Code Course name CFU Year G6403B Info not available 3 1
|
|
|
- Kelly Owen
- 5 years ago
- Views:
Transcription
1 Basic aims The aim of the course is an in-depth discussion of the structureactivity relationships of the main classes of biological molecules. Strategies for synthesis, isolation and structural characterization of biomolecules will be treated. Potential applications in biotechnology will also be discussed, particularly those more closely related to the concept of bioeconomy. Acquired skills The student will be able to critically discuss the structure-activity relationships of the molecular classes presented in the course. He will also acquire knowledge and skills on the most important extraction, preparation/purification and structural characterization methodologies. Finally, the student will be able to use the main experimental and computational methodologies for understanding the mechanisms of macromolecule-ligand interaction. Course contents A general review on structure and chemistry of the main classes of biomolecules: aminoacids and peptides, carbohydrates, nucleic acids and lipids. In particular, the course deals with advanced and up-to-date synthetic methods in solution and in the solid phase. In addition, case studies on specific biomolecules (e.g. insulin) and selected topics will be treated. In addition, the course provides a general introduction to the main spectroscopic techniques for chemical structural analysis of small organic molecules and biomolecules: UV-Vis spectroscopy, chiroptical methods, Nuclear Magnetic Resonance. Particular emphasis is dedicated to practical aspects and applications in structural biology (secondary and tertiary structural analysis, solution structure of small proteins and oligonucleotides, structure-activity relationship studies) and in the food and biotechnology industry (process control).
2 Program Teaching unit 1: Biomolecular Spectroscopy (teacher: Prof. E. M. Ragg) - General introduction. (0.25CFU) Main spectroscopic techniques for chemical structural analysis. The mechanism of interaction between radiation and matter. The electromagnetic spectrum. Fundamental and excited states. Absorption and emission spectroscopies. - Ultraviolet-Visible (UV-VIS) Spectroscopy and Chiroptical methods (0.5 CFU) Electronic transitions and UV-vis absorbance. Electronic, vibrational and rotational energy levels. Molecular orbitals and electronic transitions in isolated and conjugated alkenes, in carbonyl compounds, in benzene and heteroaromatic compounds. Spectral features of purines, pyrimidines, aromatic aminoacids. Chiroptical methods: optical rotation, rotatory optical dispersion, circular dichroism. Applications in biotechnology: determination of melting temperatures of PCR primers. Use of fluorescent probes for DNA staining and binding studies. Secondary structure analysis of proteins and nucleic acids by circular dichroism. - Nuclear Magnetic Resonance (NMR) (1.25CFU). Magnetic properties of the atomic nucleus. Energy and frequency of nuclear spin transitions. Pulsed NMR and Fourier Transform NMR. The chemical shift. Structural factors influencing the chemical shift. Signal multiplicity. Geminal and vicinal couplings. "long range" couplings. Chemical and magnetic equivalence. Examples of analysis of the main spin-systems. The Nuclear Overhauser Effect for the determination of proton-proton distances. - Heteronuclear spectroscopy: 13C-NMR. 31P-NMR, 15N-NMR. Coupled and decoupled spectra. Application examples in the biotechnology and food industry - 2D-NMR spectroscopy. Introduction. The main homo- and hetero-nuclear correlation experiments - Application of NMR spectroscopy for solution structure determination of nucleic acids and proteins and their complexes with small molecules: secondary and tertiary structure analysis
3 of proteins and DNA by NMR; determination of acidity constants by means of chemical shifts ph variations. Thermal stability analysis of biomolecules. Association studies in small moleculebiomolecule interactions Tutorials (1CFU) Class (0.5 CFU): Structure determination of unknown compounds by joint analysis of UV and 1H/13C-NMR spectra Lab.(0.5 CFU): Bioinformatics laboratory on computer analysis of X-Ray and NMR biomolecular structures
4 CFU subdivision Lectures Practise in class Practise in lab. Laboratory Seminar Other 2 0,5 Prerequisites Basic knowledge of the structure and nomenclature of the main functional groups in organic chemistry. Basic knowledge of biochemistry. For any question and further details please contact the teacher at enzio.ragg@unimi.it or andrea.pinto@unimi.it. Preparatory instructions none Learning materials a) E. Rossi, D. Nava, G. Abbiati,G. Celentano,S. Pandini Structure determination of organic compounds, practical exercise Ed. EdiSES; b) Biochemistry- 4th Edition Voet & Voet Wiley; c) Organic Chemistry - 4thEdition - Clayden, Greeves, Warren and Wothers-Oxford University Press, Wiley and Sons; d) Slides of lessons available through the Ariel Web Site Other info MODALITÀ DI ESAME ED ALTRE INFORMAZIONI: The examination is in the written form and will deal with all topics inherent to both course units. Unit 1: a) the student should be able to describe the structure of a simple organic molecule on the basis of the provided spectroscopic data; b) a theoretical question about general aspects of molecular spectroscopy. Unit 2: two questions related to synthetic aspects and the description of structure-activity relationships of specific classes of compounds described in the course. Students should answer to all questions within 2 hours. To each question a formal maximum value of 8 points will be assigned. The exam is considered sufficient when at least 9 points for each unit have been
5 assigned. At least 7 exam sessions per year will be guaranteed. The date of the exam will be published on SIFA platform.
Course Syllabus. Department: Science & Technology. Date: April I. Course Prefix and Number: CHM 212. Course Name: Organic Chemistry II
 Department: Science & Technology Date: April 2012 I. Course Prefix and Number: CHM 212 Course Name: Organic Chemistry II Course Syllabus Credit Hours and Contact Hours: 5 credit hours and 7 (3:3:1) contact
Department: Science & Technology Date: April 2012 I. Course Prefix and Number: CHM 212 Course Name: Organic Chemistry II Course Syllabus Credit Hours and Contact Hours: 5 credit hours and 7 (3:3:1) contact
CHEMISTRY (CHEM) CHEM 208. Introduction to Chemical Analysis II - SL
 Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 100. Elements of General Chemistry Prerequisite(s): Completion of general education requirement in mathematics recommended. Description: The basic concepts of general
Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 100. Elements of General Chemistry Prerequisite(s): Completion of general education requirement in mathematics recommended. Description: The basic concepts of general
Principles of Physical Biochemistry
 Principles of Physical Biochemistry Kensal E. van Hold e W. Curtis Johnso n P. Shing Ho Preface x i PART 1 MACROMOLECULAR STRUCTURE AND DYNAMICS 1 1 Biological Macromolecules 2 1.1 General Principles
Principles of Physical Biochemistry Kensal E. van Hold e W. Curtis Johnso n P. Shing Ho Preface x i PART 1 MACROMOLECULAR STRUCTURE AND DYNAMICS 1 1 Biological Macromolecules 2 1.1 General Principles
The Chemistry department approved by the American Chemical Society offers a Chemistry degree in the following concentrations:
 Chemistry 1 Chemistry 203-C Materials Science Building Telephone: 256.824.6153 Email: chem.admin@uah.edu (chem@uah.edu) The Chemistry department approved by the American Chemical Society offers a Chemistry
Chemistry 1 Chemistry 203-C Materials Science Building Telephone: 256.824.6153 Email: chem.admin@uah.edu (chem@uah.edu) The Chemistry department approved by the American Chemical Society offers a Chemistry
Chemistry (CHEM) Chemistry (CHEM) 1
 Chemistry (CHEM) 1 Chemistry (CHEM) CHEM 1000: Introductory Chemistry Introductory course for students with no high school background in chemistry. Covers fundamental principles of scientific measurement,
Chemistry (CHEM) 1 Chemistry (CHEM) CHEM 1000: Introductory Chemistry Introductory course for students with no high school background in chemistry. Covers fundamental principles of scientific measurement,
CHEMISTRY (CHEM) Chemistry (CHEM) 1
 Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 100 Chemistry: Issues and Answers (3 crs) Prerequisite: No credit if taken after CHEM 101, CHEM 103, or CHEM 115. An exploration into the world of atoms and molecules,
Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 100 Chemistry: Issues and Answers (3 crs) Prerequisite: No credit if taken after CHEM 101, CHEM 103, or CHEM 115. An exploration into the world of atoms and molecules,
CHEMISTRY. CHEM 0100 PREPARATION FOR GENERAL CHEMISTRY 3 cr. CHEM 0110 GENERAL CHEMISTRY 1 4 cr. CHEM 0120 GENERAL CHEMISTRY 2 4 cr.
 CHEMISTRY CHEM 0100 PREPARATION FOR GENERAL CHEMISTRY 3 cr. Designed for those students who intend to take chemistry 0110 and 0120, but whose science and mathematical backgrounds are judged by their advisors
CHEMISTRY CHEM 0100 PREPARATION FOR GENERAL CHEMISTRY 3 cr. Designed for those students who intend to take chemistry 0110 and 0120, but whose science and mathematical backgrounds are judged by their advisors
Biophysical Chemistry: NMR Spectroscopy
 Nuclear Magnetism Vrije Universiteit Brussel 21st October 2011 Outline 1 Overview and Context 2 3 Outline 1 Overview and Context 2 3 Context Proteins (and other biological macromolecules) Functional characterisation
Nuclear Magnetism Vrije Universiteit Brussel 21st October 2011 Outline 1 Overview and Context 2 3 Outline 1 Overview and Context 2 3 Context Proteins (and other biological macromolecules) Functional characterisation
Chemistry Courses -1
 Chemistry Courses -1 Guide to Terms annually = course offered at least once a year during fall or spring semester occasionally = course offered less frequently than once a year, contact advisor if interested
Chemistry Courses -1 Guide to Terms annually = course offered at least once a year during fall or spring semester occasionally = course offered less frequently than once a year, contact advisor if interested
NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY
 NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY A STUDENT SHOULD BE ABLE TO: 1. Identify and explain the processes involved in proton ( 1 H) and carbon-13 ( 13 C) nuclear magnetic resonance
NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY A STUDENT SHOULD BE ABLE TO: 1. Identify and explain the processes involved in proton ( 1 H) and carbon-13 ( 13 C) nuclear magnetic resonance
COURSE UNIT DESCRIPTION. Dept. Organic Chemistry, Vilnius University. Type of the course unit
 Course unit title Organic Chemistry II Lecturer(s) Rimantas Vaitkus COURSE UNIT DESCRIPTION Department Dept. Organic Chemistry, Vilnius University Cycle First Type of the course unit Mode of delivery Period
Course unit title Organic Chemistry II Lecturer(s) Rimantas Vaitkus COURSE UNIT DESCRIPTION Department Dept. Organic Chemistry, Vilnius University Cycle First Type of the course unit Mode of delivery Period
MS731M: Chemical Analysis of Materials
 (Revised) Course Code and Title Course Coordinator Details of Course MS731M: Chemical Analysis of Materials Dr. Eileen Fong (Course Coordinator) Dr. Thresen Matthew Rationale for introducing this course
(Revised) Course Code and Title Course Coordinator Details of Course MS731M: Chemical Analysis of Materials Dr. Eileen Fong (Course Coordinator) Dr. Thresen Matthew Rationale for introducing this course
Information on a Course offered by Department of Biology and Chemistry with effect from Semester A 2015 / 2016
 City University of Hong Kong Information on a Course offered by Department of Biology and Chemistry with effect from Semester A 2015 / 2016 Part I Course Title: Course Code: Course Duration: No. of Credit
City University of Hong Kong Information on a Course offered by Department of Biology and Chemistry with effect from Semester A 2015 / 2016 Part I Course Title: Course Code: Course Duration: No. of Credit
The lecture schedule is only a rough guide and will be likely changed as needed.
 CEHM 239 ORGANIC CHEMISTRY II Spring 2010 Instructor: Professor Hyun-Soon Chong Chemistry Division, BCPS Dept, IIT, LS 398, Chong@iit.edu, 312-567-3235 Course Hours: TR 1:50pm-3:05pm in LS 111 Office Hours:
CEHM 239 ORGANIC CHEMISTRY II Spring 2010 Instructor: Professor Hyun-Soon Chong Chemistry Division, BCPS Dept, IIT, LS 398, Chong@iit.edu, 312-567-3235 Course Hours: TR 1:50pm-3:05pm in LS 111 Office Hours:
Advanced Structural Analysis. Code: ECTS Credits: 6. Degree Type Year Semester
 2017/2018 Advanced Structural Analysis Code: 100907 ECTS Credits: 6 Degree Type Year Semester 2500252 Biochemistry OT 4 0 Contact Name: Ester Boix Borrás Email: Ester.Boix@uab.cat Teachers Use of languages
2017/2018 Advanced Structural Analysis Code: 100907 ECTS Credits: 6 Degree Type Year Semester 2500252 Biochemistry OT 4 0 Contact Name: Ester Boix Borrás Email: Ester.Boix@uab.cat Teachers Use of languages
Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers
 Chapter Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers Ch 1-Structure and bonding Ch 2-Polar covalent bonds: Acids and bases McMurry, J. (2004) Organic Chemistry 6 th Edition
Chapter Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers Ch 1-Structure and bonding Ch 2-Polar covalent bonds: Acids and bases McMurry, J. (2004) Organic Chemistry 6 th Edition
1.- GENERAL INFORMATION
 1.- GENERAL INFORMATION Learning unit Analytical Chemistry Instrumentation I Prerequisites(P) Corequisites (CO) Analytical Chemistry II Analytical Chemistry Instrumentation Lab I Type Basic Particular
1.- GENERAL INFORMATION Learning unit Analytical Chemistry Instrumentation I Prerequisites(P) Corequisites (CO) Analytical Chemistry II Analytical Chemistry Instrumentation Lab I Type Basic Particular
Contents. xiii. Preface v
 Contents Preface Chapter 1 Biological Macromolecules 1.1 General PrincipIes 1.1.1 Macrornolecules 1.2 1.1.2 Configuration and Conformation Molecular lnteractions in Macromolecular Structures 1.2.1 Weak
Contents Preface Chapter 1 Biological Macromolecules 1.1 General PrincipIes 1.1.1 Macrornolecules 1.2 1.1.2 Configuration and Conformation Molecular lnteractions in Macromolecular Structures 1.2.1 Weak
CIM Report May 8, :01pm
 ATTACHMENT C CIM Report May 8, 2017 3:01pm Course Changes Pending Approval from Graduate Committee Code Field Old Value New Value ARTS 4623 ARTS 4963 4123 4213 Deleted code ARTS 4613 ARTS 4963 Course Catalog
ATTACHMENT C CIM Report May 8, 2017 3:01pm Course Changes Pending Approval from Graduate Committee Code Field Old Value New Value ARTS 4623 ARTS 4963 4123 4213 Deleted code ARTS 4613 ARTS 4963 Course Catalog
COURSE MASTER CATALOG
 COURSE MASTER CATALOG CHEM Graduate courses in Chemistry are listed as, 7000-, or 8000- and 9000-level courses. Students in the PhD program are required to take the three core courses, 7021, 7022 and 7023,
COURSE MASTER CATALOG CHEM Graduate courses in Chemistry are listed as, 7000-, or 8000- and 9000-level courses. Students in the PhD program are required to take the three core courses, 7021, 7022 and 7023,
CHEMISTRY (CHEM) CHEM 5800 Principles Of Materials Chemistry. Tutorial in selected topics in materials chemistry. S/U grading only.
 Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 5100 Principles of Organic and Inorganic Chemistry Study of coordination compounds with a focus on ligand bonding, electron counting, molecular orbital theory,
Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 5100 Principles of Organic and Inorganic Chemistry Study of coordination compounds with a focus on ligand bonding, electron counting, molecular orbital theory,
UNIT 3 CHEMISTRY. Fundamental Principles in Chemistry
 UNIT 3 CHEMISTRY NOTE: This list has been compiled based on the topics covered in the 2016 Master Class program. Once all of the 2017 Chemistry program materials have been finalised, this summary will
UNIT 3 CHEMISTRY NOTE: This list has been compiled based on the topics covered in the 2016 Master Class program. Once all of the 2017 Chemistry program materials have been finalised, this summary will
CHEMISTRY (CHM) Chemistry (CHM) 1
 Chemistry (CHM) 1 CHEMISTRY (CHM) CHM 099. Preparatory Chemistry 1. 3 Credit A description of atoms and periodicity, structure of the atom, atomic orbital, the Aufbau principle, combining atoms to make
Chemistry (CHM) 1 CHEMISTRY (CHM) CHM 099. Preparatory Chemistry 1. 3 Credit A description of atoms and periodicity, structure of the atom, atomic orbital, the Aufbau principle, combining atoms to make
A SURVEY OF ORGANIC CHEMISTRY CHEMISTRY 1315 TuTr 9:35-10:55 am, Boggs B6
 GEORGIA INSTITUTE OF TECHNOLOGY School of Chemistry and Biochemistry Spring 2004 A SURVEY OF ORGANIC CHEMISTRY CHEMISTRY 1315 TuTr 9:35-10:55 am, Boggs B6 Instructor: Marcus Weck Office: Boggs 3-85 Phone:
GEORGIA INSTITUTE OF TECHNOLOGY School of Chemistry and Biochemistry Spring 2004 A SURVEY OF ORGANIC CHEMISTRY CHEMISTRY 1315 TuTr 9:35-10:55 am, Boggs B6 Instructor: Marcus Weck Office: Boggs 3-85 Phone:
Prerequisites: MATH 103 or a Math Placement Test score for MATH 104 or 106. Prerequisites: CHEM 109
 Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 101 Career Opportunities in Chemistry Description: Introduction to chemistry careers and faculty research interests in the Department of Chemistry. This course
Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 101 Career Opportunities in Chemistry Description: Introduction to chemistry careers and faculty research interests in the Department of Chemistry. This course
Increasing energy. ( 10 4 cm -1 ) ( 10 2 cm -1 )
 The branch of science which deals with the interaction of electromagnetic radiation with matter is called spectroscopy The energy absorbed or emitted in each transition corresponds to a definite frequency
The branch of science which deals with the interaction of electromagnetic radiation with matter is called spectroscopy The energy absorbed or emitted in each transition corresponds to a definite frequency
Introduction. The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants
 Introduction The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants Spectroscopy and the Electromagnetic Spectrum Unlike mass spectrometry,
Introduction The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants Spectroscopy and the Electromagnetic Spectrum Unlike mass spectrometry,
CHEMISTRY COURSE INFORMATION Chemistry Department, CB 213,
 CHEMISTRY COURSE INFORMATION Chemistry Department, CB 213, 784-6041 www.unr.edu/chemistry/advising Contents 1. Chemistry placement guidelines by program 2. Chemistry course prerequisites and mathematics
CHEMISTRY COURSE INFORMATION Chemistry Department, CB 213, 784-6041 www.unr.edu/chemistry/advising Contents 1. Chemistry placement guidelines by program 2. Chemistry course prerequisites and mathematics
Basic One- and Two-Dimensional NMR Spectroscopy
 Horst Friebolin Basic One- and Two-Dimensional NMR Spectroscopy Third Revised Edition Translated by Jack K. Becconsall WILEY-VCH Weinheim New York Chichester Brisbane Singapore Toronto Contents XV 1 The
Horst Friebolin Basic One- and Two-Dimensional NMR Spectroscopy Third Revised Edition Translated by Jack K. Becconsall WILEY-VCH Weinheim New York Chichester Brisbane Singapore Toronto Contents XV 1 The
COURSE DESCRIPTIONS CHEM 050 CHEM 101 CHEM 111 CHEM 112 CHEM 121 CHEM 253 CHEM 254 CHEM 275 CHEM 276 CHEM 277 CHEM 278 CHEM 299
 COURSE DESCRIPTIONS Note: Students may not register for a course lower in number than that required by their major. When a change in degree requirements forces a student to take a higher number course
COURSE DESCRIPTIONS Note: Students may not register for a course lower in number than that required by their major. When a change in degree requirements forces a student to take a higher number course
Lecture 6: Physical Methods II. UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy
 Lecture 6: Physical Methods II UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy Physical Methods used in bioinorganic chemistry X ray crystallography X ray absorption (XAS)
Lecture 6: Physical Methods II UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy Physical Methods used in bioinorganic chemistry X ray crystallography X ray absorption (XAS)
Georgia Gwinnett College CHEM 2212 Organic Chemistry II Course Syllabus Summer MTWR, 9-11am, A1640 (class); MTW, pm, A1290 (lab)
 Georgia Gwinnett College CHEM 2212 Organic Chemistry II Course Syllabus Summer 2008 Class Meetings: Instructor: Office: E-Mail: Phone Number: Wiki page: MTWR, 9-11am, A1640 (class); MTW, 1.15-4pm, A1290
Georgia Gwinnett College CHEM 2212 Organic Chemistry II Course Syllabus Summer 2008 Class Meetings: Instructor: Office: E-Mail: Phone Number: Wiki page: MTWR, 9-11am, A1640 (class); MTW, 1.15-4pm, A1290
Chemistry. Courses. Chemistry 1
 Chemistry 1 Chemistry Courses CHEM 1011 Chemistry in the Environment: 3 semester This course examines the role of chemistry in the environment and the application of chemistry to our understanding of society.
Chemistry 1 Chemistry Courses CHEM 1011 Chemistry in the Environment: 3 semester This course examines the role of chemistry in the environment and the application of chemistry to our understanding of society.
Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy
 Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy Mitesh Shrestha Electron Spin Resonance Electron paramagnetic
Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy Mitesh Shrestha Electron Spin Resonance Electron paramagnetic
Modern Optical Spectroscopy
 Modern Optical Spectroscopy With Exercises and Examples from Biophysics and Biochemistry von William W Parson 1. Auflage Springer-Verlag Berlin Heidelberg 2006 Verlag C.H. Beck im Internet: www.beck.de
Modern Optical Spectroscopy With Exercises and Examples from Biophysics and Biochemistry von William W Parson 1. Auflage Springer-Verlag Berlin Heidelberg 2006 Verlag C.H. Beck im Internet: www.beck.de
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis)
 Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Infrared Spectroscopy
 Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
INTRODUCTION TO MODERN VIBRATIONAL SPECTROSCOPY
 INTRODUCTION TO MODERN VIBRATIONAL SPECTROSCOPY MAX DIEM Department of Chemistry City University of New York Hunter College A Wiley-Interscience Publication JOHN WILEY & SONS New York Chichester Brisbane
INTRODUCTION TO MODERN VIBRATIONAL SPECTROSCOPY MAX DIEM Department of Chemistry City University of New York Hunter College A Wiley-Interscience Publication JOHN WILEY & SONS New York Chichester Brisbane
Kingdom of Saudi Arabia. The National Commission for Academic Accreditation & Assessment
 ATTACHMENT 2 (e) Course Specifications Kingdom of Saudi Arabia The National Commission for Academic Accreditation & Assessment Course Specifications (CS)!1 Course Specifications Institution Date University
ATTACHMENT 2 (e) Course Specifications Kingdom of Saudi Arabia The National Commission for Academic Accreditation & Assessment Course Specifications (CS)!1 Course Specifications Institution Date University
MODULE CONTENT YEAR TERM CREDITS TYPE. I - Fundamentals Chemistry BASIC. Postal address, telephone n o, address
 SUBJECT GUIDE Academic year 2015-2016 ORGANIC CHEMISTRY MODULE CONTENT YEAR TERM CREDITS TYPE I - Fundamentals Chemistry 1 1 6 BASIC LECTURER(S) Prof. Mónica Díaz Gavilán (MDG) Prof. Francisco Franco Montalbán
SUBJECT GUIDE Academic year 2015-2016 ORGANIC CHEMISTRY MODULE CONTENT YEAR TERM CREDITS TYPE I - Fundamentals Chemistry 1 1 6 BASIC LECTURER(S) Prof. Mónica Díaz Gavilán (MDG) Prof. Francisco Franco Montalbán
Protein Structure Analysis and Verification. Course S Basics for Biosystems of the Cell exercise work. Maija Nevala, BIO, 67485U 16.1.
 Protein Structure Analysis and Verification Course S-114.2500 Basics for Biosystems of the Cell exercise work Maija Nevala, BIO, 67485U 16.1.2008 1. Preface When faced with an unknown protein, scientists
Protein Structure Analysis and Verification Course S-114.2500 Basics for Biosystems of the Cell exercise work Maija Nevala, BIO, 67485U 16.1.2008 1. Preface When faced with an unknown protein, scientists
Spectroscopy Chapter 13
 Spectroscopy Chapter 13 Electromagnetic Spectrum Electromagnetic spectrum in terms of wavelength, frequency and Energy c=λν c= speed of light in a vacuum 3x108 m/s v= frequency in Hertz (Hz s-1 ) λ= wavelength
Spectroscopy Chapter 13 Electromagnetic Spectrum Electromagnetic spectrum in terms of wavelength, frequency and Energy c=λν c= speed of light in a vacuum 3x108 m/s v= frequency in Hertz (Hz s-1 ) λ= wavelength
CHEMISTRY. CHEM 0100 PREPARATION FOR GENERAL CHEMISTRY 3 cr. CHEM 0110 GENERAL CHEMISTRY 1 4 cr. CHEM 0120 GENERAL CHEMISTRY 2 4 cr.
 CHEMISTRY CHEM 0100 PREPARATION FOR GENERAL CHEMISTRY 3 cr. Course for students who intend to take CHEM 0110 later, but whose science and mathematics backgrounds are weak. Through intensive drill, this
CHEMISTRY CHEM 0100 PREPARATION FOR GENERAL CHEMISTRY 3 cr. Course for students who intend to take CHEM 0110 later, but whose science and mathematics backgrounds are weak. Through intensive drill, this
12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy
 12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy Determining the Structure of an Organic Compound The analysis of the outcome of a reaction requires that we know the full structure
12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy Determining the Structure of an Organic Compound The analysis of the outcome of a reaction requires that we know the full structure
COURSE: ADVANCED ORGANIC SYNTHESIS
 GENERAL CHARACTERISTICS * Type: Basic training, Compulsory Optional Final Project Work, External Practices Duration: Semester Semester / s: 1 Number of ECTS credits: 5 Language: Spanish, Catalán and English
GENERAL CHARACTERISTICS * Type: Basic training, Compulsory Optional Final Project Work, External Practices Duration: Semester Semester / s: 1 Number of ECTS credits: 5 Language: Spanish, Catalán and English
CHEMISTRY (CHE) CHE 104 General Descriptive Chemistry II 3
 Chemistry (CHE) 1 CHEMISTRY (CHE) CHE 101 Introductory Chemistry 3 Survey of fundamentals of measurement, molecular structure, reactivity, and organic chemistry; applications to textiles, environmental,
Chemistry (CHE) 1 CHEMISTRY (CHE) CHE 101 Introductory Chemistry 3 Survey of fundamentals of measurement, molecular structure, reactivity, and organic chemistry; applications to textiles, environmental,
ORGANIC CHEMISTRY. Wiley STUDY GUIDE AND SOLUTIONS MANUAL TO ACCOMPANY ROBERT G. JOHNSON JON ANTILLA ELEVENTH EDITION. University of South Florida
 STUDY GUIDE AND SOLUTIONS MANUAL TO ACCOMPANY ORGANIC CHEMISTRY ELEVENTH EDITION T. W. GRAHAM SOLOMONS University of South Florida CRAIG B. FRYHLE Pacific Lutheran University SCOTT A. SNYDER Columbia University
STUDY GUIDE AND SOLUTIONS MANUAL TO ACCOMPANY ORGANIC CHEMISTRY ELEVENTH EDITION T. W. GRAHAM SOLOMONS University of South Florida CRAIG B. FRYHLE Pacific Lutheran University SCOTT A. SNYDER Columbia University
7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text , , 12.10)
 2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
Spectroscopy of Polymers
 Spectroscopy of Polymers Jack L. Koenig Case Western Reserve University WOMACS Professional Reference Book American Chemical Society, Washington, DC 1992 Contents Preface m xiii Theory of Polymer Characterization
Spectroscopy of Polymers Jack L. Koenig Case Western Reserve University WOMACS Professional Reference Book American Chemical Society, Washington, DC 1992 Contents Preface m xiii Theory of Polymer Characterization
Calculate a rate given a species concentration change.
 Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Chapter 13. R.F.----µ-wave----I.R. (Heat)------Visible------U.V X-Ray------γ-Ray SPECTROSCOPY. Definition: Types to Be Covered:
 hamras Glendale ommunity ollege rganic hemistry 105 Exam 4 Materials hapter 13 SPETRSPY Definition: Types to Be overed: A) Infrared Spectroscopy (IR) B) Nuclear Magnetic Resonance Spectroscopy (NMR) )
hamras Glendale ommunity ollege rganic hemistry 105 Exam 4 Materials hapter 13 SPETRSPY Definition: Types to Be overed: A) Infrared Spectroscopy (IR) B) Nuclear Magnetic Resonance Spectroscopy (NMR) )
Homework Due by 5PM September 20 (next class) Does everyone have a topic that has been approved by the faculty?
 Howdy Folks. Homework Due by 5PM September 20 (next class) 5-Problems Every Week due 1 week later. Does everyone have a topic that has been approved by the faculty? Practice your presentation as I will
Howdy Folks. Homework Due by 5PM September 20 (next class) 5-Problems Every Week due 1 week later. Does everyone have a topic that has been approved by the faculty? Practice your presentation as I will
Spring Term 2012 Dr. Williams (309 Zurn, ex 2386)
 Chemistry 242 Organic Chemistry II Spring Term 2012 Dr. Williams (309 Zurn, ex 2386) Web Page: http://math.mercyhurst.edu/~jwilliams/ jwilliams@mercyhurst.edu (or just visit Department web site and look
Chemistry 242 Organic Chemistry II Spring Term 2012 Dr. Williams (309 Zurn, ex 2386) Web Page: http://math.mercyhurst.edu/~jwilliams/ jwilliams@mercyhurst.edu (or just visit Department web site and look
Circular Dichroism & Optical Rotatory Dispersion. Proteins (KCsa) Polysaccharides (agarose) DNA CHEM 305. Many biomolecules are α-helical!
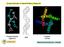 Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
3) In CE separation is based on what two properties of the solutes? (3 pts)
 Final Exam Chem 311 Fall 2002 December 16 Name 1) (3 pts) In GC separation is based on the following two properties of the solutes a) polarity and size b) vapor pressure and molecular weight c) vapor pressure
Final Exam Chem 311 Fall 2002 December 16 Name 1) (3 pts) In GC separation is based on the following two properties of the solutes a) polarity and size b) vapor pressure and molecular weight c) vapor pressure
Biochemistry 530 NMR Theory and Practice
 Biochemistry 530 NMR Theory and Practice Gabriele Varani Department of Biochemistry and Department of Chemistry University of Washington Lecturer: Gabriele Varani Biochemistry and Chemistry Room J479 and
Biochemistry 530 NMR Theory and Practice Gabriele Varani Department of Biochemistry and Department of Chemistry University of Washington Lecturer: Gabriele Varani Biochemistry and Chemistry Room J479 and
Electronic Spectra of Complexes
 Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
Alabama Department of Postsecondary Education
 Date Adopted: July 1, 1998 Date Reviewed: December 1, 1999 Date Revised: 1999, 2007, 2011 Alabama Department of Postsecondary Education Representing Alabama s Public Two-Year College System Jefferson State
Date Adopted: July 1, 1998 Date Reviewed: December 1, 1999 Date Revised: 1999, 2007, 2011 Alabama Department of Postsecondary Education Representing Alabama s Public Two-Year College System Jefferson State
The Fundamentals of Spectroscopy: Theory BUILDING BETTER SCIENCE AGILENT AND YOU
 The Fundamentals of Spectroscopy: Theory BUILDING BETTER SCIENCE AGILENT AND YOU 1 Agilent is committed to the educational community and is willing to provide access to company-owned material. This slide
The Fundamentals of Spectroscopy: Theory BUILDING BETTER SCIENCE AGILENT AND YOU 1 Agilent is committed to the educational community and is willing to provide access to company-owned material. This slide
Chemistry. Faculty Kent Davis, chair; Pablo Hilario, Denise Lee-Haye, Marie Pak, Robert Wilson Departmental Office: 355 Chan Shun Hall; (707)
 Faculty Kent Davis, chair; Pablo Hilario, Denise Lee-Haye, Marie Pak, Robert Wilson Departmental Office: 355 Chan Shun Hall; (707) 965-7597 Degrees and Programs Chemistry, B.S....77 Chemistry, B.A....78
Faculty Kent Davis, chair; Pablo Hilario, Denise Lee-Haye, Marie Pak, Robert Wilson Departmental Office: 355 Chan Shun Hall; (707) 965-7597 Degrees and Programs Chemistry, B.S....77 Chemistry, B.A....78
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II. 5 Credit Hours. Prepared by: Richard A. Pierce
 JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
on-line spectroscopy 1!!! Chemistry 1B Fall 2013 Chemistry 1B, Fall 2013 Lecture Spectroscopy Lectures Spectroscopy Fall 2013
 Lecture 190 Spectroscopy Flipping the lecture discussion Flip teaching (or flipped classroom) is a form of blended learning in which students learn new content online by watching video lectures, usually
Lecture 190 Spectroscopy Flipping the lecture discussion Flip teaching (or flipped classroom) is a form of blended learning in which students learn new content online by watching video lectures, usually
INSTRUMENTAL ANALYSIS-I
 Course Title Course Code INSTRUMENTAL ANALYSIS-I PH604 Lecture : 3 Course Credit Practical : 3 Tutorial : 0 Total : 6 Course Objectives On the completion of the course, students will be able to: 1. Understand
Course Title Course Code INSTRUMENTAL ANALYSIS-I PH604 Lecture : 3 Course Credit Practical : 3 Tutorial : 0 Total : 6 Course Objectives On the completion of the course, students will be able to: 1. Understand
Lectures Spectroscopy. Fall 2012
 Lectures 19-20 Spectroscopy Fall 2012 1 spectroscopic principles (Chem 1M/1N exps. #6 and #11) 4 1 spectroscopic excitations ( E = h = hc/ ; = c ) (nm) (sec -1 ) radiation technique molecular excitation
Lectures 19-20 Spectroscopy Fall 2012 1 spectroscopic principles (Chem 1M/1N exps. #6 and #11) 4 1 spectroscopic excitations ( E = h = hc/ ; = c ) (nm) (sec -1 ) radiation technique molecular excitation
COURSE DESCRIPTIONS. Vertically-related courses in this subject field are: Chem ;
 COURSE DESCRIPTIONS Ray von Wandruszka, Chair, Dept. of Chemistry (116 Malcolm M. Renfrew Hall, PO Box 442342, Moscow 83844-2343; phone 208/885-6552; chemoff@uidaho.edu). Note: Students may not register
COURSE DESCRIPTIONS Ray von Wandruszka, Chair, Dept. of Chemistry (116 Malcolm M. Renfrew Hall, PO Box 442342, Moscow 83844-2343; phone 208/885-6552; chemoff@uidaho.edu). Note: Students may not register
Lectures Spectroscopy. Fall 2012
 Lectures 19-20 Spectroscopy Fall 2012 1 spectroscopic principles (Chem 1M/1N exps. #6 and #11) 4 spectroscopic excitations ( E = h = hc/ ; = c ) (nm) (sec -1 ) radiation technique molecular excitation
Lectures 19-20 Spectroscopy Fall 2012 1 spectroscopic principles (Chem 1M/1N exps. #6 and #11) 4 spectroscopic excitations ( E = h = hc/ ; = c ) (nm) (sec -1 ) radiation technique molecular excitation
1.1 Is the following molecule aromatic or not aromatic? Give reasons for your answer.
 Page 1 QUESTION ONE 1.1 Is the following molecule aromatic or not aromatic? Give reasons for your answer. 1.2 List four criteria which compounds must meet in order to be considered aromatic. Page 2 QUESTION
Page 1 QUESTION ONE 1.1 Is the following molecule aromatic or not aromatic? Give reasons for your answer. 1.2 List four criteria which compounds must meet in order to be considered aromatic. Page 2 QUESTION
CHEMISTRY MAJOR. Suggested Course Sequence. 2nd-Class Year
 CHEMISTRY MAJOR Suggested Course Sequence 3rd-Class Year Biology 210 (BioChem) Chem 233 (All) Chem 234 (All) Chem 243 (All) Econ 201 (All) English 211 (All) Engr Mech 220 (All) Law 220 (All) Math 243 (All)
CHEMISTRY MAJOR Suggested Course Sequence 3rd-Class Year Biology 210 (BioChem) Chem 233 (All) Chem 234 (All) Chem 243 (All) Econ 201 (All) English 211 (All) Engr Mech 220 (All) Law 220 (All) Math 243 (All)
NPTEL/IITM. Molecular Spectroscopy Lectures 1 & 2. Prof.K. Mangala Sunder Page 1 of 15. Topics. Part I : Introductory concepts Topics
 Molecular Spectroscopy Lectures 1 & 2 Part I : Introductory concepts Topics Why spectroscopy? Introduction to electromagnetic radiation Interaction of radiation with matter What are spectra? Beer-Lambert
Molecular Spectroscopy Lectures 1 & 2 Part I : Introductory concepts Topics Why spectroscopy? Introduction to electromagnetic radiation Interaction of radiation with matter What are spectra? Beer-Lambert
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee
 Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 6 Spectroscopic Techniques Lecture - 2 UV-Visible Spectroscopy
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 6 Spectroscopic Techniques Lecture - 2 UV-Visible Spectroscopy
Course Home Page. Why is PHYSICS important in life science? Why is biophysics important?
 Course Home Page http://www.physics.nus.edu.sg/~bioph ysics/pc3267/indexa.html Password: pc3267 Please familiarize yourselves with the information there, especially with respect to the tutorials and other
Course Home Page http://www.physics.nus.edu.sg/~bioph ysics/pc3267/indexa.html Password: pc3267 Please familiarize yourselves with the information there, especially with respect to the tutorials and other
CHEMISTRY (CHEM) CHEM 5. Chemistry for Nurses. 5 Units. Prerequisite(s): One year high school algebra; high school chemistry
 Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 1A. General Chemistry I. 5 Units Prerequisite(s): High school chemistry and college algebra; sufficient performance on the college algebra diagnostic test, or equivalent;
Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 1A. General Chemistry I. 5 Units Prerequisite(s): High school chemistry and college algebra; sufficient performance on the college algebra diagnostic test, or equivalent;
Required Materials For complete material(s) information, refer to
 Butler Community College Science, Technology, Engineering, and Math Division Robert Carlson Revised Fall 2017 Implemented Spring 2018 COURSE OUTLINE Organic Chemistry 2 Course Description CH 245. Organic
Butler Community College Science, Technology, Engineering, and Math Division Robert Carlson Revised Fall 2017 Implemented Spring 2018 COURSE OUTLINE Organic Chemistry 2 Course Description CH 245. Organic
Chemistry Courses. Courses. Chemistry Courses 1
 Chemistry Courses 1 Chemistry Courses Courses CHEM 1105. Laboratory for CHEM 1305.. Prerequisite(s): (CHEM 1305 w/d or better) CHEM 1106. Laboratory for CHEM 1306.. Prerequisite(s): (CHEM 1306 w/d or better)
Chemistry Courses 1 Chemistry Courses Courses CHEM 1105. Laboratory for CHEM 1305.. Prerequisite(s): (CHEM 1305 w/d or better) CHEM 1106. Laboratory for CHEM 1306.. Prerequisite(s): (CHEM 1306 w/d or better)
CHM1001, 1002, 1051, <PHY1101, 1102, 1001, 1002> or <BIO1101, 1102, 1105, 1106> Total 16 cr.
 Major: (1) credits: - Multiple majors: minimum of 4 credits - Single major: minimum of 48 credits - Teacher training program: minimum of 51 credits () Required courses: 0 credits Required: CHM101, 151,
Major: (1) credits: - Multiple majors: minimum of 4 credits - Single major: minimum of 48 credits - Teacher training program: minimum of 51 credits () Required courses: 0 credits Required: CHM101, 151,
CHEMISTRY (CHEM) CHEM 1200 Problem Solving In General Chemistry
 Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 1090 Elementary Chemistry [0-3 credit hours (0-2, 0, 0-1)] For students who major in science, engineering or other fields which require chemistry as a prerequisite
Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 1090 Elementary Chemistry [0-3 credit hours (0-2, 0, 0-1)] For students who major in science, engineering or other fields which require chemistry as a prerequisite
CHE 230 ORGANIC CHEMISTRY I. (3) Fundamental principles and theories of organic chemistry. Prereq: CHE 107 and 115.
 104 INTRODUCTORY GENERAL MISTRY. (3) A study of the general principles including laws of definite and multiple proportions, stoichiometry, gases, electronic structure, chemical bonding, periodic relationships,
104 INTRODUCTORY GENERAL MISTRY. (3) A study of the general principles including laws of definite and multiple proportions, stoichiometry, gases, electronic structure, chemical bonding, periodic relationships,
COURSE UNIT DESCRIPTION. Type of the course unit. Mode of delivery Period of delivery Language of instruction Face to face Autumn English
 Course unit title Organic Chemistry I Lecturer(s) Dr. Rimantas Vaitkus COURSE UNIT DESCRIPTION Department Dept. Organic Chemistry, Vilnius University Cycle First Type of the course unit Mode of delivery
Course unit title Organic Chemistry I Lecturer(s) Dr. Rimantas Vaitkus COURSE UNIT DESCRIPTION Department Dept. Organic Chemistry, Vilnius University Cycle First Type of the course unit Mode of delivery
CHEM CHEMISTRY. Note: See beginning of Section H for abbreviations, course numbers and coding.
 CHEM CHEMISTRY Valid WHMIS (Workplace Hazardous Materials Information System) certification is required for all students who wish to take CHEM laboratory courses. WHMIS certification workshops will be
CHEM CHEMISTRY Valid WHMIS (Workplace Hazardous Materials Information System) certification is required for all students who wish to take CHEM laboratory courses. WHMIS certification workshops will be
Visible and IR Absorption Spectroscopy. Andrew Rouff and Kyle Chau
 Visible and IR Absorption Spectroscopy Andrew Rouff and Kyle Chau The Basics wavelength= (λ) original intensity= Ι o sample slab thickness= dl Final intensity= I f ε = molar extinction coefficient -di=
Visible and IR Absorption Spectroscopy Andrew Rouff and Kyle Chau The Basics wavelength= (λ) original intensity= Ι o sample slab thickness= dl Final intensity= I f ε = molar extinction coefficient -di=
Classification of spectroscopic methods
 Introduction Spectroscopy is the study of the interaction between the electromagnetic radiation and the matter. Spectrophotometry is the measurement of these interactions i.e. the measurement of the intensity
Introduction Spectroscopy is the study of the interaction between the electromagnetic radiation and the matter. Spectrophotometry is the measurement of these interactions i.e. the measurement of the intensity
Switching to OCR A from AQA
 Switching to OCR A from AQA The content within the OCR Chemistry A specification covers the key concepts of chemistry and will be very familiar. We ve laid it out in a logical progression to support co-teaching
Switching to OCR A from AQA The content within the OCR Chemistry A specification covers the key concepts of chemistry and will be very familiar. We ve laid it out in a logical progression to support co-teaching
Chiroptical Spectroscopy
 Chiroptical Spectroscopy Theory and Applications in Organic Chemistry Lecture 3: (Crash course in) Theory of optical activity Masters Level Class (181 041) Mondays, 8.15-9.45 am, NC 02/99 Wednesdays, 10.15-11.45
Chiroptical Spectroscopy Theory and Applications in Organic Chemistry Lecture 3: (Crash course in) Theory of optical activity Masters Level Class (181 041) Mondays, 8.15-9.45 am, NC 02/99 Wednesdays, 10.15-11.45
Advanced Studies in Structural Chemistry
 Advanced Studies in Structural Chemistry Biological Mass Spectrometry * 784634S Lectures 24 Exercises 6 4th spring 784627S Interpretation of Mass Spectra or the textbook: Davis, R. and Fearson, M.: Mass
Advanced Studies in Structural Chemistry Biological Mass Spectrometry * 784634S Lectures 24 Exercises 6 4th spring 784627S Interpretation of Mass Spectra or the textbook: Davis, R. and Fearson, M.: Mass
Prerequisites: CHEM 1312 and CHEM 1112, or CHEM 1412 General Chemistry II (Lecture and Laboratory)
 Course Syllabus CHEM 2423 Organic Chemistry I Revision Date: 8/19/2013 Catalog Description: Fundamental principles of organic chemistry will be studied, including the structure, bonding, properties, and
Course Syllabus CHEM 2423 Organic Chemistry I Revision Date: 8/19/2013 Catalog Description: Fundamental principles of organic chemistry will be studied, including the structure, bonding, properties, and
College of Science (CSCI) CSCI EETF Assessment Year End Report, June, 2017
 College of Science (CSCI) North Science 135 25800 Carlos Bee Boulevard, Hayward CA 94542 2016-2017 CSCI EETF Assessment Year End Report, June, 2017 Program Name(s) EETF Faculty Rep Department Chair Chemistry/Biochemistry
College of Science (CSCI) North Science 135 25800 Carlos Bee Boulevard, Hayward CA 94542 2016-2017 CSCI EETF Assessment Year End Report, June, 2017 Program Name(s) EETF Faculty Rep Department Chair Chemistry/Biochemistry
Chemistry Brigham Young University Idaho
 Department of Chemistry Chemistry Introduction Chemistry is the study of matter, energy, and their transformations. The principles of this discipline serve as a theoretical basis for a wide variety of
Department of Chemistry Chemistry Introduction Chemistry is the study of matter, energy, and their transformations. The principles of this discipline serve as a theoretical basis for a wide variety of
4.3A: Electronic transitions
 Ashley Robison My Preferences Site Tools Popular pages MindTouch User Guide FAQ Sign Out If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it
Ashley Robison My Preferences Site Tools Popular pages MindTouch User Guide FAQ Sign Out If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it
Keynotes in Organic Chemistry
 Keynotes in Organic Chemistry Second Edition ANDREW F. PARSONS Department of Chemistry, University of York, UK Wiley Contents Preface xi 1 Structure and bonding 1 1.1 Ionic versus covalent bonds 1 1.2
Keynotes in Organic Chemistry Second Edition ANDREW F. PARSONS Department of Chemistry, University of York, UK Wiley Contents Preface xi 1 Structure and bonding 1 1.1 Ionic versus covalent bonds 1 1.2
CHEMISTRY (CHEM) CHEM 5. Chemistry for Nurses. 5 Units. Prerequisite(s): One year high school algebra; high school chemistry
 Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 1A. General Chemistry I. 5 Units Prerequisite(s): High school chemistry and college algebra; sufficient performance on the college algebra diagnostic test, or equivalent;
Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 1A. General Chemistry I. 5 Units Prerequisite(s): High school chemistry and college algebra; sufficient performance on the college algebra diagnostic test, or equivalent;
SUBJECT: ADVANCED ORGANIC SYNTHESIS
 Page 1 of 8 GENERAL CHARACTERISTICS * Type: DESCRIPTION Basic training, Compulsory Optional Final Project Work, External Practices Duration: Semester Semester / s: 1 Number of ECTS credits: 5 Language:
Page 1 of 8 GENERAL CHARACTERISTICS * Type: DESCRIPTION Basic training, Compulsory Optional Final Project Work, External Practices Duration: Semester Semester / s: 1 Number of ECTS credits: 5 Language:
Nuclear magnetic resonance spectroscopy II. 13 C NMR. Reading: Pavia Chapter , 6.7, 6.11, 6.13
 Nuclear magnetic resonance spectroscopy II. 13 NMR Reading: Pavia hapter 6.1-6.5, 6.7, 6.11, 6.13 1. General - more/better/additional structural information for larger compounds -problems: a) isotopes
Nuclear magnetic resonance spectroscopy II. 13 NMR Reading: Pavia hapter 6.1-6.5, 6.7, 6.11, 6.13 1. General - more/better/additional structural information for larger compounds -problems: a) isotopes
Graduate Course Offerings
 Graduate Course Offerings CHM 400. First Year Graduate Seminar Course (0) First year graduate student seminar course and introduction to research. Topics include: research opportunities in the department,
Graduate Course Offerings CHM 400. First Year Graduate Seminar Course (0) First year graduate student seminar course and introduction to research. Topics include: research opportunities in the department,
CHEMISTRY (CHEMSTRY) Prereqs/Coreqs: P: math placement score of 15 or higher Typically Offered: Fall/Spring
 Chemistry (CHEMSTRY) 1 CHEMISTRY (CHEMSTRY) CHEMSTRY 1010 Introduction to College Life for Chemistry Majors 1 Credit This course is designed to provide a student with some of the academic and social skills
Chemistry (CHEMSTRY) 1 CHEMISTRY (CHEMSTRY) CHEMSTRY 1010 Introduction to College Life for Chemistry Majors 1 Credit This course is designed to provide a student with some of the academic and social skills
Tentative Chem 125 Schedule
 Tentative Chem 125 Schedule Introduction Monday, Aug 26 th As we have a shortened class schedule on the first day, we are move to ASC 202 & 204 to where you will take the Colorado Survey, log into Connect,
Tentative Chem 125 Schedule Introduction Monday, Aug 26 th As we have a shortened class schedule on the first day, we are move to ASC 202 & 204 to where you will take the Colorado Survey, log into Connect,
Chemistry 200: Basic Chemistry and Applications Course Syllabus: Spring
 Chemistry 200: Basic Chemistry and Applications Course Syllabus: Spring 2017 2018 Course Instructors Faraj Hasanayn; Office of Faraj Hasanayn: Chem Bldg. Rm 522 Office Hours: TBA Email: fh19@aub.edu.lb.
Chemistry 200: Basic Chemistry and Applications Course Syllabus: Spring 2017 2018 Course Instructors Faraj Hasanayn; Office of Faraj Hasanayn: Chem Bldg. Rm 522 Office Hours: TBA Email: fh19@aub.edu.lb.
ORGAN IC STRUCTURAL SPECTROSCOPY
 ORGAN IC STRUCTURAL SPECTROSCOPY JOSEPH B. LAMBERT Northwestern University HERBERT F. SHURVELL Queen's University DAVID A. LIGHTNER University of Nevada at Reno R. GRAHAM COOKS Purdue University Prentice
ORGAN IC STRUCTURAL SPECTROSCOPY JOSEPH B. LAMBERT Northwestern University HERBERT F. SHURVELL Queen's University DAVID A. LIGHTNER University of Nevada at Reno R. GRAHAM COOKS Purdue University Prentice
CHEM2077 HONORS ORGANIC CHEMISTRY SYLLABUS
 CHEM2077 HONORS ORGANIC CHEMISTRY SYLLABUS 1. STRUCTURE AND BONDING a] Atomic structure and bonding b] Hybridization and MO Theory c] Drawing chemical structures 2. POLAR COVALENT BONDS: ACIDS AND BASES
CHEM2077 HONORS ORGANIC CHEMISTRY SYLLABUS 1. STRUCTURE AND BONDING a] Atomic structure and bonding b] Hybridization and MO Theory c] Drawing chemical structures 2. POLAR COVALENT BONDS: ACIDS AND BASES
CH Exam #4 (Take Home) Date Due: 11/25,26/2013
 CH2710 - Exam #4 (Take Home) Date Due: 11/25,26/2013 Section I - Multiple Choice - Choose the BEST answer from the choices given and place the letter of you choice in the space provided. 1. Energy absorbed
CH2710 - Exam #4 (Take Home) Date Due: 11/25,26/2013 Section I - Multiple Choice - Choose the BEST answer from the choices given and place the letter of you choice in the space provided. 1. Energy absorbed
CHEMISTRY (CHEM) CHEM Fundamentals of Biochemistry
 Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 100 - Introductory Chemistry An introductory lecture and laboratory course in modern chemistry for non-science majors intended to make chemistry understandable,
Chemistry (CHEM) 1 CHEMISTRY (CHEM) CHEM 100 - Introductory Chemistry An introductory lecture and laboratory course in modern chemistry for non-science majors intended to make chemistry understandable,
