A Theoretical Study of Oxidation of Phenoxy and Benzyl Radicals by HO 2
|
|
|
- Jason Flynn
- 5 years ago
- Views:
Transcription
1 A Theoretical Study of xidation of Phenoxy and Benzyl adicals by 2 S. Skokov, A. Kazakov, and F. L. Dryer Department of Mechanical and Aerospace Engineering Princeton University, Princeton, NJ We present a quantum chemical study of oxidation of phenoxy and benzyl radicals by 2. All calculations were carried out using the modified Perdew-Wang (MPW1K) density functional method. We found that adsorption of 2 in the ortho and para positions of the aromatic ring leads to stable phenyl- and benzyl- adducts with dissociation energies from 16 to 27 kcal/mol. Calculations indicate that the barriers for various decomposition pathways for phenoxy- and benzyl- complexes are at least 10 kcal/mol above the dissociation thresholds, thus precluding the possibility of chemically activated decomposition. The main products from the decomposition of phenoxy- adduct are found to be phenol, 1,4- benzoquinone, and 1,2-benzoquinone. Similarly, the decomposition of benzyl- leads to toluene and methylenecyclohexadienone. 1,2-benzoquinone is the least stable specie among these compounds. We found that subsequent thermal decomposition of 1,2-benzoquinone into carbon monoxide and cyclopentadienone proceeds via a 48 kcal/mol barrier. I. Introduction An accurate modeling of toluene combustion is currently hindered by the absence of credible chemical kinetic mechanisms for oxidation of benzyl and phenoxy radicals. Benzyl radicals are formed at the initial stages of toluene combustion via abstraction of by 2 from the side chain of toluene. 1 Phenoxy radicals are believed to be formed further down the reaction chain via benzaldehyde, benzoyl radical, and phenyl radicals. The main consumption channel for phenoxy radicals is the thermal decomposition into cyclopentadienyl radical and carbon monoxide. owever, this reaction has a high activation energy, about 50 kcal/mol, 2 and will be slow in low temperature combustion systems. Consumption of benzyl radicals is modeled via barrierless decomposition of the benzylhydroperoxide complex. 3 While this proposition has received some prior theoretical support, 4 most of the details remain unknown. The purpose of the present study was to investigate the reaction mechanism for decomposition of benzyl and phenoxy radicals under low temperature combustion conditions. Therefore, we focused on the recombination of benzyl and phenoxy radicals with 2, rather than on the unimolecular decomposition of these radicals, or their reactions with highly energetic radicals such as,, or. II. Computational methods All quantum chemical calculations were carried out using Gaussian98 code. 5 Geometries were optimized using modified Perdew-Wang 1-parameter for kinetics, MPW1K/6-31G+(d,p), method proposed by Linch and Truhlar. 6 This method was shown to be superior to other popular hybrid density functional methods in calculation of transition state geometries and energy barriers. At the MPW1K/6-31+G(d,p) level of theory, the mean unsigned error in barriers heights for the set of 22 reactions was reported to be only 1.6 kcal/mol. 6,7 Vibrational frequency calculations for gas phase species were performed for all ground and transition states at the same MPW1K/6-31+G(d,p) levels of theory and rescaled by the factor, Intrinsic reaction coordinate 8 (IC) calculations were performed for all transition states to verify that they connect correct reactants and products. eaction rate constants were computed using Variflex computer code. 9 eactions with tight transition states were treated using a canonical non-variational transition state theory. eactions with loose transition states were treated using a canonical variable reaction coordinate-transition state theory (VC-TST) formalism. 10 In the VC-TST calculations a potential energy along reaction coordinate (assumed to be distance between bonding atoms) was represented by Varshni potential 11 V (r) = D e [1 r e 2 r e α(r r 2 ) e ] 2 D e (1) ere D e is the electronic dissociation energy, r e is the equilibrium bond distance, and α is the parameter controlling tightness of the potential well. To facilitate the use of the reaction rate constants for chemical kinetics modeling, we fitted all rate constants in the temperature range from 300 to 1000 K to the Arrhenius type expression: k = AT n exp( E b k B T ) (2) ere A is the pre exponential factor, n is the fitting parameter, k B is the Boltzmann constant, E b is the energy barrier, and T is the temperature.
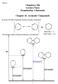
2 III. esults and discussion The recombination of phenoxy and benzyl radicals with 2 leads to stable phenoxy- and benzyl- adducts as shown in Figure 1. These adducts, denoted as A1 and A2 in Figure 1, are formed via barrierless adsorption of 2 in ortho and para position, respectively A1, E=-27.4 (-16.3) E=0.0 A2, E=-26.3 (-19.2) Figure 1. ecombination of benzyl ( = C 2 ) and phenoxy ( = ) radicals with 2 radical. Numbers are relative energies in kcal/mol, including ZPE, at the MPW1K/6-31+G(d,p) level of theory for phenoxy + 2 reaction. Numbers in parentheses are for benzyl + 2 reaction. Both reactions are exothermic with reaction energies in the case of the phenoxy- adduct computed to be 27.4 and 26.3 kcal/mol for A1 and A2, respectively. In the case of the benzyl- adduct, reaction energies were computed to be 16.3 and 19.2 kcal/mol for A1 and A2, respectively. elatively modest reaction energies are due to destabilization of the aromatic ring in A1 and A2 adducts. For comparison, the exothermicity of the addition of 2 to the side chain of benzyl radical was computed to be 49.8 kcal/mol. Note that our value falls between the previously reported 4 values of 39 and 78 kcal/mol computed at the B3LYP and MP2 levels of theory, respectively. We defer further discussion of this compound to future studies; here we consider only the chemical reactivity of A1 and A2 complexes. The mechanisms for subsequent decomposition of phenoxy- and benzyl- adducts with in ortho position (A1) are shown in Figure 2. Both mechanisms are very similar, and we discuss in detail only the case of the phenoxy- adduct. The lowest energy pathway is for transformation of A1 into 1,2 benzoquinone via TS8, intermediate compound F1, and TS9. Visual inspection of TS8 geometry suggests that this reaction is due to simultaneous shift of two hydrogen atoms. ne hydrogen atom moves from the group to the = group while another moves from a backbone carbon atom to the group. Double hydrogen shift is also apparent from the analysis of the lowest vibrational eigenstate. Indeed, we found that the imaginary frequency is quite large, 1576 cm -1. educed mass association with motion along this mode is close to the hydrogen mass, 1.1 a.m.u., and largest contributions to this eigenstate are from two hydrogen atoms. n the next step, TS9, atom from the group and fragment from the group form a product water molecule. Calculations indicate that a dominant contribution to the reaction coordinate is from the motion of atom with an imaginary frequency equal to 2200 cm -1. An alternative pathway for the formation of 1,2-benzoquinone is the direct elimination of water via TS10 with an energy barrier of 51.6 kcal/mol. The second lowest energy pathway in Figure 2 is the transformation of A1 into phenol via TS5 with an energy barrier of 42 kcal/mol. This reaction involves transfer from the group to the = group with simultaneous elimination of an oxygen molecule. An analysis of bond distances in TS5 indicates that this reaction path is associated with concerted motion of all atoms of the - group. Indeed, C- bond is stretched from Å in A1 to Å in TS5, bond is stretched from to Å while - bond is reduced from to Å. An additional indication of concerted motion is a low imaginary frequency mode, 445 cm -1, and high reduced mass, 6.1 a.m.u., associated with a motion along the reaction coordinate. An alternative pathway via TS1-B1-TS4 has a much higher energy barrier, and therefore is of little importance. The ring opening reactions, TS3 and TS6, proceed via attack of a neighboring carbon atom by atom of the group. Both reactions have high energy barriers, 60.9 and 65.6 kcal/mol for TS3 and TS6, respectively. The alternative pathway for the ring opening via TS1, 46 kcal/mol, and TS2, 31.8 kcal/mol, is more viable.
3 E=-35.5 (-38.6) + 2 TS5, E=14.6 (19.9) TS6, E=38.2 (48.9) D1, E=-62.9 (-63.7) B1, E=-13.8 (-13.0) TS4, E=32.8 (33.4) A1, E=-27.4 (-16.3) TS1 E=18.6 (30.8) TS7 E=55.7 (61.7) E1, E=-71.8 (-64.8) TS2, E=18.1 (20.0) C1, E=-84.7 (-79.9) TS3, E=33.5 TS8, E=10.9 (32.1) TS10, E=24.2 (36.3) F1, E=-50.5 (-52.2) + 2 TS9=-42.9 (-11.6) E=-80.8 (-85.2) Figure 2. eaction mechanism for decomposition of A1 intermediate of benzyl ( = C 2 ) and phenoxy ( = ) radicals. Numbers are relative energies in kcal/mol, including ZPE, at the MPW1K/6-31+G(d,p) level of theory for phenoxy- 2 adduct. Numbers in parentheses are for benzyl- 2 adduct. D2, E=-69.0 (-63.0) A2, E=-26.3 (-19.2) TS11 E=40.4 (51.1) TS12 E=54.9 (62.5) TS13, E=24.1 (34.0) E2, E=-75.5 (-62.7) + 2 E=-88.8 (-88.0) Figure 3. eaction mechanism for decomposition of A2 intermediate of benzyl ( = C 2 ) and phenoxy ( = ) radicals. Numbers are relative energies in kcal/mol, including ZPE, at the MPW1K/6-31+G(d,p) level of theory for phenoxy- 2 adduct. Numbers in parentheses are for benzyl- 2 adduct.
4 TS (58.1) TS (80.8) I C TS (67.3) + C 25.0 (47.0) 15.3 (31.4) 14.2 (19.7) 0.0 Figure 4. eaction mechanism for decomposition of 2-methylene-3,5-cyclohexadiene-1-one ( = C 2 ) and 1,2- benzoquinone ( = ). Numbers are relative energies in kcal/mol including ZPE at the MPW1K/6-31+G(d,p) level of theory for 1,2-benzoquinone. Numbers in parentheses are 2-methylene-3,5-cyclohexadiene-1-one. TABLE 1. ate constants, k inf, for phenoxy + 2 ( = ) and benzyl + 2 ( = C 2 ) reaction mechanisms. Units are s, cm 3, K, kcal/mol. Species notations are given in Figures 1-4. eaction TS ate constant, = ate constant, = C A n E b A n E b C A1-3.2e e C A2-1.5e e A1 C e e A2 C e e A1 B1 TS1 1.5e e B1 C1 TS2 1.6e e A1 C1 TS3 1.5e B1 C TS4 2.5e e A1 C TS5 3.2e e A1 D1 TS6 1.6e e A1 E1 TS7 6.2e e A1 F1 TS8 6.3e e F1 A1 TS8 7.9e e F1 1,2-C TS9 9.2e e A1 1,2-C TS10 1.8e e A2 D2 TS11 6.1e e A2 E2 TS12 9.4e e A2 1,4-C TS13 7.8e e ,2-C 6 4 C 2 TS14 7.5e e ,2-C 6 4 TS14 3.8e e I TS15 5.1e e I TS15 5.4e e I C C TS16 5.2e e
5 Finally, an epoxy-like structure, E1, can be formed via TS7. This reaction proceeds via transfer of the group to the para position. Due to the very high energy barrier, 83.1 kcal/mol, this reaction is of little importance for low temperature oxidation. In the case of the benzyl- adduct A1, the lowest energy pathway with 36.2 kcal/mol barrier leads to the formation of toluene, while formation of 2-methylene-3,5-cyclohexadiene-1-one (analog of 1,2-benzoquinone) proceeds via TS10 with an energy barrier of 52.6 kcal/mol or via TS8 and TS9 with energy barriers of 48.5 and 40.6 kcal/mol, respectively. verall, decomposition of the benzyl- A1 adduct proceeds via somewhat higher energy barriers as compared to those for the phenoxy- A1 adduct. The mechanism of decomposition of phenoxy- and benzyl- compounds with in the para position (A2) is shown in Figure 3. This mechanism is much simpler than decomposition of A1 due to an absence of interaction between the adsorbed group and the side group. The lowest energy pathway leads to 1,4-benzoquinone and 4- methylene-2,5-cyclohexadiene-1-one from the phenoxy- and benzyl- adducts, respectively. This reaction is analogous to decomposition of A1 adduct via TS10 and has a similar reaction barrier of 50.4 kcal/mol for phenoxy- and 53.2 kcal/mol for benzyl-. ing opening via TS11 is an analog of ring opening for the A1 adduct via TS6, and formation of the epoxy-type compound E2 via TS12 is an analog of formation of E1 compound via TS7. Simple comparison of barriers heights for different reaction channels indicates that benzoquinones and methylene substituted cyclohexadienones formed via TS13, TS8 and TS19, and TS10 will be the dominant intermediate compounds. Further investigation of the decomposition of these compounds is therefore warranted. The barrier for thermal decomposition of 1,4-benzoquinone into cyclopentadienone and C was estimated previously by Frank et. al. 12 to be 59 kcal/mol. owever, no data are available for 1,2-benzoquinone or methylene substituted cyclohexadienones. The mechanism for thermal decomposition of these compounds is shown in Figure 4. The first step in this mechanism is the ring opening via TS14, followed by formation of fused 5 and 3-member ring intermediates (I) via TS15. Finally, compound I ejects a C molecule and forms cyclopentadienone or 5-methylene-1,3-cyclopentadiene (a.k.a. fulvene) via TS16. For 1,2- benzoquinone decomposition, the highest energy barrier is 48.2 kcal/mol above the reactant and the overall reaction energy is 14.2 kcal/mol. In contrast, 2-methylene-3,5-cyclohexadiene-1-one appears to be more stable with the highest barrier being 80.8 kcal/mol above the reactant and with the overall reaction energy of 19.7 kcal/mol. IV. Summary We have conducted a density functional study of oxidation of phenoxy and benzyl radicals by 2. It was found that 2 can adsorb into the ortho and para positions leading to the formation of benzyl- and phenoxy- adducts. The dissociation energies for benzyl- and phenoxy- adducts are relatively low, between 16 and 27 kcal/mol, as compared to typical values for radical recombination reactions. This is due to destabilization of the aromatic ring in the benzyl- and phenoxy- compounds. We also studied a number of the decomposition pathways for benzyl- and phenoxy- adducts. Calculations indicate a wide range of possible reactions such as ring openings, elimination of 2, and formation of hetero rings. The proximity of side groups in A1 type ( in the ortho position) adducts leads to a number of addition reactions such as transfer between side groups and elimination of 2 and 2. The main products from the decomposition of phenoxy- adducts were found to be phenol and benzoquinone. Similarly, the decomposition of benzyl- adducts leads to toluene and methylene-cyclohexadienone. owever, energy barriers for all decomposition pathways identified in the present study are higher than dissociation energies of benzyl- and phenoxy- adducts. This result precludes the possibility of chemically activated decomposition of these adducts. The rate constants for chemical mechanisms shown in Figures 1-4 are summarized in Table 1. Inspection of these data indicates that the most likely decomposition pathway is the formation of 1,2-benzoquinone and its subsequent decomposition into carbon monooxide and cyclopentadienone. The energy barrier for 1,2-benzoquinone decomposition was computed to be 48 kcal/mol. The effect of these reactions on the chemical kinetics of toluene oxidation is currently under investigation and will be reported in the future. Finally, we found that adsorption of 2 into the side chain of benzyl radical leads to a stable adduct with dissociation energy about 50 kcal/mol. Large adsorption energy opens the possibility for fast chemically activated reactions. 4 The reactivity of this adduct is currently under investigation and will be reported elsewhere. Acknowledgements This work was supported by the Chemical Sciences, Geosciences and Biosciences Division, ffice of Basic Energy Sciences, ffice of Science, U.S. Department of Energy under Grant No. DE-FG02-86E13503.
6 eferences (1) Eng,. A.; Fittschen, C.; ibomvschi, P.; ippler,.; Unterreiner, A.-N. Proc. Combust. Inst. 1998, (2) Liu,.; Morokuma, K.; Mebel, A.M.; Lin, M.C. J. Phys. Chem. 1996, 100, (3) Klotz, S.D.; Brezinsky, K.; Glassman, I. Symp. (Int.) Comb. Proc. 1998, 27, 337. (4) Davis, W.M.; eck, S.M.; Pritchard,.. J. Chem. Soc. Faraday Trans. 1998, 94, (5) Frisch, M. J., et. al. Gaussian 98, evision A.6, Gaussian, Inc., Pittsburgh PA, (6) Lynch, B. J. : Truhlar, D. G. J. Phys. Chem. A 2001, 105, (7) Lynch, B. J.: Truhlar, D. G. J. Phys. Chem. A 2002, 106, 842. (8) Gonzalez, C.; Schlegel,. B. J. Chem. Phys. 1998, 90, (9) Klippenstein, S. J.; Wagner, A.F.; Dunbar,. C.; Wardlaw, D.M.; obertson, S.. VAIFLEX, Version 1.0, (10) obertson, S..; Wagner, A. F.; Wardlaw, D. M. Faraday Disc. Chem. Soc. 1995, 102, 65; (11) Varshni, V. P. ev. Mod. Phys. 1957, 29, 664. (12) Frank, P.; erzler, J.; Just, T.; Wahl, C. Symp. (Int.) Combust. Proc. 1994, 25, 833.
Computational Study on the Recombination Reaction between Benzyl and Propargyl Radicals
 The 7 th International Conference on Chemical Kinetics July 11, 2011 Computational Study on the Recombination Reaction between Benzyl and Propargyl Radicals Akira Matsugi and Akira Miyoshi Department of
The 7 th International Conference on Chemical Kinetics July 11, 2011 Computational Study on the Recombination Reaction between Benzyl and Propargyl Radicals Akira Matsugi and Akira Miyoshi Department of
Theorical study of the thermal decomposition mechanism of phenylperoxy radical.
 Theorical study of the thermal decomposition mechanism of phenylperoxy radical. B. Sirjean (1), M.F. Ruiz-Lopez (1)*, P.A. Glaude (2), F. Battin-Leclerc (2), R. Fournet (2) (1) Equipe de Chimie et Biochimie
Theorical study of the thermal decomposition mechanism of phenylperoxy radical. B. Sirjean (1), M.F. Ruiz-Lopez (1)*, P.A. Glaude (2), F. Battin-Leclerc (2), R. Fournet (2) (1) Equipe de Chimie et Biochimie
Ab Initio MO and TST Calculations for the Rate Constant of the HNO NO : HONO NO
 Ab Initio M and TST Calculations for the Rate Constant of the N N : N N 2 Reaction A. M. MEBEL,* M. C. LIN, K. MRKUMA Cherry L. Emerson Center for Scientific Computation and Department of Chemistry, Emory
Ab Initio M and TST Calculations for the Rate Constant of the N N : N N 2 Reaction A. M. MEBEL,* M. C. LIN, K. MRKUMA Cherry L. Emerson Center for Scientific Computation and Department of Chemistry, Emory
Rate Constant for the NH 3 NO 2. HONO Reaction: Comparison of Kinetically Modeled and Predicted Results
 Rate Constant for the NH 3 HONO Reaction: Comparison of Kinetically Modeled and Predicted Results A. GRANT THAXTON, C.-C. HSU, M. C. LIN Department of Chemistry, Emory University, Atlanta, Georgia 30322
Rate Constant for the NH 3 HONO Reaction: Comparison of Kinetically Modeled and Predicted Results A. GRANT THAXTON, C.-C. HSU, M. C. LIN Department of Chemistry, Emory University, Atlanta, Georgia 30322
Hydrogen Abstraction/Acetylene Addition Revealed
 Hydrogen Abstraction/Acetylene Addition Revealed Dorian S. N. Parker, Ralf I. Kaiser,* Tyler P. Troy, and Musahid Ahmed* University of Hawaii at Manoa, USA Lawrence Berkeley National Laboratory, USA Angew.
Hydrogen Abstraction/Acetylene Addition Revealed Dorian S. N. Parker, Ralf I. Kaiser,* Tyler P. Troy, and Musahid Ahmed* University of Hawaii at Manoa, USA Lawrence Berkeley National Laboratory, USA Angew.
Formation of CO precursors during char gasification with O 2,CO 2 and H 2 O
 Fuel Processing Technology 77 78 (2002) 125 130 www.elsevier.com/locate/fuproc Formation of CO precursors during char gasification with O 2,CO 2 and H 2 O Alejandro Montoya a, *, Fanor Mondragón a, Thanh
Fuel Processing Technology 77 78 (2002) 125 130 www.elsevier.com/locate/fuproc Formation of CO precursors during char gasification with O 2,CO 2 and H 2 O Alejandro Montoya a, *, Fanor Mondragón a, Thanh
240 Chem. Aromatic Compounds. Chapter 6
 240 Chem Aromatic Compounds Chapter 6 1 The expressing aromatic compounds came to mean benzene and derivatives of benzene. Structure of Benzene: Resonance Description C 6 H 6 1.It contains a six-membered
240 Chem Aromatic Compounds Chapter 6 1 The expressing aromatic compounds came to mean benzene and derivatives of benzene. Structure of Benzene: Resonance Description C 6 H 6 1.It contains a six-membered
Research Article Theoretical Mechanism Study on the Reaction of FOO Radical with NO
 Chemistry Volume 2016, Article ID 5387853, 6 pages http://dx.doi.org/10.1155/2016/5387853 Research Article Theoretical Mechanism Study on the Reaction of Radical with ShunLi u Yang, 1 anan Wu, 1,2 JingYao
Chemistry Volume 2016, Article ID 5387853, 6 pages http://dx.doi.org/10.1155/2016/5387853 Research Article Theoretical Mechanism Study on the Reaction of Radical with ShunLi u Yang, 1 anan Wu, 1,2 JingYao
Kinetics of nitric oxide desorption from carbonaceous surfaces
 Fuel Processing Technology 77 78 (2002) 453 458 www.elsevier.com/locate/fuproc Kinetics of nitric oxide desorption from carbonaceous surfaces Alejandro Montoya a, Fanor Mondragón a, Thanh N. Truong b,
Fuel Processing Technology 77 78 (2002) 453 458 www.elsevier.com/locate/fuproc Kinetics of nitric oxide desorption from carbonaceous surfaces Alejandro Montoya a, Fanor Mondragón a, Thanh N. Truong b,
Direct ab initio dynamics studies of N H 2^NH H reaction
 JOURNAL OF CHEMICAL PHYSICS VOLUME 113, NUMBER 15 15 OCTOBER 2000 Direct ab initio dynamics studies of N H 2^NH H reaction Shaowen Zhang and Thanh N. Truong a) Henry Eyring Center for Theoretical Chemistry,
JOURNAL OF CHEMICAL PHYSICS VOLUME 113, NUMBER 15 15 OCTOBER 2000 Direct ab initio dynamics studies of N H 2^NH H reaction Shaowen Zhang and Thanh N. Truong a) Henry Eyring Center for Theoretical Chemistry,
Theoretical Studies of Reaction Mechanisms Relevant to Hydrocarbon Growth in Titan s Atmosphere
 Theoretical Studies of Reaction Mechanisms Relevant to Hydrocarbon Growth in Titan s Atmosphere Alexander M. Mebel, Adeel Jamal, Alexander Landera, and Ralf I. Kaiser Department of Chemistry and Biochemistry,
Theoretical Studies of Reaction Mechanisms Relevant to Hydrocarbon Growth in Titan s Atmosphere Alexander M. Mebel, Adeel Jamal, Alexander Landera, and Ralf I. Kaiser Department of Chemistry and Biochemistry,
Chemical Kinetics of HC Combustion
 Spark Ignition Engine Combustion MAK65E Chemical Kinetics of HC Combustion Prof.Dr. Cem Soruşbay Istanbul Technical University Chemical Kinetics of HC Combustion Introduction Elementary reactions Multi-step
Spark Ignition Engine Combustion MAK65E Chemical Kinetics of HC Combustion Prof.Dr. Cem Soruşbay Istanbul Technical University Chemical Kinetics of HC Combustion Introduction Elementary reactions Multi-step
Supporting Information
 Supporting Information Indirect Four-Electron Oxygen Reduction Reaction on Carbon Materials Catalysts in Acidic Solutions Guo-Liang Chai* 1, Mauro Boero 2, Zhufeng Hou 3, Kiyoyuki Terakura 3,4 and Wendan
Supporting Information Indirect Four-Electron Oxygen Reduction Reaction on Carbon Materials Catalysts in Acidic Solutions Guo-Liang Chai* 1, Mauro Boero 2, Zhufeng Hou 3, Kiyoyuki Terakura 3,4 and Wendan
Supporting Information
 Supporting Information Conflict in the Mechanism and Kinetics of the Barrierless Reaction between SH and NO 2 Radicals Ramanpreet Kaur and Vikas * Quantum Chemistry Group, Department of Chemistry & Centre
Supporting Information Conflict in the Mechanism and Kinetics of the Barrierless Reaction between SH and NO 2 Radicals Ramanpreet Kaur and Vikas * Quantum Chemistry Group, Department of Chemistry & Centre
Ion-Molecule Reactions in a Nitrogen-Benzene Plasma: Implications for the Destruction of Aromatic Compounds
 Ion-Molecule eactions in a Nitrogen-Benzene Plasma: Implications for the Destruction of Aromatic Compounds S. Williams, S. Arnold, A. Viggiano, and. Morris Air Force esearch Laboratory / Space Vehicles
Ion-Molecule eactions in a Nitrogen-Benzene Plasma: Implications for the Destruction of Aromatic Compounds S. Williams, S. Arnold, A. Viggiano, and. Morris Air Force esearch Laboratory / Space Vehicles
Jeff Turner Dr Scanlon Dr. Scanlon Ripon College
 Jeff Turner Dr Scanlon Dr. Scanlon Ripon College Importance of Nitrene Insertion Carbon Nitrogen bonds are useful in a chemical synthesis. Biomolecules Pharmaceuticals i l Specialized materials Formation
Jeff Turner Dr Scanlon Dr. Scanlon Ripon College Importance of Nitrene Insertion Carbon Nitrogen bonds are useful in a chemical synthesis. Biomolecules Pharmaceuticals i l Specialized materials Formation
Mechanism for Unimolecular Decomposition of HMX (1,3,5,7-Tetranitro-1,3,5,7-tetrazocine), an ab Initio Study
 1302 J. Phys. Chem. A 2001, 105, 1302-1314 Mechanism for Unimolecular Decomposition of HMX (1,3,5,7-Tetranitro-1,3,5,7-tetrazocine), an ab Initio Study Debashis Chakraborty, Richard P. Muller, Siddharth
1302 J. Phys. Chem. A 2001, 105, 1302-1314 Mechanism for Unimolecular Decomposition of HMX (1,3,5,7-Tetranitro-1,3,5,7-tetrazocine), an ab Initio Study Debashis Chakraborty, Richard P. Muller, Siddharth
Theoretical Gas Phase Chemical Kinetics. Stephen J. Klippenstein
 Theoretical Gas Phase Chemical Kinetics Stephen J. Klippenstein Goal Contribute to Improving the Accuracy of Mechanisms Theoretical Kinetics Predictions for Key Reactions Guided by Modeling Efforts Butanol
Theoretical Gas Phase Chemical Kinetics Stephen J. Klippenstein Goal Contribute to Improving the Accuracy of Mechanisms Theoretical Kinetics Predictions for Key Reactions Guided by Modeling Efforts Butanol
Figure 1: Transition State, Saddle Point, Reaction Pathway
 Computational Chemistry Workshops West Ridge Research Building-UAF Campus 9:00am-4:00pm, Room 009 Electronic Structure - July 19-21, 2016 Molecular Dynamics - July 26-28, 2016 Potential Energy Surfaces
Computational Chemistry Workshops West Ridge Research Building-UAF Campus 9:00am-4:00pm, Room 009 Electronic Structure - July 19-21, 2016 Molecular Dynamics - July 26-28, 2016 Potential Energy Surfaces
Chapter 4: Aromatic Compounds. Bitter almonds are the source of the aromatic compound benzaldehyde
 Chapter 4: Aromatic Compounds Bitter almonds are the source of the aromatic compound benzaldehyde Sources of Benzene Benzene, C 6 H 6, is the parent hydrocarbon of the especially stable compounds known
Chapter 4: Aromatic Compounds Bitter almonds are the source of the aromatic compound benzaldehyde Sources of Benzene Benzene, C 6 H 6, is the parent hydrocarbon of the especially stable compounds known
Density Functional Theory Study on Mechanism of Forming Spiro-Geheterocyclic Ring Compound from Me 2 Ge=Ge: and Acetaldehyde
 CHINESE JURNAL F CHEMICAL PHYSICS VLUME 26, NUMBER 1 FEBRUARY 27, 2013 ARTICLE Density Functional Theory Study on Mechanism of Forming Spiro-Geheterocyclic Ring Compound from Me 2 Ge=Ge: and Acetaldehyde
CHINESE JURNAL F CHEMICAL PHYSICS VLUME 26, NUMBER 1 FEBRUARY 27, 2013 ARTICLE Density Functional Theory Study on Mechanism of Forming Spiro-Geheterocyclic Ring Compound from Me 2 Ge=Ge: and Acetaldehyde
Supplementary Information
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Supplementary Information Insights into the Synergistic Role of Metal-Lattice
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Supplementary Information Insights into the Synergistic Role of Metal-Lattice
New Reaction Classes in the Kinetic Modeling of Low Temperature Oxidation of n-alkanes
 Supplemental Material for paper New Reaction Classes in the Kinetic Modeling of Low Temperature Oxidation of n-alkanes Eliseo Ranzi, Carlo Cavallotti, Alberto Cuoci, Alessio Frassoldati, Matteo Pelucchi,
Supplemental Material for paper New Reaction Classes in the Kinetic Modeling of Low Temperature Oxidation of n-alkanes Eliseo Ranzi, Carlo Cavallotti, Alberto Cuoci, Alessio Frassoldati, Matteo Pelucchi,
Theoretical investigation of mechanism for the gas-phase reaction of OH radical and ethane
 J. At. Mol. Sci. doi: 10.4208/jams.122810.011811a Vol. 2, No. 3, pp. 225-233 August 2011 Theoretical investigation of mechanism for the gas-phase reaction of OH radical and ethane Xiao-Ping Hu a, Bing-Xing
J. At. Mol. Sci. doi: 10.4208/jams.122810.011811a Vol. 2, No. 3, pp. 225-233 August 2011 Theoretical investigation of mechanism for the gas-phase reaction of OH radical and ethane Xiao-Ping Hu a, Bing-Xing
Applications of Computational Chemistry to the Reactions of Lignin
 Applications of Computational Chemistry to the Reactions of Lignin Thomas Elder USDA-Forest Service Southern Research Station ctober 30-ovember 2, 2012 Frontiers in Biorefining, Chemicals and Products
Applications of Computational Chemistry to the Reactions of Lignin Thomas Elder USDA-Forest Service Southern Research Station ctober 30-ovember 2, 2012 Frontiers in Biorefining, Chemicals and Products
Role of Quantum Chemistry in Atmospheric Chemical Mechanism Development
 Role of Quantum Chemistry in Atmospheric Chemical Mechanism Development Renyi Zhang and Jun Zhao Department of Atmospheric Sciences Texas A&M University College Station, TX 77843 Presented at the international
Role of Quantum Chemistry in Atmospheric Chemical Mechanism Development Renyi Zhang and Jun Zhao Department of Atmospheric Sciences Texas A&M University College Station, TX 77843 Presented at the international
Theoretical study of the BF 3-promoted rearrangement of oxiranyl N-methyliminodiacetic acid boronates
 Theoretical study of the BF 3-promoted rearrangement of oxiranyl N-methyliminodiacetic acid boronates Margarita M. Vallejos a* and Silvina C. Pellegrinet b* a Laboratorio de Química Orgánica, IQUIBA-NEA,
Theoretical study of the BF 3-promoted rearrangement of oxiranyl N-methyliminodiacetic acid boronates Margarita M. Vallejos a* and Silvina C. Pellegrinet b* a Laboratorio de Química Orgánica, IQUIBA-NEA,
Lecture Topics: I. Electrophilic Aromatic Substitution (EAS)
 Reactions of Aromatic Compounds Reading: Wade chapter 17, sections 17-1- 17-15 Study Problems: 17-44, 17-46, 17-47, 17-48, 17-51, 17-52, 17-53, 17-59, 17-61 Key Concepts and Skills: Predict and propose
Reactions of Aromatic Compounds Reading: Wade chapter 17, sections 17-1- 17-15 Study Problems: 17-44, 17-46, 17-47, 17-48, 17-51, 17-52, 17-53, 17-59, 17-61 Key Concepts and Skills: Predict and propose
Temperature and pressure dependent rate coefficients for the reaction of vinyl radical with molecular oxygen
 Paper # 070RK-0274 Topic: Reaction Kinetics 8 th U. S. National Combustion Meeting rganized by the Western States Section of the Combustion Institute and hosted by the University of Utah May 19-22, 2013
Paper # 070RK-0274 Topic: Reaction Kinetics 8 th U. S. National Combustion Meeting rganized by the Western States Section of the Combustion Institute and hosted by the University of Utah May 19-22, 2013
An Aromatics and cc 5 H 5-8 Model with Improved Transport for Flat Flames, Flow Reactors and Shock Tubes.
 8 th US National Combustion Meeting Organized by the Western States Section of the Combustion Institute and hosted by the University of Utah May 19-22, 2013 An Aromatics and cc 5 H 5-8 Model with Improved
8 th US National Combustion Meeting Organized by the Western States Section of the Combustion Institute and hosted by the University of Utah May 19-22, 2013 An Aromatics and cc 5 H 5-8 Model with Improved
Energy Barriers and Rates - Transition State Theory for Physicists
 Energy Barriers and Rates - Transition State Theory for Physicists Daniel C. Elton October 12, 2013 Useful relations 1 cal = 4.184 J 1 kcal mole 1 = 0.0434 ev per particle 1 kj mole 1 = 0.0104 ev per particle
Energy Barriers and Rates - Transition State Theory for Physicists Daniel C. Elton October 12, 2013 Useful relations 1 cal = 4.184 J 1 kcal mole 1 = 0.0434 ev per particle 1 kj mole 1 = 0.0104 ev per particle
Pathway and Kinetic Analysis on the Propyl Radical + O2 Reaction System
 URL-J-126375 Rev. 1 PREPRINT Pathway and Kinetic Analysis on the Propyl Radical + 2 Reaction System J.W. Bozzelli W.J. Pitz This paper was prepared for submittal to the Fourth International onference on
URL-J-126375 Rev. 1 PREPRINT Pathway and Kinetic Analysis on the Propyl Radical + 2 Reaction System J.W. Bozzelli W.J. Pitz This paper was prepared for submittal to the Fourth International onference on
Chapter 16 Aromatic Compounds. Discovery of Benzene
 Chapter 16 Aromatic Compounds Discovery of Benzene Isolated in 1825 by Michael Faraday who determined C: ratio to be 1:1. Synthesized in 1834 by Eilhard Mitscherlich who determined molecular formula to
Chapter 16 Aromatic Compounds Discovery of Benzene Isolated in 1825 by Michael Faraday who determined C: ratio to be 1:1. Synthesized in 1834 by Eilhard Mitscherlich who determined molecular formula to
Chemical Sciences and Engineering Department, Argonne National Laboratory, 9700 S. Cass Ave., Argonne, IL 60439
 8 th U. S. National Combustion Meeting rganized by the Western States Section of the Combustion Institute and hosted by the University of Utah May 19-22, 2013 A Shock Tube Laser Schlieren Study of Methyl
8 th U. S. National Combustion Meeting rganized by the Western States Section of the Combustion Institute and hosted by the University of Utah May 19-22, 2013 A Shock Tube Laser Schlieren Study of Methyl
AD-A i ELECTE. (*Sandia National Laboratories, Livermore, CA) OFFICE OF NAVAL RESEARCH. R&T Project Code 413 a 001
 AD-A262 1 1 5 OFFICE OF NAVAL RESEARCH R&T Project Code 413 a 001 Contract No. NOOO14-89-J-1235 Technical Report No. 16 Kinetics and Thermochemistry of Tetramethyl and Tetraethyl Orthosilicates In the
AD-A262 1 1 5 OFFICE OF NAVAL RESEARCH R&T Project Code 413 a 001 Contract No. NOOO14-89-J-1235 Technical Report No. 16 Kinetics and Thermochemistry of Tetramethyl and Tetraethyl Orthosilicates In the
Chemistry 204: Benzene and Aromaticity
 Chemistry 204: Benzene and Aromaticity Structure of and Bonding in Benzene benzene, C 6 H 6, was first isolated in 1825 (Michael Faraday), but it was not until more than 100 years later that an adequate
Chemistry 204: Benzene and Aromaticity Structure of and Bonding in Benzene benzene, C 6 H 6, was first isolated in 1825 (Michael Faraday), but it was not until more than 100 years later that an adequate
DFT Study on the Reaction of Molybdenum with Acetaldehyde in Gas Phase
 Asian Journal of Chemistry; Vol. 25, No. 1 (2013), 89-94 http://dx.doi.org/10.14233/ajchem.2013.12753 DFT Study on the Reaction of Molybdenum with Acetaldehyde in Gas Phase YONG WANG 1,2 and GUO-LIANG
Asian Journal of Chemistry; Vol. 25, No. 1 (2013), 89-94 http://dx.doi.org/10.14233/ajchem.2013.12753 DFT Study on the Reaction of Molybdenum with Acetaldehyde in Gas Phase YONG WANG 1,2 and GUO-LIANG
2 Reaction kinetics in gases
 2 Reaction kinetics in gases October 8, 2014 In a reaction between two species, for example a fuel and an oxidizer, bonds are broken up and new are established in the collision between the species. In
2 Reaction kinetics in gases October 8, 2014 In a reaction between two species, for example a fuel and an oxidizer, bonds are broken up and new are established in the collision between the species. In
Supplementary information. Nitrogen backbone oligomers
 Supplementary information Nitrogen backbone oligomers Hongbo Wang 1,2, Mikhail I. Eremets 1 *, Ivan Troyan 1,3, Hanyu Liu 2, Yanming Ma 2, Luc Vereecken 1 1 Max Planck Institute for Chemistry, Biogeochemistry
Supplementary information Nitrogen backbone oligomers Hongbo Wang 1,2, Mikhail I. Eremets 1 *, Ivan Troyan 1,3, Hanyu Liu 2, Yanming Ma 2, Luc Vereecken 1 1 Max Planck Institute for Chemistry, Biogeochemistry
Notes. Rza Abbasoglu*, Abdurrahman Atalay & Ahmet Şenocak. Indian Journal of Chemistry Vol. 53A, March, 2014, pp
 Indian Journal of Chemistry Vol. 53A, March, 2014, pp. 288-293 Notes DFT investigations of the Diels-Alder reaction of fulvene with tetracyclo[6.2.2.1 3,6.0 2,7 ]trideca-4,9,11-triene- 9,10-dicarboxylic
Indian Journal of Chemistry Vol. 53A, March, 2014, pp. 288-293 Notes DFT investigations of the Diels-Alder reaction of fulvene with tetracyclo[6.2.2.1 3,6.0 2,7 ]trideca-4,9,11-triene- 9,10-dicarboxylic
Chapter 09 Benzene and Its Derivatives
 Chapter 09 Benzene and Its Derivatives Benzene First isolated in 1825 from whale oil by Michael Faraday Unsaturated hydrocarbon but did not have the typical reactivity of alkenes or alkynes. CM 240: Fall
Chapter 09 Benzene and Its Derivatives Benzene First isolated in 1825 from whale oil by Michael Faraday Unsaturated hydrocarbon but did not have the typical reactivity of alkenes or alkynes. CM 240: Fall
Radical Reactions. Radical Stability!!! bond dissociation energies X Y X + Y. bond BDE (kcal/mol) bond BDE (kcal/mol) CH 3 CH 3 CH 2 95 O H R 2 C H
 adical eactions adical Stability!!! bond dissociation energies X Y X Y bond BDE (kcal/mol) bond BDE (kcal/mol) C 3 104 108 C 3 C 2 98 110 95 2 C 102 (-) 93 (C-) 92 C 3 C 3 36 89 85 C 3 C 3 80 adical eactions
adical eactions adical Stability!!! bond dissociation energies X Y X Y bond BDE (kcal/mol) bond BDE (kcal/mol) C 3 104 108 C 3 C 2 98 110 95 2 C 102 (-) 93 (C-) 92 C 3 C 3 36 89 85 C 3 C 3 80 adical eactions
Chapter 5. Aromatic Compounds
 Chapter 5. Aromatic Compounds 5.1 Structure of Benzene: The Kekule Proposal Mid-1800s, benzene was known to have the molecular formula C 6 6. Benzene reacts with 2 in the presence of iron to give substitution
Chapter 5. Aromatic Compounds 5.1 Structure of Benzene: The Kekule Proposal Mid-1800s, benzene was known to have the molecular formula C 6 6. Benzene reacts with 2 in the presence of iron to give substitution
4 - BENZENE: AROMATICITY, CONJUGATION AND ASSOCIATED REACTIVITY
 4 - BENZENE: AROMATICITY, CONJUGATION AND ASSOCIATED REACTIVITY During the early 1800's, a group of compounds of natural origin became collectively known as aromatic compounds. As several of these compounds
4 - BENZENE: AROMATICITY, CONJUGATION AND ASSOCIATED REACTIVITY During the early 1800's, a group of compounds of natural origin became collectively known as aromatic compounds. As several of these compounds
Thermal Degradation of Polymers
 Thermal Degradation of Polymers David A. Gallagher, CAChe Group, Fujitsu America, Inc. The following is a summary of the article "Computer-aided Chemistry Techniques Reveal Thermal Degradation mechanisms
Thermal Degradation of Polymers David A. Gallagher, CAChe Group, Fujitsu America, Inc. The following is a summary of the article "Computer-aided Chemistry Techniques Reveal Thermal Degradation mechanisms
Chap. 8 Substitution Reactions
 Chap. 8 Substitution Reactions Y + R X R' Y + X Nucleophilic not necessarily the same as R Electrophilic S N 1 slow (C 3 ) 3 CCl (C + Cl - 3 ) 3 C + (C 3 ) 3 C + OC 2 C 3 C 3 C 2 O C 3 C 2 O d[( C ) 3CCl]
Chap. 8 Substitution Reactions Y + R X R' Y + X Nucleophilic not necessarily the same as R Electrophilic S N 1 slow (C 3 ) 3 CCl (C + Cl - 3 ) 3 C + (C 3 ) 3 C + OC 2 C 3 C 3 C 2 O C 3 C 2 O d[( C ) 3CCl]
Foundations of Chemical Kinetics. Lecture 12: Transition-state theory: The thermodynamic formalism
 Foundations of Chemical Kinetics Lecture 12: Transition-state theory: The thermodynamic formalism Marc R. Roussel Department of Chemistry and Biochemistry Breaking it down We can break down an elementary
Foundations of Chemical Kinetics Lecture 12: Transition-state theory: The thermodynamic formalism Marc R. Roussel Department of Chemistry and Biochemistry Breaking it down We can break down an elementary
3 - CONJUGATION. More than one double bond can be in a given compound: n=0
 3 - NJUGATIN 1. Terminology and Nomenclature (SF 13.1 13.6; SFS 13.1 13.6) A compound containing a double bond is called an alkene, olefin or maybe simply "ene". There are often other names associated
3 - NJUGATIN 1. Terminology and Nomenclature (SF 13.1 13.6; SFS 13.1 13.6) A compound containing a double bond is called an alkene, olefin or maybe simply "ene". There are often other names associated
Mechanisms of H- and OH-assisted CO activation as well as C-C coupling on the flat Co(0001) surface Revisited
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2016 Mechanisms of H- and OH-assisted CO activation as well as C-C coupling on
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2016 Mechanisms of H- and OH-assisted CO activation as well as C-C coupling on
Benzene and Aromaticity
 Benzene and Aromaticity Why this Chapter? Reactivity of substituted aromatic compounds is tied to their structure Aromatic compounds provide a sensitive probe for studying relationship between structure
Benzene and Aromaticity Why this Chapter? Reactivity of substituted aromatic compounds is tied to their structure Aromatic compounds provide a sensitive probe for studying relationship between structure
Theoretical Models for Chemical Kinetics
 Theoretical Models for Chemical Kinetics Thus far we have calculated rate laws, rate constants, reaction orders, etc. based on observations of macroscopic properties, but what is happening at the molecular
Theoretical Models for Chemical Kinetics Thus far we have calculated rate laws, rate constants, reaction orders, etc. based on observations of macroscopic properties, but what is happening at the molecular
ReaxFF Reactive Force Field for Molecular Dynamics Simulations of Hydrocarbon Oxidation
 1040 J. Phys. Chem. A 2008, 112, 1040-1053 ReaxFF Reactive Force Field for Molecular Dynamics Simulations of Hydrocarbon Oxidation Kimberly Chenoweth, Adri C. T. van Duin, and William A. Goddard, III*
1040 J. Phys. Chem. A 2008, 112, 1040-1053 ReaxFF Reactive Force Field for Molecular Dynamics Simulations of Hydrocarbon Oxidation Kimberly Chenoweth, Adri C. T. van Duin, and William A. Goddard, III*
I5 ELECTROPHILIC SUBSTITUTIONS OF
 Section I Aromatic chemistry I5 ELECTPILIC SUBSTITUTINS F MN-SUBSTITUTED AMATIC INGS Key Notes ortho, meta and para substitution Substituent effect eaction profile Activating groups inductive o/p Deactivating
Section I Aromatic chemistry I5 ELECTPILIC SUBSTITUTINS F MN-SUBSTITUTED AMATIC INGS Key Notes ortho, meta and para substitution Substituent effect eaction profile Activating groups inductive o/p Deactivating
Organic Chemistry. Second Edition. Chapter 18 Aromatic Compounds. David Klein. Klein, Organic Chemistry 2e
 Organic Chemistry Second Edition David Klein Chapter 18 Aromatic Compounds Copyright 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2e 18.1 Introduction to Aromatic Compounds
Organic Chemistry Second Edition David Klein Chapter 18 Aromatic Compounds Copyright 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2e 18.1 Introduction to Aromatic Compounds
A multifacet mechanism for the OH HNO 3 reaction: An ab initio molecular orbitalõstatistical theory study
 JOURNAL OF CHEMICAL PHYSICS VOLUME 114, NUMBER 10 8 MARCH 2001 A multifacet mechanism for the OH HNO 3 reaction: An ab initio molecular orbitalõstatistical theory study W. S. Xia a) and M. C. Lin b) Department
JOURNAL OF CHEMICAL PHYSICS VOLUME 114, NUMBER 10 8 MARCH 2001 A multifacet mechanism for the OH HNO 3 reaction: An ab initio molecular orbitalõstatistical theory study W. S. Xia a) and M. C. Lin b) Department
Reaction Mechanism of Polar Diels-Alder Reactions between 3-Nitrofuran and different Dienes. A Theoretical Study.
 Reaction Mechanism of Polar Diels-Alder Reactions between 3-Nitrofuran and different Dienes. A Theoretical Study. MAUR CAINELLI, CARLA RMACHEA, PEDR MANCINI, MARÍA KNEETEMAN* Área Química rgánica Departamento
Reaction Mechanism of Polar Diels-Alder Reactions between 3-Nitrofuran and different Dienes. A Theoretical Study. MAUR CAINELLI, CARLA RMACHEA, PEDR MANCINI, MARÍA KNEETEMAN* Área Química rgánica Departamento
The Abstraction of Iodine from Aromatic Iodides by Alkyl Radicals. Steric and Electronic Effects 1
 Supporting Information: The Abstraction of Iodine from Aromatic Iodides by Alkyl Radicals. Steric and Electronic Effects 1 Darko Dolenc* and Božo Plesničar University of Ljubljana, Faculty of Chemistry
Supporting Information: The Abstraction of Iodine from Aromatic Iodides by Alkyl Radicals. Steric and Electronic Effects 1 Darko Dolenc* and Božo Plesničar University of Ljubljana, Faculty of Chemistry
Understanding Chemical Reactions through Computer Modeling. Tyler R. Josephson University of Delaware 4/21/16
 Understanding Chemical Reactions through Computer Modeling Tyler R. Josephson University of Delaware 4/21/16 A little about me B.S. in Chem E from U of M, 2011 Currently, Ph.D. student at University of
Understanding Chemical Reactions through Computer Modeling Tyler R. Josephson University of Delaware 4/21/16 A little about me B.S. in Chem E from U of M, 2011 Currently, Ph.D. student at University of
Physical Chemistry Chemical Physics
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2018 Physical Chemistry Chemical Physics Supporting Information Thermochemistry of the
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2018 Physical Chemistry Chemical Physics Supporting Information Thermochemistry of the
Aromatic Compounds. A number of these compounds had a distinct odor. Hence these compounds were called aromatic
 Aromatic Compounds Early in the history of organic chemistry (late 18 th, early 19 th century) chemists discovered a class of compounds which were unusually stable A number of these compounds had a distinct
Aromatic Compounds Early in the history of organic chemistry (late 18 th, early 19 th century) chemists discovered a class of compounds which were unusually stable A number of these compounds had a distinct
Classification of Reactions by:
 lassification of Reactions by: 1) Functional group 2) Kind a) Addition: A + B > b) Elimination: A > B + c) Substitution: A-B + -D > A- + B-D d) Rearrangement: A > B, where B is a constitutional isomer
lassification of Reactions by: 1) Functional group 2) Kind a) Addition: A + B > b) Elimination: A > B + c) Substitution: A-B + -D > A- + B-D d) Rearrangement: A > B, where B is a constitutional isomer
12/27/2010. Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
Electronic Supplementary Information. for
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information for Two Chiral Catalysts in Action: Insights on Cooperativity
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information for Two Chiral Catalysts in Action: Insights on Cooperativity
Chapter 16: Aromatic Compounds
 Chamras Chemistry 106 Lecture otes xamination 2 Materials Chapter 16: Aromatic Compounds Benzene, the Most Commonly Known Aromatic Compound: The aromatic nature of benzene stabilizes it 36 kcal.mol 1.
Chamras Chemistry 106 Lecture otes xamination 2 Materials Chapter 16: Aromatic Compounds Benzene, the Most Commonly Known Aromatic Compound: The aromatic nature of benzene stabilizes it 36 kcal.mol 1.
Molecular Orbital (MO) Theory
 Molecular rbital (M) Theory Molecular orbital mixing takes place at all times along reaction coordinate. (S N 2) + + C- σ As - approaches, and as - departs, orbitals mix. sp 3 C- σ Molecular rbital (M)
Molecular rbital (M) Theory Molecular orbital mixing takes place at all times along reaction coordinate. (S N 2) + + C- σ As - approaches, and as - departs, orbitals mix. sp 3 C- σ Molecular rbital (M)
Journal of Chemical and Pharmaceutical Research
 Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 The controversial reaction mechanism of Stevens rearrangement: A review Soumendranath
Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 The controversial reaction mechanism of Stevens rearrangement: A review Soumendranath
This paper is part of the following report: UNCLASSIFIED
 UNCLASSIFIED Defense Technical Information Center Compilation Part Notice ADP023624 TITLE: Ignition Kinetics in Fuels Oxidation DISTRIBUTION: Approved for public release, distribution unlimited This paper
UNCLASSIFIED Defense Technical Information Center Compilation Part Notice ADP023624 TITLE: Ignition Kinetics in Fuels Oxidation DISTRIBUTION: Approved for public release, distribution unlimited This paper
Computational Chemistry Problem Set 4 Due Monday, February 22, 2011 (at the start of class) Total Number of Points = 85
 Computational Chemistry Problem Set 4 Due Monday, February 22, 2011 (at the start of class) Total Number of Points = 85 Basic Technical Notes: (1) For security reasons, you are allowed to log into the
Computational Chemistry Problem Set 4 Due Monday, February 22, 2011 (at the start of class) Total Number of Points = 85 Basic Technical Notes: (1) For security reasons, you are allowed to log into the
Chapter 8: Alkene Structure and Preparation via Elimination Reactions
 Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
Author(s) Mendes, Jorge; Zhou, Chong-Wen; Curran, Henry J.
 Provided by the author(s) and NUI Galway in accordance with publisher policies. Please cite the published version when available. Title Theoretical study of the rate constants for the hydrogen atom abstraction
Provided by the author(s) and NUI Galway in accordance with publisher policies. Please cite the published version when available. Title Theoretical study of the rate constants for the hydrogen atom abstraction
Chapter 17. Preview. Lesson Starter Objectives Reaction Mechanisms Collision Theory Activation Energy The Activated Complex Sample Problem A
 Preview Lesson Starter Objectives Reaction Mechanisms Collision Theory Activation Energy The Activated Complex Sample Problem A Section 1 The Reaction Process Lesson Starter The reaction H 2 + I 2 2HI
Preview Lesson Starter Objectives Reaction Mechanisms Collision Theory Activation Energy The Activated Complex Sample Problem A Section 1 The Reaction Process Lesson Starter The reaction H 2 + I 2 2HI
Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution
 John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution Paul D. Adams University of Arkansas Substitution Reactions of Benzene and Its Derivatives
John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution Paul D. Adams University of Arkansas Substitution Reactions of Benzene and Its Derivatives
11/30/ Substituent Effects in Electrophilic Substitutions. Substituent Effects in Electrophilic Substitutions
 Chapter 9 Problems: 9.1-29, 32-34, 36-37, 39-45, 48-56, 58-59, 61-69, 71-72. 9.8 Substituent effects in the electrophilic substitution of an aromatic ring Substituents affect the reactivity of the aromatic
Chapter 9 Problems: 9.1-29, 32-34, 36-37, 39-45, 48-56, 58-59, 61-69, 71-72. 9.8 Substituent effects in the electrophilic substitution of an aromatic ring Substituents affect the reactivity of the aromatic
The carbon-carbon double bond is the distinguishing feature of alkenes.
 Alkenes: Structure & Properties Alkane (acyclic): n 2n+2 > saturated. Alkene (acyclic): n 2n > unsaturated. eg ethylene (IUPA: ethene), 2 4 : 2 = 2 The carbon-carbon double bond is the distinguishing feature
Alkenes: Structure & Properties Alkane (acyclic): n 2n+2 > saturated. Alkene (acyclic): n 2n > unsaturated. eg ethylene (IUPA: ethene), 2 4 : 2 = 2 The carbon-carbon double bond is the distinguishing feature
CHEM 344 Molecular Modeling
 CHEM 344 Molecular Modeling The Use of Computational Chemistry to Support Experimental Organic Chemistry Part 1: Molecular Orbital Theory, Hybridization, & Formal Charge * all calculation data obtained
CHEM 344 Molecular Modeling The Use of Computational Chemistry to Support Experimental Organic Chemistry Part 1: Molecular Orbital Theory, Hybridization, & Formal Charge * all calculation data obtained
Ab Initio Molecular Orbital Calculations of Electronic Effects on the Kinetics of Cyclopropylcarbinyl Radical Ring Openings
 3618 J. Org. Chem. 1998, 63, 3618-3623 Ab Initio Molecular Orbital Calculations of Electronic Effects on the Kinetics of Cyclopropylcarbinyl Radical Ring Openings Felix N. Martinez, H. Bernhard Schlegel,*
3618 J. Org. Chem. 1998, 63, 3618-3623 Ab Initio Molecular Orbital Calculations of Electronic Effects on the Kinetics of Cyclopropylcarbinyl Radical Ring Openings Felix N. Martinez, H. Bernhard Schlegel,*
Investigation of the Mechanism of the Reaction between Atomic Oxygen Radical Anion and Benzene
 ACTA PHYSICO-CHIMICA SINICA Volume 22, Issue 9, September 2006 Online English edition of the Chinese language journal. RESEARCH PAPER Investigation of the Mechanism of the Reaction between Atomic Oxygen
ACTA PHYSICO-CHIMICA SINICA Volume 22, Issue 9, September 2006 Online English edition of the Chinese language journal. RESEARCH PAPER Investigation of the Mechanism of the Reaction between Atomic Oxygen
COMBUSTION CHEMISTRY COMBUSTION AND FUELS
 COMBUSTION CHEMISTRY CHEMICAL REACTION AND THE RATE OF REACTION General chemical reaction αa + βb = γc + δd A and B are substracts and C and are products, α, β, γ and δ are stoichiometric coefficients.
COMBUSTION CHEMISTRY CHEMICAL REACTION AND THE RATE OF REACTION General chemical reaction αa + βb = γc + δd A and B are substracts and C and are products, α, β, γ and δ are stoichiometric coefficients.
Reactions in the Al-H-Cl System Studied by ab Initio Molecular Orbital and Density Functional Methods
 264 J. Phys. Chem. A 2001, 105, 264-273 Reactions in the Al-H-Cl System Studied by ab Initio Molecular Orbital and Density Functional Methods Mark T. Swihart* Department of Chemical Engineering, UniVersity
264 J. Phys. Chem. A 2001, 105, 264-273 Reactions in the Al-H-Cl System Studied by ab Initio Molecular Orbital and Density Functional Methods Mark T. Swihart* Department of Chemical Engineering, UniVersity
Supporting information for: On the atmospheric fate of methacrolein: 1. Peroxy. radical isomerization following addition of OH and O 2
 Supporting information for: On the atmospheric fate of methacrolein: 1. Peroxy radical isomerization following addition of OH and O 2 John D. Crounse,, Hasse C. Knap, Kristian B. Ørnsø, Solvejg Jørgensen,
Supporting information for: On the atmospheric fate of methacrolein: 1. Peroxy radical isomerization following addition of OH and O 2 John D. Crounse,, Hasse C. Knap, Kristian B. Ørnsø, Solvejg Jørgensen,
Chapter 15. Reactions of Aromatic Compounds. 1. Electrophilic Aromatic Substitution Reactions
 hapter 15 eactions of Aromatic ompounds 1. Electrophilic Aromatic Substitution eactions v verall reaction reated by Professor William Tam & Dr. Phillis hang opyright S 3 2 S 4 S 3 2. A General Mechanism
hapter 15 eactions of Aromatic ompounds 1. Electrophilic Aromatic Substitution eactions v verall reaction reated by Professor William Tam & Dr. Phillis hang opyright S 3 2 S 4 S 3 2. A General Mechanism
( ) Thermodynamic selectivity (reversible reactions)
 II. Reactivity A) General Principles Fundamental Equations G o Ea / RT = "RT ln K eq k = Ae 1) Kinetics vs Thermodynamics Kinetic selectivity (irreversible reactions) [ P 1 ] [ ] = ln k 1 P 2 ( ) = " #G
II. Reactivity A) General Principles Fundamental Equations G o Ea / RT = "RT ln K eq k = Ae 1) Kinetics vs Thermodynamics Kinetic selectivity (irreversible reactions) [ P 1 ] [ ] = ln k 1 P 2 ( ) = " #G
Ground State Calculations for Protonated PAHs
 15 Chapter 2 Ground State Calculations for Protonated PAHs 2.1 Introduction Ab initio ground electronic state calculations can be used to determine geometries and energies of all possible protonated PAH
15 Chapter 2 Ground State Calculations for Protonated PAHs 2.1 Introduction Ab initio ground electronic state calculations can be used to determine geometries and energies of all possible protonated PAH
CHEM 251 (4 credits): Description
 CHEM 251 (4 credits): Intermediate Reactions of Nucleophiles and Electrophiles (Reactivity 2) Description: An understanding of chemical reactivity, initiated in Reactivity 1, is further developed based
CHEM 251 (4 credits): Intermediate Reactions of Nucleophiles and Electrophiles (Reactivity 2) Description: An understanding of chemical reactivity, initiated in Reactivity 1, is further developed based
An ab initio study of the mechanism of the cycloaddition reaction forming bicyclic compounds between vinylidene (H 2 C=C:) and ethylene
 J. Serb. Chem. Soc. 76 (5) 743 749 (2011) UDC 547.313.2+541.124: JSCS 4155 66.095.252.091.7:536.722 Original scientific paper An ab initio study of the mechanism of the cycloaddition reaction forming bicyclic
J. Serb. Chem. Soc. 76 (5) 743 749 (2011) UDC 547.313.2+541.124: JSCS 4155 66.095.252.091.7:536.722 Original scientific paper An ab initio study of the mechanism of the cycloaddition reaction forming bicyclic
Supplementary Information for:
 Supplementary Information for: Towards Active and Stable Oxygen Reduction Cathode: A Density Functional Theory Survey on Pt 2 M skin alloys Guang-Feng Wei and Zhi-Pan Liu* Shanghai Key Laboratory of lecular
Supplementary Information for: Towards Active and Stable Oxygen Reduction Cathode: A Density Functional Theory Survey on Pt 2 M skin alloys Guang-Feng Wei and Zhi-Pan Liu* Shanghai Key Laboratory of lecular
Supporting Information for
 Supporting Information for Factors Controlling the Reactivity and Chemoselectivity of Resonance Destabilized Amides in Ni-catalyzed Decarbonylative and Non-decarbonylative Suzuki-Miyaura Coupling Chong-Lei
Supporting Information for Factors Controlling the Reactivity and Chemoselectivity of Resonance Destabilized Amides in Ni-catalyzed Decarbonylative and Non-decarbonylative Suzuki-Miyaura Coupling Chong-Lei
Prediction of Absolute Rate Constants for the Reactions of NH 2 with Alkanes from ab Initio G2M/TST Calculations
 2088 J. Phys. Chem. A 1999, 103, 2088-2096 Prediction of Absolute Rate Constants for the Reactions of NH 2 with Alkanes from ab Initio G2M/TST Calculations A. M. Mebel, and M. C. Lin*, Institute of Atomic
2088 J. Phys. Chem. A 1999, 103, 2088-2096 Prediction of Absolute Rate Constants for the Reactions of NH 2 with Alkanes from ab Initio G2M/TST Calculations A. M. Mebel, and M. C. Lin*, Institute of Atomic
Chapter 8: Alkene Structure and Preparation via Elimination Reactions
 1. Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C bond length bond strength 3 C C 3 3 C C 3 3 C C 3 3 C 2 C C 2 3 C a C=C double bond
1. Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C bond length bond strength 3 C C 3 3 C C 3 3 C C 3 3 C 2 C C 2 3 C a C=C double bond
Structure, Electronic and Nonlinear Optical Properties of Furyloxazoles and Thienyloxazoles
 Journal of Physics: Conference Series PAPER OPEN ACCESS Structure, Electronic and Nonlinear Optical Properties of Furyls and Thienyls To cite this article: Ozlem Dagli et al 6 J. Phys.: Conf. Ser. 77 View
Journal of Physics: Conference Series PAPER OPEN ACCESS Structure, Electronic and Nonlinear Optical Properties of Furyls and Thienyls To cite this article: Ozlem Dagli et al 6 J. Phys.: Conf. Ser. 77 View
Chemical Kinetics. Topic 7
 Chemical Kinetics Topic 7 Corrosion of Titanic wrec Casón shipwrec 2Fe(s) + 3/2O 2 (g) + H 2 O --> Fe 2 O 3.H 2 O(s) 2Na(s) + 2H 2 O --> 2NaOH(aq) + H 2 (g) Two examples of the time needed for a chemical
Chemical Kinetics Topic 7 Corrosion of Titanic wrec Casón shipwrec 2Fe(s) + 3/2O 2 (g) + H 2 O --> Fe 2 O 3.H 2 O(s) 2Na(s) + 2H 2 O --> 2NaOH(aq) + H 2 (g) Two examples of the time needed for a chemical
Chapter 19: Aromatic Substitution Reactions
 Chem A225 Notes Page 52 Chapter 19: Aromatic Substitution Reactions Topic One: lectrophilic Aromatic Substitution I. Introduction to lectrophilic Aromatic Substitution (AS) A. eneral Reaction Pattern B.
Chem A225 Notes Page 52 Chapter 19: Aromatic Substitution Reactions Topic One: lectrophilic Aromatic Substitution I. Introduction to lectrophilic Aromatic Substitution (AS) A. eneral Reaction Pattern B.
Quantitative Understanding of Advanced Oxidation Processes for the Treatment of Emerging Contaminants
 Quantitative Understanding of Advanced Oxidation Processes for the Treatment of Emerging Contaminants Daisuke Minakata and John Crittenden School of Civil and Environmental Engineering Brook Byers Institute
Quantitative Understanding of Advanced Oxidation Processes for the Treatment of Emerging Contaminants Daisuke Minakata and John Crittenden School of Civil and Environmental Engineering Brook Byers Institute
3. RATE LAW AND STOICHIOMETRY
 Page 1 of 39 3. RATE LAW AND STOICHIOMETRY Professional Reference Shelf R3.2 Abbreviated Lecture Notes Full Lecture Notes I. Overview II. Introduction A. The Transition State B. Procedure to Calculate
Page 1 of 39 3. RATE LAW AND STOICHIOMETRY Professional Reference Shelf R3.2 Abbreviated Lecture Notes Full Lecture Notes I. Overview II. Introduction A. The Transition State B. Procedure to Calculate
Pericyclic Reactions and Organic Photochemistry S. Sankararaman Department of Chemistry Indian Institute of Technology, Madras
 Pericyclic Reactions and Organic Photochemistry S. Sankararaman Department of Chemistry Indian Institute of Technology, Madras Module No. #02 Lecture No. #09 Pericyclic Reactions Cycloaddition Reactions
Pericyclic Reactions and Organic Photochemistry S. Sankararaman Department of Chemistry Indian Institute of Technology, Madras Module No. #02 Lecture No. #09 Pericyclic Reactions Cycloaddition Reactions
Classes of Alkenes. Alkenes and Alkynes. Saturated compounds (alkanes): Have the maximum number of hydrogen atoms attached to each carbon atom.
 Alkenes and Alkynes Saturated compounds (alkanes): ave the maximum number of hydrogen atoms attached to each carbon atom. Unsaturated compounds: ave fewer hydrogen atoms attached to the carbon chain than
Alkenes and Alkynes Saturated compounds (alkanes): ave the maximum number of hydrogen atoms attached to each carbon atom. Unsaturated compounds: ave fewer hydrogen atoms attached to the carbon chain than
Solution problem 22: Non-Benzoid Aromatic Sytems
 Solution problem 22: on-enzoid Aromatic Sytems 22.1 & 22.2 Each double bond and each heteroatom (, ) with lone pairs donates 2 π- electrons as well as a negative charge. oron or a positive charge does
Solution problem 22: on-enzoid Aromatic Sytems 22.1 & 22.2 Each double bond and each heteroatom (, ) with lone pairs donates 2 π- electrons as well as a negative charge. oron or a positive charge does
Problem 1 (2 points) Lone pair on N may donate to an empty p orbital of B on another molecule of aminoborane, leading to oligomers.
 Problem 1 (2 points) B N Lone pair on N may donate to an empty p orbital of B on another molecule of aminoborane, leading to oligomers. B N x 3 B N 3 3x 2 + (BN) x MW( 3 B N 3 ) = 30.87 g/mol MW( 2 ) =
Problem 1 (2 points) B N Lone pair on N may donate to an empty p orbital of B on another molecule of aminoborane, leading to oligomers. B N x 3 B N 3 3x 2 + (BN) x MW( 3 B N 3 ) = 30.87 g/mol MW( 2 ) =
Formation of aromatics in rich methane flames doped by unsaturated compounds
 Formation of aromatics in rich methane flames doped by unsaturated compounds H.A. Gueniche, P.A. Glaude *, R. Fournet and F. Battin-Leclerc Département de Chimie-Physique des Réactions, UMR n 7630 CNRS,
Formation of aromatics in rich methane flames doped by unsaturated compounds H.A. Gueniche, P.A. Glaude *, R. Fournet and F. Battin-Leclerc Département de Chimie-Physique des Réactions, UMR n 7630 CNRS,
CHEMICAL KINETICS (RATES OF REACTION)
 Kinetics F322 1 CHEMICAL KINETICS (RATES OF REACTION) Introduction Chemical kinetics is concerned with the dynamics of chemical reactions such as the way reactions take place and the rate (speed) of the
Kinetics F322 1 CHEMICAL KINETICS (RATES OF REACTION) Introduction Chemical kinetics is concerned with the dynamics of chemical reactions such as the way reactions take place and the rate (speed) of the
