Raman spectroelectrochemical study of electrodeposited Prussian blue layer: dependence of spectral intensity on underlying electrode roughness
|
|
|
- Sophie Allison Waters
- 5 years ago
- Views:
Transcription
1 chemija vol. 28. No. 1. P lietuvos mokslų akademija, 2017 Raman spectroelectrochemical study of electrodeposited Prussian blue layer: dependence of spectral intensity on underlying electrode roughness Regina Mažeikienė, Gediminas Niaura, Albertas Malinauskas* Institute of Chemistry, Center for Physical Sciences and Technology, Saulėtekio Ave. 3, LT Vilnius, Lithuania Prussian blue (PB) has been electrodeposited at a smooth and at an electrochemically roughened gold electrodes as a thin layer of a few tens of nmol/cm 2 coverage (or a few tens of nanometers thick), and studied by Raman spectroelectrochemical technique using 785-nm laser excitation. It has been shown that the spectra obtained from a roughened electrode are by one order of magnitude more intense than those obtained from a smooth electrode. This shows that even at a relative thick layer of a modifier PB the surface enhancement of Raman spectra plays an important role in obtaining well-defined spectra from modified electrodes. Keywords: Raman spectroscopy, spectroelectrochemistry, modified electrode, Prussian blue INTRODUCTION Surface enhanced Raman scattering (SERS) is an important phenomenon, widely applied in different fields of research and technology [1]. Localised surface plasmon resonance as a response to optical excitation by visible and near infrared light has been known long ago. As a result of this resonance, strong electromagnetic fields are generated, and the intensity of inelastic Raman scattering from molecules present at a nanostructurized noble metal surface is highly increased. A total enhancement ranging from 10 5 to can be obtained [1]. Many efforts have been made through decades to create SERS-active structures capable to enhance Raman scattering from adsorbed molecules or particles. In particular, noble metal nanoparticles as plasmonic nanosensors possessing extremely high enhancement, even reaching a single molecule detection level, are widely used and well reviewed [2 4]. Also, e-beam lithography and related techniques are used to obtain repeatable sensitive substrates [5, 6]. Morphological diversity of silver nanomaterials has been shown to highly influence sensitive and selective detection schemes based on SERS [7]. It is well known that the most intense Raman scattering is obtained from the nearest layer of adsorbate adjacent to * Corresponding author. albertas.malinauskas@ftmc.lt the nanostructured interface. For electrochemical applications, like sensors, batteries or electrocatalytic systems, however, much thicker layers of electrode modifiers are often used. Then, the problem is whether the data obtained by Raman spectroscopy, especially by SERS, relate to a thin, even a monomolecular layer at the electrode surface, or to a thicker one? Or: How does Raman scattering from a monolayer of adsorbate compete with that from the bulk of the modifier layer? Previously, for relatively thin layers of adsorbed azine type dyes Toluidine blue [8] or Neutral red [9] we obtained Raman spectra closely related to those from electropolymerized layers of these species. In this case, however, electropolymerized layers do not differ significantly by their thickness from adsorbed layers. A different situation would be observed for thicker layers of an electrode modifier, like e. g. transition metal hexacyanoferrate complexes, often used for electrocatalytic sensors and biosensors [10, 11]. In such cases, the main question is as follows: Do the spectral features and their changes observed relate to a thin layer nearest to the electrode surface, or to an entire thick layer of a modifier at the electrode? There should be many theoretical aspects of this important problem, however, our present study has been aimed at more practically related aspects. In the present study, we concerned about Prussian blue (ferrous hexacyanoferrate) modified electrode, well known and widely applied in electrocatalysis and electroanalytical systems.
2 29 EXPERIMENTAL A BASi-Epsilon model potentiostat (Bioanalytical Systems Inc., USA) was used in electrochemical experiments. As a working electrode, a flat circular gold electrode of 5 mm in diameter, press-fitted into a Teflon holder, was used. Platinum wire, as a counter electrode, and a KCl saturated Ag/AgCl reference electrode were used to accomplish an electrochemical circuit. All potential values reported below refer to this reference electrode. Commercially available analytical or reagent grade chemicals were used as received. The electrode preparation procedure consisted of etching for 1 h in a Piranha solution (a mixture of 30% hydrogen peroxide and sulfuric acid, approx. 3:1 by volume), and the next following cleaning by ultrasonication for 2 min in ethanol and water mixture. Following this way, a clean smooth electrode was prepared, designed below as an Au electrode. For preparation of a roughened SERS active electrode (designed below as Au(SERS) electrode), the Au electrode was additionally electrochemically roughened in a 0.1 M solution of KCl by potential cycling for 50 cycles within the limits of 0.3 and 1.4 V at a scan rate of 0.2 V/s. The deposition of a Prussian blue layer onto the Au or Au(SERS) working electrode has been performed in a freshly prepared solution containing 0.1 M KCl, 0.1 M HCl, 2.5 mm K 3 [Fe(CN) 6 ], and 2.5 mm FeCl 3 (ph 0.95) by applying a controlled potential of 0.4 V for 1 min. After that, the electrode was rinsed with water and transferred into a background electrolyte containing 0.1 M KH 2 and 0.1 M KCl (ph 5.4). All spectroelectrochemical experiments were done in this background electrolyte. Raman spectroelectrochemical experiments were done in a cylinder-shaped three electrode cell, equipped with a gold working electrode (as indicated above), a platinum wire counter electrode, and a KCl saturated Ag/AgCl reference electrode. Raman spectra were recorded using an Echelle type spectrometer RamanFlex 400 (PerkinElmer, Inc.) equipped with a thermoelectrically cooled up to 50 C CCD camera and a fiber-optic cable for excitation and collection of the Raman spectra. The 785-nm beam of the diode laser was used as the excitation source. The 180 scattering geometry was employed. The laser power at the sample was restricted to 30 mw and the beam was focused to a 0.2 mm diameter spot on the electrode. The integration time was 100 s. The working electrode was placed at approx. 3 mm distance from the cell window. In order to reduce photo- and thermoeffects, and a possible degradation of the Prussian blue film by the incident light as well, the cell holder was moved periodically with respect to the laser beam at ca mm/s with the help of custom built equipment [12]. RESULTS AND DISCUSSION Figure 1 compares the cyclic voltammograms for two Prussian blue (PB) modified electrodes a smooth cleaned gold Current, ma Current, ma a b Potential, V (vs. Ag/AgCl) Fig. 1. Cyclic voltammograms of Prussian blue modified Au (a, at the top) and Au(SERS) (b, at the bottom) electrodes, as obtained in an electrolyte containing 0.1 M of KH 2 and 0.1 M of KCl within the potential scan limits of 0.1 to 0.5 V at different potential scan rate ranging from 5 to 200 mv/s (as indicated) electrode, and a rougnened, prepared for SERS studies gold electrode. For both electrodes, the well defined anodic and cathodic peaks are observed. Within an entire potential scan rate range studied the peak pairs show a clearly defined separation on the potential scale ranging from 0.03 V at the lowest scan rate of 5 mv/s to 0.22 or 0.26 V at the fastest scan of 200 mv/s for PB/Au and PB/Au(SERS) electrodes, respectively. Taking into account that both anodic and cathodic peaks are obtained from the surface-bound PB layers, it could be concluded that the rate of the electrochemical redox process is controlled by the diffusion of some species through the PB layer, most probably potassium ions according to the equation: Fe (N)4 I [Fe (C) (CN) 6 ] 3 + 4e + 4K + K 4 Fe (N)4 [Fe (C) (CN) 6 ] 3. Here Fe (N) and Fe (C) represent Fe ions, coordinated either to N (high-spin Fe ions), or to C (low-spin Fe ions) atoms, respectively. Also, the closely related midpotential (E pa + E pc )/2 values were obtained for both types of electrodes. For the PB/ Au electrode, midpotential values of and V were obtained for a potential scan rate of 5 and 200 mv/s,
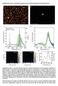
3 30 a respectively, whereas values of and V were obtained for the PB/Au(SERS) electrode, respectively. The obtained values coincide within the error limits with those reported previously [10]. A linear dependence of the peak potential and peak current for both electrodes on the square rooth of the potential scan rate was obtained, as shown in Fig. 2. Extrapolation of the dependence of the peak potential on v 1/2 to the zero potential scan rate yields again nearly coinciding values of a midpotential of V for both types of the modified electrode. A linear dependence of the peak current on v 1/2, as observed for the cathodic and anodic peaks for both electrodes (Fig. 2), confirms the diffusion-controlled nature of both redox processes. By integration of anodic and cathodic peak currents, a specific electric charge characteristic of redox processes of both types of the modified electrode could be obtained. For the smooth PB/Au electrode, a mean specific redox charge of 3.02 mc/cm 2 was obtained both from anodic and cathodic peaks, and for all potential scan rate values studied. For the roughened PB/Au(SERS) electrode, a somewhat higher value of a specific charge of 3.67 mc/cm 2 was calculated. Probably, this higher redox capacity of the latter electrode corresponds to somewhat higher surface area, obtained by the roughening procedure. Despite of this difference, the redox capacity for both types of the modified electrode appears closely related, indicating nearly equal amounts of Prussian blue deposited during the preparation procedure. The results presented are well reproducible during the repeated preparation of modified electrodes. Based on the redox charge observed, the mean thickness of the PB layer, as well as the mean coverage could be calculated. As for electrochemically prepared PB layers, it is well known that a redox charge of 5 mc/ cm 2 corresponds to 100 nm of layer thickness [13]. Therefore, it could be calculated that the thickness of the PB layer of 60 and 73 nm is characteristic of the prepared PB/Au and PB/Au(SERS) electrodes, respectively. Alternatively, turning to the molar coverage of PB modified electrodes, the values of 31 and 38 nmol/cm 2 can be obtained for the PB/Au and PB/Au(SERS) electrodes, respectively. It could be concluded that nanometer-sized well-reproducible PB layers containing a few tens of nanomoles of PB per cm 2 were prepared by the electrodeposition procedure applied for both smooth and roughened gold electrodes. The electrodes prepared were subjected to a combined Raman electrochemical study. Figure 3 shows the characteristic Raman spectra, obtained from the smooth PB/Au electrode. Two different redox forms of PB are well discernible. At the applied potential of 0.1 V, the reduced form of PB, viz. Prussian white (PW), prevails. Within the most characteristic Raman spectral range of cm 1, stretching vibrations of the tripple C N bonds are observed. Different frequencies of C N bond stretchings are caused by a different valence state of the Fe ions, to which the CN ligands are coordinated. For PW, the presence of a characteristic frag- Peak current, ma Peak potential, V b Raman intensity, arb. v 1/2 /(mv/s) 1/2 Raman shift, cm 1 Fig. 2. Dependence of the peak potential (a, at the top) and peak potential (b, at the bottom) on the square rooth of potential scan rate, as obtained for the cathodic and anodic peaks of Prussian blue modified Au and Au(SERS) electrodes (as indicated) Fig. 3. Raman spectra, presented within wavenumber ranges of and cm 1, from the Prussian blue modified Au electrode, obtained in a solution containing 0.1 M of KH 2 and 0.1 M of KCl using 785 nm laser excitation at electrode potential of 0.1 and 0.3 V (as indicated)
4 31 Raman intensity, arb. Raman shift, cm 1 Fig. 4. Raman spectra, presented within a broad wavenumber range of cm 1, from the Prussian blue modified Au and Au(SERS) electrodes (as indicated), as obtained at the electrode potential of 0.1 V ment Fe CN Fe results in three Raman bands within this region: a strong band centered at 2132 cm 1, a mid-intense band at 2095 cm 1, and a weak band at 2048 cm 1. Within the lower frequency range, the well-defined mid-intense bands observed at 599 and 530 cm 1 (and probably some other low-intense bands within this range) correspond most probably to the Fe C stretching vibrations, and the band at 255 cm 1 corresponds to the deformation vibrations of Fe C N fragments [14]. At the applied potential of 0.3 V, the oxidised form of PB prevails. Within a high frequency range, a mid-intense band located at 2157 cm 1, and a weak band at 2092 cm 1 are observed (Fig. 3). Also, changes within the low-frequency range are evident by turning from PW to PB. The intensity of Raman bands for PB appears lower than for PW (Fig. 3). Nearly same results regarding Raman band frequencies were obtained from both PB/Au and PB/Au(SERS) electrodes, except for some minor bands within the lowfrequency range. However, a drastic difference in spectral intensity was observed for these two types of electrodes, as depicted in Fig. 4. The spectrum obtained from the PB/ Au(SERS) electrode is much more intense than that from the unroughened PB/Au electrode. By integration of band intensities within a range of cm 1, an enhancement factor of ca. 11 could be obtained for the roughened PB/Au(SERS) electrode. This shows that even at a relative thick layer of a modifier PB, consisting of a few tens of nanometers, or, in other terms, a few tens of nmol/cm 2 for surface coverage by an electrochemically active substance, the surface enhancement of Raman spectra plays an important role in obtaining well-defined spectra from modified electrodes. It could be speculated whether the thinnest (or even a monomolecular) layer at the electrode surface or the rest bulk layer add more or less to the net intensity of spectra, however, it is seen that the position of Raman bands as well as their relative intensity ratio do not practically differ for both types of electrodes. From a practical point of view, the results obtained show that even for relative thick modifier layers like those obtained by electrodeposition or electropolymerization procedures, next to monolayers of adsorbate, it is expedient to apply a surface roughening procedure in order to get more intense Raman spectra. CONCLUSIONS When deposited at a roughened gold electrode surface, Prussian blue layers show approximately ten times more intense Raman scattering as compared to those layers deposited at a smooth gold electrode. This clearly shows that even for relatively thick modifier layers of a few tens of nanometers, surface Raman enhancement plays an important role. References Received 22 December 2016 Accepted 29 December Y. Cao, J. Zhang, Y. Yang, Z. Huang, N. V. Long, C. Fu, Appl. Spectrosc. Rev., 50, 499 (2015). 2. J. Langer, S. M. Novikov, L. M. Liz-Marzan, Nanotechnology, 26, (2015). 3. S. K. Chauhan, S. Mukherji, Mater. Technol., 30, B167 (2015). 4. H. Wei, S. M. H. Abtahi, P. J. Vikesland, Environ. Sci. Nano, 2, 120 (2015). 5. N. A. Cinel, S. Cakmakyapan, S. Butun, G. Ertas, E. Ozbay, Phot. Nano. Fund. Appl., 15, 109 (2015). 6. A. Shiohara, Y. S. Wang, L. M. Liz-Marzan, J. Photochem. Photobiol., C, 21, 2 (2014). 7. J. S. Lee, Nanotechnol. Rev., 3, 499 (2014). 8. R. Mažeikienė, G. Niaura, O. Eicher-Lorka, A. Malinauskas, Vibrat. Spectrosc., 47, 105 (2008). 9. R. Mažeikienė, K. Balskus, O. Eicher-Lorka, G. Niaura, R. Meškys, A. Malinauskas, Vibrat. Spectrosc., 51, 238 (2009). 10. R. Mažeikienė, G. Niaura, A. Malinauskas, J. Electroanal. Chem., 660, 140 (2011). 11. R. Mažeikienė, G. Niaura, A. Malinauskas, J. Electroanal. Chem., 719, 60 (2014). 12. G. Niaura, A. K. Gaigalas, V. L. Vilker, J. Raman Spectrosc., 28, 1009 (1997). 13. K. Itaya, H. Akahoshi, S. Toshima, J. Electrochem. Soc., 129, 1498 (1982). 14. K. Nakamoto, Infrared Spectra of Inorganic and Coordination Compounds, Wiley, New York (1963).
5 32 ELEKTRONU SODINTO BERLYNO MĖLIO SLUOKSNIO TYRIMAS RAMANO SPEKTROELEKTROCHEMINIU METODU: ELEKTRODO ŠIURKŠTUMO ĮTAKA SPEKTRŲ INTENSYVUMUI Santrauka Ploni Berlyno mėlio sluoksniai elektrocheminiu būdu nusodinti ant lygaus ir elektrochemiškai šiurkštinto aukso elektrodo paviršių. Gautieji sluoksniai, kurių storis sudaro kelias dešimtis nmol/cm 2 (arba kelias dešimtis nanometrų), buvo tiriami Ramano spektroelektrochemijos metodu, taikant 785 nm bangos ilgio spektro sužadinimą. Nustatyta, kad spektrai, gauti nuo šiurkštinto elektrodo paviršiaus, yra maždaug viena eile intensyvesni nei spektrai, gauti nuo lygaus elektrodo paviršiaus. Prieita prie išvados, kad, net esant gana storam elektrodo padengimui Berlyno mėlio sluoksniu, paviršiaus stiprinimas yra svarbus veiksnys, lemiantis bendrą Ramano spektro intensyvumą.
In situ time-resolved Raman spectroelectrochemical study of aniline polymerization at platinum and gold electrodes
 CHEMIJA. 2018. Vol. 29. No. 2. P. 81 88 Lietuvos mokslų akademija, 2018 In situ time-resolved Raman spectroelectrochemical study of aniline polymerization at platinum and gold electrodes Regina Mažeikienė,
CHEMIJA. 2018. Vol. 29. No. 2. P. 81 88 Lietuvos mokslų akademija, 2018 In situ time-resolved Raman spectroelectrochemical study of aniline polymerization at platinum and gold electrodes Regina Mažeikienė,
Shell-isolated nanoparticle-enhanced Raman spectroscopy
 Shell-isolated nanoparticle-enhanced Raman spectroscopy Jian Feng Li, Yi Fan Huang, Yong Ding, Zhi Lin Yang, Song Bo Li, Xiao Shun Zhou, Feng Ru Fan, Wei Zhang, Zhi You Zhou, De Yin Wu, Bin Ren, Zhong
Shell-isolated nanoparticle-enhanced Raman spectroscopy Jian Feng Li, Yi Fan Huang, Yong Ding, Zhi Lin Yang, Song Bo Li, Xiao Shun Zhou, Feng Ru Fan, Wei Zhang, Zhi You Zhou, De Yin Wu, Bin Ren, Zhong
SUPPORTING INFORMATION. Direct Observation on Reaction Intermediates and the Role of. Cu Surfaces
 SUPPORTING INFORMATION Direct Observation on Reaction Intermediates and the Role of Bicarbonate Anions in CO 2 Electrochemical Reduction Reaction on Cu Surfaces Shangqian Zhu, Bei Jiang, Wen-Bin Cai, Minhua
SUPPORTING INFORMATION Direct Observation on Reaction Intermediates and the Role of Bicarbonate Anions in CO 2 Electrochemical Reduction Reaction on Cu Surfaces Shangqian Zhu, Bei Jiang, Wen-Bin Cai, Minhua
A new concept of charging supercapacitors based on a photovoltaic effect
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Electronic supporting information (ESI) A new concept of charging supercapacitors based on a photovoltaic
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Electronic supporting information (ESI) A new concept of charging supercapacitors based on a photovoltaic
Supplementary Information for. Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings
 Supplementary Information for Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings Supplementary Figure 1. Simulated from pristine graphene gratings at different Fermi energy
Supplementary Information for Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings Supplementary Figure 1. Simulated from pristine graphene gratings at different Fermi energy
Preparation of Prussian blue-modified screen-printed electrodes via a chemical deposition for mass production of stable hydrogen peroxide sensors
 Procedure 7 Preparation of Prussian blue-modified screen-printed electrodes via a chemical deposition for mass production of stable hydrogen peroxide sensors Francesco Ricci, Danila Moscone and Giuseppe
Procedure 7 Preparation of Prussian blue-modified screen-printed electrodes via a chemical deposition for mass production of stable hydrogen peroxide sensors Francesco Ricci, Danila Moscone and Giuseppe
Nickel Sulfides Freestanding Holey Films as Air-Breathing Electrodes for. Flexible Zn-Air Batteries
 Nickel Sulfides Freestanding Holey Films as Air-Breathing Electrodes for Flexible Zn-Air Batteries Kyle Marcus, 1,# Kun Liang, 1,# Wenhan Niu, 1,# Yang Yang 1,* 1 NanoScience Technology Center, Department
Nickel Sulfides Freestanding Holey Films as Air-Breathing Electrodes for Flexible Zn-Air Batteries Kyle Marcus, 1,# Kun Liang, 1,# Wenhan Niu, 1,# Yang Yang 1,* 1 NanoScience Technology Center, Department
Material Analysis. What do you want to know about your sample? How do you intend to do for obtaining the desired information from your sample?
 Material Analysis What do you want to know about your sample? How do you intend to do for obtaining the desired information from your sample? Why can you acquire the proper information? Symmetrical stretching
Material Analysis What do you want to know about your sample? How do you intend to do for obtaining the desired information from your sample? Why can you acquire the proper information? Symmetrical stretching
Supporting Information
 Supporting Information NiFe-Layered Double Hydroxide Nanosheet Arrays Supported on Carbon Cloth for Highly Sensitive Detection of Nitrite Yue Ma,, Yongchuang Wang,, Donghua Xie,, Yue Gu,, Haimin Zhang,
Supporting Information NiFe-Layered Double Hydroxide Nanosheet Arrays Supported on Carbon Cloth for Highly Sensitive Detection of Nitrite Yue Ma,, Yongchuang Wang,, Donghua Xie,, Yue Gu,, Haimin Zhang,
Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering
 Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering Yukun Han, 1 Hai Xiao, 2 and Hai-Lung Tsai 1, * 1 Department of Mechanical and Aerospace
Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering Yukun Han, 1 Hai Xiao, 2 and Hai-Lung Tsai 1, * 1 Department of Mechanical and Aerospace
Nanojet and Surface Enhanced Raman Spectroscopy (NASERS) for Highly Reproducible and Controllable Single Molecule Detection
 Nanojet and Surface Enhanced Raman Spectroscopy (NASERS) for Highly Reproducible and Controllable Single Molecule Detection Te-Wei Chang, Manas Ranjan Gartia and Gang Logan Liu Department of Electrical
Nanojet and Surface Enhanced Raman Spectroscopy (NASERS) for Highly Reproducible and Controllable Single Molecule Detection Te-Wei Chang, Manas Ranjan Gartia and Gang Logan Liu Department of Electrical
Supplementary Figure 1. Characterization of immobilized cobalt protoporphyrin electrode. The cyclic voltammogram of: (a) pyrolytic graphite
 Supplementary Figure 1. Characterization of immobilized cobalt protoporphyrin electrode. The cyclic voltammogram of: (a) pyrolytic graphite electrode; (b) pyrolytic graphite electrode with 100 µl 0.5 mm
Supplementary Figure 1. Characterization of immobilized cobalt protoporphyrin electrode. The cyclic voltammogram of: (a) pyrolytic graphite electrode; (b) pyrolytic graphite electrode with 100 µl 0.5 mm
HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS
 www.arpapress.com/volumes/vol19issue1/ijrras_19_1_06.pdf HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS M. Eslamifar Physics Department, BehbahanKhatamAl-Anbia
www.arpapress.com/volumes/vol19issue1/ijrras_19_1_06.pdf HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS M. Eslamifar Physics Department, BehbahanKhatamAl-Anbia
Voltammetric Comparison of the Electrochemical Oxidation of Toluene on Monolithic and Reticulated Glassy Carbon Electrodes in Aqueous Medium
 Portugaliae Electrochimica Acta 2010, 28(6), 397-404 DOI: 10.4152/pea.201006397 PORTUGALIAE ELECTROCHIMICA ACTA ISSN 1647-1571 Voltammetric Comparison of the Electrochemical Oxidation of Toluene on Monolithic
Portugaliae Electrochimica Acta 2010, 28(6), 397-404 DOI: 10.4152/pea.201006397 PORTUGALIAE ELECTROCHIMICA ACTA ISSN 1647-1571 Voltammetric Comparison of the Electrochemical Oxidation of Toluene on Monolithic
Lithium-ion Batteries Based on Vertically-Aligned Carbon Nanotubes and Ionic Liquid
 Electronic Supplementary Information Lithium-ion Batteries Based on Vertically-Aligned Carbon Nanotubes and Ionic Liquid Electrolytes Wen Lu, * Adam Goering, Liangti Qu, and Liming Dai * 1. Synthesis of
Electronic Supplementary Information Lithium-ion Batteries Based on Vertically-Aligned Carbon Nanotubes and Ionic Liquid Electrolytes Wen Lu, * Adam Goering, Liangti Qu, and Liming Dai * 1. Synthesis of
Università degli Studi di Bari "Aldo Moro"
 Università degli Studi di Bari "Aldo Moro" Table of contents 1. Introduction to Atomic Force Microscopy; 2. Introduction to Raman Spectroscopy; 3. The need for a hybrid technique Raman AFM microscopy;
Università degli Studi di Bari "Aldo Moro" Table of contents 1. Introduction to Atomic Force Microscopy; 2. Introduction to Raman Spectroscopy; 3. The need for a hybrid technique Raman AFM microscopy;
Supplementary Figure 1 Morphology and composition of the original carbon nanotube (CNT) sample. (a, b) TEM images of CNT; (c) EDS of CNT.
 1 Supplementary Figure 1 Morphology and composition of the original carbon nanotube (CNT sample. (a, b TEM images of CNT; (c EDS of CNT. Cobalt is not detected in the original CNT sample (Note: The accidentally
1 Supplementary Figure 1 Morphology and composition of the original carbon nanotube (CNT sample. (a, b TEM images of CNT; (c EDS of CNT. Cobalt is not detected in the original CNT sample (Note: The accidentally
Supporting Information. High Wettable and Metallic NiFe-Phosphate/Phosphide Catalyst Synthesized by
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information High Wettable and Metallic NiFe-Phosphate/Phosphide
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information High Wettable and Metallic NiFe-Phosphate/Phosphide
Surface-enhanced raman scattering from a layer of gold nanoparticles
 VNU Journal of Science, Mathematics - Physics 26 (2010) 187-192 Surface-enhanced raman scattering from a layer of gold nanoparticles Nguyen The Binh *, Nguyen Thanh Dinh, Nguyen Quang Dong, Vu Thi Khanh
VNU Journal of Science, Mathematics - Physics 26 (2010) 187-192 Surface-enhanced raman scattering from a layer of gold nanoparticles Nguyen The Binh *, Nguyen Thanh Dinh, Nguyen Quang Dong, Vu Thi Khanh
Supplementary Information. For. A Universal Method for Preparing Functional ITO Electrodes with Ultrahigh Stability
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary Information For A Universal Method for Preparing Functional ITO Electrodes with Ultrahigh
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary Information For A Universal Method for Preparing Functional ITO Electrodes with Ultrahigh
Application note. SERS study of EMImTFSI on gold surfaces. (c) rhd instruments GmbH & Co. KG Mareike Länger
 Application note SERS study of EMImTFSI on gold surfaces (c) 2013-2018 rhd instruments GmbH & Co. KG Mareike Länger Introduction Vibrational spectroscopy techniques like infrared or Raman spectroscopy
Application note SERS study of EMImTFSI on gold surfaces (c) 2013-2018 rhd instruments GmbH & Co. KG Mareike Länger Introduction Vibrational spectroscopy techniques like infrared or Raman spectroscopy
Gold nanothorns macroporous silicon hybrid structure: a simple and ultrasensitive platform for SERS
 Supporting Information Gold nanothorns macroporous silicon hybrid structure: a simple and ultrasensitive platform for SERS Kamran Khajehpour,* a Tim Williams, b,c Laure Bourgeois b,d and Sam Adeloju a
Supporting Information Gold nanothorns macroporous silicon hybrid structure: a simple and ultrasensitive platform for SERS Kamran Khajehpour,* a Tim Williams, b,c Laure Bourgeois b,d and Sam Adeloju a
Supporting Information
 Supporting Information Superstructural Raman Nanosensors with Integrated Dual Functions for Ultrasensitive Detection and Tunable Release of Molecules Jing Liu #, Jianhe Guo #, Guowen Meng and Donglei Fan*
Supporting Information Superstructural Raman Nanosensors with Integrated Dual Functions for Ultrasensitive Detection and Tunable Release of Molecules Jing Liu #, Jianhe Guo #, Guowen Meng and Donglei Fan*
Supporting Information
 Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2018. Supporting Information for Small, DOI: 10.1002/smll.201801523 Ultrasensitive Surface-Enhanced Raman Spectroscopy Detection Based
Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2018. Supporting Information for Small, DOI: 10.1002/smll.201801523 Ultrasensitive Surface-Enhanced Raman Spectroscopy Detection Based
"Surface-Enhanced Raman Scattering
 SMR: 1643/11 WINTER COLLEGE ON OPTICS ON OPTICS AND PHOTONICS IN NANOSCIENCE AND NANOTECHNOLOGY ( 7-18 February 2005) "Surface-Enhanced Raman Scattering presented by: Martin Moskovits University of California,
SMR: 1643/11 WINTER COLLEGE ON OPTICS ON OPTICS AND PHOTONICS IN NANOSCIENCE AND NANOTECHNOLOGY ( 7-18 February 2005) "Surface-Enhanced Raman Scattering presented by: Martin Moskovits University of California,
Supporting Information
 Supporting Information Highly Sensitive, Reproducible, and Stable SERS Sensors Based on Well-Controlled Silver Nanoparticles Decorated Silicon Nanowire Building Blocks Xue Mei Han, Hui Wang, Xue Mei Ou,
Supporting Information Highly Sensitive, Reproducible, and Stable SERS Sensors Based on Well-Controlled Silver Nanoparticles Decorated Silicon Nanowire Building Blocks Xue Mei Han, Hui Wang, Xue Mei Ou,
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 214 Electronic Supplementary Information Ultrathin and High-Ordered CoO Nanosheet
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 214 Electronic Supplementary Information Ultrathin and High-Ordered CoO Nanosheet
Goals. The laboratory instructor has already purged the solutions of dissolved. Purging the from these solutions prevents spurious
 Goals 41 Cyclic Voltammetry XXGoals The goals of this experiment are to: Learn how to set up a screen-printed electrode Learn how to operate the Gamry potentiostat Determine the redox potential of potassium
Goals 41 Cyclic Voltammetry XXGoals The goals of this experiment are to: Learn how to set up a screen-printed electrode Learn how to operate the Gamry potentiostat Determine the redox potential of potassium
Supporting Information
 Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2017. Supporting Information for Adv. Energy Mater., DOI: 10.1002/aenm.201701456 Selective Etching of Nitrogen-Doped Carbon by Steam
Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2017. Supporting Information for Adv. Energy Mater., DOI: 10.1002/aenm.201701456 Selective Etching of Nitrogen-Doped Carbon by Steam
Flexible, Transparent and Highly Sensitive SERS. Substrates with Cross-nanoporous Structures for
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 supplementary information Flexible, Transparent and Highly Sensitive SERS Substrates with Cross-nanoporous
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 supplementary information Flexible, Transparent and Highly Sensitive SERS Substrates with Cross-nanoporous
ECE280: Nano-Plasmonics and Its Applications. Week8
 ECE280: Nano-Plasmonics and Its Applications Week8 Surface Enhanced Raman Scattering (SERS) and Surface Plasmon Amplification by Stimulated Emission of Radiation (SPASER) Raman Scattering Chandrasekhara
ECE280: Nano-Plasmonics and Its Applications Week8 Surface Enhanced Raman Scattering (SERS) and Surface Plasmon Amplification by Stimulated Emission of Radiation (SPASER) Raman Scattering Chandrasekhara
Supplementary Information
 Electronic Supplementary Material (ESI) for Lab on a Chip. This journal is The Royal Society of Chemistry 217 Supplementary Information Continuous Fabrication of Plasmonic Photonic Microcapsules with Controllable
Electronic Supplementary Material (ESI) for Lab on a Chip. This journal is The Royal Society of Chemistry 217 Supplementary Information Continuous Fabrication of Plasmonic Photonic Microcapsules with Controllable
CHAPTER-5 CYCLIC VOLTAMETRIC STUDIES OF NOVEL INDOLE ANALOGUES PREPARED IN THE PRESENT STUDY
 CHAPTER-5 CYCLIC VOLTAMETRIC STUDIES OF NOVEL INDOLE ANALOGUES PREPARED IN THE PRESENT STUDY Page No. 175-187 5.1 Introduction 5.2 Theoretical 5.3 Experimental 5.4 References 5. 1 Introduction Electrochemical
CHAPTER-5 CYCLIC VOLTAMETRIC STUDIES OF NOVEL INDOLE ANALOGUES PREPARED IN THE PRESENT STUDY Page No. 175-187 5.1 Introduction 5.2 Theoretical 5.3 Experimental 5.4 References 5. 1 Introduction Electrochemical
Figure 1. Contact mode AFM (A) and the corresponding scanning Kelvin probe image (B) of Pt-TiN surface.
 Synopsis Synopsis of the thesis entitled Titanium Nitride-ased Electrode Materials for Oxidation of Small Molecules: pplications in Electrochemical Energy Systems submitted by Muhammed Musthafa O. T under
Synopsis Synopsis of the thesis entitled Titanium Nitride-ased Electrode Materials for Oxidation of Small Molecules: pplications in Electrochemical Energy Systems submitted by Muhammed Musthafa O. T under
Label-free SERS Selective Detection of Dopamine and Serotonin. Using Graphene-Au Nanopyramid Heterostructure
 Supporting nformation for Label-free Selective Detection of Dopamine and Serotonin Using Graphene-Au Nanopyramid Heterostructure Pu Wang 1, Ming Xia 1, Owen Liang 1, Ke Sun 1,2, Aaron F. Cipriano 3, Thomas
Supporting nformation for Label-free Selective Detection of Dopamine and Serotonin Using Graphene-Au Nanopyramid Heterostructure Pu Wang 1, Ming Xia 1, Owen Liang 1, Ke Sun 1,2, Aaron F. Cipriano 3, Thomas
Nanosphere Lithography
 Nanosphere Lithography Derec Ciafre 1, Lingyun Miao 2, and Keita Oka 1 1 Institute of Optics / 2 ECE Dept. University of Rochester Abstract Nanosphere Lithography is quickly emerging as an efficient, low
Nanosphere Lithography Derec Ciafre 1, Lingyun Miao 2, and Keita Oka 1 1 Institute of Optics / 2 ECE Dept. University of Rochester Abstract Nanosphere Lithography is quickly emerging as an efficient, low
Electronic Supplementary Material (ESI) for Chemical Communications This journal is The Royal Society of Chemistry 2011
 Supplementary Information for Selective adsorption toward toxic metal ions results in selective response: electrochemical studies on polypyrrole/reduced graphene oxide nanocomposite Experimental Section
Supplementary Information for Selective adsorption toward toxic metal ions results in selective response: electrochemical studies on polypyrrole/reduced graphene oxide nanocomposite Experimental Section
Chapter - 8. Summary and Conclusion
 Chapter - 8 Summary and Conclusion The present research explains the synthesis process of two transition metal oxide semiconductors SnO 2 and V 2 O 5 thin films with different morphologies and studies
Chapter - 8 Summary and Conclusion The present research explains the synthesis process of two transition metal oxide semiconductors SnO 2 and V 2 O 5 thin films with different morphologies and studies
Solution Purging. Goals. 1. Purge both solutions with an inert gas (preferably N 2
 Goals 43 Cyclic Voltammetry XXGoals The goals of this experiment are to: Learn how to set up a screen-printed electrode Learn how to operate the Gamry potentiostat Determine the redox potential of potassium
Goals 43 Cyclic Voltammetry XXGoals The goals of this experiment are to: Learn how to set up a screen-printed electrode Learn how to operate the Gamry potentiostat Determine the redox potential of potassium
[Supplementary Information] One-Pot Synthesis and Electrocatalytic Activity of Octapodal Au-Pd Nanoparticles
![[Supplementary Information] One-Pot Synthesis and Electrocatalytic Activity of Octapodal Au-Pd Nanoparticles [Supplementary Information] One-Pot Synthesis and Electrocatalytic Activity of Octapodal Au-Pd Nanoparticles](/thumbs/78/77448401.jpg) [Supplementary Information] One-Pot Synthesis and Electrocatalytic Activity of Octapodal Au-Pd Nanoparticles Jong Wook Hong, Young Wook Lee, Minjung Kim, Shin Wook Kang, and Sang Woo Han * Department of
[Supplementary Information] One-Pot Synthesis and Electrocatalytic Activity of Octapodal Au-Pd Nanoparticles Jong Wook Hong, Young Wook Lee, Minjung Kim, Shin Wook Kang, and Sang Woo Han * Department of
Supporting Information s for
 Supporting Information s for # Self-assembling of DNA-templated Au Nanoparticles into Nanowires and their enhanced SERS and Catalytic Applications Subrata Kundu* and M. Jayachandran Electrochemical Materials
Supporting Information s for # Self-assembling of DNA-templated Au Nanoparticles into Nanowires and their enhanced SERS and Catalytic Applications Subrata Kundu* and M. Jayachandran Electrochemical Materials
Evaluating nanogaps in Ag and Au nanoparticle clusters for SERS applications using COMSOL Multiphysics
 Evaluating nanogaps in Ag and Au nanoparticle clusters for SERS applications using COMSOL Multiphysics Ramesh Asapu 1, Radu-George Ciocarlan 2, Nathalie Claes 3, Natan Blommaerts 1, Sara Bals 3, Pegie
Evaluating nanogaps in Ag and Au nanoparticle clusters for SERS applications using COMSOL Multiphysics Ramesh Asapu 1, Radu-George Ciocarlan 2, Nathalie Claes 3, Natan Blommaerts 1, Sara Bals 3, Pegie
Supplementary Materials
 Atomic layer-deposited tunnel oxide stabilizes silicon photoanodes for water oxidation Yi Wei Chen 1, Jonathan D. Prange 2, Simon Dühnen 2, Yohan Park 1, Marika Gunji 1, Christopher E. D. Chidsey 2, and
Atomic layer-deposited tunnel oxide stabilizes silicon photoanodes for water oxidation Yi Wei Chen 1, Jonathan D. Prange 2, Simon Dühnen 2, Yohan Park 1, Marika Gunji 1, Christopher E. D. Chidsey 2, and
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Phosphorus-Doped CoS 2 Nanosheet Arrays as
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Phosphorus-Doped CoS 2 Nanosheet Arrays as
Carbon nanotubes and conducting polymer composites
 University of Wollongong Thesis Collections University of Wollongong Thesis Collection University of Wollongong Year 4 Carbon nanotubes and conducting polymer composites May Tahhan University of Wollongong
University of Wollongong Thesis Collections University of Wollongong Thesis Collection University of Wollongong Year 4 Carbon nanotubes and conducting polymer composites May Tahhan University of Wollongong
Electrodeposited nickel hydroxide on nickel foam with ultrahigh. capacitance
 Electrodeposited nickel hydroxide on nickel foam with ultrahigh capacitance Guang-Wu Yang, Cai-Ling Xu* and Hu-Lin Li* College of Chemistry and Chemical Engineering, Lanzhou University, 73 (PR China) 1.
Electrodeposited nickel hydroxide on nickel foam with ultrahigh capacitance Guang-Wu Yang, Cai-Ling Xu* and Hu-Lin Li* College of Chemistry and Chemical Engineering, Lanzhou University, 73 (PR China) 1.
Department of Chemistry, University of Idaho, Moscow, Idaho Christine M. Pharr and Peter R. Griffiths*
 Anal. Chem. 1997, 69, 4673-4679 Infrared Spectroelectrochemical Analysis of Adsorbed Hexacyanoferrate Species Formed during Potential Cycling in the Ferrocyanide/ Ferricyanide Redox Couple Christine M.
Anal. Chem. 1997, 69, 4673-4679 Infrared Spectroelectrochemical Analysis of Adsorbed Hexacyanoferrate Species Formed during Potential Cycling in the Ferrocyanide/ Ferricyanide Redox Couple Christine M.
GRAPHENE EFFECT ON EFFICIENCY OF TiO 2 -BASED DYE SENSITIZED SOLAR CELLS (DSSC)
 Communications in Physics, Vol. 26, No. 1 (2016), pp. 43-49 DOI:10.15625/0868-3166/26/1/7961 GRAPHENE EFFECT ON EFFICIENCY OF TiO 2 -BASED DYE SENSITIZED SOLAR CELLS (DSSC) NGUYEN THAI HA, PHAM DUY LONG,
Communications in Physics, Vol. 26, No. 1 (2016), pp. 43-49 DOI:10.15625/0868-3166/26/1/7961 GRAPHENE EFFECT ON EFFICIENCY OF TiO 2 -BASED DYE SENSITIZED SOLAR CELLS (DSSC) NGUYEN THAI HA, PHAM DUY LONG,
Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering
 Supporting Information Cyclic Electroplating and Stripping of Silver on Au@SiO 2 Core/Shell Nanoparticles for Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering Dan Li a, Da-Wei Li
Supporting Information Cyclic Electroplating and Stripping of Silver on Au@SiO 2 Core/Shell Nanoparticles for Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering Dan Li a, Da-Wei Li
Fundamental molecular electrochemistry - potential sweep voltammetry
 Fundamental molecular electrochemistry - potential sweep voltammetry Potential (aka voltammetric) sweep methods are the most common electrochemical methods in use by chemists today They provide an efficient
Fundamental molecular electrochemistry - potential sweep voltammetry Potential (aka voltammetric) sweep methods are the most common electrochemical methods in use by chemists today They provide an efficient
Natallia Strekal. Plasmonic films of noble metals for nanophotonics
 Natallia Strekal Plasmonic films of noble metals for nanophotonics The aim of our investigation is the mechanisms of light interactions with nanostructure and High Tech application in the field of nanophotonics
Natallia Strekal Plasmonic films of noble metals for nanophotonics The aim of our investigation is the mechanisms of light interactions with nanostructure and High Tech application in the field of nanophotonics
Supporting Information. Electropolymerization of aniline on nickel-based electrocatalysts substantially
 Supporting Information Electropolymerization of aniline on nickel-based electrocatalysts substantially enhances their performance for hydrogen evolution Fuzhan Song, Wei Li, Guanqun Han, and Yujie Sun*
Supporting Information Electropolymerization of aniline on nickel-based electrocatalysts substantially enhances their performance for hydrogen evolution Fuzhan Song, Wei Li, Guanqun Han, and Yujie Sun*
Application of Raman Spectroscopy for Noninvasive Detection of Target Compounds. Kyung-Min Lee
 Application of Raman Spectroscopy for Noninvasive Detection of Target Compounds Kyung-Min Lee Office of the Texas State Chemist, Texas AgriLife Research January 24, 2012 OTSC Seminar OFFICE OF THE TEXAS
Application of Raman Spectroscopy for Noninvasive Detection of Target Compounds Kyung-Min Lee Office of the Texas State Chemist, Texas AgriLife Research January 24, 2012 OTSC Seminar OFFICE OF THE TEXAS
Highly efficient SERS test strips
 Electronic Supplementary Information (ESI) for Highly efficient SERS test strips 5 Ran Zhang, a Bin-Bin Xu, a Xue-Qing Liu, a Yong-Lai Zhang, a Ying Xu, a Qi-Dai Chen, * a and Hong-Bo Sun* a,b 5 10 Experimental
Electronic Supplementary Information (ESI) for Highly efficient SERS test strips 5 Ran Zhang, a Bin-Bin Xu, a Xue-Qing Liu, a Yong-Lai Zhang, a Ying Xu, a Qi-Dai Chen, * a and Hong-Bo Sun* a,b 5 10 Experimental
Supplemental Information for
 Supplemental Information for Densely arranged two-dimensional silver nanoparticle assemblies with optical uniformity over vast areas as excellent surface-enhanced Raman scattering substrates Yoshimasa
Supplemental Information for Densely arranged two-dimensional silver nanoparticle assemblies with optical uniformity over vast areas as excellent surface-enhanced Raman scattering substrates Yoshimasa
RamanStation 400: a Versatile Platform for SERS Analysis
 FIELD APPLICATION REPORT Raman Spectroscopy Author: Dean H. Brown PerkinElmer, Inc. Shelton, CT USA RamanStation 400 RamanStation 400: a Versatile Platform for SERS Analysis Introduction Surface Enhanced
FIELD APPLICATION REPORT Raman Spectroscopy Author: Dean H. Brown PerkinElmer, Inc. Shelton, CT USA RamanStation 400 RamanStation 400: a Versatile Platform for SERS Analysis Introduction Surface Enhanced
6. Plasmon coupling between a flat gold interface and gold nanoparticles.
 6. Plasmon coupling between a flat gold interface and gold nanoparticles. 6.1. Introduction In this outlook oriented chapter the applicability of the multilayered system used in chapter 4.1., for the study
6. Plasmon coupling between a flat gold interface and gold nanoparticles. 6.1. Introduction In this outlook oriented chapter the applicability of the multilayered system used in chapter 4.1., for the study
Cyclic Voltammetry. Objective: To learn the basics of cyclic voltammetry with a well-behaved echem system
 Cyclic Voltammetry Objective: To learn the basics of cyclic voltammetry with a well-behaved echem system Introduction Cyclic voltammetry (CV) is a popular electroanalytical technique for its relative simplicity
Cyclic Voltammetry Objective: To learn the basics of cyclic voltammetry with a well-behaved echem system Introduction Cyclic voltammetry (CV) is a popular electroanalytical technique for its relative simplicity
High-Performance Flexible Asymmetric Supercapacitors Based on 3D. Electrodes
 Supporting Information for: High-Performance Flexible Asymmetric Supercapacitors Based on 3D Porous Graphene/MnO 2 Nanorod and Graphene/Ag Hybrid Thin-Film Electrodes Yuanlong Shao, a Hongzhi Wang,* a
Supporting Information for: High-Performance Flexible Asymmetric Supercapacitors Based on 3D Porous Graphene/MnO 2 Nanorod and Graphene/Ag Hybrid Thin-Film Electrodes Yuanlong Shao, a Hongzhi Wang,* a
PT/NI COUNTER-ELECTRODES WITH IMPROVED STABILITY FOR DYE SENSITIZED SOLAR CELLS
 PT/NI COUNTER-ELECTRODES WITH IMPROVED STABILITY FOR DYE SENSITIZED SOLAR CELLS G. Syrrokostas, G. Leftheriotis and P. Yianoulis Energy and Environment Lab, Physics Department, University of Patras, Rion,
PT/NI COUNTER-ELECTRODES WITH IMPROVED STABILITY FOR DYE SENSITIZED SOLAR CELLS G. Syrrokostas, G. Leftheriotis and P. Yianoulis Energy and Environment Lab, Physics Department, University of Patras, Rion,
Vibrational Spectroscopies. C-874 University of Delaware
 Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
Supporting Information. One-Pot Synthesis of Reduced Graphene
 Supporting Information One-Pot Synthesis of Reduced Graphene Oxide/Metal (oxide) Composites Xu Wu, Yuqian Xing, David Pierce, Julia Xiaojun Zhao* a Department of Chemistry, University of North Dakota,
Supporting Information One-Pot Synthesis of Reduced Graphene Oxide/Metal (oxide) Composites Xu Wu, Yuqian Xing, David Pierce, Julia Xiaojun Zhao* a Department of Chemistry, University of North Dakota,
Introduction to Cyclic Voltammetry Measurements *
 OpenStax-CNX module: m34669 1 Introduction to Cyclic Voltammetry Measurements * Xianyu Li Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License
OpenStax-CNX module: m34669 1 Introduction to Cyclic Voltammetry Measurements * Xianyu Li Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License
Supplementary Information. "Enhanced light-matter interactions in. graphene-covered gold nanovoid arrays"
 Supplementary Information "Enhanced light-matter interactions in graphene-covered gold nanovoid arrays" Xiaolong Zhu,, Lei Shi, Michael S. Schmidt, Anja Boisen, Ole Hansen,, Jian Zi, Sanshui Xiao,,, and
Supplementary Information "Enhanced light-matter interactions in graphene-covered gold nanovoid arrays" Xiaolong Zhu,, Lei Shi, Michael S. Schmidt, Anja Boisen, Ole Hansen,, Jian Zi, Sanshui Xiao,,, and
Diffraction Gratings as a Chemical Sensing Platform
 Diffraction Gratings as a Chemical Sensing Platform Gordon T. Mitchell Center for Process Analytical Chemistry Department of Chemistry University of Washington Grating Light Reflection Spectroscopy Refractive
Diffraction Gratings as a Chemical Sensing Platform Gordon T. Mitchell Center for Process Analytical Chemistry Department of Chemistry University of Washington Grating Light Reflection Spectroscopy Refractive
Templated electrochemical fabrication of hollow. molybdenum sulfide micro and nanostructures. with catalytic properties for hydrogen production
 Supporting Information Templated electrochemical fabrication of hollow molybdenum sulfide micro and nanostructures with catalytic properties for hydrogen production Adriano Ambrosi, Martin Pumera* Division
Supporting Information Templated electrochemical fabrication of hollow molybdenum sulfide micro and nanostructures with catalytic properties for hydrogen production Adriano Ambrosi, Martin Pumera* Division
Large-Area and Uniform Surface-Enhanced Raman. Saturation
 Supporting Information Large-Area and Uniform Surface-Enhanced Raman Spectroscopy Substrate Optimized by Enhancement Saturation Daejong Yang 1, Hyunjun Cho 2, Sukmo Koo 1, Sagar R. Vaidyanathan 2, Kelly
Supporting Information Large-Area and Uniform Surface-Enhanced Raman Spectroscopy Substrate Optimized by Enhancement Saturation Daejong Yang 1, Hyunjun Cho 2, Sukmo Koo 1, Sagar R. Vaidyanathan 2, Kelly
Flexible Organic Photovoltaics Employ laser produced metal nanoparticles into the absorption layer 1. An Introduction
 Flexible Organic Photovoltaics Employ laser produced metal nanoparticles into the absorption layer 1. An Introduction Among the renewable energy sources that are called to satisfy the continuously increased
Flexible Organic Photovoltaics Employ laser produced metal nanoparticles into the absorption layer 1. An Introduction Among the renewable energy sources that are called to satisfy the continuously increased
Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation
 Supplementary Information Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation Yu Liu, Shuping Xu, Haibo Li, Xiaoguang Jian, Weiqing Xu* State Key
Supplementary Information Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation Yu Liu, Shuping Xu, Haibo Li, Xiaoguang Jian, Weiqing Xu* State Key
Highly Surface-roughened Flower-like Silver Nanoparticles for Extremely Sensitive Substrates of Surface-enhanced Raman Scattering
 Highly Surface-roughened Flower-like Silver Nanoparticles for Extremely Sensitive Substrates of Surface-enhanced Raman Scattering By Hongyan Liang, Zhipeng Li, Wenzhong Wang, Youshi Wu, and Hongxing Xu*
Highly Surface-roughened Flower-like Silver Nanoparticles for Extremely Sensitive Substrates of Surface-enhanced Raman Scattering By Hongyan Liang, Zhipeng Li, Wenzhong Wang, Youshi Wu, and Hongxing Xu*
Effect of Spiral Microwave Antenna Configuration on the Production of Nano-crystalline Film by Chemical Sputtering in ECR Plasma
 THE HARRIS SCIENCE REVIEW OF DOSHISHA UNIVERSITY, VOL. 56, No. 1 April 2015 Effect of Spiral Microwave Antenna Configuration on the Production of Nano-crystalline Film by Chemical Sputtering in ECR Plasma
THE HARRIS SCIENCE REVIEW OF DOSHISHA UNIVERSITY, VOL. 56, No. 1 April 2015 Effect of Spiral Microwave Antenna Configuration on the Production of Nano-crystalline Film by Chemical Sputtering in ECR Plasma
Cation-Hydroxide-Water Co-Adsorption Inhibits the. Alkaline Hydrogen Oxidation Reaction
 Supporting Information Cation-Hydroxide-Water Co-Adsorption Inhibits the Alkaline Hydrogen Oxidation Reaction Hoon Taek Chung [a], Ulises Martinez [a], Ivana Matanovic [b,c] and Yu Seung Kim* [a]. [a]
Supporting Information Cation-Hydroxide-Water Co-Adsorption Inhibits the Alkaline Hydrogen Oxidation Reaction Hoon Taek Chung [a], Ulises Martinez [a], Ivana Matanovic [b,c] and Yu Seung Kim* [a]. [a]
Spectroscopy at nanometer scale
 Spectroscopy at nanometer scale 1. Physics of the spectroscopies 2. Spectroscopies for the bulk materials 3. Experimental setups for the spectroscopies 4. Physics and Chemistry of nanomaterials Various
Spectroscopy at nanometer scale 1. Physics of the spectroscopies 2. Spectroscopies for the bulk materials 3. Experimental setups for the spectroscopies 4. Physics and Chemistry of nanomaterials Various
Supporting Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Engineering Cu 2 O/NiO/Cu 2 MoS 4 Hybrid Photocathode for H 2 Generation in Water Chen Yang, a,b
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Engineering Cu 2 O/NiO/Cu 2 MoS 4 Hybrid Photocathode for H 2 Generation in Water Chen Yang, a,b
Nanoscale optical circuits: controlling light using localized surface plasmon resonances
 Nanoscale optical circuits: controlling light using localized surface plasmon resonances T. J. Davis, D. E. Gómez and K. C. Vernon CSIRO Materials Science and Engineering Localized surface plasmon (LSP)
Nanoscale optical circuits: controlling light using localized surface plasmon resonances T. J. Davis, D. E. Gómez and K. C. Vernon CSIRO Materials Science and Engineering Localized surface plasmon (LSP)
Supporting Information for: Dependence and Quantitative Modeling
 Supporting Information for: Tip-Enhanced Raman Voltammetry: Coverage Dependence and Quantitative Modeling Michael Mattei, Gyeongwon Kang, Guillaume Goubert, Dhabih V. Chulhai, George C. Schatz, Lasse Jensen,
Supporting Information for: Tip-Enhanced Raman Voltammetry: Coverage Dependence and Quantitative Modeling Michael Mattei, Gyeongwon Kang, Guillaume Goubert, Dhabih V. Chulhai, George C. Schatz, Lasse Jensen,
Correlating Hydrogen Evolution Reaction Activity in Alkaline Electrolyte to Hydrogen Binding Energy on Monometallic Surfaces
 Supplemental Materials for Correlating Hydrogen Evolution Reaction Activity in Alkaline Electrolyte to Hydrogen Binding Energy on Monometallic Surfaces Wenchao Sheng, a MyatNoeZin Myint, a Jingguang G.
Supplemental Materials for Correlating Hydrogen Evolution Reaction Activity in Alkaline Electrolyte to Hydrogen Binding Energy on Monometallic Surfaces Wenchao Sheng, a MyatNoeZin Myint, a Jingguang G.
Surface Chemistry and Reaction Dynamics of Electron Beam Induced Deposition Processes
 Surface Chemistry and Reaction Dynamics of Electron Beam Induced Deposition Processes e -? 2 nd FEBIP Workshop Thun, Switzerland 2008 Howard Fairbrother Johns Hopkins University Baltimore, MD, USA Outline
Surface Chemistry and Reaction Dynamics of Electron Beam Induced Deposition Processes e -? 2 nd FEBIP Workshop Thun, Switzerland 2008 Howard Fairbrother Johns Hopkins University Baltimore, MD, USA Outline
Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries
 Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries Supplementary Figure 1: Dark field scattering properties of individual nanoparticle on mirror geometries separated
Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries Supplementary Figure 1: Dark field scattering properties of individual nanoparticle on mirror geometries separated
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Experimental section Synthesis of Ni-Co Prussian
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Experimental section Synthesis of Ni-Co Prussian
An Optimal Substrate Design for SERS: Dual-Scale Diamond-Shaped Gold Nano-Structures Fabricated via Interference Lithography
 Supporting Information An Optimal Substrate Design for SERS: Dual-Scale Diamond-Shaped Gold Nano-Structures Fabricated via Interference Lithography Hyo-Jin Ahn a, Pradheep Thiyagarajan a, Lin Jia b, Sun-I
Supporting Information An Optimal Substrate Design for SERS: Dual-Scale Diamond-Shaped Gold Nano-Structures Fabricated via Interference Lithography Hyo-Jin Ahn a, Pradheep Thiyagarajan a, Lin Jia b, Sun-I
ELECTROCATALYSIS OF THE HYDROGEN-EVOLUTION REACTION BY ELECTRODEPOSITED AMORPHOUS COBALT SELENIDE FILMS
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Supplementary Information for: ELECTROCATALYSIS OF THE HYDROGEN-EVOLUTION
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Supplementary Information for: ELECTROCATALYSIS OF THE HYDROGEN-EVOLUTION
UV-vis Analysis of the Effect of Sodium Citrate on the Size and the Surface Plasmon Resonance of Au NPs. Eman Mousa Alhajji
 UV-vis Analysis of the Effect of Sodium Citrate on the Size and the Surface Plasmon Resonance of Au NPs Eman Mousa Alhajji North Carolina State University Department of Materials Science and Engineering
UV-vis Analysis of the Effect of Sodium Citrate on the Size and the Surface Plasmon Resonance of Au NPs Eman Mousa Alhajji North Carolina State University Department of Materials Science and Engineering
The effects of probe boundary conditions and propagation on nano- Raman spectroscopy
 The effects of probe boundary conditions and propagation on nano- Raman spectroscopy H. D. Hallen,* E. J. Ayars** and C. L. Jahncke*** * Physics Department, North Carolina State University, Raleigh, NC
The effects of probe boundary conditions and propagation on nano- Raman spectroscopy H. D. Hallen,* E. J. Ayars** and C. L. Jahncke*** * Physics Department, North Carolina State University, Raleigh, NC
Electrochemically-assisted self-assembly of mesoporous silica thin films SUPPLEMENTARY INFORMATION A. WALCARIUS, E. SIBOTTIER, M. ETIENNE, J.
 SUPPLEMENTARY INFORMATION Electrochemically-assisted self-assembly of mesoporous silica thin films A. WALCARIUS, E. SIBOTTIER, M. ETIENNE, J. GHANBAJA 50 nm Fig. S1. TEM image of an electrodeposited surfactant-templated
SUPPLEMENTARY INFORMATION Electrochemically-assisted self-assembly of mesoporous silica thin films A. WALCARIUS, E. SIBOTTIER, M. ETIENNE, J. GHANBAJA 50 nm Fig. S1. TEM image of an electrodeposited surfactant-templated
Carbon powder modification. Preparation of NS1, NS2, NS3 and NS4.
 SUPPORTING INFORMATION EXPERIMENTAL SECTION Reagents. Carbon powder (Norit-S50) was purchased from Norit, 4-aminobenzene sulfonic acid (99%), lithium perchlorate (99%, potassium ferricyanide (99%) and
SUPPORTING INFORMATION EXPERIMENTAL SECTION Reagents. Carbon powder (Norit-S50) was purchased from Norit, 4-aminobenzene sulfonic acid (99%), lithium perchlorate (99%, potassium ferricyanide (99%) and
Influence of Self-Assembling Redox Mediators on Charge Transfer at Hydrophobic Electrodes
 Supporting Information Influence of Self-Assembling Redox Mediators on Charge Transfer at Hydrophobic Electrodes Timothy J. Smith, Chenxuan Wang, and Nicholas L. Abbott* Department of Chemical and Biological
Supporting Information Influence of Self-Assembling Redox Mediators on Charge Transfer at Hydrophobic Electrodes Timothy J. Smith, Chenxuan Wang, and Nicholas L. Abbott* Department of Chemical and Biological
Surface-Enhanced Raman Spectroscopy
 GENERAL ARTICLE Surface-Enhanced Raman Spectroscopy Recent Advancement of Raman Spectroscopy Ujjal Kumar Sur Ujjal Kumar Sur works in the Netaji Nagar Day College, Kolkata, West Bengal. He is also a visiting
GENERAL ARTICLE Surface-Enhanced Raman Spectroscopy Recent Advancement of Raman Spectroscopy Ujjal Kumar Sur Ujjal Kumar Sur works in the Netaji Nagar Day College, Kolkata, West Bengal. He is also a visiting
Carbon Quantum Dots/NiFe Layered Double Hydroxide. Composite as High Efficient Electrocatalyst for Water
 Supplementary Information Carbon Quantum Dots/NiFe Layered Double Hydroxide Composite as High Efficient Electrocatalyst for Water Oxidation Di Tang, Juan Liu, Xuanyu Wu, Ruihua Liu, Xiao Han, Yuzhi Han,
Supplementary Information Carbon Quantum Dots/NiFe Layered Double Hydroxide Composite as High Efficient Electrocatalyst for Water Oxidation Di Tang, Juan Liu, Xuanyu Wu, Ruihua Liu, Xiao Han, Yuzhi Han,
Electronic Supplementary Information. for. Discrimination of dopamine from ascorbic acid and uric acid on thioglycolic. acid modified gold electrode
 Electronic Supplementary Information for Discrimination of dopamine from ascorbic acid and uric acid on thioglycolic acid modified gold electrode Guangming Liu,* a Jingjing Li, b Li Wang b, Nana Zong b,
Electronic Supplementary Information for Discrimination of dopamine from ascorbic acid and uric acid on thioglycolic acid modified gold electrode Guangming Liu,* a Jingjing Li, b Li Wang b, Nana Zong b,
Cu 2 graphene oxide composite for removal of contaminants from water and supercapacitor
 Electronic Supplementary Information (ESI) for Cu 2 O@reduced graphene oxide composite for removal of contaminants from water and supercapacitor Baojun Li, a Huaqiang Cao,* a Gui Yin, b Yuexiang Lu, a
Electronic Supplementary Information (ESI) for Cu 2 O@reduced graphene oxide composite for removal of contaminants from water and supercapacitor Baojun Li, a Huaqiang Cao,* a Gui Yin, b Yuexiang Lu, a
High-Flux CO Reduction Enabled by Three-Dimensional Nanostructured. Copper Electrodes
 Supporting Information High-Flux CO Reduction Enabled by Three-Dimensional Nanostructured Copper Electrodes Yuxuan Wang, David Raciti, Chao Wang * Department of Chemical and Biomolecular Engineering, Johns
Supporting Information High-Flux CO Reduction Enabled by Three-Dimensional Nanostructured Copper Electrodes Yuxuan Wang, David Raciti, Chao Wang * Department of Chemical and Biomolecular Engineering, Johns
Fig. S1 The Structure of RuCE(Left) and RuCA (Right)
 Supporting information Fabrication of CZTS and CZTSSe photocathode CZTS photocathode was fabricated by sulfurization of a stacked film containing Cu, Zn and Sn. The stacked film was fabricated on Mo coated
Supporting information Fabrication of CZTS and CZTSSe photocathode CZTS photocathode was fabricated by sulfurization of a stacked film containing Cu, Zn and Sn. The stacked film was fabricated on Mo coated
Study on Aqueous CO 2 Detection to Monitor the Potential Leakage of CO 2 Stored in the Ocean
 tudy on Aqueous CO 2 Detection to Monitor the Potential Leakage of CO 2 tored in the Ocean ATO iroshi : Ph. D., P. E. Jp (Applied cience), Advanced Applied cience Department, Research Laboratory, Corporate
tudy on Aqueous CO 2 Detection to Monitor the Potential Leakage of CO 2 tored in the Ocean ATO iroshi : Ph. D., P. E. Jp (Applied cience), Advanced Applied cience Department, Research Laboratory, Corporate
Infrared Spectroscopy: Identification of Unknown Substances
 Infrared Spectroscopy: Identification of Unknown Substances Suppose a white powder is one of the four following molecules. How can they be differentiated? H N N H H H H Na H H H H H A technique that is
Infrared Spectroscopy: Identification of Unknown Substances Suppose a white powder is one of the four following molecules. How can they be differentiated? H N N H H H H Na H H H H H A technique that is
Lecture 4. Conductance sensors. ChemFET. Electrochemical Impedance Spectroscopy. py Practical consideration for electrochemical biosensors.
 Lecture 4 Conductance sensors. ChemFET. Electrochemical Impedance Spectroscopy. py Practical consideration for electrochemical biosensors. Conductivity I V = I R=, L - conductance L= κa/, l Λ= κ /[ C]
Lecture 4 Conductance sensors. ChemFET. Electrochemical Impedance Spectroscopy. py Practical consideration for electrochemical biosensors. Conductivity I V = I R=, L - conductance L= κa/, l Λ= κ /[ C]
International Journal of Pure and Applied Sciences and Technology
 Int. J. Pure Appl. Sci. Technol., 9(1) (2012), pp. 1-8 International Journal of Pure and Applied Sciences and Technology ISSN 2229-6107 Available online at www.ijopaasat.in Research Paper Preparation,
Int. J. Pure Appl. Sci. Technol., 9(1) (2012), pp. 1-8 International Journal of Pure and Applied Sciences and Technology ISSN 2229-6107 Available online at www.ijopaasat.in Research Paper Preparation,
Three-dimensional Visualization and Quantification of Gold Nanomaterial Deposition and Aggregation in Porous Media via Raman Spectroscopy
 Raman Spectroscopy Nanomaterials Exposure? Three-dimensional Visualization and Quantification of Gold Nanomaterial Deposition and Aggregation in Porous Media via Raman Spectroscopy Matthew Y. Chan, Weinan
Raman Spectroscopy Nanomaterials Exposure? Three-dimensional Visualization and Quantification of Gold Nanomaterial Deposition and Aggregation in Porous Media via Raman Spectroscopy Matthew Y. Chan, Weinan
Visualizing the bi-directional electron transfer in a Schottky junction consisted of single CdS nanoparticles and a planar gold film
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Visualizing the bi-directional electron transfer in
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Visualizing the bi-directional electron transfer in
Supporting Information Inherent Electrochemistry and Charge Transfer Properties of Few-Layer Two Dimensional Ti 3 C 2 T x MXene
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 Supporting Information Inherent Electrochemistry and Charge Transfer Properties of Few-Layer Two
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 Supporting Information Inherent Electrochemistry and Charge Transfer Properties of Few-Layer Two
