Supplementary information
|
|
|
- Oswin Willis
- 5 years ago
- Views:
Transcription
1 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supplementary information Real-time imaging and elemental mapping of AgAu nanoparticle transformations E. A. Lewis, T. J. A. Slater, E. Prestat, A. Macedo, P. O Brien, P. H. C. Camargo and S. J. Haigh Discussion of the oxidation mechanism Assuming the starting material is a spherical particle 30 nm in diameter with composition Ag 96% Au 4%. The particle contains x10-18 moles of silver and has a surface area of 2.83 x m 2. The flux of an ideal gas is given by where P is the gas pressure, m is mass of the molecule, k the Boltzmann constant, and T the temperature. At a temperature of K and a pressure of 9x10-8 Torr (approximate column pressure in the microscope used in this work); the flux of O 2 molecules is 1.09 x m -2 s -1. This would result in 307 O 2 molecules s -1 hitting the surface of the 30 nm particle. If every collision was successful (i.e. results in the generation of 2Ag 2 O), complete conversion of the particle to the oxide would require 673 s. However, the literature contains a number of studies of oxygen chemisorption on Ag and Au surfaces which suggest the probability of a successful collision is very low. The dissociative adsorption of O 2 on a clean Ag surface is believed to have a low sticking probability, between 10-3 and The sticking probability of oxygen on gold is believed to be below Even if we use a generous value of 10-3, a 30 nm particle would require ~187 hours to completely oxidise. As we see significant oxidation occurring in ~400 s (e.g. Figure S1), atmospheric oxygen clearly doesn t play a significant role in the oxidation process observed in this study. We must therefore conclude that oxygen is not primarily supplied from the atmosphere but from the chemical species found on the surface of the nanoparticles and the surrounding region of substrate. The primary candidate is polyvinylpyrrolidone (PVP), a polymer employed in the nanoparticle synthesis which is found on the surface of the particles and deposited on the surrounding substrate during drop casting. 5 It has previously been observed that electron beam irradiation of Co nanoparticles coated in the oxygen containing ligand lauric acid result in partial oxidation of the particles, forming cobalt oxide shells. 6 However, this beam induced reaction didn t show the characteristic features of the Kirkendall effect, and in fact the beam induced shell formation was shown to inhibit ex situ Kirkendall hollowing. 6 We propose that an analogous oxidation mechanism occurs here with the electron beam interacting with the PVP to generate reactive oxygen species. The electron beam is commonly observed to draw organic contamination into the scan area. This process allows additional PVP to be drawn to the particle from the surrounding substrate during imaging.
2 Supplementary Figures Figure S1. Shell growth and void formation are observed for a range of initial morphologies. Sequences of HAADF STEM images show the beam induced structural evolution of 5 particles. In all cases the particles were exposed to a constant dose rate of ~2550 eå 2 s -1. All particles belong to the same sample of Ag-Au nanoparticles synthesised by the galvanic replacement reaction and containing ~6at.% Au 94at.% Ag. While some particles (e.g. a and b) are solid and approximately spherical, other particles show some hollowing (c) or faceting (d) in their initial structure. Despite different initial morphologies a number of universal mechanistic features are observed during the beam induced reaction. Initially shell growth is non uniform, with islands of Ag 2O emerging on the particle s surface which eventually coalesce to form a complete shell. Shell growth is accompanied by the formation and growth of voids in the core. In some cases a single void grows in the particles core, in other cases several voids form initially before coalescing to form a single void.
3 Figure S2. Quantitative analysis of void growth and coalescence. The nominal areas of the voids within a particle were measured with automated image thresholding. A self-consistent thresholding limit was applied to a series of 23 HAADF STEM images (Video S2) acquired as beam induced void growth and coalescence occurred. The particle studied is that shown in Figure 1. The total areal growth rate is found to remain approximately constant at ~0.74 nm 2 s -1. However, the trajectories of individual voids are less predictable, for example void 4 doesn t increase in size at any point, shrinking before eventually coalescing with Void 1. The growth of Void 1 shows distinct jumps in size corresponding to the points where it consumes neighbouring voids.
4 Figure S3. EDX spectrum imaging reveals growth of an Ag and O rich shell and inward diffusion of Au. EDX spectrum images of the same particles were recorded at different stages of the beam induced oxidation. (a) shows the particle before significant changes have occurred and (b) and (c) show later points of the beam induced reaction. Unlike the particle shown in Figure 1 this particle has some initial hollowing, this is a consequence of the galvanic replacement reaction used to synthesise the Ag-Au starting material. The spectrum images reveal that as the reaction proceeds, the area containing Ag grows. The Ag maps correlate strongly with the O maps suggesting the outward diffusion of Ag is driven by oxidation. Simultaneously the area containing Au decreases, it is believed that inward diffusion of Au occurs due to the increased concentration of vacancies in the core which accompanies the outward diffusion of Ag. Throughout this process the Ag:Au ratio remains constant (94at.% Ag, 6at.% Au) however, the amount of oxygen present (O/Ag at.% ratio) increases ~137% between (a) and (c), further supporting the oxidation hypothesis. The growth of an Ag 2O shell and the inward diffusion of Au are further demonstrated by the line scans shown in the right hand column.
5 a) b) c) Figure S4. The lattice resolution images in Figure 3 confirm that shell growth is due to Ag 2O formation. EDX spectrum images acquired for the particle shown in Figure 3 at various stages of the beam induced reaction show the changes in elemental distribution that accompany the formation of Ag 2O. The changing distributions of Ag, Au and O are similar to those seen in Figures 1 and S3, with Ag diffusing outwards while Au diffuses inwards and a strong correlation between the distributions of Ag and O.
6 Figure S5. Oxidation is substrate dependent. Upon heating to 400 C particles on amorphous carbon immediately alloy. However, after quenching, no beam induced restructuring occurred when exposed to the same dose rates that caused the transformations shown in Figures 1 and 4. The absence of void formation and hollowing cannot be attributed to a shortage of oxygen; quantification of spectrum images reveals that O/Ag at.% ratio increased 622% from an initial value of 0.67 (a) to a final value of 4.82 (c) (probably a result of the electron beam drawing PVP to the scan area) while the Ag:Au ratio remains constant at 97at.% Ag 3at.% Au. In fact, the final O/Ag ratio is actually considerably higher than that seen in comparable examples where reactions have occurred on Si 3N 4 grids (e.g. in Figure 1 the final ratio is 2.18), but despite more oxygen accumulating in the scanned area during imaging there is no evidence of shell growth and hollowing. Mapping the oxygen counts reveals that, in contrast to samples on Si 3N 4 (where O and Ag counts are clearly correlated), the support surrounding the particles contains more O than the area occupied by the AgAu particle. It is therefore postulated that the amorphous carbon support reacts preferentially with oxygen containing species, preventing Ag 2O formation.
7 Figure S6. Elemental maps corresponding to figure 4. (a) shows the initial Au surface segregated particles, (b) shows the homogeneously alloyed AgAu particles formed by heating to 400 C, (c-e) show the different stages of the beam induced shell growth and hollowing that occurs after quenching to room temperature. The maps of O and Ag counts for (e) suggest that the increase in oxygen counts (Figure 4f) that accompanies the beam induced reaction is due to the growth of an Ag 2O shell.
8 Figure S7. Heating Au surface segregated particles to 400 C results in alloying and some morphological changes (rounding of faceted edges and some loss of hollowing), however, the size and shape of the particle is largely retained and the ratio of Ag:Au remains identical to that measured at room temperature. In contrast, we observe a dramatic change in particle size, shape, and composition upon heating to 650 C. The particle shown in this figure shrinks dramatically upon heating, EDX quantification reveals that this shrinkage corresponds to a dramatic loss of Ag. The particle is initially 78at.% Ag 22at.% Au (coming from a different sample to the lower Au content particles used elsewhere in this paper) but the heated particle is 3at.% Ag 97at.% Au. This is consistent with previous reports which show that at high temperatures the lower melting point component of a bimetallic nanoparticle system will leech out, improving the particles high temperature stability. 7 The melting points of bulk Ag and Au are C and 1064 C respectively but nanoparticles show significant melting point depression. Pure Ag particles of a similar size to those studied here have been observed, by in situ TEM heating experiments, to sublime at C. 8 Supplementary Videos Video S1. Video of early stages of growth. Video comprised of a sequence of HAADF STEM images capturing the particle shown in Figure 1 during the early stages of the beam induced reaction. This video covers the period between the images shown Figures 1a and 1c. The particle is subjected to a constant dose rate of ~4990 eå 2 s -1 during the entire video. The Ag 2 O shell appears to emerge from at least 3 distinct sites upon the particle s surface. The video clip, available in the electronic supplementary material, plays at 4x real speed. Video S2. Video of void coalescence. Video comprised of a sequence of HAADF STEM images showing the growth and coalescence of voids in the particle shown in Figure 1. This video covers the period between the images shown Figures 1d and 1e. The particle is subjected to a constant dose rate of ~4990 eå 2 s -1 during the entire video. By this point in the growth trajectory, the multiple Ag 2 O islands seen in Video S1 have combined to form a complete shell and multiple voids have formed in the core. As the beam induced reaction proceeds the voids continue to grow and eventually coalesce. A quantitative analysis of this image sequence can be found in supplementary Figure S2. The video clip, available in the electronic supplementary material, plays at 4x real speed. Video S3. Video comprised of a sequence of HAADF STEM images showing shell growth and void formation in a cluster of alloyed AgAu nanoparticles. This video covers the period between the images shown in Figures 4d and 4e. The particles were exposed to a constant dose rate of ~2550 eå 2 s -1.Particles alloyed by heating undergo a beam induced reaction after they are returned to room temperature. This reaction shows similar characteristics to the reaction observed in Au surface segregated particles. However, the proximity of particles to other particles undergoing the same beam induced reaction influences the final morphology, with the growing shells of neighbouring particles contacting and merging. The video clip, available in the electronic supplementary material, plays at ~9x real speed. Supporting References 1. P. J. Goddard and R. M. Lambert, Surface Science, 1981, 107, G. Rovida and F. Pratesi, Surface Science, 1975, 52, W.-X. Li, C. Stampfl and M. Scheffler, Physical Review B, 2003, 67, N. D. S. Canning, D. Outka and R. J. Madix, Surface Science, 1984, 141, T. J. A. Slater, A. Macedo, S. L. M. Schroeder, M. G. Burke, P. O Brien, P. H. C. Camargo and S. J. Haigh, Nano Letters, 2014, 14, Z. Yang, M. Walls, I. Lisiecki and M.-P. Pileni, Chemistry of Materials, 2013, 25, A. Cao and G. Veser, Nature Materials, 2010, 9, M. A. Asoro, D. Kovar and P. J. Ferreira, ACS Nano, 2013, 7,
Advanced electron microscopy techniques for mechanistic studies of the growth and transformation of nanocrystals
 The University of Manchester Advanced electron microscopy techniques for mechanistic studies of the growth and transformation of nanocrystals Edward A. Lewis 2016 1 Contents Publication list... 5 2016...
The University of Manchester Advanced electron microscopy techniques for mechanistic studies of the growth and transformation of nanocrystals Edward A. Lewis 2016 1 Contents Publication list... 5 2016...
Justin G. Railsback, Aaron C. Johnston-Peck, Junwei Wang, and Joseph B. Tracy DOI: Sreeprasad T. S
 Size-Dependent Nanoscale Kirkendall Effect During the Oxidation of Nickel Nanoparticles Justin G. Railsback, Aaron C. Johnston-Peck, Junwei Wang, and Joseph B. Tracy Department of Materials Science and
Size-Dependent Nanoscale Kirkendall Effect During the Oxidation of Nickel Nanoparticles Justin G. Railsback, Aaron C. Johnston-Peck, Junwei Wang, and Joseph B. Tracy Department of Materials Science and
Special Properties of Au Nanoparticles
 Special Properties of Au Nanoparticles Maryam Ebrahimi Chem 7500/750 March 28 th, 2007 1 Outline Introduction The importance of unexpected electronic, geometric, and chemical properties of nanoparticles
Special Properties of Au Nanoparticles Maryam Ebrahimi Chem 7500/750 March 28 th, 2007 1 Outline Introduction The importance of unexpected electronic, geometric, and chemical properties of nanoparticles
Effect of Metal Concentration on Shape and Composition Changes in Gold-Silver Bimetallic Systems Md. Jahangir Alam
 Noto-are 15542466: Chemical technology. 2013-07-15. Effect of Metal Concentration on Shape and Composition Changes in Gold-Silver Bimetallic Systems Md. Jahangir Alam Department of Agronomy and Agricultural
Noto-are 15542466: Chemical technology. 2013-07-15. Effect of Metal Concentration on Shape and Composition Changes in Gold-Silver Bimetallic Systems Md. Jahangir Alam Department of Agronomy and Agricultural
Supplementary. respectively.
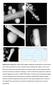 Figure S1. HAADF STEM images of Ag NRod interacting with Au NP (a and b) and Au NRod with Ag NP (c and d). Interface areas are labeled with ellipse. Itt can be seen thatt when interface was oriented parallel
Figure S1. HAADF STEM images of Ag NRod interacting with Au NP (a and b) and Au NRod with Ag NP (c and d). Interface areas are labeled with ellipse. Itt can be seen thatt when interface was oriented parallel
Supplementary Information (ESI) Synthesis of Ultrathin Platinum Nanoplates for Enhanced Oxygen Reduction Activity
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2017 Supplementary Information (ESI) Synthesis of Ultrathin Platinum Nanoplates for Enhanced
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2017 Supplementary Information (ESI) Synthesis of Ultrathin Platinum Nanoplates for Enhanced
Characterisation of Nanoparticle Structure by High Resolution Electron Microscopy
 Journal of Physics: Conference Series OPEN ACCESS Characterisation of Nanoparticle Structure by High Resolution Electron Microscopy To cite this article: Robert D Boyd et al 2014 J. Phys.: Conf. Ser. 522
Journal of Physics: Conference Series OPEN ACCESS Characterisation of Nanoparticle Structure by High Resolution Electron Microscopy To cite this article: Robert D Boyd et al 2014 J. Phys.: Conf. Ser. 522
k T m 8 B P m k T M T
 I. INTRODUCTION AND OBJECTIVE OF THE EXPERIENT The techniques for evaporation of chemicals in a vacuum are widely used for thin film deposition on rigid substrates, leading to multiple applications: production
I. INTRODUCTION AND OBJECTIVE OF THE EXPERIENT The techniques for evaporation of chemicals in a vacuum are widely used for thin film deposition on rigid substrates, leading to multiple applications: production
Electronic supplementary information for:
 Electronic supplementary information for: Charge-transfer-induced suppression of galvanic replacement and synthesis of (Au@Ag)@Au double shell nanoparticles for highly uniform, robust and sensitive bioprobes
Electronic supplementary information for: Charge-transfer-induced suppression of galvanic replacement and synthesis of (Au@Ag)@Au double shell nanoparticles for highly uniform, robust and sensitive bioprobes
Rh 3d. Co 2p. Binding Energy (ev) Binding Energy (ev) (b) (a)
 Co 2p Co(0) 778.3 Rh 3d Rh (0) 307.2 810 800 790 780 770 Binding Energy (ev) (a) 320 315 310 305 Binding Energy (ev) (b) Supplementary Figure 1 Photoemission features of a catalyst precursor which was
Co 2p Co(0) 778.3 Rh 3d Rh (0) 307.2 810 800 790 780 770 Binding Energy (ev) (a) 320 315 310 305 Binding Energy (ev) (b) Supplementary Figure 1 Photoemission features of a catalyst precursor which was
One-Pot Synthesis of Core-Shell-like Pt 3 Co Nanoparticle Electrocatalyst with Pt-enriched Surface for Oxygen Reduction Reaction in Fuel Cells
 Electronic Supplementary Information for One-Pot Synthesis of Core-Shell-like 3 Co Nanoparticle Electrocatalyst with -enriched Surface for Oxygen Reduction Reaction in Fuel Cells Ji-Hoon Jang b, Juyeong
Electronic Supplementary Information for One-Pot Synthesis of Core-Shell-like 3 Co Nanoparticle Electrocatalyst with -enriched Surface for Oxygen Reduction Reaction in Fuel Cells Ji-Hoon Jang b, Juyeong
Supplementary information
 Supplementary information Supplementary Figures Supplementary Figure 1. CO 2 light off curve obtained from the 5 wt% Pt/Al 2 O 3 catalyst obtained through heating the catalyst under a 50 ml.min -1 flow
Supplementary information Supplementary Figures Supplementary Figure 1. CO 2 light off curve obtained from the 5 wt% Pt/Al 2 O 3 catalyst obtained through heating the catalyst under a 50 ml.min -1 flow
Plasma Deposition (Overview) Lecture 1
 Plasma Deposition (Overview) Lecture 1 Material Processes Plasma Processing Plasma-assisted Deposition Implantation Surface Modification Development of Plasma-based processing Microelectronics needs (fabrication
Plasma Deposition (Overview) Lecture 1 Material Processes Plasma Processing Plasma-assisted Deposition Implantation Surface Modification Development of Plasma-based processing Microelectronics needs (fabrication
Supporting Information
 Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 217 Supporting Information Catalyst preparation A certain of aqueous NiCl 2 6H 2 O (2 mm), H 2 PtCl
Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 217 Supporting Information Catalyst preparation A certain of aqueous NiCl 2 6H 2 O (2 mm), H 2 PtCl
Supporting Information
 Supporting Information Experimental Materials: For the gold nanocuboids, the chemicals gold (ш) chloride trihydrate, sodium borohydride, cetyltrimethylammonium bromide, copper sulfate, L-ascorbic acid
Supporting Information Experimental Materials: For the gold nanocuboids, the chemicals gold (ш) chloride trihydrate, sodium borohydride, cetyltrimethylammonium bromide, copper sulfate, L-ascorbic acid
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Supporting Information Sustainable photocatalytic production of hydrogen
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Supporting Information Sustainable photocatalytic production of hydrogen
Magnetically-driven selective synthesis of Au clusters on Fe 3 O 4 Nanoparticles
 Electronic Supplementary Material (ESI) for Chemical Communications Magnetically-driven selective synthesis of Au clusters on Fe 3 O 4 Nanoparticles Víctor Sebastian, M. Pilar Calatayud, Gerardo F. Goya
Electronic Supplementary Material (ESI) for Chemical Communications Magnetically-driven selective synthesis of Au clusters on Fe 3 O 4 Nanoparticles Víctor Sebastian, M. Pilar Calatayud, Gerardo F. Goya
Xin Zhang, Weiwei Chen, Jianjun Wang, Yang Shen*, Yuanhua Lin, and Ce-Wen
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supporting information to Hierarchical Interfaces Induce High-Dielectric Permittivity in Nanocomposites
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supporting information to Hierarchical Interfaces Induce High-Dielectric Permittivity in Nanocomposites
Molecular Dynamics on the Angstrom Scale
 Probing Interface Reactions by STM: Molecular Dynamics on the Angstrom Scale Zhisheng Li Prof. Richard Osgood Laboratory for Light-Surface Interactions, Columbia University Outline Motivation: Why do we
Probing Interface Reactions by STM: Molecular Dynamics on the Angstrom Scale Zhisheng Li Prof. Richard Osgood Laboratory for Light-Surface Interactions, Columbia University Outline Motivation: Why do we
Supporting Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supporting Information Stefanos Mourdikoudis, a,b Mariana Chirea, a Daniele Zanaga, c Thomas Altantzis,
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supporting Information Stefanos Mourdikoudis, a,b Mariana Chirea, a Daniele Zanaga, c Thomas Altantzis,
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Probing the Surface Reactivity of
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Probing the Surface Reactivity of
Review of Nature Communications, NCOMMS In situ tracking of heterogeneous nanocatalytic processes. Reviewers' comments:
 Reviewers' comments: Reviewer #1 (Remarks to the Author): This manuscript describes measurement of surface enhanced Raman spectra (SERS) during CO oxidation catalysis on metal catalysts (E.g, Pt, FePt,
Reviewers' comments: Reviewer #1 (Remarks to the Author): This manuscript describes measurement of surface enhanced Raman spectra (SERS) during CO oxidation catalysis on metal catalysts (E.g, Pt, FePt,
Determination of redox reaction rates and orders by in-situ liquid cell. electron microscopy of Pd and Au solution growth
 BNL-107596-2014-JA Determination of redox reaction rates and orders by in-situ liquid cell electron microscopy of Pd and Au solution growth Eli A. Sutter * and Peter W. Sutter Center for Functional Nanomaterials,
BNL-107596-2014-JA Determination of redox reaction rates and orders by in-situ liquid cell electron microscopy of Pd and Au solution growth Eli A. Sutter * and Peter W. Sutter Center for Functional Nanomaterials,
LOW-TEMPERATURE Si (111) HOMOEPITAXY AND DOPING MEDIATED BY A MONOLAYER OF Pb
 LOW-TEMPERATURE Si (111) HOMOEPITAXY AND DOPING MEDIATED BY A MONOLAYER OF Pb O.D. DUBON, P.G. EVANS, J.F. CHERVINSKY, F. SPAEPEN, M.J. AZIZ, and J.A. GOLOVCHENKO Division of Engineering and Applied Sciences,
LOW-TEMPERATURE Si (111) HOMOEPITAXY AND DOPING MEDIATED BY A MONOLAYER OF Pb O.D. DUBON, P.G. EVANS, J.F. CHERVINSKY, F. SPAEPEN, M.J. AZIZ, and J.A. GOLOVCHENKO Division of Engineering and Applied Sciences,
Secondary Ion Mass Spectroscopy (SIMS)
 Secondary Ion Mass Spectroscopy (SIMS) Analyzing Inorganic Solids * = under special conditions ** = semiconductors only + = limited number of elements or groups Analyzing Organic Solids * = under special
Secondary Ion Mass Spectroscopy (SIMS) Analyzing Inorganic Solids * = under special conditions ** = semiconductors only + = limited number of elements or groups Analyzing Organic Solids * = under special
Efficient Hydrogen Evolution. University of Central Florida, 4000 Central Florida Blvd. Orlando, Florida, 32816,
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2017 MoS 2 /TiO 2 Heterostructures as Nonmetal Plasmonic Photocatalysts for Highly
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2017 MoS 2 /TiO 2 Heterostructures as Nonmetal Plasmonic Photocatalysts for Highly
Supplementary Information
 Supplementary Information Supplementary Figure 1. X-ray diffraction patterns of (a) pure LDH, (b) AuCl 4 ion-exchanged LDH and (c) the Au/LDH hybrid catalyst. The refined cell parameters for pure, ion-exchanged,
Supplementary Information Supplementary Figure 1. X-ray diffraction patterns of (a) pure LDH, (b) AuCl 4 ion-exchanged LDH and (c) the Au/LDH hybrid catalyst. The refined cell parameters for pure, ion-exchanged,
Supplementary Figure 1 In-situ and ex-situ XRD. a) Schematic of the synchrotron
 Supplementary Figure 1 In-situ and ex-situ XRD. a) Schematic of the synchrotron based XRD experimental set up for θ-2θ measurements. b) Full in-situ scan of spot deposited film for 800 sec at 325 o C source
Supplementary Figure 1 In-situ and ex-situ XRD. a) Schematic of the synchrotron based XRD experimental set up for θ-2θ measurements. b) Full in-situ scan of spot deposited film for 800 sec at 325 o C source
Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supplementary Information AgPd@Pd/TiO 2 nanocatalyst synthesis by microwave
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supplementary Information AgPd@Pd/TiO 2 nanocatalyst synthesis by microwave
Harnessing the Synergy Between Upconverting Nanoparticles and Lanthanide Complexes in a Multi-Wavelength Responsive Hybrid System.
 Electronic Supporting Information for: Harnessing the Synergy Between Upconverting Nanoparticles and Lanthanide Complexes in a Multi-Wavelength Responsive Hybrid System. Riccardo Marin, a Ilias Halimi,
Electronic Supporting Information for: Harnessing the Synergy Between Upconverting Nanoparticles and Lanthanide Complexes in a Multi-Wavelength Responsive Hybrid System. Riccardo Marin, a Ilias Halimi,
Characterisation of Nanoparticles using Advanced Electron Microscopy
 Wright State University CORE Scholar Special Session 5: Carbon and Oxide Based Nanostructured Materials (2013) Special Session 5 6-2013 Characterisation of Core@Shell Nanoparticles using Advanced Electron
Wright State University CORE Scholar Special Session 5: Carbon and Oxide Based Nanostructured Materials (2013) Special Session 5 6-2013 Characterisation of Core@Shell Nanoparticles using Advanced Electron
Chapter 5: Radiation induced synthesis of anisotropic gold nanoparticles and their characterization
 Chapter 5: Radiation induced synthesis of anisotropic gold nanoparticles and their characterization 5.1. Introduction Fine metal particles with nanometer scale dimensions are of current interest due to
Chapter 5: Radiation induced synthesis of anisotropic gold nanoparticles and their characterization 5.1. Introduction Fine metal particles with nanometer scale dimensions are of current interest due to
Supplementary information
 1 2 Supplementary information 3 4 5 6 Supplementary Figure 1 7 8 Supplementary Figure 1 ǀ Characterization of the lysozyme fibrils by atomic force microscopy 9 (AFM) and scanning electron microscopy (SEM).
1 2 Supplementary information 3 4 5 6 Supplementary Figure 1 7 8 Supplementary Figure 1 ǀ Characterization of the lysozyme fibrils by atomic force microscopy 9 (AFM) and scanning electron microscopy (SEM).
Focused Ion Beam Induced Local Tungsten Deposition
 Focused Ion Beam Induced Local Tungsten Deposition H. Langfischer, B. Basnar, E. Bertagnolli Institute for Solid State Electronics, Vienna University of Technology, Floragasse 7, 1040 ien, Austria H. Hutter
Focused Ion Beam Induced Local Tungsten Deposition H. Langfischer, B. Basnar, E. Bertagnolli Institute for Solid State Electronics, Vienna University of Technology, Floragasse 7, 1040 ien, Austria H. Hutter
Tunable Nanoparticle Arrays at Charged Interfaces
 Tunable Nanoparticle Arrays at Charged Interfaces Supporting Material Sunita Srivastava 1, Dmytro Nykypanchuk 1, Masafumi Fukuto 2 and Oleg Gang 1* 1 Center for Functional Nanomaterials, Brookhaven National
Tunable Nanoparticle Arrays at Charged Interfaces Supporting Material Sunita Srivastava 1, Dmytro Nykypanchuk 1, Masafumi Fukuto 2 and Oleg Gang 1* 1 Center for Functional Nanomaterials, Brookhaven National
Supplementary Figure 1. HRTEM images of PtNi / Ni-B composite exposed to electron beam. The. scale bars are 5 nm.
 Supplementary Figure 1. HRTEM images of PtNi / Ni-B composite exposed to electron beam. The scale bars are 5 nm. S1 Supplementary Figure 2. TEM image of PtNi/Ni-B composite obtained under N 2 protection.
Supplementary Figure 1. HRTEM images of PtNi / Ni-B composite exposed to electron beam. The scale bars are 5 nm. S1 Supplementary Figure 2. TEM image of PtNi/Ni-B composite obtained under N 2 protection.
In-situ environmental (scanning) transmission electron microscopy of catalysts at the atomic level
 Journal of Physics: Conference Series OPEN ACCESS In-situ environmental (scanning) transmission electron microscopy of catalysts at the atomic level To cite this article: P L Gai and E D Boyes 2014 J.
Journal of Physics: Conference Series OPEN ACCESS In-situ environmental (scanning) transmission electron microscopy of catalysts at the atomic level To cite this article: P L Gai and E D Boyes 2014 J.
Supplementary Information. Atomic Layer Deposition of Platinum Catalysts on Nanowire Surfaces for Photoelectrochemical Water Reduction
 Supplementary Information Atomic Layer Deposition of Platinum Catalysts on Nanowire Surfaces for Photoelectrochemical Water Reduction Neil P. Dasgupta 1 ǂ, Chong Liu 1,2 ǂ, Sean Andrews 1,2, Fritz B. Prinz
Supplementary Information Atomic Layer Deposition of Platinum Catalysts on Nanowire Surfaces for Photoelectrochemical Water Reduction Neil P. Dasgupta 1 ǂ, Chong Liu 1,2 ǂ, Sean Andrews 1,2, Fritz B. Prinz
25th Anniversary Article: Galvanic Replacement: A Simple and Versatile Route to Hollow Nanostructures with Tunable and Well-Controlled Properties
 25th Anniversary Article: Galvanic Replacement: A Simple and Versatile Route to Hollow Nanostructures with Tunable and Well-Controlled Properties Xiaohu Xia, Yi Wang, Aleksey Ruditskiy, and Younan Xia
25th Anniversary Article: Galvanic Replacement: A Simple and Versatile Route to Hollow Nanostructures with Tunable and Well-Controlled Properties Xiaohu Xia, Yi Wang, Aleksey Ruditskiy, and Younan Xia
Sub-Stoichiometric Germanium Sulfide Thin-Films as a High-Rate Lithium Storage Material
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Sub-Stoichiometric Germanium Sulfide Thin-Films as a High-Rate Lithium
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Sub-Stoichiometric Germanium Sulfide Thin-Films as a High-Rate Lithium
Small-Angle X-ray Scattering (SAXS)/X-ray Absorption Near Edge Spectroscopy (XANES).
 S1 Small-Angle X-ray Scattering (SAXS)/X-ray Absorption Near Edge Spectroscopy (XANES). The combined SAXS/XANES measurements were carried out at the µspot beamline at BESSY II (Berlin, Germany). The beamline
S1 Small-Angle X-ray Scattering (SAXS)/X-ray Absorption Near Edge Spectroscopy (XANES). The combined SAXS/XANES measurements were carried out at the µspot beamline at BESSY II (Berlin, Germany). The beamline
Q1. The electronic structure of the atoms of five elements are shown in the figure below.
 Q1. The electronic structure of the atoms of five elements are shown in the figure below. The letters are not the symbols of the elements. Choose the element to answer the question. Each element can be
Q1. The electronic structure of the atoms of five elements are shown in the figure below. The letters are not the symbols of the elements. Choose the element to answer the question. Each element can be
Exploring the Formation of Symmetric Gold Nanostars by Liquid-Cell Transmission Electron Microscopy
 Supplementary information Exploring the Formation of Symmetric Gold Nanostars by Liquid-Cell Transmission Electron Microscopy Nabeel Ahmad, 1,2 Guillaume Wang, 1 Jaysen Nelayah, 1 Christian Ricolleau,
Supplementary information Exploring the Formation of Symmetric Gold Nanostars by Liquid-Cell Transmission Electron Microscopy Nabeel Ahmad, 1,2 Guillaume Wang, 1 Jaysen Nelayah, 1 Christian Ricolleau,
Size-selected Metal Cluster Deposition on Oxide Surfaces: Impact Dynamics and Supported Cluster Chemistry
 Size-selected Metal Cluster Deposition on Oxide Surfaces: Impact Dynamics and Supported Cluster Chemistry Sungsik Lee, Masato Aizawa, Chaoyang Fan, Tianpin Wu, and Scott L. Anderson Support: AFOSR, DOE
Size-selected Metal Cluster Deposition on Oxide Surfaces: Impact Dynamics and Supported Cluster Chemistry Sungsik Lee, Masato Aizawa, Chaoyang Fan, Tianpin Wu, and Scott L. Anderson Support: AFOSR, DOE
A Novel Electroless Method for the Deposition of Single-Crystalline Platinum Nanoparticle Films On
 Supplementary Information A Novel Electroless Method for the Deposition of Single-Crystalline Platinum Nanoparticle Films On an Organic Solid Matrix in the Presence of Gold Single Crystals Khaleda Banu,,,*
Supplementary Information A Novel Electroless Method for the Deposition of Single-Crystalline Platinum Nanoparticle Films On an Organic Solid Matrix in the Presence of Gold Single Crystals Khaleda Banu,,,*
Supporting Information. Emergent, Collective Oscillations of Self- Mobile Particles and Patterned Surfaces under Redox Conditions
 Supporting Information Emergent, Collective Oscillations of Self- Mobile Particles and Patterned Surfaces under Redox Conditions Michael E. Ibele, a Paul E. Lammert, b Vincent H. Crespi,* b and Ayusman
Supporting Information Emergent, Collective Oscillations of Self- Mobile Particles and Patterned Surfaces under Redox Conditions Michael E. Ibele, a Paul E. Lammert, b Vincent H. Crespi,* b and Ayusman
Supporting Information: Analysis of protein coatings on gold nanoparticles by XPS and liquid-based particle sizing techniques
 Supporting Information: Analysis of protein coatings on gold nanoparticles by XPS and liquid-based particle sizing techniques Natalie A. Belsey, a) Alex G. Shard a) and Caterina Minelli a),b) National
Supporting Information: Analysis of protein coatings on gold nanoparticles by XPS and liquid-based particle sizing techniques Natalie A. Belsey, a) Alex G. Shard a) and Caterina Minelli a),b) National
Design of a new family of catalytic support based on thiol containing plasma polymer films
 Design of a new family of catalytic support based on thiol containing plasma polymer films Dr. D. Thiry damien.thiry@umons.ac.be Chimie des Interactions Plasma Surface (ChIPS), CIRMAP, University of Mons,
Design of a new family of catalytic support based on thiol containing plasma polymer films Dr. D. Thiry damien.thiry@umons.ac.be Chimie des Interactions Plasma Surface (ChIPS), CIRMAP, University of Mons,
Supplementary Information. Rong Fu, Xiumei Jin, Jinglun Liang, Weishi Zheng, Jiaqi Zhuang and Wensheng Yang*
 Supplementary Information Preparation of Nearly Monodispersed Fe 3 O 4 /SiO 2 composite particles from aggregates of Fe 3 O 4 nanoparticles Rong Fu, Xiumei Jin, Jinglun Liang, Weishi Zheng, Jiaqi Zhuang
Supplementary Information Preparation of Nearly Monodispersed Fe 3 O 4 /SiO 2 composite particles from aggregates of Fe 3 O 4 nanoparticles Rong Fu, Xiumei Jin, Jinglun Liang, Weishi Zheng, Jiaqi Zhuang
Conversion of Methane and Light Alkanes to Chemicals over Heterogeneous Catalysts Lessons Learned from Experiment and Theory
 Conversion of Methane and Light Alkanes to Chemicals over Heterogeneous Catalysts Lessons Learned from Experiment and Theory March 8, 201 6 Alexis T. Bell Department of Chemical and Biomolecular Engineering
Conversion of Methane and Light Alkanes to Chemicals over Heterogeneous Catalysts Lessons Learned from Experiment and Theory March 8, 201 6 Alexis T. Bell Department of Chemical and Biomolecular Engineering
SUPPORTING INFORMATION
 Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2017 SUPPORTING INFORMATION Synthesis of Circular and Triangular Gold Nanorings with
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2017 SUPPORTING INFORMATION Synthesis of Circular and Triangular Gold Nanorings with
In situ studies on dynamic properties of carbon nanotubes with metal clusters
 In situ studies on dynamic properties of carbon nanotubes with metal clusters Jason Chang, Yuan-Chih Chang, Der-Hsien Lien, Shaw-Chieh Wang*, Tung Hsu*, and Tien T. Tsong Institute of Physics, Academia
In situ studies on dynamic properties of carbon nanotubes with metal clusters Jason Chang, Yuan-Chih Chang, Der-Hsien Lien, Shaw-Chieh Wang*, Tung Hsu*, and Tien T. Tsong Institute of Physics, Academia
An account of our efforts towards air quality monitoring in epitaxial graphene on SiC
 European Network on New Sensing Technologies for Air Pollution Control and Environmental Sustainability - EuNetAir COST Action TD1105 2 nd International Workshop EuNetAir on New Sensing Technologies for
European Network on New Sensing Technologies for Air Pollution Control and Environmental Sustainability - EuNetAir COST Action TD1105 2 nd International Workshop EuNetAir on New Sensing Technologies for
Nanoscale voxel spectroscopy by simultaneous EELS and EDS tomography
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supplementary Information Nanoscale voxel spectroscopy by simultaneous EELS and EDS tomography
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supplementary Information Nanoscale voxel spectroscopy by simultaneous EELS and EDS tomography
Single-walled carbon nanotubes as nano-electrode and nanoreactor to control the pathways of a redox reaction
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 014 Supporting information Single-walled carbon nanotubes as nano-electrode and nanoreactor to control
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 014 Supporting information Single-walled carbon nanotubes as nano-electrode and nanoreactor to control
Nova 600 NanoLab Dual beam Focused Ion Beam IITKanpur
 Nova 600 NanoLab Dual beam Focused Ion Beam system @ IITKanpur Dual Beam Nova 600 Nano Lab From FEI company (Dual Beam = SEM + FIB) SEM: The Electron Beam for SEM Field Emission Electron Gun Energy : 500
Nova 600 NanoLab Dual beam Focused Ion Beam system @ IITKanpur Dual Beam Nova 600 Nano Lab From FEI company (Dual Beam = SEM + FIB) SEM: The Electron Beam for SEM Field Emission Electron Gun Energy : 500
2.3 Modeling Interatomic Interactions Pairwise Potentials Many-Body Potentials Studying Biomolecules: The Force
 Contents 1 Introduction to Computational Meso-Bio-Nano (MBN) Science and MBN EXPLORER.... 1 1.1 Meso-Bio-Nano Science: A Novel Field of Interdisciplinary Research.... 1 1.1.1 Structure and Dynamics of
Contents 1 Introduction to Computational Meso-Bio-Nano (MBN) Science and MBN EXPLORER.... 1 1.1 Meso-Bio-Nano Science: A Novel Field of Interdisciplinary Research.... 1 1.1.1 Structure and Dynamics of
Q1. The electronic structure of the atoms of five elements are shown in the figure below.
 Q. The electronic structure of the atoms of five elements are shown in the figure below. The letters are not the symbols of the elements. Choose the element to answer the question. Each element can be
Q. The electronic structure of the atoms of five elements are shown in the figure below. The letters are not the symbols of the elements. Choose the element to answer the question. Each element can be
Supplementary Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
4.2.1 Chemical bonds, ionic, covalent and metallic
 4.2 Bonding, structure, and the properties of matter Chemists use theories of structure and bonding to explain the physical and chemical properties of materials. Analysis of structures shows that atoms
4.2 Bonding, structure, and the properties of matter Chemists use theories of structure and bonding to explain the physical and chemical properties of materials. Analysis of structures shows that atoms
Supporting Information:
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supporting Information:. FigS1:TEM images of Pd conacve nanocube of three different sizes synthesised
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supporting Information:. FigS1:TEM images of Pd conacve nanocube of three different sizes synthesised
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/NCHEM.2491 Experimental Realization of Two-dimensional Boron Sheets Baojie Feng 1, Jin Zhang 1, Qing Zhong 1, Wenbin Li 1, Shuai Li 1, Hui Li 1, Peng Cheng 1, Sheng Meng 1,2, Lan Chen 1 and
DOI: 10.1038/NCHEM.2491 Experimental Realization of Two-dimensional Boron Sheets Baojie Feng 1, Jin Zhang 1, Qing Zhong 1, Wenbin Li 1, Shuai Li 1, Hui Li 1, Peng Cheng 1, Sheng Meng 1,2, Lan Chen 1 and
X-ray photoelectron spectroscopic characterization of molybdenum nitride thin films
 Korean J. Chem. Eng., 28(4), 1133-1138 (2011) DOI: 10.1007/s11814-011-0036-2 INVITED REVIEW PAPER X-ray photoelectron spectroscopic characterization of molybdenum nitride thin films Jeong-Gil Choi Department
Korean J. Chem. Eng., 28(4), 1133-1138 (2011) DOI: 10.1007/s11814-011-0036-2 INVITED REVIEW PAPER X-ray photoelectron spectroscopic characterization of molybdenum nitride thin films Jeong-Gil Choi Department
Electronic Supplementary Information. Facile synthesis of polypyrrole coated copper nanowire: new concept to engineered core-shell structures
 Electronic Supplementary Information Facile synthesis of polypyrrole coated copper nanowire: new concept to engineered core-shell structures Yang Liu, a Zhen Liu, a Ning Lu, b Elisabeth Preiss, a Selcuk
Electronic Supplementary Information Facile synthesis of polypyrrole coated copper nanowire: new concept to engineered core-shell structures Yang Liu, a Zhen Liu, a Ning Lu, b Elisabeth Preiss, a Selcuk
LECTURE 5 SUMMARY OF KEY IDEAS
 LECTURE 5 SUMMARY OF KEY IDEAS Etching is a processing step following lithography: it transfers a circuit image from the photoresist to materials form which devices are made or to hard masking or sacrificial
LECTURE 5 SUMMARY OF KEY IDEAS Etching is a processing step following lithography: it transfers a circuit image from the photoresist to materials form which devices are made or to hard masking or sacrificial
Chapter 4. Surface defects created by kev Xe ion irradiation on Ge
 81 Chapter 4 Surface defects created by kev Xe ion irradiation on Ge 4.1. Introduction As high energy ions penetrate into a solid, those ions can deposit kinetic energy in two processes: electronic excitation
81 Chapter 4 Surface defects created by kev Xe ion irradiation on Ge 4.1. Introduction As high energy ions penetrate into a solid, those ions can deposit kinetic energy in two processes: electronic excitation
Electronic Supplementary Information (ESI)
 Electronic Supplementary Information (ESI) Experimental details 1. Preparation of KIT-6 mesoporous silica: Conc. HCl solution (37 wt%) and Pluronic P123 were dissolved in water. After the dissolution,
Electronic Supplementary Information (ESI) Experimental details 1. Preparation of KIT-6 mesoporous silica: Conc. HCl solution (37 wt%) and Pluronic P123 were dissolved in water. After the dissolution,
A nanoscale perspective on the effect of acid washing of carbon catalyst supports
 A nanoscale perspective on the effect of acid washing of carbon catalyst supports Emir Bouleghlimat, Philip R. Davies, Robert J. Davies, Jiri Kulahvy, David J. Morgan, School of Chemistry, Cardiff University
A nanoscale perspective on the effect of acid washing of carbon catalyst supports Emir Bouleghlimat, Philip R. Davies, Robert J. Davies, Jiri Kulahvy, David J. Morgan, School of Chemistry, Cardiff University
Carbonaceous contaminants on support films for transmission electron microscopy
 PERGAMON Carbon 39 (2001) 909 913 Carbonaceous contaminants on support films for transmission electron microscopy P.J.F. Harris Department of Chemistry, University of Reading, Whiteknights, Reading RG6
PERGAMON Carbon 39 (2001) 909 913 Carbonaceous contaminants on support films for transmission electron microscopy P.J.F. Harris Department of Chemistry, University of Reading, Whiteknights, Reading RG6
Supporting Information for. Revealing Surface Elemental Composition and Dynamic Processes
 Supporting Information for Revealing Surface Elemental Composition and Dynamic Processes Involved in Facet-dependent Oxidation of Pt 3 Co Nanoparticles via in-situ Transmission Electron Microscopy Sheng
Supporting Information for Revealing Surface Elemental Composition and Dynamic Processes Involved in Facet-dependent Oxidation of Pt 3 Co Nanoparticles via in-situ Transmission Electron Microscopy Sheng
Enduring Understandings & Essential Knowledge for AP Chemistry
 Enduring Understandings & Essential Knowledge for AP Chemistry Big Idea 1: The chemical elements are fundamental building materials of matter, and all matter can be understood in terms of arrangements
Enduring Understandings & Essential Knowledge for AP Chemistry Big Idea 1: The chemical elements are fundamental building materials of matter, and all matter can be understood in terms of arrangements
Impact of Contact Evolution on the Shelf Life of Organic Solar Cells
 Impact of Contact Evolution on the Shelf Life of Organic Solar Cells By Matthew T. Lloyd, Dana C. Olson, Ping Lu, Erica Fang, Diana L. Moore, Matthew S. White, Matthew O. Reese, David S. Ginley, and Julia
Impact of Contact Evolution on the Shelf Life of Organic Solar Cells By Matthew T. Lloyd, Dana C. Olson, Ping Lu, Erica Fang, Diana L. Moore, Matthew S. White, Matthew O. Reese, David S. Ginley, and Julia
Supplementary Information
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Supplementary Information Insights into the Synergistic Role of Metal-Lattice
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Supplementary Information Insights into the Synergistic Role of Metal-Lattice
OCR Chemistry Checklist
 Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Topic 1. Particles Video: The Particle Model Describe the main features of the particle model in terms of states of matter. Explain in terms of the particle model the distinction between physical changes
Word of the Day for August 27, Definition - any property of a substance that must be observed during a chemical change.
 Word of the Day for August 27, 2008 Chemical Property Definition - any property of a substance that must be observed during a chemical change. One chemical property of magnesium is that it will combine
Word of the Day for August 27, 2008 Chemical Property Definition - any property of a substance that must be observed during a chemical change. One chemical property of magnesium is that it will combine
Fabrication Methods: Chapter 4. Often two methods are typical. Top Down Bottom up. Begins with atoms or molecules. Begins with bulk materials
 Fabrication Methods: Chapter 4 Often two methods are typical Top Down Bottom up Begins with bulk materials Begins with atoms or molecules Reduced in size to nano By thermal, physical Chemical, electrochemical
Fabrication Methods: Chapter 4 Often two methods are typical Top Down Bottom up Begins with bulk materials Begins with atoms or molecules Reduced in size to nano By thermal, physical Chemical, electrochemical
Supplementary Information. for. Controlled Scalable Synthesis of Uniform, High-Quality Monolayer and Fewlayer
 Supplementary Information for Controlled Scalable Synthesis of Uniform, High-Quality Monolayer and Fewlayer MoS 2 Films Yifei Yu 1, Chun Li 1, Yi Liu 3, Liqin Su 4, Yong Zhang 4, Linyou Cao 1,2 * 1 Department
Supplementary Information for Controlled Scalable Synthesis of Uniform, High-Quality Monolayer and Fewlayer MoS 2 Films Yifei Yu 1, Chun Li 1, Yi Liu 3, Liqin Su 4, Yong Zhang 4, Linyou Cao 1,2 * 1 Department
In-vessel Tritium Inventory in ITER Evaluated by Deuterium Retention of Carbon Dust
 FT/P1-19 In-vessel Tritium Inventory in ITER Evaluated by Deuterium Retention of Carbon Dust T. Hino 1), H. Yoshida 1), M. Akiba 2), S. Suzuki 2), Y. Hirohata 1) and Y. Yamauchi 1) 1) Laboratory of Plasma
FT/P1-19 In-vessel Tritium Inventory in ITER Evaluated by Deuterium Retention of Carbon Dust T. Hino 1), H. Yoshida 1), M. Akiba 2), S. Suzuki 2), Y. Hirohata 1) and Y. Yamauchi 1) 1) Laboratory of Plasma
Structural Analysis of Nanoparticles Using Scanning Transmission Electron Microscopy
 Structural Analysis of Nanoparticles Using Scanning Transmission Electron Microscopy Shinya Maenosono and Derrick Mott School of Materials Science, Japan Advanced Institute of Science and Technology (JAIST)
Structural Analysis of Nanoparticles Using Scanning Transmission Electron Microscopy Shinya Maenosono and Derrick Mott School of Materials Science, Japan Advanced Institute of Science and Technology (JAIST)
Supporting Information
 Supporting Information Remarkable Photothermal Effect of Interband Excitation on Nanosecond Laser-induced Reshaping and Size Reduction of Pseudo-spherical Gold Nanoparticles in Aqueous Solution Daniel
Supporting Information Remarkable Photothermal Effect of Interband Excitation on Nanosecond Laser-induced Reshaping and Size Reduction of Pseudo-spherical Gold Nanoparticles in Aqueous Solution Daniel
Chapter 5: Nanoparticle Production from Cathode Sputtering. in High-Pressure Microhollow Cathode and Arc Discharges
 96 Chapter 5: Nanoparticle Production from Cathode Sputtering in High-Pressure Microhollow Cathode and Arc Discharges 5.1. Introduction Sputtering is a fundamental aspect of plasma operation and has been
96 Chapter 5: Nanoparticle Production from Cathode Sputtering in High-Pressure Microhollow Cathode and Arc Discharges 5.1. Introduction Sputtering is a fundamental aspect of plasma operation and has been
Catalytic oxidations: finding the optimum composition of AuPd coreshell nanoparticle catalysts
 Brazilian ChemComm Symposium Chemistry and Sustainable Energy 5 th November 2012, São Paulo, Brazil Catalytic oxidations: finding the optimum composition of AuPd coreshell nanoparticle catalysts Prof.
Brazilian ChemComm Symposium Chemistry and Sustainable Energy 5 th November 2012, São Paulo, Brazil Catalytic oxidations: finding the optimum composition of AuPd coreshell nanoparticle catalysts Prof.
7. Oxidation of gold by oxygen-ion sputtering
 7. Oxidation of gold by oxygen-ion sputtering Up to now, relatively little attention has been paid to oxygen phases obtained by sputtering of gold surfaces with oxygen ions. Nevertheless, the high oxygen
7. Oxidation of gold by oxygen-ion sputtering Up to now, relatively little attention has been paid to oxygen phases obtained by sputtering of gold surfaces with oxygen ions. Nevertheless, the high oxygen
 CHAPTER 6 CHEMICAL BONDING SHORT QUESTION WITH ANSWERS Q.1 Dipole moments of chlorobenzene is 1.70 D and of chlorobenzene is 2.5 D while that of paradichlorbenzene is zero; why? Benzene has zero dipole
CHAPTER 6 CHEMICAL BONDING SHORT QUESTION WITH ANSWERS Q.1 Dipole moments of chlorobenzene is 1.70 D and of chlorobenzene is 2.5 D while that of paradichlorbenzene is zero; why? Benzene has zero dipole
Worksheet 1.1. Chapter 1: Quantitative chemistry glossary
 Worksheet 1.1 Chapter 1: Quantitative chemistry glossary Amount The number of moles of a substance present in a sample. Aqueous solution A solution with water as the solvent. Atmosphere The unit atmosphere
Worksheet 1.1 Chapter 1: Quantitative chemistry glossary Amount The number of moles of a substance present in a sample. Aqueous solution A solution with water as the solvent. Atmosphere The unit atmosphere
An Introduction to Chemical Kinetics
 An Introduction to Chemical Kinetics Michel Soustelle WWILEY Table of Contents Preface xvii PART 1. BASIC CONCEPTS OF CHEMICAL KINETICS 1 Chapter 1. Chemical Reaction and Kinetic Quantities 3 1.1. The
An Introduction to Chemical Kinetics Michel Soustelle WWILEY Table of Contents Preface xvii PART 1. BASIC CONCEPTS OF CHEMICAL KINETICS 1 Chapter 1. Chemical Reaction and Kinetic Quantities 3 1.1. The
Thermal behaviour of 2 nanostructures in various gas environments: a combined 3D and in-situ TEM approach
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 Supporting Information Thermal behaviour of Pd@SiO 2 nanostructures in various gas environments:
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 Supporting Information Thermal behaviour of Pd@SiO 2 nanostructures in various gas environments:
Supplementary Information for. Silver Nanoparticles Embedded Anti-microbial Paints Based on Vegetable Oil
 Supplementary Information for Silver Nanoparticles Embedded Anti-microbial Paints Based on Vegetable Oil Ashavani Kumar #, Praveen Kumar Vemula #, Pulickel M. Ajayan, George John * Department of Chemistry,
Supplementary Information for Silver Nanoparticles Embedded Anti-microbial Paints Based on Vegetable Oil Ashavani Kumar #, Praveen Kumar Vemula #, Pulickel M. Ajayan, George John * Department of Chemistry,
Supporting Information for. Selectivity and Activity in Catalytic Methanol Oxidation in the Gas Phase
 1 / 5 Supporting Information for The Influence of Size-Induced Oxidation State of Platinum Nanoparticles on Selectivity and Activity in Catalytic Methanol Oxidation in the Gas Phase Hailiang Wang, Yihai
1 / 5 Supporting Information for The Influence of Size-Induced Oxidation State of Platinum Nanoparticles on Selectivity and Activity in Catalytic Methanol Oxidation in the Gas Phase Hailiang Wang, Yihai
Large Scale Direct Synthesis of Graphene on Sapphire and Transfer-free Device Fabrication
 Supplementary Information Large Scale Direct Synthesis of Graphene on Sapphire and Transfer-free Device Fabrication Hyun Jae Song a, Minhyeok Son a, Chibeom Park a, Hyunseob Lim a, Mark P. Levendorf b,
Supplementary Information Large Scale Direct Synthesis of Graphene on Sapphire and Transfer-free Device Fabrication Hyun Jae Song a, Minhyeok Son a, Chibeom Park a, Hyunseob Lim a, Mark P. Levendorf b,
Supplementary Information. In colloidal drop drying processes, multi-ring depositions are formed due to the stick-slip
 Electronic Supplementary Material (ESI for Soft Matter. This journal is The Royal Society of Chemistry 14 Supplementary Information A1. Contact line receding velocity of an evaporating drop In colloidal
Electronic Supplementary Material (ESI for Soft Matter. This journal is The Royal Society of Chemistry 14 Supplementary Information A1. Contact line receding velocity of an evaporating drop In colloidal
Supporting Information
 Supporting Information Defects and Surface Structural Stability of MoTe 2 Under Vacuum Annealing Hui Zhu, Qingxiao Wang, Lanxia Cheng, Rafik Addou, Jiyoung Kim, Moon J. Kim*, Robert M. Wallace* Department
Supporting Information Defects and Surface Structural Stability of MoTe 2 Under Vacuum Annealing Hui Zhu, Qingxiao Wang, Lanxia Cheng, Rafik Addou, Jiyoung Kim, Moon J. Kim*, Robert M. Wallace* Department
Cation exchange induced transformation of InP magic-sized clusters
 Supplementary Information to Accompany: Cation exchange induced transformation of InP magic-sized clusters Jennifer L. Stein, Molly I. Steimle Maxwell W. Terban, Alessio Petrone, Simon J. L. Billinge,
Supplementary Information to Accompany: Cation exchange induced transformation of InP magic-sized clusters Jennifer L. Stein, Molly I. Steimle Maxwell W. Terban, Alessio Petrone, Simon J. L. Billinge,
4.2 Bonding, structure, and the properties of matter
 4.2 Bonding, structure, and the properties of matter Chemists use theories of structure and bonding to explain the physical and chemical properties of materials. Analysis of structures shows that atoms
4.2 Bonding, structure, and the properties of matter Chemists use theories of structure and bonding to explain the physical and chemical properties of materials. Analysis of structures shows that atoms
Encapsulation. Battelle Technology. Introduction
 Encapsulation Introduction The encapsulation methods reported in the literature 1-7 for the production of microcapsules are generally achieved using one of the following techniques: 1. Phase separation
Encapsulation Introduction The encapsulation methods reported in the literature 1-7 for the production of microcapsules are generally achieved using one of the following techniques: 1. Phase separation
Crystalline Surfaces for Laser Metrology
 Crystalline Surfaces for Laser Metrology A.V. Latyshev, Institute of Semiconductor Physics SB RAS, Novosibirsk, Russia Abstract: The number of methodological recommendations has been pronounced to describe
Crystalline Surfaces for Laser Metrology A.V. Latyshev, Institute of Semiconductor Physics SB RAS, Novosibirsk, Russia Abstract: The number of methodological recommendations has been pronounced to describe
Fabrication at the nanoscale for nanophotonics
 Fabrication at the nanoscale for nanophotonics Ilya Sychugov, KTH Materials Physics, Kista silicon nanocrystal by electron beam induced deposition lithography Outline of basic nanofabrication methods Devices
Fabrication at the nanoscale for nanophotonics Ilya Sychugov, KTH Materials Physics, Kista silicon nanocrystal by electron beam induced deposition lithography Outline of basic nanofabrication methods Devices
Conceptual Chemistry
 Conceptual Chemistry Objective 1 Describe, at the molecular level, the difference between a gas, liquid, and solid phase. Solids Definite shape Definite volume Particles are vibrating and packed close
Conceptual Chemistry Objective 1 Describe, at the molecular level, the difference between a gas, liquid, and solid phase. Solids Definite shape Definite volume Particles are vibrating and packed close
Gaetano L Episcopo. Scanning Electron Microscopy Focus Ion Beam and. Pulsed Plasma Deposition
 Gaetano L Episcopo Scanning Electron Microscopy Focus Ion Beam and Pulsed Plasma Deposition Hystorical background Scientific discoveries 1897: J. Thomson discovers the electron. 1924: L. de Broglie propose
Gaetano L Episcopo Scanning Electron Microscopy Focus Ion Beam and Pulsed Plasma Deposition Hystorical background Scientific discoveries 1897: J. Thomson discovers the electron. 1924: L. de Broglie propose
Characterization of individual free-standing nanoobjects by cluster SIMS in transmission
 Characterization of individual free-standing nanoobjects by cluster SIMS in transmission Running title: Characterization of individual free-standing nano-objects by cluster SIMS in transmission Running
Characterization of individual free-standing nanoobjects by cluster SIMS in transmission Running title: Characterization of individual free-standing nano-objects by cluster SIMS in transmission Running
