Supplementary Figure 1. Calculated Ca-oxalate cluster concentration. Red line show the
|
|
|
- Frank Walsh
- 6 years ago
- Views:
Transcription
1 Supplementary Figure 1. Calculated Ca-oxalate cluster concentration. Red line show the calcium oxalate cluster concentration calculated from ISE measurements and the blue line that calculated using conductivity measurements and assuming a oxalate to calcium ratio of 2 (n) in the clusters. Error bars s.d.
2 Supplementary Figure 2. Calcium complexation by citrate. Time development of the free Ca 2+ concentration in titration experiments in aqueous solutions (no oxalate in the reaction media) in the presence of different concentrations of citrate at ph 6.2. The black line refers to the amount of Ca 2+ added.
3 Supplementary Figure 3. Stability of calcium oxalate clusters as a function of citrate concentration. Gibbs standard energy for the formation of calcium/oxalate ion pairs in calcium oxalate clusters, as a function of citrate concentration.
4 Supplementary Figure 4. Amorphous calcium oxalate in control runs. TEM photomicrograph of calcium oxalate nanoparticles formed after 1600 s in the titration experiments (control runs). SAED pattern showing their amorphous nature in inset. Scale bar: 200 nm. Scale bar in the inset: 2 nm -1
5 Supplementary Figure 5. X-Ray diffraction patterns of solids collected at the end of titration experiments. Black line: control run (i.e. no citrate in the reaction media). Red line: experiment performed in the presence of 10 mm citrate.
6 Supplementary Figure 6. STEM line profile analysis of ACO nanoparticles. TEM annular dark field images of the edge of an ACO nanoparticle displaying a ca. 20 nm thick rim and lineprofile EDS analysis (along the line A-A depicted in the TEM image) showing an increase in C in the outer part of the sphere is likely due to the presence of an adsorbed/incorporated layer of citrate.
7 Supplementary Figure 7. Schematic diagram showing the ionic concentration around the negatively charged calcium oxalate clusters. Note that this is just a sketch to illustrate our model explaining the slightly positive zeta potential measure in the clusters in the absence of citrate, as well as citrate binding, as PNCs actually do not have surfaces that separate them from the surrounding solution.
8 Supplementary Figure 8. Analytical Ultracentrifugation analysis (I). Sedimentation coefficients measured for samples drawn from citrate buffer ph 6.4 (1,2), control runs (no citrate in the reaction media) with samples taken after 500 s (3,4), titration runs performed in the presence of 10 mm citrate taken after 6000 s (5, 6) and s (7-9). Two different species were found: the black dots are the smallest species and the red ones are the next bigger species that are found in the presence of citrate.
9 Supplementary Figure 9. Analytical Ultracentrifugation analysis (II). Apparent hydrodynamic diameters calculated from the sedimentation coefficients in figure S5 using v = ml/g for citrate and v = ml/g for CaC 2 O 4.
10 Supplementary Figure 10. Sketch of the citrate molecule. The longest distance (0.74 nm) is marked by the dashed green line.
11 Supplementary Figure 11. Thermogravimetric analysis of calcium oxalate precipitates. TG traces of (a, b) calcium oxalate crystals precipitated (a) in the absence and (b) in the presence of 10 mm citrate. The different steps in the thermal decomposition correspond to: (1) the first weight loss at temperatures ranging C corresponds to the loss of water of
12 crystallization, (2) the second weight loss at temperatures ranging C, corresponds to the decomposition of anhydrous calcium oxalate, with the loss of carbon monoxide, (3) the third and final weight loss, at temperatures ranging C, corresponds to the decomposition of calcium carbonate to calcium oxide with the loss of carbon dioxide. (c) TG trace of sodium citrate, showing partial overlapping with some of the decomposition steps of calcium oxalate precipitates.
a b c Supplementary Figure S1
 a b c Supplementary Figure S1 AFM measurements of MoS 2 nanosheets prepared from the electrochemical Liintercalation and exfoliation. (a) AFM measurement of a typical MoS 2 nanosheet, deposited on Si/SiO
a b c Supplementary Figure S1 AFM measurements of MoS 2 nanosheets prepared from the electrochemical Liintercalation and exfoliation. (a) AFM measurement of a typical MoS 2 nanosheet, deposited on Si/SiO
Osteopontin Stabilizes Metastable States Prior to Nucleation during Apatite Formation
 Supplemental Osteopontin Stabilizes Metastable States Prior to Nucleation during Apatite Formation Casper Jon Steenberg Ibsen1, Denis Gebauer2, Henrik Birkedal1 * 1 inano and Department of Chemistry, Aarhus
Supplemental Osteopontin Stabilizes Metastable States Prior to Nucleation during Apatite Formation Casper Jon Steenberg Ibsen1, Denis Gebauer2, Henrik Birkedal1 * 1 inano and Department of Chemistry, Aarhus
It is the purpose of this paper to demonstrate that the detection limit is directly related to the sample size, for both TG and TG/FTIR.
 BIGGER IS BETTER: PUSHING THE LIMIT OF TG AND TG/FTIR Abstract In Thermogravimetry (TG) and TG/FTIR systems, there are many variables that can affect the detection limit of the system. It is demonstrated
BIGGER IS BETTER: PUSHING THE LIMIT OF TG AND TG/FTIR Abstract In Thermogravimetry (TG) and TG/FTIR systems, there are many variables that can affect the detection limit of the system. It is demonstrated
Hydrogen Titanium Oxide Hydrate: Excellent Performance. on Degradation of Methyl Blue in Aqueous Solutions
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supplementary information Hydrogen Titanium Oxide Hydrate: Excellent Performance on Degradation
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supplementary information Hydrogen Titanium Oxide Hydrate: Excellent Performance on Degradation
Introduction to Chemical Bonding Chemical Bond
 Introduction to Chemical Bonding Chemical Bond Mutual attraction between the and electrons of different atoms that binds the atoms together. Ionic Bond o that results from the attraction between large
Introduction to Chemical Bonding Chemical Bond Mutual attraction between the and electrons of different atoms that binds the atoms together. Ionic Bond o that results from the attraction between large
Supporting Information
 Supporting Information Visualizing the Effect of Partial Oxide Formation on Single Silver Nanoparticle Electrodissolution Vignesh Sundaresan, Joseph W. Monaghan, and Katherine A. Willets* Department of
Supporting Information Visualizing the Effect of Partial Oxide Formation on Single Silver Nanoparticle Electrodissolution Vignesh Sundaresan, Joseph W. Monaghan, and Katherine A. Willets* Department of
Supplementary Figure 1. A photographic image of directionally grown perovskite films on a glass substrate (size: cm).
 Supplementary Figure 1. A photographic image of directionally grown perovskite films on a glass substrate (size: 1.5 4.5 cm). 1 Supplementary Figure 2. Optical microscope images of MAPbI 3 films formed
Supplementary Figure 1. A photographic image of directionally grown perovskite films on a glass substrate (size: 1.5 4.5 cm). 1 Supplementary Figure 2. Optical microscope images of MAPbI 3 films formed
Construction of nanoantennas on the outer bacterial membrane
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Construction of nanoantennas on the outer bacterial membrane
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Construction of nanoantennas on the outer bacterial membrane
UNIT 3 IB MATERIAL BONDING, MOLES & STOICHIOMETRY
 UNIT 3 IB MATERIAL Name: BONDING, MOLES & STOICHIOMETRY ESSENTIALS: Know, Understand, and Be Able To Apply the mole concept to substances. Determine the number of particles and the amount of substance
UNIT 3 IB MATERIAL Name: BONDING, MOLES & STOICHIOMETRY ESSENTIALS: Know, Understand, and Be Able To Apply the mole concept to substances. Determine the number of particles and the amount of substance
Supplementary Figure S1: Particle size distributions of the Pt ML /Pd 9 Au 1 /C
 a 2 15 before cycle test mean particle size: 3.8 ± 1.2 nm b 2 15 after.6v - 1.V 1k cycle test mean particle size: 4.1 ± 1.5 nm Number 1 total number: 558 Number 1 total number: 554 5 5 1 2 3 4 5 6 7 8
a 2 15 before cycle test mean particle size: 3.8 ± 1.2 nm b 2 15 after.6v - 1.V 1k cycle test mean particle size: 4.1 ± 1.5 nm Number 1 total number: 558 Number 1 total number: 554 5 5 1 2 3 4 5 6 7 8
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Photochemical Reduction of Carbon
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Photochemical Reduction of Carbon
size (nm) Supplementary Figure 1. Hydrodynamic size distribution in a H 2O:MeOH 50% v/v
 Intensity (%) 20 15 10 [0a] [0b] CoTPMA: 3 eq CoTPMA: 10 eq CoTPMA: 20 eq CoTPMA : 40 eq CoTPMA: 60 eq, [1] CoTPMA: 85 eq CoTPMA: 125 eq CoTPMA: 170 eq CoTPMA: 210 eq 5 0 10 100 size (nm) Supplementary
Intensity (%) 20 15 10 [0a] [0b] CoTPMA: 3 eq CoTPMA: 10 eq CoTPMA: 20 eq CoTPMA : 40 eq CoTPMA: 60 eq, [1] CoTPMA: 85 eq CoTPMA: 125 eq CoTPMA: 170 eq CoTPMA: 210 eq 5 0 10 100 size (nm) Supplementary
Supporting Information
 Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2015. Supporting Information for Adv. Mater., DOI: 10.1002/adma.201502134 Stable Metallic 1T-WS 2 Nanoribbons Intercalated with Ammonia
Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2015. Supporting Information for Adv. Mater., DOI: 10.1002/adma.201502134 Stable Metallic 1T-WS 2 Nanoribbons Intercalated with Ammonia
Supporting Information: Time- and Size-Resolved Plasmonic Evolution with nm Resolution of Galvanic Replacement Reaction in AuAg Nanoshells Synthesis
 Supporting Information: Time- and Size-Resolved Plasmonic Evolution with nm Resolution of Galvanic Replacement Reaction in AuAg Nanoshells Synthesis Lorenzo Russo, Florind Merkoçi, Javier Patarroyo, Jordi
Supporting Information: Time- and Size-Resolved Plasmonic Evolution with nm Resolution of Galvanic Replacement Reaction in AuAg Nanoshells Synthesis Lorenzo Russo, Florind Merkoçi, Javier Patarroyo, Jordi
Synthesis and Bioconjugation of 2 and 3 nm-diameter Gold Cluster Compounds
 Supplementary Information Synthesis and Bioconjugation of 2 and 3 nm-diameter Gold Cluster Compounds Christopher J. Ackerson, Pablo D. Jadzinsky, Jonathan Z. Sexton and Roger D. Kornberg Department of
Supplementary Information Synthesis and Bioconjugation of 2 and 3 nm-diameter Gold Cluster Compounds Christopher J. Ackerson, Pablo D. Jadzinsky, Jonathan Z. Sexton and Roger D. Kornberg Department of
SIR MICHELANGELO REFALO
 SIR MICELANGELO REFALO SIXT FORM alf-yearly Exam 2014 Name: CEMISTRY ADV 1 ST 3 hrs ANSWER ANY 7 QUESTIONS. All questions carry equal marks. You are reminded of the importance of clear presentation in
SIR MICELANGELO REFALO SIXT FORM alf-yearly Exam 2014 Name: CEMISTRY ADV 1 ST 3 hrs ANSWER ANY 7 QUESTIONS. All questions carry equal marks. You are reminded of the importance of clear presentation in
Supporting information. Stability Issues in Pd-based Catalysts: The Role of Surface Pt in Improving the Stability
 Supporting information Stability Issues in Pd-based Catalysts: The Role of Surface Pt in Improving the Stability and Oxygen Reduction Reaction (ORR) Activity R. K. Singh, R. Rahul, M. Neergat 1 Department
Supporting information Stability Issues in Pd-based Catalysts: The Role of Surface Pt in Improving the Stability and Oxygen Reduction Reaction (ORR) Activity R. K. Singh, R. Rahul, M. Neergat 1 Department
Chemical Bonds, Lewis Structures, Bond Order, and Formal Charge
 Chemical Bonds, Lewis Structures, Bond Order, and Formal Charge PRELAB ASSIGNMENT Read the entire laboratory write up. Write an objective, any hazards associated with this lab, and answer the following
Chemical Bonds, Lewis Structures, Bond Order, and Formal Charge PRELAB ASSIGNMENT Read the entire laboratory write up. Write an objective, any hazards associated with this lab, and answer the following
There are two main requirements for atoms to form a covalent bond and make a molecule:
 HOW ATOMS BOND TO EACH OTHER Covalent bonding Remember that a hydrogen atom has 1 proton and 1 electron and that the electron and the proton are attracted to each other. But if the atoms get close enough
HOW ATOMS BOND TO EACH OTHER Covalent bonding Remember that a hydrogen atom has 1 proton and 1 electron and that the electron and the proton are attracted to each other. But if the atoms get close enough
4 CO O 2. , how many moles of KCl will be produced? Use the unbalanced equation below: PbCl 2. PbSO 4
 Honors Chemistry Practice Final 2017 KEY 1. Acetylene gas, C 2, is used in welding because it generates an extremely hot flame when combusted with oxygen. How many moles of oxygen are required to react
Honors Chemistry Practice Final 2017 KEY 1. Acetylene gas, C 2, is used in welding because it generates an extremely hot flame when combusted with oxygen. How many moles of oxygen are required to react
THE BIG IDEA: ELECTRONS AND THE STRUCTURE OF ATOMS. BONDING AND INTERACTIONS.
 HONORS CHEMISTRY - CHAPTER 9 CHEMICAL NAMES AND FORMULAS OBJECTIVES AND NOTES - V18 NAME: DATE: PAGE: THE BIG IDEA: ELECTRONS AND THE STRUCTURE OF ATOMS. BONDING AND INTERACTIONS. Essential Questions 1.
HONORS CHEMISTRY - CHAPTER 9 CHEMICAL NAMES AND FORMULAS OBJECTIVES AND NOTES - V18 NAME: DATE: PAGE: THE BIG IDEA: ELECTRONS AND THE STRUCTURE OF ATOMS. BONDING AND INTERACTIONS. Essential Questions 1.
Supplementary Information
 Supplementary Information Time-dependent growth of zinc hydroxide nanostrands and their crystal structure Xinsheng Peng, ab Jian Jin, a Noriko Kobayashi, a Wolfgang Schmitt, c and Izumi Ichinose* a a Organic
Supplementary Information Time-dependent growth of zinc hydroxide nanostrands and their crystal structure Xinsheng Peng, ab Jian Jin, a Noriko Kobayashi, a Wolfgang Schmitt, c and Izumi Ichinose* a a Organic
Chapter 1 Section 1- Pages 4-7: Electrons and Chemical Bonding COMBINING ATOMS THROUGH CHEMICAL BONDING
 Study Guide Chapter 1 and 2 Interactions of Matter Chapter 1 Section 1- Pages 4-7: Electrons and Chemical Bonding COMBINING ATOMS THROUGH CHEMICAL BONDING 1. Which of these substances is a combination
Study Guide Chapter 1 and 2 Interactions of Matter Chapter 1 Section 1- Pages 4-7: Electrons and Chemical Bonding COMBINING ATOMS THROUGH CHEMICAL BONDING 1. Which of these substances is a combination
ELECTRONIC SUPPLEMENTARY INFORMATION (ESI)
 Electronic Supplementary Material (ESI) for Environmental Science: Nano. This journal is The Royal Society of Chemistry 216 ELECTRNIC SUPPLEMENTARY INFRMATIN (ESI) Spectral Characterization and Surface
Electronic Supplementary Material (ESI) for Environmental Science: Nano. This journal is The Royal Society of Chemistry 216 ELECTRNIC SUPPLEMENTARY INFRMATIN (ESI) Spectral Characterization and Surface
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supporting Information An Anode Buffer Layer with Size-Controlled Ag Nanoparticles for Polymer
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supporting Information An Anode Buffer Layer with Size-Controlled Ag Nanoparticles for Polymer
Nucleation rate (m -3 s -1 ) Radius of water nano droplet (Å) 1e+00 1e-64 1e-128 1e-192 1e-256
 Supplementary Figures Nucleation rate (m -3 s -1 ) 1e+00 1e-64 1e-128 1e-192 1e-256 Calculated R in bulk water Calculated R in droplet Modified CNT 20 30 40 50 60 70 Radius of water nano droplet (Å) Supplementary
Supplementary Figures Nucleation rate (m -3 s -1 ) 1e+00 1e-64 1e-128 1e-192 1e-256 Calculated R in bulk water Calculated R in droplet Modified CNT 20 30 40 50 60 70 Radius of water nano droplet (Å) Supplementary
Chemistry Summer Holiday Homework Year Y9 & 10
 Chemistry Summer Holiday Homework Year Y9 & 10 1. An atom of aluminium has the symbol (a) Give the number of protons, neutrons and electrons in this atom of aluminium. Number of protons... Number of neutrons...
Chemistry Summer Holiday Homework Year Y9 & 10 1. An atom of aluminium has the symbol (a) Give the number of protons, neutrons and electrons in this atom of aluminium. Number of protons... Number of neutrons...
CHEMISTRY 2815/01 Trends and Patterns
 THIS IS A LEGACY SPECIFICATION ADVANCED GCE CHEMISTRY 2815/01 Trends and Patterns *CUP/T76203* Candidates answer on the question paper A calculator may be used for this paper OCR Supplied Materials: Data
THIS IS A LEGACY SPECIFICATION ADVANCED GCE CHEMISTRY 2815/01 Trends and Patterns *CUP/T76203* Candidates answer on the question paper A calculator may be used for this paper OCR Supplied Materials: Data
Supplementary Materials: Localization and Spectroscopic Analysis of the Cu(I) Binding Site in Wheat Metallothionein Ec-1
 S1 of S8 Supplementary Materials: Localization and Spectroscopic Analysis of the Cu(I) Binding Site in Wheat Metallothionein Ec-1 Katsiaryna Tarasava, Jens Loebus and Eva Freisinger Figure S1. Deconvoluted
S1 of S8 Supplementary Materials: Localization and Spectroscopic Analysis of the Cu(I) Binding Site in Wheat Metallothionein Ec-1 Katsiaryna Tarasava, Jens Loebus and Eva Freisinger Figure S1. Deconvoluted
Chiral Nematic Cellulose / Gold Nanoparticle Composites from Mesoporous Photonic Cellulose
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Chiral Nematic Cellulose / Gold Nanoparticle Composites from Mesoporous Photonic Cellulose Maik
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Chiral Nematic Cellulose / Gold Nanoparticle Composites from Mesoporous Photonic Cellulose Maik
Supporting Information
 Supporting Information Study of molecular conformation and activity-related properties of lipase immobilized onto core-shell structured polyacrylic acid-coated magnetic silica nanocomposite particles Parvaneh
Supporting Information Study of molecular conformation and activity-related properties of lipase immobilized onto core-shell structured polyacrylic acid-coated magnetic silica nanocomposite particles Parvaneh
Review Topic 8: Phases of Matter and Mixtures
 Name: Score: 24 / 24 points (100%) Review Topic 8: Phases of Matter and Mixtures Multiple Choice Identify the choice that best completes the statement or answers the question. C 1. Soda water is a solution
Name: Score: 24 / 24 points (100%) Review Topic 8: Phases of Matter and Mixtures Multiple Choice Identify the choice that best completes the statement or answers the question. C 1. Soda water is a solution
Valence bond theory accounts, at least qualitatively, for the stability of the covalent bond in terms of overlapping atomic orbitals.
 Molecular Orbital Theory Valence bond theory accounts, at least qualitatively, for the stability of the covalent bond in terms of overlapping atomic orbitals. Using the concept of hybridization, valence
Molecular Orbital Theory Valence bond theory accounts, at least qualitatively, for the stability of the covalent bond in terms of overlapping atomic orbitals. Using the concept of hybridization, valence
Small-Angle X-ray Scattering (SAXS)/X-ray Absorption Near Edge Spectroscopy (XANES).
 S1 Small-Angle X-ray Scattering (SAXS)/X-ray Absorption Near Edge Spectroscopy (XANES). The combined SAXS/XANES measurements were carried out at the µspot beamline at BESSY II (Berlin, Germany). The beamline
S1 Small-Angle X-ray Scattering (SAXS)/X-ray Absorption Near Edge Spectroscopy (XANES). The combined SAXS/XANES measurements were carried out at the µspot beamline at BESSY II (Berlin, Germany). The beamline
For Practice 4.1 Magnesium hydroxide, the active ingredient in milk of magnesia, neutralizes stomach acid, primarily HCl, according to the reaction:
 Stoichiometry For Practice 4.1 Magnesium hydroxide, the active ingredient in milk of magnesia, neutralizes stomach acid, primarily HCl, according to the reaction: What mass of HCl, in grams, is neutralized
Stoichiometry For Practice 4.1 Magnesium hydroxide, the active ingredient in milk of magnesia, neutralizes stomach acid, primarily HCl, according to the reaction: What mass of HCl, in grams, is neutralized
1-amino-9-octadecene, HAuCl 4, hexane, ethanol 55 o C, 16h AuSSs on GO
 Supplementary Figures GO Supplementary Figure S1 1-amino-9-octadecene, HAuCl 4, hexane, ethanol 55 o C, 16h AuSSs on GO Schematic illustration of synthesis of Au square sheets on graphene oxide sheets.
Supplementary Figures GO Supplementary Figure S1 1-amino-9-octadecene, HAuCl 4, hexane, ethanol 55 o C, 16h AuSSs on GO Schematic illustration of synthesis of Au square sheets on graphene oxide sheets.
Copyright Elite Minds Academy 2015 All rights reserved. No part of this publication may be reproduced, stored in or introduced into a retrieval
 1 TOPIC 1 WEEK 1 - THE CHEMICAL EARTH CONTEXTUAL OUTLINE ASSUMED KNOWLEDGE Domain: knowledge and understanding The Earth includes a clearly identifiable biosphere, lithosphere, hydrosphere and atmosphere.
1 TOPIC 1 WEEK 1 - THE CHEMICAL EARTH CONTEXTUAL OUTLINE ASSUMED KNOWLEDGE Domain: knowledge and understanding The Earth includes a clearly identifiable biosphere, lithosphere, hydrosphere and atmosphere.
Stoichiometry. Introduction. Rx between Hydrogen and Oxygen can be described as: Balanced equation: Or Avogadros Number: (number of Molecules)
 Stoichiometry Introduction Rx between Hydrogen and Oxygen can be described as: Balanced equation: Or Or Avogadros Number: (number of Molecules) Or Moles (amount of a substance containing avogadros number
Stoichiometry Introduction Rx between Hydrogen and Oxygen can be described as: Balanced equation: Or Or Avogadros Number: (number of Molecules) Or Moles (amount of a substance containing avogadros number
burette filled with sulphuric acid conical flask 25.0 cm 3 of sodium hydroxide(aq) concentration 2.24 mol / dm 3
 1 Crystals of sodium sulphate-10-water, Na 2 SO 4.10H 2 O, are prepared by titration. burette filled with sulphuric acid conical flask 25.0 cm 3 of sodium hydroxide(aq) concentration 2.24 mol / dm 3 (a)
1 Crystals of sodium sulphate-10-water, Na 2 SO 4.10H 2 O, are prepared by titration. burette filled with sulphuric acid conical flask 25.0 cm 3 of sodium hydroxide(aq) concentration 2.24 mol / dm 3 (a)
Heteroagglomeration of nanosilver with colloidal SiO2 and clay
 , 14, 1 8 Supplementary material Heteroagglomeration of nanosilver with colloidal SiO2 and clay Sébastien Maillette, A Caroline Peyrot, A Tapas Purkait, B Muhammad Iqbal, B Jonathan G. C. Veinot B and
, 14, 1 8 Supplementary material Heteroagglomeration of nanosilver with colloidal SiO2 and clay Sébastien Maillette, A Caroline Peyrot, A Tapas Purkait, B Muhammad Iqbal, B Jonathan G. C. Veinot B and
Elucidating the Role of AgCl in the Nucleation and Growth of Silver Nanoparticles in Ethylene Glycol
 Supplementary Data and Descriptions Elucidating the Role of AgCl in the Nucleation and Growth of Silver Nanoparticles in Ethylene Glycol Suyue Chen, Jesse L. Carey III, David R. Whitcomb, Philippe Bühlmann,
Supplementary Data and Descriptions Elucidating the Role of AgCl in the Nucleation and Growth of Silver Nanoparticles in Ethylene Glycol Suyue Chen, Jesse L. Carey III, David R. Whitcomb, Philippe Bühlmann,
Facile synthesis of polymer and carbon spheres decorated with highly dispersed metal nanoparticles
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 1 Facile synthesis of polymer and carbon spheres decorated with highly dispersed metal nanoparticles
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 1 Facile synthesis of polymer and carbon spheres decorated with highly dispersed metal nanoparticles
Precious metal recovery from nanowaste for sustainable nanotechnology: Current challenges and life cycle considerations
 Precious metal recovery from nanowaste for sustainable nanotechnology: Current challenges and life cycle considerations Dr. Peter Vikesland Dr. Sean McGinnis Paramjeet Pati pvikes@vt.edu smcginn@vt.edu
Precious metal recovery from nanowaste for sustainable nanotechnology: Current challenges and life cycle considerations Dr. Peter Vikesland Dr. Sean McGinnis Paramjeet Pati pvikes@vt.edu smcginn@vt.edu
Ionic or Covalent: Track Those Electrons
 Electrons Goal Use rings and diagrams to model the formation of ionic compounds and covalent compounds. What to Do Use a blank piece of paper and rings to model electron transfers and electron sharing,
Electrons Goal Use rings and diagrams to model the formation of ionic compounds and covalent compounds. What to Do Use a blank piece of paper and rings to model electron transfers and electron sharing,
NOTE: This practice exam contains more than questions than the real final.
 NOTE: This practice exam contains more than questions than the real final. 1. The wavelength of light emitted from a green laser pointer is 5.32 10 2 nm. What is the wavelength in meters? 2. What is the
NOTE: This practice exam contains more than questions than the real final. 1. The wavelength of light emitted from a green laser pointer is 5.32 10 2 nm. What is the wavelength in meters? 2. What is the
Name: Unit 1: Nature of Science
 Name: Unit 1: Nature of Science 1. Using the picture above, list lab safety rules that are being ignored. 2. What are the steps to the scientific method? 3. Convert the following into scientific notation:
Name: Unit 1: Nature of Science 1. Using the picture above, list lab safety rules that are being ignored. 2. What are the steps to the scientific method? 3. Convert the following into scientific notation:
IMADUDDIN SCHOOL Second Term Examination 2017
 Index Register Class number number Name IMADUDDIN SCHOOL Second Term Examination 2017 GRADE 9 CHEMISTRY 5070/02 Paper 2 Theory Nov 2017 TIME 1 hour 30 minutes INSTRUCTIONS TO CANDIDATES Write your name,
Index Register Class number number Name IMADUDDIN SCHOOL Second Term Examination 2017 GRADE 9 CHEMISTRY 5070/02 Paper 2 Theory Nov 2017 TIME 1 hour 30 minutes INSTRUCTIONS TO CANDIDATES Write your name,
Physisorption of Antibodies using BioReady Bare Nanoparticles
 TECHNICAL RESOURCE Lateral Flow Immunoassays Physisorption of Antibodies using BioReady Bare Nanoparticles Introduction For more than 20 years, lateral flow immunoassay diagnostic tests have provided a
TECHNICAL RESOURCE Lateral Flow Immunoassays Physisorption of Antibodies using BioReady Bare Nanoparticles Introduction For more than 20 years, lateral flow immunoassay diagnostic tests have provided a
Supporting Information
 Elucidating the Effects of Polyprotic Acid Speciation in Calcium Oxalate Crystallization Jihae Chung, Ricardo Sosa, and Jeffrey D. Rimer * University of Houston, Department of Chemical and Biomolecular
Elucidating the Effects of Polyprotic Acid Speciation in Calcium Oxalate Crystallization Jihae Chung, Ricardo Sosa, and Jeffrey D. Rimer * University of Houston, Department of Chemical and Biomolecular
IONIC COVALENT BONDING WEBQUEST. IONIC BONDING WEBSITE 1:
 IONIC COVALENT BONDING WEBQUEST IONIC BONDING WEBSITE 1: http://visionlearning.com/en/library/chemistry/1/chemical-bonding/55 1. Approximately how many elements are represented on the periodic table? 2.
IONIC COVALENT BONDING WEBQUEST IONIC BONDING WEBSITE 1: http://visionlearning.com/en/library/chemistry/1/chemical-bonding/55 1. Approximately how many elements are represented on the periodic table? 2.
CHEMISTRY (SALTERS) 2854/01 Chemistry by Design
 THIS IS A LEGACY SPECIFICATIN ADVANCED GCE CHEMISTRY (SALTERS) 2854/01 Chemistry by Design *CUP/T69315* Candidates answer on the question paper A calculator may be used for this paper CR Supplied Materials:
THIS IS A LEGACY SPECIFICATIN ADVANCED GCE CHEMISTRY (SALTERS) 2854/01 Chemistry by Design *CUP/T69315* Candidates answer on the question paper A calculator may be used for this paper CR Supplied Materials:
Supporting Information
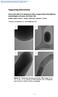 Supporting Information Interaction effects of mesoporous silica nanoparticles with different morphologies on human red blood cells Madhura Joglekar, Robert A. Roggers, Yannan Zhao, and Brian G. Trewyn
Supporting Information Interaction effects of mesoporous silica nanoparticles with different morphologies on human red blood cells Madhura Joglekar, Robert A. Roggers, Yannan Zhao, and Brian G. Trewyn
The Chemistry of Life Chapter 2. Prof. J. Dodd
 The Chemistry of Life Chapter 2 Prof. J. Dodd Why should we study chemistry in C, H, O, N Atoms are composed of 3 main particles: (subatomic particles) Protons (+) Neutrons Electrons (-) Protons and Neutrons
The Chemistry of Life Chapter 2 Prof. J. Dodd Why should we study chemistry in C, H, O, N Atoms are composed of 3 main particles: (subatomic particles) Protons (+) Neutrons Electrons (-) Protons and Neutrons
Moon Il Kim a, Jongmin Shim b, Taihua Li a, Min-Ah Woo a, Daeyeon Cho c, Jinwoo Lee b, Hyun Gyu Park a, *
 Colorimetric quantification of galactose using a nanostructured multi-catalyst system entrapping galactose oxidase and magnetic nanoparticles as peroxidase mimetics Moon Il Kim a, Jongmin Shim b, Taihua
Colorimetric quantification of galactose using a nanostructured multi-catalyst system entrapping galactose oxidase and magnetic nanoparticles as peroxidase mimetics Moon Il Kim a, Jongmin Shim b, Taihua
- Use the STEM NAME of the element, then add "-ide" suffix
 65 ANIONS 2 kinds Main-group nonmetals - Use the STEM NAME of the element, then add "-ide" suffix N : "nitride" ion P : "phosphide ion" O : "oxide ion" F : "fluoride ion" Polyatomic ions - Memorize list.(see
65 ANIONS 2 kinds Main-group nonmetals - Use the STEM NAME of the element, then add "-ide" suffix N : "nitride" ion P : "phosphide ion" O : "oxide ion" F : "fluoride ion" Polyatomic ions - Memorize list.(see
Supplementary Figure 1 a-c, The viscosity fitting curves of high-molecular-weight poly(vinyl alcohol) (HMW-PVA) (a), middle-molecular-weight
 Supplementary Figure 1 a-c, The viscosity fitting curves of high-molecular-weight poly(vinyl alcohol) (HMW-PVA) (a), middle-molecular-weight poly(vinyl alcohol) (MMW-PVA) (b) and low-molecular-weight poly(vinyl
Supplementary Figure 1 a-c, The viscosity fitting curves of high-molecular-weight poly(vinyl alcohol) (HMW-PVA) (a), middle-molecular-weight poly(vinyl alcohol) (MMW-PVA) (b) and low-molecular-weight poly(vinyl
Supplementary Information: Triggered self-assembly of magnetic nanoparticles
 Supplementary Information: Triggered self-assembly of magnetic nanoparticles L. Ye 1,3, T. Pearson 1, Y. Cordeau 2, O.T. Mefford 2, and T. M. Crawford 1 1 Smart State Center for Experimental Nanoscale
Supplementary Information: Triggered self-assembly of magnetic nanoparticles L. Ye 1,3, T. Pearson 1, Y. Cordeau 2, O.T. Mefford 2, and T. M. Crawford 1 1 Smart State Center for Experimental Nanoscale
Kinetically Controlled Seeded Growth Synthesis of Citrate Stabilized Gold. Nanoparticles up to 200 nm: Size Focusing versus.
 Kinetically Controlled Seeded Growth Synthesis of Citrate Stabilized Gold Nanoparticles up to 200 nm: Size Focusing versus. Ostwald Ripening Neus G. Bastús, Joan Comenge and Víctor Puntes Additional Results
Kinetically Controlled Seeded Growth Synthesis of Citrate Stabilized Gold Nanoparticles up to 200 nm: Size Focusing versus. Ostwald Ripening Neus G. Bastús, Joan Comenge and Víctor Puntes Additional Results
Jahresbericht 2003 der Arbeitsgruppe Experimentalphysik Prof. Dr. Michael Farle
 olloidal Synthesis of Magnetic Nanoparticles V. Salgueirino Maceira and M. Farle 1 Institut für Physik, Universität Duisburg-Essen, Lotharstr. 1, 47048 Duisburg 1. Introduction 1 The synthesis of monodisperse
olloidal Synthesis of Magnetic Nanoparticles V. Salgueirino Maceira and M. Farle 1 Institut für Physik, Universität Duisburg-Essen, Lotharstr. 1, 47048 Duisburg 1. Introduction 1 The synthesis of monodisperse
SPECIMEN. Candidate Number
 Advanced Subsidiary GCE CHEMISTRY A F321 QP Unit F321: Atoms, Bonds and Groups Specimen Paper Candidates answer on the question paper. Additional Materials: Data Sheet for Chemistry (Inserted) Scientific
Advanced Subsidiary GCE CHEMISTRY A F321 QP Unit F321: Atoms, Bonds and Groups Specimen Paper Candidates answer on the question paper. Additional Materials: Data Sheet for Chemistry (Inserted) Scientific
TOPIC: Chemical Bonds
 TOPIC: Chemical Bonds H O bond H a water molecule In elements and compounds, the atoms are held together by chemical bonds. Forming a bond makes an atom more stable, so atoms form as many bonds are they
TOPIC: Chemical Bonds H O bond H a water molecule In elements and compounds, the atoms are held together by chemical bonds. Forming a bond makes an atom more stable, so atoms form as many bonds are they
مدرسة القديسة مريم الكاثوليكية الثانوية - ST. MARY S CATHOLIC HIGH SCHOOL, DUBAI REVISION WORKSHEET YEAR-10 JANUARY 2019
 مدرسة القديسة مريم الكاثوليكية الثانوية - دبي ST. MARY S CATHOLIC HIGH SCHOOL, DUBAI REVISION WORKSHEET YEAR-10 JANUARY 2019 1.Calculate the relative formula mass (Mr) of aluminium sulfate, Al 2 (SO 4
مدرسة القديسة مريم الكاثوليكية الثانوية - دبي ST. MARY S CATHOLIC HIGH SCHOOL, DUBAI REVISION WORKSHEET YEAR-10 JANUARY 2019 1.Calculate the relative formula mass (Mr) of aluminium sulfate, Al 2 (SO 4
Supplementary Figure 1: ADF images and profiles for several types of atomic chains encapsulated in DWNTs. (a d) ADF images of NaI, CsF, CsCl, and CsI
 Supplementary Figure 1: ADF images and profiles for several types of atomic chains encapsulated in DWNTs. (a d) ADF images of NaI, CsF, CsCl, and CsI atomic chains encapsulated in DWNTs, respectively.
Supplementary Figure 1: ADF images and profiles for several types of atomic chains encapsulated in DWNTs. (a d) ADF images of NaI, CsF, CsCl, and CsI atomic chains encapsulated in DWNTs, respectively.
S 8 + F 2 SF 6 4/9/2014. iclicker Participation Question: Balance the following equation by inspection: H + + Cr 2 O 7 + C 2 H 5 OH
 Today: Redox Reactions Oxidations Reductions Oxidation Numbers Half Reactions Balancing in Acidic Solution Balancing in Basic Solution QUIZ 3 & EXAM 3 moved up by one day: Quiz 3 Wednesday/Thursday next
Today: Redox Reactions Oxidations Reductions Oxidation Numbers Half Reactions Balancing in Acidic Solution Balancing in Basic Solution QUIZ 3 & EXAM 3 moved up by one day: Quiz 3 Wednesday/Thursday next
Q1. The chart shows the processes involved in the manufacture of nitric acid from ammonia.
 Q1. The chart shows the processes involved in the manufacture of nitric acid from ammonia. (a) Complete the word equation for the reaction that takes place in the first reaction vessel. ammonia +... nitrogen
Q1. The chart shows the processes involved in the manufacture of nitric acid from ammonia. (a) Complete the word equation for the reaction that takes place in the first reaction vessel. ammonia +... nitrogen
Rh 3d. Co 2p. Binding Energy (ev) Binding Energy (ev) (b) (a)
 Co 2p Co(0) 778.3 Rh 3d Rh (0) 307.2 810 800 790 780 770 Binding Energy (ev) (a) 320 315 310 305 Binding Energy (ev) (b) Supplementary Figure 1 Photoemission features of a catalyst precursor which was
Co 2p Co(0) 778.3 Rh 3d Rh (0) 307.2 810 800 790 780 770 Binding Energy (ev) (a) 320 315 310 305 Binding Energy (ev) (b) Supplementary Figure 1 Photoemission features of a catalyst precursor which was
IONIC AND METALLIC BONDING
 7 IONIC AND METALLIC BONDING Chapter Test B A. Matching Match each term in Column B with the correct description in Column A. Write the letter of the correct term on the line. Column A Column B 1. compound
7 IONIC AND METALLIC BONDING Chapter Test B A. Matching Match each term in Column B with the correct description in Column A. Write the letter of the correct term on the line. Column A Column B 1. compound
rinds for 8 Loihi rocks. Sample spectra, fits and model compounds are shown in Figure S5. All
 SUPPLEMENTARY INFORMATION Supplementary Table 1 Sample Location %Mats (M34, M39) 2L-FH (III)- smectite Vivianite Basalt glass red chi 2 J2-369-R2 Marker 17a 30% 21% - 11% 38% 0.06 J2-369-R5 Marker 17a
SUPPLEMENTARY INFORMATION Supplementary Table 1 Sample Location %Mats (M34, M39) 2L-FH (III)- smectite Vivianite Basalt glass red chi 2 J2-369-R2 Marker 17a 30% 21% - 11% 38% 0.06 J2-369-R5 Marker 17a
Supporting Information
 Supporting Information Extraordinary Off-stoichiometric Bismuth Telluride for Enhanced n- type Thermoelectric Power Factor Kunsu Park,,,# Kyunghan Ahn,,# Joonil Cha,, Sanghwa Lee,, Sue In Chae,, Sung-
Supporting Information Extraordinary Off-stoichiometric Bismuth Telluride for Enhanced n- type Thermoelectric Power Factor Kunsu Park,,,# Kyunghan Ahn,,# Joonil Cha,, Sanghwa Lee,, Sue In Chae,, Sung-
Supporting Information. One-Pot Synthesis of Reduced Graphene
 Supporting Information One-Pot Synthesis of Reduced Graphene Oxide/Metal (oxide) Composites Xu Wu, Yuqian Xing, David Pierce, Julia Xiaojun Zhao* a Department of Chemistry, University of North Dakota,
Supporting Information One-Pot Synthesis of Reduced Graphene Oxide/Metal (oxide) Composites Xu Wu, Yuqian Xing, David Pierce, Julia Xiaojun Zhao* a Department of Chemistry, University of North Dakota,
NAMING IONIC COMPOUNDS. ammonium sulfide. iron(ii) carbonate. barium phosphate. titanium(iv) sulfide. Spelling matters!
 67 NAMING IONIC COMPOUNDS ammonium sulfide iron(ii) carbonate titanium(iv) sulfide barium phosphate Spelling matters! barium phosphide 68 DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The
67 NAMING IONIC COMPOUNDS ammonium sulfide iron(ii) carbonate titanium(iv) sulfide barium phosphate Spelling matters! barium phosphide 68 DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The
Chapter 4: Forces Between Particles
 Chapter 4: Forces Between Particles NOBLE GAS CONFIGURATIONS An electronic configuration that is characterized by two electrons in the valence shell of helium and eight electrons in the valence shell of
Chapter 4: Forces Between Particles NOBLE GAS CONFIGURATIONS An electronic configuration that is characterized by two electrons in the valence shell of helium and eight electrons in the valence shell of
Supporting Information
 Supporting Information Characterizing the Effect of Salt and Surfactant Concentration on the Counter-ion Atmosphere around Surfactant Stabilized SWCNTs using Analytical Ultracentrifugation Stephanie Lam
Supporting Information Characterizing the Effect of Salt and Surfactant Concentration on the Counter-ion Atmosphere around Surfactant Stabilized SWCNTs using Analytical Ultracentrifugation Stephanie Lam
Chemistry course ACME Faculty, EHVE course B.Sc. Studies, I year, I semester. Inorganic chemistry. Oxidation state. Oxidation state.
 -- Chemistry course ACME Faculty, EHVE course B.Sc. Studies, I year, I semester Leszek Niedzicki, PhD, Eng. Inorganic chemistry Particle Particle can be a simple atom system, i.e.: atom (usually of metal
-- Chemistry course ACME Faculty, EHVE course B.Sc. Studies, I year, I semester Leszek Niedzicki, PhD, Eng. Inorganic chemistry Particle Particle can be a simple atom system, i.e.: atom (usually of metal
Lower Sixth Chemistry. Sample Entrance Examination
 Lower Sixth Chemistry Sample Entrance Examination Time allowed: 60 minutes Name: Total : 60 Marks INSTRUCTIONS : Answer all questions Answers should be written in the spaces provided Dictionaries or reference
Lower Sixth Chemistry Sample Entrance Examination Time allowed: 60 minutes Name: Total : 60 Marks INSTRUCTIONS : Answer all questions Answers should be written in the spaces provided Dictionaries or reference
Cambridge International Examinations Cambridge Ordinary Level
 Cambridge International Examinations Cambridge Ordinary Level *6218899063* CHEMISTRY 5070/41 Paper 4 Alternative to Practical October/November 2015 1 hour Candidates answer on the Question Paper. No Additional
Cambridge International Examinations Cambridge Ordinary Level *6218899063* CHEMISTRY 5070/41 Paper 4 Alternative to Practical October/November 2015 1 hour Candidates answer on the Question Paper. No Additional
Chemical Bonds, Molecular Models, and Molecular Shapes
 Chemical Bonds, Molecular Models, and Molecular Shapes PRELAB ASSINGMENT Read the entire laboratory write up and answer the following questions before coming to lab. Read the entire laboratory write up
Chemical Bonds, Molecular Models, and Molecular Shapes PRELAB ASSINGMENT Read the entire laboratory write up and answer the following questions before coming to lab. Read the entire laboratory write up
Atoms with a complete outer shell do not react with other atoms. The outer shell is called the valence shell. Its electrons are valence electrons.
 Bonding and the Outer Shell Use this table for reference: http://www.dreamwv.com/primer/page/s_pertab.html Atoms with incomplete shells react with others in a way that allows it to complete the outer shell.
Bonding and the Outer Shell Use this table for reference: http://www.dreamwv.com/primer/page/s_pertab.html Atoms with incomplete shells react with others in a way that allows it to complete the outer shell.
Elements in the Periodic Table show a periodic trend in atomic radius. In your answer you should use appropriate technical terms, spelled correctly.
 1 The Periodic Table is arranged in periods and groups (a) Elements in the Periodic Table show a periodic trend in atomic radius State and explain the trend in atomic radius from Li to F In your answer
1 The Periodic Table is arranged in periods and groups (a) Elements in the Periodic Table show a periodic trend in atomic radius State and explain the trend in atomic radius from Li to F In your answer
Selective Activation of Methane on Single-Atom Catalyst of Rhodium Dispersed on Zirconia for Direct Conversion
 Supporting Information Selective Activation of Methane on Single-Atom Catalyst of Rhodium Dispersed on Zirconia for Direct Conversion Yongwoo Kwon a, Tae Yong Kim b, Gihun Kwon a, Jongheop Yi b *, and
Supporting Information Selective Activation of Methane on Single-Atom Catalyst of Rhodium Dispersed on Zirconia for Direct Conversion Yongwoo Kwon a, Tae Yong Kim b, Gihun Kwon a, Jongheop Yi b *, and
This packet contains review material from Pre-AP Chemistry. Be prepared to take a quiz over this material during the first week of school.
 This packet contains review material from Pre-AP Chemistry. Be prepared to take a quiz over this material during the first week of school. How many significant figures (digits) are represented by each
This packet contains review material from Pre-AP Chemistry. Be prepared to take a quiz over this material during the first week of school. How many significant figures (digits) are represented by each
NAMING IONIC COMPOUNDS
 NAMING IONIC COMPOUNDS iron(ii) carbonate ammonium sulfide titanium (IV) sulfide barium phosphate Spelling matters! calcium nitrate sodium phosphide DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE
NAMING IONIC COMPOUNDS iron(ii) carbonate ammonium sulfide titanium (IV) sulfide barium phosphate Spelling matters! calcium nitrate sodium phosphide DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE
Draw one line from each solution to the ph value of the solution. Solution ph value of the solution
 1 The ph scale is a measure of the acidity or alkalinity of a solution. (a) Draw one line from each solution to the ph value of the solution. Solution ph value of the solution 5 Acid 7 9 Neutral 11 13
1 The ph scale is a measure of the acidity or alkalinity of a solution. (a) Draw one line from each solution to the ph value of the solution. Solution ph value of the solution 5 Acid 7 9 Neutral 11 13
Harnessing the Synergy Between Upconverting Nanoparticles and Lanthanide Complexes in a Multi-Wavelength Responsive Hybrid System.
 Electronic Supporting Information for: Harnessing the Synergy Between Upconverting Nanoparticles and Lanthanide Complexes in a Multi-Wavelength Responsive Hybrid System. Riccardo Marin, a Ilias Halimi,
Electronic Supporting Information for: Harnessing the Synergy Between Upconverting Nanoparticles and Lanthanide Complexes in a Multi-Wavelength Responsive Hybrid System. Riccardo Marin, a Ilias Halimi,
Supplementary figure 1 Application of tmfret in LeuT. (a) To assess the feasibility of using tmfret for distance-dependent measurements in LeuT, a
 Supplementary figure 1 Application of tmfret in LeuT. (a) To assess the feasibility of using tmfret for distance-dependent measurements in LeuT, a series of tmfret-pairs comprised of single cysteine mutants
Supplementary figure 1 Application of tmfret in LeuT. (a) To assess the feasibility of using tmfret for distance-dependent measurements in LeuT, a series of tmfret-pairs comprised of single cysteine mutants
Chapter 9 Bonding - 1. Dr. Sapna Gupta
 Chapter 9 Bonding - 1 Dr. Sapna Gupta Lewis Dot Symbol Lewis dot symbols is a notation where valence electrons are shown as dots. Draw the electrons symmetrically around the sides (top, bottom, left and
Chapter 9 Bonding - 1 Dr. Sapna Gupta Lewis Dot Symbol Lewis dot symbols is a notation where valence electrons are shown as dots. Draw the electrons symmetrically around the sides (top, bottom, left and
Supporting Information (Journal of Materials Chemistry A) for. via a scalable limited space chemical vapor deposition technique
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information (Journal of Materials Chemistry A) for Development
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information (Journal of Materials Chemistry A) for Development
Unit 3 Ray Tedder s Chemistry I Test Prep Guide page 1
 Unit 3 Ray Tedder s Chemistry I Test Prep Guide page 1 Bonding Unit 3: Chemistry I In this unit all students must be able to Understand that the structure of molecules is the result of nonmetals sharing
Unit 3 Ray Tedder s Chemistry I Test Prep Guide page 1 Bonding Unit 3: Chemistry I In this unit all students must be able to Understand that the structure of molecules is the result of nonmetals sharing
Chapter 12. Chemical Bonding
 Chapter 12 Chemical Bonding Chapter 12 Introduction to Chemical Bonding Chemical Bonding Valence electrons are the electrons in the outer shell (highest energy level) of an atom. A chemical bond is a mutual
Chapter 12 Chemical Bonding Chapter 12 Introduction to Chemical Bonding Chemical Bonding Valence electrons are the electrons in the outer shell (highest energy level) of an atom. A chemical bond is a mutual
Supporting Information
 Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2016 Supporting Information Single-crystalline Pd square nanoplates enclosed by {100}
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2016 Supporting Information Single-crystalline Pd square nanoplates enclosed by {100}
Chem!stry. Atomic Structure and Chemical Bonding Concepts: Change, Models & Systems
 Chem!stry Name: ( ) Class: Date: / / Atomic Structure and Chemical Bonding Concepts: Change, Models & Systems All matter is essentially composed of three different particles electrons, neutrons and protons
Chem!stry Name: ( ) Class: Date: / / Atomic Structure and Chemical Bonding Concepts: Change, Models & Systems All matter is essentially composed of three different particles electrons, neutrons and protons
Citric acid adsorption on cerium oxide nanoparticles in aqueous suspensions at acidic and. circumneutral ph
 Citric acid adsorption on cerium oxide nanoparticles in aqueous suspensions at acidic and circumneutral ph Abstract Citric acid is a naturally abundant organic acid, which can play an important role in
Citric acid adsorption on cerium oxide nanoparticles in aqueous suspensions at acidic and circumneutral ph Abstract Citric acid is a naturally abundant organic acid, which can play an important role in
4-4 Chemical changes Chemistry
 4-4 Chemical changes Chemistry.0 A student investigated the reaction of sodium carbonate with dilute hydrochloric acid. The student used the apparatus shown in Figure. Figure Sodium carbonate This is the
4-4 Chemical changes Chemistry.0 A student investigated the reaction of sodium carbonate with dilute hydrochloric acid. The student used the apparatus shown in Figure. Figure Sodium carbonate This is the
-:Vijay Singh(09CEB023)
 Heterogeneous Semiconductor Photocatalyst -:Vijay Singh(09CEB023) Guided by Azrina Abd Aziz Under Dr. Saravanan Pichiah Preparation of TiO 2 Nanoparticle TiO 2 was prepared by hydrolysis and poly-condensation
Heterogeneous Semiconductor Photocatalyst -:Vijay Singh(09CEB023) Guided by Azrina Abd Aziz Under Dr. Saravanan Pichiah Preparation of TiO 2 Nanoparticle TiO 2 was prepared by hydrolysis and poly-condensation
Supporting Information:
 Supporting Information: Supplementary Figure 1. (a) TEM and (b) HRTEM image of the Pd nanocube seeds. Inset of (b): the FFT pattern. (c) Magnified image of the square region highlighted in (b). Supplementary
Supporting Information: Supplementary Figure 1. (a) TEM and (b) HRTEM image of the Pd nanocube seeds. Inset of (b): the FFT pattern. (c) Magnified image of the square region highlighted in (b). Supplementary
Personalised Learning Checklists Edexcel Combined: Chemistry Paper 1
 Edexcel (combined) Chemistry Topics (1SC0) from 2016 - Paper 1 (Topic 1 parts a&b) Topic Student Checklist R A G Describe how the Dalton model of an atom has changed over time because of the discovery
Edexcel (combined) Chemistry Topics (1SC0) from 2016 - Paper 1 (Topic 1 parts a&b) Topic Student Checklist R A G Describe how the Dalton model of an atom has changed over time because of the discovery
Supplementary Information
 Supplementary Information Supplementary Table 1. Atomic details for the crystal structures of silver closo-boranes. See Table 1 for further details. α Ag 2 B 10 H 10 Wyckoff x y z U / Å 2 Occ. Ag 4d 0.250
Supplementary Information Supplementary Table 1. Atomic details for the crystal structures of silver closo-boranes. See Table 1 for further details. α Ag 2 B 10 H 10 Wyckoff x y z U / Å 2 Occ. Ag 4d 0.250
Supplementary Material
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supplementary Material Spin crossover and reversible single-crystal to single-crystal
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supplementary Material Spin crossover and reversible single-crystal to single-crystal
SUPPLEMENTARY INFORMATION
 Supplementary Table 1: Amplitudes of three current levels. Level 0 (pa) Level 1 (pa) Level 2 (pa) TrkA- TrkH WT 200 K 0.01 ± 0.01 9.5 ± 0.01 18.7 ± 0.03 200 Na * 0.001 ± 0.01 3.9 ± 0.01 12.5 ± 0.03 200
Supplementary Table 1: Amplitudes of three current levels. Level 0 (pa) Level 1 (pa) Level 2 (pa) TrkA- TrkH WT 200 K 0.01 ± 0.01 9.5 ± 0.01 18.7 ± 0.03 200 Na * 0.001 ± 0.01 3.9 ± 0.01 12.5 ± 0.03 200
Stoichiometry Chapter 9 Practice Assessment B
 NAME Hour Date Stoichiometry Chapter 9 Practice Assessment B Objective 1: Interpret balanced chemical equations in terms of interacting moles, representative particles, masses, and gas volume at STP. Directions:
NAME Hour Date Stoichiometry Chapter 9 Practice Assessment B Objective 1: Interpret balanced chemical equations in terms of interacting moles, representative particles, masses, and gas volume at STP. Directions:
