COPYRIGHTED MATERIAL. Production of Net Magnetization. Chapter 1
|
|
|
- Winfred Green
- 5 years ago
- Views:
Transcription
1 Chapter 1 Production of Net Magnetization Magnetic resonance (MR) is a measurement technique used to examine atoms and molecules. It is based on the interaction between an applied magnetic field and a particle that possesses spin. While electrons and other subatomic particles possess spin (or more precisely, spin angular momentum) and can be examined using MR techniques, this book focuses on nuclei and the use of MR techniques for their study, formally known as nuclear magnetic resonance (NMR). Nuclear spin, or more precisely, nuclear spin angular momentum, is one of several intrinsic properties of an atom, and its value depends on the precise atomic composition. Every element in the periodic table except argon and cerium has at least one naturally occurring isotope that possesses spin. Thus, in principle, nearly every element can be examined using MR, and the basic ideas of resonance absorption and relaxation are common for all of these elements. The precise details will vary from nucleus to nucleus and from system to system. The structure of an atom is an essential component of the MR experiment. Atoms consist of three fundamental particles: protons, which possess a positive charge; neutrons, which have no charge; and electrons, which have a negative charge. The protons and neutrons are located in the nucleus or core of an atom, and the electrons are located in shells or orbitals surrounding the nucleus. The characteristic chemical reactions of elements depend on the particular number of each of these particles. The properties most commonly COPYRIGHTED MATERIAL MRI: Basic Principles and Applications, Fourth Edition, By Mark A. Brown and Richard C. Semelka Copyright 2010 Wiley-Blackwell 1
2 2 PRODUCTION OF NET MAGNETIZATION N B S (a) (b) Figure 1-1. A rotating nucleus (spin) with a positive charge produces a magnetic fi eld known as the magnetic moment oriented parallel to the axis of rotation (a). This arrangement is analogous to a bar magnet in which the magnetic fi eld is considered to be oriented from the south to the north pole (b). used to categorize elements are the atomic number and the atomic weight. The atomic number is the number of protons in the nucleus and is the primary index used to differentiate atoms. All atoms of an element have the same atomic number and undergo the same chemical reactions. The atomic weight is the sum of the number of protons and the number of neutrons. Atoms with the same atomic number but different atomic weights are called isotopes. Isotopes of an element will undergo the same chemical reactions but at different reaction rates. A third property of the nucleus is spin, or intrinsic spin angular momentum. Nuclei with spin can be considered to be constantly rotating about an axis at a constant rate or velocity. This self - rotation axis is perpendicular to the direction of rotation (Figure 1-1 ). A limited number of values for the spin are found in nature; that is, the spin, I, is quantized to certain discrete values, which depend on the atomic number and atomic weight of the particular nucleus. There are three groups of values for I : zero, integral, and half - integral values. A nucleus has no spin ( I = 0) if it has an even - number atomic weight and an even atomic number: for example, 12 C (6 protons and 6 neutrons) or 16 O (8 protons and 8 neutrons). Such a nucleus does not interact with an external magnetic field and cannot be studied using MR. A nucleus has an integral value for I (e.g., 1, 2, 3) if it has an even atomic weight and an odd atomic number: for example, 2 H (1 proton and 1 neutron) or 6 Li (3 protons and 3 neutrons). A nucleus has a half - integral value for I (e.g., 1/2, 3/2, 5/2) if it has an odd atomic weight. Table 1-1 lists the spin and isotopic composition for several elements commonly found in biological systems. The 1 H nucleus, consisting of a single proton, is a natural choice for probing the body using MR
3 PRODUCTION OF NET MAGNETIZATION 3 TABLE 1-1 Constants for Selected Nuclei of Biological Interest Element Nuclear Composition Protons Neutrons Nuclear Spin, I Gyromagnetic Ratio, γ (MHz T 1 ) % Natural Abundance ω at 1.5 T (MHz) 1 H, protium 1 0 1/ H, deuterium He 2 1 1/ Li Li 3 4 3/ C C 6 7 1/ N N 7 8 1/ O O 8 9 5/ F / Na / P / Xe / Source: Adapted from Mills (1989). techniques, for several reasons. It has a spin of 1/2 and is the most abundant isotope for hydrogen. Its response to an applied magnetic field is one of the largest found in nature. Since the body is composed of tissues that contain primarily water and fat, both of which contain hydrogen, a significant MR signal can be produced naturally by normal tissues. Whereas a rigorous mathematical description of a nucleus with spin and its interactions requires the use of quantum mechanical principles, most of MR can be described using the concepts of classical mechanics, particularly in describing the actions of a nucleus with spin. Subsequent discussions of MR phenomena in this book use a classical approach. In addition, although the concepts of resonance absorption and relaxation apply to all nuclei with spin, the descriptions in this book focus on 1 H (commonly referred to as a proton ) since most imaging experiments visualize the 1 H nucleus. Recall that the nucleus is the location of the protons. For many nuclei, the arrangement of the protons is asymmetric. When this asymmetric charge arrangement rotates due to the nuclear spin, a local magnetic field or magnetic moment is induced about the nucleus. This magnetic moment will be oriented parallel to the axis of rotation. Since the nuclear spin is constant in magnitude and orientation or direction, its associated magnetic moment will also be constant in magnitude and orientation. This magnetic moment is fundamental to MR. A bar magnet provides a useful analogy. A bar magnet has a north and a south pole, or more precisely, a magnitude and an orientation to the magnetic field can be defined. The axis of rotation for a nucleus with spin can be viewed as a vector with a definite orientation and magnitude (Figure 1-1 ). This orientation of the nuclear spin and the changes induced in it due to the
4 4 PRODUCTION OF NET MAGNETIZATION 0 Figure 1-2. Microscopic and macroscopic pictures of a collection of spins in the absence of an external magnetic fi eld. In the absence of a magnetic fi eld, the spins have their axes oriented randomly (microscopic picture, left side of fi gure). The vector sum of these spin vectors is zero (macroscopic picture, right side). experimental manipulations that the nucleus undergoes provide the basis for the MR signal. In general, MR measurements are made on collections of similar spins rather than on an individual spin. It is convenient to consider such a collection both as individual spins acting independently (a microscopic picture) and as a single entity (a macroscopic picture). For many concepts, the two pictures provide equivalent results, even though the microscopic picture is more complete. Conversion between the two pictures requires the principles of statistical mechanics. Although necessary for a complete understanding of MR phenomena, the nature of this conversion is beyond the scope of this book. However, the macroscopic picture is sufficient for an adequate description of most concepts presented in this book. When necessary, the microscopic picture will be used. Consider an arbitrary volume of tissue containing hydrogen atoms (protons). Each proton has a spin vector of equal magnitude. However, the spin vectors for the entire collection of protons within the tissue are oriented randomly in all directions; there is a continuous distribution of the spin orientations. Performing a vector addition (head to toe) of these spin vectors produces a zero sum; that is, no net magnetization is observed in the tissue (Figure 1-2 ). If the tissue is placed inside a magnetic field B 0, 1 the individual protons begin to rotate perpendicular to, or precess about, the magnetic field. The spin vectors for the protons are tilted slightly away from the axis of the magnetic field, but their axis of precession is parallel to B 0. This precession is at a constant rate and occurs because of the interaction of the magnetic field with the spinning positive charge of the nucleus. By convention, B 0 and the axis of precession are defined to be oriented in the z direction of a Cartesian coordinate system. (This convention is not followed universally, but it is the 1 In this book, vector quantities with direction and magnitude are indicated by boldface italic type, while scalar quantities that are magnitude only are indicated by italic type.
5 PRODUCTION OF NET MAGNETIZATION 5 z w 0 B 0 z component y y component x component x Figure 1-3. Inside a magnetic fi eld, a proton precesses or revolves about the magnetic fi eld. The precessional axis is parallel to the main magnetic fi eld, B 0. The z component of the spin vector (projection of the spin onto the z axis) is the component of interest because it does not change in magnitude or direction as the proton precesses. The x and y components vary with time at a frequency ω 0 proportional to B 0 as expressed by equation (1-1). prevailing convention.) The motion of each proton can be described by a unique set of coordinates perpendicular ( x and y ) and parallel ( z ) to B 0. The perpendicular or transverse coordinates are nonzero but vary with time as the proton precesses, while the parallel or longitudinal coordinate is constant with time (Figure 1-3 ). The rate or frequency of precession is proportional to the strength of the magnetic field and is expressed by the Larmor equation : γ 0 ω0 = B 2π (1-1) where ω 0 is the Larmor frequency in megahertz (MHz), 2 B 0 is the magnetic field strength in tesla (T) that the proton experiences, and γ is a constant for each nucleus in s 1 T 1, known as the gyromagnetic ratio. Values for γ and ω at 1.5 T for several nuclei are tabulated in Table The factor 2π in equation (1-1) is necessary to convert from angular to cyclical frequency. In many discussions of MR, ω (Greek letter omega) is used to represent angular frequency, with units of s 1, while cyclical frequency, in units of hertz, is represented by either ν (Greek letter nu) or f. The Larmor equation is more properly written as ν 0 = γ B 0 /2π. In imaging derivations, the Larmor equation is usually expressed as equation (1-1) using ω but with units of hertz. To minimize confusion, we follow the imaging tradition throughout this book.
6 6 PRODUCTION OF NET MAGNETIZATION Higher Energy B 0 ΔE Lower Energy Figure 1-4. Zeeman diagram. In the absence of a magnetic fi eld (left side of fi gure), a collection of spins will have the confi gurations of z components equal in energy so that there is no preferential alignment between the spin - up and spin - down orientations. In the presence of a magnetic fi eld (right side), the spin - up orientation (parallel to B 0 ) is of lower energy and its confi guration contains more spins than does the higher - energy spin - down confi guration. The difference in energy Δ E between the two levels is proportional to B 0. If a vector addition is performed, as before, for the spin vectors inside the magnetic field, the results will be slightly different than those for the sum outside the field. In the direction perpendicular to B 0, the spin orientations are still randomly distributed just as they were outside the magnetic field, despite the time - varying nature of each transverse component. There is still no net magnetization perpendicular to B 0. However, in the direction parallel to B 0, there is a different result. Because there is an orientation to the precessional axis of the proton that is constant with time, there is a constant, nonzero interaction or coupling between the proton and B 0, known as the Zeeman interaction. Instead of a continuous range of values, this coupling causes the z component to be quantized to a limited or discrete number of values. For the 1 H nucleus, there are only two possible values for the z component: parallel or along B 0 and antiparallel or against B 0. This coupling also causes a difference in energy Δ E between these two orientations that is proportional to B 0 (Figure 1-4 ). The result of the Zeeman interaction is that spins in the two orientations, parallel (also known as spin up ) and antiparallel ( spin down ), have different energies. Those spins oriented parallel to B 0 are of lower energy than those oriented antiparallel. For a collection of protons, more will be oriented parallel to B 0 than antiparallel; that is, there is residual polarization of the spins induced parallel to the magnetic field (Figure 1-5 a). The exact number of protons in each energy level can be predicted by a distribution function known as the Boltzmann distribution : N N UPPER LOWER = e Δ EkT (1-2) where k is Boltzmann s constant, J K 1 and N UPPER and N LOWER are the number of protons in the upper and lower energy levels, respectively.
7 PRODUCTION OF NET MAGNETIZATION 7 z z y y B 0 B 0 M x x (a) (b) Figure 1-5. Microscopic (a) and macroscopic (b) pictures of a collection of spins in the presence of an external magnetic fi eld. Each spin precesses about the magnetic fi eld. If a rotating frame of reference is used with a rotation rate of ω 0, the collection of protons appears stationary. Whereas the z components are one of two values (one positive and one negative), the x and y components can be any value, positive or negative. The spins will appear to track along two cones, one with a positive z component and one with a negative z component. Because there are more spins in the upper cone, there will be a nonzero vector sum M 0, the net magnetization. It will be of constant magnitude and parallel to B 0. Since the separation between the energy levels Δ E depends on the field strength B 0, the exact number of spins in each level also depends on B 0 and increases with increasing B 0. For a collection of protons at body temperature (310 K) at 1.5 T, there will typically be an excess of about protons in the lower level out of the approximately protons within the tissue. This unequal number of protons in each energy level means that the vector sum of spins will be nonzero and will point along the magnetic field. In other words, the tissue will become polarized or magnetized in the presence of B 0 with a value M 0, known as the net magnetization. The orientation of this net magnetization will be in the same direction as B 0 and will be constant with respect to time (Figure 1-5 b). For tissues in the body, the magnitude of M 0 is proportional to B 0 : M = χ B (1-3) 0 0 where χ is known as the bulk magnetic susceptibility or simply the magnetic susceptibility. This arrangement with M 0 aligned along the magnetic field with no transverse component is the normal, or equilibrium, configuration for the protons. This configuration of spins has the lowest energy and is the arrangement to which the protons will naturally try to return following any perturbations, such as energy absorption. This induced magnetization, M 0, is the source of signal for all of the MR experiments. Consequently, all other things being equal, the greater the field strength, the greater the value of M 0 and the greater the potential MR signal.
8 8 PRODUCTION OF NET MAGNETIZATION The magnetic susceptibility describes the response of a substance to the applied magnetic field. Although a complete analysis of the origin of this response is beyond the scope of the book, it is useful to describe the three levels of response that are encountered in MR. A diamagnetic response is found in all materials, arising from the electrons surrounding the nuclei. It is a very weak response ( χ is very small) and is the only response for most materials, including tissue. A paramagnetic response is larger in magnitude than a diamagnetic response but is still relatively weak ( χ is small). It is found in molecules where there are unpaired electrons. The final response level is known as ferromagnetic, typically found in certain metals, in which χ is very large. Whereas diamagnetic and paramagnetic materials are safe to be in or near MR scanners, ferromagnetic materials should remain outside the scan room. An alternative picture known as a rotating frame of reference or rotating coordinate system is often used in MR. It is a convenient view when describing objects that undergo rotational motion. When viewed in a rotating frame of reference, the coordinate system rotates about one axis while the other two axes vary with time. By choosing a suitable axis and rate of rotation for the coordinate system, the moving object appears stationary. For MR experiments, a convenient rotating frame uses the z axis, parallel to B 0, as the axis of rotation, while the x and y axes rotate at the Larmor frequency, ω 0. When viewed in this fashion, the precessing spin appears stationary in space with a fixed set of x, y, and z coordinates. If the entire collection of protons in the tissue volume is examined, a complete range of x and y values will be found, both positive and negative, but only two z values. There will be an equal number of positive and negative x and y values but a slight excess of positive z values, as just described. If a vector sum is performed on this collection of protons, the x and y components sum to zero but a nonzero, positive z component will be left, the net magnetization M 0. In addition, since the z axis is the axis of rotation, M 0 does not vary with time. Regardless of whether a stationary or fixed coordinate system is used, M 0 is of fixed amplitude and is parallel to the main magnetic field. For all subsequent discussions in this book, a rotating frame of reference with the rotation axis parallel to B 0 is used when describing the motion of the protons.
Fundamental MRI Principles Module 2 N. Nuclear Magnetic Resonance. X-ray. MRI Hydrogen Protons. Page 1. Electrons
 Fundamental MRI Principles Module 2 N S 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons positively charged neutrons no significant charge electrons negatively charged Protons
Fundamental MRI Principles Module 2 N S 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons positively charged neutrons no significant charge electrons negatively charged Protons
Fundamental MRI Principles Module Two
 Fundamental MRI Principles Module Two 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons neutrons electrons positively charged no significant charge negatively charged Protons
Fundamental MRI Principles Module Two 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons neutrons electrons positively charged no significant charge negatively charged Protons
The Basics of Magnetic Resonance Imaging
 The Basics of Magnetic Resonance Imaging Nathalie JUST, PhD nathalie.just@epfl.ch CIBM-AIT, EPFL Course 2013-2014-Chemistry 1 Course 2013-2014-Chemistry 2 MRI: Many different contrasts Proton density T1
The Basics of Magnetic Resonance Imaging Nathalie JUST, PhD nathalie.just@epfl.ch CIBM-AIT, EPFL Course 2013-2014-Chemistry 1 Course 2013-2014-Chemistry 2 MRI: Many different contrasts Proton density T1
A Hands on Introduction to NMR Lecture #1 Nuclear Spin and Magnetic Resonance
 A Hands on Introduction to NMR 22.920 Lecture #1 Nuclear Spin and Magnetic Resonance Introduction - The aim of this short course is to present a physical picture of the basic principles of Nuclear Magnetic
A Hands on Introduction to NMR 22.920 Lecture #1 Nuclear Spin and Magnetic Resonance Introduction - The aim of this short course is to present a physical picture of the basic principles of Nuclear Magnetic
Introduction of Key Concepts of Nuclear Magnetic Resonance
 I have not yet lost that sense of wonder, and delight, that this delicate motion should reside in all ordinary things around us, revealing itself only to those who looks for it. E. M. Purcell, Nobel Lecture.
I have not yet lost that sense of wonder, and delight, that this delicate motion should reside in all ordinary things around us, revealing itself only to those who looks for it. E. M. Purcell, Nobel Lecture.
Basic p rinciples COPYRIGHTED MATERIAL. Introduction. Atomic s tructure
 1 Basic p rinciples Introduction 1 Atomic structure 1 Motion in the atom 2 MR active nuclei 2 The hydrogen nucleus 4 Alignment 4 Precession 8 The Larmor equation 9 Introduction The basic principles of
1 Basic p rinciples Introduction 1 Atomic structure 1 Motion in the atom 2 MR active nuclei 2 The hydrogen nucleus 4 Alignment 4 Precession 8 The Larmor equation 9 Introduction The basic principles of
The Theory of Nuclear Magnetic Resonance Behind Magnetic Resonance Imaging. Catherine Wasko Physics 304 Physics of the Human Body May 3, 2005
 The Theory of Nuclear Magnetic Resonance Behind Magnetic Resonance Imaging Catherine Wasko Physics 304 Physics of the Human Body May 3, 2005 Magnetic resonance imaging (MRI) is a tool utilized in the medical
The Theory of Nuclear Magnetic Resonance Behind Magnetic Resonance Imaging Catherine Wasko Physics 304 Physics of the Human Body May 3, 2005 Magnetic resonance imaging (MRI) is a tool utilized in the medical
MRI Physics I: Spins, Excitation, Relaxation
 MRI Physics I: Spins, Excitation, Relaxation Douglas C. Noll Biomedical Engineering University of Michigan Michigan Functional MRI Laboratory Outline Introduction to Nuclear Magnetic Resonance Imaging
MRI Physics I: Spins, Excitation, Relaxation Douglas C. Noll Biomedical Engineering University of Michigan Michigan Functional MRI Laboratory Outline Introduction to Nuclear Magnetic Resonance Imaging
Magnetic Resonance Imaging (MRI)
 Magnetic Resonance Imaging Introduction The Components The Technology (MRI) Physics behind MR Most slides taken from http:// www.slideworld.org/ viewslides.aspx/magnetic- Resonance-Imaging- %28MRI%29-MR-Imaging-
Magnetic Resonance Imaging Introduction The Components The Technology (MRI) Physics behind MR Most slides taken from http:// www.slideworld.org/ viewslides.aspx/magnetic- Resonance-Imaging- %28MRI%29-MR-Imaging-
Lecture 02 Nuclear Magnetic Resonance Spectroscopy Principle and Application in Structure Elucidation
 Application of Spectroscopic Methods in Molecular Structure Determination Prof. S. Sankararaman Department of Chemistry Indian Institution of Technology Madras Lecture 02 Nuclear Magnetic Resonance Spectroscopy
Application of Spectroscopic Methods in Molecular Structure Determination Prof. S. Sankararaman Department of Chemistry Indian Institution of Technology Madras Lecture 02 Nuclear Magnetic Resonance Spectroscopy
The NMR Inverse Imaging Problem
 The NMR Inverse Imaging Problem Nuclear Magnetic Resonance Protons and Neutrons have intrinsic angular momentum Atoms with an odd number of proton and/or odd number of neutrons have a net magnetic moment=>
The NMR Inverse Imaging Problem Nuclear Magnetic Resonance Protons and Neutrons have intrinsic angular momentum Atoms with an odd number of proton and/or odd number of neutrons have a net magnetic moment=>
Basic principles COPYRIGHTED MATERIAL. Introduction. Introduction 1 Precession and precessional
 1 Basic principles Introduction 1 Precession and precessional Atomic structure 2 (Larmor) frequency 10 Motion in the atom 2 Precessional phase 13 MR-active nuclei 4 Resonance 13 The hydrogen nucleus 5
1 Basic principles Introduction 1 Precession and precessional Atomic structure 2 (Larmor) frequency 10 Motion in the atom 2 Precessional phase 13 MR-active nuclei 4 Resonance 13 The hydrogen nucleus 5
Physical Background Of Nuclear Magnetic Resonance Spectroscopy
 Physical Background Of Nuclear Magnetic Resonance Spectroscopy Michael McClellan Spring 2009 Department of Physics and Physical Oceanography University of North Carolina Wilmington What is Spectroscopy?
Physical Background Of Nuclear Magnetic Resonance Spectroscopy Michael McClellan Spring 2009 Department of Physics and Physical Oceanography University of North Carolina Wilmington What is Spectroscopy?
ELECTRON SPIN RESONANCE & MAGNETIC RESONANCE TOMOGRAPHY
 ELECTRON SPIN RESONANCE & MAGNETIC RESONANCE TOMOGRAPHY 1. AIM OF THE EXPERIMENT This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment
ELECTRON SPIN RESONANCE & MAGNETIC RESONANCE TOMOGRAPHY 1. AIM OF THE EXPERIMENT This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment
Topics. The concept of spin Precession of magnetic spin Relaxation Bloch Equation. Bioengineering 280A Principles of Biomedical Imaging
 Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2006 MRI Lecture 1 Topics The concept of spin Precession of magnetic spin Relaxation Bloch Equation 1 Spin Intrinsic angular momentum of
Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2006 MRI Lecture 1 Topics The concept of spin Precession of magnetic spin Relaxation Bloch Equation 1 Spin Intrinsic angular momentum of
10.3 NMR Fundamentals
 10.3 NMR Fundamentals nuclear spin calculations and examples NMR properties of selected nuclei the nuclear magnetic moment and precession around a magnetic field the spin quantum number and the NMR transition
10.3 NMR Fundamentals nuclear spin calculations and examples NMR properties of selected nuclei the nuclear magnetic moment and precession around a magnetic field the spin quantum number and the NMR transition
NMR, the vector model and the relaxation
 NMR, the vector model and the relaxation Reading/Books: One and two dimensional NMR spectroscopy, VCH, Friebolin Spin Dynamics, Basics of NMR, Wiley, Levitt Molecular Quantum Mechanics, Oxford Univ. Press,
NMR, the vector model and the relaxation Reading/Books: One and two dimensional NMR spectroscopy, VCH, Friebolin Spin Dynamics, Basics of NMR, Wiley, Levitt Molecular Quantum Mechanics, Oxford Univ. Press,
Magnetic Resonance Imaging
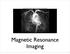 http://www.qldxray.com.au/filelibrary/mri_cardiovascular_system_ca_0005.jpg Magnetic Resonance Imaging 1 Overview 1. The magnetic properties of nuclei, and how they behave in strong magnetic fields. 2.
http://www.qldxray.com.au/filelibrary/mri_cardiovascular_system_ca_0005.jpg Magnetic Resonance Imaging 1 Overview 1. The magnetic properties of nuclei, and how they behave in strong magnetic fields. 2.
Ferdowsi University of Mashhad
 Spectroscopy in Inorganic Chemistry Nuclear Magnetic Resonance Spectroscopy spin deuterium 2 helium 3 The neutron has 2 quarks with a -e/3 charge and one quark with a +2e/3 charge resulting in a total
Spectroscopy in Inorganic Chemistry Nuclear Magnetic Resonance Spectroscopy spin deuterium 2 helium 3 The neutron has 2 quarks with a -e/3 charge and one quark with a +2e/3 charge resulting in a total
Nuclear Magnetic Resonance Spectroscopy
 Nuclear Magnetic Resonance Spectroscopy Ecole Polytechnique Département de Chimie CHI 551 Dr. Grégory Nocton Bureau 01 30 11 A Tel: 44 02 Ecole polytechnique / CNRS Laboratoire de Chimie Moléculaire E-mail:
Nuclear Magnetic Resonance Spectroscopy Ecole Polytechnique Département de Chimie CHI 551 Dr. Grégory Nocton Bureau 01 30 11 A Tel: 44 02 Ecole polytechnique / CNRS Laboratoire de Chimie Moléculaire E-mail:
The Nuclear Emphasis
 The Nuclear Emphasis Atoms are composed of electrons and nuclei we ll focus almost exclusively on the physical properties of the nucleus and the chemicoelectronic attributes of its environment. The nucleus
The Nuclear Emphasis Atoms are composed of electrons and nuclei we ll focus almost exclusively on the physical properties of the nucleus and the chemicoelectronic attributes of its environment. The nucleus
Magnetic Resonance Spectroscopy EPR and NMR
 Magnetic Resonance Spectroscopy EPR and NMR A brief review of the relevant bits of quantum mechanics 1. Electrons have spin, - rotation of the charge about its axis generates a magnetic field at each electron.
Magnetic Resonance Spectroscopy EPR and NMR A brief review of the relevant bits of quantum mechanics 1. Electrons have spin, - rotation of the charge about its axis generates a magnetic field at each electron.
Magnetic Resonance Imaging. Pål Erik Goa Associate Professor in Medical Imaging Dept. of Physics
 Magnetic Resonance Imaging Pål Erik Goa Associate Professor in Medical Imaging Dept. of Physics pal.e.goa@ntnu.no 1 Why MRI? X-ray/CT: Great for bone structures and high spatial resolution Not so great
Magnetic Resonance Imaging Pål Erik Goa Associate Professor in Medical Imaging Dept. of Physics pal.e.goa@ntnu.no 1 Why MRI? X-ray/CT: Great for bone structures and high spatial resolution Not so great
Introduction to NMR! Ravinder Reddy!
 Introduction to NMR! Ravinder Reddy! Brief History of NMR! First detection of NMR! MSNMR! FT NMR! 2D NMR! 2D-NMR and protein structure! Development of MRI! Outline! Concept of SPIN! Spin angular momentum!
Introduction to NMR! Ravinder Reddy! Brief History of NMR! First detection of NMR! MSNMR! FT NMR! 2D NMR! 2D-NMR and protein structure! Development of MRI! Outline! Concept of SPIN! Spin angular momentum!
G Medical Imaging. Outline 4/13/2012. Physics of Magnetic Resonance Imaging
 G16.4426 Medical Imaging Physics of Magnetic Resonance Imaging Riccardo Lattanzi, Ph.D. Assistant Professor Department of Radiology, NYU School of Medicine Department of Electrical and Computer Engineering,
G16.4426 Medical Imaging Physics of Magnetic Resonance Imaging Riccardo Lattanzi, Ph.D. Assistant Professor Department of Radiology, NYU School of Medicine Department of Electrical and Computer Engineering,
NMR Spectroscopy. for 1 st B.Tech INTRODUCTION Lecture -1 Indian Institute of Technology, Dhanbad
 NMR Spectroscopy for 1 st B.Tech Lecture -1 Indian Institute of Technology, Dhanbad by Dr. R P John & Dr. C. Halder INTRODUCTION Nucleus of any atom has protons and neutrons Both Proton and Neutron has
NMR Spectroscopy for 1 st B.Tech Lecture -1 Indian Institute of Technology, Dhanbad by Dr. R P John & Dr. C. Halder INTRODUCTION Nucleus of any atom has protons and neutrons Both Proton and Neutron has
CHAPTER 2 MAGNETISM. 2.1 Magnetic materials
 CHAPTER 2 MAGNETISM Magnetism plays a crucial role in the development of memories for mass storage, and in sensors to name a few. Spintronics is an integration of the magnetic material with semiconductor
CHAPTER 2 MAGNETISM Magnetism plays a crucial role in the development of memories for mass storage, and in sensors to name a few. Spintronics is an integration of the magnetic material with semiconductor
Topics. Spin. The concept of spin Precession of magnetic spin Relaxation Bloch Equation
 Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2005 MRI Lecture 1 Topics The concept of spin Precession of magnetic spin Relaation Bloch Equation Spin Intrinsic angular momentum of elementary
Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2005 MRI Lecture 1 Topics The concept of spin Precession of magnetic spin Relaation Bloch Equation Spin Intrinsic angular momentum of elementary
I Basic Spin Physics. 1. Nuclear Magnetism
 I Basic Spin Physics Lecture notes by Assaf Tal The simplest example of a magnetic moment is the refrigerator magnet. We ll soon meet other, much smaller and weaker magnetic moments, when we discuss the
I Basic Spin Physics Lecture notes by Assaf Tal The simplest example of a magnetic moment is the refrigerator magnet. We ll soon meet other, much smaller and weaker magnetic moments, when we discuss the
Nuclear Magnetic Resonance (NMR) Spectroscopy Introduction:
 Nuclear Magnetic Resonance (NMR) Spectroscopy Introduction: Nuclear magnetic resonance spectroscopy (NMR) is the most powerful tool available for organic structure determination. Like IR spectroscopy,
Nuclear Magnetic Resonance (NMR) Spectroscopy Introduction: Nuclear magnetic resonance spectroscopy (NMR) is the most powerful tool available for organic structure determination. Like IR spectroscopy,
Chemistry 431. Lecture 23
 Chemistry 431 Lecture 23 Introduction The Larmor Frequency The Bloch Equations Measuring T 1 : Inversion Recovery Measuring T 2 : the Spin Echo NC State University NMR spectroscopy The Nuclear Magnetic
Chemistry 431 Lecture 23 Introduction The Larmor Frequency The Bloch Equations Measuring T 1 : Inversion Recovery Measuring T 2 : the Spin Echo NC State University NMR spectroscopy The Nuclear Magnetic
Introduction to MRI. Spin & Magnetic Moments. Relaxation (T1, T2) Spin Echoes. 2DFT Imaging. K-space & Spatial Resolution.
 Introduction to MRI Spin & Magnetic Moments Relaxation (T1, T2) Spin Echoes 2DFT Imaging Selective excitation, phase & frequency encoding K-space & Spatial Resolution Contrast (T1, T2) Acknowledgement:
Introduction to MRI Spin & Magnetic Moments Relaxation (T1, T2) Spin Echoes 2DFT Imaging Selective excitation, phase & frequency encoding K-space & Spatial Resolution Contrast (T1, T2) Acknowledgement:
NMR Spectroscopy Laboratory Experiment Introduction. 2. Theory
 1. Introduction 64-311 Laboratory Experiment 11 NMR Spectroscopy Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful and theoretically complex analytical tool. This experiment will introduce to
1. Introduction 64-311 Laboratory Experiment 11 NMR Spectroscopy Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful and theoretically complex analytical tool. This experiment will introduce to
Spin. Nuclear Spin Rules
 Spin Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2012 MRI Lecture 1 Intrinsic angular momentum of elementary particles -- electrons, protons, neutrons. Spin is quantized. Key concept
Spin Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2012 MRI Lecture 1 Intrinsic angular momentum of elementary particles -- electrons, protons, neutrons. Spin is quantized. Key concept
Chem 325 NMR Intro. The Electromagnetic Spectrum. Physical properties, chemical properties, formulas Shedding real light on molecular structure:
 Physical properties, chemical properties, formulas Shedding real light on molecular structure: Wavelength Frequency ν Wavelength λ Frequency ν Velocity c = 2.998 10 8 m s -1 The Electromagnetic Spectrum
Physical properties, chemical properties, formulas Shedding real light on molecular structure: Wavelength Frequency ν Wavelength λ Frequency ν Velocity c = 2.998 10 8 m s -1 The Electromagnetic Spectrum
Nuclear Magnetic Resonance Imaging
 Nuclear Magnetic Resonance Imaging Simon Lacoste-Julien Electromagnetic Theory Project 198-562B Department of Physics McGill University April 21 2003 Abstract This paper gives an elementary introduction
Nuclear Magnetic Resonance Imaging Simon Lacoste-Julien Electromagnetic Theory Project 198-562B Department of Physics McGill University April 21 2003 Abstract This paper gives an elementary introduction
4/4/11. Particles possess intrinsic angular momentum. Spin angular momentum is quantized (it can only take on discrete values)
 For the completely filled shells, subshell (4d 10 ) the orbital magnetic momentum is zero; for the 5s orbital M L is also zero. Hypothesis: the argent atom possesses no magnetic momentum >> they move in
For the completely filled shells, subshell (4d 10 ) the orbital magnetic momentum is zero; for the 5s orbital M L is also zero. Hypothesis: the argent atom possesses no magnetic momentum >> they move in
Introduction to Biomedical Imaging
 Alejandro Frangi, PhD Computational Imaging Lab Department of Information & Communication Technology Pompeu Fabra University www.cilab.upf.edu MRI advantages Superior soft-tissue contrast Depends on among
Alejandro Frangi, PhD Computational Imaging Lab Department of Information & Communication Technology Pompeu Fabra University www.cilab.upf.edu MRI advantages Superior soft-tissue contrast Depends on among
VIII. NUCLEAR MAGNETIC RESONANCE (NMR) SPECTROSCOPY
 1 VIII. NUCLEAR MAGNETIC RESONANCE (NMR) SPECTROSCOPY Molecules are extremely small entities; thus, their direct detection and direct investigation is still almost impossible. For the detection and detailed
1 VIII. NUCLEAR MAGNETIC RESONANCE (NMR) SPECTROSCOPY Molecules are extremely small entities; thus, their direct detection and direct investigation is still almost impossible. For the detection and detailed
NMR BMB 173 Lecture 16, February
 NMR The Structural Biology Continuum Today s lecture: NMR Lots of slides adapted from Levitt, Spin Dynamics; Creighton, Proteins; And Andy Rawlinson There are three types of particles in the universe Quarks
NMR The Structural Biology Continuum Today s lecture: NMR Lots of slides adapted from Levitt, Spin Dynamics; Creighton, Proteins; And Andy Rawlinson There are three types of particles in the universe Quarks
Biophysical Chemistry: NMR Spectroscopy
 Nuclear Magnetism Vrije Universiteit Brussel 21st October 2011 Outline 1 Overview and Context 2 3 Outline 1 Overview and Context 2 3 Context Proteins (and other biological macromolecules) Functional characterisation
Nuclear Magnetism Vrije Universiteit Brussel 21st October 2011 Outline 1 Overview and Context 2 3 Outline 1 Overview and Context 2 3 Context Proteins (and other biological macromolecules) Functional characterisation
MR Fundamentals. 26 October Mitglied der Helmholtz-Gemeinschaft
 MR Fundamentals 26 October 2010 Mitglied der Helmholtz-Gemeinschaft Mitglied der Helmholtz-Gemeinschaft Nuclear Spin Nuclear Spin Nuclear magnetic resonance is observed in atoms with odd number of protons
MR Fundamentals 26 October 2010 Mitglied der Helmholtz-Gemeinschaft Mitglied der Helmholtz-Gemeinschaft Nuclear Spin Nuclear Spin Nuclear magnetic resonance is observed in atoms with odd number of protons
Chapter 7. Nuclear Magnetic Resonance Spectroscopy
 Chapter 7 Nuclear Magnetic Resonance Spectroscopy I. Introduction 1924, W. Pauli proposed that certain atomic nuclei have spin and magnetic moment and exposure to magnetic field would lead to energy level
Chapter 7 Nuclear Magnetic Resonance Spectroscopy I. Introduction 1924, W. Pauli proposed that certain atomic nuclei have spin and magnetic moment and exposure to magnetic field would lead to energy level
With that first concept in mind, it is seen that a spinning nucleus creates a magnetic field, like a bar magnet
 NMR SPECTROSCOPY This section will discuss the basics of NMR (nuclear magnetic resonance) spectroscopy. Most of the section will discuss mainly 1H or proton spectroscopy but the most popular nuclei in
NMR SPECTROSCOPY This section will discuss the basics of NMR (nuclear magnetic resonance) spectroscopy. Most of the section will discuss mainly 1H or proton spectroscopy but the most popular nuclei in
Medical Imaging Physics Spring Quarter Week 9-1
 Medical Imaging Physics Spring Quarter Week 9-1 NMR and MRI Davor Balzar balzar@du.edu www.du.edu/~balzar Intro MRI Outline NMR & MRI Guest lecturer fmri Thursday, May 22 Visit to CUHSC It s not mandatory
Medical Imaging Physics Spring Quarter Week 9-1 NMR and MRI Davor Balzar balzar@du.edu www.du.edu/~balzar Intro MRI Outline NMR & MRI Guest lecturer fmri Thursday, May 22 Visit to CUHSC It s not mandatory
Magnetic resonance imaging MRI
 Magnetic resonance imaging MRI Introduction What is MRI MRI is an imaging technique used primarily in medical settings that uses a strong magnetic field and radio waves to produce very clear and detailed
Magnetic resonance imaging MRI Introduction What is MRI MRI is an imaging technique used primarily in medical settings that uses a strong magnetic field and radio waves to produce very clear and detailed
An Introduction to Hyperfine Structure and Its G-factor
 An Introduction to Hyperfine Structure and Its G-factor Xiqiao Wang East Tennessee State University April 25, 2012 1 1. Introduction In a book chapter entitled Model Calculations of Radiation Induced Damage
An Introduction to Hyperfine Structure and Its G-factor Xiqiao Wang East Tennessee State University April 25, 2012 1 1. Introduction In a book chapter entitled Model Calculations of Radiation Induced Damage
Biochemistry 530 NMR Theory and Practice
 Biochemistry 530 NMR Theory and Practice Gabriele Varani Department of Biochemistry and Department of Chemistry University of Washington Lecturer: Gabriele Varani Biochemistry and Chemistry Room J479 and
Biochemistry 530 NMR Theory and Practice Gabriele Varani Department of Biochemistry and Department of Chemistry University of Washington Lecturer: Gabriele Varani Biochemistry and Chemistry Room J479 and
Magnetic Resonance Imaging in a Nutshell
 Magnetic Resonance Imaging in a Nutshell Oliver Bieri, PhD Department of Radiology, Division of Radiological Physics, University Hospital Basel Department of Biomedical Engineering, University of Basel,
Magnetic Resonance Imaging in a Nutshell Oliver Bieri, PhD Department of Radiology, Division of Radiological Physics, University Hospital Basel Department of Biomedical Engineering, University of Basel,
PHY331 Magnetism. Lecture 3
 PHY331 Magnetism Lecture 3 Last week Derived magnetic dipole moment of a circulating electron. Discussed motion of a magnetic dipole in a constant magnetic field. Showed that it precesses with a frequency
PHY331 Magnetism Lecture 3 Last week Derived magnetic dipole moment of a circulating electron. Discussed motion of a magnetic dipole in a constant magnetic field. Showed that it precesses with a frequency
Measuring Spin-Lattice Relaxation Time
 WJP, PHY381 (2009) Wabash Journal of Physics v4.0, p.1 Measuring Spin-Lattice Relaxation Time L.W. Lupinski, R. Paudel, and M.J. Madsen Department of Physics, Wabash College, Crawfordsville, IN 47933 (Dated:
WJP, PHY381 (2009) Wabash Journal of Physics v4.0, p.1 Measuring Spin-Lattice Relaxation Time L.W. Lupinski, R. Paudel, and M.J. Madsen Department of Physics, Wabash College, Crawfordsville, IN 47933 (Dated:
V27: RF Spectroscopy
 Martin-Luther-Universität Halle-Wittenberg FB Physik Advanced Lab Course V27: RF Spectroscopy ) Electron spin resonance (ESR) Investigate the resonance behaviour of two coupled LC circuits (an active rf
Martin-Luther-Universität Halle-Wittenberg FB Physik Advanced Lab Course V27: RF Spectroscopy ) Electron spin resonance (ESR) Investigate the resonance behaviour of two coupled LC circuits (an active rf
Lecture 2 nmr Spectroscopy
 Lecture 2 nmr Spectroscopy Pages 427 430 and Chapter 13 Molecular Spectroscopy Molecular spectroscopy: the study of the frequencies of electromagnetic radiation that are absorbed or emitted by substances
Lecture 2 nmr Spectroscopy Pages 427 430 and Chapter 13 Molecular Spectroscopy Molecular spectroscopy: the study of the frequencies of electromagnetic radiation that are absorbed or emitted by substances
MRI Homework. i. (0.5 pt each) Consider the following arrangements of bar magnets in a strong magnetic field.
 MRI Homework 1. While x-rays are used to image bones, magnetic resonance imaging (MRI) is used to examine tissues within the body by detecting where hydrogen atoms (H atoms) are and their environment (e.g.
MRI Homework 1. While x-rays are used to image bones, magnetic resonance imaging (MRI) is used to examine tissues within the body by detecting where hydrogen atoms (H atoms) are and their environment (e.g.
Electromagnetism - Lecture 10. Magnetic Materials
 Electromagnetism - Lecture 10 Magnetic Materials Magnetization Vector M Magnetic Field Vectors B and H Magnetic Susceptibility & Relative Permeability Diamagnetism Paramagnetism Effects of Magnetic Materials
Electromagnetism - Lecture 10 Magnetic Materials Magnetization Vector M Magnetic Field Vectors B and H Magnetic Susceptibility & Relative Permeability Diamagnetism Paramagnetism Effects of Magnetic Materials
Topics. The History of Spin. Spin. The concept of spin Precession of magnetic spin Relaxation
 Topics Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2008 MRI Lecture 1 The concept of spin Precession of magnetic spin Relaation Spin The History of Spin Intrinsic angular momentum
Topics Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2008 MRI Lecture 1 The concept of spin Precession of magnetic spin Relaation Spin The History of Spin Intrinsic angular momentum
NUCLEAR MAGNETIC RESONANCE. The phenomenon of nuclear magnetic resonance will be used to study magnetic moments of nuclei.
 14 Sep 11 NMR.1 NUCLEAR MAGNETIC RESONANCE The phenomenon of nuclear magnetic resonance will be used to study magnetic moments of nuclei. Theory: In addition to its well-known properties of mass, charge,
14 Sep 11 NMR.1 NUCLEAR MAGNETIC RESONANCE The phenomenon of nuclear magnetic resonance will be used to study magnetic moments of nuclei. Theory: In addition to its well-known properties of mass, charge,
Doppler echocardiography & Magnetic Resonance Imaging. Doppler echocardiography. History: - Langevin developed sonar.
 1 Doppler echocardiography & Magnetic Resonance Imaging History: - Langevin developed sonar. - 1940s development of pulse-echo. - 1950s development of mode A and B. - 1957 development of continuous wave
1 Doppler echocardiography & Magnetic Resonance Imaging History: - Langevin developed sonar. - 1940s development of pulse-echo. - 1950s development of mode A and B. - 1957 development of continuous wave
Classical behavior of magnetic dipole vector. P. J. Grandinetti
 Classical behavior of magnetic dipole vector Z μ Y X Z μ Y X Quantum behavior of magnetic dipole vector Random sample of spin 1/2 nuclei measure μ z μ z = + γ h/2 group μ z = γ h/2 group Quantum behavior
Classical behavior of magnetic dipole vector Z μ Y X Z μ Y X Quantum behavior of magnetic dipole vector Random sample of spin 1/2 nuclei measure μ z μ z = + γ h/2 group μ z = γ h/2 group Quantum behavior
L z L L. Think of it as also affecting the angle
 Quantum Mechanics and Atomic Physics Lecture 19: Quantized Angular Momentum and Electron Spin http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Last time Raising/Lowering angular momentum
Quantum Mechanics and Atomic Physics Lecture 19: Quantized Angular Momentum and Electron Spin http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Last time Raising/Lowering angular momentum
Lecture #6 NMR in Hilbert Space
 Lecture #6 NMR in Hilbert Space Topics Review of spin operators Single spin in a magnetic field: longitudinal and transverse magnetiation Ensemble of spins in a magnetic field RF excitation Handouts and
Lecture #6 NMR in Hilbert Space Topics Review of spin operators Single spin in a magnetic field: longitudinal and transverse magnetiation Ensemble of spins in a magnetic field RF excitation Handouts and
Electron spin resonance
 Quick reference guide Introduction This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment of the electron spin with a superimposed direct
Quick reference guide Introduction This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment of the electron spin with a superimposed direct
Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy
 Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy Mitesh Shrestha Electron Spin Resonance Electron paramagnetic
Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy Mitesh Shrestha Electron Spin Resonance Electron paramagnetic
μ (vector) = magnetic dipole moment (not to be confused with the permeability μ). Magnetism Electromagnetic Fields in a Solid
 Magnetism Electromagnetic Fields in a Solid SI units cgs (Gaussian) units Total magnetic field: B = μ 0 (H + M) = μ μ 0 H B = H + 4π M = μ H Total electric field: E = 1/ε 0 (D P) = 1/εε 0 D E = D 4π P
Magnetism Electromagnetic Fields in a Solid SI units cgs (Gaussian) units Total magnetic field: B = μ 0 (H + M) = μ μ 0 H B = H + 4π M = μ H Total electric field: E = 1/ε 0 (D P) = 1/εε 0 D E = D 4π P
10.4 Continuous Wave NMR Instrumentation
 10.4 Continuous Wave NMR Instrumentation coherent detection bulk magnetization the rotating frame, and effective magnetic field generating a rotating frame, and precession in the laboratory frame spin-lattice
10.4 Continuous Wave NMR Instrumentation coherent detection bulk magnetization the rotating frame, and effective magnetic field generating a rotating frame, and precession in the laboratory frame spin-lattice
Lecture 12 February 11, 2016
 MATH 262/CME 372: Applied Fourier Analysis and Winter 2016 Elements of Modern Signal Processing Lecture 12 February 11, 2016 Prof. Emmanuel Candes Scribe: Carlos A. Sing-Long, Edited by E. Bates 1 Outline
MATH 262/CME 372: Applied Fourier Analysis and Winter 2016 Elements of Modern Signal Processing Lecture 12 February 11, 2016 Prof. Emmanuel Candes Scribe: Carlos A. Sing-Long, Edited by E. Bates 1 Outline
Spin. Nuclear Spin Rules
 Spin Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 203 MRI Lecture Intrinsic angular momentum of elementary particles -- electrons, protons, neutrons. Spin is quantized. Key concept
Spin Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 203 MRI Lecture Intrinsic angular momentum of elementary particles -- electrons, protons, neutrons. Spin is quantized. Key concept
Basic MRI physics and Functional MRI
 Basic MRI physics and Functional MRI Gregory R. Lee, Ph.D Assistant Professor, Department of Radiology June 24, 2013 Pediatric Neuroimaging Research Consortium Objectives Neuroimaging Overview MR Physics
Basic MRI physics and Functional MRI Gregory R. Lee, Ph.D Assistant Professor, Department of Radiology June 24, 2013 Pediatric Neuroimaging Research Consortium Objectives Neuroimaging Overview MR Physics
Geology 228/378 Applied & Environmental Geophysics Lecture 8. Induced Polarization (IP) and Nuclear Magnetic Resonance (NMR)
 Geology 228/378 Applied & Environmental Geophysics Lecture 8 Induced Polarization (IP) and Nuclear Magnetic Resonance (NMR) Induced Polarization (IP) and Nuclear Magnetic Resonance (NMR) 1. Time domain
Geology 228/378 Applied & Environmental Geophysics Lecture 8 Induced Polarization (IP) and Nuclear Magnetic Resonance (NMR) Induced Polarization (IP) and Nuclear Magnetic Resonance (NMR) 1. Time domain
NMR Nuclear Magnetic Resonance Spectroscopy p. 83. a hydrogen nucleus (a proton) has a charge, spread over the surface
 NMR Nuclear Magnetic Resonance Spectroscopy p. 83 a hydrogen nucleus (a proton) has a charge, spread over the surface a spinning charge produces a magnetic moment (a vector = direction + magnitude) along
NMR Nuclear Magnetic Resonance Spectroscopy p. 83 a hydrogen nucleus (a proton) has a charge, spread over the surface a spinning charge produces a magnetic moment (a vector = direction + magnitude) along
RADIOLOGIV TECHNOLOGY 4912 COMPREHENSEIVE REVIEW/MRI WORSHEET #1- PATIENT CARE AND SAFETY/PHYSICAL PRINCIPLES
 RADIOLOGIV TECHNOLOGY 4912 COMPREHENSEIVE REVIEW/MRI WORSHEET #1- PATIENT CARE AND SAFETY/PHYSICAL PRINCIPLES 1. What are potential consequences to patients and personnel should there be a release of gaseous
RADIOLOGIV TECHNOLOGY 4912 COMPREHENSEIVE REVIEW/MRI WORSHEET #1- PATIENT CARE AND SAFETY/PHYSICAL PRINCIPLES 1. What are potential consequences to patients and personnel should there be a release of gaseous
Line spectrum (contd.) Bohr s Planetary Atom
 Line spectrum (contd.) Hydrogen shows lines in the visible region of the spectrum (red, blue-green, blue and violet). The wavelengths of these lines can be calculated by an equation proposed by J. J. Balmer:
Line spectrum (contd.) Hydrogen shows lines in the visible region of the spectrum (red, blue-green, blue and violet). The wavelengths of these lines can be calculated by an equation proposed by J. J. Balmer:
Basis of MRI Contrast
 Basis of MRI Contrast MARK A. HORSFIELD Department of Cardiovascular Sciences University of Leicester Leicester LE1 5WW UK Tel: +44-116-2585080 Fax: +44-870-7053111 e-mail: mah5@le.ac.uk 1 1.1 The Magnetic
Basis of MRI Contrast MARK A. HORSFIELD Department of Cardiovascular Sciences University of Leicester Leicester LE1 5WW UK Tel: +44-116-2585080 Fax: +44-870-7053111 e-mail: mah5@le.ac.uk 1 1.1 The Magnetic
Magnetic Resonance Spectroscopy ( )
 Magnetic Resonance Spectroscopy In our discussion of spectroscopy, we have shown that absorption of E.M. radiation occurs on resonance: When the frequency of applied E.M. field matches the energy splitting
Magnetic Resonance Spectroscopy In our discussion of spectroscopy, we have shown that absorption of E.M. radiation occurs on resonance: When the frequency of applied E.M. field matches the energy splitting
III.4 Nuclear Magnetic Resonance
 III.4 Nuclear Magnetic Resonance Radiofrequency (rf) spectroscopy on nuclear spin states in a uniaxial constant magnetic field B = B 0 z (III.4.1) B 0 is on the order of 1-25 T The rf frequencies vary
III.4 Nuclear Magnetic Resonance Radiofrequency (rf) spectroscopy on nuclear spin states in a uniaxial constant magnetic field B = B 0 z (III.4.1) B 0 is on the order of 1-25 T The rf frequencies vary
Physical fundamentals of magnetic resonance imaging
 Physical fundamentals of magnetic resonance imaging Stepan Sereda University of Bonn 1 / 26 Why? Figure 1 : Full body MRI scan (Source: [4]) 2 / 26 Overview Spin angular momentum Rotating frame and interaction
Physical fundamentals of magnetic resonance imaging Stepan Sereda University of Bonn 1 / 26 Why? Figure 1 : Full body MRI scan (Source: [4]) 2 / 26 Overview Spin angular momentum Rotating frame and interaction
INTRODUCTION TO NMR and NMR QIP
 Books (NMR): Spin dynamics: basics of nuclear magnetic resonance, M. H. Levitt, Wiley, 2001. The principles of nuclear magnetism, A. Abragam, Oxford, 1961. Principles of magnetic resonance, C. P. Slichter,
Books (NMR): Spin dynamics: basics of nuclear magnetic resonance, M. H. Levitt, Wiley, 2001. The principles of nuclear magnetism, A. Abragam, Oxford, 1961. Principles of magnetic resonance, C. P. Slichter,
Nuclear Magnetic Resonance (NMR)
 Nuclear Magnetic Resonance (NMR) Nuclear Magnetic Resonance (NMR) The Nuclear Magnetic Resonance Spectroscopy (NMR) is one of the most important spectroscopic methods to explore the structure and dynamic
Nuclear Magnetic Resonance (NMR) Nuclear Magnetic Resonance (NMR) The Nuclear Magnetic Resonance Spectroscopy (NMR) is one of the most important spectroscopic methods to explore the structure and dynamic
Biomedical Imaging Magnetic Resonance Imaging
 Biomedical Imaging Magnetic Resonance Imaging Charles A. DiMarzio & Eric Kercher EECE 4649 Northeastern University May 2018 Background and History Measurement of Nuclear Spins Widely used in physics/chemistry
Biomedical Imaging Magnetic Resonance Imaging Charles A. DiMarzio & Eric Kercher EECE 4649 Northeastern University May 2018 Background and History Measurement of Nuclear Spins Widely used in physics/chemistry
Condensed Matter Physics Prof. G. Rangarajan Department of Physics Indian Institute of Technology, Madras
 (Refer Slide Time: 00:22) Condensed Matter Physics Prof. G. Rangarajan Department of Physics Indian Institute of Technology, Madras Lecture 25 Pauli paramagnetism and Landau diamagnetism So far, in our
(Refer Slide Time: 00:22) Condensed Matter Physics Prof. G. Rangarajan Department of Physics Indian Institute of Technology, Madras Lecture 25 Pauli paramagnetism and Landau diamagnetism So far, in our
Spin Dynamics Basics of Nuclear Magnetic Resonance. Malcolm H. Levitt
 Spin Dynamics Basics of Nuclear Magnetic Resonance Second edition Malcolm H. Levitt The University of Southampton, UK John Wiley &. Sons, Ltd Preface xxi Preface to the First Edition xxiii Introduction
Spin Dynamics Basics of Nuclear Magnetic Resonance Second edition Malcolm H. Levitt The University of Southampton, UK John Wiley &. Sons, Ltd Preface xxi Preface to the First Edition xxiii Introduction
NMR of CeCoIn5. AJ LaPanta 8/15/2016
 NMR of CeCoIn5 AJ LaPanta 8/15/2016 In Co-NMR measurements on CeCoIn5, we see an increasing peak width below 50K. We interpret this as the growth of antiferromagnetic regions surrounding Cadmium dopants
NMR of CeCoIn5 AJ LaPanta 8/15/2016 In Co-NMR measurements on CeCoIn5, we see an increasing peak width below 50K. We interpret this as the growth of antiferromagnetic regions surrounding Cadmium dopants
Nuclear Magnetic Resonance
 Nuclear Magnetic Resonance PRINCIPLES OF NMR SPECTROSCOPY Contents Principles of nuclear magnetic resonance The nmr spectrometer Basic principles in nmr application NMR tools used to obtain information
Nuclear Magnetic Resonance PRINCIPLES OF NMR SPECTROSCOPY Contents Principles of nuclear magnetic resonance The nmr spectrometer Basic principles in nmr application NMR tools used to obtain information
Introductory MRI Physics
 C HAPR 18 Introductory MRI Physics Aaron Sodickson EXRNAL MAGNETIC FIELD, PROTONS AND EQUILIBRIUM MAGNETIZATION Much of the bulk of the magnetic resonance imaging (MRI) scanner apparatus is dedicated to
C HAPR 18 Introductory MRI Physics Aaron Sodickson EXRNAL MAGNETIC FIELD, PROTONS AND EQUILIBRIUM MAGNETIZATION Much of the bulk of the magnetic resonance imaging (MRI) scanner apparatus is dedicated to
Lecture 19: Building Atoms and Molecules
 Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r y even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r y even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
Nuclear Magnetic Resonance Imaging
 Nuclear Magnetic Resonance Imaging Jeffrey A. Fessler EECS Department The University of Michigan NSS-MIC: Fundamentals of Medical Imaging Oct. 20, 2003 NMR-0 Background Basic physics 4 magnetic fields
Nuclear Magnetic Resonance Imaging Jeffrey A. Fessler EECS Department The University of Michigan NSS-MIC: Fundamentals of Medical Imaging Oct. 20, 2003 NMR-0 Background Basic physics 4 magnetic fields
Physics 221A Fall 1996 Notes 13 Spins in Magnetic Fields
 Physics 221A Fall 1996 Notes 13 Spins in Magnetic Fields A nice illustration of rotation operator methods which is also important physically is the problem of spins in magnetic fields. The earliest experiments
Physics 221A Fall 1996 Notes 13 Spins in Magnetic Fields A nice illustration of rotation operator methods which is also important physically is the problem of spins in magnetic fields. The earliest experiments
Atomic Structure, Periodic Table, and Other Effects: Chapter 8 of Rex and T. Modern Physics
 Atomic Structure, Periodic Table, and Other Effects: Chapter 8 of Rex and T Modern Physics 11/16 and 11/19/2018 1 Introduction In Chapter 7, we studied the hydrogen atom. What about other elements, e.g.,
Atomic Structure, Periodic Table, and Other Effects: Chapter 8 of Rex and T Modern Physics 11/16 and 11/19/2018 1 Introduction In Chapter 7, we studied the hydrogen atom. What about other elements, e.g.,
Chapter 8 Magnetic Resonance
 Chapter 8 Magnetic Resonance 9.1 Electron paramagnetic resonance 9.2 Ferromagnetic resonance 9.3 Nuclear magnetic resonance 9.4 Other resonance methods TCD March 2007 1 A resonance experiment involves
Chapter 8 Magnetic Resonance 9.1 Electron paramagnetic resonance 9.2 Ferromagnetic resonance 9.3 Nuclear magnetic resonance 9.4 Other resonance methods TCD March 2007 1 A resonance experiment involves
NMR Spectroscopy. Guangjin Hou
 NMR Spectroscopy Guangjin Hou 22-04-2009 NMR History 1 H NMR spectra of water H NMR spectra of water (First NMR Spectra on Water, 1946) 1 H NMR spectra ethanol (First bservation of the Chemical Shift,
NMR Spectroscopy Guangjin Hou 22-04-2009 NMR History 1 H NMR spectra of water H NMR spectra of water (First NMR Spectra on Water, 1946) 1 H NMR spectra ethanol (First bservation of the Chemical Shift,
7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text , , 12.10)
 2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
DETECTION OF UNPAIRED ELECTRONS
 DETECTION OF UNPAIRED ELECTRONS There are experimental methods for the detection of unpaired electrons. One of the hallmarks of unpaired electrons in materials is interaction with a magnetic field. That
DETECTION OF UNPAIRED ELECTRONS There are experimental methods for the detection of unpaired electrons. One of the hallmarks of unpaired electrons in materials is interaction with a magnetic field. That
Lecture 5. Chapters 3 & 4. Induced magnetization: that which is induced in the presence of an applied magnetic field. diamagnetic.
 Lecture 5 Induced magnetization: that which is induced in the presence of an applied magnetic field diamagnetic paramagnetic Remanent magnetization: that which remains in the absence of an external field
Lecture 5 Induced magnetization: that which is induced in the presence of an applied magnetic field diamagnetic paramagnetic Remanent magnetization: that which remains in the absence of an external field
Principles of Magnetic Resonance Imaging
 Principles of Magnetic Resonance Imaging Hi Klaus Scheffler, PhD Radiological Physics University of 1 Biomedical Magnetic Resonance: 1 Introduction Magnetic Resonance Imaging Contents: Hi 1 Introduction
Principles of Magnetic Resonance Imaging Hi Klaus Scheffler, PhD Radiological Physics University of 1 Biomedical Magnetic Resonance: 1 Introduction Magnetic Resonance Imaging Contents: Hi 1 Introduction
Magentic Energy Diagram for A Single Electron Spin and Two Coupled Electron Spins. Zero Field.
 7. Examples of Magnetic Energy Diagrams. There are several very important cases of electron spin magnetic energy diagrams to examine in detail, because they appear repeatedly in many photochemical systems.
7. Examples of Magnetic Energy Diagrams. There are several very important cases of electron spin magnetic energy diagrams to examine in detail, because they appear repeatedly in many photochemical systems.
Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure. Nuclear Magnetic Resonance (NMR)
 Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure Nuclear Magnetic Resonance (NMR) !E = h" Electromagnetic radiation is absorbed when the energy of photon corresponds
Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure Nuclear Magnetic Resonance (NMR) !E = h" Electromagnetic radiation is absorbed when the energy of photon corresponds
5.61 Physical Chemistry Lecture #36 Page
 5.61 Physical Chemistry Lecture #36 Page 1 NUCLEAR MAGNETIC RESONANCE Just as IR spectroscopy is the simplest example of transitions being induced by light s oscillating electric field, so NMR is the simplest
5.61 Physical Chemistry Lecture #36 Page 1 NUCLEAR MAGNETIC RESONANCE Just as IR spectroscopy is the simplest example of transitions being induced by light s oscillating electric field, so NMR is the simplest
Introduction to Relaxation Theory James Keeler
 EUROMAR Zürich, 24 Introduction to Relaxation Theory James Keeler University of Cambridge Department of Chemistry What is relaxation? Why might it be interesting? relaxation is the process which drives
EUROMAR Zürich, 24 Introduction to Relaxation Theory James Keeler University of Cambridge Department of Chemistry What is relaxation? Why might it be interesting? relaxation is the process which drives
Saturation Absorption Spectroscopy of Rubidium Atom
 Saturation Absorption Spectroscopy of Rubidium Atom Jayash Panigrahi August 17, 2013 Abstract Saturated absorption spectroscopy has various application in laser cooling which have many relevant uses in
Saturation Absorption Spectroscopy of Rubidium Atom Jayash Panigrahi August 17, 2013 Abstract Saturated absorption spectroscopy has various application in laser cooling which have many relevant uses in
CHAPTER 8 Atomic Physics
 CHAPTER 8 Atomic Physics 8.1 Atomic Structure and the Periodic Table 8.2 Total Angular Momentum 8.3 Anomalous Zeeman Effect What distinguished Mendeleev was not only genius, but a passion for the elements.
CHAPTER 8 Atomic Physics 8.1 Atomic Structure and the Periodic Table 8.2 Total Angular Momentum 8.3 Anomalous Zeeman Effect What distinguished Mendeleev was not only genius, but a passion for the elements.
