CHEM J-2 June 2006 HCO 2. Calculate the osmotic pressure of a solution of 1.0 g of glucose (C 6 H 12 O 6 ) in 1500 ml of water at 37 C.
|
|
|
- Daniella McLaughlin
- 6 years ago
- Views:
Transcription
1 CEM J-2 June 2006 Draw Lewis structures of ozone, 3, and the formate anion, C 2, including resonance hybrids where appropriate. 3 C 2 3 C C Calculate the osmotic pressure of a solution of 1.0 g of glucose (C ) in 1500 ml of water at 37 C. 4 The osmotic pressure π = crt where c is the concentration. The molar mass of glucose is: ( (C)) + ( ()) + ( ()) = g of glucose corresponds to mass 1.0 = = mol molar mass The concentration when this amount is dissolved in 1500 ml = 1.5 L is: c = number of moles = = M volume 1.5 ence, π = crt = (0.0037) ( ) ( ) = atm. Answer: atm Explain why a drip for intravenous administration of fluids is made of a solution of acl at a particular concentration rather than pure water. Blood plasma is isotonic with cells (same osmotic pressure). Using saline drip of same osmotic pressure as blood prevents haemolysis or crenation of red blood cells. Write down the ground state electron configuration of the iron atom. 1 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 6
2 CEM J-3 June 2006 The balanced equation for the complete oxidation of glucose to carbon dioxide and water is given below. C (s) (g) 6C 2 (g) (l) Calculate the mass of carbon dioxide produced by the complete oxidation of 1.00 g of glucose. 3 The molar mass of glucose is: ( (C)) + ( ()) + ( ()) = g of glucose corresponds to mass 1.00 = = mol molar mass From the chemical equation, oxidation of 1 mol of glucose leads 6 mol of C 2. ence the number of moles of C 2 produced is = mol. The molar mass of C 2 is (12.01 (C)) + ( ()) = Therefore, the number of mass of C 2 produced is: mass = number of moles molar mass = = 1.47 g Answer: 1.47 g Calculate the volume of this mass of carbon dioxide at 0.50 atm pressure and 37 C. The ideal gas law gives PV = nrt, hence: V = nrt (0.0333) ( ) (273+37) = = 1.69L P (0.50) Answer: 1.69 L Explain, in terms of chemical bonding and intermolecular forces, the following trend in melting points: C 4 < I 2 < acl < silica (Si 2 ) 3 There are only dispersion forces between the molecules in C 4 and I 2. The I atom is a large, many-electron atom so its electron cloud is more easily polarised than the C or in C 4 and therefore I 2 has stronger dispersion forces and the higher melting point. acl is an ionic compound with strong coulombic attraction between the a + ions and the Cl ions packed together in the solid. Silica is a covalent network solid. Melting it requires breaking of the very strong covalent Si bonds, so it has the highest melting point.
3 CEM J-4 June 2006 Draw a Lewis structure and thus determine the geometry of the ICl 4 ion. (The I is the central atom.) 2 Cl Cl I Cl Cl There are two lone pairs and 4 bonds around the iodine: the geometry is based on an octahedron with the lone pairs located opposite to one another to minimise repulsion between them. The geometry of the actual molecule is therefore square planar. Explain briefly, in terms of intermolecular forces, why an analogue of DA could not be made with phosphorus atoms replacing some nitrogen atoms, while still retaining a double-helical structure. 3 The double helical structure is held together by hydrogen bonding between the cytosine and guanine (C G) and the adenine and thymine (A=T) base pairs. o -bonding would occur if the electronegative atoms in these bases were replaced with P atoms. The solubility of nitrogen in water at 25 C and 1.0 atm is g L 1. What is its solubility at 0.50 atm and 25 C? 1 The equilibrium of interest is 2 (g) 2 (aq) with equilibrium constant: [ 2 (aq)] K = [ 2 (g)] From the perfect gas law, PV = nrt or concentration = n = P V RT. As a result, doubling the pressure doubles the concentration of 2 (g). As the temperature is unchanged, the equilibrium constant does not change and so [ 2 (aq)] must halve to ensure that K p is constant. The solubility is thus halved to ½ = g L -1 Answer: g L -1
4 CEM J-5 June 2006 Glucose is a common food source. The net reaction for its metabolism in humans is: C (s) (g) 6C 2 (g) (l) Calculate for this reaction given the following heats of formation. f (C (s)) = 1274 kj mol 1, f (C 2 (g)) = 393 kj mol 1 and f ( 2 (l)) = 285 kj mol 1 5 Using o o o rxn f f = m (products) n (reac tan ts) : rxn = [6 f (C 2(g)) + 6 f (2(l))] [ f (C612 6(g))] 1 = [(6 393) + (6 285)] [( 1274)] = 2794kJ mol (ote that f ( 2(g)) = 0 as it is an element in its standard state). Answer: kj mol -1 If the combustion of glucose is carried out in air, water is produced as a vapour. Calculate the for the combustion of glucose in air given that 2 (l) 2 (g) = +44 kj mol 1 As vaporising liquid water requires energy (+44 kj mol -1 ), the combustion enthalpy is reduced. Six moles of 2 are produced in the combustion so the enthalpy of combustion is reduced to: (6 +44) = kj mol -1 Answer: kj mol -1 Will S be different for the two oxidation reactions? If so, how will it differ and why? As gaseous molecules have higher entropy than liquid phase molecules, oxidation to produce 2 (g) will lead to a higher value for S than oxidation to produce 2 (l).
5 CEM J-6 June 2006 Butyric acid, C 3 C 2 C 2 C, is found in rancid butter and parmesan cheese. The pk a of butyric acid is (a) What is the p of a 0.10 M water solution of butyric acid? 6 As pk a = -logk a = 4.83, K a = Denoting butyric acid as A, the initial concentration of [A(aq)] = 0.10 M. The reaction table is then: [A(aq)] [ + (aq)] [A - (aq)] t = change -x +x +x equilibrium 0.10 x x x ence, K a = + - [ (aq)][a (aq)] [A(aq)] = (x)(x) 0.10-x 2 x = 0.10-x As K z is small, the amount of dissociation, x, is also small so 0.10 x ~ x Using this approximation, K z = =10 hence x = M As x = [ + (aq)], p = -log[ + (aq)] = -log( ) = 2.92 Answer: 2.92 (b) Calculate the p of the solution formed when mol of a(s) is added to 1.0 L of 0.10 M butyric acid. As a is a strong base, it will dissociate completely and each mole of - will react with butyric acid to form one mole of A - (aq). 1.0 L of 0.10 M A contains 0.10 mol. After addition of mol of -, the number of moles of A = ( ) = 0.05 mol and the number of moles of A - = mol. As 1.0 L of solution is present, [A(aq)] = 0.05 M and [A - (aq)] = 0.05 M. Substituting into the expression for K a gives: K a = + - [ (aq)][a (aq)] [A(aq)] + [ (aq)] (0.05) = = (0.05) so [ + (aq)] = 1.5 M ence, p = -log[ + (aq)] = 4.83 Answer: 4.83
6 (c) Using equations, comment on how the final solution in (b) will respond to additions of small amounts of acid or base in comparison to 1 L of water. Solution (b) consists of a mixture of a weak acid and its conjugate base: it is a buffer system and will resist changes in p. If acid is added, the system can respond by removing it using A - : + (aq) + A - (aq) A(aq) If base is added, the system can respond by removing it using A: - (aq) + A(aq) 2 (l) + A - (aq)
7 CEM J-7 June 2006 Consider the reaction of 2 (g) with I 2 (g) at 298 K to give I(g). 2 (g) + I 2 (g) 2I(g) K p = 2.24 If partial pressures of 0.20 atm of all three gases are mixed, in which direction will the reaction proceed? 3 The reaction quotient Q = 2 2 (p I ) (0.20) = =1.0 (p )(p ) (0.20) (0.20) 2 I2 As Q < K p, the reaction will proceed towards I, to increase the partial pressure of I and decrease the partial pressure of 2 and I 2, until Q = K. Calculate G for this reaction at 298 K. Answer: towards products Using G = -RTlnK p : G = -(8.314) (298) ln(2.24) = J mol -1 = kj mol -1 Answer: J mol -1 = kj mol -1
8 CEM J-8 June 2006 Draw the constitutional formula(s) of the major organic product(s) of the following reactions Cr 2 7 / S S + AD / S + AD + excess C 3 / 3 C C 3 Br + Br 2 Br Cl + excess 2 C 3 C 3 What are the requirements for a molecule to be aromatic? Give one example of an aromatic heterocycle. 2 The molecule must be cyclic. The ring system must have a conjugated π bond system. The number of electrons in the π bond system must be 4n+2 where n=integer. All atoms in the ring system must be sp 2 hybridised. Examples of heterocyclic heterocycles include: S
9
10 CEM J-9 June 2006 Adenine and thymine have the structures shown below C adenine Draw a tautomer of the shown structure of adenine. thymine In DA, adenine forms a base pair with thymine. Explain what is meant by base pair and indicate the point(s) of interaction between adenine and thymine. Four different bases are found in DA chains. The two strands in the double helix are held together by 3 hydrogen bonds between guanine (G) and cytosine (C), and by 2 hydrogen bonds between adenine (A) and thymine (T). The complementary bases C and G are called a base pair. A and T are another base pair. adenine = A thymine = T
11 CEM J-10 June 2006 The open chain form of D-glucose has the structure shown. C C 2 5 Draw the aworth projection of β-d-glucopyranose. Draw the major organic product of the reaction of D-glucose with the following reagents. 1. ab 4 2. / 2 C 2 C 2 [Ag( 3 ) 2 ] / C 2 C 2 Would you expect D-glucose to be water soluble? Why? D-glucose will be water soluble as it has numerous alcohol functional groups which can hydrogen bond with the water molecules.
12 CEM J-11 June 2006 L-Tyrosine is a naturally occurring amino acid with the following side-chain. 6 C 2 The pk a values of tyrosine are 2.20 (α-c), 9.19 (α- 3 ) and (sidechain). Draw the Fischer projection of L-tyrosine indicating the correct charge state at physiological p. C 2 3 C C 2 What is the absolute stereochemistry of L-tyrosine? Write (R) or (S). S What is the value of the pi of L-tyrosine? pi = ½ ( ) = 5.70 What does pi represent? pi represents the isoelectric point - the p at which there is no net charge on the molecule.
13 Account for the difference in acidity of the carboxylic acid group and the phenol. Acid strength is dependent on the stability of the conjugate base. The carboxylate anion is resonance stabilised, with the charge being spread over the electronegative atoms: R C R C The phenoxide anion is resonance stabilised also, but the charge in the resonance contributers is spead over the C atoms in the ring. C is not as electronegative as, so these contributers are not as significant as that with the charge on the. Resonance stabilisation is not as great as for carboxylate and therefore phenol is weaker acid than carboxylic acid.
14 CEM J-12 June 2006 Draw the products of acid hydrolysis of the following peptide, indicating the correct charge state under these conditions. 3 3 C C 3 C C C 2 C 2 C 2 C 2 3 C C 3 C C C 3 2 C C 2 C
CHEM J-3 June Calculate the osmotic pressure of a 0.25 M aqueous solution of sucrose, C 12 H 22 O 11, at 37 C
 CHEM1405 003-J-3 June 003 Calculate the osmotic pressure of a 0.5 M aqueous solution of sucrose, C 1 H O 11, at 37 C The osmotic pressure for strong electrolyte solutions is given by: Π = i (crt) where
CHEM1405 003-J-3 June 003 Calculate the osmotic pressure of a 0.5 M aqueous solution of sucrose, C 1 H O 11, at 37 C The osmotic pressure for strong electrolyte solutions is given by: Π = i (crt) where
Number of d electrons CO 2 carbon dioxide +IV or +4 0
 CEM1611 2007-J-2 June 2007 Complete the following table. Give, as required, the formula, the systematic name, the oxidation number of the underlined atom and, where indicated, the number of d electrons
CEM1611 2007-J-2 June 2007 Complete the following table. Give, as required, the formula, the systematic name, the oxidation number of the underlined atom and, where indicated, the number of d electrons
Chap 10 Part 4Ta.notebook December 08, 2017
 Chapter 10 Section 1 Intermolecular Forces the forces between molecules or between ions and molecules in the liquid or solid state Stronger Intermolecular forces cause higher melting points and boiling
Chapter 10 Section 1 Intermolecular Forces the forces between molecules or between ions and molecules in the liquid or solid state Stronger Intermolecular forces cause higher melting points and boiling
AP CHEMISTRY 2009 SCORING GUIDELINES
 2009 SCORING GUIDELINES Question 1 (10 points) Answer the following questions that relate to the chemistry of halogen oxoacids. (a) Use the information in the table below to answer part (a)(i). Acid HOCl
2009 SCORING GUIDELINES Question 1 (10 points) Answer the following questions that relate to the chemistry of halogen oxoacids. (a) Use the information in the table below to answer part (a)(i). Acid HOCl
Assessment Schedule 2014 Scholarship Chemistry (93102) Evidence Statement
 Assessment Schedule 2014 Scholarship Chemistry (93102) Evidence Statement Scholarship Chemistry (93102) 2014 page 1 of 10 Question ONE (a)(i) Evidence Na(s) to Na(g) will require less energy than vaporisation
Assessment Schedule 2014 Scholarship Chemistry (93102) Evidence Statement Scholarship Chemistry (93102) 2014 page 1 of 10 Question ONE (a)(i) Evidence Na(s) to Na(g) will require less energy than vaporisation
CHEM J-3 June 2012
 CEM1611 2012-J-3 June 2012 In a standard acid-base titration, 25.00 ml of 0.1043 M a solution was found to react exactly with 28.45 ml of an Cl solution of unknown concentration. What is the p of the unknown
CEM1611 2012-J-3 June 2012 In a standard acid-base titration, 25.00 ml of 0.1043 M a solution was found to react exactly with 28.45 ml of an Cl solution of unknown concentration. What is the p of the unknown
Intermolecular Forces & Condensed Phases
 Intermolecular Forces & Condensed Phases CHEM 107 T. Hughbanks READING We will discuss some of Chapter 5 that we skipped earlier (Van der Waals equation, pp. 145-8), but this is just a segue into intermolecular
Intermolecular Forces & Condensed Phases CHEM 107 T. Hughbanks READING We will discuss some of Chapter 5 that we skipped earlier (Van der Waals equation, pp. 145-8), but this is just a segue into intermolecular
CHM 151 Practice Final Exam
 CM 151 Practice Final Exam 1. ow many significant figures are there in the result of 5.52 divided by 3.745? (a) 1 (b) 2 (c) 3 (d) 4 (e) 5 2. ow many significant figures are there in the answer when 9.021
CM 151 Practice Final Exam 1. ow many significant figures are there in the result of 5.52 divided by 3.745? (a) 1 (b) 2 (c) 3 (d) 4 (e) 5 2. ow many significant figures are there in the answer when 9.021
Name: Score: /100. Part I. Multiple choice. Write the letter of the correct answer for each problem. 3 points each
 Name: Score: /100 Part I. Multiple choice. Write the letter of the correct answer for each problem. 3 points each 1. Which of the following contains the greatest number of moles of O? A) 2.3 mol H 2 O
Name: Score: /100 Part I. Multiple choice. Write the letter of the correct answer for each problem. 3 points each 1. Which of the following contains the greatest number of moles of O? A) 2.3 mol H 2 O
READING. Review of Intermolecular Forces & Liquids (Chapter 12) Ion-Ion Forces. Ion-Dipole Energies
 Review of Intermolecular Forces & Liquids (Chapter 12) CEM 102 T. ughbanks READIG We will very briefly review the underlying concepts from Chapters 12 on intermolecular forces since it is relevant to Chapter
Review of Intermolecular Forces & Liquids (Chapter 12) CEM 102 T. ughbanks READIG We will very briefly review the underlying concepts from Chapters 12 on intermolecular forces since it is relevant to Chapter
Chapter 1 The Atomic Nature of Matter
 Chapter 1 The Atomic Nature of Matter 1-1 Chemistry: Science of Change 1-2 The Composition of Matter 1-3 The Atomic Theory of Matter 1-4 Chemical Formulas and Relative Atomic Masses 1-5 The Building Blocks
Chapter 1 The Atomic Nature of Matter 1-1 Chemistry: Science of Change 1-2 The Composition of Matter 1-3 The Atomic Theory of Matter 1-4 Chemical Formulas and Relative Atomic Masses 1-5 The Building Blocks
CHEM 1211K Test IV. 3) The phase diagram of a substance is given above. This substance is a at 25 o C and 1.0 atm.
 CEM 1211K Test IV A MULTIPLE COICE. ( points) 1) A sample of a gas (5.0 mol) at 1.0 atm is expanded at constant temperature from 10 L to 15 L. The final pressure is atm. A). B) 1.5 C) 15 D) 7.5 E) 0.67
CEM 1211K Test IV A MULTIPLE COICE. ( points) 1) A sample of a gas (5.0 mol) at 1.0 atm is expanded at constant temperature from 10 L to 15 L. The final pressure is atm. A). B) 1.5 C) 15 D) 7.5 E) 0.67
CHEM J-9 June 2014
 CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
Questions 1-2 Consider the atoms of the following elements. Assume that the atoms are in the ground state. a. S b. Ca c. Ga d. Sb e.
 AP Chemistry Fall Semester Practice Exam 5 MULTIPLE CHOICE PORTION: Write the letter for the correct answer to the following questions on the provided answer sheet. Each multiple choice question is worth
AP Chemistry Fall Semester Practice Exam 5 MULTIPLE CHOICE PORTION: Write the letter for the correct answer to the following questions on the provided answer sheet. Each multiple choice question is worth
CHEMISTRY 110 Final EXAM Dec 17, 2012 FORM A
 CEMISTRY 110 Final EXAM Dec 17, 2012 FORM A 1. Given the following reaction which of the following statements is true? Fe(s) + CuCl 2 (aq)! Cu(s) + FeCl 2 (aq) A. Iron is oxidized and copper is reduced.
CEMISTRY 110 Final EXAM Dec 17, 2012 FORM A 1. Given the following reaction which of the following statements is true? Fe(s) + CuCl 2 (aq)! Cu(s) + FeCl 2 (aq) A. Iron is oxidized and copper is reduced.
Some Basic Concepts of Chemistry
 0 Some Basic Concepts of Chemistry Chapter 0: Some Basic Concept of Chemistry Mass of solute 000. Molarity (M) Molar mass volume(ml).4 000 40 500 0. mol L 3. (A) g atom of nitrogen 8 g (B) 6.03 0 3 atoms
0 Some Basic Concepts of Chemistry Chapter 0: Some Basic Concept of Chemistry Mass of solute 000. Molarity (M) Molar mass volume(ml).4 000 40 500 0. mol L 3. (A) g atom of nitrogen 8 g (B) 6.03 0 3 atoms
NChO 2006 ANNOTATED ANSWERS
 NChO 2006 ANNOTATED ANSWERS 1. D HCl H 2 and CH 4 are nonpolar and will dissolve very little in polar water. CO is polar and will dissolve OK, but HCl is very polar and dissolves amazingly well in water.
NChO 2006 ANNOTATED ANSWERS 1. D HCl H 2 and CH 4 are nonpolar and will dissolve very little in polar water. CO is polar and will dissolve OK, but HCl is very polar and dissolves amazingly well in water.
molality: m = = 1.70 m
 C h e m i s t r y 1 2 U n i t 3 R e v i e w P a g e 1 Chem 12: Chapters 10, 11, 12, 13, 14 Unit 3 Worksheet 1. What is miscible? Immiscible? Miscible: two or more substances blend together for form a solution
C h e m i s t r y 1 2 U n i t 3 R e v i e w P a g e 1 Chem 12: Chapters 10, 11, 12, 13, 14 Unit 3 Worksheet 1. What is miscible? Immiscible? Miscible: two or more substances blend together for form a solution
STD-XI-Science-Chemistry Chemical Bonding & Molecular structure
 STD-XI-Science-Chemistry Chemical Bonding & Molecular structure Chemical Bonding Question 1 What is meant by the term chemical bond? How does Kessel-Lewis approach of bonding differ from the modern views?
STD-XI-Science-Chemistry Chemical Bonding & Molecular structure Chemical Bonding Question 1 What is meant by the term chemical bond? How does Kessel-Lewis approach of bonding differ from the modern views?
GENERAL BONDING REVIEW
 GENERAL BONDING REVIEW Chapter 8 November 2, 2016 Questions to Consider 1. What is meant by the term chemical bond? 2. Why do atoms bond with each other to form compounds? 3. How do atoms bond with each
GENERAL BONDING REVIEW Chapter 8 November 2, 2016 Questions to Consider 1. What is meant by the term chemical bond? 2. Why do atoms bond with each other to form compounds? 3. How do atoms bond with each
SIR MICHELANGELO REFALO
 SIR MICELANGELO REFALO SIXT FORM alf-yearly Exam 2014 Name: CEMISTRY ADV 1 ST 3 hrs ANSWER ANY 7 QUESTIONS. All questions carry equal marks. You are reminded of the importance of clear presentation in
SIR MICELANGELO REFALO SIXT FORM alf-yearly Exam 2014 Name: CEMISTRY ADV 1 ST 3 hrs ANSWER ANY 7 QUESTIONS. All questions carry equal marks. You are reminded of the importance of clear presentation in
 CHAPTER 6 CHEMICAL BONDING SHORT QUESTION WITH ANSWERS Q.1 Dipole moments of chlorobenzene is 1.70 D and of chlorobenzene is 2.5 D while that of paradichlorbenzene is zero; why? Benzene has zero dipole
CHAPTER 6 CHEMICAL BONDING SHORT QUESTION WITH ANSWERS Q.1 Dipole moments of chlorobenzene is 1.70 D and of chlorobenzene is 2.5 D while that of paradichlorbenzene is zero; why? Benzene has zero dipole
Name AP CHEM / / Collected AP Exam Essay Answers for Chapter 16
 Name AP CHEM / / Collected AP Exam Essay Answers for Chapter 16 1980 - #7 (a) State the physical significance of entropy. Entropy (S) is a measure of randomness or disorder in a system. (b) From each of
Name AP CHEM / / Collected AP Exam Essay Answers for Chapter 16 1980 - #7 (a) State the physical significance of entropy. Entropy (S) is a measure of randomness or disorder in a system. (b) From each of
4 States of matter. N Goalby chemrevise.org 1. Ideal Gas Equation
 4 States of matter Ideal Gas Equation The ideal gas equation applies to all gases and mixtures of gases. If a mixture of gases is used the value n will be the total moles of all gases in the mixture. The
4 States of matter Ideal Gas Equation The ideal gas equation applies to all gases and mixtures of gases. If a mixture of gases is used the value n will be the total moles of all gases in the mixture. The
Downloaded from
 I.I.T.Foundation - XI Chemistry MCQ #4 Time: 45 min Student's Name: Roll No.: Full Marks: 90 Chemical Bonding I. MCQ - Choose Appropriate Alternative 1. The energy required to break a chemical bond to
I.I.T.Foundation - XI Chemistry MCQ #4 Time: 45 min Student's Name: Roll No.: Full Marks: 90 Chemical Bonding I. MCQ - Choose Appropriate Alternative 1. The energy required to break a chemical bond to
Topic 04 Bonding 4.3 Intermolecular Forces. IB Chemistry T04D05
 Topic 04 Bonding 4.3 Intermolecular orces IB Chemistry T04D05 Intermolecular orces 2 hrs 4.3.1 Describe the types of intermolecular forces (attractions between molecules that have temporary dipoles, permanent
Topic 04 Bonding 4.3 Intermolecular orces IB Chemistry T04D05 Intermolecular orces 2 hrs 4.3.1 Describe the types of intermolecular forces (attractions between molecules that have temporary dipoles, permanent
UNIT 7 p-block ELEMENTS
 UNIT 7 p-blck ELEMENTS 1 MARK QUESTINS Q 1 Arrange the following in Acidic strength : HN 3, H 3 4, H 3 As 4 The order of acidity will be : HN 3 > H 3 4 > H 3 As 4 N > > As Electronegativity of elements
UNIT 7 p-blck ELEMENTS 1 MARK QUESTINS Q 1 Arrange the following in Acidic strength : HN 3, H 3 4, H 3 As 4 The order of acidity will be : HN 3 > H 3 4 > H 3 As 4 N > > As Electronegativity of elements
Midterm II Material/Topics Autumn 2010
 1 Midterm II Material/Topics Autumn 2010 Supplemental Material: Resonance Structures Ch 5.8 Molecular Geometry Ch 5.9 Electronegativity Ch 5.10 Bond Polarity Ch 5.11 Molecular Polarity Ch 5.12 Naming Binary
1 Midterm II Material/Topics Autumn 2010 Supplemental Material: Resonance Structures Ch 5.8 Molecular Geometry Ch 5.9 Electronegativity Ch 5.10 Bond Polarity Ch 5.11 Molecular Polarity Ch 5.12 Naming Binary
Chem GENERAL CHEMISTRY II
 GENERAL CHEMISTRY II CHEM 206 /2 51 Final Examination December 19, 2006 1900-2200 Dr. Cerrie ROGERS x x programmable calculators must be reset Chem 206 --- GENERAL CHEMISTRY II LAST NAME: STUDENT NUMBER:
GENERAL CHEMISTRY II CHEM 206 /2 51 Final Examination December 19, 2006 1900-2200 Dr. Cerrie ROGERS x x programmable calculators must be reset Chem 206 --- GENERAL CHEMISTRY II LAST NAME: STUDENT NUMBER:
2: CHEMICAL COMPOSITION OF THE BODY
 1 2: CHEMICAL COMPOSITION OF THE BODY Although most students of human physiology have had at least some chemistry, this chapter serves very well as a review and as a glossary of chemical terms. In particular,
1 2: CHEMICAL COMPOSITION OF THE BODY Although most students of human physiology have had at least some chemistry, this chapter serves very well as a review and as a glossary of chemical terms. In particular,
Chapter 16 Covalent Bonding
 Chemistry/ PEP Name: Date: Chapter 16 Covalent Bonding Chapter 16: 1 26; 28, 30, 31, 35-37, 40, 43-46, Extra Credit: 50-53, 55, 56, 58, 59, 62-67 Section 16.1 The Nature of Covalent Bonding Practice Problems
Chemistry/ PEP Name: Date: Chapter 16 Covalent Bonding Chapter 16: 1 26; 28, 30, 31, 35-37, 40, 43-46, Extra Credit: 50-53, 55, 56, 58, 59, 62-67 Section 16.1 The Nature of Covalent Bonding Practice Problems
Level 3 Chemistry Demonstrate understanding of thermochemical principles and the properties of particles and substances
 1 ANSWERS Level 3 Chemistry 91390 Demonstrate understanding of thermochemical principles and the properties of particles and substances Credits: Five Achievement Achievement with Merit Achievement with
1 ANSWERS Level 3 Chemistry 91390 Demonstrate understanding of thermochemical principles and the properties of particles and substances Credits: Five Achievement Achievement with Merit Achievement with
Solutions. Chapter 14 Solutions. Ion-Ion Forces (Ionic Bonding) Attraction Between Ions and Permanent Dipoles. Covalent Bonding Forces
 Solutions Chapter 14 1 Brief Review of Major Topics in Chapter 13, Intermolecular forces Ion-Ion Forces (Ionic Bonding) 2 Na + Cl - in salt These are the strongest forces. Lead to solids with high melting
Solutions Chapter 14 1 Brief Review of Major Topics in Chapter 13, Intermolecular forces Ion-Ion Forces (Ionic Bonding) 2 Na + Cl - in salt These are the strongest forces. Lead to solids with high melting
Molecular Structure and Bonding- 2. Assis.Prof.Dr.Mohammed Hassan Lecture 3
 Molecular Structure and Bonding- 2 Assis.Prof.Dr.Mohammed Hassan Lecture 3 Hybridization of atomic orbitals Orbital hybridization was proposed to explain the geometry of polyatomic molecules. Covalent
Molecular Structure and Bonding- 2 Assis.Prof.Dr.Mohammed Hassan Lecture 3 Hybridization of atomic orbitals Orbital hybridization was proposed to explain the geometry of polyatomic molecules. Covalent
Upon successful completion of this unit, the students should be able to:
 Unit 9. Liquids and Solids - ANSWERS Upon successful completion of this unit, the students should be able to: 9.1 List the various intermolecular attractions in liquids and solids (dipole-dipole, London
Unit 9. Liquids and Solids - ANSWERS Upon successful completion of this unit, the students should be able to: 9.1 List the various intermolecular attractions in liquids and solids (dipole-dipole, London
Chapter 11 Properties of Solutions
 Chapter 11 Properties of Solutions Solutions Homogeneous mixtures of two or more substances Composition is uniform throughout the sample No chemical reaction between the components of the mixture Solvents
Chapter 11 Properties of Solutions Solutions Homogeneous mixtures of two or more substances Composition is uniform throughout the sample No chemical reaction between the components of the mixture Solvents
Chapter #16 Liquids and Solids
 Chapter #16 Liquids and Solids 16.1 Intermolecular Forces 16.2 The Liquid State 16.3 An Introduction to Structures and Types of Solids 16.4 Structure and Bonding of Metals 16.5 Carbon and Silicon: Network
Chapter #16 Liquids and Solids 16.1 Intermolecular Forces 16.2 The Liquid State 16.3 An Introduction to Structures and Types of Solids 16.4 Structure and Bonding of Metals 16.5 Carbon and Silicon: Network
KEY: FREE RESPONSE. Question Ammonium carbamate, NH 4CO 2NH 2, reversibly decomposes into CO 2 and NH 3 as shown by the balanced equation below.
 KEY: FREE RESPONSE Question 1 1. Ammonium carbamate, NH 4CO 2NH 2, reversibly decomposes into CO 2 and NH 3 as shown by the balanced equation below. NH 4CO 2NH 2(s) 2 NH 3(g) + CO 2(g) K p =? A student
KEY: FREE RESPONSE Question 1 1. Ammonium carbamate, NH 4CO 2NH 2, reversibly decomposes into CO 2 and NH 3 as shown by the balanced equation below. NH 4CO 2NH 2(s) 2 NH 3(g) + CO 2(g) K p =? A student
What are covalent bonds?
 Covalent Bonds What are covalent bonds? Covalent Bonds A covalent bond is formed when neutral atoms share one or more pairs of electrons. Covalent Bonds Covalent bonds form between two or more non-metal
Covalent Bonds What are covalent bonds? Covalent Bonds A covalent bond is formed when neutral atoms share one or more pairs of electrons. Covalent Bonds Covalent bonds form between two or more non-metal
List, with an explanation, the three compounds in order of increasing carbon to oxygen bond length (shortest first).
 T4-2P1 [226 marks] 1. Which statement best describes the intramolecular bonding in HCN(l)? A. Electrostatic attractions between H + and CN ions B. Only van der Waals forces C. Van der Waals forces and
T4-2P1 [226 marks] 1. Which statement best describes the intramolecular bonding in HCN(l)? A. Electrostatic attractions between H + and CN ions B. Only van der Waals forces C. Van der Waals forces and
CHEM N-2 November 2014
 CHEM1612 2014-N-2 November 2014 Explain the following terms or concepts. Le Châtelier s principle 1 Used to predict the effect of a change in the conditions on a reaction at equilibrium, this principle
CHEM1612 2014-N-2 November 2014 Explain the following terms or concepts. Le Châtelier s principle 1 Used to predict the effect of a change in the conditions on a reaction at equilibrium, this principle
Topics in the November 2009 Exam Paper for CHEM1101
 November 2009 Topics in the November 2009 Exam Paper for CHEM1101 Click on the links for resources on each topic. 2009-N-2: 2009-N-3: 2009-N-4: 2009-N-5: 2009-N-6: 2009-N-7: 2009-N-8: 2009-N-9: 2009-N-10:
November 2009 Topics in the November 2009 Exam Paper for CHEM1101 Click on the links for resources on each topic. 2009-N-2: 2009-N-3: 2009-N-4: 2009-N-5: 2009-N-6: 2009-N-7: 2009-N-8: 2009-N-9: 2009-N-10:
Chapter 2: Chemical Basis of Life
 Chapter 2: Chemical Basis of Life Chemistry is the scientific study of the composition of matter and how composition changes. In order to understand human physiological processes, it is important to understand
Chapter 2: Chemical Basis of Life Chemistry is the scientific study of the composition of matter and how composition changes. In order to understand human physiological processes, it is important to understand
4. What is the number of unpaired electrons in Ni? Chemistry 12 Final Exam Form A May 4, 2001
 Chemistry 12 Final Exam Form A May 4, 2001 In all questions involving gases, assume that the ideal-gas laws hold, unless the question specifically refers to the non-ideal behavior. 1. Which of the following
Chemistry 12 Final Exam Form A May 4, 2001 In all questions involving gases, assume that the ideal-gas laws hold, unless the question specifically refers to the non-ideal behavior. 1. Which of the following
I can calculate the rate of reaction from graphs of a changing property versus time, e.g. graphs of volume against time
 UNIT 1 CONTROLLING THE RATE OF REACTION I can calculate the rate of reaction from graphs of a changing property versus time, e.g. graphs of volume against time I can use the reciprocal of to calculate
UNIT 1 CONTROLLING THE RATE OF REACTION I can calculate the rate of reaction from graphs of a changing property versus time, e.g. graphs of volume against time I can use the reciprocal of to calculate
Spanish Fork High School Unit Topics and I Can Statements AP Chemistry
 Spanish Fork High School 2014-15 Unit Topics and I Can Statements AP Chemistry Properties of Elements I can describe how mass spectroscopy works and use analysis of elements to calculate the atomic mass
Spanish Fork High School 2014-15 Unit Topics and I Can Statements AP Chemistry Properties of Elements I can describe how mass spectroscopy works and use analysis of elements to calculate the atomic mass
A solution is a homogeneous mixture of two or more substances.
 UNIT (5) SOLUTIONS A solution is a homogeneous mixture of two or more substances. 5.1 Terminology Solute and Solvent A simple solution has two components, a solute, and a solvent. The substance in smaller
UNIT (5) SOLUTIONS A solution is a homogeneous mixture of two or more substances. 5.1 Terminology Solute and Solvent A simple solution has two components, a solute, and a solvent. The substance in smaller
CHEM N-2 November 2012
 CHEM1101 2012-N-2 November 2012 Nitrogen dioxide, NO 2, is formed in the atmosphere from industrial processes and automobile exhaust. It is an indicator of poor quality air and is mostly responsible for
CHEM1101 2012-N-2 November 2012 Nitrogen dioxide, NO 2, is formed in the atmosphere from industrial processes and automobile exhaust. It is an indicator of poor quality air and is mostly responsible for
Topics in the November 2008 Exam Paper for CHEM1612
 November 2008 Topics in the November 2008 Exam Paper for CHEM1612 Click on the links for resources on each topic. 2008-N-2: 2008-N-3: 2008-N-4: 2008-N-5: 2008-N-6: 2008-N-7: 2008-N-8: 2008-N-9: 2008-N-10:
November 2008 Topics in the November 2008 Exam Paper for CHEM1612 Click on the links for resources on each topic. 2008-N-2: 2008-N-3: 2008-N-4: 2008-N-5: 2008-N-6: 2008-N-7: 2008-N-8: 2008-N-9: 2008-N-10:
H O H. Chapter 3: Outline-2. Chapter 3: Outline-1
 Chapter 3: utline-1 Molecular Nature of Water Noncovalent Bonding Ionic interactions van der Waals Forces Thermal Properties of Water Solvent Properties of Water ydrogen Bonds ydrophilic, hydrophobic,
Chapter 3: utline-1 Molecular Nature of Water Noncovalent Bonding Ionic interactions van der Waals Forces Thermal Properties of Water Solvent Properties of Water ydrogen Bonds ydrophilic, hydrophobic,
Name: Score: /100. Part I. Multiple choice. Write the letter of the correct answer for each problem. 3 points each
 Name: Score: /100 Part I. Multiple choice. Write the letter of the correct answer for each problem. 3 points each 1. Which of the following contains the greatest number of moles of O? A) 2.3 mol H 2 O
Name: Score: /100 Part I. Multiple choice. Write the letter of the correct answer for each problem. 3 points each 1. Which of the following contains the greatest number of moles of O? A) 2.3 mol H 2 O
CHEMISTRY HIGHER LEVEL
 *P15* Pre-Leaving Certificate Examination, 2012 Triailscrúdú na hardteistiméireachta, 2012 CHEMISTRY HIGHER LEVEL TIME: 3 HOURS 400 MARKS Answer eight questions in all These must include at least two questions
*P15* Pre-Leaving Certificate Examination, 2012 Triailscrúdú na hardteistiméireachta, 2012 CHEMISTRY HIGHER LEVEL TIME: 3 HOURS 400 MARKS Answer eight questions in all These must include at least two questions
Week 8 Intermolecular Forces
 NO CALCULATORS MAY BE USED FOR THESE QUESTIONS Questions 1-3 refer to the following list. (A) Cu (B) PH 3 (C) C (D) SO 2 (E) O 2 1. Contains instantaneous dipole moments. 2. Forms covalent network solids.
NO CALCULATORS MAY BE USED FOR THESE QUESTIONS Questions 1-3 refer to the following list. (A) Cu (B) PH 3 (C) C (D) SO 2 (E) O 2 1. Contains instantaneous dipole moments. 2. Forms covalent network solids.
Chapter 14. Liquids and Solids
 Chapter 14 Liquids and Solids Section 14.1 Water and Its Phase Changes Reviewing What We Know Gases Low density Highly compressible Fill container Solids High density Slightly compressible Rigid (keeps
Chapter 14 Liquids and Solids Section 14.1 Water and Its Phase Changes Reviewing What We Know Gases Low density Highly compressible Fill container Solids High density Slightly compressible Rigid (keeps
Big Idea #5: The laws of thermodynamics describe the essential role of energy and explain and predict the direction of changes in matter.
 KUDs for Unit 6: Chemical Bonding Textbook Reading: Chapters 8 & 9 Big Idea #2: Chemical and physical properties of materials can be explained by the structure and the arrangement of atoms, ion, or molecules
KUDs for Unit 6: Chemical Bonding Textbook Reading: Chapters 8 & 9 Big Idea #2: Chemical and physical properties of materials can be explained by the structure and the arrangement of atoms, ion, or molecules
Topic 1: Quantitative chemistry
 covered by A-Level Chemistry products Topic 1: Quantitative chemistry 1.1 The mole concept and Avogadro s constant 1.1.1 Apply the mole concept to substances. Moles and Formulae 1.1.2 Determine the number
covered by A-Level Chemistry products Topic 1: Quantitative chemistry 1.1 The mole concept and Avogadro s constant 1.1.1 Apply the mole concept to substances. Moles and Formulae 1.1.2 Determine the number
exothermic reaction and that ΔH c will therefore be a negative value. Heat change, q = mcδt q = m(h 2
 Worked solutions hapter 5 Exercises 1 B If the temperature drops, the process must be endothermic. Δ for endothermic reactions is always positive. 2 B All exothermic reactions give out heat. While there
Worked solutions hapter 5 Exercises 1 B If the temperature drops, the process must be endothermic. Δ for endothermic reactions is always positive. 2 B All exothermic reactions give out heat. While there
Chem 112 Dr. Kevin Moore
 Chem 112 Dr. Kevin Moore Gas Liquid Solid Polar Covalent Bond Partial Separation of Charge Electronegativity: H 2.1 Cl 3.0 H Cl δ + δ - Dipole Moment measure of the net polarity in a molecule Q Q magnitude
Chem 112 Dr. Kevin Moore Gas Liquid Solid Polar Covalent Bond Partial Separation of Charge Electronegativity: H 2.1 Cl 3.0 H Cl δ + δ - Dipole Moment measure of the net polarity in a molecule Q Q magnitude
IB Chemistry Solutions Gasses and Energy
 Solutions A solution is a homogeneous mixture it looks like one substance. An aqueous solution will be a clear mixture with only one visible phase. Be careful with the definitions of clear and colourless.
Solutions A solution is a homogeneous mixture it looks like one substance. An aqueous solution will be a clear mixture with only one visible phase. Be careful with the definitions of clear and colourless.
CHEM J-8 June /01(a) With 3 C-O bonds and no lone pairs on the C atom, the geometry is trigonal planar.
 CHEM1001 2014-J-8 June 2014 22/01(a) What is the molecular geometry of the formate ion? Marks 7 With 3 C-O bonds and no lone pairs on the C atom, the geometry is trigonal planar. Write the equilibrium
CHEM1001 2014-J-8 June 2014 22/01(a) What is the molecular geometry of the formate ion? Marks 7 With 3 C-O bonds and no lone pairs on the C atom, the geometry is trigonal planar. Write the equilibrium
Properties of Solutions
 Properties of Solutions The Solution Process A solution is a homogeneous mixture of solute and solvent. Solutions may be gases, liquids, or solids. Each substance present is a component of the solution.
Properties of Solutions The Solution Process A solution is a homogeneous mixture of solute and solvent. Solutions may be gases, liquids, or solids. Each substance present is a component of the solution.
Midterm Exam III. CHEM 181: Introduction to Chemical Principles November 25, 2014 Answer Key
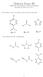 Midterm Exam III EM 181: Introduction to hemical Principles ovember 25, 2014 Answer Key 1. For reference, here are the pk a values for three weak acids: 3 pk a = 7.2 pk a = 4.8 pk a = 15 ow consider this
Midterm Exam III EM 181: Introduction to hemical Principles ovember 25, 2014 Answer Key 1. For reference, here are the pk a values for three weak acids: 3 pk a = 7.2 pk a = 4.8 pk a = 15 ow consider this
Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks =
 Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks = 1. (12%) Compound X contains 2.239% hydrogen, 26.681% carbon and 71.080 % oxygen by mass. The titration of 0.154 g of this compound
Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks = 1. (12%) Compound X contains 2.239% hydrogen, 26.681% carbon and 71.080 % oxygen by mass. The titration of 0.154 g of this compound
Topics in the November 2014 Exam Paper for CHEM1101
 November 2014 Topics in the November 2014 Exam Paper for CHEM1101 Click on the links for resources on each topic. 2014-N-2: 2014-N-3: 2014-N-4: 2014-N-5: 2014-N-7: 2014-N-8: 2014-N-9: 2014-N-10: 2014-N-11:
November 2014 Topics in the November 2014 Exam Paper for CHEM1101 Click on the links for resources on each topic. 2014-N-2: 2014-N-3: 2014-N-4: 2014-N-5: 2014-N-7: 2014-N-8: 2014-N-9: 2014-N-10: 2014-N-11:
CHEMISTRY 1A Fall 2010 Final Exam Key
 CHEMISTRY 1A Fall 2010 Final Exam Key YOU MIGHT FIND THE FOLLOWING USEFUL; 0.008314 kj H E ( n)rt R = K mol 0.00418 kj q C cal m w T g C H rxn = H f (products) H f (reactants) Electronegativities H 2.2
CHEMISTRY 1A Fall 2010 Final Exam Key YOU MIGHT FIND THE FOLLOWING USEFUL; 0.008314 kj H E ( n)rt R = K mol 0.00418 kj q C cal m w T g C H rxn = H f (products) H f (reactants) Electronegativities H 2.2
The following practice quiz contains 26 questions. The actual quiz will also contain 26 questions valued at 1 point/question
 hem 121 Quiz 3 Practice Winter 2018 The following practice quiz contains 26 questions. The actual quiz will also contain 26 questions valued at 1 point/question Name KEY G = T S PV = nrt P 1 V 1 = P 2
hem 121 Quiz 3 Practice Winter 2018 The following practice quiz contains 26 questions. The actual quiz will also contain 26 questions valued at 1 point/question Name KEY G = T S PV = nrt P 1 V 1 = P 2
Entropy, Free Energy, and Equilibrium
 Entropy, Free Energy, and Equilibrium Chapter 17 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Spontaneous Physical and Chemical Processes A waterfall runs
Entropy, Free Energy, and Equilibrium Chapter 17 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Spontaneous Physical and Chemical Processes A waterfall runs
51. Pi bonding occurs in each of the following species EXCEPT (A) CO 2 (B) C 2 H 4 (C) CN (D) C 6 H 6 (E) CH 4
 Name AP Chemistry: Bonding Multiple Choice 41. Which of the following molecules has the shortest bond length? (A) N 2 (B) O 2 (C) Cl 2 (D) Br 2 (E) I 2 51. Pi bonding occurs in each of the following species
Name AP Chemistry: Bonding Multiple Choice 41. Which of the following molecules has the shortest bond length? (A) N 2 (B) O 2 (C) Cl 2 (D) Br 2 (E) I 2 51. Pi bonding occurs in each of the following species
Chapter 8. Basic Concepts of Chemical Bonding
 Chapter 8. Basic Concepts of Chemical Bonding 8.1 Chemical Bonds, Lewis Symbols, and the Octet Rule 8.2 Ionic Bonding positive and negative ions form an ionic lattice, in which each cation is surrounded
Chapter 8. Basic Concepts of Chemical Bonding 8.1 Chemical Bonds, Lewis Symbols, and the Octet Rule 8.2 Ionic Bonding positive and negative ions form an ionic lattice, in which each cation is surrounded
CHEM J-2 June In the spaces provided, briefly explain the meaning of the following terms.
 CHEM1611 2013-J-2 June 2013 In the spaces provided, briefly explain the meaning of the following terms. Effective nuclear charge 4 The force of attraction experienced by the outer electrons of an atom.
CHEM1611 2013-J-2 June 2013 In the spaces provided, briefly explain the meaning of the following terms. Effective nuclear charge 4 The force of attraction experienced by the outer electrons of an atom.
Phase Change DIagram
 States of Matter Phase Change DIagram Phase Change Temperature remains during a phase change. Water phase changes Phase Diagram What is a phase diagram? (phase diagram for water) Normal melting point:
States of Matter Phase Change DIagram Phase Change Temperature remains during a phase change. Water phase changes Phase Diagram What is a phase diagram? (phase diagram for water) Normal melting point:
States of Matter. The Solid State. Particles are tightly packed, very close together (strong cohesive forces) Low kinetic energy (energy of motion)
 States of Matter The Solid State Particles are tightly packed, very close together (strong cohesive forces) Low kinetic energy (energy of motion) Fixed shape and volume Crystalline or amorphous structure
States of Matter The Solid State Particles are tightly packed, very close together (strong cohesive forces) Low kinetic energy (energy of motion) Fixed shape and volume Crystalline or amorphous structure
Lecture 21 Cations, Anions and Hydrolysis in Water:
 2P32 Principles of Inorganic Chemistry Dr. M. Pilkington Lecture 21 Cations, Anions and ydrolysis in Water: 1. ydration.energy 2. ydrolysis of metal cations 3. Categories of acidity and observable behavior
2P32 Principles of Inorganic Chemistry Dr. M. Pilkington Lecture 21 Cations, Anions and ydrolysis in Water: 1. ydration.energy 2. ydrolysis of metal cations 3. Categories of acidity and observable behavior
PROPERTIES OF SOLIDS SCH4U1
 PROPERTIES OF SOLIDS SCH4U1 Intra vs. Intermolecular Bonds The properties of a substance are influenced by the force of attraction within and between the molecules. Intra vs. Intermolecular Bonds Intramolecular
PROPERTIES OF SOLIDS SCH4U1 Intra vs. Intermolecular Bonds The properties of a substance are influenced by the force of attraction within and between the molecules. Intra vs. Intermolecular Bonds Intramolecular
Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks =
 Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks = 1. Which salt is colorless? (A) KMn 4 (B) BaS 4 (C) Na 2 Cr 4 (D) CoCl 2 2. Which 0.10 M aqueous solution exhibits the lowest
Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks = 1. Which salt is colorless? (A) KMn 4 (B) BaS 4 (C) Na 2 Cr 4 (D) CoCl 2 2. Which 0.10 M aqueous solution exhibits the lowest
Sample Exam Solutions. Section A. The answers provided are suggestions only and do not represent directly or otherwise official VCAA answers.
 1 Sample Exam 1 2008 Solutions The answers provided are suggestions only and do not represent directly or otherwise official VAA answers. Section A Q1 n(agl) = 4.85 / (107.9+35.5) = 0.00338 mol n(kl) =
1 Sample Exam 1 2008 Solutions The answers provided are suggestions only and do not represent directly or otherwise official VAA answers. Section A Q1 n(agl) = 4.85 / (107.9+35.5) = 0.00338 mol n(kl) =
Chapter 17. Free Energy and Thermodynamics. Chapter 17 Lecture Lecture Presentation. Sherril Soman Grand Valley State University
 Chapter 17 Lecture Lecture Presentation Chapter 17 Free Energy and Thermodynamics Sherril Soman Grand Valley State University First Law of Thermodynamics You can t win! The first law of thermodynamics
Chapter 17 Lecture Lecture Presentation Chapter 17 Free Energy and Thermodynamics Sherril Soman Grand Valley State University First Law of Thermodynamics You can t win! The first law of thermodynamics
We study bonding since it plays a central role in the understanding of chemical reactions and understanding the chemical & physical properties.
 AP Chapter 8 Notes Bonding We study bonding since it plays a central role in the understanding of chemical reactions and understanding the chemical & physical properties. Chemical Bond: holding atoms together
AP Chapter 8 Notes Bonding We study bonding since it plays a central role in the understanding of chemical reactions and understanding the chemical & physical properties. Chemical Bond: holding atoms together
Chemistry: The Central Science
 Chemistry: The Central Science Fourteenth Edition Chapter 8 Basic Concepts of Chemical Bonding Chemical Bonds Three basic types of bonds Ionic Electrostatic attraction between ions Covalent Sharing of
Chemistry: The Central Science Fourteenth Edition Chapter 8 Basic Concepts of Chemical Bonding Chemical Bonds Three basic types of bonds Ionic Electrostatic attraction between ions Covalent Sharing of
Chem 12: Chapters 10, 11, 12, 13, 14 Unit 3 Worksheet
 C h e m i s t r y 1 2 U n i t 3 R e v i e w P a g e 1 Chem 12: Chapters 10, 11, 12, 13, 14 Unit 3 Worksheet 1. What is miscible? Immiscible? 2. What is saturated? Unsaturated? Supersaturated? 3. How does
C h e m i s t r y 1 2 U n i t 3 R e v i e w P a g e 1 Chem 12: Chapters 10, 11, 12, 13, 14 Unit 3 Worksheet 1. What is miscible? Immiscible? 2. What is saturated? Unsaturated? Supersaturated? 3. How does
Water, water everywhere,; not a drop to drink. Consumption resulting from how environment inhabited Deforestation disrupts water cycle
 Chapter 3 Water: The Matrix of Life Overview n n n Water, water everywhere,; not a drop to drink Only 3% of world s water is fresh How has this happened Consumption resulting from how environment inhabited
Chapter 3 Water: The Matrix of Life Overview n n n Water, water everywhere,; not a drop to drink Only 3% of world s water is fresh How has this happened Consumption resulting from how environment inhabited
Intermolecular Forces
 Intermolecular Forces H covalent bond (stronger) Cl H Cl intermolecular attraction (weaker) The attractions between molecules are not nearly as strong as the covalent bonds that hold atoms together. They
Intermolecular Forces H covalent bond (stronger) Cl H Cl intermolecular attraction (weaker) The attractions between molecules are not nearly as strong as the covalent bonds that hold atoms together. They
Chapter 11. Liquids and Intermolecular Forces
 Chapter 11 Liquids and Intermolecular Forces States of Matter The three states of matter are 1) Solid Definite shape Definite volume 2) Liquid Indefinite shape Definite volume 3) Gas Indefinite shape Indefinite
Chapter 11 Liquids and Intermolecular Forces States of Matter The three states of matter are 1) Solid Definite shape Definite volume 2) Liquid Indefinite shape Definite volume 3) Gas Indefinite shape Indefinite
OFB Chapter 6 Condensed Phases and Phase Transitions
 OFB Chapter 6 Condensed Phases and Phase Transitions 6-1 Intermolecular Forces: Why Condensed Phases Exist 6- The Kinetic Theory of Liquids and Solids 6-3 Phase Equilibrium 6-4 Phase Transitions 6-5 Phase
OFB Chapter 6 Condensed Phases and Phase Transitions 6-1 Intermolecular Forces: Why Condensed Phases Exist 6- The Kinetic Theory of Liquids and Solids 6-3 Phase Equilibrium 6-4 Phase Transitions 6-5 Phase
16 years ago TODAY (9/11) at 8:46, the first tower was hit at 9:03, the second tower was hit. Lecture 2 (9/11/17)
 16 years ago TODAY (9/11) at 8:46, the first tower was hit at 9:03, the second tower was hit By Anthony Quintano - https://www.flickr.com/photos/quintanomedia/15071865580, CC BY 2.0, https://commons.wikimedia.org/w/index.php?curid=38538291
16 years ago TODAY (9/11) at 8:46, the first tower was hit at 9:03, the second tower was hit By Anthony Quintano - https://www.flickr.com/photos/quintanomedia/15071865580, CC BY 2.0, https://commons.wikimedia.org/w/index.php?curid=38538291
Ch 9 Liquids & Solids (IMF) Masterson & Hurley
 Ch 9 Liquids & Solids (IMF) Masterson & Hurley Intra- and Intermolecular AP Questions: 2005 Q. 7, 2005 (Form B) Q. 8, 2006 Q. 6, 2007 Q. 2 (d) and (c), Periodic Trends AP Questions: 2001 Q. 8, 2002 Q.
Ch 9 Liquids & Solids (IMF) Masterson & Hurley Intra- and Intermolecular AP Questions: 2005 Q. 7, 2005 (Form B) Q. 8, 2006 Q. 6, 2007 Q. 2 (d) and (c), Periodic Trends AP Questions: 2001 Q. 8, 2002 Q.
Copyright McGraw-Hill Education. Permission required for reproduction or display : A force that holds atoms together in a molecule or compound
 : Chemical Bonding 8-1 8.1 Types of Bonds : A force that holds atoms together in a molecule or compound Two types of chemical bonds Ionic Bonds Covalent Bonds 8-2 1 8.1 Types of Bonds 8-3 8.1 Types of
: Chemical Bonding 8-1 8.1 Types of Bonds : A force that holds atoms together in a molecule or compound Two types of chemical bonds Ionic Bonds Covalent Bonds 8-2 1 8.1 Types of Bonds 8-3 8.1 Types of
Chapter 10. Dipole Moments. Intermolecular Forces (IMF) Polar Bonds and Polar Molecules. Polar or Nonpolar Molecules?
 Polar Bonds and Polar Molecules Chapter 10 Liquids, Solids, and Phase Changes Draw Lewis Structures for CCl 4 and CH 3 Cl. What s the same? What s different? 1 Polar Covalent Bonds and Dipole Moments Bonds
Polar Bonds and Polar Molecules Chapter 10 Liquids, Solids, and Phase Changes Draw Lewis Structures for CCl 4 and CH 3 Cl. What s the same? What s different? 1 Polar Covalent Bonds and Dipole Moments Bonds
Review for Final Exam
 Review for Final Exam Chapter 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Types of Changes A physical change does not alter the composition or identity
Review for Final Exam Chapter 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Types of Changes A physical change does not alter the composition or identity
Full file at
 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following is an uncharged particle found in the nucleus of 1) an atom and which has
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following is an uncharged particle found in the nucleus of 1) an atom and which has
Name Date Class MOLECULAR COMPOUNDS. Distinguish molecular compounds from ionic compounds Identify the information a molecular formula provides
 8.1 MOLECULAR COMPOUNDS Section Review Objectives Distinguish molecular compounds from ionic compounds Identify the information a molecular formula provides Vocabulary covalent bond molecule diatomic molecule
8.1 MOLECULAR COMPOUNDS Section Review Objectives Distinguish molecular compounds from ionic compounds Identify the information a molecular formula provides Vocabulary covalent bond molecule diatomic molecule
Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks =
 Name:. orrect Questions = Wrong Questions =.. Unattempt Questions = Marks = 1. (11%) A(g) + 3B(g) r 2(g) Use the tabulated data to answer the questions about this reaction, which is carried out in a 1.0
Name:. orrect Questions = Wrong Questions =.. Unattempt Questions = Marks = 1. (11%) A(g) + 3B(g) r 2(g) Use the tabulated data to answer the questions about this reaction, which is carried out in a 1.0
Chapter 8 Covalent Boding
 Chapter 8 Covalent Boding Molecules & Molecular Compounds In nature, matter takes many forms. The noble gases exist as atoms. They are monatomic; monatomic they consist of single atoms. Hydrogen chloride
Chapter 8 Covalent Boding Molecules & Molecular Compounds In nature, matter takes many forms. The noble gases exist as atoms. They are monatomic; monatomic they consist of single atoms. Hydrogen chloride
States of matter Part 1
 Physical pharmacy I 1. States of matter (2 Lectures) 2. Thermodynamics (2 Lectures) 3. Solution of non-electrolyte 4. Solution of electrolyte 5. Ionic equilibria 6. Buffered and isotonic solution Physical
Physical pharmacy I 1. States of matter (2 Lectures) 2. Thermodynamics (2 Lectures) 3. Solution of non-electrolyte 4. Solution of electrolyte 5. Ionic equilibria 6. Buffered and isotonic solution Physical
General Chemistry I. Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University. Module 4: Chemical Thermodynamics
 General Chemistry I Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University Module 4: Chemical Thermodynamics Zeroth Law of Thermodynamics. First Law of Thermodynamics (state quantities:
General Chemistry I Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University Module 4: Chemical Thermodynamics Zeroth Law of Thermodynamics. First Law of Thermodynamics (state quantities:
2.2.2 Bonding and Structure
 2.2.2 Bonding and Structure Ionic Bonding Definition: Ionic bonding is the electrostatic force of attraction between oppositely charged ions formed by electron transfer. Metal atoms lose electrons to form
2.2.2 Bonding and Structure Ionic Bonding Definition: Ionic bonding is the electrostatic force of attraction between oppositely charged ions formed by electron transfer. Metal atoms lose electrons to form
Definition: An Ionic bond is the electrostatic force of attraction between oppositely charged ions formed by electron transfer.
 3 Bonding Definition An Ionic bond is the electrostatic force of attraction between oppositely charged ions formed by electron transfer. Metal atoms lose electrons to form +ve ions. on-metal atoms gain
3 Bonding Definition An Ionic bond is the electrostatic force of attraction between oppositely charged ions formed by electron transfer. Metal atoms lose electrons to form +ve ions. on-metal atoms gain
Chapter 9 MODELS OF CHEMICAL BONDING
 Chapter 9 MODELS OF CHEMICAL BONDING 1 H H A + B H H A B A comparison of metals and nonmetals 2 9.1 Atomic Properties & Chemical Bonds Chemical bond: A force that holds atoms together in a molecule or
Chapter 9 MODELS OF CHEMICAL BONDING 1 H H A + B H H A B A comparison of metals and nonmetals 2 9.1 Atomic Properties & Chemical Bonds Chemical bond: A force that holds atoms together in a molecule or
SUPPLEMENTAL HOMEWORK SOLUTIONS WEEK 8
 SUPPLEMETAL MEWRK SLUTIS WEEK 8 Assignment for Tuesday, March 7 th 7.36 a) + + b) + + c) 6 14 4 + 6 14 4 + + d) 6 5 3 7 + 6 5 7 + Be sure to write the correct charges for the products. 7.4 a) l + l + b)
SUPPLEMETAL MEWRK SLUTIS WEEK 8 Assignment for Tuesday, March 7 th 7.36 a) + + b) + + c) 6 14 4 + 6 14 4 + + d) 6 5 3 7 + 6 5 7 + Be sure to write the correct charges for the products. 7.4 a) l + l + b)
4. Based on the following thermochemical equation below, which statement is false? 2 NH 3 (g) N 2 (g) + 3 H 2 (g) H = kj
 CHEM 101 WINTER 09-10 EXAM 3 On the answer sheet (Scantron) write you name, student ID number, and recitation section number. Choose the best (most correct) answer for each question and enter it on your
CHEM 101 WINTER 09-10 EXAM 3 On the answer sheet (Scantron) write you name, student ID number, and recitation section number. Choose the best (most correct) answer for each question and enter it on your
