Chemistry 2541, Fall 2017 Midterm Exam 2 (100 points)
|
|
|
- Winfred Simmons
- 6 years ago
- Views:
Transcription
1 Chemistry 2541, Fall 2017 Midterm Exam 2 (100 points) Important notes: - Please use the provided Scantron form for your answers; you can keep the sheet with the questions and can use it as scratch paper - Do not forget to write your name on the Scantron form - You will not receive credit for unmarked answers or for more than one mark on answer line - Your scores will be posted on egradebook; graded Scantron forms will not be returned to students. Questions 1-28 (84 pts): Please mark the appropriate box on the front of the Scantron form (3 pts each). 1. What is the IUPAC name for the compound shown in the box C A) (2S,3S)-2-bromo-3-chlorobutane B) (2S,3 R)-2-bromo-3-chlorobutane C Cl C) (2R,3S)-2-bromo-3-chlorobutane D) (2R,3 R)-2-bromo-3-chlorobutane 2. Consider the following orders of priority (highest to lowest). Which order is incorrect A) > C2 > C > C=C2 B) C > C=C2 > C2 > C2C3 C) > C2Cl > C2C3 > C3 D) N2 > C2 > C2C2S > C2C2C3 3. What is the relationship of structure I to the other structures shown in the box Which one of the following pairs is a pair of diastereomers I) C II) C III) C A) I and II B) I and III IV) C V) C C) I and IV D) I and V 1
2 4. What is the configuration (R or S) at the carbon atoms 2 and 3 of the molecule shown in the box 2 3 C C 2 A) 2R,3R B) 2S,3R C) 2R,3S D) 2S,3S 5. Which one of the following molecules is a meso compound C 2 A) B) C 2 C 2 6. Which one of the following molecules is chiral A) Cl B) Cl Cl 7. Which of the statements below is NT true about stereoisomers A. They have the same connectivity of atoms B. They have the same molecular weight C. They have the same orientation of atoms in space D. They have the same molecular formula 8. What is the energy diagram for the reaction in the box: A) B) I NaS S 9. What is the rate equation for the reaction in the box (excess) A) Rate = k [R][ ] B) Rate = k [R] C) Rate = k [R][ ] D) Rate = k [ ] 2
3 10. Which structure best represents the transition state for the reaction in the box δ C A) 3 B) C NaCN CN CN δ C) C D) 11. What is the structure of an intermediate in the reaction shown in the box C δ+ C CN δ+ 2 (excess) + A) B) What is the IUPAC name of the major product for the reaction shown in the box KI A) cis-1-bromo-4-methylcyclohexane B) trans-1-bromo-4-methylcyclohexane C) cis-1-iodo-4-methylcyclohexane D) trans-1-iodo-4-methylcyclohexane 13. What is the main product of the reaction shown in the box C SNa C S A) B) C SNa C S C SNa 14. What is the main product of the E2-elimination reaction shown in the box C C 2 Na 6 5 C 6 5 C 6 5 C C A) C C B) C C C) C C D) C C C 2 3
4 15. What is the main product of the reaction shown in the box A) B) Na 16. Which of the following bromoalkanes reacts the fastest with sodium cyanide, NaCN, in acetonitrile A) bromocyclohexane B) 2-bromo-2-methylbutane C) 1-bromo-3-methylcyclobutane D) bromoethane 17. Which reagent can be used for the reaction shown in the box I reagent A) NaCN B) C CNa acetonitrile C) NaN 3 D) C CNa 18. Which of the following alkenes has an E-configuration of the double bond F C 2 C(C 2 ) 2 A) B) 2 N C 2 C 2 C 2 B 2 C( ) What is the IUPAC name for the compound shown in the box F A) (E)-3-ethyl-2,5,5-trimethyl-1,3-hexadiene B) (Z)-3-ethyl-2,5,5-trimethyl-1,3-hexadiene C) (E)-3-vinyl-2,5,5-trimethyl-3-hexene D) (Z)-3-vinyl-2,5,5-trimethyl-3-hexene 20. What is the structure of an intermediate in the reaction shown in the box 2 CCl 4, dark, 20 o C A) B) C) D) 4
5 21. What is the structure of an intermediate in the rearrangement reaction shown in the box 2 S 4, 2 A) B) 22. What is the main product of the reaction shown in the box A) B) 1) B 3 2) 2 2 Na, 2 C) D) 23. What alkene when treated with ozone and then with (C3)2S gives the products shown in the box + A) trans-2-butene B) (E)-3-methyl-2-pentene C) 2-methyl-2-butene D) (Z)-3-methyl-2-pentene 24. Which is the least stable alkene A) 1-pentene B) trans-2-pentene C) cis-2-pentene D) 2,3-dimethyl-2-pentene 25. What is the IUPAC name of the major product for the reaction shown in the box + N light or heat C 2 Cl 2 A) 1-bromocyclohexene B) 4-bromocyclohexene C) 3-bromocyclohexene D) bromocyclohexane 5
6 26. What is the IUPAC name of the major product for the reaction shown in the box RR (peroxides) A) 1-bromo-2-methylpropane B) 2-bromo-2-methylpropane C) 3-bromo-2-methyl-1-propene D) 1,2-dibromo-2-methylpropane 27. Which sequence of reagents can be used for the reaction shown in the box reagent (1), hν reagent (2) C 2 Cl 2 acetonitrile N 3 A) (1), (2) NaN 3 B) (1) NBS, (2) NaN 3 C) (1) NaN 2, (2) 2 D) (1) NaN, (2) 28. Which sequence of reagents can be used for the reaction shown in the box C A) (1) NBS, (2) N 3 C3 3 reagent (1) reagent (2) B) (1), (2) N 2 peroxides N 3 C) (1), (2) N 3 D) (1) 2, (2) N 2 Question 29: Please write your answers in boxes on the back of the Scantron form 29. Provide the reagents that give the indicated products in high yield (4 pts each): 1. line ( ) 2 S 1. line NaS 3, 2 line 68, hν line 69 N 3 6
Chemistry 2541, Fall 2017 Midterm Exam 3 (100 points)
 Important notes: Chemistry 2541, Fall 2017 Midterm Exam 3 (100 points) - Please use the provided Scantron form for your answers; you can keep the sheet with the questions and can use it as scratch paper
Important notes: Chemistry 2541, Fall 2017 Midterm Exam 3 (100 points) - Please use the provided Scantron form for your answers; you can keep the sheet with the questions and can use it as scratch paper
CHEM Exam 2 ANSWER KEY
 CEM 3311-200 Exam 2 ANSWER KEY ctober 23, 2012 Time: 2 ours Please copy and sign the onor Pledge on the scantron sheet in the space below the double lines. I pledge that n my honor, as a University of
CEM 3311-200 Exam 2 ANSWER KEY ctober 23, 2012 Time: 2 ours Please copy and sign the onor Pledge on the scantron sheet in the space below the double lines. I pledge that n my honor, as a University of
CH Organic Chemistry I (Katz) Practice Exam #3- Fall 2013
 C2710 - rganic Chemistry I (Katz) Practice Exam #3- all 2013 Name: Score: Part I - Choose the best answer and write the letter of your choice in the space provided. (32 pts) 1. f the following, which reaction
C2710 - rganic Chemistry I (Katz) Practice Exam #3- all 2013 Name: Score: Part I - Choose the best answer and write the letter of your choice in the space provided. (32 pts) 1. f the following, which reaction
Chemistry 2541, Fall 2017 Midterm Exam 1 (100 points)
 hemistry 2541, all 2017 Midterm Exam 1 (100 points) Important notes: - Please use the provided Scantron form for your answers; you can keep the sheet with the questions and can use it as scratch paper
hemistry 2541, all 2017 Midterm Exam 1 (100 points) Important notes: - Please use the provided Scantron form for your answers; you can keep the sheet with the questions and can use it as scratch paper
Chemistry Fall Semester 2007; Midterm 3 Exam
 1 Chemistry 2521 Fall Semester 2007; Midterm 3 Exam December 7, Friday, 1:00 to 1:50 pm This exam has 7 problems (100 pts) on 9 pages. Make sure your copy is complete and correct. Printed Name (Last, First)
1 Chemistry 2521 Fall Semester 2007; Midterm 3 Exam December 7, Friday, 1:00 to 1:50 pm This exam has 7 problems (100 pts) on 9 pages. Make sure your copy is complete and correct. Printed Name (Last, First)
Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron sheet.
 Test 3 Chemistry 2321 April 25, 2003 McMurry, Chapters 9-11 103 Total Points Name Please Print Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron
Test 3 Chemistry 2321 April 25, 2003 McMurry, Chapters 9-11 103 Total Points Name Please Print Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron
As time allows, additional practice questions for Exam 2 follow. This is an incomplete collection. Content & emphasis will vary.
 Chem 226 Exam 2 Fall 2005: will cover Bruice, Chaps 3 through 6; Note: review e-mail & inclass quizzes and Worksheets, suggest on-line practice questions and quizzes plus ACS rganic Chemistry Guide As
Chem 226 Exam 2 Fall 2005: will cover Bruice, Chaps 3 through 6; Note: review e-mail & inclass quizzes and Worksheets, suggest on-line practice questions and quizzes plus ACS rganic Chemistry Guide As
B. A transition state represents a maximum on the reaction path diagram and can be isolated.
 Practice Hour Exam 2, Chemistry 2210, Organic Chemistry I 1. The most stable carbocation is: 2. Which of the following statements is true of transition states? A. A transition state represents a minimum
Practice Hour Exam 2, Chemistry 2210, Organic Chemistry I 1. The most stable carbocation is: 2. Which of the following statements is true of transition states? A. A transition state represents a minimum
Final Exam Professor R. Hoenigman
 I pledge to uphold the CU onor Code: CEM 3311-100 Spring 2007 Final Exam Professor R. oenigman Signature Name (printed) Last four digits of your student ID number Recitation TA Recitation number, day,
I pledge to uphold the CU onor Code: CEM 3311-100 Spring 2007 Final Exam Professor R. oenigman Signature Name (printed) Last four digits of your student ID number Recitation TA Recitation number, day,
CH320/328 M Spring 2014
 CH320/328 M Spring 2014 HW Set #3 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. Carefully record your
CH320/328 M Spring 2014 HW Set #3 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. Carefully record your
Exam 2 Chem 109a Fall 2004
 Exam 2 Chem 109a Fall 2004 Please put your name and perm number on both the exam and the scantron sheet. Next, answer the following 34 multiple-choice questions on the scantron sheet. Then choose one A-type
Exam 2 Chem 109a Fall 2004 Please put your name and perm number on both the exam and the scantron sheet. Next, answer the following 34 multiple-choice questions on the scantron sheet. Then choose one A-type
8. What is the slow, rate-determining step, in the acidcatalyzed dehydration of 2-methyl-2-propanol?
 CHEMISTRY 313-03 MIDTERM # 2 answer key October 25, 2011 Statistics: Average: 68 pts (68%); Highest: 100 pts (100%); Lowest: 30 pts (30%) Number of students performing at or above average: 56 (54%) Number
CHEMISTRY 313-03 MIDTERM # 2 answer key October 25, 2011 Statistics: Average: 68 pts (68%); Highest: 100 pts (100%); Lowest: 30 pts (30%) Number of students performing at or above average: 56 (54%) Number
Chemistry 51 Exam #3. Name KEY November 20, 2001
 Chemistry 51 Exam #3 Name KEY November 20, 2001 This exam has nine (9) questions. Please check before beginning to make sure no questions are missing. All scratch work must be done on the attached blank
Chemistry 51 Exam #3 Name KEY November 20, 2001 This exam has nine (9) questions. Please check before beginning to make sure no questions are missing. All scratch work must be done on the attached blank
Departmental Final Examination. Organic Chemistry I Caffein
 Departmental Final Examination rganic Chemistry I 2423 Caffein Name CEMISTRY 2423 FINAL EXAM FALL, 2005 DIRECTINS: A periodic table is attached at the end of this exam. Please answer all questions as completely
Departmental Final Examination rganic Chemistry I 2423 Caffein Name CEMISTRY 2423 FINAL EXAM FALL, 2005 DIRECTINS: A periodic table is attached at the end of this exam. Please answer all questions as completely
CHE 321 Summer 2012 Exam 2 Form Select the correct number of chirality centers in heroin, the illicit drug shown below.
 Multiple Choice Questions: 50 points 1. Select the correct number of chirality centers in heroin, the illicit drug shown below. A 4 B 5 C 6 D 7 E 8 F more than 8 2. Choose the correct IUPAC name for the
Multiple Choice Questions: 50 points 1. Select the correct number of chirality centers in heroin, the illicit drug shown below. A 4 B 5 C 6 D 7 E 8 F more than 8 2. Choose the correct IUPAC name for the
Name. Department of Chemistry SUNY/Oneonta. Chem Organic Chemistry I. Examination #3 - November 8, 2004 ANSWERS
 Name Department of hemistry SUNY/neonta hem 221 - rganic hemistry I Examination #3 - November 8, 2004 ANSWERS INSTRUTINS --- This examination has two parts. The first part is in multiple choice format;
Name Department of hemistry SUNY/neonta hem 221 - rganic hemistry I Examination #3 - November 8, 2004 ANSWERS INSTRUTINS --- This examination has two parts. The first part is in multiple choice format;
CHE 321 Summer 2010 Exam 2 Form Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III
 CE 321 Summer 2010 Exam 2 orm 2 Multiple Choice Questions: 60 points 1. Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III (A) I only (B) II only (C) I and II only II
CE 321 Summer 2010 Exam 2 orm 2 Multiple Choice Questions: 60 points 1. Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III (A) I only (B) II only (C) I and II only II
Chemistry 232 PRACTICE Midterm 3 October 2010 Your name: 1
 Chemistry 232 PRACTICE Midterm 3 October 2010 Your name: 1 You will need to be able to show picture ID to take the test. DO NOT OPEN TIS TEST UNTIL EVERYONE AS ONE I encourage following instructions: ten
Chemistry 232 PRACTICE Midterm 3 October 2010 Your name: 1 You will need to be able to show picture ID to take the test. DO NOT OPEN TIS TEST UNTIL EVERYONE AS ONE I encourage following instructions: ten
Chemistry 2321 OLD TEST QUESTIONS CH 3
 Test No. 2 hemistry 2321 LD TEST QUESTNS Professor M. Pomerantz 1. n the following reaction the electrophile is: 3 3 + the group none of these 2. Given the reaction shown and the bond dissociation energies
Test No. 2 hemistry 2321 LD TEST QUESTNS Professor M. Pomerantz 1. n the following reaction the electrophile is: 3 3 + the group none of these 2. Given the reaction shown and the bond dissociation energies
Chemistry 143 Exam #2 November 15, Name: Student Number: Section Number: TA: INSTRUCTIONS:
 Chemistry 143 Exam #2 November 15, 2017 Name: Student Number: Section Number: TA: INSTRUCTINS: This exam consists of 32 questions on 9 pages. Please make certain that your exam is complete. Write your
Chemistry 143 Exam #2 November 15, 2017 Name: Student Number: Section Number: TA: INSTRUCTINS: This exam consists of 32 questions on 9 pages. Please make certain that your exam is complete. Write your
PLEASE DO NOT OPEN THIS EXAM UNTIL YOU ARE INSTRUCTED TO DO SO.
 CEMISTRY 0 :5 AM Section EXAM 2 Nov 2009 Name: Note: Your exam should consist of 5 pages including the cover page and grade tabulation sheet. Skim the entire exam, and solve the easiest problems first.
CEMISTRY 0 :5 AM Section EXAM 2 Nov 2009 Name: Note: Your exam should consist of 5 pages including the cover page and grade tabulation sheet. Skim the entire exam, and solve the easiest problems first.
CHEM 3311 Exam 2 ANSWER KEY
 CEM 3311 Exam 2 ANSWER KEY March 11, 2014 Time: 2 ours Please sign the onor Pledge h in this sheet with your scantron. I pledge that n my honor, as a University of Colorado-Boulder student, I have neither
CEM 3311 Exam 2 ANSWER KEY March 11, 2014 Time: 2 ours Please sign the onor Pledge h in this sheet with your scantron. I pledge that n my honor, as a University of Colorado-Boulder student, I have neither
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams.
 2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
CHEMISTRY 341. Final Exam Tuesday, December 16, Problem 1 15 pts Problem 9 8 pts. Problem 2 5 pts Problem pts
 CEMISTRY 341 Final Exam Tuesday, December 16, 1997 Name NAID Problem 1 15 pts Problem 9 8 pts Problem 2 5 pts Problem 10 21 pts Problem 3 26 pts Problem 11 15 pts Problem 4 10 pts Problem 12 6 pts Problem
CEMISTRY 341 Final Exam Tuesday, December 16, 1997 Name NAID Problem 1 15 pts Problem 9 8 pts Problem 2 5 pts Problem 10 21 pts Problem 3 26 pts Problem 11 15 pts Problem 4 10 pts Problem 12 6 pts Problem
Final Exam Professor R. Hoenigman
 I pledge to uphold the CU onor Code: CEM 3311-200 Fall 2006 Final Exam Professor. oenigman Average Score = 145 igh Score = 235 Low Score = 27 Signature Name (printed) Last four digits of your student ID
I pledge to uphold the CU onor Code: CEM 3311-200 Fall 2006 Final Exam Professor. oenigman Average Score = 145 igh Score = 235 Low Score = 27 Signature Name (printed) Last four digits of your student ID
CHEM 2312 practice final. Version - II
 EM 2312 practice final Version - II The following standardized final examination covers the entire introductory year of organic chemistry (EM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
EM 2312 practice final Version - II The following standardized final examination covers the entire introductory year of organic chemistry (EM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
CHEM 2311 HW1. COMPLETELY FILL THE BOXES IN DARK PENCIL, i.e.:, NOT or or STRUCTURE
 EM 2311 W1 Provide answers to the following multiple choice questions on the Scantron form provided in lecture. Make sure that you put your name and bubble in your GTID # on the answer form. MPLETELY FILL
EM 2311 W1 Provide answers to the following multiple choice questions on the Scantron form provided in lecture. Make sure that you put your name and bubble in your GTID # on the answer form. MPLETELY FILL
diene. If stereoisomeric mixtures form, indicate by drawing one and writing "+ enantiomer" and/or "+ diastereomer" in the box.
 I. (9 points) Page A. Consider how varying the conditions of a reaction can vastly influence the rate of a reaction as well as the product(s) formed. First draw the product(s) that form in the reference
I. (9 points) Page A. Consider how varying the conditions of a reaction can vastly influence the rate of a reaction as well as the product(s) formed. First draw the product(s) that form in the reference
Department of Chemistry SUNY/Oneonta. Chem Organic Chemistry I. Examination #2 - October 18, 2004 ANSWERS
 Department of hemistry SUNY/Oneonta hem 221 - Organic hemistry I Examination #2 - October 18 2004 ANSWERS INSTRUTIONS This examination is in multiple choice format; the questions are in this Exam Booklet
Department of hemistry SUNY/Oneonta hem 221 - Organic hemistry I Examination #2 - October 18 2004 ANSWERS INSTRUTIONS This examination is in multiple choice format; the questions are in this Exam Booklet
10:30 AM 1:00 PM December 13, 2016 in MATH 100
 CEM 3311 ARRIGT Exam 4 10:30 AM 1:00 PM December 13, 2016 in MAT 100 Instructions. o notes, books, laptops, phones, calculators, models, or stencils are allowed. Periodic Table, Electronegativity Chart,
CEM 3311 ARRIGT Exam 4 10:30 AM 1:00 PM December 13, 2016 in MAT 100 Instructions. o notes, books, laptops, phones, calculators, models, or stencils are allowed. Periodic Table, Electronegativity Chart,
Solutions. Department of Chemistry SUNY/Oneonta. Chem Organic Chemistry I. Examination #2 - October 23, 2000
 Solutions Department of hemistry SUNY/Oneonta hem 221 - Organic hemistry I Examination #2 - October 23, 2000 INSTRUTIONS --- This examination has two parts. The first part is in multiple choice format;
Solutions Department of hemistry SUNY/Oneonta hem 221 - Organic hemistry I Examination #2 - October 23, 2000 INSTRUTIONS --- This examination has two parts. The first part is in multiple choice format;
CHEM 241 ALKYNES CHAP 9 ASSIGN
 HEM 241 ALKYNES HAP 9 ASSIGN 1. What is the IUPA name of the following compound A. 5-propyl-3-heptyne B. 5-isopropyl-3-heptyne. 5-ethyl-3-octyne D. 4-ethyl-5-octyne 2. What is the correct IUPA name for
HEM 241 ALKYNES HAP 9 ASSIGN 1. What is the IUPA name of the following compound A. 5-propyl-3-heptyne B. 5-isopropyl-3-heptyne. 5-ethyl-3-octyne D. 4-ethyl-5-octyne 2. What is the correct IUPA name for
CHEMISTRY FINAL EXAM June 25, 2005
 CEMISTRY 313-61 FINAL EXAM June 25, 2005 Name... The total number of points is 100. The total exam time is 120 min (2 h). Good luck! PART I: CNCEPTS 1. (10 pts) Mark as true (T) or false (F) the following
CEMISTRY 313-61 FINAL EXAM June 25, 2005 Name... The total number of points is 100. The total exam time is 120 min (2 h). Good luck! PART I: CNCEPTS 1. (10 pts) Mark as true (T) or false (F) the following
1. What is the letter designation given to dumbbell shaped orbitals like the one depicted below?
 1. What is the letter designation given to dumbbell shaped orbitals like the one depicted below? 2. Which of the following does not have an octet of electrons surrounding the central atom? A. B 3 B. C
1. What is the letter designation given to dumbbell shaped orbitals like the one depicted below? 2. Which of the following does not have an octet of electrons surrounding the central atom? A. B 3 B. C
Identification Sticker
 Chemistry 1A Midterm Exam 1 September 20, 2001 Page 1 of 10. (Closed Book, 90 minutes, 150 points) Version D Name: TA: SID: Section: Identification Sticker Test-taking strategy: PLEASE READ THIS FIRST!
Chemistry 1A Midterm Exam 1 September 20, 2001 Page 1 of 10. (Closed Book, 90 minutes, 150 points) Version D Name: TA: SID: Section: Identification Sticker Test-taking strategy: PLEASE READ THIS FIRST!
CHE1502. Tutorial letter 201/1/2016. General Chemistry 1B. Semester 1. Department of Chemistry CHE1502/201/1/2016
 CE1502/201/1/2016 Tutorial letter 201/1/2016 General Chemistry 1B CE1502 Semester 1 Department of Chemistry This tutorial letter contains the answers to the questions in assignment 1. FIRST SEMESTER: KEY
CE1502/201/1/2016 Tutorial letter 201/1/2016 General Chemistry 1B CE1502 Semester 1 Department of Chemistry This tutorial letter contains the answers to the questions in assignment 1. FIRST SEMESTER: KEY
SECTION-I (SINGLE CORRECT CHOICE)
 SECTI-I (SIGLE CRRECT CICE) 1. Which of the following phrases are not correctly associated with an S1 reaction? 1. Rearrangement is possible. 2. Rate is affected by solvent polarity. 3. The strength of
SECTI-I (SIGLE CRRECT CICE) 1. Which of the following phrases are not correctly associated with an S1 reaction? 1. Rearrangement is possible. 2. Rate is affected by solvent polarity. 3. The strength of
Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat #
 Test 4 Name Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat # Part 1. Multiple Choice. Choose the one best answer, and clearly mark that answer
Test 4 Name Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat # Part 1. Multiple Choice. Choose the one best answer, and clearly mark that answer
Your course TA (Steve, Jenn-C, Jenn-P):
 CHM 1321 Second Midterm April 3, 2008 1 Your Name: Student #: Your course TA (Steve, Jenn-C, Jenn-P): Exercise 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 key 1. Deliver only the solution
CHM 1321 Second Midterm April 3, 2008 1 Your Name: Student #: Your course TA (Steve, Jenn-C, Jenn-P): Exercise 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 key 1. Deliver only the solution
Chemistry 2321 OLD TEST QUESTIONS
 hemistry 2321 Test No. 3 Professor M. Pomerantz LD TEST QUESTINS Select the best name for the compounds shown. 1. l 2 3 2 2-chloro-4-ethyl-5-heptyne 6-chloro-4-ethyl-2-heptyne 4-(2-chloropropyl)-2-hexyne
hemistry 2321 Test No. 3 Professor M. Pomerantz LD TEST QUESTINS Select the best name for the compounds shown. 1. l 2 3 2 2-chloro-4-ethyl-5-heptyne 6-chloro-4-ethyl-2-heptyne 4-(2-chloropropyl)-2-hexyne
1. (6 points) Provide IUPAC accepted names for the following compounds. 2. (6 points) Provide a structure for the following compounds.
 Chem52 omework Set 1 This homework set is similar to a Chem 51 final exam. Please provide answers in the spaces provided or, preferably, on attached sheets. Point values are listed only to give you the
Chem52 omework Set 1 This homework set is similar to a Chem 51 final exam. Please provide answers in the spaces provided or, preferably, on attached sheets. Point values are listed only to give you the
trans-cyclooctene 4-fluoro-1-butanol 2-cyclohexenol 4. (5 pts) Provide structural formula for each of the following molecules: Br OH Cl OH
 CEMISTRY 313-01 MIDTERM # 2 answer key March 12, 2009 Statistics: Average: 72 pts (72%); ighest: 98 pts (98%); Lowest: 21 pts (21%) umber of students performing at or above average: 22 (50%) umber of students
CEMISTRY 313-01 MIDTERM # 2 answer key March 12, 2009 Statistics: Average: 72 pts (72%); ighest: 98 pts (98%); Lowest: 21 pts (21%) umber of students performing at or above average: 22 (50%) umber of students
THE UNIVERSITY OF CALGARY FACULTY OF SCIENCE FINAL EXAMINATION CHEMISTRY 351 READ ALL THE INSTRUCTIONS CAREFULLY
 TE UNIVERSITY F CALGARY FACULTY F SCIENCE FINAL EXAMINATIN CEMISTRY 351 DECEMBER 22nd 1999 Time: 3 ours READ ALL TE INSTRUCTINS CAREFULLY PLEASE WRITE YUR NAME, STUDENT I.D. NUMBER N BT YUR EXAM ANSWER
TE UNIVERSITY F CALGARY FACULTY F SCIENCE FINAL EXAMINATIN CEMISTRY 351 DECEMBER 22nd 1999 Time: 3 ours READ ALL TE INSTRUCTINS CAREFULLY PLEASE WRITE YUR NAME, STUDENT I.D. NUMBER N BT YUR EXAM ANSWER
- NH2 or NH 3 H 2 O or CH 3 COO - BF 3 or F - I Br. or Br
 CEMSTRY 313-02 MDTERM # 3 answer key November 09, 2004 Statistics: Average: 71 pts (71%); ighest: 90 pts (90%); Lowest: 49 pts (49%) Number of students perfming at above average: 22 (51%) 1. (8 pts) Mark
CEMSTRY 313-02 MDTERM # 3 answer key November 09, 2004 Statistics: Average: 71 pts (71%); ighest: 90 pts (90%); Lowest: 49 pts (49%) Number of students perfming at above average: 22 (51%) 1. (8 pts) Mark
ALKANES STRUCTURE, PROPERTIES, AND SYNTHESIS A STUDENT WHO HAS MASTERED THE MATERIAL IN THIS SECTION SHOULD BE ABLE TO:
 ALKANES STRUCTURE, PROPERTIES, AND SYNTESIS A STUDENT WO AS MASTERED TE MATERIAL IN TIS SECTION SOULD BE ABLE TO: 1. Predict relative boiling points of alkanes, in comparison with other alkanes and with
ALKANES STRUCTURE, PROPERTIES, AND SYNTESIS A STUDENT WO AS MASTERED TE MATERIAL IN TIS SECTION SOULD BE ABLE TO: 1. Predict relative boiling points of alkanes, in comparison with other alkanes and with
1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound?
 CEM 331: Chapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N C 2 C N C 2 C N 1 2 3 4 1: three sigma bonds and
CEM 331: Chapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N C 2 C N C 2 C N 1 2 3 4 1: three sigma bonds and
Organic Chemistry 1 CHM 2210 Exam 3 (November 9, 2001)
 Organic Chemistry 1 CM 2210 Exam 3 (November 9, 2001) Name (print): Signature: Student ID Number: There are 14 multiple choice problems (5 points each) on this exam, however, you will only be graded out
Organic Chemistry 1 CM 2210 Exam 3 (November 9, 2001) Name (print): Signature: Student ID Number: There are 14 multiple choice problems (5 points each) on this exam, however, you will only be graded out
Chem 1120 Midterm points Dr. Luther Giddings
 Chem 1120 Midterm 1 100 points Dr. Luther Giddings Name Instructions: This is a closed book, closed notebook test. You may not discuss this exam with anyone, either during or after the exam, until it has
Chem 1120 Midterm 1 100 points Dr. Luther Giddings Name Instructions: This is a closed book, closed notebook test. You may not discuss this exam with anyone, either during or after the exam, until it has
CHE 275 REACTIONS OF ALKENES: ADDITION REACTIONS CHAP 8 ASSIGN
 HE 275 REATINS F ALKENES: ADDITIN REATINS HAP 8 ASSIGN 1. Which reagent or test could be used to distinguish between 2-methyl-2-pentene and 2-methylpentane? A. Br 2/l 4 B. KMn 4,. oncd. H 2S 4 D. Two of
HE 275 REATINS F ALKENES: ADDITIN REATINS HAP 8 ASSIGN 1. Which reagent or test could be used to distinguish between 2-methyl-2-pentene and 2-methylpentane? A. Br 2/l 4 B. KMn 4,. oncd. H 2S 4 D. Two of
Department of Chemistry SUNY/Oneonta. Chem Organic Chemistry I. Examination #2 - October 14, 2002
 Department of hemistry SUNY/Oneonta hem 221 - Organic hemistry Examination #2 - October 14, 2002 ANSWERS NSTRUTONS This examination is in multiple choice format; the questions are in this Exam Booklet
Department of hemistry SUNY/Oneonta hem 221 - Organic hemistry Examination #2 - October 14, 2002 ANSWERS NSTRUTONS This examination is in multiple choice format; the questions are in this Exam Booklet
B. (2 pts) The regiochemistry of alcohol dehydration is determined by the: a. Zaitsev rule; b. Hammond postulate;
 CEMISTRY 313-01 MIDTERM # answer key ctober 1, 008 Statistics: Average: 73 pts (73%); ighest: 98 pts (98%); Lowest: 3 pts (3%) Number of students performing at or above average: 36 (58%) Number of students
CEMISTRY 313-01 MIDTERM # answer key ctober 1, 008 Statistics: Average: 73 pts (73%); ighest: 98 pts (98%); Lowest: 3 pts (3%) Number of students performing at or above average: 36 (58%) Number of students
2. Which functional groups and structural features are present in the following molecule (strychnine)?
 Chapter 1-2: 1. Which of the following species has a negative charge but NO lone pair of valance shell nonbonding electrons? [all atoms have complete valance shell of electrons, but lone pairs are not
Chapter 1-2: 1. Which of the following species has a negative charge but NO lone pair of valance shell nonbonding electrons? [all atoms have complete valance shell of electrons, but lone pairs are not
UNIVERSITY OF SWAZILAND FINAL EXAMINATION 2013, DECEMBER
 UNIVERSITY OF SWAZILAND FINAL EXAMINATION 2013, DECEMBER TITLE OF PAPER COURSE NUMBER Introductory Organic Chemistry C203 TIME Three Hours INSTRUCTIONS Section A is compulsory. Answer any three questions
UNIVERSITY OF SWAZILAND FINAL EXAMINATION 2013, DECEMBER TITLE OF PAPER COURSE NUMBER Introductory Organic Chemistry C203 TIME Three Hours INSTRUCTIONS Section A is compulsory. Answer any three questions
FALL 2018 CEM 251: Organic Chemistry I Sections September 25, 2018
 FALL 2018 CEM 251: Organic Chemistry Sections 25-36 TUE-TU 8:00 9:20 AM September 25, 2018 Name: Section: PD: TA: This is a closed book and note examination. f boxes are provided for your answers, only
FALL 2018 CEM 251: Organic Chemistry Sections 25-36 TUE-TU 8:00 9:20 AM September 25, 2018 Name: Section: PD: TA: This is a closed book and note examination. f boxes are provided for your answers, only
1) (100 pts) 5) (20 pts) 3) (35 pts) 4) (25pts. Total (200 pts) More Tutorial at
 Name: Perm number: Question 1) (100 pts) 2) (20 pts) 3) (35 pts) 4) (25pts 5) (20 pts) Total (200 pts) Your score 1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers
Name: Perm number: Question 1) (100 pts) 2) (20 pts) 3) (35 pts) 4) (25pts 5) (20 pts) Total (200 pts) Your score 1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers
tbuo OtBu Br 1) B 2 H 6 /THF OH 2) OH - + H 2 O 2 NaNH 2 NH3 Na NH 3 /-35 C CH 3 CCH 2 CHCH 3
 EM 12A Name KEY EXAM II all 2003 1.(16 pts) Draw the structure of the major product expected from each of the following sets of reactants. Specify stereochemistry where appropriate. + 2 Pt + 2 2 2 + enantiomer
EM 12A Name KEY EXAM II all 2003 1.(16 pts) Draw the structure of the major product expected from each of the following sets of reactants. Specify stereochemistry where appropriate. + 2 Pt + 2 2 2 + enantiomer
Homework Problem Set 9 Iverson CH320M/328M Due Friday, November 30
 omework Problem Set 9 Iverson C0M/8M Due Friday, November 0 NAME (Print): SIGNATURE: Chemistry 0M/8M Dr. ent Iverson 9th omework November 9, 08 Please print the first three letters of your last name in
omework Problem Set 9 Iverson C0M/8M Due Friday, November 0 NAME (Print): SIGNATURE: Chemistry 0M/8M Dr. ent Iverson 9th omework November 9, 08 Please print the first three letters of your last name in
a) 1. O 3 2. (CH 3 ) 2 S
 Name: 1 Chemistry 250B Final Exam December 19, 2008 Show non-zero formal charges on all atoms for all structures. There are 11 pages. 1. (42 pts) Complete the following reactions. Show the stereochemistry
Name: 1 Chemistry 250B Final Exam December 19, 2008 Show non-zero formal charges on all atoms for all structures. There are 11 pages. 1. (42 pts) Complete the following reactions. Show the stereochemistry
Study Union: CHM 2210 Final Exam Review 1. The following molecules are related in what manner?
 Study Union: CHM 2210 Final Exam Review 1. The following molecules are related in what manner? A) They are isotopes B) They are constitutional isomers C) They are the same structure D) They are composed
Study Union: CHM 2210 Final Exam Review 1. The following molecules are related in what manner? A) They are isotopes B) They are constitutional isomers C) They are the same structure D) They are composed
Partial Periodic Table
 CHEM 33-00, Fall 03 Professor Walba Third Hour Exam November 9, 03 scores: ) ) 3) 4) 5) CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither given nor received
CHEM 33-00, Fall 03 Professor Walba Third Hour Exam November 9, 03 scores: ) ) 3) 4) 5) CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither given nor received
CHEMISTRY MIDTERM # 2 October 27, The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck!
 EMISTRY 313-01 MIDTERM # 2 ctober 27, 2005 Name... The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck! 1. (7 pts) Mark as true (T) or false (F) the following
EMISTRY 313-01 MIDTERM # 2 ctober 27, 2005 Name... The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck! 1. (7 pts) Mark as true (T) or false (F) the following
Partial Periodic Table
 Easily Legible Printed Name: CEM 3351 (100), Fall 2015 Professor Walba First our Exam September 22, 2015 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither
Easily Legible Printed Name: CEM 3351 (100), Fall 2015 Professor Walba First our Exam September 22, 2015 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither
Chemistry 233 Exam 3 (Green) The Periodic Table
 Name: Last First MI Chemistry Exam (Green) Summer 7 Dr. J. sbourn Instructions: The first questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
Name: Last First MI Chemistry Exam (Green) Summer 7 Dr. J. sbourn Instructions: The first questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2
 C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
Part C- section 1 p-bonds as nucleophiles
 Part C- section 1 p-bonds as nucleophiles Chemistry of Alkenes (Ch 8, 9, 10) - the double bond prevents free rotation - isomerism cis and trans - nomenclature E and Z (3 or 4 different substituents around
Part C- section 1 p-bonds as nucleophiles Chemistry of Alkenes (Ch 8, 9, 10) - the double bond prevents free rotation - isomerism cis and trans - nomenclature E and Z (3 or 4 different substituents around
Chem 2320 Exam 1. January 30, (Please print)
 Chem 2320 Exam 1 January 30, 2006 Name: (first) (last) (Please print) Last 4 digits of I.D. I. Multiple Choice ( /20) Score /60 II /15 III /25 Total score /100 I. Multiple choice questions. (3 points each).
Chem 2320 Exam 1 January 30, 2006 Name: (first) (last) (Please print) Last 4 digits of I.D. I. Multiple Choice ( /20) Score /60 II /15 III /25 Total score /100 I. Multiple choice questions. (3 points each).
Practice Multiple-Choice Questions for Ending Chem 21 and/or Starting Chem 22
 Practice Multiple-Choice Questions for Ending Chem 21 and/or Starting Chem 22 Includes information from chapters 5-10 of the third edition of Klein s Organic Chemistry text. 1. Which of the following compounds
Practice Multiple-Choice Questions for Ending Chem 21 and/or Starting Chem 22 Includes information from chapters 5-10 of the third edition of Klein s Organic Chemistry text. 1. Which of the following compounds
----_._--_._.._._----_.._ _..._----_..._-
 UNIVERSITY OF SWAZILAND FINAL EXAMINATION 2016, DECEMBER..-.._._ TITLE OF PAPER Functional group chemistry & stereochemistry or Introductory Organic Chemistry COURSE NUMBER CE 232/ C 203 TIME Three ours
UNIVERSITY OF SWAZILAND FINAL EXAMINATION 2016, DECEMBER..-.._._ TITLE OF PAPER Functional group chemistry & stereochemistry or Introductory Organic Chemistry COURSE NUMBER CE 232/ C 203 TIME Three ours
CEM 351 2nd EXAM/Version A Friday, October 17, :50 2:40 p.m. Room 138, Chemistry
 Name (print) Signature Student # Section Number CEM 351 2nd EXAM/Version A Friday, October 17, 2003 1:50 2:40 p.m. Room 138, Chemistry Ann Swerkey Grade? 1.(20 2.(20. 3.(20 4.(20 5.(20 6.(20 TOTAL 100
Name (print) Signature Student # Section Number CEM 351 2nd EXAM/Version A Friday, October 17, 2003 1:50 2:40 p.m. Room 138, Chemistry Ann Swerkey Grade? 1.(20 2.(20. 3.(20 4.(20 5.(20 6.(20 TOTAL 100
FALL 2018 CEM 251: Organic Chemistry I Sections September 25, 2018
 ALL 2018 CEM 251: rganic Chemistry Sections 25-36 TUE-TU 8:00 9:20 AM September 25, 2018 Name: Section: PD: TA: This is a closed book and note examination. f boxes are provided for your answers, only what
ALL 2018 CEM 251: rganic Chemistry Sections 25-36 TUE-TU 8:00 9:20 AM September 25, 2018 Name: Section: PD: TA: This is a closed book and note examination. f boxes are provided for your answers, only what
EXAMINATION 2 Chemistry 3A
 1 EXAMINATIN 2 Chemistry 3A Name: KEY Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
1 EXAMINATIN 2 Chemistry 3A Name: KEY Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound?
 EM 331: hapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N 2 N 2 N 1 2 3 4 2. What hybrid orbitals are used to
EM 331: hapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N 2 N 2 N 1 2 3 4 2. What hybrid orbitals are used to
H C C C C H C H C C H H. Cl H CH 3 O. Br CH 3 OH H 3 C. H Br H
 CEMISTRY 313-61 MITERM # 3 answer key June 17, 2005 Statistics: Average: 69 pts (69%); ighest: 96 pts (96%); Lowest: 46 pts (46%) Number of students perfming at above average: 16 (53%) 1. (10 pts) Mark
CEMISTRY 313-61 MITERM # 3 answer key June 17, 2005 Statistics: Average: 69 pts (69%); ighest: 96 pts (96%); Lowest: 46 pts (46%) Number of students perfming at above average: 16 (53%) 1. (10 pts) Mark
3. (6 pts) Provide an acceptable name for each of the following molecules: OH
 CEMISTRY 313-61 MIDTERM # 2 answer key June 07, 2005 Statistics: Average: 71 pts (71%); ighest: 99 pts (99%); Lowest: 45 pts (45%) Number of students performing at or above average: 15 (48%) 1. (11 pts)
CEMISTRY 313-61 MIDTERM # 2 answer key June 07, 2005 Statistics: Average: 71 pts (71%); ighest: 99 pts (99%); Lowest: 45 pts (45%) Number of students performing at or above average: 15 (48%) 1. (11 pts)
Columbia University C99ORG12.DOC S3443D Summer 99 Professor Grace B. Borowitz Exam No. 2 June 14, 1999
 olumbia University 99RG12.D SD Summer 99 Professor Grace B. Borowitz Exam No. 2 June 1, 1999 Name: Sterie hemistry Grade: Please use a non-red pen. Answer questions in the provided space. If you write
olumbia University 99RG12.D SD Summer 99 Professor Grace B. Borowitz Exam No. 2 June 1, 1999 Name: Sterie hemistry Grade: Please use a non-red pen. Answer questions in the provided space. If you write
Exam 3 November 17, 2005 Professor Rebecca Hoenigman
 EM 3311-200, Fall 2005 Exam 3 November 17, 2005 Professor Rebecca oenigman Average Score = 54.8 igh Score = 98 Low Score = 8 I pledge to uphold the U onor ode: Signature Name (printed) Last four digits
EM 3311-200, Fall 2005 Exam 3 November 17, 2005 Professor Rebecca oenigman Average Score = 54.8 igh Score = 98 Low Score = 8 I pledge to uphold the U onor ode: Signature Name (printed) Last four digits
C h a p t e r E i g h t: Alkenes: Structure and Preparation via Elimination Reactions. 5-Androstene, the parent alkene for most anabolic steroids
 C h a p t e r E i g h t: Alkenes: Structure and Preparation via Elimination Reactions 5-Androstene, the parent alkene for most anabolic steroids CHM 321: Summary of Important Concepts YConcepts for Chapter
C h a p t e r E i g h t: Alkenes: Structure and Preparation via Elimination Reactions 5-Androstene, the parent alkene for most anabolic steroids CHM 321: Summary of Important Concepts YConcepts for Chapter
Study Union: CHM 2210 Final Exam Review 1. The following molecules are related in what manner?
 Study Union: CHM 2210 Final Exam Review 1. The following molecules are related in what manner? A) They are isotopes B) They are constitutional isomers C) They are the same structure D) They are composed
Study Union: CHM 2210 Final Exam Review 1. The following molecules are related in what manner? A) They are isotopes B) They are constitutional isomers C) They are the same structure D) They are composed
CHEM Exam 1
 CEM 3311-100 Exam 1 February 12, 2013 Time: 2 ours By printing my name and signing on this cover page, I pledge that On my honor, as a University of Colorado-Boulder student, I have neither given nor received
CEM 3311-100 Exam 1 February 12, 2013 Time: 2 ours By printing my name and signing on this cover page, I pledge that On my honor, as a University of Colorado-Boulder student, I have neither given nor received
10:30 AM 1:00 PM December 13, 2016 in MATH 100
 CEM 3311 ARRIGT Exam 4 KEY 10:30 AM 1:00 PM December 13, 016 in MAT 100 Instructions. o notes, books, laptops, phones, calculators, models, or stencils are allowed. Periodic Table, Electronegativity Chart,
CEM 3311 ARRIGT Exam 4 KEY 10:30 AM 1:00 PM December 13, 016 in MAT 100 Instructions. o notes, books, laptops, phones, calculators, models, or stencils are allowed. Periodic Table, Electronegativity Chart,
More Tutorial at
 1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question (50 pts). 1) Which of the following statements about propene, CH3CH CH2, is 1) correct? A) There
1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question (50 pts). 1) Which of the following statements about propene, CH3CH CH2, is 1) correct? A) There
Chemistry 123: Physical and Organic Chemistry Topic 1: Organic Chemistry
 Concept Check: Topic 1: Conformation Winter 2009 Page 112 Concept Check: Topic 1: Conformation Winter 2009 Page 113 1 STEREOCHEMISTRY Winter 2009 Page 114 We have already covered two kinds of isomerism:
Concept Check: Topic 1: Conformation Winter 2009 Page 112 Concept Check: Topic 1: Conformation Winter 2009 Page 113 1 STEREOCHEMISTRY Winter 2009 Page 114 We have already covered two kinds of isomerism:
Final Exam CHM1321-B. Date: April 19 Length: 3 hrs Last Name: Professor Sandro Gambarotta Name:
 Faculté des sciences Faculty of Science Département de chimie et science biomoléculaire Department of Chemistry and Biomolecular Sciences Pavillon d Iorio Hall 10 Marie-Curie Ottawa ON Canada K1N 6N5 '
Faculté des sciences Faculty of Science Département de chimie et science biomoléculaire Department of Chemistry and Biomolecular Sciences Pavillon d Iorio Hall 10 Marie-Curie Ottawa ON Canada K1N 6N5 '
1. Name the following two compounds [8 pts]. Where it is important, points are explicitly allocated for stereochemistry.
![1. Name the following two compounds [8 pts]. Where it is important, points are explicitly allocated for stereochemistry. 1. Name the following two compounds [8 pts]. Where it is important, points are explicitly allocated for stereochemistry.](/thumbs/74/70778069.jpg) 1. Name the following two compounds [8 pts]. Where it is important, points are explicitly allocated for stereochemistry. 2. raw the structures of the following two compounds [8 pts]. cis-1-bromo-2,2-dimethyl-4-octene.
1. Name the following two compounds [8 pts]. Where it is important, points are explicitly allocated for stereochemistry. 2. raw the structures of the following two compounds [8 pts]. cis-1-bromo-2,2-dimethyl-4-octene.
CHEM 2423 Unit 8 Homework Answers
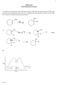 1 CEM 2423 Unit 8 omework Answers 1. Show the mechanism of the following reaction. Label the rate-limiting step. Draw the potential energy diagram for this reaction. (c) Explain the regiochemistry of this
1 CEM 2423 Unit 8 omework Answers 1. Show the mechanism of the following reaction. Label the rate-limiting step. Draw the potential energy diagram for this reaction. (c) Explain the regiochemistry of this
Chapter Organic Chemistry Basics
 29 Chapter Organic Chemistry Basics 2. A similarity between optical and geometrical isomerism is that [2002] (a) each forms equal number of isomers for a given compound (b) if in a compound one is present
29 Chapter Organic Chemistry Basics 2. A similarity between optical and geometrical isomerism is that [2002] (a) each forms equal number of isomers for a given compound (b) if in a compound one is present
STEREOCHEMISTRY A STUDENT SHOULD BE ABLE TO:
 A STUDENT SHOULD BE ABLE TO: STEREOCHEMISTRY 1. Determine the relationship between two given structures (which may be any of the kinds below). Also, define the following terms, and give examples of pairs
A STUDENT SHOULD BE ABLE TO: STEREOCHEMISTRY 1. Determine the relationship between two given structures (which may be any of the kinds below). Also, define the following terms, and give examples of pairs
CHEMISTRY 332 FINAL EXAM. December 14, I. (18 points) II. (48 points) III. (50 points) IV. (40 points) V. (26 points) VI.
 Seat No. Name (please print and circle your last name) CEMISTRY 332 FINAL EXAM December 14, 2005 I. (18 points) II. (48 points) III. (50 points) IV. (40 points) V. (26 points) VI. (18 points) TTAL (200
Seat No. Name (please print and circle your last name) CEMISTRY 332 FINAL EXAM December 14, 2005 I. (18 points) II. (48 points) III. (50 points) IV. (40 points) V. (26 points) VI. (18 points) TTAL (200
H C C C C H C H C C H OH. Cl H CH 3. Br CH 3. H Br H OH H 3 C. CHEMISTRY MIDTERM # 3 June 17, Name...
 Name... CEMISTRY 313-61 MIDTERM # 3 June 17, 2005 The total number of points in this midterm is 100. The total exam time is 90 min (1.5 h). Good luck! 1. (10 pts) Mark as true (T) false () the following
Name... CEMISTRY 313-61 MIDTERM # 3 June 17, 2005 The total number of points in this midterm is 100. The total exam time is 90 min (1.5 h). Good luck! 1. (10 pts) Mark as true (T) false () the following
FINAL EXAMINATION Chemistry 3A
 1 FINAL EXAMINATION Chemistry 3A Key Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt May 11, 2016 GSI (if you are taking Chem 3AL): Please provide the following information
1 FINAL EXAMINATION Chemistry 3A Key Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt May 11, 2016 GSI (if you are taking Chem 3AL): Please provide the following information
CH 3 C 2 H 5. Tetrahedral Stereochemistry
 Ch 5 Tetrahedral Stereochemistry Enantiomers - Two non-superimposable mirror image molecules - They are stereoisomers with the same atoms and bonds, but different spatial geometries. - The two molecules
Ch 5 Tetrahedral Stereochemistry Enantiomers - Two non-superimposable mirror image molecules - They are stereoisomers with the same atoms and bonds, but different spatial geometries. - The two molecules
CHEM1902/ N-9 November 2014
 CEM1902/4 2014-N-9 November 2014 The elimination of 2 O from alcohol A can form the isomeric alkenes B and C. Elimination of Br from the alkyl halide D can generate the same two alkenes. 7 Assign the absolute
CEM1902/4 2014-N-9 November 2014 The elimination of 2 O from alcohol A can form the isomeric alkenes B and C. Elimination of Br from the alkyl halide D can generate the same two alkenes. 7 Assign the absolute
Chemistry Exam 2. The Periodic Table
 Name: Last First MI Chemistry 234-002 Exam 2 Spring 2017 Dr. J. sbourn Instructions: The first 18 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in
Name: Last First MI Chemistry 234-002 Exam 2 Spring 2017 Dr. J. sbourn Instructions: The first 18 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in
CHEM 241 ALKANES AND CYCLOALKANES CHAP 3 ASSIGN H H
 CEM 241 ALKANES AND CYCLOALKANES CAP 3 ASSIGN COMFORMATIONS AND cis-trans STEREOISOMERS 1. trans-1,2-dibromocyclohexane is represented by structure(s): D. II and III E. I and II 2. cis-1,3-dibromocyclohexane
CEM 241 ALKANES AND CYCLOALKANES CAP 3 ASSIGN COMFORMATIONS AND cis-trans STEREOISOMERS 1. trans-1,2-dibromocyclohexane is represented by structure(s): D. II and III E. I and II 2. cis-1,3-dibromocyclohexane
CHEM 2312 practice final. Version I
 CEM 2312 practice final Version The following standardized final examination covers the entire introductory year of organic chemistry (CEM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
CEM 2312 practice final Version The following standardized final examination covers the entire introductory year of organic chemistry (CEM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
Chemistry 201. MW 12:00pm 1:15pm Examination #2 August 15 th Bronco ID. Question Score Possible Points. 1 (12pts) 2 (24pts) 3 (25pts)
 Chemistry 201 MW 12:00pm 1:15pm Examination #2 August 15 th 2016 Name Bronco ID. Question Score Possible Points 1 (12pts) 2 (24pts) 3 (25pts) 4... (12pts) 5 (27pts). Total (100pts) 1. Read each question
Chemistry 201 MW 12:00pm 1:15pm Examination #2 August 15 th 2016 Name Bronco ID. Question Score Possible Points 1 (12pts) 2 (24pts) 3 (25pts) 4... (12pts) 5 (27pts). Total (100pts) 1. Read each question
FOURTH EXAMINATION. (1)..20 pts... (2)..24 pts... (3)..18 pts... (4)..12 pts... (5)..12 pts... (6).. 6 pts... (7).. 8 pts... Bonus 4 pts...
 Name: CHEM 331 FURTH EXAMINATIN All answers should be written on the exam in the spaces provided. Clearly indicate your answers in the spaces provided; if I have to guess as to what or where your answer
Name: CHEM 331 FURTH EXAMINATIN All answers should be written on the exam in the spaces provided. Clearly indicate your answers in the spaces provided; if I have to guess as to what or where your answer
Chemistry 233 Exam 3. The Periodic Table
 Name: Last First MI Chemistry 233 Exam 3 Fall 2017 Dr. J. sbourn Instructions: The first 8 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
Name: Last First MI Chemistry 233 Exam 3 Fall 2017 Dr. J. sbourn Instructions: The first 8 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
Assign (R) or (S) configurations to the chiral carbons in the following molecules: enantiomers
 CAPTER 5: STERECEMISTRY (cont.) Assign (R) or (S) configurations to the chiral carbons in the following molecules: 3 C 3 C N 2 Molecules With Multiple Chiral Atoms. 1-chloro-2-methylcyclohexane has four
CAPTER 5: STERECEMISTRY (cont.) Assign (R) or (S) configurations to the chiral carbons in the following molecules: 3 C 3 C N 2 Molecules With Multiple Chiral Atoms. 1-chloro-2-methylcyclohexane has four
CONJUGATED PI SYSTEMS AND PERICYCLIC REACTIONS
 CNJUGATED PI SYSTEMS AND PERICYCLIC REACTINS A STUDENT SULD BE ABLE T: 1. Recognize and give examples of conjugated, isolated, and cumulated dienes, and of allylic intermediates. Also, name given the structure,
CNJUGATED PI SYSTEMS AND PERICYCLIC REACTINS A STUDENT SULD BE ABLE T: 1. Recognize and give examples of conjugated, isolated, and cumulated dienes, and of allylic intermediates. Also, name given the structure,
