MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
|
|
|
- Alvin Stanley
- 6 years ago
- Views:
Transcription
1 A.P. Chemistry Practice Test - Ch. 7, Atomic Structure and Periodicity Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Ham radio operators often broadcast on the 6-meter band. The frequency of this electromagnetic radiation is MHz. A) 50 B) 20 C) 2.0 D) 200 E) 500 2) What is the frequency of light (s-1) that has a wavelength of 1.23 x 10-6 cm? A) 2.44 x 1016 B) 1.04 x C) 9.62 x 1012 D) 3.69 E) 4.10 x ) The wavelength of a photon that has an energy of 5.25 x J is m. A) 4.21 x B) 2.38 x 1023 C) 2.64 x 106 D) 3.79 x 107 E) 3.79 x ) The frequency of a photon that has an energy of 3.7 x J is s-1. A) 5.4 x 10-8 B) 5.6 x 1015 C) 1.8 x D) 2.5 x E) 2.5 x ) A mole of red photons of wavelength 725 nm has kj of energy. A) 6.05 x 10-3 B) 2.74 x C) 4.56 x D) 227 E) 165 6) Of the following transitions in the Bohr hydrogen atom, the transition results in the emission of the highest-energy photon. A) n = 6 n = 1 B) n = 1 n = 6 C) n = 3 n = 6 D) n = 6 n = 3 E) n = 1 n = 4 7) Using Bohr's equation for the energy levels of the electron in the hydrogen atom, determine the energy (J) of an electron in the n = 4 level. A) x B) x C) x D) x E) x ) The lines in the emission spectrum of hydrogen result from. A) electrons given off by hydrogen as it cools B) decomposing hydrogen atoms C) electrons given off by hydrogen when it burns D) energy given off in the form of visible light when an electron moves from a higher energy state to a lower energy state E) protons given off when hydrogen burns 9) When the electron in a hydrogen atom moves from n = 6 to n = 2, light with a wavelength of nm is emitted. A) 657 B) 93.8 C) 411 D) 434 E) ) What is the De Broglie wavelength (m) of a 2.0 kg object moving at a speed of 50 m/s? A) 1.5 x 1035 B) 3.8 x 1034 C) 2.6 x D) 5.3 x E) 6.6 x
2 11) All of the orbitals in a given electron shell (energy level) have the same value of the quantum number. A) spin B) principal C) azimuthal D) magnetic E) psi 12) All of the orbitals in a given subshell (energy sublevel) have the same value of the quantum number. A) azimuthal B) magnetic C) principal D) A and B E) B and C 13) There are possible values for the magnetic quantum number of an electron in a 5f subshell. A) 7 B) 3 C) 14 D) 5 E) 1 14) Which of the subshells below do not exist due to the constraints upon the azimuthal quantum number? A) 2p B) 2s C) 2d D) all of the above E) none of the above 15) -orbitals are spherically symmetrical. A) d B) g C) p D) f E) s 2
3 16) Which sketch represents an orbital with the quantum numbers n = 3, l = 0, and ml = 0? A) B) C) D) E) 3
4 SHORT ANSWER. Write the word or phrase that best completes each statement or answers the question. 17) On the axes below, draw the general shape of a py orbital. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 18) Each p-orbital can accommodate a maximum of electrons. A) 5 B) 3 C) 1 D) 2 E) 6 19) Which one of the following represents an acceptable possible set of quantum numbers (in the order n, l, ml, ms) for an electron in an atom? A) 2, 1, 0, 0 B) 2, 2, 0, 1/2 C) 2, 0, 2, +1/2 D) 2, 0, 1, -1/2 E) 2, 1, -1, 1/2 4
5 20) Which electron configuration represents a violation of the Pauli exclusion principle? A) B) C) D) E) 21) The electron configuration of a ground-state Ag atom is. A) [Kr]5s14d10 B) [Kr]5s24d10 C) [Ar]4s24d9 D) [Kr]5s23d9 E) [Ar]4s14d10 5
6 22) Which electron configuration denotes an atom in its ground state? A) B) C) D) E) 23) The ground state electron configuration of Fe is. A) 1s22s23s23p63d6 B) 1s22s22p63s23p64s24d6 C) 1s22s22p63s23p64s2 D) 1s22s22p63s23p63d64s2 E) 1s22s23s23p10 24) Which one of the following configurations depicts an excited carbon atom? A) 1s22s22p3 B) 1s22s22p2 C) 1s22s23s1 D) 1s22s22p1 E) 1s22s22p13s1 Consider the following electron configurations to answer the questions that follow: (i) 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 (ii) 1s2 2s2 2p6 3s2 3p5 (iii) 1s2 2s2 2p6 3s2 3p6 4s2 3d8 (iv) 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 (v) 1s2 2s2 2p4 3s1 25) An example of an electron configuration of a transition metal is. A) (i) B) (ii) C) (iii) D) (iv) E) (v) 6
7 26) An example of an excited state electron configuration for fluorine is. A) (i) B) (ii) C) (iii) D) (iv) E) (v) 27) Elements in group have a np6 electron configuration in the outer shell. A) 4A B) 7A C) 5A D) 6A E) 8A 28) The electron configuration of the valence electrons of an atom in its ground state is ns2np1. This atom is a group element. A) q B) r C) s D) t E) y 29) In which set of elements would all members be expected to have very similar chemical properties? A) S, Se, Si B) O, S, Se C) Ne, Na, Mg D) N, O, F E) Na, Mg, K 30) Atomic radius generally increases as we move. A) up a group and from right to left across a period B) down a group; the period position has no effect C) up a group and from left to right across a period D) down a group and from right to left across a period E) down a group and from left to right across a period 31) The atomic radius of main-group elements generally increases down a group because. A) effective nuclear charge increases down a group B) the principal quantum number of the valence orbitals increases C) effective nuclear charge decreases down a group D) effective nuclear charge zigzags down a group E) both effective nuclear charge increases down a group and the principal quantum number of the valence orbitals increases 32) Screening by core electrons in atoms is. A) more efficient than that by valence electrons B) responsible for a general decrease in atomic radius going down a group C) essentially identical to that by valence electrons D) less efficient than that by valence electrons E) both essentially identical to that by valence electrons and responsible for a general decrease in atomic radius going down a group 33) Which one of the following has the smallest radius? A)P B)Na C)Br D)Cl E)Fe 7
8 34) Which one of the following atoms has the largest radius? A) Co B) Sr C) I D) Ca E) Ba Consider the following electron configurations to answer the questions that follow: (i) [Kr] 5s1 (ii) [Ne] 3s2 3p5 (iii) [Ar] 4s2 3d10 4p4 (iv) [Ne] 3s2 3p6 (v) [Ar] 4s1 35) The electron configuration of the atom that is expected to have the lowest first ionization energy is. A) (i) B) (ii) C) (iii) D) (iv) E) (v) 36) The electron configuration of the atom that is expected to have the highest first ionization energy is. A) (i) B) (ii) C) (iii) D) (iv) E) (v) 37) Of the choices below, which gives the order for first ionization energies? A) Cl > S > Al > Ar > Si B) S > Si > Cl > Al > Ar C) Al > Si > S > Cl > Ar D) Cl > S > Al > Si > Ar E) Ar > Cl > S > Si > Al 38) The first ionization energies of the elements as you go from left to right across a period of the periodic table, and as you go from the bottom to the top of a group in the table. A) increase, increase B) increase, decrease C) decrease, increase D) decrease, decrease E) are completely unpredictable 39) have the lowest first ionization energies of the groups listed. A) Transition elements B) Halogens C) Alkaline earth metals D) Alkali metals E) Noble gases 40) Which of the following has the largest second ionization energy? A)P B)Al C)Mg D)Na E)Si 8
9 41) Which of the following correctly represents the second ionization of calcium? A) Ca+ (g) Ca2+ (g) + e- B) Ca (g) Ca+ (g) + e- C) Ca- (g) + e- Ca2- (g) D) Ca+ (g) + e- Ca (g) E) Ca+ (g) + e- Ca2+ (g) 42) Which ion in the isoelectronic series below has the smallest radius in a crystal? A) O2- B) N3- C) Na+ D) Al3+ E) F- 43) Which of the following sets contains species that are isoelectronic? A) Cl, Ar, K B) F-, Ne, Na+ C) F, Ne, Na D) Al3+, S2-, Ar E) P3-, S2-, Ar- 44) Of the following elements, has the most negative electron affinity. A) Be B) N C) F D) Li E) Na 45) Sodium is much more apt to exist as a cation than is chlorine. This is because. A) chlorine is a gas and sodium is a solid B) chlorine is more metallic than sodium C) chlorine has a greater ionization energy than sodium does D) chlorine has a greater electron affinity than sodium does E) chlorine is bigger than sodium 46) Of the elements below, is the most metallic. A) sodium B) barium C) calcium D) cesium E) magnesium 9
10 Answer Key Testname: CH_07_PRAC_TEST.TST MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) A ID: chem9b ) A ID: chem9b ) E ID: chem9b ) B ID: chem9b ) E ID: chem9b ) A ID: chem9b ) C ID: chem9b ) D ID: chem9b ) C ID: chem9b ) E ID: chem9b ) B ID: chem9b ) D ID: chem9b ) A ID: chem9b ) C ID: chem9b ) E ID: chem9b ) D ID: chem9b
11 Answer Key Testname: CH_07_PRAC_TEST.TST SHORT ANSWER. Write the word or phrase that best completes each statement or answers the question. 17) ID: chem9b MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 18) D ID: chem9b ) E ID: chem9b ) A ID: chem9b ) A ID: chem9b ) D ID: chem9b ) D ID: chem9b ) E ID: chem9b ) C ID: chem9b ) E ID: chem9b ) E ID: chem9b ) D ID: chem9b ) B ID: chem9b
12 Answer Key Testname: CH_07_PRAC_TEST.TST 30) D ID: chem9b ) B ID: chem9b ) A ID: chem9b ) D ID: chem9b ) E ID: chem9b ) A ID: chem9b ) D ID: chem9b ) E ID: chem9b ) A ID: chem9b ) D ID: chem9b ) D ID: chem9b ) A ID: chem9b ) D ID: chem9b ) B ID: chem9b ) C ID: chem9b ) C ID: chem9b ) D ID: chem9b
13
14
15
A.P. Chemistry Practice Test - Ch. 7, Atomic Structure and Periodicity
 A.P. Chemistry Practice Test - Ch. 7, Atomic Structure and Periodicity 1) Ham radio operators often broadcast on the 6-meter band. The frequency of this electromagnetic radiation is MHz. A) 50 B) 20 C)
A.P. Chemistry Practice Test - Ch. 7, Atomic Structure and Periodicity 1) Ham radio operators often broadcast on the 6-meter band. The frequency of this electromagnetic radiation is MHz. A) 50 B) 20 C)
Key Equations. Determining the smallest change in an atom's energy.
 ATOMIC STRUCTURE AND PERIODICITY Matter and Energy Key Equations λν = c ΔE = hν Relating speed of a wave to its wavelength and frequency. Determining the smallest change in an atom's energy. H( λ =R n
ATOMIC STRUCTURE AND PERIODICITY Matter and Energy Key Equations λν = c ΔE = hν Relating speed of a wave to its wavelength and frequency. Determining the smallest change in an atom's energy. H( λ =R n
CDO AP Chemistry Unit 5
 1. a. Calculate the wavelength of electromagnetic radiation that has a frequency of 5.56 MHz. b. Calculate the frequency of electromagnetic radiation that has a wavelength equal to 667 nm. 2. Electromagnetic
1. a. Calculate the wavelength of electromagnetic radiation that has a frequency of 5.56 MHz. b. Calculate the frequency of electromagnetic radiation that has a wavelength equal to 667 nm. 2. Electromagnetic
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Test 4 Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the subshells below do not exist due to the constraints upon the azimuthal
Test 4 Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the subshells below do not exist due to the constraints upon the azimuthal
AP Chapter 6 Study Questions
 Class: Date: AP Chapter 6 Study Questions True/False Indicate whether the statement is true or false. 1. The wavelength of radio waves can be longer than a football field. 2. Black body radiation is the
Class: Date: AP Chapter 6 Study Questions True/False Indicate whether the statement is true or false. 1. The wavelength of radio waves can be longer than a football field. 2. Black body radiation is the
1. Ham radio operators often broadcast on the 6 meter band. The frequency of this electromagnetic radiation is MHz. a. 500 b. 200 c. 50 d. 20 e. 2.
 Name: Score: 0 / 60 points (0%) [1 open ended question not graded] Chapters 6&7 Practice Exam Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Ham radio
Name: Score: 0 / 60 points (0%) [1 open ended question not graded] Chapters 6&7 Practice Exam Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Ham radio
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Test 3 Fall 2011 General Chemistry I Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) What is the electron configuration for the Co2+ ion?
Test 3 Fall 2011 General Chemistry I Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) What is the electron configuration for the Co2+ ion?
Unit 7. Atomic Structure
 Unit 7. Atomic Structure Upon successful completion of this unit, the students should be able to: 7.1 List the eight regions of the electromagnetic spectrum in the designated order and perform calculations
Unit 7. Atomic Structure Upon successful completion of this unit, the students should be able to: 7.1 List the eight regions of the electromagnetic spectrum in the designated order and perform calculations
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which one of the following is not one of the postulates of Dalton's atomic theory? A)
Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which one of the following is not one of the postulates of Dalton's atomic theory? A)
Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT.
 ELECTRONS IN ATOMS Chapter Quiz Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. 1. The orbitals of a principal energy level are lower in energy than the orbitals
ELECTRONS IN ATOMS Chapter Quiz Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. 1. The orbitals of a principal energy level are lower in energy than the orbitals
Honors Chemistry: Chapter 4- Problem Set (with some 6)
 Honors Chemistry: Chapter 4- Problem Set (with some 6) All answers and work on a separate sheet of paper! Classify the following as always true (AT), sometimes true (ST), or never true (NT) 1. Atoms of
Honors Chemistry: Chapter 4- Problem Set (with some 6) All answers and work on a separate sheet of paper! Classify the following as always true (AT), sometimes true (ST), or never true (NT) 1. Atoms of
Chapter 11 Prep Test CLASS SET!!!! Matching
 CLASS SET!!!! ID: A Chapter 11 Prep Test Matching Match each item with the correct statement below. A electronegativity D period B ionization energy E transition metal C atomic radius F group 1 type of
CLASS SET!!!! ID: A Chapter 11 Prep Test Matching Match each item with the correct statement below. A electronegativity D period B ionization energy E transition metal C atomic radius F group 1 type of
CHM 1045 Test #4 December 4, 2000
 CHM 1045 Test #4 December 4, 2000 1. The scientist who was first to propose that electrons in an atom could have only certain energies was a. Planck. b. Einstein. c. Bohr. d. Rydberg. 2. Select the arrangement
CHM 1045 Test #4 December 4, 2000 1. The scientist who was first to propose that electrons in an atom could have only certain energies was a. Planck. b. Einstein. c. Bohr. d. Rydberg. 2. Select the arrangement
CHEMISTRY - BROWN 13E CH.7 - PERIODIC PROPERTIES OF THE ELEMENTS
 !! www.clutchprep.com CONCEPT: EFFECTIVE NUCLEAR CHARGE & SLATER S RULES When looking at any particular electron within an atom it experiences two major forces. A(n) force from the nucleus and a(n) force
!! www.clutchprep.com CONCEPT: EFFECTIVE NUCLEAR CHARGE & SLATER S RULES When looking at any particular electron within an atom it experiences two major forces. A(n) force from the nucleus and a(n) force
Particle Behavior of Light 1. Calculate the energy of a photon, mole of photons 2. Find binding energy of an electron (know KE) 3. What is a quanta?
 Properties of Electromagnetic Radiation 1. What is spectroscopy, a continuous spectrum, a line spectrum, differences and similarities 2. Relationship of wavelength to frequency, relationship of E to λ
Properties of Electromagnetic Radiation 1. What is spectroscopy, a continuous spectrum, a line spectrum, differences and similarities 2. Relationship of wavelength to frequency, relationship of E to λ
Gilbert Kirss Foster. Chapter3. Atomic Structure. Explaining the Properties of Elements
 Gilbert Kirss Foster Chapter3 Atomic Structure Explaining the Properties of Elements Chapter Outline 3.1 Waves of Light 3.2 Atomic Spectra 3.3 Particles of Light: Quantum Theory 3.4 The Hydrogen Spectrum
Gilbert Kirss Foster Chapter3 Atomic Structure Explaining the Properties of Elements Chapter Outline 3.1 Waves of Light 3.2 Atomic Spectra 3.3 Particles of Light: Quantum Theory 3.4 The Hydrogen Spectrum
Name Date Period Unit 3 Review: Electrons and the periodic table
 Name Date Period Unit 3 Review: Electrons and the periodic table G Chem; Coleman SHOW YOUR WORK ON ANY AND ALL CALCULATIONS. SIG FIGS MATTER. UNITS MATTER. General Questions: 1. Use the following terms
Name Date Period Unit 3 Review: Electrons and the periodic table G Chem; Coleman SHOW YOUR WORK ON ANY AND ALL CALCULATIONS. SIG FIGS MATTER. UNITS MATTER. General Questions: 1. Use the following terms
Test bank chapter (7)
 Test bank chapter (7) Choose the most correct answer 1. The lowest energy state of an atom is referred to as its a) bottom state. b) ground state. c) fundamental state. d) original state. 2. All s orbitals
Test bank chapter (7) Choose the most correct answer 1. The lowest energy state of an atom is referred to as its a) bottom state. b) ground state. c) fundamental state. d) original state. 2. All s orbitals
Notes: Unit 6 Electron Configuration and the Periodic Table
 Name KEY Block Notes: Unit 6 Electron Configuration and the Periodic Table In the 1790's Antoine Lavoisier compiled a list of the known elements at that time. There were only 23 elements. By the 1870's
Name KEY Block Notes: Unit 6 Electron Configuration and the Periodic Table In the 1790's Antoine Lavoisier compiled a list of the known elements at that time. There were only 23 elements. By the 1870's
Honors Unit 6 Atomic Structure
 Honors Unit 6 Atomic Structure Miss Adams Honors Chemistry 1 Name: 1 Homework #1: Frequency, Wavelength and the Speed of Light Show all of your work for the problems, including the initial formula, substitution
Honors Unit 6 Atomic Structure Miss Adams Honors Chemistry 1 Name: 1 Homework #1: Frequency, Wavelength and the Speed of Light Show all of your work for the problems, including the initial formula, substitution
Ch. 4 Sec. 1-2, Ch. 3 sec.6-8 ENERGY CHANGES AND THE QUANTUM THEORY THE PERIODIC TABLE
 Ch. 4 Sec. 1-2, Ch. 3 sec.6-8 ENERGY CHANGES AND THE QUANTUM THEORY THE PERIODIC TABLE What Makes Red Light Red? (4.1) Electromagnetic Radiation: energy that travels in waves (light) Waves Amplitude: height
Ch. 4 Sec. 1-2, Ch. 3 sec.6-8 ENERGY CHANGES AND THE QUANTUM THEORY THE PERIODIC TABLE What Makes Red Light Red? (4.1) Electromagnetic Radiation: energy that travels in waves (light) Waves Amplitude: height
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) In nature, sulfur is most commonly found in. 1) A) pure elemental sulfur B) H2S C) sulfuric
Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) In nature, sulfur is most commonly found in. 1) A) pure elemental sulfur B) H2S C) sulfuric
Ch 9 Electrons in Atoms & the Periodic Table Study Sheet Acc. Chemistry SCANTRON. Name /99. 3) Light is a type of matter. 3)
 Ch 9 Electrons in Atoms & the Periodic Table Study Sheet Acc. Chemistry SCANTRON Name /99 TRUE/FALSE. Write 'T' if the statement is true and 'F' if the statement is false. 1) When the elements are arranged
Ch 9 Electrons in Atoms & the Periodic Table Study Sheet Acc. Chemistry SCANTRON Name /99 TRUE/FALSE. Write 'T' if the statement is true and 'F' if the statement is false. 1) When the elements are arranged
A. 24 B. 27 C. 30 D. 32 E. 33. A. It is impossible to tell from the information given. B. 294 mm C. 122 mm D. 10 mm E. 60 mm A. 1 H B. C. D. 19 F " E.
 CHEMISTRY 110 EXAM 1 Sept. 24, 2012 FORM A 1. A microwave oven uses 2.45! 10 9 Hz electromagnetic waves to heat food. What is the wavelength of this radiation in mm? A. It is impossible to tell from the
CHEMISTRY 110 EXAM 1 Sept. 24, 2012 FORM A 1. A microwave oven uses 2.45! 10 9 Hz electromagnetic waves to heat food. What is the wavelength of this radiation in mm? A. It is impossible to tell from the
CHEMISTRY 110 EXAM 1 SEPTEMBER 20, 2010 FORM A
 CHEMISTRY 110 EXAM 1 SEPTEMBER 20, 2010 FORM A 1. What are the correct numbers of protons, neutrons and electrons in a 39 K + ion? p n e A. 20 19 18 B. 20 19 19 C. 19 20 18 D. 19 20 19 E. 20 19 20 2. Which
CHEMISTRY 110 EXAM 1 SEPTEMBER 20, 2010 FORM A 1. What are the correct numbers of protons, neutrons and electrons in a 39 K + ion? p n e A. 20 19 18 B. 20 19 19 C. 19 20 18 D. 19 20 19 E. 20 19 20 2. Which
Sample Exercise 6.1 Concepts of Wavelength and Frequency
 Sample Exercise 6.1 Concepts of Wavelength and Frequency Two electromagnetic waves are represented in the margin. (a) Which wave has the higher frequency? (b) If one wave represents visible light and the
Sample Exercise 6.1 Concepts of Wavelength and Frequency Two electromagnetic waves are represented in the margin. (a) Which wave has the higher frequency? (b) If one wave represents visible light and the
What is the energy of a photon with wavelength 232 nm?
 EMISTRY 110 EXAM 1 February 6, 2012 FRM A 1 ow many single covalent bonds must a sulfur atom form to have a complete octet in its valence shell? A. 3 B. 4. 1 D. 2 E. 0 2. What are the correct numbers of
EMISTRY 110 EXAM 1 February 6, 2012 FRM A 1 ow many single covalent bonds must a sulfur atom form to have a complete octet in its valence shell? A. 3 B. 4. 1 D. 2 E. 0 2. What are the correct numbers of
The Electronic Structures of Atoms Electromagnetic Radiation The wavelength of electromagnetic radiation has the symbol λ.
 CHAPTER 7 Atomic Structure Chapter 8 Atomic Electron Configurations and Periodicity 1 The Electronic Structures of Atoms Electromagnetic Radiation The wavelength of electromagnetic radiation has the symbol
CHAPTER 7 Atomic Structure Chapter 8 Atomic Electron Configurations and Periodicity 1 The Electronic Structures of Atoms Electromagnetic Radiation The wavelength of electromagnetic radiation has the symbol
White Light. Chapter 7 Electron Structure of the Atom
 Chapter 7 Electron Structure of the Atom Electromagnetic Radiation and Energy The Bohr Model of the Hydrogen Atom The Modern Model of the Atom Periodicity of Electron Configurations Valence Electrons for
Chapter 7 Electron Structure of the Atom Electromagnetic Radiation and Energy The Bohr Model of the Hydrogen Atom The Modern Model of the Atom Periodicity of Electron Configurations Valence Electrons for
Test Bank for General Chemistry Atoms First 2nd Edition by John E. McMurry and Robert C. Fay
 Test Bank for General Chemistry Atoms First 2nd Edition by John E. McMurry and Robert C. Fay Link download full: https://digitalcontentmarket.org/download/test-bank-for-general-chemistry-atoms-f irst-2nd-edition-by-mcmurry-and-fay/
Test Bank for General Chemistry Atoms First 2nd Edition by John E. McMurry and Robert C. Fay Link download full: https://digitalcontentmarket.org/download/test-bank-for-general-chemistry-atoms-f irst-2nd-edition-by-mcmurry-and-fay/
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Elements in Group 8A are known as the. A) alkali metals B) alkaline earth metals C) noble
Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Elements in Group 8A are known as the. A) alkali metals B) alkaline earth metals C) noble
Atomic Structure. 1. For a hydrogen atom which electron transition requires the largest amount of energy?
 Atomic Structure 1. For a hydrogen atom which electron transition requires the largest amount of energy? A. n = 4 to n = 10 B. n = 3 to n = 2 C. n = 3 to n = 4 D. n = 1 to n = 3 E. n = 2 to n = 4 2. Which
Atomic Structure 1. For a hydrogen atom which electron transition requires the largest amount of energy? A. n = 4 to n = 10 B. n = 3 to n = 2 C. n = 3 to n = 4 D. n = 1 to n = 3 E. n = 2 to n = 4 2. Which
1) What type of relationship (direct or indirect) exists between wavelength, frequency, and photon energy?
 Chapter 7 Study Guide Name: AP Chemistry 6 points DUE AT TEST (Mon., 11/20/17) Date: Topics/people to be covered on the test: wavelength frequency c=ëí ideas of Max Planck E=hí quanta photons relationship
Chapter 7 Study Guide Name: AP Chemistry 6 points DUE AT TEST (Mon., 11/20/17) Date: Topics/people to be covered on the test: wavelength frequency c=ëí ideas of Max Planck E=hí quanta photons relationship
Chapter 6 Part 3; Many-electron atoms
 Chapter 6 Part 3; Many-electron atoms Read: BLB 6.7 6.9 HW: BLB 6:59,63,64,67,71b-d,74,75,90,97; Packet 6:10 14 Know: s & atoms with many electrons Spin quantum number m s o Pauli exclusion principle o
Chapter 6 Part 3; Many-electron atoms Read: BLB 6.7 6.9 HW: BLB 6:59,63,64,67,71b-d,74,75,90,97; Packet 6:10 14 Know: s & atoms with many electrons Spin quantum number m s o Pauli exclusion principle o
Professor K. Section 8 Electron Configuration Periodic Table
 Professor K Section 8 Electron Configuration Periodic Table Schrödinger Cannot be solved for multielectron atoms We must assume the orbitals are all hydrogen-like Differences In the H atom, all subshells
Professor K Section 8 Electron Configuration Periodic Table Schrödinger Cannot be solved for multielectron atoms We must assume the orbitals are all hydrogen-like Differences In the H atom, all subshells
1. Electronic Structure 2. Electron Configuration 3. Core Notation 4. EC Relationship to Periodic Table 5. Electron Configuration of Ions
 Pre-AP Chemistry 11 Atomic Theory II Name: Date: Block: 1. Electronic Structure 2. Electron Configuration 3. Core Notation 4. EC Relationship to Periodic Table 5. Electron Configuration of Ions Electronic
Pre-AP Chemistry 11 Atomic Theory II Name: Date: Block: 1. Electronic Structure 2. Electron Configuration 3. Core Notation 4. EC Relationship to Periodic Table 5. Electron Configuration of Ions Electronic
Section 11: Electron Configuration and Periodic Trends
 Section 11: Electron Configuration and Periodic Trends The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 11.01 The Bohr Model of the Atom
Section 11: Electron Configuration and Periodic Trends The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 11.01 The Bohr Model of the Atom
Assessment Chapter 5 Pre-Test Chapter: The Periodic Law Use the periodic table below to answer the questions in this Chapter Test.
 Assessment Chapter 5 Pre-Test Chapter: The Periodic Law Use the periodic table below to answer the questions in this Chapter Test. In the space provided, write the letter of the term or phrase that best
Assessment Chapter 5 Pre-Test Chapter: The Periodic Law Use the periodic table below to answer the questions in this Chapter Test. In the space provided, write the letter of the term or phrase that best
3. Write ground-state electron configurations for any atom or ion using only the Periodic Table. (Sections 8.3 & 9.2)
 Lecture 2: learning objectives, readings, topics, and resources: 1. Understand the significance of the quantum numbers, understand how they can be used to code for the electron energy levels within atoms
Lecture 2: learning objectives, readings, topics, and resources: 1. Understand the significance of the quantum numbers, understand how they can be used to code for the electron energy levels within atoms
10 Modern Atomic Theory and the Periodic Table. Chapter Outline. ElectromagneDc RadiaDon. ElectromagneDc RadiaDon. ElectromagneDc RadiaDon
 10 Modern Atomic Theory and the Periodic Table Chapter Outline 10.1 ElectromagneDc RadiaDon 10.2 The Bohr Atom 10.3 10.4 10.5 Electron Structures and the Periodic Table The amazing colors of fireworks
10 Modern Atomic Theory and the Periodic Table Chapter Outline 10.1 ElectromagneDc RadiaDon 10.2 The Bohr Atom 10.3 10.4 10.5 Electron Structures and the Periodic Table The amazing colors of fireworks
2) The energy of a photon of light is proportional to its frequency and proportional to its wavelength.
 Advanced Chemistry Chapter 13 Review Name Per Show all work Wave Properties 1) Which one of the following is correct? A) ν + λ = c B) ν λ = c C) ν = cλ D) λ = c ν E) νλ = c 2) The energy of a photon of
Advanced Chemistry Chapter 13 Review Name Per Show all work Wave Properties 1) Which one of the following is correct? A) ν + λ = c B) ν λ = c C) ν = cλ D) λ = c ν E) νλ = c 2) The energy of a photon of
Test Review # 4. Chemistry: Form TR4-9A
 Chemistry: Form TR4-9A REVIEW Name Date Period Test Review # 4 Location of electrons. Electrons are in regions of the atom known as orbitals, which are found in subdivisions of the principal energy levels
Chemistry: Form TR4-9A REVIEW Name Date Period Test Review # 4 Location of electrons. Electrons are in regions of the atom known as orbitals, which are found in subdivisions of the principal energy levels
Name: Electrons in Atoms Chemical Periodicity Chapters 13 and 14
 Name: Electrons in Atoms Chemical Periodicity Chapters 13 and 14 1 Chapter 13 Electrons in Atoms We need to further develop our understanding of atomic structure to help us understand how atoms bond to
Name: Electrons in Atoms Chemical Periodicity Chapters 13 and 14 1 Chapter 13 Electrons in Atoms We need to further develop our understanding of atomic structure to help us understand how atoms bond to
Chemistry 1211K Test III. MULTIPLE CHOICE (3 points each, put answers on the answer sheet).
 A Chemistry 1211K Test III MULTIPLE CICE (3 points each, put answers on the answer sheet). 1) f the following, which one is a state function? A) q B) C) w D) heat E) none of the above 2) The reaction 4Al
A Chemistry 1211K Test III MULTIPLE CICE (3 points each, put answers on the answer sheet). 1) f the following, which one is a state function? A) q B) C) w D) heat E) none of the above 2) The reaction 4Al
CHAPTER STRUCTURE OF ATOM
 12 CHAPTER STRUCTURE OF ATOM 1. The spectrum of He is expected to be similar to that [1988] H Li + Na He + 2. The number of spherical nodes in 3p orbitals are [1988] one three none two 3. If r is the radius
12 CHAPTER STRUCTURE OF ATOM 1. The spectrum of He is expected to be similar to that [1988] H Li + Na He + 2. The number of spherical nodes in 3p orbitals are [1988] one three none two 3. If r is the radius
What is the total change in internal energy for a system that does 42 kj of work and absorbs 17 kj of heat?
 Question #: 1 Posting ID: 426731 Course: CHE 105 2015 SU Instructor: Sarah Edwards Which statement is false? A. Potential energy is associated with the position or composition of an object. B. Kinetic
Question #: 1 Posting ID: 426731 Course: CHE 105 2015 SU Instructor: Sarah Edwards Which statement is false? A. Potential energy is associated with the position or composition of an object. B. Kinetic
Chapter 2: The Structure of the Atom and the Periodic Table
 Chapter 2: The Structure of the Atom and the Periodic Table 1. What are the three primary particles found in an atom? A) neutron, positron, and electron B) electron, neutron, and proton C) electron, proton,
Chapter 2: The Structure of the Atom and the Periodic Table 1. What are the three primary particles found in an atom? A) neutron, positron, and electron B) electron, neutron, and proton C) electron, proton,
Modern Atomic Theory and the Periodic Table
 Modern Atomic Theory and the Periodic Table Chapter 10 the exam would have to be given earlier Hein and Arena Version 1.1 Eugene Passer Chemistry Department Bronx Community 1 College John Wiley and Sons,
Modern Atomic Theory and the Periodic Table Chapter 10 the exam would have to be given earlier Hein and Arena Version 1.1 Eugene Passer Chemistry Department Bronx Community 1 College John Wiley and Sons,
1) Which electron would be most likely to emit x-ray electromagnetic energy?
 AP Chemistry Test (Chapter 7) Multiple Choice (40%) 1) Which electron would be most likely to emit x-ray electromagnetic energy? A) n = 1 n = 6 B) n = 2 n = 3 C) n = 6 n = 1 D) n = 3 n = 2 2) Which statement
AP Chemistry Test (Chapter 7) Multiple Choice (40%) 1) Which electron would be most likely to emit x-ray electromagnetic energy? A) n = 1 n = 6 B) n = 2 n = 3 C) n = 6 n = 1 D) n = 3 n = 2 2) Which statement
CHEMISTRY - KIRSS 2E CH.3 - ATOMIC STRUCTURE: EXPLAINING THE PROPERTIES OF ELEMENTS
 !! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
!! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
The orbitals in an atom are arranged in shells and subshells. orbital 3s 3p 3d. Shell: all orbitals with the same value of n.
 Shells and Subshells The orbitals in an atom are arranged in shells and subshells. n=3 orbital 3s 3p 3d Shell: all orbitals with the same value of n n=3 3s 3p 3d Subshell: all orbitals with the same value
Shells and Subshells The orbitals in an atom are arranged in shells and subshells. n=3 orbital 3s 3p 3d Shell: all orbitals with the same value of n n=3 3s 3p 3d Subshell: all orbitals with the same value
Problems with the Wave Theory of Light (Photoelectric Effect)
 CHEM101 NOTES Properties of Light Found that the wave theory could not work for some experiments e.g. the photovoltaic effect This is because the classic EM view of light could not account for some of
CHEM101 NOTES Properties of Light Found that the wave theory could not work for some experiments e.g. the photovoltaic effect This is because the classic EM view of light could not account for some of
Modern Atomic Theory CHAPTER OUTLINE
 Chapter 3B Modern Atomic Theory 1 CHAPTER OUTLINE Waves Electromagnetic Radiation Dual Nature of Light Bohr Model of Atom Quantum Mechanical Model of Atom Electron Configuration Electron Configuration
Chapter 3B Modern Atomic Theory 1 CHAPTER OUTLINE Waves Electromagnetic Radiation Dual Nature of Light Bohr Model of Atom Quantum Mechanical Model of Atom Electron Configuration Electron Configuration
ATOMIC THEORY, PERIODICITY, and NUCLEAR CHEMISTRY
 ATOMIC THEORY, PERIODICITY, and NUCLEAR CHEMISTRY Note: For all questions referring to solutions, assume that the solvent is water unless otherwise stated. 1. The nuclide is radioactive and decays by the
ATOMIC THEORY, PERIODICITY, and NUCLEAR CHEMISTRY Note: For all questions referring to solutions, assume that the solvent is water unless otherwise stated. 1. The nuclide is radioactive and decays by the
2 e. 14 e. # e # orbitals. 10 e 5. sublevel. shape of orbital. Orbital Shapes. Notes Orbital Notation; e Config; NGN.
 How to build an atom: The bigger (more massive) the atom, the more protons (and neutrons) The bigger the atom, the more electrons Electrons fill lower energy levels first "Aufbau" Principle ("To build
How to build an atom: The bigger (more massive) the atom, the more protons (and neutrons) The bigger the atom, the more electrons Electrons fill lower energy levels first "Aufbau" Principle ("To build
CHEMISTRY - ZUMDAHL 8E CH.7 - ATOMIC STRUCTURE & PERIODICITY.
 !! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
!! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
s or Hz J atom J mol or -274 kj mol CHAPTER 4. Practice Exercises ΔE atom = ΔE mol =
 CHAPTER 4 Practice Exercises 4.1 10 1 2.1410 s or Hz 4.3 ΔE atom = ΔE mol = 4.5610 J atom 19 1 2.7410 J mol or -274 kj mol 5 1-1 4.5 excitation energy = 471 kj mol 1 + 275 kj mol 1 = 746 kj mol 1 Hg 4.7
CHAPTER 4 Practice Exercises 4.1 10 1 2.1410 s or Hz 4.3 ΔE atom = ΔE mol = 4.5610 J atom 19 1 2.7410 J mol or -274 kj mol 5 1-1 4.5 excitation energy = 471 kj mol 1 + 275 kj mol 1 = 746 kj mol 1 Hg 4.7
CHEMISTRY 113 EXAM 3(A)
 Summer 2003 CHEMISTRY 113 EXAM 3(A) 1. Specify radiation with the greatest energy from the following list: A. ultraviolet B. gamma C. infrared D. radio waves 2. The photoelectric effect is: A. reflection
Summer 2003 CHEMISTRY 113 EXAM 3(A) 1. Specify radiation with the greatest energy from the following list: A. ultraviolet B. gamma C. infrared D. radio waves 2. The photoelectric effect is: A. reflection
Unit 3: The Periodic Table and Atomic Theory
 Name: Period: Unit 3: The Periodic Table and Atomic Theory Day Page # Description IC/HW 1 2-3 Periodic Table and Quantum Model Notes IC 1 4-5 Orbital Diagrams Notes IC 1 14 3-A: Orbital Diagrams Worksheet
Name: Period: Unit 3: The Periodic Table and Atomic Theory Day Page # Description IC/HW 1 2-3 Periodic Table and Quantum Model Notes IC 1 4-5 Orbital Diagrams Notes IC 1 14 3-A: Orbital Diagrams Worksheet
The Electronic Structure of Atoms
 The Electronic Structure of Atoms Classical Hydrogen-like atoms: Atomic Scale: 10-10 m or 1 Å + - Proton mass : Electron mass 1836 : 1 Problems with classical interpretation: - Should not be stable (electron
The Electronic Structure of Atoms Classical Hydrogen-like atoms: Atomic Scale: 10-10 m or 1 Å + - Proton mass : Electron mass 1836 : 1 Problems with classical interpretation: - Should not be stable (electron
Orbitals give the probability of finding an electron in a given region of space (boundary surface encloses 90% of electron density)
 Matter Waves Find the wavelength of any object given v and m Orbitals Square of Schrödinger wave-function gives the probability density or electron density or orbital Orbitals give the probability of finding
Matter Waves Find the wavelength of any object given v and m Orbitals Square of Schrödinger wave-function gives the probability density or electron density or orbital Orbitals give the probability of finding
Chapter 1. I- Fill the following table. Element symbol and the mass no. n p n n n e. number. II - Choose the correct answer for the following: Ca-40
 Chapter 1 I- Fill the following table. Element symbol and the mass no. Ca-40 Ca 2+ -40 O-17 O 2- -16 C-12 C-13 Atomic number n p n n n e II - Choose the correct answer for the following: 1. Consider the
Chapter 1 I- Fill the following table. Element symbol and the mass no. Ca-40 Ca 2+ -40 O-17 O 2- -16 C-12 C-13 Atomic number n p n n n e II - Choose the correct answer for the following: 1. Consider the
Quantum Theory & Electronic Structure of Atoms. It s Unreal!! Check your intuition at the door.
 Quantum Theory & Electronic Structure of Atoms It s Unreal!! Check your intuition at the door. 1 Quantum Theory of the Atom Description of the atom and subatomic particles. We will focus on the electronic
Quantum Theory & Electronic Structure of Atoms It s Unreal!! Check your intuition at the door. 1 Quantum Theory of the Atom Description of the atom and subatomic particles. We will focus on the electronic
1. Draw a wave below and label the following parts: peak, trough, wavelength and amplitude
 Wave Nature of Light 1. Draw a wave below and label the following parts: peak, trough, wavelength and amplitude 2. Draw two waves with different frequencies and circle the wave that has a higher frequency.
Wave Nature of Light 1. Draw a wave below and label the following parts: peak, trough, wavelength and amplitude 2. Draw two waves with different frequencies and circle the wave that has a higher frequency.
CHEM 1211K Test III. 4) Of the following elements, which has the largest first ionization energy? A) As B) Sb C) Ge D) Se E) S
 CEM 1211K Test III MULTIPLE COICE. (3 points each) 1) The photoelectric effect is. A) a relativistic effect B) the production of current by silicon solar cells when exposed to sunlight C) the total reflection
CEM 1211K Test III MULTIPLE COICE. (3 points each) 1) The photoelectric effect is. A) a relativistic effect B) the production of current by silicon solar cells when exposed to sunlight C) the total reflection
Chapter Test B. Chapter: Arrangement of Electrons in Atoms. possible angular momentum quantum numbers? energy level? a. 4 b. 8 c. 16 d.
 Assessment Chapter Test B Chapter: Arrangement of Electrons in Atoms PART I In the space provided, write the letter of the term or phrase that best completes each statement or best answers each question
Assessment Chapter Test B Chapter: Arrangement of Electrons in Atoms PART I In the space provided, write the letter of the term or phrase that best completes each statement or best answers each question
1. What is the phenomenon that occurs when certain metals emit electrons when illuminated by particular wavelengths of light? a.
 CHEMISTRY 123-07 Midterm #3 solution key December 02, 2010 Statistics: Average: 77 p (77%); Highest: 100 p (100%); Lowest: 33 p (33%) Number of students performing at or above average: 54 (52%) Number
CHEMISTRY 123-07 Midterm #3 solution key December 02, 2010 Statistics: Average: 77 p (77%); Highest: 100 p (100%); Lowest: 33 p (33%) Number of students performing at or above average: 54 (52%) Number
8. Which of the following could be an isotope of chlorine? (A) 37 Cl 17 (B) 17 Cl 17 (C) 37 Cl 17 (D) 17 Cl 37.5 (E) 17 Cl 37
 Electronic Structure Worksheet 1 Given the following list of atomic and ionic species, find the appropriate match for questions 1-4. (A) Fe 2+ (B) Cl (C) K + (D) Cs (E) Hg + 1. Has the electron configuration:
Electronic Structure Worksheet 1 Given the following list of atomic and ionic species, find the appropriate match for questions 1-4. (A) Fe 2+ (B) Cl (C) K + (D) Cs (E) Hg + 1. Has the electron configuration:
Unit Two Test Review. Click to get a new slide. Choose your answer, then click to see if you were correct.
 Unit Two Test Review Click to get a new slide. Choose your answer, then click to see if you were correct. According to the law of definite proportions, any two samples of water, H2O, A. will be made up
Unit Two Test Review Click to get a new slide. Choose your answer, then click to see if you were correct. According to the law of definite proportions, any two samples of water, H2O, A. will be made up
Electron configurations follow the order of sublevels on the periodic table.
 Electron configurations follow the order of sublevels on the periodic table. 1 The periodic table consists of sublevel blocks arranged in order of increasing energy. Groups 1A(1)-2A(2) = s level Groups
Electron configurations follow the order of sublevels on the periodic table. 1 The periodic table consists of sublevel blocks arranged in order of increasing energy. Groups 1A(1)-2A(2) = s level Groups
Chemistry Chapter 9 Review. 2. Calculate the wavelength of a photon of blue light whose frequency is 6.3 x s -1.
 Chemistry Chapter 9 Review 1. What is the frequency of radiation that has a wavelength of 4.7 x 10-5 cm? 2. Calculate the wavelength of a photon of blue light whose frequency is 6.3 x 10 14 s -1. 3. The
Chemistry Chapter 9 Review 1. What is the frequency of radiation that has a wavelength of 4.7 x 10-5 cm? 2. Calculate the wavelength of a photon of blue light whose frequency is 6.3 x 10 14 s -1. 3. The
MODERN ATOMIC THEORY AND THE PERIODIC TABLE
 C10 04/19/2013 13:34:14 Page 114 CHAPTER 10 MODERN ATOMIC THEORY AND THE PERIODIC TABLE SOLUTIONS TO REVIEW QUESTIONS 1. Wavelength is defined as the distance between consecutive peaks in a wave. It is
C10 04/19/2013 13:34:14 Page 114 CHAPTER 10 MODERN ATOMIC THEORY AND THE PERIODIC TABLE SOLUTIONS TO REVIEW QUESTIONS 1. Wavelength is defined as the distance between consecutive peaks in a wave. It is
Electromagnetic spectrum
 All course materials, including lectures, class notes, quizzes, exams, handouts, presentations, and other materials provided to students for this course are protected intellectual property. As such, the
All course materials, including lectures, class notes, quizzes, exams, handouts, presentations, and other materials provided to students for this course are protected intellectual property. As such, the
2008 Brooks/Cole 2. Frequency (Hz)
 Electromagnetic Radiation and Matter Oscillating electric and magnetic fields. Magnetic field Electric field Chapter 7: Electron Configurations and the Periodic Table Traveling wave moves through space
Electromagnetic Radiation and Matter Oscillating electric and magnetic fields. Magnetic field Electric field Chapter 7: Electron Configurations and the Periodic Table Traveling wave moves through space
Mendeleev s Periodic Law
 Mendeleev s Periodic Law Periodic Law When the elements are arranged in order of increasing atomic mass, certain sets of properties recur periodically. Mendeleev s Periodic Law allows us to predict what
Mendeleev s Periodic Law Periodic Law When the elements are arranged in order of increasing atomic mass, certain sets of properties recur periodically. Mendeleev s Periodic Law allows us to predict what
NEET Exam Study Material Chemistry. 3. Classification of Elements and Periodicity in Properties
 NEET Exam Study Material Chemistry 3. Classification of Elements and Periodicity in Properties 1. Increasing order of atomic weights was violated (anomalous pairs) in the case of (1) Te, I (2) Ar, K (3)
NEET Exam Study Material Chemistry 3. Classification of Elements and Periodicity in Properties 1. Increasing order of atomic weights was violated (anomalous pairs) in the case of (1) Te, I (2) Ar, K (3)
What are the isotopes of hydrogen? H, 2 H, and 3 H
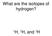 What are the isotopes of hydrogen? 1 H, 2 H, and 3 H The average mass of a boron atom is 10.81. Assuming you were able to isolate only one boron atom, the chance that you would randomly get one with a
What are the isotopes of hydrogen? 1 H, 2 H, and 3 H The average mass of a boron atom is 10.81. Assuming you were able to isolate only one boron atom, the chance that you would randomly get one with a
C H E M 1 CHEM 101-GENERAL CHEMISTRY CHAPTER 6 THE PERIODIC TABLE & ATOMIC STRUCTURE INSTR : FİLİZ ALSHANABLEH
 C H E M 1 CHEM 101-GENERAL CHEMISTRY CHAPTER 6 THE PERIODIC TABLE & ATOMIC STRUCTURE 0 1 INSTR : FİLİZ ALSHANABLEH CHAPTER 6 THE PERIODIC TABLE & ATOMIC STRUCTURE The Electromagnetic Spectrum The Wave
C H E M 1 CHEM 101-GENERAL CHEMISTRY CHAPTER 6 THE PERIODIC TABLE & ATOMIC STRUCTURE 0 1 INSTR : FİLİZ ALSHANABLEH CHAPTER 6 THE PERIODIC TABLE & ATOMIC STRUCTURE The Electromagnetic Spectrum The Wave
Practice MC Test H (Ch 6 & 7) Electrons & Periodicity Name Per
 Practice MC Test H (Ch 6 & 7) Electrons & Periodicity Name Per This is practice - Do NOT cheat yourself of finding out what you are capable of doing. Be sure you follow the testing conditions outlined
Practice MC Test H (Ch 6 & 7) Electrons & Periodicity Name Per This is practice - Do NOT cheat yourself of finding out what you are capable of doing. Be sure you follow the testing conditions outlined
Electronic Structure of Atoms and the Periodic table. Electron Spin Quantum # m s
 Electronic Structure of Atoms and the Periodic table Chapter 6 & 7, Part 3 October 26 th, 2004 Homework session Wednesday 3:00 5:00 Electron Spin Quantum # m s Each electron is assigned a spinning motion
Electronic Structure of Atoms and the Periodic table Chapter 6 & 7, Part 3 October 26 th, 2004 Homework session Wednesday 3:00 5:00 Electron Spin Quantum # m s Each electron is assigned a spinning motion
Why Patterns for Charges of Common Cations and Anions? Electrons in Atoms
 Electrons in Atoms From Light to Energy of Electrons in Atom Quantum mechanical description of Atom 1. Principal quantum number: Shell 2. Orientation (shape) of : Subshell 3. Orbitals hold electrons with
Electrons in Atoms From Light to Energy of Electrons in Atom Quantum mechanical description of Atom 1. Principal quantum number: Shell 2. Orientation (shape) of : Subshell 3. Orbitals hold electrons with
ELECTRONS IN ATOMS AND THE PERIODIC TABLE. Light and Energy. Chapter Nine
 ELECTRONS IN ATOMS AND THE PERIODIC TABLE Chapter Nine Light and Energy! Electromagnetic radiation (EM) is an especially important form of energy for scientific study.! Many types of radiant energy are
ELECTRONS IN ATOMS AND THE PERIODIC TABLE Chapter Nine Light and Energy! Electromagnetic radiation (EM) is an especially important form of energy for scientific study.! Many types of radiant energy are
Bohr Model of Hydrogen Atom
 Bohr Model of Hydrogen Atom electrons move in circular orbits around nucleus orbits can only be of certain radii each radius corresponds to different energy ( only certain energies are allowed) n - defines
Bohr Model of Hydrogen Atom electrons move in circular orbits around nucleus orbits can only be of certain radii each radius corresponds to different energy ( only certain energies are allowed) n - defines
PART 2 Electronic Structure and the Periodic Table. Reference: Chapter 7 8 in textbook
 PART 2 Electronic Structure and the Periodic Table Reference: Chapter 7 8 in textbook 1 Experiment to Discover Atom Structure -particle: He 2+ mass number = 4 Nucleus and Electron Model 2 Atomic Structure
PART 2 Electronic Structure and the Periodic Table Reference: Chapter 7 8 in textbook 1 Experiment to Discover Atom Structure -particle: He 2+ mass number = 4 Nucleus and Electron Model 2 Atomic Structure
Chapter 9. Blimps, Balloons, and Models for the Atom. Electrons in Atoms and the Periodic Table. Hindenburg. Properties of Elements Hydrogen Atoms
 Chapter 9 Electrons in Atoms and the Periodic Table Blimps, Balloons, and Models for the Atom Hindenburg Blimps, Balloons, and Models for the Atom Properties of Elements Hydrogen Atoms Helium Atoms 1 Blimps,
Chapter 9 Electrons in Atoms and the Periodic Table Blimps, Balloons, and Models for the Atom Hindenburg Blimps, Balloons, and Models for the Atom Properties of Elements Hydrogen Atoms Helium Atoms 1 Blimps,
Provide a short and specific definition in YOUR OWN WORDS. Do not use the definition from the book. Electromagnetic Radiation
 Name: Provide a short and specific definition in YOUR OWN WORDS. Do not use the definition from the book Additional Notes: Electromagnetic Radiation Electromagnetic Spectrum Wavelength Frequency Photoelectric
Name: Provide a short and specific definition in YOUR OWN WORDS. Do not use the definition from the book Additional Notes: Electromagnetic Radiation Electromagnetic Spectrum Wavelength Frequency Photoelectric
1051-2nd Chem Exam_ (A)
 1051-2nd Chem Exam_1051207(A) MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) In which of the molecules below is the carbon-carbon distance the
1051-2nd Chem Exam_1051207(A) MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) In which of the molecules below is the carbon-carbon distance the
Chapter 2 Atoms and Elements. Electromagnetic Radiation. Electromagnetic Spectrum. Electron Energy Levels. 2.6 Electron Energy Levels
 Chapter 2 Atoms and Elements Electromagnetic Radiation 2.6 Electron Energy Levels Electromagnetic radiation Consists of energy particles called photons that travel as waves. Includes low energy particles
Chapter 2 Atoms and Elements Electromagnetic Radiation 2.6 Electron Energy Levels Electromagnetic radiation Consists of energy particles called photons that travel as waves. Includes low energy particles
CHAPTER 6. Chemical Periodicity
 CHAPTER 6 Chemical Periodicity 1 Chapter Goals 1. More About the Periodic Table Periodic Properties of the Elements 2. Atomic Radii 3. Ionization Energy (IE) 4. Electron Affinity (EA) 5. Ionic Radii 6.
CHAPTER 6 Chemical Periodicity 1 Chapter Goals 1. More About the Periodic Table Periodic Properties of the Elements 2. Atomic Radii 3. Ionization Energy (IE) 4. Electron Affinity (EA) 5. Ionic Radii 6.
1051-2nd Chem Exam_ (B)
 1051-2nd Chem Exam_1051207(B) MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) have the lowest first ionization energies of the groups listed. A)
1051-2nd Chem Exam_1051207(B) MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) have the lowest first ionization energies of the groups listed. A)
Chemistry 1210, Section 3, Fall semester 2012 Second Hour Exam October 24, 2012 Dr. Scott Ensign ExamVersion 0001
 Chemistry 1210, Section 3, Fall semester 2012 Second Hour Exam October 24, 2012 Dr. Scott Ensign ExamVersion 0001 Instructions: Be sure to mark the exam version number (0001) on your scantron Do not begin
Chemistry 1210, Section 3, Fall semester 2012 Second Hour Exam October 24, 2012 Dr. Scott Ensign ExamVersion 0001 Instructions: Be sure to mark the exam version number (0001) on your scantron Do not begin
AP Chemistry - Problem Drill 10: Atomic Structures and Electron Configuration
 AP Chemistry - Problem Drill 10: Atomic Structures and Electron Configuration No. 1 of 10 Instructions: (1) Read the problem statement and answer choices carefully (2) Work the problems on paper as 1.
AP Chemistry - Problem Drill 10: Atomic Structures and Electron Configuration No. 1 of 10 Instructions: (1) Read the problem statement and answer choices carefully (2) Work the problems on paper as 1.
Chapter 3 Classification of Elements and Periodicity in Properties
 Question 3.1: What is the basic theme of organisation in the periodic table? The basic theme of organisation of elements in the periodic table is to classify the elements in periods and groups according
Question 3.1: What is the basic theme of organisation in the periodic table? The basic theme of organisation of elements in the periodic table is to classify the elements in periods and groups according
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Take Home Exam Chem 1A Fall 2008 - Chapters 6 to 9: You may us any resource you wish accept people. On your honor, you may not ask another person for help. Show your work on every answer. Partial credit
Take Home Exam Chem 1A Fall 2008 - Chapters 6 to 9: You may us any resource you wish accept people. On your honor, you may not ask another person for help. Show your work on every answer. Partial credit
ELECTRONIC STRUCTURE OF ATOMS
 ELECTRONIC STRUCTURE OF ATOMS Electron Spin The electron: spins around its own axis acts as an tiny magnet (any moving electrical charge creates a magnetic field around itself) can spin in either of 2
ELECTRONIC STRUCTURE OF ATOMS Electron Spin The electron: spins around its own axis acts as an tiny magnet (any moving electrical charge creates a magnetic field around itself) can spin in either of 2
Chemistry 121: Atomic and Molecular Chemistry Topic 3: Atomic Structure and Periodicity
 Text Chapter 2, 8 & 9 3.1 Nature of light, elementary spectroscopy. 3.2 The quantum theory and the Bohr atom. 3.3 Quantum mechanics; the orbital concept. 3.4 Electron configurations of atoms 3.5 The periodic
Text Chapter 2, 8 & 9 3.1 Nature of light, elementary spectroscopy. 3.2 The quantum theory and the Bohr atom. 3.3 Quantum mechanics; the orbital concept. 3.4 Electron configurations of atoms 3.5 The periodic
Electron Arrangement - Part 2
 Brad Collins Electron Arrangement - Part 2 Chapter 9 Some images Copyright The McGraw-Hill Companies, Inc. Review Energy Levels Multi-electron 4d 4d 4d 4d 4d n = 4 4s 4p 4p 4p 3d 3d 3d 3d 3d n=3, l = 2
Brad Collins Electron Arrangement - Part 2 Chapter 9 Some images Copyright The McGraw-Hill Companies, Inc. Review Energy Levels Multi-electron 4d 4d 4d 4d 4d n = 4 4s 4p 4p 4p 3d 3d 3d 3d 3d n=3, l = 2
To review Rutherford s model of the atom To explore the nature of electromagnetic radiation To see how atoms emit light
 Objectives To review Rutherford s model of the atom To explore the nature of electromagnetic radiation To see how atoms emit light 1 A. Rutherford s Atom.but there is a problem here!! 2 Using Rutherford
Objectives To review Rutherford s model of the atom To explore the nature of electromagnetic radiation To see how atoms emit light 1 A. Rutherford s Atom.but there is a problem here!! 2 Using Rutherford
Because light behaves like a wave, we can describe it in one of two ways by its wavelength or by its frequency.
 Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
Name Date Class MODELS OF THE ATOM
 5.1 MODELS OF THE ATOM Section Review Objectives Identify inadequacies in the Rutherford atomic model Identify the new assumption in the Bohr model of the atom Describe the energies and positions of electrons
5.1 MODELS OF THE ATOM Section Review Objectives Identify inadequacies in the Rutherford atomic model Identify the new assumption in the Bohr model of the atom Describe the energies and positions of electrons
