Combustion Theory and Applications in CFD
|
|
|
- Preston Cannon
- 5 years ago
- Views:
Transcription
1 Combustion Theory and Applications in CFD Princeton Combustion Summer School 2018 Prof. Dr.-Ing. Heinz Pitsch Copyright by Heinz Pitsch. This material is not to be sold, reproduced or distributed without prior written permission of the owner, Heinz Pitsch.
2 Combustion Applications Mobility & Transport Institute for Combustion Technology Heinz Pitsch
3 Combustion Applications Power Generation Mobility & Transport Institute for Combustion Technology Heinz Pitsch
4 Combustion Applications Power Generation Mobility & Transport Industry & Household Institute for Combustion Technology Heinz Pitsch
5 Combustion Applications Power Generation Fire Safety & Explosions Mobility & Transport Industry & Household Institute for Combustion Technology Heinz Pitsch
6 Combustion Applications Power Generation Combustion is omnipresent! Mobility & Transport Industry & Household But causes emissions of Harmful pollutants Greenhouse gases Design challenge Combustion extremely complex, multi-scale & multi-physics Devices usually big and high power Measurement in severe environments difficult Computational engineering very important Fire Safety & Explosions Institute for Combustion Technology Heinz Pitsch
7 Combustion Science Combustion research started many years ago Why is combustion science still important? Why is combustion science interesting? HPC as an important step Institute for Combustion Technology Heinz Pitsch
8 US DOE s International Energy Outlook 2016 World Energy Consumption Increase in world wide energy consumption from 2012 until 2040: 50% Fossil fuels > 75% by 2040 World Energy Consumption by Fuel [10 15 Btu] Large numbers 120 million tons daily CO2 emissions in kg daily per person 10 billion liter daily fuel consumption 1.3 liter daily liquid fuel use Clean combustion extremely important! Institute for Combustion Technology Heinz Pitsch Source: EIA s International Energy Outlook, 2016
9 IEA s World Energy Outlook 2015 Primary Energy Demand by Region & Source in toe per person 6.7 toe per person 1.5 toe per person 2.8 toe per person 1.1 toe per person * Institute for Combustion Technology Heinz Pitsch * Computed as energy 2040 / people 2017
10 IEA s World Energy Outlook 2015 Primary Energy Demand by Region & Source in toe per person 6.7 toe per person 1.5 toe per person 2.8 toe per person 1.1 toe per person * Institute for Combustion Technology Heinz Pitsch * Computed as energy 2040 / people 2017
11 IEA s World Energy Outlook 2015 Primary Energy Demand by Region & Source in toe per person 6.7 toe per person 1.5 toe per person 2.8 toe per person 1.1 toe per person * Institute for Combustion Technology Heinz Pitsch * Computed as energy 2040 / people 2017
12 DOE s International Energy Outlook 2016 Transport Sector World Energy Consumption by Source Fraction of Battery Electric Vehicle (BEV) in 2040 still small 6% BEV for 450 scenario Quadrillion Btu Institute for Combustion Technology Heinz Pitsch
13 DOE s International Energy Outlook 2016 World Net Electricity Generation by Source For positive effect of BEV, electric power should be from renewable sources Trillion kwh Institute for Combustion Technology Heinz Pitsch
14 IEA s World Energy Outlook 2015 World Energy Demand in Mtoe by Fuel & Sector in 2040 GHG emissions can be reduced by considering 1. Energy use 2. Primary energy 3. Conversion process Institute for Combustion Technology Heinz Pitsch
15 Mitigation of GHG Emissions Energy Use Opportunities Electrification, but o electrical power has to be from renewable sources Also, not all relevant applications can be electrified, i.e. o industrial processes requiring high process temperatures o applications requiring high energy densities o Institute for Combustion Technology Heinz Pitsch
16 Mitigation of GHG Emissions Primary Energy and Conversion Process Opportunities Cleaner fuels, e.g. natural gas Almost 50% lower specific carbon emissions compared with coal Biofuels CoE Tailor-Made Fuels from Biomass Carbon-free fossil fuel combustion CCS, CCU (SFB Oxyflame) Renewable electricity Storage E-fuels Ammonia Hydrogen Hydrogen Storage Institute for Combustion Technology Heinz Pitsch
17 What is Combustion? What is the difference between combustion and fuel oxidation in a fuel cell? In contrast to isothermal chemically reacting flows Heat release induces temperature increase Thereby combustion is self accelerating Important Each chemical or physical process has associated time scale Interaction of flow (transport) and chemistry Laminar and turbulent combustion New dimensionless groups (similar to Reynolds number) 17 Damköhler number, Karlovitz number,
18 Combustion Applications: Examples Premixed combustion Spark-ignition engine Premixed Example: SI-engine Non-premixed combustion Diesel engine Aircraft engine Example: Aircraft engine 18
19 Reduction of Greenhouse Gas Emissions Various approaches: Hydrogen economy CO 2 -sequestration (Carbon Capture and Storage, CCS) Bio-fuels Increase in efficiency Combustion Theory 24
20 Aim of this Course Develop understanding of combustion processes from physical and chemical perspectives Fundamentals: Thermodynamics (Kinetics see parallel course) Fluid mechanics Heat and mass transfer Applications: Reciprocating engines Gas turbines Furnaces 26
21 Course Overview Part I: Fundamentals and Laminar Flames Part II: Turbulent Combustion 27
22 Course Overview Part I: Fundamentals and Laminar Flames Introduction Fundamentals and mass balances of combustion systems Thermodynamics, flame temperature, and equilibrium Governing equations Laminar premixed flames: Kinematics and Burning Velocity Laminar premixed flames: Flame structure Laminar diffusion flames FlameMaster flame calculator 28 Request access and download at
23 Course Overview Part II: Turbulent Combustion Turbulence Turbulent Premixed Combustion Turbulent Non-Premixed Combustion Turbulent Combustion Modeling Applications 29
24 Fundamentals and Mass Balances of Combustion Systems Princeton Combustion Summer School 2018 Prof. Dr.-Ing. Heinz Pitsch
25 Thermodynamics The final state (after very long time) of a homogeneous system is governed by the classical laws of thermodynamics! Prerequisites: Definitions of concentrations and thermodynamic variables Mass and energy balances for multicomponent systems 31
26 Course Overview Part I: Fundamentals and Laminar Flames Introduction Fundamentals and mass balances of combustion systems Thermodynamics, flame temperature, and equilibrium Governing equations Laminar premixed flames: Kinematics and burning velocity Laminar premixed flames: Flame structure Laminar diffusion flames FlameMaster flame calculator 32 Definitions, Equation of State, Mass Balance Elementary and Global Reactions Coupling Functions Stoichiometry Mixture Fraction Burke-Schumann Solution
27 Definitions, Equation of State, Mass Balance In chemical reactions mass and chemical elements are conserved Combustion always in (gas) mixtures The mole fraction Multi-component system with k different chemical species Mole: molecules are defined as one mole Avogadro number N A Number of moles of species i: n i Total number of moles: Mole fraction of species i: 33
28 The mass fraction Mass m i of all molecules of species i is related to its number of moles by where W i is the molecular weight of species i Total mass of all molecules in the mixture: Mass fraction of species i: Mean molecular weight W: Mass fraction and mole fraction: 34
29 The mass fraction of elements Mass fractions of elements are very useful in combustion Mass of the species changes due to chemical reactions, but mass of the elements is conserved Number of atoms of element j in a molecule of species i: a ij Mass of all atoms j in the system: where k e is the total number of elements in the system, W j is molecular weight of element j 35
30 The mass fraction of elements Mass fraction of element j is then From definitions above it follows 36
31 The partial molar density (concentration) Number of moles per volume V or partial molar density, the concentration: Total molar density of the system is then 37
32 The Partial Density Density and partial density are defined Partial molar density is related to partial density and mass fraction by (relation often important for evaluation of reaction rates) 38
33 The ideal gas thermal equation of state In most combustion systems, thermally ideal gas law is valid Even for high pressure combustion this is a sufficiently accurate approximation, because the temperatures are typically also very high In mixture of ideal gases, molecules of species i exert on the surrounding walls of the vessel the partial pressure Universal gas constant equal to 39
34 Dalton's law For an ideal gas the total pressure is equal to the sum of the partial pressures Thermal equation of state for a mixture of ideal gases x x x x x + x x x o From this follows o o o And for the volume = o x o x x x o x x x o x 40
35 *Example: Methane/Air Mixture Known: CH 4 -air-mixture; 5 mass percent CH 4, 95 mass percent air Air: 21% (volume fraction) O 2, 79% N 2 (approximately) Unknown: Mole fractions and element mass fractions Solution: Molar masses: Mass fractions in the air: In the mixture: Mean molar mass: 41
36 *Example: Methane/Air Mixture Mole fractions of Components: Molar mass of elements: with: Mass fractions of elements: Simplification: Whole numbers for values of the molar masses 42
37 Course Overview Part I: Fundamentals and Laminar Flames Introduction Fundamentals and mass balances of combustion systems Thermodynamics, flame temperature, and equilibrium Governing equations Laminar premixed flames: Kinematics and burning velocity Laminar premixed flames: Flame structure Laminar diffusion flames FlameMaster flame calculator 43 Definitions, Equation of State, Mass Balance Elementary and Global Reactions Coupling Functions Stoichiometry Mixture Fraction Burke-Schumann Solution
38 Elementary and Global Reactions Distinction between elementary reactions and global reactions important! Elementary reactions Describe actual micro-process of chemical reaction Only take place, if collisions between reactants take place Reaction velocities can be determined experimentally oder theoretically Global reactions Conversion of educts to products Ratios of amounts of substance Does not represent a chemical micro-process Temporal process of the reaction cannot be given 44
39 Elementary Reactions Observe the conservation of elements Chemical changes due to collisions of components Transition from educts to products symbolized by arrow Example: Bimolecular elementary reaction Elementary reactions also proceed backwards: Often symbolized by a double arrow: 45
40 Global reactions Conservation of elements Global ratios of amounts of substance Do not take place on atomic scale Global balance of a variety of elementary reactions Equality sign for global reactions Example for global reaction: meaning that 2 mol H 2 react with 1 mol O 2, yielding 2 mol H 2 O 46
41 Global reactions Multiples of the equation are also valid: This does not hold for elementary reactions! Multiplication of the equation of the global reaction by the molar masses Mass balance during combustion Example: Combustion of H 2 using the foregoing equation 47
42 Global reactions Stoichiometric coefficient of reactants i: Stoichiometric coefficient of products i: Example: ν CH4 = 1, ν H2O = 2 Stoichiometric coefficient of a component (only for global reactions): Example: Rate of change for reactants negative Note: Stoichiometric coefficients ν i of reactants are negative! ν i are defined to be positive! 48
43 Global reactions Formulation of global reactions: Combustion of hydrocarbon fuel or an alcohol Atoms in the fuel: Carbon, hydrogen and oxygen Number of atoms in the fuel Stochiometric coefficients of the global reaction are derived from ν B Balances of atoms C: H: O: Example: 49
44 Course Overview Part I: Fundamentals and Laminar Flames Introduction Fundamentals and mass balances of combustion systems Thermodynamics, flame temperature, and equilibrium Governing equations Laminar premixed flames: Kinematics and burning velocity Laminar premixed flames: Flame structure Laminar diffusion flames FlameMaster flame calculator 50 Definitions, Equation of State, Mass Balance Elementary and Global Reactions Coupling Functions Stoichiometry Mixture Fraction Burke-Schumann Solution
45 Coupling functions Example: Global reaction 4 H 2 + 2O 2 = 4H 2 O Consider conversion of 8 moles of H 2 How many moles of O 2 have been converted? Reaction has taken place how many times? In equations: or Change of n i /ν i same for all species 51
46 Coupling functions Global reaction, e.g.: ν 1 F + ν 2 O = ν 3 P Conversion of: n 1 moles of component 1 n i moles of component i Reaction has taken place n 1 /ν 1 or n i /ν i times n 1 /ν 1 = n i /ν i Differential notation: Integrating, e.g. for fuel and oxygen from the unburnt state Coupling function: 52
47 Coupling functions Example H 2 Coupling function: Example: A closed system contains H 2 und O 2. Through combustion reactions, 1 kg H 2 is consumed. What mass of O 2 has been converted? How much air is needed? 1. Multiply coupling function by total mass 2. Determination of For burning 1kg H 2, 8kg 76.7/23.3 = 26.3 kg of air is needed 53
48 Coupling functions Example Gasoline Example: How much CO 2 is formed when burning 1 liter of gasoline? Assumptions: Density of liquid gasoline is roughly 0.75 kg/l Gasoline can be approximated by iso-octane 1. Starting point: One-step global reaction I-C 8 H O 2 = 8 CO H 2 O 2. Coupling function between fuel and CO 2 3. Integrations leads to During combustion of 1liter Gasoline, 2.3 kg CO 2 are produced Under standard conditions, this is roughly 1.3 m 3 CO 2 54
49 Course Overview Part I: Fundamentals and Laminar Flames Introduction Fundamentals and mass balances of combustion systems Thermodynamics, flame temperature, and equilibrium Governing equations Laminar premixed flames: Kinematics and burning velocity Laminar premixed flames: Flame structure Laminar diffusion flames FlameMaster flame calculator 55 Definitions, Equation of State, Mass Balance Elementary and Global Reactions Coupling Functions Stoichiometry Mixture Fraction Burke-Schumann Solution
50 Stoichiometry Stoichiometric: - Fuel-to-oxygen ratio such that both are entirely consumed when combustion to CO 2 and H 2 O is completed For example, - Global reaction describing combustion of a single component hydrocarbon fuel C m H n (subscript F for fuel) - Stoichiometric coefficients are where may be chosen arbitrarily to unity 56
51 Stoichiometric Mass Ratio Mole number ratio for stoichiometric condition or in terms of mass fractions where ν is called the stoichiometric mass ratio Typical values: Methane: ν = 4; N-Dodecane: ν = 3.5 Mass ratio ν Fuel and oxidizer are both consumed when combustion is completed 57
52 Stoichiometric Mass Ratio This is consistent with coupling function, since leads to Complete consumption of fuel and oxygen leads to 58
53 *Extra: Minimum oxygen requirement Minimum oxygen requirement (molar): o min,m Fuel/air mole number ratio before combustion at stoichiometric conditions Ratio of the stoichiometric coefficients Minumum oxygen requirement (mass): o min 59
54 *Extra: Minimum air requirement Minimum air requirement: Mass of air per mass of fuel in complete combustion Relation between minimum oxygen and minimum air requirement: with: Mass fraction Y O2,air = 0,232 Mole fraction X O2,air = 0,21 60
55 The equivalence ratio The equivalence ratio is the ratio of fuel to oxidizer ratio in the unburnt to that of a stoichiometric mixture For combustion with oxygen Can be written also in terms of Fuel to air ratio Mole fractions Stoichiometric mass ratio ν obtained from global reaction 61
56 Course Overview Part I: Fundamentals and Laminar Flames Introduction Fundamentals and mass balances of combustion systems Thermodynamics, flame temperature, and equilibrium Governing equations Laminar premixed flames: Kinematics and burning velocity Laminar premixed flames: Flame structure Laminar diffusion flames FlameMaster flame calculator 62 Definitions, equation of state, mass balance Elementary and Global Reactions Coupling Functions Stoichiometry Mixture Fraction Burke-Schumann Solution
57 The mixture fraction Equivalence ratio important parameter in combustion Mixture fraction quantifies local fuel-air ratio in non-premixed combustion Consider two separate feed streams of Fuel Oxidizer (air, pure oxygen) Streams mix and burn Fuel stream Often consists of one component only In general does not contain oxidizer Oxidizer stream Generally does not contain fuel 63
58 The mixture fraction In the following: Fuel stream: Subscript 1 Oxidizer stream: Subscript 2 Definition mixture fraction Mass fraction of the fuel stream in the mixture: where m 1 and m 2 are the local mass originating from the individual streams Mixture fraction always between zero and one Fuel stream: Z = 1 Oxidizer stream: Z = 0 64
59 The mixture fraction Note: Index B means fuel Mass fraction of fuel in the fuel stream: Y B,1 Mass fraction of oxygen in the oxidizer stream: Y O 2,2 Two-stream system before combustion: Divide for stream 1 by total mass Mixture fraction linear with fuel mass Insert into coupling function: 65
60 The mixture fraction Mixture fraction: For stoichiometric composition: The first two terms in the numerator have to cancel out Stoichiometric mixture fraction: 66
61 Mixture fraction definition by Bilger Consider elements C, H, O in combustion of a C m H n fuel with oxygen or air ν F C m H n + ν O2 O 2 = Products Changes in elements or in terms of element mass fraction Coupling function: β should not change by chemical reactions 67
62 Mixture fraction definition by Bilger Normalizing this such that Z = 1 in the fuel stream and Z = 0 in the oxidizer stream, one obtains Bilger's definition or Because elements are conserved during combustion, element mass fractions calculated from do not change 68
63 Relation of mixture fraction with equivalence ratio Fuel-air equivalence ratio Introducing and into leads with to a unique relation between the equivalence ratio and the mixture fraction 69
64 The equivalence ratio This relation is also valid for multicomponent fuels (see exercise below) It illustrates that the mixture fraction is simply another expression for the local equivalence ratio Exercise: - The element mass fractions of a mixture of hydrocarbons and its mean molecular weight W are assumed to be known - Determine its stoichiometric mixture fraction in air - Hint: 70
65 Course Overview Part I: Fundamentals and Laminar Flames Introduction Fundamentals and mass balances of combustion systems Thermodynamics, flame temperature, and equilibrium Governing equations Laminar premixed flames: Kinematics and burning velocity Laminar premixed flames: Flame structure Laminar diffusion flames FlameMaster flame calculator 71 Definitions, equation of state, mass balance Elementary and Global Reactions Coupling Functions Stoichiometry Mixture Fraction Burke-Schumann Solution
66 Burke-Schumann Solution Diffusion Flame Structure at Complete Conversion Profiles of Y F and Y O in the unburnt gas 2 72
67 Diffusion Flame Structure at Complete Conversion Stoichiometric composition If Z < Z st, fuel is deficient Mixture is fuel lean Combustion terminates when all fuel is consumed: (burnt gas, subscript b) Remaining oxygen mass fraction in the burnt gas is calculated from as 73
68 Diffusion Flame Structure at Complete Conversion If Z > Z st oxygen is deficient Mixture is fuel rich Combustion then terminates when all the oxygen is consumed: leading to 74
69 Diffusion Flame Structure at Complete Conversion For hydrocarbon fuel C m H n, the element mass fractions in the unburnt mixture are For the burnt gas, these are for the hydrocarbon fuel considered above Elements are conserved, hence Z j,u = Z j,b 75
70 Diffusion Flame Structure at Complete Conversion This leads with and for and for to piecewise linear relations of the product mass fractions in terms of Z: where 76
71 Diffusion Flame Structure at Complete Conversion Profiles in the burning mixture Burke-Schumann Solution: 77 Infinitely fast, irreversible chemistry
72 Summary Part I: Fundamentals and Laminar Flames Introduction Fundamentals and mass balances of combustion systems Thermodynamics, flame temperature, and equilibrium Governing equations Laminar premixed flames: Kinematics and burning velocity Laminar premixed flames: Flame structure Laminar diffusion flames FlameMaster flame calculator 78 Definitions, Equation of State, Mass Balance Elementary and Global Reactions Coupling Functions Stoichiometry Mixture Fraction Burke-Schumann Solution
Laminar Premixed Flames: Flame Structure
 Laminar Premixed Flames: Flame Structure Combustion Summer School 2018 Prof. Dr.-Ing. Heinz Pitsch Course Overview Part I: Fundamentals and Laminar Flames Introduction Fundamentals and mass balances of
Laminar Premixed Flames: Flame Structure Combustion Summer School 2018 Prof. Dr.-Ing. Heinz Pitsch Course Overview Part I: Fundamentals and Laminar Flames Introduction Fundamentals and mass balances of
Turbulent Premixed Combustion
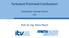 Turbulent Premixed Combustion Combustion Summer School 2018 Prof. Dr.-Ing. Heinz Pitsch Example: LES of a stationary gas turbine velocity field flame 2 Course Overview Part II: Turbulent Combustion Turbulence
Turbulent Premixed Combustion Combustion Summer School 2018 Prof. Dr.-Ing. Heinz Pitsch Example: LES of a stationary gas turbine velocity field flame 2 Course Overview Part II: Turbulent Combustion Turbulence
Combustion. Indian Institute of Science Bangalore
 Combustion Indian Institute of Science Bangalore Combustion Applies to a large variety of natural and artificial processes Source of energy for most of the applications today Involves exothermic chemical
Combustion Indian Institute of Science Bangalore Combustion Applies to a large variety of natural and artificial processes Source of energy for most of the applications today Involves exothermic chemical
Stoichiometry: Chemical Calculations. Chapter 3-4
 Chapters 3-4 Stoichiometry: Chemical Calculations Slide 1 of 48 Molecular Masses And Formula Masses Molecular Masses Molecular mass is the sum of the masses of the atoms represented in a molecular formula.
Chapters 3-4 Stoichiometry: Chemical Calculations Slide 1 of 48 Molecular Masses And Formula Masses Molecular Masses Molecular mass is the sum of the masses of the atoms represented in a molecular formula.
Fundamentals Of Combustion (Part 1) Dr. D.P. Mishra Department of Aerospace Engineering Indian Institute of Technology, Kanpur
 Fundamentals Of Combustion (Part 1) Dr. D.P. Mishra Department of Aerospace Engineering Indian Institute of Technology, Kanpur Lecture 09 Stoichiometric calculations for air-gas mixture Let us start this
Fundamentals Of Combustion (Part 1) Dr. D.P. Mishra Department of Aerospace Engineering Indian Institute of Technology, Kanpur Lecture 09 Stoichiometric calculations for air-gas mixture Let us start this
AAE COMBUSTION AND THERMOCHEMISTRY
 5. COMBUSTIO AD THERMOCHEMISTRY Ch5 1 Overview Definition & mathematical determination of chemical equilibrium, Definition/determination of adiabatic flame temperature, Prediction of composition and temperature
5. COMBUSTIO AD THERMOCHEMISTRY Ch5 1 Overview Definition & mathematical determination of chemical equilibrium, Definition/determination of adiabatic flame temperature, Prediction of composition and temperature
Department of Mechanical Engineering BM 7103 FUELS AND COMBUSTION QUESTION BANK UNIT-1-FUELS
 Department of Mechanical Engineering BM 7103 FUELS AND COMBUSTION QUESTION BANK UNIT-1-FUELS 1. Define the term fuels. 2. What are fossil fuels? Give examples. 3. Define primary fuels. Give examples. 4.
Department of Mechanical Engineering BM 7103 FUELS AND COMBUSTION QUESTION BANK UNIT-1-FUELS 1. Define the term fuels. 2. What are fossil fuels? Give examples. 3. Define primary fuels. Give examples. 4.
Chapter 5. Stoichiometry
 Chapter 5 Stoichiometry Chapter 5 Table of Contents (5-1) Counting by weighing (5-2) Atomic masses (5-3) Learning to solve problems (5-4) The mole (5-5) Molar mass (5-6) Percent composition of compounds
Chapter 5 Stoichiometry Chapter 5 Table of Contents (5-1) Counting by weighing (5-2) Atomic masses (5-3) Learning to solve problems (5-4) The mole (5-5) Molar mass (5-6) Percent composition of compounds
Thermodynamic and Stochiometric Principles in Materials Balance
 Thermodynamic and Stochiometric Principles in Materials Balance Typical metallurgical engineering problems based on materials and energy balance NiO is reduced in an open atmosphere furnace by excess carbon
Thermodynamic and Stochiometric Principles in Materials Balance Typical metallurgical engineering problems based on materials and energy balance NiO is reduced in an open atmosphere furnace by excess carbon
A comparison between two different Flamelet reduced order manifolds for non-premixed turbulent flames
 8 th U. S. National Combustion Meeting Organized by the Western States Section of the Combustion Institute and hosted by the University of Utah May 19-22, 2013 A comparison between two different Flamelet
8 th U. S. National Combustion Meeting Organized by the Western States Section of the Combustion Institute and hosted by the University of Utah May 19-22, 2013 A comparison between two different Flamelet
Quantity Relationships in Chemical Reactions
 Chapter 10 Relationships in Chemical Reactions Section 10.1 Conversion Factors from a Chemical Equation Goal 1 The coefficients in a chemical equation give us the conversion factors to get from the number
Chapter 10 Relationships in Chemical Reactions Section 10.1 Conversion Factors from a Chemical Equation Goal 1 The coefficients in a chemical equation give us the conversion factors to get from the number
Combustion: Flame Theory and Heat Produced. Arthur Anconetani Oscar Castillo Everett Henderson
 Combustion: Flame Theory and Heat Produced Arthur Anconetani Oscar Castillo Everett Henderson What is a Flame?! Reaction Zone! Thermo/Chemical characteristics Types of Flame! Premixed! Diffusion! Both
Combustion: Flame Theory and Heat Produced Arthur Anconetani Oscar Castillo Everett Henderson What is a Flame?! Reaction Zone! Thermo/Chemical characteristics Types of Flame! Premixed! Diffusion! Both
N L N G : C O M B U S T I O N
 N L N G : C O M B U S T I O N G R A N T I N G R A M 6 T H D E C E M B E R 2 0 1 7 This short lecture provides an introduction into the basic principles of combustion and some of the background to the use
N L N G : C O M B U S T I O N G R A N T I N G R A M 6 T H D E C E M B E R 2 0 1 7 This short lecture provides an introduction into the basic principles of combustion and some of the background to the use
Chapter Objectives. Chapter 9 Energy and Chemistry. Chapter Objectives. Energy Use and the World Economy. Energy Use and the World Economy
 Chapter Objectives Larry Brown Tom Holme www.cengage.com/chemistry/brown Chapter 9 Energy and Chemistry Explain the economic importance of conversions between different forms of energy and the inevitability
Chapter Objectives Larry Brown Tom Holme www.cengage.com/chemistry/brown Chapter 9 Energy and Chemistry Explain the economic importance of conversions between different forms of energy and the inevitability
Thermochemistry X.S. Bai Thermochemistry
 Lecture 2 Thermochemistry Design a power plant X.S. Bai Thermochemistry When we study a combustion device, what do we want to know? heat generated power production combustion efficiency combustion control
Lecture 2 Thermochemistry Design a power plant X.S. Bai Thermochemistry When we study a combustion device, what do we want to know? heat generated power production combustion efficiency combustion control
Fluid Dynamics and Balance Equations for Reacting Flows
 Fluid Dynamics and Balance Equations for Reacting Flows Combustion Summer School 2018 Prof. Dr.-Ing. Heinz Pitsch Balance Equations Basics: equations of continuum mechanics balance equations for mass and
Fluid Dynamics and Balance Equations for Reacting Flows Combustion Summer School 2018 Prof. Dr.-Ing. Heinz Pitsch Balance Equations Basics: equations of continuum mechanics balance equations for mass and
Chemical Equations 10/30/13. Types of Chemical Reactions. Types of Chemical Reactions. Types of Chemical Reactions. Types of Chemical Reactions
 Chemical Equations A chemical equation just like a mathematical equation is a way to express, in symbolic form, the reactions occurring in a chemical system. n Balancing chemical equations n Reaction stoichiometry
Chemical Equations A chemical equation just like a mathematical equation is a way to express, in symbolic form, the reactions occurring in a chemical system. n Balancing chemical equations n Reaction stoichiometry
Lecture 11 - Stoichiometry. Lecture 11 - Introduction. Lecture 11 - The Mole. Lecture 11 - The Mole. Lecture 11 - The Mole
 Chem 103, Section F0F Unit IV - Stoichiometry of Formulas and Equations Lecture 11 The concept of a mole, which is a very large group of atoms or molecules Determining the formulas for a compound Stoichiometry
Chem 103, Section F0F Unit IV - Stoichiometry of Formulas and Equations Lecture 11 The concept of a mole, which is a very large group of atoms or molecules Determining the formulas for a compound Stoichiometry
Chapter 5. Chemistry for Changing Times, Chemical Accounting. Lecture Outlines. John Singer, Jackson Community College. Thirteenth Edition
 Chemistry for Changing Times, Thirteenth Edition Lecture Outlines Chemical Accounting John Singer, Jackson Community College Chemical Sentences: Equations Chemical equations represent the sentences in
Chemistry for Changing Times, Thirteenth Edition Lecture Outlines Chemical Accounting John Singer, Jackson Community College Chemical Sentences: Equations Chemical equations represent the sentences in
ADVANCED DES SIMULATIONS OF OXY-GAS BURNER LOCATED INTO MODEL OF REAL MELTING CHAMBER
 ADVANCED DES SIMULATIONS OF OXY-GAS BURNER LOCATED INTO MODEL OF REAL MELTING CHAMBER Ing. Vojtech Betak Ph.D. Aerospace Research and Test Establishment Department of Engines Prague, Czech Republic Abstract
ADVANCED DES SIMULATIONS OF OXY-GAS BURNER LOCATED INTO MODEL OF REAL MELTING CHAMBER Ing. Vojtech Betak Ph.D. Aerospace Research and Test Establishment Department of Engines Prague, Czech Republic Abstract
AP Chemistry Chapter 3. Stoichiometry
 AP Chemistry Chapter 3 Stoichiometry Stoichiometry Is the study of the quantities of substances consumed and produced in chemical reactions Derived from the Greek words stoicheion meaning element and metron
AP Chemistry Chapter 3 Stoichiometry Stoichiometry Is the study of the quantities of substances consumed and produced in chemical reactions Derived from the Greek words stoicheion meaning element and metron
Usual Atomic Charges of Main Group Elements
 Usual Atomic Charges of Main Group Elements +1 +2 +3 +4 +5 +6 +7-5 -4-3 -2-1 Examples SO 3 sulfur trioxide CO 2 carbon dioxide Al 2 O 3 aluminum trioxide IF 7 iodine heptafluoride Fig. 2-6, p.63 Chemical
Usual Atomic Charges of Main Group Elements +1 +2 +3 +4 +5 +6 +7-5 -4-3 -2-1 Examples SO 3 sulfur trioxide CO 2 carbon dioxide Al 2 O 3 aluminum trioxide IF 7 iodine heptafluoride Fig. 2-6, p.63 Chemical
Environmental chemistry
 Page 1 of 5 Environmental chemistry Almost every pollution problem that we face has a chemical basis. Even the qualitative descriptions of such problems as the greenhouse effect, ozone depletion, toxic
Page 1 of 5 Environmental chemistry Almost every pollution problem that we face has a chemical basis. Even the qualitative descriptions of such problems as the greenhouse effect, ozone depletion, toxic
Asymptotic Structure of Rich Methane-Air Flames
 Asymptotic Structure of Rich Methane-Air Flames K. SESHADRI* Center for Energy and Combustion Research, Department of Mechanical and Aerospace Engineering, University of California at San Diego, La Jolla,
Asymptotic Structure of Rich Methane-Air Flames K. SESHADRI* Center for Energy and Combustion Research, Department of Mechanical and Aerospace Engineering, University of California at San Diego, La Jolla,
Chapter 3: STOICHIOMETRY: MASS, FORMULAS, AND REACTIONS
 Chapter 3: STOICHIOMETRY: MASS, FORMULAS, AND REACTIONS Problems: 3.1-3.8, 3.11, 3.14-3.90, 3.103-3.120, 3.122-3.125, 3.128-3.131, 3.134, 3.137-36.138, 3.140-3.142 3.2 THE MOLE Stoichiometry (STOY-key-OM-e-tree):
Chapter 3: STOICHIOMETRY: MASS, FORMULAS, AND REACTIONS Problems: 3.1-3.8, 3.11, 3.14-3.90, 3.103-3.120, 3.122-3.125, 3.128-3.131, 3.134, 3.137-36.138, 3.140-3.142 3.2 THE MOLE Stoichiometry (STOY-key-OM-e-tree):
Dr Ali Jawarneh Department of Mechanical Engineering Hashemite University
 Chapter 15 CHEMICAL REACTIONS Dr Ali Jawarneh Department of Mechanical Engineering Hashemite University 2 Objectives Give an overview of fuels and combustion. Apply the conservation of mass to reacting
Chapter 15 CHEMICAL REACTIONS Dr Ali Jawarneh Department of Mechanical Engineering Hashemite University 2 Objectives Give an overview of fuels and combustion. Apply the conservation of mass to reacting
MOLE CONCEPT AND STOICHIOMETRY
 MOLE CONCEPT AND STOICHIOMETRY Dear Reader You have studied about the term 'mole' in your previous class. It is defined as the amount of a substance containing as many constituting particles (atoms, molecules
MOLE CONCEPT AND STOICHIOMETRY Dear Reader You have studied about the term 'mole' in your previous class. It is defined as the amount of a substance containing as many constituting particles (atoms, molecules
CHEMICAL FORMULA COEFFICIENTS AND SUBSCRIPTS 3O 2 2O 3. ! Formula that gives the TOTAL number of elements in a molecule or formula unit.
 CHEMICAL FORMULA! Formula that gives the TOTAL number of elements in a molecule or formula unit. No Score from Exam 1? Go to 210 Whitmore and speak with Mike Joyce to get it straightened out. Which Skill
CHEMICAL FORMULA! Formula that gives the TOTAL number of elements in a molecule or formula unit. No Score from Exam 1? Go to 210 Whitmore and speak with Mike Joyce to get it straightened out. Which Skill
Lecture 6 Asymptotic Structure for Four-Step Premixed Stoichiometric Methane Flames
 Lecture 6 Asymptotic Structure for Four-Step Premixed Stoichiometric Methane Flames 6.-1 Previous lecture: Asymptotic description of premixed flames based on an assumed one-step reaction. basic understanding
Lecture 6 Asymptotic Structure for Four-Step Premixed Stoichiometric Methane Flames 6.-1 Previous lecture: Asymptotic description of premixed flames based on an assumed one-step reaction. basic understanding
Chemical Reactions. Chapter 17
 Chemical Reactions Chapter 17 Chemical Equations C+O 2 CO 2 C (s) +O 2 (g) CO 2 (g) Reactants on left, products on right Each are balanced because same number of atoms of reactants as products Some equations
Chemical Reactions Chapter 17 Chemical Equations C+O 2 CO 2 C (s) +O 2 (g) CO 2 (g) Reactants on left, products on right Each are balanced because same number of atoms of reactants as products Some equations
Chapter 3. Stoichiometry: Calculations with Chemical Formulas and Equations
 Chapter 3. Stoichiometry: Calculations with Chemical Formulas and Equations Lecture Outline 3.1 Chemical Equations The quantitative nature of chemical formulas and reactions is called stoichiometry. Lavoisier
Chapter 3. Stoichiometry: Calculations with Chemical Formulas and Equations Lecture Outline 3.1 Chemical Equations The quantitative nature of chemical formulas and reactions is called stoichiometry. Lavoisier
The following gas laws describes an ideal gas, where
 Alief ISD Chemistry STAAR Review Reporting Category 4: Gases and Thermochemistry C.9.A Describe and calculate the relations between volume, pressure, number of moles, and temperature for an ideal gas as
Alief ISD Chemistry STAAR Review Reporting Category 4: Gases and Thermochemistry C.9.A Describe and calculate the relations between volume, pressure, number of moles, and temperature for an ideal gas as
AE 205 Materials and Energy Balances Asst. Prof. Dr. Tippabust Eksangsri. Chapter 4 Stoichiometry and MB with Reactions
 AE 205 Materials and Energy Balances Asst. Prof. Dr. Tippabust Eksangsri Chapter 4 Stoichiometry and MB with Reactions Stoichiometry Stoichiometry provides a quantitative means of relating the amount of
AE 205 Materials and Energy Balances Asst. Prof. Dr. Tippabust Eksangsri Chapter 4 Stoichiometry and MB with Reactions Stoichiometry Stoichiometry provides a quantitative means of relating the amount of
First Law of Thermodynamics: energy cannot be created or destroyed.
 1 CHEMICAL THERMODYNAMICS ANSWERS energy = anything that has the capacity to do work work = force acting over a distance Energy (E) = Work = Force x Distance First Law of Thermodynamics: energy cannot
1 CHEMICAL THERMODYNAMICS ANSWERS energy = anything that has the capacity to do work work = force acting over a distance Energy (E) = Work = Force x Distance First Law of Thermodynamics: energy cannot
COMBUSTION OF FUEL 12:57:42
 COMBUSTION OF FUEL The burning of fuel in presence of air is known as combustion. It is a chemical reaction taking place between fuel and oxygen at temperature above ignition temperature. Heat is released
COMBUSTION OF FUEL The burning of fuel in presence of air is known as combustion. It is a chemical reaction taking place between fuel and oxygen at temperature above ignition temperature. Heat is released
9/14/ Chemistry Second Edition Julia Burdge. Stoichiometry: Ratios of Combination. Molecular and Formula Masses
 9/14/1 Chemistry Second Edition Julia Burdge Stoichiometry: Ratios of Combination Copyright (c) The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Stoichiometry: Ratios
9/14/1 Chemistry Second Edition Julia Burdge Stoichiometry: Ratios of Combination Copyright (c) The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Stoichiometry: Ratios
Fundamentals of Combustion
 Fundamentals of Combustion Lec 3: Chemical Thermodynamics Dr. Zayed Al-Hamamre Content Process Heat Transfer 1-3 Process Heat Transfer 1-4 Process Heat Transfer 1-5 Theoretical and Excess Air Combustion
Fundamentals of Combustion Lec 3: Chemical Thermodynamics Dr. Zayed Al-Hamamre Content Process Heat Transfer 1-3 Process Heat Transfer 1-4 Process Heat Transfer 1-5 Theoretical and Excess Air Combustion
Mass Relationships in Chemical Reactions
 Mass Relationships in Chemical Reactions Chapter 3 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Micro World atoms & molecules Macro World grams Atomic mass
Mass Relationships in Chemical Reactions Chapter 3 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Micro World atoms & molecules Macro World grams Atomic mass
SUPPLEMENTARY TOPIC 3 ENERGY AND CHEMICAL REACTIONS
 SUPPLEMENTARY TOPIC 3 ENERGY AND CHEMICAL REACTIONS Rearranging atoms. In a chemical reaction, bonds between atoms in one or more molecules (reactants) break and new bonds are formed with other atoms to
SUPPLEMENTARY TOPIC 3 ENERGY AND CHEMICAL REACTIONS Rearranging atoms. In a chemical reaction, bonds between atoms in one or more molecules (reactants) break and new bonds are formed with other atoms to
AP Chemistry A. Allan Chapter Six Notes - Thermochemistry
 AP Chemistry A. Allan Chapter Six Notes - Thermochemistry 6.1 The Nature of Energy A. Definition 1. Energy is the capacity to do work (or to produce heat*) a. Work is a force acting over a distance (moving
AP Chemistry A. Allan Chapter Six Notes - Thermochemistry 6.1 The Nature of Energy A. Definition 1. Energy is the capacity to do work (or to produce heat*) a. Work is a force acting over a distance (moving
L = 6.02 x mol Determine the number of particles and the amount of substance (in moles)
 1.1 The Mole 1.1.1 - Apply the mole concept to substances A mole is the name given to a certain quantity. It represents 6.02 x 10 23 particles. This number is also known as Avogadro's constant, symbolised
1.1 The Mole 1.1.1 - Apply the mole concept to substances A mole is the name given to a certain quantity. It represents 6.02 x 10 23 particles. This number is also known as Avogadro's constant, symbolised
Balancing chemical reaction equations (stoichiometry)
 Balancing chemical reaction equations (stoichiometry) This worksheet and all related files are licensed under the Creative Commons Attribution License, version 1.0. To view a copy of this license, visit
Balancing chemical reaction equations (stoichiometry) This worksheet and all related files are licensed under the Creative Commons Attribution License, version 1.0. To view a copy of this license, visit
Chapter 6 Thermochemistry 許富銀
 Chapter 6 Thermochemistry 許富銀 6.1 Chemical Hand Warmers Thermochemistry: the study of the relationships between chemistry and energy Hand warmers use the oxidation of iron as the exothermic reaction: Nature
Chapter 6 Thermochemistry 許富銀 6.1 Chemical Hand Warmers Thermochemistry: the study of the relationships between chemistry and energy Hand warmers use the oxidation of iron as the exothermic reaction: Nature
Chemistry. Chapter 17
 Chemistry Chapter 17 Chemical Equations C+O 2 CO 2 C (s) +O 2 (g) CO 2 (g) Reactants on left, products on right Each are balanced because same number of atoms of reactants as products Balancing Chemical
Chemistry Chapter 17 Chemical Equations C+O 2 CO 2 C (s) +O 2 (g) CO 2 (g) Reactants on left, products on right Each are balanced because same number of atoms of reactants as products Balancing Chemical
Lecture 8 Laminar Diffusion Flames: Diffusion Flamelet Theory
 Lecture 8 Laminar Diffusion Flames: Diffusion Flamelet Theory 8.-1 Systems, where fuel and oxidizer enter separately into the combustion chamber. Mixing takes place by convection and diffusion. Only where
Lecture 8 Laminar Diffusion Flames: Diffusion Flamelet Theory 8.-1 Systems, where fuel and oxidizer enter separately into the combustion chamber. Mixing takes place by convection and diffusion. Only where
Stoichiometry. Please take out your notebooks
 Stoichiometry Please take out your notebooks Stoichiometry stochio = Greek for element metry = measurement Stoichiometry is about measuring the amounts of elements and compounds involved in a reaction.
Stoichiometry Please take out your notebooks Stoichiometry stochio = Greek for element metry = measurement Stoichiometry is about measuring the amounts of elements and compounds involved in a reaction.
Name Date Class STUDY GUIDE FOR CONTENT MASTERY
 Stoichiometry Section 12.1 What is stoichiometry? In your textbook, read about stoichiometry and the balanced equation. For each statement below, write true or false. 1. The study of the quantitative relationships
Stoichiometry Section 12.1 What is stoichiometry? In your textbook, read about stoichiometry and the balanced equation. For each statement below, write true or false. 1. The study of the quantitative relationships
Reacting Gas Mixtures
 Reacting Gas Mixtures Reading Problems 15-1 15-7 15-21, 15-32, 15-51, 15-61, 15-74 15-83, 15-91, 15-93, 15-98 Introduction thermodynamic analysis of reactive mixtures is primarily an extension of the principles
Reacting Gas Mixtures Reading Problems 15-1 15-7 15-21, 15-32, 15-51, 15-61, 15-74 15-83, 15-91, 15-93, 15-98 Introduction thermodynamic analysis of reactive mixtures is primarily an extension of the principles
Copyrighted by Gabriel Tang B.Ed., B.Sc.
 Unit 5: Chemical Equations and Reactions & Stoichiometry Chemistry Chapter 9: Stoichiometry 9.1: Calculating Quantities in Reactions Avogadro s Number: - a group of (6.0 10 ) molecules = 1 mole Stoichiometry:
Unit 5: Chemical Equations and Reactions & Stoichiometry Chemistry Chapter 9: Stoichiometry 9.1: Calculating Quantities in Reactions Avogadro s Number: - a group of (6.0 10 ) molecules = 1 mole Stoichiometry:
TOPICAL PROBLEMS OF FLUID MECHANICS 97
 TOPICAL PROBLEMS OF FLUID MECHANICS 97 DOI: http://dx.doi.org/10.14311/tpfm.2016.014 DESIGN OF COMBUSTION CHAMBER FOR FLAME FRONT VISUALISATION AND FIRST NUMERICAL SIMULATION J. Kouba, J. Novotný, J. Nožička
TOPICAL PROBLEMS OF FLUID MECHANICS 97 DOI: http://dx.doi.org/10.14311/tpfm.2016.014 DESIGN OF COMBUSTION CHAMBER FOR FLAME FRONT VISUALISATION AND FIRST NUMERICAL SIMULATION J. Kouba, J. Novotný, J. Nožička
Asymptotic Analysis of the Structure of Moderately Rich Methane-Air Flames
 Asymptotic Analysis of the Structure of Moderately Rich Methane-Air Flames K. SESHADRI,* X. S. BAI,** H. PITSCH, and N. PETERS Institut für Technische Mechanik, RWTH Aachen, D-52056 Aachen, Federal Republic
Asymptotic Analysis of the Structure of Moderately Rich Methane-Air Flames K. SESHADRI,* X. S. BAI,** H. PITSCH, and N. PETERS Institut für Technische Mechanik, RWTH Aachen, D-52056 Aachen, Federal Republic
Stoichiometry Rockets
 Stoichiometry Rockets The objective of this lab is to to: calculate the needed volume of fuel to react with a given volume of gas and result in a productive explosion determine the heat of the reaction
Stoichiometry Rockets The objective of this lab is to to: calculate the needed volume of fuel to react with a given volume of gas and result in a productive explosion determine the heat of the reaction
Mole: base unit for an amount of substance A mole contains Avogadro s number (N A ) of particles (atoms, molecules, ions, formula units )
 Mole: base unit for an amount of substance A mole contains Avogadro s number (N A ) of particles (atoms, molecules, ions, formula units ) N A 6.0 10 mol -1 1 mol substance contains N A Molar mass (g/mol)
Mole: base unit for an amount of substance A mole contains Avogadro s number (N A ) of particles (atoms, molecules, ions, formula units ) N A 6.0 10 mol -1 1 mol substance contains N A Molar mass (g/mol)
Thermochemistry. Using Heats of Reaction - Hess s Law - Standard Enthalpies of Formation - Fuels Foods, Commercial Fuels, and Rocket Fuels
 Thermochemistry Understanding Heats of Reaction - Energy and Its Units - Heat of Reaction - Enthalpy and Enthalpy Change - Thermochemical Equations - Applying Stoichiometry to Heats of Reaction - Measuring
Thermochemistry Understanding Heats of Reaction - Energy and Its Units - Heat of Reaction - Enthalpy and Enthalpy Change - Thermochemical Equations - Applying Stoichiometry to Heats of Reaction - Measuring
Mass Relationships in Chemical Reactions
 Mass Relationships in Chemical Reactions Chapter 3 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Micro World atoms & molecules Macro World grams Atomic mass
Mass Relationships in Chemical Reactions Chapter 3 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Micro World atoms & molecules Macro World grams Atomic mass
Chapter 1 Introduction and Basic Concepts
 Chapter 1 Introduction and Basic Concepts 1-1 Thermodynamics and Energy Application Areas of Thermodynamics 1-2 Importance of Dimensions and Units Some SI and English Units Dimensional Homogeneity Unity
Chapter 1 Introduction and Basic Concepts 1-1 Thermodynamics and Energy Application Areas of Thermodynamics 1-2 Importance of Dimensions and Units Some SI and English Units Dimensional Homogeneity Unity
Mass Relationships in Chemical Reactions
 Mass Relationships in Chemical Reactions Chapter 3 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Micro World atoms & molecules Macro World grams Atomic mass
Mass Relationships in Chemical Reactions Chapter 3 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Micro World atoms & molecules Macro World grams Atomic mass
Chapter 3. Mass Relationships in Chemical Reactions
 Chapter 3 Mass Relationships in Chemical Reactions In this chapter, Chemical structure and formulas in studying the mass relationships of atoms and molecules. To explain the composition of compounds and
Chapter 3 Mass Relationships in Chemical Reactions In this chapter, Chemical structure and formulas in studying the mass relationships of atoms and molecules. To explain the composition of compounds and
3 Stoichiometry: Calculations with Chemical Formulas and Equations
 3 Stoichiometry: Calculations with Chemical Formulas and Equations 3.1 Chemical Equations Balance chemical equations. 3. Simple Patterns of Reactivity Predict products of a chemical reaction in a combination
3 Stoichiometry: Calculations with Chemical Formulas and Equations 3.1 Chemical Equations Balance chemical equations. 3. Simple Patterns of Reactivity Predict products of a chemical reaction in a combination
Chapter 3 Calculations with Chemical Formulas and Equations
 Chapter 3 Calculations with Chemical Formulas and Equations Contents and Concepts Mass and Moles of Substances Here we will establish a critical relationship between the mass of a chemical substance and
Chapter 3 Calculations with Chemical Formulas and Equations Contents and Concepts Mass and Moles of Substances Here we will establish a critical relationship between the mass of a chemical substance and
Lecture Presentation. Chapter 6. Thermochemistry. Sherril Soman Grand Valley State University Pearson Education, Inc.
 Lecture Presentation Chapter 6 Thermochemistry Sherril Soman Grand Valley State University Chemical Hand Warmers Most hand warmers work by using the heat released from the slow oxidation of iron 4 Fe(s)
Lecture Presentation Chapter 6 Thermochemistry Sherril Soman Grand Valley State University Chemical Hand Warmers Most hand warmers work by using the heat released from the slow oxidation of iron 4 Fe(s)
Gestão de Sistemas Energéticos 2017/2018
 Gestão de Sistemas Energéticos 2017/2018 Exergy Analysis Prof. Tânia Sousa taniasousa@tecnico.ulisboa.pt Conceptualizing Chemical Exergy C a H b O c enters the control volume at T 0, p 0. O 2 and CO 2,
Gestão de Sistemas Energéticos 2017/2018 Exergy Analysis Prof. Tânia Sousa taniasousa@tecnico.ulisboa.pt Conceptualizing Chemical Exergy C a H b O c enters the control volume at T 0, p 0. O 2 and CO 2,
4.3 Reaction Stoichiometry
 198 Chapter 4 Stoichiometry of Chemical Reactions 4.3 Reaction Stoichiometry By the end of this section, you will be able to: Explain the concept of stoichiometry as it pertains to chemical reactions Use
198 Chapter 4 Stoichiometry of Chemical Reactions 4.3 Reaction Stoichiometry By the end of this section, you will be able to: Explain the concept of stoichiometry as it pertains to chemical reactions Use
Chapter 4. Chemical Quantities and Aqueous Reactions
 Chapter 4 Chemical Quantities and Aqueous Reactions Stoichiometry The study of the numerical relationship between chemical quantities in a chemical reaction Making Pizza The number of pizzas you can make
Chapter 4 Chemical Quantities and Aqueous Reactions Stoichiometry The study of the numerical relationship between chemical quantities in a chemical reaction Making Pizza The number of pizzas you can make
Outcomes: Interpret a balanced chemical equation in terms of moles, mass and volume of gases. Solve stoichiometric problems involving: moles, mass,
 Stoichiometry Outcomes: Interpret a balanced chemical equation in terms of moles, mass and volume of gases. Solve stoichiometric problems involving: moles, mass, volume, and heat of reaction. Stoichiometry
Stoichiometry Outcomes: Interpret a balanced chemical equation in terms of moles, mass and volume of gases. Solve stoichiometric problems involving: moles, mass, volume, and heat of reaction. Stoichiometry
05/04/2011 Tarik Al-Shemmeri 2
 05/04/2011 Tarik Al-Shemmeri 1 05/04/2011 Tarik Al-Shemmeri 2 05/04/2011 Tarik Al-Shemmeri 3 05/04/2011 Tarik Al-Shemmeri 4 05/04/2011 Tarik Al-Shemmeri 5 05/04/2011 Tarik Al-Shemmeri 6 05/04/2011 Tarik
05/04/2011 Tarik Al-Shemmeri 1 05/04/2011 Tarik Al-Shemmeri 2 05/04/2011 Tarik Al-Shemmeri 3 05/04/2011 Tarik Al-Shemmeri 4 05/04/2011 Tarik Al-Shemmeri 5 05/04/2011 Tarik Al-Shemmeri 6 05/04/2011 Tarik
Chemistry 101 Chapter 10 Energy
 Chemistry 101 Chapter 10 Energy Energy: the ability to do work or produce heat. Kinetic energy (KE): is the energy of motion. Any object that is moving has kinetic energy. Several forms of kinetic energy
Chemistry 101 Chapter 10 Energy Energy: the ability to do work or produce heat. Kinetic energy (KE): is the energy of motion. Any object that is moving has kinetic energy. Several forms of kinetic energy
Chapter 3. Stoichiometry: Calculations with Chemical Formulas and Equations
 Chapter 3 Stoichiometry: Calculations with Chemical Formulas and Equations Matter Matter is anything that has mass and takes up space 2 Composition of Matter Atom number of protons = atomic number (Z)
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and Equations Matter Matter is anything that has mass and takes up space 2 Composition of Matter Atom number of protons = atomic number (Z)
Chapter 6: Thermochemistry
 Chem 1045 General Chemistry by Ebbing and Gammon, 8th Edition George W.J. Kenney, Jr Last Update: 24-Oct-2008 Chapter 6: Thermochemistry These Notes are to SUPPLIMENT the Text, They do NOT Replace reading
Chem 1045 General Chemistry by Ebbing and Gammon, 8th Edition George W.J. Kenney, Jr Last Update: 24-Oct-2008 Chapter 6: Thermochemistry These Notes are to SUPPLIMENT the Text, They do NOT Replace reading
association adilca FUEL COMBUSTION
 FUEL COMBUSTION Where does the energy that drives the motors come from? What are hydrocarbons made of? Why does it need air to burn fuel? What is the amount of energy released by combustion? How to calculate
FUEL COMBUSTION Where does the energy that drives the motors come from? What are hydrocarbons made of? Why does it need air to burn fuel? What is the amount of energy released by combustion? How to calculate
ASSORTED STOICHIOMETRY PRACTICE PROBLEMS PART I
 ASSORTED STOICHIOMETRY PRACTICE PROBLEMS PART I (1) A 1.2156-gram sample of a mixture of CaCO 3 and Na 2 SO 4 was analyzed by dissolving the sample and completely precipitating the Ca 2+ as CaC 2 O 4.
ASSORTED STOICHIOMETRY PRACTICE PROBLEMS PART I (1) A 1.2156-gram sample of a mixture of CaCO 3 and Na 2 SO 4 was analyzed by dissolving the sample and completely precipitating the Ca 2+ as CaC 2 O 4.
3.2 Alkanes. Refining crude oil. N Goalby chemrevise.org 40 C 110 C 180 C. 250 C fuel oil 300 C 340 C. Fractional Distillation: Industrially
 3.2 Alkanes Refining crude oil Fractional Distillation: Industrially Petroleum is a mixture consisting mainly of alkane hydrocarbons Petroleum fraction: mixture of hydrocarbons with a similar chain length
3.2 Alkanes Refining crude oil Fractional Distillation: Industrially Petroleum is a mixture consisting mainly of alkane hydrocarbons Petroleum fraction: mixture of hydrocarbons with a similar chain length
Most hand warmers work by using the heat released from the slow oxidation of iron: The amount your hand temperature rises depends on several factors:
 Lecture Presentation Chapter 6 Thermochemistry Chemical Hand Warmers Most hand warmers work by using the heat released from the slow oxidation of iron: Exothermic reaction 4 Fe(s) + 3 O 2 (g) 2 Fe 2 O
Lecture Presentation Chapter 6 Thermochemistry Chemical Hand Warmers Most hand warmers work by using the heat released from the slow oxidation of iron: Exothermic reaction 4 Fe(s) + 3 O 2 (g) 2 Fe 2 O
Understanding Equations
 Chemical Reactions Chemical reaction: a process of chemically changing both the physical and chemical properties of a substance to a new substance with different physical and chemical properties. Video
Chemical Reactions Chemical reaction: a process of chemically changing both the physical and chemical properties of a substance to a new substance with different physical and chemical properties. Video
Balancing Chemical Reactions. CHAPTER 3: Quantitative Relationships in Chemical Reactions. Zn + HCl ZnCl 2 + H 2. reactant atoms product atoms
 CHAPTER 3: Quantitative Relationships in Chemical Reactions Stoichiometry: Greek for measure elements Stoichiometry involves calculations based on chemical formulas and chemical equations (reactions) quantitative.
CHAPTER 3: Quantitative Relationships in Chemical Reactions Stoichiometry: Greek for measure elements Stoichiometry involves calculations based on chemical formulas and chemical equations (reactions) quantitative.
Chapter 6 Thermochemistry
 Chapter 6 Thermochemistry Contents and Concepts Understanding Heats of Reaction The first part of the chapter lays the groundwork for understanding what we mean by heats of reaction. 1. Energy and Its
Chapter 6 Thermochemistry Contents and Concepts Understanding Heats of Reaction The first part of the chapter lays the groundwork for understanding what we mean by heats of reaction. 1. Energy and Its
Chapter 15. In the preceding chapters we limited our consideration to CHEMICAL REACTIONS. Objectives
 Chapter 15 CHEMICAL REACTIONS In the preceding chapters we limited our consideration to nonreacting systems systems whose chemical composition remains unchanged during a process. This was the case even
Chapter 15 CHEMICAL REACTIONS In the preceding chapters we limited our consideration to nonreacting systems systems whose chemical composition remains unchanged during a process. This was the case even
Fuel, Air, and Combustion Thermodynamics
 Chapter 3 Fuel, Air, and Combustion Thermodynamics 3.1) What is the molecular weight, enthalpy (kj/kg), and entropy (kj/kg K) of a gas mixture at P = 1000 kpa and T = 500 K, if the mixture contains the
Chapter 3 Fuel, Air, and Combustion Thermodynamics 3.1) What is the molecular weight, enthalpy (kj/kg), and entropy (kj/kg K) of a gas mixture at P = 1000 kpa and T = 500 K, if the mixture contains the
Chapter 12 Stoichiometry
 12.2 Chemical Calculations > Chapter 12 Stoichiometry 12.1 The Arithmetic of Equations 12.22 Chemical Calculations 12.3 Limiting Reagent and Percent Yield 1 Copyright Pearson Education, Inc., or its affiliates.
12.2 Chemical Calculations > Chapter 12 Stoichiometry 12.1 The Arithmetic of Equations 12.22 Chemical Calculations 12.3 Limiting Reagent and Percent Yield 1 Copyright Pearson Education, Inc., or its affiliates.
Chemistry 40S Chemical Kinetics (This unit has been adapted from
 Chemistry 40S Chemical Kinetics (This unit has been adapted from https://bblearn.merlin.mb.ca) Name: 1 2 Lesson 1: Introduction to Kinetics Goals: Identify variables used to monitor reaction rate. Formulate
Chemistry 40S Chemical Kinetics (This unit has been adapted from https://bblearn.merlin.mb.ca) Name: 1 2 Lesson 1: Introduction to Kinetics Goals: Identify variables used to monitor reaction rate. Formulate
Mass Relationships in Chemical Reactions
 Mass Relationships in Chemical Reactions Chapter 3 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Micro World atoms & molecules Macro World grams Atomic mass
Mass Relationships in Chemical Reactions Chapter 3 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Micro World atoms & molecules Macro World grams Atomic mass
Class XI Chapter 1 Some Basic Concepts of Chemistry Chemistry
 Question 1.1: Calculate the molecular mass of the following: (i) H 2 O (ii) CO 2 (iii) CH 4 (i) H 2 O: The molecular mass of water, H 2 O = (2 Atomic mass of hydrogen) + (1 Atomic mass of oxygen) = [2(1.0084)
Question 1.1: Calculate the molecular mass of the following: (i) H 2 O (ii) CO 2 (iii) CH 4 (i) H 2 O: The molecular mass of water, H 2 O = (2 Atomic mass of hydrogen) + (1 Atomic mass of oxygen) = [2(1.0084)
Chapter 8: Quantities in Chemical Reactions
 C h e m i s t r y 1 2 C h 8 : Q u a n t i t i e s i n C h e m i c a l R e a c t i o n s P a g e 1 Chapter 8: Quantities in Chemical Reactions Read Chapter 8 and Check MasteringChemistry due dates. Stoichiometry:
C h e m i s t r y 1 2 C h 8 : Q u a n t i t i e s i n C h e m i c a l R e a c t i o n s P a g e 1 Chapter 8: Quantities in Chemical Reactions Read Chapter 8 and Check MasteringChemistry due dates. Stoichiometry:
Reactions and Reactors
 Reactions and Reactors ChE 400 - Reactive Process Engineering If we want to run a chemical process in order to convert some reactants (the reactor feed) to some product (the reactor effluent), we have
Reactions and Reactors ChE 400 - Reactive Process Engineering If we want to run a chemical process in order to convert some reactants (the reactor feed) to some product (the reactor effluent), we have
Best Practice Guidelines for Combustion Modeling. Raphael David A. Bacchi, ESSS
 Best Practice Guidelines for Combustion Modeling Raphael David A. Bacchi, ESSS PRESENTATION TOPICS Introduction; Combustion Phenomenology; Combustion Modeling; Reaction Mechanism; Radiation; Case Studies;
Best Practice Guidelines for Combustion Modeling Raphael David A. Bacchi, ESSS PRESENTATION TOPICS Introduction; Combustion Phenomenology; Combustion Modeling; Reaction Mechanism; Radiation; Case Studies;
Gas Volumes and the Ideal Gas Law
 Section 3, 9B s Gases react in whole-number ratios. Equal volumes of gases under the same conditions contain equal numbers of molecules. All gases have a volume of 22.4 L under standard conditions. In
Section 3, 9B s Gases react in whole-number ratios. Equal volumes of gases under the same conditions contain equal numbers of molecules. All gases have a volume of 22.4 L under standard conditions. In
Name AP Chemistry / / Chapter 5 Collected AP Exam Free Response Questions Answers
 Name AP Chemistry / / Chapter 5 Collected AP Exam Free Response Questions 1980 2010 - Answers 1982 - #5 (a) From the standpoint of the kinetic-molecular theory, discuss briefly the properties of gas molecules
Name AP Chemistry / / Chapter 5 Collected AP Exam Free Response Questions 1980 2010 - Answers 1982 - #5 (a) From the standpoint of the kinetic-molecular theory, discuss briefly the properties of gas molecules
11 Stoichiometry. Section 11.1 What is stoichiometry?
 11 Stoichiometry Section 11.1 What is stoichiometry? In your textbook, read about stoichiometry and the balanced equation. For each statement below, write true or false. 1.. 3. 4. 5. The study of the quantitative
11 Stoichiometry Section 11.1 What is stoichiometry? In your textbook, read about stoichiometry and the balanced equation. For each statement below, write true or false. 1.. 3. 4. 5. The study of the quantitative
TECHNICAL SCIENCE DAS12703 ROZAINITA BT. ROSLEY PUSAT PENGAJIAN DIPLOMA UNVERSITI TUN HUSSEIN ONN MALAYSIA
 TECHNICAL SCIENCE DAS12703 ROZAINITA BT. ROSLEY PUSAT PENGAJIAN DIPLOMA UNVERSITI TUN HUSSEIN ONN MALAYSIA ii TABLE OF CONTENTS TABLE OF CONTENTS... i LIST OF FIGURES... iii Chapter 1... 4 SOLUTIONS...
TECHNICAL SCIENCE DAS12703 ROZAINITA BT. ROSLEY PUSAT PENGAJIAN DIPLOMA UNVERSITI TUN HUSSEIN ONN MALAYSIA ii TABLE OF CONTENTS TABLE OF CONTENTS... i LIST OF FIGURES... iii Chapter 1... 4 SOLUTIONS...
Chemistry B11 Chapter 5 Chemical reactions
 Chapter 5 Chemical reactions Chemical reactions are classified into five groups: A + B AB Synthesis reactions (Combination) H + O H O AB A + B Decomposition reactions (Analysis) NaCl Na +Cl A + BC AC +
Chapter 5 Chemical reactions Chemical reactions are classified into five groups: A + B AB Synthesis reactions (Combination) H + O H O AB A + B Decomposition reactions (Analysis) NaCl Na +Cl A + BC AC +
Fundamentals Of Combustion (Part 1) Dr. D.P. Mishra Department of Aerospace Engineering Indian Institute of Technology, Kanpur
 Fundamentals Of Combustion (Part 1) Dr. D.P. Mishra Department of Aerospace Engineering Indian Institute of Technology, Kanpur Lecture 10 Mixture fraction calculation for diffusion flames Let us start
Fundamentals Of Combustion (Part 1) Dr. D.P. Mishra Department of Aerospace Engineering Indian Institute of Technology, Kanpur Lecture 10 Mixture fraction calculation for diffusion flames Let us start
Chapter 3. Stoichiometry:
 Chapter 3. Stoichiometry: Watch Bozeman Videos & other videos on my website for additional help: Big Idea 1: Chemical Analysis Conservation of Atoms Balancing Equations Symbolic Representation Mole Big
Chapter 3. Stoichiometry: Watch Bozeman Videos & other videos on my website for additional help: Big Idea 1: Chemical Analysis Conservation of Atoms Balancing Equations Symbolic Representation Mole Big
Lecture Outline. 5.1 The Nature of Energy. Kinetic Energy and Potential Energy. 1 mv
 Chapter 5. Thermochemistry Common Student Misconceptions Students confuse power and energy. Students confuse heat with temperature. Students fail to note that the first law of thermodynamics is the law
Chapter 5. Thermochemistry Common Student Misconceptions Students confuse power and energy. Students confuse heat with temperature. Students fail to note that the first law of thermodynamics is the law
Introduction to laser-based combustion diagnostics
 Introduction to laser-based combustion diagnostics (Lecture 1b) Lecture prepared for course in laser-based combustion diagnostics by Per-Erik Bengtsson and Joakim Bood Division of Combustion Physics at
Introduction to laser-based combustion diagnostics (Lecture 1b) Lecture prepared for course in laser-based combustion diagnostics by Per-Erik Bengtsson and Joakim Bood Division of Combustion Physics at
Mass Relationships in Chemical Reactions
 Reading Assignments: Chapter 3 in R. Chang, Chemistry, 8th Ed., McGraw-Hill, 2005 Mass Relationships in Chemical Reactions Or Related topics in other textbooks. Consultation outside lecture room: Office
Reading Assignments: Chapter 3 in R. Chang, Chemistry, 8th Ed., McGraw-Hill, 2005 Mass Relationships in Chemical Reactions Or Related topics in other textbooks. Consultation outside lecture room: Office
Quantitative Relationships in Chemical Reactions Chapter 7
 Quantitative Relationships in Chemical Reactions Chapter 7 The burning of charcoal releases heat (thermal energy) that grills our food. But the combustion of charcoal and fossil fuels also releases CO
Quantitative Relationships in Chemical Reactions Chapter 7 The burning of charcoal releases heat (thermal energy) that grills our food. But the combustion of charcoal and fossil fuels also releases CO
General Chemistry. Chapter 3. Mass Relationships in Chemical Reactions CHEM 101 (3+1+0) Dr. Mohamed El-Newehy 10/12/2017
 General Chemistry CHEM 101 (3+1+0) Dr. Mohamed El-Newehy http://fac.ksu.edu.sa/melnewehy Chapter 3 Mass Relationships in Chemical Reactions 1 In this chapter, Chemical structure and formulas in studying
General Chemistry CHEM 101 (3+1+0) Dr. Mohamed El-Newehy http://fac.ksu.edu.sa/melnewehy Chapter 3 Mass Relationships in Chemical Reactions 1 In this chapter, Chemical structure and formulas in studying
Example Exercise 10.1 Interpreting Chemical Equation Calculations
 Example Exercise 10.1 Interpreting Chemical Equation Calculations Given the chemical equation for the combustion of methane, CH 4, balance the equation and interpret the coefficients in terms of (a) moles
Example Exercise 10.1 Interpreting Chemical Equation Calculations Given the chemical equation for the combustion of methane, CH 4, balance the equation and interpret the coefficients in terms of (a) moles
Chapter 7: Stoichiometry - Mass Relations in Chemical Reactions
 Chapter 7: Stoichiometry - Mass Relations in Chemical Reactions How do we balance chemical equations? How can we used balanced chemical equations to relate the quantities of substances consumed and produced
Chapter 7: Stoichiometry - Mass Relations in Chemical Reactions How do we balance chemical equations? How can we used balanced chemical equations to relate the quantities of substances consumed and produced
The Effect of Initial Pressure on Explosions of Hydrogen- Enriched Methane/Air Mixtures
 The Effect of Initial Pressure on Explosions of Hydrogen- Enriched Methane/Air Mixtures Cammarota F. 1, Di Benedetto A. 1, Di Sarli V. 1, Salzano E. 1, Russo G. 2 1 Istituto di Ricerche sulla Combustione,
The Effect of Initial Pressure on Explosions of Hydrogen- Enriched Methane/Air Mixtures Cammarota F. 1, Di Benedetto A. 1, Di Sarli V. 1, Salzano E. 1, Russo G. 2 1 Istituto di Ricerche sulla Combustione,
