legendre Functions Dr. Vasileios Lempesis Introduction Legendre functions or Legendre polynomials are the solutions of Legendre s differential equation that appear when we separate
PHYS 502 Lecture 8: Legendre Functions Dr. Vasileios Lempesis Introduction Legendre functions or Legendre polynomials are the solutions of Legendre s differential equation that appear when we separate
PDEs in Spherical and Circular Coordinates
 Introduction to Partial Differential Equations part of EM, Scalar and Vector Fields module (PHY2064) This lecture Laplacian in spherical & circular polar coordinates Laplace s PDE in electrostatics Schrödinger
Introduction to Partial Differential Equations part of EM, Scalar and Vector Fields module (PHY2064) This lecture Laplacian in spherical & circular polar coordinates Laplace s PDE in electrostatics Schrödinger
Separation of Variables in Linear PDE: One-Dimensional Problems
 Separation of Variables in Linear PDE: One-Dimensional Problems Now we apply the theory of Hilbert spaces to linear differential equations with partial derivatives (PDE). We start with a particular example,
Separation of Variables in Linear PDE: One-Dimensional Problems Now we apply the theory of Hilbert spaces to linear differential equations with partial derivatives (PDE). We start with a particular example,
Quantum Mechanics in Three Dimensions
 8 Quantum Mechanics in Three Dimensions Chapter Outline 8.1 Particle in a Three-Dimensional Box 8. Central Forces and Angular Momentum 8.3 Space Quantization 8.4 Quantization of Angular Momentum and Energy
8 Quantum Mechanics in Three Dimensions Chapter Outline 8.1 Particle in a Three-Dimensional Box 8. Central Forces and Angular Momentum 8.3 Space Quantization 8.4 Quantization of Angular Momentum and Energy
Time part of the equation can be separated by substituting independent equation
 Lecture 9 Schrödinger Equation in 3D and Angular Momentum Operator In this section we will construct 3D Schrödinger equation and we give some simple examples. In this course we will consider problems where
Lecture 9 Schrödinger Equation in 3D and Angular Momentum Operator In this section we will construct 3D Schrödinger equation and we give some simple examples. In this course we will consider problems where
Project: Vibration of Diatomic Molecules
 Project: Vibration of Diatomic Molecules Objective: Find the vibration energies and the corresponding vibrational wavefunctions of diatomic molecules H 2 and I 2 using the Morse potential. equired Numerical
Project: Vibration of Diatomic Molecules Objective: Find the vibration energies and the corresponding vibrational wavefunctions of diatomic molecules H 2 and I 2 using the Morse potential. equired Numerical
Lecture 12: Particle in 1D boxes & Simple Harmonic Oscillator
 Lecture 12: Particle in 1D boxes & Simple Harmonic Oscillator U(x) E Dx y(x) x Dx Lecture 12, p 1 Properties of Bound States Several trends exhibited by the particle-in-box states are generic to bound
Lecture 12: Particle in 1D boxes & Simple Harmonic Oscillator U(x) E Dx y(x) x Dx Lecture 12, p 1 Properties of Bound States Several trends exhibited by the particle-in-box states are generic to bound
1 Solutions in cylindrical coordinates: Bessel functions
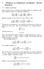 1 Solutions in cylindrical coordinates: Bessel functions 1.1 Bessel functions Bessel functions arise as solutions of potential problems in cylindrical coordinates. Laplace s equation in cylindrical coordinates
1 Solutions in cylindrical coordinates: Bessel functions 1.1 Bessel functions Bessel functions arise as solutions of potential problems in cylindrical coordinates. Laplace s equation in cylindrical coordinates
Physics 218 Quantum Mechanics I Assignment 6
 Physics 218 Quantum Mechanics I Assignment 6 Logan A. Morrison February 17, 2016 Problem 1 A non-relativistic beam of particles each with mass, m, and energy, E, which you can treat as a plane wave, is
Physics 218 Quantum Mechanics I Assignment 6 Logan A. Morrison February 17, 2016 Problem 1 A non-relativistic beam of particles each with mass, m, and energy, E, which you can treat as a plane wave, is
Properties of Commutators and Schroedinger Operators and Applications to Quantum Computing
 International Journal of Engineering and Advanced Research Technology (IJEART) Properties of Commutators and Schroedinger Operators and Applications to Quantum Computing N. B. Okelo Abstract In this paper
International Journal of Engineering and Advanced Research Technology (IJEART) Properties of Commutators and Schroedinger Operators and Applications to Quantum Computing N. B. Okelo Abstract In this paper
Harmonic Oscillator with raising and lowering operators. We write the Schrödinger equation for the harmonic oscillator in one dimension as follows:
 We write the Schrödinger equation for the harmonic oscillator in one dimension as follows: H ˆ! = "!2 d 2! + 1 2µ dx 2 2 kx 2! = E! T ˆ = "! 2 2µ d 2 dx 2 V ˆ = 1 2 kx 2 H ˆ = ˆ T + ˆ V (1) where µ is
We write the Schrödinger equation for the harmonic oscillator in one dimension as follows: H ˆ! = "!2 d 2! + 1 2µ dx 2 2 kx 2! = E! T ˆ = "! 2 2µ d 2 dx 2 V ˆ = 1 2 kx 2 H ˆ = ˆ T + ˆ V (1) where µ is
Harmonic Oscillator I
 Physics 34 Lecture 7 Harmonic Oscillator I Lecture 7 Physics 34 Quantum Mechanics I Monday, February th, 008 We can manipulate operators, to a certain extent, as we would algebraic expressions. By considering
Physics 34 Lecture 7 Harmonic Oscillator I Lecture 7 Physics 34 Quantum Mechanics I Monday, February th, 008 We can manipulate operators, to a certain extent, as we would algebraic expressions. By considering
The Hydrogen Atom. Nucleus charge +Ze mass m 1 coordinates x 1, y 1, z 1. Electron charge e mass m 2 coordinates x 2, y 2, z 2
 The Hydrogen Ato The only ato that can be solved exactly. The results becoe the basis for understanding all other atos and olecules. Orbital Angular Moentu Spherical Haronics Nucleus charge +Ze ass coordinates
The Hydrogen Ato The only ato that can be solved exactly. The results becoe the basis for understanding all other atos and olecules. Orbital Angular Moentu Spherical Haronics Nucleus charge +Ze ass coordinates
The 3 dimensional Schrödinger Equation
 Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
Time-Independent Perturbation Theory
 4 Phys46.nb Time-Independent Perturbation Theory.. Overview... General question Assuming that we have a Hamiltonian, H = H + λ H (.) where λ is a very small real number. The eigenstates of the Hamiltonian
4 Phys46.nb Time-Independent Perturbation Theory.. Overview... General question Assuming that we have a Hamiltonian, H = H + λ H (.) where λ is a very small real number. The eigenstates of the Hamiltonian
Brief review of Quantum Mechanics (QM)
 Brief review of Quantum Mechanics (QM) Note: This is a collection of several formulae and facts that we will use throughout the course. It is by no means a complete discussion of QM, nor will I attempt
Brief review of Quantum Mechanics (QM) Note: This is a collection of several formulae and facts that we will use throughout the course. It is by no means a complete discussion of QM, nor will I attempt
E = hν light = hc λ = ( J s)( m/s) m = ev J = ev
 Problem The ionization potential tells us how much energy we need to use to remove an electron, so we know that any energy left afterwards will be the kinetic energy of the ejected electron. So first we
Problem The ionization potential tells us how much energy we need to use to remove an electron, so we know that any energy left afterwards will be the kinetic energy of the ejected electron. So first we
Quantum mechanics (QM) deals with systems on atomic scale level, whose behaviours cannot be described by classical mechanics.
 A 10-MINUTE RATHER QUICK INTRODUCTION TO QUANTUM MECHANICS 1. What is quantum mechanics (as opposed to classical mechanics)? Quantum mechanics (QM) deals with systems on atomic scale level, whose behaviours
A 10-MINUTE RATHER QUICK INTRODUCTION TO QUANTUM MECHANICS 1. What is quantum mechanics (as opposed to classical mechanics)? Quantum mechanics (QM) deals with systems on atomic scale level, whose behaviours
Harmonic Oscillator Eigenvalues and Eigenfunctions
 Chemistry 46 Fall 217 Dr. Jean M. Standard October 4, 217 Harmonic Oscillator Eigenvalues and Eigenfunctions The Quantum Mechanical Harmonic Oscillator The quantum mechanical harmonic oscillator in one
Chemistry 46 Fall 217 Dr. Jean M. Standard October 4, 217 Harmonic Oscillator Eigenvalues and Eigenfunctions The Quantum Mechanical Harmonic Oscillator The quantum mechanical harmonic oscillator in one
Physics 221A Fall 2017 Notes 27 The Variational Method
 Copyright c 2018 by Robert G. Littlejohn Physics 221A Fall 2017 Notes 27 The Variational Method 1. Introduction Very few realistic problems in quantum mechanics are exactly solvable, so approximation methods
Copyright c 2018 by Robert G. Littlejohn Physics 221A Fall 2017 Notes 27 The Variational Method 1. Introduction Very few realistic problems in quantum mechanics are exactly solvable, so approximation methods
Solutions Final exam 633
 Solutions Final exam 633 S.J. van Enk (Dated: June 9, 2008) (1) [25 points] You have a source that produces pairs of spin-1/2 particles. With probability p they are in the singlet state, ( )/ 2, and with
Solutions Final exam 633 S.J. van Enk (Dated: June 9, 2008) (1) [25 points] You have a source that produces pairs of spin-1/2 particles. With probability p they are in the singlet state, ( )/ 2, and with
6.2 Unitary and Hermitian operators
 6.2 Unitary and Hermitian operators Slides: Video 6.2.1 Using unitary operators Text reference: Quantum Mechanics for Scientists and Engineers Section 4.10 (starting from Changing the representation of
6.2 Unitary and Hermitian operators Slides: Video 6.2.1 Using unitary operators Text reference: Quantum Mechanics for Scientists and Engineers Section 4.10 (starting from Changing the representation of
Physics 342 Lecture 30. Solids. Lecture 30. Physics 342 Quantum Mechanics I
 Physics 342 Lecture 30 Solids Lecture 30 Physics 342 Quantum Mechanics I Friday, April 18th, 2008 We can consider simple models of solids these highlight some special techniques. 30.1 An Electron in a
Physics 342 Lecture 30 Solids Lecture 30 Physics 342 Quantum Mechanics I Friday, April 18th, 2008 We can consider simple models of solids these highlight some special techniques. 30.1 An Electron in a
Joint Entrance Examination for Postgraduate Courses in Physics EUF
 Joint Entrance Examination for Postgraduate Courses in Physics EUF Second Semester 013 Part 1 3 April 013 Instructions: DO NOT WRITE YOUR NAME ON THE TEST. It should be identified only by your candidate
Joint Entrance Examination for Postgraduate Courses in Physics EUF Second Semester 013 Part 1 3 April 013 Instructions: DO NOT WRITE YOUR NAME ON THE TEST. It should be identified only by your candidate
CHMY 564 Homework #3 2Feb17 Due Friday, 10Feb17
 564-17 Lec 11 Mon 6feb17 CHMY 564 Homework #3 2Feb17 Due Friday, 10Feb17 1. (a) Find the eigenfunctions and eigenvalues of the x component of linear momentum. (b) Why can a momentum eigenfunction never
564-17 Lec 11 Mon 6feb17 CHMY 564 Homework #3 2Feb17 Due Friday, 10Feb17 1. (a) Find the eigenfunctions and eigenvalues of the x component of linear momentum. (b) Why can a momentum eigenfunction never
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer. Lecture 9, February 8, 2006
 Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer Lecture 9, February 8, 2006 The Harmonic Oscillator Consider a diatomic molecule. Such a molecule
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer Lecture 9, February 8, 2006 The Harmonic Oscillator Consider a diatomic molecule. Such a molecule
1 Introduction. Green s function notes 2018
 Green s function notes 8 Introduction Back in the "formal" notes, we derived the potential in terms of the Green s function. Dirichlet problem: Equation (7) in "formal" notes is Φ () Z ( ) ( ) 3 Z Φ (
Green s function notes 8 Introduction Back in the "formal" notes, we derived the potential in terms of the Green s function. Dirichlet problem: Equation (7) in "formal" notes is Φ () Z ( ) ( ) 3 Z Φ (
QUANTUM MECHANICS A (SPA 5319) The Finite Square Well
 QUANTUM MECHANICS A (SPA 5319) The Finite Square Well We have already solved the problem of the infinite square well. Let us now solve the more realistic finite square well problem. Consider the potential
QUANTUM MECHANICS A (SPA 5319) The Finite Square Well We have already solved the problem of the infinite square well. Let us now solve the more realistic finite square well problem. Consider the potential
LECTURE 10: REVIEW OF POWER SERIES. 1. Motivation
 LECTURE 10: REVIEW OF POWER SERIES By definition, a power series centered at x 0 is a series of the form where a 0, a 1,... and x 0 are constants. For convenience, we shall mostly be concerned with the
LECTURE 10: REVIEW OF POWER SERIES By definition, a power series centered at x 0 is a series of the form where a 0, a 1,... and x 0 are constants. For convenience, we shall mostly be concerned with the
one-dimensional box with harmonic interaction
 On the symmetry of four particles in a arxiv:1607.00977v [quant-ph] 8 Jul 016 one-dimensional box with harmonic interaction Francisco M. Fernández INIFTA (CONICET, UNLP), División Química Teórica Blvd.
On the symmetry of four particles in a arxiv:1607.00977v [quant-ph] 8 Jul 016 one-dimensional box with harmonic interaction Francisco M. Fernández INIFTA (CONICET, UNLP), División Química Teórica Blvd.
Physics 342 Lecture 23. Radial Separation. Lecture 23. Physics 342 Quantum Mechanics I
 Physics 342 Lecture 23 Radial Separation Lecture 23 Physics 342 Quantum Mechanics I Friday, March 26th, 2010 We begin our spherical solutions with the simplest possible case zero potential. Aside from
Physics 342 Lecture 23 Radial Separation Lecture 23 Physics 342 Quantum Mechanics I Friday, March 26th, 2010 We begin our spherical solutions with the simplest possible case zero potential. Aside from
1 Mathematical Preliminaries
 Mathematical Preliminaries We shall go through in this first chapter all of the mathematics needed for reading the rest of this book. The reader is expected to have taken a one year course in differential
Mathematical Preliminaries We shall go through in this first chapter all of the mathematics needed for reading the rest of this book. The reader is expected to have taken a one year course in differential
Quantum Physics 130A. April 1, 2006
 Quantum Physics 130A April 1, 2006 2 1 HOMEWORK 1: Due Friday, Apr. 14 1. A polished silver plate is hit by beams of photons of known energy. It is measured that the maximum electron energy is 3.1 ± 0.11
Quantum Physics 130A April 1, 2006 2 1 HOMEWORK 1: Due Friday, Apr. 14 1. A polished silver plate is hit by beams of photons of known energy. It is measured that the maximum electron energy is 3.1 ± 0.11
