Trilogy - Science Revision
|
|
|
- Kristin Watkins
- 5 years ago
- Views:
Transcription
1 Trilogy - Science Revision Sept June 2018 Year 11 Subject Unit Date AM/PM Name: Biology Paper 1 Biology Paper 2 Chemistry Paper 1 Chemistry Paper 2 Physics Paper 1 Physics Paper 2 Target: Tuesday 15 May pm Monday 11 June am Thursday 17 May am Wednesday 13 June am Wednesday 23 May pm Friday 15 June am
2 2 1 Draw and label a plant & animal cell. Write the function of each cell part. Define term specialised cell & give examples. Describe the processes that happen during the cell cycle, including mitosis. Describe stem cells & the uses of stem cells, including sources of stem cells in plants and animals and their roles Explain the processes of osmosis, active transport and diffusion. Describe how to prepare an uncontaminated culture using aseptic technique. Describe the structure of the human heart and lungs. Describe the digestive enzymes, including their names, sites of production and actions Give the charge and mass of each part of an atom. Calculate the number of p, n & e for lithium Li. Define the term isotope. Describe the trends and properties in Groups 1, 7 & 0 of the Periodic Table. Explain why elements in the same group have similar properties and how to use the periodic table to predict the reactivity of elements Describe the three main types of bonds: ionic bonds, covalent bonds and metallic bonds in terms of electrostatic forces and the transfer or sharing of electrons Compare nano dimensions to typical dimensions of atoms and molecules. Explain how the structure of metals and alloys affects their properties, including explaining why they are good conductors Explain why alloys are harder than pure metals Week 1-5: Paper 1 State the equation that shows the kinetic energy of an object. State the equation that shows the gravitational potential energy gained by an object raised above ground level. Define the term 'specific heat capacity' State the equation for efficiency. State the law of conservation of energy. Explain what wasted energy is and give examples. List the main renewable and non-renewable energy resources and define what a renewable energy resource is. Compare ways that different energy resources are used, including uses in transport, electricity generation and heating State the equation that links charge and current. Define the terms electric current and resistance State the equation that links current, potential difference or resistance Define the term power and state the two equations that calculate it. Write the word and balanced symbol equation which shows the reaction between hydrogen and chlorine, forming hydrochloric acid. Cells- describe what differentiation is. Describe how the rate of cooling of a building can be increased. Describe the early attempts to classify elements. Explain the creation and attributes of Mendeleev s periodic table. Create a table showing the potential risks, benefits and issues with using stem cells in medical research/treatments (inc diabetes and paralysis) Evaluate the use of different energy resources, taking into account any ethical and environmental issues Draw dot and cross diagrams for the molecules of hydrogen,cl2, O2, N2, hydrogen chloride, H2O, NH3 and methane Explain how the effectiveness of an exchange surface can be increased Define the term ohmic conductor Explain how the structure of ionic compounds affects their properties Describe what cancer is and explain the difference between benign and malignant tumours Describe how patients can be treated in the case of heart failure (markscheme available) r (markscheme available) r (markscheme available) assess areas t hat require development from this weeks require development from this weeks require development from this weeks
3 4 3 Week 1-5: Paper 1 Describe plant tissues (e.g epidermal, palisade mesophyll, and describe their functions Describe the role of stomata and guard cells. Describe how plant diseases can be detected and identified Describe what the relative formula mass (Mr) of a compound is and calculate the relative formula mass of a compound, given its formula Explain how the concentration of a solution in mol/dm3 is related to the mass of the solute and the volume of the solution Show by calculation and explanation that parts in series have the same current passing through them Solve problems for circuits which include resistors in series using the concept of equivalent resistance. Explain why whenever a measurement is made there is always some uncertainty about the result obtained Explain how root hair cells, xylem and phloem are adapted to their functions Describe defences that stop pathogens entering the human body. Describe how white blood cells destroy pathogens. Describe how vaccination works Explain what a mole is with reference to relative formula mass and Avogadro's constant Identify and describe the function of each wire in a three-core cable connected to the mains. Explain the difference between direct and alternating voltage and current, stating what UK mains is. Use moles to write a balanced equation when given the masses of reactants and products Describe how new drugs are tested, Describe what happens in photosynthesis, including using a word equation and recognise the chemical formulas for carbon dioxide, water, oxygen & glucose Use the reactivity series to predict the outcome of displacement reactions Describe the difference compared with Group 1 in melting points, densities, strength, hardness and reactivity with oxygen, water and halogens. Exemplify these general properties by reference to Cr, Mn, Fe, Co, Ni, Cu. Describe how to carry out titrations using strong acids and strong alkalis only (sulfuric, hydrochloric and nitric acids only) to find the reacting volumes accurately State the two equations used to calculate power. Describe the National Grid. Explain why and how some metals are extracted through reduction with carbon. Give the definition of oxidation and reduction. Explain how the limiting factors of photosynthesis interact, inc graphical interpretation (two/three factors) Describe what happens in respiration including using a word equation and recognise the chemical formulas for carbon dioxide, water, oxygen & glucose Describe how monoclonal antibodies are produced. Define the terms acid and alkali Describe the use of universal indicator to measure the approximate ph of a solution and use the ph scale to identify acidic or alkaline solutions calculate the percentage yield of a product from the actual yield of a reaction State the equation used to calculate density. Recognise/draw simple diagrams to model the difference between solids, liquids and gases Use the particle model to explain why a change of state is reversible and affects the properties of a substance, but not its mass. Use and explain the terms dilute and concentrated (in terms of amount of substance) and weak and strong (in terms of the degree of ionisation) in relation to acids Explain what happens to accumulated lactic acid in the body END OF BIOLOGY
4 6 5 Week 1-5: Paper 2 State what the internal energy of a system is. Interpret and draw heating and cooling graphs that include changes of state Describe the process of electrolysis Describe how ionic compounds can conduct electricity when dissolved in water and describe these solutions as electrolytes Explain, with reference to the particle model, the effect of changing the temperature of a gas held at constant volume on its pressure Describe the electrolysis of aqueous solutions and predict the products of the electrolysis of aqueous solutions containing single ionic compounds Explain the term internal energy Explain why the molecules of a gas are in constant random motion and that the higher the temperature of a gas, the greater the particles average kinetic energy Define electrons, neutrons, protons, isotopes and ions Describe how the atomic model has changed over time due to new experimental evidence Describe the common features of all control systems Explain exothermic and endothermic reactions on the basis of the temperature change of the surroundings and give examples of everyday uses Describe the penetration through materials, the range in air and the ionising power for alpha particles, beta particles and gamma rays Week 6-10: Paper 2 Explain the energy changes in breaking and making bonds and calculate the overall energy change using bond energies END OF CHEMISTRY Define half-life of a radioactive isotope END OF PHYSICS r Identify the cerebral cortex, cerebellum and medulla on a diagram of the brain, and describe their functions. Explain some of the difficulties of investigating brain function and treating brain damage and disease. Describe how different factors affect the rate of a chemical reaction, including the conc., pressure, SA, temp and catalysts State the equation that calculates weight. Identify and describe scalar quantities and vector quantities. Calculate the resultant of two forces that act in a straight line Use vector diagrams to illustrate resolution of forces, equilibrium situations and determine the resultant of two forces, to include both magnitude and direction. Draw and interpret reaction profiles for catalysed reactions Describe what homeostasis is and why it is important stating specific examples from the human body Relate the structures of the eye to their functions. Including: accommodation to focus on near or distant objects adaptation to dim light.
5 8 7 Week 6-10: Paper 2 Describe how information passes through the nervous system. Describe what happens in a reflex action and why reflex actions are important Explain mechanisms to lower or raise body temperature in a given context. Explain what a reversible reaction is, including how the direction can be changed Describe energy transfers involved when work is done and calculate the work done by recalling and using the equation: [ W = Fs ] Explain why work done against the frictional forces acting on an object causes a rise in the temperature of the object. Explain and predict the effect of a change in concentration of reactants or products, temperature, or pressure of gases on the equilibrium position of a reaction. Describe how body temperature is monitored and controlled Describe the endocrine system, the location of pituitary, pancreas, thyroid, aderenal gland, ovary and testis and the role of hormones Describe the consequences of losing or gaining too much water for body cells Describe the process of fractional distillation. Describe and write balanced chemical equations for the complete combustion of hydrocarbon fuels Describe the extension of an elastic object below the limit of proportionality and calculate it by recalling and applying the equation: [ F = ke ] Describe examples of the forces involved in stretching, bending or compressing an object Explain why cracking is useful and why modern life depends on the uses of hydrocarbons. Explain how hormones are used to treat infertility, inc the steps in IVF Explain how the different hormones interact to control the menstrual cycle and ovulation Describe what happens during meiosis and compare to mitosis. Describe how the chromosomes are arranged in human body cells, including the function of the sex chromosomes Describe chromatography, including the terms stationary phase and mobile phase and identify pure substances using paper chromatography Define and apply Newton's second law relating to the acceleration of an object Describe factors that can effect a drivers reactions time Explain how to test for the presence of hydrogen, oxygen, carbon dioxide and chlorine HT ONLY: Construct Punnet square diagrams to predict the outcomes of a monohybrid cross Describe what variation is and how it can be caused within a population. Describe how new species can be formed Describe what genetic engineering is, including examples, and how it is carried out Describe the composition of gases in the Earth's atmosphere using percentages, fractions or ratios Describe examples of momentum in a collision Describe what inertia is and give a definition. Represent motion using velocity time graphs, finding the acceleration from its gradient and distance travelled from the area underneath Explain and apply the idea that, in a closed system, the total momentum before an event is equal to the total momentum after the event. Explain why the levels of carbon dioxide in the atmosphere changes as the oceans were formed HT ONLY: Explain the process of genetic engineering, to include knowledge of enzymes and vectors
6 10 9 Week 6-10: Paper 2 Describe the effects of some plant hormones and different ways people use them to control growth. Describe how organisms are named and classified in the Linnaean system. Describe and interpret evolutionary trees State some potential side effects of global climate change, including discussing scale, risk and environmental implications Define amplitude, wavelength, frequency, period and wave speed and Identify them where appropriate on diagrams List the groups of electromagnetic waves in order of wavelength State the uses of each group of EM wave explaining why wave is suitable for its applications Evaluate arguments for and against the idea that human activities cause a rise in temperature that results in global climate change Explain how scientific advances have led to the proposal of new models of classification, inc three-domain system Describe structural, behavioural and functional adaptations of organisms Describe what an extremophile is. Describe which resources animals and plants compete for, and why they do this Describe the properties and effects of carbon monoxide, sulfur dioxide and particulates in the atmosphere Describe a method to measure the speed of sound waves in air. Explain how different wavelengths of electromagnetic radiation are reflected, refracted, absorbed or transmitted differently by different substances and types of surface. Describe and explain the problems caused by increased amounts of these pollutants in the air Explain how a change in an abiotic or biotic factor might affect a community Describe the processes involved in the carbon cycle & water cycle. Represent the feeding relationships within a community using a food chain and describe these relationships. Describe methods to produce potable water, including desalination of salty water or sea water and the potential problems of desalination Draw the magnetic field pattern of a bar magnet, showing how field strength and direction are indicated and change from one point to another Name and describe alternative biological methods for extracting metals, including phytomining and bioleaching. Explain how and why ecologists use quadrats and transects Describe what biodiversity is, why it is important, and how human activities affect it. Describe how the composition of the atmosphere is changing, and the impact of this on global warming Describe DNA structure and the impact of Mutations. Describe, carry out and interpret a simple comparative life cycle assessment (LCA) of materials or products. Discuss the advantages and disadvantages of LCAs Draw the magnetic field pattern for a straight wire carrying a current and for a solenoid (showing the direction of the field) Explain how the behaviour of a magnetic compass is related to evidence that the core of the Earth must be magnetic. State and use Fleming's left-hand rule and explain what the size of the induced force depends on. Explain how rotation is caused in an electric motor. Discuss how to reduce the consumption of raw resources and explain how reusing and recycling reduces energy use (inc environmental impacts) Explain the consequences of deforestation and peat bog destruction.
7 PAPER 1 WEEK TOPIC 1 TOPIC 2 TOPIC 3 1 Cell structure A simple model of the atom, symbols, relative atomic mass, electronic charge and isotopes Energy changes in a system, and the ways energy is stored before and after such changes Cell division The periodic table Conservation and dissipation of energy 2 Transport in cells Chemical bonds, ionic, covalent and metallic National and global energy resources Principles of organisation & Animal tissues, organs How bonding and structure are related to the properties of Current, potential difference and resistance and organ systems substances 3 Plant tissues, organs and system Chemical measurements, conservation of mass and the Series and parallel circuits quantitative interpretation Communicable diseases Use of amount of substance in relation to masses of pure Domestic uses and safety substances 4 Photosynthesis Reactivity of metals Energy transfers Respiration Reactions of acids Changes of state and the particle model 5 Particle model and pressure Electrolysis Internal energy and energy transfers Atoms and isotopes Exothermic and endothermic reactions Atoms and nuclear radiation PAPER 2 6 Homeostasis Rate of reaction Forces and their interactions 7 The human nervous system Reversible reactions and dynamic equilibrium Work done and energy transfer Hormonal coordination in humans Carbon compounds as fuels and feedstock Forces and elasticity 8 Reproduction Purity, formulations and chromatograph & ID of gases Forces and motion Variation and evolution & The development of The composition and evolution of the Earth's atmosphere Momentum understanding of genetics and evolution 9 Classification Carbon dioxide and methane as greenhouse gases Waves in air, fluids and solids Adaptations, interdependence and competition Common atmospheric pollutants and their sources Electromagnetic waves 10 Organisation of an ecosystem Using the Earth's resources and obtaining potable water Permanent and induced magnetism, magnetic forces and fields Biodiversity and the effect of human interaction on ecosystems Life cycle assessment and recycling The motor effect LEARNING GAPS What am I stuck on? Resources Created?
Biology Paper 1 1hr 15mins 70 marks
 Biology Paper 1 1hr 15mins 70 marks Cell Biology (Yr9) Cell Organisation (Yr9) Infection and Response (Yr10) Bioenergetics (Yr10) Cells Cell Organisation Competition Photosynthesis Microscopy Enzymes Abiotic
Biology Paper 1 1hr 15mins 70 marks Cell Biology (Yr9) Cell Organisation (Yr9) Infection and Response (Yr10) Bioenergetics (Yr10) Cells Cell Organisation Competition Photosynthesis Microscopy Enzymes Abiotic
Biology Combined Chemistry Combined Physics Combined. Content Hours Content Hours Content Hours
 Biology Combined Chemistry Combined Physics Combined Content Hours Content Hours Content Hours Half Term.7.. Communities Competition Ecosystems Interdependence ASSESSED HOMEWORK (Communities).7.. Abiotic
Biology Combined Chemistry Combined Physics Combined Content Hours Content Hours Content Hours Half Term.7.. Communities Competition Ecosystems Interdependence ASSESSED HOMEWORK (Communities).7.. Abiotic
AQA TRILOGY Chemistry (8464) from 2016 Topics T5.1 Atomic structure and the periodic table (Paper 1) To pic. Student Checklist
 Personalised Learning Checklist AQA TRILOGY Chemistry (8464) from 2016 s T5.1 Atomic structure and the periodic table (Paper 1) State that everything is made of atoms and recall what they are 5.1.1 A simple
Personalised Learning Checklist AQA TRILOGY Chemistry (8464) from 2016 s T5.1 Atomic structure and the periodic table (Paper 1) State that everything is made of atoms and recall what they are 5.1.1 A simple
This unit will help you define health, learn about some pathogens and the diseases they cause, medicines and about the immune system.
 YEAR 10 Biology Units 1-5 Biology Unit CB1 Key biological concepts [Paper 1 & CB2 Cells and Control CB3 Genetics CB4 Natural Selection and Genetic Modification CB5 Health, Disease and the Development of
YEAR 10 Biology Units 1-5 Biology Unit CB1 Key biological concepts [Paper 1 & CB2 Cells and Control CB3 Genetics CB4 Natural Selection and Genetic Modification CB5 Health, Disease and the Development of
Science 9-1 Grades Guidance
 U1 U2 U3 Science 9-1 Grades Guidance This guide is designed to help explain the level that a student is working at using examples of the curriculum that a student should be able to access. This is not
U1 U2 U3 Science 9-1 Grades Guidance This guide is designed to help explain the level that a student is working at using examples of the curriculum that a student should be able to access. This is not
Topic Student Checklist R A G
 Personalised Learning Checklist AQA TRILOGY Physics (8464) from 2016 Topics T6.1. Energy Topic Student Checklist R A G 6.1.1 Energy changes in a system, and the ways energy is stored before and after such
Personalised Learning Checklist AQA TRILOGY Physics (8464) from 2016 Topics T6.1. Energy Topic Student Checklist R A G 6.1.1 Energy changes in a system, and the ways energy is stored before and after such
AQA Chemistry (Combined Science) Specification Checklists. Name: Teacher:
 AQA Chemistry (Combined Science) Specification Checklists Name: Teacher: Paper 1-4.1 Atomic structure and the periodic table 4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic
AQA Chemistry (Combined Science) Specification Checklists Name: Teacher: Paper 1-4.1 Atomic structure and the periodic table 4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic
GCSE CHEMISTRY REVISION LIST
 GCSE CHEMISTRY REVISION LIST OCR Gateway Chemistry (J248) from 2016 Topic C1: Particles C1.1 Describe the main features of the particle model in terms of states of matter and change of state Explain, in
GCSE CHEMISTRY REVISION LIST OCR Gateway Chemistry (J248) from 2016 Topic C1: Particles C1.1 Describe the main features of the particle model in terms of states of matter and change of state Explain, in
AQA Chemistry Checklist
 Topic 1. Atomic structure Video: Atoms, elements, compounds, mixtures Use the names and symbols of the first 20 elements in the periodic table, the elements in Groups 1 and 7, and other elements in this
Topic 1. Atomic structure Video: Atoms, elements, compounds, mixtures Use the names and symbols of the first 20 elements in the periodic table, the elements in Groups 1 and 7, and other elements in this
HASTINGS HIGH SCHOOL
 Subject HASTINGS HIGH SCHOOL YEAR 11 EXAMINATION GUIDE 20167-19 COMBINED SCIENCE TRILOGY Physics Course code AQA GCSE COMBINED SCIENCE TRILOGY 8464 Website address Provisional examination dates http://www.aqa.org.uk/subjects/science/gcse/combined-science-trilogy-
Subject HASTINGS HIGH SCHOOL YEAR 11 EXAMINATION GUIDE 20167-19 COMBINED SCIENCE TRILOGY Physics Course code AQA GCSE COMBINED SCIENCE TRILOGY 8464 Website address Provisional examination dates http://www.aqa.org.uk/subjects/science/gcse/combined-science-trilogy-
OCR Chemistry Checklist
 Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Edexcel Chemistry Checklist
 Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
Science Years 9 to 10
 Boardworks Contents Guide Boardworks Presentations: Acids and metal oxides 10 slides Reactions of metal oxides with acids. Adapting to changes 9 slides Ways that animals adapt to their habitats. Air pollution
Boardworks Contents Guide Boardworks Presentations: Acids and metal oxides 10 slides Reactions of metal oxides with acids. Adapting to changes 9 slides Ways that animals adapt to their habitats. Air pollution
Personalised Learning Checklists AQA Chemistry Paper 2
 AQA Chemistry (8462) from 2016 Topics C4.6 The rate and extent of chemical change Calculate the rate of a chemical reaction over time, using either the quantity of reactant used or the quantity of product
AQA Chemistry (8462) from 2016 Topics C4.6 The rate and extent of chemical change Calculate the rate of a chemical reaction over time, using either the quantity of reactant used or the quantity of product
Subject: GCSE Physics
 Subject: GCSE Physics Density States of matter SHC Atoms and radiation Discovery of the nucleus Changes in the nucleus Alpha, beta and gamma Activity and half life Nuclear radiation and medicine Nuclear
Subject: GCSE Physics Density States of matter SHC Atoms and radiation Discovery of the nucleus Changes in the nucleus Alpha, beta and gamma Activity and half life Nuclear radiation and medicine Nuclear
Personalised Learning Checklists AQA Chemistry Paper 1
 AQA Chemistry (8462) from 2016 Topics C4.1 Atomic structure and the periodic table State that everything is made of atoms and recall what they are Describe what elements and compounds are State that elements
AQA Chemistry (8462) from 2016 Topics C4.1 Atomic structure and the periodic table State that everything is made of atoms and recall what they are Describe what elements and compounds are State that elements
Time (s) Velocity (m/s)
 29.01.18 22.01.18 15.01.18 w/c Weekly Combined Science revision countdown plan (revision guide / workbook page numbers) *content across both paper 1 and paper 2 15 14 13 Revise *Key concepts in biology
29.01.18 22.01.18 15.01.18 w/c Weekly Combined Science revision countdown plan (revision guide / workbook page numbers) *content across both paper 1 and paper 2 15 14 13 Revise *Key concepts in biology
Paper Atomic structure and the periodic table
 Paper 1 4.1 Atomic structure and the periodic table 4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic charge and isotopes Use the names and symbols of the first 20 elements in
Paper 1 4.1 Atomic structure and the periodic table 4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic charge and isotopes Use the names and symbols of the first 20 elements in
Physics GCSE (9-1) Energy
 Topic Student Checklist R A G Define a system as an object or group of objects and State examples of changes in the way energy is stored in a system Describe how all the energy changes involved in an energy
Topic Student Checklist R A G Define a system as an object or group of objects and State examples of changes in the way energy is stored in a system Describe how all the energy changes involved in an energy
Personalised Learning Checklists AQA Biology Paper 2
 AQA Biology (8461) from 2016 Topic B4.5 Homeostasis and response Topic Student Checklist R A G Describe what homeostasis is and why it is important stating specific examples from the human body 4.5.1 Homeostasis
AQA Biology (8461) from 2016 Topic B4.5 Homeostasis and response Topic Student Checklist R A G Describe what homeostasis is and why it is important stating specific examples from the human body 4.5.1 Homeostasis
AQA GCSE Chemistry (Combined Science Trilogy) 15 Week Revision Timetable
 Exam advice o READ THE WHOLE QUESTION CAREFULLY before starting to write an answer. o Make sure you have all the necessary equipment o It s ok to draw diagrams even if there are lines for writing. Don
Exam advice o READ THE WHOLE QUESTION CAREFULLY before starting to write an answer. o Make sure you have all the necessary equipment o It s ok to draw diagrams even if there are lines for writing. Don
Lymm High School- KS3 Life after levels - Science Y9
 Biology BRONZE SILVER GOLD PLATINUM D and below= GCSE 1,2,3 C= GCSE 4 C/B= GCSE 5,6 A/A*= GCSE 7,8,9 Explain that cells are very Demonstrate an small and a microscope is understanding of the scale needed
Biology BRONZE SILVER GOLD PLATINUM D and below= GCSE 1,2,3 C= GCSE 4 C/B= GCSE 5,6 A/A*= GCSE 7,8,9 Explain that cells are very Demonstrate an small and a microscope is understanding of the scale needed
Personalised Learning Checklists AQA Physics Paper 2
 6.5.1 Forces and their interactions 6.5.2 Work done and energy transfer AQA TRILOGY Physics (8464) from 2016 Topics T6.5. Forces Topic Student Checklist R A G Identify and describe scalar quantities and
6.5.1 Forces and their interactions 6.5.2 Work done and energy transfer AQA TRILOGY Physics (8464) from 2016 Topics T6.5. Forces Topic Student Checklist R A G Identify and describe scalar quantities and
AQA Physics Checklist
 Topic 1. Energy Video: Energy changes in a system To understand the ways in which energy can be stored in a system and can be transferred from one energy store to another within a system To understand
Topic 1. Energy Video: Energy changes in a system To understand the ways in which energy can be stored in a system and can be transferred from one energy store to another within a system To understand
OCR Chemistry Checklist
 Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Personalised Learning Checklists AQA Trilogy Chemistry Paper 1
 AQA TRILOGY Chemistry (8464) from 2016 Topics T5.1 Atomic structure and the periodic table State that everything is made of atoms and recall what they are Describe what elements and compounds are State
AQA TRILOGY Chemistry (8464) from 2016 Topics T5.1 Atomic structure and the periodic table State that everything is made of atoms and recall what they are Describe what elements and compounds are State
Personalised Learning Checklist
 Personalised Learning Checklist AQA TRILOGY Biology (8464) from 2016 Topic T4.1 Cell biology (Bio Paper 1) 4.1.1 Cell structure 4.1.2 Cell division Use the terms 'eukaryotic' and 'prokaryotic' to describe
Personalised Learning Checklist AQA TRILOGY Biology (8464) from 2016 Topic T4.1 Cell biology (Bio Paper 1) 4.1.1 Cell structure 4.1.2 Cell division Use the terms 'eukaryotic' and 'prokaryotic' to describe
Use estimations and explain when they should be used to judge the relative size or area of sub-cellular structures
 Trilogy Biology Paper 1 Topic Student Checklist R A G Use the terms 'eukaryotic' and 'prokaryotic' to describe types of cells Describe the features of bacterial (prokaryotic) cells Demonstrate an understanding
Trilogy Biology Paper 1 Topic Student Checklist R A G Use the terms 'eukaryotic' and 'prokaryotic' to describe types of cells Describe the features of bacterial (prokaryotic) cells Demonstrate an understanding
OCR Chemistry Checklist
 Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
HASTINGS HIGH SCHOOL
 Subject ASTINGS IG SCOOL YEAR 11 EXAMINATION GUIDE 2017-19 COMBINED SCIENCE: TRILOGY- BIOLOGY Course code AQA GCSE BIOLOGY 8464 Website address Provisional examination dates http://www.aqa.org.uk/subjects/science/gcse/combined-science-trilogy-
Subject ASTINGS IG SCOOL YEAR 11 EXAMINATION GUIDE 2017-19 COMBINED SCIENCE: TRILOGY- BIOLOGY Course code AQA GCSE BIOLOGY 8464 Website address Provisional examination dates http://www.aqa.org.uk/subjects/science/gcse/combined-science-trilogy-
Cecil Jones Academy Science Fundamentals Map
 Fundamentals: Year 9 Biology Knowledge Unit 1 Skills B1 Cell Structure and Transport Unit 2 Use the terms 'eukaryotic' and 'prokaryotic' to describe types of cells Describe the features of bacterial (prokaryotic)
Fundamentals: Year 9 Biology Knowledge Unit 1 Skills B1 Cell Structure and Transport Unit 2 Use the terms 'eukaryotic' and 'prokaryotic' to describe types of cells Describe the features of bacterial (prokaryotic)
OCR Biology Checklist
 Topic 1. Cell level systems Video: Eukaryotic and prokaryotic cells Compare the structure of animal and plant cells. Label typical and atypical prokaryotic cells. Compare prokaryotic and eukaryotic cells.
Topic 1. Cell level systems Video: Eukaryotic and prokaryotic cells Compare the structure of animal and plant cells. Label typical and atypical prokaryotic cells. Compare prokaryotic and eukaryotic cells.
OCR Biology Checklist
 Topic 1. Cell level systems Video: Eukaryotic and prokaryotic cells Compare the structure of animal and plant cells. Label typical and atypical prokaryotic cells. Compare prokaryotic and eukaryotic cells.
Topic 1. Cell level systems Video: Eukaryotic and prokaryotic cells Compare the structure of animal and plant cells. Label typical and atypical prokaryotic cells. Compare prokaryotic and eukaryotic cells.
4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic charge and isotopes. Unit 1 Unit 2 Unit 3. C2.1.1a Structure and bonding
 Summary of changes This resource outlines the main changes that have been made to the assessment and subject content from our previous GCSE Chemistry (4402) to the new specification (8462). Our new specifications
Summary of changes This resource outlines the main changes that have been made to the assessment and subject content from our previous GCSE Chemistry (4402) to the new specification (8462). Our new specifications
Level 789 Pathway: Combined Science Double Award
 Level 789 Pathway: Combined Science Double Award Yr Combined Science Targets: Pathway 8 11 Biology a) Explain in detail the role of the hormone ADH on the nephron b) Explain in detail the process by which
Level 789 Pathway: Combined Science Double Award Yr Combined Science Targets: Pathway 8 11 Biology a) Explain in detail the role of the hormone ADH on the nephron b) Explain in detail the process by which
Describe how the inter-conversion of solids, liquids and gases are achieved and recall names used for these inter-conversions
 Understand the arrangements, movements and energy of the particle in each of the 3 states of matter : solid, liquid and gas Describe how the inter-conversion of solids, liquids and gases are achieved and
Understand the arrangements, movements and energy of the particle in each of the 3 states of matter : solid, liquid and gas Describe how the inter-conversion of solids, liquids and gases are achieved and
KS4 CURRICULUM. Science - BTEC
 KS4 CURRICULUM Science - BTEC Students have four lessons per fortnight completing a BTEC in the Principles of Applied Science. This is the equivalent to one GCSE. Unit One Principles of Science; externally
KS4 CURRICULUM Science - BTEC Students have four lessons per fortnight completing a BTEC in the Principles of Applied Science. This is the equivalent to one GCSE. Unit One Principles of Science; externally
GCSE PHYSICS REVISION LIST
 GCSE PHYSICS REVISION LIST OCR Gateway Physics (J249) from 2016 Topic P1: Matter P1.1 Describe how and why the atomic model has changed over time Describe the structure of the atom and discuss the charges
GCSE PHYSICS REVISION LIST OCR Gateway Physics (J249) from 2016 Topic P1: Matter P1.1 Describe how and why the atomic model has changed over time Describe the structure of the atom and discuss the charges
PLC Booklet for. Paper 1
 PLC Booklet for Combined Science Paper 1 This personalised learning checklist (PLC) booklet has been put together to support your preparation for Biology Paper 1, Chemistry Paper 1 and Physics Paper1.
PLC Booklet for Combined Science Paper 1 This personalised learning checklist (PLC) booklet has been put together to support your preparation for Biology Paper 1, Chemistry Paper 1 and Physics Paper1.
Science. synthesis 2 Analysis and synthesis Using physics to make things work 3 Magnetic fields to keep things moving Energy calculations 4 Energy
 Year 11 (Triple science) 2016-17 Half-term Topic Week 1 Week 2 Week 3 Week 4 Week 5 Week 6 Week 7 1 Exchange of materials Keeping internal conditions constant Analysis and synthesis 2 Analysis and synthesis
Year 11 (Triple science) 2016-17 Half-term Topic Week 1 Week 2 Week 3 Week 4 Week 5 Week 6 Week 7 1 Exchange of materials Keeping internal conditions constant Analysis and synthesis 2 Analysis and synthesis
Part 4- Chemistry Paper 1 Bonding Knowledge Questions
 Part 4- Chemistry Paper 1 Bonding Knowledge Questions How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative atomic mass, electronic charge
Part 4- Chemistry Paper 1 Bonding Knowledge Questions How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative atomic mass, electronic charge
Cell Structure. Cell Structure. Investigating Cells. Investigating Cells. Cell Division. Cell Division. Transport In and Out of Cells
 Cell Structure What are the two main types of cell? Cell Structure The two main types of cell are prokaryotic and eukaryotic. 1 1 Investigating Cells How is magnification calculated? 2 Investigating Cells
Cell Structure What are the two main types of cell? Cell Structure The two main types of cell are prokaryotic and eukaryotic. 1 1 Investigating Cells How is magnification calculated? 2 Investigating Cells
Curriculum overview Key Stage 2 Living things and their habitats: Animals: Properties and changes of materials: Earth and Space: Forces: Evolution:
 Curriculum overview: Science (Foundation) Key Stage 2 Living things and their habitats: Life cycles of a mammal, an amphibian, an insect and a bird Reproduction in some plants and animals. Classification
Curriculum overview: Science (Foundation) Key Stage 2 Living things and their habitats: Life cycles of a mammal, an amphibian, an insect and a bird Reproduction in some plants and animals. Classification
Lesson Aiming for 4 Aiming for 6 Aiming for 8. I can use the periodic table to find the relative atomic mass of all elements.
 Chemical calculations C4.1 Relative masses and moles I can use the periodic table to identify the relative atomic mass for the first 20 elements. I can calculate the relative formula mass for familiar
Chemical calculations C4.1 Relative masses and moles I can use the periodic table to identify the relative atomic mass for the first 20 elements. I can calculate the relative formula mass for familiar
Lesson Target 4 Target 6 Target 8. atom.
 Student checklist C1 Atomic structure Lesson Target 4 Target 6 Target 8 C1.1 Atoms I can define the word element. I can classify familiar substances as elements or compounds. I can use the periodic table
Student checklist C1 Atomic structure Lesson Target 4 Target 6 Target 8 C1.1 Atoms I can define the word element. I can classify familiar substances as elements or compounds. I can use the periodic table
Switching to AQA from OCR Gateway Science B and Additional Science B
 Switching to AQA from OCR Gateway Science B and Additional Science B If you re thinking of switching to AQA from OCR Science B (Gateway) and Additional Science B (J261 and J262) for teaching from September
Switching to AQA from OCR Gateway Science B and Additional Science B If you re thinking of switching to AQA from OCR Science B (Gateway) and Additional Science B (J261 and J262) for teaching from September
Reproduction Chemical Reactions. 8J Light 8G Metals & Their Uses 8C Breathing & Respiration 8D Unicellular Organisms
 Science: Key Stage 3 Based on the Exploring Science Scheme of Learning Term 1 & 2 Term 3 & 4 Term 5 & 6 Year 7 Cells, Tissues & Organs Particles Forces & Motion Reproduction Chemical Reactions Chemical
Science: Key Stage 3 Based on the Exploring Science Scheme of Learning Term 1 & 2 Term 3 & 4 Term 5 & 6 Year 7 Cells, Tissues & Organs Particles Forces & Motion Reproduction Chemical Reactions Chemical
AQA Chemistry Checklist
 Topic 1. Atomic structure Video: Atoms, elements, compounds, mixtures Use the names and symbols of the first 20 elements in the periodic table, the elements in Groups 1 and 7, and other elements in this
Topic 1. Atomic structure Video: Atoms, elements, compounds, mixtures Use the names and symbols of the first 20 elements in the periodic table, the elements in Groups 1 and 7, and other elements in this
Curriculum overview Key Stage 2 Living things and their habitats: Animals: Properties and changes of materials: Earth and Space: Forces: Evolution:
 Curriculum overview: Science (Separate award) Key Stage 2 Living things and their habitats: Life cycles of a mammal, an amphibian, an insect and a bird Reproduction in some plants and animals. Classification
Curriculum overview: Science (Separate award) Key Stage 2 Living things and their habitats: Life cycles of a mammal, an amphibian, an insect and a bird Reproduction in some plants and animals. Classification
Part 5- Chemistry Paper 2 Rate and Extent of Chemical Change Combined Science Application
 Part 5- Chemistry Paper 2 Rate and Extent of Chemical Change Combined Science Application How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative
Part 5- Chemistry Paper 2 Rate and Extent of Chemical Change Combined Science Application How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative
Queen s Park High School Key Stage 3 Assessment Science - Earth IA6 target.
 Year 7 Enquiry Skills Queen s Park High School Key Stage 3 Assessment Science - Earth IA6 target. EARTH STRUCTURE AND UNIVERSE Draw simple graphs outlining information about planets Construct models of
Year 7 Enquiry Skills Queen s Park High School Key Stage 3 Assessment Science - Earth IA6 target. EARTH STRUCTURE AND UNIVERSE Draw simple graphs outlining information about planets Construct models of
Review Chemistry Paper 1
 Atomic Structure Topic Define an atom and element. Use scientific conventions to identify chemical symbols Identify elements by chemical symbols Define compound Use chemical formulae to show different
Atomic Structure Topic Define an atom and element. Use scientific conventions to identify chemical symbols Identify elements by chemical symbols Define compound Use chemical formulae to show different
Summary of changes (certificate to new GCSE)
 Summary of changes (certificate to new GCSE) This resource outlines the main changes that have been made to the assessment and subject content from our legacy Level 1/2 Certificate in Biology (8401) to
Summary of changes (certificate to new GCSE) This resource outlines the main changes that have been made to the assessment and subject content from our legacy Level 1/2 Certificate in Biology (8401) to
Part 5- Chemistry Paper 1 Atomic Structure Knowledge Questions
 Part 5- Chemistry Paper 1 Atomic Structure Knowledge Questions How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative atomic mass, electronic
Part 5- Chemistry Paper 1 Atomic Structure Knowledge Questions How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative atomic mass, electronic
Edexcel Chemistry Checklist
 Topic 1. Key concepts in chemistry Video: Atomic Structure Recall the different charges of the particles that make up an atom. Describe why atoms have no overall charge. Use the periodic table to identify
Topic 1. Key concepts in chemistry Video: Atomic Structure Recall the different charges of the particles that make up an atom. Describe why atoms have no overall charge. Use the periodic table to identify
Edexcel Chemistry Checklist
 Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
Combined Science: Trilogy
 Co-teaching GCSE Chemistry and GCSE Combined Science: Trilogy This high level co-teaching guide will help you plan your route through the course. You ll be able to see what common themes and topics span
Co-teaching GCSE Chemistry and GCSE Combined Science: Trilogy This high level co-teaching guide will help you plan your route through the course. You ll be able to see what common themes and topics span
Lesson title Lesson objectives AQA specification reference 1.1 Elements and compounds
 1.1 Elements and compounds 1.2 Atoms, formulae and equations Identify symbols of elements from the periodic table Recognise the properties of elements and compounds. Identify the elements in a compound
1.1 Elements and compounds 1.2 Atoms, formulae and equations Identify symbols of elements from the periodic table Recognise the properties of elements and compounds. Identify the elements in a compound
PLC Booklet for. Separate Science. Paper
 PLC Booklet for Separate Science Paper 1 This personalised learning checklist (PLC) booklet has been put together to support your preparation for Biology Paper 1, Chemistry Paper 1 and Physics Paper 1.
PLC Booklet for Separate Science Paper 1 This personalised learning checklist (PLC) booklet has been put together to support your preparation for Biology Paper 1, Chemistry Paper 1 and Physics Paper 1.
Part 8- Chemistry Paper 2 Using Resources Triple Science
 Part 8- Chemistry Paper 2 Using Resources Triple Science How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative atomic mass, electronic charge
Part 8- Chemistry Paper 2 Using Resources Triple Science How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative atomic mass, electronic charge
Same theme covered in Combined but extra content Extra parts atomic symbols (first 20, Group 1 and Group 7)
 Co-teaching document new ELC Science 5960 and Foundation Level GCSE Combined Science: Trilogy (8464) Chemistry: Component 3 Elements, mixtures and compounds ELC Outcomes Summary of content covered in ELC
Co-teaching document new ELC Science 5960 and Foundation Level GCSE Combined Science: Trilogy (8464) Chemistry: Component 3 Elements, mixtures and compounds ELC Outcomes Summary of content covered in ELC
Personalised Learning Checklists AQA Physics Paper 1
 6.1.1 Energy changes in a system, and the ways energy is stored before and after such changes AQA TRILOGY Physics (8464) from 2016 Topics T6.1. Energy Topic Student Checklist R A G Define a system as an
6.1.1 Energy changes in a system, and the ways energy is stored before and after such changes AQA TRILOGY Physics (8464) from 2016 Topics T6.1. Energy Topic Student Checklist R A G Define a system as an
Combined GCSE OCR Revision Science magnification tur tur magnification ell Struc ell Struc ells ells Combined GCSE OCR Revision Science
 Cell Structures How is the magnification of a light microscope calculated? Cell Structures Total magnification is calculated by multiplying the magnification of the eyepiece lens by the magnification of
Cell Structures How is the magnification of a light microscope calculated? Cell Structures Total magnification is calculated by multiplying the magnification of the eyepiece lens by the magnification of
Departmental Curriculum Planning
 Department: Btec Subject: Biology Key Stage: 4 Year Group: 10 Learning aim A: Investigate the relationships that different organisms have with each other and with their environment Learning aim B: Demonstrate
Department: Btec Subject: Biology Key Stage: 4 Year Group: 10 Learning aim A: Investigate the relationships that different organisms have with each other and with their environment Learning aim B: Demonstrate
YEAR 7. St Edmund Arrowsmith CCFL Science Department Curriculum Map New AQA Course Started November 2016
 YEAR 7 St Edmund Arrowsmith CCFL Science Department Curriculum Map 201-2017 New AQA Course Started November 2016 05/09/2016 12/09/2016 19/09/2016 26/09/2016 03/10/2016 10/10/2016 17/10/2016 24/10/2016
YEAR 7 St Edmund Arrowsmith CCFL Science Department Curriculum Map 201-2017 New AQA Course Started November 2016 05/09/2016 12/09/2016 19/09/2016 26/09/2016 03/10/2016 10/10/2016 17/10/2016 24/10/2016
Curriculum Mapping Subject: Science Year Group Autumn Term 1a Autumn Term 1b Spring Term 2a Spring Term 2b Summer Term 3a Summer Term 3b
 Curriculum Mapping Subject: Science Year Group Autumn Term 1a Autumn Term 1b Spring Term 2a Spring Term 2b Summer Term 3a Summer Term 3b Year 11 Topic(s) Forces and Energy Atomic Structure Electromagnetism
Curriculum Mapping Subject: Science Year Group Autumn Term 1a Autumn Term 1b Spring Term 2a Spring Term 2b Summer Term 3a Summer Term 3b Year 11 Topic(s) Forces and Energy Atomic Structure Electromagnetism
Y8 Science Controlled Assessment Topics & Keywords
 Y8 Science Controlled Assessment Topics & Biology Respiration. Know that respiration in living organisms can be aerobic or anaerobic The word equation for aerobic respiration The process of anaerobic respiration
Y8 Science Controlled Assessment Topics & Biology Respiration. Know that respiration in living organisms can be aerobic or anaerobic The word equation for aerobic respiration The process of anaerobic respiration
Science Year 10 Unit 1 Biology
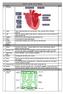 Week 1: 1. The Heart Science Year 10 Unit 1 Biology RAG 2. Artery Takes oxygenated blood away from the heart. Thick, muscular walls to withstand pressure. 3. Vein Takes deoxygenated blood towards the heart.
Week 1: 1. The Heart Science Year 10 Unit 1 Biology RAG 2. Artery Takes oxygenated blood away from the heart. Thick, muscular walls to withstand pressure. 3. Vein Takes deoxygenated blood towards the heart.
Edexcel Physics Checklist
 Topic 1. Key concepts of physics Video: Key concepts of Physics Know the units which will be used throughout the GCSE physics course Remember and use metric prefixes (from nano to giga) Understand and
Topic 1. Key concepts of physics Video: Key concepts of Physics Know the units which will be used throughout the GCSE physics course Remember and use metric prefixes (from nano to giga) Understand and
AQA GCSE (9-1) Combined Chemistry Three Year Scheme of Work
 Year 9 Term 1 1/2 1.1 Elements and compounds Year 9 Term 1 1/2 1.2 Atoms, formulae and equations Chapter 1: Atomic structure and the periodic table Identify symbols of elements from the periodic table
Year 9 Term 1 1/2 1.1 Elements and compounds Year 9 Term 1 1/2 1.2 Atoms, formulae and equations Chapter 1: Atomic structure and the periodic table Identify symbols of elements from the periodic table
Part 6- Chemistry Paper 1 Bonding Application Questions Triple Science
 Part 6- Chemistry Paper 1 Bonding Application Questions Triple Science How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative atomic mass, electronic
Part 6- Chemistry Paper 1 Bonding Application Questions Triple Science How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative atomic mass, electronic
Year 8 Tracking Document. Year 8 Science National Curriculum. Health and Lifestyle
 Health and Lifestyle Describe the components of a healthy diet Explain the role of each food group in the body Describe the test for starch, lipids, sugar and proteins Describe the positive test for each
Health and Lifestyle Describe the components of a healthy diet Explain the role of each food group in the body Describe the test for starch, lipids, sugar and proteins Describe the positive test for each
YEAR 10- Chemistry Term 1 plan
 YEAR 10- Chemistry Term 1 plan 2016-2017 Week Topic Learning outcomes 1 1. The particulate nature of matter State the distinguishing properties of solids, liquids and gases. Describe the structure of solids,
YEAR 10- Chemistry Term 1 plan 2016-2017 Week Topic Learning outcomes 1 1. The particulate nature of matter State the distinguishing properties of solids, liquids and gases. Describe the structure of solids,
GCSE OCR Revision Chemistry. GCSE OCR Revision Chemistry. GCSE OCR Revision Chemistry. Bonding. GCSE OCR Revision Chemistry
 Particle Model and Atomic Structure The following symbols describe two different substances. Deduce all the information you can from these symbols. 13 C 12 6 6 C 1 Particle Model and Atomic Structure The
Particle Model and Atomic Structure The following symbols describe two different substances. Deduce all the information you can from these symbols. 13 C 12 6 6 C 1 Particle Model and Atomic Structure The
Department Curriculum and Assessment Outline
 Department: Science Year Group: 10 Teaching, learning and assessment during the course: Combined Science 1 2 B1 Key concepts in Biology B2 Cells and control What are the structure and function of cells.
Department: Science Year Group: 10 Teaching, learning and assessment during the course: Combined Science 1 2 B1 Key concepts in Biology B2 Cells and control What are the structure and function of cells.
PROGRAMME OF STUDY FOR SCIENCE BECKET KEYS CHURCH OF ENGLAND SECONDARY SCHOOL. Term 1 Term 2 Term 3 Term 4 Term 5 Term 6 Matter. Genes.
 Year 7 Matter Genes Earth Ecosystems Revision of year 7 Particle model Variation Earth Structure Interdependence Separating materials Human Reproduction Universe Plant reproduction (GFE Plan) (PTU-Review)
Year 7 Matter Genes Earth Ecosystems Revision of year 7 Particle model Variation Earth Structure Interdependence Separating materials Human Reproduction Universe Plant reproduction (GFE Plan) (PTU-Review)
AQA GCSE Chemistry Checklist
 AQA GCSE Chemistry Checklist Subject content 4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic charge and isotopes Students will be supplied with a periodic table for the exam
AQA GCSE Chemistry Checklist Subject content 4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic charge and isotopes Students will be supplied with a periodic table for the exam
Combined GCSE OCR Revision Science magnification tur tur magnification ell Struc ell Struc ells ells Combined GCSE OCR Revision Science
 Cell Structures How is the magnification of a light microscope calculated? Cell Structures Total magnification is calculated by multiplying the magnification of the eyepiece lens by the magnification of
Cell Structures How is the magnification of a light microscope calculated? Cell Structures Total magnification is calculated by multiplying the magnification of the eyepiece lens by the magnification of
Continuous Development Cycle Curriculum & Assessment Overview
 Year 7 Long-term Plan - Science 1 2 3 4 5 6 Working Scientifically FORCES Elements, Atoms & Compounds - revisit Body Systems - revisit Reproduction revisit Sound - ENERGY Chemical Reactions revisit Light
Year 7 Long-term Plan - Science 1 2 3 4 5 6 Working Scientifically FORCES Elements, Atoms & Compounds - revisit Body Systems - revisit Reproduction revisit Sound - ENERGY Chemical Reactions revisit Light
GCSE Science: Progression in key ideas
 The Collins GCSE Science (9-1) Five Year Scheme of Work: Identifies ten key scientific concepts Outlines the development of students understanding in these concepts over the five years from age 11 to 16
The Collins GCSE Science (9-1) Five Year Scheme of Work: Identifies ten key scientific concepts Outlines the development of students understanding in these concepts over the five years from age 11 to 16
Chemistry (separate) for November PPE
 1.1 Elements and 1.2 Atoms, formulae and Chapter 1 Atomic Structure and Periodic Table Identify symbols of elements from the periodic table Recognise the properties of elements and. Identify the elements
1.1 Elements and 1.2 Atoms, formulae and Chapter 1 Atomic Structure and Periodic Table Identify symbols of elements from the periodic table Recognise the properties of elements and. Identify the elements
Part 7- Quantitative Chemistry Application Questions Triple Science
 Part 7- Quantitative Chemistry Application Questions Triple Science How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative atomic mass, electronic
Part 7- Quantitative Chemistry Application Questions Triple Science How bonding and structure are related to the properties of substances A simple model of the atom, symbols, relative atomic mass, electronic
Atoms, Elements, Atoms, Elements, Compounds and Mixtures. Compounds and Mixtures. Atoms and the Periodic Table. Atoms and the.
 Atoms, Elements, Compounds and Mixtures Explain how fractional distillation can be used to separate a mixture. 1 Atoms, Elements, Compounds and Mixtures Fractional distillation is used to separate components
Atoms, Elements, Compounds and Mixtures Explain how fractional distillation can be used to separate a mixture. 1 Atoms, Elements, Compounds and Mixtures Fractional distillation is used to separate components
Parkside Science Department Year 11 GCSE Support 2018
 Parkside Science Department Year 11 GCSE Support 2018 Name: These are your BIOLOGY science exam dates for your BIOLOGY topics Biology Paper 1 15 th May 2018 Biology Paper 2 11 th June 2018 These are your
Parkside Science Department Year 11 GCSE Support 2018 Name: These are your BIOLOGY science exam dates for your BIOLOGY topics Biology Paper 1 15 th May 2018 Biology Paper 2 11 th June 2018 These are your
AQA Biology Checklist
 Topic 1. Cell biology Video: Eukaryotic and prokaryotic cells Distinguish between eukaryotic and prokaryotic cells. Compare animal and plant cells. Relate cell structures to their functions. Video: Specialised
Topic 1. Cell biology Video: Eukaryotic and prokaryotic cells Distinguish between eukaryotic and prokaryotic cells. Compare animal and plant cells. Relate cell structures to their functions. Video: Specialised
Additional Science. Important exam information and revision booklet
 Additional Science Important exam information and revision booklet CONTENTS PAGE Page Introduction 3 Section One When are my exams? What will they test 4 Useful places to help you to revise Additional
Additional Science Important exam information and revision booklet CONTENTS PAGE Page Introduction 3 Section One When are my exams? What will they test 4 Useful places to help you to revise Additional
8 th Grade Integrated Science Curriculum
 Date Hobbs Science By being embedded throughout the curriculum, these Processing Skills will be addressed throughout the year. 8.1 Scientific Thinking and Practice 1. Use scientific methods to develop
Date Hobbs Science By being embedded throughout the curriculum, these Processing Skills will be addressed throughout the year. 8.1 Scientific Thinking and Practice 1. Use scientific methods to develop
Switching to AQA from OCR: Science A and Additional Science A (Chemistry)
 Switching to AQA from OCR: Science A and Additional Science A (Chemistry) If you're thinking of switching to AQA from OCR Science A and Additional Science A (J241 and J242) for teaching from September
Switching to AQA from OCR: Science A and Additional Science A (Chemistry) If you're thinking of switching to AQA from OCR Science A and Additional Science A (J241 and J242) for teaching from September
AQA GCSE (9-1) Chemistry Three Year Scheme of Work
 This 3-Year Scheme of Work offers a flexible approach for KS4. It is based on three science lessons per fortnight (assuming a two week timetable of two lessons one week and one lesson in the other). Lessons
This 3-Year Scheme of Work offers a flexible approach for KS4. It is based on three science lessons per fortnight (assuming a two week timetable of two lessons one week and one lesson in the other). Lessons
K-12 Science Content Areas
 K-12 Science Content Areas 100 Nature of Science 2000 Earth Systems 200 Science & Technology 2100 Astronomy 300 Science, Health & Environment 2200 Meteorology 400 Measurement & Calculation in Science 2300
K-12 Science Content Areas 100 Nature of Science 2000 Earth Systems 200 Science & Technology 2100 Astronomy 300 Science, Health & Environment 2200 Meteorology 400 Measurement & Calculation in Science 2300
AQA GCSE CHEMISTRY (9-1) Topic 1: Atomic Structure and the Periodic Table
 AQA GCSE CHEMISTRY (9-1) Topic 1: Atomic Structure and the Periodic Table 4.1.1 Atoms, elements and compounds 4.1.1.1 Atoms, elements and compounds All substances are made of atoms. An atom is the smallest
AQA GCSE CHEMISTRY (9-1) Topic 1: Atomic Structure and the Periodic Table 4.1.1 Atoms, elements and compounds 4.1.1.1 Atoms, elements and compounds All substances are made of atoms. An atom is the smallest
Year 7 Science - Learning Aims
 Year 7 Science - Learning Aims Learning Aim 1 - Beginning 2 - Working towards 3 - Secure 4 - Mastered 1 Living Organisms. List the seven life processes. Define sensitivity, excretion, respiration and nutrition.
Year 7 Science - Learning Aims Learning Aim 1 - Beginning 2 - Working towards 3 - Secure 4 - Mastered 1 Living Organisms. List the seven life processes. Define sensitivity, excretion, respiration and nutrition.
KS3 Science Levelness Posters
 KS Science Levelness Posters Contents Year Year 8 Year 9 A. Cells 8A. Food and digestion 9A. Inheritance and selection B. Reproduction 8B. Respiration 9B. Fit and healthy C. Environment and feeding 8C.
KS Science Levelness Posters Contents Year Year 8 Year 9 A. Cells 8A. Food and digestion 9A. Inheritance and selection B. Reproduction 8B. Respiration 9B. Fit and healthy C. Environment and feeding 8C.
Colorado High School Physical Science Standards Foundations of Physical Science, 3rd Edition
 11/13/2014 Page 1 of 34 PS.1.A Physical Science 81 vectors and velocity 22 find speed of car Newton s laws of motion and gravitation describe the relationships among forces acting on and between objects,
11/13/2014 Page 1 of 34 PS.1.A Physical Science 81 vectors and velocity 22 find speed of car Newton s laws of motion and gravitation describe the relationships among forces acting on and between objects,
Plant Biology. 2. Explain why energy is lost between each trophic level (triple only).
 1. Calculate the % of energy lost between each level. A-B = B-C = C-D = 80%+ Describe quantitatively the proportion of energy transferred between trophic levels and use this to calculate efficiency (triple
1. Calculate the % of energy lost between each level. A-B = B-C = C-D = 80%+ Describe quantitatively the proportion of energy transferred between trophic levels and use this to calculate efficiency (triple
LONGMAN STUDY GUIDES. Science. Di Barton. SUB Q6ttlngen B1842 LONGMAN
 LONGMAN STUDY GUIDES Science Di Barton SUB Q6ttlngen 206 451 652 97B1842 LONGMAN CONTENTS Editors' Preface vii Acknowledgements vii Information about this book viii 1 Revision, examinations and coursework
LONGMAN STUDY GUIDES Science Di Barton SUB Q6ttlngen 206 451 652 97B1842 LONGMAN CONTENTS Editors' Preface vii Acknowledgements vii Information about this book viii 1 Revision, examinations and coursework
0654 CO-ORDINATED SCIENCES
 CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International General Certificate of Secondary Education MARK SCHEME for the May/June 2015 series 0654 CO-ORDINATED SCIENCES 0654/31 Paper 3 (Extended Theory),
CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International General Certificate of Secondary Education MARK SCHEME for the May/June 2015 series 0654 CO-ORDINATED SCIENCES 0654/31 Paper 3 (Extended Theory),
Year 11 Science Learning Cycle 3 Overview
 Year 11 Science Learning Cycle 3 Overview Learning Cycle Overview: Biology Hypothesis 1 Hypothesis 2 Hypothesis 3 Hypothesis 4 Hypothesis 5 Hypothesis 6 Hypothesis 7 Hypothesis 8 Hypothesis 9 How does
Year 11 Science Learning Cycle 3 Overview Learning Cycle Overview: Biology Hypothesis 1 Hypothesis 2 Hypothesis 3 Hypothesis 4 Hypothesis 5 Hypothesis 6 Hypothesis 7 Hypothesis 8 Hypothesis 9 How does
Combined Science Physics Academic Overview
 Combined Science Physics Academic Overview 2018-19 Science Term 1.1 Term 1.2 Term 2.1 Term 2.2 Term 3.1 Term 3.1 Year 9 Motion Forces 1 Newton s Laws Forces 2 Momentum and Safety Energy Waves End of year
Combined Science Physics Academic Overview 2018-19 Science Term 1.1 Term 1.2 Term 2.1 Term 2.2 Term 3.1 Term 3.1 Year 9 Motion Forces 1 Newton s Laws Forces 2 Momentum and Safety Energy Waves End of year
Unit C1: Chemistry in our world Page 1 of 5
 Unit C1: Chemistry in our world Page 1 of 5 Lesson Specification learning outcomes Edexcel 360 Science Specification match Edexcel 360 Science GCSE Science Students Book page reference Additional information
Unit C1: Chemistry in our world Page 1 of 5 Lesson Specification learning outcomes Edexcel 360 Science Specification match Edexcel 360 Science GCSE Science Students Book page reference Additional information
