Raman characterization of metal-alkanethiolates
|
|
|
- Brent Jacobs
- 6 years ago
- Views:
Transcription
1 Spectrochimica Acta Part A 55 (1999) Raman characterization of metal-alkanethiolates F. Bensebaa a, *, Y. Zhou b,c, A.G. Brolo d, D.E. Irish d, Y. Deslandes a, E. Kruus b, T.H. Ellis c a ICPET, NRC of Canada, Building M-12, Ottawa Ont. K1A OR6, Canada b Département de Chimie et de Biochimie, UQAM, Montréal Quc. H3C 3P8, Canada c Département de Chimie, Uni ersité de Montréal, Montréal Quc. HC3 3J7, Canada d Department of Chemistry, Uni ersity of Waterloo, Waterloo Ont. N2L 3G1, Canada Received 27 April 1998; received in revised form 9 September 1998; accepted 9 September 1998 Abstract Raman spectroscopy has been used to characterize neat alkanethiol and various metal-alkanethiolate materials. Neat alkanethiol gives rise to two C S stretching peaks at 662 and 735 cm 1, assigned to gauche and trans rotamers respectively. Only one C S stretching peak positioned at 725 cm 1 was found from CuC 12 and AgC 12 layered compounds, implying the absence of gauche rotamer near the thiolate group. An all-trans conformation of the chain is inferred from the peak position values of the C C stretching modes of CuC 12 and AgC 12 layered compounds. Gauche rotamers were observed in silver colloids capped with alkanethiolate Elsevier Science B.V. All rights reserved. 1. Introduction A new approach to the stabilization of gold nanoparticles using alkanethiol chains has been recently reported [1,2]. The possibility of controlling the size of the core metal [3], the length of the capping chain [4,5], the end group [2,6,7], and the nature of the metal [8 12] has opened a new range of fundamental studies and potential applications [13,14]. Nanoparticles with sizes ranging from 20 to 200 Å have been stabilized with HS(CH 2 ) n 1 CH 3 (abbreviated to C n ) chains [3]. * Corresponding author. Tel.: ; Fax: address: faird.bensebaa@nrc.ca (F. Bensebaa) The high curvature of these materials gives rise to substantial free inter-chain volume, allowing the possibility of the occurrence of the gauche conformation. However, as for the case of the densely packed self-assembled monolayers (SAMs) on flat substrate [15], very few gauche rotamers are found [4]. For example, the position of the infrared CH 2 anti-symmetric stretch ( as ) peak from gold nanoparticles capped with C n alkanethiolate was found at about 2918 cm 1 for n 11 [4]. This implies the prevalence of all-trans chains [5,16]. Interdigitation [4] and the faceted core metal [17] could play a critical role in attaining the conformational order in these nanoparticles capped alkanethiolate /99/$ - see front matter 1999 Elsevier Science B.V. All rights reserved. PII: S (98)
2 1230 F. Bensebaa et al. / Spectrochimica Acta Part A 55 (1999) Some structural differences between layered compound and nanoparticle in one hand and alkanethiolate self-assembled on flat substrate in the other hand have been suggested [3,4,9,11,12]. In particular, the difference in the metal sulfur ratio would lead to conformational difference around the thiolate group. Raman spectroscopy has been used in the past to characterize the conformation and structure of various alkanethiolates adsorbed on Ag and Au flat substrates [18 24]. Vibrational modes of S Au, S Ag, C S, and C C bonds give rise to characteristic Raman bands with features sensitive to the structural and conformational details. Sandroff et al. (1983) identified two different C S stretching peaks in the Raman spectra of an alkanethiolate monolayer [18]. Based on the C S peak position, they attributed these two peaks to gauche and to trans conformation of the C C bond near the C S bond [18]. Bryant and Pemberton (1991) [20] used the C S and C C peak positions to follow the conformational and structural changes of the alkanethiolate molecule as a function of the chain length and the degree of roughness of the metal substrate. An alternative approach to characterize metalalkanethiolate monolayers is to study related materials, which contain metal-thiolate bonds. Layered compounds composed of thiolate-metalthiolate sheets have been prepared and characterized [9,11,12,23]. Their planar structure and high degree of order make them excellent analogs for alkanethiolate monolayers. There are effectively no bulk metal atoms, as there is a 1:1 metal:sulfur stoichiometry [9,12]. An obvious advantage in studying theses new materials over the SAMs on flat substrate is the increase in surface area. Very few studies comparing directly the structural and conformational properties of nanoparticles and layered metal-alkanethiolate have been so far reported [11]. As schematically shown in Fig. 1, better packing is expected in layered compounds than with the corresponding nanoparticle material. The overall structure of these layered compounds is suggested to be similar to the flat substrate based alkanethiolate SAMs [9,23]. In the present study, Raman spectroscopy is used to characterize the conformational and structural properties of different metal-based alkanethiolate samples. Preliminary Raman results of various metal-alkanethiolate materials are reported in this paper. Particular attention will be paid to the following peculiarities; (i) the difference between Raman features of the neat alkanethiol molecule and the metal capped alkanethiolate; (ii) the influence of the metal nature (Cu, Ag, and Au) on the Raman features of these metal-alkanethiolate materials; (iii) the difference between Raman features of layered compounds and the corresponding colloid alkanethiolates. Fig. 1. Schematic representation of: (a) a layered silveralkanethiolate 9 ; and (b) colloidal gold alkanethiolate. 5 Open rectangle and open oval correspond to the metal layer sheet and the metal colloid respectively. Thick lines correspond to the alkanethiolate chains. Dimensions are not in scale.
3 F. Bensebaa et al. / Spectrochimica Acta Part A 55 (1999) Experimental Previously published recipes have been used to prepare gold colloids capped with C 12 and SH(CH 2 ) 15 CO 2 H (abbreviated to C 15 CO 2 H) [1], silver colloid capped with C 12 chain, and also 1:1 C 2 and C 12 silver layered compounds [12]. A new recipe has been devised to prepare the copper alkanethiolate layered compound [12]. In the following the 1:1 metal-alkanethiolate layered compound will be abbreviated by MC n, where M is either Ag or Cu, and n stands for the total number of carbon atoms within the alkanethiolate chain. These samples present some characteristic color, the AgC 2 layered compound is yellow, AgC 12 is pale yellow, and the CuC 12 compound is green. Silver nanoparticles capped with C 12 are dark grey, and gold colloids capped with C 12 or C 15 CO 2 H look black and shiny. Morphologically, the CuC 12 and AgC 12 layered compounds are also different. The silver layered compounds are powder-like. The copper layered compound is composed of sheets of about l mm thick and up to 1 cm long. More details on preparation and characterization of these materials can be found in a forthcoming paper [12]. For Raman analysis in the backscattering mode, samples were deposited on a glass microscope slide, and covered with a very thin glass slide. Raman spectra were measured with a Renishaw-1000 Raman microscope system equipped with a thermoelectrically cooled CCD array detector. Raman spectra were measured in two ranges, ( cm 1 ) and (0 800 cm 1 ). Raman peak positions are reproducible within 1 2 cm 1. A 50 times microscope objective lens with ca. 6 mm focal length was used. The spot size was about 1 m. A Melles Griot He Ne laser (wavelength of nm) was used as an excitation source. The maximum power at the laser head was 35 mw. At this power most of the silver layered compound burns and turns blackish at and around the laser spot. Neutral density filters, with optical density ranging from 0.3 (50% transmission) to 2 (1% transmission), were used to attenuate the laser power and consequently avoid decomposition. 3. Results and discussion Raman spectra from 50 to 3300 cm 1 of a neat alkanethiol powder and four metal alkanethiolate materials measured under similar conditions are displayed in Fig. 2. Also displayed in Fig. 3 are the Raman spectra of three of the samples recorded from 50 to 800 cm 1. The Raman spectrum of neat C 15 CO 2 H alkanethiol shows some characteristic lines (Fig. 2a). Lines attributed to; C H stretching (2921, 2883, and 2852 cm 1 ); S H stretching (2575 cm 1 ); C H deformation (1466, 1442 and 1411 cm 1 ); CH 2 Wagging (1370, 1296 and 1276 cm 1 ); C C stretching (1118 and 1061 cm 1 ); CH 2 rocking (708 and 905 cm 1 ) and; C S stretching (662 and 735 cm 1 ) modes have been detected. Most of these (and the following) assignments are based on previously reported investigations of neat alkanethiol and alkanethiolate self-assembled on flat metal substrates [18 24]. A summary of peak assignments of the different samples is given in Table 1. The Raman spectra of the different metal alkanethiolate materials show also some characteristic features. In contrast to the neat alkanethiol, no S H stretching peak is observed. This confirms the cleavage of the S H bond upon formation of the thiolate bond. The relative intensities of the different Raman peaks seem to vary quite drastically from one alkanethiolate material to another. Given the fact that the same volume of sample (defined by the laser spot size) is probed, the sample density (or the number of bonds per unit volume) is obviously a factor that could influence the Raman signal intensity. This could contribute to the overall more intense Raman signal observed on neat alkanethiol and on CuC 12 layered compound as well as to a weaker signal from colloids in comparison with 1:1 compounds. Besides sample density, the type of metal (Ag versus Cu) and the microscopic shape (nanoparticles versus. layered compound) may affect the Raman signal intensity. The copper layered compound contains some interesting Raman features. Besides the stretching, deformation and rocking peaks from CH 2 and CH 3 groups (see Table 1), sharp peaks assigned to C S stretching, C C stretching, and to wagging
4 1232 F. Bensebaa et al. / Spectrochimica Acta Part A 55 (1999) Fig. 2. Raman spectra of: (a) HSC 15 H 28 CO 2 H, (b) CuC 12 layered compound; (c) AgC 12 layered compound; (d) silver colloid capped with C 12 (or Ag*C 12 ); and (e) gold colloid capped with C 15 CO 2 H (or Au*C 15 CO 2 H). modes are also observed (Fig. 2b) at 725, 1062 (and 1128) and 1213 (1240, 1265, 1297, and 1320) cm 1, respectively. The presence of distinct and sharp peaks assigned to the wagging progression modes is a strong indication of high crystallinity [25]. In contrast, the Raman spectra of the two silver-based alkanethiolate samples are relatively different. Signal intensities are weaker especially in the case of the colloid samples. Except for the wagging progression bands, all other characteristic bands are not observed on the silver-alkanethiolate colloids. The absence of wagging bands may suggest that both the morphology and the type of the supporting metal affect the Raman signal. The trend in the signal-to-noise ratio (S/N) of the Raman spectra from the different metal-alkanethiolate materials is difficult to explain. The eventual occurrence of Raman signal enhancement in these layered compounds is a very interesting phenomenon. The intensity ratios I(C S)/I(C H) and I(C S)/I(C C) involving C S, C H and C C stretching peaks are estimated in the case of neat alkanethiol (Fig. 2a) and the copper layered compound alkanethiolate (Fig. 2b). Both ratios are found to be about five times larger in the CuC 12 layered compound. A similar Raman signal enhancement was also observed on the silver layered compound. In both cases, the signal enhancement is probably related to the charge transfer involving the sulfur and metal atoms, following the thiolate bond formation. This could explain why the signal enhancement is limited to the C S stretching peak. Furthermore, it has been shown in the past that the Raman intensity decreases with the distance between the bond and the interface [24]. Given that only a single atomic layer is present, we may speculate that this effect is probably more extreme in the layered compounds. However it is difficult to attribute this signal enhancement to a SERS (Surface enhanced Raman spectroscopy) effect [26 28] on these layered compounds. Firstly, the SERS effect has been coined as a surface phenomenon involving bulk metals or relatively large clusters. In the present study, only a single layer of atoms is involved [9,12,23]. Secondly, the enhancement factor is much weaker than what was observed on bulky metal surfaces. In the following, we will compare the prominent Raman bands of the different samples of alkanethiol and metal alkanethiolates.
5 F. Bensebaa et al. / Spectrochimica Acta Part A 55 (1999) Fig. 3. Raman spectra of AgC 2 layered compound, CuC 12 layered compound and gold colloid capped with C 12 (or Au*C 12 ) M S stretch region The metal sulfur (M S) stretching mode is critical, since the observation of its corresponding Raman peak is a unique direct spectroscopic proof of the thiolate bond formation. Vibrational peaks attributed to the M S stretching mode have been observed in the region between 200 and 235 cm 1. The relatively wide variation in the peak position of the M S stretching peak is not yet understood. Sexton and Nyberg (1986) reported an EELS (electron energy loss spectroscopy) vibrational peak at about 200 cm 1 from dimethyl sulfide adsorbed on Cu (100) [29]. They ascribed this peak to the Cu S stretching mode. Nuzzo et al. (1987) [30] also observed a strong band at 220 and 235 cm 1 on methanethiol and dimethyl disulfide adsorbed on Au (111), respectively. Using FT-SERS (Fourier transform surface enhanced Raman spectroscopy) Dai et al. (1995) [22] observed a peak at 218 cm 1 from phenyl disulfide adsorbed on a silver surface. Two factors could influence the M S peak position; (i) the nature of the metal and; (ii) the composition of the adsorbed molecule, particularly the type of chemical group near the sulfur. As shown in Fig. 3 very different Raman spectra features have been observed on three different metal alkanethiolates. Of the two peaks observed from the AgC 2 layered compound at 218 and 250 cm 1, only the former one is straightforwardly assigned to an S Ag stretching mode. The second peak is probably due to the C C S deformation [23,31 33]. It is worth noting that no Raman peak is observed between 200 and 220 cm 1 in the neat alkanethiol sample. At least two peaks (200 and 214 cm 1 ) are also observed in the M S stretching region on the CuC 12 layered compound. We could not presently explain the presence of these two peaks. The absence of a Raman peak from the S Au stretching mode is probably due to a weak Raman scattering for gold colloids. This could not be due to the absence of alkanethiolate. Indeed, a Raman peak is observed in the C H stretching region. Furthermore, XPS measurements of the same sample in the S2p spectral region have given binding energy values characteristics of thiolate species [11] C S stretch region Two C S stretching peaks positioned at 735 and 662 cm 1 were observed from the neat alka-
6 1234 F. Bensebaa et al. / Spectrochimica Acta Part A 55 (1999) Table 1 Position and assignment of the Raman peaks from neat alkanethiol and different alkanethiolate samples a HSC 15 CO 2 H CuC 12 AgC 12 Ag*C 12 Au*C 15 CO 2 H a M S 200 (215) CCC/CCS deformation 326; 424 (435) C S T d C S G 662 c 700 b 642 d CC T 1061; ; C C G CH 2 rocking 708; l4 c CH 2 wagging band pro- 1296; 1276 c ; 1213; 1240; 1265; 1297; 1320 c ; gression 1370 c 1342 CH deformation 1411 c ; 1442; ; 1450; ; 1441; 1429; 1442 l43l c 1464 SH stretch 2575 CH stretch 2852; 2883; ; 2884; ; ; ; 2910 a Silver or gold colloid capped with alkanethiolate. b Weak shoulder. c Weak peak. d Broad peak. nethiol sample. The high frequency mode (735 cm 1 ) is assigned to alkyl chains with the C C bond in the trans conformation [18,20]. The low frequency peak is assigned to the C S bond from the alkyl chain with the C C bond in the gauche conformation [18,20]. C and C correspond to the two nearest carbon atoms nearest to the sulfur atom. The relatively small peak at 708 cm 1 is probably due to the CH 2 rocking mode [20,34]. In most samples, the high frequency C S stretching peak is downshifted by at least 10 cm 1, upon thiolate formation. This observation confirms that a change occurred to the thiol group, as suggested by the disappearance of the S H Raman peak and the appearance of new Raman peaks in the M S stretching region. A similar observation has been reported in the case of alkanethiolate self-assembled on flat Ag and Au substrate [18 20]. This shift is probably due to a weakening of the C S bond following the charge transfer following the thiolate bond formation. The single C S peak observed on CuC 12 and AgC 12 layered compounds positioned at 725 cm 1 is attributed to the trans rotamer. This is in line with the geometrical model of the layered compound [9] showing ample similarity to SAMs [15]. As shown in Fig. 3, the AgC 2 layered compound contains at least two rotamers. Indeed two C S stretching peaks positioned at 644 and 720 cm 1 are observed. This observation contrasts with AgC 12 data showing only one band in this region. The low frequency band is observed only in small chain alkanethiolate. A similar feature has been observed in colloid alkanethiolate [34] and in alkanethiolate SAMs on flat substrate [15,16]. Most of the silver layered compounds based on short alkanethiolate chains (with n 7) look yellow or bright yellow in color. Based on the work of Fijolek et al. (1997) [23], this suggests that they contain gauche defects, and this seems consistent with the present work C C stretch region The Raman spectrum of neat alkanethiol in the C C stretching region shows the presence of two prominent peaks at 1061 and 1118 cm 1. They are attributed to trans rotamers. Two strong peaks (at 1062 an d 1128 cm 1 ), also attributed to the trans C C rotamers, are observed on the CuC 12 layered compound. In both samples a relatively weak peak is observed at 1084 cm 1. This peak is attributed to the gauche C C rotamers. Given the value of the C S stretching peak, it is
7 F. Bensebaa et al. / Spectrochimica Acta Part A 55 (1999) more likely that gauche rotamers are located at the chain end. Silver colloid capped with C 12 is the only sample with relatively high concentration of gauche rotamers. Indeed the Raman peak positioned at about 1080 cm 1 is more intense than the one at 1125 cm C H stretch and deformation region C H stretching and C H deformation modes give rise to very strong Raman peaks. The Raman bands are better defined for the copper and silver layered compounds than for the silver colloid sample. In the case of gold colloids, only weak C H stretching peaks have been detected using Raman. Particularly interesting is the difference between the C H stretching peak shape of layered and colloid silver alkanethiolate. Indeed, C H stretching peaks are better defined in the former sample. This is in agreement with IR data showing that higher order is obtained with the layered compounds [4,12,34]. 5. Conclusions We have studied a series of metal alkanethiolates using Raman spectroscopy. The results indicate that following the thiolate bond formation, the S H bond cleaves and new bands appear in the metal sulphur stretching region. Based on the value of the peak position of the C S stretching mode, we concluded that the rotamer near the thiolate group adopts the trans conformation, for both C 12 alkanethiolate colloidal and layered compound. However contrarily to the layered compound, the colloid sample contain some gauche rotamer. The relative intensity of the C S Raman peak increased by a factor of five following the thiolate bond formation in the case of CuC 12 and AgC 12 layered compounds. Such signal enhancement is still not well understood. Acknowledgements This work was supported by the National Research Council of Canada and by the Natural Sciences and Engineering Research Council of Canada. References [1] M. Brust, M. Walker, D. Bethell, D.J. Schiffrin, R. Whyman, J. Chem. Soc. Chem. Commun. (1994) [2] M. Brust, J. Fink, D. Bethel, D.J. Schiffrin, C. Kiely, J. Chem. Soc. Chem. Soc. Commun. (1995) [3] D.V. Leff, P.C. Ohara, J.R. Heath, W.M. Gelbart, J. Phys. Chem. 99 (1995) [4] A. Badia, L. Cuccia, L. Demers, F. Morin, R.B. Lennox, J. Am. Chem. Soc. 119 (1997) [5] R.H. Terrill, T.A. Postlethwaite, C. Chen, J. Am. Chem. Soc. 117 (1997) [6] D.V. Leff, L. Brandt, J.R. Heath, Langmuir 12 (1996) [7] K.S. Mayya, V. Partil, M. Sastry, Langmuir 13 (1997) [8] S. Murthy, T.P. Bigioni, Z.L. Wang, J.T. Khoury, R.L. Whetten, Mater. Lett. 30 (1997) [9] I.G. Dance, K.J. Fisher, R.M. Herath Banda, M.L. Scudder, Inorg. Chem. 30 (1992) [10] K.V. Sarathy, G.U. Kulkarni, C.N.R. Rao, Chem. Commun. (1997) [11] F. Bensebaa, Y. Zhou, Y. Deslandes, E. Kruus, T.H. Ellis, Surf. Sci. 405 (1998) [12] F. Bensebaa, T.H. Ellis, E. Kruus, R. Voicu, Y. Zhou, Can J. Chem. (in press). [13] A.P. Alivatos, Endeavou 21 (1997) [14] J.J. Storhoff, R.C. Mucic, C.A. Mirkin, J. Cluster Sci. 8 (1997) [15] A. Ulman, Chem. Rev. 96 (1996) [16] F. Bensebaa, C. Bakoyannis, T.H. Ellis, Mikrochim. Acta 14 (1997) [17] R.L. Whetten, J.T. Khoury, M.M. Alvarez, et al., Adv. Mater. 8 (1996) [18] C.S. Sandroff, S. Garoff, K.P. Leung, Chem. Phys. Lett. 96 (1983) [19] T.H. Joo, K. Kim, M.S. Kim, J. Mol. Struct. 158 (1987) [20] M.A. Byant, J.E. Pemberton, J. Am. Chem. Soc. 113 (1991) [21] S.D. Evans, T.L. Freeman, T.M. Flynn, D.N. Batchelder, A. Ulman, Thin Solid Films 244 (1994) [22] Q. Dai, C. Xue, G. Xue, L. Jiang, J. Adhes. Sci. Technol. 9 (1995) [23] H.G. Fijolek, J.R. Grohal, J.L. Sample, M.J. Natan, Inorg. Chem. 36 (1997) [24] M. Tsen, L. Sun, Anal. Chim. Acta 307 (1995) [25] L. Senak, D. Moore, R. Mendelsohn, J. Phys. Chem. 96 (1992)
8 1236 F. Bensebaa et al. / Spectrochimica Acta Part A 55 (1999) [26] E.S. Brandt, T.M. Cotton, Investigations of surfaces and interfaces, in: B.W. Rossier, R.C. Baetzold (Eds.), Physical Methods of Chemistry Volume IXB 2nd ed., Wiley, New York, 1993, pp [27] M. Moscovits, Rev. Mod. Phys. 57 (1985) [28] A.G. Brolo, D.E. Irish, B.D Smith, J. Mol. Struct. 405 (1997) [29] B.A. Sexton, G.L. Nyberg, Surf. Sci. 165 (1986) [30] R.G. Nuzzo, B.R. Zegarski, L.H. Dubois, J. Am. Chem. Soc. 109 (1987) [31] H.E. Van Wart, H.A. Scheraga, J. Phys. Chem. 80 (1976) [32] M. Hayshi, Y. Shiro, H. Murata, Bull. Chem. Soc. Jpn 39 (1966) [33] N. Nogami, H. Sugeta, T. Miyazawa, Bull. Chem. Soc. Jpn 48 (1975) [34] M.J. Hoestetler, J. Stokes, R.W. Murray, Langmuir 12 (1996)
Supplemental Information for
 Supplemental Information for Densely arranged two-dimensional silver nanoparticle assemblies with optical uniformity over vast areas as excellent surface-enhanced Raman scattering substrates Yoshimasa
Supplemental Information for Densely arranged two-dimensional silver nanoparticle assemblies with optical uniformity over vast areas as excellent surface-enhanced Raman scattering substrates Yoshimasa
Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering
 Supporting Information Cyclic Electroplating and Stripping of Silver on Au@SiO 2 Core/Shell Nanoparticles for Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering Dan Li a, Da-Wei Li
Supporting Information Cyclic Electroplating and Stripping of Silver on Au@SiO 2 Core/Shell Nanoparticles for Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering Dan Li a, Da-Wei Li
Highly efficient SERS test strips
 Electronic Supplementary Information (ESI) for Highly efficient SERS test strips 5 Ran Zhang, a Bin-Bin Xu, a Xue-Qing Liu, a Yong-Lai Zhang, a Ying Xu, a Qi-Dai Chen, * a and Hong-Bo Sun* a,b 5 10 Experimental
Electronic Supplementary Information (ESI) for Highly efficient SERS test strips 5 Ran Zhang, a Bin-Bin Xu, a Xue-Qing Liu, a Yong-Lai Zhang, a Ying Xu, a Qi-Dai Chen, * a and Hong-Bo Sun* a,b 5 10 Experimental
Structure and Thermodynamics of Self-Assembled Monolayers on Gold Nanocrystallites
 6566 J. Phys. Chem. B 1998, 102, 6566-6572 Structure and Thermodynamics of Self-Assembled Monolayers on Gold Nanocrystallites W. D. Luedtke and Uzi Landman* School of Physics, Georgia Institute of Technology,
6566 J. Phys. Chem. B 1998, 102, 6566-6572 Structure and Thermodynamics of Self-Assembled Monolayers on Gold Nanocrystallites W. D. Luedtke and Uzi Landman* School of Physics, Georgia Institute of Technology,
Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation
 Supplementary Information Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation Yu Liu, Shuping Xu, Haibo Li, Xiaoguang Jian, Weiqing Xu* State Key
Supplementary Information Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation Yu Liu, Shuping Xu, Haibo Li, Xiaoguang Jian, Weiqing Xu* State Key
Self-Assembled Monolayers of Alkanethiols on Clean Copper Surfaces
 748 Bull. Korean Chem. Soc. 2001, Vol. 22, No. 7 Myung M. Sung and Yunsoo Kim Self-Assembled Monolayers of Alkanethiols on Clean Copper Surfaces Myung M. Sung * and Yunsoo Kim Department of Chemistry,
748 Bull. Korean Chem. Soc. 2001, Vol. 22, No. 7 Myung M. Sung and Yunsoo Kim Self-Assembled Monolayers of Alkanethiols on Clean Copper Surfaces Myung M. Sung * and Yunsoo Kim Department of Chemistry,
Supporting Information s for
 Supporting Information s for # Self-assembling of DNA-templated Au Nanoparticles into Nanowires and their enhanced SERS and Catalytic Applications Subrata Kundu* and M. Jayachandran Electrochemical Materials
Supporting Information s for # Self-assembling of DNA-templated Au Nanoparticles into Nanowires and their enhanced SERS and Catalytic Applications Subrata Kundu* and M. Jayachandran Electrochemical Materials
3D Monolayers of 1,4-Benzenedimethanethiol on Au and Ag Clusters: Distinct Difference in Adsorption Geometry with the Corresponding 2D Monolayers
 Journal of Colloid and Interface Science 216, 134 142 (1999) Article ID jcis.1999.6260, available online at http://www.idealibrary.com on 3D Monolayers of 1,4-Benzenedimethanethiol on Au and Ag Clusters:
Journal of Colloid and Interface Science 216, 134 142 (1999) Article ID jcis.1999.6260, available online at http://www.idealibrary.com on 3D Monolayers of 1,4-Benzenedimethanethiol on Au and Ag Clusters:
Supporting Information
 Supporting Information Highly Sensitive, Reproducible, and Stable SERS Sensors Based on Well-Controlled Silver Nanoparticles Decorated Silicon Nanowire Building Blocks Xue Mei Han, Hui Wang, Xue Mei Ou,
Supporting Information Highly Sensitive, Reproducible, and Stable SERS Sensors Based on Well-Controlled Silver Nanoparticles Decorated Silicon Nanowire Building Blocks Xue Mei Han, Hui Wang, Xue Mei Ou,
HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS
 www.arpapress.com/volumes/vol19issue1/ijrras_19_1_06.pdf HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS M. Eslamifar Physics Department, BehbahanKhatamAl-Anbia
www.arpapress.com/volumes/vol19issue1/ijrras_19_1_06.pdf HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS M. Eslamifar Physics Department, BehbahanKhatamAl-Anbia
Gold nanoparticles surface-terminated with bifunctional ligands
 Colloids and Surfaces A: Physicochem. Eng. Aspects 246 (2004) 109 113 Gold nanoparticles surface-terminated with bifunctional ligands Weili Shi a, Y. Sahoo b, Mark T. Swihart a, a Department of Chemical
Colloids and Surfaces A: Physicochem. Eng. Aspects 246 (2004) 109 113 Gold nanoparticles surface-terminated with bifunctional ligands Weili Shi a, Y. Sahoo b, Mark T. Swihart a, a Department of Chemical
Chapter - 5 SELF ASSEMBLED MONOLAYERS
 Chapter - 5 SELF ASSEMBLED MONOLAYERS (a) (b) (c) Figure 1. Langmuir Blodgett methodology. a) Surfactant in water. The black dot represents th head and the line represents the tail. The molecules are disordered.
Chapter - 5 SELF ASSEMBLED MONOLAYERS (a) (b) (c) Figure 1. Langmuir Blodgett methodology. a) Surfactant in water. The black dot represents th head and the line represents the tail. The molecules are disordered.
Self-Assembled Monolayers
 Self-Assembled Monolayers Literature and Further Information Surface Chemistry: www.chem.qmul.ac.uk/surfaces/scc/ www.nottingham.ac.uk/~ppzpjm/amshome.htm venables.asu.edu/grad/lectures.html SAM s: www.ifm.liu.se/applphys/ftir/sams.html
Self-Assembled Monolayers Literature and Further Information Surface Chemistry: www.chem.qmul.ac.uk/surfaces/scc/ www.nottingham.ac.uk/~ppzpjm/amshome.htm venables.asu.edu/grad/lectures.html SAM s: www.ifm.liu.se/applphys/ftir/sams.html
Scanned Chemical Enhancement of Surface-Enhanced Raman. Scattering Using a Charge-Transfer Complex
 Supporting Information Scanned Chemical Enhancement of Surface-Enhanced Raman Scattering Using a Charge-Transfer Complex Wei Ji, a Xiangxin Xue, a Weidong Ruan, a Chunxu Wang, a Nan Ji, a Lei Chen, a Zhishi
Supporting Information Scanned Chemical Enhancement of Surface-Enhanced Raman Scattering Using a Charge-Transfer Complex Wei Ji, a Xiangxin Xue, a Weidong Ruan, a Chunxu Wang, a Nan Ji, a Lei Chen, a Zhishi
Supporting information
 Supporting information Polymer-Single-Crystal@Nanoparticle Nanosandwich for Surface Enhanced Raman Spectroscopy Bin Dong, Wenda Wang, David L. Miller, Christopher Y. Li* Department of Material Science
Supporting information Polymer-Single-Crystal@Nanoparticle Nanosandwich for Surface Enhanced Raman Spectroscopy Bin Dong, Wenda Wang, David L. Miller, Christopher Y. Li* Department of Material Science
Supplementary Figure 1 Experimental setup for crystal growth. Schematic drawing of the experimental setup for C 8 -BTBT crystal growth.
 Supplementary Figure 1 Experimental setup for crystal growth. Schematic drawing of the experimental setup for C 8 -BTBT crystal growth. Supplementary Figure 2 AFM study of the C 8 -BTBT crystal growth
Supplementary Figure 1 Experimental setup for crystal growth. Schematic drawing of the experimental setup for C 8 -BTBT crystal growth. Supplementary Figure 2 AFM study of the C 8 -BTBT crystal growth
Supplementary Information
 Electronic Supplementary Material (ESI) for Lab on a Chip. This journal is The Royal Society of Chemistry 217 Supplementary Information Continuous Fabrication of Plasmonic Photonic Microcapsules with Controllable
Electronic Supplementary Material (ESI) for Lab on a Chip. This journal is The Royal Society of Chemistry 217 Supplementary Information Continuous Fabrication of Plasmonic Photonic Microcapsules with Controllable
PECULIARITIES OF THE SERS SPECTRA OF THE HYDROQUINONE MOLECULE ADSORBED ON TITANIUM DIOXID
 PECULIARITIES OF THE SERS SPECTRA OF THE HYDROQUINONE MOLECULE ADSORBED ON TITANIUM DIOXID A.M. Polubotko *, V.P. Chelibanov** *A.F. Ioffe Physico-Technical Institute Russian Academy of Sciences, Politechnicheskaya
PECULIARITIES OF THE SERS SPECTRA OF THE HYDROQUINONE MOLECULE ADSORBED ON TITANIUM DIOXID A.M. Polubotko *, V.P. Chelibanov** *A.F. Ioffe Physico-Technical Institute Russian Academy of Sciences, Politechnicheskaya
Light-Controlled Shrinkage of Large-Area Gold Nanoparticles Monolayer Film for Tunable SERS Activity
 Light-Controlled Shrinkage of Large-Area Gold Nanoparticles Monolayer Film for Tunable SERS Activity Xuefei Lu a,b, Youju Huang b,c,d, *, Baoqing Liu a,b, Lei Zhang b,c, Liping Song b,c, Jiawei Zhang b,c,
Light-Controlled Shrinkage of Large-Area Gold Nanoparticles Monolayer Film for Tunable SERS Activity Xuefei Lu a,b, Youju Huang b,c,d, *, Baoqing Liu a,b, Lei Zhang b,c, Liping Song b,c, Jiawei Zhang b,c,
International Journal of Scientific & Engineering Research, Volume 5, Issue 3, March-2014 ISSN
 156 Copper Nanoparticles: Green Synthesis Characterization Y.Suresh*1, S.Annapurna*2, G.Bhikshamaiah*3, A.K.Singh#4 Abstract Present work describes the synthesis nanoparticles using papaya extract as a
156 Copper Nanoparticles: Green Synthesis Characterization Y.Suresh*1, S.Annapurna*2, G.Bhikshamaiah*3, A.K.Singh#4 Abstract Present work describes the synthesis nanoparticles using papaya extract as a
Using Visible Laser Based Raman Spectroscopy to Identify the Surface Polarity of Silicon Carbide
 Supporting Information for Using Visible Laser Based Raman Spectroscopy to Identify the Surface Polarity of Silicon Carbide Submitted by Yi-Chuan Tseng, a Yu-Chia Cheng, a Yang-Chun Lee, a Dai-Liang Ma,
Supporting Information for Using Visible Laser Based Raman Spectroscopy to Identify the Surface Polarity of Silicon Carbide Submitted by Yi-Chuan Tseng, a Yu-Chia Cheng, a Yang-Chun Lee, a Dai-Liang Ma,
Supporting Information for J. Am. Chem. Soc., 1991, 113(19), , DOI: /ja00019a011 LAIBINIS
 Supporting Information for J. Am. Chem. Soc., 1991, 113(19), 7152-7167, DOI: 10.1021/ja00019a011 LAIBINIS 7152-7167 I Terms& Conditions Electronic Supporting Information files are available without a subscription
Supporting Information for J. Am. Chem. Soc., 1991, 113(19), 7152-7167, DOI: 10.1021/ja00019a011 LAIBINIS 7152-7167 I Terms& Conditions Electronic Supporting Information files are available without a subscription
Supplementary Information for. Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings
 Supplementary Information for Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings Supplementary Figure 1. Simulated from pristine graphene gratings at different Fermi energy
Supplementary Information for Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings Supplementary Figure 1. Simulated from pristine graphene gratings at different Fermi energy
The Student Seminar Series. Jinseok Heo. Recent Applications of Surface Enhanced Raman Scattering in Analytical Chemistry
 Abstract The Student Seminar Series Presents a seminar by Jinseok Heo Department of Chemistry Texas A&M University Recent Applications of Surface Enhanced Raman Scattering in Analytical Chemistry 4:00
Abstract The Student Seminar Series Presents a seminar by Jinseok Heo Department of Chemistry Texas A&M University Recent Applications of Surface Enhanced Raman Scattering in Analytical Chemistry 4:00
Supplementary Information. depending on the atomic thickness of intrinsic and chemically doped. MoS 2
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supplementary Information Confocal absorption spectral imaging of MoS 2 : Optical transitions
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supplementary Information Confocal absorption spectral imaging of MoS 2 : Optical transitions
Mercury(II) detection by SERS based on a single gold microshell
 Mercury(II) detection by SERS based on a single gold microshell D. Han, S. Y. Lim, B. J. Kim, L. Piao and T. D. Chung* Department of Chemistry, Seoul National University, Seoul, Korea. 2010, 46, 5587-558
Mercury(II) detection by SERS based on a single gold microshell D. Han, S. Y. Lim, B. J. Kim, L. Piao and T. D. Chung* Department of Chemistry, Seoul National University, Seoul, Korea. 2010, 46, 5587-558
Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering
 Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering Yukun Han, 1 Hai Xiao, 2 and Hai-Lung Tsai 1, * 1 Department of Mechanical and Aerospace
Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering Yukun Han, 1 Hai Xiao, 2 and Hai-Lung Tsai 1, * 1 Department of Mechanical and Aerospace
Vibrational Spectroscopies. C-874 University of Delaware
 Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
CHAPTER 5. FORMATION OF SAMs CONRTOLLED BY STERIC EFFECTS. The steric effect is an important subject in chemistry. It arises from the fact that
 CHAPTER 5 FRMATIN F SAMs CNRTLLED BY STERIC EFFECTS 5.1 Motivation The steric effect is an important subject in chemistry. It arises from the fact that each atom within a molecule occupies a certain volume
CHAPTER 5 FRMATIN F SAMs CNRTLLED BY STERIC EFFECTS 5.1 Motivation The steric effect is an important subject in chemistry. It arises from the fact that each atom within a molecule occupies a certain volume
Imaging Methods: Scanning Force Microscopy (SFM / AFM)
 Imaging Methods: Scanning Force Microscopy (SFM / AFM) The atomic force microscope (AFM) probes the surface of a sample with a sharp tip, a couple of microns long and often less than 100 Å in diameter.
Imaging Methods: Scanning Force Microscopy (SFM / AFM) The atomic force microscope (AFM) probes the surface of a sample with a sharp tip, a couple of microns long and often less than 100 Å in diameter.
Surface-enhanced raman scattering from a layer of gold nanoparticles
 VNU Journal of Science, Mathematics - Physics 26 (2010) 187-192 Surface-enhanced raman scattering from a layer of gold nanoparticles Nguyen The Binh *, Nguyen Thanh Dinh, Nguyen Quang Dong, Vu Thi Khanh
VNU Journal of Science, Mathematics - Physics 26 (2010) 187-192 Surface-enhanced raman scattering from a layer of gold nanoparticles Nguyen The Binh *, Nguyen Thanh Dinh, Nguyen Quang Dong, Vu Thi Khanh
Characterisation of vibrational modes of adsorbed species
 17.7.5 Characterisation of vibrational modes of adsorbed species Infrared spectroscopy (IR) See Ch.10. Infrared vibrational spectra originate in transitions between discrete vibrational energy levels of
17.7.5 Characterisation of vibrational modes of adsorbed species Infrared spectroscopy (IR) See Ch.10. Infrared vibrational spectra originate in transitions between discrete vibrational energy levels of
Supporting Information
 Supporting Information Wiley-VCH 2011 69451 Weinheim, Germany Silver Nanocrystals with Concave Surfaces and Their Optical and Surface-Enhanced Raman Scattering Properties** Xiaohu Xia, Jie Zeng, Brenden
Supporting Information Wiley-VCH 2011 69451 Weinheim, Germany Silver Nanocrystals with Concave Surfaces and Their Optical and Surface-Enhanced Raman Scattering Properties** Xiaohu Xia, Jie Zeng, Brenden
Evaluating nanogaps in Ag and Au nanoparticle clusters for SERS applications using COMSOL Multiphysics
 Evaluating nanogaps in Ag and Au nanoparticle clusters for SERS applications using COMSOL Multiphysics Ramesh Asapu 1, Radu-George Ciocarlan 2, Nathalie Claes 3, Natan Blommaerts 1, Sara Bals 3, Pegie
Evaluating nanogaps in Ag and Au nanoparticle clusters for SERS applications using COMSOL Multiphysics Ramesh Asapu 1, Radu-George Ciocarlan 2, Nathalie Claes 3, Natan Blommaerts 1, Sara Bals 3, Pegie
Shell-isolated nanoparticle-enhanced Raman spectroscopy
 Shell-isolated nanoparticle-enhanced Raman spectroscopy Jian Feng Li, Yi Fan Huang, Yong Ding, Zhi Lin Yang, Song Bo Li, Xiao Shun Zhou, Feng Ru Fan, Wei Zhang, Zhi You Zhou, De Yin Wu, Bin Ren, Zhong
Shell-isolated nanoparticle-enhanced Raman spectroscopy Jian Feng Li, Yi Fan Huang, Yong Ding, Zhi Lin Yang, Song Bo Li, Xiao Shun Zhou, Feng Ru Fan, Wei Zhang, Zhi You Zhou, De Yin Wu, Bin Ren, Zhong
Gold nanothorns macroporous silicon hybrid structure: a simple and ultrasensitive platform for SERS
 Supporting Information Gold nanothorns macroporous silicon hybrid structure: a simple and ultrasensitive platform for SERS Kamran Khajehpour,* a Tim Williams, b,c Laure Bourgeois b,d and Sam Adeloju a
Supporting Information Gold nanothorns macroporous silicon hybrid structure: a simple and ultrasensitive platform for SERS Kamran Khajehpour,* a Tim Williams, b,c Laure Bourgeois b,d and Sam Adeloju a
Bi-Axial Growth Mode of Au-TTF Nanowires. Induced by Tilted Molecular Column Stacking
 Supporting Information Bi-Axial Growth Mode of Au-TTF Nanowires Induced by Tilted Molecular Column Stacking Yanlong Xing Eugen Speiser * Dheeraj K. Singh Petra S. Dittrich and Norbert Esser Leibniz-Institut
Supporting Information Bi-Axial Growth Mode of Au-TTF Nanowires Induced by Tilted Molecular Column Stacking Yanlong Xing Eugen Speiser * Dheeraj K. Singh Petra S. Dittrich and Norbert Esser Leibniz-Institut
ECE280: Nano-Plasmonics and Its Applications. Week8
 ECE280: Nano-Plasmonics and Its Applications Week8 Surface Enhanced Raman Scattering (SERS) and Surface Plasmon Amplification by Stimulated Emission of Radiation (SPASER) Raman Scattering Chandrasekhara
ECE280: Nano-Plasmonics and Its Applications Week8 Surface Enhanced Raman Scattering (SERS) and Surface Plasmon Amplification by Stimulated Emission of Radiation (SPASER) Raman Scattering Chandrasekhara
Heterogeneity of molecular structure of Ag photo-diffused Ge 30 Se 70 thin films
 Available online at www.sciencedirect.com Journal of Non-Crystalline Solids 354 (2008) 2719 2723 www.elsevier.com/locate/jnoncrysol Heterogeneity of molecular structure of Ag photo-diffused Ge 30 Se 70
Available online at www.sciencedirect.com Journal of Non-Crystalline Solids 354 (2008) 2719 2723 www.elsevier.com/locate/jnoncrysol Heterogeneity of molecular structure of Ag photo-diffused Ge 30 Se 70
The CdS and CdMnS nanocrystals have been characterized using UV-visible spectroscopy, TEM, FTIR, Particle Size Measurement and Photoluminiscence.
 Synthesis of CdS and CdMns Nanocrystals in Organic phase Usha Raghavan HOD, Dept of Information Technology VPM s Polytechnic, Thane Maharashtra Email id: usharagha@gmail.com Abstract: The present work
Synthesis of CdS and CdMns Nanocrystals in Organic phase Usha Raghavan HOD, Dept of Information Technology VPM s Polytechnic, Thane Maharashtra Email id: usharagha@gmail.com Abstract: The present work
Università degli Studi di Bari "Aldo Moro"
 Università degli Studi di Bari "Aldo Moro" Table of contents 1. Introduction to Atomic Force Microscopy; 2. Introduction to Raman Spectroscopy; 3. The need for a hybrid technique Raman AFM microscopy;
Università degli Studi di Bari "Aldo Moro" Table of contents 1. Introduction to Atomic Force Microscopy; 2. Introduction to Raman Spectroscopy; 3. The need for a hybrid technique Raman AFM microscopy;
Fast ph-assisted functionalization of silver nanoparticles with monothiolated DNA
 Supporting Information for Fast ph-assisted functionalization of silver nanoparticles with monothiolated DNA Xu Zhang ab, Mark R. Servos b, and Juewen Liu* a a Department of Chemistry and Waterloo Institute
Supporting Information for Fast ph-assisted functionalization of silver nanoparticles with monothiolated DNA Xu Zhang ab, Mark R. Servos b, and Juewen Liu* a a Department of Chemistry and Waterloo Institute
Spectroscopy at nanometer scale
 Spectroscopy at nanometer scale 1. Physics of the spectroscopies 2. Spectroscopies for the bulk materials 3. Experimental setups for the spectroscopies 4. Physics and Chemistry of nanomaterials Various
Spectroscopy at nanometer scale 1. Physics of the spectroscopies 2. Spectroscopies for the bulk materials 3. Experimental setups for the spectroscopies 4. Physics and Chemistry of nanomaterials Various
Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using
 Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using 633 nm laser excitation at different powers and b) the
Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using 633 nm laser excitation at different powers and b) the
Understanding Aqueous Dispersibility of Graphene Oxide and Reduced Graphene Oxide through pka Measurements
 Understanding Aqueous Dispersibility of Graphene xide and Reduced Graphene xide through pka Measurements Bharathi Konkena and Sukumaran Vasudevan * Department of Inorganic and Physical Chemistry Indian
Understanding Aqueous Dispersibility of Graphene xide and Reduced Graphene xide through pka Measurements Bharathi Konkena and Sukumaran Vasudevan * Department of Inorganic and Physical Chemistry Indian
Supporting Information. for. Angew. Chem. Int. Ed. Z Wiley-VCH 2003
 Supporting Information for Angew. Chem. Int. Ed. Z52074 Wiley-VCH 2003 69451 Weinheim, Germany Kinetic and Thermodynamic Control via Chemical Bond Rearrangement on Si(001) Surface Chiho Hamai, Akihiko
Supporting Information for Angew. Chem. Int. Ed. Z52074 Wiley-VCH 2003 69451 Weinheim, Germany Kinetic and Thermodynamic Control via Chemical Bond Rearrangement on Si(001) Surface Chiho Hamai, Akihiko
Bincy Jose, Colm T. Mallon, Robert J. Forster & Tia E. Keyes School of Chemical Sciences, Dublin City University, Dublin 9, Ireland
 Supplementary material for The Application of Selective Surface Modification of Nanocavities Arrays to Compare Surface vs Cavity Plasmons in SERS enhancement Bincy Jose, Colm T. Mallon, Robert J. Forster
Supplementary material for The Application of Selective Surface Modification of Nanocavities Arrays to Compare Surface vs Cavity Plasmons in SERS enhancement Bincy Jose, Colm T. Mallon, Robert J. Forster
Electronic supplementary information for:
 Electronic supplementary information for: Charge-transfer-induced suppression of galvanic replacement and synthesis of (Au@Ag)@Au double shell nanoparticles for highly uniform, robust and sensitive bioprobes
Electronic supplementary information for: Charge-transfer-induced suppression of galvanic replacement and synthesis of (Au@Ag)@Au double shell nanoparticles for highly uniform, robust and sensitive bioprobes
Dithiocarbamate Self-Assembled Monolayers as Efficient Surface Modifiers for Low Work Function Noble Metals
 Dithiocarbamate Self-Assembled Monolayers as Efficient Surface Modifiers for Low Work Function Noble Metals Dominik Meyer*,1, Tobias Schäfer 1, Philip Schulz 1,2,3, Sebastian Jung 1, Daniel Mokros 1, Ingolf
Dithiocarbamate Self-Assembled Monolayers as Efficient Surface Modifiers for Low Work Function Noble Metals Dominik Meyer*,1, Tobias Schäfer 1, Philip Schulz 1,2,3, Sebastian Jung 1, Daniel Mokros 1, Ingolf
Luminescent molecular clusters of noble metals
 Established in 1959 Luminescent molecular clusters of noble metals T. Pradeep Department of Chemistry Indian Institute of Technology Madras Chennai 600 036 pradeep@iitm.ac.in Quantum clusters: Au 25, Au
Established in 1959 Luminescent molecular clusters of noble metals T. Pradeep Department of Chemistry Indian Institute of Technology Madras Chennai 600 036 pradeep@iitm.ac.in Quantum clusters: Au 25, Au
Gold and Silver Nanoparticles Functionalized by the Adsorption of Dialkyl Disulfides
 7378 Langmuir 1998, 14, 7378-7386 Gold and Silver Nanoparticles Functionalized by the Adsorption of Dialkyl Disulfides Lon A. Porter, Jr., David Ji, Sarah L. Westcott, Michael Graupe, Roman S. Czernuszewicz,
7378 Langmuir 1998, 14, 7378-7386 Gold and Silver Nanoparticles Functionalized by the Adsorption of Dialkyl Disulfides Lon A. Porter, Jr., David Ji, Sarah L. Westcott, Michael Graupe, Roman S. Czernuszewicz,
Formation of complex films with water-soluble CTAB molecules
 Spectrochimica Acta Part A 65 (2006) 628 632 Formation of complex films with water-soluble CTAB molecules S. Biswas a,b, S.A. Hussain a,s.deb a, R.K. Nath b, D. Bhattacharjee a, a Department of Physics,
Spectrochimica Acta Part A 65 (2006) 628 632 Formation of complex films with water-soluble CTAB molecules S. Biswas a,b, S.A. Hussain a,s.deb a, R.K. Nath b, D. Bhattacharjee a, a Department of Physics,
Simple Method for the Preparation of
 JOURNAL OF RAMAN SPECTROSCOPY, VOL. 9, 359È363 (1998) Simple Method for the Preparation of Surface-Enhanced Raman-Active Gold Films N. Sandhyarani, K. V. G. K. Murty and T. Pradeep* Department of Chemistry
JOURNAL OF RAMAN SPECTROSCOPY, VOL. 9, 359È363 (1998) Simple Method for the Preparation of Surface-Enhanced Raman-Active Gold Films N. Sandhyarani, K. V. G. K. Murty and T. Pradeep* Department of Chemistry
RamanStation 400: a Versatile Platform for SERS Analysis
 FIELD APPLICATION REPORT Raman Spectroscopy Author: Dean H. Brown PerkinElmer, Inc. Shelton, CT USA RamanStation 400 RamanStation 400: a Versatile Platform for SERS Analysis Introduction Surface Enhanced
FIELD APPLICATION REPORT Raman Spectroscopy Author: Dean H. Brown PerkinElmer, Inc. Shelton, CT USA RamanStation 400 RamanStation 400: a Versatile Platform for SERS Analysis Introduction Surface Enhanced
Influence of temperature and voltage on electrochemical reduction of graphene oxide
 Bull. Mater. Sci., Vol. 37, No. 3, May 2014, pp. 629 634. Indian Academy of Sciences. Influence of temperature and voltage on electrochemical reduction of graphene oxide XIUQIANG LI, DONG ZHANG*, PEIYING
Bull. Mater. Sci., Vol. 37, No. 3, May 2014, pp. 629 634. Indian Academy of Sciences. Influence of temperature and voltage on electrochemical reduction of graphene oxide XIUQIANG LI, DONG ZHANG*, PEIYING
Supporting Information
 Supporting Information Direct Chemical Vapor Deposition-Derived Graphene Glasses Targeting Wide Ranged Applications Jingyu Sun, Yubin Chen, Manish Kr. Priydarshi, Zhang Chen, Alicja Bachmatiuk,, Zhiyu
Supporting Information Direct Chemical Vapor Deposition-Derived Graphene Glasses Targeting Wide Ranged Applications Jingyu Sun, Yubin Chen, Manish Kr. Priydarshi, Zhang Chen, Alicja Bachmatiuk,, Zhiyu
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Facile Synthesis of High Quality Graphene Nanoribbons Liying Jiao, Xinran Wang, Georgi Diankov, Hailiang Wang & Hongjie Dai* Supplementary Information 1. Photograph of graphene
SUPPLEMENTARY INFORMATION Facile Synthesis of High Quality Graphene Nanoribbons Liying Jiao, Xinran Wang, Georgi Diankov, Hailiang Wang & Hongjie Dai* Supplementary Information 1. Photograph of graphene
Electrostatic interactions to modulate the reflective assembly of nanoparticles at the oilwater
 Supplemental Information Electrostatic interactions to modulate the reflective assembly of nanoparticles at the oilwater interface Mingxiang Luo, Gloria K. Olivier, and Joelle Frechette* Department of
Supplemental Information Electrostatic interactions to modulate the reflective assembly of nanoparticles at the oilwater interface Mingxiang Luo, Gloria K. Olivier, and Joelle Frechette* Department of
Effect of the Molecule-Metal Interface on the Surface Enhanced Raman Scattering of 1,4-Benzenedithiol
 Supporting Information Effect of the Molecule-Metal Interface on the Surface Enhanced Raman Scattering of 1,4-Benzenedithiol Sho Suzuki, Satoshi Kaneko*, Shintaro Fujii, Santiago Marqués-González, Tomoaki
Supporting Information Effect of the Molecule-Metal Interface on the Surface Enhanced Raman Scattering of 1,4-Benzenedithiol Sho Suzuki, Satoshi Kaneko*, Shintaro Fujii, Santiago Marqués-González, Tomoaki
Metal-Catalyzed Chemical Reaction of. Single Molecules Directly Probed by. Vibrational Spectroscopy
 Supporting Information to: Metal-Catalyzed Chemical Reaction of Single Molecules Directly Probed by Vibrational Spectroscopy Han-Kyu Choi, Won-Hwa Park, Chan Gyu Park, Hyun-Hang Shin, Kang Sup Lee and
Supporting Information to: Metal-Catalyzed Chemical Reaction of Single Molecules Directly Probed by Vibrational Spectroscopy Han-Kyu Choi, Won-Hwa Park, Chan Gyu Park, Hyun-Hang Shin, Kang Sup Lee and
Theta Probe: A tool for characterizing ultra thin films and self assembled monolayers using parallel angle resolved XPS (ARXPS)
 Theta Probe: A tool for characterizing ultra thin films and self assembled monolayers using parallel angle resolved XPS (ARXPS) C. E. Riley, P. Mack, T. S. Nunney and R. G. White Thermo Fisher Scientific
Theta Probe: A tool for characterizing ultra thin films and self assembled monolayers using parallel angle resolved XPS (ARXPS) C. E. Riley, P. Mack, T. S. Nunney and R. G. White Thermo Fisher Scientific
Supporting Information. Temperature dependence on charge transport behavior of threedimensional
 Supporting Information Temperature dependence on charge transport behavior of threedimensional superlattice crystals A. Sreekumaran Nair and K. Kimura* University of Hyogo, Graduate School of Material
Supporting Information Temperature dependence on charge transport behavior of threedimensional superlattice crystals A. Sreekumaran Nair and K. Kimura* University of Hyogo, Graduate School of Material
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/2/7/e1600322/dc1 Supplementary Materials for Ultrasensitive molecular sensor using N-doped graphene through enhanced Raman scattering Simin Feng, Maria Cristina
advances.sciencemag.org/cgi/content/full/2/7/e1600322/dc1 Supplementary Materials for Ultrasensitive molecular sensor using N-doped graphene through enhanced Raman scattering Simin Feng, Maria Cristina
Electronic Supplemental Information. Metal nanoparticle catalyzed cyclobutane cleavage reaction
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Electronic Supplemental Information Metal nanoparticle catalyzed cyclobutane cleavage reaction
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Electronic Supplemental Information Metal nanoparticle catalyzed cyclobutane cleavage reaction
Supporting Information. for. Advanced Materials, adma Wiley-VCH 2005
 Supporting Information for Advanced Materials, adma.200501482 Wiley-VCH 2005 69451 Weinheim, Germany Supplementary Material Characterization of SAMs The HBCS monolayers were characterized by XPS, NEXAFS
Supporting Information for Advanced Materials, adma.200501482 Wiley-VCH 2005 69451 Weinheim, Germany Supplementary Material Characterization of SAMs The HBCS monolayers were characterized by XPS, NEXAFS
Cu 2 O/g-C 3 N 4 nanocomposites: An insight into the band structure tuning and catalytic efficiencies
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 216 Cu 2 O/g-C 3 N 4 nanocomposites: An insight into the band structure tuning and catalytic efficiencies
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 216 Cu 2 O/g-C 3 N 4 nanocomposites: An insight into the band structure tuning and catalytic efficiencies
New Chemistry of Gold
 Established in 1959 New Chemistry of Gold T. Pradeep Department of Chemistry Indian Institute of Technology Madras Chennai 600 036 pradeep@iitm.ac.in Quantum clusters: Au 25, Au 23, Au 22, Au 8, Ag 8,
Established in 1959 New Chemistry of Gold T. Pradeep Department of Chemistry Indian Institute of Technology Madras Chennai 600 036 pradeep@iitm.ac.in Quantum clusters: Au 25, Au 23, Au 22, Au 8, Ag 8,
Supplementary Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Supplementary Information Cross-linker Mediated Formation of Sulfur-functionalized V 2 O 5 /Graphene
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Supplementary Information Cross-linker Mediated Formation of Sulfur-functionalized V 2 O 5 /Graphene
Supplementary Information for
 Supplementary Information for Facile transformation of low cost thiourea into nitrogen-rich graphitic carbon nitride nanocatalyst with high visible light photocatalytic performance Fan Dong *a, Yanjuan
Supplementary Information for Facile transformation of low cost thiourea into nitrogen-rich graphitic carbon nitride nanocatalyst with high visible light photocatalytic performance Fan Dong *a, Yanjuan
Mass spectrometric determination of the surface compositions of ethanol water mixtures
 International Journal of Mass Spectrometry 212 (2001) 267 271 www.elsevier.com/locate/ijms Cluster/kinetic method Mass spectrometric determination of the surface compositions of ethanol water mixtures
International Journal of Mass Spectrometry 212 (2001) 267 271 www.elsevier.com/locate/ijms Cluster/kinetic method Mass spectrometric determination of the surface compositions of ethanol water mixtures
Achieve a deeper understanding of polymeric systems
 The nanoscale spectroscopy company The world leader in nanoscale IR spectroscopy Achieve a deeper understanding of polymeric systems nanoir spectroscopy uniquely and unambiguously identifies the chemical
The nanoscale spectroscopy company The world leader in nanoscale IR spectroscopy Achieve a deeper understanding of polymeric systems nanoir spectroscopy uniquely and unambiguously identifies the chemical
6. Plasmon coupling between a flat gold interface and gold nanoparticles.
 6. Plasmon coupling between a flat gold interface and gold nanoparticles. 6.1. Introduction In this outlook oriented chapter the applicability of the multilayered system used in chapter 4.1., for the study
6. Plasmon coupling between a flat gold interface and gold nanoparticles. 6.1. Introduction In this outlook oriented chapter the applicability of the multilayered system used in chapter 4.1., for the study
Two Dimensional Graphene/SnS 2 Hybrids with Superior Rate Capability for Lithium ion Storage
 Electronic Supplementary Information Two Dimensional Graphene/SnS 2 Hybrids with Superior Rate Capability for Lithium ion Storage Bin Luo, a Yan Fang, a Bin Wang, a Jisheng Zhou, b Huaihe Song, b and Linjie
Electronic Supplementary Information Two Dimensional Graphene/SnS 2 Hybrids with Superior Rate Capability for Lithium ion Storage Bin Luo, a Yan Fang, a Bin Wang, a Jisheng Zhou, b Huaihe Song, b and Linjie
Effects of methanol on crystallization of water in the deeply super cooled region
 Effects of methanol on crystallization of water in the deeply super cooled region Ryutaro Souda Nanoscale Materials Center National Institute for Materials Science Japan PHYSICAL REVIEW B 75, 184116, 2007
Effects of methanol on crystallization of water in the deeply super cooled region Ryutaro Souda Nanoscale Materials Center National Institute for Materials Science Japan PHYSICAL REVIEW B 75, 184116, 2007
IR LASER-INDUCED CARBOTHERMAL REDUCTION OF TITANIUM MONOXIDE: CARBON- PHASE SHIELD TO NANOSIZED TiO OXIDATION
 IR LASER-INDUCED CARBOTHERMAL REDUCTION OF TITANIUM MONOXIDE: CARBON- PHASE SHIELD TO NANOSIZED TiO OXIDATION Věra JANDOVÁ a, Zdeněk BASTL b, Jan ŠUBRT c, Josef POLA a a Institute of Chemical Process Fundamentals,
IR LASER-INDUCED CARBOTHERMAL REDUCTION OF TITANIUM MONOXIDE: CARBON- PHASE SHIELD TO NANOSIZED TiO OXIDATION Věra JANDOVÁ a, Zdeněk BASTL b, Jan ŠUBRT c, Josef POLA a a Institute of Chemical Process Fundamentals,
Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries
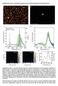 Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries Supplementary Figure 1: Dark field scattering properties of individual nanoparticle on mirror geometries separated
Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries Supplementary Figure 1: Dark field scattering properties of individual nanoparticle on mirror geometries separated
Microstructure and formation mechanism of titanium dioxide nanotubes
 Chemical Physics Letters 365 (2002) 427 431 www.elsevier.com/locate/cplett Microstructure and formation mechanism of titanium dioxide nanotubes Y.Q. Wang a,b, *, G.Q. Hu a, X.F. Duan a, H.L. Sun c, Q.K.
Chemical Physics Letters 365 (2002) 427 431 www.elsevier.com/locate/cplett Microstructure and formation mechanism of titanium dioxide nanotubes Y.Q. Wang a,b, *, G.Q. Hu a, X.F. Duan a, H.L. Sun c, Q.K.
Electrochemistry. in situ in situ. in situ. Key Words. electrochemical surface science. flame annealing and quenching method.
 in situ in situ in situ Key Words electrochemical surface science flame annealing and quenching method in situ in situ in situ not only but also in situ Q Q Q in situ δ q r in situ in situ et al in situ
in situ in situ in situ Key Words electrochemical surface science flame annealing and quenching method in situ in situ in situ not only but also in situ Q Q Q in situ δ q r in situ in situ et al in situ
Nanojet and Surface Enhanced Raman Spectroscopy (NASERS) for Highly Reproducible and Controllable Single Molecule Detection
 Nanojet and Surface Enhanced Raman Spectroscopy (NASERS) for Highly Reproducible and Controllable Single Molecule Detection Te-Wei Chang, Manas Ranjan Gartia and Gang Logan Liu Department of Electrical
Nanojet and Surface Enhanced Raman Spectroscopy (NASERS) for Highly Reproducible and Controllable Single Molecule Detection Te-Wei Chang, Manas Ranjan Gartia and Gang Logan Liu Department of Electrical
Electron Rutherford Backscattering, a versatile tool for the study of thin films
 Electron Rutherford Backscattering, a versatile tool for the study of thin films Maarten Vos Research School of Physics and Engineering Australian National University Canberra Australia Acknowledgements:
Electron Rutherford Backscattering, a versatile tool for the study of thin films Maarten Vos Research School of Physics and Engineering Australian National University Canberra Australia Acknowledgements:
Self-Assembled Monolayers
 CHE499 : A Nanotechnology Course in Chemical & Materials Engineering Spring 2006 Self-Assembled Monolayers By Drs. Lloyd Lee, Winny Dong 5GD6ER Self-Assembled Monolayers (SAMs) History Nuzzo, R. G.; Allara,
CHE499 : A Nanotechnology Course in Chemical & Materials Engineering Spring 2006 Self-Assembled Monolayers By Drs. Lloyd Lee, Winny Dong 5GD6ER Self-Assembled Monolayers (SAMs) History Nuzzo, R. G.; Allara,
Spectroscopy at nanometer scale
 Spectroscopy at nanometer scale 1. Physics of the spectroscopies 2. Spectroscopies for the bulk materials 3. Experimental setups for the spectroscopies 4. Physics and Chemistry of nanomaterials Various
Spectroscopy at nanometer scale 1. Physics of the spectroscopies 2. Spectroscopies for the bulk materials 3. Experimental setups for the spectroscopies 4. Physics and Chemistry of nanomaterials Various
A Novel Electroless Method for the Deposition of Single-Crystalline Platinum Nanoparticle Films On
 Supplementary Information A Novel Electroless Method for the Deposition of Single-Crystalline Platinum Nanoparticle Films On an Organic Solid Matrix in the Presence of Gold Single Crystals Khaleda Banu,,,*
Supplementary Information A Novel Electroless Method for the Deposition of Single-Crystalline Platinum Nanoparticle Films On an Organic Solid Matrix in the Presence of Gold Single Crystals Khaleda Banu,,,*
Direct Measurement of Electron Transfer through a Hydrogen Bond
 Supporting Information Direct Measurement of Electron Transfer through a Hydrogen Bond between Single Molecules Tomoaki Nishino,*, Nobuhiko Hayashi, and Phuc T. Bui Nanoscience and Nanotechnology Research
Supporting Information Direct Measurement of Electron Transfer through a Hydrogen Bond between Single Molecules Tomoaki Nishino,*, Nobuhiko Hayashi, and Phuc T. Bui Nanoscience and Nanotechnology Research
Self Assembled Monolayers
 Nanotechnology for engineers Winter semester 2004-2005 Nanotechnology for Engineers : J. Brugger (LMIS-1) & P. Hoffmann (IOA) Outlook Introduction (gas phase solution) Large molecules SAMs Small molecules
Nanotechnology for engineers Winter semester 2004-2005 Nanotechnology for Engineers : J. Brugger (LMIS-1) & P. Hoffmann (IOA) Outlook Introduction (gas phase solution) Large molecules SAMs Small molecules
For more information, please contact: or +1 (302)
 Introduction Graphene Raman Analyzer: Carbon Nanomaterials Characterization Dawn Yang and Kristen Frano B&W Tek Carbon nanomaterials constitute a variety of carbon allotropes including graphene, graphene
Introduction Graphene Raman Analyzer: Carbon Nanomaterials Characterization Dawn Yang and Kristen Frano B&W Tek Carbon nanomaterials constitute a variety of carbon allotropes including graphene, graphene
Chapter - 8 MONOLAYER PROTECTED NANOPARTICLES
 Chapter - 8 MONOLAYER PROTECTED NANOPARTICLES Figure 1. Schematic showing the Brust method of preparing monolayer protected clusters. Figure 2. UV-vis spectrum of Au@hexanethiol showing the presence of
Chapter - 8 MONOLAYER PROTECTED NANOPARTICLES Figure 1. Schematic showing the Brust method of preparing monolayer protected clusters. Figure 2. UV-vis spectrum of Au@hexanethiol showing the presence of
PHYSICAL AND CHEMICAL PROPERTIES OF ATMOSPHERIC PRESSURE PLASMA POLYMER FILMS
 PHYSICAL AND CHEMICAL PROPERTIES OF ATMOSPHERIC PRESSURE PLASMA POLYMER FILMS O. Goossens, D. Vangeneugden, S. Paulussen and E. Dekempeneer VITO Flemish Institute for Technological Research, Boeretang
PHYSICAL AND CHEMICAL PROPERTIES OF ATMOSPHERIC PRESSURE PLASMA POLYMER FILMS O. Goossens, D. Vangeneugden, S. Paulussen and E. Dekempeneer VITO Flemish Institute for Technological Research, Boeretang
Surface Chemistry and Reaction Dynamics of Electron Beam Induced Deposition Processes
 Surface Chemistry and Reaction Dynamics of Electron Beam Induced Deposition Processes e -? 2 nd FEBIP Workshop Thun, Switzerland 2008 Howard Fairbrother Johns Hopkins University Baltimore, MD, USA Outline
Surface Chemistry and Reaction Dynamics of Electron Beam Induced Deposition Processes e -? 2 nd FEBIP Workshop Thun, Switzerland 2008 Howard Fairbrother Johns Hopkins University Baltimore, MD, USA Outline
Supplementary Information for Self-assembled, monodispersed, flowerlike γ-alooh
 Supplementary Information for Self-assembled, monodispersed, flowerlike γ-alooh hierarchical superstructures for greatly fast removal of heavy metal ions with high efficiency Yong-Xing Zhang, a,b Yong
Supplementary Information for Self-assembled, monodispersed, flowerlike γ-alooh hierarchical superstructures for greatly fast removal of heavy metal ions with high efficiency Yong-Xing Zhang, a,b Yong
Efficient Hydrogen Evolution. University of Central Florida, 4000 Central Florida Blvd. Orlando, Florida, 32816,
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2017 MoS 2 /TiO 2 Heterostructures as Nonmetal Plasmonic Photocatalysts for Highly
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2017 MoS 2 /TiO 2 Heterostructures as Nonmetal Plasmonic Photocatalysts for Highly
Chapter 11. Structures and Dynamics of Self-Assembled Surface Monolayers
 325 Chapter 11 Structures and Dynamics of Self-Assembled Surface Monolayers adapted from C.-Y. Ruan, D.-S. Yang, A. H. Zewail, J. Am. Chem. Soc. 126, 12797 (2004). 326 Introduction When a beam of ultrashort
325 Chapter 11 Structures and Dynamics of Self-Assembled Surface Monolayers adapted from C.-Y. Ruan, D.-S. Yang, A. H. Zewail, J. Am. Chem. Soc. 126, 12797 (2004). 326 Introduction When a beam of ultrashort
Portable Raman Spectroscopy for the Study of Polymorphs and Monitoring Polymorphic Transitions
 Introduction Portable Raman Spectroscopy for the Study of Polymorphs and Monitoring Polymorphic Transitions Philip Zhou and Katherine A. Bakeev, B&W Tek, Inc., Newark, DE Materials can exist in different
Introduction Portable Raman Spectroscopy for the Study of Polymorphs and Monitoring Polymorphic Transitions Philip Zhou and Katherine A. Bakeev, B&W Tek, Inc., Newark, DE Materials can exist in different
Melting Transition of Directly-Linked Gold Nanoparticle DNA Assembly arxiv:physics/ v1 [physics.bio-ph] 10 Mar 2005
![Melting Transition of Directly-Linked Gold Nanoparticle DNA Assembly arxiv:physics/ v1 [physics.bio-ph] 10 Mar 2005 Melting Transition of Directly-Linked Gold Nanoparticle DNA Assembly arxiv:physics/ v1 [physics.bio-ph] 10 Mar 2005](/thumbs/74/69791072.jpg) Physica A (25) to appear. Melting Transition of Directly-Linked Gold Nanoparticle DNA Assembly arxiv:physics/539v [physics.bio-ph] Mar 25 Abstract Y. Sun, N. C. Harris, and C.-H. Kiang Department of Physics
Physica A (25) to appear. Melting Transition of Directly-Linked Gold Nanoparticle DNA Assembly arxiv:physics/539v [physics.bio-ph] Mar 25 Abstract Y. Sun, N. C. Harris, and C.-H. Kiang Department of Physics
Biosensing based on slow plasmon nanocavities
 iosensing based on slow plasmon nanocavities. Sepulveda, 1, Y. Alaverdyan,. rian, M. Käll 1 Nanobiosensors and Molecular Nanobiophysics Group Research Center on Nanoscience and Nanotechnolog (CIN)CSIC-ICN
iosensing based on slow plasmon nanocavities. Sepulveda, 1, Y. Alaverdyan,. rian, M. Käll 1 Nanobiosensors and Molecular Nanobiophysics Group Research Center on Nanoscience and Nanotechnolog (CIN)CSIC-ICN
Type of file: PDF Title of file for HTML: Peer Review File Description:
 Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures, Supplementary Table, Supplementary Notes and Supplementary References. Type of file: PDF Title of
Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures, Supplementary Table, Supplementary Notes and Supplementary References. Type of file: PDF Title of
Probing the Kinetics of Ligand Exchange on Colloidal Gold. Nanoparticles by Surface-Enhanced Raman Scattering
 -Supporting Information- Probing the Kinetics of Ligand Exchange on Colloidal Gold Nanoparticles by Surface-Enhanced Raman Scattering Yuhua Feng, Shuangxi Xing, Jun Xu, Hong Wang, Jun Wei Lim, and Hongyu
-Supporting Information- Probing the Kinetics of Ligand Exchange on Colloidal Gold Nanoparticles by Surface-Enhanced Raman Scattering Yuhua Feng, Shuangxi Xing, Jun Xu, Hong Wang, Jun Wei Lim, and Hongyu
Surface-enhanced Raman scattering study on non-activity of Ag-Cl clusters
 Acta Physica Sinica (Overseas Edition) Surface-enhanced Raman scattering study on non-activity of Ag-Cl clusters To cite this article: Fang Yan and Li Qin 1997 Acta Phys. Sin. (Overseas Edn) 6 183 View
Acta Physica Sinica (Overseas Edition) Surface-enhanced Raman scattering study on non-activity of Ag-Cl clusters To cite this article: Fang Yan and Li Qin 1997 Acta Phys. Sin. (Overseas Edn) 6 183 View
Fluorescence Enhancement on Silver Nanoplate at the. Single- and Sub-Nanoparticle Level
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry Supporting 2015 Information Fluorescence Enhancement on Silver Nanoplate at the Single- and Sub-Nanoparticle
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry Supporting 2015 Information Fluorescence Enhancement on Silver Nanoplate at the Single- and Sub-Nanoparticle
Secondary Ion Mass Spectrometry (SIMS)
 CHEM53200: Lecture 10 Secondary Ion Mass Spectrometry (SIMS) Major reference: Surface Analysis Edited by J. C. Vickerman (1997). 1 Primary particles may be: Secondary particles can be e s, neutral species
CHEM53200: Lecture 10 Secondary Ion Mass Spectrometry (SIMS) Major reference: Surface Analysis Edited by J. C. Vickerman (1997). 1 Primary particles may be: Secondary particles can be e s, neutral species
