Big Bang Nucleosynthesis
|
|
|
- Clifton Wells
- 5 years ago
- Views:
Transcription
1 Big Bang Nucleosynthesis Chris Williams Student HET th September 2004 Introduction The origin of the stuff from which the Universe is made has been of interest for many centuries. In recent scientific history several possible scenarios for the origin of the 92 natural elements, consistent with Hubble s observation of an expanding Universe, have been proposed. A steady-state theory in which the Universe is eternal, elementary matter is spontaneously created everywhere to maintain a uniform density, and stars convert this to the elements we see, was championed by Hoyle. An alternate theory, supported by Gamow, postulated that all elementary matter was created in a single primordial explosion, the so-called Big Bang, and that the current state has evolved from there. Observational evidence has since discounted Hoyle s model by detecting artifacts of the Big Bang. A key element of both these Universal models is a process called nucleosynthesis. The specifics of this process were of relevance to discounting Hoyle s model. This essay addresses nucleosynthesis, its role in the Universe, and the constraints placed on the conditions of the Big Bang in order to match observational evidence. Atoms and Nuclei All the material around us is built from atoms. Each atom, or element, has a structure consisting of a nucleus surrounded by a cloud of electrons (negative particles). The nucleus consists of positively charged protons and electrically neutral particles called neutrons (collectively called nucleons). The number of protons in the nucleus determines the type of element the atom represents. For example, Hydrogen atoms contain a single proton, Helium contains two, and Carbon contains six. The number of protons in a nucleus is called the atomic number (A). The protons within a nucleus may be accompanied by a number of neutrons, particles which have no electric charge and roughly the same mass as a proton. The protons and neutrons combined provide the 1
2 bulk of the atom s mass; the sum of proton and neutron counts is called the atomic mass (Z). A shorthand is used to indicate the atomic number and mass of atoms, A ZEl, e.g. 1 1 H for the Hydrogen atom or 4 2He for the Helium nucleus. Atoms with the same atomic number but varying neutron counts are called isotopes of the atom or element. For example, Hydrogen comes in isotopes with zero ( 1 1 H), one (2 1 H), or two (3 1H) neutrons, with Z of one, two, or three as a result. The heavier Hydrogen isotopes are named Deuterium and Tritium, but typically an isotope is known only by its atomic mass number, for example Carbon-12 and Carbon-14 of Carbon-dating fame. Nucleosynthesis Nucleosynthesis is the process by which the defining structure of atoms, the nucleus, is constructed from smaller building blocks, protons and neutrons. There are two main processes by which an atom s nucleus may convert from one element to another: fusion, and neutron capture followed by decay. In their large 1957 paper, Burbidge, Burbidge, Fowler, and Hoyle ([2], hereafter BBFH) outlined in substantial detail the processes of nucleosynthesis in the environments expected at the core of stars. Conditions in stellar cores are extreme by everyday standards, with our Sun harbouring a 15, 000, 000 C core temperature and a pressure in the order of 10 8 atmospheres. Under such conditions protons have sufficient energy (motion) to overcome the repulsion, induced by the electrical charges of other protons, and fuse together. The resulting nucleus increases its atomic number by one and becomes a heavier element. For example, the capture of a proton by a Deuterium nucleus, with one proton and one neutron, gives a Helium isotope. Capture can also occur between more complex nuclei, e.g. between two Helium nuclei forming a Beryllium nucleus, but this requires more energy to overcome repulsion. At various stages in the lives of particularly heavy stars conditions can support fusion of nuclei as heavy as Silicon ( Si) to form Iron isotopes ( x 26 Fe). BBFH also outlined another mechanism that could modify the nucleus of atoms in the end stages of stellar lifetimes, particularly in the catastrophic explosion of heavy stars (supernovæ). The process of neutron capture allows formation of a heavier isotope of the same element by adding a neutron e.g. a 3 2He nucleus may capture a neutron to become the most common form of Helium ( 4 2 He). Neutron capture does not have to overcome electrical repulsion and can occur with lower energy. Neutrons have only a short life span outside of a nucleus, which limits likelihood of capture, therefore supplies of free neutrons in quantity are required order to build up nuclei. Our world is, for the large part, populated by long-term stable elements, and it is clear that nature favours certain arrangements of protons and neutrons over others. Isotopes that are not stable, such as all the radium iso- 2
3 topes, are termed radioactive and are subject to decay until they reach a stable configuration. Many possible configurations created by fusion or neutron capture are unstable over relatively short periods and eject a particle or change in other ways. A typical decay might eject two neutrons and two protons, a Helium nucleus or α-particle, to become an isotope of a lighter element. Another form of decay that affects the nucleus is when a neutron decays by emitting an electron and anti-neutrino to become a proton (β decay). β decay increases the nucleus atomic number by one and changes the arrangement of nucleons, maybe to a more stable state. In environments where neutron capture is occurring more slowly than β decay the capture process is called the s-process (slow ), and can build nuclei as heavy as Lead ( Pb) and Bismuth ( Bi). When neutron capture rates exceed β decay rates, the process is called the r-process (rapid ), and may result in nuclei heavier than the s-process, e.g. Uranium ( U). Chains of the processes described could, on the face of it, allow the construction of any element from only Hydrogen given the correct environment. BBFH concluded that fusion processes could reasonably account for the abundances of naturally occurring elements as heavy as iron. Elements heavier than iron were created by the s and r-processes in the violent explosion of massive stars. BBFH also admitted that hard evidence was needed to bolster the case. Unfortunately, the stellar processes alone could not account for the unexpectedly high observed Helium concentration, which leads to the other place that nucleosynthesis could occur. Big Bang Nucleosynthesis outside of stars is a pivotal part of the Big Bang model of Universal creation. In the Big Bang model our Universe was once contained within an infinitely dense point that exploded to form the space and time that we know. All of the energy and matter contained in our Universe was present in this original point so, shortly after the explosion, temperature and density would have been extreme and a candidate for processes of nucleosynthesis. In the moments following the initial explosion of the singularity, nucleons as we know them did not exist because they were dissociated into even smaller particles called quarks and gluons. To form neutrons and protons a mechanism was required to dramatically cool the Universe. The theory calls for a period of rapid inflation in order to produce the even distribution of matter we see, and this is also a cooling factor. As the expansion progressed and temperatures dropped, neutrons were able to form from the quark soup. Neutrons are not stable for long period outside of atomic nuclei due to β decay. However, until the temperature and density of the Universe dropped sufficiently, any neutron that decayed into a proton would quickly reabsorb an electron (inverse β process), leading to an equilibrium state. 3
4 Ultimately the Universe cooled sufficiently that the equilibrium was broken and protons became more common, and neutrons less so. The Universe now had the building blocks of nucleosynthesis. In 1946, George Gamow [3] outlined reasoning supporting the formation of the basic elements in a short period following the initial event. Alpher, Bethe, and Gamow [1] expanded on this work to flesh out the consequences of this model on the abundance curve of the elements. Gamow et al. considered that the initial gamut of elements was determined between when conditions allowed free neutrons to exist (T Kelvin) and was largely fixed by the time that Deuterium nuclei could form without immediately being disassembled (T Kelvin). In this time period neutrons were decaying into protons, a process that would cease when there were no longer free neutrons to decay. Once Deuterium became stable Gamow determined that all the available neutrons would be rapidly absorbed into larger nuclei. At the point the process ceased, the ratio of protons to neutrons would be fixed; with expectations of about ten to one. When Deuterium nuclei became stable conditions were such that fusion of nuclei was still possible. However, this remained so only until expansion once more removed temperature and diluted density. If expansion was slow enough conditions could remain suitable for fusion of Helium and Lithium, and heavier elements would follow. If expansion was faster then less Helium would form, and remaining Deuterium would decay back into Hydrogen. The only stable isotopes of mass of eight or less are 1 1 H, 4 2 He, and 7 3Li, and it is these that we observe in the early Universe. While unstable isotopes 2 1 H, 3 1 H, 3 2 He, 5 2 He, 5 3 Li, 8 4Be could form, they would all decay on short timescales. The end point of the process is strongly linked to the expansion rate, which is given a lower limit by observed Helium to Hydrogen ratios. The ratio of protons to neutrons and the dominance of stable Helium as a heavier nucleus leads directly to relative abundances of Hydrogen and Helium. Working with the 10:1 estimated ratio, each 4 2He nuclei must be matched by eighteen Hydrogen nuclei to maintain the ratio. In the example, the Helium nucleus is 18% of the mass. Current estimates of the primordial Helium abundance, based on distant, younger galaxies much closer to original abundances, are 23 24% (e.g. Izotov & Thuan [4]). This corresponds to a proton/neutron ratio of seven or eight to one. Universal expansion between the time that neutron-proton equilibrium broke and the formation of Deuterium must be within a narrow range to arrive at these ratios. The observed Helium abundance therefore constrains the conditions of the early Big Bang model. 4
5 Conclusion Everything around us is made from atoms, and these atoms originated in the Big Bang. The processes of nucleosynthesis operated to build larger nuclei from protons and neutrons, but the process was limited by expansion. The precise ratio of material in the primordial Big Bang material constrains the conditions of the early Universe, particularly the rate of expansion. Only Hydrogen ( 77%), Helium ( 23%) and minute traces of Lithium are observed in regions close to this period. Stars have since taken the rudimentary building blocks from the Big Bang and produced the pantheon of elements we see now using largely the same processes that occurred briefly during the Big Bang. For this reason, we can truly be said to be made from the stuff of stars. References [1] R. A. Alpher, H. Bethe, and G. Gamow. The Origin of Chemical Elements. Physical Review, 73: , April [2] E. M. Burbidge, G. R. Burbidge, W. A. Fowler, and F. Hoyle. Synthesis of the Elements in Stars. Reviews of Modern Physics, 29: , [3] G. Gamow. Expanding Universe and the Origin of Elements. Physical Review, 70: , October [4] Y. I. Izotov and T. X. Thuan. The Primordial Abundance of 4He Revisited. Astrophysical Journal, 500:188 +, June
In the Beginning. After about three minutes the temperature had cooled even further, so that neutrons were able to combine with 1 H to form 2 H;
 In the Beginning Obviously, before we can have any geochemistry we need some elements to react with one another. The most commonly held scientific view for the origin of the universe is the "Big Bang"
In the Beginning Obviously, before we can have any geochemistry we need some elements to react with one another. The most commonly held scientific view for the origin of the universe is the "Big Bang"
Interactions. Laws. Evolution
 Lecture Origin of the Elements MODEL: Origin of the Elements or Nucleosynthesis Fundamental Particles quarks, gluons, leptons, photons, neutrinos + Basic Forces gravity, electromagnetic, nuclear Interactions
Lecture Origin of the Elements MODEL: Origin of the Elements or Nucleosynthesis Fundamental Particles quarks, gluons, leptons, photons, neutrinos + Basic Forces gravity, electromagnetic, nuclear Interactions
Neutron-to-proton ratio
 Neutron-to-proton ratio After one second, the Universe had cooled to 10 13 K. The Universe was filled with protons, neutrons, electrons, and neutrinos. The temperature was high enough that they interconverted
Neutron-to-proton ratio After one second, the Universe had cooled to 10 13 K. The Universe was filled with protons, neutrons, electrons, and neutrinos. The temperature was high enough that they interconverted
How to Build a Habitable Planet Summary. Chapter 1 The Setting
 How to Build a Habitable Planet Summary Chapter 1 The Setting The universe as we know it began about 15 billion years ago with an explosion that is called the big bang. There is no record of any prior
How to Build a Habitable Planet Summary Chapter 1 The Setting The universe as we know it began about 15 billion years ago with an explosion that is called the big bang. There is no record of any prior
ORIGIN OF THE ELEMENETS
 VISUAL PHYSICS ONLINE ORIGIN OF THE ELEMENETS Watch Video: The Origin of the Elements The ordinary matter in our universe (known as baryonic matter) is made up of 94 naturally occurring elements. It is
VISUAL PHYSICS ONLINE ORIGIN OF THE ELEMENETS Watch Video: The Origin of the Elements The ordinary matter in our universe (known as baryonic matter) is made up of 94 naturally occurring elements. It is
Primordial (Big Bang) Nucleosynthesis
 Primordial (Big Bang) Nucleosynthesis H Li Be Which elements? He METALS - 1942: Gamow suggests a Big Bang origin of the elements. - 1948: Alpher, Bethe & Gamow: all elements are synthesized minutes after
Primordial (Big Bang) Nucleosynthesis H Li Be Which elements? He METALS - 1942: Gamow suggests a Big Bang origin of the elements. - 1948: Alpher, Bethe & Gamow: all elements are synthesized minutes after
BIG BANG SUMMARY NOTES
 BIG BANG SUMMARY NOTES BIG BANG THEORY Studies of red-shifts of distant galaxies show that the universe is expanding. This and other observations has led to the Big Bang Theory The Big Bang Theory claims
BIG BANG SUMMARY NOTES BIG BANG THEORY Studies of red-shifts of distant galaxies show that the universe is expanding. This and other observations has led to the Big Bang Theory The Big Bang Theory claims
Nucleosynthesis. W. F. McDonough 1. Neutrino Science, Tohoku University, Sendai , Japan. (Dated: Tuesday 24 th April, 2018)
 Nucleosynthesis W. F. McDonough 1 1 Department of Earth Sciences and Research Center for Neutrino Science, Tohoku University, Sendai 980-8578, Japan (Dated: Tuesday 24 th April, 2018) Goals Where were
Nucleosynthesis W. F. McDonough 1 1 Department of Earth Sciences and Research Center for Neutrino Science, Tohoku University, Sendai 980-8578, Japan (Dated: Tuesday 24 th April, 2018) Goals Where were
IB Test. Astrophysics HL. Name_solution / a) Describe what is meant by a nebula [1]
![IB Test. Astrophysics HL. Name_solution / a) Describe what is meant by a nebula [1] IB Test. Astrophysics HL. Name_solution / a) Describe what is meant by a nebula [1]](/thumbs/73/69495220.jpg) IB Test Astrophysics HL Name_solution / 47 1. a) Describe what is meant by a nebula [1] an intergalactic cloud of gas and dust where all stars begin to form b) Explain how the Jeans criterion applies to
IB Test Astrophysics HL Name_solution / 47 1. a) Describe what is meant by a nebula [1] an intergalactic cloud of gas and dust where all stars begin to form b) Explain how the Jeans criterion applies to
Stellar processes, nucleosynthesis OUTLINE
 Stellar processes, nucleosynthesis OUTLINE Reading this week: White 313-326 and 421-464 Today 1. Stellar processes 2. Nucleosynthesis Powerpoint credit: Using significant parts of a WHOI ppt 1 Question
Stellar processes, nucleosynthesis OUTLINE Reading this week: White 313-326 and 421-464 Today 1. Stellar processes 2. Nucleosynthesis Powerpoint credit: Using significant parts of a WHOI ppt 1 Question
Lecture 19 Big Bang Nucleosynthesis
 Lecture 19 Big Bang Nucleosynthesis As with all course material (including homework, exams), these lecture notes are not be reproduced, redistributed, or sold in any form. The CMB as seen by the WMAP satellite.!2
Lecture 19 Big Bang Nucleosynthesis As with all course material (including homework, exams), these lecture notes are not be reproduced, redistributed, or sold in any form. The CMB as seen by the WMAP satellite.!2
Week 4: Nuclear physics relevant to stars
 Week 4: Nuclear physics relevant to stars So, in week 2, we did a bit of formal nuclear physics just setting out the reaction rates in terms of cross sections, but not worrying about what nuclear reactions
Week 4: Nuclear physics relevant to stars So, in week 2, we did a bit of formal nuclear physics just setting out the reaction rates in terms of cross sections, but not worrying about what nuclear reactions
Fundamental Stellar Parameters. Radiative Transfer. Stellar Atmospheres. Equations of Stellar Structure
 Fundamental Stellar Parameters Radiative Transfer Stellar Atmospheres Equations of Stellar Structure Nuclear Reactions in Stellar Interiors Binding Energy Coulomb Barrier Penetration Hydrogen Burning Reactions
Fundamental Stellar Parameters Radiative Transfer Stellar Atmospheres Equations of Stellar Structure Nuclear Reactions in Stellar Interiors Binding Energy Coulomb Barrier Penetration Hydrogen Burning Reactions
Stars and their properties: (Chapters 11 and 12)
 Stars and their properties: (Chapters 11 and 12) To classify stars we determine the following properties for stars: 1. Distance : Needed to determine how much energy stars produce and radiate away by using
Stars and their properties: (Chapters 11 and 12) To classify stars we determine the following properties for stars: 1. Distance : Needed to determine how much energy stars produce and radiate away by using
The origin of the light elements in the early Universe
 1 HUBERT REEVES* The origin of the light elements in the early Universe Shortly after World War II, George Gamov and his collaborators (Alpher et al. 148) considered the possibility that all chemical elements
1 HUBERT REEVES* The origin of the light elements in the early Universe Shortly after World War II, George Gamov and his collaborators (Alpher et al. 148) considered the possibility that all chemical elements
Matter vs. Antimatter in the Big Bang. E = mc 2
 Matter vs. Antimatter in the Big Bang Threshold temperatures If a particle encounters its corresponding antiparticle, the two will annihilate: particle + antiparticle ---> radiation * Correspondingly,
Matter vs. Antimatter in the Big Bang Threshold temperatures If a particle encounters its corresponding antiparticle, the two will annihilate: particle + antiparticle ---> radiation * Correspondingly,
Lecture 3: Big Bang Nucleosynthesis The First Three Minutes
 Lecture 3: Big Bang Nucleosynthesis The First Three Minutes Last time: particle anti-particle soup --> quark soup --> neutron-proton soup p / n ratio at onset of 2 D formation Today: Form 2 D and 4 He
Lecture 3: Big Bang Nucleosynthesis The First Three Minutes Last time: particle anti-particle soup --> quark soup --> neutron-proton soup p / n ratio at onset of 2 D formation Today: Form 2 D and 4 He
The Big Bang Theory, General Timeline. The Planck Era. (Big Bang To 10^-35 Seconds) Inflationary Model Added. (10^-35 to 10^-33 Of A Second)
 The Big Bang Theory, General Timeline The Planck Era. (Big Bang To 10^-35 Seconds) The time from the exact moment of the Big Bang until 10^-35 of a second later is referred to as the Planck Era. While
The Big Bang Theory, General Timeline The Planck Era. (Big Bang To 10^-35 Seconds) The time from the exact moment of the Big Bang until 10^-35 of a second later is referred to as the Planck Era. While
Section 12. Nuclear reactions in stars Introduction
 Section 12 Nuclear reactions in stars 12.1 Introduction Consider two types of nuclei, A and B, number densities n(a), n(b). The rate at which a particular (nuclear) reaction occurs is r(v) = n(a)n(b)v
Section 12 Nuclear reactions in stars 12.1 Introduction Consider two types of nuclei, A and B, number densities n(a), n(b). The rate at which a particular (nuclear) reaction occurs is r(v) = n(a)n(b)v
Chapter 17 Lecture. The Cosmic Perspective Seventh Edition. Star Stuff Pearson Education, Inc.
 Chapter 17 Lecture The Cosmic Perspective Seventh Edition Star Stuff Star Stuff 17.1 Lives in the Balance Our goals for learning: How does a star's mass affect nuclear fusion? How does a star's mass affect
Chapter 17 Lecture The Cosmic Perspective Seventh Edition Star Stuff Star Stuff 17.1 Lives in the Balance Our goals for learning: How does a star's mass affect nuclear fusion? How does a star's mass affect
MODERN ASTROPHYSICS PCES 4.55
 MODERN ASTROPHYSICS PCES 4.55 The 20 th century brought an appreciation of the colossal scale of the universe, and an explanation of how it all worked. The understanding of the stars came from nuclear
MODERN ASTROPHYSICS PCES 4.55 The 20 th century brought an appreciation of the colossal scale of the universe, and an explanation of how it all worked. The understanding of the stars came from nuclear
Lecture 3: Big Bang Nucleosynthesis The First Three Minutes Last time:
 Lecture 3: Big Bang Nucleosynthesis The First Three Minutes Last time: particle anti-particle soup --> quark soup --> neutron-proton soup p / n ratio at onset of 2 D formation Today: Form 2 D and 4 He
Lecture 3: Big Bang Nucleosynthesis The First Three Minutes Last time: particle anti-particle soup --> quark soup --> neutron-proton soup p / n ratio at onset of 2 D formation Today: Form 2 D and 4 He
The first 400,000 years
 The first 400,000 years All about the Big Bang Temperature Chronology of the Big Bang The Cosmic Microwave Background (CMB) The VERY early universe Our Evolving Universe 1 Temperature and the Big Bang
The first 400,000 years All about the Big Bang Temperature Chronology of the Big Bang The Cosmic Microwave Background (CMB) The VERY early universe Our Evolving Universe 1 Temperature and the Big Bang
Chapter 21 Stellar Explosions
 Chapter 21 Stellar Explosions Units of Chapter 21 21.1 XXLife after Death for White Dwarfs (not on exam) 21.2 The End of a High-Mass Star 21.3 Supernovae Supernova 1987A The Crab Nebula in Motion 21.4
Chapter 21 Stellar Explosions Units of Chapter 21 21.1 XXLife after Death for White Dwarfs (not on exam) 21.2 The End of a High-Mass Star 21.3 Supernovae Supernova 1987A The Crab Nebula in Motion 21.4
Astronomy Ch. 21 Stellar Explosions. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Name: Period: Date: Astronomy Ch. 21 Stellar Explosions MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) A surface explosion on a white dwarf, caused
Name: Period: Date: Astronomy Ch. 21 Stellar Explosions MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) A surface explosion on a white dwarf, caused
High Mass Stars. Dr Ken Rice. Discovering Astronomy G
 High Mass Stars Dr Ken Rice High mass star formation High mass star formation is controversial! May form in the same way as low-mass stars Gravitational collapse in molecular clouds. May form via competitive
High Mass Stars Dr Ken Rice High mass star formation High mass star formation is controversial! May form in the same way as low-mass stars Gravitational collapse in molecular clouds. May form via competitive
Stellar Interior: Physical Processes
 Physics Focus on Astrophysics Focus on Astrophysics Stellar Interior: Physical Processes D. Fluri, 29.01.2014 Content 1. Mechanical equilibrium: pressure gravity 2. Fusion: Main sequence stars: hydrogen
Physics Focus on Astrophysics Focus on Astrophysics Stellar Interior: Physical Processes D. Fluri, 29.01.2014 Content 1. Mechanical equilibrium: pressure gravity 2. Fusion: Main sequence stars: hydrogen
Lecture 3: Big Bang Nucleosynthesis
 Lecture 3: Big Bang Nucleosynthesis Last time: particle anti-particle soup --> quark soup --> neutron-proton soup. Today: Form 2 D and 4 He Form heavier nuclei? Discuss primordial abundances X p, Y p,
Lecture 3: Big Bang Nucleosynthesis Last time: particle anti-particle soup --> quark soup --> neutron-proton soup. Today: Form 2 D and 4 He Form heavier nuclei? Discuss primordial abundances X p, Y p,
Chapter 22. Preview. Objectives Properties of the Nucleus Nuclear Stability Binding Energy Sample Problem. Section 1 The Nucleus
 Section 1 The Nucleus Preview Objectives Properties of the Nucleus Nuclear Stability Binding Energy Sample Problem Section 1 The Nucleus Objectives Identify the properties of the nucleus of an atom. Explain
Section 1 The Nucleus Preview Objectives Properties of the Nucleus Nuclear Stability Binding Energy Sample Problem Section 1 The Nucleus Objectives Identify the properties of the nucleus of an atom. Explain
Origin of heavier elements, origin of universe
 Origin of heavier elements, origin of universe Like we said earlier It takes higher and higher temperatures to make larger and larger nuclei fuse together What happens when a star cannot maintain fusion
Origin of heavier elements, origin of universe Like we said earlier It takes higher and higher temperatures to make larger and larger nuclei fuse together What happens when a star cannot maintain fusion
The slides with white background you need to know. The slides with blue background just have some cool information.
 The slides with white background you need to know. The slides with blue background just have some cool information. The Big Bang cosmology the study of the origin, properties, processes, and evolution
The slides with white background you need to know. The slides with blue background just have some cool information. The Big Bang cosmology the study of the origin, properties, processes, and evolution
Fission and Fusion Book pg cgrahamphysics.com 2016
 Fission and Fusion Book pg 286-287 cgrahamphysics.com 2016 Review BE is the energy that holds a nucleus together. This is equal to the mass defect of the nucleus. Also called separation energy. The energy
Fission and Fusion Book pg 286-287 cgrahamphysics.com 2016 Review BE is the energy that holds a nucleus together. This is equal to the mass defect of the nucleus. Also called separation energy. The energy
NSCI 314 LIFE IN THE COSMOS
 NSCI 314 LIFE IN THE COSMOS 2 BASIC ASTRONOMY, AND STARS AND THEIR EVOLUTION Dr. Karen Kolehmainen Department of Physics CSUSB COURSE WEBPAGE: http://physics.csusb.edu/~karen MOTIONS IN THE SOLAR SYSTEM
NSCI 314 LIFE IN THE COSMOS 2 BASIC ASTRONOMY, AND STARS AND THEIR EVOLUTION Dr. Karen Kolehmainen Department of Physics CSUSB COURSE WEBPAGE: http://physics.csusb.edu/~karen MOTIONS IN THE SOLAR SYSTEM
A Short History of the Universe. The universe in a grain of sand Particles and forces The Big Bang Early history of the universe Nucleosynthesis
 A Short History of the Universe The universe in a grain of sand Particles and forces The Big Bang Early history of the universe Nucleosynthesis Molecular Level Quartz SiO 2 Composed of linked spirals of
A Short History of the Universe The universe in a grain of sand Particles and forces The Big Bang Early history of the universe Nucleosynthesis Molecular Level Quartz SiO 2 Composed of linked spirals of
NUCLEI. Atomic mass unit
 13 NUCLEI Atomic mass unit It is a unit used to express the mass of atoms and particles inside it. One atomic mass unit is the mass of atom. 1u = 1.660539 10. Chadwick discovered neutron. The sum of number
13 NUCLEI Atomic mass unit It is a unit used to express the mass of atoms and particles inside it. One atomic mass unit is the mass of atom. 1u = 1.660539 10. Chadwick discovered neutron. The sum of number
Lecture 24: Cosmology: The First Three Minutes. Astronomy 111 Monday November 27, 2017
 Lecture 24: Cosmology: The First Three Minutes Astronomy 111 Monday November 27, 2017 Reminders Last star party of the semester tomorrow night! Online homework #11 due Monday at 3pm The first three minutes
Lecture 24: Cosmology: The First Three Minutes Astronomy 111 Monday November 27, 2017 Reminders Last star party of the semester tomorrow night! Online homework #11 due Monday at 3pm The first three minutes
LECTURE 25 NUCLEAR STRUCTURE AND STABILITY. Instructor: Kazumi Tolich
 LECTURE 25 NUCLEAR STRUCTURE AND STABILITY Instructor: Kazumi Tolich Lecture 25 2 30.1 Nuclear structure Isotopes Atomic mass 30.2 Nuclear stability Biding energy 30.3 Forces and energy in the nucleus
LECTURE 25 NUCLEAR STRUCTURE AND STABILITY Instructor: Kazumi Tolich Lecture 25 2 30.1 Nuclear structure Isotopes Atomic mass 30.2 Nuclear stability Biding energy 30.3 Forces and energy in the nucleus
ASTR 1120 General Astronomy: Stars & Galaxies. OUR Universe: Accelerating Universe
 ASTR 1120 General Astronomy: Stars & Galaxies FINAL: Saturday, Dec 12th, 7:30pm, HERE ALTERNATE FINAL: Monday, Dec 7th, 5:30pm in Muenzinger E131 Last OBSERVING session, Tue, Dec.8th, 7pm Please check
ASTR 1120 General Astronomy: Stars & Galaxies FINAL: Saturday, Dec 12th, 7:30pm, HERE ALTERNATE FINAL: Monday, Dec 7th, 5:30pm in Muenzinger E131 Last OBSERVING session, Tue, Dec.8th, 7pm Please check
Nuclear Physics Questions. 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of?
 Nuclear Physics Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
Nuclear Physics Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
ASTR 101 General Astronomy: Stars & Galaxies
 ASTR 101 General Astronomy: Stars & Galaxies ANNOUNCEMENTS FINAL EXAM: THURSDAY, May 14 th, 11:15am Last Astronomy public talk, May 8 th (up to 3% Extra class credit (see Blackboard announcement for details)
ASTR 101 General Astronomy: Stars & Galaxies ANNOUNCEMENTS FINAL EXAM: THURSDAY, May 14 th, 11:15am Last Astronomy public talk, May 8 th (up to 3% Extra class credit (see Blackboard announcement for details)
Astro-2: History of the Universe. Lecture 12; May
 Astro-2: History of the Universe Lecture 12; May 23 2013 Previously on astro-2 The four fundamental interactions are? Strong, weak, electromagnetic and gravity. We think they are unified at high energies,
Astro-2: History of the Universe Lecture 12; May 23 2013 Previously on astro-2 The four fundamental interactions are? Strong, weak, electromagnetic and gravity. We think they are unified at high energies,
Doppler Effect. Sound moving TOWARDS. Sound moving AWAY 9/22/2017. Occurs when the source of sound waves moves towards or away
 Burkey- ESS QUIZ Thursday At the instant of the Big Bang, all the matter, energy, time, & space in the Universe was condensed into a single, tiny point. We call this Singularity. Doppler Effect Occurs
Burkey- ESS QUIZ Thursday At the instant of the Big Bang, all the matter, energy, time, & space in the Universe was condensed into a single, tiny point. We call this Singularity. Doppler Effect Occurs
:Lecture 27: Stellar Nucleosynthesis. Cassieopia A
 :Lecture 27: Stellar Nucleosynthesis Cassieopia A Major nuclear burning processes Common feature is release of energy by consumption of nuclear fuel. Rates of energy release vary enormously. Nuclear processes
:Lecture 27: Stellar Nucleosynthesis Cassieopia A Major nuclear burning processes Common feature is release of energy by consumption of nuclear fuel. Rates of energy release vary enormously. Nuclear processes
Lecture 24: Testing Stellar Evolution Readings: 20-6, 21-3, 21-4
 Lecture 24: Testing Stellar Evolution Readings: 20-6, 21-3, 21-4 Key Ideas HR Diagrams of Star Clusters Ages from the Main Sequence Turn-off Open Clusters Young clusters of ~1000 stars Blue Main-Sequence
Lecture 24: Testing Stellar Evolution Readings: 20-6, 21-3, 21-4 Key Ideas HR Diagrams of Star Clusters Ages from the Main Sequence Turn-off Open Clusters Young clusters of ~1000 stars Blue Main-Sequence
Lecture PowerPoints. Chapter 33 Physics: Principles with Applications, 7 th edition Giancoli
 Lecture PowerPoints Chapter 33 Physics: Principles with Applications, 7 th edition Giancoli This work is protected by United States copyright laws and is provided solely for the use of instructors in teaching
Lecture PowerPoints Chapter 33 Physics: Principles with Applications, 7 th edition Giancoli This work is protected by United States copyright laws and is provided solely for the use of instructors in teaching
Stellar energy generation on the main sequence
 Stellar energy generation on the main sequence Once fusion reactions begin at the center of a cloud of gas, we call the object a star. For the bulk of its lifetime, a star fuses hydrogen into helium in
Stellar energy generation on the main sequence Once fusion reactions begin at the center of a cloud of gas, we call the object a star. For the bulk of its lifetime, a star fuses hydrogen into helium in
The Nucleosynthesis of Chemical Elements
 The Nucleosynthesis of Chemical Elements Dr. Adriana Banu, James Madison University January 22, Saturday Morning Physics 11 Big questions We knew for long time that our energy comes from Sun! 1.But what
The Nucleosynthesis of Chemical Elements Dr. Adriana Banu, James Madison University January 22, Saturday Morning Physics 11 Big questions We knew for long time that our energy comes from Sun! 1.But what
Cosmology. What is Cosmology?
 Cosmology What is Cosmology? The study of the structure and evolution of the entire universe The idea is to form picture of the entire Universe: origin, size, and future We will make assumptions that what
Cosmology What is Cosmology? The study of the structure and evolution of the entire universe The idea is to form picture of the entire Universe: origin, size, and future We will make assumptions that what
Cosmic Fission: the Synthesis of the Heavy Elements and the role of Fission
 Cosmic Fission: the Synthesis of the Heavy Elements and the role of Fission Sunniva Johanne Rose Trial lecture 29th March 2017 In 2002, the American Research Council made a list over the 11 Greatest Unanswered
Cosmic Fission: the Synthesis of the Heavy Elements and the role of Fission Sunniva Johanne Rose Trial lecture 29th March 2017 In 2002, the American Research Council made a list over the 11 Greatest Unanswered
Late stages of stellar evolution for high-mass stars
 Late stages of stellar evolution for high-mass stars Low-mass stars lead a relatively peaceful life in their old age: although some may gently blow off their outer envelopes to form beautiful planetary
Late stages of stellar evolution for high-mass stars Low-mass stars lead a relatively peaceful life in their old age: although some may gently blow off their outer envelopes to form beautiful planetary
Lecture 36: The First Three Minutes Readings: Sections 29-1, 29-2, and 29-4 (29-3)
 Lecture 36: The First Three Minutes Readings: Sections 29-1, 29-2, and 29-4 (29-3) Key Ideas Physics of the Early Universe Informed by experimental & theoretical physics Later stages confirmed by observations
Lecture 36: The First Three Minutes Readings: Sections 29-1, 29-2, and 29-4 (29-3) Key Ideas Physics of the Early Universe Informed by experimental & theoretical physics Later stages confirmed by observations
Low mass stars. Sequence Star Giant. Red. Planetary Nebula. White Dwarf. Interstellar Cloud. White Dwarf. Interstellar Cloud. Planetary Nebula.
 Low mass stars Interstellar Cloud Main Sequence Star Red Giant Planetary Nebula White Dwarf Interstellar Cloud Main Sequence Star Red Giant Planetary Nebula White Dwarf Low mass stars Interstellar Cloud
Low mass stars Interstellar Cloud Main Sequence Star Red Giant Planetary Nebula White Dwarf Interstellar Cloud Main Sequence Star Red Giant Planetary Nebula White Dwarf Low mass stars Interstellar Cloud
Big Bang Theory How the Universe was Formed
 Big Bang Theory How the Universe was Formed Objectives Explain the Big Bang Theory. Give evidence to support the Big Bang Theory. Dispel misconceptions about the Big Bang Theory. Explain problems with
Big Bang Theory How the Universe was Formed Objectives Explain the Big Bang Theory. Give evidence to support the Big Bang Theory. Dispel misconceptions about the Big Bang Theory. Explain problems with
Basic Nuclear Theory. Lecture 1 The Atom and Nuclear Stability
 Basic Nuclear Theory Lecture 1 The Atom and Nuclear Stability Introduction Nuclear power is made possible by energy emitted from either nuclear fission or nuclear fusion. Current nuclear power plants utilize
Basic Nuclear Theory Lecture 1 The Atom and Nuclear Stability Introduction Nuclear power is made possible by energy emitted from either nuclear fission or nuclear fusion. Current nuclear power plants utilize
Atomic and Nuclear Physics. Topic 7.3 Nuclear Reactions
 Atomic and Nuclear Physics Topic 7.3 Nuclear Reactions Nuclear Reactions Rutherford conducted experiments bombarding nitrogen gas with alpha particles from bismuth-214. He discovered that fast-moving particles
Atomic and Nuclear Physics Topic 7.3 Nuclear Reactions Nuclear Reactions Rutherford conducted experiments bombarding nitrogen gas with alpha particles from bismuth-214. He discovered that fast-moving particles
12 Big Bang Nucleosynthesis. introduc)on to Astrophysics, C. Bertulani, Texas A&M-Commerce 1
 12 Big Bang Nucleosynthesis introduc)on to Astrophysics, C. Bertulani, Texas A&M-Commerce 1 12.1 The Early Universe According to the accepted cosmological theories: The Universe has cooled during its expansion
12 Big Bang Nucleosynthesis introduc)on to Astrophysics, C. Bertulani, Texas A&M-Commerce 1 12.1 The Early Universe According to the accepted cosmological theories: The Universe has cooled during its expansion
Physics 142 Modern Physics 2 Page 1. Nuclear Physics
 Physics 142 Modern Physics 2 Page 1 Nuclear Physics The Creation of the Universe was made possible by a grant from Texas Instruments. Credit on a PBS Program Overview: the elements are not elementary The
Physics 142 Modern Physics 2 Page 1 Nuclear Physics The Creation of the Universe was made possible by a grant from Texas Instruments. Credit on a PBS Program Overview: the elements are not elementary The
Chapter CHAPTER 11 ORIGIN OF THE ELEMENTS
 Chapter 11 165 CHAPTER 11 ORIGIN OF THE ELEMENTS The nuclear reactions of the early universe lead to the production of light nuclei like 2 H and 4 He. There are few reaction pathways leading to nuclei
Chapter 11 165 CHAPTER 11 ORIGIN OF THE ELEMENTS The nuclear reactions of the early universe lead to the production of light nuclei like 2 H and 4 He. There are few reaction pathways leading to nuclei
Cosmology: Building the Universe.
 Cosmology: Building the Universe. The term has several different meanings. We are interested in physical cosmology - the study of the origin and development of the physical universe, and all the structure
Cosmology: Building the Universe. The term has several different meanings. We are interested in physical cosmology - the study of the origin and development of the physical universe, and all the structure
Astronomy 182: Origin and Evolution of the Universe
 Astronomy 182: Origin and Evolution of the Universe Prof. Josh Frieman Lecture 11 Nov. 13, 2015 Today Cosmic Microwave Background Big Bang Nucleosynthesis Assignments This week: read Hawley and Holcomb,
Astronomy 182: Origin and Evolution of the Universe Prof. Josh Frieman Lecture 11 Nov. 13, 2015 Today Cosmic Microwave Background Big Bang Nucleosynthesis Assignments This week: read Hawley and Holcomb,
The Evolution of High-mass Stars
 The Evolution of High-mass Stars For stars with initial main-sequence mass greater than around 6M o the evolution is much faster and fundamentally different. 1M o 3M o 15M o 25M o 10 x 10 9 years 500 x
The Evolution of High-mass Stars For stars with initial main-sequence mass greater than around 6M o the evolution is much faster and fundamentally different. 1M o 3M o 15M o 25M o 10 x 10 9 years 500 x
Activity 12: Energy from Nuclear Reactions
 Name Section Activity 12: Energy from Nuclear Reactions 12.1 A Model of the Composition of Nucleons 1) Formation of Nucleons Nucleons consist of quark trios. a) Place orange or green quarks into the metal
Name Section Activity 12: Energy from Nuclear Reactions 12.1 A Model of the Composition of Nucleons 1) Formation of Nucleons Nucleons consist of quark trios. a) Place orange or green quarks into the metal
Zach Meisel, PAN 2016
 Nuclear Astrophysics Zach Meisel, PAN 2016 Nuclear Astrophysics is the study of: Energy generation in stars and stellar explosions Extremely dense matter The origin of the elements If the sun were powered
Nuclear Astrophysics Zach Meisel, PAN 2016 Nuclear Astrophysics is the study of: Energy generation in stars and stellar explosions Extremely dense matter The origin of the elements If the sun were powered
Lecture 17: the CMB and BBN
 Lecture 17: the CMB and BBN As with all course material (including homework, exams), these lecture notes are not be reproduced, redistributed, or sold in any form. Peering out/back into the Universe As
Lecture 17: the CMB and BBN As with all course material (including homework, exams), these lecture notes are not be reproduced, redistributed, or sold in any form. Peering out/back into the Universe As
Computational Applications in Nuclear Astrophysics using JAVA
 Computational Applications in Nuclear Astrophysics using JAVA Lecture: Friday 10:15-11:45 Room NB 6/99 Jim Ritman and Elisabetta Prencipe j.ritman@fz-juelich.de e.prencipe@fz-juelich.de Computer Lab: Friday
Computational Applications in Nuclear Astrophysics using JAVA Lecture: Friday 10:15-11:45 Room NB 6/99 Jim Ritman and Elisabetta Prencipe j.ritman@fz-juelich.de e.prencipe@fz-juelich.de Computer Lab: Friday
The Sun = Typical Star
 The Sun = Typical Star Some Properties Diameter - 109 times Earth s Volume - about 1,000,000 times Earth s Mass - about 300,000 times Earth s 99.8% of Solar System Density = Mass/Volume = 1.4 g/cm 3 The
The Sun = Typical Star Some Properties Diameter - 109 times Earth s Volume - about 1,000,000 times Earth s Mass - about 300,000 times Earth s 99.8% of Solar System Density = Mass/Volume = 1.4 g/cm 3 The
Nuclear Astrophysics - I
 Nuclear Astrophysics - I Carl Brune Ohio University, Athens Ohio Exotic Beam Summer School 2016 July 20, 2016 Astrophysics and Cosmology Observations Underlying Physics Electromagnetic Spectrum: radio,
Nuclear Astrophysics - I Carl Brune Ohio University, Athens Ohio Exotic Beam Summer School 2016 July 20, 2016 Astrophysics and Cosmology Observations Underlying Physics Electromagnetic Spectrum: radio,
Alta Chemistry CHAPTER 25. Nuclear Chemistry: Radiation, Radioactivity & its Applications
 CHAPTER 25 Nuclear Chemistry: Radiation, Radioactivity & its Applications Nuclear Chemistry Nuclear Chemistry deals with changes in the nucleus The nucleus of an atom contains Protons Positively Charged
CHAPTER 25 Nuclear Chemistry: Radiation, Radioactivity & its Applications Nuclear Chemistry Nuclear Chemistry deals with changes in the nucleus The nucleus of an atom contains Protons Positively Charged
THE NUCLEUS: A CHEMIST S VIEW Chapter 20
 THE NUCLEUS: A CHEMIST S VIEW Chapter 20 "For a long time I have considered even the craziest ideas about [the] atom[ic] nucleus... and suddenly discovered the truth." [shell model of the nucleus]. Maria
THE NUCLEUS: A CHEMIST S VIEW Chapter 20 "For a long time I have considered even the craziest ideas about [the] atom[ic] nucleus... and suddenly discovered the truth." [shell model of the nucleus]. Maria
Particles in the Early Universe
 Particles in the Early Universe David Morrissey Saturday Morning Physics, October 16, 2010 Using Little Stuff to Explain Big Stuff David Morrissey Saturday Morning Physics, October 16, 2010 Can we explain
Particles in the Early Universe David Morrissey Saturday Morning Physics, October 16, 2010 Using Little Stuff to Explain Big Stuff David Morrissey Saturday Morning Physics, October 16, 2010 Can we explain
R Process Nucleosynthesis And Its Site. Mario A. Riquelme Theore<cal Seminar Fall 2009
 R Process Nucleosynthesis And Its Site Mario A. Riquelme Theore
R Process Nucleosynthesis And Its Site Mario A. Riquelme Theore
Matter and Energy. Previous studies have taught us that matter and energy cannot be created nor destroyed We balance equations to obey this law.
 Fission & Fusion Matter and Energy Previous studies have taught us that matter and energy cannot be created nor destroyed We balance equations to obey this law. 2 H 2 O 2 H 2 + O 2 We now need to understand
Fission & Fusion Matter and Energy Previous studies have taught us that matter and energy cannot be created nor destroyed We balance equations to obey this law. 2 H 2 O 2 H 2 + O 2 We now need to understand
Lecture Outlines Chapter 32. Physics, 3 rd Edition James S. Walker
 Lecture Outlines Chapter 32 Physics, 3 rd Edition James S. Walker 2007 Pearson Prentice Hall This work is protected by United States copyright laws and is provided solely for the use of instructors in
Lecture Outlines Chapter 32 Physics, 3 rd Edition James S. Walker 2007 Pearson Prentice Hall This work is protected by United States copyright laws and is provided solely for the use of instructors in
13 Synthesis of heavier elements. introduc)on to Astrophysics, C. Bertulani, Texas A&M-Commerce 1
 13 Synthesis of heavier elements introduc)on to Astrophysics, C. Bertulani, Texas A&M-Commerce 1 The triple α Reaction When hydrogen fusion ends, the core of a star collapses and the temperature can reach
13 Synthesis of heavier elements introduc)on to Astrophysics, C. Bertulani, Texas A&M-Commerce 1 The triple α Reaction When hydrogen fusion ends, the core of a star collapses and the temperature can reach
Life of a High-Mass Stars
 Life of a High-Mass Stars 1 Evolutionary Tracks Paths of high-mass stars on the HR Diagram are different from those of low-mass stars. Once these stars leave the main sequence, they quickly grow in size
Life of a High-Mass Stars 1 Evolutionary Tracks Paths of high-mass stars on the HR Diagram are different from those of low-mass stars. Once these stars leave the main sequence, they quickly grow in size
Earlier in time, all the matter must have been squeezed more tightly together and a lot hotter AT R=0 have the Big Bang
 Re-cap from last lecture Discovery of the CMB- logic From Hubble s observations, we know the Universe is expanding This can be understood theoretically in terms of solutions of GR equations Earlier in
Re-cap from last lecture Discovery of the CMB- logic From Hubble s observations, we know the Universe is expanding This can be understood theoretically in terms of solutions of GR equations Earlier in
Subir Sarkar
 Trinity 2016 Oxford ² The universe observed ² Relativistic world models ² Reconstructing the thermal history ² Big bang nucleosynthesis ² Dark matter: astrophysical observations ² Dark matter: relic particles
Trinity 2016 Oxford ² The universe observed ² Relativistic world models ² Reconstructing the thermal history ² Big bang nucleosynthesis ² Dark matter: astrophysical observations ² Dark matter: relic particles
Cosmology. Big Bang and Inflation
 Cosmology Big Bang and Inflation What is the Universe? Everything we can know about is part of the universe. Everything we do know about is part of the universe. Everything! The Universe is expanding If
Cosmology Big Bang and Inflation What is the Universe? Everything we can know about is part of the universe. Everything we do know about is part of the universe. Everything! The Universe is expanding If
Unit 1 Atomic Structure
 Unit 1 Atomic Structure 3-1 The Atom: From Philosophical Idea to Scientific Theory I. Atomic Theory A. Modern Atomic Theory 1. All matter is made up of very tiny particles called atoms 2. Atoms of the
Unit 1 Atomic Structure 3-1 The Atom: From Philosophical Idea to Scientific Theory I. Atomic Theory A. Modern Atomic Theory 1. All matter is made up of very tiny particles called atoms 2. Atoms of the
Lecture notes 8: Nuclear reactions in solar/stellar interiors
 Lecture notes 8: Nuclear reactions in solar/stellar interiors Atomic Nuclei We will henceforth often write protons 1 1p as 1 1H to underline that hydrogen, deuterium and tritium are chemically similar.
Lecture notes 8: Nuclear reactions in solar/stellar interiors Atomic Nuclei We will henceforth often write protons 1 1p as 1 1H to underline that hydrogen, deuterium and tritium are chemically similar.
Stellar Evolution. The lives of low-mass stars. And the lives of massive stars
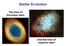 Stellar Evolution The lives of low-mass stars And the lives of massive stars Stars of High Mass High mass stars fuse H He, but do so in a different reaction: the CNO cycle Carbon is a catalyst, speeding
Stellar Evolution The lives of low-mass stars And the lives of massive stars Stars of High Mass High mass stars fuse H He, but do so in a different reaction: the CNO cycle Carbon is a catalyst, speeding
The expansion of the Universe, and the big bang
 The expansion of the Universe, and the big bang Q: What is Hubble s law? A. The larger the galaxy, the faster it is moving way from us. B. The farther away the galaxy, the faster it is moving away from
The expansion of the Universe, and the big bang Q: What is Hubble s law? A. The larger the galaxy, the faster it is moving way from us. B. The farther away the galaxy, the faster it is moving away from
Planetary Nebulae White dwarfs
 Life of a Low-Mass Star AST 101 Introduction to Astronomy: Stars & Galaxies Planetary Nebulae White dwarfs REVIEW END STATE: PLANETARY NEBULA + WHITE DWARF WHAS IS A WHITE DWARF? Exposed core of a low-mass
Life of a Low-Mass Star AST 101 Introduction to Astronomy: Stars & Galaxies Planetary Nebulae White dwarfs REVIEW END STATE: PLANETARY NEBULA + WHITE DWARF WHAS IS A WHITE DWARF? Exposed core of a low-mass
Outline - March 18, H-R Diagram Review. Protostar to Main Sequence Star. Midterm Exam #2 Tuesday, March 23
 Midterm Exam #2 Tuesday, March 23 Outline - March 18, 2010 Closed book Will cover Lecture 8 (Special Relativity) through Lecture 14 (Star Formation) only If a topic is in the book, but was not covered
Midterm Exam #2 Tuesday, March 23 Outline - March 18, 2010 Closed book Will cover Lecture 8 (Special Relativity) through Lecture 14 (Star Formation) only If a topic is in the book, but was not covered
Forces and Nuclear Processes
 Forces and Nuclear Processes To understand how stars generate the enormous amounts of light they produce will require us to delve into a wee bit of physics. First we will examine the forces that act at
Forces and Nuclear Processes To understand how stars generate the enormous amounts of light they produce will require us to delve into a wee bit of physics. First we will examine the forces that act at
Chapter 22: Cosmology - Back to the Beginning of Time
 Chapter 22: Cosmology - Back to the Beginning of Time Expansion of Universe implies dense, hot start: Big Bang Future of universe depends on the total amount of dark and normal matter Amount of matter
Chapter 22: Cosmology - Back to the Beginning of Time Expansion of Universe implies dense, hot start: Big Bang Future of universe depends on the total amount of dark and normal matter Amount of matter
Binding Energy. Bởi: OpenStaxCollege
 Binding Energy Bởi: OpenStaxCollege The more tightly bound a system is, the stronger the forces that hold it together and the greater the energy required to pull it apart. We can therefore learn about
Binding Energy Bởi: OpenStaxCollege The more tightly bound a system is, the stronger the forces that hold it together and the greater the energy required to pull it apart. We can therefore learn about
1) Radioactive Decay, Nucleosynthesis, and Basic Geochronology
 1) Radioactive Decay, Nucleosynthesis, and Basic Geochronology Reading (all from White s Notes) Lecture 1: Introduction And Physics Of The Nucleus: Skim Lecture 1: Radioactive Decay- Read all Lecture 3:
1) Radioactive Decay, Nucleosynthesis, and Basic Geochronology Reading (all from White s Notes) Lecture 1: Introduction And Physics Of The Nucleus: Skim Lecture 1: Radioactive Decay- Read all Lecture 3:
17.3 Life as a High-Mass Star
 17.3 Life as a High-Mass Star Our goals for learning: What are the life stages of a high-mass star? How do high-mass stars make the elements necessary for life? How does a high-mass star die? What are
17.3 Life as a High-Mass Star Our goals for learning: What are the life stages of a high-mass star? How do high-mass stars make the elements necessary for life? How does a high-mass star die? What are
Atoms have two separate parts. The nucleus and the electron cloud.
 Name Ch. 5 - Atomic Structure Pre-AP Modern Atomic Theory All atoms are made of three subatomic (smaller than the atom) particles: the protons, the electrons and the neutrons. (P.E.N. s) There are particles
Name Ch. 5 - Atomic Structure Pre-AP Modern Atomic Theory All atoms are made of three subatomic (smaller than the atom) particles: the protons, the electrons and the neutrons. (P.E.N. s) There are particles
Unit 1 Atomic Structure
 Unit 1 Atomic Structure Defining the Atom I. Atomic Theory A. Modern Atomic Theory 1. All matter is made up of very tiny particles called atoms 2. Atoms of the same element are chemically alike 3. Individual
Unit 1 Atomic Structure Defining the Atom I. Atomic Theory A. Modern Atomic Theory 1. All matter is made up of very tiny particles called atoms 2. Atoms of the same element are chemically alike 3. Individual
Electromagnetic Waves
 Big Bang Theory OUTLINE First 25 minutes of the video Notes on waves, Doppler effect, and red/blue shift Mini lab on spectrums and how they are used to understand red and blue shift Mini lab on the expanding
Big Bang Theory OUTLINE First 25 minutes of the video Notes on waves, Doppler effect, and red/blue shift Mini lab on spectrums and how they are used to understand red and blue shift Mini lab on the expanding
Heavy Element Nucleosynthesis. A summary of the nucleosynthesis of light elements is as follows
 Heavy Element Nucleosynthesis A summary of the nucleosynthesis of light elements is as follows 4 He Hydrogen burning 3 He Incomplete PP chain (H burning) 2 H, Li, Be, B Non-thermal processes (spallation)
Heavy Element Nucleosynthesis A summary of the nucleosynthesis of light elements is as follows 4 He Hydrogen burning 3 He Incomplete PP chain (H burning) 2 H, Li, Be, B Non-thermal processes (spallation)
The light curves for a nova look like the following.
 End of high mass stars Nova Supernova Type I Type II Stellar nucleo-synthesis Stellar Recycling Nova (means new): is a star that suddenly increases greatly in brightness, then slowly fades back to its
End of high mass stars Nova Supernova Type I Type II Stellar nucleo-synthesis Stellar Recycling Nova (means new): is a star that suddenly increases greatly in brightness, then slowly fades back to its
1920s 1990s (from Friedmann to Freedman)
 20 th century cosmology 1920s 1990s (from Friedmann to Freedman) theoretical technology available, but no data 20 th century: birth of observational cosmology Hubble s law ~1930 Development of astrophysics
20 th century cosmology 1920s 1990s (from Friedmann to Freedman) theoretical technology available, but no data 20 th century: birth of observational cosmology Hubble s law ~1930 Development of astrophysics
Nuclear Physics and Radioactivity
 Nuclear Physics and Radioactivity Structure and Properties of the Nucleus Nucleus is made of protons and neutrons Proton has positive charge: Neutron is electrically neutral: Neutrons and protons are collectively
Nuclear Physics and Radioactivity Structure and Properties of the Nucleus Nucleus is made of protons and neutrons Proton has positive charge: Neutron is electrically neutral: Neutrons and protons are collectively
NJCTL.org 2015 AP Physics 2 Nuclear Physics
 AP Physics 2 Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
AP Physics 2 Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
Chapter 17 Cosmology
 Chapter 17 Cosmology Over one thousand galaxies visible The Universe on the Largest Scales No evidence of structure on a scale larger than 200 Mpc On very large scales, the universe appears to be: Homogenous
Chapter 17 Cosmology Over one thousand galaxies visible The Universe on the Largest Scales No evidence of structure on a scale larger than 200 Mpc On very large scales, the universe appears to be: Homogenous
Chapter 27 The Early Universe Pearson Education, Inc.
 Chapter 27 The Early Universe Units of Chapter 27 27.1 Back to the Big Bang 27.2 The Evolution of the Universe More on Fundamental Forces 27.3 The Formation of Nuclei and Atoms 27.4 The Inflationary Universe
Chapter 27 The Early Universe Units of Chapter 27 27.1 Back to the Big Bang 27.2 The Evolution of the Universe More on Fundamental Forces 27.3 The Formation of Nuclei and Atoms 27.4 The Inflationary Universe
10/26/ Star Birth. Chapter 13: Star Stuff. How do stars form? Star-Forming Clouds. Mass of a Star-Forming Cloud. Gravity Versus Pressure
 10/26/16 Lecture Outline 13.1 Star Birth Chapter 13: Star Stuff How do stars form? Our goals for learning: How do stars form? How massive are newborn stars? Star-Forming Clouds Stars form in dark clouds
10/26/16 Lecture Outline 13.1 Star Birth Chapter 13: Star Stuff How do stars form? Our goals for learning: How do stars form? How massive are newborn stars? Star-Forming Clouds Stars form in dark clouds
