Supplementary Information Surface energy and wettability of van der Waals structures
|
|
|
- Daisy Blankenship
- 5 years ago
- Views:
Transcription
1 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Supplementary Material (ESI) for Nanoscale Supplementary Information Surface energy and wettability of van der Waals structures Meenakshi Annamalai a, Kalon Gopinadhan *a,b, Sang A Han c,d, Surajit Saha a,e, Hye Jeong Park c, Brijesh Kumar #a, Abhijeet Patra a,f, Sang-Woo Kim *c,d, T Venkatesan *a,b,e,f,g a NUSNNI-NanoCore, National University of Singapore (NUS), Singapore b Department of Electrical and Computer Engineering, National University of Singapore (NUS), Singapore c Nanoelectronic Science and Engineering Laboratory, Department of Advanced Materials Science & Engineering, School of Engineering, Sungkyunkwan University, Suwon , Republic of Korea d SKKU Advanced Institute of Nanotechnology (SAINT), Center for Human Interface Nanotechnology (HINT), Sungkyunkwan University, Suwon , Republic of Korea e Department of Physics, Faculty of Science, National University of Singapore (NUS), Singapore f NUS Graduate School for Integrative Sciences and Engineering (NGS), National University of Singapore (NUS), Singapore g Department of Materials Science and Engineering, National University of Singapore (NUS), Singapore # Currently at Centre for Nanoscience and Technology, School of Engineering and Technology, Amity University, Gurgaon , India Corresponding author venky@nus.edu.sg; kimsw1@skku.edu; gopinadhan@iitkalumni.org
2 Macroscale contact angle and surface energy measurement Wettability of surfaces by liquids is of great interest in a number of fields ranging from engineering to medicine. Wetting phenomena on a macroscopic scale can be illustrated using Young s equation. 1 γ lv cosθ = γ sv γ sl (1) where θ is the contact angle, γ sl γ is solid/liquid interfacial free energy, lv is liquid/vapour γ interfacial tension (liquid surface tension) and sv solid surface free energy. The contact angle is estimated using the sessile drop technique by measuring the angle between the tangent lines along solid-liquid interface and liquid-vapour interface of the liquid contour as shown in Fig. S1. A contact angle of 0 and 180 correspond to complete wetting and non-wetting respectively. Surfaces exhibiting contact angles below 90 are called hydrophilic and those above 90 are called hydrophobic. In the past few decades many semiempirical analytical models have been developed to compute surface free energy from measured contact angles such as Fowkes, 2 Owens Wendt Rabel and Kaelbel (OWRK), 3 Van Oss-Chaudhury Good/Lewis acid base theory, 4 Zisman 5 and Neumann, 6 to name a few. Each approach is targeted for measuring surface energies of either low surface energy materials or high surface materials or both. Also, the essence and physical interpretation of these approaches are different and therefore subsequently provide information on total surface energy or individual components (polar, dispersive, hydrogen bonding etc.) of surface energy or both. In our work, the OWRK method has been adopted which is suitable for universal systems. Figure S1. Schematic diagram of a liquid drop on a solid surface showing the interfacial tensions at three phase boundary. Description of OWRK method Owens, Wendt, Rabel and Kaelble developed a two component model to separate the interfacial tension according to the underlying interactions between the molecules. These interactions are defined as polar and dispersive interactions. The total surface energy of the solid is the sum of the two parts. The polar interactions arise due to the permanent dipole permanent dipole interactions or Keesom forces. 7 They are stronger and only exist in polar molecules. Dispersive component also known as London forces are weak and arise due to
3 random fluctuations in the electron density in an electron cloud and hence lead to temporary/induced dipole interactions. 8 In OWRK method, at least two liquids with known dispersive and polar parts of surface tensions are needed to compute the solid surface free energy as there are two unknowns (solid/liquid interfacial free energy and solid surface free energy). The combining rule proposed by OWRK model is indicated below. γ sl = γ sv + γ lv 2( γ D sv γd lv + γ P sv γp lv) Where γ sv D and γ lvd are dispersive components and γ sv P and γ lv P are polar components of solid and liquid surface energies respectively. Substituting for γ sl from equation (1), (2) γ D sv γd lv + γ D sv γd lv + γ P sv γp lv = 1 2 [γ sv + γ lv (γ sv γ lv cosθ)] γ sv P γp lv = 1 2 [γ lv (1 + cosθ)] (3) (4) By dividing in equation (4), we get, (5) γ D sv + γ P sv γ P lv = 1 2 [γ lv (1 + cosθ] The above equation can be represented in the linear form, c + mx = y (6) where in, c = γ D sv m = γ P sv x = γp lv A graphical representation of the OWRK method is shown in Fig. S2 for MoS 2 on SiO 2 -Si. The polar and dispersive components of total surface tension of the liquids used in this study are known (Table S1) and are substituted to compute the polar and dispersive components of the surface free energy of the solid. The slope of the graph gives the polar component and the vertical intercept gives the dispersive component of the solid surface free energy.
4 Figure S2. A representative surface energy (polar and dispersive) calculation graph based on OWRK model for MoS 2 /SiO 2 /Si sample. Table S1. Surface tensions (SFT) of the test liquids Solvent SFT (Total) SFT (Dispersive) SFT (Polar) Values mn/m mn/m mn/m Adapted From Water Chen et al. Ethylene Glycol Gebhardt et al. Diiodomethane Busscher et al. Formamide Rabel et al. For determining the thickness of h-bn, h-bn has been transferred to a copper grid for TEM characterization. The high resolution TEM image on the edge of h-bn is shown Fig. S3. The image shows the crystalline and layered nature of h-bn where the number of BN layers is determined to be ~10. Figure S3. A transmission electron microscope (TEM) cross sectional image of h-bn.
5 Measured contact angles with four test liquids on the fabricated 2D structures are shown in Table S2. The standard deviation has been calculated based on the variations in the measured left and right contact angles and also taking in to account the instrumental uncertainty of 0.1. Figure S4 shows the contact angle images obtained for ethylene glycol, diiodomethane and formamide on the 2D structures. Table S2. Contact angles obtained from four different solvents on the fabricated surfaces S/N Sample Details Water CA Ethylene Water CA Diiodomethane Formamide Repeat Glycol CA (degrees) CA (degrees) CA (degrees) (degrees) (degrees) 1 Graphene/h- BN/SiO 2 /Si 79.1± ± ± ± ±0.3 2 MoS 2 /h-bn/sio 2 /Si 61.6± ± ± ± ±0.6 3 WS 2 /h-bn/sio 2 /Si 66.4± ± ± ± ±1.5 4 MoS 2 /WS 2 /h- BN/SiO 2 /Si 63.3± ± ± ± ±0.4 5 Graphene/SiO 2 /Si 86.3± ± ± ± ±0.3 6 MoS 2 /SiO 2 /Si 74.3± ± ± ± ±1.8 7 WS 2 /SiO 2 /Si 71.2± ± ±1 36.7± ±0.2 8 MoS 2 /WS 2 /SiO 2 /Si 89.9± ± ±1 45.4± ±0.5 Figure S4. Contact angles obtained on various fabricated structures with ethylene glycol, diiodomethane and formamide.
6 From earlier reports on wetting of 2D materials we also believe that airborne contaminants have a pronounced impact on wettability and can significantly modify the water contact angle. The contact angle measurements were carried out in a controlled class 10,000 clean room with 45 % RH condition. Annealing was also performed in the same clean room environment and the samples were measured immediately after the samples were cooled down to room temperature. Surfaces are believed to adsorb hydrocarbons from ambient air and such adsorption reduces the surface energy and increases the hydrophobicity of the material. In order to study such an influence of time over the wettability, we performed water contact angle (WCA) measurements over time (t = 0 to 24 hours) on MoS 2 /SiO 2 /Si. For annealing, we followed the procedure adopted in electrical measurements. For example, annealing at 400 K recovers the intrinsic surface of graphene/bn system as evident from the zero charge neutrality point. 9 The samples used in this study were thermally annealed at 150 C to remove any aromatic hydrocarbons and moisture that might be present on the surface before the contact angle measurements and with the samples exposed to the controlled clean room environment and even with ageing the WCA seems to be almost constant (see Fig. S5). Figure S5. Water contact angle of MoS 2 /SiO 2 /Si (thermally annealed at 150 C) over time. Any presence of MoO 3 should be reflected as a strong Raman peak at ~820 cm -1. A Raman spectrum of MoS 2 after 150 C annealing showing absence of any peak near 820 cm -1 is shown in Fig. S6. So we can rule out the oxidation process at 150 C.
7 References Figure S6. Raman spectrum of MoS 2 after 150 C thermal annealing. 1. T. Young, Phil. Trans. R. Soc. Lond., 1805, 95, F. M. Fowkes, J. Colloid Interface Sci., 1968, 28, D. K. Owens and R. C. Wendt, J. Appl. Polym. Sci., 1969, 13, C. J. Van Oss, R. J. Good and M. K. Chaudhury, J. Colloid Interface Sci., 1986, 111, H. Fox and W. Zisman, J. Colloid Sci., 1952, 7, M. Zenkiewicz, Polimery, 2006, 51, W. Keesom, Proc. R. Acad. Amsterdam, F. London, Trans. Faraday Soc., 1937, 33, 8b D. A. Abanin, S. V. Morozov, L. A. Ponomarenko, R. V. Gorbachev, A. S. Mayorov, M. I. Katsnelson, K. Watanabe, T. Taniguchi, K. S. Novoselov, L. S. Levitov and A. K. Geim, Science, 2011, 332,
PAPER. On the nature of wettability of van der Waals heterostructures INTRODUCTION
 PAPER On the nature of wettability of van der Waals heterostructures Meenakshi Annamalai a, Kalon Gopinadhan *a,b, Sang A Han f,g, Surajit Saha a,e, Hye Jeong Park f, Eun Bi Cho f, Brijesh Kumar #a, Sang-Woo
PAPER On the nature of wettability of van der Waals heterostructures Meenakshi Annamalai a, Kalon Gopinadhan *a,b, Sang A Han f,g, Surajit Saha a,e, Hye Jeong Park f, Eun Bi Cho f, Brijesh Kumar #a, Sang-Woo
Changes of polymer material wettability by surface discharge
 Changes of polymer material wettability by surface discharge Surface discharge and material treatment Surface treatment of materials in low temperature plasma belongs to the modern and very perspective
Changes of polymer material wettability by surface discharge Surface discharge and material treatment Surface treatment of materials in low temperature plasma belongs to the modern and very perspective
Research Article Validation of Methods for the Optical Characterisation of the Wettability of Polymeric Films for Food Packaging
 Industrial Engineering, Article ID 6235, 6 pages http://dx.doi.org/.1155/14/6235 Research Article Validation of Methods for the Optical Characterisation of the Wettability of Polymeric Films for Food Packaging
Industrial Engineering, Article ID 6235, 6 pages http://dx.doi.org/.1155/14/6235 Research Article Validation of Methods for the Optical Characterisation of the Wettability of Polymeric Films for Food Packaging
Surface Characteristics of a Polyimide Film Treated with a Dielectric Barrier Discharge Plasma
 Journal of the Korean Physical Society, Vol. 54, No. 3, March 2009, pp. 11561160 Surface Characteristics of a Polyimide Film Treated with a Dielectric Barrier Discharge Plasma S. M. Kang, W. J. Park and
Journal of the Korean Physical Society, Vol. 54, No. 3, March 2009, pp. 11561160 Surface Characteristics of a Polyimide Film Treated with a Dielectric Barrier Discharge Plasma S. M. Kang, W. J. Park and
Surface Free Energy Effects in Sputter-Deposited WN x Films
 Materials Transactions, Vol. 48, No. 9 (2007) pp. 2449 to 2453 #2007 The Japan Institute of Metals Surface Free Energy Effects in Sputter-Deposited WN x Films Chun-Wei Fan* and Shih-Chin Lee Department
Materials Transactions, Vol. 48, No. 9 (2007) pp. 2449 to 2453 #2007 The Japan Institute of Metals Surface Free Energy Effects in Sputter-Deposited WN x Films Chun-Wei Fan* and Shih-Chin Lee Department
Supporting Information
 Electronic Supplementary Material (ESI) for Biomaterials Science. This journal is The Royal Society of Chemistry 2014 Supporting Information Biomimetic Honeycomb-patterned Surface as the Tunable Cell Adhesion
Electronic Supplementary Material (ESI) for Biomaterials Science. This journal is The Royal Society of Chemistry 2014 Supporting Information Biomimetic Honeycomb-patterned Surface as the Tunable Cell Adhesion
Microfluidics 2 Surface tension, contact angle, capillary flow
 MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
Wetting Properties of XLPE based Inorganic Hybrid Nanocomposites: Contact Angle Studies
 Chapter 8 Wetting Properties of XLPE based Inorganic Hybrid Nanocomposites: Contact Angle Studies Abstract: Wetting properties and surface energy characteristics of nanocomposites are studied using contact
Chapter 8 Wetting Properties of XLPE based Inorganic Hybrid Nanocomposites: Contact Angle Studies Abstract: Wetting properties and surface energy characteristics of nanocomposites are studied using contact
Wettability of polymeric solids by aqueous solution of surfactants
 A N N A L E S U N I V E R S I T A T I S M A R I A E C U R I E - S K Ł O D O W S K A L U B L I N P O L O N I A VOL. LX, 8 SECTIO AA 2005 Wettability of polymeric solids by aqueous solution of surfactants
A N N A L E S U N I V E R S I T A T I S M A R I A E C U R I E - S K Ł O D O W S K A L U B L I N P O L O N I A VOL. LX, 8 SECTIO AA 2005 Wettability of polymeric solids by aqueous solution of surfactants
Surface and Interfacial Tensions. Lecture 1
 Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
ATOMIC FORCE MICROSCOPY METHOD DEVELOPMENT FOR SURFACE ENERGY ANALYSIS
 University of Kentucky UKnowledge University of Kentucky Doctoral Dissertations Graduate School 2011 ATOMIC FORCE MICROSCOPY METHOD DEVELOPMENT FOR SURFACE ENERGY ANALYSIS Clare Aubrey Medendorp University
University of Kentucky UKnowledge University of Kentucky Doctoral Dissertations Graduate School 2011 ATOMIC FORCE MICROSCOPY METHOD DEVELOPMENT FOR SURFACE ENERGY ANALYSIS Clare Aubrey Medendorp University
DLVO interaction between the spheres
 DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
Activity coefficient at infinite dilution of aqueous solutions of nonionic hydrocarbon and fluorocarbon surfactants as well as their ternary mixtures
 10.1515/umcschem-2015-0008 ANNALES UNIVERSITATIS ARIAE CURIE-SKŁODOWSKA LUBLIN POLONIA VOL. LXX, 1 SECTIO AA 2015 Activity coefficient at infinite dilution of aqueous solutions of nonionic hydrocarbon
10.1515/umcschem-2015-0008 ANNALES UNIVERSITATIS ARIAE CURIE-SKŁODOWSKA LUBLIN POLONIA VOL. LXX, 1 SECTIO AA 2015 Activity coefficient at infinite dilution of aqueous solutions of nonionic hydrocarbon
Influence of Surface Energy of Polymer Films on Spreading and Adhesion of UV-Flexo Inks
 acta graphica 219 short communication received: 08-04-2013. accepted: 17-07-2013. Influence of Surface Energy of Polymer Films on Spreading and Adhesion of UV-Flexo Inks Authors Vyacheslav Repeta Ukrainian
acta graphica 219 short communication received: 08-04-2013. accepted: 17-07-2013. Influence of Surface Energy of Polymer Films on Spreading and Adhesion of UV-Flexo Inks Authors Vyacheslav Repeta Ukrainian
Chapter 11. Intermolecular forces. Chapter 11 1
 Chapter 11 Intermolecular Attractions and the Properties of Liquids and Solids 1 2 Intermolecular forces Forces of attraction between molecules Directly dependent on the distance between the molecules
Chapter 11 Intermolecular Attractions and the Properties of Liquids and Solids 1 2 Intermolecular forces Forces of attraction between molecules Directly dependent on the distance between the molecules
Chapter Intermolecular attractions
 Chapter 11 11.2 Intermolecular attractions Intermolecular Attractions and the Properties of Liquids and Solids Intermolecular forces control the physical properties of the substance. Intramolecular forces
Chapter 11 11.2 Intermolecular attractions Intermolecular Attractions and the Properties of Liquids and Solids Intermolecular forces control the physical properties of the substance. Intramolecular forces
Lecture 7 Contact angle phenomena and wetting
 Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
The role of charge traps in inducing hysteresis: capacitance voltage measurements on top gated bilayer graphene
 The role of charge traps in inducing hysteresis: capacitance voltage measurements on top gated bilayer graphene Gopinadhan Kalon, Young Jun Shin, Viet Giang Truong, Alan Kalitsov, and Hyunsoo Yang a) Department
The role of charge traps in inducing hysteresis: capacitance voltage measurements on top gated bilayer graphene Gopinadhan Kalon, Young Jun Shin, Viet Giang Truong, Alan Kalitsov, and Hyunsoo Yang a) Department
Dispersion of rgo in Polymeric Matrices by Thermodynamically Favorable Self-Assembly of GO at Oil-Water Interfaces
 Supporting information for Dispersion of rgo in Polymeric Matrices by Thermodynamically Favorable Self-Assembly of GO at Oil-Water Interfaces Saeed Zajforoushan Moghaddam, Sina Sabury and Farhad Sharif*
Supporting information for Dispersion of rgo in Polymeric Matrices by Thermodynamically Favorable Self-Assembly of GO at Oil-Water Interfaces Saeed Zajforoushan Moghaddam, Sina Sabury and Farhad Sharif*
Praktikum zur. Materialanalytik
 Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Continuous Growth of Hexagonal Graphene and Boron Nitride In-Plane Heterostructures by Atmospheric Pressure Chemical Vapor Deposition
 SUPPORTING INFORMATION FOR Continuous Growth of Hexagonal Graphene and Boron Nitride In-Plane Heterostructures by Atmospheric Pressure Chemical Vapor Deposition Gang Hee Han 1, 3, Julio A. Rodríguez-Manzo
SUPPORTING INFORMATION FOR Continuous Growth of Hexagonal Graphene and Boron Nitride In-Plane Heterostructures by Atmospheric Pressure Chemical Vapor Deposition Gang Hee Han 1, 3, Julio A. Rodríguez-Manzo
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Porous MoS 2 @C hetero shell with Si yolk structure
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Porous MoS 2 @C hetero shell with Si yolk structure
CHAPTER 6 Intermolecular Forces Attractions between Particles
 CHAPTER 6 Intermolecular Forces Attractions between Particles Scientists are interested in how matter behaves under unusual circumstances. For example, before the space station could be built, fundamental
CHAPTER 6 Intermolecular Forces Attractions between Particles Scientists are interested in how matter behaves under unusual circumstances. For example, before the space station could be built, fundamental
Effect of Non-Ionic Surfactants on Dispersion and. Polar Interactions in the Adsorption of Cellulases. onto Lignin
 Supporting Information Effect of Non-Ionic Surfactants on Dispersion and Polar Interactions in the Adsorption of Cellulases onto Lignin Feng Jiang, Chen Qian, Alan R. Esker and Maren Roman, * Macromolecules
Supporting Information Effect of Non-Ionic Surfactants on Dispersion and Polar Interactions in the Adsorption of Cellulases onto Lignin Feng Jiang, Chen Qian, Alan R. Esker and Maren Roman, * Macromolecules
Surface Plasmon-Induced Hot Carrier Effect on Catalytic Activity of CO oxidation on Cu 2 O/Hexoctahedral Au Inverse Catalyst
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Surface Plasmon-Induced Hot Carrier Effect on Catalytic Activity
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Surface Plasmon-Induced Hot Carrier Effect on Catalytic Activity
Biomaterial Scaffolds
 Biomaterial Scaffolds Biomaterial Properties Surface properties Bulk properties Biological properties Types of Biomaterials Biological materials Synthetic materials Surface Properties The body reads the
Biomaterial Scaffolds Biomaterial Properties Surface properties Bulk properties Biological properties Types of Biomaterials Biological materials Synthetic materials Surface Properties The body reads the
Seminars in Nanosystems - I
 Seminars in Nanosystems - I Winter Semester 2011/2012 Dr. Emanuela Margapoti Emanuela.Margapoti@wsi.tum.de Dr. Gregor Koblmüller Gregor.Koblmueller@wsi.tum.de Seminar Room at ZNN 1 floor Topics of the
Seminars in Nanosystems - I Winter Semester 2011/2012 Dr. Emanuela Margapoti Emanuela.Margapoti@wsi.tum.de Dr. Gregor Koblmüller Gregor.Koblmueller@wsi.tum.de Seminar Room at ZNN 1 floor Topics of the
CHAPTER 10. Characteristics of the Surfaces of Biomaterials
 CHAPTER 10 Characteristics of the Surfaces of Biomaterials 10.1 Surface Characteristics Related to Chemical Bonding 10.2 Surface Chemistry Related to Bonding of Biological Molecules 10.3 Porosity 10.4
CHAPTER 10 Characteristics of the Surfaces of Biomaterials 10.1 Surface Characteristics Related to Chemical Bonding 10.2 Surface Chemistry Related to Bonding of Biological Molecules 10.3 Porosity 10.4
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai
 Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Surfaces and Interfaces Defining of interfacial region Types of interfaces: surface vs interface Surface
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Surfaces and Interfaces Defining of interfacial region Types of interfaces: surface vs interface Surface
Contents. Preface XI Symbols and Abbreviations XIII. 1 Introduction 1
 V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
The Origins of Surface and Interfacial Tension
 The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
Hydrophilization of Fluoropolymers and Silicones
 2017 Adhesive and Sealant Council Spring Meeting Hydrophilization of Fluoropolymers and Silicones Aknowledgements: Wei Chen Mount Holyoke College NSF, NIH, Dreyfus, ACS-RF, MHC Bryony Coupe, Mamle Quarmyne,
2017 Adhesive and Sealant Council Spring Meeting Hydrophilization of Fluoropolymers and Silicones Aknowledgements: Wei Chen Mount Holyoke College NSF, NIH, Dreyfus, ACS-RF, MHC Bryony Coupe, Mamle Quarmyne,
Supporting Information. Golf ball-shaped PLGA microparticles with internal pores fabricated by simple O/W emulsion
 Supplementary Material (ESI) for Chemical Communications Supporting Information Golf ball-shaped PLGA microparticles with internal pores fabricated by simple O/W emulsion Mi Ri Kim, a Seungwoo Lee, b Jung-Ki
Supplementary Material (ESI) for Chemical Communications Supporting Information Golf ball-shaped PLGA microparticles with internal pores fabricated by simple O/W emulsion Mi Ri Kim, a Seungwoo Lee, b Jung-Ki
CHAPTER 10. Characteristics of the Surfaces of Biomaterials
 CHAPTER 10 Characteristics of the Surfaces of Biomaterials 10.1 Surface Characteristics Related to Chemical Bonding 10.2 Surface Chemistry Related to Bonding of Biological Molecules 10.3 Porosity 10.4
CHAPTER 10 Characteristics of the Surfaces of Biomaterials 10.1 Surface Characteristics Related to Chemical Bonding 10.2 Surface Chemistry Related to Bonding of Biological Molecules 10.3 Porosity 10.4
Supplementary Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Overview. Carbon in all its forms. Background & Discovery Fabrication. Important properties. Summary & References. Overview of current research
 Graphene Prepared for Solid State Physics II Pr Dagotto Spring 2009 Laurene Tetard 03/23/09 Overview Carbon in all its forms Background & Discovery Fabrication Important properties Overview of current
Graphene Prepared for Solid State Physics II Pr Dagotto Spring 2009 Laurene Tetard 03/23/09 Overview Carbon in all its forms Background & Discovery Fabrication Important properties Overview of current
Surface Hydrophilic Treatment of Polyester Films via UV irradiation
 Surface Hydrophilic Treatment of Polyester Films via UV irradiation Gwang Hoe Koo, Hae Sung Lee and Jinho Jang School of Advanced Materials and System Engineering, Kumoh National Institute of Technology,
Surface Hydrophilic Treatment of Polyester Films via UV irradiation Gwang Hoe Koo, Hae Sung Lee and Jinho Jang School of Advanced Materials and System Engineering, Kumoh National Institute of Technology,
Module 3: "Components of Surface Energy" Lecture 14: "" The Lecture Contains: Surface Energy. Spcial Cases. Objectives_template
 The Lecture Contains: Surface Energy Spcial Cases file:///e /courses/colloid_interface_science/lecture14/14_1.htm[6/16/2012 1:37:24 PM] Surface Energy (3.20) On the RHS, the first term is the apolar (or
The Lecture Contains: Surface Energy Spcial Cases file:///e /courses/colloid_interface_science/lecture14/14_1.htm[6/16/2012 1:37:24 PM] Surface Energy (3.20) On the RHS, the first term is the apolar (or
Influence of the ambient temperature on water and diiodomethane contact angle with quartz surface
 10.1515/umcschem-2015-0009 ANNALES UNIVERSITATIS MARIAE CURIE-SKŁODOWSKA LUBLIN POLONIA VOL. LXX, 1 SECTIO AA 2015 Influence of the ambient temperature on water and diiodomethane contact angle with quartz
10.1515/umcschem-2015-0009 ANNALES UNIVERSITATIS MARIAE CURIE-SKŁODOWSKA LUBLIN POLONIA VOL. LXX, 1 SECTIO AA 2015 Influence of the ambient temperature on water and diiodomethane contact angle with quartz
Abstract. The principles and applicability of surface structure and hydrophobicity of polymers (PS, PDMS),
 Contact Angle Goniometer: Hydrophobicity of Biomaterial Surfaces and Protein Coatings Eman Mousa Alhajji North Carolina State University Department of Materials Science and Engineering MSE 255 Lab Report
Contact Angle Goniometer: Hydrophobicity of Biomaterial Surfaces and Protein Coatings Eman Mousa Alhajji North Carolina State University Department of Materials Science and Engineering MSE 255 Lab Report
MOBILE SURFACE ANALYZER MSA MOBILE MEASUREMENT OF SURFACE FREE ENERGY WITH ONLY ONE CLICK
 MOBILE SURFACE ANALYZER MSA MOBILE MEASUREMENT OF SURFACE FREE ENERGY WITH ONLY ONE CLICK WE RE SETTING NEW STANDARDS FOR QUALITY CONTROL WITH THE MOBILE MEASUREMENT OF SURFACE FREE ENERGY One-click measurement
MOBILE SURFACE ANALYZER MSA MOBILE MEASUREMENT OF SURFACE FREE ENERGY WITH ONLY ONE CLICK WE RE SETTING NEW STANDARDS FOR QUALITY CONTROL WITH THE MOBILE MEASUREMENT OF SURFACE FREE ENERGY One-click measurement
CHEMISTRY. Chapter 11 Intermolecular Forces Liquids and Solids
 CHEMISTRY The Central Science 8 th Edition Chapter 11 Liquids and Solids Kozet YAPSAKLI States of Matter difference between states of matter is the distance between particles. In the solid and liquid states
CHEMISTRY The Central Science 8 th Edition Chapter 11 Liquids and Solids Kozet YAPSAKLI States of Matter difference between states of matter is the distance between particles. In the solid and liquid states
INTERMOLECULAR FORCES: LIQUIDS, SOLIDS & PHASE CHANGES (Silberberg, Chapter 12)
 INTERMOLECULAR FORCES: LIQUIDS, SOLIDS & PASE CANGES (Silberberg, Chapter 12) Intermolecular interactions Ideal gas molecules act independently PV=nRT Real gas molecules attract/repulse one another 2 n
INTERMOLECULAR FORCES: LIQUIDS, SOLIDS & PASE CANGES (Silberberg, Chapter 12) Intermolecular interactions Ideal gas molecules act independently PV=nRT Real gas molecules attract/repulse one another 2 n
Atomic and molecular interaction forces in biology
 Atomic and molecular interaction forces in biology 1 Outline Types of interactions relevant to biology Van der Waals interactions H-bond interactions Some properties of water Hydrophobic effect 2 Types
Atomic and molecular interaction forces in biology 1 Outline Types of interactions relevant to biology Van der Waals interactions H-bond interactions Some properties of water Hydrophobic effect 2 Types
States of Matter. Intermolecular Forces. The States of Matter. Intermolecular Forces. Intermolecular Forces
 Intermolecular Forces Have studied INTRAmolecular forces the forces holding atoms together to form compounds. Now turn to forces between molecules INTERmolecular forces. Forces between molecules, between
Intermolecular Forces Have studied INTRAmolecular forces the forces holding atoms together to form compounds. Now turn to forces between molecules INTERmolecular forces. Forces between molecules, between
CHARACTERIZATION OF FLUID-ROCK INTERACTION BY ADSORPTION CALORIMETRY
 SCA2018_053 1/8 CHARACTERIZATION OF FLUID-ROCK INTERACTION BY ADSORPTION CALORIMETRY D. Korobkov 1, V. Pletneva 1, E. Dyshlyuk 2 1 Schlumberger, Moscow Research Center and 2 Dahran Carbonate Research Center
SCA2018_053 1/8 CHARACTERIZATION OF FLUID-ROCK INTERACTION BY ADSORPTION CALORIMETRY D. Korobkov 1, V. Pletneva 1, E. Dyshlyuk 2 1 Schlumberger, Moscow Research Center and 2 Dahran Carbonate Research Center
The Wilhelmy balance. How can we measure surface tension? Surface tension, contact angles and wettability. Measuring surface tension.
 ow can we measure surface tension? Surface tension, contact angles and wettability www.wikihow.com/measure-surface-tension Measuring surface tension The Wilhelmy balance F Some methods: Wilhelmy plate
ow can we measure surface tension? Surface tension, contact angles and wettability www.wikihow.com/measure-surface-tension Measuring surface tension The Wilhelmy balance F Some methods: Wilhelmy plate
Chapter 11 Intermolecular Forces, Liquids, and Solids
 Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten Chapter 11, Liquids, and Solids John D. Bookstaver St. Charles Community College Cottleville,
Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten Chapter 11, Liquids, and Solids John D. Bookstaver St. Charles Community College Cottleville,
Droplet Migration during Condensation on Chemically Patterned. Micropillars
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry Please do 2016 not adjust margins RSC Advances ELECTRONIC SUPPORTING INFORMATION (ESI) Droplet Migration
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry Please do 2016 not adjust margins RSC Advances ELECTRONIC SUPPORTING INFORMATION (ESI) Droplet Migration
Lecture Presentation. Chapter 11. Liquids and Intermolecular Forces. John D. Bookstaver St. Charles Community College Cottleville, MO
 Lecture Presentation Chapter 11 Liquids and Intermolecular Forces John D. Bookstaver St. Charles Community College Cottleville, MO Properties of Gases, Liquids, and Solids State Volume Shape of State Density
Lecture Presentation Chapter 11 Liquids and Intermolecular Forces John D. Bookstaver St. Charles Community College Cottleville, MO Properties of Gases, Liquids, and Solids State Volume Shape of State Density
SURFACE FREE ENERGY OF VISCOELASTIC THERMAL COMPRESSED WOOD AND ITS WETTABILITY BY THE PHENOL-FORMALDEHYDE ADHESIVE
 SURFACE FREE ENERGY OF VISCOELASTIC THERMAL COMPRESSED WOOD AND ITS WETTABILITY BY THE PHENOL-FORMALDEHYDE ADHESIVE Marko Petrič 1*, Andreja Kutnar 1, Borut Kričej 1, Matjaž Pavlič 1, Frederick A. Kamke
SURFACE FREE ENERGY OF VISCOELASTIC THERMAL COMPRESSED WOOD AND ITS WETTABILITY BY THE PHENOL-FORMALDEHYDE ADHESIVE Marko Petrič 1*, Andreja Kutnar 1, Borut Kričej 1, Matjaž Pavlič 1, Frederick A. Kamke
Temperature Dependent Current-voltage Characteristics of P- type Crystalline Silicon Solar Cells Fabricated Using Screenprinting
 Temperature Dependent Current-voltage Characteristics of P- type Crystalline Silicon Solar Cells Fabricated Using Screenprinting Process Hyun-Jin Song, Won-Ki Lee, Chel-Jong Choi* School of Semiconductor
Temperature Dependent Current-voltage Characteristics of P- type Crystalline Silicon Solar Cells Fabricated Using Screenprinting Process Hyun-Jin Song, Won-Ki Lee, Chel-Jong Choi* School of Semiconductor
Chapter 11 Intermolecular Forces, Liquids, and Solids
 Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten Chapter 11, Liquids, and Solids States of Matter The fundamental difference between states of
Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten Chapter 11, Liquids, and Solids States of Matter The fundamental difference between states of
Fibre-matrix interfacial adhesion on composite properties in natural fibre composites
 Fibre-matrix interfacial adhesion on composite properties in natural fibre composites Le Quan Ngoc TRAN 1,2,a, Xiaowen YUAN 1,b*, Debes BHATTACHARYYA 3,c, Carlos A. FUENTES 2,d, Aart Willem VAN VUURE 2,e,
Fibre-matrix interfacial adhesion on composite properties in natural fibre composites Le Quan Ngoc TRAN 1,2,a, Xiaowen YUAN 1,b*, Debes BHATTACHARYYA 3,c, Carlos A. FUENTES 2,d, Aart Willem VAN VUURE 2,e,
INTERFACE PHENOMENA OF CATIONIZED COTTON WITH EPTAC
 OF CATIONIZED COTTON WITH EPTAC Ana Marija GRANCARIĆ Anita TARBUK Marijn M.C.G. WARMOESKERKEN INTRODUCTION The most important phase in cotton pretreatment and finishing is adsorption of chemical substances
OF CATIONIZED COTTON WITH EPTAC Ana Marija GRANCARIĆ Anita TARBUK Marijn M.C.G. WARMOESKERKEN INTRODUCTION The most important phase in cotton pretreatment and finishing is adsorption of chemical substances
Specific ion effects on the interaction of. hydrophobic and hydrophilic self assembled
 Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Colloid Chemistry. La chimica moderna e la sua comunicazione Silvia Gross.
 Colloid Chemistry La chimica moderna e la sua comunicazione Silvia Gross Istituto Dipartimento di Scienze di e Scienze Tecnologie Chimiche Molecolari ISTM-CNR, Università Università degli Studi degli Studi
Colloid Chemistry La chimica moderna e la sua comunicazione Silvia Gross Istituto Dipartimento di Scienze di e Scienze Tecnologie Chimiche Molecolari ISTM-CNR, Università Università degli Studi degli Studi
Improving Adhesive Bonding of Composites Through Surface Characterization Using Inverse Gas Chromatography (IGC) Methods
 Improving Adhesive Bonding of Composites Through Surface Characterization Using Inverse Gas Chromatography (IGC) Methods Rita Taitano Johnson Brian Flinn University of Washington Anaheim Convention Center
Improving Adhesive Bonding of Composites Through Surface Characterization Using Inverse Gas Chromatography (IGC) Methods Rita Taitano Johnson Brian Flinn University of Washington Anaheim Convention Center
Highly doped and exposed Cu(I)-N active sites within graphene towards. efficient oxygen reduction for zinc-air battery
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) for Energy & Environmental Science.
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) for Energy & Environmental Science.
Question 2 Identify the phase transition that occurs when CO 2 solid turns to CO 2 gas as it is heated.
 For answers, send email to: admin@tutor-homework.com. Include file name: Chemistry_Worksheet_0039 Price: $4 (c) 2012 www.tutor-homework.com: Tutoring, homework help, help with online classes. Chapter 11
For answers, send email to: admin@tutor-homework.com. Include file name: Chemistry_Worksheet_0039 Price: $4 (c) 2012 www.tutor-homework.com: Tutoring, homework help, help with online classes. Chapter 11
MOBILE SURFACE ANALYZER MSA MOBILE MEASUREMENT OF SURFACE FREE ENERGY WITH ONLY ONE CLICK
 MOBILE SURFACE ANALYZER MSA MOBILE MEASUREMENT OF SURFACE FREE ENERGY WITH ONLY ONE CLICK WE RE SETTING NEW STANDARDS FOR QUALITY CONTROL WITH THE MOBILE MEASUREMENT OF SURFACE FREE ENERGY One-click measurement
MOBILE SURFACE ANALYZER MSA MOBILE MEASUREMENT OF SURFACE FREE ENERGY WITH ONLY ONE CLICK WE RE SETTING NEW STANDARDS FOR QUALITY CONTROL WITH THE MOBILE MEASUREMENT OF SURFACE FREE ENERGY One-click measurement
A Novel Approach to the Layer Number-Controlled and Grain Size- Controlled Growth of High Quality Graphene for Nanoelectronics
 Supporting Information A Novel Approach to the Layer Number-Controlled and Grain Size- Controlled Growth of High Quality Graphene for Nanoelectronics Tej B. Limbu 1,2, Jean C. Hernández 3, Frank Mendoza
Supporting Information A Novel Approach to the Layer Number-Controlled and Grain Size- Controlled Growth of High Quality Graphene for Nanoelectronics Tej B. Limbu 1,2, Jean C. Hernández 3, Frank Mendoza
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa.
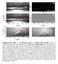 Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Application Report. Textile cleaning, washing, rewetting, surfactant, cotton, interfacial tension
 Application Report Cleaning of textiles Application report: AR231e Industry section: Textiles Author: C. Bilke-Krause Date: 01/2003 Method: Force Tensiometer K100 Drop Shape Analysis System DSA10 Keywords:
Application Report Cleaning of textiles Application report: AR231e Industry section: Textiles Author: C. Bilke-Krause Date: 01/2003 Method: Force Tensiometer K100 Drop Shape Analysis System DSA10 Keywords:
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2017 Supporting Information Upper Critical Solution Temperature Thermo-Responsive
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2017 Supporting Information Upper Critical Solution Temperature Thermo-Responsive
Wettability of CaCO 3 surfaces
 Colloids and Surfaces A: Physicochemical and Engineering Aspects 157 (1999) 333 340 www.elsevier.nl/locate/colsurfa Wettability of CaCO 3 surfaces Malvina G. Orkoula a,b, Petros G. Koutsoukos a,b, *, Michel
Colloids and Surfaces A: Physicochemical and Engineering Aspects 157 (1999) 333 340 www.elsevier.nl/locate/colsurfa Wettability of CaCO 3 surfaces Malvina G. Orkoula a,b, Petros G. Koutsoukos a,b, *, Michel
Hybrid Surface-Phonon-Plasmon Polariton Modes in Graphene /
 Supplementary Information: Hybrid Surface-Phonon-Plasmon Polariton Modes in Graphene / Monolayer h-bn stacks Victor W. Brar 1,2, Min Seok Jang 3,, Michelle Sherrott 1, Seyoon Kim 1, Josue J. Lopez 1, Laura
Supplementary Information: Hybrid Surface-Phonon-Plasmon Polariton Modes in Graphene / Monolayer h-bn stacks Victor W. Brar 1,2, Min Seok Jang 3,, Michelle Sherrott 1, Seyoon Kim 1, Josue J. Lopez 1, Laura
Chapter 11 Intermolecular Forces, Liquids, and Solids
 Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten Chapter 11 Intermolecular Forces, Liquids, and Solids John D. Bookstaver St. Charles Community
Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten Chapter 11 Intermolecular Forces, Liquids, and Solids John D. Bookstaver St. Charles Community
Supporting Information. Fast Synthesis of High-Performance Graphene by Rapid Thermal Chemical Vapor Deposition
 1 Supporting Information Fast Synthesis of High-Performance Graphene by Rapid Thermal Chemical Vapor Deposition Jaechul Ryu, 1,2, Youngsoo Kim, 4, Dongkwan Won, 1 Nayoung Kim, 1 Jin Sung Park, 1 Eun-Kyu
1 Supporting Information Fast Synthesis of High-Performance Graphene by Rapid Thermal Chemical Vapor Deposition Jaechul Ryu, 1,2, Youngsoo Kim, 4, Dongkwan Won, 1 Nayoung Kim, 1 Jin Sung Park, 1 Eun-Kyu
When Sessile Drops Are No Longer Small: Transitions from Spherical to Fully Flattened
 purify When Sessile Drops Are No Longer Small: Transitions from Spherical to Fully Flattened protect transport Chuck Extrand & Sung In Moon October 00 Outline Introduction Objectives Materials Method Analysis
purify When Sessile Drops Are No Longer Small: Transitions from Spherical to Fully Flattened protect transport Chuck Extrand & Sung In Moon October 00 Outline Introduction Objectives Materials Method Analysis
Intermolecular and Intramolecular Forces. Introduction
 Intermolecular and Intramolecular Forces Introduction Atoms can form stable units called molecules by sharing electrons. The formation of molecules is the result of intramolecular bonding (within the molecule)
Intermolecular and Intramolecular Forces Introduction Atoms can form stable units called molecules by sharing electrons. The formation of molecules is the result of intramolecular bonding (within the molecule)
Chapter 11 Intermolecular Forces, Liquids, and Solids
 Surveying the Chapter: Page 442 Chapter 11 Intermolecular Forces, Liquids, and Solids We begin with a brief comparison of solids, liquids, and gases from a molecular perspective, which reveals the important
Surveying the Chapter: Page 442 Chapter 11 Intermolecular Forces, Liquids, and Solids We begin with a brief comparison of solids, liquids, and gases from a molecular perspective, which reveals the important
Liquids and Solids The Condensed States of Matter
 Liquids and Solids The Condensed States of Matter AP Chemistry Ms. Grobsky Where We Have Been And Where We Are Going In the last few chapters, we saw that atoms can form stable units called molecules by
Liquids and Solids The Condensed States of Matter AP Chemistry Ms. Grobsky Where We Have Been And Where We Are Going In the last few chapters, we saw that atoms can form stable units called molecules by
Supplementary Information. depending on the atomic thickness of intrinsic and chemically doped. MoS 2
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supplementary Information Confocal absorption spectral imaging of MoS 2 : Optical transitions
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supplementary Information Confocal absorption spectral imaging of MoS 2 : Optical transitions
Surface Tension and its measurements
 Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading
 Loughborough University Institutional Repository Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading This item was submitted
Loughborough University Institutional Repository Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading This item was submitted
Solid-liquid interface
 Lecture Note #9 (Spring, 2017) Solid-liquid interface Reading: Shaw, ch. 6 Contact angles and wetting Wetting: the displacement from a surface of one fluid by another. A gas is displaced by a liquid at
Lecture Note #9 (Spring, 2017) Solid-liquid interface Reading: Shaw, ch. 6 Contact angles and wetting Wetting: the displacement from a surface of one fluid by another. A gas is displaced by a liquid at
Organic Chemistry. Introduction to Organic Molecules and Functional Groups
 For updated version, please click on http://ocw.ump.edu.my Organic Chemistry Introduction to Organic Molecules and Functional Groups by Dr. Seema Zareen & Dr. Izan Izwan Misnon Faculty Industrial Science
For updated version, please click on http://ocw.ump.edu.my Organic Chemistry Introduction to Organic Molecules and Functional Groups by Dr. Seema Zareen & Dr. Izan Izwan Misnon Faculty Industrial Science
Work-Function Decrease of Graphene Sheet. Using Alkali Metal Carbonates
 Supporting Information Work-Function Decrease of Graphene Sheet Using Alkali Metal Carbonates Ki Chang Kwon and Kyoung Soon Choi School of Chemical Engineering and Materials Science, Chung-Ang University
Supporting Information Work-Function Decrease of Graphene Sheet Using Alkali Metal Carbonates Ki Chang Kwon and Kyoung Soon Choi School of Chemical Engineering and Materials Science, Chung-Ang University
International Journal of Advancements in Research & Technology, Volume 3, Issue 11, November ISSN
 International Journal of Advancements in Research & Technology, Volume 3, Issue 11, November -2014 30 HEAT TRANSFER INTENSIFICATION USING NANOFLUIDS INTRODUCTION Prof. B.N. Havaraddi Assistant Professor
International Journal of Advancements in Research & Technology, Volume 3, Issue 11, November -2014 30 HEAT TRANSFER INTENSIFICATION USING NANOFLUIDS INTRODUCTION Prof. B.N. Havaraddi Assistant Professor
SUPPLEMENTARY INFORMATION
 Effect of airborne contaminants on the wettability of supported graphene and graphite Zhiting Li 1,ǂ, Yongjin Wang 2, ǂ, Andrew Kozbial 2, Ganesh Shenoy 1, Feng Zhou 1, Rebecca McGinley 2, Patrick Ireland
Effect of airborne contaminants on the wettability of supported graphene and graphite Zhiting Li 1,ǂ, Yongjin Wang 2, ǂ, Andrew Kozbial 2, Ganesh Shenoy 1, Feng Zhou 1, Rebecca McGinley 2, Patrick Ireland
Critical Micellization Concentration Determination using Surface Tension Phenomenon
 Critical Micellization Concentration Determination using Phenomenon 1. Introduction Surface-active agents (surfactants) were already known in ancient times, when their properties were used in everyday
Critical Micellization Concentration Determination using Phenomenon 1. Introduction Surface-active agents (surfactants) were already known in ancient times, when their properties were used in everyday
INTERMOLECULAR AND SURFACE FORCES
 INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
Supplementary Information for
 Supplementary Information for Highly Stable, Dual-Gated MoS 2 Transistors Encapsulated by Hexagonal Boron Nitride with Gate-Controllable Contact Resistance and Threshold Voltage Gwan-Hyoung Lee, Xu Cui,
Supplementary Information for Highly Stable, Dual-Gated MoS 2 Transistors Encapsulated by Hexagonal Boron Nitride with Gate-Controllable Contact Resistance and Threshold Voltage Gwan-Hyoung Lee, Xu Cui,
CH676 Physical Chemistry: Principles and Applications. CH676 Physical Chemistry: Principles and Applications
 CH676 Physical Chemistry: Principles and Applications History of Nanotechnology: Time Line Democritus in ancient Greece: concept of atom 1900 : Rutherford : discovery of atomic nucleus The first TEM was
CH676 Physical Chemistry: Principles and Applications History of Nanotechnology: Time Line Democritus in ancient Greece: concept of atom 1900 : Rutherford : discovery of atomic nucleus The first TEM was
Bioassay on a Robust and Stretchable Extreme Wetting. Substrate through Vacuum-Based Droplet Manipulation
 Supporting Information for A Single-Droplet Multiplex Bioassay on a Robust and Stretchable Extreme Wetting Substrate through Vacuum-Based Droplet Manipulation Heetak Han, Jung Seung Lee, Hyunchul Kim,
Supporting Information for A Single-Droplet Multiplex Bioassay on a Robust and Stretchable Extreme Wetting Substrate through Vacuum-Based Droplet Manipulation Heetak Han, Jung Seung Lee, Hyunchul Kim,
coatings ISSN
 Coatings 2014, 4, 527-552; doi:10.3390/coatings4030527 Article OPEN ACCESS coatings ISSN 2079-6412 www.mdpi.com/journal/coatings Wetting, Solubility and Chemical Characteristics of Plasma-Polymerized 1-Isopropyl-4-Methyl-1,4-Cyclohexadiene
Coatings 2014, 4, 527-552; doi:10.3390/coatings4030527 Article OPEN ACCESS coatings ISSN 2079-6412 www.mdpi.com/journal/coatings Wetting, Solubility and Chemical Characteristics of Plasma-Polymerized 1-Isopropyl-4-Methyl-1,4-Cyclohexadiene
Previous Faraday Discussions
 Previous Faraday Discussions All previous volumes can be viewed at Faraday Discussions from 2011 onwards are listed at Faraday Discussions 2000-2010 147: Chemistry of the Planets Introductory Lecture:
Previous Faraday Discussions All previous volumes can be viewed at Faraday Discussions from 2011 onwards are listed at Faraday Discussions 2000-2010 147: Chemistry of the Planets Introductory Lecture:
Proton-Conducting Nanocomposites and Hybrid Polymers
 Proton-onducting Nanocomposites and Hybrid Polymers Y.D. Premchand 1, M.L. Di Vona 2, and P. Knauth 1 1 Introduction This chapter is about proton-conducting nanocomposites and hybrid polymers. Before beginning
Proton-onducting Nanocomposites and Hybrid Polymers Y.D. Premchand 1, M.L. Di Vona 2, and P. Knauth 1 1 Introduction This chapter is about proton-conducting nanocomposites and hybrid polymers. Before beginning
Atomic Structure and Cohesion
 Nanotechnology for engineers Winter semester 2005-2006 Atomic Structure and Cohesion Chemical bonds Absorption and adsorption of particles on a surface Microscopic and Macroscopic Van der Waals (VdW) forces
Nanotechnology for engineers Winter semester 2005-2006 Atomic Structure and Cohesion Chemical bonds Absorption and adsorption of particles on a surface Microscopic and Macroscopic Van der Waals (VdW) forces
Atoms can form stable units called molecules by sharing electrons.
 Atoms can form stable units called molecules by sharing electrons. The formation of molecules is the result of intramolecular bonding (within the molecule) e.g. ionic, covalent. Forces that cause the aggregation
Atoms can form stable units called molecules by sharing electrons. The formation of molecules is the result of intramolecular bonding (within the molecule) e.g. ionic, covalent. Forces that cause the aggregation
Perfect mixing of immiscible macromolecules at fluid interfaces
 Perfect mixing of immiscible macromolecules at fluid interfaces Sergei S. Sheiko, 1* Jing Zhou, 1 Jamie Boyce, 1 Dorota Neugebauer, 2+ Krzysztof Matyjaszewski, 2 Constantinos Tsitsilianis, 4 Vladimir V.
Perfect mixing of immiscible macromolecules at fluid interfaces Sergei S. Sheiko, 1* Jing Zhou, 1 Jamie Boyce, 1 Dorota Neugebauer, 2+ Krzysztof Matyjaszewski, 2 Constantinos Tsitsilianis, 4 Vladimir V.
Production of Graphite Chloride and Bromide Using Microwave Sparks
 Supporting Information Production of Graphite Chloride and Bromide Using Microwave Sparks Jian Zheng, Hongtao Liu, Bin Wu, Chong-an Di, Yunlong Guo, Ti Wu, Gui Yu, Yunqi Liu, * and Daoben Zhu Key Laboratory
Supporting Information Production of Graphite Chloride and Bromide Using Microwave Sparks Jian Zheng, Hongtao Liu, Bin Wu, Chong-an Di, Yunlong Guo, Ti Wu, Gui Yu, Yunqi Liu, * and Daoben Zhu Key Laboratory
Supporting Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 207 Supporting Information Carbon Nanoscroll- Silk Crystallite Hybrid Structure with Controllable Hydration
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 207 Supporting Information Carbon Nanoscroll- Silk Crystallite Hybrid Structure with Controllable Hydration
Appendix A SPR Sensorgrams for Interactions of 3 Serum Proteins with Gold Nanoparticles
 Appendix A SPR Sensorgrams for Interactions of 3 Serum Proteins with Gold Nanoparticles Further from Sect. 5.3.2 (Figs. A.1, A.2 and A.3). Springer International Publishing Switzerland 2017 A. Patra, Quantifying
Appendix A SPR Sensorgrams for Interactions of 3 Serum Proteins with Gold Nanoparticles Further from Sect. 5.3.2 (Figs. A.1, A.2 and A.3). Springer International Publishing Switzerland 2017 A. Patra, Quantifying
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Zoltán Nagy Department of Physical Chemistry
 Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Zoltán Nagy Department of Physical Chemistry Surfaces and Interfaces Defining of interfacial region Types
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Zoltán Nagy Department of Physical Chemistry Surfaces and Interfaces Defining of interfacial region Types
Supplementary Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Supplementary Information Cross-linker Mediated Formation of Sulfur-functionalized V 2 O 5 /Graphene
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Supplementary Information Cross-linker Mediated Formation of Sulfur-functionalized V 2 O 5 /Graphene
Liquids & Solids. For the condensed states the ave KE is less than the attraction between molecules so they are held together.
 Liquids & Solids Intermolecular Forces Matter exists in 3 states. The state of matter is influenced by the physical properties of a substance. For liquids & solids, the condensed states, many of the physical
Liquids & Solids Intermolecular Forces Matter exists in 3 states. The state of matter is influenced by the physical properties of a substance. For liquids & solids, the condensed states, many of the physical
Atomistics of the Lithiation of Oxidized Silicon. Dynamics Simulations
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2016 Electronic Supplementary Information (ESI) Atomistics of the Lithiation of Oxidized
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2016 Electronic Supplementary Information (ESI) Atomistics of the Lithiation of Oxidized
Atomic Force Microscopy Characterization of Room- Temperature Adlayers of Small Organic Molecules through Graphene Templating
 Atomic Force icroscopy Characterization of Room- Temperature Adlayers of Small Organic olecules through Graphene Templating Peigen Cao, Ke Xu,2, Joseph O. Varghese, and James R. Heath *. Kavli Nanoscience
Atomic Force icroscopy Characterization of Room- Temperature Adlayers of Small Organic olecules through Graphene Templating Peigen Cao, Ke Xu,2, Joseph O. Varghese, and James R. Heath *. Kavli Nanoscience
