Combined GCSE OCR Revision Science magnification tur tur magnification ell Struc ell Struc ells ells Combined GCSE OCR Revision Science
|
|
|
- Evelyn Hoover
- 6 years ago
- Views:
Transcription
1 Cell Structures How is the magnification of a light microscope calculated? Cell Structures Total magnification is calculated by multiplying the magnification of the eyepiece lens by the magnification of the objective lens. 1 1 What Happens in Cells What term is used to describe the shape of a DNA molecule? 2 What Happens in Cells A DNA molecule forms the shape of a double helix. 2 Respiration What is the word equation for aerobic respiration? Respiration glucose + oxygen carbon dioxide + water (+ energy released) 3 3 Photosynthesis What are the products of photosynthesis? Photosynthesis The products of photosynthesis are glucose and oxygen. 4 4 Supplying the Cell What is the process by which water molecules move across a partially permeable membrane from a dilute solution to a more concentrated one? 5 Supplying the Cell Osmosis is the movement of water molecules from a dilute to a more concentrated solution across a partially permeable membrane. 5
2 The Challenges of Size Exchange surfaces allow efficient transport of materials. What are the three main features of an efficient exchange system? 6 The Challenges of Size An efficient exchange system should have: (1) a large surface-area-to-volume ratio; (2) thin membranes so the diffusion distance is short; (3) a good supply of transport medium (e.g. blood, air, etc.). 6 The Heart and Blood Cells What are the three types of blood vessel? The Heart and Blood Cells The three types of blood vessel are arteries, capillaries and veins. 7 7 Plants, Water and Minerals What is translocation? 8 Plants, Water and Minerals Translocation is the movement of glucose from the leaf (where it is produced) to other parts of the plant (where it is needed) via the phloem. 8 Coordination and Control The nervous system is composed of two parts. What are they? 9 Coordination and Control The nervous system is composed of the central nervous system (the brain and the spinal cord) and the peripheral nervous system (all the other nerve cells that connect to the central nervous system). 9 The Endocrine System Hormones are chemical messengers produced by glands. How do they reach their target organ? 10 The Endocrine System Hormones are released directly into the blood, which transports them to their target organ. 10
3 Hormones and Their Uses What two hormones are combined in most contraceptive pills? Hormones and Their Uses Most contraceptive pills contain oestrogen and progesterone Maintaining Internal Environments What triggers the pancreas to produce insulin? 12 Maintaining Internal Environments High blood sugar levels cause the pancreas to produce insulin. 12 Recycling Photosynthesis removes carbon dioxide from the atmosphere. What two processes release carbon dioxide to the Recycling Respiration (animal, plant and microbial) and combustion release carbon dioxide to the atmosphere. atmosphere? Interdependence What is the name of the relationship between two organisms where both depend on each other and both benefit? 14 Interdependence Mutualism. In a mutualistic relationship, both organisms depend on each other and both benefit. 14 The allele for brown eyes is dominant to Genes the allele for blue eyes. What will be the phenotype of a person who is heterozygous Genes The person will have brown eyes. for eye colour? 15 15
4 Genetics and Reproduction What type of cell division produces gametes? Genetics and Reproduction Meiosis produces gametes Natural Selection and Evolution For evolution to occur, there must be genetic variation in a population. How might genetic variation arise? 17 Natural Selection and Evolution Variation can arise through mutations in genes. 17 Monitoring and Maintaining the Environment What piece of equipment is used to sample the number of plant species in a field? Monitoring and Maintaining the Environment A quadrat is used to sample the number of plant species in a field Investigations In an investigation, how would you make sure your measurements are reliable and identify any that might be anomalous? Investigations All measurements should be repeated. If a single reading is very different to the others (anomalous), this might indicate that an error has been made in measuring Feeding the Human Race How is selective breeding different from genetic engineering? 20 Feeding the Human Race Selective breeding is the traditional, natural process of breeding plants and animals with certain, desirable genetic features. Genetic engineering is a modern, faster way of bringing about changes in organisms. It is the artificial process of transplanting genes for a desired characteristic into an organism. 20
5 Monitoring and Maintaining Health What are the four main human defences to stop microorganisms entering the body? 21 Monitoring and Maintaining Health Microorganisms are prevented from entering the body by the skin (a physical barrier), platelets that help the blood to clot and seal wounds, mucous in the lungs that traps microorganisms and acid in the stomach that destroys microorganisms. 21 Prevention and Treatment of Disease Why wouldn t you use antibiotics to treat a cold caused by a virus? 22 Prevention and Treatment of Disease Antibiotics are used for bacterial infections. They have no effect on viruses, which are found inside the cell. 22 Non-Communicable Diseases What three options can be used to treat cardiovascular disease? 23 Non-Communicable Diseases Cardiovascular disease can be treated with lifestyle changes (healthy eating and exercise, stopping smoking), surgery (heart transplants or stents) and medications (statins or aspirin). 23 Particle Model and Atomic Structure The following symbols describe two different substances. Deduce all the information you can from these symbols. 13 C 12 C Particle Model and Atomic Structure The substances are isotopes of the same element, carbon. The atomic number of carbon is 6 and the mass numbers of the isotopes are 13 and 12. An atom of carbon-13 contains 6 protons and 7 neutrons. An atom of carbon-12 contains 6 protons and 6 neutrons. 24 Purity and Separating Mixtures Explain how the three different types of chromatography can be used to separate a mixture. 25 Purity and Separating Mixtures Chromatography separates mixtures using a stationary phase and a mobile phase. Paper chromatography is used to separate mixtures of coloured dyes in solution. Thin layer chromatography uses a thin layer of an inert solid as the stationary phase. Gas chromatography separates mixtures of gases by passing them through a solid stationary phase. 25
6 Bonding Describe the two main types of chemical bond that can form between two different elements. 26 Bonding An ionic bond is formed when one or more electrons are donated from an atom of one element to an atom of another element, so both atoms have full outer electron shells. The atoms form electrically charged ions. A covalent bond is formed when two atoms share electrons to complete the outer electron shells of both atoms. 26 Models of Bonding Explain the differences between simple molecules, polymers and metals, referring to how they are formed and the bonding that takes place. 27 Models of Bonding Simple molecules are formed when two or more atoms share electrons and form covalent bonds. Polymers are formed when repeated units of smaller molecules bond covalently to form a long chain. Metal atoms have outermost electrons that can move freely from one metal atom to another. They are held together by strong metallic bonds. 27 Properties of Materials Explain the difference between intermolecular forces and intramolecular forces. 28 Properties of Materials Intermolecular forces are the forces between molecules. Intramolecular forces are the forces between the atoms inside a molecule, such as covalent bonds. 28 Introducing Chemical Complete the table. State of substance State symbol (s) liquid ( ) (g) ( ) dissolved ( ) in water 29 Introducing Chemical State of substance solid liquid gas (aqueous) dissolved in water State symbol (s) (l) (g) (aq) 29 Chemical Equations Simplify and balance the following ionic equation, which shows the reaction between copper sulfate and sodium hydroxide solutions to form solid copper hydroxide. Cu 2+ (aq) + SO 4 2 (aq) + Na + (aq) + OH (aq) Cu(OH) 2 (s) + Na + (aq) + SO 4 2 (aq) 30 Chemical Equations Cu 2+ (aq) + 2OH (aq) Cu(OH) 2 (s) (The sodium and sulfate ions are spectator ions that do not change during the reaction, so they can be deleted from both sides.) 30
7 Moles and Mass Which of the following sentences are true and which are false? A. Two moles of calcium atoms contain a total of just over atoms. B. Carbon has a lower relative atomic mass than calcium, so two moles of carbon atoms contain fewer than atoms. 31 Moles and Mass A is true: one mole is atoms, so = atoms B is false: one mole of any element always contains atoms 31 Energetics What is the activation energy of a reaction and why is it not the same as the total energy given out or taken in during the reaction? 32 Energetics For most chemical reactions, energy is needed to break chemical bonds so the reaction can start. This is the activation energy. Any new bonds that form will cause some energy to be given out, so the total energy of the reaction is not the same as the activation energy. 32 Types of Chemical Use these words to complete the sentences that follow. (You do not need to use all the words.) separately oxygen together reduction gains In oxidation reactions, a substance often oxygen. In reactions, a substance often loses. 33 These two types of reaction always occur. Types of Chemical In oxidation reactions, a substance often gains oxygen. In reduction reactions, a substance often loses oxygen. These two types of reaction always occur together. 33 ph, Acids and Neutralisation Explain the difference between a strong acid and a weak acid. 34 ph, Acids and Neutralisation A strong acid easily forms H + ions, so the acids fully ionise. A weak acid forms an equilibrium mixture, so that some of the ions formed can recombine into the original acid. 34 Electrolysis Use these words to complete the sentences that follow. positive negative anode dissociate electrolyte cathode In electrolysis, the solution containing the ionic compound is called the. In solution, the ions in the compound. The negative electrode is the and attracts ions. The positive electrode is the 35 and attracts ions. Electrolysis In electrolysis, the solution containing the ionic compound is called the electrolyte. In solution, the ions in the compound dissociate. The negative electrode is the cathode and attracts positive ions. The positive electrode is the anode and attracts negative ions. 35
8 Predicting Chemical Explain the differences in electron shells between atoms of Group 1, Group 7 and Group 0 elements and suggest what happens to these elements in chemical reactions. 36 Predicting Chemical Group 1 elements all have one electron in their outer shell. In a chemical reaction, they tend to lose this outer electron so that the new outer shell is complete. Group 7 elements all have seven electrons in their outer shell. They tend to gain one electron so the outer shell is complete. Group 0 elements all have a complete outer shell of 36 electrons. They are unreactive. Controlling Chemical A sample of solid calcium carbonate is divided precisely into two equal masses. One half is a single solid piece, which is then reacted with an acid. The other half is broken into small pieces and reacted with a fresh sample of the same acid. Which half will react faster, and why? 37 Controlling Chemical The half that is broken into small pieces will react faster. This is because small pieces have a large surface area in relation to their volume. More solid particles are exposed to contact with acid particles, so there are more collisions and a faster reaction. 37 Catalysts and Activation Energy What is a catalyst? 38 Catalysts and Activation Energy A catalyst is a substance that speeds up the rate of a chemical reaction without being used up or changed in the reaction. 38 Equilibria State Le Chatelier s principle. Equilibria Le Chatelier s principle: When the conditions of a system are altered, the position of the equilibrium changes to try and restore the original conditions Improving Processes and Products How is carbon useful in the extraction of metals? 40 Improving Processes and Products Most metals are found naturally as minerals (compounds). Carbon can displace less reactive metals from their mineral oxides. The carbon is heated with the metal oxide, and the pure metal is extracted. 40
9 Life Cycle Assessments and Recycling According to a life cycle assessment, what are the four stages in the life of a product? 41 Life Cycle Assessments and Recycling 1 Obtaining raw materials. 2 Manufacture of the product. 3 Use of the product. 4 Disposal of the product when it is no longer useful. 41 Crude Oil Describe the conditions needed for the reaction called cracking, and explain why this reaction is useful. 42 Crude Oil Cracking requires a catalyst, high temperature and high pressure. Cracking breaks down some of the large molecules in crude oil to form smaller, more useful molecules. 42 Interpreting and Interacting with Earth s Systems Suggest three ways in which we could slow down climate change. 43 Interpreting and Interacting with Earth s Systems Any three from: Use less fossil fuels. Develop and use alternative energy sources. Improve energy efficiency/cut down on wasted energy. Plant new forests that can change carbon dioxide into oxygen. Reduce the amount of waste we produce, to cut down the amount of methane 43 gas in the air. Air Pollution and Potable Water Why have many governments passed laws restricting the amounts of particulates that can be emitted? 44 Air Pollution and Potable Water Particulates in the air can cause lung problems and respiratory diseases. They can coat buildings and trees. Laws to restrict their emissions aim to improve air quality. 44 Matter, Models and Density What is a typical size of an atom? Choose from the following m m m Matter, Models and Density The size of an atom is of the order of m
10 Temperature and State What is the difference between the specific heat capacity and the specific latent heat of a material? 46 Temperature and State Specific heat capacity is the energy needed to raise the temperature of 1 kg of the material by 1 C, with no change of state. Specific latent heat is the energy needed to change the state of 1 kg of the material, with no change in temperature. 46 Journeys State the equation for calculating the kinetic energy of a moving object, and give the unit of each quantity. 47 Journeys Kinetic energy = 0.5 mass (speed) 2 Unit of kinetic energy: joule (J) Unit of mass: kilogram (kg) Unit of speed: metres per second (m/s) 47 Forces Which equation is a statement of Newton s second law? Forces Force = mass acceleration is a statement of Newton s second law Force, Energy and Power What is meant by work in physics? 49 Force, Energy and Power Work is done on an object when a force causes the object to move through a distance. Work done = force distance (along the line of action of the force) The work done is equal to the energy transferred. 49 Changes of Shape What is the difference between elastic deformation and plastic deformation? 50 Changes of Shape Elastic deformation: forces make an object change shape, but it returns to its original shape when the forces are removed. Plastic deformation: forces make an object change shape, and the object keeps its new shape when the forces are removed. 50
11 Friction transfers electrons Electric Charge How does friction cause objects to become charged? 51 Electric Charge between two objects that are rubbed together. This leaves one object with an excess of electrons (making it negatively charged) and the other object with a shortage of electrons (making it positively charged). 51 Circuits What is the relationship between the potential difference across, the current through and the resistance of Circuits Potential difference = current resistance a component in a circuit? Resistors and Energy Transfers State the equation for calculating the electrical power of a device in terms of its resistance and the current through it, and state the unit of power. 53 Resistors and Energy Transfers Power = (current) 2 resistance The unit of power is the watt, W (equivalent to J/s). 53 Magnetic Fields and Motors Describe the basic structure of an electromagnet and how it works. 54 Magnetic Fields and Motors An electromagnet is a coil of wire of many turns wound on an iron core. When current is passed through the coil, a strong magnetic field is set up through the core and around the coil. 54 Wave Behaviour What is a wavelength and what is its unit? 55 Wave Behaviour A wavelength is the distance from one point on a wave to the equivalent point on the next wave. Its unit is metre (m). 55
12 Electromagnetic Radiation List the types of radiation in the electromagnetic spectrum, in order of increasing frequency. 56 Electromagnetic Radiation In order of increasing frequency: radio waves, microwaves, infrared radiation, visible light, ultraviolet (UV) radiation, X-rays, gamma rays. 56 Nuclei of Atoms Which type of radioactive emission is least penetrating, and why? alpha beta gamma Nuclei of Atoms Alpha radiation is least penetrating because it loses its energy in the shortest distance, by strongly ionising the atoms of a material Half-Life Define the half-life of a radioactive material, and explain why radioactive decay can be considered random. Half-Life The half-life of a radioactive material is the time taken for the number of undecayed nuclei in a sample of the material to reduce by half. The decay of a particular nucleus is unpredictable, so the decay is described as random Systems and Transfers True or false? If all of the electrical energy supplied to an efficient kettle is used to heat the water, this equation determines the change in temperature of the water. potential difference current time = mass of water specific heat capacity of water change in temperature 59 Systems and Transfers True. The electrical energy supplied to the kettle is potential difference charge = potential difference current time. The rise in temperature of the water depends on the mass and the specific heat capacity of the water. The thermal energy change of water is mass specific heat capacity change in temperature. 59 Energy, Power and Efficiency When a device transfers energy, some energy is wasted. How does the energy transfer obey the law of conservation of energy? 60 Energy, Power and Efficiency Energy is said to be wasted when the useful output energy of a device is less than the input (supplied) energy. This does not contravene the law of conservation of energy because the wasted energy is dissipated to the surroundings, raising the temperature. 60
13 Physics on the Road What is a typical speed for a cyclist on a clear flat road? Choose from: 1 m/s 10 m/s 100 m/s 61 Physics on the Road A typical speed for a cyclist on a clear flat road is 10 m/s. A person walking slowly would have a speed of about 1 m/s. A plane, or an extremely fast train, might have a speed of 100 m/s. 61 Energy for the World Give some types of bio-fuel, and explain whether bio-fuels are renewable or non-renewable energy resources. 62 Energy for the World Types of bio-fuel include: wood; oils and bio-diesels from crops such as rape and palm; also bio-ethanol from crops such as sugar cane. Bio-fuels are renewable energy resources, because we can plant more trees and crops. 62 Energy at Home Why are transformers used in the national grid? 63 Energy at Home Transformers are used in the national grid to increase (step up) the generated voltage to a high value for transmission around the country, because there is then less energy loss from the cables. Transformers are then used to reduce (step down) the voltage to a safer and more convenient voltage for the user. 63
Combined GCSE OCR Revision Science magnification tur tur magnification ell Struc ell Struc ells ells Combined GCSE OCR Revision Science
 Cell Structures How is the magnification of a light microscope calculated? Cell Structures Total magnification is calculated by multiplying the magnification of the eyepiece lens by the magnification of
Cell Structures How is the magnification of a light microscope calculated? Cell Structures Total magnification is calculated by multiplying the magnification of the eyepiece lens by the magnification of
GCSE OCR Revision Chemistry. GCSE OCR Revision Chemistry. GCSE OCR Revision Chemistry. Bonding. GCSE OCR Revision Chemistry
 Particle Model and Atomic Structure The following symbols describe two different substances. Deduce all the information you can from these symbols. 13 C 12 6 6 C 1 Particle Model and Atomic Structure The
Particle Model and Atomic Structure The following symbols describe two different substances. Deduce all the information you can from these symbols. 13 C 12 6 6 C 1 Particle Model and Atomic Structure The
GCSE OCR Revision Physics. GCSE OCR Revision Physics. GCSE OCR Revision Physics. GCSE OCR Revision Physics. Journeys. GCSE OCR Revision Physics
 Matter, Models and Density What is a typical size of an atom? Choose from the following. 10 15 m 10 12 m 10 10 m Matter, Models and Density The size of an atom is of the order of 10 10 m. 1 1 Temperature
Matter, Models and Density What is a typical size of an atom? Choose from the following. 10 15 m 10 12 m 10 10 m Matter, Models and Density The size of an atom is of the order of 10 10 m. 1 1 Temperature
Cell Structure. Cell Structure. Investigating Cells. Investigating Cells. Cell Division. Cell Division. Transport In and Out of Cells
 Cell Structure What are the two main types of cell? Cell Structure The two main types of cell are prokaryotic and eukaryotic. 1 1 Investigating Cells How is magnification calculated? 2 Investigating Cells
Cell Structure What are the two main types of cell? Cell Structure The two main types of cell are prokaryotic and eukaryotic. 1 1 Investigating Cells How is magnification calculated? 2 Investigating Cells
Biology Paper 1 1hr 15mins 70 marks
 Biology Paper 1 1hr 15mins 70 marks Cell Biology (Yr9) Cell Organisation (Yr9) Infection and Response (Yr10) Bioenergetics (Yr10) Cells Cell Organisation Competition Photosynthesis Microscopy Enzymes Abiotic
Biology Paper 1 1hr 15mins 70 marks Cell Biology (Yr9) Cell Organisation (Yr9) Infection and Response (Yr10) Bioenergetics (Yr10) Cells Cell Organisation Competition Photosynthesis Microscopy Enzymes Abiotic
Science Years 9 to 10
 Boardworks Contents Guide Boardworks Presentations: Acids and metal oxides 10 slides Reactions of metal oxides with acids. Adapting to changes 9 slides Ways that animals adapt to their habitats. Air pollution
Boardworks Contents Guide Boardworks Presentations: Acids and metal oxides 10 slides Reactions of metal oxides with acids. Adapting to changes 9 slides Ways that animals adapt to their habitats. Air pollution
Edexcel Chemistry Checklist
 Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
OCR Chemistry Checklist
 Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
AQA Chemistry Checklist
 Topic 1. Atomic structure Video: Atoms, elements, compounds, mixtures Use the names and symbols of the first 20 elements in the periodic table, the elements in Groups 1 and 7, and other elements in this
Topic 1. Atomic structure Video: Atoms, elements, compounds, mixtures Use the names and symbols of the first 20 elements in the periodic table, the elements in Groups 1 and 7, and other elements in this
Subject: GCSE Physics
 Subject: GCSE Physics Density States of matter SHC Atoms and radiation Discovery of the nucleus Changes in the nucleus Alpha, beta and gamma Activity and half life Nuclear radiation and medicine Nuclear
Subject: GCSE Physics Density States of matter SHC Atoms and radiation Discovery of the nucleus Changes in the nucleus Alpha, beta and gamma Activity and half life Nuclear radiation and medicine Nuclear
This unit will help you define health, learn about some pathogens and the diseases they cause, medicines and about the immune system.
 YEAR 10 Biology Units 1-5 Biology Unit CB1 Key biological concepts [Paper 1 & CB2 Cells and Control CB3 Genetics CB4 Natural Selection and Genetic Modification CB5 Health, Disease and the Development of
YEAR 10 Biology Units 1-5 Biology Unit CB1 Key biological concepts [Paper 1 & CB2 Cells and Control CB3 Genetics CB4 Natural Selection and Genetic Modification CB5 Health, Disease and the Development of
Biology Combined Chemistry Combined Physics Combined. Content Hours Content Hours Content Hours
 Biology Combined Chemistry Combined Physics Combined Content Hours Content Hours Content Hours Half Term.7.. Communities Competition Ecosystems Interdependence ASSESSED HOMEWORK (Communities).7.. Abiotic
Biology Combined Chemistry Combined Physics Combined Content Hours Content Hours Content Hours Half Term.7.. Communities Competition Ecosystems Interdependence ASSESSED HOMEWORK (Communities).7.. Abiotic
Atoms, Elements, Atoms, Elements, Compounds and Mixtures. Compounds and Mixtures. Atoms and the Periodic Table. Atoms and the.
 Atoms, Elements, Compounds and Mixtures Explain how fractional distillation can be used to separate a mixture. 1 Atoms, Elements, Compounds and Mixtures Fractional distillation is used to separate components
Atoms, Elements, Compounds and Mixtures Explain how fractional distillation can be used to separate a mixture. 1 Atoms, Elements, Compounds and Mixtures Fractional distillation is used to separate components
Introduction. Introduction. Forces An. Forces An. Forces in Action. Forces in Action. Pressure and Pressure. Pressure and Pressure.
 Forces An Introduction A force is a vector quantity. What does this mean? Forces An Introduction A vector quantity, such as force, has a direction as well as a magnitude. 1 1 Forces in Action The moment
Forces An Introduction A force is a vector quantity. What does this mean? Forces An Introduction A vector quantity, such as force, has a direction as well as a magnitude. 1 1 Forces in Action The moment
Science. synthesis 2 Analysis and synthesis Using physics to make things work 3 Magnetic fields to keep things moving Energy calculations 4 Energy
 Year 11 (Triple science) 2016-17 Half-term Topic Week 1 Week 2 Week 3 Week 4 Week 5 Week 6 Week 7 1 Exchange of materials Keeping internal conditions constant Analysis and synthesis 2 Analysis and synthesis
Year 11 (Triple science) 2016-17 Half-term Topic Week 1 Week 2 Week 3 Week 4 Week 5 Week 6 Week 7 1 Exchange of materials Keeping internal conditions constant Analysis and synthesis 2 Analysis and synthesis
Science 9-1 Grades Guidance
 U1 U2 U3 Science 9-1 Grades Guidance This guide is designed to help explain the level that a student is working at using examples of the curriculum that a student should be able to access. This is not
U1 U2 U3 Science 9-1 Grades Guidance This guide is designed to help explain the level that a student is working at using examples of the curriculum that a student should be able to access. This is not
AQA Chemistry (Combined Science) Specification Checklists. Name: Teacher:
 AQA Chemistry (Combined Science) Specification Checklists Name: Teacher: Paper 1-4.1 Atomic structure and the periodic table 4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic
AQA Chemistry (Combined Science) Specification Checklists Name: Teacher: Paper 1-4.1 Atomic structure and the periodic table 4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic
OCR Biology Checklist
 Topic 1. Cell level systems Video: Eukaryotic and prokaryotic cells Compare the structure of animal and plant cells. Label typical and atypical prokaryotic cells. Compare prokaryotic and eukaryotic cells.
Topic 1. Cell level systems Video: Eukaryotic and prokaryotic cells Compare the structure of animal and plant cells. Label typical and atypical prokaryotic cells. Compare prokaryotic and eukaryotic cells.
OCR Biology Checklist
 Topic 1. Cell level systems Video: Eukaryotic and prokaryotic cells Compare the structure of animal and plant cells. Label typical and atypical prokaryotic cells. Compare prokaryotic and eukaryotic cells.
Topic 1. Cell level systems Video: Eukaryotic and prokaryotic cells Compare the structure of animal and plant cells. Label typical and atypical prokaryotic cells. Compare prokaryotic and eukaryotic cells.
Switching to AQA from OCR Gateway Science B and Additional Science B
 Switching to AQA from OCR Gateway Science B and Additional Science B If you re thinking of switching to AQA from OCR Science B (Gateway) and Additional Science B (J261 and J262) for teaching from September
Switching to AQA from OCR Gateway Science B and Additional Science B If you re thinking of switching to AQA from OCR Science B (Gateway) and Additional Science B (J261 and J262) for teaching from September
Science Year 10 Unit 1 Biology
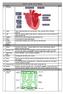 Week 1: 1. The Heart Science Year 10 Unit 1 Biology RAG 2. Artery Takes oxygenated blood away from the heart. Thick, muscular walls to withstand pressure. 3. Vein Takes deoxygenated blood towards the heart.
Week 1: 1. The Heart Science Year 10 Unit 1 Biology RAG 2. Artery Takes oxygenated blood away from the heart. Thick, muscular walls to withstand pressure. 3. Vein Takes deoxygenated blood towards the heart.
Lymm High School- KS3 Life after levels - Science Y9
 Biology BRONZE SILVER GOLD PLATINUM D and below= GCSE 1,2,3 C= GCSE 4 C/B= GCSE 5,6 A/A*= GCSE 7,8,9 Explain that cells are very Demonstrate an small and a microscope is understanding of the scale needed
Biology BRONZE SILVER GOLD PLATINUM D and below= GCSE 1,2,3 C= GCSE 4 C/B= GCSE 5,6 A/A*= GCSE 7,8,9 Explain that cells are very Demonstrate an small and a microscope is understanding of the scale needed
GCSE CHEMISTRY REVISION LIST
 GCSE CHEMISTRY REVISION LIST OCR Gateway Chemistry (J248) from 2016 Topic C1: Particles C1.1 Describe the main features of the particle model in terms of states of matter and change of state Explain, in
GCSE CHEMISTRY REVISION LIST OCR Gateway Chemistry (J248) from 2016 Topic C1: Particles C1.1 Describe the main features of the particle model in terms of states of matter and change of state Explain, in
KS4 CURRICULUM. Science - BTEC
 KS4 CURRICULUM Science - BTEC Students have four lessons per fortnight completing a BTEC in the Principles of Applied Science. This is the equivalent to one GCSE. Unit One Principles of Science; externally
KS4 CURRICULUM Science - BTEC Students have four lessons per fortnight completing a BTEC in the Principles of Applied Science. This is the equivalent to one GCSE. Unit One Principles of Science; externally
8/24/2018. Bio 1101 Lecture 2 (guided) Chapters 2: Essential Chemistry. Chapter 2: Essential Chemistry for Biology
 1 2 3 4 5 Bio 1101 Lecture 2 (guided) Chapters 2: Essential Chemistry Chapter 2: Essential Chemistry for Biology Levels of biological organization Ecosystem (e.g. savanna) Community (e.g. the organisms
1 2 3 4 5 Bio 1101 Lecture 2 (guided) Chapters 2: Essential Chemistry Chapter 2: Essential Chemistry for Biology Levels of biological organization Ecosystem (e.g. savanna) Community (e.g. the organisms
0654 CO-ORDINATED SCIENCES
 CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education MARK SCHEME for the May/June 2013 series 0654 CO-ORDINATED SCIENCES 0654/22 Paper 2 (Core Theory), maximum
CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education MARK SCHEME for the May/June 2013 series 0654 CO-ORDINATED SCIENCES 0654/22 Paper 2 (Core Theory), maximum
AQA TRILOGY Chemistry (8464) from 2016 Topics T5.1 Atomic structure and the periodic table (Paper 1) To pic. Student Checklist
 Personalised Learning Checklist AQA TRILOGY Chemistry (8464) from 2016 s T5.1 Atomic structure and the periodic table (Paper 1) State that everything is made of atoms and recall what they are 5.1.1 A simple
Personalised Learning Checklist AQA TRILOGY Chemistry (8464) from 2016 s T5.1 Atomic structure and the periodic table (Paper 1) State that everything is made of atoms and recall what they are 5.1.1 A simple
AQA Physics Checklist
 Topic 1. Energy Video: Energy changes in a system To understand the ways in which energy can be stored in a system and can be transferred from one energy store to another within a system To understand
Topic 1. Energy Video: Energy changes in a system To understand the ways in which energy can be stored in a system and can be transferred from one energy store to another within a system To understand
YEAR 7. St Edmund Arrowsmith CCFL Science Department Curriculum Map New AQA Course Started November 2016
 YEAR 7 St Edmund Arrowsmith CCFL Science Department Curriculum Map 201-2017 New AQA Course Started November 2016 05/09/2016 12/09/2016 19/09/2016 26/09/2016 03/10/2016 10/10/2016 17/10/2016 24/10/2016
YEAR 7 St Edmund Arrowsmith CCFL Science Department Curriculum Map 201-2017 New AQA Course Started November 2016 05/09/2016 12/09/2016 19/09/2016 26/09/2016 03/10/2016 10/10/2016 17/10/2016 24/10/2016
OCR Chemistry Checklist
 Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Level 789 Pathway: Combined Science Double Award
 Level 789 Pathway: Combined Science Double Award Yr Combined Science Targets: Pathway 8 11 Biology a) Explain in detail the role of the hormone ADH on the nephron b) Explain in detail the process by which
Level 789 Pathway: Combined Science Double Award Yr Combined Science Targets: Pathway 8 11 Biology a) Explain in detail the role of the hormone ADH on the nephron b) Explain in detail the process by which
GCSE PHYSICS REVISION LIST
 GCSE PHYSICS REVISION LIST OCR Gateway Physics (J249) from 2016 Topic P1: Matter P1.1 Describe how and why the atomic model has changed over time Describe the structure of the atom and discuss the charges
GCSE PHYSICS REVISION LIST OCR Gateway Physics (J249) from 2016 Topic P1: Matter P1.1 Describe how and why the atomic model has changed over time Describe the structure of the atom and discuss the charges
Describe how the inter-conversion of solids, liquids and gases are achieved and recall names used for these inter-conversions
 Understand the arrangements, movements and energy of the particle in each of the 3 states of matter : solid, liquid and gas Describe how the inter-conversion of solids, liquids and gases are achieved and
Understand the arrangements, movements and energy of the particle in each of the 3 states of matter : solid, liquid and gas Describe how the inter-conversion of solids, liquids and gases are achieved and
KS3 Science Levelness Posters
 KS Science Levelness Posters Contents Year Year 8 Year 9 A. Cells 8A. Food and digestion 9A. Inheritance and selection B. Reproduction 8B. Respiration 9B. Fit and healthy C. Environment and feeding 8C.
KS Science Levelness Posters Contents Year Year 8 Year 9 A. Cells 8A. Food and digestion 9A. Inheritance and selection B. Reproduction 8B. Respiration 9B. Fit and healthy C. Environment and feeding 8C.
Year 8 Tracking Document. Year 8 Science National Curriculum. Health and Lifestyle
 Health and Lifestyle Describe the components of a healthy diet Explain the role of each food group in the body Describe the test for starch, lipids, sugar and proteins Describe the positive test for each
Health and Lifestyle Describe the components of a healthy diet Explain the role of each food group in the body Describe the test for starch, lipids, sugar and proteins Describe the positive test for each
Topic Student Checklist R A G
 Personalised Learning Checklist AQA TRILOGY Physics (8464) from 2016 Topics T6.1. Energy Topic Student Checklist R A G 6.1.1 Energy changes in a system, and the ways energy is stored before and after such
Personalised Learning Checklist AQA TRILOGY Physics (8464) from 2016 Topics T6.1. Energy Topic Student Checklist R A G 6.1.1 Energy changes in a system, and the ways energy is stored before and after such
Paper Atomic structure and the periodic table
 Paper 1 4.1 Atomic structure and the periodic table 4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic charge and isotopes Use the names and symbols of the first 20 elements in
Paper 1 4.1 Atomic structure and the periodic table 4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic charge and isotopes Use the names and symbols of the first 20 elements in
Personalised Learning Checklists Edexcel Combined: Chemistry Paper 1
 Edexcel (combined) Chemistry Topics (1SC0) from 2016 - Paper 1 (Topic 1 parts a&b) Topic Student Checklist R A G Describe how the Dalton model of an atom has changed over time because of the discovery
Edexcel (combined) Chemistry Topics (1SC0) from 2016 - Paper 1 (Topic 1 parts a&b) Topic Student Checklist R A G Describe how the Dalton model of an atom has changed over time because of the discovery
Y8 Science Controlled Assessment Topics & Keywords
 Y8 Science Controlled Assessment Topics & Biology Respiration. Know that respiration in living organisms can be aerobic or anaerobic The word equation for aerobic respiration The process of anaerobic respiration
Y8 Science Controlled Assessment Topics & Biology Respiration. Know that respiration in living organisms can be aerobic or anaerobic The word equation for aerobic respiration The process of anaerobic respiration
Time (s) Velocity (m/s)
 29.01.18 22.01.18 15.01.18 w/c Weekly Combined Science revision countdown plan (revision guide / workbook page numbers) *content across both paper 1 and paper 2 15 14 13 Revise *Key concepts in biology
29.01.18 22.01.18 15.01.18 w/c Weekly Combined Science revision countdown plan (revision guide / workbook page numbers) *content across both paper 1 and paper 2 15 14 13 Revise *Key concepts in biology
OCR Chemistry Checklist
 Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Same theme covered in Combined but extra content Extra parts atomic symbols (first 20, Group 1 and Group 7)
 Co-teaching document new ELC Science 5960 and Foundation Level GCSE Combined Science: Trilogy (8464) Chemistry: Component 3 Elements, mixtures and compounds ELC Outcomes Summary of content covered in ELC
Co-teaching document new ELC Science 5960 and Foundation Level GCSE Combined Science: Trilogy (8464) Chemistry: Component 3 Elements, mixtures and compounds ELC Outcomes Summary of content covered in ELC
What is the resultant force acting on the object? BASE jumpers jump from very high buildings and mountains for sport.
 Forces Easier (a) The diagram shows two forces acting on an object. What is the resultant force acting on the object? Tick ( ) one box. 8 N to the right 8 N to the left 4 N to the right 4 N to the left
Forces Easier (a) The diagram shows two forces acting on an object. What is the resultant force acting on the object? Tick ( ) one box. 8 N to the right 8 N to the left 4 N to the right 4 N to the left
5 Energy from chemicals
 5 Energy from chemicals Content 5.1 Enthalpy 5.2 Hydrogen fuel cell Learning Outcomes Candidates should be able to: (a) (b) (c) (d) (e) describe the meaning of enthalpy change in terms of exothermic (H
5 Energy from chemicals Content 5.1 Enthalpy 5.2 Hydrogen fuel cell Learning Outcomes Candidates should be able to: (a) (b) (c) (d) (e) describe the meaning of enthalpy change in terms of exothermic (H
Reproduction Chemical Reactions. 8J Light 8G Metals & Their Uses 8C Breathing & Respiration 8D Unicellular Organisms
 Science: Key Stage 3 Based on the Exploring Science Scheme of Learning Term 1 & 2 Term 3 & 4 Term 5 & 6 Year 7 Cells, Tissues & Organs Particles Forces & Motion Reproduction Chemical Reactions Chemical
Science: Key Stage 3 Based on the Exploring Science Scheme of Learning Term 1 & 2 Term 3 & 4 Term 5 & 6 Year 7 Cells, Tissues & Organs Particles Forces & Motion Reproduction Chemical Reactions Chemical
Cambridge International Examinations Cambridge International General Certificate of Secondary Education. Published
 Cambridge International Examinations Cambridge International General Certificate of Secondary Education CO-ORDINATED SCIENCES 0654/4 Paper 4 Theory (Extended) May/June 017 MARK SCHEME Maximum Mark: 10
Cambridge International Examinations Cambridge International General Certificate of Secondary Education CO-ORDINATED SCIENCES 0654/4 Paper 4 Theory (Extended) May/June 017 MARK SCHEME Maximum Mark: 10
Unit 1: Cells, Tissues, Organs, and Systems
 Unit 1: Cells, Tissues, Organs, and Systems Big Ideas The cell is the basic scientific unit of all living things. Cells must interact with the external environment to meet their basic needs. Your health
Unit 1: Cells, Tissues, Organs, and Systems Big Ideas The cell is the basic scientific unit of all living things. Cells must interact with the external environment to meet their basic needs. Your health
Physics GCSE (9-1) Energy
 Topic Student Checklist R A G Define a system as an object or group of objects and State examples of changes in the way energy is stored in a system Describe how all the energy changes involved in an energy
Topic Student Checklist R A G Define a system as an object or group of objects and State examples of changes in the way energy is stored in a system Describe how all the energy changes involved in an energy
0654 CO-ORDINATED SCIENCES
 CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education MARK SCHEME for the May/June 2013 series 0654 CO-DINATED SCIENCES 0654/32 Paper 3 (Extended Theory), maximum
CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education MARK SCHEME for the May/June 2013 series 0654 CO-DINATED SCIENCES 0654/32 Paper 3 (Extended Theory), maximum
PAF Chapter Comprehensive Worksheet May 2016 Science Class 7 (Answering Key)
 The City School PAF Chapter Comprehensive Worksheet May 2016 Science Class 7 (Answering Key) The City School/ PAF Chapter/ Comprehensive Worksheet/ May 2016/ Science/ Class 7 /Ans Key Page 1 of 7 OBJECTIVE
The City School PAF Chapter Comprehensive Worksheet May 2016 Science Class 7 (Answering Key) The City School/ PAF Chapter/ Comprehensive Worksheet/ May 2016/ Science/ Class 7 /Ans Key Page 1 of 7 OBJECTIVE
Edexcel Chemistry Checklist
 Topic 1. Key concepts in chemistry Video: Atomic Structure Recall the different charges of the particles that make up an atom. Describe why atoms have no overall charge. Use the periodic table to identify
Topic 1. Key concepts in chemistry Video: Atomic Structure Recall the different charges of the particles that make up an atom. Describe why atoms have no overall charge. Use the periodic table to identify
Edexcel Chemistry Checklist
 Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
The City School PAF Chapter
 The City School PAF Chapter Comprehensive Worksheet May 2016 Science Class 7 Candidate Name: Index Number: Section: Branch/Campus: Date: INSTRUCTIONS: Write your name, index number, section, branch/campus
The City School PAF Chapter Comprehensive Worksheet May 2016 Science Class 7 Candidate Name: Index Number: Section: Branch/Campus: Date: INSTRUCTIONS: Write your name, index number, section, branch/campus
1.4 recall and use the relationship between acceleration, velocity and time: 1.6 determine acceleration from the gradient of a velocity-time graph
 Physics Section 1: Forces and motion b) Movement and position c) Forces, movement and shape d) Astronomy 1.1 use the following units: kilogram (kg), metre (m), metre/second (m/s), metre/second 2 (m/s 2
Physics Section 1: Forces and motion b) Movement and position c) Forces, movement and shape d) Astronomy 1.1 use the following units: kilogram (kg), metre (m), metre/second (m/s), metre/second 2 (m/s 2
LONGMAN STUDY GUIDES. Science. Di Barton. SUB Q6ttlngen B1842 LONGMAN
 LONGMAN STUDY GUIDES Science Di Barton SUB Q6ttlngen 206 451 652 97B1842 LONGMAN CONTENTS Editors' Preface vii Acknowledgements vii Information about this book viii 1 Revision, examinations and coursework
LONGMAN STUDY GUIDES Science Di Barton SUB Q6ttlngen 206 451 652 97B1842 LONGMAN CONTENTS Editors' Preface vii Acknowledgements vii Information about this book viii 1 Revision, examinations and coursework
Department Curriculum and Assessment Outline
 Department: Science Year Group: 10 Teaching, learning and assessment during the course: Combined Science 1 2 B1 Key concepts in Biology B2 Cells and control What are the structure and function of cells.
Department: Science Year Group: 10 Teaching, learning and assessment during the course: Combined Science 1 2 B1 Key concepts in Biology B2 Cells and control What are the structure and function of cells.
0654 CO-ORDINATED SCIENCES
 CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education MARK SCHEME for the May/June 2014 series 0654 CO-ORDINATED SCIENCES 0654/23 Paper 2 (Core Theory), maximum
CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education MARK SCHEME for the May/June 2014 series 0654 CO-ORDINATED SCIENCES 0654/23 Paper 2 (Core Theory), maximum
Unit C1: Chemistry in our world Page 1 of 5
 Unit C1: Chemistry in our world Page 1 of 5 Lesson Specification learning outcomes Edexcel 360 Science Specification match Edexcel 360 Science GCSE Science Students Book page reference Additional information
Unit C1: Chemistry in our world Page 1 of 5 Lesson Specification learning outcomes Edexcel 360 Science Specification match Edexcel 360 Science GCSE Science Students Book page reference Additional information
for sodium ion (Na + )
 3.4 Unit 2 Chemistry 2 Throughout this unit candidates will be expected to write word equations for reactions specified. Higher tier candidates will also be expected to write and balance symbol equations
3.4 Unit 2 Chemistry 2 Throughout this unit candidates will be expected to write word equations for reactions specified. Higher tier candidates will also be expected to write and balance symbol equations
Coimisiún na Scrúduithe Stáit State Examinations Commission
 2009. M 37 Coimisiún na Scrúduithe Stáit State Examinations Commission LEAVING CERTIFICATE EXAMINATION 2009 PHYSICS AND CHEMISTRY ORDINARY LEVEL MONDAY, 15 JUNE MORNING 9:30 TO 12:30 Six questions to be
2009. M 37 Coimisiún na Scrúduithe Stáit State Examinations Commission LEAVING CERTIFICATE EXAMINATION 2009 PHYSICS AND CHEMISTRY ORDINARY LEVEL MONDAY, 15 JUNE MORNING 9:30 TO 12:30 Six questions to be
Appendix 1. Periodic Table and Atomic Structure. History of the idea of elements.
 Appendix 1 Detailed list of additions and deletions This appendix provides a detailed list of additions and deletions compared with the former (1983) Leaving Certificate Chemistry syllabus. Completely
Appendix 1 Detailed list of additions and deletions This appendix provides a detailed list of additions and deletions compared with the former (1983) Leaving Certificate Chemistry syllabus. Completely
YEAR 10- Chemistry Term 1 plan
 YEAR 10- Chemistry Term 1 plan 2016-2017 Week Topic Learning outcomes 1 1. The particulate nature of matter State the distinguishing properties of solids, liquids and gases. Describe the structure of solids,
YEAR 10- Chemistry Term 1 plan 2016-2017 Week Topic Learning outcomes 1 1. The particulate nature of matter State the distinguishing properties of solids, liquids and gases. Describe the structure of solids,
An important fuel is methane, natural gas. The equation for its combustion is as follows. CO 2 + 2H 2 O
 1 (a Exothermic reactions produce heat energy. An important fuel is methane, natural gas. The equation for its combustion is as follows. CH 4 + 2O 2 CO 2 + 2H 2 O (i) In chemical reactions bonds are broken
1 (a Exothermic reactions produce heat energy. An important fuel is methane, natural gas. The equation for its combustion is as follows. CH 4 + 2O 2 CO 2 + 2H 2 O (i) In chemical reactions bonds are broken
Date: SCH 4U Name: ENTHALPY CHANGES
 Date: SCH 4U Name: ENTHALPY CHANGES Enthalpy (H) = heat content of system (heat, latent heat) Enthalpy = total energy of system + pressure volume H = E + PV H = E + (PV) = final conditions initial conditions
Date: SCH 4U Name: ENTHALPY CHANGES Enthalpy (H) = heat content of system (heat, latent heat) Enthalpy = total energy of system + pressure volume H = E + PV H = E + (PV) = final conditions initial conditions
Electrodes are normally made out of inert (unreactive) materials. Graphite and platinum are common electrode materials.
 Electrolysis Electrolysis is using an electric current to break up an ionic compound to form elements. Covalent compounds can t be split up by electrolysis. Terms used in electrolysis: Electrolyte - the
Electrolysis Electrolysis is using an electric current to break up an ionic compound to form elements. Covalent compounds can t be split up by electrolysis. Terms used in electrolysis: Electrolyte - the
0653 COMBINED SCIENCE
 CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International General Certificate of Secondary Education MARK SCHEME for the October/November 2014 series 0653 COMBINED SCIENCE 0653/31 Paper 3 (Extended
CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International General Certificate of Secondary Education MARK SCHEME for the October/November 2014 series 0653 COMBINED SCIENCE 0653/31 Paper 3 (Extended
Summary of changes (certificate to new GCSE)
 Summary of changes (certificate to new GCSE) This resource outlines the main changes that have been made to the assessment and subject content from our legacy Level 1/2 Certificate in Biology (8401) to
Summary of changes (certificate to new GCSE) This resource outlines the main changes that have been made to the assessment and subject content from our legacy Level 1/2 Certificate in Biology (8401) to
Lesson title Lesson objectives AQA specification reference 1.1 Elements and compounds
 1.1 Elements and compounds 1.2 Atoms, formulae and equations Identify symbols of elements from the periodic table Recognise the properties of elements and compounds. Identify the elements in a compound
1.1 Elements and compounds 1.2 Atoms, formulae and equations Identify symbols of elements from the periodic table Recognise the properties of elements and compounds. Identify the elements in a compound
Foundation Support Workbook AQA GCSE Combined Science Chemistry topics. Sunetra Berry
 Foundation Workbook AQA GCSE Combined Science Chemistry topics Sunetra Berry 224708 Foundation Workbook_Sample_Chemistry.indd 1 4/22/16 4:17 PM Contents Section 1 Atomic structure and the periodic table
Foundation Workbook AQA GCSE Combined Science Chemistry topics Sunetra Berry 224708 Foundation Workbook_Sample_Chemistry.indd 1 4/22/16 4:17 PM Contents Section 1 Atomic structure and the periodic table
4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic charge and isotopes. Unit 1 Unit 2 Unit 3. C2.1.1a Structure and bonding
 Summary of changes This resource outlines the main changes that have been made to the assessment and subject content from our previous GCSE Chemistry (4402) to the new specification (8462). Our new specifications
Summary of changes This resource outlines the main changes that have been made to the assessment and subject content from our previous GCSE Chemistry (4402) to the new specification (8462). Our new specifications
100 Physics Facts. 1. The standard international unit (SI unit) for mass (m) is. kg (kilograms) s (seconds)
 100 Physics Facts 1. The standard international unit (SI unit) for mass (m) is. kg (kilograms) 2. The standard international unit (SI unit) for time (t) is. s (seconds) 3. The standard international unit
100 Physics Facts 1. The standard international unit (SI unit) for mass (m) is. kg (kilograms) 2. The standard international unit (SI unit) for time (t) is. s (seconds) 3. The standard international unit
State the position of protons, neutrons and electrons in the atom
 2.1 The Atom 2.1.1 - State the position of protons, neutrons and electrons in the atom Atoms are made up of a nucleus containing positively charged protons and neutral neutrons, with negatively charged
2.1 The Atom 2.1.1 - State the position of protons, neutrons and electrons in the atom Atoms are made up of a nucleus containing positively charged protons and neutral neutrons, with negatively charged
Combined Science: Trilogy
 Co-teaching GCSE Chemistry and GCSE Combined Science: Trilogy This high level co-teaching guide will help you plan your route through the course. You ll be able to see what common themes and topics span
Co-teaching GCSE Chemistry and GCSE Combined Science: Trilogy This high level co-teaching guide will help you plan your route through the course. You ll be able to see what common themes and topics span
Section A Multiple Choice
 Section Multiple hoice 1. Two atoms have the same atomic number but different mass numbers. The atoms are two different elements have different numbers of electrons have different numbers of protons are
Section Multiple hoice 1. Two atoms have the same atomic number but different mass numbers. The atoms are two different elements have different numbers of electrons have different numbers of protons are
Electrolysis. Specification points. Year 11 Electrolysis
 Electrolysis Specification points Year Electrolysis The process of electrolysis When an ionic compound is melted or dissolved in water, the ions are free to move about within the liquid or solution (the
Electrolysis Specification points Year Electrolysis The process of electrolysis When an ionic compound is melted or dissolved in water, the ions are free to move about within the liquid or solution (the
YEAR 9 - AQA GCSE COMBINED SCIENCE (9-1) Cell Biology
 YEAR 9 - AQA GCSE COMBINED SCIENCE (9-1) Cell Biology Cell structure Eukaryotes and prokaryotes Plant and animal cells (eukaryotic cells) have a cell membrane, cytoplasm and genetic material enclosed in
YEAR 9 - AQA GCSE COMBINED SCIENCE (9-1) Cell Biology Cell structure Eukaryotes and prokaryotes Plant and animal cells (eukaryotic cells) have a cell membrane, cytoplasm and genetic material enclosed in
GCE O' LEVEL PURE CHEMISTRY (5073/02) Suggested Answers for 2016 O Level Pure Chemistry Paper 2
 Section A (50 M) Aa) trend The number of electron shell increases The number of valence electrons increases Proton number increases There is a change in character from metallic to non-metallic Only true
Section A (50 M) Aa) trend The number of electron shell increases The number of valence electrons increases Proton number increases There is a change in character from metallic to non-metallic Only true
Combined Science Chemistry Academic Overview
 Combined Science Chemistry Academic Overview 2018-2019 Science Term 1.1 Term 1.2 Term 2.1 Term 2.2 Term 3.1 Term 3.2 Year 9 States of Matter Methods of Separating and Purifying Substances Atomic Structure
Combined Science Chemistry Academic Overview 2018-2019 Science Term 1.1 Term 1.2 Term 2.1 Term 2.2 Term 3.1 Term 3.2 Year 9 States of Matter Methods of Separating and Purifying Substances Atomic Structure
UNIT 9A Inheritance and Selection
 UNIT 9A Inheritance and Selection Know what inheritance means Know what characteristics are Know that sexual reproduction needs two parents Know that animals and plants can be bred for their characteristics
UNIT 9A Inheritance and Selection Know what inheritance means Know what characteristics are Know that sexual reproduction needs two parents Know that animals and plants can be bred for their characteristics
Matter mass space atoms solid, a liquid, a gas, or plasm elements compounds mixtures atoms Compounds chemically combined Mixtures not chemically
 SOL PS.2 THE NATURE OF MATTER Matter is anything that has mass and occupies space. All matter is made up of small particles called atoms. Matter can exist as a solid, a liquid, a gas, or plasma. Matter
SOL PS.2 THE NATURE OF MATTER Matter is anything that has mass and occupies space. All matter is made up of small particles called atoms. Matter can exist as a solid, a liquid, a gas, or plasma. Matter
Trilogy - Science Revision
 Trilogy - Science Revision Sept 2015- June 2018 Year 11 Subject Unit Date AM/PM Name: Biology Paper 1 Biology Paper 2 Chemistry Paper 1 Chemistry Paper 2 Physics Paper 1 Physics Paper 2 Target: Tuesday
Trilogy - Science Revision Sept 2015- June 2018 Year 11 Subject Unit Date AM/PM Name: Biology Paper 1 Biology Paper 2 Chemistry Paper 1 Chemistry Paper 2 Physics Paper 1 Physics Paper 2 Target: Tuesday
Q1. Fresh milk is a mixture of compounds including lipid, protein and about 5% lactose sugar.
 Q1. Fresh milk is a mixture of compounds including lipid, protein and about 5% lactose sugar. Lactose must be digested by the enzyme lactase, before the products can be absorbed. Lactase can be added to
Q1. Fresh milk is a mixture of compounds including lipid, protein and about 5% lactose sugar. Lactose must be digested by the enzyme lactase, before the products can be absorbed. Lactase can be added to
- know that in multi-cellular organisms cells are massed together to form tissues, and tissues can be massed together to form organs
 Science Age 12-13 (A) BIOLOGY: ORGANISMS, THEIR BEHAVIOUR AND THE ENVIRONMENT 1. Cells and their functions - know that in multi-cellular organisms cells are massed together to form tissues, and tissues
Science Age 12-13 (A) BIOLOGY: ORGANISMS, THEIR BEHAVIOUR AND THE ENVIRONMENT 1. Cells and their functions - know that in multi-cellular organisms cells are massed together to form tissues, and tissues
BTEC L3 Diploma in Applied Science. Pre-Course Knowledge Assessment Task
 Name: BTEC L3 Diploma in Applied Science Pre-Course Knowledge Assessment Task Aim: For you to self-assess your current science knowledge and address any areas of weakness. Objective: To complete the self-assessment
Name: BTEC L3 Diploma in Applied Science Pre-Course Knowledge Assessment Task Aim: For you to self-assess your current science knowledge and address any areas of weakness. Objective: To complete the self-assessment
0654 CO-ORDINATED SCIENCES
 CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International General Certificate of Secondary Education MARK SCHEME for the May/June 2015 series 0654 CO-ORDINATED SCIENCES 0654/31 Paper 3 (Extended Theory),
CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International General Certificate of Secondary Education MARK SCHEME for the May/June 2015 series 0654 CO-ORDINATED SCIENCES 0654/31 Paper 3 (Extended Theory),
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 BIO130 Pre-Requisite Chemistry and Metric System Take Home Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. Transfer your final answer to
BIO130 Pre-Requisite Chemistry and Metric System Take Home Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. Transfer your final answer to
Paget High School. Preparing for A level Biology
 Paget High School Preparing for A level Biology You will need a copy of the Head Start guide above and use it to make notes on the learning outcomes below. Topic areas Learning outcome Describe with the
Paget High School Preparing for A level Biology You will need a copy of the Head Start guide above and use it to make notes on the learning outcomes below. Topic areas Learning outcome Describe with the
The response made by part of a plant, by which it grow towards
 Word/phrase Biology Nutrition Excretion Sensitivity Growth Reproduction Movement Digestion Enzyme Partially permeable Membrane Egestion Ingestion Photosynthesis Absorbtion Transpiration Translocation Respiration
Word/phrase Biology Nutrition Excretion Sensitivity Growth Reproduction Movement Digestion Enzyme Partially permeable Membrane Egestion Ingestion Photosynthesis Absorbtion Transpiration Translocation Respiration
HASTINGS HIGH SCHOOL
 Subject HASTINGS HIGH SCHOOL YEAR 11 EXAMINATION GUIDE 20167-19 COMBINED SCIENCE TRILOGY Physics Course code AQA GCSE COMBINED SCIENCE TRILOGY 8464 Website address Provisional examination dates http://www.aqa.org.uk/subjects/science/gcse/combined-science-trilogy-
Subject HASTINGS HIGH SCHOOL YEAR 11 EXAMINATION GUIDE 20167-19 COMBINED SCIENCE TRILOGY Physics Course code AQA GCSE COMBINED SCIENCE TRILOGY 8464 Website address Provisional examination dates http://www.aqa.org.uk/subjects/science/gcse/combined-science-trilogy-
Science. Biology Year 7. Chemistry Year 7. Physics Year 7
 Science Biology Year 7 Chemistry Year 7 Physics Year 7 Using a microscope Slide Preparation o Prepare specimens of plant cells and animal cells o Describe the role of stain in specimen preparation o Draw
Science Biology Year 7 Chemistry Year 7 Physics Year 7 Using a microscope Slide Preparation o Prepare specimens of plant cells and animal cells o Describe the role of stain in specimen preparation o Draw
Unit 4: Chemical Changes (Higher Content)
 Metals react with oxygen to produce metal oxides. E.g. Copper + Oxygen > Copper Oxide The reactions are oxidation reactions because the metals gain oxygen. Reactivity of Metals Metal Extraction Metals
Metals react with oxygen to produce metal oxides. E.g. Copper + Oxygen > Copper Oxide The reactions are oxidation reactions because the metals gain oxygen. Reactivity of Metals Metal Extraction Metals
Complete and balance these equations to show the reactions during electrolysis. Na Na (2)
 Q1. The diagram shows electrolysis of sodium chloride solution. (a) Complete and balance these equations to show the reactions during electrolysis. At the positive electrode Cl e Cl At the negative electrode
Q1. The diagram shows electrolysis of sodium chloride solution. (a) Complete and balance these equations to show the reactions during electrolysis. At the positive electrode Cl e Cl At the negative electrode
Foundation Year Programme
 Foundation Year Programme Entrance Tests PHYSICS SPECIFICATION Standard ATS sample material 2 3 Physics 1. Electricity 1.1 Electrostatics: a. charging of insulators by friction b. object gaining electrons
Foundation Year Programme Entrance Tests PHYSICS SPECIFICATION Standard ATS sample material 2 3 Physics 1. Electricity 1.1 Electrostatics: a. charging of insulators by friction b. object gaining electrons
The electrolysis of sodium chloride solution produces useful substances. covalent ionic non-metallic
 1 The electrolysis of sodium chloride solution produces useful substances. (a) (i) Choose a word from the box to complete the sentence. covalent ionic non-metallic Electrolysis takes place when electricity
1 The electrolysis of sodium chloride solution produces useful substances. (a) (i) Choose a word from the box to complete the sentence. covalent ionic non-metallic Electrolysis takes place when electricity
8. atomic mass: the mass of the element and represents the. 9. Atomic number: on top of the atomic symbol; represents the.
 Intro To Chemistry Study online at quizlet.com/_2xw51k 1....:... 2. alkali metals: 8. atomic mass: the mass of the element and represents the number of protons plus the number of neutrons. 9. Atomic number:
Intro To Chemistry Study online at quizlet.com/_2xw51k 1....:... 2. alkali metals: 8. atomic mass: the mass of the element and represents the number of protons plus the number of neutrons. 9. Atomic number:
Optics Definitions. The apparent movement of one object relative to another due to the motion of the observer is called parallax.
 Optics Definitions Reflection is the bouncing of light off an object Laws of Reflection of Light: 1. The incident ray, the normal at the point of incidence and the reflected ray all lie in the same plane.
Optics Definitions Reflection is the bouncing of light off an object Laws of Reflection of Light: 1. The incident ray, the normal at the point of incidence and the reflected ray all lie in the same plane.
Lesson Target 4 Target 6 Target 8. atom.
 Student checklist C1 Atomic structure Lesson Target 4 Target 6 Target 8 C1.1 Atoms I can define the word element. I can classify familiar substances as elements or compounds. I can use the periodic table
Student checklist C1 Atomic structure Lesson Target 4 Target 6 Target 8 C1.1 Atoms I can define the word element. I can classify familiar substances as elements or compounds. I can use the periodic table
What happens during nuclear decay? During nuclear decay, atoms of one element can change into atoms of a different element altogether.
 When Henri Becquerel placed uranium salts on a photographic plate and then developed the plate, he found a foggy image. The image was caused by rays that had not been observed before. For his discovery
When Henri Becquerel placed uranium salts on a photographic plate and then developed the plate, he found a foggy image. The image was caused by rays that had not been observed before. For his discovery
S4 CHEMISTRY SUMMARY NOTES
 S4 CHEMISTRY SUMMARY NOTES 1. The Mole One mole of a substance = GRAM FORMULA MASS e.g. H 2 SO 4 RAM from databook pg.7 2H 2 x 1 = 2 1S 1 x 32 = 32 4O 4 x 16 = 64 98g Mass = number of moles x Mass of 1
S4 CHEMISTRY SUMMARY NOTES 1. The Mole One mole of a substance = GRAM FORMULA MASS e.g. H 2 SO 4 RAM from databook pg.7 2H 2 x 1 = 2 1S 1 x 32 = 32 4O 4 x 16 = 64 98g Mass = number of moles x Mass of 1
