Doping-induced valence change in Yb 5 Ge 4 x (Sb, Ga) x : (x 1)
|
|
|
- Bryce Eaton
- 5 years ago
- Views:
Transcription
1 Hyperfine Interact (2012) 208:59 63 DOI /s Doping-induced valence change in Yb 5 Ge 4 x (Sb, Ga) x : (x 1) D. H. Ryan N. R. Lee-Hone J. M. Cadogan Published online: 26 October 2011 Springer Science+Business Media B.V Abstract Following our earlier observation that the ytterbium valence distribution in Yb 5 Ge 4 x Si x was independent of x, weuse 170 Yb Mössbauer spectroscopy to follow the Yb valence distribution in Sb and Ga doped Yb 5 Ge 4. The crystal structure does not change for either dopant and the Yb valence, while affected, proves quite resistant to change, with only one third of the electrons added or removed by the dopants leading to Yb 3+ Yb 2+ conversion. Keywords Lanthanide Valence Mössbauer spectroscopy 1 Introduction The R 5 X 4 (R = Y, La Lu; X = Si, Ge, Sn) compounds became the subject of intense investigation after the discovery of a giant magnetocaloric effect in Gd 5 Si 2 Ge 2 [1]. Systematic phase relationship and structural data have been obtained for most of D. H. Ryan N. R. Lee-Hone Department of Physics, McGill University, Montreal, QC, H3A2T8, Canada J. M. Cadogan (B) Department of Physics and Astronomy, University of Manitoba, Winnipeg, MB, R3T2N2, Canada cadogan@physics.umanitoba.ca Ames Laboratory of the U.S. Department of Energy, Iowa State University, Ames, IA , USA Department of Materials Science and Engineering, Iowa State University, Ames, IA , USA
2 60 D.H. Ryan et al. the R 5 Ge 4 x Si x systems and two key parameters have been identified which affect the balance between the magnetic and structural energies in the R 5 X 4 phases. The first is the ratio of the ionic radius of the rare earth, r R, to the weighted average radius of the metalloid elements, r X [2] and the second is the valence electron concentration [3]. As the ratio r R /r X decreases, the crystal structures of most R 5 X 4 compounds change in the sequence: tetragonal Zr 5 Si 4 orthorhombic Gd 5 Si 4 monoclinic Gd 5 Si 2 Ge 2 orthorhombic Sm 5 Ge 4 [2]. The Yb 5 X 4 compounds are of particular interest because Yb can occur in two valence states: Yb 2+ (r R = 1.94 Å) and Yb 3+ (r R = 1.74 Å). This allows us to explore the effects of both the valence electron concentration and the relative metallic radii (r R /r X ). Surprisingly, Yb 5 Ge 4 x Si x adopts the orthorhombic Gd 5 Si 4 -type (Pnma #62) structure, with a constant Yb 2+ :Yb 3+ population ratio of 1.09(4) for all x [4]. In this paper, we investigate the effects of Sb and Ga doping in Yb 5 Ge 4. Sb and Ga have respectively one more and one less electron in their outer shell than Ge, allowing us to drive Yb 3+ Yb 2+ interconversion directly, while the very different metallic radii of Sb (1.59 Å) and Ga (1.41 Å), both larger that that of Ge (1.37 Å), could drive the Yb valence towards the larger Yb 2+, effectively maintaining an approximately constant r R /r X ratio. 2 Experimental methods The samples used in this study (Yb 5 Ge 4 x Sb x (x = 0.5, 0.75, 1), Yb 5 Ge 4 x Ga x (x = 0.5, 1) and Yb 5 Ge 4 ) were prepared at the Ames Laboratory by induction melting stoichiometric mixtures of the pure elements following the standard procedure described elsewhere [5]. X-ray powder diffraction was used to verify the structure and phase composition of the materials. All of the materials were found to crystallize in the orthorhombic Gd 5 Si 4 -type Pnma structure. A second phase (11:10) is present in the Sb doped samples, amounting to 5 15 mol % of the materials. 170 Yb Mössbauer spectroscopy was used to determine the relative proportions of Yb 2+ and Yb 3+. A 20mCi 170 Tm source was prepared by neutron activation of 20 mg of Tm as a 10 wt% alloy in aluminium. The source and sample were mounted vertically in a helium flow cryostat and the drive was operated in sine mode. The kev Mössbauer γ-photons were isolated from the various x-rays emitted by the source using a high-purity Ge detector. The spectrometer s drive velocity was calibrated with a laser interferometer and velocities were cross-checked against 57 CoRh/α-Fe at room temperature. All spectra were obtained at 5 K (except for Yb 5 Ge 4 which was part of the earlier study [4] and was taken at 8 K) and fitted using a nonlinear least-squares minimization routine with line positions and intensities derived from an exact solution to the full Hamiltonian [6]. 3 Results and discussion The kev transition in 170 Yb involves a rotational change at almost constant nuclear volume, so the isomer shift is essentially independent of valence. However, the electric field gradients (EFG) associated with the two valence
3 Doping-induced valence change in Yb 5 Ge 4 x (Sb, Ga) x : (x 1) 61 Fig. 1 (Left) 170 Yb Mössbauer spectra of Yb 5 Ge 4 x (Sb, Ga) x : (x 1), obtained at 5 K (except for Yb 5 Ge 4, taken at 8 K). The solid lines are fits to a full Hamiltonian solution as described in the text. For the top spectrum (Yb 5 GaGe 3 ) we also show the two components Yb 2+ (red) andyb 3+ (blue). (Right) Dopant dependence of the Yb 2+ :Yb 3+ area ratio, deduced from fits to the 170 Yb Mössbauer spectra states are quite different. Yb 2+ has a nonmagnetic, closed shell 4 f 14 electronic configuration, leaving only a small contribution to the EFG from the lattice, whereas Yb 3+ has a 4 f 13 configuration which results in a significant 4f contribution to the EFG. The 170 Yb Mössbauer spectra of the studied compounds are shown in the left panel of Fig. 1. Each spectrum consists of two paramagnetic components, corresponding to the two valence states of Yb. As with Yb 5 Ge 4 x Si x [4], it was not possible to resolve the contributions from the three ytterbium sites, and only a valence separation was possible. The right panel of Fig. 1 shows the Yb 2+ :Yb 3+ subspectral area ratio plotted against the dopant content. Doping with Sb strongly increases the Yb 2+ :Yb 3+ ratio, whereas Ga doping produces a smaller decrease. This difference in the strength of the doping effects may derive from the larger size of the dopants supporting (in the case of Sb) or competing with (in the case of Ga) the valence shift. One way to separate out the effects of dopant size (r X ) is to combine our data with those from Yb 5 Ge 4 x Si x [4] in the left panel of Fig. 2, where it is clear that doping with the larger Ga actually leads to a cell volume decrease while the increase with Sb doping is slower than expected. Both departures from the trend established by Yb 5 Ge 4 x Si x are due to Yb 3+ Yb 2+ conversion. Finally, in the right panel of Fig. 2 we show the total valence electron count, per formula unit, as a function of dopant (Ga = 3; Ge = 4; Sb = 5 with the Yb value derived from the measured Yb 2+ :Yb 3+ ratio). No valence conversion would give a slope of unity, while full dopant-induced conversion would yield zero slope. The
4 62 D.H. Ryan et al. Fig. 2 (Left) Cell volume dependence of the atomic radii ratio (r R /r X ). (green) and the dashed line show the trend for Yb 5 Ge 4 x Si x [4]. (red) and (blue) show data for gallium and antimony doping respectively. (Right) Dopant dependence of the weighted valence electron count fitted slope of 0.63(6) indicates that the Yb valence distribution is rather robust, with only about one-third of the valence electrons donated by the dopants actually leading to Yb 3+ Yb 2+ valence conversion, with the other two-thirds entering the conduction band. In order to obtain more information on the relative importance of dopant size and valence, we plan to investigate the effects of In (over-sized acceptor) and As (size-matched donor) doping in this system. 4 Conclusions We have examined the effect of Ga and Sb doping on the Yb 2+ :Yb 3+ population ratio in the Yb 5 (Ge, Ga, Sb) 4 system by 170 Yb Mössbauer spectroscopy. Sb doping has a large effect on the Yb valence ratio, due to the combined effects of the increased atom size and the increase in valence electron concentration. In the case of Ga doping, the competing effects of the increased atom size and the reduced electron valence concentration lead to significantly smaller changes in the Yb 2+ :Yb 3+ ratio. Finally, the crystallographic cell volume increases with Sb doping, and decreases with Ga doping, an effect which is explained in part by the change in average weighted volume of the Yb ions, due to the change in the Yb 2+ :Yb 3+ ratio. Acknowledgements JMC acknowledges support from the Canada Research Chairs programme. Financial support for various stages of this work was provided by the Natural Sciences and Engineering Research Council of Canada and Fonds Québécois de la Recherche sur la Nature et les Technologies. Source activation was by M. Butler at the McMaster Nuclear Reactor (MNR), Hamilton, Ontario. Work at Ames Laboratory is supported by the U.S. Department of Energy, Office of Basic Energy Sciences, Division of Materials Sciences and Engineering under Contract No. DE-AC02-07CH11358 with Iowa State University.
5 Doping-induced valence change in Yb 5 Ge 4 x (Sb, Ga) x : (x 1) 63 References 1. Pecharsky, V.K., Gschneidner, K.A. Jr.: Giant magnetocaloric effect in Gd 5 Si 2 Ge 2. Phys. Rev. Lett. 78, (1997) 2. Pecharsky, A.O., Pecharsky, V.K., Gschneidner, K.A. Jr.: Phase relationships and low temperature heat capacities of alloys in the Y 5 Si 4 -Y 5 Ge 4 pseudobinary system. J. Alloys Compd. 379, 127 (2004) 3. Choe, W., Pecharsky, V.K., Pecharsky, A.O., Gschneidner, K.A. Jr., Young, V.G., Miller, G.J.: Making and breaking covalent bonds across the magnetic transition in the giant magnetocaloric material Gd 5 (Si 2 Ge 2 ). Phys. Rev. Lett. 84, 4617 (2000) 4. Voyer, C.J., Ryan, D.H., Ahn, K., Gschneidner, K.A. Jr., Pecharsky, V.K.: Valence and magnetic ordering in the Yb 5 Si x Ge 4 x pseudobinary system. Phys. Rev. B 73, (2006) 5. Ahn, K., Tsokol, A.O., Mozharivskyj, Y., Gschneidner, K.A. Jr., Pecharsky, V.K.: Phase relationships and structural, magnetic and thermodynamic properties of the Yb 5 Si 4 Yb 5 Ge 4 pseudobinary system. Phys. Rev. B 72, (2005) 6. Voyer, C.J., Ryan, D.H.: A complete solution to the Mössbauer problem, all in one place. Hyperfine Interact. 170, (2006)
Valence and magnetic ordering in YbMn 2 Si 2 x Ge x
 INSTITUTE OF PHYSICSPUBLISHING JOURNAL OFPHYSICS: CONDENSED MATTER J. Phys.: Condens. Matter 16 (2004) 6129 6138 PII: S0953-8984(04)79691-5 Valence and magnetic ordering in YbMn 2 Si 2 x Ge x DHRyan 1,JMCadogan
INSTITUTE OF PHYSICSPUBLISHING JOURNAL OFPHYSICS: CONDENSED MATTER J. Phys.: Condens. Matter 16 (2004) 6129 6138 PII: S0953-8984(04)79691-5 Valence and magnetic ordering in YbMn 2 Si 2 x Ge x DHRyan 1,JMCadogan
Observation of giant magnetocaloric effect in HoCoSi
 Materials Letters 113, 195 (2013) Observation of giant magnetocaloric effect in ocosi Sachin Gupta and K.G. Suresh * Department of Physics, Indian Institute of Technology Bombay, Mumbai-400076, India Abstract
Materials Letters 113, 195 (2013) Observation of giant magnetocaloric effect in ocosi Sachin Gupta and K.G. Suresh * Department of Physics, Indian Institute of Technology Bombay, Mumbai-400076, India Abstract
A proof-of-principle experiment of EIT with gamma radiation in FePSe 3 single crystal
 Hyperfine Interact DOI 1.17/s1751-11-8-8 A proof-of-principle experiment of EIT with gamma radiation in FePSe 3 single crystal Hisakazu Muramatsu S. Nakajo K. Nakagami K. Nagata S. Mochizuki H. Suzuki
Hyperfine Interact DOI 1.17/s1751-11-8-8 A proof-of-principle experiment of EIT with gamma radiation in FePSe 3 single crystal Hisakazu Muramatsu S. Nakajo K. Nakagami K. Nagata S. Mochizuki H. Suzuki
Metals and Nonmetals
 The Periodic Table and Atomic Properties The periodic table originally came from the observation that when the elements are arranged by atomic mass, properties recur periodically. (Mendeleev) Now we understand
The Periodic Table and Atomic Properties The periodic table originally came from the observation that when the elements are arranged by atomic mass, properties recur periodically. (Mendeleev) Now we understand
Metals and Nonmetals. Metals and Nonmetals. The Periodic Table and Atomic Properties
 The Periodic Table and Atomic Properties The periodic table originally came from the observation that when the elements are arranged by atomic mass, properties recur periodically. (Mendeleev) Now we understand
The Periodic Table and Atomic Properties The periodic table originally came from the observation that when the elements are arranged by atomic mass, properties recur periodically. (Mendeleev) Now we understand
Magnetic Order versus superconductivity in the Iron-based
 Magnetic Order versus superconductivity in the Iron-based layered La(O 1-x F x )FeAs systems Clarina de la Cruz 1,2, Q. Huang 3, J. W. Lynn 3, Jiying Li 3,4, W. Ratcliff II 3, J. L. Zarestky 5, H. A. Mook
Magnetic Order versus superconductivity in the Iron-based layered La(O 1-x F x )FeAs systems Clarina de la Cruz 1,2, Q. Huang 3, J. W. Lynn 3, Jiying Li 3,4, W. Ratcliff II 3, J. L. Zarestky 5, H. A. Mook
X-Ray and Mössbauer Spectra and Electronic Structure of ScFe 2 Si 2 Compound
 Journal of Materials Science and Engineering B 5 (1-2) (2015) 42-49 doi: 10.17265/2161-6221/2015.1-2.004 D DAVID PUBLISHING X-Ray and Mössbauer Spectra and Electronic Structure of ScFe 2 Si 2 Compound
Journal of Materials Science and Engineering B 5 (1-2) (2015) 42-49 doi: 10.17265/2161-6221/2015.1-2.004 D DAVID PUBLISHING X-Ray and Mössbauer Spectra and Electronic Structure of ScFe 2 Si 2 Compound
ECE 335: Electronic Engineering Lecture 2: Semiconductors
 Faculty of Engineering ECE 335: Electronic Engineering Lecture 2: Semiconductors Agenda Intrinsic Semiconductors Extrinsic Semiconductors N-type P-type Carrier Transport Drift Diffusion Semiconductors
Faculty of Engineering ECE 335: Electronic Engineering Lecture 2: Semiconductors Agenda Intrinsic Semiconductors Extrinsic Semiconductors N-type P-type Carrier Transport Drift Diffusion Semiconductors
3.1 - The Periodic Table
 3.1 - The Periodic Table 3.1.1 - Describe the arrangement of elements in the periodic table in order of increasing atomic number Elements in the periodic table are arranged in order of increasing atomic
3.1 - The Periodic Table 3.1.1 - Describe the arrangement of elements in the periodic table in order of increasing atomic number Elements in the periodic table are arranged in order of increasing atomic
Structure, magnetism, and thermodynamics of the novel rare earth-based R 5
 Pure Appl. Chem., Vol. 79, No. 8, pp. 1383 1402, 2007. doi:10.1351/pac200779081383 2007 IUPAC Structure, magnetism, and thermodynamics of the novel rare earth-based R 5 T 4 intermetallics* V. K. Pecharsky
Pure Appl. Chem., Vol. 79, No. 8, pp. 1383 1402, 2007. doi:10.1351/pac200779081383 2007 IUPAC Structure, magnetism, and thermodynamics of the novel rare earth-based R 5 T 4 intermetallics* V. K. Pecharsky
Magnetic and structural transitions in the ironchalcogenide K0.8Fe1.76Se2.00
 Ames Laboratory Publications Ames Laboratory 2012 Magnetic and structural transitions in the ironchalcogenide high-tc superconductor: K0.8Fe1.76Se2.00 W. N. Rowan-Weetaluktuk McGill University D. H. Ryan
Ames Laboratory Publications Ames Laboratory 2012 Magnetic and structural transitions in the ironchalcogenide high-tc superconductor: K0.8Fe1.76Se2.00 W. N. Rowan-Weetaluktuk McGill University D. H. Ryan
Phase Transformation Driven by Valence Electron Concentration: Tuning Interslab Bond Distances in Gd5GaxGe4-x
 Chemistry Publications Chemistry 2003 Phase Transformation Driven by Valence Electron Concentration: Tuning Interslab Bond Distances in Gd5GaxGe4-x Yurij Mozharivskyj Iowa State University Wonyoung Choe
Chemistry Publications Chemistry 2003 Phase Transformation Driven by Valence Electron Concentration: Tuning Interslab Bond Distances in Gd5GaxGe4-x Yurij Mozharivskyj Iowa State University Wonyoung Choe
Semiconductors. Semiconductors also can collect and generate photons, so they are important in optoelectronic or photonic applications.
 Semiconductors Semiconducting materials have electrical properties that fall between true conductors, (like metals) which are always highly conducting and insulators (like glass or plastic or common ceramics)
Semiconductors Semiconducting materials have electrical properties that fall between true conductors, (like metals) which are always highly conducting and insulators (like glass or plastic or common ceramics)
Magnetic properties and magnetic entropy changes of La 1 x Pr x Fe 11.5 Si 1.5 compounds with 0 x 0.5
 Vol 16 No 12, December 2007 c 2007 Chin. Phys. Soc. 1009-1963/2007/16(12)/3848-05 Chinese Physics and IOP Publishing Ltd Magnetic properties and magnetic entropy changes of La 1 x Pr x Fe 11.5 Si 1.5 compounds
Vol 16 No 12, December 2007 c 2007 Chin. Phys. Soc. 1009-1963/2007/16(12)/3848-05 Chinese Physics and IOP Publishing Ltd Magnetic properties and magnetic entropy changes of La 1 x Pr x Fe 11.5 Si 1.5 compounds
APEX CARE INSTITUTE FOR PG - TRB, SLET AND NET IN PHYSICS
 Page 1 1. Within the nucleus, the charge distribution A) Is constant, but falls to zero sharply at the nuclear radius B) Increases linearly from the centre, but falls off exponentially at the surface C)
Page 1 1. Within the nucleus, the charge distribution A) Is constant, but falls to zero sharply at the nuclear radius B) Increases linearly from the centre, but falls off exponentially at the surface C)
A Comparative Study of the Electronic and Magnetic Properties of Gd 5 Ge 4 and Gd 5 Si 4 Compounds
 Commun. Theor. Phys. 62 (2014) 903 908 Vol. 62, No. 6, December 1, 2014 A Comparative Study of the Electronic and Magnetic Properties of Gd 5 Ge 4 and Gd 5 Si 4 Compounds Z. Momeni Larimi, 1, A. Amirabadizadeh,
Commun. Theor. Phys. 62 (2014) 903 908 Vol. 62, No. 6, December 1, 2014 A Comparative Study of the Electronic and Magnetic Properties of Gd 5 Ge 4 and Gd 5 Si 4 Compounds Z. Momeni Larimi, 1, A. Amirabadizadeh,
Structure and magnetic transition of LaFe 13 x Si x compounds
 INSTITUTE OF PHYSICSPUBLISHING JOURNAL OFPHYSICS: CONDENSED MATTER J. Phys.: Condens. Matter 15 (2003) 7385 7394 PII: S0953-8984(03)65192-1 Structure and magnetic transition of LaFe 13 x Si x compounds
INSTITUTE OF PHYSICSPUBLISHING JOURNAL OFPHYSICS: CONDENSED MATTER J. Phys.: Condens. Matter 15 (2003) 7385 7394 PII: S0953-8984(03)65192-1 Structure and magnetic transition of LaFe 13 x Si x compounds
2008 Brooks/Cole 2. Frequency (Hz)
 Electromagnetic Radiation and Matter Oscillating electric and magnetic fields. Magnetic field Electric field Chapter 7: Electron Configurations and the Periodic Table Traveling wave moves through space
Electromagnetic Radiation and Matter Oscillating electric and magnetic fields. Magnetic field Electric field Chapter 7: Electron Configurations and the Periodic Table Traveling wave moves through space
PHASE DIAGRAM AND MAGNETOCALORIC EFFECTS IN Ni 1-x Cr x MnGe 1.05
 PHASE DIAGRAM AND MAGNETOCALORIC EFFECTS IN Ni 1-x Cr x MnGe 1.05 Anil Aryal 1, Abdiel Quetz 1, Sudip Pandey 1, Michael Eubank 1, Tapas Samanta 2, Igor Dubenko 1, Shane Stadler 2, and Naushad Ali 1 1 Department
PHASE DIAGRAM AND MAGNETOCALORIC EFFECTS IN Ni 1-x Cr x MnGe 1.05 Anil Aryal 1, Abdiel Quetz 1, Sudip Pandey 1, Michael Eubank 1, Tapas Samanta 2, Igor Dubenko 1, Shane Stadler 2, and Naushad Ali 1 1 Department
Reduction of hysteresis losses in the magnetic refrigerant La0.8Ce0.2Fe11.4Si1.6 by the addition of boron
 University of Wollongong Research Online Faculty of Engineering - Papers (Archive) Faculty of Engineering and Information Sciences 2011 Reduction of hysteresis losses in the magnetic refrigerant La0.8Ce0.2Fe11.4Si1.6
University of Wollongong Research Online Faculty of Engineering - Papers (Archive) Faculty of Engineering and Information Sciences 2011 Reduction of hysteresis losses in the magnetic refrigerant La0.8Ce0.2Fe11.4Si1.6
Multifunctionality from coexistence of large magnetoresistance and magnetocaloric effect in La0.7Ca0.3MnO3
 University of Wollongong Research Online Faculty of Engineering - Papers (Archive) Faculty of Engineering and Information Sciences 2011 Multifunctionality from coexistence of large magnetoresistance and
University of Wollongong Research Online Faculty of Engineering - Papers (Archive) Faculty of Engineering and Information Sciences 2011 Multifunctionality from coexistence of large magnetoresistance and
Lecture 7: Extrinsic semiconductors - Fermi level
 Lecture 7: Extrinsic semiconductors - Fermi level Contents 1 Dopant materials 1 2 E F in extrinsic semiconductors 5 3 Temperature dependence of carrier concentration 6 3.1 Low temperature regime (T < T
Lecture 7: Extrinsic semiconductors - Fermi level Contents 1 Dopant materials 1 2 E F in extrinsic semiconductors 5 3 Temperature dependence of carrier concentration 6 3.1 Low temperature regime (T < T
Periodicity & Many-Electron Atoms
 Chap. 8 ELECTRON CONFIGURAT N & CEMICAL PERIODICITY 8.1-8.2 Periodicity & Many-Electron Atoms Understand the correlation of electron configuration and the periodic character of atomic properties such as
Chap. 8 ELECTRON CONFIGURAT N & CEMICAL PERIODICITY 8.1-8.2 Periodicity & Many-Electron Atoms Understand the correlation of electron configuration and the periodic character of atomic properties such as
The Periodic Table. Periodic Properties. Can you explain this graph? Valence Electrons. Valence Electrons. Paramagnetism
 Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Chapter 8 Magnetic Resonance
 Chapter 8 Magnetic Resonance 9.1 Electron paramagnetic resonance 9.2 Ferromagnetic resonance 9.3 Nuclear magnetic resonance 9.4 Other resonance methods TCD March 2007 1 A resonance experiment involves
Chapter 8 Magnetic Resonance 9.1 Electron paramagnetic resonance 9.2 Ferromagnetic resonance 9.3 Nuclear magnetic resonance 9.4 Other resonance methods TCD March 2007 1 A resonance experiment involves
Lecture 3 continuation from Wednesday
 Lecture 3 continuation from Wednesday Dynamical Matrix - Conduction electrons Importance of el-ph Matrix elements NiAl 50-50 compound Confirmed by H. Chou and S.M.Shapiro Phys. Rev. B48, 16088 (1993) Exp.
Lecture 3 continuation from Wednesday Dynamical Matrix - Conduction electrons Importance of el-ph Matrix elements NiAl 50-50 compound Confirmed by H. Chou and S.M.Shapiro Phys. Rev. B48, 16088 (1993) Exp.
Trace Elements - Definitions
 Trace Elements - Definitions Elements that are not stoichiometric constituents in phases in the system of interest For example, IG/MET systems would have different trace elements than aqueous systems Do
Trace Elements - Definitions Elements that are not stoichiometric constituents in phases in the system of interest For example, IG/MET systems would have different trace elements than aqueous systems Do
arxiv: v1 [cond-mat.supr-con] 9 May 2013
![arxiv: v1 [cond-mat.supr-con] 9 May 2013 arxiv: v1 [cond-mat.supr-con] 9 May 2013](/thumbs/92/109271866.jpg) Journal of the Physical Society of Japan LETTERS Superconductivity in Tetragonal LaPt 2 x Ge 2+x arxiv:135.224v1 [cond-mat.supr-con] 9 May 213 Satoki Maeda 1, Kazuaki Matano 1, Hiroki Sawaoka 1, Yoshihiko
Journal of the Physical Society of Japan LETTERS Superconductivity in Tetragonal LaPt 2 x Ge 2+x arxiv:135.224v1 [cond-mat.supr-con] 9 May 213 Satoki Maeda 1, Kazuaki Matano 1, Hiroki Sawaoka 1, Yoshihiko
CHEM 10113, Quiz 5 October 26, 2011
 CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
X-ray, Neutron and e-beam scattering
 X-ray, Neutron and e-beam scattering Introduction Why scattering? Diffraction basics Neutrons and x-rays Techniques Direct and reciprocal space Single crystals Powders CaFe 2 As 2 an example What is the
X-ray, Neutron and e-beam scattering Introduction Why scattering? Diffraction basics Neutrons and x-rays Techniques Direct and reciprocal space Single crystals Powders CaFe 2 As 2 an example What is the
Unit IV Semiconductors Engineering Physics
 Introduction A semiconductor is a material that has a resistivity lies between that of a conductor and an insulator. The conductivity of a semiconductor material can be varied under an external electrical
Introduction A semiconductor is a material that has a resistivity lies between that of a conductor and an insulator. The conductivity of a semiconductor material can be varied under an external electrical
Atoms? All matters on earth made of atoms (made up of elements or combination of elements).
 Chapter 1 Atoms? All matters on earth made of atoms (made up of elements or combination of elements). Atomic Structure Atom is the smallest particle of an element that can exist in a stable or independent
Chapter 1 Atoms? All matters on earth made of atoms (made up of elements or combination of elements). Atomic Structure Atom is the smallest particle of an element that can exist in a stable or independent
ECE 142: Electronic Circuits Lecture 3: Semiconductors
 Faculty of Engineering ECE 142: Electronic Circuits Lecture 3: Semiconductors Agenda Intrinsic Semiconductors Extrinsic Semiconductors N-type P-type Carrier Transport Drift Diffusion Semiconductors A semiconductor
Faculty of Engineering ECE 142: Electronic Circuits Lecture 3: Semiconductors Agenda Intrinsic Semiconductors Extrinsic Semiconductors N-type P-type Carrier Transport Drift Diffusion Semiconductors A semiconductor
EXTRINSIC SEMICONDUCTOR
 EXTRINSIC SEMICONDUCTOR In an extrinsic semiconducting material, the charge carriers originate from impurity atoms added to the original material is called impurity [or] extrinsic semiconductor. This Semiconductor
EXTRINSIC SEMICONDUCTOR In an extrinsic semiconducting material, the charge carriers originate from impurity atoms added to the original material is called impurity [or] extrinsic semiconductor. This Semiconductor
ELECTRONIC I Lecture 1 Introduction to semiconductor. By Asst. Prof Dr. Jassim K. Hmood
 ELECTRONIC I Lecture 1 Introduction to semiconductor By Asst. Prof Dr. Jassim K. Hmood SOLID-STATE ELECTRONIC MATERIALS Electronic materials generally can be divided into three categories: insulators,
ELECTRONIC I Lecture 1 Introduction to semiconductor By Asst. Prof Dr. Jassim K. Hmood SOLID-STATE ELECTRONIC MATERIALS Electronic materials generally can be divided into three categories: insulators,
Solid State Spectroscopy Problem Set 7
 Solid State Spectroscopy Problem Set 7 Due date: June 29th, 2015 Problem 5.1 EXAFS Study of Mn/Fe substitution in Y(Mn 1-x Fe x ) 2 O 5 From article «EXAFS, XANES, and DFT study of the mixed-valence compound
Solid State Spectroscopy Problem Set 7 Due date: June 29th, 2015 Problem 5.1 EXAFS Study of Mn/Fe substitution in Y(Mn 1-x Fe x ) 2 O 5 From article «EXAFS, XANES, and DFT study of the mixed-valence compound
Chapter 10: Modern Atomic Theory and the Periodic Table. How does atomic structure relate to the periodic table? 10.1 Electromagnetic Radiation
 Chapter 10: Modern Atomic Theory and the Periodic Table How does atomic structure relate to the periodic table? 10.1 Electromagnetic Radiation Electromagnetic (EM) radiation is a form of energy that exhibits
Chapter 10: Modern Atomic Theory and the Periodic Table How does atomic structure relate to the periodic table? 10.1 Electromagnetic Radiation Electromagnetic (EM) radiation is a form of energy that exhibits
1 Review of semiconductor materials and physics
 Part One Devices 1 Review of semiconductor materials and physics 1.1 Executive summary Semiconductor devices are fabricated using specific materials that offer the desired physical properties. There are
Part One Devices 1 Review of semiconductor materials and physics 1.1 Executive summary Semiconductor devices are fabricated using specific materials that offer the desired physical properties. There are
Effects of Crystal Structure on Microwave Dielectric Properties of Ceramics
 Journal of the Korean Ceramic Society Vol. 5 No. 5 pp. 5~55 008. Review Effects of Crystal Structure on Microwave Dielectric Properties of Ceramics Eung Soo Kim Chang Jun Jeon Sung Joo Kim and Su Jung
Journal of the Korean Ceramic Society Vol. 5 No. 5 pp. 5~55 008. Review Effects of Crystal Structure on Microwave Dielectric Properties of Ceramics Eung Soo Kim Chang Jun Jeon Sung Joo Kim and Su Jung
Gd5 xyxtt4 (Tt = Si or Ge): Effect of Metal Substitution on Structure, Bonding, and Magnetism
 Chemistry Publications Chemistry 2008 Gd5 xyxtt4 (Tt = Si or Ge): Effect of Metal Substitution on Structure, Bonding, and Magnetism Sumohan Misra Iowa State University, misra@iastate.edu Gordon J. Miller
Chemistry Publications Chemistry 2008 Gd5 xyxtt4 (Tt = Si or Ge): Effect of Metal Substitution on Structure, Bonding, and Magnetism Sumohan Misra Iowa State University, misra@iastate.edu Gordon J. Miller
Gd5Si4 xpx: Targeted Structural Changes through Increase in Valence Electron Count
 Chemistry Publications Chemistry 2009 Gd5Si4 xpx: Targeted Structural Changes through Increase in Valence Electron Count Volodymyr Svitlyk McMaster University Gordon J. Miller Iowa State University, gmiller@iastate.edu
Chemistry Publications Chemistry 2009 Gd5Si4 xpx: Targeted Structural Changes through Increase in Valence Electron Count Volodymyr Svitlyk McMaster University Gordon J. Miller Iowa State University, gmiller@iastate.edu
The effect of varying the crystal structure on the magnetism, electronic
 The effect of varying the crystal structure on the magnetism, electronic structure and thermodynamics in the Gd 5 (Si x Ge 1-x ) 4 system near x = 0.5 V.K. Pecharsky 1,*,#, G.D. Samolyuk *, V.P. Antropov
The effect of varying the crystal structure on the magnetism, electronic structure and thermodynamics in the Gd 5 (Si x Ge 1-x ) 4 system near x = 0.5 V.K. Pecharsky 1,*,#, G.D. Samolyuk *, V.P. Antropov
A Perturbed-Angular-Correlation Study of Hyperfine Interactions at 181 Ta in -Fe 2 O 3
 Hyperfine Interactions (2005) DOI 10.1007/s10751-005-9061-z # Springer 2005 A Perturbed-Angular-Correlation Study of Hyperfine Interactions at 181 Ta in -Fe 2 O 3 A. F. PASQUEVICH 1, *,a, A. C. JUNQUEIRA
Hyperfine Interactions (2005) DOI 10.1007/s10751-005-9061-z # Springer 2005 A Perturbed-Angular-Correlation Study of Hyperfine Interactions at 181 Ta in -Fe 2 O 3 A. F. PASQUEVICH 1, *,a, A. C. JUNQUEIRA
Chapter 9 Ionic and Covalent Bonding
 Chem 1045 Prof George W.J. Kenney, Jr General Chemistry by Ebbing and Gammon, 8th Edition Last Update: 06-April-2009 Chapter 9 Ionic and Covalent Bonding These Notes are to SUPPLIMENT the Text, They do
Chem 1045 Prof George W.J. Kenney, Jr General Chemistry by Ebbing and Gammon, 8th Edition Last Update: 06-April-2009 Chapter 9 Ionic and Covalent Bonding These Notes are to SUPPLIMENT the Text, They do
MOSELEY and MODERN PERIODIC TABLE (designed by atomic numbers of elements)
 MOSELEY and MODERN PERIODIC TABLE (designed by atomic numbers of elements) 1 PERIODS: Period number = Number of basic energy levels = The principal quantum number The horizontal lines in the periodic system
MOSELEY and MODERN PERIODIC TABLE (designed by atomic numbers of elements) 1 PERIODS: Period number = Number of basic energy levels = The principal quantum number The horizontal lines in the periodic system
CLASS TEST GRADE 11. PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials 1
 CLASS TEST GRADE PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials MARKS: 45 TIME: hour INSTRUCTIONS AND INFORMATION. Answer ALL the questions. 2. You may use non-programmable calculators. 3. You
CLASS TEST GRADE PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials MARKS: 45 TIME: hour INSTRUCTIONS AND INFORMATION. Answer ALL the questions. 2. You may use non-programmable calculators. 3. You
 lectures accompanying the book: Solid State Physics: An Introduction, by Philip ofmann (2nd edition 2015, ISBN-10: 3527412824, ISBN-13: 978-3527412822, Wiley-VC Berlin. www.philiphofmann.net 1 Bonds between
lectures accompanying the book: Solid State Physics: An Introduction, by Philip ofmann (2nd edition 2015, ISBN-10: 3527412824, ISBN-13: 978-3527412822, Wiley-VC Berlin. www.philiphofmann.net 1 Bonds between
Studying Metal to Insulator Transitions in Solids using Synchrotron Radiation-based Spectroscopies.
 PY482 Lecture. February 28 th, 2013 Studying Metal to Insulator Transitions in Solids using Synchrotron Radiation-based Spectroscopies. Kevin E. Smith Department of Physics Department of Chemistry Division
PY482 Lecture. February 28 th, 2013 Studying Metal to Insulator Transitions in Solids using Synchrotron Radiation-based Spectroscopies. Kevin E. Smith Department of Physics Department of Chemistry Division
Chapter 1 Overview of Semiconductor Materials and Physics
 Chapter 1 Overview of Semiconductor Materials and Physics Professor Paul K. Chu Conductivity / Resistivity of Insulators, Semiconductors, and Conductors Semiconductor Elements Period II III IV V VI 2 B
Chapter 1 Overview of Semiconductor Materials and Physics Professor Paul K. Chu Conductivity / Resistivity of Insulators, Semiconductors, and Conductors Semiconductor Elements Period II III IV V VI 2 B
The periodic properties of elements. The stable filled electron configuration
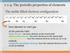 1-1-4. The periodic properties of elements The stable filled electron configuration Inert element or inert gas At the periodic table Alkali element : one more electron on the closed shell. Alkaline earth
1-1-4. The periodic properties of elements The stable filled electron configuration Inert element or inert gas At the periodic table Alkali element : one more electron on the closed shell. Alkaline earth
Topic 3: Periodic Trends and Atomic Spectroscopy
 Topic 3: Periodic Trends and Atomic Spectroscopy Introduction Valence Electrons are those in the outer most shell of an element and are responsible for the bonding characteristics of that element. Core
Topic 3: Periodic Trends and Atomic Spectroscopy Introduction Valence Electrons are those in the outer most shell of an element and are responsible for the bonding characteristics of that element. Core
EXTRINSIC SEMICONDUCTOR
 EXTRINSIC SEMICONDUCTOR EXTRINSIC SEMICONDUCTOR A semiconductor in which the impurity atoms are added by doping process is called Extrinsic semiconductor. The addition of impurities increases the carrier
EXTRINSIC SEMICONDUCTOR EXTRINSIC SEMICONDUCTOR A semiconductor in which the impurity atoms are added by doping process is called Extrinsic semiconductor. The addition of impurities increases the carrier
Magnetic structure of Gd 5 Si 2 Ge 2 and Gd 5 Si 2 Ge 1.9 M 0.1 (M = Ga, Cu)
 Magnetic structure of Gd 5 Si 2 Ge 2 and Gd 5 Si 2 Ge 1.9 M 0.1 (M = Ga, Cu) E Palacios 1, J A Rodríguez-Velamazán 1,2, G F Wang 1, R Burriel 1, G Cuello 2 and J Rodríguez-Carvajal 2 1 Instituto de Ciencia
Magnetic structure of Gd 5 Si 2 Ge 2 and Gd 5 Si 2 Ge 1.9 M 0.1 (M = Ga, Cu) E Palacios 1, J A Rodríguez-Velamazán 1,2, G F Wang 1, R Burriel 1, G Cuello 2 and J Rodríguez-Carvajal 2 1 Instituto de Ciencia
Correlation of the Fermi Energy and Others to the Electron Charge-Discharge Characteristics of Atoms
 SSRG International Journal of Material Science and Engineering (SSRG-IJMSE) volume 3 Issue 3 Sep to Dec 7 Correlation of the rmi Energy and Others to the Electron Charge-Discharge Characteristics of Atoms
SSRG International Journal of Material Science and Engineering (SSRG-IJMSE) volume 3 Issue 3 Sep to Dec 7 Correlation of the rmi Energy and Others to the Electron Charge-Discharge Characteristics of Atoms
1 of 5 14/10/ :21
 X-ray absorption s, characteristic X-ray lines... 4.2.1 Home About Table of Contents Advanced Search Copyright Feedback Privacy You are here: Chapter: 4 Atomic and nuclear physics Section: 4.2 Absorption
X-ray absorption s, characteristic X-ray lines... 4.2.1 Home About Table of Contents Advanced Search Copyright Feedback Privacy You are here: Chapter: 4 Atomic and nuclear physics Section: 4.2 Absorption
Mossbauer Effect and Spectroscopy. Kishan Sinha Xu Group Department of Physics and Astronomy University of Nebraska-Lincoln
 Mossbauer Effect and Spectroscopy Kishan Sinha Xu Group Department of Physics and Astronomy University of Nebraska-Lincoln Emission E R γ-photon E transition hν = E transition - E R Photon does not carry
Mossbauer Effect and Spectroscopy Kishan Sinha Xu Group Department of Physics and Astronomy University of Nebraska-Lincoln Emission E R γ-photon E transition hν = E transition - E R Photon does not carry
Laser Spectroscopy on Bunched Radioactive Ion Beams
 Laser Spectroscopy on Bunched Radioactive Ion Beams Jon Billowes University of Manchester Balkan School on Nuclear Physics, Bodrum 2004 Lecture 1. 1.1 Nuclear moments 1.2 Hyperfine interaction in free
Laser Spectroscopy on Bunched Radioactive Ion Beams Jon Billowes University of Manchester Balkan School on Nuclear Physics, Bodrum 2004 Lecture 1. 1.1 Nuclear moments 1.2 Hyperfine interaction in free
Lattice dynamics, phase transitions and spin relaxation in [Fe(C 5 H 5 ) 2 ]PF 6
![Lattice dynamics, phase transitions and spin relaxation in [Fe(C 5 H 5 ) 2 ]PF 6 Lattice dynamics, phase transitions and spin relaxation in [Fe(C 5 H 5 ) 2 ]PF 6](/thumbs/96/126746830.jpg) Hyperfine Interact (2016) 237:100 DOI 10.1007/s10751-016-1310-9 Lattice dynamics, phase transitions and spin relaxation in [Fe(C 5 H 5 ) 2 ]PF 6 R. H. Herber 1 I. Felner 1 I. Nowik 1 Springer International
Hyperfine Interact (2016) 237:100 DOI 10.1007/s10751-016-1310-9 Lattice dynamics, phase transitions and spin relaxation in [Fe(C 5 H 5 ) 2 ]PF 6 R. H. Herber 1 I. Felner 1 I. Nowik 1 Springer International
Unit WorkBook 3 Level 4 ENG U3 Engineering Science LO3 Engineering Materials 2018 UniCourse Ltd. All Rights Reserved. Sample
 Pearson BTEC Levels 4 and 5 Higher Nationals in Engineering (RQF) Unit 3: Engineering Science (core) Unit Workbook 3 in a series of 4 for this unit Learning Outcome 3 Engineering Materials Page 1 of 27
Pearson BTEC Levels 4 and 5 Higher Nationals in Engineering (RQF) Unit 3: Engineering Science (core) Unit Workbook 3 in a series of 4 for this unit Learning Outcome 3 Engineering Materials Page 1 of 27
This work was performed under the auspices of the U.S. Department of Energy by Lawrence Livermore National Laboratory under contract
 This work was performed under the auspices of the U.S. Department of Energy by under contract DE-AC52-7NA27344. Lawrence Livermore National Security, LLC The ITER tokamak Tungsten (W) is attractive as
This work was performed under the auspices of the U.S. Department of Energy by under contract DE-AC52-7NA27344. Lawrence Livermore National Security, LLC The ITER tokamak Tungsten (W) is attractive as
Magnetic properties and magnetocaloric effect in layered NdMn1.9Ti0.1Si2
 University of Wollongong Research Online Australian Institute for Innovative Materials - Papers Australian Institute for Innovative Materials 214 Magnetic properties and magnetocaloric effect in layered
University of Wollongong Research Online Australian Institute for Innovative Materials - Papers Australian Institute for Innovative Materials 214 Magnetic properties and magnetocaloric effect in layered
PART 1 Introduction to Theory of Solids
 Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:1 Trim:165 240MM TS: Integra, India PART 1 Introduction to Theory of Solids Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:2
Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:1 Trim:165 240MM TS: Integra, India PART 1 Introduction to Theory of Solids Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:2
Nihal İKİZOĞLU. MOSELEY and MODERN PERIODIC TABLE (designed by atomic numbers of elements) kimyaakademi.com 1
 MOSELEY and MODERN PERIODIC TABLE (designed by atomic numbers of elements) kimyaakademi.com 1 PERIODS: Period number = Number of basic energy levels = The principal quantum number The horizontal lines
MOSELEY and MODERN PERIODIC TABLE (designed by atomic numbers of elements) kimyaakademi.com 1 PERIODS: Period number = Number of basic energy levels = The principal quantum number The horizontal lines
Earth Materials I Crystal Structures
 Earth Materials I Crystal Structures Isotopes same atomic number, different numbers of neutrons, different atomic mass. Ta ble 1-1. Su mmar y of quantu m num bers Name Symbol Values Principal n 1, 2,
Earth Materials I Crystal Structures Isotopes same atomic number, different numbers of neutrons, different atomic mass. Ta ble 1-1. Su mmar y of quantu m num bers Name Symbol Values Principal n 1, 2,
Conclusion. 109m Ag isomer showed that there is no such broadening. Because one can hardly
 Conclusion This small book presents a description of the results of studies performed over many years by our research group, which, in the best period, included 15 physicists and laboratory assistants
Conclusion This small book presents a description of the results of studies performed over many years by our research group, which, in the best period, included 15 physicists and laboratory assistants
Semiconductors 1. Explain different types of semiconductors in detail with necessary bond diagrams. Intrinsic semiconductors:
 Semiconductors 1. Explain different types of semiconductors in detail with necessary bond diagrams. There are two types of semi conductors. 1. Intrinsic semiconductors 2. Extrinsic semiconductors Intrinsic
Semiconductors 1. Explain different types of semiconductors in detail with necessary bond diagrams. There are two types of semi conductors. 1. Intrinsic semiconductors 2. Extrinsic semiconductors Intrinsic
ELEMENTARY BAND THEORY
 ELEMENTARY BAND THEORY PHYSICIST Solid state band Valence band, VB Conduction band, CB Fermi energy, E F Bloch orbital, delocalized n-doping p-doping Band gap, E g Direct band gap Indirect band gap Phonon
ELEMENTARY BAND THEORY PHYSICIST Solid state band Valence band, VB Conduction band, CB Fermi energy, E F Bloch orbital, delocalized n-doping p-doping Band gap, E g Direct band gap Indirect band gap Phonon
Engineering 2000 Chapter 8 Semiconductors. ENG2000: R.I. Hornsey Semi: 1
 Engineering 2000 Chapter 8 Semiconductors ENG2000: R.I. Hornsey Semi: 1 Overview We need to know the electrical properties of Si To do this, we must also draw on some of the physical properties and we
Engineering 2000 Chapter 8 Semiconductors ENG2000: R.I. Hornsey Semi: 1 Overview We need to know the electrical properties of Si To do this, we must also draw on some of the physical properties and we
Regan & Johnston Chemistry Unit 3 Exam: The Periodic Table Class Period
 Regan & Johnston Name Chemistry Unit 3 Exam: The Periodic Table Class Period 1. An atom of which element has the largest atomic radius? (1) Si (2) Fe (3) Zn (4) Mg 2. Which characteristics both generally
Regan & Johnston Name Chemistry Unit 3 Exam: The Periodic Table Class Period 1. An atom of which element has the largest atomic radius? (1) Si (2) Fe (3) Zn (4) Mg 2. Which characteristics both generally
Lecture 1. OUTLINE Basic Semiconductor Physics. Reading: Chapter 2.1. Semiconductors Intrinsic (undoped) silicon Doping Carrier concentrations
 Lecture 1 OUTLINE Basic Semiconductor Physics Semiconductors Intrinsic (undoped) silicon Doping Carrier concentrations Reading: Chapter 2.1 EE105 Fall 2007 Lecture 1, Slide 1 What is a Semiconductor? Low
Lecture 1 OUTLINE Basic Semiconductor Physics Semiconductors Intrinsic (undoped) silicon Doping Carrier concentrations Reading: Chapter 2.1 EE105 Fall 2007 Lecture 1, Slide 1 What is a Semiconductor? Low
EPR in Kagome Staircase Compound Mg Co V 2 O 8
 Vol. 111 (2007) ACTA PHYSICA POLONICA A No. 1 Proceedings of the Symposium K: Complex Oxide Materials for New Technologies of E-MRS Fall Meeting 2006, Warsaw, September 4 8, 2006 EPR in Kagome Staircase
Vol. 111 (2007) ACTA PHYSICA POLONICA A No. 1 Proceedings of the Symposium K: Complex Oxide Materials for New Technologies of E-MRS Fall Meeting 2006, Warsaw, September 4 8, 2006 EPR in Kagome Staircase
Homework Packet Unit 2. b. Al 3+, F, Na +, Mg 2+, O 2
 Name Period Homework Packet Unit 2 1. Which of the following is the correct empirical formula for a compound that has 37.5% C, 12.6% H, and 49.9% O? (A) C 2 H 4 O (B) CH 4 O 2 (C) CH 5 O 2 (D) CH 4 O (E)
Name Period Homework Packet Unit 2 1. Which of the following is the correct empirical formula for a compound that has 37.5% C, 12.6% H, and 49.9% O? (A) C 2 H 4 O (B) CH 4 O 2 (C) CH 5 O 2 (D) CH 4 O (E)
I. Introduction II. Solid State Physics Detection of Light Bernhard Brandl 1
 Detection of Light I. Introduction II. Solid State Physics 4-2-2015 Detection of Light Bernhard Brandl 1 4-2-2015 Detection of Light Bernhard Brandl 2 Blabla Recommended 4-2-2015 Detection of Light Bernhard
Detection of Light I. Introduction II. Solid State Physics 4-2-2015 Detection of Light Bernhard Brandl 1 4-2-2015 Detection of Light Bernhard Brandl 2 Blabla Recommended 4-2-2015 Detection of Light Bernhard
Fe Co Si. Fe Co Si. Ref. p. 59] d elements and C, Si, Ge, Sn or Pb Alloys and compounds with Ge
![Fe Co Si. Fe Co Si. Ref. p. 59] d elements and C, Si, Ge, Sn or Pb Alloys and compounds with Ge Fe Co Si. Fe Co Si. Ref. p. 59] d elements and C, Si, Ge, Sn or Pb Alloys and compounds with Ge](/thumbs/72/66917236.jpg) Ref. p. 59] 1.5. 3d elements and C, Si, Ge, Sn or Pb 7 1.75 1.50 Co Si 0.8 0. 3.50 3.5 Co Si 0.8 0. H cr Magnetic field H [koe] 1.5 1.00 0.75 0.50 0.5 C C IF "A" P Frequency ωγ / e [koe] 3.00.75.50.5.00
Ref. p. 59] 1.5. 3d elements and C, Si, Ge, Sn or Pb 7 1.75 1.50 Co Si 0.8 0. 3.50 3.5 Co Si 0.8 0. H cr Magnetic field H [koe] 1.5 1.00 0.75 0.50 0.5 C C IF "A" P Frequency ωγ / e [koe] 3.00.75.50.5.00
Bonding in Solids. What is the chemical bonding? Bond types: Ionic (NaCl vs. TiC?) Covalent Van der Waals Metallic
 Bonding in Solids What is the chemical bonding? Bond types: Ionic (NaCl vs. TiC?) Covalent Van der Waals Metallic 1 Ions and Ionic Radii LiCl 2 Ions (a) Ions are essentially spherical. (b) Ions may be
Bonding in Solids What is the chemical bonding? Bond types: Ionic (NaCl vs. TiC?) Covalent Van der Waals Metallic 1 Ions and Ionic Radii LiCl 2 Ions (a) Ions are essentially spherical. (b) Ions may be
CHEM 172 EXAMINATION 2. February 12, Dr. Kimberly M. Broekemeier NAME: l = 2r l = 8 1/2 r l = (4/3 1/2 )r. h = 6.
 EM 17 EXAMINATION February 1, 009 Dr. Kimberly M. Broekemeier NAME: P 1 1 P1 R T T1 ln = - ( - ) l = r l = 8 1/ r l = (4/3 1/ )r onstants: c = 3.00 X 10 8 m/s h = 6.63 X 10-34 J x s R = 0.0806 L x atm/mol
EM 17 EXAMINATION February 1, 009 Dr. Kimberly M. Broekemeier NAME: P 1 1 P1 R T T1 ln = - ( - ) l = r l = 8 1/ r l = (4/3 1/ )r onstants: c = 3.00 X 10 8 m/s h = 6.63 X 10-34 J x s R = 0.0806 L x atm/mol
Review of Semiconductor Physics
 Solid-state physics Review of Semiconductor Physics The daunting task of solid state physics Quantum mechanics gives us the fundamental equation The equation is only analytically solvable for a handful
Solid-state physics Review of Semiconductor Physics The daunting task of solid state physics Quantum mechanics gives us the fundamental equation The equation is only analytically solvable for a handful
Lecture 6 - Bonding in Crystals
 Lecture 6 onding in Crystals inding in Crystals (Kittel Ch. 3) inding of atoms to form crystals A crystal is a repeated array of atoms Why do they form? What are characteristic bonding mechanisms? How
Lecture 6 onding in Crystals inding in Crystals (Kittel Ch. 3) inding of atoms to form crystals A crystal is a repeated array of atoms Why do they form? What are characteristic bonding mechanisms? How
Introduction to Engineering Materials ENGR2000. Dr.Coates
 Introduction to Engineering Materials ENGR2000 Chapter 18: Electrical Properties Dr.Coates 18.2 Ohm s Law V = IR where R is the resistance of the material, V is the voltage and I is the current. l R A
Introduction to Engineering Materials ENGR2000 Chapter 18: Electrical Properties Dr.Coates 18.2 Ohm s Law V = IR where R is the resistance of the material, V is the voltage and I is the current. l R A
Drickamer type. Disk containing the specimen. Pressure cell. Press
 ε-fe Drickamer type Press Pressure cell Disk containing the specimen Low Temperature Cryostat Diamond Anvil Cell (DAC) Ruby manometry Re gasket for collimation Small size of specimen space High-density
ε-fe Drickamer type Press Pressure cell Disk containing the specimen Low Temperature Cryostat Diamond Anvil Cell (DAC) Ruby manometry Re gasket for collimation Small size of specimen space High-density
Recent progress in exploring magnetocaloric materials
 Recent progress in exploring magnetocaloric materials B. G. Shen,* J. R. Sun, F. X. Hu, H. W. Zhang, and Z. H. Cheng State Key Laboratory for Magnetism, Institute of Physics, Chinese Academy of Sciences,
Recent progress in exploring magnetocaloric materials B. G. Shen,* J. R. Sun, F. X. Hu, H. W. Zhang, and Z. H. Cheng State Key Laboratory for Magnetism, Institute of Physics, Chinese Academy of Sciences,
LN 3 IDLE MIND SOLUTIONS
 IDLE MIND SOLUTIONS 1. Let us first look in most general terms at the optical properties of solids with band gaps (E g ) of less than 4 ev, semiconductors by definition. The band gap energy (E g ) can
IDLE MIND SOLUTIONS 1. Let us first look in most general terms at the optical properties of solids with band gaps (E g ) of less than 4 ev, semiconductors by definition. The band gap energy (E g ) can
Chemistry Higher level Paper 1
 N15/4/EMI/PM/ENG/TZ0/XX hemistry igher level Paper 1 Friday 13 November 2015 (afternoon) 1 hour Instructions to candidates Do not open this examination paper until instructed to do so. Answer all the questions.
N15/4/EMI/PM/ENG/TZ0/XX hemistry igher level Paper 1 Friday 13 November 2015 (afternoon) 1 hour Instructions to candidates Do not open this examination paper until instructed to do so. Answer all the questions.
2011 CHEM 120: CHEMICAL REACTIVITY
 2011 CHEM 120: CHEMICAL REACTIVITY INORGANIC CHEMISTRY SECTION Lecturer: Dr. M.D. Bala Textbook by Petrucci, Harwood, Herring and Madura 15 Lectures (4/10-29/10) 3 Tutorials 1 Quiz 1 Take-home test https://chemintra.ukzn.ac.za/
2011 CHEM 120: CHEMICAL REACTIVITY INORGANIC CHEMISTRY SECTION Lecturer: Dr. M.D. Bala Textbook by Petrucci, Harwood, Herring and Madura 15 Lectures (4/10-29/10) 3 Tutorials 1 Quiz 1 Take-home test https://chemintra.ukzn.ac.za/
Periodic Relationships
 Periodic Relationships 1 Tabulation of Elements Mendeleev (1869) Arranged by mass Tabulation by chem.& physical properties Predicted missing elements and properties 2 Modern Periodic Table Argon vs. potassium
Periodic Relationships 1 Tabulation of Elements Mendeleev (1869) Arranged by mass Tabulation by chem.& physical properties Predicted missing elements and properties 2 Modern Periodic Table Argon vs. potassium
EECS130 Integrated Circuit Devices
 EECS130 Integrated Circuit Devices Professor Ali Javey 8/30/2007 Semiconductor Fundamentals Lecture 2 Read: Chapters 1 and 2 Last Lecture: Energy Band Diagram Conduction band E c E g Band gap E v Valence
EECS130 Integrated Circuit Devices Professor Ali Javey 8/30/2007 Semiconductor Fundamentals Lecture 2 Read: Chapters 1 and 2 Last Lecture: Energy Band Diagram Conduction band E c E g Band gap E v Valence
A semiconductor is an almost insulating material, in which by contamination (doping) positive or negative charge carriers can be introduced.
 Semiconductor A semiconductor is an almost insulating material, in which by contamination (doping) positive or negative charge carriers can be introduced. Page 2 Semiconductor materials Page 3 Energy levels
Semiconductor A semiconductor is an almost insulating material, in which by contamination (doping) positive or negative charge carriers can be introduced. Page 2 Semiconductor materials Page 3 Energy levels
Debye temperature Θ D K x = 3 83I
 substance: LaH x property: crystal structure, physical properties crystal structure cubic (O 5 h Fm3m) 55M, 57S, 59S semiconductor: x = 2.7 T < 239 K 79B, 79Z semiconductor: x = 3.0 T < 241 K 83I energy
substance: LaH x property: crystal structure, physical properties crystal structure cubic (O 5 h Fm3m) 55M, 57S, 59S semiconductor: x = 2.7 T < 239 K 79B, 79Z semiconductor: x = 3.0 T < 241 K 83I energy
Modified from: Larry Scheffler Lincoln High School IB Chemistry 1-2.1
 Modified from: Larry Scheffler Lincoln High School IB Chemistry 1-2.1 The development of the periodic table brought a system of order to what was otherwise an collection of thousands of pieces of information.
Modified from: Larry Scheffler Lincoln High School IB Chemistry 1-2.1 The development of the periodic table brought a system of order to what was otherwise an collection of thousands of pieces of information.
Test Topics: Periodic Table, Atomic Theory, Physical/Chemical Properties, Atom, Isotopes, Average Atomic Mass
 Elemental Properties Review Worksheet Test Topics: Periodic Table, Atomic Theory, Physical/Chemical Properties, Atom, Isotopes, Average Atomic Mass Periodic Table 1. List the element symbols for the following
Elemental Properties Review Worksheet Test Topics: Periodic Table, Atomic Theory, Physical/Chemical Properties, Atom, Isotopes, Average Atomic Mass Periodic Table 1. List the element symbols for the following
Lecture 2. Semiconductor Physics. Sunday 4/10/2015 Semiconductor Physics 1-1
 Lecture 2 Semiconductor Physics Sunday 4/10/2015 Semiconductor Physics 1-1 Outline Intrinsic bond model: electrons and holes Charge carrier generation and recombination Intrinsic semiconductor Doping:
Lecture 2 Semiconductor Physics Sunday 4/10/2015 Semiconductor Physics 1-1 Outline Intrinsic bond model: electrons and holes Charge carrier generation and recombination Intrinsic semiconductor Doping:
Atomic Structure & Interatomic Bonding
 Atomic Structure & Interatomic Bonding Chapter Outline Review of Atomic Structure Atomic Bonding Atomic Structure Atoms are the smallest structural units of all solids, liquids & gases. Atom: The smallest
Atomic Structure & Interatomic Bonding Chapter Outline Review of Atomic Structure Atomic Bonding Atomic Structure Atoms are the smallest structural units of all solids, liquids & gases. Atom: The smallest
Discovery of Elements. Dmitri Mendeleev Stanislao Canizzaro (1860) Modern Periodic Table. Henry Moseley. PT Background Information
 Discovery of Elements Development of the Periodic Table Chapter 5 Honors Chemistry 412 At the end of the 1700 s, only 30 elements had been isolated Included most currency metals and some nonmetals New
Discovery of Elements Development of the Periodic Table Chapter 5 Honors Chemistry 412 At the end of the 1700 s, only 30 elements had been isolated Included most currency metals and some nonmetals New
3 points in total correct molar mass 1 point, correct equation 1 point, correct numerical calculation starting from correct equation 1 point
 Problem 5. Bohemian garnet Bohemian garnet (pyrope) is a famous Czech blood coloured semi-precious stone. The chemical composition of natural garnets is expressed by the general stoichiometric formula
Problem 5. Bohemian garnet Bohemian garnet (pyrope) is a famous Czech blood coloured semi-precious stone. The chemical composition of natural garnets is expressed by the general stoichiometric formula
Atoms and The Periodic Table
 Atoms and The Periodic Table A. Early Models of the Atom 1. The earliest models of the atom came in the 5 th century B.C. when In the 4 th century, B.C., rejected this idea and proposed that earthly matter
Atoms and The Periodic Table A. Early Models of the Atom 1. The earliest models of the atom came in the 5 th century B.C. when In the 4 th century, B.C., rejected this idea and proposed that earthly matter
arxiv:nucl-ex/ v1 27 May 2002
 1 MEASUREMENTS OF NUCLEAR LEVEL DENSITIES AND γ-ray STRENGTH FUNCTIONS AND THEIR INTERPRETATIONS M. Guttormsen, M. Hjorth-Jensen, J. Rekstad and S. Siem Department of Physics, University of Oslo, Box 1048
1 MEASUREMENTS OF NUCLEAR LEVEL DENSITIES AND γ-ray STRENGTH FUNCTIONS AND THEIR INTERPRETATIONS M. Guttormsen, M. Hjorth-Jensen, J. Rekstad and S. Siem Department of Physics, University of Oslo, Box 1048
Exam Electrons and Periodic Table
 1-20 multiple choice. Answer on scantron. 21-25 short response. Answer on exam paper. All questions are 4 points each. 1. Which term is defined as the region in an atom where an electron is most likely
1-20 multiple choice. Answer on scantron. 21-25 short response. Answer on exam paper. All questions are 4 points each. 1. Which term is defined as the region in an atom where an electron is most likely
(Me=Mg, Zn) compounds. Solid State Physics, Department of Physics, University of Athens, Zografos, Athens, Greece
 EPR Rev.Adv.Mater.Sci. study of the Me14(7) 2 125-129 (Me=Mg, Zn) compounds 125 EPR STUDY OF THE COMPOUNDS (Me = Mg, Zn) Nikolaos Guskos 1,2, Grzegorz Zolnierkiewicz 2, Janusz Typek 2 and Monika Bosacka
EPR Rev.Adv.Mater.Sci. study of the Me14(7) 2 125-129 (Me=Mg, Zn) compounds 125 EPR STUDY OF THE COMPOUNDS (Me = Mg, Zn) Nikolaos Guskos 1,2, Grzegorz Zolnierkiewicz 2, Janusz Typek 2 and Monika Bosacka
PHYSICAL SCIENCES GRADE : 10
 PHYSICAL SCIENCES GRADE : 0 TIME : hour TOTAL : 75 INSTRUCTIONS AND INFORMATION. Write your full name on your answer book in the appropriate place. 2. The question paper consists of SEVEN questions. Answer
PHYSICAL SCIENCES GRADE : 0 TIME : hour TOTAL : 75 INSTRUCTIONS AND INFORMATION. Write your full name on your answer book in the appropriate place. 2. The question paper consists of SEVEN questions. Answer
The Periodic Table of Elements
 The Periodic Table of Elements 8 Uuo Uus Uuh (9) Uup (88) Uuq (89) Uut (8) Uub (8) Rg () 0 Ds (9) 09 Mt (8) 08 Hs (9) 0 h () 0 Sg () 0 Db () 0 Rf () 0 Lr () 88 Ra () 8 Fr () 8 Rn () 8 At (0) 8 Po (09)
The Periodic Table of Elements 8 Uuo Uus Uuh (9) Uup (88) Uuq (89) Uut (8) Uub (8) Rg () 0 Ds (9) 09 Mt (8) 08 Hs (9) 0 h () 0 Sg () 0 Db () 0 Rf () 0 Lr () 88 Ra () 8 Fr () 8 Rn () 8 At (0) 8 Po (09)
