Transmission of Batrachochytrium dendrobatidis within and between amphibian life stages
|
|
|
- Douglas Goodman
- 6 years ago
- Views:
Transcription
1 DISEASES OF AQUATIC ORGANISMS Vol. 61: 75 83, 2004 Published October 21 Dis Aquat Org Transmission of Batrachochytrium dendrobatidis within and between amphibian life stages Lara J. Rachowicz*, Vance T. Vredenburg Department of Integrative Biology, University of California, Berkeley, California , USA ABSTRACT: Chytridiomycosis is an emerging infectious disease caused by the chytrid fungus Batrachochytrium dendrobatidis, which has been implicated in amphibian declines worldwide. The mountain yellow-legged frog Rana muscosa is a declining amphibian species that can be infected by B. dendrobatidis; however, transmission between conspecifics has not been documented. Here, we present experimental evidence that R. muscosa tadpoles can be infected by fungal zoospores and that they can transmit infection to each other and to postmetamorphic animals. We compared several techniques for detecting B. dendrobatidis transmission and found that histology with serial sectioning was able to detect infection before cytology or visual inspections. We also show that R. muscosa tadpoles appear healthy with B. dendrobatidis infection, while postmetamorphic animals experience mortality. In addition, we provide guidelines for visually detecting B. dendrobatidis in R. muscosa tadpoles, which may be useful in other affected species. Field surveys of infected and uninfected populations verify this identification technique. KEY WORDS: Amphibian decline Chytridiomycosis Emerging infectious disease Rana muscosa Tadpole Resale or republication not permitted without written consent of the publisher INTRODUCTION Infectious disease, alone and in synergy with other factors, has been implicated in the decline of amphibian populations worldwide (Blaustein et al. 1994, Laurance et al. 1996, Berger et al. 1998). Although many pathogens have been observed in amphibians (reviewed by Wright & Whitaker 2001), the chytrid fungus Batrachochytrium dendrobatidis has recently been associated with severe declines and extinctions of amphibians in Australia (Berger et al. 1998), South America (Young et al. 2001), Central America (Lips 1998), Mexico (Lips et al. 2004), USA (Bradley et al. 2002, Muths et al. 2003), and Europe (Bosch et al. 2001). The disease associated with this fungus, chytridiomycosis, has recently appeared in amphibian populations and is considered an emerging infectious disease (Daszak et al. 1999). Many of the reported outbreaks have occurred in protected habitats without obvious human impacts (Berger et al. 1998, Bosch et al. 2001, Muths et al. 2003), as is the case for the mountain yellow-legged frog Rana muscosa (Fellers et al. 2001). Rana muscosa exists almost entirely on protected federal lands, occurring in montane areas around the Los Angeles Basin in southern California and in the Sierra Nevada range in California and Nevada, USA (Zweifel 1955). The populations in southern California are listed as federally endangered, and Sierra Nevada populations are in steep decline. Although this species was once the most commonly encountered vertebrate in high elevation aquatic ecosystems of the Sierra Nevada (Grinnell & Storer 1924, Zweifel 1955), it has become increasingly rare over the past several decades (Jennings 1996). The best-documented cause of declines in R. muscosa is the introduction of nonnative fish (Bradford 1989, Bradford et al. 1993, Knapp & Matthews 2000, Vredenburg 2004), but chytridiomycosis presents a new threat (Fellers et al. 2001). Mass mortalities have occurred in postmetamorphic animals infected with Batrachochytrium dendrobatidis * larar@socrates.berkeley.edu Inter-Research
2 76 Dis Aquat Org 61: 75 83, 2004 in numerous anuran species (Berger et al. 1998, Daszak et al. 1999, Bosch et al. 2001, Bradley et al. 2002); however, no mortality has been reported in infected anuran larvae. Although B. dendrobatidis has been identified in amphibian species and amphibians have been experimentally infected from laboratoryreared B. dendrobatidis zoospores (the mode of transmission) (Berger et al. 1999, Nichols et al. 2001), studies documenting transmission within and between life stages are lacking. Our focal species, Rana muscosa, has an unusually long tadpole stage, lasting 2 to 4 yr prior to metamorphosis (Zweifel 1955). Historically, R. muscosa tadpoles of various age-classes formed extremely dense aggregations in the shallow areas of high-altitude lakes during summer days (Grinnell & Storer 1924). Although the species is in decline, dense aggregations of tadpoles and postmetamorphic animals have recently been observed along several lake shorelines (Vredenburg 2004). Knowing whether transmission occurs between tadpoles and from tadpoles to postmetamorphic animals will help us predict the spread and effects of chytridiomycosis. Batrachochytrium dendrobatidis is found in the keratinized structures of infected postmetamorphic animals (skin) and tadpoles (mouthparts) (Berger et al. 1998, Pessier et al. 1999, Fellers et al. 2001). The oral discs of most tadpoles contain keratinized jaw sheaths and tooth rows that are normally heavily pigmented (Altig & McDiarmid 1999). The current non-lethal method of identifying infection in tadpoles is inspecting mouthpart pigmentation for abnormalities (Berger et al. 1999, Lips 1999, Fellers et al. 2001, Vredenburg & Summers 2001, Bradley et al. 2002). In larval Rana muscosa, a lack of pigment of these structures is strongly associated with chytridiomycosis (Fellers et al. 2001). Pigment abnormalities in tadpole mouthparts associated with chytridiomycosis also occur in other species outside of the United States (Lips 1999, Lips et al. 2003, 2004). However, there is potential for erroneous identification of chytridiomycosis based on mouthpart abnormalities because depigmentation of mouthparts is also associated with metamorphosis (Gosner 1960), exposure to chemical contaminants (Hayes et al. 1997, Rowe et al. 1998), and low temperatures (Rachowicz 2002). The objectives of this study were (1) to test the possibility of conspecific transmission between Rana muscosa tadpoles and from tadpoles to postmetamorphic animals using controlled laboratory experiments, (2) to observe the effects of infection on larval and postmetamorphic stages, and (3) to infect R. muscosa tadpoles with laboratory-reared Batrachochytrium dendrobatidis zoospores and document the resulting pattern of pigment loss caused by B. dendrobatidis over time. This pattern was contrasted with the pattern of pigment loss observed with exposure to low temperature conditions (based on Rachowicz 2002). Field surveys were conducted to verify patterns observed in the laboratory. MATERIALS AND METHODS Detection of Batrachochytrium dendrobatidis. We compared several identification techniques to detect chytridiomycosis in Rana muscosa. To detect chytridiomycosis in live animals, we used visual inspection and cytology. Visual inspection consisted of examining the pigmentation of tadpole jaw sheaths and tooth rows with a 10 hand lens (Fellers et al. 2001). We estimated the percent of the upper and lower beak lengths that were pigmented (0 to 100%), classified the width of each beak as thinly or thickly pigmented, and classified the tooth rows as fully pigmented, partially depigmented, or fully depigmented. Cytology consisted of scraping epithelial cells off the skin of postmetamorphic animals (Berger et al. 1998) or the jaw sheaths of tadpoles with the dull side of a sterile scalpel blade, mounting cells on a slide with a drop of water, and microscopically examining the cells for the presence of B. dendrobatidis sporangia. Sporangia are spherical bodies (7 to 15 µm diam.) where zoospores are produced (Longcore et al. 1999, Pessier et al. 1999). Additionally, we used histology ( standard and serial sectioning ) and culturing to detect Batrachochytrium dendrobatidis. For histological examination, all animals were euthanized with MS-222 and fixed in 10% neutral buffered formalin. We embedded mouthparts in paraffin, sectioned at 8 µm, and stained with hematoxylin and eosin (H&E). For standard histology we processed 1 to 2 sagittal sections per mouthpart, whereas for serial sectioning we processed approximately 250 sagittal sections per mouthpart. We also cultured the fungal organism from tadpole mouthparts for the detection of B. dendrobatidis, following methods previously described (Longcore et al. 1999). Briefly, culturing involved incubating small pieces of infected mouthpart on a 1% tryptone, antibiotic agar plate at room temperature. After 1 wk, sporangia were transferred to a 1% tryptone and agar plate and incubated at room temperature for 7 to 10 d. Finally, a 1 cm 2 piece of agar containing sporangia was placed in a 30 ml flask of 1% tryptone broth and maintained at 4 C. Experimental infection via zoospore inoculation. To determine whether infection of Batrachochytrium dendrobatidis-free, wild-caught Rana muscosa tadpoles was possible, we collected 10 tadpoles from Kings Canyon National Park (KCNP) and housed them indi-
3 Rachowicz & Vredenburg: Batrachochytrium dendrobatidis in tadpoles 77 vidually in plastic, aerated containers filled with 4 l of carbon-filtered water at the animal care facility, University of California, Berkeley. Tank water was changed weekly and tadpoles were fed rabbit chow twice weekly. The water temperature was kept at 13 C and tadpoles received 12 h of light per day. Six of the containers received inoculations with B. dendrobatidis zoospores and the remaining 4 containers were used as controls. Tadpoles in the inoculated and control groups were of similar developmental stages (1.5 to 2.6 cm snout vent length [SVL], 0.7 to 3.3 g) and had fully pigmented jaw sheaths and tooth rows at the start of the observation (see Fig. 1a). Tadpoles were inoculated every 2 to 3 d over a period of 1 mo for a total of 12 inoculations. Fungal inoculum was prepared by spreading 1 ml of Batrachochytrium dendrobatidis broth culture (1% tryptone) on a 1% tryptone and agar plate and incubating for 7 to 10 d at room temperature. Inoculum was isolated by J. E. Longcore from a Rana muscosa tadpole collected in Mono County, California, USA (culture identification: JEL216). The plate was then flooded with 6 ml of sterile, distilled water for 45 to 60 min to release zoospores (following Longcore et al. 1999). One ml of the 6 ml zoospore solution was added directly into the water of each tadpole tank. On average, zoospores were in each inoculation, measured using a hemacytometer. Control inoculum was prepared following the same methods but without B. dendrobatidis in the broth. To test for early infection, 2 chytrid-exposed tadpoles and 1 control were euthanized 4 wk after the first inoculation. The remaining 4 tadpoles in the exposure group were inoculated 3 more times over the next week (total 15 inoculations) and then received no further inoculations. One of each of these 4 were euthanized at Weeks 7, 19, 20, or 20 after the first inoculation. The 3 remaining control tadpoles were euthanized at Week 20 after the first inoculation. To detect infection, standard histology was performed on 5 exposed tadpoles and all controls. As another method to detect infection, we cultured the fungus from 1 exposed tadpole euthanized at Week 20. Mouthparts of each tadpole were inspected and photographed once per week, beginning on the day of first inoculation until euthanasia. Based on the photographs and visual observations, we produced illustrations to summarize the pattern of pigment loss due to chytridiomycosis. Experimental transmission from tadpoles to conspecifics. To determine whether conspecific transmission was possible within the larval stage, 5 Rana muscosa tadpoles with confirmed Batrachochytrium dendrobatidis infections were housed with 8 uninfected tadpoles (Developmental Stages 37 to 39; sensu Gosner 1960). The infected tadpoles were wild-caught from Yosemite National Park (YNP) and the uninfected tadpoles were wild-caught from KCNP. Infection status was assessed by visual inspection and cytology. The tadpoles were housed together in a container filled with 10 l of carbon-filtered water and cared for in the same manner as the tadpoles exposed to B. dendrobatidis zoospores (described in the above section). To be closer to the optimal temperature for the fungus (17 to 23 C: Longcore et al. 1999, Piotrowski et al. 2004), the water temperature was maintained at 16 C. Infected tadpoles were marked by a small notch made in their tails above the muscle to be distinguishable from the initially uninfected individuals. To determine the onset of detectable infection, 2 exposed tadpoles were haphazardly selected from the tank each week for 4 wk. They were tested for Batrachochytrium dendrobatidis by visual inspection, cytology, and histology with serial sectioning. Four control tadpoles were housed together in a separate container. One control tadpole was removed and euthanized each week and tested for the fungal pathogen using the same tests applied to the exposed tadpoles. To determine whether conspecific transmission was possible from the larval to postmetamorphic stage, 6 Batrachochytrium dendrobatidis-free, postmetamorphic Rana muscosa were housed individually with 5 tadpoles each. Four containers housed tadpoles that were infected with B. dendrobatidis and 2 contained uninfected tadpoles as controls. Postmetamorphic animals were wild-caught as tadpoles from KCNP, metamorphosed in the laboratory, and were 2.5 to 3.1 cm SVL at the beginning of the experiment. Infected tadpoles were wild-caught in YNP and were at Stages 34 to 39. Uninfected tadpoles were wild-caught from KCNP and were at Stages 30 to 36. Infection status of tadpoles was determined visually and confirmed with cytology. Each container was filled with 8 l of carbonfiltered water and a platform was placed just above the water line for the postmetamorphic animal to rest. Animals were housed and fed in the same manner as the tadpoles exposed to B. dendrobatidis zoospores described above, with the addition of twenty 1 wk old crickets added each week as food for the postmetamorphic animal. Tank water temperature was maintained at 16 C. Infection status of postmetamorphic animals was assessed at the beginning of the experiment and at Day 18 via cytology. Postmetamorphic animals and tadpoles were observed every 1 to 2 d, and any mortality was recorded until the experiment ended on Day 96. Identification of Batrachochytrium dendrobatidis in tadpoles. To verify whether the patterns of depigmentation seen in the laboratory were similar to those in the field, surveys were conducted in 2002 at 5 sites
4 78 Dis Aquat Org 61: 75 83, 2004 containing Rana muscosa populations in Yosemite, Sequoia, and Kings Canyon National Parks in the Sierra Nevada mountains of California, USA. Surveys consisted of catching tadpoles (up to 25), assessing developmental stage, and visually inspecting the mouthparts of each. B. dendrobatidis infection in each population was further investigated by testing a subset of the tadpoles caught (n = 1 to 10) with cytology, histology with serial sectioning, and/or B. dendrobatidis culturing. RESULTS Experimental infection via zoospore inoculation We successfully infected previously uninfected tadpoles in the laboratory. Of the 4 tadpoles that were exposed and held for 7+ wk, all 4 became infected with Batrachochytrium dendrobatidis; 3 infections were confirmed with standard histology and 1 by culturing the fungus from the tadpole mouthpart. In contrast, based on results from standard histology, no evidence of infection was found in the 2 exposed tadpoles that were euthanized after 4 wk or in the 4 control animals. In histology and culture, fungal sporangia were not distinguishable from those previously described for B. dendrobatidis (Longcore et al. 1999, Pessier et al. 1999). Loss of pigment was detected visually in the 4 infected tadpoles while no pigment loss was found in the 2 exposed tadpoles in which infection was not detected or in the 4 control tadpoles. Pigment loss was first detected at Week 7 after the first inoculation in 2 tadpoles, Week 11 in the third, and Week 15 in the fourth. The extent of pigment loss increased quickly after initial detection. By Weeks 20 to 21, the total area of the jaw sheath from which pigment had been lost in the 3 remaining infected animals was 45%, 90%, and 98%. There was no pigment loss in the tooth rows. Experimental transmission from tadpoles to conspecifics We confirmed transmission of Batrachochytrium dendrobatidis between infected and initially uninfected tadpoles using histology with serial sectioning; B. dendrobatidis infection was found in animals exposed for 3 and 4 wk but not in animals exposed for 1 and 2 wk. There was no pigment loss (visual inspection) in the animals exposed for 1 and 2 wk. One of the 2 infected animals removed at Week 3 showed very slight mouthpart abnormalities (5% loss of upper jaw-sheath pigment, no loss of lower jawsheath pigment, and partially depigmented tooth rows) upon visual inspection. The animals from the first 3 wk were between Stages 37 and 40. The animals from Week 4 lost mouthpart pigmentation; however, because they were at Stages 41 to 42, any pigment loss was likely due to metamorphic changes. Jaw-sheath scrapes (cytology) were negative for B. dendrobatidis. Controls were negative for B. dendrobatidis based on visual inspection (Weeks 1 to 3) and cytology (Weeks 1 to 4). We confirmed transmission from the larval to postmetamorphic stage. All postmetamorphic animals were uninfected at the start of the experiment and the 4 animals exposed to infected tadpoles were infected on Day 18. The 2 controls were uninfected on Day 18. Three of the infected postmetamorphic animals were found dead on Day 39 and the fourth on Day 50. The 2 uninfected postmetamorphic animals were alive and clinically healthy on Day 96 when the experiment was ended. All 30 tadpoles (20 infected, 10 uninfected) remained alive until the end of the experiment and appeared clinically healthy. Identification of Batrachochytrium dendrobatidis in tadpoles Fig. 1a d shows the general pattern of pigment loss caused by chytridiomycosis. Based on the above experiments and field surveys (discussed below), pigment loss began with gaps (i.e. a complete loss of pigment in a localized area of the jaw sheath surrounded by fully pigmented areas; a discontinuity in the length of the beak pigment) in the jaw sheaths that became larger over time. Some infected tadpoles examined in the field had lost all jaw-sheath or tooth-row pigment. The general pattern elucidated from tadpoles exposed to a low temperature treatment (4ºC) is illustrated in Fig. 1e h, based on descriptions in Rachowicz (2002) and field surveys (below). In contrast, this pattern began with a loss of tooth-row pigment and a continuous reduction in jaw-sheath pigment width until it was completely lost. Both patterns started with complete pigmentation (Fig. 1a,e) and ended with a loss of all mouthpart pigment (Fig. 1d,h). Tadpoles exposed to a low temperature (Fig. 2a) or infected with Batrachochytrium dendrobatidis (Fig. 2b) showed histological differences that reflect the pigment loss patterns described above. After a winter at the bottom of a frozen lake (low temperature exposure), jaw-sheath cells had the same organization as fully pigmented jaw sheaths (Fig. 2a,c). However, after exposure to low temperature, fewer keratinized cells were seen in the jaw sheath, very few were pigmented, and no keratinized cells were seen in the tooth rows in the section shown. B. dendrobatidis-infected
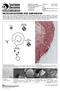
5 Rachowicz & Vredenburg: Batrachochytrium dendrobatidis in tadpoles 79 a b c d e f g h Fig. 1. Rana muscosa. Illustration of tadpole mouthparts showing 2 patterns of pigment loss over time. Upper (anterior) tooth rows (t), upper and lower (posterior) jaw sheaths (j), and 4 lower tooth rows (t) are shown. Black is pigmentation and gray is loss of pigment. (a d) The pattern of loss associated with Batrachochytrium dendrobatidis and (e h) the pattern of pigment loss associated with a low overwintering temperature. Note that some R. muscosa tadpoles have 1 to 2 additional tooth rows anterior to the upper jaw sheath. The outline of the tadpole mouthparts is from Altig et al. (1998) Fig. 2. Rana muscosa (a) Upper (anterior) jaw sheath and 2 upper tooth rows of a tadpole collected approximately 10 d after lake melt-out. Keratin is present in the jaw sheath but not in the tooth rows. The dark black on the end of the jaw sheath is the only pigmented keratin. (b) Upper jaw sheath infected with chytridiomycosis and 2 upper tooth rows. The jaw sheath contains numerous Batrachochytrium dendrobatidis sporangia (arrow). The dark black on the lower part of the jaw sheath and tooth rows is pigmented keratin. (c) Normal upper jaw sheath and 2 upper tooth rows. The dark black on the jaw sheath and tooth rows is pigmented keratin jaw sheaths (Fig. 2b) displayed histological features similar to those previously described (e.g. hyperkeratosis: Fellers et al. 2001). In the mouthpart section shown, pigmentation was only seen on part of the infected jaw-sheath section, and upon visual inspection, this area appeared to be the beginning of a gap. Our histological examinations of Rana muscosa tadpoles that were experimentally infected from conspe-
6 80 Dis Aquat Org 61: 75 83, 2004 cific tadpoles support the progression of the disease that is illustrated in Fig. 1a d. We detected very early infection (after 3 wk of exposure to infected tadpoles) using histology with serial sectioning; infection was discrete and localized within the jaw sheath, and few fungal sporangia were seen per section. After 4 wk of exposure, localized infections seen consisted of more sporangia and covered more area within the jaw sheath than Week 3 infections, and numerous localized infections had developed simultaneously within a jaw sheath. After 3 and 4 wk of exposure, sporangia were present in 3 to 20% of the jaw sheaths (lengthwise) and all sections with infection were consecutive. Stained slides showed that full pigment was preserved in tadpoles without any infection or visual pigment loss (Fig. 2c). Field surveys (Table 1) clearly showed 2 patterns of pigment loss in tadpoles: (1) low temperature-exposure pattern ( continuous ) and (2) Batrachochytrium dendrobatidis-infection pattern ( gaps ). In a survey at Site 1, which was conducted approximately 10 d after ice melted from the surface in early June 2002, tadpoles had the low temperature pattern of pigment loss, and no evidence of B. dendrobatidis was found. Sites 2 to 5 contained tadpoles with pigment loss (listed in order of progressing loss; Table 1) and confirmed Batrachochytrium dendrobatidis infections. Pigment was lacking in localized areas (gaps) of the jaw sheaths and/or an entire jaw sheath was missing pigment. When lack of pigment in the jaw-sheath length was large (>65%), tooth rows also lost part of or all pigment. DISCUSSION Chytridiomycosis is clearly associated with population collapse in several species of declining amphibians (Berger et al. 1998, Daszak et al. 1999, Lips 1999, Bradley et al. 2002, Lips et al. 2003); however, not all infected populations and species are being negatively affected (Weldon 2002, Mazzoni et al. 2003). To understand the population and species level dynamics of chytridiomycosis, studies that focus on the transmission and effects of the disease on all life stages of the host are necessary. In our experiments, we demonstrated that Rana muscosa tadpoles can be infected by fungal zoospores and that tadpoles can transmit the infection to each other and to postmetamorphic animals. We also showed that infected tadpoles remained clinically healthy while infected postmetamorphic animals died. All uninfected animals remained alive during our experiments. This result is consistent with observations of tadpoles remaining at sites where postmetamorphic anurans infected with Batrachochytrium dendrobatidis were dead or dying (Berger et al. 1998, Bradley et al. 2002, Mazzoni et al. 2003). The implications of the transmission of Batrachochytrium dendrobatidis from infected larval Rana muscosa likely go beyond this species. This study shows that transmission can occur between tadpoles and from tadpoles to postmetamorphic animals in one species, and thus raises the possibility that congregations of tadpoles may become extremely large reservoirs for the fungus, which might spread the disease to postmetamorphic animals. Our results can inform models Table 1. Rana muscosa. Examination of tadpoles at sites in Yosemite (YNP) and Sequoia-Kings Canyon (SEKI) National Parks in 2002 Site Date No. % with jaw- % of jaw- Pattern of jaw- % with No. tadpoles tested, type location examined sheath pig- sheath pig- sheath pig- tooth-row of Batrachochytrium ment loss ment lost ment loss pigment loss dendrobatidis test, and result (Gosner stage) 1-SEKI 11 June or 50 Continuous histology with serial sectioning; 4 negative (Stages 31 35) 2-YNP 22 June Gaps 8 6 histology with serial sectioning, 1 culture; 7 positive (Stages 37 39) 3-YNP 16 July Gaps 11 6 cytology, 4 histology with serial sectioning; 6 positive (Stages 30 40), 4 negative (Stages 26 28) 4-SEKI 24 June Gaps cytology, 5 culture; 8 positive (Stages 33 40) 5-YNP 26 September Gaps or no 55 1 cytology; 1 positive pigment (Stage 39)
7 Rachowicz & Vredenburg: Batrachochytrium dendrobatidis in tadpoles 81 that predict the dynamics of the disease, in particular by highlighting the need to include stage-structure to reflect the biphasic lifestyle of anurans where the larval stage is clinically healthy yet can transmit the disease to conspecifics. Simple epidemiological models show that a primary determinant of the persistence of infection is the length of the infectious period (Anderson & May 1991). Pathogens with short infectious periods do not tend to persist after an epidemic, whereas those with long infectious periods may be more likely to persist between epidemics (Keeling & Grenfell 1998). Given this, a healthy life stage may allow a disease to persist even when it causes mortality in another life stage. If infected tadpoles with long larval stages release zoospores over extended periods, long larval stages may increase the likelihood of outbreaks by preventing extinction of the fungal population. Bosch et al. (2001) found that in sympatric anurans in Spain, those with longer larval periods experienced severe epidemics while species with shorter larval periods did not. In the southwestern United States, Bradley et al. (2002) found chytridiomycosis in Rana yavapaiensis and speculated that the long larval period of this species may allow the pathogen to persist between observed seasonal outbreaks. Hopkins & Channing (2002) also speculated that the long larval period of Afrana fuscigula in South Africa makes the frog more susceptible to chytridiomycosis. R. muscosa may be vulnerable to severe chytridiomycosis in part because the pathogen is sustained in tadpoles during their 2 to 4 yr larval stage. In colder, high-altitude tropical areas of Australia and Central America, where chytridiomycosis has been associated with population declines in numerous species (Berger et al. 1998, Lips et al. 2003), individuals generally have longer larval periods than those living in the lowland, warmer tropics (Morrison & Hero 2003). Given the severity of population declines in these high-altitude regions, the role of larval-stage length in chytridiomycosis dynamics should be investigated. One notable exception is the American bullfrog R. catesbeiana; although it has a long larval period and both life stages become infected, adults retain a low level infection and seldom die (Bradley et al. 2002, Mazzoni et al. 2003). This example suggests that some species are less susceptible to the disease; species-specific innate immunity has been suggested as a factor (Carey et al. 2003). In species where the postmetamorphic animals are clinically healthy, this life stage could function as a reservoir or carrier, infecting members of sympatric species or more susceptible members of the same species. Two widely introduced postmetamorphic amphibians, the African clawed frog Xenopus laevis (Weldon 2002) and the American bullfrog Rana catesbeiana (Bradley et al. 2002, Mazzoni et al. 2003), can maintain Batrachochytrium dendrobatidis while remaining clinically healthy. Several native amphibians that now occur sympatrically with introduced bullfrogs have declined dramatically (Jennings & Hayes 1994), raising the question of the role of interspecies transmission in these declines. Identification of Batrachochytrium dendrobatidis in tadpoles Detection of early Batrachochytrium dendrobatidis infection is dependent upon the identification technique used. In the laboratory, infection was identified by histology with serial sectioning after 3 wk of exposure to infected conspecifics. Given the localized nature of an early stage of infection, false negative results are likely unless serial sections are examined. The 2 zoospore-inoculated tadpoles in which no infection was found were euthanized early and examined by histology using 2 sections. They may have been at an early stage of infection and serial sectioning may have allowed the detection of localized infected areas. By the time gaps were visible in jaw sheaths of our zoospore-inoculated tadpoles, however, 1 or 2 sections were sufficient to detect chytridiomycosis. To identify chytridiomycosis in the wild, simple, nonlethal detection techniques are necessary. Epithelial cell scrapes of tadpole mouthparts and postmetamorphic skin and visual inspection of tadpole mouthparts are possibilities (Berger et al. 1998, Fellers et al. 2001). In this study we detected Batrachochytrium dendrobatidis by identifying a specific pattern of pigmentation loss that can be distinguished from other types of loss in Rana muscosa, and confirmed B. dendrobatidis in animals with this pattern by other methods. Currently, an important weakness in chytridiomycosis research is the lack of a reliable, non-lethal diagnostic test that can confirm lack of infection. Based on our findings, we recommend that visual inspections in Rana muscosa (1) take place at least 3 wk after lake melt-out, (2) target older tadpoles (that have had a longer period to become infected), but those at less than Stage 41, and (3) focus on pigmentation gaps in the jaw sheaths. Although differences in the pattern of pigment loss between infection and prolonged low temperature exposure can be distinguished at many stages (Fig. 1b,c,e,f), mouthparts may look similar when jaw sheaths are fully depigmented (Fig. 1d,h); thus, surveys should not be conducted until mouthpart pigment has had sufficient time to return after the water temperatures warm. Older tadpoles may be more likely to be infected. At Sites 4 and 6 (Table 1), tadpoles showing pigment loss were primarily at
8 82 Dis Aquat Org 61: 75 83, 2004 Stages 35 to 40, whereas tadpoles at Stages 28 to 32 had not lost pigment. This may be caused by longer exposure times and/or larger mouthparts providing a larger substrate for Batrachochytrium dendrobatidis zoospores to infect. Tadpoles begin to metamorphose at Stage 41 and mouthparts become naturally depigmented (Gosner 1960, Altig & McDiarmid 1999); therefore, the techniques described in this paper should not be used on tadpoles that are beyond Gosner Stage 40. Gaps in jaw-sheath pigment should be used as the primary indicator of chytridiomycosis. Tooth-row abnormalities are more uncertain as a means for detection because teeth can be difficult to see due to their small size and because aberrant and displaced tooth rows are common (Altig & McDiarmid 1999). However, timing of tooth-row loss (Fig. 1) may be informative; tooth rows were lost after jaw-sheath pigment in many tadpoles with B. dendrobatidis and before jaw-sheath pigment in tadpoles exposed to low temperature. Because mouthpart depigmentation that is not associated with chytridiomycosis has been reported in larval anurans, we recommend that visual inspections of mouthparts be corroborated with histological analysis or other techniques in voucher specimens when possible. Exposure to DDT and corticosterone induce a regression of tissue (a hole ) that results in a complete loss of the upper mouthparts (Hayes et al. 1997). Additionally, bullfrog tadpoles exposed to coal-ash pollution during egg development had a significantly higher frequency of some portion of at least one tooth row missing than those that developed in an unpolluted pond (Rowe et al. 1998). Mouthpart loss in R. muscosa caused by chemical pollution has not been investigated. Others have found tadpole mouthpart abnormalities at sites where postmetamorphic animals are experiencing chytridiomycosis-related die-offs (hylids and centrolenids: Lips 1999, Lips et al. 2003; ranids: Fellers et al. 2001, Lips et al. 2004). However, researchers have not found abnormal tadpole mouthparts in some locations where the postmetamorphic animals are infected with Batrachochytrium dendrobatidis (Bufo canorus: Green & Sherman 2001; B. boreas: Muths et al. 2003; Rana yavapaiensis: Bradley et al. 2002). Whether B. dendrobatidis infects the tadpoles in these species is unknown. When chytridiomycosis was first recognized in Australia, abnormal mouthparts were not seen (Berger et al. 1999); however, pigmentation loss has recently been found in several Australian species (D. Mendez & P. Symonds pers. comm.). Visual inspection is probably relevant to other species, but investigations of the association between B. dendrobatidis infection and mouthpart pigment loss in other species should be conducted to verify the usefulness of visual inspection as a widescale detection technique. Acknowledgements. This work was funded by the NIH National Institutes of Environmental Health Sciences, Ecology of Infectious Diseases program (grant R01 ES ), and grants from the Canon National Parks Science Scholars Program, the Cleveland Zoological Society, and the Department of Integrative Biology, University of California, Berkeley. We are grateful to M. Daugherty, E. Knight, M. Stice, and T. Tunstall for excellent field and laboratory support and C. Briggs, M. Hoopes, R. Knapp, J. Morgan, J. Parker, M. Wake, and anonymous reviewers for useful comments. We thank P. Ustach for providing the initial illustrations of tadpole mouthparts, and D. Mendez and P. Symonds for personal communications. We especially thank J. E. Longcore for teaching us proper culturing technique and providing a fungal isolate. All animal procedures were approved by the Animal Care and Use Committee, University of California, Berkeley, USA. LITERATURE CITED Altig R, McDiarmid RW (1999) Body plan: development and morphology. In: McDiarmid RW, Altig R (eds) Tadpoles: the biology of anuran larvae. University of Chicago Press, Chicago, p Altig R, McDiarmid RW, Nichols KA, Ustach PC (1998) A key to the anuran tadpoles of the United States and Canada. Contemporary Herpetology Information Series 2 ( Anderson RM, May RM (1991) Infectious diseases of humans: dynamics and control, 1st edn. Oxford University Press, New York Berger L, Speare R, Daszak P, Green DE and 10 others (1998) Chytridiomycosis causes amphibian mortality associated with population declines in the rain forests of Australia and Central America. Proc Natl Acad Sci 95: Berger L, Speare R, Hyatt A (1999) Chytrid fungi and amphibian declines: overview, implications and future directions. Environment Australia, Canberra Blaustein AR, Hokit DG, O Hara RK (1994) Pathogenic fungus contributed to amphibian losses in the Pacific Northwest. Biol Conserv 67: Bosch J, Martinez-Solano I, Garcia-Paris M (2001) Evidence of a chytrid fungus infection involved in the decline of the common midwife toad (Alytes obstetricans) in protected areas of central Spain. Biol Conserv 97: Bradford DF (1989) Allotopic distribution of native frogs and introduced fish in high Sierra Nevada lakes of California: implications of the negative impact of fish introductions. Copeia 1989: Bradford DF, Tabatabai F, Graber DM (1993) Isolation of remaining populations of the native frog, Rana muscosa, by introduced fishes in Sequoia and Kings Canyon National Parks, California. Conserv Biol 7: Bradley GA, Rosen PC, Sredl MJ, Jones TR, Longcore JE (2002) Chytridiomycosis in native Arizona frogs. J Wildl Dis 38: Carey C, Pessier AP, Peace AD (2003) Pathogens, infectious disease, and immune defenses. In: Semlitsch RD (ed) Amphibian conservation. Smithsonian Books, Washington, DC, p Daszak P, Berger L, Cunningham AA, Hyatt AD, Green DE, Speare R (1999) Emerging infectious diseases and amphibian population declines. Emerg Infect Dis 5: Fellers GM, Green DE, Longcore JE (2001) Oral chytridio-
9 Rachowicz & Vredenburg: Batrachochytrium dendrobatidis in tadpoles 83 mycosis in the mountain yellow-legged frog (Rana muscosa). Copeia 2001: Gosner KL (1960) A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16: Green DE, Sherman CK (2001) Diagnostic histological findings in Yosemite toads (Bufo canorus) from a die-off in the 1970s. J Herpetol 35: Grinnell J, Storer T (1924) Animal life in the Yosemite. University of California Press, Berkeley, CA Hayes TB, Wu TH, Gill TN (1997) DDT-like effects as a result of corticosterone treatment in an anuran amphibian: is DDT a corticoid mimic or a stressor? Environ Toxicol Chem 16: Hopkins S, Channing A (2002) Chytrdiomycosis in Northern and Western Cape frog populations, South Africa. Herp- Digest 3: ( Jennings MR (1996) Status of amphibians. In: Sierra Nevada ecosystem project: final report to Congress, Vol 2. Assessments and scientific basis for management options. University of California, Centers for Water and Wildland Resources, Davis, p Jennings MR, Hayes MP (1994) Amphibian and reptile species of special concern in California. California Department of Fish and Game, Inland Fisheries Division, Rancho Cordova Keeling MJ, Grenfell BT (1998) Effect of variability in infection period on the persistence and spatial spread of infectious diseases. Math Biosci 147: Knapp RA, Matthews KR (2000) Non-native fish introductions and the decline of the mountain yellow-legged frog from within protected areas. Conserv Biol 14: Laurance WF, McDonald KR, Speare R (1996) Epidemic disease and the catastrophic decline of Australian rain forest frogs. Conserv Biol 10: Lips KR (1998) Decline of a tropical montane amphibian fauna. Conserv Biol 12: Lips KR (1999) Mass mortality and population declines of anurans at an upland site in western Panama. Conserv Biol 13: Lips KR, Green DE, Papendick R (2003) Chytridiomycosis in wild frogs from southern Costa Rica. J Herpetol 37: Lips KR, Mendelson III JR, Munoz-Alonso A, Canseco- Márquez L, Mulcahy DG (2004) Amphibian population declines in montane southern Mexico: resurveys of historical localities. Biol Conserv 119(4): Editorial responsibility: Peernel Zwart, Utrecht, The Netherlands Longcore JE, Pessier AP, Nichols DK (1999) Batrachochytrium dendrobatidis gen. et sp. nov., a chytrid pathogenic to amphibians. Mycologia 91: Mazzoni R, Cunningham AA, Daszak P, Apolo A, Perdomo E, Speranza G (2003) Emerging pathogen of wild amphibians in frogs (Rana catesbeiana) farmed for international trade. Emerg Infect Dis 9: Morrison C, Hero JM (2003) Geographic variation in lifehistory characteristics of amphibians: a review. J Anim Ecol 72: Muths E, Corn PS, Pessier AP, Green DE (2003) Evidence for disease-related amphibian decline in Colorado. Biol Conserv 110: Nichols DK, Lamirande EW, Pessier AP, Longcore JE (2001) Experimental transmission of cutaneous chytridiomycosis in dendrobatid frogs. J Wildl Dis 37:1 11 Pessier AP, Nichols DK, Longcore JE, Fuller MS (1999) Cutaneous chytridiomycosis in poison dart frogs (Dendrobates spp.) and White s tree frogs (Litoria caerulea). J Vet Diagn Invest 11: Piotrowski JS, Annis SL, Longcore JE (2004) Physiology of Batrachochytrium dendrobatidis, a chytrid pathogen of amphibians. Mycologia 96:9 15 Rachowicz LJ (2002) Mouthpart pigmentation in Rana muscosa tadpoles: seasonal changes without chytridiomycosis. Herpetol Rev 33: Rowe CL, Kinney OM, Congdon JD (1998) Oral deformities in tadpoles of the bullfrog (Rana catesbeiana) caused by conditions in a polluted habitat. Copeia 1: Vredenburg VT (2004) Reversing introduced species effects: experimental removal of introduced fish leads to rapid recovery of a declining frog. Proc Natl Acad Sci 101: Vredenburg VT, Summers AP (2001) Field identification of chytridiomycosis in Rana muscosa (Camp 1915). Herpetol Rev 32: Weldon C (2002) Chytridiomycosis survey in South Africa. Froglog 51:1 2 Wright KM, Whitaker BR (2001) Amphibian medicine and captive husbandry. Krieger Publishing, Malabar Young BE, Lips KR, Reaser JK, Ibanez R and 10 others (2001) Population declines and priorities for amphibian conservation in Latin America. Conserv Biol 15: Zweifel RG (1955) Ecology, distribution and systematics of frogs of the Rana boylii group. Univ Calif Publ Zool 54: Submitted: March 7, 2004; Accepted: June 21, 2004 Proofs received from author(s): September 14, 2004
EMERGING INFECTIOUS DISEASE AS A PROXIMATE CAUSE OF AMPHIBIAN MASS MORTALITY
 Ecology, 87(7), 2006, pp. 1671 1683 Ó 2006 by the Ecological Society of America EMERGING INFECTIOUS DISEASE AS A PROXIMATE CAUSE OF AMPHIBIAN MASS MORTALITY LARA J. RACHOWICZ, 1,5 ROLAND A. KNAPP, 2 JESS
Ecology, 87(7), 2006, pp. 1671 1683 Ó 2006 by the Ecological Society of America EMERGING INFECTIOUS DISEASE AS A PROXIMATE CAUSE OF AMPHIBIAN MASS MORTALITY LARA J. RACHOWICZ, 1,5 ROLAND A. KNAPP, 2 JESS
INVESTIGATING THE POPULATION-LEVEL EFFECTS OF CHYTRIDIOMYCOSIS: AN EMERGING INFECTIOUS DISEASE OF AMPHIBIANS
 Ecology, 86(12), 2005, pp. 3149 3159 2005 by the Ecological Society of America INVESTIGATING THE POPULATION-LEVEL EFFECTS OF CHYTRIDIOMYCOSIS: AN EMERGING INFECTIOUS DISEASE OF AMPHIBIANS CHERYL J. BRIGGS,
Ecology, 86(12), 2005, pp. 3149 3159 2005 by the Ecological Society of America INVESTIGATING THE POPULATION-LEVEL EFFECTS OF CHYTRIDIOMYCOSIS: AN EMERGING INFECTIOUS DISEASE OF AMPHIBIANS CHERYL J. BRIGGS,
Effect of Temperature on Host Response to Batrachochytrium dendrobatidis Infection in the Mountain Yellow-legged Frog (Rana muscosa)
 Effect of Temperature on Host Response to Batrachochytrium dendrobatidis Infection in the Mountain Yellow-legged Frog (Rana muscosa) Authors: Sara E. Andre, John Parker, and Cheryl J. Briggs Source: Journal
Effect of Temperature on Host Response to Batrachochytrium dendrobatidis Infection in the Mountain Yellow-legged Frog (Rana muscosa) Authors: Sara E. Andre, John Parker, and Cheryl J. Briggs Source: Journal
Use of Immunohistochemistry to Diagnose Chytridiomycosis in Dyeing Poison Dart Frogs (Dendrobates tinctorius)
 Use of Immunohistochemistry to Diagnose Chytridiomycosis in Dyeing Poison Dart Frogs (Dendrobates tinctorius) Author(s): Tracy Van Ells, James Stanton, Ann Strieby, Peter Daszak, Alex D. Hyatt, and Corrie
Use of Immunohistochemistry to Diagnose Chytridiomycosis in Dyeing Poison Dart Frogs (Dendrobates tinctorius) Author(s): Tracy Van Ells, James Stanton, Ann Strieby, Peter Daszak, Alex D. Hyatt, and Corrie
Evan H Campbell Grant Northeast Amphibian Research and Monitoring Initiative US Geological Survey Patuxent Wildlife Research Center
 Evan H Campbell Grant Northeast Amphibian Research and Monitoring Initiative US Geological Survey Patuxent Wildlife Research Center New England Chapter of the Wildlife Society Spring Workshop 2015 Color
Evan H Campbell Grant Northeast Amphibian Research and Monitoring Initiative US Geological Survey Patuxent Wildlife Research Center New England Chapter of the Wildlife Society Spring Workshop 2015 Color
Principal Investigator: Roland Knapp; Co-principal Investigator: Vance Vredenburg
 P092: Effectiveness of reintroductions and probiotic treatment as tools to restore the endangered Sierra Nevada yellow-legged frog (Rana sierrae) to the Lake Tahoe Basin Project period: July 1, 2012 to
P092: Effectiveness of reintroductions and probiotic treatment as tools to restore the endangered Sierra Nevada yellow-legged frog (Rana sierrae) to the Lake Tahoe Basin Project period: July 1, 2012 to
Ecotoxicology of Declining Amphibian Populations
 Ecotoxicology of Declining Amphibian Populations Some Questions What does the term declining amphibians mean? decrease in number of individuals in an area decrease in number of sites occupied by breeding
Ecotoxicology of Declining Amphibian Populations Some Questions What does the term declining amphibians mean? decrease in number of individuals in an area decrease in number of sites occupied by breeding
INTRODUCTION. Blacksburg, Virginia 24061, USA 4 Corresponding author: (
 Journal of Wildlife Diseases, 43(3), 2007, pp. 450 460 # Wildlife Disease Association 2007 PREVALENCE OF BATRACHOCHYTRIUM DENDROBATIDIS IN AMERICAN BULLFROG AND SOUTHERN LEOPARD FROG LARVAE FROM WETLANDS
Journal of Wildlife Diseases, 43(3), 2007, pp. 450 460 # Wildlife Disease Association 2007 PREVALENCE OF BATRACHOCHYTRIUM DENDROBATIDIS IN AMERICAN BULLFROG AND SOUTHERN LEOPARD FROG LARVAE FROM WETLANDS
Species-level correlates of susceptibility to the pathogenic amphibian fungus Batrachochytrium dendrobatidis in the United States
 Biodivers Conserv (2011) 20:1911 1920 DOI 10.1007/s10531-011-0066-4 ORIGINAL PAPER Species-level correlates of susceptibility to the pathogenic amphibian fungus Batrachochytrium dendrobatidis in the United
Biodivers Conserv (2011) 20:1911 1920 DOI 10.1007/s10531-011-0066-4 ORIGINAL PAPER Species-level correlates of susceptibility to the pathogenic amphibian fungus Batrachochytrium dendrobatidis in the United
Possible modes of dissemination of the amphibian chytrid Batrachochytrium dendrobatidis in the environment
 DISEASES OF AQUATIC ORGANISMS Vol. 65: 181 186, 2005 Published July 18 Dis Aquat Org Possible modes of dissemination of the amphibian chytrid Batrachochytrium dendrobatidis in the environment Megan L.
DISEASES OF AQUATIC ORGANISMS Vol. 65: 181 186, 2005 Published July 18 Dis Aquat Org Possible modes of dissemination of the amphibian chytrid Batrachochytrium dendrobatidis in the environment Megan L.
Nowhere to hide: impact of a temperature-sensitive amphibian pathogen along an elevation gradient in the temperate zone
 Nowhere to hide: impact of a temperature-sensitive amphibian pathogen along an elevation gradient in the temperate zone ROLAND A. KNAPP, 1, CHERYL J. BRIGGS, 2 THOMAS C. SMITH, 2 AND JEFF R. MAURER 3,4
Nowhere to hide: impact of a temperature-sensitive amphibian pathogen along an elevation gradient in the temperate zone ROLAND A. KNAPP, 1, CHERYL J. BRIGGS, 2 THOMAS C. SMITH, 2 AND JEFF R. MAURER 3,4
Saprolegnia: Amphibian Killer? Discussion Outline. What is Saprolegnia? 4/6/2009. What is Saprolegnia? What does Saprolegnia do to amphibians?
 Saprolegnia: Amphibian Killer? Brendan Van Mater Discussion Outline What is Saprolegnia? What does Saprolegnia do to amphibians? How is Saprolegnia connected to amphibian declines? Why is Saprolegnia the
Saprolegnia: Amphibian Killer? Brendan Van Mater Discussion Outline What is Saprolegnia? What does Saprolegnia do to amphibians? How is Saprolegnia connected to amphibian declines? Why is Saprolegnia the
Methodologies for the Care, Maintenance, and Breeding of Tropical Poison Frogs
 JOURNAL OF APPLIED ANIMAL WELFARE SCIENCE, 6(2), 95 102 Copyright 2003, Lawrence Erlbaum Associates, Inc. Methodologies for the Care, Maintenance, and Breeding of Tropical Poison Frogs Scott P. McRobert
JOURNAL OF APPLIED ANIMAL WELFARE SCIENCE, 6(2), 95 102 Copyright 2003, Lawrence Erlbaum Associates, Inc. Methodologies for the Care, Maintenance, and Breeding of Tropical Poison Frogs Scott P. McRobert
Interspecific Variation in Susceptibility of Frog Tadpoles to the Pathogenic Fungus Batrachochytrium dendrobatidis
 Interspecific Variation in Susceptibility of Frog Tadpoles to the Pathogenic Fungus Batrachochytrium dendrobatidis ANDREW R. BLAUSTEIN, JOHN M. ROMANSIC, ERIN A. SCHEESSELE, BARBARA A. HAN, ALLAN P. PESSIER,
Interspecific Variation in Susceptibility of Frog Tadpoles to the Pathogenic Fungus Batrachochytrium dendrobatidis ANDREW R. BLAUSTEIN, JOHN M. ROMANSIC, ERIN A. SCHEESSELE, BARBARA A. HAN, ALLAN P. PESSIER,
Impact of a Chytrid-related mortality event on a population of the Green and Golden Bell Frog Litoria aurea
 University of Wollongong Research Online Faculty of Science - Papers (Archive) Faculty of Science, Medicine and Health 2008 Impact of a Chytrid-related mortality event on a population of the Green and
University of Wollongong Research Online Faculty of Science - Papers (Archive) Faculty of Science, Medicine and Health 2008 Impact of a Chytrid-related mortality event on a population of the Green and
[Byline: Erica Bree Rosenblum1*, Jamie Voyles2, Thomas J. Poorten1, Jason E. Stajich3,4
 2010-02-03-35 Chytrid fungus, frogs - worldwide: review article To: (05) Zoonoses, general; (23) Veterinary education ************************************************* CHYTRID FUNGUS, FROGS - WORLDWIDE:
2010-02-03-35 Chytrid fungus, frogs - worldwide: review article To: (05) Zoonoses, general; (23) Veterinary education ************************************************* CHYTRID FUNGUS, FROGS - WORLDWIDE:
Annual assessment of Chytrid fungus prevalence amongst endangered native Fijian ground frog populations on Viwa Fiji Islands Project ID:
 Annual assessment of Chytrid fungus prevalence amongst endangered native Fijian ground frog populations on Viwa Fiji Islands Project ID: 29.07.09 Final Report Submitted by Dr. Edward Narayan Fiji Islands
Annual assessment of Chytrid fungus prevalence amongst endangered native Fijian ground frog populations on Viwa Fiji Islands Project ID: 29.07.09 Final Report Submitted by Dr. Edward Narayan Fiji Islands
Amphibian chytridiomycosis: strategies for captive management and conservation
 REVIEW: AMPHIBIANS & CHYTRIDIOMYCOSIS: STRATEGIES FOR CAPTIVE MANAGEMENT 85 Int. Zoo Yb. (2007) 41: 85 95 DOI:10.1111/j.1748-1090.2007.00010.x Amphibian chytridiomycosis: strategies for captive management
REVIEW: AMPHIBIANS & CHYTRIDIOMYCOSIS: STRATEGIES FOR CAPTIVE MANAGEMENT 85 Int. Zoo Yb. (2007) 41: 85 95 DOI:10.1111/j.1748-1090.2007.00010.x Amphibian chytridiomycosis: strategies for captive management
Introduction. Abstract
 Temperature alters reproductive life history patterns in Batrachochytrium dendrobatidis, a lethal pathogen associated with the global loss of amphibians Jamie Voyles 1,4, Leah R. Johnson 2,3, Cheryl J.
Temperature alters reproductive life history patterns in Batrachochytrium dendrobatidis, a lethal pathogen associated with the global loss of amphibians Jamie Voyles 1,4, Leah R. Johnson 2,3, Cheryl J.
Distribution Limits of Batrachochytrium dendrobatidis: A Case Study in the Rocky Mountains, USA
 Journal of Wildlife Diseases, 45(4), 2009, pp. 1198 1202 # Wildlife Disease Association 2009 Distribution Limits of Batrachochytrium dendrobatidis: A Case Study in the Rocky Mountains, USA Blake R. Hossack,
Journal of Wildlife Diseases, 45(4), 2009, pp. 1198 1202 # Wildlife Disease Association 2009 Distribution Limits of Batrachochytrium dendrobatidis: A Case Study in the Rocky Mountains, USA Blake R. Hossack,
Amphibian Population Declines and deformities are useful phenomena to illustrate concepts in evolutionary biology
 Amphibian Population Declines and deformities are useful phenomena to illustrate concepts in evolutionary biology Today, I will focus on a particular aspect of the amphibian population decline phenomenon
Amphibian Population Declines and deformities are useful phenomena to illustrate concepts in evolutionary biology Today, I will focus on a particular aspect of the amphibian population decline phenomenon
Tadpole requirements?
 Tadpole Husbandry Peter Harlow Tadpole requirements? We need to know the maximum, minimum and optimum values for these important variables for each species we keep: Water Quality; ph Temperature Nitrate
Tadpole Husbandry Peter Harlow Tadpole requirements? We need to know the maximum, minimum and optimum values for these important variables for each species we keep: Water Quality; ph Temperature Nitrate
Survival of three species of anuran metamorphs exposed to UV-B radiation and the pathogenic fungus Batrachochytrium dendrobatidis
 DISEASES OF AQUATIC ORGANISMS Vol. 72: 163 169, 2006 Published October 17 Dis Aquat Org Survival of three species of anuran metamorphs exposed to UV-B radiation and the pathogenic fungus Batrachochytrium
DISEASES OF AQUATIC ORGANISMS Vol. 72: 163 169, 2006 Published October 17 Dis Aquat Org Survival of three species of anuran metamorphs exposed to UV-B radiation and the pathogenic fungus Batrachochytrium
Effects of the Amphibian Chytrid Fungus and Four Insecticides on Pacific Treefrogs (Pseudacris regilla)
 Journal of Herpetology, Vol. 46, No. 4, 625 631, 2012 Copyright 2012 Society for the Study of Amphibians and Reptiles Effects of the Amphibian Chytrid Fungus and Four Insecticides on Pacific Treefrogs
Journal of Herpetology, Vol. 46, No. 4, 625 631, 2012 Copyright 2012 Society for the Study of Amphibians and Reptiles Effects of the Amphibian Chytrid Fungus and Four Insecticides on Pacific Treefrogs
The ecology and impact of chytridiomycosis: an emerging disease of amphibians
 Review The ecology and impact of chytridiomycosis: an emerging disease of amphibians A. Marm Kilpatrick 1, Cheryl J. Briggs 2 and Peter Daszak 3 1 Department of Ecology and Evolutionary Biology, University
Review The ecology and impact of chytridiomycosis: an emerging disease of amphibians A. Marm Kilpatrick 1, Cheryl J. Briggs 2 and Peter Daszak 3 1 Department of Ecology and Evolutionary Biology, University
Methodologies for the Care, Maintenance, and Breeding of Tropical Poison Frogs Scott P. McRobert Published online: 04 Jun 2010.
 This article was downloaded by: [Dr Kenneth Shapiro] On: 08 June 2015, At: 09:45 Publisher: Routledge Informa Ltd Registered in England and Wales Registered Number: 1072954 Registered office: Mortimer
This article was downloaded by: [Dr Kenneth Shapiro] On: 08 June 2015, At: 09:45 Publisher: Routledge Informa Ltd Registered in England and Wales Registered Number: 1072954 Registered office: Mortimer
4/18/2013 FUNGUS MAP WHAT IS SAPROLEGNIASIS
 SAPROLEGNIASIS: WE VE GOT OUR MCYETES ON YOU Christina Saidak WFS 433/533 Spring Semester 2013 FUNGUS MAP What is Saprolegniasis? Saprolegnia spp. Defined Saprolegnia Life Cycle Saprolegniasis In Amphibians
SAPROLEGNIASIS: WE VE GOT OUR MCYETES ON YOU Christina Saidak WFS 433/533 Spring Semester 2013 FUNGUS MAP What is Saprolegniasis? Saprolegnia spp. Defined Saprolegnia Life Cycle Saprolegniasis In Amphibians
PATHOGENICITY OF BATRACHOCHYTRIUM DENDROBATIDIS IN LARVAL AMBYSTOMATID SALAMANDERS
 Herpetological Conservation and Biology 5(2):174-182. Submitted: 18 December 2009; Accepted: 17 May 2010. PATHOGENICITY OF BATRACHOCHYTRIUM DENDROBATIDIS IN LARVAL AMBYSTOMATID SALAMANDERS MATTHEW D. VENESKY
Herpetological Conservation and Biology 5(2):174-182. Submitted: 18 December 2009; Accepted: 17 May 2010. PATHOGENICITY OF BATRACHOCHYTRIUM DENDROBATIDIS IN LARVAL AMBYSTOMATID SALAMANDERS MATTHEW D. VENESKY
Distribution of Batrachochytrium dendrobatidis and pathology in the skin of green tree frogs Litoria caerulea with severe chytridiomycosis
 DISEASES OF AQUATIC ORGANISMS Vol. 68: 65 70, 2005 Published December 30 Dis Aquat Org Distribution of Batrachochytrium dendrobatidis and pathology in the skin of green tree frogs Litoria caerulea with
DISEASES OF AQUATIC ORGANISMS Vol. 68: 65 70, 2005 Published December 30 Dis Aquat Org Distribution of Batrachochytrium dendrobatidis and pathology in the skin of green tree frogs Litoria caerulea with
MICROORGANISMS AND AMPHIBIANS
 The life cycle of the chytrid fungus Batrachochytrium dendrobatidis begins with a motile zoospore, which is the infective stage of this pathogen. During the course of infection, chytrid zoospores enter
The life cycle of the chytrid fungus Batrachochytrium dendrobatidis begins with a motile zoospore, which is the infective stage of this pathogen. During the course of infection, chytrid zoospores enter
Mechanisms behind the successful invasion of American Bullfrogs (Rana catesbeiana) in the Northwest United States
 Mechanisms behind the successful invasion of American Bullfrogs (Rana catesbeiana) in the Northwest United States Tiffany Garcia, Rebbecca Hill, Sarah Abdulkarim, and Chris Funk Department of Fisheries
Mechanisms behind the successful invasion of American Bullfrogs (Rana catesbeiana) in the Northwest United States Tiffany Garcia, Rebbecca Hill, Sarah Abdulkarim, and Chris Funk Department of Fisheries
INFLUENCE OF ANURAN PREY ON THE CONDITION AND DISTRIBUTION OF RANA MUSCOSA IN THE SIERRA NEVADA
 354 HERPETOLOGICA [Vol. 58, No. 3 Herpetologica, 58(3), 2002, 354 363 2002 by The Herpetologists League, Inc. INFLUENCE OF ANURAN PREY ON THE CONDITION AND DISTRIBUTION OF RANA MUSCOSA IN THE SIERRA NEVADA
354 HERPETOLOGICA [Vol. 58, No. 3 Herpetologica, 58(3), 2002, 354 363 2002 by The Herpetologists League, Inc. INFLUENCE OF ANURAN PREY ON THE CONDITION AND DISTRIBUTION OF RANA MUSCOSA IN THE SIERRA NEVADA
3/24/10. Amphibian community ecology. Lecture goal. Lecture concepts to know
 Amphibian community ecology Lecture goal To familiarize students with the abiotic and biotic factors that structure amphibian communities, patterns in species richness, and encourage discussion about community
Amphibian community ecology Lecture goal To familiarize students with the abiotic and biotic factors that structure amphibian communities, patterns in species richness, and encourage discussion about community
Movement Ecology and Seasonal Distribution of Mountain Yellow-Legged Frogs, Rana muscosa, in a High-Elevation Sierra Nevada Basin
 Copeia, 2001(3), pp. 787 793 Movement Ecology and Seasonal Distribution of Mountain Yellow-Legged Frogs, Rana muscosa, in a High-Elevation Sierra Nevada Basin KAREN L. POPE AND KATHLEEN R. MATTHEWS Movement
Copeia, 2001(3), pp. 787 793 Movement Ecology and Seasonal Distribution of Mountain Yellow-Legged Frogs, Rana muscosa, in a High-Elevation Sierra Nevada Basin KAREN L. POPE AND KATHLEEN R. MATTHEWS Movement
THE Mountain Yellow-legged Frog, Rana
 Copeia, 2007(4), pp. 986 993 A Skeletochronological Study of the Age Structure, Growth, and Longevity of the Mountain Yellow-legged Frog, Rana muscosa, in the Sierra Nevada, California KATHLEEN R. MATTHEWS
Copeia, 2007(4), pp. 986 993 A Skeletochronological Study of the Age Structure, Growth, and Longevity of the Mountain Yellow-legged Frog, Rana muscosa, in the Sierra Nevada, California KATHLEEN R. MATTHEWS
CHAPTER. Population Ecology
 CHAPTER 4 Population Ecology Chapter 4 TOPIC POPULATION ECOLOGY Indicator Species Serve as Biological Smoke Alarms Indicator species Provide early warning of damage to a community Can monitor environmental
CHAPTER 4 Population Ecology Chapter 4 TOPIC POPULATION ECOLOGY Indicator Species Serve as Biological Smoke Alarms Indicator species Provide early warning of damage to a community Can monitor environmental
Host species determines whether infection load increases beyond disease-causing thresholds following exposure to the amphibian chytrid fungus
 Host species determines whether infection load increases beyond disease-causing thresholds following exposure to the amphibian chytrid fungus M. P. Stockwell, J. Clulow & M. J. Mahony Animal Conservation.
Host species determines whether infection load increases beyond disease-causing thresholds following exposure to the amphibian chytrid fungus M. P. Stockwell, J. Clulow & M. J. Mahony Animal Conservation.
AMPHIBIAN CHYTRID FUNGUS (BATRACHOCHYTRIUM
 Herpetological Conservation and Biology 6(3):383 394. Submitted: 13 April 2010; Accepted: 16 September 2011; Published: 31 December 2011. AMPHIBIAN CHYTRID FUNGUS (BATRACHOCHYTRIUM DENDROBATIDIS) IN COASTAL
Herpetological Conservation and Biology 6(3):383 394. Submitted: 13 April 2010; Accepted: 16 September 2011; Published: 31 December 2011. AMPHIBIAN CHYTRID FUNGUS (BATRACHOCHYTRIUM DENDROBATIDIS) IN COASTAL
Altitudinal distribution of chytrid (Batrachochytrium dendrobatidis) infection in subtropical Australian frogs
 Austral Ecology (2008) 33, 22 32 Altitudinal distribution of chytrid (Batrachochytrium dendrobatidis) infection in subtropical Australian frogs KERRY M. KRIGER* AND JEAN-MARC HERO Griffith University,
Austral Ecology (2008) 33, 22 32 Altitudinal distribution of chytrid (Batrachochytrium dendrobatidis) infection in subtropical Australian frogs KERRY M. KRIGER* AND JEAN-MARC HERO Griffith University,
Herpetological Review
 Herpetological Review chytrid fungus in American bullfrogs (Lithobates catesbeianus) along the platte River, nebraska, usa mary J. harner Platte River Whooping Crane Critical Habitat Maintenance Trust,
Herpetological Review chytrid fungus in American bullfrogs (Lithobates catesbeianus) along the platte River, nebraska, usa mary J. harner Platte River Whooping Crane Critical Habitat Maintenance Trust,
Chytridiomycosis: a global threat to amphibians
 Rev. sci. tech. Off. int. Epiz., 2013, 32 (3),... -... Chytridiomycosis: a global threat to amphibians P.L.L. Pereira (1)*, A.M.C. Torres M. Hijosa-Valsero (2) & E. Bécares (2) (1), D.F.M. Soares (1),
Rev. sci. tech. Off. int. Epiz., 2013, 32 (3),... -... Chytridiomycosis: a global threat to amphibians P.L.L. Pereira (1)*, A.M.C. Torres M. Hijosa-Valsero (2) & E. Bécares (2) (1), D.F.M. Soares (1),
Purpose. Potential causes for amphibian declines in Puerto Rico. Three species presumably extinct! Long Term Data of Transect Surveys :
 Potential causes for amphibian declines in Puerto Rico Patricia A. Burrowes, Rafael L. Joglar and Ana Longo University of Puerto Rico Purpose Summarize findings on the current status of Puerto Rican Amphibians
Potential causes for amphibian declines in Puerto Rico Patricia A. Burrowes, Rafael L. Joglar and Ana Longo University of Puerto Rico Purpose Summarize findings on the current status of Puerto Rican Amphibians
Goal of the Lecture. Lecture Structure. Tadpole Development, Ecology, and Metamorphosis
 Tadpole Development, Ecology, and Metamorphosis Matthew J. Gray, Ph.D. College of Agricultural Sciences and Natural Resources University of Tennessee-Knoxville Goal of the Lecture To familiarize students
Tadpole Development, Ecology, and Metamorphosis Matthew J. Gray, Ph.D. College of Agricultural Sciences and Natural Resources University of Tennessee-Knoxville Goal of the Lecture To familiarize students
Effects of nonnative fish and habitat characteristics on lentic herpetofauna in Yosemite National Park, USA
 BIOLOGICAL CONSERVATION Biological Conservation 121 (25) 265 279 www.elsevier.com/locate/biocon Effects of nonnative fish and habitat characteristics on lentic herpetofauna in Yosemite National Park, USA
BIOLOGICAL CONSERVATION Biological Conservation 121 (25) 265 279 www.elsevier.com/locate/biocon Effects of nonnative fish and habitat characteristics on lentic herpetofauna in Yosemite National Park, USA
Tadpoles of Mannophryne trinitatis and M. olmonae (Anura: Aromobatidae): Further Description and Comparison
 Tadpoles of Mannophryne trinitatis and M. olmonae (Anura: Aromobatidae): Further Description and Comparison J.R. Downie and M. Nokhbatolfoghahai Downie, J.R., Nokhbatolfoghahai, M. 2014. Tadpoles of Mannophryne
Tadpoles of Mannophryne trinitatis and M. olmonae (Anura: Aromobatidae): Further Description and Comparison J.R. Downie and M. Nokhbatolfoghahai Downie, J.R., Nokhbatolfoghahai, M. 2014. Tadpoles of Mannophryne
Batrachochytrium dendrobatidis: Chytrid disease
 Batrachochytrium dendrobatidis: Chytrid disease FISH497 Fall 2008 Aquatic Invasion Ecology Final Report By: Dave Lawrence Section 1. Diagnostic information Batrachochytrium dendrobatidis, commonly called
Batrachochytrium dendrobatidis: Chytrid disease FISH497 Fall 2008 Aquatic Invasion Ecology Final Report By: Dave Lawrence Section 1. Diagnostic information Batrachochytrium dendrobatidis, commonly called
Spread of Chytridiomycosis Has Caused the Rapid Global Decline and Extinction of Frogs
 EcoHealth 4, 125 134, 2007 DOI: 10.1007/s10393-007-0093-5 Ó 2007 EcoHealth Journal Consortium Spread of Chytridiomycosis Has Caused the Rapid Global Decline and Extinction of Frogs Lee Francis Skerratt,
EcoHealth 4, 125 134, 2007 DOI: 10.1007/s10393-007-0093-5 Ó 2007 EcoHealth Journal Consortium Spread of Chytridiomycosis Has Caused the Rapid Global Decline and Extinction of Frogs Lee Francis Skerratt,
CHYTRIDIOMYCOSIS caused by the fungal pathogen
 Copeia 2009, No. 4, 653 660 An Aquatic Disease on a Terrestrial Salamander: Individual and Population Level Effects of the Amphibian Chytrid Fungus, Batrachochytrium dendrobatidis, onbatrachoseps attenuatus
Copeia 2009, No. 4, 653 660 An Aquatic Disease on a Terrestrial Salamander: Individual and Population Level Effects of the Amphibian Chytrid Fungus, Batrachochytrium dendrobatidis, onbatrachoseps attenuatus
The ecology of chytridiomycosis in red-legged frog (Rana aurora) tadpoles. Phineas Hamilton B.Sc., University of Victoria, 2006
 The ecology of chytridiomycosis in red-legged frog (Rana aurora) tadpoles by Phineas Hamilton B.Sc., University of Victoria, 2006 A Thesis Submitted in Partial Fulfillment of the Requirements for the Degree
The ecology of chytridiomycosis in red-legged frog (Rana aurora) tadpoles by Phineas Hamilton B.Sc., University of Victoria, 2006 A Thesis Submitted in Partial Fulfillment of the Requirements for the Degree
Environmental Science Chapter 13 Atmosphere and Climate Change Review
 Environmental Science Chapter 13 Atmosphere and Climate Change Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Climate in a region is a. the long-term,
Environmental Science Chapter 13 Atmosphere and Climate Change Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Climate in a region is a. the long-term,
A SURVEY FOR CHYTRID FUNGUS IN THAI AMPHIBIANS
 THE RAFFLES BULLETIN OF ZOOLOGY 2008 THE RAFFLES BULLETIN OF ZOOLOGY 2008 56(1): 199 204 Date of Publication: 29 Feb.2008 National University of Singapore A SURVEY FOR CHYTRID FUNGUS IN THAI AMPHIBIANS
THE RAFFLES BULLETIN OF ZOOLOGY 2008 THE RAFFLES BULLETIN OF ZOOLOGY 2008 56(1): 199 204 Date of Publication: 29 Feb.2008 National University of Singapore A SURVEY FOR CHYTRID FUNGUS IN THAI AMPHIBIANS
Phenotypic variation 3/6/17. Phenotypic plasticity in amphibians
 Phenotypic plasticity in amphibians Goals Overview of phenotypic plasticity Summary of different types of plasticity Discuss costs and benefits of plasticity Discuss complexity of plasticity Readings Wells:
Phenotypic plasticity in amphibians Goals Overview of phenotypic plasticity Summary of different types of plasticity Discuss costs and benefits of plasticity Discuss complexity of plasticity Readings Wells:
Integrative and objective science is the best link between amphibian decline research and conservation on the ground
 Alytes, 2012, 29 (1 4): 119-132. 119 Integrative and objective science is the best link between amphibian decline research and conservation on the ground Carlos A. Navas a, Catherine R. Bevier b & Ana
Alytes, 2012, 29 (1 4): 119-132. 119 Integrative and objective science is the best link between amphibian decline research and conservation on the ground Carlos A. Navas a, Catherine R. Bevier b & Ana
amphibian decline, Batrachochytrium dendrobatidis, biodiversity, historical ecology, local ecological knowledge, oral history, reintroduction
 Received: 8 April 2017 Revised: 6 August 2017 Accepted: 19 August 2017 DOI: 10.1002/ece3.3468 ORIGINAL RESEARCH Rapid extirpation of a North American frog coincides with an increase in fungal pathogen
Received: 8 April 2017 Revised: 6 August 2017 Accepted: 19 August 2017 DOI: 10.1002/ece3.3468 ORIGINAL RESEARCH Rapid extirpation of a North American frog coincides with an increase in fungal pathogen
BEFORE THE SECRETARY OF INTERIOR ) ) ) ) ) ) ) February 28, 2000
 BEFORE THE SECRETARY OF INTERIOR CENTER FOR BIOLOGICAL DIVERSITY AND PACIFIC RIVERS COUNCIL Petitioners ) ) ) ) ) ) ) PETITION TO LIST THE YOSEMITE TOAD (BUFO CANORUS) AS AN ENDANGERED SPECIES UNDER THE
BEFORE THE SECRETARY OF INTERIOR CENTER FOR BIOLOGICAL DIVERSITY AND PACIFIC RIVERS COUNCIL Petitioners ) ) ) ) ) ) ) PETITION TO LIST THE YOSEMITE TOAD (BUFO CANORUS) AS AN ENDANGERED SPECIES UNDER THE
TROPICAL BIOLOGY AND CONSERVATION MANAGEMENT - Vol. VIII - Threats To Amphibians In Tropical Regions - Jean-Marc Hero and Kerry Kriger
 THREATS TO AMPHIBIANS IN TROPICAL REGIONS Centre for Innovative Conservation Strategies, Griffith University, PMB 50 Gold Coast Mail Centre, Queensland 9726, Australia Keywords: amphibian, species, populations,
THREATS TO AMPHIBIANS IN TROPICAL REGIONS Centre for Innovative Conservation Strategies, Griffith University, PMB 50 Gold Coast Mail Centre, Queensland 9726, Australia Keywords: amphibian, species, populations,
What Shapes an Ecosystem? Section 4-2 pgs 90-97
 What Shapes an Ecosystem? Section 4-2 pgs 90-97 What Shapes an Ecosystem? If you ask an ecologist where a particular organism lives, that person might say the organism lives on a Caribbean coral reef,
What Shapes an Ecosystem? Section 4-2 pgs 90-97 What Shapes an Ecosystem? If you ask an ecologist where a particular organism lives, that person might say the organism lives on a Caribbean coral reef,
Biological Conservation
 Biological Conservation 144 (2011) 2913 2921 Contents lists available at SciVerse ScienceDirect Biological Conservation journal homepage: www.elsevier.com/locate/biocon Factors related to the distribution
Biological Conservation 144 (2011) 2913 2921 Contents lists available at SciVerse ScienceDirect Biological Conservation journal homepage: www.elsevier.com/locate/biocon Factors related to the distribution
AMPHIBIAN AND REPTILE DISEASES
 AMPHIBIAN AND REPTILE DISEASES 37 AMPHIBIAN AND REPTILE DISEASES Herpetological Review, 2018, 49(1), 37 41. 2018 by Society for the Study of Amphibians and Reptiles Expanding Knowledge of Batrachochytrium
AMPHIBIAN AND REPTILE DISEASES 37 AMPHIBIAN AND REPTILE DISEASES Herpetological Review, 2018, 49(1), 37 41. 2018 by Society for the Study of Amphibians and Reptiles Expanding Knowledge of Batrachochytrium
Do emerging infectious diseases have a role in the decline and extinction of species? 1. Global loss of biodiversity 2. Emerging infectious diseases
 The Role of Infectious Disease in Amphibian Population Decline and Extinction James P. Collins School of Life Sciences Arizona State University University of Tennessee Department of Forestry, Wildlife
The Role of Infectious Disease in Amphibian Population Decline and Extinction James P. Collins School of Life Sciences Arizona State University University of Tennessee Department of Forestry, Wildlife
Impacts of Changes in Extreme Weather and Climate on Wild Plants and Animals. Camille Parmesan Integrative Biology University of Texas at Austin
 Impacts of Changes in Extreme Weather and Climate on Wild Plants and Animals Camille Parmesan Integrative Biology University of Texas at Austin Species Level: Climate extremes determine species distributions
Impacts of Changes in Extreme Weather and Climate on Wild Plants and Animals Camille Parmesan Integrative Biology University of Texas at Austin Species Level: Climate extremes determine species distributions
Higher temperature variability increases the impact of Batrachochytrium dendrobatidis and shifts interspecific interactions in tadpole mesocosms
 Higher temperature variability increases the impact of Batrachochytrium dendrobatidis and shifts interspecific interactions in tadpole mesocosms Phineas T. Hamilton 1, Jean M.L. Richardson 1, Purnima Govindarajulu
Higher temperature variability increases the impact of Batrachochytrium dendrobatidis and shifts interspecific interactions in tadpole mesocosms Phineas T. Hamilton 1, Jean M.L. Richardson 1, Purnima Govindarajulu
If you Build It, They Will Come
 If you Build It, They Will Come Use of Temporary Microbiota as an Environmentally sound alternative to the traditional frog life cycle project in home education settings. Allison Metler Questions Can temporary
If you Build It, They Will Come Use of Temporary Microbiota as an Environmentally sound alternative to the traditional frog life cycle project in home education settings. Allison Metler Questions Can temporary
Teacher s Resource Pack
 Teacher s Resource Pack Stage 4 & Stage 5 Geography Book your excursion today! Call (02) 8251 7801 Welcome to SEA LIFE Sydney Aquarium s Geography Student Resources Teacher Notes A trip to SEA LIFE Sydney
Teacher s Resource Pack Stage 4 & Stage 5 Geography Book your excursion today! Call (02) 8251 7801 Welcome to SEA LIFE Sydney Aquarium s Geography Student Resources Teacher Notes A trip to SEA LIFE Sydney
Evolution and pathogenicity in the deadly chytrid pathogen of amphibians. Erica Bree Rosenblum UC Berkeley
 Evolution and pathogenicity in the deadly chytrid pathogen of amphibians Erica Bree Rosenblum UC Berkeley Emerging infectious disease Emerging infectious disease EID events have risen significantly over
Evolution and pathogenicity in the deadly chytrid pathogen of amphibians Erica Bree Rosenblum UC Berkeley Emerging infectious disease Emerging infectious disease EID events have risen significantly over
EFFECTS OF CANOPY COVER ON THE LANDSCAPE EPIDEMIOLOGY OF AN AMPHIBIAN CHYTRID FUNGUS STACY ELIZABETH BEYER THESIS
 EFFECTS OF CANOPY COVER ON THE LANDSCAPE EPIDEMIOLOGY OF AN AMPHIBIAN CHYTRID FUNGUS BY STACY ELIZABETH BEYER THESIS Submitted in partial fulfillment of the requirements for the degree of Master of Science
EFFECTS OF CANOPY COVER ON THE LANDSCAPE EPIDEMIOLOGY OF AN AMPHIBIAN CHYTRID FUNGUS BY STACY ELIZABETH BEYER THESIS Submitted in partial fulfillment of the requirements for the degree of Master of Science
Host species composition influences infection severity among amphibians in the absence of spillover transmission
 Host species composition influences infection severity among amphibians in the absence of spillover transmission Han, B. A., Kerby, J. L., Searle, C. L., Storfer, A., & Blaustein, A. R. (2015). Host species
Host species composition influences infection severity among amphibians in the absence of spillover transmission Han, B. A., Kerby, J. L., Searle, C. L., Storfer, A., & Blaustein, A. R. (2015). Host species
Fire effects on tinajas and amphibian habitat at Saguaro National Park
 Fire effects on tinajas and amphibian habitat at Saguaro National Park Don Swann, Kara O Brien, and Chuck Perger Saguaro National Park, Tucson, Arizona Thanks to: Mike Sredl (Arizona Game and Fish) Ann
Fire effects on tinajas and amphibian habitat at Saguaro National Park Don Swann, Kara O Brien, and Chuck Perger Saguaro National Park, Tucson, Arizona Thanks to: Mike Sredl (Arizona Game and Fish) Ann
A SURVEY FOR THE PRESENCE OF THE AMPHIBIAN CHYTRID FUNGUS (BATRACHOCHYTRIUM DENDROBATIDIS) IN TASMANIA
 Papers and Proceedings of the Royal Society of Tasmania, Volume 140, 2006 25 A SURVEY FOR THE PRESENCE OF THE AMPHIBIAN CHYTRID FUNGUS (BATRACHOCHYTRIUM DENDROBATIDIS) IN TASMANIA by David Obendorf and
Papers and Proceedings of the Royal Society of Tasmania, Volume 140, 2006 25 A SURVEY FOR THE PRESENCE OF THE AMPHIBIAN CHYTRID FUNGUS (BATRACHOCHYTRIUM DENDROBATIDIS) IN TASMANIA by David Obendorf and
Climate Change Vulnerability Assessment for Species
 Climate Change Vulnerability Assessment for Species SPECIES: Specify whether you are assessing the entire species or particular populations: This tool assesses the vulnerability or resilience of species
Climate Change Vulnerability Assessment for Species SPECIES: Specify whether you are assessing the entire species or particular populations: This tool assesses the vulnerability or resilience of species
National Wildland Significant Fire Potential Outlook
 National Wildland Significant Fire Potential Outlook National Interagency Fire Center Predictive Services Issued: September, 2007 Wildland Fire Outlook September through December 2007 Significant fire
National Wildland Significant Fire Potential Outlook National Interagency Fire Center Predictive Services Issued: September, 2007 Wildland Fire Outlook September through December 2007 Significant fire
Chytrid fungus parasitizing the wild amphibian Leptodactylus ocellatus (Anura: Leptodactylidae) in Argentina
 DISEASES OF AQUATIC ORGANISMS Vol. 64: 247 252, 2005 Published May 20 Dis Aquat Org Chytrid fungus parasitizing the wild amphibian Leptodactylus ocellatus (Anura: Leptodactylidae) in Argentina Raúl A.
DISEASES OF AQUATIC ORGANISMS Vol. 64: 247 252, 2005 Published May 20 Dis Aquat Org Chytrid fungus parasitizing the wild amphibian Leptodactylus ocellatus (Anura: Leptodactylidae) in Argentina Raúl A.
The Effects of Temperature Frequency and Magnitude on Lithobates sylvaticus and Hyla versicolor
 University of Connecticut DigitalCommons@UConn Holster Scholar Projects Honors Scholar Program 2013 The Effects of Temperature Frequency and Magnitude on Lithobates sylvaticus and Hyla versicolor Diana
University of Connecticut DigitalCommons@UConn Holster Scholar Projects Honors Scholar Program 2013 The Effects of Temperature Frequency and Magnitude on Lithobates sylvaticus and Hyla versicolor Diana
2011 National Seasonal Assessment Workshop for the Eastern, Southern, & Southwest Geographic Areas
 2011 National Seasonal Assessment Workshop for the Eastern, Southern, & Southwest Geographic Areas On January 11-13, 2011, wildland fire, weather, and climate met virtually for the ninth annual National
2011 National Seasonal Assessment Workshop for the Eastern, Southern, & Southwest Geographic Areas On January 11-13, 2011, wildland fire, weather, and climate met virtually for the ninth annual National
Biology and Ecology of Forest Health. Climate Change and Tree Health
 Biology and Ecology of Forest Health Climate Change and Tree Health Assume classic UKCIP scenario: ca 3 o C warming in 50-80 yrs; warmer winters/summers; increased winter rain and summer drought; perturbations
Biology and Ecology of Forest Health Climate Change and Tree Health Assume classic UKCIP scenario: ca 3 o C warming in 50-80 yrs; warmer winters/summers; increased winter rain and summer drought; perturbations
CBA Practice Exam - Ecology
 CBA Practice Exam - Ecology For the following two questions, use the diagram below: 1. (TEKS 11B) The organisms in the illustration are all part of a pond community. What would likely happen to the fish
CBA Practice Exam - Ecology For the following two questions, use the diagram below: 1. (TEKS 11B) The organisms in the illustration are all part of a pond community. What would likely happen to the fish
Supporting Information
 Supporting Information Ryan et al. 10.1073/pnas.0902252106 SI Materials and Methods Mesocosm Preparation. Mesocosm tanks were 300 gallon polyethylene cattle watering tanks manufactured by Rotonics Manufacturing
Supporting Information Ryan et al. 10.1073/pnas.0902252106 SI Materials and Methods Mesocosm Preparation. Mesocosm tanks were 300 gallon polyethylene cattle watering tanks manufactured by Rotonics Manufacturing
The impact of the Amphibian Chytrid Fungus Batrachochytrium dendrobatidis on a Green and Golden Bell Frog Litoria aurea
 The impact of the Amphibian Chytrid Fungus Batrachochytrium dendrobatidis on a Green and Golden Bell Frog Litoria aurea reintroduction program at the Hunter Wetlands Centre Australia in the Hunter Region
The impact of the Amphibian Chytrid Fungus Batrachochytrium dendrobatidis on a Green and Golden Bell Frog Litoria aurea reintroduction program at the Hunter Wetlands Centre Australia in the Hunter Region
Vanishing Species 5.1. Before You Read. Read to Learn. Biological Diversity. Section. What do biodiversity studies tell us?
 Vanishing Species Before You Read Dinosaurs are probably the most familiar organisms that are extinct, or no longer exist. Many plants and animals that are alive today are in danger of dying out. Think
Vanishing Species Before You Read Dinosaurs are probably the most familiar organisms that are extinct, or no longer exist. Many plants and animals that are alive today are in danger of dying out. Think
March 1, 2003 Western Snowpack Conditions and Water Supply Forecasts
 Natural Resources Conservation Service National Water and Climate Center 101 SW Main Street, Suite 1600 Portland, OR 97204-3224 Date: March 17, 2003 Subject: March 1, 2003 Western Snowpack Conditions and
Natural Resources Conservation Service National Water and Climate Center 101 SW Main Street, Suite 1600 Portland, OR 97204-3224 Date: March 17, 2003 Subject: March 1, 2003 Western Snowpack Conditions and
California: Land and People Lesson 1: Locating California
 California: Land and People Lesson 1: Locating California Hemisphere One half of the earth or another sphere Video on Hemispheres click here Relative location Where a place is compared to one or more other
California: Land and People Lesson 1: Locating California Hemisphere One half of the earth or another sphere Video on Hemispheres click here Relative location Where a place is compared to one or more other
Background. North Cascades Ecosystem Grizzly Bear Restoration Plan/ Environmental Impact Statement. Steve Rochetta
 Grizzly Bear Restoration Plan/ Environmental Impact Statement Steve Rochetta Background Situated in the core of the North Cascades ecosystem (NCE), the North Cascades National Park Complex is surrounded
Grizzly Bear Restoration Plan/ Environmental Impact Statement Steve Rochetta Background Situated in the core of the North Cascades ecosystem (NCE), the North Cascades National Park Complex is surrounded
The North American bullfrog as a reservoir for the spread of Batrachochytrium dendrobatidis in Brazil
 Animal Conservation. Print ISSN 1367-9430 The North American bullfrog as a reservoir for the spread of Batrachochytrium dendrobatidis in Brazil L. M. Schloegel 1,2, C. M. Ferreira 3, T. Y. James 4, M.
Animal Conservation. Print ISSN 1367-9430 The North American bullfrog as a reservoir for the spread of Batrachochytrium dendrobatidis in Brazil L. M. Schloegel 1,2, C. M. Ferreira 3, T. Y. James 4, M.
Low Prevalence of Chytrid Fungus (Batrachochytrium dendrobatidis) in Amphibians of U.S. Headwater Streams
 Journal of Herpetology, Vol. 44, No. 2, pp. 253 260, 2010 Copyright 2010 Society for the Study of Amphibians and Reptiles Low Prevalence of Chytrid Fungus (Batrachochytrium dendrobatidis) in Amphibians
Journal of Herpetology, Vol. 44, No. 2, pp. 253 260, 2010 Copyright 2010 Society for the Study of Amphibians and Reptiles Low Prevalence of Chytrid Fungus (Batrachochytrium dendrobatidis) in Amphibians
Kingdom Animalia. Zoology the study of animals
 Kingdom Animalia Zoology the study of animals Summary Animals are multicellular and eukaryotic. consume and digest organic materials thereby being heterotrophs. Most are motile at some time in their lives.
Kingdom Animalia Zoology the study of animals Summary Animals are multicellular and eukaryotic. consume and digest organic materials thereby being heterotrophs. Most are motile at some time in their lives.
Diet comparison in three tadpole species, Rana sylvatica, Bufo americanus, and Pseudacris crucifer, in a northern temperate climate
 Diet comparison in three tadpole species, Rana sylvatica, Bufo americanus, and Pseudacris crucifer, in a northern temperate climate Abstract Jennifer K. Quammen and Dr. Richard D. Durtsche Department of
Diet comparison in three tadpole species, Rana sylvatica, Bufo americanus, and Pseudacris crucifer, in a northern temperate climate Abstract Jennifer K. Quammen and Dr. Richard D. Durtsche Department of
SUSCEPTIBILITY TO THE AMPHIBIAN CHYTRID FUNGUS VARIES WITH ONTOGENY IN THE DIRECT-DEVELOPING FROG ELEUTHERODACTYLUS COQUI
 DOI: 10.7589/2013-10-268 Journal of Wildlife Diseases, 50(3), 2014, pp. 000 000 # Wildlife Disease Association 2014 SUSCEPTIBILITY TO THE AMPHIBIAN CHYTRID FUNGUS VARIES WITH ONTOGENY IN THE DIRECT-DEVELOPING
DOI: 10.7589/2013-10-268 Journal of Wildlife Diseases, 50(3), 2014, pp. 000 000 # Wildlife Disease Association 2014 SUSCEPTIBILITY TO THE AMPHIBIAN CHYTRID FUNGUS VARIES WITH ONTOGENY IN THE DIRECT-DEVELOPING
Adverse effects of environmentally relevant dietary mercury exposure in larvae of the southern leopard frog, Rana sphenocephala
 Adverse effects of environmentally relevant dietary mercury exposure in larvae of the southern leopard frog, Rana sphenocephala JASON UNRINE, CHARLES JAGOE, WILLIAM HOPKINS AND HEATHER BRANT Savannah River
Adverse effects of environmentally relevant dietary mercury exposure in larvae of the southern leopard frog, Rana sphenocephala JASON UNRINE, CHARLES JAGOE, WILLIAM HOPKINS AND HEATHER BRANT Savannah River
Repercussions of Global Change
 CAUSES ELEVEN Repercussions of Global Change JAMIE K. REASER AND ANDREW BLAUSTEIN Living organisms must track the climate regimes appropriate for their survival, adapt to new conditions, or go extinct.
CAUSES ELEVEN Repercussions of Global Change JAMIE K. REASER AND ANDREW BLAUSTEIN Living organisms must track the climate regimes appropriate for their survival, adapt to new conditions, or go extinct.
CAPTIVE REARING STUDY OF THE THERMONECTUS MARMORATUS. Tim O Sullivan. Keeper, Invertebrates, St. Louis Zoo
 CAPTIVE REARING STUDY OF THE THERMONECTUS MARMORATUS Tim O Sullivan Keeper, Invertebrates, St. Louis Zoo 1 Government Drive, St. Louis, MO 63110 USA INTRODUCTION Thermonectus marmoratus, commonly referred
CAPTIVE REARING STUDY OF THE THERMONECTUS MARMORATUS Tim O Sullivan Keeper, Invertebrates, St. Louis Zoo 1 Government Drive, St. Louis, MO 63110 USA INTRODUCTION Thermonectus marmoratus, commonly referred
Station #5: Evolution. Read over the Theory of Evolution study guide Answer the following questions:
 Station #5: Evolution Read over the Theory of Evolution study guide Answer the following questions: 1. Which of the following does not contribute to genetic variation among offspring? a. Division of cells
Station #5: Evolution Read over the Theory of Evolution study guide Answer the following questions: 1. Which of the following does not contribute to genetic variation among offspring? a. Division of cells
Ch 5. Evolution, Biodiversity, and Population Ecology. Part 1: Foundations of Environmental Science
 Ch 5 Evolution, Biodiversity, and Population Ecology Part 1: Foundations of Environmental Science PowerPoint Slides prepared by Jay Withgott and Heidi Marcum Copyright 2006 Pearson Education, Inc., publishing
Ch 5 Evolution, Biodiversity, and Population Ecology Part 1: Foundations of Environmental Science PowerPoint Slides prepared by Jay Withgott and Heidi Marcum Copyright 2006 Pearson Education, Inc., publishing
Tasmanian Frog and Chytrid monitoring 2014: Executive summary
 Tasmanian Frog and Chytrid monitoring 2014: Executive summary December 2014 Supported and funded by: Full citation Sinn, D. and Philips, A. (2014). Tasmanian Frog and Chytrid monitoring 2014: sound recording,
Tasmanian Frog and Chytrid monitoring 2014: Executive summary December 2014 Supported and funded by: Full citation Sinn, D. and Philips, A. (2014). Tasmanian Frog and Chytrid monitoring 2014: sound recording,
Alligator mississippiensis.
 Alligator mississippiensis http://www.birdsasart.com/bn201.htm Core Case Study: Why Should We Care about the American Alligator? Largest reptile in North America 1930s: Hunters and poachers Importance
Alligator mississippiensis http://www.birdsasart.com/bn201.htm Core Case Study: Why Should We Care about the American Alligator? Largest reptile in North America 1930s: Hunters and poachers Importance
What is insect forecasting, and why do it
 Insect Forecasting Programs: Objectives, and How to Properly Interpret the Data John Gavloski, Extension Entomologist, Manitoba Agriculture, Food and Rural Initiatives Carman, MB R0G 0J0 Email: jgavloski@gov.mb.ca
Insect Forecasting Programs: Objectives, and How to Properly Interpret the Data John Gavloski, Extension Entomologist, Manitoba Agriculture, Food and Rural Initiatives Carman, MB R0G 0J0 Email: jgavloski@gov.mb.ca
Resistance, tolerance and environmental transmission dynamics determine host extinction risk in a load-dependent amphibian disease
 LETTER Ecology Letters, (2017) 20: 1169 1181 doi: 10.1111/ele.12814 Resistance, tolerance and environmental transmission dynamics determine host extinction risk in a load-dependent amphibian disease Mark
LETTER Ecology Letters, (2017) 20: 1169 1181 doi: 10.1111/ele.12814 Resistance, tolerance and environmental transmission dynamics determine host extinction risk in a load-dependent amphibian disease Mark
Curriculum Vitae John Matthew Romansic 1203 E. Curtis St. Tampa, FL (813)
 Curriculum Vitae John Matthew Romansic 1203 E. Curtis St. Tampa, FL 33603 (813) 545-6500 jromansi@cas.usf.edu Education 2001-2007 Oregon State University Ph.D. - Zoology Advisor: Professor Andrew R. Blaustein
Curriculum Vitae John Matthew Romansic 1203 E. Curtis St. Tampa, FL 33603 (813) 545-6500 jromansi@cas.usf.edu Education 2001-2007 Oregon State University Ph.D. - Zoology Advisor: Professor Andrew R. Blaustein
Effects of an Infectious Fungus, Batrachochytrium dendrobatidis, on Amphibian Predator-Prey Interactions
 Effects of an Infectious Fungus, Batrachochytrium dendrobatidis, on Amphibian Predator-Prey Interactions Barbara A. Han 1 *, Catherine L. Searle 2, Andrew R. Blaustein 2 1 Odum School of Ecology, University
Effects of an Infectious Fungus, Batrachochytrium dendrobatidis, on Amphibian Predator-Prey Interactions Barbara A. Han 1 *, Catherine L. Searle 2, Andrew R. Blaustein 2 1 Odum School of Ecology, University
Evidence for disease-related amphibian decline in Colorado
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln USGS Staff -- Published Research US Geological Survey 2003 Evidence for disease-related amphibian decline in Colorado Erin
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln USGS Staff -- Published Research US Geological Survey 2003 Evidence for disease-related amphibian decline in Colorado Erin
Linking manipulative experiments to field data to test the dilution effect
 Journal of Animal Ecology 2014, 83, 557 565 doi: 10.1111/1365-2656.12159 Linking manipulative experiments to field data to test the dilution effect Matthew D. Venesky 1, *, Xuan Liu 2, Erin L. Sauer 1
Journal of Animal Ecology 2014, 83, 557 565 doi: 10.1111/1365-2656.12159 Linking manipulative experiments to field data to test the dilution effect Matthew D. Venesky 1, *, Xuan Liu 2, Erin L. Sauer 1
