States of Matter 1 of 21 Boardworks Ltd 2016
|
|
|
- Osborne Lambert
- 5 years ago
- Views:
Transcription
1 States of Matter 1 of 21 Boardworks Ltd 2016
2 States of Matter 2 of 21 Boardworks Ltd 2016
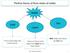
3 What are the three states of matter? 3 of 21 Boardworks Ltd 2016 At any given temperature, all substances exist in one of the three states of matter. solid liquid gas
4 Particles 4 of 21 Boardworks Ltd 2016 Solids, liquids and gases can be easily represented by the simple particle theory: All substances are made up of millions of particles. Particles are represented by small, solid spheres. The spheres can move around and they are described as having kinetic energy. The spheres have no forces between them. Particle theory can help to explain melting, boiling, freezing and condensing. Can you identify the limitations of the particle theory?
5 Properties of solids 5 of 21 Boardworks Ltd 2016 The properties of solids include: the particles are packed very close together and so solids have a high density they cannot be compressed as there is a very small amount of empty space between particles they have a fixed shape as the particles are held very tightly together cannot diffuse as the particles are not able to move.
6 Properties of liquids 6 of 21 Boardworks Ltd 2016 The properties of liquids include: the particles are packed quite close together and so liquids have a fairly high density cannot be compressed because there is very little empty space between particles they take up the shape of their container because the particles are free to move can diffuse because the particles are able to change places.
7 Properties of gases 7 of 21 Boardworks Ltd 2016 The properties of gases include: the particles are spaced far apart and so have a low density can be compressed because there is space between particles they have no fixed shape because the particles move about rapidly in all directions can diffuse because the particles are free to move in all directions due to high collision frequency, they exert pressure on the walls of a container.
8 How do particles move? 8 of 21 Boardworks Ltd 2016
9 Limitations of particle theory 9 of 21 Boardworks Ltd 2016 In reality, particles are not solid spheres at all. They are not completely solid. They are not fully spherical. They have forces acting between them. The nature of real particles depends on many things, including the type of bonding and structure of the substance. Particle theory is a useful way of thinking about solids, liquids and gases but it is not completely accurate! Atoms themselves do not have the same properties as the material they are found in. The interaction of atoms gives a material its bulk properties.
10 States of Matter 10 of 21 Boardworks Ltd 2016
11 Changes of state 11 of 21 Boardworks Ltd 2016 The changes of state happen at different temperatures: melting evaporation happen happen at the at the melting boiling ice point water point steam freezing condensation Changes of state occur at different temperatures for different substances.
12 Particle theory and changes of state 12 of 21 Boardworks Ltd 2016 Particle theory is useful for thinking about changes of state and gives a clear idea about what might happen. It represents atoms as small, inelastic spheres with no forces acting between them i.e. like small bowling balls. It does not take into account the nature of the real particles involved. Different particles will have a different: mass shape size force acting between them. All of these properties will affect changes of state.
13 Changing state 13 of 21 Boardworks Ltd 2016 Particles have forces acting between them. The stronger the forces between the particles, the higher the melting point and boiling point of the substance. Why is this? The amount of energy needed to change the state from solid to liquid (melting) or liquid to gas (boiling) increases as the strength of the forces between the particles increases.
14 Different substances 14 of 21 Boardworks Ltd 2016 What state are the following substances in at 25 C? Substance Water (H 2 O) Chlorine (Cl 2 ) Melting point ( C) Boiling point ( C) Force between the particles Quite strong Weak Iron Very strong Why are the forces between the particles different strengths? Is water (H 2 O) broken down into hydrogen (H 2 ) and oxygen (O) at its melting and boiling points?
15 Melting point and boiling point 15 of 21 Boardworks Ltd 2016 The temperature at which a substance changes from a solid to a liquid is called its melting point. What is the melting point of pure water? The temperature at which a substance changes from a liquid to a gas is called its boiling point. What is the boiling point of pure water? What state is water in at 25 C, 300 C and 100 C?
16 Particles during melting and boiling 16 of 21 Boardworks Ltd 2016 What happens to the particles in a substance when it changes state? When a solid is heated, the particles gain energy and vibrate faster. The hotter the solid gets, the faster the particles vibrate. When the particles have enough energy, they can break the strong forces holding them together and move around freely. This is called the melting point. What do you think happens to the particles when the liquid freezes? What about when a liquid evaporates?
17 State symbols 17 of 21 Boardworks Ltd 2016 State symbols are added to a symbol equation to show which state the reactants and products are in. Solid state symbol is (s). Liquid state symbol is (l). Gas state symbol is (g). Dissolved in water/aqueous state symbol is (aq). This symbol equation now shows that the sulfur is a solid, the oxygen is a gas and the sulfur dioxide is also a gas.
18 States of Matter 18 of 21 Boardworks Ltd 2016
19 Glossary 19 of 21 Boardworks Ltd 2016
20 Solids, liquids and gases: which one? 20 of 21 Boardworks Ltd 2016
21 Multiple-choice quiz 21 of 21 Boardworks Ltd 2016
THE PHASES OF MATTER. Solid: holds its shape and does not flow. The molecules in a solid vibrate in place, but on average, don t move very far.
 THE QUESTIONS What are the phases of matter? What makes these phases different from each other? What is the difference between melting, freezing, boiling and condensation? How do you interpret a Temperature
THE QUESTIONS What are the phases of matter? What makes these phases different from each other? What is the difference between melting, freezing, boiling and condensation? How do you interpret a Temperature
Solids (cont.) Describe the movement of particles in a solid and the forces between them.
 Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Section 16.3 Phase Changes
 Section 16.3 Phase Changes Solid Liquid Gas 3 Phases of Matter Density of Matter How packed matter is (The amount of matter in a given space) Solid: Liquid: Gas: High Density Medium Density Low Density
Section 16.3 Phase Changes Solid Liquid Gas 3 Phases of Matter Density of Matter How packed matter is (The amount of matter in a given space) Solid: Liquid: Gas: High Density Medium Density Low Density
STATES OF MATTER. The Four States of Ma/er. Four States. Solid Liquid Gas Plasma
 STATES OF MATTER The Four States of Ma/er Solid Liquid Gas Plasma Four States STATES OF MATTER Ø What makes a substance a par:cular state of ma
STATES OF MATTER The Four States of Ma/er Solid Liquid Gas Plasma Four States STATES OF MATTER Ø What makes a substance a par:cular state of ma
Chemistry A: States of Matter Packet Name: Hour: Page 1. Chemistry A States of Matter Packet
 Chemistry A: States of Matter Packet Name: Hour: Page 1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page 2 Worksheet #1: States of Matter In this packet we will
Chemistry A: States of Matter Packet Name: Hour: Page 1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page 2 Worksheet #1: States of Matter In this packet we will
Pg , Syllabus
 Pg. 169 171, 173-175 Syllabus 5.7 5.14 www.cgrahamphysics.com What do you remember? End www.cgrahamphysics.com How do particles move? 3 of 30 Boardworks Ltd 2012 4 of 30 Boardworks Ltd 2012 States of matter
Pg. 169 171, 173-175 Syllabus 5.7 5.14 www.cgrahamphysics.com What do you remember? End www.cgrahamphysics.com How do particles move? 3 of 30 Boardworks Ltd 2012 4 of 30 Boardworks Ltd 2012 States of matter
THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES
 THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES The particle model of a gas A gas has no fixed shape or volume, but always spreads out to fill any container. There are
THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES The particle model of a gas A gas has no fixed shape or volume, but always spreads out to fill any container. There are
Chemistry A: States of Matter Packet Name: Hour: Page!1. Chemistry A States of Matter Packet
 Chemistry A: States of Matter Packet Name: Hour: Page!1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page!2 Worksheet #1: States of Matter In this packet we will
Chemistry A: States of Matter Packet Name: Hour: Page!1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page!2 Worksheet #1: States of Matter In this packet we will
Matter. Gas. Solid Liquid. Both shape and volume are not fixed. It has a fixed shape and a fixed volume.
 Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Chapter 10 States of Matter
 Chapter 10 States of Matter 1 Section 10.1 The Nature of Gases Objectives: Describe the assumptions of the kinetic theory as it applies to gases. Interpret gas pressure in terms of kinetic theory. Define
Chapter 10 States of Matter 1 Section 10.1 The Nature of Gases Objectives: Describe the assumptions of the kinetic theory as it applies to gases. Interpret gas pressure in terms of kinetic theory. Define
Conservation of Mass
 Conservation of Mass 1 of 35 Boardworks Ltd 2016 Conservation of Mass 2 of 35 Boardworks Ltd 2016 What is a chemical reaction? 3 of 35 Boardworks Ltd 2016 A chemical reaction is a change that takes place
Conservation of Mass 1 of 35 Boardworks Ltd 2016 Conservation of Mass 2 of 35 Boardworks Ltd 2016 What is a chemical reaction? 3 of 35 Boardworks Ltd 2016 A chemical reaction is a change that takes place
CHM Solids, Liquids, and Phase Changes (r15) Charles Taylor 1/9
 CHM 111 - Solids, Liquids, and Phase Changes (r15) - 2015 Charles Taylor 1/9 Introduction In CHM 110, we used kinetic theory to explain the behavior of gases. Now, we will discuss solids and liquids. While
CHM 111 - Solids, Liquids, and Phase Changes (r15) - 2015 Charles Taylor 1/9 Introduction In CHM 110, we used kinetic theory to explain the behavior of gases. Now, we will discuss solids and liquids. While
Ch(3)Matter & Change. John Dalton
 Ch(3)Matter & Change John Dalton What is Matter? Matter is anything that contains mass & volume (takes up space) Energy, such as light, heat, and sound, is NOT matter. The Particle Theory of Matter 1.
Ch(3)Matter & Change John Dalton What is Matter? Matter is anything that contains mass & volume (takes up space) Energy, such as light, heat, and sound, is NOT matter. The Particle Theory of Matter 1.
Some notes on sigma and pi bonds:
 Some notes on sigma and pi bonds: SIGMA bonds are formed when orbitals overlap along the axis between two atoms. These bonds have good overlap between the bonding orbitals, meaning that they are strong.
Some notes on sigma and pi bonds: SIGMA bonds are formed when orbitals overlap along the axis between two atoms. These bonds have good overlap between the bonding orbitals, meaning that they are strong.
Chapter 7: Kinetic Molecular Theory. 7.1 States of Matter
 Chapter 7: Kinetic Molecular Theory 7.1 States of Matter 7.1 KMT and Changes in State Matter: anything with mass and volume Mass: quantity of matter that a substance or object contains (g or kg) Volume:
Chapter 7: Kinetic Molecular Theory 7.1 States of Matter 7.1 KMT and Changes in State Matter: anything with mass and volume Mass: quantity of matter that a substance or object contains (g or kg) Volume:
OUTLINE. States of Matter, Forces of Attraction Phase Changes Gases The Ideal Gas Law Gas Stoichiometry
 UNIT 6 GASES OUTLINE States of Matter, Forces of Attraction Phase Changes Gases The Ideal Gas Law Gas Stoichiometry STATES OF MATTER Remember that all matter exists in three physical states: Solid Liquid
UNIT 6 GASES OUTLINE States of Matter, Forces of Attraction Phase Changes Gases The Ideal Gas Law Gas Stoichiometry STATES OF MATTER Remember that all matter exists in three physical states: Solid Liquid
4.2.1 Chemical bonds, ionic, covalent and metallic
 4.2 Bonding, structure, and the properties of matter Chemists use theories of structure and bonding to explain the physical and chemical properties of materials. Analysis of structures shows that atoms
4.2 Bonding, structure, and the properties of matter Chemists use theories of structure and bonding to explain the physical and chemical properties of materials. Analysis of structures shows that atoms
Chapter 3. States of Matter
 Chapter 3 States of Matter 1. Solid 2. Liquid 3. Gas States of Matter Two More (discuss later) Plasma Bose-Einstein condensate States of Matter Solid (definite shape and volume) Particles are tightly packed
Chapter 3 States of Matter 1. Solid 2. Liquid 3. Gas States of Matter Two More (discuss later) Plasma Bose-Einstein condensate States of Matter Solid (definite shape and volume) Particles are tightly packed
Draw the Lewis structures of all 7 diatomic elements
 Warm up Draw the Lewis structures of all 7 diatomic elements States of Matter - Part 1 - Gasses Definitions kinetic-molecular theory particles of matter are always in motion ideal gas hypothetical gas
Warm up Draw the Lewis structures of all 7 diatomic elements States of Matter - Part 1 - Gasses Definitions kinetic-molecular theory particles of matter are always in motion ideal gas hypothetical gas
2. If the volume of a container holding a gas is reduced, what will happen to the presure within the container?
 1. Which gas law states that the volume of a fixed mass of a gas is directly proportional to its Kelvin temperature if the pressure is kept constant? A. Boyle s law B. Charles law C. Dalton s law D. Gay-Lussac
1. Which gas law states that the volume of a fixed mass of a gas is directly proportional to its Kelvin temperature if the pressure is kept constant? A. Boyle s law B. Charles law C. Dalton s law D. Gay-Lussac
Chapter 8. Chapter 8. Preview. Bellringer. Chapter 8. Particles of Matter. Objectives. Chapter 8. Particles of Matter, continued
 States of Matter Preview Bellringer Section 2 Behavior of Gases In the kitchen, you might find three different forms of water. What are these three forms of water, and where exactly in the kitchen would
States of Matter Preview Bellringer Section 2 Behavior of Gases In the kitchen, you might find three different forms of water. What are these three forms of water, and where exactly in the kitchen would
Thermal Physics. Temperature (Definition #1): a measure of the average random kinetic energy of all the particles of a system Units: o C, K
 Thermal Physics Internal Energy: total potential energy and random kinetic energy of the molecules of a substance Symbol: U Units: J Internal Kinetic Energy: arises from random translational, vibrational,
Thermal Physics Internal Energy: total potential energy and random kinetic energy of the molecules of a substance Symbol: U Units: J Internal Kinetic Energy: arises from random translational, vibrational,
Ionic Compounds 1 of 31 Boardworks Ltd 2016
 Ionic Compounds 1 of 31 Boardworks Ltd 2016 Ionic Compounds 2 of 31 Boardworks Ltd 2016 3 of 31 Boardworks Ltd 2016 Elements and compounds Elements are made up of just one type of atom. Some elements exist
Ionic Compounds 1 of 31 Boardworks Ltd 2016 Ionic Compounds 2 of 31 Boardworks Ltd 2016 3 of 31 Boardworks Ltd 2016 Elements and compounds Elements are made up of just one type of atom. Some elements exist
Chapter 2: the Structure of the Atom
 Chapter 2: the Structure of the Atom exists in MATTER made of goes through Solid Liquid Gas differs in Melting Freezing Boiling Condensation Sublimation consists of Atoms Arrangement of particles Movement
Chapter 2: the Structure of the Atom exists in MATTER made of goes through Solid Liquid Gas differs in Melting Freezing Boiling Condensation Sublimation consists of Atoms Arrangement of particles Movement
Name Date Class STATES OF MATTER
 13 STATES OF MATTER Chapter Test A A. Matching Match each description in Column B with the correct term in Column A. Write the letter of the correct description on the line. Column A Column B 1. amorphous
13 STATES OF MATTER Chapter Test A A. Matching Match each description in Column B with the correct term in Column A. Write the letter of the correct description on the line. Column A Column B 1. amorphous
Chapter 3. Preview. Section 1 Three States of Matter. Section 2 Behavior of Gases. Section 3 Changes of State. States of Matter.
 States of Matter Preview Section 1 Three States of Matter Section 2 Behavior of Gases Section 3 Changes of State Concept Mapping Section 1 Three States of Matter Bellringer In the kitchen, you might find
States of Matter Preview Section 1 Three States of Matter Section 2 Behavior of Gases Section 3 Changes of State Concept Mapping Section 1 Three States of Matter Bellringer In the kitchen, you might find
Unit 13 Lesson 1 What Are Solids, Liquids, and Gases? Copyright Houghton Mifflin Harcourt Publishing Company
 Unit 13 Lesson 1 What Are Solids, Liquids, and Gases? Copyright Houghton Mifflin Harcourt Publishing Company What s the Matter? Matter has mass and volume. It cannot be created or destroyed. Mass is the
Unit 13 Lesson 1 What Are Solids, Liquids, and Gases? Copyright Houghton Mifflin Harcourt Publishing Company What s the Matter? Matter has mass and volume. It cannot be created or destroyed. Mass is the
States of Matter. Solids Liquids Gases
 States of Matter Solids Liquids Gases 1 Solid vs. Liquid vs. Gas Depends on only two things: What? Attractions Kinetic between particles vs Energy of particles 2 Intermolecular Forces (Molecular Attractions)
States of Matter Solids Liquids Gases 1 Solid vs. Liquid vs. Gas Depends on only two things: What? Attractions Kinetic between particles vs Energy of particles 2 Intermolecular Forces (Molecular Attractions)
Matter: Properties and Changes. Chapter 3.1: Properties of Matter
 Matter: Properties and Changes Chapter 3.1: Properties of Matter Substances Review: Matter is anything that has mass and takes up space. Matter with uniform and unchanging composition is pure substance.
Matter: Properties and Changes Chapter 3.1: Properties of Matter Substances Review: Matter is anything that has mass and takes up space. Matter with uniform and unchanging composition is pure substance.
Kinetic Theory of Matter notes 2012
 Kinetic Theory of Matter notes 2012 Kinetic Theory of Matter 3 parts: 1) All matter is made up of and that act as tiny 2) These tiny particles are always in. State of matter depends on its molecular motion
Kinetic Theory of Matter notes 2012 Kinetic Theory of Matter 3 parts: 1) All matter is made up of and that act as tiny 2) These tiny particles are always in. State of matter depends on its molecular motion
KINETIC PARTICLE THEORY
 KINETIC PARTICLE THEORY IMPORTANT DEFINITIONS: The mixing process in gases or solutions due to the random motion of particles is called Diffusion. The process by which a liquid changes into a vapour at
KINETIC PARTICLE THEORY IMPORTANT DEFINITIONS: The mixing process in gases or solutions due to the random motion of particles is called Diffusion. The process by which a liquid changes into a vapour at
Covalent Compounds 1 of 30 Boardworks Ltd 2016
 Covalent Compounds 1 of 30 Boardworks Ltd 2016 Covalent Compounds 2 of 30 Boardworks Ltd 2016 What are covalent bonds? 3 of 30 Boardworks Ltd 2016 When atoms share pairs of electrons, they form covalent
Covalent Compounds 1 of 30 Boardworks Ltd 2016 Covalent Compounds 2 of 30 Boardworks Ltd 2016 What are covalent bonds? 3 of 30 Boardworks Ltd 2016 When atoms share pairs of electrons, they form covalent
States of Matter. Solids Liquids Gases
 States of Matter Solids Liquids Gases 1 Solid vs. Liquid vs. Gas Depends on only two things: What? Attractions Kinetic between particles vs Energy of particles 2 Intermolecular Forces (Molecular Attractions)
States of Matter Solids Liquids Gases 1 Solid vs. Liquid vs. Gas Depends on only two things: What? Attractions Kinetic between particles vs Energy of particles 2 Intermolecular Forces (Molecular Attractions)
Matter & Energy. Kinetic Theory of Matter. Kinetic Theory of Matter. Kinetic Theory of Matter. Kinetic Theory of Matter. Temperature.
 Matter & Energy 1) All matter is made up of atoms and molecules that act as tiny particles. 1 2 2) These tiny particles are always in motion. State of matter depends on its molecular motion as measured
Matter & Energy 1) All matter is made up of atoms and molecules that act as tiny particles. 1 2 2) These tiny particles are always in motion. State of matter depends on its molecular motion as measured
Draw the Lewis structures of all 7 diatomic elements
 Warm up Draw the Lewis structures of all 7 diatomic elements Jan 27-7:44 AM States of Matter - Part 1 - Gases Definitions kinetic-molecular theory particles of matter are always in motion ideal gas hypothetical
Warm up Draw the Lewis structures of all 7 diatomic elements Jan 27-7:44 AM States of Matter - Part 1 - Gases Definitions kinetic-molecular theory particles of matter are always in motion ideal gas hypothetical
Chemistry Physical, Chemical, and Nuclear Changes
 Chemistry 1010 Physical, Chemical, and Nuclear Changes Review Which state of matter matches the following pictures? gas solid liquid What could the circles in these pictures represent? usually molecules,
Chemistry 1010 Physical, Chemical, and Nuclear Changes Review Which state of matter matches the following pictures? gas solid liquid What could the circles in these pictures represent? usually molecules,
Physical Science Exam 3 Study Guide. Dr. Karoline Rostamiani. Chapter 3
 Chapter 3 Section 1 States of Matter What is matter made of? What are the three most common states of matter? How do particles behave in each state of matter? Solids, Liquids, and Gases Materials can be
Chapter 3 Section 1 States of Matter What is matter made of? What are the three most common states of matter? How do particles behave in each state of matter? Solids, Liquids, and Gases Materials can be
Physical Properties of Matter. Examples of Physical Properties. QUESTION: How could you find the volume of air in an "empty" room?
 QUESTION: How could you find the volume of air in an "empty" room? The volume of regularly shaped solids can be calculated from their dimensions. For example, the volume of a rectangular solid is the product
QUESTION: How could you find the volume of air in an "empty" room? The volume of regularly shaped solids can be calculated from their dimensions. For example, the volume of a rectangular solid is the product
4.2 Bonding, structure, and the properties of matter
 4.2 Bonding, structure, and the properties of matter Chemists use theories of structure and bonding to explain the physical and chemical properties of materials. Analysis of structures shows that atoms
4.2 Bonding, structure, and the properties of matter Chemists use theories of structure and bonding to explain the physical and chemical properties of materials. Analysis of structures shows that atoms
Chapter 7.1. States of Matter
 Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
Unit 3: States of Matter, Heat and Gas Laws
 Unit 3 - Stevens 1 Unit 3: States of Matter, Heat and Gas Laws Vocabulary: Solid Term Definition Example Liquid Gas No definite shape, but definite volume; Particles close together, but can move around
Unit 3 - Stevens 1 Unit 3: States of Matter, Heat and Gas Laws Vocabulary: Solid Term Definition Example Liquid Gas No definite shape, but definite volume; Particles close together, but can move around
Copyright 2015 Edmentum - All rights reserved. During which of the following phase changes is there a gain in energy? I.
 Study Island Copyright 2015 Edmentum - All rights reserved. Generation Date: 03/16/2015 Generated By: Kristina Brown 1. Examine the phase-change diagram below. During which of the following phase changes
Study Island Copyright 2015 Edmentum - All rights reserved. Generation Date: 03/16/2015 Generated By: Kristina Brown 1. Examine the phase-change diagram below. During which of the following phase changes
Ch. 7 Foundations of Chemistry
 Ch. 7 Foundations of Chemistry Classifying Matter Matter is anything that has mass and takes up space. Atom - smallest particle of matter 2 Main Classifications of Matter Substance Mixtures Substances
Ch. 7 Foundations of Chemistry Classifying Matter Matter is anything that has mass and takes up space. Atom - smallest particle of matter 2 Main Classifications of Matter Substance Mixtures Substances
Term Info Picture. Anything that has mass and takes up space; everything is made of matter.
 Characteristics, Changes, and States of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. B. Develop and use models to describe the movement of particles
Characteristics, Changes, and States of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. B. Develop and use models to describe the movement of particles
C L A S S I F I C AT I O N O F M AT T E R
 UNIT 2: C L A S S I F I C AT I O N O F M AT T E R MATTER Reflect: What is matter? MATTER Matter: Anything that has mass and occupies space. You can describe matter in terms of quantity by mass NOT weight.
UNIT 2: C L A S S I F I C AT I O N O F M AT T E R MATTER Reflect: What is matter? MATTER Matter: Anything that has mass and occupies space. You can describe matter in terms of quantity by mass NOT weight.
Section 2: Changes of State (p. 68) 20 HOLT SCIENCE AND TECHNOLOGY
 Plasmas (p. 67) 24. More than 99 percent of the known matter in the universe is in the plasma state. 25. Plasmas are made up of particles that have broken apart. 26. Plasmas have a definite shape and volume.
Plasmas (p. 67) 24. More than 99 percent of the known matter in the universe is in the plasma state. 25. Plasmas are made up of particles that have broken apart. 26. Plasmas have a definite shape and volume.
STATES OF MATTER STATES OF MATTER. The Four States of Matter 3/5/2015. Solid. Liquid Commonly found on Gas Earth Plasma
 Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) STATES OF MATTER The Four States of Matter Solid } Liquid Commonly found on Gas Earth Plasma STATES OF MATTER Based upon particle
Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) STATES OF MATTER The Four States of Matter Solid } Liquid Commonly found on Gas Earth Plasma STATES OF MATTER Based upon particle
STATES OF MATTER STATES OF MATTER. The Four States of Matter 3/5/2015
 The Four States of Matter Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) Solid } Liquid Commonly found on Gas Earth Plasma Based upon particle arrangement Based upon energy of
The Four States of Matter Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) Solid } Liquid Commonly found on Gas Earth Plasma Based upon particle arrangement Based upon energy of
SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES
 30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
Chapter Practice Test Grosser
 Class: Date: Chapter 10-11 Practice Test Grosser Multiple Choice Identify the choice that best completes the statement or answers the question. 1. According to the kinetic-molecular theory, particles of
Class: Date: Chapter 10-11 Practice Test Grosser Multiple Choice Identify the choice that best completes the statement or answers the question. 1. According to the kinetic-molecular theory, particles of
CHAPTER 10. States of Matter
 CHAPTER 10 States of Matter Kinetic Molecular Theory Kinetikos - Moving Based on the idea that particles of matter are always in motion The motion has consequences Explains the behavior of Gases, Liquids,
CHAPTER 10 States of Matter Kinetic Molecular Theory Kinetikos - Moving Based on the idea that particles of matter are always in motion The motion has consequences Explains the behavior of Gases, Liquids,
CHAPTER 10. Kinetic Molecular Theory. Five Assumptions of the KMT. Atmospheric Pressure
 Kinetic Molecular Theory CHAPTER 10 States of Matter Kinetikos - Moving Based on the idea that particles of matter are always in motion The motion has consequences Explains the behavior of Gases, Liquids,
Kinetic Molecular Theory CHAPTER 10 States of Matter Kinetikos - Moving Based on the idea that particles of matter are always in motion The motion has consequences Explains the behavior of Gases, Liquids,
What is Matter? How can matter be classified? Every sample of matter is either an element, a compound, or a mixture.
 Matter Section 1 What is Matter? How can matter be classified? Every sample of matter is either an element, a compound, or a mixture. matter: anything that has mass and takes up space Matter Section 1
Matter Section 1 What is Matter? How can matter be classified? Every sample of matter is either an element, a compound, or a mixture. matter: anything that has mass and takes up space Matter Section 1
Changing Reaction Rates
 Changing Reaction Rates 1 of 30 Boardworks Ltd 2016 Changing Reaction Rates 2 of 30 Boardworks Ltd 2016 Rates of reaction 3 of 30 Boardworks Ltd 2016 Why are some reactions faster than others? Reactions,
Changing Reaction Rates 1 of 30 Boardworks Ltd 2016 Changing Reaction Rates 2 of 30 Boardworks Ltd 2016 Rates of reaction 3 of 30 Boardworks Ltd 2016 Why are some reactions faster than others? Reactions,
Chemistry Day 5. Friday, August 31 st Tuesday, September 4 th, 2018
 Chemistry Day 5 Friday, August 31 st Tuesday, September 4 th, 2018 Do-Now Title: BrainPOP: States of Matter 1. Write down today s FLT 2. List two examples of gases 3. List two examples of things that are
Chemistry Day 5 Friday, August 31 st Tuesday, September 4 th, 2018 Do-Now Title: BrainPOP: States of Matter 1. Write down today s FLT 2. List two examples of gases 3. List two examples of things that are
CHEMISTRY Matter and Change. Chapter 12: States of Matter
 CHEMISTRY Matter and Change Chapter 12: States of Matter CHAPTER 12 States of Matter Section 12.1 Section 12.2 Section 12.3 Section 12.4 Gases Forces of Attraction Liquids and Solids Phase Changes Click
CHEMISTRY Matter and Change Chapter 12: States of Matter CHAPTER 12 States of Matter Section 12.1 Section 12.2 Section 12.3 Section 12.4 Gases Forces of Attraction Liquids and Solids Phase Changes Click
Name: 1. Which of the following is probably true about 300 ml of sand and 300 ml of water?
 Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Name Date Class MATTER AND CHANGE. SECTION 2.1 PROPERTIES OF MATTER (pages 39 42)
 2 MATTER AND CHANGE SECTION 2.1 PROPERTIES OF MATTER (pages 39 42) This section helps you distinguish extensive from intensive properties and identify substances by their properties. It teaches you how
2 MATTER AND CHANGE SECTION 2.1 PROPERTIES OF MATTER (pages 39 42) This section helps you distinguish extensive from intensive properties and identify substances by their properties. It teaches you how
Water SECTION The properties of water in all phases are determined by its structure.
 SECTION 10.5 Water Water commonly exists in all three physical states on Earth, where it is by far the most abundant liquid. It covers nearly three-quarters of Earth s surface. Water is an essential component
SECTION 10.5 Water Water commonly exists in all three physical states on Earth, where it is by far the most abundant liquid. It covers nearly three-quarters of Earth s surface. Water is an essential component
4 Discuss and evaluate the 5th state of matter. 3 - Differentiate among the four states of matter in terms of energy,
 Goal: Differentiate among the four states of matter in terms of energy, particle motion, and phase transitions. 4 States of Mater Sections 3.1, 3.2 4 Discuss and evaluate the 5 th state of matter. 3 -
Goal: Differentiate among the four states of matter in terms of energy, particle motion, and phase transitions. 4 States of Mater Sections 3.1, 3.2 4 Discuss and evaluate the 5 th state of matter. 3 -
CHAPTER 1: Chemistry, An Introduction
 CHAPTER 1: Chemistry, An Introduction science: the study of nature to explain what one observes 1.4 THE SCIENTIFIC METHOD: How Chemists Think Applying the Scientific Method 1. Make an observation, and
CHAPTER 1: Chemistry, An Introduction science: the study of nature to explain what one observes 1.4 THE SCIENTIFIC METHOD: How Chemists Think Applying the Scientific Method 1. Make an observation, and
Science 8 Chapter 7 Section 1
 Science 8 Chapter 7 Section 1 Describing Fluids (pp. 268-277) What is a fluid? Fluid: any thing that flows; a liquid or a gas While it would seem that some solids flow (sugar, salt, etc), they are not
Science 8 Chapter 7 Section 1 Describing Fluids (pp. 268-277) What is a fluid? Fluid: any thing that flows; a liquid or a gas While it would seem that some solids flow (sugar, salt, etc), they are not
Lesson 9: States of Matter
 Lesson 9: States of Matter Do Now 6O, 6S 11.8.18 Take out HW 6.14 to be checked. Copy info into CJ keep CJ out and open on desk throughout class. On Do Now Page #5, copy and answer: 1. If you use a magnet
Lesson 9: States of Matter Do Now 6O, 6S 11.8.18 Take out HW 6.14 to be checked. Copy info into CJ keep CJ out and open on desk throughout class. On Do Now Page #5, copy and answer: 1. If you use a magnet
INTRODUCTION TO LESSON CLUSTER 7
 INTRODUCTION TO LESSON CLUSTER 7 EXPLAINING MELTING AND SOLIDIFYING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain melting and solidifying, by reference to the molecular
INTRODUCTION TO LESSON CLUSTER 7 EXPLAINING MELTING AND SOLIDIFYING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain melting and solidifying, by reference to the molecular
The Can Demonstration
 The Can Demonstration With your table, make a prediction as to what will happen to the can. Discuss why you think that the can imploded. What are some reasons? https://www.youtube.com/watch?v=xg5niowf_zw&t=28s
The Can Demonstration With your table, make a prediction as to what will happen to the can. Discuss why you think that the can imploded. What are some reasons? https://www.youtube.com/watch?v=xg5niowf_zw&t=28s
Matter and Change. Introductory Concepts for Physical Science or Chemistry
 Matter and Change Introductory Concepts for Physical Science or Chemistry Properties of Matter Matter Anything that has mass and occupies space VOLUME WEIGHT RELIABILITY How does one describe the quantities
Matter and Change Introductory Concepts for Physical Science or Chemistry Properties of Matter Matter Anything that has mass and occupies space VOLUME WEIGHT RELIABILITY How does one describe the quantities
ENERGY IN CHEMISTRY. R. Ashby Duplication by permission only.
 CH 11 TOPIC 28 CHANGING STATES OF MATTER 1 You have mastered this topic when you can: 1) define or describe: ENERGY, POTENTIAL ENERGY, KINETIC ENERGY & KINETIC MOLECULAR THEORY 2) define or describe HEAT
CH 11 TOPIC 28 CHANGING STATES OF MATTER 1 You have mastered this topic when you can: 1) define or describe: ENERGY, POTENTIAL ENERGY, KINETIC ENERGY & KINETIC MOLECULAR THEORY 2) define or describe HEAT
Intensive Properties are Independent.
 Chapter 3: Matter- Properties and Changes Section 3.1: Properties of Matter SUBSTANCE: matter that has an uniform and unchanging composition EXAMPLES: gold, pure water, iron, aluminum PHYSICAL PROPERTIES:
Chapter 3: Matter- Properties and Changes Section 3.1: Properties of Matter SUBSTANCE: matter that has an uniform and unchanging composition EXAMPLES: gold, pure water, iron, aluminum PHYSICAL PROPERTIES:
SOLIDS, LIQUIDS AND GASES
 CHEMIS TRY CONTENTS 17 SOLIDS, LIQUIDS AND GASES 17 Solids, Liquids and Gases 147 18 Solutions and Crystallisation 155 19 Separating Mixtures 162 20 Elements, Compounds and Mixtures 171 All materials exist
CHEMIS TRY CONTENTS 17 SOLIDS, LIQUIDS AND GASES 17 Solids, Liquids and Gases 147 18 Solutions and Crystallisation 155 19 Separating Mixtures 162 20 Elements, Compounds and Mixtures 171 All materials exist
Unit 3 Lesson 4 Ionic, Covalent, and Metallic Bonding. Copyright Houghton Mifflin Harcourt Publishing Company
 Opposites Attract What is an ion? An atom has a neutral charge because it has an equal number of electrons and protons. An ion is a particle with a positive or negative charge. An ion forms when an atom
Opposites Attract What is an ion? An atom has a neutral charge because it has an equal number of electrons and protons. An ion is a particle with a positive or negative charge. An ion forms when an atom
SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES
 30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
Name Date Class THE NATURE OF GASES
 13.1 THE NATURE OF GASES Section Review Objectives Describe the assumptions of the kinetic theory as it applies to gases Interpret gas pressure in terms of kinetic theory Define the relationship between
13.1 THE NATURE OF GASES Section Review Objectives Describe the assumptions of the kinetic theory as it applies to gases Interpret gas pressure in terms of kinetic theory Define the relationship between
NAME: ACTIVITY SHEETS PHYSICS AND CHEMISTRY (SECONDARY 3 rd YEAR)
 NAME: ACTIVITY SHEETS PHYSICS AND CHEMISTRY (SECONDARY 3 rd YEAR) ACTIVITY 1: Matter Lesson 2 THE PARTICULATE NATURE OF MATTER 1-What is matter? 2-What is a particle (corpuscle)? Set some examples 3-What
NAME: ACTIVITY SHEETS PHYSICS AND CHEMISTRY (SECONDARY 3 rd YEAR) ACTIVITY 1: Matter Lesson 2 THE PARTICULATE NATURE OF MATTER 1-What is matter? 2-What is a particle (corpuscle)? Set some examples 3-What
THE CORPUSCULAR NATURE OF MATTER AND ITS PHYSICAL STATES
 THE CORPUSCULAR NATURE OF MATTER AND ITS PHYSICAL STATES In this unit we are going to study the matter from a microscopic point of view using the kinetic theory. We will understand the properties of the
THE CORPUSCULAR NATURE OF MATTER AND ITS PHYSICAL STATES In this unit we are going to study the matter from a microscopic point of view using the kinetic theory. We will understand the properties of the
Types of bonding: OVERVIEW
 1 of 43 Boardworks Ltd 2009 Types of bonding: OVERVIEW 2 of 43 Boardworks Ltd 2009 There are three types of bond that can occur between atoms: an ionic bond occurs between a metal and non-metal atom (e.g.
1 of 43 Boardworks Ltd 2009 Types of bonding: OVERVIEW 2 of 43 Boardworks Ltd 2009 There are three types of bond that can occur between atoms: an ionic bond occurs between a metal and non-metal atom (e.g.
Properties and Structure of Matter
 Properties and Structure of Matter Chapter 10 You can use a spider map to organize the main ideas and supporting details of a topic such as properties of matter. Look at the example shown below. The central
Properties and Structure of Matter Chapter 10 You can use a spider map to organize the main ideas and supporting details of a topic such as properties of matter. Look at the example shown below. The central
Ch 2.1 (Properties of Matter)
 Ch 2.1 (Properties of Matter) matter Anything that has mass and takes up space is matter http://www.chem4kids.com/files/matter_intro.html Matter is described by two types of properties; 1. Physical Properties
Ch 2.1 (Properties of Matter) matter Anything that has mass and takes up space is matter http://www.chem4kids.com/files/matter_intro.html Matter is described by two types of properties; 1. Physical Properties
Chapter 14 9/21/15. Solids, Liquids & Gasses. Essential Questions! Kinetic Theory! Gas State! Gas State!
 Chapter 14 Solids, Liquids & Gasses Essential Questions What is the kinetic theory of matter? How do particles move in the different states of matter? How do particles behave at the boiling and melting
Chapter 14 Solids, Liquids & Gasses Essential Questions What is the kinetic theory of matter? How do particles move in the different states of matter? How do particles behave at the boiling and melting
P5 Heat and Particles Revision Kinetic Model of Matter: States of matter
 P5 Heat and Particles Revision Kinetic Model of Matter: States of matter State Size Shape Solid occupies a fixed volume has a fixed shape Liquid occupies a fixed volume takes the shape of its container
P5 Heat and Particles Revision Kinetic Model of Matter: States of matter State Size Shape Solid occupies a fixed volume has a fixed shape Liquid occupies a fixed volume takes the shape of its container
Chemistry States of Matter Lesson 9 Lesson Plan David V. Fansler
 Chemistry States of Matter Lesson 9 Lesson Plan David V. Fansler States of Matter The Nature of Gases Objectives: Describe the motion of gas particles according to the kinetic theory; Interpret gas pressure
Chemistry States of Matter Lesson 9 Lesson Plan David V. Fansler States of Matter The Nature of Gases Objectives: Describe the motion of gas particles according to the kinetic theory; Interpret gas pressure
CHAPTER 3: MATTER. Active Learning Questions: 1-6, 9, 13-14; End-of-Chapter Questions: 1-18, 20, 24-32, 38-42, 44, 49-52, 55-56, 61-64
 CHAPTER 3: MATTER Active Learning Questions: 1-6, 9, 13-14; End-of-Chapter Questions: 1-18, 20, 24-32, 38-42, 44, 49-52, 55-56, 61-64 3.1 MATTER Matter: Anything that has mass and occupies volume We study
CHAPTER 3: MATTER Active Learning Questions: 1-6, 9, 13-14; End-of-Chapter Questions: 1-18, 20, 24-32, 38-42, 44, 49-52, 55-56, 61-64 3.1 MATTER Matter: Anything that has mass and occupies volume We study
Test Review Unit 3_2 Chemical reactions. Fundamentals Identify the letter of the choice that best completes the statement or answers the question.
 Test Review Unit 3_2 Chemical reactions. Fundamentals Identify the letter of the choice that best completes the statement or answers the question. 1. Which of the following are chemical changes: a) Sugar
Test Review Unit 3_2 Chemical reactions. Fundamentals Identify the letter of the choice that best completes the statement or answers the question. 1. Which of the following are chemical changes: a) Sugar
Work hard. Be nice. 100% EVERYDAY.
 Name: Period: Date: UNIT 10: Energy Lesson 1: Entropy vs. Enthalpy By the end of today, you will have an answer to: What conditions are favored during reactions? Definition Enthalpy is another word for
Name: Period: Date: UNIT 10: Energy Lesson 1: Entropy vs. Enthalpy By the end of today, you will have an answer to: What conditions are favored during reactions? Definition Enthalpy is another word for
Matter, Atoms & Molecules
 Matter, Atoms & Molecules Matter is anything that has mass and takes up space. All matter is made of tiny particles called atoms, which are too small to see with the naked eye. Matter Matter is anything
Matter, Atoms & Molecules Matter is anything that has mass and takes up space. All matter is made of tiny particles called atoms, which are too small to see with the naked eye. Matter Matter is anything
Section A-Gases 1A. Circle the correct answer (18 pts total) (2 points each)
 Section A-Gases 1A. Circle the correct answer (18 pts total) (2 points each) (b) If a sample of gas is warmed in a rigid container (i.e., with fixed volume), which is true? i) the density of the gas remains
Section A-Gases 1A. Circle the correct answer (18 pts total) (2 points each) (b) If a sample of gas is warmed in a rigid container (i.e., with fixed volume), which is true? i) the density of the gas remains
2 Changes of State KEY IDEAS READING TOOLBOX ADDING AND REMOVING ENERGY. States of Matter. As you read this section, keep these questions in mind:
 CHAPTER 3 States of Matter 2 Changes of State SECTION KEY IDEAS As you read this section, keep these questions in mind: What happens when a substance changes from one state of matter to another? What happens
CHAPTER 3 States of Matter 2 Changes of State SECTION KEY IDEAS As you read this section, keep these questions in mind: What happens when a substance changes from one state of matter to another? What happens
Chapter 11. Freedom of Motion. Comparisons of the States of Matter. Liquids, Solids, and Intermolecular Forces
 Liquids, Solids, and Intermolecular Forces Chapter 11 Comparisons of the States of Matter The solid and liquid states have a much higher density than the gas state The solid and liquid states have similar
Liquids, Solids, and Intermolecular Forces Chapter 11 Comparisons of the States of Matter The solid and liquid states have a much higher density than the gas state The solid and liquid states have similar
States of Matter. Essential Question: How does the movement of atoms and molecules relate to matter s different phases?
 States of Matter Essential Question: How does the movement of atoms and molecules relate to matter s different phases? These notes come from pages 60 to 73 in your Physical Science Textbook All Matter
States of Matter Essential Question: How does the movement of atoms and molecules relate to matter s different phases? These notes come from pages 60 to 73 in your Physical Science Textbook All Matter
Classification of Matter. Chapter 10 Classification of Matter
 Chapter 10 Classification of Matter Grade 7 Classification of Matter Matter is anything that has mass and occupies space. We can classify matter based on whether it s solid, liquid, or gas. 2 1 Understanding
Chapter 10 Classification of Matter Grade 7 Classification of Matter Matter is anything that has mass and occupies space. We can classify matter based on whether it s solid, liquid, or gas. 2 1 Understanding
Regents Review Sheet 4 Naming Chemical Compounds, Shapes of Compounds and Balancing Chemical Equations
 Regents Review Sheet 4 Naming Chemical Compounds, Shapes of Compounds and Balancing Chemical Equations These are some important points to remember about Naming Compounds. Use this sheet when you do the
Regents Review Sheet 4 Naming Chemical Compounds, Shapes of Compounds and Balancing Chemical Equations These are some important points to remember about Naming Compounds. Use this sheet when you do the
Module 5: Rise and Fall of the Clockwork Universe. You should be able to demonstrate and show your understanding of:
 OCR B Physics H557 Module 5: Rise and Fall of the Clockwork Universe You should be able to demonstrate and show your understanding of: 5.2: Matter Particle model: A gas consists of many very small, rapidly
OCR B Physics H557 Module 5: Rise and Fall of the Clockwork Universe You should be able to demonstrate and show your understanding of: 5.2: Matter Particle model: A gas consists of many very small, rapidly
Formulae and Equations
 Formulae and Equations 1 of 41 Boardworks Ltd 2016 Formulae and Equations 2 of 41 Boardworks Ltd 2016 Forming different compounds 3 of 41 Boardworks Ltd 2016 Elements are made up of just one type of atom.
Formulae and Equations 1 of 41 Boardworks Ltd 2016 Formulae and Equations 2 of 41 Boardworks Ltd 2016 Forming different compounds 3 of 41 Boardworks Ltd 2016 Elements are made up of just one type of atom.
CIE Physics IGCSE. Topic 2: Thermal Physics
 CIE Physics IGCSE Topic 2: Thermal Physics Summary Notes Simple kinetic molecular model of matter Molecular model Solids Molecules close together in regular pattern Strong intermolecular forces of attraction
CIE Physics IGCSE Topic 2: Thermal Physics Summary Notes Simple kinetic molecular model of matter Molecular model Solids Molecules close together in regular pattern Strong intermolecular forces of attraction
Electrolysis 1 of 21 Boardworks Ltd 2016
 Electrolysis 1 of 21 Boardworks Ltd 2016 Electrolysis 2 of 21 Boardworks Ltd 2016 What are ionic compounds? 3 of 21 Boardworks Ltd 2016 Ionic compounds are made up of positive ions and negative ions. What
Electrolysis 1 of 21 Boardworks Ltd 2016 Electrolysis 2 of 21 Boardworks Ltd 2016 What are ionic compounds? 3 of 21 Boardworks Ltd 2016 Ionic compounds are made up of positive ions and negative ions. What
2. THE STATES OF MATTER
 2. THE STATES OF MATTER 2.1. THE THREE STATES OF MATTER Every substance can take on several distinct forms called phases or states of aggregation of matter. Four states of matter are observable in everyday
2. THE STATES OF MATTER 2.1. THE THREE STATES OF MATTER Every substance can take on several distinct forms called phases or states of aggregation of matter. Four states of matter are observable in everyday
Chapter 3 Phases of Matter Physical Science
 Chapter 3 Phases of Matter Physical Science CH 3- States of Matter 1 What makes up matter? What is the difference between a solid, a liquid, and a gas? What kind of energy do all particles of matter have?
Chapter 3 Phases of Matter Physical Science CH 3- States of Matter 1 What makes up matter? What is the difference between a solid, a liquid, and a gas? What kind of energy do all particles of matter have?
Chapter 14: Liquids and Solids
 I. Phases of matter and phase changes a. Recall the three main phases of matter: Chapter 14: Liquids and Solids Energy is involved during the transition from one phase of matter to another. You should
I. Phases of matter and phase changes a. Recall the three main phases of matter: Chapter 14: Liquids and Solids Energy is involved during the transition from one phase of matter to another. You should
Atoms and molecules are in motion and have energy
 Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Gases: Properties and Behaviour
 SECTION 11.1 Gases: Properties and Behaviour Key Terms kinetic molecular theory of gases ideal gas On Earth, matter typically exists in three physical states: solid, liquid, and gas. All three states of
SECTION 11.1 Gases: Properties and Behaviour Key Terms kinetic molecular theory of gases ideal gas On Earth, matter typically exists in three physical states: solid, liquid, and gas. All three states of
Calorimetry. A calorimeter is a device in which this energy transfer takes place
 Calorimetry One technique for measuring specific heat involves heating a material, adding it to a sample of water, and recording the final temperature This technique is known as calorimetry A calorimeter
Calorimetry One technique for measuring specific heat involves heating a material, adding it to a sample of water, and recording the final temperature This technique is known as calorimetry A calorimeter
