1. The idea that matter is made from atoms can be traced to the early Greek philosophers.
|
|
|
- Marvin McKinney
- 5 years ago
- Views:
Transcription
1 UNIT 3 WHAT IS THE BASIC STRUCTURE OF THE ATOM? (pp 52-53) Atomic Theory 1. The idea that matter is made from atoms can be traced to the early Greek philosophers. 2. The first theories were explanations based on their own life experiences so they concluded that matter was composed of air, earth, fire, and water. 3. One philosopher, DEn'10Ce.lil)J, believed otherwise and proposed that matter was composed of tiny particles called atomos (atoms). His ideas were: - Matter is composed of empty space through which atoms move. - Atoms are solid, homogeneous, indestructible and indivisible. - Different kinds of atoms have different sizes and shapes. - The differing properties of matter are due to the size, shape and movement of atoms. - Apparent changes in matter result from changes in the groupings of atoms and not from changes in the atoms themselves. 4. Unfortunately, Aristotle did not agree. Since he was more influential the ideas of Democritus were discarded and the air, earth, fire and water theory lasted much longer. Evidence Supporting Atomic Theory I. Observations of compounds and chemical reactions led to three laws that describe how compounds are formed. :DE'FI~r~ 2. U~~J Cl= COfJrVtS 17K).") states that a compound contains the same elements in exactly the same proportions by mass regardless of the size of the sample or source of the compound. C lc 1-IIl.C\ c CO,>J.St:IV/A flo,,j 0 I:: 3. L0,t.,v OJ;:: rvv t~s states that when two or more elements react to produce a compound, the total mass of the compound is the same as the sum of the masses of the individual elements. 3 (S) -\ 0,,1 ~9) --7 5D;; 19) Il(()Ln,:J l!; 4. U'l,J Of.=: fr,(j/br..i7l~jj applies to different compounds made from the same elements. The mass ratio for one of the elements that combines with a fixed mass of the other element can be expressed in small whole numbers. H:O If._ 0,- H',O cd '. I OVER
2 .John Dalton 1. Showed that, collectively, the laws demonstrate the existence of atoms. He argued that these laws could not hold true if atoms did not exist. 2. This led to the development of Dalton's Atomic Theory: - all matter is made up of indivisible and indestructible atoms. - atoms of a given element are identical in their physical and chemical properties. - atoms of different elements have different physical and chemical properties. - atoms of different elements combine in simple, whole number ratios to form chemical compounds. - atoms cannot be subdivided, created or destroyed when they are combined, separated or rearranged in chemical reactions. THE STRUCTURE OF THE ATOM (pp ) Electrons I. Scientists studying electrical current discovered that current would flow from the negative cathode to the positive cathode in a tube that was a near vacuum. 2. The remaining gas would glow and the beam was capable of moving a small paddle wheel. 3. This suggested the cathode ray consists of small individual particles. 4. J.J. Thomson studied the cathode ray. He discovered magnetic and electrical fields could deflect the rays. By carefully measuring the effect of the fields, Thomson calculated the CHfl'l.0E - TV~ 1/f1,q.~iSIl.q16 the charged particles. 5. Thomson compared his results with other known information and found that his particles were smaller than an atom. He had identified the first subatomic particle: the ELECn2().J 6. In 1909, Robert Millikan conducted an experiment, known as the () I L.. D!WP /E,./)Qe,irlll::7VT. This experiment determined the actual charge on the electron. This was set at.- I 7. He then used this information and Thomson's work to calculate the actual mass of an electron. ~ ly nvi SS O~ e ~ o, JJ 'J.. 10 a- 8. Since matter is neutrally charged, atoms must be neutral. But if all atoms contain electrons, how could this be? 9. Thomson theorized that the atom was a ball of positive charge with the negatively charged electrons embedded in the sides. This model became known as the PL1j (]r) PUDDIJoJG 1.(00&:'1-. Nucleus I. In 1911, Ernest Rutherford and a group of scientists set out to study how alpha particles would interact with solid matter. Alpha particles are radioactive and consist of the nucleus of the helium atom. Therefore, the particle has a positive charge.
3 2. They decided to fire a beam of alpha particles at a thin sheet of gold foil. A screen coated with zinc sulfide surrounded the foil. The thought was that the particles would cause a flash of light on the screen when they struck it. 3. Rutherford, knowing about Thomson's model, thought the particles would be deflected slightly, ifat all. 4. He discovered that some of the particles had very large deflections. Some even came straight back toward the source of the alpha particles. 5. Because of this he concluded the plum pudding model was incorrect and set out to develop a new model. 6. Rutherford's model states the atom is mostly empty space through which the electrons travel. He concluded there is a tiny, dense region of positive charge, which he called the rjvclt:.::",s. The electrons travel around the nucleus. 7. Rutherford also concluded that most of the atom's mass resides in the nucleus. Protons and Neutrons I. Rutherford refined his model and concluded the nucleus contained positively charged particles called Pt.;; '11f,",j,s 2. The proton's charge is equal to, but opposite that of an electron. 3. Scientists still had a dilemma because the mass of the known elements, except hydrogen, had been found to be heavier than that predicted by the protons and electrons. 4. In I932, James Chadwick showed that the nucleus contained another particle. This particle had the same mass as the proton, but had no charge. It was called a oi/el//lloa/ PROPERTIES OF SUBATOMIC PARTICLES PARTICLE SYMBOL LOCATION RELATIVE, RELATIVE ACTUAL CHARGE MASS MASS (2:) t'(i..~1.,_\ electron _L. e -.,.. -u,\j(,'cu;v-'s /3'1/0 q.if'f.if) proton (J -i- '.,-,l.'1 {'JIX..l8J..I +1 I (, fc 7 '00 neutron o,\) v ~2.'1 Cl!:l.JS ci I I, It; 7 't./o n HOW ATOMS DIFFER Atomic Number l. Not long after Rutherford's gold foil experiment, one of his fellow scientists beamed X rays at samples of the elements and discovered that the wavelength of the resultant X rays decreased with increasing atomic weight. 2. This scientist, Henry Moseley, concluded that the number of electrons around the nucleus must have been increasing. If this were so, then the number of protons in the nucleus must also increase in order to maintain a neutral charge. 3. Atoms of different elements contain a unique positive charge and therefore a unique number of protons in the nucleus. OVER
4 4. The number of protons in the nucleus is referred to as the A TDml C tj u mbe)t 5. If one knows the atomic number, one also knows the number of electrons present since the atom is neutral. Isotopes and Mass Number (pp ) 1. While the number of protons in an atom is fixed, the number of neutrons in the nucleus can vary. Even for the same element. 2. Atoms of the same element that have the same umber of protons, but different neutrons are called.isoto PES 3. Despite this, isotopes still have very similar chemical properties. 4. To make it easier to identify each of the various isotopes of an element, chemists add a number after each's name. The number is called the MASS tj\,1()1tlb7l fi,~i\ss AlVfV/,(j(:;71:= #,4e1.Jf7),.,J.S -+ # ;Jf=lJ77Z0dS ***** Determine the mass number for the isotope of potassium containing 19 protons and 20 neutrons. Write its name. ***** Determine the mass number of the isotope of mercury containing 80 protons and 124 neutrons. Write its name. tv\ A s.s J:t z: I ;;llf ;; ao If 5. An isotope can also be represented by a symbol. It looks like: ***** Write the isotopic symbol for each of the following: uranium-238 Th ' IS sym b0i' IS k nown as an.s: "...so TOm~ "-_'{_I... 3Ynl8bL; _ ':0 (\""""" h".,\ss tt iron r: J7 re ca\cium-46
5 ***** One of the most harmful components of nuclear waste is a radioactive isotope ofstrontium-90. It can be deposited in your bones where it replaces the calcium. Write the isotopic symbol for strontium-90. How many protons are in the nucleus? How many neutrons? p,ectojj-s,,; 3i qo.s. q ~(' S ;18 r.08)~1--j5 z: (J - vcr-'=' ;> Mass of an Atom I. Because the actual mass of a proton, neutron and electron are so small, the actual mass of an atom is a very small number. 2. These number are hard to work with so chemists developed a method of measuring the mass of an atom relative to the mass of a specifically chosen atomic standard. 3. The standard is the O.J.~otJ -/3. exactly 12 atomic mass units. 4. One f\tvil~1 ( n'y-\s.s UN lr (all1ujs equal to atom. Scientists assigned the carbon-12 atom a mass of.l I d the mass of the carbon-12 atom. 5. The atomic masses of the elements are listed on the periodic table, but they are not whole numbers. 6. This is because the ATlln1lt W/-4SJ.. of an element is the weighted average mass of that element. I<.8-ADVt: 7. Weighted averages depend on two factors: the VI'1 As.s and the A~\J.uMI\.I CE of each isotope: (n-v\>1) ( ~tl. (~S(}.J) + (iw\$5) er.a. ~'~IiN).1 A'T1)i1'liC n114ss -= It) 0 ***** Calculate the average atomic mass for silicon. Silicon has three naturally occurring isotopes: silicon-28 silicon-29 silicon amu 28/98 amu amu ,~ ~ % 4.700% 3.090% log ( OJ y, 17) ( a. (I 9t;!) ***** Calculate the atomic mass of oxygen. Oxygen has three naturally occurring isotopes: oxygen-l 6 oxygen-i 7 oxygen-i amu amu amu 99.76% % % o = /00 IS,qCjaI'Vlti OVER
6 CALCULA nons AND THE RULES FOR SIGNIFICANT FIGURES I. Each measured quantity has some degree of error in it. When measurements are added, subtracted, multiplied and divided small error may either cancel each other or add up to a larger error. 2. To avoid the possibility of adding extra error, rules for significant figures are used in calculations. The rules for addition and subtractions are different from the rules for multiplication and division. 3. When adding or subtracting: The answer can have no more digits to the right of the decimal point than there are in the measurement with the smallest number of digits to the right of the decimal point. ***** Add the following number together and round the answer to the proper number of significant figures: : &;)0,'-/;)1.:: ;;: Vd,{P z; &3 4. When multiplying or dividing: The answer can have no more significant figures than there are in the measurement with the smallest number of significant figures. * * * * * Perform the following calculations and express the answer in the proper number of significant figures:. (l2.257)x(1.162) ~ IlL ;Jl(,J(P z: (5.61) x (7.891) [(0.871)x(0.23)]+(5.871)- O.() 3lf/&IQSSq s: o,03v ***** Perform the following calculations and express the answer in the correct number of significant figures : <(t:,fbis'= 1./0" ;0,3/,'-1'.:: /3/ :-: tl,yn.fof~{q.: '/. V:) [(257) x(3.1)] +(547) s: f,!/s~i/rc;qi/j: I. S
7 [(12.4) x (7.943)] [(246.83) + (26.3)] :: 3"; ::,p-. C If SCIENTIFIC NOTATION 1. Sometimes known as exponential notation. 2. This system helps us avoid the uncertainty of whether zeros at the end of a number are significant. 3. The exponent tells us how many decimal places to move the decimal point. The sign (+) or (-) tells us whether to move the decimal point to the left or the right. 4. Scientific notation is a method for making very large or very small numbers more compact and easier to write. 5. It expresses a number as a :-,1,-~2=:,O,-::'D::-l_~C..:... -=-' ofa number between 1_4--,--"--'_'.::. 1_0 and the appropriate "(0 j,~t:;1l.. of 10 _ ***** q, ;3 X {O 7 6. When the decimal point is moved to the Ll.:;.i=='T, the power often is PO.:iITlifl: ***** (), 0001'<>7 ::: 7. When the decimal point is moved ill 0 HT, the power often is tj. E6177~/t7 ***** Express the following numbers to three significant figures in scientific notation:, ~ ~ 3,500,000 3, SO'/. ( /.Jl( Y If s. s o '/ , To convert a number expressed in scientific notation into its normal expanded form simply move the decimal point the appropriate number of spaces in the direction indicated by the sign on the power often. ***** Express the following numbers in their normal expanded form X X 10-3 C) OVER ~ _----_... _ ~ ----
8 9. To add or subtract numbers expressed in scientific notation, the exponents must be the same: 5.3 ;J,OO "" oo~IO..:3 :3 ::;.;J ~ o. 0...,.. I Y.. I 0 ~ a. BO 'f... (0 10. When multiplying numbers expressed in scientific notation, carry out the operation and then add the exponents together..3 (3,O'j;10 5 )(4,O"'f.IO-'2..)-:.,~ 1-.,0 z: /.~"'IOIf -,),) ( 4) (I.O~~'O J.b? "f...fo :: Q).:J. } L/ X When dividing, carry out the operation and then subtract the exponent in the denominator from the exponent in the numerator.,/ --if 1?,O'IC(O-~ &'.O'f...IO -l/_(-l.} = -2 :. ';;,0)",10 ;."",'0-2- f,foyio When raising a number expressed in scientific notation to a power, carry out the operation, then multiply the exponent by the power. -~ ~. -~ (,?, 0 -.,../0) z: (o~ 'f.. 10 :: 0.l(X/O -1 3) 3 (q. (03,'" fo = qq, cl S OlJCf7 X 10 7 = qq,,3 '/.10 i 10
Early Atomic Models. Atoms: the smallest particle of an element that retains the properties of that element.
 Chapter 5 Early Atomic Models Atoms: the smallest particle of an element that retains the properties of that element. (Greek: atomos = indivisible) Democritus (Greek teacher in the 4 th century BC) First
Chapter 5 Early Atomic Models Atoms: the smallest particle of an element that retains the properties of that element. (Greek: atomos = indivisible) Democritus (Greek teacher in the 4 th century BC) First
Chapter 4 Atomic Structure. Chemistry- Lookabaugh Moore High School
 Chapter 4 Atomic Structure Chemistry- Lookabaugh Moore High School Section 4.1 Defining the Atom Democritus (460 B.C 370 B.C.) first used the term atomon to describe the smallest particle of matter possible.
Chapter 4 Atomic Structure Chemistry- Lookabaugh Moore High School Section 4.1 Defining the Atom Democritus (460 B.C 370 B.C.) first used the term atomon to describe the smallest particle of matter possible.
Chemistry. - Many philosophers concluded that matter was composed of things such as earth, -Democritus: -Aristotle: -Dalton:
 Chemistry Made by Saleem Abu-Tayeh Chapter 4 - Many philosophers concluded that matter was composed of things such as earth, water, air, and fire. It was also commonly accepted that matter could be endlessly
Chemistry Made by Saleem Abu-Tayeh Chapter 4 - Many philosophers concluded that matter was composed of things such as earth, water, air, and fire. It was also commonly accepted that matter could be endlessly
Chapter 4: Atomic Structure Section 4.1 Defining the Atom
 Chapter 4: Atomic Structure Section 4.1 Defining the Atom Early Models of the Atom atom the smallest particle of an element that retains its identity in a chemical reaction Democritus s Atomic Philosophy
Chapter 4: Atomic Structure Section 4.1 Defining the Atom Early Models of the Atom atom the smallest particle of an element that retains its identity in a chemical reaction Democritus s Atomic Philosophy
Chapter 4. Atomic Structure
 Chapter 4 Atomic Structure Warm Up We have not yet discussed this material, but what do you know already?? What is an atom? What are electron, neutrons, and protons? Draw a picture of an atom from what
Chapter 4 Atomic Structure Warm Up We have not yet discussed this material, but what do you know already?? What is an atom? What are electron, neutrons, and protons? Draw a picture of an atom from what
DescribeDemocritus s Democritus s ideas
 Atomic Structure Section 4.1 Defining the Atom DescribeDemocritus s Democritus s ideas about atoms. Section 4.1 Defining the Atom Explain Dalton s atomic theory. Section 4.1 Defining the Atom Identifywhat
Atomic Structure Section 4.1 Defining the Atom DescribeDemocritus s Democritus s ideas about atoms. Section 4.1 Defining the Atom Explain Dalton s atomic theory. Section 4.1 Defining the Atom Identifywhat
Atomic Structure. ppst.com
 Atomic Structure ppst.com Defining the Atom The Greek philosopher (460 B.C. 370 B.C.) was among the first to suggest the existence of atoms (from the Greek word ) He believed that atoms were and His ideas
Atomic Structure ppst.com Defining the Atom The Greek philosopher (460 B.C. 370 B.C.) was among the first to suggest the existence of atoms (from the Greek word ) He believed that atoms were and His ideas
Chapter 4. The structure of the atom. AL-COS Objectives 1, 2,3,4,7, 10, 15, 20, 21, 22, 27and 28
 Chapter 4 The structure of the atom AL-COS Objectives 1, 2,3,4,7, 10, 15, 20, 21, 22, 27and 28 You ll learn to Identify the experiments that led to the development of the nuclear model of atomic structure
Chapter 4 The structure of the atom AL-COS Objectives 1, 2,3,4,7, 10, 15, 20, 21, 22, 27and 28 You ll learn to Identify the experiments that led to the development of the nuclear model of atomic structure
Get out your diagram from your research paper. Get out a sheet of paper to take some notes on.
 Bellwork: Get out your diagram from your research paper. Get out a sheet of paper to take some notes on. Fill in the Following Table in your notes (assume an atom unless otherwise stated: Symbol Protons
Bellwork: Get out your diagram from your research paper. Get out a sheet of paper to take some notes on. Fill in the Following Table in your notes (assume an atom unless otherwise stated: Symbol Protons
Atomic Structure. For thousands of years, people had many ideas about matter Ancient Greeks believed that everything was made up of the four elements
 An atom is the smallest particle of an element that retains its identity in a chemical reaction. Although early philosophers and scientists could not observe individual atoms, they were still able to propose
An atom is the smallest particle of an element that retains its identity in a chemical reaction. Although early philosophers and scientists could not observe individual atoms, they were still able to propose
Origins of the Atom. Atoms: The Building Blocks of Matter. Let s Get Ready to Rumble. Aristotle s Theory of the Atom CHAPTER 3
 Origins of the Atom CHAPTER 3 Atoms: The Building Blocks of Matter Let s Get Ready to Rumble The idea of the atom was met with great skepticism, especially among great thinkers. The most vocal critic of
Origins of the Atom CHAPTER 3 Atoms: The Building Blocks of Matter Let s Get Ready to Rumble The idea of the atom was met with great skepticism, especially among great thinkers. The most vocal critic of
Dalton Thompson Rutherford Bohr Modern Model ("Wave. Models of the Atom
 Dalton Thompson Rutherford Bohr Modern Model ("Wave Models of the Atom Mechanical" Model) Aim: To discuss the scientists and their contributions to the current atomic model. Focus: Rutherford's Gold Foil
Dalton Thompson Rutherford Bohr Modern Model ("Wave Models of the Atom Mechanical" Model) Aim: To discuss the scientists and their contributions to the current atomic model. Focus: Rutherford's Gold Foil
Chemistry Chapter 3. Atoms: The Building Blocks of Matter
 Chemistry Chapter 3 Atoms: The Building Blocks of Matter I. From Philosophical Idea to Scientific Theory History of the Atom The Ancient Greeks were the first to come up with the idea of the atom. Democritus
Chemistry Chapter 3 Atoms: The Building Blocks of Matter I. From Philosophical Idea to Scientific Theory History of the Atom The Ancient Greeks were the first to come up with the idea of the atom. Democritus
CHAPTER 3. Atoms: The Building Blocks of Matter
 CHAPTER 3 Atoms: The Building Blocks of Matter Origins of the Atom Democritus: Greek philosopher (460 BC - 370 BC) Coined the term atom from the Greek word atomos Democritus believes that atoms were indivisible
CHAPTER 3 Atoms: The Building Blocks of Matter Origins of the Atom Democritus: Greek philosopher (460 BC - 370 BC) Coined the term atom from the Greek word atomos Democritus believes that atoms were indivisible
SNC1D1 History of the Atom
 SNC1D1 History of the Atom What is the atom? Atoms are the building block for all matter: Atoms make up elements! Elements combine to make compounds!2 ATOMIC MODEL TIMELINE 400 B.C PRESENT DAY ATOMIC MODEL
SNC1D1 History of the Atom What is the atom? Atoms are the building block for all matter: Atoms make up elements! Elements combine to make compounds!2 ATOMIC MODEL TIMELINE 400 B.C PRESENT DAY ATOMIC MODEL
4-1 Notes. Defining the Atom
 4-1 Notes Defining the Atom Early Models of the Atom All matter is composed of atoms Atoms are the smallest particles of an element that retains their identity in a chemical reaction Greek philosopher
4-1 Notes Defining the Atom Early Models of the Atom All matter is composed of atoms Atoms are the smallest particles of an element that retains their identity in a chemical reaction Greek philosopher
CHEMISTRY. Matter and Change. Table Of Contents. Section 4.1 Early Ideas About Matter. Unstable Nuclei and Radioactive Decay
 CHEMISTRY 4 Table Of Contents Matter and Change Section 4.1 Early Ideas About Matter Chapter 4: The Structure of the Atom Section 4.2 Section 4.3 Section 4.4 Defining the Atom How Atoms Differ Unstable
CHEMISTRY 4 Table Of Contents Matter and Change Section 4.1 Early Ideas About Matter Chapter 4: The Structure of the Atom Section 4.2 Section 4.3 Section 4.4 Defining the Atom How Atoms Differ Unstable
Scientist wanted to understand how the atom looked. It was known that matter was neutral. It was known that matter had mass
 Atom Models Scientist wanted to understand how the atom looked It was known that matter was neutral It was known that matter had mass They used these ideas to come up with their models, however science
Atom Models Scientist wanted to understand how the atom looked It was known that matter was neutral It was known that matter had mass They used these ideas to come up with their models, however science
Chapter 3 https://youtu.be/thndxfdkzzs?list=pl8dpuualjx tphzzyuwy6fyeax9mqq8ogr
 Chapter 3 https://youtu.be/thndxfdkzzs?list=pl8dpuualjx tphzzyuwy6fyeax9mqq8ogr The smallest particle of an element that retains the chemical properties of that element. Regions: Nucleus: very small region
Chapter 3 https://youtu.be/thndxfdkzzs?list=pl8dpuualjx tphzzyuwy6fyeax9mqq8ogr The smallest particle of an element that retains the chemical properties of that element. Regions: Nucleus: very small region
Atoms and their structure
 Atoms and their structure History of atomic theory Not the history of atom, but the idea of the atom Original idea Ancient Greece (400 B.C..) Democritus and Leucippus Greek philosophers Another Greek Aristotle
Atoms and their structure History of atomic theory Not the history of atom, but the idea of the atom Original idea Ancient Greece (400 B.C..) Democritus and Leucippus Greek philosophers Another Greek Aristotle
History of Atomic Theory
 Unit 2 The Atom History of Atomic Theory A. Democritus and Aristotle Democritus named the "atom" - means indivisible Dalton (with work of Lavoisier, Proust, and Gay-Lussac) 1. atomic theory - first based
Unit 2 The Atom History of Atomic Theory A. Democritus and Aristotle Democritus named the "atom" - means indivisible Dalton (with work of Lavoisier, Proust, and Gay-Lussac) 1. atomic theory - first based
Chapter 3 Atoms: The Building Blocks of Matter. Honors Chemistry 412
 Chapter 3 Atoms: The Building Blocks of Matter Honors Chemistry 412 Foundations of Atomic Theory Democritus Greek Philosopher 460-370 B.C. Stated Matter could be divided into smaller & smaller particles
Chapter 3 Atoms: The Building Blocks of Matter Honors Chemistry 412 Foundations of Atomic Theory Democritus Greek Philosopher 460-370 B.C. Stated Matter could be divided into smaller & smaller particles
Democritus thought atoms were indivisible & indestructible Lacked experimental support 4 th century B.C.
 Chapter 5 Democritus thought atoms were indivisible & indestructible Lacked experimental support 4 th century B.C. Democritus thought atoms were indivisible & indestructible Lacked experimental support
Chapter 5 Democritus thought atoms were indivisible & indestructible Lacked experimental support 4 th century B.C. Democritus thought atoms were indivisible & indestructible Lacked experimental support
Chapter 4 The Atom. Philosophers and scientists have proposed many ideas on the structure of atoms.
 Chapter4 TheAtom 4.1 Early Models of the Atom An atom is the smallest particle of an element that retains its identity in a chemical reaction. Philosophers and scientists have proposed many ideas on the
Chapter4 TheAtom 4.1 Early Models of the Atom An atom is the smallest particle of an element that retains its identity in a chemical reaction. Philosophers and scientists have proposed many ideas on the
THE ATOM Pearson Education, Inc.
 THE ATOM Title and Highlight Right Side NOTES ONLY TN Ch 4.1-4.2 Topic: EQ: Date Reflect Question: Reflect on the material by asking a question (its not suppose to be answered from notes) NOTES: Write
THE ATOM Title and Highlight Right Side NOTES ONLY TN Ch 4.1-4.2 Topic: EQ: Date Reflect Question: Reflect on the material by asking a question (its not suppose to be answered from notes) NOTES: Write
Nuclear Chemistry. Atomic Structure Notes Start on Slide 20 from the second class lecture
 Nuclear Chemistry Atomic Structure Notes Start on Slide 20 from the second class lecture The Birth of an Idea Democritus, 400 B.C. coined the term atom If you divide matter into smaller and smaller pieces,
Nuclear Chemistry Atomic Structure Notes Start on Slide 20 from the second class lecture The Birth of an Idea Democritus, 400 B.C. coined the term atom If you divide matter into smaller and smaller pieces,
The Atom. protons, neutrons, and electrons oh my!
 The Atom protons, neutrons, and electrons oh my! What s an Atom? An atom is the smallest physical particle of an element that still retains the properties of that element. How Big is an Atom? At sea level,
The Atom protons, neutrons, and electrons oh my! What s an Atom? An atom is the smallest physical particle of an element that still retains the properties of that element. How Big is an Atom? At sea level,
CHEMISTRY 11 UNIT REVIEW: ATOMIC THEORY & PERIODIC TRENDS
 CHEMISTRY 11 UNIT REVIEW: ATOMIC THEORY & PERIODIC TRENDS Atoms Atoms have protons and neutrons located in the nucleus of the atom. Electrons orbit around the nucleus in well-defined paths. Protons have
CHEMISTRY 11 UNIT REVIEW: ATOMIC THEORY & PERIODIC TRENDS Atoms Atoms have protons and neutrons located in the nucleus of the atom. Electrons orbit around the nucleus in well-defined paths. Protons have
Name Date Class DEFINING THE ATOM
 4.1 DEFINING THE ATOM Section Review Objectives Describe Democritus s ideas about atoms Explain Dalton s atomic theory Describe the size of an atom Vocabulary atom Dalton s atomic theory Part A Completion
4.1 DEFINING THE ATOM Section Review Objectives Describe Democritus s ideas about atoms Explain Dalton s atomic theory Describe the size of an atom Vocabulary atom Dalton s atomic theory Part A Completion
General Chemistry Standard : Identify the significance of the various outcomes of Thomson s and Rutherford s experiments
 Not the history of the atom, but the idea of the atom The atom was not discovered until recently Original Idea Ancient Greece (400 BC) Proposed by lesser-known scientists They looked at a beach made of
Not the history of the atom, but the idea of the atom The atom was not discovered until recently Original Idea Ancient Greece (400 BC) Proposed by lesser-known scientists They looked at a beach made of
1. Based on Dalton s evidence, circle the drawing that demonstrates Dalton s model.
 Various models of the ATOM Dalton Model John Dalton developed the first atomic model in 1808. Before him people, mostly philosophers, had speculated about the smallest unit of matter and two theories prevailed.
Various models of the ATOM Dalton Model John Dalton developed the first atomic model in 1808. Before him people, mostly philosophers, had speculated about the smallest unit of matter and two theories prevailed.
ATOMIC STRUCTURE. Name: Period: Date: 1) = a generalization of scientific observations that what happens (does explain)
 ATOMIC STRUCTURE Name: Period: Date: I. LAW vs. THEORY: 1) = a generalization of scientific observations that what happens (does explain) 2) (model) = a set of assumptions used to explain observations
ATOMIC STRUCTURE Name: Period: Date: I. LAW vs. THEORY: 1) = a generalization of scientific observations that what happens (does explain) 2) (model) = a set of assumptions used to explain observations
Greek Philosophers (cont.)
 Greek Philosophers (cont.) Many ancient scholars believed matter was composed of such things as earth, water, air, and fire. Many believed matter could be endlessly divided into smaller and smaller pieces.
Greek Philosophers (cont.) Many ancient scholars believed matter was composed of such things as earth, water, air, and fire. Many believed matter could be endlessly divided into smaller and smaller pieces.
Early Atomic Theory. Alchemy. The atom
 Early Atomic Theory Chapter 3 Democritus 460 BC- ~ 370 BC Nothing exists except atoms and empty space; everything else is opinion. Matter is composed of small indivisible particles, atomos meaning Indivisible
Early Atomic Theory Chapter 3 Democritus 460 BC- ~ 370 BC Nothing exists except atoms and empty space; everything else is opinion. Matter is composed of small indivisible particles, atomos meaning Indivisible
Development of Atomic Theory Elements of chemistry- Atoms, the building blocks of matter Video
 Development of Atomic Theory Elements of chemistry- Atoms, the building blocks of matter Video 2 CH 4- Atoms 1 Discovering the Atom In this lesson we will take a look at the scientists who explored the
Development of Atomic Theory Elements of chemistry- Atoms, the building blocks of matter Video 2 CH 4- Atoms 1 Discovering the Atom In this lesson we will take a look at the scientists who explored the
Topic III Quest Study Guide
 Topic III Quest Study Guide A. Early Concepts: Democritus: Democritus: Greek Philosopher 400 B.C. Matter is composed of atoms, which move through empty space Atoms are solid, homogeneous indestructible
Topic III Quest Study Guide A. Early Concepts: Democritus: Democritus: Greek Philosopher 400 B.C. Matter is composed of atoms, which move through empty space Atoms are solid, homogeneous indestructible
Democritus & Leucippus (~400 BC) Greek philosophers: first to propose that matter is made up of particles called atomos, the Greek word for atoms
 AP Chemistry Ms. Ye Name Date Block The Evolution of the Atomic Model Since atoms are too small to see even with a very powerful microscope, scientists rely upon indirect evidence and models to help them
AP Chemistry Ms. Ye Name Date Block The Evolution of the Atomic Model Since atoms are too small to see even with a very powerful microscope, scientists rely upon indirect evidence and models to help them
Early Models of the Atom
 Early Models of the Atom An atom is the smallest particle of an element that retains its identity in a chemical reaction. Although early philosophers and scientists could not observe individual atoms,
Early Models of the Atom An atom is the smallest particle of an element that retains its identity in a chemical reaction. Although early philosophers and scientists could not observe individual atoms,
Democritus 460 BC 370 BC. First scholar to suggest that atoms existed. Believed that atoms were indivisible and indestructible.
 Democritus 460 BC 370 BC First scholar to suggest that atoms existed. Believed that atoms were indivisible and indestructible. Democritus 460 BC 370 BC Problems with theory: 1. Did not explain chemical
Democritus 460 BC 370 BC First scholar to suggest that atoms existed. Believed that atoms were indivisible and indestructible. Democritus 460 BC 370 BC Problems with theory: 1. Did not explain chemical
Chapter 4. History of the atom. History of Atom Smallest possible piece? Atomos - not to be cut. Atoms and their structure
 Chapter 4 Atoms and their structure History of the atom Not the history of atom, but the idea of the atom. Original idea Ancient Greece (400 B.C.) Democritus and Leucippus Greek philosophers. Looked at
Chapter 4 Atoms and their structure History of the atom Not the history of atom, but the idea of the atom. Original idea Ancient Greece (400 B.C.) Democritus and Leucippus Greek philosophers. Looked at
The History of the Atom. How did we learn about the atom?
 The History of the Atom How did we learn about the atom? The Atomic Theory of Matter All matter is made up of fundamental particles. What does fundamental mean? The Greek Philosophers, 400 B.C. Democritus
The History of the Atom How did we learn about the atom? The Atomic Theory of Matter All matter is made up of fundamental particles. What does fundamental mean? The Greek Philosophers, 400 B.C. Democritus
4.2 Structure of the Nuclear Atom > Chapter 4 Atomic Structure. 4.2 Structure of the Nuclear Atom. 4.1 Defining the Atom
 Chapter 4 Atomic Structure 4.1 Defining the Atom 4.2 Structure of the Nuclear Atom 4.3 Distinguishing Among Atoms 1 CHEMISTRY & YOU How did scientists determine the structures that are inside an atom?
Chapter 4 Atomic Structure 4.1 Defining the Atom 4.2 Structure of the Nuclear Atom 4.3 Distinguishing Among Atoms 1 CHEMISTRY & YOU How did scientists determine the structures that are inside an atom?
Chapter 11 Study Questions Name: Class:
 Chapter 11 Study Questions Name: Class: Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. The discovery of which particle proved that the atom
Chapter 11 Study Questions Name: Class: Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. The discovery of which particle proved that the atom
NOTES ON CHAPTER 4: ELEMENTS AND THE PERIODIC TABLE. 4.1 Introduction to Atoms
 NOTES ON CHAPTER 4: ELEMENTS AND THE PERIODIC TABLE 4.1 Introduction to Atoms The first people to think about the nature of matter were the ancient Greeks. Around 430 B.C, Democritus, a Greek philosopher,
NOTES ON CHAPTER 4: ELEMENTS AND THE PERIODIC TABLE 4.1 Introduction to Atoms The first people to think about the nature of matter were the ancient Greeks. Around 430 B.C, Democritus, a Greek philosopher,
Chemistry. Robert Taggart
 Chemistry Robert Taggart Table of Contents To the Student..................................................v Unit 1: Matter and Measurement Lesson 1: Chemistry and the Scientific Method...................3
Chemistry Robert Taggart Table of Contents To the Student..................................................v Unit 1: Matter and Measurement Lesson 1: Chemistry and the Scientific Method...................3
8.5 Atomic Structure
 Views of the Atom Democritus (Greek philosopher from 460 BC to 370 BC) Believed that atoms were indivisible and indestructible. His approach was simply philosophy and was not based upon the scientific
Views of the Atom Democritus (Greek philosopher from 460 BC to 370 BC) Believed that atoms were indivisible and indestructible. His approach was simply philosophy and was not based upon the scientific
Glencoe: Chapter 4. The Structure of the Atom
 Glencoe: Chapter 4 The Structure of the Atom Section One: Early Ideas about Matter Atomists and Democritus : 400 B.C. From Thrace in Greece. Atoms- Uncut-Table Indivisible parts which cannot be broken
Glencoe: Chapter 4 The Structure of the Atom Section One: Early Ideas about Matter Atomists and Democritus : 400 B.C. From Thrace in Greece. Atoms- Uncut-Table Indivisible parts which cannot be broken
Vocabulary QUIZ: 1. The total number of particles in the nucleus 2. 1 / 12
 Sep 29 11:29 AM Vocabulary QUIZ: 1. The total number of particles in the nucleus 2. 1 / 12 th of the mass of a carbon atom 3. The weighted average mass of all the isotopes of a particular element 4. A
Sep 29 11:29 AM Vocabulary QUIZ: 1. The total number of particles in the nucleus 2. 1 / 12 th of the mass of a carbon atom 3. The weighted average mass of all the isotopes of a particular element 4. A
DEMOCRITUS - A philosopher in the year 400 B.C. - He didn t do experiments and he wondered if atoms kept on being divided, that there would only be
 DEMOCRITUS A philosopher in the year 400 B.C. He didn t do experiments and he wondered if atoms kept on being divided, that there would only be one undividable particle left. He discovered that this was
DEMOCRITUS A philosopher in the year 400 B.C. He didn t do experiments and he wondered if atoms kept on being divided, that there would only be one undividable particle left. He discovered that this was
Passing an electric current makes a beam appear to move from the negative to the positive end.
 Chapter 4 Atoms and their structure History of the atom Not the history of atom, but the idea of the atom. Original idea Ancient Greece (400 B.C.) Democritus and Leucippus Greek philosophers. Smallest
Chapter 4 Atoms and their structure History of the atom Not the history of atom, but the idea of the atom. Original idea Ancient Greece (400 B.C.) Democritus and Leucippus Greek philosophers. Smallest
Physics 30 Modern Physics Unit: Atomic Basics
 Physics 30 Modern Physics Unit: Atomic Basics Models of the Atom The Greeks believed that if you kept dividing matter into smaller and smaller pieces, you would eventually come to a bit of matter that
Physics 30 Modern Physics Unit: Atomic Basics Models of the Atom The Greeks believed that if you kept dividing matter into smaller and smaller pieces, you would eventually come to a bit of matter that
Atomic Structure. 1. Democritus 2. Dalton 3. Thompson 4. Millikan 5. Moseley 6. Bohr 7. Rutherford 8. Schrodinger. October 03, 2014.
 October 03, 2014 Atomic Structure Chapter 4 Democritus's Atomic Philosophy 1. Democritus 2. Dalton 3. Thompson 4. Millikan 5. Moseley 6. Bohr 7. Rutherford 8. Schrodinger Aristotle -Rejected the idea of
October 03, 2014 Atomic Structure Chapter 4 Democritus's Atomic Philosophy 1. Democritus 2. Dalton 3. Thompson 4. Millikan 5. Moseley 6. Bohr 7. Rutherford 8. Schrodinger Aristotle -Rejected the idea of
Particle Theory of Matter. By the late 1700s, scientists had adopted the Particle Theory of Matter. This theory states that:
 Particle Theory of Matter By the late 1700s, scientists had adopted the Particle Theory of Matter. This theory states that: all matter is made up of very tiny particles each pure substance has its own
Particle Theory of Matter By the late 1700s, scientists had adopted the Particle Theory of Matter. This theory states that: all matter is made up of very tiny particles each pure substance has its own
9/23/2012. Democritus 400 B.C. Greek philosopher Proposed that all materials are made from atoms. Coined Greek word atmos, meaning indivisible.
 Mr. Sudbury Atoms are too small to see with your eyes. Atoms are too small to see with the most powerful microscopes. Scientist use models to explain atoms. A scientific model is an representation containing
Mr. Sudbury Atoms are too small to see with your eyes. Atoms are too small to see with the most powerful microscopes. Scientist use models to explain atoms. A scientific model is an representation containing
ATOM. Rich -Paradis. Early Thoughts Aristotle-- Continuous theory. Matter can be divided indefinitely. Greeks
 ATOM Early Thoughts Aristotle-- Continuous theory Greeks Matter can be divided indefinitely matter is made up of particles--4 elements 4 elements --air--fire--water- -- earth Democritus --Discontinuous
ATOM Early Thoughts Aristotle-- Continuous theory Greeks Matter can be divided indefinitely matter is made up of particles--4 elements 4 elements --air--fire--water- -- earth Democritus --Discontinuous
Chapter 4 Jeopardy Review
 Chapter 4 Jeopardy Review Atom Models of the Atom Atomic Theory Calculating Subatomic Particles Isotopes 100 100 100 100 100 200 200 200 200 200 300 300 300 300 300 400 400 400 400 400 500 500 500 500
Chapter 4 Jeopardy Review Atom Models of the Atom Atomic Theory Calculating Subatomic Particles Isotopes 100 100 100 100 100 200 200 200 200 200 300 300 300 300 300 400 400 400 400 400 500 500 500 500
CHAPTER 4 Atomic Structure
 CHAPTER 4 Atomic Structure 4.1 Early Theories of Matter Earth, Water, Air, Fire Matter was thought to be infinitely divisible No method was available to test theories Democritus (460 B.C. 370 B.C.) First
CHAPTER 4 Atomic Structure 4.1 Early Theories of Matter Earth, Water, Air, Fire Matter was thought to be infinitely divisible No method was available to test theories Democritus (460 B.C. 370 B.C.) First
Accelerated Chemistry Study Guide Atomic Structure, Chapter 3
 Accelerated Chemistry Study Guide Atomic Structure, Chapter 3 Terms and definitions atom ion law of constant composition isotope atomic theory of matter mass number cathode ray tube atomic mass electron
Accelerated Chemistry Study Guide Atomic Structure, Chapter 3 Terms and definitions atom ion law of constant composition isotope atomic theory of matter mass number cathode ray tube atomic mass electron
7.1 Development of a Modern Atomic Theory
 7.1 Development of a Modern Atomic Theory Development of the Atomic Theory Many scientists in different countries have contributed to the understanding of matter - atoms John Dalton Credited with developing
7.1 Development of a Modern Atomic Theory Development of the Atomic Theory Many scientists in different countries have contributed to the understanding of matter - atoms John Dalton Credited with developing
The structure of Atom III
 The structure of Atom III Atomic Structure If, in some cataclysm, all of scientific knowledge were to be destroyed, and only one sentence passed on to the next generations of creatures, what statement
The structure of Atom III Atomic Structure If, in some cataclysm, all of scientific knowledge were to be destroyed, and only one sentence passed on to the next generations of creatures, what statement
Early Atomic Theories and the Origins of Quantum Theory. Chapter 3.1
 Early Atomic Theories and the Origins of Quantum Theory Chapter 3.1 What is Matter Made of? People have wondered about the answer to this question for thousands of years Philosophers Matter is composed
Early Atomic Theories and the Origins of Quantum Theory Chapter 3.1 What is Matter Made of? People have wondered about the answer to this question for thousands of years Philosophers Matter is composed
Atomic Structure. How do you discover and study something you can t see?
 Atomic Structure How do you discover and study something you can t see? WHAT IS A THEORY? A hypothesis is a proposed explanation made as a starting point for further investigation (It s bright outside
Atomic Structure How do you discover and study something you can t see? WHAT IS A THEORY? A hypothesis is a proposed explanation made as a starting point for further investigation (It s bright outside
HIGH SCHOOL SCIENCE. Physical Science 9: Atomic Structure
 HIGH SCHOOL SCIENCE Physical Science 9: Atomic Structure WILLMAR PUBLIC SCHOOL 2013-2014 EDITION CHAPTER 9 Atomic Structure In this chapter you will: 1. Compare and contrast quarks, leptons, and bosons.
HIGH SCHOOL SCIENCE Physical Science 9: Atomic Structure WILLMAR PUBLIC SCHOOL 2013-2014 EDITION CHAPTER 9 Atomic Structure In this chapter you will: 1. Compare and contrast quarks, leptons, and bosons.
Bravo 15,000 kilotons
 Bravo 15,000 kilotons All matter is composed of atoms Atoms cannot be subdivided, created, or destroyed in ordinary chemical reactions. However, these changes CAN occur in nuclear reactions! Atoms of an
Bravo 15,000 kilotons All matter is composed of atoms Atoms cannot be subdivided, created, or destroyed in ordinary chemical reactions. However, these changes CAN occur in nuclear reactions! Atoms of an
2 Copyright Pearson Education, Inc., or its affiliates. All Rights Reserved.
 CHEMISTRY & YOU Chapter 4 Atomic Structure 4.1 Defining the Atom 4.2 Structure of the Nuclear Atom How did scientists determine the structures that are inside an atom? X-rays are used to see structures
CHEMISTRY & YOU Chapter 4 Atomic Structure 4.1 Defining the Atom 4.2 Structure of the Nuclear Atom How did scientists determine the structures that are inside an atom? X-rays are used to see structures
EARLY VIEWS: The Ancient Greeks
 Feb 7 11:59 AM EARLY VIEWS: The Ancient Greeks Empedocles (c. 450 B.C.) proposed Four Element theory he thought that matter was composed of four elements: AIR, EARTH, FIRE and WATER elements mixed together
Feb 7 11:59 AM EARLY VIEWS: The Ancient Greeks Empedocles (c. 450 B.C.) proposed Four Element theory he thought that matter was composed of four elements: AIR, EARTH, FIRE and WATER elements mixed together
Atomic Theory. Democritus to the Planetary Model
 Atomic Theory Democritus to the Planetary Model Democritus Greek philosopher (460-370 BCE) Believed in the philosophy of materialism With Leucippus, they though that matter can not be divided infinitely.
Atomic Theory Democritus to the Planetary Model Democritus Greek philosopher (460-370 BCE) Believed in the philosophy of materialism With Leucippus, they though that matter can not be divided infinitely.
Fill in the Following Table in your notes (assume an atom unless otherwise stated: Symbol Protons Electrons Neutrons Atomic # Mass # 24 Na
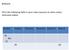 Bellwork: Fill in the Following Table in your notes (assume an atom unless otherwise stated: Symbol Protons Electrons Neutrons Atomic # Mass # 24 Na + 11 29 63 Bellwork: Fill in the Following Table in
Bellwork: Fill in the Following Table in your notes (assume an atom unless otherwise stated: Symbol Protons Electrons Neutrons Atomic # Mass # 24 Na + 11 29 63 Bellwork: Fill in the Following Table in
Unit 2 continued-chemical Foundations Atoms, Ions, &Elements
 Unit 2 continuedchemical Foundations Atoms, Ions, &Elements The Elements Most abundant elements in/on Earth: Oxygen 49.2% Silicon25.7% Most abundant in the human body: Oxygen65.0% Carbon18.0 % Hydrogen10.0%
Unit 2 continuedchemical Foundations Atoms, Ions, &Elements The Elements Most abundant elements in/on Earth: Oxygen 49.2% Silicon25.7% Most abundant in the human body: Oxygen65.0% Carbon18.0 % Hydrogen10.0%
Chapter 5. Early Atomic Theory and Structure
 Chapter 5 Early Atomic Theory and Structure 1 5.1 Early thoughts ~ 400 B.C. Greek philosophers Empedocles all matter was composed of four elements earth, air, water, fire Democritus all forms of matter
Chapter 5 Early Atomic Theory and Structure 1 5.1 Early thoughts ~ 400 B.C. Greek philosophers Empedocles all matter was composed of four elements earth, air, water, fire Democritus all forms of matter
Chapter 3: Atoms: The Building Blocks of Ma;er
 Chapter 3: Atoms: The Building Blocks of Ma;er The Atom: From Philosophical Idea to Scien7fic Theory The Greek philosopher Democritus (460 B.C. 370 B.C.) was among the first to suggest the existence of
Chapter 3: Atoms: The Building Blocks of Ma;er The Atom: From Philosophical Idea to Scien7fic Theory The Greek philosopher Democritus (460 B.C. 370 B.C.) was among the first to suggest the existence of
Chapter 4. Atomic Structure
 Chapter 4 Atomic Structure Warm Up We have not discussed this material, what do you know already?? What is an atom? What are electron, neutrons, and protons? Draw a picture of an atom from what you know
Chapter 4 Atomic Structure Warm Up We have not discussed this material, what do you know already?? What is an atom? What are electron, neutrons, and protons? Draw a picture of an atom from what you know
Democritus of Abdera. John Dalton. Dalton s Atom. Dalton s Atomic Theory Ancient Greece - 4th century BC. Eaglesfield, England
 Democritus of Abdera Ancient Greece - 4th century BC first suggested the existence of tiny fundamental particles that make up matter. atoms = indestructible did not agree with the current sci theory -
Democritus of Abdera Ancient Greece - 4th century BC first suggested the existence of tiny fundamental particles that make up matter. atoms = indestructible did not agree with the current sci theory -
Chapter #1 - Atomic Structure
 Chapter #1 - Atomic Structure Atomic Theories Democritus (460-340 BC) Democritus believed that all matter consisted of extremely small particles that could not be divided. He called them atoms from the
Chapter #1 - Atomic Structure Atomic Theories Democritus (460-340 BC) Democritus believed that all matter consisted of extremely small particles that could not be divided. He called them atoms from the
Do Now: Recall 1. What is an atom? What have you learned about the word atom so far this semester?
 Chemistry Ms. Ye Name Date Block Do Now: Recall 1. What is an atom? What have you learned about the word atom so far this semester? Atoms Video: 1. Proper Portioned Giant Atom Model of Science: Structure
Chemistry Ms. Ye Name Date Block Do Now: Recall 1. What is an atom? What have you learned about the word atom so far this semester? Atoms Video: 1. Proper Portioned Giant Atom Model of Science: Structure
Atomic Structure. History of Atomic Theory
 Atomic Structure History of Atomic Theory Democritus (460-370 BC) Was the to come up with the idea of atom Believed that all matter was composed of Which is derived from the Greek word Atomos meaning He
Atomic Structure History of Atomic Theory Democritus (460-370 BC) Was the to come up with the idea of atom Believed that all matter was composed of Which is derived from the Greek word Atomos meaning He
Ch. 4 Notes THE STRUCTURE OF THE ATOM NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics.
 Ch. 4 Notes THE STRUCTURE OF THE ATOM NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Early Ideas About Matter A. atom the smallest particle of an element retaining
Ch. 4 Notes THE STRUCTURE OF THE ATOM NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Early Ideas About Matter A. atom the smallest particle of an element retaining
Do Now: Recall 1. What is an atom? What have you learned about the word atom so far this semester?
 Chemistry Ms. Ye Name Date Block Do Now: Recall 1. What is an atom? What have you learned about the word atom so far this semester? Atoms Video: 1. Proper Portioned Giant Atom Model of Science: Structure
Chemistry Ms. Ye Name Date Block Do Now: Recall 1. What is an atom? What have you learned about the word atom so far this semester? Atoms Video: 1. Proper Portioned Giant Atom Model of Science: Structure
Ancient Greek Models of Atoms
 Atomic Theory Ancient Greek Models of Atoms The philosopher Democritus believed that all matter consisted of extremely small particles that could not be divided. He called these particles atoms from the
Atomic Theory Ancient Greek Models of Atoms The philosopher Democritus believed that all matter consisted of extremely small particles that could not be divided. He called these particles atoms from the
5 Early Atomic Theory and Structure
 5 Early Atomic Theory and Structure Chapter Outline 5.1 5.2 Electric Charge A. Discovery of Ions 5.3 Subatomic Parts of the Atom Lightning occurs when electrons move to neutralize charge difference between
5 Early Atomic Theory and Structure Chapter Outline 5.1 5.2 Electric Charge A. Discovery of Ions 5.3 Subatomic Parts of the Atom Lightning occurs when electrons move to neutralize charge difference between
4.1 Structure of the Atom
 4.1 Structure of the Atom How do atoms differ from each other? What are atoms composed of? What are the subatomic particles? 2-1 Structure of the Atom Atoms actually are divisible. They are composed of
4.1 Structure of the Atom How do atoms differ from each other? What are atoms composed of? What are the subatomic particles? 2-1 Structure of the Atom Atoms actually are divisible. They are composed of
PROGRESSION OF THE ATOMIC MODEL
 PROGRESSION OF THE ATOMIC MODEL By 1808, it was widely accepted that matter was made up of ELEMENTS, which consisted of tiny PARTICLES called ATOMS. After 2000 years - DEMOCRITUS was right all along John
PROGRESSION OF THE ATOMIC MODEL By 1808, it was widely accepted that matter was made up of ELEMENTS, which consisted of tiny PARTICLES called ATOMS. After 2000 years - DEMOCRITUS was right all along John
Early Ideas About Matter
 Early Ideas About Matter Democritus (460 370 BC) believed that matter is made of small, solid objects called atomos, from which the English word atom is derived. Early Ideas About Matter (cont.) Aristotle
Early Ideas About Matter Democritus (460 370 BC) believed that matter is made of small, solid objects called atomos, from which the English word atom is derived. Early Ideas About Matter (cont.) Aristotle
Name Chemistry-PAP Per. Notes: Atomic Structure
 Name Chemistry-PAP Per. I. Historical Development of the Atomic Model Ancient Greek Model Notes: Atomic Structure Democritus (460-370 BC) was an ancient Greek philosopher credited with the first particle
Name Chemistry-PAP Per. I. Historical Development of the Atomic Model Ancient Greek Model Notes: Atomic Structure Democritus (460-370 BC) was an ancient Greek philosopher credited with the first particle
Atomic Theory. Past and Present: pieces of a puzzle
 Atomic Theory Past and Present: pieces of a puzzle The First Atomic Hypothesis Democritus (460 370 BC): Greek philosopher Speculated that matter is composed of atoms which move through empty space Atoms
Atomic Theory Past and Present: pieces of a puzzle The First Atomic Hypothesis Democritus (460 370 BC): Greek philosopher Speculated that matter is composed of atoms which move through empty space Atoms
Chapter 2 Atoms and Elements
 1 Chapter 2 Atoms and Elements Sec 2.3 Modern Atomic Theory and the Laws That Led to it Law of conservation of mass states that in a chemical reaction, matter is neither created nor destroyed. Law of definite
1 Chapter 2 Atoms and Elements Sec 2.3 Modern Atomic Theory and the Laws That Led to it Law of conservation of mass states that in a chemical reaction, matter is neither created nor destroyed. Law of definite
Atomic Structure. A model uses familiar ideas to explain unfamiliar facts observed in nature.
 Atomic Structure 1 2 This model of the atom may look familiar to you. This is the Bohr model. In this model, the nucleus is orbited by electrons, which are in different energy levels. A model uses familiar
Atomic Structure 1 2 This model of the atom may look familiar to you. This is the Bohr model. In this model, the nucleus is orbited by electrons, which are in different energy levels. A model uses familiar
In Atomic Theory, if we were to zoom in on a copper wire, it might look like the picture below. What is between the atoms?
 History of the Atom In Atomic Theory, if we were to zoom in on a copper wire, it might look like the picture below. What is between the atoms? What did be believe before we thought there were atoms? We
History of the Atom In Atomic Theory, if we were to zoom in on a copper wire, it might look like the picture below. What is between the atoms? What did be believe before we thought there were atoms? We
Atomic Theory: Early Models of the Atom:
 Atomic Theory: Our next job in Chemistry 11 is to learn about what matter is made of. After we have done this, we can start to understand why matter behaves the way it does. Everything that has volume
Atomic Theory: Our next job in Chemistry 11 is to learn about what matter is made of. After we have done this, we can start to understand why matter behaves the way it does. Everything that has volume
The Beginning of the Atom
 The Beginning of the Atom Democritus s Atomic Philosophy 460BC 370BC ~ Believed atoms were indivisible and indestructible Dalton took it and ran with it 2000 yrs later! Jan 31 3:33 PM Dalton and the Atomic
The Beginning of the Atom Democritus s Atomic Philosophy 460BC 370BC ~ Believed atoms were indivisible and indestructible Dalton took it and ran with it 2000 yrs later! Jan 31 3:33 PM Dalton and the Atomic
Atom s Structure. Chemistry is the study of the structure, characteristics and behavior of matter. The basic
 AlMarzooqi 1 Hamad AlMarzooqi Professor Kafle ESL 015 26 February 2013 Atom s Structure Chemistry is the study of the structure, characteristics and behavior of matter. The basic unit of matter is an atom,
AlMarzooqi 1 Hamad AlMarzooqi Professor Kafle ESL 015 26 February 2013 Atom s Structure Chemistry is the study of the structure, characteristics and behavior of matter. The basic unit of matter is an atom,
PreAP Chemistry. Unit 4 Atomic Structure, the Periodic Table, and Nuclear Radiation
 PreAP Chemistry Unit 4 Atomic Structure, the Periodic Table, and Nuclear Radiation Democritus A Greek who lived ~400 BC, was the first to suggest the existence of atoms. He believed atoms to be indivisible
PreAP Chemistry Unit 4 Atomic Structure, the Periodic Table, and Nuclear Radiation Democritus A Greek who lived ~400 BC, was the first to suggest the existence of atoms. He believed atoms to be indivisible
4. The mass of a proton is approximately equal to the mass of A an alpha particle C a positron. B a beta particle D a neutron
 1. Which particles have approximately the same mass? A an electron and an alpha particle B an electron and a proton C a neutron and an alpha particle D a neutron and a proton 2. Which phrase describes
1. Which particles have approximately the same mass? A an electron and an alpha particle B an electron and a proton C a neutron and an alpha particle D a neutron and a proton 2. Which phrase describes
Memorial to a Scientist
 Memorial to a Scientist 1. My Question of Inquiry: Use this sheet to outline how you will collect and present the information to the class. My Group s Scientist: 1 Part I: Memorial to a Scientist: John
Memorial to a Scientist 1. My Question of Inquiry: Use this sheet to outline how you will collect and present the information to the class. My Group s Scientist: 1 Part I: Memorial to a Scientist: John
I. History and Development of the Atom
 Unit 3: The Atom I. History and Development of the Atom A. Democritus (around 400 B.C.) Based on his observations of the natural world around him, Democritus was the first to suggest that all matter was
Unit 3: The Atom I. History and Development of the Atom A. Democritus (around 400 B.C.) Based on his observations of the natural world around him, Democritus was the first to suggest that all matter was
History of the OBJECTIVES. ESSENTIAL QUESTION What evidence is there for the existence of atoms and their sub-atomic particles?
 History of the 09/15/2016 OBJECTIVES Understand the law of definite proportions. Define a scientific law and identify how observations become a law. Explain that a scientific theory is not established
History of the 09/15/2016 OBJECTIVES Understand the law of definite proportions. Define a scientific law and identify how observations become a law. Explain that a scientific theory is not established
Title: Chem Review 2 TOPIC: DISCOVERY OF ATOM
 Title: Chem Review 2 TOPIC: DISCOVERY OF ATOM EQ: How were the 3 subatomic particles discovered? Title and Highlight Topic: EQ: Date Reflect Question: Reflect on the material by asking a question (its
Title: Chem Review 2 TOPIC: DISCOVERY OF ATOM EQ: How were the 3 subatomic particles discovered? Title and Highlight Topic: EQ: Date Reflect Question: Reflect on the material by asking a question (its
Chapter 5. Early Atomic Theory and Structure
 Chapter 5 Early Atomic Theory and Structure Introduction to General, Organic, and Biochemistry 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison, and Susan Arena Lightning occurs when electrons move
Chapter 5 Early Atomic Theory and Structure Introduction to General, Organic, and Biochemistry 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison, and Susan Arena Lightning occurs when electrons move
tomic tructure Chapter 3
 tomic tructure Chapter 3 Early Theories of Matter 460 BC Democritus Proposed the matter was not infinitely divisible. Believed matter composed of particles called atoms. Early Theories of Matter Aristotle
tomic tructure Chapter 3 Early Theories of Matter 460 BC Democritus Proposed the matter was not infinitely divisible. Believed matter composed of particles called atoms. Early Theories of Matter Aristotle
Chapter 2. Atoms and Ions
 Chapter 2 Atoms and Ions A History of Atomic Models 400 B.C.E. (Democritus, a early atomist) 1804 (Dalton) Law of Conservation of Mass Antoine Lavoisier 1743-1794 In a chemical reaction, matter is neither
Chapter 2 Atoms and Ions A History of Atomic Models 400 B.C.E. (Democritus, a early atomist) 1804 (Dalton) Law of Conservation of Mass Antoine Lavoisier 1743-1794 In a chemical reaction, matter is neither
